Abstract
Metabolic dysfunction-associated steatotic liver disease (MASLD) and obstructive sleep apnea (OSA) share overlapping metabolic and inflammatory pathways, yet population-level evidence linking MASLD and incident OSA remains limited. Using a nationwide cohort of 265,452 Korean adults aged ≥ 40 years, we evaluated OSA risk across five mutually exclusive phenotypes defined by steatosis (fatty liver index [FLI] ≥ 30), cardiometabolic risk factors (CMRFs), and alcohol intake: no steatotic liver disease (SLD) without CMRFs, no SLD with CMRFs, MASLD without alcohol, MASLD with alcohol intake below the metabolic-associated alcohol-related liver disease (MetALD) threshold (men < 210 g/week, women < 140 g/week), and MetALD. During a mean follow-up of 9.5 years, 1,025 participants developed OSA. Compared with the reference group, adjusted hazard ratios (aHRs) for OSA were 1.18 (95% confidence intervals [CI] 0.93–1.50) in individuals with CMRFs alone, 1.46 (95% CI 1.12–1.91) in MASLD without alcohol, 1.52 (95% CI 1.17–1.98) in MASLD with alcohol, and 1.40 (95% CI 1.01–1.94) in MetALD. Model-based absolute risk differences (ARDs) at 9.5 years showed consistent patterns (+ 0.05%, + 0.14%, + 0.16%, and + 0.12%, respectively). Sensitivity analyses using stricter steatosis criteria (FLI ≥ 60 or hepatic steatosis index ≥ 36) demonstrated a clearer dose-related gradient, with progressively higher OSA risk across MASLD without alcohol, MASLD with alcohol, and MetALD. These findings highlight MASLD—particularly alcohol-associated phenotypes—as important risk markers for OSA and underscore the need for targeted screening and early intervention strategies in this increasingly prevalent population.
Similar content being viewed by others
Introduction
OSA is a common and increasingly recognized sleep disorder characterized by recurrent episodes of partial or complete upper airway obstruction during sleep. These episodes result in intermittent hypoxia, sleep fragmentation, and sympathetic nervous system activation, contributing to a wide range of adverse health consequences. OSA is estimated to affect 9–38% of the global adult population, with higher prevalence among older adults, males, and individuals with obesity1,2. It is independently associated with increased risks of cardiovascular disease, stroke, insulin resistance, and all-cause mortality3,4. Despite its clinical significance, OSA remains underdiagnosed and undertreated, particularly in populations with coexisting metabolic disorders.
OSA has been closely linked to several metabolic conditions, with particularly strong associations observed with type 2 diabetes mellitus (T2DM), hypertension, and obesity. For instance, OSA has been shown to increase insulin resistance and worsen glycemic control in patients with T2DM through sympathetic overactivity and nocturnal hypoxemia5. Similarly, obesity—a major risk factor for both OSA and metabolic syndrome—exacerbates upper airway collapsibility and systemic inflammation6. Nonalcoholic fatty liver disease (NAFLD), the hepatic manifestation of metabolic syndrome, has also been increasingly associated with OSA. Previous studies have reported a bidirectional relationship between OSA and NAFLD, with intermittent hypoxia accelerating liver injury and hepatic steatosis, while fatty liver disease exacerbates cardiometabolic profiles that predispose to OSA7,8,9,10.
Recently, the term MASLD was introduced to replace NAFLD so as to better reflect the underlying pathophysiology and clinical heterogeneity of fatty liver disease. MASLD is diagnosed based on evidence of hepatic steatosis in the presence of at least one of five cardinal cardiometabolic risk factors (CMRFs): overweight/obesity, hypertension, hypertriglyceridemia, low HDL-C, or T2DM11,12,13. This redefinition has garnered substantial interest and has prompted renewed investigations into the role of MASLD in systemic metabolic and cardiovascular diseases14.
