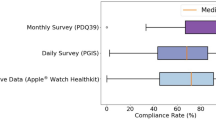
Abstract
Remote, internet-based deep brain stimulation programming for Parkinson’s disease accelerates clinical benefits postoperatively by improving access to therapy adjustments compared to in-clinic optimization. After completion of the initial digital programming phase, we show that clinical outcomes, quality of life, and safety remain sustained over at least twelve months under routine care conditions. Embedding a randomized trial within a larger cohort study enables long-term, real-world evaluation, offering a scalable and pragmatic model for assessing complex digital interventions in routine clinical care. (NCT05269862 registered on 2022-03-08 and NCT04071847 registered on 2019-08-28).
Similar content being viewed by others
Introduction
Digital health interventions hold considerable promise for improving healthcare delivery by expanding access, enhancing personalization, and reducing resource demands1. Technologies such as mobile applications, remote monitoring platforms, and algorithm-guided tools are increasingly integrated into clinical care, yet their evaluation presents unique methodological challenges2,3. Unlike conventional interventions, digital therapeutics are often deployed in real-world settings where variability in user engagement, delivery contexts, and measurement fidelity can impact outcomes4,5. These complexities make it difficult for traditional randomized trial designs to fully capture the long-term effectiveness, sustainability, and generalizability of digital approaches5,6.
This challenge is particularly acute in device-assisted therapies, where access to specialist-guided care is essential7. Deep brain stimulation (DBS) is an established therapy for managing motor symptoms in Parkinson’s disease, but optimal outcomes depend on individualized programming and continued follow-up, typically conducted in specialized centers8. For many patients, these services are difficult to access due to travel burden, cost, or geographic limitations9,10. Digital models of care, such as remote DBS programming and virtual consultations, offer a promising solution11,12,13,14,15. However, little is known about their long-term clinical effectiveness and safety.
To address this gap, we implemented a multicenter, prospective hybrid study design by embedding a three-month randomized controlled trial (RCT) within a five-year observational cohort. The previously reported RCT compared remote, internet-based DBS programming via a virtual clinic platform with standard in-clinic follow-up, showing that virtual programming accelerated clinical improvement without compromising safety or patient satisfaction in the first three months after therapy initiation16. To evaluate the durability and generalizability of these effects, the embedded study design enabled ongoing follow-up of both groups under routine care conditions (Figs. 1 and 2). After the initial 3-month RCT phase, all participants had access to remote programming, with the choice between remote or in-clinic visits left to patients and/or their physicians, allowing for a real-world assessment of long-term outcomes (Table 1).
Data collection was decentralized across both study arms. Initial programing was conducted in-clinic. Titration visits followed either in-clinic or remotely via virtual clinic (RIBA arm). Long-term follow-up occurred at 6 months ±60 days after initial programming, and 1-year ± 60 days after initial programming.
Study design overview16: Schematic representation of the hybrid ROAM-DBS/ADROIT study design. Following baseline enrollment, participants underwent DBS implantation, initial programming, and eligibility screening. After providing informed consent, patients were randomized into either the Virtual Clinic Arm (remote and/or in-clinic titration visits) or the In-Clinic Only Arm (titration visits conducted exclusively in clinic) for a 3-month randomized controlled trial (RCT) period. All participants were then followed in a long-term observational cohort (ADROIT) for five years with in-clinic follow-up visits. Here we report the findings at 6 and 12 months (±60 days) after the initial programming visit.
This study was not designed to demonstrate replacement of in-clinic care by remote programming. After the RCT, remote programming became part of routine care, and withholding this approved option would not have been ethically justifiable. Patients with advanced Parkinson’s disease also require multidisciplinary management, including pharmacological adjustments and treatment of non-motor symptoms, and at most sites they attend routine in-clinic visits every six to twelve months independent of DBS therapy. Remote programming was therefore implemented as a complementary option rather than a substitute for in-clinic care.
