Abstract
Antibiotic therapy is essential for sepsis management, but the optimal empirical strategy remains uncertain. This study evaluated the effects of first-line antibiotic preference and initiation timing on in-hospital mortality among intensive care units (ICU) patients with sepsis. Using the MIMIC-IV database, we emulated a sequential target trial comparing patients who received antibiotics within 48 h of sepsis diagnosis versus delayed initiation. Randomization was approximated through a clone–censor–weight process to address confounding by indication. The primary outcome was in-hospital mortality. Weighted Cox regression estimated hazard ratios (HRs), and sensitivity analyses tested robustness. Among 3,669 eligible patients, 3,568 (97%) received antibiotics within 48 h. After weighting, covariate balance was achieved. Beta-lactam use was associated with lower in-hospital mortality (HR 0.88, 95% CI 0.78–0.95), with consistent reductions at 7, 14, and 60 days. Timing within the 48-hour window did not modify outcomes for either beta-lactams or glycopeptides. Empirical beta-lactam therapy was linked to improved survival among ICU sepsis patients, whereas timing of initiation showed no significant impact. These findings support prioritizing beta-lactam–based regimens as first-line empirical coverage in early sepsis management.
Similar content being viewed by others
Introduction
Sepsis, a severe systemic inflammatory response syndrome caused by infection, is often considered an excessive response of the body to infection or other forms of trauma1,2,3. The mortality rate for sepsis patients ranges from 25% to 30% and is responsible for up to half of all in-hospital deaths in the United States4,5. Standard sepsis management involves antimicrobial treatment, hemodynamic stabilization (e.g., fluid resuscitation, vasopressors, acid-base correction), and advanced life support (e.g., mechanical ventilation, renal replacement therapy)6. Among these, antimicrobial treatment is applied to all cases of septicemia and is considered the foundation of treatment. Although antibiotics have significantly improved sepsis outcomes, the optimal strategy for their initiation and selection remains debated7. Earlier SSC guidance advocated administration within 1 h for sepsis8, a position that drew criticism regarding evidence strength and potential unintended consequences of rigid timing metrics9. The 2021 SSC guideline refined this issue by stratifying recommendations based on clinical context: for adults with septic shock or a high likelihood of sepsis, antimicrobials should be administered immediately, whereas for adults with possible sepsis without shock, a time-limited rapid investigation is suggested and antimicrobials should be given within 3 h if concern for infection persists; delaying antimicrobials may be reasonable when infection likelihood is low and shock is absent10. This evolution underscores the tension between avoiding harmful delays and minimizing unnecessary broad-spectrum exposure.
In clinical practice, sepsis treatment often begins with empirical broad-spectrum antibiotics or combination therapies to avoid delays in targeting the causative pathogens11. However, overly potent antibiotics may cause adverse drug effects, disrupt the patient’s microbiota balance, or lead to antibiotic resistance or persistent infections12. The optimal strategy is not always straightforward given the risks associated with antibiotic type and the potential conflict over delayed administration. This seems to be case especially in patients without septic shock. Perhaps for this reason, the use of antibiotics is more based on empirical evidence rather than strict evidence-based medicine in actual practice. Moreover, ethical considerations have prevented trials comparing delayed versus early antibiotic administration strategies. In this setting, using observational data to emulate a potential randomized trial could provide valuable insights13.
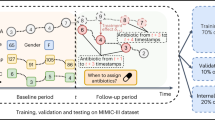
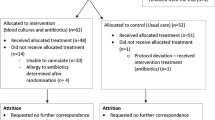
Therefore, this study leverages the MIMIC-IV database to evaluate how first-line coverage preference, defined by the initial choice between glycopeptides and beta-lactams, and the timing of antibiotic initiation influence outcomes in sepsis patients. Our objective is to assess different empirical treatment preferences to inform evidence-based decision-making in early sepsis management (Figs. 1 and 2).
Flowchart for study selection process.
Study design diagram for a TTE steps including clone-censor-weight three approaches, b Comparing different antibiotic types, c Comparing different time intervals using glycopeptide as an example, d Exposure and follow-up windows.