Despite this redefinition, few large-scale epidemiological studies have examined the association between MASLD and OSA. Given their shared metabolic origins and overlapping pathophysiological mechanisms, understanding this association has important clinical and public health implications. To address this gap, we conducted a nationwide retrospective cohort study using data from the Korean National Health Insurance Service (NHIS) to assess the risk of OSA in individuals with MASLD, with and without alcohol-related hepatic involvement. Furthermore, we sought to disentangle the independent and alcohol-modified components of MASLD by applying recently established MASLD–MetALD classifications. This approach enables a more refined assessment of how hepatic steatosis and alcohol exposure jointly shape OSA susceptibility.
Results
Baseline characteristics of the study population
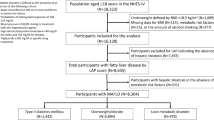
Baseline characteristics of the study population are presented in Table 1. After applying the exclusion criteria, a total of 265,452 participants were included in the final analysis (Fig. 1). Overall, 53.4% of participants were male. The mean age of participants was 58.9 ± 8.8 years. The mean body mass index (BMI) and waist circumference were 23.9 ± 3.0 kg/m2 and 81.5 ± 8.3 cm, respectively. Participants were classified into five mutually exclusive groups according to the presence of SLD and CMRFs: (1) no SLD without CMRF (n = 26,067), (2) no SLD with CMRFs (n = 138,580), (3) MASLD without alcohol consumption (n = 50,526), (4) MASLD with alcohol consumption (n = 38,207), and (5) MetALD (n = 12,072).
Flow diagram of the study population.
Notable differences in demographic and metabolic characteristics were observed across the groups. The MASLD with alcohol and MetALD groups exhibited the highest proportion of males (91.6% and 95.7%, respectively) and the youngest mean ages (56.9 ± 7.6 years and 56.2 ± 7.3 years). In contrast, the MASLD without alcohol group had the highest mean BMI (26.5 ± 2.6 kg/m2) and waist circumference (88.4 ± 6.4 cm). The prevalence of hypertension, diabetes, and dyslipidemia was substantially higher in all MASLD phenotypes than in the no-SLD groups. Additionally, liver enzymes (aspartate aminotransferase [AST], alanine aminotransferase [ALT], and γ-glutamyl transferase [γ-GTP]), triglycerides, and the Charlson Comorbidity Index (CCI) were elevated among MASLD groups, whereas HDL-C levels were lower, reflecting a more adverse metabolic profile consistent with metabolic dysfunction-associated liver disease.
Association between SLD and OSA
As shown in Table 2, during a mean follow-up of 9.5 years, 1025 participants developed OSA. Compared with the reference group (no SLD and no CMRF), the adjusted HR for OSA in individuals with CMRFs alone was not significant (aHR 1.18; 95% CI 0.93–1.50; p = 0.179). In contrast, all MASLD groups demonstrated significantly higher OSA risk. The aHRs were 1.46 (95% CI 1.12–1.91; p = 0.006) for MASLD without alcohol, 1.52 (95% CI 1.17–1.98; p = 0.002) for MASLD with alcohol below MetALD thresholds, and 1.40 (95% CI 1.01–1.94; p = 0.042) for MetALD. Model-based ARDs showed similar patterns (MASLD without alcohol: +0.14%; MASLD with alcohol: +0.16%; MetALD: +0.12%).
Dose–response relationship between the number of CMRFs and OSA risk
Among individuals without SLD, a clear dose–response pattern in OSA risk according to the number of CMRFs was not observed. Compared with the reference group (no SLD without CMRF), adjusted HRs varied modestly across CMRF strata, but none reached statistical significance (one CMRF: aHR 1.24, 95% CI 0.95–1.63; two CMRFs: aHR 1.06, 95% CI 0.81–1.40; ≥3 CMRFs: aHR 1.26, 95% CI 0.95–1.68). In contrast, individuals with MASLD (aHR 1.49, 95% CI 1.17–1.91; p = 0.001) and MetALD (aHR 1.40, 95% CI 1.01–1.94; p = 0.044) showed significantly higher risks of OSA compared with the reference group (Table 3).
