Abstract
Osimertinib has been one of the most commonly prescribed first-line treatments for EGFR-mutated advanced NSCLC for the last several years for patients with EGFR-mutated non-small cell lung cancer (NSCLC), yet clinical outcomes among racial and ethnic minority patients remain poorly characterized due to underrepresentation in pivotal trials. We conducted a retrospective study of 174 patients with advanced EGFR-mutated NSCLC treated at the Montefiore Einstein Comprehensive Cancer Center (MECCC) from 2015 to 2024 to evaluate real-world survival and treatment adherence. The cohort included 32.2% Hispanic, 28.7% non-Hispanic Black (NHB), 26.4% non-Hispanic White (NHW), and 12.6% Asian patients. In multivariable analyses, NHB patients experienced significantly worse overall survival compared with NHW patients, both in the overall cohort (Hazard ratio (HR) 1.83, 95% confidence interval (CI) 1.02–3.27) and in first-line osimertinib users (HR 3.42, 95% CI 1.48–7.88), despite similar adherence rates. Hispanic and Asian patients also showed trends toward inferior outcomes. These findings highlight survival disparities and underscore the need for inclusive trials and targeted strategies.
Similar content being viewed by others
Introduction
Lung cancer remains the leading cause of cancer-related deaths in the United States1. Approximately 15–20% of patients with NSCLC in the U.S. have tumors harboring actionable EGFR alterations2. Osimertinib, a third-generation EGFR tyrosine kinase inhibitor (TKI), is the most commonly used standard frontline therapy for EGFR-mutated advanced NSCLC and an adjuvant therapy after resection, based on improved survival demonstrated in pivotal trials such as FLAURA3,4 and ADAURA5. However, those trials had limited participation of racial/ethnic minorities (only ~1–5% Black or Hispanic), raising concerns about the generalizability of their findings to underrepresented populations in real-world practice3,4,5. Moreover, recent randomized trials have expanded first-line options, with FLAURA2 demonstrating improved outcomes for osimertinib plus platinum–pemetrexed versus monotherapy, and MARIPOSA showing superiority of amivantamab–lazertinib versus osimertinib; we therefore focused this analysis specifically on osimertinib monotherapy to isolate outcomes for this widely used regimen.
Previous studies have suggested racial disparities in lung cancer outcomes, with Black patients often experiencing worse survival than White patients6,7,8. Additionally, the prevalence of EGFR mutations might differ by ancestry: most analyses report similar or lower EGFR mutation rates in African American patients compared to Whites, and similar or higher rates in Hispanic patients9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24,25. Notably, a prior study at our institution found that Black patients with EGFR-mutant NSCLC had inferior survival on first- or second-generation EGFR TKIs compared to White patients13. However, data on osimertinib access, adherence, and outcomes in racial/ethnic minority populations remain limited. Given that the Montefiore Einstein Comprehensive Cancer Center (MECCC) serves a predominantly underrepresented minority patient population (>30% Black and >35% Hispanic overall), we sought to evaluate treatment adherence and clinical outcomes of osimertinib in this diverse cohort, to determine whether racial/ethnic disparities observed in earlier generation therapies persist with osimertinib.
Results
Patient demographics and socioeconomic factors
A total of 174 EGFR-mutated advanced/metastatic patients with NSCLC treated with osimertinib met the inclusion criteria. Median age was 69 years, and 69.5% were female. The cohort was racially and ethnically diverse: 26.4% were non-Hispanic White (NHW), 28.7% non-Hispanic Black (NHB), 32.2% Hispanic, and 12.6% Asian. Overall, 55.7% were documented to be never smokers with a significantly higher proportion of never-smokers among Asian patients (81.8%) compared to other groups (p < 0.001). Baseline body weight and BMI differed by race: NHB and Hispanic patients had higher median weight/BMI than their NHW and Asian counterparts (e.g., median BMI 25.2 and 24.9 for NHB and Hispanic vs. 22.2 and 22.4 for NHW and Asian, p = 0.011 for BMI difference). There were no significant differences in age or sex distribution across the groups.
Several socioeconomic factors revealed significant differences across racial and ethnic groups. The majority of Hispanic (75.0%) and NHB (54.0%) patients lived in neighborhoods with a median annual household income of less than $50,000 USD, compared to 8.7% of NHW patients (p < 0.001 for income distribution). Insurance coverage also varied: 32% of NHB, 25% of Hispanic, and 31.8% of Asian patients were on Medicaid, versus only 8.7% of NHW patients (p = 0.025). Among 63 patients who obtained osimertinib through our institution’s specialty pharmacy (enabling detailed cost data), the median total cost for osimertinib per patient per month was $16,734 USD. However, due to insurance and patient assistance programs, the mean out-of-pocket cost to patients was approximately $59.30 USD, with a median out-of-pocket cost of $0 USD for these patients. There were no significant differences in the cost of osimertinib or out-of-pocket expenses across racial and ethnic groups.
