Abstract
Effect of telemedicine-based interventions have been designed to enhance in-hospital interprofessional collaboration. But their effect on healthcare processes and patient outcomes remain variable. We conducted a systematic review to explore the impact of telemedicine facilitated interprofessional collaboration. We searched six databases and clinical trial registries through April 2, 2025. Twenty-nine studies were included, representing 179,612 patients. Most studies reported improved care processes (e.g., timeliness, treatment appropriateness, diagnostic accuracy). Effects on outcomes like mortality and readmission were mixed. Meta-analysis of randomized trials showed reduced hospital length of stay (mean difference -1.03 days, 95% confidence interval (CI): -1.48 to -0.59) but increased readmissions (risk ratio 1.18, 95% CI: 1.05 to 1.32). These findings suggested that in-hospital telemedicine functions as structural changes that can support improved care processes and, in some cases, better outcomes. Their effectiveness varies based on patients’ conditions, application settings, interprofessional collaboration model and workflows.
Similar content being viewed by others
Introduction
Acute critical illness and complex chronic conditions such as sepsis, acute kidney injury (AKI), and stroke remain significant challenges in patient care within hospital campus1,2,3,4,5. Timely recognition and appropriate response determine outcomes and often require support from other experts6. Notably, for these conditions in general hospital care, early recognition and treatment are often suboptimal, which contributes to a high rate of “failure to rescue”7,8. For instance, a meta-analysis reported a pooled incidence of 21% for in-hospital AKI4, and the missed diagnosis rate was found to be as high as 15.1%6.
Interprofessional collaboration is a potential approach to achieving more accurate diagnoses and optimal, timely interventions9,10,11. Traditional models of interprofessional collaboration—such as rapid response services (RRS) or specialist consultations—depend heavily on bedside staff to recognize deterioration and initiate escalation, which can lead to inconsistent reactions and delayed interventions12,13,14. Barriers such as inefficient information transfer, delays in activating off-site teams, and physical separation between providers are known to hinder effective collaboration12.
Telemedicine has been widely adopted to facilitate remote communication between providers and outpatients, demonstrating potential to improve outcomes15. More recently, studies have explored the use of telemedicine to enhance interprofessional collaboration by enabling communication between off-site specialists and on-site care teams within hospitals16,17,18. Systems that incorporate remote patient monitoring, clinical decision support, and automated activation of interprofessional teams may help close existing gaps in recognition and response12,18. However, evidence on their impact on in-hospital care processes and outcomes remains variable6,19,20.
To better understand the impact of telemedicine on in-hospital interprofessional collaboration, we conducted a systematic review focusing on telemedicine tools that support remote patient monitoring and interprofessional communication. We applied Donabedian’s structure–process–outcome model as a guiding framework to organize and interpret our findings21,22. Anchoring in this framework, the objectives of this review are (1) to systemically summarize the structural features of telemedicine for interventions, including its type of technology used, application settings, human resources, and collaborative workflow; (2) to evaluate their impact on in-hospital care processes; (3) to assess their impact on patient outcomes, aiming to provide evidence-based guidance for clinical application and insights for future research.
Results
Study selection and characteristics
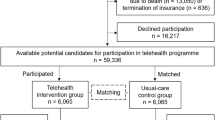
Figure 1 presents the PRISMA flow diagram. From 9800 citations searched via databases and reference lists, a total of 29 studies were included, including nine randomized controlled trials (RCTs) (31%), 19 before-after studies (65.5%), and one retrospective cohort study (3.5%). The included studies were published from 2004 to 2025. A total of 179,612 participants were included (83,032 in the intervention arms; 96,580 in the control arms). The studies’ characteristics are presented in Table 1 and Supplementary Tables 1 and 2.
Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flow chart.
Risk of bias and quality of evidence
As shown by the Newcastle-Ottawa Scale (NOS) assessment (Fig. 2A), the 20 non-randomized studies were of moderate-to-high quality. For the nine RCTs assessed by RoB 2, two studies were judged to have low risk of bias in all domains. Only one study showed a high risk of bias in the performance domain. Six studies had moderate concerns in the domains of random sequence generation, allocation concealment, and blinding (Fig. 2B, C).
A Results of NOS assessment for non-RCT; B each risk of bias item for individual study; C each risk of bias item presented as percentage across all included studies. B, C were created using Review Manager (RevMan) 5.4, developed by the Cochrane Collaboration.
