Abstract
An ‘on-farm’ hybrid priming treatment, that combines hydropriming and biopriming with Bacillus subtilis spores was developed for the neglected legumes grass pea, forage pea, and fenugreek. The seed response to treatments highlighted the predominant effect of hydropriming. In the attempt to improve the efficacy of biopriming and enhance the overall value of the combined protocol, an in-depth investigation of the seed-spore interaction was performed. Scanning electron microscopy was used to analyse the seed sculpture roughness. Multispectral imaging analysis was applied to monitor the presence of increasing doses of Bacillus subtilis spores and test the potential of this tool for the optimization of biopriming protocols. Within the wavelengths applied using multispectral imaging, only 645 nm, 660 nm, and 690 nm were able to detect the presence of the spores in the bioprimed seeds, providing a range of analysis potentially effective for discrimination. Grass pea varieties, showing the most contrasting roughness profiles, were selected for multispectral imaging analysis. The investigation highlighted the relevance of the seed sculpture roughness in determining the reflectance of untreated and bioprimed seeds, and the occurrence of variety-specific profiles. Results are discussed in view of the potential use of this model to expand the current applications of multispectral imaging in the context of seed biopriming design.
Similar content being viewed by others
Introduction
The transition towards sustainable, climate-resilient agrifood systems is an essential component of the Sustainable Development Goals, with relevant implications at the socio-economic level1. Orphan crops, which are neglected or underutilized species, have a minimal market impact but are highly valuable to small local farmers in the most climate-vulnerable regions of developing countries2,3. The genetic resources of these minor crops, still under-investigated, represent a unique source of agrobiodiversity in terms of traits for abiotic and biotic stress resilience, and nutritional and medicinal properties4,5,6.
The roadmap towards ‘climate-ready’ crops requires the selection of highly resilient pre-breeding germplasm to be used for developing lines with improved stress tolerance7. Among neglected legume species, grass pea (Lathyrus sativus L.) has been included in the list of potential ‘climate-smart’ crops, due to its strong adaptation to adverse environments and high stress resilience. This annual cool-season grain legume crop has become a model for sustainable agriculture and cropping systems diversification in climate-sensitive regions7,8,9. Fenugreek (Trigonella foenum-graecum L.) is a multi-purpose crop that offers the value as a food, feed, spice and medicinal plant with enhanced tolerance to biotic and abiotic stress, leading to high yield stability under low-input agricultural systems10,11,12. Forage pea (Pisum sativum ssp. arvense L.) is a cool-season annual legume crop directly grazeable or harvestable and used for human consumption, as ruminal silage, or as green fertilizer13. The strong nutritional value and adaptation potential represent ideal features for sustainability of pasture systems and as part of crop rotations, ensuring resilient and productive farming practices14. Yield quantity and quality are however decreased in response to salinity and drought stresses15.
The availability of high-quality seeds, providing efficient germination even under climate crisis, has become an utmost priority, since germination and early seedling establishment are extremely sensitive to environmental stresses16. Technologies for seed quality enhancement have shown to contribute to boost the field performance of crop plants. In the specific case of orphan legumes, tailored seed priming protocols can further improve germination of high-quality seeds17. A careful design of priming approaches must consider crucial factors, such as type and concentration of priming agents, time and temperature of their application18. Hydropriming, biopriming and hybrid priming19,20 and chemical priming21 have been implemented in L. sativus seeds. As for Pisum sativum L., seed priming has been extensively documented for garden pea (var. sativum)22 whereas applications in forage pea (var. arvense) are scant23. Nanopriming, silicon-based seed priming, osmopriming, hormopriming, and chemopriming have been reported also for fenugreek24,25,26,27, and the overall benefits of these treatments on germination performance discussed28.
Among the seed priming technologies, biopriming exploits the potential of beneficial microbes (plant growth promoting bacteria, PGPBs) to promote growth and stress resistance29. The spore-forming Bacilli, including Bacillus subtilis, are prominent PGPBs well adapted to colonize dry and saline soils, supporting plant growth under stress conditions, and preventing pathogen infection30,31. These PGPBs promote seed germination, enhancing shoot and root growth. Biopriming can also be used in combination with other seed priming agents to develop novel hybrid priming strategies with a higher impact on seed performance19,32. Among the research questions regarding orphan crops that still need to be addressed, seed quality issues represent a crucial point. Innovation in seed technology is envisaged as an effective strategy to boost seed resilience and support productivity under the climate challenges.
In the present work, an ‘on-farm’ hybrid priming treatment was developed, tailored for seeds of three neglected legume species, namely grass pea, forage pea, and fenugreek. For each species, two varieties were investigated and tested with a hybrid priming protocol that combines hydropriming and biopriming with B. subtilis spores. The seed response to treatments was monitored in terms of germination performance and seedling phenotyping, highlighting the predominant effect of hydropriming. In the attempt to improve the efficacy of the biopriming treatment and, thus, enhance the overall value of the combined protocol, an additional investigation was performed, focusing on parameters that could influence the seed-spore interaction. Scanning electron microscopy (SEM) was used to visualize the seed sculpture and the associated roughness value, for each variety. Grass pea seeds of the tested varieties showed the most contrastive roughness patterns and for this reason they were selected for multispectral imaging analysis (MSI), in an attempt to support the design of improved biopriming treatments.
Materials and methods
Seeds and bacterial strain
Seeds of Lathyrus sativus L. var. ‘Maleme-107’ and var. ‘Sofades’ (Lat-M and Lat-S) were supplied by Hellenic Agricultural Organization - Demeter/Industrial & Forage Crops Institute (Larissa, Greece) and Agroland S.A. (Sofades, Greece). Seeds of Pisum sativum var. arvense var. ‘Forrimax’ and var. ‘Guifredo’ (Pis-F and Pis-G) were supplied by Semillas Batlle S.A. (Barcelona, Spain). Seeds of Trigonella foenum-graecum L. var. ‘Rayhane’ and var. ‘Tborsek’ (Tri-R and Tri-T) were provided by INGC (Institut National des Grandes Cultures, Bou Salem, Tunisia) (Fig. 1A). The seed lots used in this study were harvested in 2023. Bacillus subtilis strain NCBI 3610 (ATTC 6051) was obtained from the Bacillus genetic stock center (https://bgsc.org). Spores were obtained by nutrient exhaustion as previously described19.
(a) Seeds of Lathyrus sativus L. ‘Maleme-107’ and ‘Sofades’ (Lat-M and Lat-S), Pisum sativum var. arvense ‘Forrimax’ and ‘Guifredo’ (Pis-F and Pis-G) and Trigonella foenum-graecum L. ‘Rayhane’ and ‘Tborsek’ (Tri-R and Tri-T). (b) The experimental system included four different treatments: non-primed control (NP, T1), hydroprimed (HP, T2), bioprimed (NP-ST-SP, T3), and hybrid primed (HP-ST-SP, T4) seeds. NP and HP seeds treated with the sticker solution (ST) are also indicated as NP-ST and HP-ST, respectively. HP was applied for 8 h followed by dry-back (24 h). Germination was monitored every 12 h for seven days.