Clinical and tumor characteristics
Nearly all patients (96.6%) had adenocarcinoma histology. Regarding EGFR genotypes, 110 patients’ tumors (63.2%) had an exon 19 deletion and 45 (25.9%) had an exon 21 L858R point mutation. The remaining 10.9% (19 patients) harbored uncommon EGFR mutations. Baseline disease burden and performance status (PS) were similar among groups: at osimertinib initiation, 74.1% of all patients had Eastern Cooperative Oncology Group (ECOG) PS 0–1, and 25.9% had ECOG PS ≥ 2, with no significant difference in PS distribution by race/ethnicity (p = 0.573). Brain metastases were present in 39.7% of patients overall at the start of osimertinib (not significantly different between groups, p = 0.157).
Treatment adherence to osimertinib was generally high across the cohort. Among the 76 patients with available pharmacy refill data, 81.6% achieved proportion of days covered (PDC) > 80% and were considered adherent. Adherence rates were 83.3% among NHW patients, 74.1% among NHB patients, 82.8% among Hispanic patients, and 100% among Asian patients. Although these differences were not statistically significant (p = 0.412), the approximately 10% lower adherence observed among NHB patients may warrant further investigation. Overall, no definitive racial disparities in medication adherence were identified.
Osimertinib was used as first-line therapy in 111 patients (63.8%), while 63 patients (36.2%) received it as second-line or later. Prior therapies in those who received osimertinib beyond first line included platinum-based chemotherapy in 17.5% and a first- or second-generation EGFR TKI (erlotinib, gefitinib, or afatinib) in 28.2% of patients. The distribution of prior treatments did not differ notably by race/ethnicity (Table 1).
Clinical outcomes
At a median follow-up time of 34.3 months, the median real-world overall survival (rwOS) for the entire cohort was 32.7 months. When stratified by race/ethnicity, median rwOS was shortest in NHB patients at 21.9 months, compared to 40.2 months in NHW, 35.3 months in Hispanic, and 32.7 months in Asian patients (Table 2). Although the log-rank test for overall survival by race/ethnicity did not reach significance (p = 0.27), Cox proportional hazards analysis revealed a significant association between NHB race and mortality. In univariable analysis (Fig. 1A), compared to NHW, NHB patients had significantly worse rwOS (Hazard ratio (HR): 1.76, 95% CI: 1.00–3.08; p = 0.049), while Hispanic and Asian patients did not show a significant difference in rwOS. In the multivariable model adjusting for sex, BMI, ECOG PS, EGFR mutation type, and line of therapy (Fig. 1B), NHB race remained independently associated with worse rwOS (HR 1.83, 95% confidence interval (CI) 1.02–3.27; p = 0.042), while Hispanic and Asian patients remained non-significant. Notably, median household income (as a binary ≥$50,000 vs. < $50,000) was not significantly associated with rwOS (p = 0.378), nor was insurance type (p = 0.391 for Medicaid vs. others), suggesting that while insurance and income were not independently associated with survival in our models, they may act as mediators rather than confounders and could still contribute to the observed disparities. The Kaplan–Meier plots of rwOS for the entire cohort and by racial and ethnic groups are shown in Figs. 2 and 3. Among other covariates, in multivariable analysis, ECOG PS was strongly predictive of rwOS (ECOG ≥ 2 vs. ECOG 0-1: HR: 3.97, 95% CI: 2.4–6.54; p < 0.001). Underweight with low BMI (<18.5) was also associated with significantly worse rwOS (HR 2.30, 95% CI 1.26–4.19; p = 0.007).
A Univariable Cox model results for risk of death among patients with metastatic NSCLC on osimertinib (B) Multivariable Cox model results (adjusted for multiple covariates simultaneously) (C) Univariable Cox model results for risk of death among patients with metastatic NSCLC on first-line osimertinib (D) Multivariable Cox model results (adjusted for multiple covariates simultaneously) in patients receiving first-line osimertinib therapy.
Kaplan-Meier estimates of real-world overall survival for the entire cohort.
A Total cohort. B First-line osimertinib cohort.