Sensitivity analyses
Sensitivity analyses using stricter steatosis definitions yielded consistent results (Supplementary Table 2). Using FLI ≥ 60, a similar pattern of elevated risk across MASLD phenotypes was observed (aHR 1.70, 1.61, and 1.62, respectively). Similarly, analyses based on hepatic steatosis index (HSI) ≥ 36 showed increasing OSA risk across metabolic-alcohol phenotypes (aHR 1.67, 1.64, and 2.09, respectively). These results reinforce the robustness of the primary findings. In stratified analyses, the association between MASLD and incident OSA was generally consistent across sex, age, and smoking status, with no statistically significant effect modification observed (Fig. 2).
Stratified and interaction analyses of the association between metabolic dysfunction-associated steatotic liver disease (MASLD) and incident obstructive sleep apnea (OSA). *Adjusted hazard ratios (aHRs) and 95% confidence intervals are shown across predefined subgroups, including sex, age, and smoking status. Estimates were derived from fully adjusted Cox proportional hazards models. P values for interaction represent effect modification between MASLD and each subgroup variable.
Graphical representation of risk gradients
Figure 3 presents restricted cubic spline analyses demonstrating positive associations between BMI, waist circumference, and OSA risk. The alcohol–OSA association exhibited a J-shaped pattern, with an apparent decline at very high intake due to sparse data and wider confidence intervals.
Restricted cubic spline of hazard ratio with 95% confidence intervals for OSA. *The model was adjusted for age, sex, income level, residence area, Charlson comorbidity index, hemoglobin level, glomerular filtration rate, and smoking and regular exercise status.
The Kaplan–Meier analysis (Fig. 4) illustrated clear visual differences in the cumulative incidence of OSA across the five exposure groups. The MASLD with alcohol and MetALD groups displayed the highest cumulative incidence throughout follow-up, followed by the MASLD without alcohol group. Both MASLD phenotypes showed higher incidence trajectories than the two non-SLD groups, with the lowest incidence observed in the reference category. Log-rank testing indicated statistically significant differences in OSA incidence across the groups (p < 0.001).
Kaplan–Meier curves for the association between SLD and OSA.
Discussion
In this large, nationwide cohort of Korean adults, we found that MASLD was associated with a significantly higher risk of incident OSA, whereas CMRFs in the absence of steatosis did not confer a statistically meaningful increase in risk. Although modest increases in OSA incidence were observed with a greater number of CMRFs among individuals without SLD, the lack of a clear dose–response pattern suggests that CMRFs alone may not fully capture the metabolic phenotype linked to elevated OSA susceptibility, consistent with prior research highlighting the multifactorial nature of OSA beyond traditional metabolic predictors1,2,3,4,5,6. In contrast, all MASLD and MetALD phenotypes exhibited significantly higher OSA risk, supporting the notion that the metabolic–hepatic phenotype identified by MASLD may serve as a more clinically informative marker of OSA risk than CMRFs alone7,8,9,10,15,16,−17.
Alcohol intake appeared to further modify OSA risk among individuals with MASLD. Although differences between alcohol consumption strata did not reach statistical significance in subgroup analyses, point estimates suggested a tendency toward higher OSA risk with increasing levels of alcohol exposure. This observation is consistent with established evidence indicating that alcohol increases upper-airway collapsibility and exacerbates sleep-disordered breathing18,19,20. Several factors may explain the lack of statistical significance, including reduced statistical power in the highest alcohol category (MetALD) and the well-known tendency for under-reporting of alcohol consumption in large-scale cohorts. Importantly, sensitivity analyses using stricter definitions of steatosis demonstrated a clearer alcohol-related gradient, reinforcing the possibility that alcohol may act as an additive contributor to OSA risk in the presence of metabolic liver disease. Notably, the HSI-based definition produced a higher effect estimate for MetALD (aHR 2.09), suggesting that indices with stronger metabolic weighting may better capture OSA-related risk among heavy drinkers.