Results
Among 22,119 sepsis episodes, the majority received either glycopeptides or beta-lactams as the first antibiotic. Initial aminoglycoside use was infrequent (389/22,119, 1.8%), and macrolide use was also low (1,255/22,119, 5.7%). Survival curves for the four antibiotic groups such as glycopeptides, macrolides, aminoglycosides, and beta-lactams (Supplementary Fig. 1) revealed in-hospital mortality rates of 22% (354/1597), 20% (18/88), 5% (1/21), and 13% (230/1825), respectively (P < 0.001). Bonferroni-adjusted pairwise comparisons indicated a significant difference in prognosis between glycopeptide and beta-lactam treatments (P.adjusted < 0.001, Supplementary Table 2).
Primary analysis
From 2008 to 2019, 3,669 out of 20,475 newly diagnosed sepsis patients met eligibility criteria, with 3,568 (97%) receiving antibiotic treatment in 48 h. Table 1 presents patient characteristics, with an average age of 65.35 years and a predominance of males (58%). Compared to glycopeptide-treated patients, those with beta-lactams had lower rates of kidney disease (297 vs. 335, P = 0.001), thrombosis (70 vs. 99, P = 0.002), malignancy (199 vs. 215, P = 0.036), chronic obstructive pulmonary disease (196 vs. 222, P = 0.009), and septic shock (291 vs. 618, P < 0.001). They also had lower SOFA scores (3.53 vs. 3.93, p < 0.001) and SAPS II scores (38.83 vs. 42.80, P < 0.001), suggesting generally milder illness and fewer comorbidities among the beta-lactam group.
After excluding any missing values for covariates, 3,510 patients remained, of whom 1,857 (52.9%) received glycopeptides and 1,653 (47.1%) received beta-lactams. IPTW weighting balance tables and love plots showed covariates are well balanced between patients before and after censoring (Fig. 3, Supplementary Table 3). In this weighted cohort, beta-lactam treatment resulted in a lower risk of admission mortality (HR: 0.88, 95% CI: 0.78–0.95) and reduced mortality at 7, 14, and 60 days, with HRs of 0.78 (0.67–0.92), 0.86 (0.76–0.94), and 0.86 (0.78–0.90), respectively (Table 2). The beta-lactam group recorded 234 deaths (13%), while the glycopeptide group had 368 deaths (22%). Survival curves (Fig. 4) highlighted improved outcomes for beta-lactam treatment, with median survival times of 56.99 versus 40.38 days (P < 0.001).
Love plot for patients before and after censoring. The dashed line represents a value of 0.1. If the absolute standardized mean difference of the weighted variables is within the dashed line, then the result shows that covariates are well balanced.
Weighted survival curves between glycopeptides and beta-lactams.
Using the same approach, we assessed the timing of antibiotic initiation for glycopeptides and beta-lactams. Supplementary Tables 4 and Supplementary Fig. 2 illustrate the number of censored cases per hour and covariate balance within the first hour. In hourly target trial emulations, neither glycopeptides nor beta-lactams showed that time played a significant role in the potential benefit of initiating antibiotic therapy (Table 3, Supplementary Table 5).
Sensitivity and subgroup analyses
Sensitivity and subgroup analysis results are provided in the supplementary materials (Supplementary Tables 6–8, Supplementary Fig. 3). Key findings confirmed that the results remained robust with a 24-hour grace period, multiple imputation for missing data, and truncation of extreme weights. Moreover, restriction to patients receiving monotherapy only within the first 48 h yielded results consistent with the main analysis: beta-lactam was associated with a lower hazard (HR 0.63, 95% CI: 0.49–0.80). Subgroup analyses by age, sex, SOFA score, and SAPS II score showed trends consistent with the primary findings.
Discussion
This study leverages a real-world observational cohort and a target trial emulation to assess the effects of antibiotic type and timing on outcomes in sepsis patients. Controlling for demographics, comorbidities, and sepsis severity, we found that beta-lactams were associated with a 12% reduction in in-hospital mortality risk compared to glycopeptides. However, we observed no significant association between the timing of antibiotic initiation and patient outcomes for either glycopeptides or beta-lactams. Sensitivity analyses supported these findings and stratified by age, sex, SOFA score, and SAPS II score yielded consistent results, with broader confidence intervals but no change in effect size or direction, affirming the robustness of our findings.