The median real-world progression-free survival (rwPFS) for the overall cohort was 16.3 months. There were no statistically significant differences in rwPFS or time to treatment discontinuation (TTD) across racial/ethnic groups. However, NHB patients had numerically shorter, but not statistically significant, rwPFS (NHW: 17.3 months; NHB: 15.6 months; Hispanic: 16.1 months; Asian: 17.1 months) and TTD (NHW: 20.5 months; NHB: 19.7 months; Hispanic: 19.6 months; Asian: 23.5 months). In univariable and multivariable analysis, none of the racial/ethnic variables were significantly associated with PFS (Table 2 and Supplementary Tables 1 and 2). These findings suggest that while minority patients had numerically shorter PFS, the study may have been underpowered to detect modest differences. The Kaplan–Meier curves for rwPFS by group are available in Supplementary Figs. 1 and 2. ECOG PS ≥ 2 (multivariable HR of 2.43, 95% CI 1.64–3.60; p < 0.001) was the only clinical factor significantly associated with poorer rwPFS.
In subgroup analysis of the 111 patients who received osimertinib as first-line therapy, racial disparities in rwOS outcomes persisted: NHB patients (n = 31) had significantly worse rwOS compared to NHW (n = 28) in both univariable (HR: 2.16, 95% CI: 1.00–4.65; p = 0.049; Fig. 1C) and multivariable analyses (HR: 3.42, 95% CI: 1.48–7.88; p = 0.004; Fig. 1D). In multivariable analyses, Hispanic patients also showed a trend toward worse rwOS (HR: 2.19, 95% CI: 0.90–5.35; p = 0.085), while outcomes for Asian patients were more comparable to NHW (HR: 1.60, 95% CI: 0.54–4.77; p = 0.396). Analyses of rwPFS among first-line osimertinib users did not differ significantly in either univariable (Supplementary Table 3) or multivariable analyses (Supplementary Table 4).
Of 128 patients with disease progression on osimertinib, 51 received a subsequent line of therapy (Supplementary Table 5A). Among these 51 patients, the median rwPFS2 was 4.7 months (Supplementary Table 5B). Median rwPFS2 did not differ significantly among racial and ethnic groups (p = 0.483). There was also no significant difference in the proportion of patients receiving next-line therapy (p = 0.416), with 38.9% of NHW, 32.4% of NHB, 50% of Hispanic, and 33.3% of Asian patients receiving next-line treatment.
In the sensitivity analysis using a multivariable model that included all variables except osimertinib adherence, we observed similar results. NHB patients continued to show numerically worse OS (HR 1.74, 95 percent CI 0.92–3.32; p = 0.090). Hispanic (HR 1.26, 95 percent CI 0.62–2.59; p = 0.524) and Asian patients (HR 1.27, 95 percent CI 0.53–3.03; p = 0.597) also demonstrated patterns consistent with the original multivariable model, although these associations were not statistically significant (Supplementary Fig. 3).
Osimertinib-related adverse events (AEs) were generally low-grade, with 58% of patients experiencing rash and 42% reporting diarrhea. There were no significant differences in AE incidence or severity by race (e.g., grade≥3: p = 0.380), suggesting similar tolerability across racial/ethnic groups.
Discussion
Despite osimertinib being one of the standard first-line therapies for EGFR-mutated NSCLC, racial and ethnic minority patients, particularly NHB and Hispanic individuals, remain underrepresented in key clinical trials3,4. In the present study involving a racially and ethnically diverse U.S. cohort, NHB patients had significantly shorter rwOS, including among those receiving first-line osimertinib for advanced disease (21.9 months), compared to NHW (40.2 months) and the benchmark from the FLAURA trial (38.6 months)13. Hispanic and Asian patients did not exhibit significantly shorter rwOS. The rwPFS did not significantly differ by race, which may be attributable to limited statistical power due to sample size constraints. Alternatively, the pattern of similar PFS but divergent OS may indicate that differences in long-term outcomes are potentially influenced by post-progression factors, such as comorbidities or other unmeasured variables. Baseline ECOG PS and stage at initiation did not differ by race, suggesting diagnostic delay may not fully explain the disparity.
