Abstract
Despite advances in metastatic colorectal cancer (mCRC) treatment, long-term survival remains poor, particularly in right-sided colorectal cancer (RCRC), which has a worse prognosis compared to left-sided CRC (LCRC). This disparity is driven by the complex biological diversity of these malignancies. RCRC and LCRC differ not only in clinical presentation and outcomes but also in their underlying molecular and genetic profiles. This article offers a detailed literature review focusing on the distinctions between RCRC and LCRC. We explore key differences across embryology, anatomy, pathology, omics, and the tumor microenvironment (TME), providing insights into how these factors contribute to prognosis and therapeutic responses. Furthermore, we examine the therapeutic implications of these differences, considering whether the conventional classification of CRC into right- and left-sided forms should be refined. Recent molecular findings suggest that this binary classification may overlook critical biological complexities. Therefore, we propose that future approaches should integrate molecular insights to better guide personalized treatments, especially anti-EGFR therapies, and improve patient outcomes.
Similar content being viewed by others
Introduction
Colorectal cancer (CRC) is one of the most frequent malignancies and accounts for approximately 9% of cancer-associated deaths worldwide1,2. Around 20% of CRC cases are diagnosed at advanced stages, where curative options are limited. For most of these patients, treatment is palliative, aiming to improve quality of life and prolong survival. In the last two decades, targeted therapies have emerged as a transformative approach in the management of metastatic CRC (mCRC)3, markedly enhancing overall survival (OS) rates. Agents, such as anti-epithelial growth factor receptor (EGFR) antibodies cetuximab4 and panitumumab5, as well as anti-vascular endothelial growth factor receptor (VEGFR) antibodies bevacizumab6, ramucirumab7, and aflibercept8 have become pivotal components of systemic treatment for mCRC. These targeted therapies exert their effects by selectively inhibiting key molecular pathways (e.g., MAPK signaling) of cancer proliferation and progression, and their addition to systemic therapy results in improved outcomes compared to conventional chemotherapy regimens9,10. Recent evidence suggests that the efficacy of anti-EGFR antibodies in combination with first-line chemotherapy is notably prolonged in patients with RAS wild-type (wt) primary tumors originating specifically from the left side of the colon (LCRC) compared to those that derive from the right side (RCRC)11,12. Treatment implications according to sidedness (e.g., using anti-EGFR in LCRC) has recently been prospectively corroborated in the randomized phase III PARADIGM trial13. Consequently, the current European Society of Medical Oncology guidelines support using anti-EGFR antibodies in patients with RAS wt/BRAF wt newly diagnosed metastatic LCRCs. In RAS wt RCRC and/or BRAF mutant cases, bevacizumab is considered as the preferred upfront option14. However, additional liquid-biopsy biomarker analyses derived from the PARADIGM and FIRE-4 trials challenge first-line decision-making solely based on tumor–tissue results and sidedness15,16, since molecular findings suggest that this binary classification may overlook fundamental biological complexities and processes. Despite recent insights on treatment optimization according to the primary’s sidedness, the underlying biological differences between RCRC and LCRC largely remain uncharacterized. This article will summarize underlying biological discrepancies between RCRC and LCRC, discussing major physiological and pathological differences between the right and LCRC. Consequently, the influence of these differences on the presence of molecular alterations and their implications for further therapeutic refinement will also be discussed.
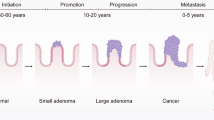
Embryonic development of large intestine
The large intestine originates from the endodermal layer of the developing gastrointestinal tract (GIT) during gastrulation in the third week of gestation. The gut tube forms through complex morphogenetic movements of the endoderm17,18, later it incorporates tissue from all three germ layers: the endoderm forms the epithelial layer of the intestinal mucosa; the mesoderm forms the muscular layer, the lamina propria, mesentery, connective tissue, and blood vessels; the ectoderm creates the enteric nervous system which develops from neural crest cells19,20. By week 4, the primitive gut tube differentiates into the foregut, midgut, and hindgut, each evolving to the different compartments of the GIT21. The foregut gives rise to the esophagus, stomach, and proximal duodenum (up to papilla of Vater). The midgut develops to the distal duodenum, jejunum, ileum, cecum, appendix, ascending colon, and proximal two-thirds of the transverse colon21. The hindgut to the remaining transverse colon, descending colon, sigmoid colon, rectum, and the superior part of the anal canal. These three divisions are later distinguished by their different arterial supply: celiac trunk, superior mesenteric artery (SMA), and inferior mesenteric artery (IMA)21. Venous drainage follows arterial supply with superior mesenteric vein and inferior mesenteric vein. The segment of the colon transversum derived from the midgut and that originating from the hindgut is indicated by the change in blood supply from SMA to IMA21.
Altogether, the development of the large intestine involves the coordinated differentiation of the endoderm, mesoderm, and ectoderm during early gestation, resulting in the formation of its complex structure and function. The distinct arterial supplies from the SMA and IMA highlight the evolutionary division of the large intestine into midgut and hindgut.
Implications of embryonic tissue origin
Studies show that the molecular heterogeneity of CRC does not follow a strict right–left pattern, but rather a continuum along the foregut-hindgut axis. Liu et al. 22 demonstrated that MSI tumors with CIMP-H and MLH1 silencing occur mainly in the proximal colon, while MSS tumors with CIN and KRAS mutations are more common in the distal colon and rectum. Importantly, their analysis revealed that these molecular alterations follow a continuous gradient along the gastrointestinal axis rather than an abrupt transition. RCRC exhibits more gastric-like molecular properties, while LCRC shares similarities with rectal carcinomas or intestinal-type gastric cancer, suggesting that the underlying molecular diversity arises from the foregut-hindgut developmental gradient rather than being dictated by the anatomical left-right axis22. Joanito et al. refined this classification using scRNA sequencing, identifying two intrinsic tumor cell states, iCMS2 and iCMS3, with show distinct distributions along the intestine. Unlike the previous CMS classification, which included tumor and stromal components, this approach focused solely on epithelial tumor cells and uncovered a left–right bias that may stem from the type of crypt progenitor cells giving rise to tumorigenesis23.
Therefore, CRC cannot be adequately categorized by a simple right-left dichotomy but instead follows a molecular continuum along the foregut–hindgut axis, with significant implications for classification and therapy.
Tumor localization affects histopathological disease characteristics and tumor morphology
Compared to LCRC, RCRCs show a tendency toward larger diameter and therefore higher T-stages upon diagnosis. This might be explained by the fact that RCRC usually becomes symptomatic at later stages that hinders earlier detection24,25. Consequently, a higher rate of tumor-infiltrated lymph nodes24 and more advanced local disease stages24,26 at diagnosis in RCRC is observed. In addition to the local disease spread, the metastatic pattern differs considerably: LCRC tumors are associated with a higher prevalence of synchronous hepatic and pulmonary metastases27,28, whereas peritoneal metastases are more common in RCRC24, which again is likely a consequence of the higher incidence of advanced T-stages24. RCRCs show a lower grade of differentiation24,25 and several studies on large CRC cohorts report of differences in morphologic subtypes distribution according to tumor localization24,25,29. In RCRC, a higher rate of histomorphologic subtypes like mucinous carcinoma, signet cell carcinoma, and medullary carcinoma are observed compared to LCRC24,25,29,30,31. Regarding histomorphologic findings, LCRC is characterized by a higher rate of tumor necrosis32, and entosis33. Conflicting results exists with respect to tumor budding, where the majority of studies show an association with LCRC34,35, although this association cannot be reproduced in all studies36.
Significant differences in the composition of the TME between LCRC and RCRC were also identified, such as fewer macrophages37 and tumor-infiltrating lymphocytes in LCRC25,38. This finding is in line with the observation of higher numbers of inflammatory cells39,40 and an increase in desmoplastic reaction in RCRCs41. In contrast, other morphological characteristics such as stromal reactive invasion front areas42, tumor-stroma ratios35,43,44,45, or vascular and perineural invasion25 do not appear to be associated with tumor location.
Tumor-sidedness influences pathological diagnosis
Tumor localization should be considered in the pathological examination of resection specimens. Fewer lymph nodes are examined in LCRC compared to RCRC24,46,47, which might be explained due to the smaller size of lymph nodes observed in LCRC46,48 as the distribution of lymph nodes in both resection specimens is comparable46. Even though a low number of examined lymph nodes is already considered as a risk factor in stage II CRC pointing in favor of adjuvant treatment, side-specific cut-offs might potentially improve treatment selection in this specific treatment setting49. Another important consideration is the association of Immunohistochemistry markers with primary tumor localization. Commonly employed markers to secure a primary of colonic origin in metastatic disease, such as CDX2, SATB2, CK20, and CK7 may be differentially expressed between LCRC and RCRC. For instance, CDX2 is more often positive in LCRC26,50,51,52 as is SATB252,53,54 and CK254. In contrast, the frequency of CK7-positive tumors is higher in RCRC54.
Molecular alterations based on tumor sidedness
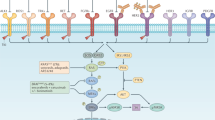
CRC is a genetically and biologically heterogeneous disease, with RCRC and LCRC presenting distinct genomic and transcriptomic patterns. Attempting to harmonize a molecular classification, the four consensus molecular subtypes (CMS)55 were defined based on bulk-RNA sequencing, genomic variants, and methylation status. While the CMS2 (high somatic copy number alterations (SCNA) with subsequent WNT, MYC, and SRC activation) and CMS4 (mesenchymal subtype, associated to epithelial-mesenchymal transition (EMT), pro-angiogenic pathways and TGF-β activation) subtypes show higher prevalence in LCRCs, RCRCs are characterized by a predominance of the CMS1 subtype (high rates of MSI-H and distinct methylation patterns and BRAFV600E mutations) (Fig. 1). No predominance of side is found for the CMS3 subtype (metabolic deregulation, low SCNA, and low CIMP, mixed MSI status, high rate of KRAS mutations). Focusing solely on somatic driver mutations, LCRCs predominantly follow the prototypical adenoma-carcinoma sequence characterized by the stepwise acquisition of mutations in the tumor-suppressor APC (60–80%), followed by mutations in the proto-oncogene KRAS (~40%) and the tumor-suppressor TP53 (~60–70%). Furthermore, mutations in PIK3CA (~11–15%), FBXW7 (~12.5%), or SMAD4 (~10%) are frequently observed56. As comprehensively reviewed by Testa et al., these sequential genetic alterations underscore the clonal evolution and molecular heterogeneity inherent to CRC, providing a basis for its diverse clinical behavior57. Furthermore, emerging evidence suggests that hormonal signaling—particularly differential estrogen receptor expression—adds another layer of molecular heterogeneity in CRC58. Conversely, RCRC has been consistently associated with higher rates of BRAF mutations (~25%) and lower rates of APC (~64%) and TP53 (~35–60%) mutations but slightly higher rates of KRAS (45–50%) mutations59. Additionally, RCRCs are enriched in mutations for RNF43 (14.3% vs 3.1%), PIK3CA (22–27%), FBXW7 (~23%), and SMAD4 (15%)56 (Fig. 2).