The restricted cubic spline analyses further illustrated nonlinear associations between continuous metabolic indices and OSA risk. BMI and waist circumference showed strong, monotonic increases in risk, in line with extensive epidemiologic evidence identifying obesity as the most prominent modifiable determinant of OSA21–22. FLI demonstrated a positive association as well, but its interpretation warrants caution because FLI incorporates several metabolic components—including BMI, waist circumference, and triglycerides—that are themselves strongly related to OSA risk. Thus, the spline findings likely reflect composite metabolic burden rather than the isolated effect of hepatic steatosis. Nonetheless, the consistent elevation of OSA risk across MASLD phenotypes highlights the potential value of MASLD as a risk-identifying phenotype, echoing prior work on MASLD-related systemic inflammation, oxidative stress, and metabolic dysfunction6,9,15,23,24,25,26,27.
Kaplan–Meier analyses tended to show higher cumulative OSA incidence across the metabolic–hepatic phenotypes, with MASLD with alcohol and MetALD showing the steepest incidence curves over time. Model-based ARDs further supported these findings by providing a clinically interpretable measure of excess risk attributable to each phenotype. Although ARDs were modest, their consistency across multiple analytic approaches suggests that these estimates reflect true population-level associations. This pattern aligns with the high global burden of OSA1,2 and the increasing prevalence of MASLD across diverse populations11,12,13,14.
Several mechanisms may underlie the association between MASLD and OSA. Intermittent hypoxia, a hallmark of OSA, promotes hepatic steatosis progression, inflammation, oxidative stress, and fibrosis7,8,9,15,25,26,28, while metabolic dysfunction characteristic of MASLD may exacerbate ventilatory instability and impair neuromuscular control of the upper airway6,27,29. These overlapping pathophysiological pathways suggest a bidirectional relationship between OSA and metabolic liver disease, as proposed in recent reviews and expert consensus reports15,29. This notion is also supported by emerging work on liver–brain circadian signaling pathways30.
This study has notable strengths, including a large and nationally representative sample, standardized health screening data, rigorous phenotypic classification based on recent MASLD–MetALD consensus definitions11,12,13, and multiple complementary analytic approaches incorporating HRs, ARDs, and spline analyses. The long follow-up period and robust sensitivity analyses further enhance confidence in the observed associations. Notably, the association between MASLD and OSA remained consistent across multiple clinically relevant subgroups, supporting the robustness of our findings and mitigating concerns regarding residual confounding or exposure misclassification. However, limitations include reliance on surrogate indices rather than imaging for steatosis assessment, claims-based OSA diagnoses that likely underestimate mild cases, potential under-reporting of alcohol consumption, and residual confounding from unmeasured factors.
In conclusion, MASLD was associated with a higher risk of developing OSA, and alcohol exposure may further amplify this risk, particularly when steatosis is more severe or metabolic dysfunction is pronounced. These findings underscore the importance of recognizing MASLD—especially alcohol-associated phenotypes—as clinically relevant risk markers for OSA and support targeted screening strategies in this high-risk population.
Methods
Study population and design
This retrospective cohort study was based on data from the NHIS, a nationwide claims database that provides universal healthcare coverage. The database includes eligibility information, diagnoses (based on ICD-10 code), prescriptions, mortality data, and biennial national health screening results for adults aged ≥ 40 years. Health screening data comprise anthropometric measurements, laboratory test results, and standardized self-reported questionnaires on lifestyle behaviors (e.g., alcohol consumption, smoking and regular exercise status), with all components subject to national quality control standards. Using this dataset, we initially identified 377,641 individuals who underwent health examinations between January 1, 2009, and December 31, 2010. Among them, 86,577 were excluded due to diagnoses of viral hepatitis, autoimmune hepatitis, toxic liver disease, Wilson’s disease, biliary cholangitis, or other autoimmune and systemic inflammatory conditions with potential to cause chronic liver disease or confound the hepatic phenotype (detailed criteria provided in Supplementary Table 1). An additional 1,550 individuals were excluded due to a prior diagnosis of OSA. Further exclusions were applied for decompensated liver cirrhosis (n = 6,880), incomplete health screening data (n = 11,973), and extreme values of AST/ALT ratio (n = 5,209). After applying these criteria, a total of 265,452 individuals remained and were followed from the date of their baseline examination until the earliest of the following: diagnosis of OSA, death, or the end of the study period (December 31, 2019) (Fig. 1).