Previous studies have not consistently identified such disparities with osimertinib, likely due in part to limited minority representation and/or differing study settings. For instance, Sabari et al.26 found no OS difference between Black and White patients in a large U.S. dataset (adjusted HR ~ 1.08, p = 0.57); however, minority patients comprised a relatively small proportion of the cohort, which may limit power to detect subgroup differences. Similarly, Cardona et al.27 found no significant racial/ethnic differences among Hispanic patients, and Li et al.28 reported no race-associated genomic predictors of therapeutic response. In contrast, our study is based on a population where approximately two-thirds of patients identify as Black or Hispanic (substantially higher than the <15% minority representation in prior studies), potentially offering greater power to detect disparities driven by healthcare inequities. Notably, the rwOS among our NHW patients (~40 months) aligns with FLAURA trial outcomes, whereas the significantly poorer rwOS observed in NHB patients underscores an urgent need for further investigation and targeted interventions.
Our subgroup analyses indicated that socioeconomic factors known to influence cancer outcomes may also affect osimertinib use29. Despite its high cost30,31, few studies have explored the impact of cost, insurance status, or income in NSCLC, though prior research suggests private insurance may improve survival in other cancers32,33. In our cohort, insurance type, household income, and neighborhood socioeconomic status were not significantly associated with rwOS or rwPFS. Broad access to osimertinib, regardless of race, likely minimized financial barriers and supported treatment access and continuation.
We also assessed treatment adherence, a well-known determinant of outcomes in oral cancer therapies34,35,36,37. Prior studies involving earlier-generation EGFR TKIs have linked lower adherence to high costs and socioeconomic disadvantage, disproportionately affecting Black patients38,39,40,41,42, In our cohort, overall adherence to osimertinib was high across all groups (~82% with PDC > 80%). While adherence among NHB patients was modestly lower (74%) compared to other groups (~86%), this difference did not reach statistical significance. Still, the 12% gap may warrant further study to assess its role in outcome disparities.
Several additional factors may contribute to the observed survival disparities beyond access, cost, and adherence. Biological differences, particularly in pharmacogenomics, may play a role. Osimertinib is metabolized by CYP3A4/5, and Black patients are more likely to carry CYP3A5 and other polymorphisms associated with increased drug clearance, potentially leading to subtherapeutic exposure43,44,45,46,47,48,49. Similar findings have been reported with other TKIs, including EGFR TKIs, such as erlotinib, raising concerns about suboptimal dosing in Black patients50,51,52. Moreover, NHB and Hispanic patients in our cohort had higher average body weight/BMI, raising concerns about underdosing with osimertinib’s fixed-dose regimen. Pharmacokinetic modeling suggests both weight and ethnicity may affect drug levels, and CYP3A variants have been linked to blood concentrations and outcomes44.
Tumor genomic differences, such as a higher prevalence of co-mutations like TP53, though not fully captured in our dataset, could further explain differential outcomes. Additional factors, including unmeasured comorbidities, poorer PS, subtle differences in care delivery, or lower rates of second-line therapy, may also contribute. Taken together, these findings suggest that the survival gap likely reflects a multifactorial interplay of pharmacological, biological, and healthcare system influences.
Our study has several limitations. As a retrospective analysis, findings may not be generalizable, and selection bias is possible, particularly given our institution’s high proportion of minority and lower socioeconomic status patients. The modest sample size, especially in subgroups, may limit statistical power and increase variability. Socioeconomic status was estimated using neighborhood income and insurance type, which may not fully capture individual or community-level factors, allowing for residual confounding. Adherence and pharmacy cost data were limited to patients using our specialty pharmacy, leading to high missingness that correlated with race/ethnicity. This limitation introduces potential bias and requires cautious interpretation of adherence-related analyses. Some clinical data, such as ctDNA and co-mutation profiles (including TP53), were incomplete, limiting our ability to assess genomic contributors to disparities. Lastly, although follow-up was mature (median 34 months), long-term outcomes for patients on newer therapies may be underrepresented.
Despite these limitations, our study offers novel insights into real-world outcomes with osimertinib in a racially diverse cohort. To our knowledge, it is the first to quantify a significant survival disadvantage for Black patients with EGFR-mutated NSCLC on this therapy. The findings highlight the urgent need for more inclusive clinical trials and targeted efforts to close outcome gaps. Minority patients must be proactively included in research to ensure equitable benefit from emerging cancer therapies.
In conclusion, our real-world analysis shows that racial disparities seen with older EGFR TKIs persist in the osimertinib era, with Non-Hispanic Black patients experiencing significantly worse survival despite similar access, cost, and adherence. This suggests underlying biological or systemic factors. Addressing these disparities requires inclusive research, personalized dosing strategies, and deeper investigation into pharmacogenomics and other risk modifiers. Future studies should explore combination therapies, such as osimertinib with chemotherapy or novel agents, for high-risk subgroups. We have initiated a prospective real-world study on osimertinib and are participating in the ECOG-ACRIN prE1702 trial to better understand outcomes in diverse populations. These efforts reflect growing recognition that equitable precision oncology depends on diverse trial representation and integration of genomic and pharmacologic insights.