Results
Here, we report six- and twelve-month findings demonstrating sustained clinical benefits and similar improvements in outcomes, quality of life, and safety across both groups, supporting the ecological validity and scalability of this evaluation framework. Across all endpoints, outcomes remained consistently favorable and closely aligned supporting the clinical long-term equivalence of the remote and in-person care models used during the initial 3-month period. The observational phase was not intended to preserve strict group separation, since all patients had access to both modalities after three months. The purpose was to test whether the accelerated improvement observed during the titration phase with remote programming would persist once universal access was provided. The results confirm this, as outcomes converged and all patients followed a favorable trajectory at six and twelve months.
Specifically, patient- and clinician-reported global improvement (PGI/CGI) remained favorable over time. Mean PGI change scores (Fig. 3a, Supplementary Table 1) were 2.3 ± 1.4 (in-clinic) and 2.8 ± 1.3 (virtual clinic), while CGI change scores (Fig. 3b, Supplementary Table 2) were 1.8 ± 1.1 and 2.2 ± 0.9, respectively, both consistent with sustained perceptions of improvement. Symptom severity improved comparably, with PGI severity scores (Fig. 3c, Supplementary Table 3) decreasing from 5.1 to 3.5 ± 1.6 in the in-clinic group and to 3.9 ± 1.3 in the virtual clinic group. CGI severity scores (Supplementary Table 4) mirrored these results, decreasing from 5.3 ± 0.9 to 3.4 ± 1.3 and from 5.5 ± 0.8 to 3.5 ± 1.2, respectively. The generalized mixed-effects model (Supplementary Tables 9–12) indicated significant longitudinal improvements in PGI-C and CGI-C (p-values for PGI-C and CGI-C were 0.04 and 0.0071, respectively), with no significant between-group differences (group effect p-values for PGI-C and CGI-C were 0.9839 and 0.5890, respectively). Quality of life, as measured by the PDQ-39 Summary Index (Fig. 4a, Supplementary Table 5), also improved in both groups (–4.3 ± 12.5 and –5.9 ± 13.1). The longitudinal analysis using a linear mixed-effects model (Supplementary Tables 13–14) revealed a significant improvement in PDQ-39 Summary Index scores from baseline (p = 0.0473), with no evidence of a group-by-time interaction (p = 0.9306). Motor function, assessed via MDS-UPDRS Part III (Supplementary Table 7), further improved in the MedON/StimON condition at twelve months, with similar scores between groups (15.3 ± 14.3 vs. 15.5 ± 9.4.5). Levodopa equivalent dose (LED) showed a trend toward reduction in the virtual clinic group (–64.8 ± 265.8 mg) and a slight increase in the in-clinic group (+99.1 ± 315.8 mg), though with high variability and overlapping confidence intervals (Fig. 4b, Supplementary Table 6).
a Patient Global Impression (PGI) of Change: Mean PGI-C scores ± standard deviation are shown for patients at 3 months, 6 months, and 1 year after initial programming, comparing those who received in-clinic care to those who received remote, internet-based care via the virtual clinic platform. PGI-C is a 7-point patient-reported scale assessing overall improvement (1 = very much improved to 7 = very much worse). The number of patients (n) included at each time point is indicated in parentheses. Bracketed values represent 95% confidence intervals. Between-group differences were small and not statistically significant at any time point. 1By normal approximation. Additional information is included in Supplementary Table 1. b Clinician Global Impression (CGI) Change: Mean CGI-C scores at 3 months, 6 months, and 1 year as rated by clinicians for participants in the in-clinic and virtual clinic arms. The CGI-C is a 7-point scale evaluating overall clinical improvement (1 = very much improved to 7 = very much worse). Both care models demonstrated sustained clinician-rated improvement over 12 months. Differences between groups were small and not statistically significant at any timepoint. Data are shown as mean ± SD with corresponding 95% confidence intervals. Additional information is included in Supplementary Table 2. c Patient Global Impression (PGI) Severity: Mean PGI-S scores at baseline, 3 months, 6 months, and 1 year for participants in the in-clinic and virtual clinic arms, along with changes from baseline. Scores reflect patient-reported symptom severity on a 7-point scale (1 = not present to 7 = extremely severe). Both groups showed comparable improvements over time, with no statistically significant differences between groups across all timepoints. Data are presented as mean ± SD with corresponding 95% confidence intervals. Additional information is included in Supplementary Table 3. d Clinician Global Impression (CGI) Severity: Mean CGI-S scores assessed by clinicians at baseline, 3 months, 6 months, and 1 year for participants in the in-clinic and virtual clinic groups. The CGI-S is a 7-point scale evaluating overall symptom severity (1 = not present to 7 = extremely severe). Both groups showed sustained improvement from baseline, with similar reductions in perceived symptom severity over time. Differences between arms remained small and statistically non-significant across all timepoints. Data are presented as mean ± SD, with associated 95% confidence intervals. Additional information is included in Supplementary Table 4.