Group classification using CMRFs and FLI
The CMRFs used in this study were defined based on the five components of the diagnostic criteria for MASLD. These included (1) a BMI ≥ 23 kg/m2 or a waist circumference ≥ 90 cm for men and ≥ 85 cm for women, as specified in the Korean Obesity Study Guidelines; (2) a fasting blood glucose level ≥ 100 mg/dL or the diagnosis and treatment of T2DM; (3) a blood pressure reading of ≥ 130/85 mmHg or the use of antihypertensive medication; (4) a serum triglyceride level ≥ 150 mg/dL or the use of lipid-lowering therapy; and (5) an HDL-C level ≤ 40 mg/dL for men or ≤ 50 mg/dL for women, or the use of lipid-lowering therapy. The presence of at least one of the above criteria qualified an individual as having CMRF.
SLD was defined using FLI, a validated non-invasive marker calculated from BMI, waist circumference, triglyceride levels, and γ-GTP. A threshold of FLI ≥ 30 was used to define hepatic steatosis, in accordance with established epidemiological practice. Based on the presence or absence of SLD, CMRFs, and alcohol consumption, participants were categorized into five mutually exclusive groups: (1) individuals with no SLD and no CMRFs (FLI < 30 and no CMRFs); (2) individuals with no SLD but with one or more CMRFs (FLI < 30 and ≥ 1 CMRF); (3) individuals with MASLD without alcohol consumption (FLI ≥ 30 and ≥ 1 CMRF); and (4) individuals classified as MASLD with alcohol consumption below the MetALD threshold (FLI ≥ 30 and ≥ 1 CMRF, with alcohol intake < 210 g/week for men and < 140 g/week for women); and (5) individuals classified as MetALD, defined as hepatic steatosis with alcohol intake at or above 210 g/week for men and 140 g/week for women. This classification follows the recent international consensus definitions proposed by Eslam et al.11 and Rinella et al.13.
To further investigate the relationship between the number of accompanying CMRFs and the risk of OSA, the following subclassification was applied to non-SLD individuals according to CMRF burden, while MASLD and MetALD were analyzed as separate steatotic phenotypes: (1) those without SLD and without any CMRFs; (2) those without SLD but with one CMRF; (3) those without SLD and two CMRFs; (4) those without SLD and three to four CMRFs; and (5) those with MASLD, regardless of the number of CMRFs.
Outcome definition
OSA was defined using ICD-10 code G47.3, requiring (1) at least one inpatient or outpatient claim with G47.3 and (2) at least one additional G47.3-coded outpatient visit within 12 months to improve diagnostic specificity, consistent with the operational definition used in the Supplementary Table 1.
Covariates
Baseline demographic and clinical variables included age, sex, household income (quartile), region (urban vs. rural), and CCI. Comorbidities such as hypertension, T2DM, and dyslipidemia were identified using ICD-10 codes and prescription records. Laboratory and clinical measures included BMI, waist circumference, systolic and diastolic blood pressure, fasting glucose, lipid profile, liver enzymes (AST, ALT, γ-GTP), hemoglobin, and estimated glomerular filtration rate (eGFR). Lifestyle behaviors—including alcohol consumption, smoking and regular exercise status—were assessed through standardized self-reported questionnaires. Covariates were selected based on prior epidemiologic evidence linking demographic, lifestyle, and metabolic parameters with OSA risk, including age, sex, socioeconomic status, smoking and regular exercise status21,22. Alcohol consumption was additionally included to minimize confounding related to alcohol-associated liver injury, given its established effects on upper-airway collapsibility and its biological interaction with hepatic steatosis. Hemoglobin, eGFR, and the CCI were incorporated to account for systemic disease burden. To evaluate the robustness of our findings, supplementary models were constructed with further adjustment for BMI and individual cardiometabolic comorbidities, which allowed assessment of potential residual confounding arising from metabolic risk factors that are also components of MASLD diagnostic criteria. Detailed results of these supplementary models are presented in Supplementary Table 2.