Methods
Patient selection and data collection
We conducted a retrospective review of the MECCC cancer registry, identifying 6,019 patients diagnosed with lung cancer (ICD-10 C34.0–C34.9) between January 1, 2015, and December 31, 2024. From these, we included patients who met the following inclusion criteria: (1) histologically or cytologically confirmed advanced or metastatic unresectable NSCLC; (2) age 18 years or older; (3) presence of an EGFR actionable mutation (confirmed by PCR or next-generation sequencing on tumor tissue or plasma); (4) at least two clinical visits at MECCC; and (5) receipt of osimertinib therapy. Patients receiving osimertinib in combination with chemotherapy were excluded from this analysis. EGFR mutations were categorized as common (Exon 19 deletions, L858R) or other (e.g., Exon 20 insertions, S768I, L861Q, G719X).
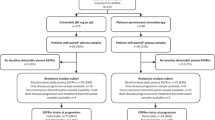
Baseline patient data were extracted from electronic medical records (EMR), including age, gender, smoking status, weight, body mass index (BMI) at osimertinib initiation, insurance status, median annual income (based on the ZIP code from the patient’s EMR address and U.S. Census Bureau data53), and tumor histology as reported in the pathology report. Additional clinical factors at osimertinib initiation included Eastern Cooperative Oncology Group (ECOG) PS, presence of brain metastases, line of therapy (first vs. later line) for osimertinib, EGFR mutation subtype, and any prior systemic treatment before osimertinib initiation. A flowchart summarizing the study cohort is presented in Fig. 4.
Patient selection flowchart: Schematic of cohort selection for patients with advanced NSCLC treated with osimertinib at MECCC.
This study was reviewed and approved by the MECCC institutional review board (IRB Protocol 2022-13806: Assessing clinical outcomes and defining treatment barriers in EGFR-mutated NSCLC patients receiving osimertinib). The current manuscript reports only the retrospective cohort component of this approved protocol. For this retrospective chart review, the requirement for obtaining individual informed consent was formally waived because the study used existing data collected as part of routine clinical care and posed no more than minimal risk to participants.
Pharmacy and adherence data
We collected pharmacy records for osimertinib prescriptions, including fill dates, quantity dispensed, total drug cost, and out-of-pocket cost to patients. Medication adherence was assessed using the PDC method, endorsed by the Pharmacy Quality Alliance as a standard for medication adherence measurement. PDC was calculated as the number of days the patient had osimertinib on hand, divided by the number of days in the observation period (from the first fill to the last fill). We included only patients with at least two fills to accurately calculate PDC. If patients refilled early, resulting in an overlap of medication supply, the excess days were carried over (ensuring PDC did not exceed 100%). Consistent with prior research and the Utilization Review Accreditation Commission guidelines, we defined patients as adherent if PDC > 80%38,54,55,56. We also recorded each patient’s total osimertinib drug cost and their out-of-pocket cost after insurance, per our institution’s specialty pharmacy records.
Clinical outcomes
rwOS was defined as the time from the date of osimertinib initiation, with death events ascertained via our institutional EMR linked to regional hospital data and obituary/nursing facility records to the date of death from any cause or last contact. rwPFS was defined as the time from osimertinib initiation to disease progression (as determined by clinical assessment and radiographic reports in EMR), or death, whichever occurred first. This was calculated in accordance with the methods used in prior retrospective studies31,32. Treatment duration was measured as time to discontinuation (TTD), defined as the number of days between the first and last dates of osimertinib administration. Additionally, data on osimertinib-related AEs were collected from EMR and categorized by system and grade when available (using CTCAE v5.0 terminology when documented).
Statistical analysis
Demographic and clinical characteristics were compared between racial and ethnic groups using the Wilcoxon rank-sum test or Kruskal–Wallis test for continuous variables, and the chi-squared test or Fisher’s exact test for categorical variables. Survival analyses were conducted using Kaplan–Meier curves with log-rank tests for rwOS and rwPFS. Cox proportional hazards models were used to assess differences in rwOS, rwPFS, and TTD. HRs and 95% CIs were calculated. rwOS and rwPFS were evaluated using both univariable and multivariable models, with adjustments for clinically relevant covariates, including gender, race/ethnicity, BMI, ECOG PS, EGFR mutation type, and line of therapy. We also conducted a sensitivity analysis using the full multivariable model to assess the robustness of our findings. Additionally, the real-world progression-free survival for the next line of therapy after osimertinib (rwPFS2), as well as the subsequent treatment type, were determined and analyzed. All statistical analyses were performed using R version 4.4.0, with a significance level set at p < 0.05 using two-sided tests.