a Parkinson’s Disease Questionnaire (PDQ-39) Summary Index (SI): Mean PDQ-39 Summary Index scores at baseline, and at 1, 2, 3, 6, and 12 months after treatment initiation in the in-clinic and virtual clinic groups. The PDQ-39 is a validated, patient-reported questionnaire assessing Parkinson’s disease-specific health status across eight domains, with higher scores indicating greater impairment and lower scores reflecting better quality of life. Both groups showed improvement from baseline over time. The virtual clinic group had slightly higher baseline impairment but demonstrated comparable or slightly greater reductions at follow-ups. Differences between groups were small and not statistically significant. Data are shown as mean ± SD, with 95% confidence intervals. Additional information is included in Supplementary Table 5. b Levodopa Equivalent Dose (LED): Mean LED values at baseline, 3, 6, and 12 months for the in-clinic and virtual clinic groups. LED provides a standardized method to quantify total dopaminergic medication load in patients with Parkinson’s disease. Both groups showed stable LED levels over the 12-month follow-up period, with modest increases observed at 12 months. Differences between groups were small and not statistically significant at most timepoints, although a higher mean LED was noted in the virtual clinic group throughout. Values are presented as mean ± SD, with 95% confidence intervals. 1The difference (mean comparison) is calculated by: (the average of virtual clinic – the average of in-clinic). Additional information is included in Supplementary Table 6.
Safety profiles were favorable and comparable across arms. No deaths were reported during the twelve-month period. Eight serious adverse device effects (SADEs) were reported in six patients. Three SADEs were related to the DBS procedure (all in the remote arm) and five to the impulse generator (four in the in-clinic arm and one in the remote arm). Two of the latter SADEs were related to DBS stimulation, both in the in-clinic group. One non-serious adverse device effect (ADE) in the in-clinic group was stimulation-related and resolved with re-programming. Healthcare utilization metrics (Supplementary Table 8), including emergency room visits (n = 13 per group) and hospitalization rates (n = 4 in-clinic, n = 3 virtual clinic), were similar across arms, indicating that remote care did not increase the burden on acute services. Protocol deviations (n = 57) were mostly related to scheduling or missing assessments and were evenly distributed without impact on outcomes or safety. None were related to the virtual clinic platform, further supporting its feasibility for routine use.
Outcome measures
To assess the clinical impact and sustainability of remote DBS programming, a range of validated outcome measures were administered at multiple time points throughout the study. Assessments occurred at 3, 6, and 12 months after treatment initiation and covered patient-reported outcomes, clinician-rated evaluations, motor symptom severity, medication intake, quality of life, safety, and healthcare utilization. The 3-month timepoint corresponded to the end of the RCT phase, while the 6- and 12-month assessments were conducted during the subsequent open-label observational period.
Patient and clinician perspectives on treatment benefit and symptom burden were captured using the Patient Global Impression of Change (PGI-C) and Clinician Global Impression of Change (CGI-C), both 7-point scales rating perceived improvement from “very much improved” (1) to “very much worse” (7). Symptom severity was similarly rated using the Patient Global Impression of Severity (PGI-S) and Clinician Global Impression of Severity (CGI-S), ranging from “not present” (1) to “extremely severe” (7).