This pie chart illustrates the distribution of CMS by tumor laterality, with right-sided CRCs predominantly exhibiting the CMS1 subtype and left-sided CRCs showing a higher proportion of CMS2 (based on ref. 55).
This figure presents a comparative overview of the most frequent genetic alterations found in left-sided (LCRC) and right-sided colorectal cancer (RCRC), displaying the relative frequencies for key genes associated with each subtype. By showcasing these genetic distinctions, the figure emphasizes the unique molecular characteristics between LCRC and RCRC, providing insights into how these differences may affect tumor behavior and treatment options. Data based on the study by Tokunaga et al.162. LCRC Left-sided colorectal cancer, RCRC Right-sided colorectal cancer.
Although genetic differences seem to distinguish right- and left-sided CRCs, Yamauchi et al.60,61 showed that molecular alterations occur gradually along the colon—a concept termed the colorectal continuum model. Their analysis of over 1400 CRC samples demonstrated a continuous transition of molecular alterations instead of a strict division. Similarly, an analysis of 522 CRC from The Cancer Genome Atlas revealed continuous gradients in gene alterations and metabolite–gene interactions across the colon, underscoring how anatomic biases in the tumor ecosystem contribute to carcinogenesis62. These findings support the need for individualized therapy based on molecular profiles60,61. Additionally, a large-scale survival analysis by Ugai et al. 63 shows that primary tumor location interacts with molecular features (e.g., MSI, CIMP) to shape prognosis. Recent evidence also links high miR-31 expression with CIMP-high status in serrated lesions harboring BRAF mutations, with frequencies increasing from the rectum to the cecum64. An important molecular marker in this context is KRAS, whose mutations are associated not only with tumor location but also with specific signaling pathway alterations. In KRAS-mutated CRC, mutations are predominantly observed in Exon 12 (~60%). The most common single nucleotide polymorphism variants identified are G12D (40%), G12V (35%), and G12C (10%). KRASG13 (17.5%) or KRASQ61 (5.5%) mutations are found at a much lower frequency65. Mutations in Exon 12 or 13 affect glycine residues in the GTP-binding pocket, ultimately resulting in a constitutively active kinase enhancing downstream signaling66. Importantly, to date, there has been no comprehensive large-scale analysis conducted on the frequency and distribution of distinct KRAS mutations comparing RCRCs and LCRCs. Such an investigation would provide crucial insights for patient stratification, particularly in light of the advent of mutant selective KRAS inhibitors67,68.
In the context of the MAPK signaling upstream of KRAS, mutations in EGFR are rarely observed in CRC. Instead, the deregulation of the EGFR signaling pathway is more commonly represented by protein overexpression, which appears to be more prevalent in RCRCs compared to LCRCs69. Conversely, HER2 amplification and HER2 overexpression are more prevalent in more distant LCRCs69.
Direct comparisons of bulk- and single-cell RNA-Seq between RCRC and LCRC are limited but reveal key differences between sides. One bulk RNA-Seq study found genes involved in carcinogen breakdown downregulated in RCRC, suggesting a more genotoxic environment. Such an environment could result in a more aggressive phenotype, potentially explaining the differences in the mutational landscape, but mechanistic studies are lacking70. Using at higher resolution, a single-cell RNA Sequencing analysis directly comparing LCRCs and RCRCs identified differences in regulation of genes involved in cell adhesion, innate immune functions, and immune cell composition, such as an enrichment in naïve CD4+ T cells and exhausted CD8+T-Cells in RCRCs70.
Enhanced T-cell function, marked by increased infiltration of CD8+ cytotoxic T-cells and Th1 cells into the TME, improves clinical outcomes in CRC. CD8+ T cells directly kill malignant cells, while Th1 cells bolster anti-tumor immunity, contributing to better survival rates and lower relapse risks71. Another single-cell study found transcriptomic signatures associated with higher T-cell function in LCRCs, suggesting an explanation for better clinical outcomes in LCRCs. Focusing on the gene regulation level, microRNA profiling found miRNAs associated with regulation of mTOR, WNT, and PI3K/AKT signaling predominantly in LCRCs, while RCRCs were enriched in miRNAs associated with MSI-H or BRAF signaling, corroborating the CMS classification72 (Fig. 3).
Understanding these differences is crucial, as they can influence the development of tailored therapeutic strategies. For instance, variations in genetic mutations, signaling pathways, and tumor microenvironments may affect treatment responses and prognoses, underscoring the need for personalized approaches in managing colorectal cancer effectively. CRC colorectal cancer, MSI microsatellite instability, TMB tumor mutational burden.
In conclusion, RCRCs and LCRCs differ not only by anatomical location but also through distinct molecular patterns, influencing their clinical behavior and response to treatments. Recent advances in methodologies, such as single-cell analysis of genomic variants combined with transcriptomic or proteomic data, could offer promising avenues for deeper insights into the underlying mechanisms of these cancers, potentially guiding more precise therapeutic approaches.
Anatomic location of CRC influences metabolic and proteomic profile
Tumor cells rely on dynamic metabolic and proteomic processes for proliferation and metastasis, with alterations in these pathways being a hallmark of cancer73. Recent studies highlight significant metabolomic differences between RCRC and LCRC. Research suggests significant metabolic heterogeneity in CRC, including differences in amino acid levels that vary within traditional right- and left-sided classifications. This heterogeneity may influence the clinical presentation and treatment responses74. Another study using ultra-performance liquid chromatography-mass spectrometry found six potential diagnostic biomarkers specific to cancerous tissue, which could help differentiate early-stage LCRC from RCRC75. Additionally, in liver metastases—the most common site of spread—RCRC showed increased levels of reactive oxygen species and bile acids76. Elevated primary bile acids and secondary bile acids like glycodeoxycholic acid were noted, indicating a connection to tumor growth and therapeutic resistance76. While these findings suggest distinct metabolic features between RCRC and LCRC, reliance on peripheral blood analyses rather than tumor tissue limits conclusions. Future studies should incorporate tumor sidedness to enhance understanding and inform treatment strategies targeting metabolic pathways77.
Proteomics, the large-scale study of proteins, is essential for understanding cancer biology, as proteins play key roles in cellular functions and disease progression. By identifying and quantifying protein biomarkers, proteomics offers insights into cancer pathophysiology, aiding in early detection, risk classification, and treatment monitoring78. In CRC, the first proteomic studies highlight significant molecular differences between LCRC and RCRC tumors79. While both show hyperproliferation driven by pre- and post-transcriptional changes, proteins linked to tumor growth are more expressed in LCRC, whereas immune-related pathways are more active in RCRC70. These findings underscore distinct biological behaviors between LCRC and RCRC, offering potential targets for personalized therapies.
Proteomics and metabolomics represent emerging members of the “OMICS” family, offering significant potential to expand the biological understanding of CRC beyond the scope of genomics and transcriptomics80. However, the current body of research on CRC, particularly regarding proteomic and metabolomic distinctions between RCRC and LCRC tumors, remains sparse80. Comprehensive investigations are required to elucidate these molecular differences and their implications for tumor biology and therapeutic strategies.
Differences in the TME according to primary sidedness
The TME is composed of a wide variety of cell types including cancer cells, immune cells, and ECM components. These components influence tumor progression, outcome and therapeutic response through altering the signaling pathways (such as chemokines and proteins)81. Consequently, a comprehensive understanding of the genetic, transcriptional, and post-transcriptional impact on tumor development and progression is essential to draw a detailed outline of the TME landscape and to identify potential differences in individual tumor entities. The individuality of tumors environment becomes even clearer when taking a closer look at CRCs. Comprehensive genome- and transcriptome-wide analyses have shown that the TME shows key differences between RCRC and LCRCs70,82. For instance, Hong et al. found that the TME of LCRC patients harbors more antitumor immune subsets, like M1 macrophages and CD8+T cells, whereas in RCRC patients the TME displays stronger pro-tumor functions by showing a higher abundance of M0 macrophages and CD4+ naive T-cells. These differences in tumor immunity may also contribute to the poorer clinical prognosis of RCRC patients83. Guo et al. showed that high infiltration of plasma cells, dendritic cells, and mast cells, coupled with low infiltration of activated memory CD4+ T cells and M1/M2 macrophages, correlated with better prognosis in CRC. These findings led to the development of an immune cell infiltration (ICI) score. Patients with a low ICI score were more likely to have a LCRC and showed a better prognosis than patients with a high ICI score which was more abundant in the RCRC cohort84. In high-ICI patients, upregulation of cell cycle progression and DNA damage repair pathways was noted. Furthermore, the tumor mutational burden (TMB) was significantly higher in patients with a high ICI score and was associated with poor prognosis. The authors suggest that the ICI score may provide an independent predictor of prognosis in patients with LCRC and RCRC, and subsequently may predict TMB and treatment sensitivity to immune checkpoint inhibitors84.
In conclusion, the TME significantly influences tumor progression, therapeutic response, and clinical outcomes in CRC. Distinct immune cell compositions and signaling pathways between RCRC and LCRC tumors underscore the need for individualized treatment approaches. Tools like the ICI score provide insights into patient prognosis and potential responses to immunotherapies, emphasizing the importance of further research to optimize personalized treatment strategies in CRC.