Data availability
Due to patient privacy and institutional ethical regulations, the datasets from this study are not publicly available but can be requested from the corresponding author upon a reasonable request. The underlying code for this study is not publicly available but may be made available to qualified researchers on reasonable request from the corresponding author.
Code availability
The underlying code for this study is not publicly available but may be made available to qualified researchers on reasonable request from the corresponding author.
References
Siegel, R. L., Giaquinto, A. N. & Jemal, A. Cancer statistics, 2024. CA Cancer J. Clin. 74, 12–49 (2024).
Siegelin, M. D. & Borczuk, A. C. Epidermal growth factor receptor mutations in lung adenocarcinoma. Lab. Investig. 94, 129–137 (2014).
Soria, J. C. et al. Osimertinib in untreated EGFR-mutated advanced non-small-cell lung cancer. N. Engl. J. Med. 378, 113–125 (2018).
Ramalingam, S. S. et al. Overall survival with osimertinib in untreated, EGFR-mutated advanced NSCLC. N. Engl. J. Med. 382, 41–50 (2020).
Tsuboi, M. et al. Overall survival with osimertinib in resected EGFR-mutated NSCLC. N. Engl. J. Med. 389, 137–147 (2023).
Zeng C. et al. Disparities by race, age, and sex in the improvement of survival for major cancers. JAMA Oncol. 1, 88–96 (2015).
Saeed, A. M. et al. The influence of Hispanic ethnicity on nonsmall cell lung cancer histology and patient survival: an analysis of the survival, epidemiology, and end results database. Cancer 118, 4495–4501 (2012).
Klugman, M., Xue, X. & Hosgood, H. D. 3rd Race/ethnicity and lung cancer survival in the United States: a meta-analysis. Cancer Causes Control 30, 1231–1241 (2019).
Araujo, L. H. et al. Somatic mutation spectrum of non-small-cell lung cancer in African Americans: a pooled analysis. J. Thorac. Oncol. 10, 1430–1436 (2015).
Arrieta, O. et al. Different mutation profiles and clinical characteristics among Hispanic patients with non-small cell lung cancer could explain the “Hispanic paradox. Lung Cancer 90, 161–166 (2015).
Bauml, J. et al. Frequency of EGFR and KRAS mutations in patients with non small cell lung cancer by racial background: do disparities exist? Lung Cancer 81, 347–353 (2013).
Campbell, J. D. et al. Comparison of prevalence and types of mutations in lung cancers among black and white populations. JAMA Oncol. 3, 801–809 (2017).
Cheng, H. et al. Survival disparities in black patients with EGFR-mutated non-small-cell lung cancer. Clin. Lung Cancer 21, 177–185 (2020).
Choudhury, N. J. et al. Fewer actionable mutations but higher tumor mutational burden characterizes NSCLC in black patients at an urban academic medical center. Oncotarget 10, 5817–5823 (2019).
Costa, P. A. et al. Prevalence of targetable mutations in black patients with lung cancer: a systematic review and meta-analysis. JCO Oncol. Pract. 17, e629–e636 (2021).
Dogan, S. et al. Molecular epidemiology of EGFR and KRAS mutations in 3,026 lung adenocarcinomas: higher susceptibility of women to smoking-related KRAS-mutant cancers. Clin. Cancer Res. 18, 6169–6177 (2012).
Errihani, H. et al. Frequency and type of epidermal growth factor receptor mutations in moroccan patients with lung adenocarcinoma. J. Thorac. Oncol. 8, 1212–1214 (2013).
Graham, R. P. et al. Worldwide frequency of commonly detected EGFR mutations. Arch. Pathol. Lab. Med. 142, 163–167 (2018).
Hsu, R., Herrmann, A., Gaur, K., Xia, B. & Nieva, J. J. Evaluating real world mutational differences between Hispanics and Asians in NSCLC at a Large Academic Institution in Los Angeles. Clin. Lung Cancer 23, e443–e452 (2022).
Leduc, N. et al. Lung adenocarcinoma survival in EGFR-mutated African-Caribbean patients: a multicenter study in the French West Indies. Target Oncol. 12, 689–693 (2017).