Structural features of tele-interprofessional collaboration
Among the included studies, the largest subgroup focused on patients at risk of physiological deterioration (five studies). Four studies focused on patients with large cerebral vessel occlusion, and another four on AKI. The most frequently involved collaboration teams are infarct teams for cerebrovascular events and rapid response teams (six studies for each). The nephrology team is featured in four studies, ID specialists in three, and pharmacology, anesthesiology, and eICU in two each, with nutrition support and tele-sepsis teams in one study (Fig. 3A, B).
A Proportions of each participant population; inner circle calculated based on patients’ number and outer circle calculated based on the study count; B outer circle shows proportions of collaboration teams and inner circle shows proportions of each model applied, both based on the study count; C three models summarized across included studies. TM telemedicine.
According to how telemedicine techniques function within interprofessional workflows, we proposed three models used in the included studies (Fig. 3C). In Model 1 (applied in 15 studies), telemedicine supports both the afferent and efferent arms of interprofessional collaboration by enabling remote patient monitoring and facilitating communication between bedside staff and off-site specialists. Model 2 (applied in 11 studies) targets urgent situations by incorporating telemedicine for patient monitoring to support off-site, real-time, parallel communication. Once the off-site specialist is automatically notified, they directly contact patients or on-site staff. This model is primarily used to manage large vessel occlusion, STEMI, and physiological deterioration. Model 3 is characterized by telemedicine notifications sent to bedside staff. The ultimate decision to notify an off-site specialist and activate interprofessional collaboration remains at the bedside physician’s discretion. This model was identified in three studies.
Regarding the types of technology leveraged in the telemedicine interventions, the most commonly used systems were communication platforms (reported in 14 studies), followed by EHR-based messaging tools (reported in 9 studies). Other technologies included auto-transmitted emails, text messaging, paging systems, and fax communication. A summary of the specific technologies applied in each study is presented in Table 1.
Impact of tele-interprofessional collaboration on healthcare process
The reported outcomes of included studies and the impact of telemedicine are summarized in Fig. 4.
Red grid with upward arrow indicates telemedicine increases this outcome with statistical significance; green grid with downward arrow indicates telemedicine decreases this outcome with statistical significance; white grid with circle indicates no statistically significant effect was observed; gray grid indicates unreported outcomes.
Timeliness of care
Eleven studies (nparticipants = 4882) investigated the effect of telemedicine on enhancing the timeliness of care for ID, AKI, STEMI, LOV, respiratory failure, and hyponatremia. Seven studies (nparticipants = 812) showed that for patients with suspected LOV or STEMI, implementing Viz.ai, a tele-ECG triage system, or real-time diagnostic support service significantly reduced the door-to-balloon time (86 [75–95]min vs. 125 [90–127]min, P < 0.001), ECG-to-infarct team activation time (7 [4–12] min vs. 25 [11–25] min, P < 0.001), and increased the proportion of door-to-balloon times <90 min (76% vs. 44% P = 0.0001). However, no statistical significance was found in time-to-reperfusion (55 [34–76] min vs. 74 [39–123] min, P = 0.07) when using the real-time remote STEMI diagnostic support service in the MORACS study.
Two studies found that electronic alerts with automated consultations led to a mean reduction of 10 h to specialist consultation (14.5 h vs. 4 h, P < 0.001) and promoted appropriate antimicrobial prescriptions and regimen switches. For AKI patients in one RCT, the automated alerts to bedside staff or pharmacists did not shorten time to renal consultation (1.61 [0.36–4.07] days vs.1.78 [0.74–4.41] days, P = 0.33). Similarly, automated postoperative monitoring by a respiratory therapist (RT) after extubation was not shown to shorten time to reintubation (31 [11.5-59.5] h vs. 28 [16.1–96.8] h, P = 0.56). Likewise, targeted automatic electronic consultations from nephrologists did not alter time to hyponatremia resolution.