Changes between RCRC and LCRC in terms of gut microbiota
The gut microbiota, a complex microbial ecosystem comprising bacteria, fungi, archaea, protozoa, and viruses, is located close to the colorectal epithelium and exerts a substantial influence on the development and progression of CRC85,86,87. The microbiome’s impact on CRC outcomes is multifaceted: microbes can directly promote carcinogenesis, influence cancer signaling pathways, and interact with other cancer hallmarks, thereby (positively or negatively) modulating tumor inflammation, genome instability, and response to anticancer therapies88.
Numerous studies have revealed a global shift in the microbiome composition among patients with CRC, characterized by a reduction in butyrate-producing commensal species and an enrichment of opportunistic, often pro-inflammatory pathogens, including oral microbes87. Common microbial signatures enriched in CRC include genera, such as Fusobacterium, Porphyromonas, Parvimonas, Peptostreptococcus, Gemella, Prevotella, and Solobacterium89,90, alongside a reduction in potentially protective taxa like Bifidobacterium and Roseburia91. However, many microbiome studies have predominantly relied on fecal samples, reflecting the luminal microbiota, thereby precluding site-specific distinctions. Notably, while fecal microbiota can indicate disease status compared to healthy controls, it only partially reflects the mucosa-associated microbial composition in CRC patients92,93. Indeed, comparative analyses of tumor-associated microbiomes in CRC patients have unveiled significant differences among rectal, distal, and proximal microbiota92,93. These analyses unveiled three distinct oncomicrobial community subtypes, with differential representation of oral pathogens including Fusobacterium nucleatum (Fn) in RCRC, and tumor-enriched Proteobacteria species, including Escherichia/Pseudescherichia/Shigella spp., in LCRC94. Additionally, the role of the ileum, serving as the site of intestinal immune surveillance and establishing a microbiome-dependent immune tonus, might be relevant for CRC outcomes, particularly RCRC, and response to therapy93,95,96.
The strongest link between CRC development and specific bacterial species indeed arises from studies focusing on Fn, enterotoxigenic Bacteroides fragilis (ETBF), and polyketide synthase (pks)+ Escherichia (E.) coli87,97,98. Fn, a common constituent of the oral microbiome linked to periodontitis99 but not typically found in the healthy colon, selectively colonizes CRC tumor tissue100,101,102,103, with multiple studies suggesting the oral cavity as its source104,105. Fn engages with epithelial and immune cells via its lectins Fap2, thereby facilitating tumorigenesis100,106. Several studies have associated increased Fn abundance with proximal CRC, and Fn has also been correlated with shorter survival94,107,108. Recent studies have identified a distinct Fn subtype, Fn subspecies animalis clade C2, prevalent in the CRC tumor niche, characterized by enhanced virulence attributes and Patho adaptation109.
Bacterial biofilms, dense microbial aggregates embedded in an extracellular polymeric matrix110, have also been implicated in colon cancer pathogenesis and are enriched in right-sided tumors, suggesting a site-specific effect111,112. Invasive polymicrobial biofilms, composed of Fusobacterium spp., Lachnospiraceae, Bacteroidetes, and Proteobacteria exhibit pro-cancerogenic potential by activating epithelial signaling pathways relevant to tumorigenesis113,114,115. In patients with familial adenomatous polyposis, colonic biofilms primarily consist of ETBF and pks + E. coli, the latter capable of producing genotoxic colibactin116. Studies have demonstrated the mutagenic potential of these E. coli strains by inducing DNA damage, thereby promoting CRC117,118. Although biofilms are more prevalent in the right-sided colon, an increased abundance of pks + E. coli has also been observed in LCRC119,120. Thus, whether E. coli promotes CRC in a site-specific manner, needs to be further investigated.
In contrast to the mutagenic properties of pks + E. coli, the contributions of ETBF to CRC are less clear. Besides its implication in diarrheal disease and colitis, ETBF enrichment in colonic mucosa has been linked to both, early and late-stage CRC121,122,123. Notably, there is no direct evidence suggesting a site-specific induction of CRC by ETBF, as it colonizes various regions of the colon without tumors necessarily co-localizing with its abundance123. Instead, ETBF appears to foster a tumor-permissive environment by potently inducing TH17-based mucosal inflammation, thereby promoting colonic tumorigenesis, albeit through yet to be fully understood87.
Furthermore, other microbial species have been linked to CRC, although their precise mechanistic roles in colon tumorigenesis remain poorly understood85,87. In essence, while the significance of the microbiome in CRC is undeniable, the specific regional variations and metabolic capabilities influencing the development of site-specific CRC represent significant gaps in our current knowledge. Nevertheless, unraveling this information holds immense importance for the advancement of targeted and personalized therapeutic strategies to combat CRC.
Implication of sidedness on systemic treatment in mCRC
As described above, the behavior of metastatic mCRC is significantly influenced by its anatomical location, which in turn affects its molecular and immunological properties. Accordingly, the response to treatment is highly variable between LCRC and RCRC. In recent years, several randomized phase II and III studies, including CALGB/SWOG 8040512, FIRE-3124, TRIBE125, CRYSTAL126, PARADIGM13, PEAK127, and PRIME128 trials, have aimed to investigate the efficacy of different therapeutic approaches in mCRC, with a focus on the combination of chemotherapy and targeted therapies.
The CALGB/SWOG 8040512, CRYSTAL126, and FIRE-3124 trials compared the efficacy of standard chemotherapy (5-FU, leucovorin, and irinotecan or oxaliplatin) in combination with either the anti-EGFR antibody cetuximab or with the anti-VEGF antibody bevacizumab as first-line therapy in patients with mCRC irrespective of the pan-RAS status. Retrospective post-hoc analyses of these studies revealed that the OS in patients with LCRC was significantly prolonged with the addition of cetuximab to doublet chemotherapy compared to doublet chemotherapy in combination with bevacizumab (38.3 months vs. 28.0 months, hazard ratio (HR) = 0.63; P = 0.002)11,129. Similarly, in the CRYSTAL and CALGB/SWOG 80405 trials an improvement of PFS was also observed for patients with LCRC vs. patients with RCRC who were treated with anti-EGFR-based therapy11,126.
The PRIME, PEAK and PARADIGM studies investigated the efficacy of the anti-EGFR antibody panitumumab in combination with chemotherapy (PRIME: FOLFOX4; PEAK + PARADIGM: mFOLFOX6) compared to chemotherapy alone (PRIME) or in combination with an anti-VEGF antibody (PEAK, PARADIGM) in patients with mCRC. All three studies have shown that first-line treatment with panitumumab plus chemotherapy results in longer PFS and OS compared to chemotherapy with or without bevacizumab in the subgroup of patients with pan-RAS wt LCRC (PEAK: PFS 14.6 vs. 11.5 months HR = 0.65, P = 0.0514; OS: 43.4 vs. 32 months; HR = 0.77, P = 0.3125; PRIME: PFS: 12.9 vs. 9.2 months HR = 0.72, P = 0.0048; OS: 30.3 vs. 23.6 months HR = 0.73, P = 0.0112)130. Of note, the PARDIGM trial represents the first study prospectively evaluating sidedness as a stratification factor in mCRC undergoing chemotherapy + anti-EGFR treatment13. Collectively, these studies have shown that anti-EGFR therapy in combination with double chemotherapy should be the preferred choice for patients with RAS/BRAF wt metastatic LCRC12,131. Post-hoc analyses of the FIRE-3 and CRYSTAL trials revealed that patients with RCRC derived less benefit from an anti-EGFR blockade compared to anti-VEGF therapy (FIRE-3: 18.3 months vs. 23.0 months, HR = 1,44; P = 0,28)11. The latter findings were corroborated in a real-world cohort of the KRAS Registry of the Austrian Group of Medical Tumor Therapy30. Moreover, the CALGB/SWOG 80405 study retrospectively suggested that FOLFIRI or FOLFOX in combination with an anti-VEGF antibody significantly improved OS in patients with RCRC compared to FOLFIRI/FOLFOX plus anti-EGFR therapy129. In the PEAK and PRIME studies, the additive effects of panitumumab in combination with chemotherapy on PFS and OS were less pronounced in RCRC patients than in the control group13,127.
For this reason, doublet chemotherapy plus anti-EGFR treatment is considered as the preferred option in fit patients with RAS/BRAF wt LCRC131,132. In patients with RCRC, the inherently poor prognosis and the lack of benefit in terms of PFS and OS from anti-EGFR therapy in particular argue in favor of treatment with doublet or triplet chemotherapy plus bevacizumab131. The addition of bevacizumab should therefore be preferred in patients with RCRC tumors irrespective of pan-RAS status131,133. However, caution is warranted when interpreting these treatment recommendations due to study limitations. In the FIRE‑4 trial15, only a subset of patients initially received FOLFIRI alone before cetuximab was added, which may have reduced statistical power, and the follow‑up duration might not capture long‑term survival. Likewise, in PARADIGM13, although the overall sample was large, key subgroups—such as patients with RCRC or particular ctDNA profiles—were relatively small, resulting in wide confidence intervals. These factors call for a careful interpretation of the treatment benefits.
Moreover, the traditional dichotomous classification of mCRC into right- and left-sided tumors has been increasingly challenged due to its oversimplification of tumor biology. Further research highlights the continuous disadvantage of anti-EGFR therapy along a spectrum from distal left to proximal right, emphasizing the need for more refined strategies to optimize patient selection133. This understanding naturally leads to the concepts of liquid biopsy and negative hyperselection, which provide a framework for identifying right-sided patients who may benefit from anti-EGFR therapy and left-sided patients for whom such therapies may be less effective133.
To address this complexity, molecular diagnostics tailored to tumor location can provide actionable insights, as illustrated in Fig. 4. It illustrates potential future directions for molecular testing in mCRC stratified by tumor location. These suggestions aim to provide a framework for diagnostics, with a focus on key markers, such as RAS, BRAF, and MSI. By aligning molecular testing with tumor location, this approach seeks to support precision medicine strategies and improve patient outcomes, particularly when considering the selection between anti-EGFR and anti-VEGF therapies. However, as treatment progresses to second-line therapy and beyond, the clear distinction between LCRC and RCRC becomes less clear. Evidence suggests that anti-EGFR therapy may also be appropriate for a subset of RCRC14,134 cases, highlighting that anatomical location alone is not a decisive factor for selecting patients for anti-EGFR or anti-VEGF(R) therapeutics.