Sepsis, a condition involving organ dysfunction from an excessive host response to infection, is managed with early detection, prompt antibiotic therapy, and fluid resuscitation to prevent septic shock. Standard empirical treatments include beta-lactams and vancomycin. Beta-lactams provide broad-spectrum coverage against both gram-positive and gram-negative bacteria, while adding vancomycin targets methicillin-resistant staphylococcus aureus14.
Evidence strongly supports beta-lactam efficacy in sepsis treatment15,16. Veiga et al.16 suggested beta-lactams can be safely administered at or above standard doses without accumulation risks, enhancing pharmacodynamics but with no obvious increased toxicity. Similarly, a meta-analysis by Vardakas et al.15 reported a 30% reduction in mortality for sepsis patients receiving prolonged beta-lactam infusions versus shorter ones. Vancomycin, a glycopeptide antibiotic, has potent activity against gram-positive bacteria but carries nephrotoxicity17. Higher vancomycin levels have been linked to an increased risk of acute kidney injury, and indiscriminate use may promote antibiotic resistance by favoring multidrug-resistant organisms18. Our findings indicate that beta-lactams, when adjusted for illness severity and other confounding factors, are associated with a lower mortality risk in sepsis patients, supporting improved clinical outcomes. This aligns with antibiotic de-escalation (ADE) principles, which aim to prevent resistance, reduce costs, and minimize overtreatment in sepsis19,20. Shifting from empirical vancomycin to beta-lactams may be less effective than initial beta-lactam therapy. Therefore, efficacy, adverse events, resistance risk, and cost must be weighed in starting antibiotic selection, with combination therapy considered when necessary.
Some studies have reported that delayed antibiotic treatment can worsen outcomes, including higher mortality rates and an increased risk of progression to septic shock21,22,23,24. However, these findings are primarily based on retrospective studies, which limit causal inference. To date, no randomized controlled trials (RCTs) have directly investigated this issue. The only prehospital RCT examining early antibiotic administration in suspected sepsis showed no survival benefits25. Our results suggest that delayed treatment of beta-lactams or vancomycin does not significantly affect sepsis outcomes. This conclusion aligns with recent studies26,27. A meta-analysis by Sterling et al.26, which included 11 studies, found no significant increase in mortality risk for antibiotics beyond one hour (pooled OR: 1.46, 95% CI: 0.89–2.40) or three hours after diagnosis (pooled OR: 1.16, 95% CI: 0.92–1.46). Yunjoo et al. analyzed 3,035 patients and reported a protective effect of administration within one hour for 601 patients with septic shock (OR: 0.66, P = 0.049). However, no significant benefit was observed in non-shock patients (OR: 0.85, P = 0.300). While our study provides valuable insights, it is not without limitations. Despite its ability to address confounding, TTE remains an observational method. Moreover, it relies on “post hoc randomization,” unlike the randomized design of RCTs, which limit the strength of causal conclusions28.
Still, there are some considerations. First, the sepsis patients in our study generally exhibited milder symptoms, with few progressing to septic shock (26%). In contrast, prior studies primarily included patients with severe sepsis or septic shock23. Furthermore, while nearly 50% of patients in previous studies received antibiotics within two hours21, administration in our study was more evenly distributed across the first five hours. Finally, although delayed treatment in patients with suspected infection has been associated with an increased risk of progression to septic shock, its effect on in-hospital mortality appears less pronounced24.