Appropriateness of treatment
Among the ten studies (nparticipants = 37,662) that examined treatment appropriateness, eight demonstrated a positive effect of telemedicine, and two demonstrated no or a negative impact. Four studies (nparticipants = 4110) involving ID and sepsis patients found that telemedicine helped optimize antibiotic prescription and administration, as well as the Severe Sepsis and Septic Shock Management Bundle (SEP-1) compliance. Subbe et al. also reported an increased antibiotic administration rate (60.5% vs. 47.4%, P < 0.001) after telemedicine implementation in hospitalized patients at risk of physiological deterioration. One study reported that tele-ICU pharmacists improved the sedative interruption rate for mechanically ventilated ICU patients to facilitate patient assessment. The real-time remote STEMI diagnostic support service eliminated missed therapies for STEMI patients (0 vs. 36%, P < 0.001). A virtual nutrition support team significantly increased prescribing appropriateness for patients receiving parenteral nutrition (97.2% vs. 58.9%, P < 0.001). However, one RCT showed that an intraoperative telemedicine program (the Anesthesiology Control Tower, ACT) only increased the rate of intraoperative glucose measurement (68.7% vs. 63.8% P = 0.02) but had no significant effect on temperature or neuromuscular monitoring, insulin per clinical guidelines, missed antibiotics, or tidal volume. In addition, for extubated patients, automated continuous monitoring and alert to an RT did not change the rate of prophylactic NIV, airway clearance, or lung expansion.
Avoiding a missed diagnosis
Three studies (nparticipants = 4521) highlighted the beneficial impact of telemedicine in facilitating interprofessional collaboration and improving diagnostic accuracy. In the MORACS study, a significant decrease in missed STEMI diagnoses occurred (0 vs. 35%, P < 0.001) via the real-time remote STEMI diagnostic support service. With the assistance of automatic AKI notification and nephrologist consultation, two studies demonstrated a reduction in overlooked AKI (3.3% vs. 12.4% and 5.9% vs. 18.1%, both P < 0.001).
Reducing adverse events
Ten studies (nparticipants = 114,060) investigated adverse event rates in patients with AKI (nstudies = 2, nparticipants = 2679), LVO (nstudies = 3, nparticipants = 469), undergoing anesthesia (nstudies = 2, nparticipants = 98,181), at risk of physiological deterioration (nstudies = 3, nparticipants = 12,731). In physiological deterioration, three studies reported that the automated notification system and activated RRS significantly reduced in-hospital cardiac arrest. For patients with AKI, automated alerts to nephrology teams reduced the risk of long-term renal replacement therapy (OR = 0.04, 95%CI: 0.00–0.32, P = 0.03), yet with no significant impact on the progression of AKI stage (OR = 1.22, 95%CI: 0.3–4.89, P = 0.783). In patients with LVO, no significant impact was shown on the symptomatic or asymptomatic intracerebral hemorrhage. Meanwhile, two large-sample size RCTs showed that ACT did not reduce postoperative complications in patients undergoing anesthesia, including delirium, respiratory failure, AKI, or incident atrial fibrillation.
Medical resource use
One study in ID patients showed a significant increase in specialty consultation rate from 70% to 100% (P < 0.001). For patients with DM, the unsolicited endocrinological e-consultation reduced the need for face-to-face visits (10% vs. 22%, P = 0.012). Three studies explored telemedicine’s impact on RRS utilization, with results varying. Three studies assessed the impact of telemedicine on rapid response system (RRS) utilization, with varying results. Sub-analysis showed that two studies implementing Model 2, which involved automatic RRS activation via telemedicine, reported an increased number of RRS activations. In contrast, one study using Model 3, which relied on bedside staff initiation, showed a decrease in RRS calls (13/1931 vs. 18/1896, P = 0.02).
The ICU admission rate was reported in eight non-RCT studies (nparticipants = 65,515). Among five studies in patients at risk of physiological deterioration, two studies (nparticipants = 40,903) showed a decrease in ICU admission rate after telemedicine implementation (3.0% vs. 3.6%, P < 0.001; adjusted HR = 0.91 (0.84–0.98), respectively). The other three studies (nparticipants = 21,780) did not show a significant impact. One study reported a rate reduction from 38% to 16% (P = 0.03) in patients with Staphylococcus aureus bacteremia after implementing an automatic notification and ID consultation program.
Impact of tele-interprofessional collaboration on patient outcomes
Favorable treatment efficacy
Eight studies (nparticipants = 74,336) assessed the impact of telemedicine on treatment efficacy. Of the four LVO-focused studies, two indicated no significant effect on post-TICI 2B-3 reperfusion rates, whereas Hassan et al. reported a 10% increase (94.1% vs. 84.9%, P = 0.036). No improvements in neurological outcomes were noted in two other studies. In patients who received parenteral nutrition, a virtual nutrition support team increased the rate of acceptable glucose control (83.5% vs. 62.2%, P < 0.001). However, among the 130 DM patients, unsolicited endocrinologist e-consultation did not influence the HbA1c over an 18-month follow-up. Similarly, ACT failed to increase normothermia or reduce hypotension in anesthetized patients, and automated nephrologist e-consultations did not alter hyponatremia resolution rates (42% vs. 29%, P = 0.304).