This figure outlines a proposed framework for molecular diagnostics in mCRC, focusing on markers like RAS, BRAF tailored to tumor location (e.g., right- vs. left-sided). mCRC metastatic colorectal cancer; CRC colorectal cancer.
Consequently, current research is shifting towards understanding primary resistance mechanisms to anti-EGFR therapy, which are likely to provide a more accurate explanation for treatment response.
The paradoxical shift in prognosis of MSI-H tumors
The prognostic significance of MSI-H status in CRC presents a notable paradox. While MSI-H in stages II and III are associated with improved survival and a lower risk of recurrence, this advantage is reversed in metastatic disease, where MSS demonstrate better outcomes55,135,136,137. In early stages, MSI-H tumors, characterized by high tumor mutation burden and strong immune infiltration, are associated with improved survival and lower recurrence rates, largely due to robust CD8+T-cell, NK cell, and Th1 responses137. The CMS classification by Guinney et al. 55 confirmed this pattern. MSI-H CRC tumors, which are predominantly assigned to the CMS1 subtype, have a significantly better prognosis in early stages of the disease and better survival rate than those with CMS4. The HR for OS indicates that CMS4 is associated with a 55% higher mortality risk compared to CMS1 (HR = 1.55, 95% CI: 1.19–2.01, p = 1.03 × 10⁻³). Similarly, recurrence rates are significantly higher in CMS4 (HR = 1.77, 95% CI: 1.34–2.34, p = 5.25 × 10⁻⁵), highlighting the strong early-stage survival advantage of CMS155. However, in the metastatic stage, this pattern reverses. While CMS4 tumors are linked to the poorest prognosis in early stages, post-relapse survival data reveal that CMS1 tumors have the worst survival outcome after recurrence. Compared to CMS4, CMS1 tumors show a 40% higher risk of death post-relapse (HR = 0.60, 95% CI: 0.40–0.88, p = 9.04 × 10⁻²)55. This shift is attributed to several factors, including immune evasion via increased expression of immune checkpoint molecules (e.g. PD-1, PD-L1, CTLA-4), stromal reorganization with enhanced fibroblastic activation and TGF-β signaling55,135,136. Also, molecular cofactors play a role: MSI-H tumors are more frequently associated with BRAF mutations, which correlate with more aggressive tumor biology and worse survival rates in the metastatic disease135. In addition, stromal factors and the microenvironment of metastases can modulate the immune response, leading to reduced effectiveness of immune mechanisms81. Another relevant aspect is therapy resistance. MSI-H responds less well to standard therapies, especially chemotherapy and anti-EGFR therapies, while MSS in the metastatic stage benefit more from targeted therapies135. In this context, dual checkpoint inhibition, as shown in the CheckMate 8HW study, significantly improved progression-free survival in metastatic MSI-H/dMMR CRC138. After a median follow-up of 31.5 months, the combination of nivolumab plus ipilimumab demonstrated significantly improved outcomes: the 24-month PFS rate was 72% versus 14% for chemotherapy (p < 0.001), and the restricted mean survival time was extended by 10.6 months.
In summary, immune evasion, stromal changes, therapy resistance and molecular cofactors contribute to MSI-H tumors having a favorable prognosis in early stages but a worse chance of survival in the metastatic stage. These mechanisms are crucial for clinical decision-making and the development of new therapeutic approaches.
Exploring resistance mechanisms in anti-EGFR therapy
Both genomic and non-genomic mechanisms both play roles in anti-EGFR resistance. However, mutations in KRAS, NRAS, BRAF, MAP2K1, or the EGFR ectodomain, which collectively account for around 40–50% of cases, do not fully explain this resistance. These points suggest to the existence of additional (in part unidentified) resistance mechanisms139,140,141. For instance, the formation of EGFR-HER3 heterodimers are known to cause secondary anti-EGFR treatment resistance by activating downstream PI3K and MAPK signaling pathways142. Another example for EGFR resistance is the MET signaling pathway via activation of hepatocyte growth factor143. These and other mechanisms allow tumors to bypass EFGR signaling pathway. Given this complexity, a promising strategy is to target multiple resistance routes simultaneously. A promising approach to specifically break these resistance mechanisms is the dual inhibition of KRASG12C and EGFR. The study by Fakih et al. 67 investigates the efficacy of sotorasib (specific KRASG12C inhibitor) in combination with panitumumab (EGFR inhibitor) in patients with mCRC harboring a KRASG12C mutation. While KRASG12C mutations occur in ~3–4% of mCRC cases, monotherapy with KRAS inhibitors has had limited success due to reactivation of the EGFR signaling pathway leading to resistance67. To address this issue, the study compared sotorasib plus panitumumab with standard treatments such as trifluridine-tipiracil or regorafenib in patients with a chemorefractory disease. Combination therapy significantly improved PFS, with a median of 5.6 months compared to 2.0 months in the standard treatment group (95% CI, 4.2–6.3 vs 95% CI, 1.9–3.9)67. These results underline that dual inhibition of KRASG12C and EGFR improves outcomes in KRASG12C-mutated mCRC and effectively overcomes the resistance observed with KRAS inhibitor monotherapy. At the same time, however, it must also be taken into account that temporal heterogeneity—i.e., the genetic changes that occur during tumor development—represents another dimension of resistance that can significantly influence therapy outcomes67. Although temporal heterogeneity has been investigated as a resistance factor, mutational analyses of KRAS, NRAS, BRAF, and PIK3CA in primary and metastatic samples show a high concordance of over 90%144.
The analysis of circulating tumor DNA (ctDNA) represents a critical approach for elucidating tumor progression and the development of resistance mechanisms to anti-EGFR and other targeted therapies145,146. Findings from the PRESSING(2) study147,148, and an exploratory biomarker analysis within the PARADIGM study149 indicate that hyperselection and ultraselection strategies can further refine the identification of patients with RAS wt mCRC who are most likely to benefit from anti-EGFR therapy.
Insights from the FIRE-4 trial15 further underscore the importance of upfront liquid biopsy for evaluating biomarker status, enabling the circumvention of temporal and spatial heterogeneity and supporting therapeutic optimization through dynamic patient selection. Hyperselection and ultraselection are strategies to optimize patient selection for anti-EGFR therapy in RAS wt mCRC by excluding individuals unlikely to respond based on molecular and clinical criteria150. Hyperselection focuses on excluding patients with well-established negative predictive biomarkers, such as RAS and BRAF mutations16,150. In contrast, ultraselection incorporates additional molecular alterations like HER2 amplifications, EGFR ectodomain mutations, PIK3CA exon 20 mutations, PTEN loss, MET amplifications, and AKT1 mutations, as well as clinical factors like tumor sidedness (LCRC), MSS, and MSI-H status150. Contrastingly, PARADIGM16 highlighted tumor sidedness as a key clinical factor, demonstrating the importance of integrating both molecular and clinical factors to improve therapeutic outcomes.
These strategies align with the results of the PARADIGM trial, which demonstrated a significantly longer OS in the negatively hyperselected population treated with anti-EGFR therapy compared to those receiving anti-VEGF therapy (40.7 vs. 34.4 months; p = 0.037), particularly in patients with LCRC149. The PRESSING studies147,148 developed a PRESSING gene panel, which includes genetic alterations previously associated with primary resistance to anti-EGFR therapies. Furthermore, the study showed that PRESSING-negative patients with LCRC receiving first-line FOLFOX plus panitumumab achieved an impressive response rate of 77.3%, with a median PFS of 13.2 months and a 2-year OS rate of 69.7%150.
In contrast, patients with confirmed genetic alterations exhibited comparable or inferior survival outcomes under anti-EGFR therapy compared to anti-VEGF therapy, regardless of tumor location147,148. In the PARADIGM study, patients with LCRC and genetic alterations treated with anti-EGFR therapy had a median OS of 24.2 months, compared to 26.4 months with anti-VEGF treatment. For patients with RCRC, OS was 14.1 months with anti-EGFR therapy and 18.5 months with anti-VEGF therapy149. Similarly, the PRESSING2 study revealed that patients with a positive PRESSING2 status had significantly worse PFS and OS compared to those with a negative status (median PFS: 7.4 vs. 13.0 months; OS: 22.6 vs. 48.8 months)150. In patients with LCRC, median PFS was 6.5 months versus 12.9 months, while median OS was 28.0 months vs 51.2 months150. For RCRC, median PFS was 6.3 months versus 9.4 months, and median OS was 18.1 months versus 27.7 months150.
Liquid biopsy data suggest that monitoring molecular changes in the ctDNA following the removal of EGFR inhibitor pressure could potentially indicate renewed sensitivity to anti-EGFR therapies, supporting its role in evaluating patients for retreatment16,151. This observation underpinned the rationale for the FIRE-4 study, which adopted a novel “switch maintenance concept”15. In the first-line setting, after an initial response to anti-EGFR therapy, an early switch to anti-VEGF treatment was implemented to achieve and sustain deep remission. In patients treated within the third-line, the efficacy of re-exposing patients to cetuximab after a period of EGFR-free therapy is being explored15.
Studies such as the CHRONOS151 and CRICKET152 trials have investigated this re-challenge strategy, demonstrating that patients who initially responded well to cetuximab may experience renewed benefit from this therapy following the development of resistance. Moreover, the rationale for using liquid biopsy has emerged to identify the subpopulation of patients who are more likely to respond again upon re-induction, by detecting ctDNA and monitoring RAS mutation status. This approach helps tailor treatment by selecting patients who may derive the most benefit from cetuximab reintroduction151,152.
The results of the FIRE-415, PRESSING147,148, and PARADIGM149 studies highlight the growing importance of molecular analyses in optimizing treatment strategies for patients with mCRC. Genetic alterations associated with resistance to anti-EGFR therapies are more frequently observed in RCRC than in LCRC (approximately 49.7% vs. 26%)149. These studies prospectively demonstrated that hyper- and ultraselection can identify RCRC patients who may still benefit from anti-EGFR therapies. This patient subset can be identified through comprehensive molecular profiling, incorporating genome-based ultraselection, elevated AREG/EREG expression, or the CMS2/epithelial subtype based on transcriptomics. In LCRC, the evaluation of PRESSING(2) alterations can further refine patient selection for anti-EGFR therapies, particularly when alternative first-line options are considered. Nevertheless, further studies are necessary to validate this approach.