SCC guidelines recommend initiating empirical broad-spectrum antibiotics within one hour of recognition29. However, this recommendation faces practical challenges29,30,31. First, clinicians must gather and assess patient data like clinical signs, laboratory results, and imaging findings within one hour, which is often unfeasible in real-world settings. Second, the use of broad-spectrum antibiotics can disrupt the gut microbiome, leading to dysbiosis, overgrowth of antibiotic-resistant organisms, and more complex clinical scenarios32,33. Third, it remains unclear how many patients genuinely require broad-spectrum empirical therapy. Rhee et al.34 revealed that more than two-thirds of patients received broad-spectrum antibiotics, but only one-sixth of those were isolated with methicillin-resistant staphylococcus aureus and that unnecessarily extensive therapy was associated with higher mortality. Fourth, the timing of antibiotic administration may influence blood culture results. A study comparing blood cultures collected before and during antibiotic treatment reported positivity rates of 50.6% and 27.7%, respectively (p < 0.001), despite similar baseline characteristics, laboratory parameters, severity distributions, and mortality rates35. This highlights the potential for reduced pathogen identification when cultures are performed after initiating antibiotics. Moreover, a retrospective analysis of suspected infection cases found that one-third were ultimately diagnosed with non-infectious or viral conditions36. Another retrospective study confirmed this phenomenon and found that using administrative data as the gold standard, 42% of sepsis treatment initiation pathways were false positives, with vancomycin being the second most overused antibiotic37. Finally, the cost-effectiveness of administering empirical broad-spectrum antibiotics within one hour of sepsis diagnosis remains uncertain and warrants further investigation.
For patients with suspected sepsis but without shock, best practices emphasize prompt evaluation to determine if the condition is infectious38. Recent advances, such as the uRAST rapid antimicrobial susceptibility testing method, have significantly reduced the turnaround time for antimicrobial susceptibility profiles by 40–60 h compared to traditional blood cultures, while maintaining high classification accuracy39. In addition, a multicenter study identified a small set of host response gene transcripts that could accurately detect sepsis up to three days before clinical symptoms appeared. The study also emphasized that infection and sepsis develop over the course of days rather than hours, thereby providing an opportunity for early targeted interventions. In addition, a multicenter study identified a small set of host response gene transcripts that could accurately detect sepsis up to three days before clinical symptoms appeared. It also emphasized that infection and sepsis develop over the course of days rather than hours, thereby providing an opportunity for early targeted interventions40,41. Guidelines also recommend adjusting antibiotic administration timing based on the likelihood of septic shock42. Taneja et al. developed a sensitivity scoring system using a random forest model to identify critically ill patients in advance and aid clinicians with the timing of administration43. Additionally, a “one-size-fits-all” approach in sepsis management has been reported in some hospitals. For instance, strict policies requiring antibiotic treatment within one hour may pressure clinicians to prescribe broad-spectrum antibiotics empirically, even for those who may not need them30. Such measures can worsen patient outcomes and lead to resource wastage44.
Study limitations
This study has several limitations. First, the MIMIC-IV dataset is a single-center cohort. Important covariates related to sepsis development, such as pre-admission infection status and infection site distribution, were underreported, which could influence decisions on antibiotic initiation and mortality risk assessments. Second, due to sample size, we did not compare single-agent versus combination therapies. Third, our data lacked information on antibiotic infusion times and dosing regimens, making it impossible to evaluate potential mortality differences across these subgroups. Despite these limitations, our study provides meaningful insights into antibiotic use in sepsis management.
Conclusion
The choice of antibiotic type significantly influences patient outcomes, with beta-lactams improving survival rates and supporting the principle of antibiotic de-escalation. For most sepsis patients, the timing of antibiotic administration does not appear to affect survival outcomes. Future research should focus on evaluating treatment timing based on risk probability and exploring differences in varied clinical scenarios. Additionally, cost-effectiveness studies are needed to assess the benefits and limitations of bundled antibiotic management, providing evidence on whether early treatment or waiting for culture results offers greater value.
Methods
Data source
The study used the Medical Information Mart for Intensive Care IV (MIMIC-IV), developed by the Massachusetts Institute of Technology (MIT). This dataset includes information from patients admitted to the emergency department or intensive care unit of Beth Israel Deaconess Medical Center from 2008 to 2019. It provides detailed information on demographics, physiological, and biochemical markers, treatments, timing, and one-year mortality following discharge.
Study design and eligibility
We conducted a retrospective cohort study using a target trial emulation (TTE) approach, following clone-censor-weight procedures to examine the effects of antibiotic type and timing on sepsis patient outcomes. Supplementary Table 1 outlines the simulation protocol with the MIMIC-IV. Patients aged over 18 with a sepsis diagnosis were eligible for inclusion. The date of the first sepsis diagnosis in the study period was designated as Day 0. Since the dataset included precise treatment start and stop times, we excluded patients with antibiotic use recorded before Day 0.