Hydropriming, biopriming and hybrid priming treatments
The experimental system included four different treatments: non-primed control seeds (NP, T1), hydroprimed seeds (HP, T2), bioprimed seeds (NP-ST-SP, T3), and hybrid primed seeds (HP-ST-SP, T4) (Fig. 1B). Additional controls were tested, namely non-primed and hydroprimed seeds treated with the sticker solution (ST) (10% w/v sucrose; Sigma Aldrich, Milan, Italy), indicated as UP-ST and HP-ST, respectively. All treatments were performed at room temperature (25 °C). For hydropriming and hybrid priming, 180 seeds for each variety were soaked in 0.5 l H2O for 8 h in a container with a volume of 2.5 l, to prevent imbibed seeds from overflowing during the treatment. Stirring was performed every 2 h to reduce anoxic conditions. Hydroprimed seeds were then spread on a layer of absorbing paper and air-dried for 24 h at 25 °C, 50% relative humidity (RH). B. subtilis spores were applied to dry untreated seeds (T3, biopriming protocol) and on seeds previously subjected to hydropriming (T4, hybrid priming protocol). The number of spores required for seed treatments was calculated as described33,34. A total of 4 × 106 spores/seed (for L. sativus and P. sativum var. arvense) and 2 × 104 spores/seed (for T. foenum-graecum) was selected based on average seed weight (Table S1). Dry spores were resuspended in the sticker solution (2 × 106 spores/µl, density for Lat-M, Lat-S, Pis-F, and Pis-G; and 2 × 104 spores/µl, density for Tri-R and Tri-T). Dry seeds were mixed with the appropriate amount of B. subtilis spores suspension using a metal spatula until the seeds were uniformly coated with the suspension and subsequently air-dried for 10 min. For multispectral imaging (MSI), the experimental system was set, using Lat-M and Lat-S. For each variety, 24 seeds were used for each treatment. Untreated, non-primed seeds (NP) and seeds treated with the sticker (10% sucrose, ST) were used as controls. Bioprimed seeds were treated with increasing spore concentrations (0.5x, 2 × 106 spores/seed; 1x, 4 × 106 spores/seed; 2x, 8 × 106 spores/seed; 4x, 1.6 × 107 spores/seed). These concentrations were selected to evaluate whether the multi-spectral imaging (MSI) approach could represent a feasible and sensitive tool for the detection of B. subtilis spores on the seed surface. Additional germination parameters were measured (Table S2) whereas no impact on germination performance and seedling growth was observed when the sticker was applied alone, as control (Table S3). For MSI analysis, seeds were collected at 0 h and 6 h from the beginning of imbibition, carried out as previously described35.
Germination tests
For germination tests, primed and non-primed (control) seeds were transferred into aluminum trays (12 cm length, 8 cm width, 5 cm height) containing sieved soil (COMPO Italia Srl, Cesano Maderno, Italy). The sieved soil was not sterilized and it was weighed using a SLMAT 200 g balance (Selling Depot Italia s.r.l., Piacenza, Italy). Soil composition was neutral sphagnum peat, compost soil improver, clay, pH 7, 90% porosity. The seeds were sown at 1 cm depth. The soil was moistened with 30 ml water and watered daily afterwards to avoid drying. The trays were kept in a growth chamber at 25 °C under light conditions with a photon flux density of 150 µmol m− 2 s− 1, a photoperiod of 16/8 h, and 70–80% relative humidity (RH). Seedling emergence, namely the phenological event when seedlings break through soil and become visible above ground, was monitored every 12 h for seven days. After seven days from the beginning of the germination test, seedlings were carefully removed from the trays and soil was washed off using water in a tray to avoid damaging or loosing roots. Biometrical analysis was performed measuring the length and fresh and dry weights of the aerial parts and roots, using a digital balance. For each condition, three independent replicates (trays) each containing 20 seeds were considered for statistical analyses. Mean germination time (MGT) was calculated according to Ranal & de Santana (2006)36.
Electron Scanning Microscopy (SEM)
A Zeiss EVO MA10 (Carl Zeiss, Oberkochen, Germany) microscope was used to perform scanning electron microscopy (SEM) measurements. For each accession and treatment, dried hydroprimed seeds, randomly sampled, were sputtered with gold and analysed in different regions (at least three zones for each seed) with secondary electrons (SE) at a working distance of 8.5 mm and a voltage of 5 kV.
Multispectral imaging
Images from each seed sample were captured using a VideometerLab4 instrument (Videometer A/S, Hørsholm, Denmark) as reported by ElMasry et al. (2021). The system is supplied with a series of light-emitting diodes (LEDs) at 20 different wavelengths (365, 405, 430, 450, 470, 490, 515, 540, 570, 590, 625, 630, 645, 660, 690, 780, 850, 880, 940, and 970 nm) that can be sequentially flashed one after another to record monochromatic images by a CCD camera at these pre-defined wavelengths. The resulting multichannel image is a cube image in HIPS format with a spatial dimension of 2992 × 4096 pixels, 30 μm/pixel of spatial resolution and 20 bands in the spectral dimension. Seeds (24 for each treatment) were placed on a tray with a blue background, in a 4-row by 4-column arrangement to ensure adequate distance among seeds and one multispectral image was acquired at each of the 20 wavelengths. Images data extraction and transformation of pixel data were conducted through the VideometerLab software version 3.26.50. The region of interest (ROI) for the reflectance measurements was defined by highlighting the whole area of the seeds that was facing the MSI camera. The measurement of mean reflectance across the 20-wavelength used by the Videometer machine was conducted per each seed. Reflectance percentage from each of the 24 seeds was obtained at 0 h and 6 h of imbibition. Reflectance percentage was used as a parameter to quantify the ability of the seed surface to reflect the radiation based on the ratio of the radiant flux reflected from a surface, Φr, to that of the radiation flux, Φi. Mean reflectance percentage was used according to Vrešak et al. (2016)37. Additionally, individual seed surface area was also measured using a similar approach to investigate any effects of surface area on the data collected for the MSI analysis38.
Seed surface topography and roughness analyses
The topography and roughness of the seed surface were assessed using SEM images through a quadrant approach, using the software ImageJ (ImageJ, Maryland, USA; https://imagej.net/ij/). A selection box with dimensions of 26 μm (width) and 20 μm (height) was placed on specific X and Y coordinates to subdivide each image in four quadrants. The X and Y coordinates for each quadrant were: 0.26 × 0.53 (Q1), 0.99 × 24.57 (Q2), 40.93 × 0.86 (Q3), and 41.39 × 24.37 (Q4). The contrast was enhanced using default settings. Afterwards, a 3-dimensional model was reconstructed through the interactive ImageJ 3D surface plot plug-in following default settings which were applied to all analysed images. The settings used were: 512 (gride size), 45 (smoothing), 0.52 (perspective), 0.92 (lighting), 0.68 (scale), 0.81 (z scale), 100% (max slider limit of the height range of the plot data) and 9% (min slider limit of the height range of the plot data). The roughness estimate of the seed surface was performed using the roughness calculation of ImageJ, focusing on roughness parameters Sq (Root Mean Square) and Ra (Arithmetic Average)39. Sq roughness is used to characterize the surface roughness, obtained by squaring each height value in the dataset, then taking the square root of the mean. Ra indicates the average surface roughness for the length of the measurement performed, such as the average difference between peaks and valleys. The Ra and Sq parameters were selected from the roughness calculation plug-in available in the ImageJ software. This is due to the direct link with the roughness estimation based on the height of peaks and depth, based on the gray values of valleys calculated by the software (through the plug-in) from the 2D pictures acquired through the scanning electron microscope (SEM).