Future perspective and conclusion
Recent advancements in understanding the molecular and immunological differences between RCRC and LCRC offer promising avenues for refining treatment approaches. Moving forward, the binary classification of CRC based on anatomical location is likely to be further nuanced by more sophisticated molecular profiling methods. As recent studies have shown, the introduction of CMS and the increasing availability of advanced genomic and proteomic tools allow for a deeper understanding of the distinct tumor behaviors that transcend anatomical distinctions55,79,83,153,154. In this context, the studies by Yamauchi et al.60,61 challenge the traditional dichotomy between proximal and distal CRC and instead proposes a continuous spectrum of molecular changes along the colon (Colorectal Continuum Model). When this model is combined with spatial multi-omics and single-cell RNA sequencing, the model captures tumor molecular diversity and cellular context in exceptional detail. This integration precisely maps cell types and reveals spatial heterogeneity of CMS and key intercellular communication events at the tumor–stroma interface. Such insights challenge traditional binary classifications and pave the way for personalized CRC therapies155. Building on this enhanced resolution, landmark studies have introduced the concept of a colorectal continuum, challenging the traditional binary classification and laying a robust foundation for numerous subsequent investigations into the molecular and clinical heterogeneity of CRC.
Such insights are paving the way for personalized therapies that go beyond the traditional categorization, potentially optimizing patient outcomes through more targeted approaches.
Despite these promising developments, translating molecular knowledge into clinical practice remains a significant challenge, particularly in RCRC, where outcomes, such as OS and response to therapies lag behind those of LCRC13.
While the application of targeted therapies, such as anti-EGFR and anti-VEGF agents, has shown clear efficacy in LCRC, particularly in RAS wt cases, the same level of success has not been achieved for RCRC11,126. One area of ongoing interest is in understanding the mechanisms driving this differential response, particularly regarding resistance to anti-EGFR therapy. A promising strategy to overcome this resistance is the dual inhibition of KRAS G12C and EGFR with anti-KRAS and anti-EGFR inhibitors (i.e., sotorasib and panitumumab)67.
Liquid-biopsy technologies and ctDNA analysis are shedding light on the temporal evolution of resistance, providing opportunities to refine treatment strategies dynamically15,151. However, these approaches are not yet fully integrated into standard of care, likely due to technical, financial, and accessibility limitations. In particular, when we delve deeper into the implementation of dynamic ctDNA monitoring, two main technical challenges become evident: first, the sensitivity of current assays is limited due to the very low concentrations of ctDNA and the small number of genome equivalents in plasma samples. Second, the high costs associated with ultra-deep sequencing and extensive bioinformatic analyses further complicate its routine implementation. To overcome these obstacles, promising solutions include integrating combined multi-omics approaches—which merge genomic, transcriptomic, proteomic, and epigenetic data—to enhance diagnostic accuracy, and optimizing ultra-deep sequencing methods to improve sensitivity while reducing costs156. Building on these technological advancements that enhance our diagnostic capabilities, there is also growing interest in translating these insights into novel therapeutic strategies. At the same time, immunotherapy represents a promising but underdeveloped frontier in CRC, particularly for RCRC. High MSI-H tumors have shown responsiveness to immune checkpoint inhibitors157,158, but the vast majority of RCRC cases do not fall into this category. However, MSI-H tumors present a prognostic paradox: while they demonstrate a survival advantage in early-stage disease due to strong immune surveillance, this reverses in metastatic settings where they exhibit poorer outcomes. Immune evasion, stromal reorganization, and therapy resistance contribute to this shift, making MSI-H tumors in stage IV particularly challenging to treat55,135,136,137. The immunosuppressive microenvironment83 observed in RCRC poses a considerable challenge for immune-based treatments, and overcoming this will require innovative approaches - possibly through combination therapies that enhance immune activation within the TME159. In this context, the Immunoscore shows great potential as a prognostic tool. As highlighted in the Atezo-Tribe160 study, paying attention to the Immunoscore may offer valuable insights for improving treatment strategies by identifying patients who are most likely to benefit from immune-based therapies. The study observed that patients with high Immunoscore Immune-Checkpoint (IC) and/or high TMB derived greater benefits from the addition of atezolizumab, improving OS in patients with mCRC160. This suggests that integrating the Immunoscore into clinical practice could help tailor treatments more effectively.
Another critical factor shaping future treatments is the role of the gut microbiome. Recent discoveries regarding its influence on both tumor progression and therapeutic resistance, particularly in RCRC, suggest that modulating the microbiome could complement existing systemic treatments88. Building on this, phage-based approaches offer promising perspectives in RCRC by not only modulating the gut microbiome to alleviate bacterial dysbiosis and enhance the tumor immune milieu, but also by leveraging a continuous spectrum model refined by spatial multi-omics technologies to capture molecular tumor profiles and interactions with pathogenic bacteria161.
Looking ahead, the next wave of CRC treatment is likely to emphasize a more integrated approach - one that combines molecular profiling, real-time monitoring through liquid-biopsies, immune modulation, and possibly microbiome-targeted therapies. While the path to clinical application is fraught with challenges, particularly regarding cost, accessibility, and patient stratification, these advances hold great promise for personalizing treatment strategies and improving outcomes, especially for patients with right-sided tumors, who have historically faced poorer prognoses. Ongoing clinical trials and technological developments will continue to redefine the treatment landscape, potentially shifting the focus from anatomical to molecular determinants of CRC progression. As our understanding deepens, the prospect of a more tailored, effective approach to CRC treatment appears within reach, heralding a new era of personalized oncology.
Data availability
No datasets were generated or analysed during the current study.
Abbreviations
- CMS:
-
Consensus molecular subtypes
- CRC:
-
Colorectal Cancer
- ctDNA:
-
Circulating tumor DNA
- EGFR:
-
Epithelial growth factor receptor
- ETBF:
-
Enterotoxigenic Bacteroides fragilis
- GIT:
-
Gastrointestinal tract
- HR:
-
Hazard Ratio
- ICI:
-
Immune cell infiltration
- IMA:
-
Inferior mesenteric artery
- LCRC:
-
Left-sided colorectal cancer
- mCRC:
-
Metastatic colorectal cancer
- MSI (-H):
-
Microsatellite instability (High)
- OS:
-
Overall survival
- PFS:
-
Progression-free survival
- RCRC:
-
Right-sided colorectal cancer
- SMA:
-
Superior mesenteric artery
- TMB:
-
Tumor mutational burden
- TME:
-
Tumor Microenvironment
- VEGF(R):
-
Vascular Endothelial Growth Factor (Receptor)
- wt:
-
Wild-type
References
Siegel, R. L., Wagle, N. S., Cercek, A., Smith, R. A. & Jemal, A. Colorectal cancer statistics, 2023. Ca. Cancer J. Clin. 73, 233–254 (2023).
Biller, L. H. & Schrag, D. Diagnosis and treatment of metastatic colorectal cancer: a review. JAMA 325, 669–685 (2021).
Seeber, A. & Gastl, G. Targeted therapy of colorectal cancer. Oncol. Res. Treat. 39, 796–802 (2016).
Stintzing, S. et al. FOLFIRI plus cetuximab versus FOLFIRI plus bevacizumab as first-line treatment for patients with metastatic colorectal cancer-subgroup analysis of patients with KRAS: mutated tumours in the randomised German AIO study KRK-0306. Ann. Oncol. 23, 1693–1699 (2012).
Douillard, J.-Y. et al. Panitumumab-FOLFOX4 treatment and RAS mutations in colorectal cancer. N. Engl. J. Med. 369, 1023–1034 (2013).
Hurwitz, H. et al. Bevacizumab plus irinotecan, fluorouracil, and leucovorin for metastatic colorectal cancer. N. Engl. J. Med. 350, 2335–2342 (2004).
Tabernero, J. et al. Ramucirumab versus placebo in combination with second-line FOLFIRI in patients with metastatic colorectal carcinoma that progressed during or after first-line therapy with bevacizumab, oxaliplatin, and a fluoropyrimidine (RAISE): a randomised, double-blind, multicentre, phase 3 study. Lancet Oncol. 16, 499–508 (2015).
Van Cutsem, E. et al. Addition of aflibercept to fluorouracil, leucovorin, and irinotecan improves survival in a phase III randomized trial in patients with metastatic colorectal cancer previously treated with an oxaliplatin-based regimen. J. Clin. Oncol. 30, 3499–3506 (2012).
Punt, C. J. A., Koopman, M. & Vermeulen, L. From tumour heterogeneity to advances in precision treatment of colorectal cancer. Nat. Rev. Clin. Oncol. 14, 235–246 (2017).
Di Nicolantonio, F. et al. Precision oncology in metastatic colorectal cancer—from biology to medicine. Nat. Rev. Clin. Oncol. 18, 506–525 (2021).
Tejpar, S. et al. Prognostic and predictive relevance of primary tumor location in patients with RAS wild-type metastatic colorectal cancer retrospective analyses of the CRYSTAL and FIRE-3 trials. JAMA Oncol. 3, 194–201 (2017).
Arnold, D. et al. Prognostic and predictive value of primary tumour side in patients with RAS wild-type metastatic colorectal cancer treated with chemotherapy and EGFR directed antibodies in six randomized trials. Ann. Oncol.28, 1713–1729 (2017).
Watanabe, J. et al. Panitumumab vs bevacizumab added to standard first-line chemotherapy and overall survival among patients with RAS wild-type, left-sided metastatic colorectal cancer: a randomized clinical trial. JAMA 329, 1271–1282 (2023).
Cervantes, A. et al. Metastatic colorectal cancer: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann. Oncol.34, 10–32 (2023).
Stintzing, S. et al. Phase III FIRE-4 study (AIO KRK-0114): Evaluation of first-line treatment efficacy of FOLFIRI/cetuximab in patients with RAS-WT mCRC receiving the first cycle of treatment with chemotherapy only. J. Clin. Oncol. 41, 100–100 (2023).
Shitara, K. et al. Baseline ctDNA gene alterations as a biomarker of survival after panitumumab and chemotherapy in metastatic colorectal cancer. Nat. Med. 30, 730–739 (2024).