Mortality
A total of 20 studies reported mortality, including seven RCTs (nparticipants = 101,527) and 13 non-RCTs (nparticipants = 69,728). Among RCTs, only one reported reduced mortality in LVO patients using Viz.ai (13% vs. 31%, P < 0.001). The meta-analysis showed no significant differences (mean RR = 0.90, 95% CI of [0.77, 1.05], P = 0.18, Fig. 5A). Among the non-RCTs, four out of five that included automatic RRS notifications reported reduced mortality in patients at risk of physiological deterioration. Two studies in critical illness also showed lower mortality after telemedicine implementation. No significant effect was observed in AKI, ID, or LVO patients.
A Mortality; B hospital LOS; C hospital readmission. The figure was created using RevMan 5.4.
Hospital and ICU LOS
Hospital LOS was reported in 16 studies, which consisted of five RCTs (nparticipants = 3022) and 11 non-RCTs (nparticipants = 28,894). The meta-analysis involving the five RCTs indicated an average 1.03-day reduction in hospital stay (Mean Difference −1.03, [95% CI: −1.48, −0.59], P < 0.001, Fig. 5B). Five out of 11 non-RCTs showed an intergroup difference in hospital LOS. Two reported a longer LOS in the intervention group, but three reported shorter ones. For ICU LOS in five studies (nparticipants = 5806), only one in critically ill patients showed that off-site eICU service significantly lowered ICU stays (3.63 [3.21–4.04] vs. 4.35 [3.93–4.78], P < 0.05).
Hospital readmission
The meta-analysis of three RCTs (nparticipants = 26,439) showed a slight increase in the risk of hospital readmission in intervention groups (mean RR = 1.18, 95% CI of [1.05, 1.32], P = 0.005, Fig. 5C). Two non-RCTs (nparticipants = 2679) found no significant difference in AKI patients with automated nephrologist collaboration.
Impact of tele-interprofessional collaboration on healthcare cost
Among four studies (nparticipants = 3877), one (in AKI) reported an average per-patient cost reduction of $2011.05, and another (in critically ill patients) showed a $2556 reduction. Two other studies reported no significant change in costs.
Discussion
The present study demonstrated that implementing telemedicine technology to facilitate in-hospital interprofessional collaboration was associated with improved healthcare timeliness, diagnosis accuracy, treatment regimen appropriateness, and reduced ICU admission rates. The meta-analysis of RCTs found a modest decrease in hospital length of stay but an ~18% relative rise in the rate of hospital readmissions compared with usual care. However, telemedicine’s effect on mortality, adverse events, and healthcare costs remains uncertain.
This review provides a structured synthesis of how in-hospital telemedicine systems, when viewed as interventions, influence care processes and patient outcomes. Despite varied patient populations and clinical conditions, most studies reported improved timeliness and appropriateness of treatment via off-site specialists collaborating through telemedicine. Notably, the Donabedian framework clarifies that structure alone is not sufficient; benefits depend on whether the intervention meaningfully improves processes. Our findings support this model. In cases where the telemedicine intervention altered the care process—such as automatic alerting to a stroke team or direct escalation to a nephrologist—timeliness, appropriateness, and diagnostic accuracy improved6,14,23. These process improvements, in turn, were associated with more favorable outcomes, including shorter hospital stays and fewer cardiac arrests.
The heterogeneous results across studies highlight key factors that influence telemedicine’s real-world efficacy. First, clinical context and severity of illness are critical. For patients with hyponatremia or type 2 DM, specialist e-consultation did not change the patient outcomes24,25, likely because the primary team already provided high-level care or because multiple non-treatment factors affect these conditions. Evans’ study, for example, showed that the highest-severity patients derived the most benefit from telemedicine-facilitated RRS26. Second, team composition matters. For AKI management, the pharmacist was the alert recipient in one study19, whereas nephrologists were notified in three others6,27,28. Studies employing nephrologist alerts yielded more positive outcomes, consistent with previous research emphasizing nephrologists’ role in early AKI management29,30. Conversely, two large RCTs31,32 found no significant advantage of an off-site anesthesiology service for surgical patients, perhaps because parallel teams with similar expertise did not add clear benefits. Third, interventions disrupted existing workflows in ways that proved beneficial. For example, the use of automated STEMI diagnosis systems in rural emergency departments bypassed conventional referral chains and led to more direct activation of tertiary cardiology teams—reducing door-to-balloon times and virtually eliminating missed diagnoses14. Finally, the collaborative model (as we summarized in three types) also impacts outcomes. Model 2 increased resource usage, such as more RRS calls, while Model 3 reduced it12,33, suggesting that requiring bedside staff to trigger alerts may avert alarm fatigue but potentially delay needed collaboration.