Statistical analysis
Data were analyzed by two-tailed Student’s t-Test with selected pairwise comparisons to highlight the effects of the treatments (T2, T3 and T4) compared with untreated controls (T1) and the effects of hybrid priming (T4) compared to hydropriming (T2), with P-values lower than 0.05 were considered statistically significant. A One-way (https://houssein-assaad.shinyapps.io/TableReport/) and Two-way (https://houssein-assaad.shinyapps.io/TwoWayANOVA/) ANOVA was performed on the data for the multi-spectral imaging (MSI) and seed surface topography and roughness analyses with a post-hoc Duncan test40. The Duncan test, commonly used in experimental designs for agricultural, biological, and food science research, reduces the chance of Type II errors and it groups treatments into statistically homogeneous subsets, thereby rendering an easy interpretation of significance.
Results
Hydropriming is the most effective component of hybrid priming
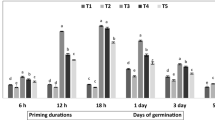
Soil emergence was measured for each variety and treatment group. As for T1, soil emergence was higher than 75% for all tested accessions (Lat-M, 95.00 ± 0.00%; Lat-S, 100 ± 0.00%; Pis-F, 86.67 ± 7.64%; Pis-G, 76.67 ± 18.93%; Tri-R, 88.33 ± 5.77%; Tri-T, 90.00 ± 13.23%) and none of the treatments induced statistically significant changes in soil emergence (Table S2). The addiction of the sticker solution to T1 and T2 did not induce significant changes in soil emergence (Table S3). Hydropriming (T2) was able to significantly (P < 0.05) decrease MGT in both grass pea Lat-M and Lat-S (80.21 ± 2.53 h and 73.93 ± 1.20 h, respectively), compared to untreated controls (T1) (91.79 ± 3.59 h and 81.00 ± 2.16 h, respectively), and a comparable accelerating effect was also maintained in the hybrid priming treatment (T4) (Fig. 2A). The only significant effect (P < 0.05) was observed for T2 in Pis-F, with a 15.4% decrease in MGT compared to T1 (from 74.27 ± 4.37 h to 62.80 ± 0.78 h). T4 significantly accelerated germination in Pis-G (83.06 ± 0.82 h), whereas, with respect to T1, a non-significant decrease was observed for both Pis-F in response to T4 and Pis-G in response to T2 (Fig. 2B). In the case of fenugreek, T2 was able to significantly decrease MGT in Tri-R accession (78.92 ± 4.86 h, P = 0.013), compared to T1 (95.29 ± 4.63 h). Comparable significant responses were observed for T4 (P = 0.009) (Fig. 2C). A positive impact of T2 was also observed in fenugreek Tri-T accession compared to T1 (69.00 ± 1.59 h vs. 83.01 ± 5.81 h, respectively; P = 0.044), with a non-significant acceleration observed for T4 (Fig. 2C).
Results from phenotyping highlighted the impact of all the priming protocols on seedling development. In the case of grass pea, shoot length was significantly (P < 0.05) enhanced in response to all treatments for Lat-M, and only to T2 and T4 for Lat-S. At the root level, only Lat-S positively responded to all treatments (Fig. 2D). As for seedlings developed from forage pea primed seeds, Pis-F revealed decreased shoot growth in response to all treatment (Fig. 2E). Tri-R showed a significant enhancement of root growth in response to T2 and T4 (Fig. 2F).
Impact of seed priming on the germination performance of Maleme-107 and Sofades (Lat-M and Lat-S) (a), Forrimax and Guifredo (Pis-F and Pis-G) (b) and Rayhane and Tborsek (Tri-R and Tri-T) (c). Impact of seed priming on seedling development of Lat-M and Lat-S (d), Pis-F and Pis-G (e), and Tri-R and Tri-T (f). T1, control non-primed. T2, hydropriming. T3, biopriming. T4, hybrid priming. MGT, Mean Germination Time. Data are presented as mean values ± standard deviation. Asterisks indicate statistically significant difference according to Student’s t-test. *, P < 0.05; **, P < 0.01; ***, P < 0.001.
Scanning electron microscopy reveals distinctive seed sculptures resulting into accession-specific surface roughness profiles
The morphology of the orphan legume seeds, particularly the seed coat topography and the surface of the outer cell wall were investigated using SEM, in an attempt to highlight some distinctive features that might influence spore adhesion and, hence the efficacy of biopriming. A tuberculate sculpture was observed for Lat-M (Fig. 3A), characterized by the occurrence of ribbed, densely distributed papillae, with peaks possibly topped with wax, as described by Gunes et al. (2011)41. The ribs appeared straight, narrow and slightly protruding42. A similar pattern was observed for Lat-S, although papillae are less visible and covered by a dusty layer, with granules of different size and shape42(Fig. 3B). In the case of the forage pea, SEM revealed a typical papillose coat ornamentation with thick ribs43(Fig. 3C and D). For fenugreek seeds, the observed micro-ornamentation on the surface of the outer cell wall was classified as ridged-tuberculated-verrucated or mounded with papillae as reported by Ceter (2012)44 and Turki et al. (2013)45(Fig. 3E and F), with a slight but visible differences between the two varieties, with Tri-T displaying a more ridged seed surface. The spores were able to adhere to the seed surface in all the tested accessions regardless of the seed sculpture (Fig. S1).
Seed coat pattern and the surface of the outer wall investigated using SEM in Maleme-107 (Lat-M) (a) and Sofades (Lat-S) (b), Forrimax (Pis-F) (c) and Guifredo (Pis-G) (d) and Rayhane (Tri-R) (e) and Tborsek (Tri-T) (f). Photographs were taken from dried non-primed seeds (T1).