Antibiotic exposure definition
Antibiotic exposure was defined based on the first antibiotic administered within 48 h of sepsis diagnosis (Day 0). Patients were classified according to the class of this first antibiotic (first-line coverage preference), regardless of any subsequent combination therapy. Treatment initiation time was calculated as the difference between Day 0 and antibiotic start, classified within a 48-hour window (e.g., if start time – Day 0 = 0.68, the patient received antibiotics within one hour). Follow-up began on Day 0 and continued until the outcome occurred. Figure 1 shows inclusion and exclusion criteria, while Fig. 2 illustrates study methods, including TTE steps, exposure and follow-up windows, and baseline variable definitions.
Treatment strategies and assignment
We compared different antibiotic strategies for patients with sepsis, focusing on several classes: glycopeptides (Vancomycin), macrolides (Zithromax Z-Pak or Erythromycin), aminoglycosides (Gentamicin, Tobramycin, Neomycin, or Amikacin), and beta-lactams (penicillins, cephalosporins, and carbapenems). Initial treatment was defined as any administration of these antibiotics within 48 h of the first sepsis diagnosis; treatment beyond 48 h was considered no antibiotic use. Patients were assigned to a treatment arm based on the first administered antibiotic, without excluding those who received additional antibiotics later.
Further, to assess the effects of early administration on survival, we simulated a trial comparing antibiotic initiation at different intervals: within the first hour, 2–6 h, 7–12 h, and later. These intervals, chosen to align with prior research, help provide greater homogeneity between patients. We initially identified eligible participants and reassessed over a 48-hour period. For example, patients receiving antibiotics in the third hour met two criteria: (1) no prior antibiotic treatment and (2) eligibility based on inclusion and exclusion criteria at that time. In this simulated trial, randomization was achieved through clone-censor-weight steps. Eligible patients were randomly assigned to any time point within the 48-hour treatment window, resulting in 48 potential strategies.
Sepsis was defined according to the Sepsis-3 criteria as suspected infection with acute organ dysfunction, operationalized as an acute increase in SOFA score ≥ 245. Suspected infection was identified using the standard EHR algorithm based on paired antibiotic administration and body-fluid culture order/collection timestamps within an asymmetric window, and we used the MIMIC-IV derived Sepsis-3 concept (mimiciv_derived.sepsis3) to implement this definition46.
Outcomes
The primary outcome of this study was in-hospital mortality, while 7, 14, and 60 day mortality were defined as secondary outcomes.
Covariates
Covariates included age, sex, admission weight, history of thrombosis, Charlson Comorbidity Index (CCI), Sequential Organ Failure Assessment (SOFA) score, Simplified Acute Physiology Score II (SAPSII), recent renal replacement therapy, recent mechanical ventilation, and recent septic shock. Additional medical history variables included cardiovascular diseases (e.g., myocardial infarction, heart failure, cerebrovascular disease, hypertension, coronary artery disease, atrial fibrillation, and stroke), kidney disease (e.g., chronic kidney disease and renal failure), liver disease (mild and severe), cancer, and chronic obstructive pulmonary disease.
Statistical analysis
Statistical analyses were performed using R (version 4.3.1). Continuous variables are presented as means with standard deviations, and categorical variables as frequencies with percentages. Group differences were tested using the Wilcoxon rank-sum test or chi-square test, as appropriate. Initially, we assessed the association between each class and in-hospital mortality, followed by pairwise comparisons with the Bonferroni adjustment to identify which antibiotics were significantly associated with prognosis. Subsequently, the clone-censor-weight approach was used to estimate the effects of two treatment strategies. For each eligible participant, two copies were created and assigned to each treatment strategy. During follow-up, we applied artificial censoring for deviations from the assigned strategy within the predefined 48-hour grace period (i.e., if the assigned antibiotic was not initiated within 48 h, the participant was censored at 48 h).