The study has several limitations. First, the studies included were conducted in various patient populations and clinical settings, which also resulted in highly heterogeneous telemedicine techniques. The association between telemedicine and healthcare processes or patient outcomes should be viewed separately. Second, only one-third of the included studies are RCTs, limiting the feasibility of meta-analysis for the interested outcomes. Third, the reliability of the quantitative analysis results in this study requires further validation, as the quality and statistical power of studies reporting these outcomes were low. Fourth, inconsistent definitions of research outcomes (e.g., timeliness, appropriateness) and low statistical power in some studies limit the reliability of quantitative estimates. Finally, our narrative summaries may introduce subjective bias.
Anchoring in a structure–process–outcome framework, this systematic review provides an overview of telemedicine as a potential facilitator of interprofessional collaboration, focusing on its impact on in-hospital care processes and patient outcomes. Despite demonstrated effectiveness, caution is warranted when applying or extrapolating results, given the variability in patient populations, teams, workflows, collaboration models, and outcome definitions. Nonetheless, our findings provide valuable insights and practical guidance for refining telemedicine interventions and highlight the need for further high-quality trials to assess the impact of telemedicine across diverse clinical contexts.
Methods
This systematic review was performed according to a prespecified protocol, which has been registered in the International Prospective Register of Systematic Reviews (PROSPERO, ID: CRD420251023429). This systematic review and meta-analysis were reported in accordance with the PRISMA (Supplementary Table 5)34.
Eligible criteria
Randomized or non-randomized clinical trials and cohort studies comparing patient care by telemedicine, which is capable of monitoring patients in the hospital and facilitating interprofessional collaborations with usual care, were included.
In this study, interprofessional collaboration was defined as a healthcare approach involving professionals from different disciplines (e.g., infectious disease specialists) or specialized medical teams (e.g., rapid response teams) who provide support to improve healthcare processes and patient outcomes. These interventions are delivered in coordination with primary care or bedside clinical staff. Usual care was defined as patients being cared for by bedside staff, and interprofessional intervention was raised by bedside staff if needed. At least the measurement of healthcare processes or patient outcomes had to be reported in eligible studies.
Eligible participants and telemedicine settings had to be on a hospital campus. Based on the fundamental function that telemedicine enables information exchange, telemedicine tools in this study were required to (1) be used in hospitalized inpatients (e.g., general wards or ICU), and outpatients receiving healthcare services in hospital campus, such as in the primary care and ED; (2) at least one off-site interprofessional specialist or medical team was connected via communication tool; (3) remotely transmit patient’s information, generate alarms or consultation requests, or transmit recommendations from off-site specialist, facilitating interprofessional collaborative management.
We applied the following exclusion criteria in this research: (1) studies involving patients outside the hospital setting or during transportation; (2) non-clinical studies, non-comparative studies, studies focused on the development of telemedicine technologies, survey or interview studies, non-original articles, studies without available full text, or retracted publications; (3) telemedicine interventions used outside the hospital campus, those not intended to facilitate interprofessional collaboration, or those designed solely for patient self-monitoring without interprofessional involvement; (4) studies that did not use usual care as a comparator or did not report outcomes related to healthcare processes or patient outcomes.
Search strategy
A medical librarian (D.G.) searched for the concepts of telemedicine, inpatients, and interprofessional collaboration. Search strategies used a combination of keywords and standardized index terms. Searches were initially run on October 7, 2024, and updated on April 2, 2025, in ClinicalTrials.gov, CINAHL, Cochrane Central, Embase, Medline, Web of Science, and WHO’s ICTRP registry. The search was limited to English-language publications with no date or publication status restrictions. Deduplication was done in Covidence. Reference lists of included articles and relevant systematic reviews were also checked. A detailed search strategy is provided in the Supplementary.