A semi-quantitative 3D-imaging approach was applied to the SEM images captured from orphan legumes seeds in order to evaluate the surface roughness, as a parameter that might influence the seed-spore interaction. As shown in Fig. 4(A, B), for each grass pea variety an area of 3060 µm2 of the seed surface was considered, starting from a selected SEM image. The region was then split into four quadrants (Figs. 1, 2, 3 and 4, A and B) that were analysed to reconstruct the corresponding 3D models. Values of roughness-related parameters were obtained on Lat-M and Lat-S seeds: (i) Sq (Root Mean Square) one of the dispersion parameters use to characterize the surface roughness, (ii) Ra (Arithmetic Average). Lat-M and Lat-S seeds are different in terms of surface roughness, as the Sq was significantly (P < 0.05) higher in Lat-S (144 ± 1.12) compared to Lat-M (140 ± 0.91) whereas no significant differences were observed for the Ra parameter (Fig. 4C). When comparing Pis-F and Pis-G, no significant differences were observed in the estimated Sq and Ra values, indicating that both varieties possess the same seed sculpture and equal roughness (Fig. S2). In the case of fenugreek, a slight but significant difference in the estimated Sq and Ra values was detected (Fig. S3), as also evidenced in Fig. 3E and F. The reported differences in seed sculpture and roughness pose the question about the potential impact of these genotype-specific structural features on the efficacy of the spore-seed interaction, particularly for the spore adhesion.
The seed sculpture of Lat-M (a) and Lat-S (b) as evidenced by SEM. The roughness profiles are shown (c). Sq, Root Mean Square Roughness. Ra, Arithmetic Average Roughness. a.u., arbitrary units. For each parameter of each accession, average values without common letters are significantly different (P < 0.05) as analyzed by one-way ANOVA.
Seed sculpture has an impact on the multispectral imaging ability to discriminate bioprimed grass pea seeds
For each grass pea variety, untreated seeds (NT), seeds treated with the sticker only (10% sucrose, ST), and bioprimed seeds treated with increasing B. subtilis spore concentrations were analysed. At the beginning of imbibition (0 h), the reflectance spectra showed treatment-dependent variability across the tested wavelength range (from 365 to 970 nm) (Fig. S4, A and B). Changes in mean reflectance in the visible range (365–780 nm) can be ascribed to differences in the seed colour, whereas changes in the NIR (Near Infra-Red) region (780–970 nm) result from differences in physico-chemical properties46. When considering the entire spectrum, both Lat-M and Lat-S seeds showed a similar trend in their reflectance patterns. In the case of Lat-M, the reflectance curves of NT seeds appeared to overlap with those of treated seeds with a change in slope only visible in the region spanning from 645 to 690 nm (Fig. S4, A). The reflectance curves of NT and treated Lat-S seeds were more distinguishable across the entire spectral range, however a difference was more evident in the region spanning from 645 to 690 nm (Fig. S4, B). Zooming into this region allowed to detect differences in reflectance profiles that could be useful to specifically discriminate bioprimed from untreated seeds (Fig. 5, A and B). In the case of Lat-M, the mean reflectance profiles were not able to clearly discriminate between control and bioprimed seeds (Fig. 5A). The mean reflectance of the sticker sample almost overlapped the value observed for the seeds treated with the 0.5x spore dose and those differences were maintained across the 645 and 690 nm range (Fig. 5B). MSI was able to distinguish between control and bioprimed grass pea seeds within the specific wavelength range of 645, 660, and 690 nm. The intrinsic variability at the single seed level might influence the reflectance values. To assess single seed variability, the distribution of reflectance for each of the 24 seeds tested was analysed (Fig. S5), showing that the resulting patterns were aligned with the data previously reported for both accessions at the three different wavelengths. Another relevant difference between the two accessions deals with the spore dose detected by MSI. For Lat-M, the discrimination between control and bioprimed seeds was observed only when the 0.5x spore dose was applied (Fig. 5A) whereas for Lat-S the discrimination between control and bioprimed seeds was observed only when the 1x spore dose was used (Fig. 5B). It is also worth noting that, in Lat-M, the recorded mean reflectance value for the untreated (NT) seeds was always lower than the values detected for the treated seeds. In the case of Lat-S, an opposite pattern was observed since the recorded mean reflectance value for the untreated (NT) seeds was always higher than the values detected for the treated seeds (Fig. 5A and B). The MSI profiles of NT seeds can be explained considering the different surface roughness levels previously calculated for the Lat-M and Lat-S (Fig. 4). Given the inverse correlation existing between surface roughness and reflectance percentage, it is correct to observe a decrease in mean reflectance percentage for seeds with higher surface roughness or viceversa.
The same analysis was performed using seeds collected at 6 h of imbibition. The reflectance spectra showed variability across the tested wavelength range (from 365 to 970 nm) (Fig. S6). For both Lat-M and Lat-S, the reflectance curves of NT and treated seeds appeared to differentiate within a narrow region covering the 645, 660 and 690 nm wavelengths (Fig. 5, C and D). The mean reflectance of the sticker sample (ST) overlapped the value observed for the seeds treated with the 1x spore dose, as already reported at 0 h. At 645 and 690 nm, the resulting reflectance profiles were able to discriminate between control and bioprimed seeds (Fig. 5C). As for Lat-S, at 645 and 690 nm, similar profiles were recorded, useful to discriminate between control and bioprimed seeds (Fig. 5D). At 6 h from the beginning of imbibition, the most discriminating wavelength was 660 nm for both varieties, however for Lat-S also 645 and 690 nm proved effective. To figure out the possibility that the intrinsic variability at the single seed level might influence the reflectance values previously reported, the distribution of reflectance for each of the 24 seeds tested was analysed, showing that the resulting patterns were aligned with the data previously reported (Fig. S7). At 6 h of imbibition, the divergent reflectance profiles of NT seeds were still mainained (Fig. 5C, D), due to the variety-specific seed surface morphologies. No influence of single seed area on the mean reflectance recorded by MSI at 0 h and 6 h, for each treatment, was observed (Fig. S8).
Average spectral fingerprints of grass pea seeds (Lat-M and Lat-S) recorded at the beginning of imbibition (0 h) (a and b) and at 6 h of imbibition (c and d), respectively, at 645, 660, and 690 nm. NT, untreated seeds. Sticker, seeds treated with 1% sucrose. Bioprimed seeds were treated with increasing B. subtilis spore concentrations (0.5x, 2 × 106 spore/seed; 1x, 4 × 106 spore/seed). For each parameter of each accession, average values without common letters are significantly different (P < 0.05) as analyzed by one-way ANOVA.