Levodopa Equivalent Dose (LED) was calculated to standardize and compare the total dopaminergic medication burden across participants, accounting for multiple medications with differing potencies.
Motor function was evaluated using Part III of the MDS-UPDRS at 6- and 12-months. This section consists of 33 item scores derived from 18 motor assessments. Each item is scored on a 5-point scale (0 = normal to 4 = severe), with higher cumulative scores indicating greater motor impairment.
To monitor health-related quality of life, the PDQ-39 was administered at 1, 2, 3, 6, and 12 months. This self-administered questionnaire evaluates eight dimensions of daily functioning (e.g., mobility, emotional well-being, cognition), with a Summary Index score reflecting the global impact of Parkinson’s disease on health status. Scores range from 0 to 100, with lower values indicating better quality of life.
Healthcare utilization and safety were monitored by documenting any emergency room visits or hospitalizations at 6 and 12 months. In addition, the incidence of device- or procedure-related serious adverse events (SAEs) was systematically recorded. SAEs were defined by standard regulatory criteria (e.g., hospitalization, death, permanent impairment). All reported safety events were adjudicated independently by a panel of three qualified clinicians.
Discussion
These findings demonstrate that remote, internet-based DBS management can achieve sustained clinical outcomes over at least twelve months, maintaining improvements in global status, symptom burden, and quality of life without compromising safety or increasing acute care utilization. The remote programming platform allowed clinicians to perform the same range of adjustments as during in-clinic sessions, including amplitude, pulse width, frequency, and contact configuration. Sessions were conducted synchronously via secure video with real-time assessment of symptom response. In the randomized phase, no adjustments made remotely required later correction in clinic, supporting the clinical equivalence of both modalities for DBS optimization. Consistent with this, large-scale real-world implementation of remote DBS programming has also demonstrated safety, feasibility, and usability, with about 800 sessions successfully conducted shortly after clinical introduction17. Importantly, the early gains observed with virtual programming within the first three months were preserved at one year, suggesting that benefits are durable and not merely driven by initial novelty or user enthusiasm. This adds real-world evidence that digital interventions can perform on par with in-clinic models when carefully implemented and supported by infrastructure18.
Beyond clinical durability, this study addresses key barriers in digital health research by illustrating a practical and efficient hybrid trial design. By embedding the RCT within an existing observational study, we minimized resource demands, reduced trial burden, and captured long-term outcomes without parallel infrastructure. This approach is particularly well-suited for evaluating digital therapeutics, which often require adaptive, user-driven delivery in naturalistic settings19. It also enhances the persistent challenges of generalizability and scalability in digital medicine, while maintaining methodological rigor. As digital health solutions become increasingly personalized, embedded hybrid study designs offer a practical blueprint for evaluating long-term effectiveness and feasibility in real-world care20,21.
Nevertheless, several limitations merit consideration. The twelve-month phase was observational and not powered for detecting between-group differences, which may have limited the ability to uncover subtle effects. Programming modality (remote versus in-clinic) was not documented during the observational phase, since after the RCT remote programming became part of standard care. This limits long-term comparisons between strictly separated groups. However, the design reflects real-world clinical practice, where both modalities are available and cannot ethically be withheld. Patients with advanced Parkinson’s disease also undergo regular in-clinic visits for multidisciplinary management beyond DBS therapy, reinforcing that remote programming should be regarded as complementary rather than substitutive.
Participant characteristics, such as higher digital literacy or willingness to engage with virtual care, may also limit generalizability. The absence of formal blinding or sham control, while typical in digital trials, restricts interpretation of subjective endpoints. Finally, some outcome variables, including medication adjustments and unscheduled visits, may have been influenced by external clinical factors beyond the study protocol. Future studies should aim to replicate these findings in broader populations and more diverse care settings.