Study selection
After deduplication was performed automatically in Covidence systematic review software (Veritas Health Innovation), a minimum of at least 2 reviewers (X.H., A.L., or S.G.) independently screened all identified publications for full-text assessment based on the reference title and abstract with Covidence. Then, the same or overlapping pairs of reviewers (X.H., A.L., or S.G.) independently assessed full texts for final inclusion, resolving disagreements by consensus or, if needed, by a third reviewer (V.H.).
Data extraction
Two reviewers (X.H., A.L., or S.G.) used Covidence for data extraction independently. Conflicts were resolved by discussion or referral to a third reviewer (V.H.). Extracted data included study design, setting, participants, telemedicine intervention details, comparison arms, and outcome measures.
Risk of bias assessment
For the randomized studies, the Cochrane Risk of Bias Assessment Tool 2 (RoB 2) was used to assess the quality of each included study by classifying bias in domains of selection, performance, detection, attrition, and reporting35. The NOS was used to assess the quality of non-randomized or cohort studies in terms of exposed and non-exposed cohort selection, comparability between cohorts, and outcome determination36. Two reviewers (X.H. and A.L.) independently performed these assessments; discrepancies were resolved by consensus or by consulting V.H. if required.
Outcomes
Three main categories of outcomes were collected and summarized: (1) In-hospital healthcare services or processes (timeliness/appropriateness of treatment, diagnosis, adverse events, resource utilization, ICU admission). (2) Patient outcomes (treatment efficacy, mortality, length of stay in hospital or ICU, readmission). (3) Healthcare cost measures. The definitions of outcomes and components in the composite outcomes are provided in Supplementary Table 4.
Data synthesis and analysis
The mean and standard deviation (SD) were used for continuous outcomes. For median and inter-quartile range, reported methods were used to convert them to mean and SD37,38. For categorical outcomes, the number of events, total number of patients in each cohort, relative risk (RR), odds ratio (OR), hazard ratio (HR), and standard error between groups were collected. If three or more randomized studies were available for a given outcome, we performed a meta-analysis to calculate the pooled RR for binary outcomes or mean difference for continuous outcomes, with 95% confidence interval (CI), to estimate the effect size. Heterogeneity was assessed using the I2 statistic; if I2 > 50%, we applied a random-effects model; otherwise, a fixed-effect model. We investigated sources of heterogeneity through sub-analyses by clinical conditions. All pooled analyses were done in RevMan 5.4. Because the count of RCTs in the meta-analysis is small, we did not analyze the reporting biases or report the certainty of evidence39. Due to the considerable heterogeneity among non-randomized studies, we only conducted narrative summaries for them.
Ethical approval
The study protocol was approved by the Institutional Review Board of Mayo Clinic (IRB#24-012843). Given the retrospective design and minimal risk, the requirement for informed consent was waived, as only existing, deidentified medical data were used.
Data availability
All data generated or analyzed during this study are included in this published article. The data used and analyzed during the current study are available from the corresponding author upon reasonable request.
References
Martin-Loeches, I., Singer, M. & Leone, M. Sepsis: key insights, future directions, and immediate goals. A review and expert opinion. Intensive Care Med. 50, 2043–2049 (2024).
Kellum, J. A. et al. Acute kidney injury. Nat. Rev. Dis. Prim. 7, 52 (2021).
Fleischmann-Struzek, C. et al. Incidence and mortality of hospital- and ICU-treated sepsis: results from an updated and expanded systematic review and meta-analysis. Intensive Care Med. 46, 1552–1562 (2020).
Mehta, R. L. et al. International Society of Nephrology’s 0by25 initiative for acute kidney injury (zero preventable deaths by 2025): a human rights case for nephrology. Lancet 385, 2616–2643 (2015).
Wu, C. et al. Predicting in-hospital outcomes of patients with acute kidney injury. Nat. Commun. 14, 3739 (2023).
Park, S. et al. Impact of electronic acute kidney injury (AKI) alerts with automated nephrologist consultation on detection and severity of AKI: a quality improvement study. Am. J. Kidney Dis. 71, 9–19 (2018).
Silber, J. H., Williams, S. V., Krakauer, H. & Schwartz, J. S. Hospital and patient characteristics associated with death after surgery. A study of adverse occurrence and failure to rescue. Med. Care 30, 615–629 (1992).
Taylor, S. P. & Kowalkowski, M. Failure to rescue as a quality measure in sepsis. JAMA 332, 11–12 (2024).