Changes in seed surface roughness impact reflectance, determining dose-dependent responses
Although MSI was able to discriminate between NT and bioprimed seeds of both grass pea varieties, a dose-dependent response was observed. As previously reported, MSI identified the presence of the spores in a dose-dependent manner but the spore concentration recognised by MSI differed for the two varieties. To gain a more precise view of the seed-spore interactions in this context, a semi-quantitative 3D-imaging approach was applied to the SEM images captured from Lat-M and Lat-S seeds treated with the 1x spore concentration (Fig. 6). For each grass pea variety an area of 3060 µm2 of the seed surface was considered, starting from a selected SEM image. The region was then split into four quadrants (Fig. 6A, B) that were analysed to reconstruct the corresponding 3D volume models. Sq and Ra parameters calculate the roughness profile of Lat-M and Lat-S seeds. As evidenced in Fig. 6C, the Sq was significantly (P < 0.05) lower in Lat-M bioprimed seeds compared to Lat-S. The Ra values also showed significant difference (P < 0.05) between Lat-M bioprimed seeds compared to Lat-S. Thus, the presence of spores changes the seed surface roughness and consequently the recorded reflectance. In the specific case of Lat-M, MSI values recorded at 660 nm were not able to discriminate between seeds treated with the sticker only and seeds treated with spore at the 1x dose (Fig. 6C): the sticker alone changed the seed surface roughness, compared to NT seeds, however when the 1x dose was administered, this spore/sticker suspension did not further change the surface roughness. In the case of Lat-S, the mean reflectance percentage recorded at 660 nm for seeds treated with sticker showed similar values compared to seeds treated with 0.5x spore dose (Fig. 6C). This can be due to the presence of the sticker alone that changed the seed surface roughness when compared to NT seeds, but the 0.5x spore/sticker suspension did not modify the surface roughness. The reported data highlight the relevance of the effects associated with the application of the sticker solution, that can itself impacts the seed surface roughness and influence the MSI values. Thus, the chemical-physical features of different types of sticker solutions (components, concentrations) should be carefully considered in this context.
The seed sculpture of Lat-M (a) and Lat-S (b) bioprimed seeds, treated with 1x spore dose, as evidenced by SEM. The roughness profiles are shown (c). Sq, Root Mean Square Roughness. a.u., arbitrary units. Ra, Arithmetic Average Roughness. For each parameter of each accession, average values without common letters are significantly different (P < 0.05) as analyzed by one-way ANOVA.
Discussion
The need of high-quality seeds, to boost vulnerable crops yields under climate change, must be addressed by exploring new avenues to improve current seed priming technologies. It is necessary to identify and understand any possible source of undesired variability, by exploring the different parameters and variables that may influence the success of seed priming treatments. In the present work, hybrid seed priming combining hydropriming and biopriming was applied to different accessions of the orphan legumes grass pea, forage pea and fenugreek. Germination tests, performed in soil under controlled conditions, highlighted the major effects of hydropriming (Fig. 2). Hydropriming represents the most effective and sustainable option for direct ‘on-farm’ treatments16,47, and these results further reinforce the effectiveness of this treatment on the target orphan legumes. However, the effect of hydropriming could be further boosted if combined with optimized biopriming. Indeed, while hydropriming promotes the seed repair response during early imbibition, biopriming provides support for growth and nutrient up-take in the later phase of germination and seedling development17,29,48. The biopriming treatment had no impact on the germination performance, however both shoot and root growth was enhanced in seedlings developed from seeds bioprimed with B. subtilis spores from strain NCBI 3610 (Fig. 2). The beneficial effects of B. subtilis as seed biopriming agent have been described in many plant species, including garden pea (Pisum sativum L.)49, common bean (Phaseolus vulgaris L.)50, Indian mustard (Brassica juncea L.)51, fenugreek and tomato (Lycopersicon esculentum L.)52. The positive impact of B. subtilis spores can also appear after radicle emergence, due to specific interactions with the seedling root tissues. According to Berlanga-Clavero et al. (2022)53, application of B. subtilis NCIB 3610 to melon (Cucumis melo L.) seeds resulted in larger radicles of five day- and 1 month-old plants, compared with those developed from untreated seeds, although no changes in germination performance were observed, compared to the untreated control. According to this study, bacterial cells accumulated in the storage tissues close to micropyle, a entry point for bacteria into seeds, exerting beneficial effects associated with ROS accumulation during seed imbibition and specific metabolic changes. It is also possible that hydropriming promotes a more conductive environment for B. subtilis colonization and beneficial activities. Furthermore, concerning the long-term impact of B. subtilis in the rhizosphere as well as in the interaction at the seedling level, it should be underlined that bacterial populations were recovered after four months from tomato seed inoculation54, and that the addition of single or multiple Bacillus species to sprouting broccoli roots was able to influence the composition of endophytic bacterial communities55.
The beneficial effects of seed treatments with B. subtilis spores are particularly evident under adverse conditions, as salinity and drought stresses49,56,57. In the specific case of grass pea accessions investigated in the present study, Lat-M and Lat-S, the seed bioproming treatment with spores from the same strain was able to enhance salt stress tolerance in plants challenged with two salinity regimes (80 and 160 mM NaCl), with a consistent impact on root development19. Although hydropriming was the main driver of improved germination performance and seedling growth, the potential value of hybrid priming may be underestimated since, in this study, all the treatments were performed under controlled conditions. Therefore, the potential for the biopriming component to significantly contribute under stress conditions warrants further investigation, as this study was conducted under controlled environments.
Attention was focused on seed sculpture: SEM analyses highlighted some distinctive features of seed surface sculpture not only at the species level but also within accessions of the same species (Fig. 3and Fig. 4). Nonetheless, it is still difficult to assess whether the observed morphological differences on seeds surface may positively or negatively influence the spore ability to become attached to them, but a correlation between these parameters might become a useful tool to tailor the biopriming system, and understand the dynamics of the interaction between spores and seeds. Based on these premises, seeds with certain roughness profiles could be preferentially selected for biopriming or tailored seed treatments could be designed taking into account the seed surface features.
It has been reported that sporulation conditions can influence the adhesion properties of spores58. On the other hand, the way spores are delivered to the seed surface, namely the inoculant formulation, requires a careful design. A precise delivery system, based on the application of exogenous material onto the seed surface, must consider the main seed features such as shape, size, and weight59. The formulation hereby applied to the orphan legume seeds was specifically designed to deliver ‘on-farm’ seed priming strategies, i.e. an easy-to-manage operational protocol suitable for farmers’ needs17,47. The amount of B. subtilis spores was calculated according to Lobo et al. (2019)34, based on seed size that was classified as large (100–180 mg, grass pea and forage pea) and small (20 mg, fenugreek), respectively. The exploitation of imaging tools to identify the best performing formulations would simplify the effective application of biopriming. Particularly, the efforts required to adjust specific components (e.g. bacterial spores of a specific strain or consortia) and dose (optimized inocula) would be reduced, as well as the number of related variables. Based on these premises, an additional investigation was performed to assess the potential of multispectral imaging to discriminate between non-treated and bioprimed seeds, as well as between different concentrations of the applied spores. To this purpose, a dedicated experimental system was designed, using the grass pea Lat-M and Lat-S seeds previously characterized for their peculiar seed sculptures. Multispectral imaging, over the wavelengths hereby tested, was previously reported as an effective approach to test seed health and detect fungal infections in cowpea (Vigna unguicolata L.)60, peanuts (Arachis hypogaea L.)61, and other relevant species62.