Legius, B., Van Den Broecke, S., Muylle, I. & Ninane, V. Driver oncogenes in Sub-Saharan African patients with non-small cell lung cancer. Lung Cancer 7, 149–152 (2016).
Leidner, R. S. et al. Genetic abnormalities of the EGFR pathway in African American patients with non-small-cell lung cancer. J. Clin. Oncol. 27, 5620–5626 (2009).
Lopez-Chavez, A. et al. EGFR Mutations in Latinos from the United States and Latin America. J. Glob. Oncol. 2, 259–267 (2016).
Reinersman, J. M. et al. Frequency of EGFR and KRAS mutations in lung adenocarcinomas in African Americans. J. Thorac. Oncol. 6, 28–31 (2011).
Zheng R. et al. Epidemiologic features of NSCLC gene alterations in Hispanic patients from Puerto Rico. Cancers 12, 3492 (2020).
Sabari J. K. et al. Overall survival in EGFR-mutant advanced NSCLC treated with first-line osimertinib: a cohort study integrating clinical and biomarker data in the United States. J. Thorac. Oncol. 20, 1268–1278 (2025).
Cardona, A. F. et al. Mechanisms of resistance to first-line osimertinib in Hispanic patients with EGFR mutant non-small cell lung cancer (FRESTON-CLICaP). Clin. Lung Cancer 23, 522–531 (2022).
Li, P. et al. Race-associated genomic correlates of therapeutic response in African American patients with non-small-cell lung cancer. JCO Precis. Oncol. 7, e2300155 (2023).
Afshar, N. et al. Differences in cancer survival by area-level socio-economic disadvantage: a population-based study using cancer registry data. PloS ONE 15, e0228551 (2020).
Roskoski, R. Jr. Cost in the United States of FDA-approved small molecule protein kinase inhibitors used in the treatment of neoplastic and non-neoplastic diseases. Pharm. Res. 199, 107036 (2024).
Vanderpoel, J. et al. Healthcare resource utilization and costs in patients with EGFR-mutated advanced non-small cell lung cancer receiving first-line treatment in the United States: an insurance claims-based descriptive analysis. PharmacoEconomics Open 7, 617–626 (2023).
Pettit, N. R., Li, X., Stewart, L. & Kline, J. Worsened outcomes of newly diagnosed cancer in patients with recent emergency care visits: a retrospective cohort study of 3699 adults in a safety net health system. Cancer Med. 12, 4832–4841 (2023).
Aghdam, N. et al. Ethnicity and insurance status predict metastatic disease presentation in prostate, breast, and non-small cell lung cancer. Cancer Med. 9, 5362–5380 (2020).
Kavookjian, J. & Wittayanukorn, S. Interventions for adherence with oral chemotherapy in hematological malignancies: a systematic review. Res. Soc. Adm. Pharm. 11, 303–314 (2015).
Marin, D. et al. Adherence is the critical factor for achieving molecular responses in patients with chronic myeloid leukemia who achieve complete cytogenetic responses on imatinib. J. Clin. Oncol. 28, 2381–2388 (2010).
Osterberg, L. & Blaschke, T. Adherence to medication. N. Engl. J. Med. 353, 487–497 (2005).
Richardson, J. L., Shelton, D. R., Krailo, M. & Levine, A. M. The effect of compliance with treatment on survival among patients with hematologic malignancies. J. Clin. Oncol. 8, 356–364 (1990).
Hess, L. M. et al. Factors associated with adherence to and treatment duration of erlotinib among patients with non-small cell lung cancer. J. Manag. Care Spec. Pharm. 23, 643–652 (2017).
Timmers, L. et al. Adherence, exposure and patients’ experiences with the use of erlotinib in non-small cell lung cancer. J. Cancer Res. Clin. Oncol. 141, 1481–1491 (2015).
Gebbia, V. et al. Treatment monitoring program for implementation of adherence to second-line erlotinib for advanced non-small-cell lung cancer. Clin. Lung Cancer 14, 390–398 (2013).
Boucher, J., Lucca, J., Hooper, C., Pedulla, L. & Berry, D. L. A structured nursing intervention to address oral chemotherapy adherence in patients with non-small cell lung cancer. Oncol. Nurs. Forum 42, 383–389 (2015).
Farias, A. J. & Du, X. L. Association between out-of-pocket costs, race/ethnicity, and adjuvant endocrine therapy adherence among medicare patients with breast cancer. J. Clin. Oncol. 35, 86–95 (2017).