In conclusion, remote digital management of DBS therapy delivers sustained and clinically meaningful benefits that match traditional care models over the long term. The hybrid trial design employed here supports a scalable, pragmatic framework for evaluating complex digital interventions in chronic disease management. These results reinforce the value of embedding digital care models into structured real-world research, offering both a clinical and methodological foundation for future innovation in digital medicine.
Methods
This study employed a prospective, multicenter hybrid design by embedding a three-month randomized controlled trial (RCT), which investigated remote internet-based adjustment of stimulation parameters (ROAM-DBS), within a larger post-market clinical follow-up cohort, which was designed to evaluate the long-term safety and effectiveness of DBS systems in real-world settings over a five-year period (ADROIT). Conducted across 17 clinical sites in the US and Europe, the trial adopted a decentralized structure, with most assessments and interventions conducted in participants’ home environments, consistent with ecological momentary assessment (EMA) principles. We obtained approval of the study protocol from Beth Israel Deaconess Medical Center Institutional Review Board, Dartmouth-Hitchcock Health Institutional Review Board, Rush University Medical Center Institutional Review Board, The Cleveland Clinic Institutional Review Board, Western Institutional Review Board-Copernicus Group (covering several US sites), Comité de Ética de La Investigación con Medicamentos del Hospital Universitario de la Princesa (covering all Spanish sites), Ethik-Kommission an der Medizinische Fakultät der Heinrich Heine Universität Düsseldorf, Ethik-Kommission bei der Landesärztekammer Rheinland-Pfalz, Ethik-Kommission an der Medizinische Fakultät der Eberhard-Karls-Universität und am Universitätsklinikum Tübingen, and the North of Scotland Research Ethics Committee. The authors complied with all relevant ethical regulations when conducting the study. Patients received detailed study information and signed an informed consent form before enrolling in the study. Both ROAM-DBS and ADROIT are registered on ClinicalTrials.gov (NCT05269862 registered on 2022-03-08 and NCT04071847 registered on 2019-08-28, respectively). The three-months results have been described previously16, here we report the twelve-months outcomes. Detailed methods have also been reported and are cited here where applicable16.
Study enrollment and allocation
All ADROIT participants were screened for inclusion in the ROAM-DBS study. Eligible patients were adults (≥21 years) with Parkinson’s disease scheduled for de novo implantation of the Infinity DBS System with the NeuroSphere™ Virtual Clinic feature (Abbott, USA). Inclusion criteria required the ability, either personally or via caregiver support, to participate in remote care sessions, including adequate internet access and technical competence.
Patients were enrolled up to six weeks before implantation and at least one week before initial programming. Baseline assessments included demographics, disease duration, medication use, Movement Disorders Society Unified Parkinson’s Disease Rating Scale (MDS-UPDRS), and Parkinson’s Disease Questionnaire-39 (PDQ-39). All but three randomized patients were provided an iPhone™ and Apple Watch™ for decentralized collection of patient-reported outcomes (PROs) and exploratory remote monitoring of motor symptoms. Delays in device provision affected three patients at study onset.
Randomization for the three-months RCT phase occurred postoperatively in a 1:1 ratio using a computer-generated, block-randomized scheme (blocks of four), managed by an independent statistician and concealed from the analysis team. Patients were allocated sequentially, independent of patient characteristics or site discretion, minimizing allocation bias. After initial in-clinic programming, patients received ongoing DBS adjustments either in clinic (IC) or via remote internet-based adjustment (RIBA). Randomization after initial programming avoided bias in initial stimulation settings. Neither patients nor clinicians were blinded to treatment allocation. All procedures followed site-specific standards of care and clinical necessity. After three months, all patients received access to RIBA, and it was thereafter at the discretion of patients and/or their physicians to choose between remote programming or in-clinic visits, with both options available to participants from both arms.