Moore, K. L., Persaud, T. V. N. & Torchia, M. G. The Developing Human: Clinically Oriented Embryology (Elsevier, 2016).
Spence, J. R., Lauf, R. & Shroyer, N. F. Vertebrate intestinal endoderm development. Dev. Dyn.240, 501–520 (2011).
Roberts, D. J. Molecular mechanisms of development of the gastrointestinal tract. Dev. Dyn.219, 109–120 (2000).
Wallace, A. S. & Burns, A. J. Development of the enteric nervous system, smooth muscle and interstitial cells of Cajal in the human gastrointestinal tract. Cell Tissue Res. 319, 367–382 (2005).
Kostouros, A. et al. Large intestine embryogenesis: molecular pathways and related disorders (Review). Int. J. Mol. Med. 46, 27–57 (2020).
Liu, Y. et al. Comparative molecular analysis of gastrointestinal adenocarcinomas. Cancer Cell 33, 721–735.e8 (2018).
Joanito, I. et al. Single-cell and bulk transcriptome sequencing identifies two epithelial tumor cell states and refines the consensus molecular classification of colorectal cancer. Nat. Genet. 54, 963–975 (2022).
Benedix, F. et al. Comparison of 17,641 patients with right- and left-sided colon cancer: differences in epidemiology, perioperative course, histology, and survival. Dis. Colon Rectum 53, 57–64 (2010).
Ghazi, S. et al. Analysis of colorectal cancer morphology in relation to sex, age, location, and family history. J. Gastroenterol. 47, 619–634 (2012).
Hansen, T. F. et al. Prognostic impact of CDX2 in stage II colon cancer: results from two nationwide cohorts. Br. J. Cancer 119, 1367–1373 (2018).
Augestad, K. M. et al. Metastatic spread pattern after curative colorectal cancer surgery. A retrospective, longitudinal analysis. Cancer Epidemiol. 39, 734–744 (2015).
Price, T. J. et al. Does the primary site of colorectal cancer impact outcomes for patients with metastatic disease?. Cancer 121, 830–835 (2015).
Li, C. et al. Prognosis of three histological subtypes of colorectal adenocarcinoma: a retrospective analysis of 8005 Chinese patients. Cancer Med. 8, 3411–3419 (2019).
Huemer, F. et al. Sidedness and TP53 mutations impact OS in anti-EGFR but not anti-VEGF treated mCRC—an analysis of the KRAS registry of the AGMT (Arbeitsgemeinschaft Medikamentöse Tumortherapie). BMC Cancer 18, 11 (2018).
Andrici, J. et al. Mismatch repair deficiency as a prognostic factor in mucinous colorectal cancer. Mod. Pathol. 29, 266–274 (2016).
Kastinen, M. et al. Immunological and prognostic significance of tumour necrosis in colorectal cancer. Br. J. Cancer 128, 2218–2226 (2023).
Bozkurt, E. et al. TRAIL signaling promotes entosis in colorectal cancer. J. Cell Biol. 220, e202010030 (2021).
Marx, A. H. et al. High-grade intratumoral tumor budding is a predictor for lymphovascular invasion and adverse outcome in stage II colorectal cancer. Int. J. Colorectal Dis. 35, 259–268 (2020).
Eriksen, A. C. et al. The prognostic value of tumour stroma ratio and tumour budding in stage II colon cancer. A nationwide population-based study. Int. J. Colorectal Dis. 33, 1115–1124 (2018).
Fan, S. et al. Prognostic value of desmoplastic stromal reaction, tumor budding and tumor-stroma ratio in stage II colorectal cancer. J. Gastrointest. Oncol. 13, (2022).
Pinto, M. L. et al. The two faces of tumor-associated macrophages and their clinical significance in colorectal cancer. Front. Immunol. 10, (2019).
Haruki, K. et al. An integrated analysis of lymphocytic reaction, tumour molecular characteristics and patient survival in colorectal cancer. Br. J. Cancer 122, 1367–1377 (2020).
Klintrup, K. et al. Inflammation and prognosis in colorectal cancer. Eur. J. Cancer 41, 2645–2654 (2005).
Briede, I., Strumfa, I., Vanags, A. & Gardovskis, J. The association between inflammation, epithelial mesenchymal transition and stemness in colorectal carcinoma. J. Inflamm. Res. 13, 15–34 (2020).
Ueno, H. et al. Prognostic value of desmoplastic reaction characterisation in stage II colon cancer: prospective validation in a Phase 3 study (SACURA Trial). Br. J. Cancer 124, 1088–1097 (2021).
Martin, B. et al. Stroma AReactive Invasion Front Areas (SARIFA)—a new easily to determine biomarker in colon cancer—results of a retrospective study. Cancers 13, 4880 (2021).
Van Pelt, G. et al. Stroma-high lymph node involvement predicts poor survival more accurately for patients with stage III colon cancer. J. Med. Surg. Pathol. 1, (2016).
Strous, M. T. A. et al. A high tumour-stroma ratio (TSR) in colon tumours and its metastatic lymph nodes predicts poor cancer-free survival and chemo resistance. Clin. Transl. Oncol. 24, 1047–1058 (2022).
Kang, G., Pyo, J.-S., Kim, N.-Y. & Kang, D.-W. Clinicopathological significances of tumor–stroma ratio (TSR) in colorectal cancers: prognostic implication of TSR compared to hypoxia-inducible factor-1α expression and microvessel density. Curr. Oncol. 28, 1314–1324 (2021).
Yamaoka, Y. et al. The distribution of lymph node metastases and their size in colon cancer. Langenbecks Arch. Surg. 402, 1213–1221 (2017).
Jess, P., Hansen, I. O., Gamborg, M. & Jess, T. A nationwide Danish cohort study challenging the categorisation into right-sided and left-sided colon cancer. BMJ Open 3, e002608 (2013).
Rössler, O. et al. Tumor size, tumor location, and antitumor inflammatory response are associated with lymph node size in colorectal cancer patients. Mod. Pathol. 30, 897–904 (2017).
Argilés, G. et al. Localised colon cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up†. Ann. Oncol. 31, 1291–1305 (2020).
Bae, J. M., Lee, T. H., Cho, N.-Y., Kim, T.-Y. & Kang, G. H. Loss of CDX2 expression is associated with poor prognosis in colorectal cancer patients. World J. Gastroenterol. 21, 1457–1467 (2015).
Konukiewitz, B. et al. Loss of CDX2 in colorectal cancer is associated with histopathologic subtypes and microsatellite instability but is prognostically inferior to hematoxylin–eosin-based morphologic parameters from the WHO classification. Br. J. Cancer 125, 1632–1646 (2021).
Ma, C. et al. SATB2 and CDX2 are prognostic biomarkers in DNA mismatch repair protein deficient colon cancer. Mod. Pathol. 32, 1217–1231 (2019).
Schmitt, M. et al. Loss of SATB2 occurs more frequently than CDX2 loss in colorectal carcinoma and identifies particularly aggressive cancers in high-risk subgroups. Cancers 13, 6177 (2021).
Hrudka, J., Fišerová, H., Jelínková, K., Matěj, R. & Waldauf, P. Cytokeratin 7 expression as a predictor of an unfavorable prognosis in colorectal carcinoma. Sci. Rep. 11, 17863 (2021).
Guinney, J. et al. The consensus molecular subtypes of colorectal cancer. Nat. Med. 21, 1350–1356 (2015).
Lee, M. S., Menter, D. G. & Kopetz, S. Right versus left colon cancer biology: integrating the consensus molecular subtypes. J. Natl Compr. Cancer Netw. 15, 411–419 (2017).
Testa, U., Pelosi, E. & Castelli, G. Colorectal cancer: genetic abnormalities, tumor progression, tumor heterogeneity, clonal evolution and tumor-initiating cells. Med. Sci.6, 31 (2018).
Caiazza, F., Ryan, E. J., Doherty, G., Winter, D. C. & Sheahan, K. Estrogen receptors and their implications in colorectal carcinogenesis. Front. Oncol. 5, 19 (2015).
Ugai, T. et al. Molecular characteristics of early-onset colorectal cancer according to detailed anatomical locations: comparison with later-onset cases. Am. J. Gastroenterol. 118, 712–726 (2023).
Yamauchi, M. et al. Colorectal cancer: a tale of two sides or a continuum? Gut 61, 794–797 (2012).
Yamauchi, M. et al. Assessment of colorectal cancer molecular features along bowel subsites challenges the conception of distinct dichotomy of proximal versus distal colorectum. Gut 61, 847–854 (2012).
Dallavilla, T. et al. Differences in the molecular organisation of tumours along the colon are linked to interactions within the tumour ecosystem. Biochim. Biophys. Acta Mol. Basis Dis. 1870, 167311 (2024).
Ugai, T. et al. Prognostic role of detailed colorectal location and tumor molecular features: analyses of 13,101 colorectal cancer patients including 2994 early-onset cases. J. Gastroenterol. 58, 229–245 (2023).
Ito, M. et al. MicroRNA-31 expression in relation to BRAF mutation, CpG island methylation and colorectal continuum in serrated lesions. Int. J. Cancer 135, 2507–2515 (2014).
Missiaglia, E. et al. Distal and proximal colon cancers differ in terms of molecular, pathological, and clinical features. Ann. Oncol. 25, 1995–2001 (2014).
Seeburg, P. H., Colby, W. W., Capon, D. J., Goeddel, D. V. & Levinson, A. D. Biological properties of human c-Ha-ras1 genes mutated at codon 12. Nature 312, 71–75 (1984).
Fakih, M. G. et al. Sotorasib plus panitumumab in refractory colorectal cancer with mutated KRAS G12C. N. Engl. J. Med. 389, 2125–2139 (2023).
Fraissenon, A. et al. Sotorasib for vascular malformations associated with KRAS G12C mutation. N. Engl. J. Med. 391, 334–342 (2024).
Salem, M. E. et al. Comparative molecular analyses of left-sided colon, right-sided colon, and rectal cancers. Oncotarget 8, 86356–86368 (2017).
Mukund, K., Syulyukina, N., Ramamoorthy, S. & Subramaniam, S. Right and left-sided colon cancers - specificity of molecular mechanisms in tumorigenesis and progression. BMC Cancer 20, 317 (2020).