McGraw, J. & Waller, D. Cytochrome P450 variations in different ethnic populations. Expert Opin. Drug Metab. Toxicol. 8, 371–382 (2012).
Brown, K. et al. Population pharmacokinetics and exposure-response of osimertinib in patients with non-small cell lung cancer. Br. J. Clin. Pharm. 83, 1216–1226 (2017).
Gao, N. et al. The influence of CYP3A4 genetic polymorphism and proton pump inhibitors on osimertinib metabolism. Front. Pharm. 13, 794931 (2022).
Liang, F., Zhang, Y., Xue, Q. & Yao, N. Exploring inter-ethnic and inter-patient variability and optimal dosing of osimertinib: a physiologically based pharmacokinetic modeling approach. Front. Pharm. 15, 1363259 (2024).
Ishikawa, E. et al. Population pharmacokinetics, pharmacogenomics, and adverse events of osimertinib and its two active metabolites, AZ5104 and AZ7550, in Japanese patients with advanced non-small cell lung cancer: a prospective observational study. Investig. New Drugs 41, 122–133 (2023).
Majam, T. et al. CYP450 and drug efflux transporters polymorphism influence clinical outcomes of Thai osimertinib-treated non-small cell lung cancer patients. Front. Pharm. 14, 1222435 (2023).
Bradford, L. D. CYP2D6 allele frequency in European Caucasians, Asians, Africans and their descendants. Pharmacogenomics 3, 229–243 (2002).
Li, J., Zhao, M., He, P., Hidalgo, M. & Baker, S. D. Differential metabolism of gefitinib and erlotinib by human cytochrome P450 enzymes. Clin. Cancer Res. 13, 3731–3737 (2007).
Rakhit, A. et al. The effects of CYP3A4 inhibition on erlotinib pharmacokinetics: computer-based simulation (SimCYP) predicts in vivo metabolic inhibition. Eur. J. Clin. Pharm. 64, 31–41 (2008).
Phelps, M. A. et al. Erlotinib in African Americans with advanced non-small cell lung cancer: a prospective randomized study with genetic and pharmacokinetic analyses. Clin. Pharm. Ther. 96, 182–191 (2014).
Bureau U. S. C. American Community Survey Data. 2021; https://www.census.gov/programs-surveys/acs/data.html (2022).
Loucks, J. et al. Proportion of days covered as a measure of medication adherence. Am. J. Health Syst. Pharm. 79, 492–496 (2022).
Academia, E. C. et al. Adherence to oral oncolytics filled through an internal health-system specialty pharmacy compared with external specialty pharmacies. J. Manag. Care Spec. Pharm. 27, 1438–1446 (2021).
Commission URA. 2023 Pharmacy benefit management measures at a glance. https://www.urac.org/wp-content/uploads/2022/12/2023_PBM_Measures-at-a-Glance.pdf (2023).
Acknowledgements
The authors acknowledge research support from AstraZeneca (to H. Cheng and J. Yang), the Montefiore Einstein Comprehensive Cancer Center Price Family Foundation Health Equity Pilot Award (to H. Cheng), and the Integrated Imaging Program for Cancer Research (IIPCR)/Evelyn Gruss Lipper Foundation (to H. Cheng, J. Yang, and B. Stiles).
Author information
Authors and Affiliations
Contributions
Conceptualization: M.L., B.H., and H.C. Methodology: M.L., J.S., B.H., and H.C. Software: J.S., S.T., and H.C. Validation: M.L., B.H., and H.C. Formal analysis: J.S. and S.T. Investigation: M.L., J.S., E.M., D.S.A., and H.C. Data curation: M.L., J.S., E.M., and D.S.A. Writing—Original draft: M.L., J.S., and H.C. Writing—Review and editing: M.L., J.S., E.M., D.S.A., C.Z., X.X., J.Y., A.D., R.G., B.S., N.O., B.H., and H.C. Visualization: M.L. and J.S. Supervision: B.H. and H.C. Project administration: H.C. Funding acquisition: J.Y., B.S., and H.C.
Corresponding author
Ethics declarations
Competing interests
H. Cheng and J. Yang received research support from AstraZeneca. The other authors declare no financial or non-financial competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Lee, M., Song, J., Miao, E. et al. Treatment adherence and clinical outcomes of osimertinib in minority patients with advanced EGFR mutated NSCLC. npj Precis. Onc. 10, 90 (2026). https://doi.org/10.1038/s41698-025-01259-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41698-025-01259-z