Reeves, S., Pelone, F., Harrison, R., Goldman, J. & Zwarenstein, M. Interprofessional collaboration to improve professional practice and healthcare outcomes. Cochrane Database Syst. Rev. 6, CD000072 (2017).
Callahan, C. M. et al. Effectiveness of collaborative care for older adults with Alzheimer disease in primary care: a randomized controlled trial. JAMA 295, 2148–2157 (2006).
Bloem, B. R. et al. Integrated and patient-centred management of Parkinson’s disease: a network model for reshaping chronic neurological care. Lancet Neurol. 19, 623–634 (2020).
Heller, A. R. et al. Detection of deteriorating patients on surgical wards outside the ICU by an automated MEWS-based early warning system with paging functionality. Ann. Surg. 271, 100–105 (2020).
Boniatti, M. M. et al. Delayed medical emergency team calls and associated outcomes. Crit. Care Med. 42, 26–30 (2014).
Dee, F. et al. Management of acute coronary syndromes in patients in rural Australia: the MORACS randomized clinical trial. JAMA Cardiol. 7, 690–698 (2022).
Kuan, P. X. et al. Efficacy of telemedicine for the management of cardiovascular disease: a systematic review and meta-analysis. Lancet Digit. Health 4, e676–e691 (2022).
Weis, S. et al. Effect of automated telephone infectious disease consultations to nonacademic hospitals on 30-day mortality among patients with Staphylococcus aureus Bacteremia: the SUPPORT cluster randomized clinical trial. JAMA Netw. Open 5, e2218515 (2022).
Gaieski, D. F. et al. Can an end-to-end telesepsis solution improve the severe sepsis and septic shock management bundle-1 metrics for sepsis patients admitted from the emergency department to the hospital?. Crit. Care Explor. 4, e0767 (2022).
Martinez-Gutierrez, J. C. et al. Automated large vessel occlusion detection software and thrombectomy treatment times: a cluster randomized clinical trial. JAMA Neurol. 80, 1182–1190 (2023).
Wilson, F. P. et al. Automated, electronic alerts for acute kidney injury: a single-blind, parallel-group, randomised controlled trial. Lancet 385, 1966–1974 (2015).
Soares, D. M., Pessanha, J. F., Sharma, A., Brocca, A. & Ronco, C. Delayed nephrology consultation and high mortality on acute kidney injury: a meta-analysis. Blood Purif. 43, 57–67 (2017).
Donabedian, A. Evaluating the quality of medical care. Milbank Meml. Fund. Q. 44, 166–206 (1966).
Tossaint-Schoenmakers, R., Versluis, A., Chavannes, N., Talboom-Kamp, E. & Kasteleyn, M. The challenge of integrating ehealth into health care: systematic literature review of the Donabedian model of structure, process, and outcome. J. Med. Internet Res. 23, e27180 (2021).
Hassan, A. E., Ringheanu, V. M., Preston, L. & Tekle, W. G. Artificial intelligence–parallel stroke workflow tool improves reperfusion rates and door-in to puncture interval. STROKE Vasc. Interv. Neurol. 2, e000224 (2022).
Oseran, A. S. et al. HbA1c-triggered endocrinology electronic consultation for type 2 diabetes management. J. Gen. Intern Med. 37, 1081–1087 (2022).
Judson, T. J. et al. Addressing inpatient hyponatremia through targeted automatic E-consults: a pilot randomized trial. J. Gen. Intern. Med. 40, 1576–1583 (2025).
Evans, R. S. et al. Automated detection of physiologic deterioration in hospitalized patients. J. Am. Med. Inf. Assoc. 22, 350–360 (2015).
Connell, A. et al. Implementation of a digitally enabled care pathway (Part 1): impact on clinical outcomes and associated health care costs. J. Med. Internet Res. 21, e13147 (2019).
Connell, A. et al. Evaluation of a digitally-enabled care pathway for acute kidney injury management in hospital emergency admissions. npj Digit. Med. 2, 67 (2019).
Yang, L. et al. Acute kidney injury in China: a cross-sectional survey. Lancet 386, 1465–1471 (2015).
Ponce, D., Zorzenon Cde, P., dos Santos, N. Y. & Balbi, A. L. Early nephrology consultation can have an impact on outcome of acute kidney injury patients. Nephrol. Dial. Transpl. 26, 3202–3206 (2011).
King, C. R. et al. An intraoperative telemedicine program to improve perioperative quality measures: the ACTFAST-3 randomized clinical trial. JAMA Netw. Open 6, e2332517 (2023).