Zheng, Z. et al. T cells in colorectal cancer: unravelling the function of different T cell subsets in the tumor microenvironment. Int. J. Mol. Sci. 24, 11673 (2023).
MicroRNAs associated with biological pathways of left- and right-sided colorectal cancer. Anticancer Res. https://ar.iiarjournals.org/content/40/7/3713/tab-figures-data.
Bergers, G. & Fendt, S.-M. The metabolism of cancer cells during metastasis. Nat. Rev. Cancer 21, 162–180 (2021).
Jain, A. et al. Charting the metabolic biogeography of the colorectum in cancer: challenging the right sided versus left sided classification. Mol. Cancer 23, 211 (2024).
Deng, K. et al. Plasma metabolomic profiling distinguishes right-sided from left-sided colon cancer. Clin. Chim. Acta Int. J. Clin. Chem. 487, 357–362 (2018).
Cai, Y. et al. Bile acid distributions, sex-specificity, and prognosis in colorectal cancer. Biol. Sex. Differ. 13, 61 (2022).
Shen, X. et al. Evaluation of peripheral blood inflammation indexes as prognostic markers for colorectal cancer metastasis. Sci. Rep. 14, 20489 (2024).
Kwon, Y. W. et al. Application of proteomics in cancer: recent trends and approaches for biomarkers discovery. Front. Med. 8, 747333 (2021).
Imperial, R. et al. Comparative proteogenomic analysis of right-sided colon cancer, left-sided colon cancer and rectal cancer reveals distinct mutational profiles. Mol. Cancer 17, 177 (2018).
Ullah, I. et al. Multi-omics approaches in colorectal cancer screening and diagnosis, recent updates and future perspectives. Cancers 14, 5545 (2022).
Bejarano, L., Jordāo, M. J. C. & Joyce, J. A. Therapeutic targeting of the tumor microenvironment. Cancer Discov. 11, 933–959 (2021).
Rajamäki, K. et al. Genetic and epigenetic characteristics of inflammatory bowel disease–associated colorectal cancer. Gastroenterology 161, 592–607 (2021).
Hong, Z. et al. Proteomic profiling and tumor microenvironment characterization reveal molecular and immunological hallmarks of left-sided and right-sided colon cancer tumorigenesis. J. Proteome Res. 22, 2973–2984 (2023).
Guo, W. et al. Resolving the difference between left-sided and right-sided colorectal cancer by single-cell sequencing. JCI Insight 7, e152616 (2022).
Tilg, H., Adolph, T. E., Gerner, R. R. & Moschen, A. R. The intestinal microbiota in colorectal cancer. Cancer Cell 33, 954–964 (2018).
Wong, S. H. & Yu, J. Gut microbiota in colorectal cancer: mechanisms of action and clinical applications. Nat. Rev. Gastroenterol. Hepatol. 16, 690–704 (2019).
White, M. T. & Sears, C. L. The microbial landscape of colorectal cancer. Nat. Rev. Microbiol. 22, 240–254 (2024).
Irrazábal, T., Belcheva, A., Girardin, S. E., Martin, A. & Philpott, D. J. The multifaceted role of the intestinal microbiota in colon cancer. Mol. Cell 54, 309–320 (2014).
Thomas, A. M. et al. Metagenomic analysis of colorectal cancer datasets identifies cross-cohort microbial diagnostic signatures and a link with choline degradation. Nat. Med. 25, 667–678 (2019).
Wirbel, J. et al. Meta-analysis of fecal metagenomes reveals global microbial signatures that are specific for colorectal cancer. Nat. Med. 25, 679–689 (2019).
Ternes, D. et al. Microbiome in colorectal cancer: how to get from meta-omics to mechanism?. Trends Microbiol. 28, 401–423 (2020).
Flemer, B. et al. Tumour-associated and non-tumour-associated microbiota in colorectal cancer. Gut 66, 633–643 (2017).
Kneis, B. et al. Colon cancer microbiome landscaping: differences in right- and left-sided colon cancer and a tumor microbiome-ileal microbiome association. Int. J. Mol. Sci. 24, 3265 (2023).
Mouradov, D. et al. Oncomicrobial community profiling identifies clinicomolecular and prognostic subtypes of colorectal cancer. Gastroenterology 165, 104–120 (2023).
Roberti, M. P. et al. Chemotherapy-induced ileal crypt apoptosis and the ileal microbiome shape immunosurveillance and prognosis of proximal colon cancer. Nat. Med. 26, 919–931 (2020).
Picard, M. et al. Ileal immune tonus is a prognosis marker of proximal colon cancer in mice and patients. Cell Death Differ. 28, 1532–1547 (2021).
Allen-Vercoe, E. & Jobin, C. Fusobacterium and Enterobacteriaceae: important players for CRC?. Immunol. Lett. 162, 54–61 (2014).
Dougherty, M. W. & Jobin, C. Intestinal bacteria and colorectal cancer: etiology and treatment. Gut Microbes 15, 2185028.
Signat, B., Roques, C., Poulet, P. & Duffaut, D. Role of fusobacterium nucleatum in periodontal health and disease. Curr. Issues Mol. Biol. 13, 25–36 (2011).
Abed, J. et al. Fap2 mediates fusobacterium nucleatum colorectal adenocarcinoma enrichment by binding to tumor-expressed Gal-GalNAc. Cell Host Microbe 20, 215–225 (2016).
Kostic, A. D., Xavier, R. J. & Gevers, D. The microbiome in inflammatory bowel disease: current status and the future ahead. Gastroenterology 146, 1489–1499 (2014).
Kostic, A. D. et al. Fusobacterium nucleatum potentiates intestinal tumorigenesis and modulates the tumor immune microenvironment. Cell Host Microbe 14, 207–215 (2013).
Segata, N. et al. Composition of the adult digestive tract bacterial microbiome based on seven mouth surfaces, tonsils, throat and stool samples. Genome Biol. 13, R42 (2012).
Komiya, Y. et al. Patients with colorectal cancer have identical strains of Fusobacterium nucleatum in their colorectal cancer and oral cavity. Gut 68, 1335–1337 (2019).
Abed, J. et al. Colon cancer-associated fusobacterium nucleatum may originate from the oral cavity and reach colon tumors via the circulatory system. Front. Cell. Infect. Microbiol. 10, 400 (2020).
Gur, C. et al. Binding of the Fap2 Protein of Fusobacterium nucleatum to human inhibitory receptor TIGIT protects tumors from immune cell attack. Immunity 42, 344–355 (2015).
Yu, J. et al. Invasive Fusobacterium nucleatum may play a role in the carcinogenesis of proximal colon cancer through the serrated neoplasia pathway. Int. J. Cancer 139, 1318–1326 (2016).
Mima, K. et al. Fusobacterium nucleatum in colorectal carcinoma tissue according to tumor location. Clin. Transl. Gastroenterol. 7, e200 (2016).
Zepeda-Rivera, M. et al. A distinct Fusobacterium nucleatum clade dominates the colorectal cancer niche. Nature 1–9 https://doi.org/10.1038/s41586-024-07182-w. (2024)
Høiby, N. et al. The clinical impact of bacterial biofilms. Int. J. Oral. Sci. 3, 55–65 (2011).
Dejea, C. M. et al. Microbiota organization is a distinct feature of proximal colorectal cancers. Proc. Natl. Acad. Sci. 111, 18321–18326 (2014).
Li, S., Konstantinov, S. R., Smits, R. & Peppelenbosch, M. P. Bacterial biofilms in colorectal cancer initiation and progression. Trends Mol. Med. 23, 18–30 (2017).
Dejea, C. M. & Sears, C. L. Do biofilms confer a pro-carcinogenic state? Gut Microbes 7, 54–57 (2016).
Drewes, J. L. et al. High-resolution bacterial 16S rRNA gene profile meta-analysis and biofilm status reveal common colorectal cancer consortia. NPJ Biofilms Microbiomes 3, 34 (2017).
Tomkovich, S. et al. Human colon mucosal biofilms from healthy or colon cancer hosts are carcinogenic. J. Clin. Investig. 129, 1699–1712.
Dejea, C. M. et al. Patients with familial adenomatous polyposis harbor colonic biofilms containing tumorigenic bacteria. Science 359, 592–597 (2018).
Pleguezuelos-Manzano, C. et al. Mutational signature in colorectal cancer caused by genotoxic pks+ E. coli. Nature 580, 269–273 (2020).
Dziubańska-Kusibab, P. J. et al. Colibactin DNA-damage signature indicates mutational impact in colorectal cancer. Nat. Med. 26, 1063–1069 (2020).
Buc, E. et al. High prevalence of mucosa-associated E. coli producing cyclomodulin and genotoxin in colon cancer. PLoS ONE 8, e56964 (2013).
Iyadorai, T. et al. Prevalence and association of pks+ Escherichia coli with colorectal cancer in patients at the University Malaya Medical Centre, Malaysia. PLoS ONE 15, e0228217 (2020).
Boleij, A. et al. The Bacteroides fragilis toxin gene is prevalent in the colon mucosa of colorectal cancer patients. Clin. Infect. Dis.60, 208–215 (2015).
Purcell, R. V. et al. Colonization with enterotoxigenic Bacteroides fragilis is associated with early-stage colorectal neoplasia. PLOS ONE 12, e0171602 (2017).
Chung, L. et al. Bacteroides fragilis toxin coordinates a pro-carcinogenic inflammatory cascade via targeting of colonic epithelial cells. Cell Host Microbe 23, 203–214.e5 (2018).
Heinemann, V. et al. FOLFIRI plus cetuximab or bevacizumab for advanced colorectal cancer: final survival and per-protocol analysis of FIRE-3, a randomised clinical trial. Br. J. Cancer 124, 587–594 (2021).
Cremolini, C. et al. Primary tumor sidedness and benefit from FOLFOXIRI plus bevacizumab as initial therapy for metastatic colorectal cancer. Retrospective analysis of the TRIBE trial by GONO. Ann. Oncol. 29, 1528–1534 (2018).
Van Cutsem, E. et al. Cetuximab plus irinotecan, fluorouracil, and leucovorin as first-line treatment for metastatic colorectal cancer: updated analysis of overall survival according to tumor KRAS and BRAF mutation status. J. Clin. Oncol. 29, 2011–2019 (2011).