To adjust for selection bias due to censoring, we applied inverse probability of treatment weighting (IPTW), with truncation to reduce the influence of extreme weights. Further details of TTE are available in Supplementary Method. We assessed covariate balance using a love plot, with standardized differences below 0.1 indicating adequate balance. Weighted Cox regression and weighted survival curves were used to evaluate in-hospital mortality, with hazard ratios (HRs) and 95% confidence intervals (CIs) reported.
In addition, we analyzed the effect of immediate glycopeptide and beta-lactam use within 48 h versus delayed. For this, we estimated the hourly probability of antibiotic administration throughout hour 1 to hour 48, producing a weighted population. Weighted Cox models were applied to assess in-hospital mortality and mortality at 7, 14, and 60 days. Results were considered statistically significant if the 95% CI for the hazard ratio did not include 1. All p-values are two-sided, with significance set at 0.05.
Sensitivity analysis
Several sensitivity analyses were conducted to evaluate the robustness of our findings. First, we shortened the grace period to 24 h to assess treatment effects with a more immediate initiation window. Second, we used multiple imputation to deal with the missing data at baseline compared with removing the data in the main method. Third, we truncated weights at 95th percentile to evaluate the stability of treatment effects in relation to extreme weights. In addition, we performed subgroup analyses by age, sex, SOFA score, and SAPSII score. Finally, we repeated the primary analysis after restricting the cohort to patients who received monotherapy only within the first 48 h, thereby assessing whether the estimated treatment effects were robust to exclusion of patients who received combination therapy.
Data availability
Publicly available datasets were analyzed in this study. This data can be found here: [https://mimic.mit.edu/](https:/mimic.mit.edu) . The codes can be found here: https://github.com/mohuaifengw/SR1ef8ce959be9/blob/main/main.R.
Abbreviations
- ICU:
-
Intensive Care Unit
- HRs:
-
Hazard ratios
- SSC:
-
Surviving Sepsis Campaign
- TTE:
-
Target trial emulation
- SOFA:
-
Sequential Organ Failure Assessment
- CCI:
-
Charlson Comorbidity Index
- SAPSII:
-
Simplified Acute Physiology Score II
- IPTW:
-
Inverse probability of treatment weighting
- CIs:
-
Confidence intervals
- ADE:
-
Antibiotic de-escalation
- RCTs:
-
Randomized controlled trials
- MIMIC-IV:
-
Medical Information Mart for Intensive Care IV
- MIT:
-
Massachusetts Institute of Technology
References
Srzić, I., Nesek Adam, V. & Tunjić Pejak, D. Sepsis definition: What’s new in the treatment guidelines. Acta Clin. Croat. 61 (Suppl 1), 67–72 (2022).
Liu, D. et al. Sepsis-induced immunosuppression: Mechanisms, diagnosis and current treatment options. Mil Med. Res. 9, 56 (2022).
Vincent, J. L. et al. Prevalence and outcomes of infection among patients in intensive care units in 2017. JAMA 323, 1478–1487 (2020).
Liu, V. et al. Hospital deaths in patients with sepsis from 2 independent cohorts. JAMA 312, 90–92 (2014).
Hou, J. et al. S1PR3 Signaling drives bacterial killing and is required for survival in bacterial sepsis. Am. J. Respir Crit. Care Med. 196, 1559–1570 (2017).
Vincent, J. L. Current sepsis therapeutics. EBioMedicine. (2022). https://doi.org/10.1016/j.ebiom.2022.104318
Klompas, M. & Rhee, C. Victories and opportunities in the surviving sepsis campaign’s antibiotic timing guidance. Crit. Care Med. 52, 1138–1141 (2024).
Seymour, C. W. et al. Assessment of clinical criteria for sepsis: For the third international consensus definitions for sepsis and septic shock (Sepsis-3). JAMA 315, 762–774 (2016).
Infectious Diseases Society of America (IDSA) POSITION STATEMENT. Why IDSA did not endorse the surviving sepsis campaign guidelines. Clin. Infect. Dis. 66, 1631–1635 (2018).
Evans, L. et al. Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock 2021. Crit. Care Med. 49 (11), e1063–e1143 (2021).
de la Fuente-Nunez, C., Cesaro, A. & Hancock, R. E. W. Antibiotic failure: Beyond antimicrobial resistance. Drug Resist. Updat. https://doi.org/10.1016/j.drup.2023.101012 (2023).