Statistical analysis
Baseline characteristics were summarized as means (standard deviations) or frequencies (percentages), and between-group differences were evaluated using ANOVA or the chi-square test, as appropriate. Cox proportional hazards regression models were used to estimate HRs and 95% CIs for incident OSA, with the “no SLD without CMRF” group serving as the reference category. Multivariable models were adjusted for age, sex, income level, region, CCI, hemoglobin level, eGFR, smoking and regular exercise status.
To enhance clinical interpretability, model-based absolute risk and ARDs were additionally calculated. Absolute risk at the mean follow-up time of 9.5 years was estimated using the riskRegression and pec packages in R, holding covariates at their sample means to allow comparability across exposure groups. ARDs were defined as the difference in predicted absolute risk relative to the reference group. 95% CIs for ARDs were obtained through 500 nonparametric bootstrap resamples, in which the Cox model was refitted and absolute risks were recomputed for each resampled dataset.
Kaplan–Meier curves were generated to compare cumulative OSA incidence between groups, and differences were evaluated using the log-rank test. Restricted cubic spline regression was used to explore potential nonlinear associations between continuous metabolic parameters—including FLI—and OSA risk. Sensitivity analyses were performed to examine the robustness of the findings: first, using a stricter FLI cutoff (≥ 60), and second, redefining steatosis using a HSI ≥ 36. In additional sensitivity analyses, we performed stratified analyses according to age group, sex, obesity and central obesity, smoking status, and alcohol consumption. Interaction terms between MASLD and each subgroup variable were included in the fully adjusted Cox models to assess potential effect modification and the consistency of the association. All analyses were performed using SAS version 9.4 and R version 4.3.0. Statistical significance was defined as a two-sided p < 0.05.
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author, C.H.P., upon reasonable request.
References
Kazeminia, M. et al. Global prevalence of obstructive sleep apnea in the elderly and related factors: a systematic review and meta-analysis. Sleep. Health. 9, 232–241 (2023).
Iannella, G. et al. The global burden of obstructive sleep apnea. Diagnostics (Basel). 15, 1088 (2025).
Javaheri, S. et al. JACC state-of-the-art review: interactions of obstructive sleep apnea with the pathophysiology of cardiovascular disease. J. Am. Coll. Cardiol. 84, 1208–1223 (2024).
Koh, H. E. et al. Effect of obstructive sleep apnea on glucose metabolism. Eur. J. Endocrinol. 186, 457–467 (2022).
Wang, C., Tan, J., Miao, Y. & Zhang, Q. Obstructive sleep apnea, prediabetes and progression of type 2 diabetes: a systematic review and meta-analysis. J. Diabetes Investig. 13, 1396–1411 (2022).
Giampà, S. et al. Obstructive sleep apnea and metabolic syndrome: a review of pathophysiology and emerging systemic connections. Obes. (Silver Spring). 31, 2261–2270 (2023).
Tang, H., Lv, F., Zhang, P., Liu, J. & Mao, J. The impact of obstructive sleep apnea on nonalcoholic fatty liver disease. Front. Endocrinol. (Lausanne). 14, 1254459 (2023).
Hany, M. et al. Effects of obstructive sleep apnea on non-alcoholic fatty liver disease in patients with obesity: a systematic review. Int. J. Obes. (Lond). 47, 1200–1213 (2023).
Fu, Y. et al. Chronic intermittent hypoxia contributes to non-alcoholic steatohepatitis progression in patients with obesity. Hepatol. Int. 16, 824–834 (2022).
Song, D., Fang, G., Greenberg, H. & Liu, S. F. Chronic intermittent hypoxia exposure-induced atherosclerosis: a brief review. Immunol. Res. 63, 121–130 (2015).
Eslam, M. et al. A new definition for metabolic dysfunction-associated fatty liver disease: an international expert consensus statement. J. Hepatol. 73, 202–209 (2020).
Tilg, H. & Effenberger, M. From NAFLD to MAFLD: when pathophysiology succeeds. Nat. Rev. Gastroenterol. Hepatol. 17, 387–388 (2020).
Rinella, M. E. et al. A multisociety Delphi consensus statement on new fatty liver disease nomenclature. Hepatology 78, 1966–1986 (2023).