Statistical analysis
Statistical analysis was completed using SAS version 9.4 (SAS Institute Inc., Cary, NC, USA). Categorical data are presented as proportions and continuous variables are summarized with means and standard deviations (SD). 95% confidence intervals (CIs) are provided to compare intervention arms for non-powered endpoints. For longitudinal clinical outcomes (PGI-C, CGI-C, PDQ-39 Summary Index) we fitted linear mixed-effects models with fixed effects for randomized group (in clinic vs virtual clinic), time, the group-by-time interaction, and a random intercept for each participant. From these models, the improvement from baseline, between-group differences, and the interaction between group and time were examined. For the mixed-effects model analysis, PGI-C and GCI-C scores were dichotomized into a binary outcome (improved vs. not improved), with scores of or below 3 classified as improvement and scores of 4 or higher classified as no improvement. Figures were developed using GraphPad Prism (GraphPad Software, La Jolla, CA, USA).
Inclusion & Ethics
The investigators involved in this study were all movement disorder specialists, with efforts made to balance representation and geographic heterogeneity. The study directly addressed two critical and commonly cited barriers to research participation: logistical constraints (such as time, caregiving responsibilities, and transportation) and the lack of clinical infrastructure in underserved areas. By utilizing the RIBA program and collecting PROs remotely, the study enabled participation without requiring clinic visits, thereby expanding access to populations traditionally underrepresented in research, including those in rural or economically disadvantaged communities. However, limited internet access remains a challenge, particularly among low-income and older adults, and continues to restrict broader inclusion. As cellular network coverage (e.g., Edge, 3 G, 4 G, and 5 G) continues to expand globally, the feasibility of remote DBS programming is expected to improve further. These findings underscore the potential of digital health solutions to reduce barriers to care and increase equity in clinical research and real-world treatment delivery.
Data availability
The datasets generated and/or analyzed during the current study are not publicly available due to restrictions associated with Abbott’s policies but are available from the corresponding author on reasonable request. Due to the proprietary nature of the custom SAS macros used, the code cannot be shared publicly. Statistical analysis was completed using SAS version 9.4 (SAS Institute Inc., Cary, NC, USA). Figures were developed using GraphPad Prism (GraphPad Software, La Jolla, CA, USA).
Code availability
Due to the proprietary nature of the custom SAS macros used, the code cannot be shared publicly. Statistical analysis was completed using SAS version 9.4 (SAS Institute Inc., Cary, NC, USA). Categorical data are presented as proportions and continuous variables are summarized with means and standard deviations (SD). 95% confidence intervals (CIs) are provided to compare intervention arms for non-powered endpoints. Figures were developed using GraphPad Prism (GraphPad Software, La Jolla, CA, USA).
References
Narayan, S.M. et al. to digital health technologies: personalized framework and global perspectives. Nat. Rev. Cardiol 23, 1–14 (2025).
Smuck, M. et al. The emerging clinical role of wearables: factors for successful implementation in healthcare. npj Digit. Med. 4, 45 (2021).
Jansen, A. J. S. et al. Device based monitoring in digital care and its impact on hospital service use. npj Digit. Med. 8, 16 (2025).
Borges do Nascimento, I. J. et al. Barriers and facilitators to utilizing digital health technologies by healthcare professionals. npj Digit. Med. 6, 161 (2023).
Kim, M. et al. The digital therapeutics real-world evidence framework: an approach for guiding evidence-based digital therapeutics design, development, testing, and monitoring. J. Med. Internet Res. 26, e49208 (2024).
Windecker, D. et al. Generalizability of FDA-approved AI-enabled medical devices for clinical use. JAMA Netw. Open 8, e258052 (2025).
Morales-Briceño, H. et al. A single centre prospective study of three device-assisted therapies for Parkinson’s disease. npj Parkinsons Dis. 9, 101 (2023).
Wagle Shukla, A., Bange, M. & Muthuraman, M. Patient, target, device, and program selection for DBS in Parkinson’s disease: advancing toward precision care. npj Parkinsons Dis. 11, 195 (2025).
Zaman, M. S., Ghahari, S. & McColl, M. A. Barriers to accessing healthcare services for people with Parkinson’s disease: a scoping review. J. Parkinsons Dis. 11, 1537–1553 (2021).