Barnhill, A. E., Brewer, M. T. & Carlson, S. A. Adverse effects of antimicrobials via predictable or idiosyncratic inhibition of host mitochondrial components. Antimicrob. Agents Chemother. 56, 4046–4051 (2012).
Hernán, M. A., Wang, W. & Leaf, D. E. Target trial emulation: A framework for causal inference from observational data. JAMA 328, 46–2447 (2022).
Tong, S. Y. C., Venkatesh, B. & McCreary, E. K. Acute kidney injury with empirical antibiotics for sepsis. JAMA 330, 531–1533 (2023).
Vardakas, K. Z., Voulgaris, G. L., Maliaros, A., Samonis, G. & Falagas, M. E. Prolonged versus short-term intravenous infusion of antipseudomonal β-lactams for patients with sepsis: A systematic review and meta-analysis of randomised trials. Lancet Infect. Dis. 18, 108–120 (2018).
Veiga, R. P. & Paiva, J. A. Pharmacokinetics-pharmacodynamics issues relevant for the clinical use of beta-lactam antibiotics in critically ill patients. Crit Care 22,233 (2018).
Hanrahan et al. Factors associated with vancomycin nephrotoxicity in the critically ill. Anaesth. Intensive Care. 43, 594–599 (2015).
Mühlberg, E. et al. Renaissance of vancomycin: Approaches for breaking antibiotic resistance in multidrug-resistant bacteria. Can. J. Microbiol. 66, 11–16 (2020).
McConeghy, K. W., Bleasdale, S. C. & Rodvold, K. A. The empirical combination of vancomycin and a β-lactam for staphylococcal bacteremia. Clin. Infect. Dis. 57, 1760–1765 (2013).
Plata-Menchaca, E. P. & Ferrer, R. Procalcitonin Is useful for antibiotic deescalation in sepsis. Crit. Care Med. 49, 693–696 (2021).
Liu, V. X. et al. The timing of early antibiotics and hospital mortality in sepsis. Am. J. Respir Crit. Care Med. 196, 856–863 (2017).
Kollef, M. H. et al. Timing of antibiotic therapy in the ICU. Crit. Care. 25, 360 (2021).
Ferrer, R. et al. Empiric antibiotic treatment reduces mortality in severe sepsis and septic shock from the first hour: Results from a guideline-based performance improvement program. Crit. Care Med. 42, 1749–1755 (2014).
Bisarya, R. et al. Antibiotic timing and progression to septic shock among patients in the ED with suspected infection. Chest 161, 112–120 (2022).
Alam, N. et al. Prehospital antibiotics in the ambulance for sepsis: A multicentre, open label, randomised trial. Lancet Respir Med. 6, 40–50 (2018).
Sterling, S. A., Miller, W. R., Pryor, J., Puskarich, M. A. & Jones, A. E. The impact of timing of antibiotics on outcomes in severe sepsis and septic shock: A systematic review and meta-analysis. Crit. Care Med. 43, 1907–1915 (2015).
Im, Y. et al. Time-to-antibiotics and clinical outcomes in patients with sepsis and septic shock: A prospective nationwide multicenter cohort study. Crit. Care. 26, 19 (2022).
Matthews, A. A., Danaei, G., Islam, N. & Kurth, T. Target trial emulation: Applying principles of randomised trials to observational studies. BMJ. (2022). https://doi.org/10.1136/bmj-2022-071108 .
Chen, A. X., Simpson, S. Q. & Pallin, D. J. Sepsis guidelines. N Engl. J. Med. 380, 1369–1371 (2019).
Einav, S., Leone, M. & Martin-Loeches, I. Sepsis and antibiotics: When should we deploy a parachute? Int. J. Antimicrob. Agents. https://doi.org/10.1016/j.ijantimicag.2023.106732 (2023).
Singer, M. Antibiotics for Sepsis: Does Each Hour Really Count, or Is It Incestuous Amplification? Am. J. Respir Crit. Care Med. 196, 800–802 (2017).
Perner, A. et al. Sepsis: Frontiers in diagnosis, resuscitation and antibiotic therapy. Intensive Care Med. 42, 1958–1969 (2016).