Chung, G. E. et al. Metabolic dysfunction-associated steatotic liver disease increases cardiovascular disease risk in young adults. Sci. Rep. 15, 5777 (2025).
Ji, Y., Liang, Y., Mak, J. C. W. & Ip, M. S. M. Obstructive sleep apnea, intermittent hypoxia and non-alcoholic fatty liver disease. Sleep. Med. 95, 16–28 (2022).
Huang, J. et al. Association between metabolic-associated fatty liver disease and obstructive sleep apnea: A cross-sectional study. Nat. Sci. Sleep. 15, 49–57 (2023).
Bettini, S. et al. Association of obstructive sleep apnea with non-alcoholic fatty liver disease in patients with obesity: an observational study. Eat. Weight Disord. 27, 335–343 (2022).
Simou, E., Britton, J. & Leonardi-Bee, J. Alcohol and the risk of sleep apnoea: a systematic review and meta-analysis. Sleep. Med. 42, 38–46 (2018).
Burgos-Sanchez, C. et al. Impact of alcohol consumption on snoring and sleep apnea: A systematic review and meta-analysis. Otolaryngol. Head Neck Surg. 163, 1078–1086 (2020).
Kim, H. J. et al. Enhanced alcohol metabolism and sleep quality with continuous positive airway pressure following alcohol consumption. Sci. Rep. 13, 16382 (2023).
Senaratna, C. V. et al. Prevalence of obstructive sleep apnea in the general population: A systematic review. Sleep. Med. Rev. 34, 70–81 (2017).
Peppard, P. E. et al. Increased prevalence of sleep-disordered breathing in adults. Am. J. Epidemiol. 177, 1006–1014 (2013).
André, S. et al. Cardiometabolic comorbidities in obstructive sleep apnea patients are related to disease severity, nocturnal hypoxemia, and decreased sleep quality. Respir. Res. 21, 35 (2020).
Gao, J., Shi, L., Zhu, X. & Liu, J. Association of obstructive sleep apnea with cardiometabolic diseases and cardiovascular mortality. Clin. Respir. J. 17, 764–770 (2023).
Lavalle, S. et al. Unraveling the complexities of oxidative stress and inflammation biomarkers in obstructive sleep apnea syndrome: A comprehensive review. Life (Basel). 14, 425 (2024).
Liu, X. et al. The relationship between inflammation and neurocognitive dysfunction in obstructive sleep apnea syndrome. J. Neuroinflamm. 17, 229 (2020).
Lv, R. et al. Pathophysiological mechanisms and therapeutic approaches in obstructive sleep apnea syndrome. Signal. Transduct. Target. Ther. 8, 218 (2023).
Zhao, Z. W. et al. Association between sleep apnea and ultrasound-defined liver fibrosis: results from NHANES 2017 to 2020. Med. (Baltim). 103, e37949 (2024).
Preshy, A. & Brown, J. A bidirectional association between obstructive sleep apnea and metabolic-associated fatty liver disease. Endocrinol. Metab. Clin. North. Am. 52, 509–520 (2023).
Gachon, F. et al. Potential bidirectional communication between the liver and the central circadian clock in MASLD. NPJ Metab. Health Dis. 3, 15 (2025).
Funding
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Ministry of Science and ICT (MSIT) (RS-2024-00342613); by the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health and Welfare (RS-2025-02223073); and by the Global Learning & Academic Research Institution for Master’s·PhD Students and Postdocs (G-LAMP) Program of the National Research Foundation of Korea (NRF), funded by the Ministry of Education (RS-2025-25440216).
Author information
Authors and Affiliations
Contributions
C.H.P. and S.Y.M. conceived and designed the study. B.K. and M.S. were responsible for data acquisition, analysis, interpretation, and drafting of the manuscript. All authors critically revised the manuscript, approved the final version, and agreed to be accountable for all aspects of the work to ensure its integrity and accuracy.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Park, C.H., Moon, S.Y., Kim, B. et al. Association between metabolic dysfunction-associated steatotic liver disease and obstructive sleep apnea: a nationwide retrospective cohort study. Sci Rep 16, 10572 (2026). https://doi.org/10.1038/s41598-026-46037-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-46037-4