The use of MSI has been extended to the evaluation of seed quality, based on the automated computer-vision of germination tests and the use of spectral signatures of single seeds as hallmarks for classifying seeds into different categories according to age, viability, seedling condition and speed of germination63. To date, no reports are available concerning the use of MSI to assist the design and optimization of biopriming protocols with bacterial spores. Results from the MSI analyses applied to Lat-M and Lat-S highlighted the potential of this tool to discriminate between control and bioprimed seeds, although variety-specific profiles were observed (Fig. 5). The semi-quantitative analysis of the 3D models generated from the SEM images of the seed sculpture allowed to describe the landscape on which the B. subtilis spores attached, in terms of roughness. It is known that a rougher surface has a lower reflectance than a smoother surface because roughness results in light scattering towards many directions, leading to multiple reflection and absorption events64. Thus, it is possible that the high roughness of the Lat-M seed sculpture, caused by the high-density protruding papillae, has a low reflectance due to increased light dispersion. This properly explains the opposite reflectance profiles observed for Lat-M and Lat-S NT seeds. This, in turn, can explain the increased reflectance values of the bioprimed seeds.
The reported data highlight the potential of MSI to reveal the presence of bacterial spores on bioprimed grass pea seeds, expanding the range of possible applications of biopriming in seed technology as well as the number of target crop species. The current biopriming protocols are developed according to a conventional workflow in which the combination between bacterial spores and seeds is determined empirically, based on the scientific literature or other technical documents as well as ‘trial and error’ steps required to adapt the protocol for the best performance (Fig. 7A). The novel biopriming workflow hereby proposed exploits (i) SEM technology to gain the quantitative profiles of innate seed roughness and (ii) MSI as a tool to measure the spore concentration, based on the reflectance value (Fig. 7B). The inverse correlation between these two parameters provides the rationale for the MSI-based analysis of bioprimed seeds. Seed surface roughness offers more precise indications about the range of spore doses that may effectively adhere to the seed whereas MSI can provide indications about the spore persistence, further narrowing the range of most suitable combinations. The added value of the proposed workflow refers to the possibility to avoid the ‘trial and error’ steps, currently needed to screen for the best performing spore/seed conbinations, speeding up the overall procedure. Given the complexity of the seed features and the variability observed in the seed response to priming treatment at both genotype and seed lot level, novel case studies shoud be expected, bringing unique features and highlighting novel relevant correlations.
Since the ability to distinguish between control and bioprimed seeds relies on the use of specific wavelengths, it is also essential to define the biophysical and biochemical bases allowing such wavelengths to be informative. The selected wavelengths, 645, 660, and 690 nm, detect the presence of chlorophyll pigments in seeds, with specific peaks around 645 nm and 662 nm65. The occurrence of chlorophyll in seeds might possibly interfere with the correct profiling of the attached bacterial spore. On the other hand, increased chlorophyll levels are typical of immature seeds and negatively correlate with seed quality, and this parameter should be also used to discriminate the seed lots suitable for treatments66. In a recent study, Ou et al. (2025)67 used MSI to discriminate bromegrass (Bromus inermis L.) seed vigour based on lipid and water content measured in the 780–970 nm range, excluded in the present investigation. The wavelengths hereby selected as the most informative for the system established with grass pea seeds can be regarded as a proper reference for the future validation across genotypes and species. However, due to the variety of seed morphological features and other components (type of sticker and spores) contributing to the overall MSI-based outputs, it is possible that further optimization will be required. The expectation is that this model might lead to advances in the design of seed biopriming treatments. The screening of the seed sculpture, expanded to a wide range of target germplasm, may be used to group genotypes sharing the same structural features whereas roughness and reflectance data will be the starting point for reference graphics (Fig. 7B).
Future efforts towards a better implementation of this tool will require to consider the innate features of the seed sculpture to optimize the administration and retention of biopriming agents and the application of coating/pelleting formulations. Moreover, the genetic and genomic tools recently developed for underutilized species can provide valuable resources and knowledge for more efficient management of novel variation for seed traits in gene banks and for the development of improved cultivars with specific seed characteristics that favor seed priming and are essential to reaching the United Nations’ Sustainable Development Goal of “zero hunger” by 203062. Climate-ready cultivars could benefit from breeding programmes focused on the selection of traits that support an effective response to seed priming, such as those traits related to improved antioxidant defences (both enzymatic and non-enzymatic components), genotoxic stress respose and desiccation tolerance.
(A) Schematic representation of the workflow currently used to design optimised biopriming protocols with Bacillus spp. spores. (B) Schematic representation of the proposed workflow that exploits SEM technology to gain the quantitative profiles of innate seed roughness and MSI as a tool to measure the spore concentration, based on the reflectance value.
Conclusion
The main novelty of the approach hereby tested lies in the break-through combination of SEM and MSI methodologies, used to deliver an in-depth profile of specific seed features relevant to develop effective biopriming protocols. The present study brings a significant contribution to the current state of the art in the field of seed technology, providing evidence of an innovative approach for the upgrade of biopriming treatments applied to seeds. The key findings relate to (i) the relevance of seed surface morphology, expressed in terms of roughness, as a parameter driving the dynamics of spore adhesion, (ii) the correlation of this parameter with MSI-based reflectance, and (iii) the possibility to exploit such a correlation to speed-up the large scale-screening of optimized biopriming protocols. Expanding the model to other crop species and genotypes and to other valuable Bacillus strains will generate data to feed algorithms for machine learning. Potential pathways for field validation can be envisaged, e.g. optimised biopriming treatments alone or applied as hybrid priming could be designed, using guidelines that support the operators’ choice for the best parameters, based on the features of seed sculpture and bacterial spores, and then assessed at the field level. With more pilot studies performed, the robustness of the dataset will be reinforced and its reliability will be fundamental to further guide seed technologists and agronomists. Besides the benefits brought to industrial seed technology, novel ‘on-farm’ protocols could be developed by local agronomists, using the same guidelines, and directly delivered to local smallholder farmers or integrated into regional seed systems with the support of key institutional actors.
Data availability
The data that support the findings of this study are openly available and provided in the Supporting Information section.
References
FAO. The state of food and agriculture 2024 - Value-driven transformation of agrifood systems. Rome. (2024). Available at: https://doi.org/10.4060/cd2616en
Popoola, J. O. et al. The exploitation of orphan legumes for food, income, and nutrition security in Sub-Saharan Africa. Front. Plant Sci. 13, 782140 (2022).
Samal, I. et al. Underutilized legumes: nutrient status and advanced breeding approaches for qualitative and quantitative enhancement. Front. Nutr. 10, 1110750 (2023).
Ogbole, O. O. et al. Nutritional and pharmacological potentials of orphan legumes: Subfamily Faboideae. Heliyon 9 (4), e15493 (2023).
Talucder, M. S. A., Ruba, U. B. & Robi, A. S. Potentiality of neglected and underutilized species (NUS) as a future resilient food: A systematic review. J. Agric. Food Res. 16(3), 101116 (2024).
Odeku, O. A., Ogunniyi, Q. A., Ogbole, O. O. & Fettke, J. Forgotten gems: Exploring the untapped benefits of underutilized legumes in agriculture, nutrition, and environmental sustainability. Plants 13(9), 1208 (2024).
Rubiales, D., Emeran, A. A. & Flores, F. Adaptation of grass pea (Lathyrus sativus) to Mediterranean environments. Agron. 10(9), 1295 (2020).