King, C. R. et al. Effect of telemedicine support for intraoperative anaesthesia care on postoperative outcomes: the TECTONICS randomised clinical trial. Br. J. Anaesth. 134, 671–680 (2025).
Subbe, C. P., Duller, B. & Bellomo, R. Effect of an automated notification system for deteriorating ward patients on clinical outcomes. Crit. Care 21, 52 (2017).
Page, M. J. et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 372, n71 (2021).
Sterne, J. A. C. et al. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ 366, l4898 (2019).
Zhang, Y. et al. The ROBINS-I and the NOS had similar reliability but differed in applicability: a random sampling observational studies of systematic reviews/meta-analysis. J. Evid. Based Med. 14, 112–122 (2021).
Luo, D., Wan, X., Liu, J. & Tong, T. Optimally estimating the sample mean from the sample size, median, mid-range, and/or mid-quartile range. Stat. Methods Med. Res. 27, 1785–1805 (2018).
Wan, X., Wang, W., Liu, J. & Tong, T. Estimating the sample mean and standard deviation from the sample size, median, range and/or interquartile range. BMC Med. Res. Methodol. 14, 135 (2014).
Egger, M., Davey Smith, G., Schneider, M. & Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 315, 629–634 (1997).
Forni, A. et al. Evaluation of the impact of a tele-ICU pharmacist on the management of sedation in critically ill mechanically ventilated patients. Ann. Pharmacother. 44, 432–438 (2010).
Umscheid, C. A. et al. Development, implementation, and impact of an automated early warning and response system for sepsis. J. Hosp. Med. 10, 26–31 (2015).
Kim, M. et al. Electronic alerts with automated consultations promote appropriate antimicrobial prescriptions. PLoS ONE 11, e0160551 (2016).
Djelic, L. et al. Automatic notification and infectious diseases consultation for patients with Staphylococcus aureus bacteremia. Diagn. Microbiol. Infect. Dis. 91, 282–283 (2018).
Kadar, R. B., Amici, D. R., Hesse, K., Bonder, A. & Ries, M. Impact of telemonitoring of critically ill emergency department patients awaiting ICU transfer. Crit. Care Med. 47, 1201–1207 (2019).
Meyer, M., Hartwell, J., Beatty, A. & Cattell, T. Creation of a virtual nutrition support team to improve quality of care for patients receiving parenteral nutrition in a multisite healthcare system. Nutr. Clin. Pract. 34, 881–886 (2019).
Nanchal, R. & Truwit, J. D. Automated identification of adults at risk for in-hospital clinical deterioration. N. Engl. J. Med. 384, 485–486 (2021).
Hassan, A. E. et al. Early experience utilizing artificial intelligence shows significant reduction in transfer times and length of stay in a hub and spoke model. Inter. Neuroradiol. 26, 615–622 (2020).
Morey, J. R. et al. Real-world experience with artificial intelligence-based triage in transferred large vessel occlusion stroke patients. Cerebrovasc. Dis. 50, 450–455 (2021).
Kaur, R. et al. Identification and prevention of extubation failure by using an automated continuous monitoring alert versus standard care. Respir. Care 67, 1282–1290 (2022).
Balshi, A. N. et al. Tele-rapid response team (Tele-RRT): the effect of implementing patient safety network system on outcomes of medical patients-A before and after cohort study. PLoS ONE 17, e0277992 (2022).
Acknowledgements
This work was supported by the Mayo Clinic Critical Care Research Subcommittee and the Critical Care Independent Multidisciplinary Program (IMP).
Author information
Authors and Affiliations
Contributions
V.H. and X.H. contributed to the conceptualization and methodology. D.G., V.H., and X.H. contributed to the citations search. X.H., A.L., and S.G. contributed to the study’s screening, data extraction, and data analysis. X.H. and A.L. drafted the manuscript. V.H. and B.P. edited and revised the manuscript. V.H. supervised the study. All authors accept responsibility for the decision to submit for publication.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, and provide a link to the Creative Commons license. You do not have permission under this license to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Hao, X., Ligocki, A., Gupta, S. et al. Control Tower in the hospital: a structure–process–outcome systemic review of telemedicine systems for interprofessional collaboration. npj Digit. Med. 8, 669 (2025). https://doi.org/10.1038/s41746-025-02043-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41746-025-02043-w
This article is cited by
-
Digital health interventions for perioperative patient-reported outcomes: a network meta-analysis
npj Digital Medicine (2026)