Asner, S. A., Desgranges, F., Schrijver, I. T. & Calandra, T. Impact of the timeliness of antibiotic therapy on the outcome of patients with sepsis and septic shock. J. Infect. 82, 125–134 (2021).
Rhee, C. et al. Prevalence of antibiotic-resistant pathogens in culture-proven sepsis and outcomes associated with inadequate and broad-spectrum empiric antibiotic use. JAMA Netw. Open. https://doi.org/10.1001/jamanetworkopen.2020.2899 (2021).
Scheer, C. S. et al. Impact of antibiotic administration on blood culture positivity at the beginning of sepsis: A prospective clinical cohort study. Clin. Microbiol. Infect. 25, 326–331 (2019).
Shappell, C. N., Klompas, M., Ochoa, A. & Rhee, C. Likelihood of bacterial infection in patients treated with broad-spectrum IV antibiotics in the emergency department. Crit. Care Med. https://doi.org/10.1097/CCM.0000000000005090 (2021).
Lukaszewski, R. A. et al. Presymptomatic diagnosis of postoperative infection and sepsis using gene expression signatures. Intensive Care Med. 48, 1133–1143 (2022).
Evans, L. et al. Executive summary: Surviving Sepsis campaign: International guidelines for the management of sepsis and septic shock 2021. Crit. Care Med. 49, 1974–1982 (2021).
Kim, T. H. et al. Blood culture-free ultra-rapid antimicrobial susceptibility testing. Nature 632, 893–902 (2024).
Taylor, S. P. et al. Development of an electronic clinical surveillance measure for unnecessary rapid antibiotic administration in suspected sepsis. Clin. Infect. Dis. https://doi.org/10.1093/cid/ciae445 (2024).
Zimmermann, T., Brealey, D. & Singer, M. Diagnosing sepsis: Where we’re at and where we’re going. Intensive Care Med. 50, 957–959 (2024).
Pak, T. R., Rhee, C. & Klompas, M. Timing and spectrum of antibiotic treatment for suspected sepsis and septic shock: Why so controversial? Infect. Dis. Clin. North. Am. 36, 719–733 (2022).
Taneja, I. et al. Diagnostic and prognostic capabilities of a biomarker and EMR-based machine learning algorithm for sepsis. Clin. Transl Sci. 14, 1578–1589 (2021).
Kadri, S. S. et al. Inappropriate empirical antibiotic therapy for bloodstream infections based on discordant in-vitro susceptibilities: A retrospective cohort analysis of prevalence, predictors, and mortality risk in US hospitals. Lancet Infect. Dis. 21, 241–251 (2021).
Singer, M. et al. The third international consensus definitions for sepsis and septic shock (Sepsis-3). JAMA 315 (8), 801–810 (2016).
Hu, W. et al. Identification of indications for albumin administration in septic patients with liver cirrhosis. Crit. Care. 27, 300 (2023).
Acknowledgements
Not applicable.
Funding
This research was funded by Hunan innovative province construction project (Grant No. 2019SK2211), Key research and development project of Hunan Province (Grant No.2020SK2089), The Natural Science Foundation of Hunan province (Grant Nos 2020JJ4833, 2019SK2211, and XY040019), Hunan Province Key Field R&D Program (Grant No. 2020SK2097), and Horizontal Project (Grant Nos KY080269, KY080262, XY080323, and XY080324).
Author information
Authors and Affiliations
Contributions
JY designed the study, analyzed the data, and drafted the manuscript. MY and QN supervised the study and revised the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
This study complied with the declaration of Helsinki. The author Jiayang Li has completed the Collaborative Institutional Training Program (CITP) exam (certification number: 13808420), and since the retrospective cohort study using de-identified public data; IRB approval waived.
Consent for publication
Since MIMIC-IV includes only anonymized information, patient consent to participate was waived at the local institution. Informed consent is not required for the secondary use of this de-identified database.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Li, J., Zhao, M. & He, Q. Association of antibiotic type and timing with sepsis mortality using target trial emulation. Sci Rep 16, 10447 (2026). https://doi.org/10.1038/s41598-026-40860-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-40860-5