Lambein, F., Travella, S., Kuo, Y.-H., Van Montagu, M. & Heijde, M. Grass pea (Lathyrus sativus L.): Orphan crop, nutraceutical or just plain food?. Planta 250(3), 821–838 (2019).
Gonçalves, L., Rubiales, D., Bronze, M. R. & Vaz Patto, M. C. Grass pea (Lathyrus sativus L.) - a sustainable and resilient answer to climate challenges. Agron. 12(6), 1324 (2022).
Acharya, S. N., Thomas, J. E. & Basu, S. K. Fenugreek: an ‘Old World’ crop for the ‘New World’. Biodiversity 7(3 – 4), 27 – 30 (2006). (2006).
Syed, Q. A. et al. Nutritional and therapeutic properties of fenugreek (Trigonella foenum-graecum): A review. Int. J. Food Prop. 23(1), 1777–1791 (2020).
Akhtary, E. et al. Fenugreek (Trigonella foenum-graecum L.) in women’s health: A review of clinical evidence and traditional use. J. Herbal Med. 43, 100816 (2024).
Sayar, M. S. & Han, Y. Forage yield performance of forage pea (Pisum sativum spp. arvense L.) genotypes and assessments using GGE biplot analysis. J. Agric. Sci. Technol. 18, 1621–1634 (2016).
Demirkol, G. & Yilmaz, N. Forage pea (Pisum sativum var. arvense L.) landraces reveal morphological and genetic diversities. Turk. J. Bot. 43(3), 7 (2019).
Sapre, S., Gontia-Mishra, I. & Tiwari, S. Plant growth-promoting Rhizobacteria ameliorates salinity stress in pea (Pisum sativum). J. Plant Growth Regul. 41(2), 647–656 (2022).
Pagano, A., Macovei, A. & Balestrazzi, A. Molecular dynamics of seed priming at the crossroads between basic and applied research. Plant Cell Rep. 42 (4), 657–688 (2023).
Balestrazzi, A. et al. Seed quality as a proxy of climate-ready orphan legumes: The need for a multidisciplinary and multi-actor vision. Front. Plant Sci. 15, 1388866 (2024).
Zaman, M. S. U., Malik, A. I., Kaur, P., Ribalta, F. M. & Erskine, W. Waterlogging tolerance at germination in field pea: Variability, genetic control, and indirect selection. Front. Plant Sci. 10, 1–11 (2019).
Goufa, M. et al. Impact of seed priming technologies on the agronomical characteristics of Lathyrus sativus L. commercial and local variety under normal and saline conditions. Appl. Sci. 15(4), 1692 (2025).
Pagano, A. et al. Exploring the genotoxic stress response in primed orphan legume seeds challenged with heat stress. Genes 16 (2), 235 (2025).
Banerjee, P. et al. Seed priming and foliar application of nutrients influence the productivity of relay grass pea (Lathyrus sativus L.) through accelerating the photosynthetically active radiation (PAR) use efficiency. Agronomy 12(5), 1125 (2022).
Galaktionova, L. V. et al. Influence of pre-sowing priming on the parameters of Pisum sativum seedlings. IOP Conf. Ser. : Earth Environ. Sci. 341, 012094 (2019).
Szafrańska, K., Reiter, R. J. & Posmyk, M. M. Melatonin application to Pisum sativum L. seeds positively influences the function of the photosynthetic apparatus in growing seedlings during Paraquat-induced oxidative stress. Front. Plant Sci. 7, 1663 (2016).
Hojjat, S. S. & Hojjat, H. Effect of nano silver on seed germination and seedling growth in fenugreek seed. ETP International Journal of Food Engineering 1(2), 106–110 (2015).
Mnafgui, W., Hajlaoui, H., Rizzo, V., Muratore, G. & Elleuch, A. Priming with EDTA, IAA and Fe alleviates Pb toxicity in Trigonella Foneum graecum L. growth: Phytochemicals and secondary metabolites. J. Biotechnol. 356, 42–50 (2022).
Hussain, S. et al. Seed priming alleviates salt stress in two fenugreek (Trigonella foenum-graecum L.) cultivars. Emirates J. Food Agric. 34 (8), 650–657 (2024).
Lamsaadi, N. et al. Enhancing fenugreek (Trigonella foenum-graecum L.) productivity and seed quality through silicon-based seed priming under salt-stressed conditions. Russ. J. Plant Physiol. 71(3), 70 (2024).
Mahmoudi, H. et al. Priming-induced changes in germination, morpho-physiological and leaf biochemical responses of fenugreek (Trigonella foenum-graecum) under salt stress.. Plant Biosyst. 154(5), 601–614 (2019).
Fiodor, A., Ajijah, N., Dziewit, L. & Pranaw, K. Biopriming of seed with plant growth-promoting bacteria for improved germination and seedling growth.. Front. Microbiol. 14, 1142966 (2023).
Blake, C., Christensen, M. N. & Kovács, Á. Molecular aspects of plant growth promotion and protection by Bacillus subtilis”.. Mol. Plant-Microbe Interact. 34(1), 15 (2021).
Lastochkina, O., Yakupova, A., Avtushenko, I., Lastochkin, A. & Yuldashev, R. R. Effect of seed priming with endophytic Bacillus subtilis on some physio-biochemical parameters of two wheat varieties exposed to drought after selective erbicide application.. Plants 12(8), 1724 (2023).
Zulueta-Rodríguez, R. et al. Effect of hydropriming and biopriming on seed germination and growth of two Mexican fir tree species in danger of extinction. Forests 6 (9), 3109–3122 (2015).
Hashmi, I. et al. Comparison of the plant growth-promotion performance of a consortium of Bacilli inoculated as endospores or as vegetative cells.. FEMS Microbiol. Ecol. 95(11), fiz147 (2019).
Lobo, C. B., Juárez Tomás, M. S., Viruel, E., Ferrero, M. A. & Lucca, M. E. Development of low-cost formulations of plant growth-promoting bacteria to be used as inoculants in beneficial agricultural technologies.. Microbiol. Res. 219, 12–25 (2019).
Pagano, A. et al. Changes in genotoxic stress response, ribogenesis and PAP (3’-phosphoadenosine 5’-phosphate) levels are associated with loss of desiccation tolerance in overprimed Medicago truncatula seeds. Plant Cell & Environment 45(5), 1457–1473 (2022).
Ranal, M. A. & Santana, D. G. How and why to measure the germination process. Revista Brasileira de Botanica 29(1), 1–11 (2006).
Vrešak, M., Halkjaer Olesen, M., Gislum, R., Bavec, F. & Ravn Jørgensen, J. The use of image-spectroscopy technology as a diagnostic method for seed health testing and variety identification. PLoS ONE 11(3), e0152011 (2016).
Egesa, A. O., Davidson, M. T., Pérez, H. E. & Begcy, K. Biochemical and physical screening using optical oxygen-sensing and multispectral imaging in sea oats seeds. Agriculture 14(16), 875 (2024).