Peeters, M. et al. Relationships between tumour response and primary tumour location, and predictors of long-term survival, in patients with RAS wild-type metastatic colorectal cancer receiving first-line panitumumab therapy: retrospective analyses of the PRIME and PEAK clinical trials. Br. J. Cancer 119, 303–312 (2018).
Douillard, J.-Y. et al. Randomized, phase III trial of panitumumab with infusional fluorouracil, leucovorin, and oxaliplatin (FOLFOX4) versus FOLFOX4 alone as first-line treatment in patients with previously untreated metastatic colorectal cancer: the PRIME study. J. Clin. Oncol. 28, 4697–4705 (2010).
Venook, A. P. et al. Impact of primary (1o) tumor location on overall survival (OS) and progression-free survival (PFS) in patients (pts) with metastatic colorectal cancer (mCRC): Analysis of CALGB/SWOG 80405 (Alliance). J. Clin. Oncol. 34, 3504–3504 (2016).
Boeckx, N. et al. Primary tumor sidedness has an impact on prognosis and treatment outcome in metastatic colorectal cancer: results from two randomized first-line panitumumab studies. Ann. Oncol. 28, 1862–1868 (2017).
Cremolini, C., Antoniotti, C., Moretto, R., Masi, G. & Falcone, A. First-line therapy for mCRC—the influence of primary tumour location on the therapeutic algorithm. Nat. Rev. Clin. Oncol. 14, 113–113 (2017).
Lonardi, S. et al. Initial panitumumab plus fluorouracil, leucovorin, and oxaliplatin or plus fluorouracil and leucovorin in elderly patients with RAS and BRAF wild-type metastatic colorectal cancer: the PANDA trial by the GONO Foundation. J. Clin. Oncol.41, 5263–5273 (2023).
Alig, A. H. S. et al. Exact primary tumor location in mCRC: prognostic value and predictive impact on anti-EGFR mAb efficacy. Cancers 14, 526 (2022).
Giordano, G. et al. Evidence-based second-line treatment in RAS wild-type/mutated metastatic colorectal cancer in the precision medicine era. Int. J. Mol. Sci. 22, 7717 (2021).
Uhlig, J. et al. Microsatellite instability and KRAS mutation in stage IV colorectal cancer: prevalence, geographic discrepancies, and outcomes from the national cancer database. J. Natl Compr. Cancer Netw. JNCCN 19, 307–318 (2021).
Tomasello, G. et al. Survival benefit with adjuvant chemotherapy in stage III microsatellite-high/deficient mismatch repair colon cancer: a systematic review and meta-analysis. Sci. Rep. 12, 1055 (2022).
Petrelli, F. et al. Microsatellite instability and survival in stage II colorectal cancer: a systematic review and meta-analysis. Anticancer Res. 39, 6431–6441 (2019).
Andre, T. et al. Nivolumab plus ipilimumab in microsatellite-instability-high metastatic colorectal cancer. N. Engl. J. Med. 391, 2014–2026 (2024).
Pietrantonio, F. et al. Heterogeneity of acquired resistance to anti-EGFR monoclonal antibodies in patients with metastatic colorectal cancer. Clin. Cancer Res. 23, 2414–2422 (2017).
Parseghian, C. M., Napolitano, S., Loree, J. M. & Kopetz, S. Mechanisms of innate and acquired resistance to anti-EGFR therapy: a review of current knowledge with a focus on rechallenge therapies. Clin. Cancer Res. J. Am. Assoc. Cancer Res. 25, 6899–6908 (2019).
Parseghian, C., Eluri, M., Kopetz, S. & Raghav, K. Mechanisms of resistance to EGFR-targeted therapies in colorectal cancer: more than just genetics. Front. Cell Dev. Biol. 11, 1176657 (2023).
Huang, S. et al. Dual targeting of EGFR and HER3 with MEHD7945A overcomes acquired resistance to EGFR inhibitors and radiation. Cancer Res. 73, 824–833 (2013).
Luraghi, P. et al. MET signaling in colon cancer stem-like cells blunts the therapeutic response to EGFR inhibitors. Cancer Res. 74, 1857–1869 (2014).
Vakiani, E. et al. Comparative genomic analysis of primary versus metastatic colorectal carcinomas. J. Clin. Oncol.30, 2956–2962 (2012).
Strickler, J. H. et al. Genomic landscape of cell-free DNA in patients with colorectal cancer. Cancer Discov. 8, 164–173 (2018).
Montagut, C. et al. Efficacy of Sym004 in patients with metastatic colorectal cancer with acquired resistance to anti-EGFR therapy and molecularly selected by circulating tumor DNA analyses: a phase 2 randomized clinical trial. JAMA Oncol. 4, e175245 (2018).
Cremolini, C. et al. Individual patient data meta-analysis of FOLFOXIRI plus bevacizumab versus doublets plus bevacizumab as initial therapy of unresectable metastatic colorectal cancer. J. Clin. Oncol. JCO2001225 https://doi.org/10.1200/JCO.20.01225 (2020)
Morano, F. et al. Negative hyperselection of patients with RAS and BRAF wild-type metastatic colorectal cancer who received panitumumab-based maintenance therapy. J. Clin. Oncol. 37, 3099–3110 (2019).
Baseline ctDNA gene alterations as a biomarker of survival after panitumumab and chemotherapy in metastatic colorectal cancer. https://pubmed.ncbi.nlm.nih.gov/38347302/.
Randon, G. et al. Negative ultraselection of patients with RAS/BRAF wild-type, microsatellite-stable metastatic colorectal cancer receiving anti-EGFR-based therapy. JCO Precis. Oncol. 6, e2200037 (2022).
Sartore-Bianchi, A. et al. Circulating tumor DNA to guide rechallenge with panitumumab in metastatic colorectal cancer: the phase 2 CHRONOS trial. Nat. Med. 28, 1612–1618 (2022).
Cremolini, C. et al. Rechallenge for patients with RAS and BRAF wild-type metastatic colorectal cancer with acquired resistance to first-line cetuximab and irinotecan: a phase 2 single-arm clinical trial. JAMA Oncol. 5, 343–350 (2019).
Long, C. et al. Weighted gene co-expression network analysis identifies the prognosis-related models of left- and right-sided colon cancer. Medicines102, e33390 (2023).
Cao, L. et al. Comparative analyses of the prognosis, tumor immune microenvironment, and drug treatment response between left-sided and right-sided colon cancer by integrating scRNA-seq and bulk RNA-seq data. Aging 15, 7098–7123 (2023).
Valdeolivas, A. et al. Profiling the heterogeneity of colorectal cancer consensus molecular subtypes using spatial transcriptomics. NPJ Precis. Oncol. 8, 10 (2024).
Dang, D. K. & Park, B. H. Circulating tumor DNA: current challenges for clinical utility. J. Clin. Investig. 132, e154941 (2022).
André, T. et al. Pembrolizumab in microsatellite-instability-high advanced colorectal cancer. N. Engl. J. Med. 383, 2207–2218 (2020).
Andre, T. et al. Nivolumab (NIVO) plus ipilimumab (IPI) vs chemotherapy (chemo) as first-line (1L) treatment for microsatellite instability-high/mismatch repair-deficient (MSI-H/dMMR) metastatic colorectal cancer (mCRC): first results of the CheckMate 8HW study. J. Clin. Oncol. 42, LBA768–LBA768 (2024).
Hamid, M. A. et al. Immunotherapy for microsatellite-stable metastatic colorectal cancer: can we close the gap between potential and practice? Curr. Oncol. Rep. https://doi.org/10.1007/s11912-024-01583-w (2024)
Antoniotti, C. et al. Upfront fluorouracil, leucovorin, oxaliplatin, and irinotecan plus bevacizumab with or without atezolizumab for patients with metastatic colorectal cancer: updated and overall survival results of the ATEZOTRIBE study. J. Clin. Oncol. 42, 2637–2644 (2024).
Shen, S., Huo, D., Ma, C., Jiang, S. & Zhang, J. Expanding the colorectal cancer biomarkers based on the human gut phageome. Microbiol. Spectr. 9, e0009021 (2021).
Tokunaga, R. et al. Molecular profiling of appendiceal adenocarcinoma and comparison with right-sided and left-sided colorectal cancer. Clin. Cancer. Res. 25, 3096–3103 (2019).
Acknowledgements
The authors declare that no specific funding was received for this study. The vector graphics in this manuscript were created using Inkscape.
Author information
Authors and Affiliations
Contributions
AH.M., P.L.M., O.S., R.J.M., H.F., and S.A. contributed equally to the majority of the work. They were primarily responsible for the conceptualization and structuring of the review. This included creating and refining the figures, drafting the main text of the manuscript, and conducting multiple rounds of thorough review and revision to ensure the accuracy and clarity of the final manuscript. The remaining authors (G.M., A.A., G.R.A., M.A., N.F.I., O.S., Z.K., G.R.R., K.F, V.S.M. and W.D.), contributed by writing specific sections of the manuscript relevant to their areas of expertize. All authors collectively reviewed the final manuscript, provided feedback on its content, and approved it for submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no personal relationships and no competing financial or academic conflicts of interest related to the submitted project, except for two authors who have declared relationships with pharmaceutical companies. These competing interests, detailed below, did not influence the results, interpretations, or conclusions of this review.FH: Honoraria: Lilly, Pierre Fabre, Amgen, Servier, Daiichi Sankyo, BMS, Merck, Sanofi, Roche, Janssen, PharmaMar, Astellas; Travel support: Servier, BMS, Roche, Merck, PharmaMar, Pfizer, Pierre Fabre, Sanofi, Daiichi Sankyo, Gilead; Stock and other ownership interests: Guardant Health, Natera, Revolution Medicines JR: Other interactions with: Amgen, BMS, MSD, Merck, Eli Lilly, Roche, Servier, Pierre Fabre; Research grants: Roche, Servier.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Abdel Hamid, M., Pammer, L.M., Oberparleiter, S. et al. Multidimensional differences of right- and left-sided colorectal cancer and their impact on targeted therapies. npj Precis. Onc. 9, 116 (2025). https://doi.org/10.1038/s41698-025-00892-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41698-025-00892-y