Chinga-Carrasco, G. et al. New advances in the 3D characterization of mineral coating layers on paper. J. Microsc. 232(2), 212–224 (2008).
Assaad, H. I., Hou, Y., Zhou, L., Carrol, R. J. & Wu, G. Rapid publication-ready MS-Word tables for Two-Way ANOVA. SpringerPlus 4, 33 (2015).
Gunes, F. & Ali, C. Seed characteristics and testa textures of some taxa of genus Lathyrus L. (Fabaceae). Int. J. Agric. Biol. 13(6), 888–894 (2011).
Tosheva, A. & Tonkov, S. Seed characters of the species from Section Lathyrus (Genus Lathyrus, Fabaceae) in Bulgaria. Proc. Bulg. Acad. Sci. 62(3), 341–348 (2009).
Aydin, H. et al. Phylogenetic relationships among Pisum L. species from Asia Minor inferred from pollen and seed coat morphology. Grana 60(5), 347–355 (2021).
Ceter, T. Comparative seed morphology of Trigonella L. species (Leguminosae) in Turkey. Afr. J. Agric. Res. 7(3), 509–522 (2012).
Turki, Z., El-Shayeb, F. & Abozeid, A. Seed morphology of some Trigonella L. species (Fabaceae) and its taxonomic significance. International Journal of Science and Research 3(12), 940–948 (2014).
Shrestha, S., Deleuran, L. C. & Gislum, R. Classification of different tomato seed cultivars by multispectral visible-near infrared spectroscopy and chemometrics. J. Spectr. Imaging 5, 1–9 (2016).
Carrillo-Reche, J., Vallejo-Marín, M. & Quilliam, R. S. Quantifying the potential of ‘on-farm’ seed priming to increase crop performance in developing countries. A meta-analysis. Agron. Sustain. Dev. 38, 64 (2018).
Srivastava et al. Seed biopriming as a promising approach for stress tolerance and enhancement of crop productivity: a review. J. Sci. Food. Agric. 104 (3), 1244–1257 (2023).
Miljaković, D. et al. Bio-priming with Bacillus isolates suppresses seed infection and improves the germination of garden peas in the presence of Fusarium strains. J. Fungi 10(5), 358 (2024).
Lastochkina, O. et al. Seed priming with endophytic Bacillus subtilis strain-specifically improves growth of Phaseolus vulgaris plants under normal and salinity conditions and exerts anti-stress effect through induced lignin deposition in roots and decreased oxidative and osmotic damages. J. Plant Physiol. 263, 153462 (2021).
Singh, S., Sarkar, D., Singh, R. K. & Rakshit, A. Biopriming with Bacillus subtilis enhanced the sulphur use efficiency of Indian Mustard under graded levels of sulphur fertilization. Agronomy 13(4), 974 (2023).
Kumar, P. et al. Seed bio-priming with tri-species consortia of phosphate solubilizing rhizobacteria (PSR) and its effect on plant growth promotion. Heliyon 6 (12), e05701 (2020).
Berlanga-Clavero, M. V. et al. Bacillus subtilis biofilm matrix components target seed oil bodies to promote growth and anti-fungal resistance in melon. Nat. Microbiol. 7(7), 1001–1015 (2022).
Sarti, G. C. et al. Inoculation with biofilm of Bacillus subtilis is a safe and sustainable alternative to promote tomato (Solanum lycopersicum) growth. Environments 11, 54 (2024).
Gadhave, K. R., Devlin, P. F., Ebertz, A., Ross, A. & Gange, A. C. Soil inoculation with Bacillus spp. modifies root endophytic bacterial diversity, evenness, and community composition in a context-specific manner. Microb. Ecol. 76, 741–750 (2018).
Li, H. et al. Seed biostimulant Bacillus sp. MGW9 improves the salt tolerance of maize during seed germination. AMB Express 11, 74 (2021).
Lubyanova, A. R., Allagulova, C. R. & Lastochkina, O. V. The effects of seed pretreatment with endophytic bacteria Bacillus subtilis on the water balance of spring and winter wheat seedlings under short-time water deficit. Plants 12(14), 2684 (2023).
Hamiot, A., Lemy, C., Krzewinski, F., Faille, C. & Dubois, T. Sporulation conditions influence the surface and adhesion properties of Bacillus subtilis spores. Front. Microbiol. 14, 1219581 (2023).
Rocha, I. et al. Seed coating: A tool for delivering beneficial microbes to agricultural crops. Front. Plant Sci. 10, 1357 (2019).
Rego, C. H. Q. et al. Using multispectral imaging for detecting seed-borne fungi in cowpea. Agriculture 10(8), 361 (2020).
Sudki, J. M. et al. Fungal identification in peanuts seeds through multispectral images: Technological advances to enhance sanitary quality. Front. Plant Sci. 14, 1112916 (2023).
Boelt, B. et al. Multispectral imaging - A new tool in seed quality assessment?. Seed Sci. Res. 28(3), 222–228 (2018).
ElMasry, G. et al. Utilization of computer vision and multispectral imaging techniques for classification of cowpea (Vigna unguiculata) seeds. Plant Methods 15, 24 (2019).
Sauli, Z., Retnasamy, V. & Say, O. T. Analysis on surface roughness and surface reflectance through DOE. Appl. Mech. Mater. 487, 37–40 (2014).
Grulichova, M., Sedlakova, V., Trojan, V., Hanacek, P. & Vyhnanek, T. Correlation of photosynthetic pigments content with indicators of seed quality in the seeds of carrot, celery, dill, parsley, and parsnip. J. Seed Sci. 44, e202244031 (2022).
Picinini Petronilio, A. C. et al. Smart and accurate: A new tool to identify stressed soybean seeds based on multispectral images and machine learning model. Smart Agricultural Technology 12, 101042 (2025).
Ou, C. et al. A novel approach integrating multispectral imaging and machine learning to identify seed maturity and vigor in smooth bromegrass. Plant Methods 21, 45 (2025).
Funding
This work was supported by PRIMA (Partnership for Research and Innovation in the Mediterranean Area) Programme-H2020, Call Multi-Topic 2021, Research & Innovation Activities-RIA, Project “Boosting technologies of orphan legumes towards resilient farming systems in the Greater Mediterranean Region: from bench to open field (BENEFIT-Med) ID: 1726 (2022–2025). AP and CDJr has been awarded a Post-Doctoral Fellowship in the frame of the BENEFIT-Med project.
Author information
Authors and Affiliations
Contributions
CD, AP, AM, and AB conceptualized and wrote the manuscript. CD, AP, CC and NK designed and validated the experimental system. CM, AG, and CD performed SEM analyses. FH and AM supervised the MSI experimental activity. AE and BK designed the fenugreek treatments. ET and DC designed the grass pea treatments. CD, AP, AM, and AB supervised and finalized the manuscript. All the authors have read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Dueñas, C., Pagano, A., Calvio, C. et al. Novel approaches for understanding and improving the effectiveness of seed biopriming. Sci Rep 16, 10965 (2026). https://doi.org/10.1038/s41598-026-46096-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-46096-7