Frassica, M. et al. Racial disparities in access to DBS: results of a real-world U.S. claims data analysis. Front. Neurol. 14, 1233684 (2023).
Esper, C. D. et al. Necessity and feasibility of remote tele-programming of deep brain stimulation systems in Parkinson’s disease. Park. Relat Disord. 96, 38–42 (2022).
Pinter, D., Jardahazi, E., Janszky, J. & Kovacs, N. Potential clinical and economic benefits of remote deep brain stimulation programming. Sci. Rep. 12, 17420 (2022).
Li, D. et al. Remotely programmed deep brain stimulation of the bilateral subthalamic nucleus for the treatment of primary parkinson disease: a randomized controlled trial investigating the safety and efficacy of a novel deep brain stimulation system. Stereotact Funct. Neurosurg. 95, 174–182 (2017).
Chen, S. et al. Remote programming for subthalamic deep brain stimulation in Parkinson’s disease. Front. Neurol. 13, 1061274 (2022).
Zhang, J. et al. Implementation of a novel bluetooth technology for remote deep brain stimulation programming: the pre- and post-COVID-19 Beijing experience. Mov. Disord. 35, 909–910 (2020).
Gharabaghi, A. et al. Accelerated symptom improvement in Parkinson’s disease via remote internet-based optimization of deep brain stimulation therapy: a randomized controlled multicenter trial. Commun. Med. 5, 31 (2025).
Silburn, P. et al. Rapid development of an integrated remote programming platform for neuromodulation systems through the biodesign process. Sci. Rep. 12, 2269 (2022).
Kambeitz-Ilankovic, L. et al. A systematic review of digital and face-to-face cognitive behavioral therapy for depression. npj Digit. Med. 5, 144 (2022).
Perski, O. & Short, C. E. Acceptability of digital health interventions: embracing the complexity. Transl. Behav. Med. 11, 1473–1480 (2021).
Wilson, B. E. & Booth, C. M. Real-world data: bridging the gap between clinical trials and practice. EClinicalMedicine 78, 102915 (2024).
Ford, I. & Norrie, J. Pragmatic trials. N. Engl. J. Med. 375, 454–463 (2016).
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
A.G., S.G., E.C., A.S., L.M.D., V.L.M., J.K., L.Z., R.A., M.S.F., M.J.S., L.L., B.L.W., C.W., H.L., D.M.H., C.L., and D.W. supervised data collection and participated in the conduct of the study. D.N. and C.A.S. participated in analyzing the patient data and aided in drafting the manuscript in collaboration with the academic authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare the following competing interests: Author AG was supported by the German Federal Ministry of Education and Research, the European Union Joint Program – Neurodegenerative Disease Research, the Else Kröner Medical Scientists Kolleg Artifical Intelligence for Clinical Brain Research, and by research grants from Medtronic, Abbott, Boston Scientific, all of which were unrelated to this work. Author SG reports research funding from patients’ groups, BMBF, Deutsche Forschungsgemeinschaft (DFG; SPP2177 Radiomics), UM Mainz, Abbott, Boston Scientific, Böhringer Foundation, Magventure, National MS Society, Precisis, Innovationsfond GBA (01NVF22107, INSPIRE – PNRM+). He also received lectures fees from Abbott, Abbvie, Bial, BVDN, IPSEN, Stada, UCB. Author AS has been serving as a consultant for Abbvie, Abbott, and Zambon. He received speaker honoraria from Abbvie, Abbott, Alexion, BSH medical communication, GE Healthcare, Kyowa Kirin, and Novartis. Author RA has received travel expenses and honoraria from Abbott for speaking in a training course. Author MS has received advisory/consulting fees from AbbVie, Abbott, Amneal Pharmaceuticals, Merz therapeutics, Medtronic, Neurocrine, Supernus. He has received research support from AbbVie, Abbott, Cerevel therapeutics, Cerevance, CND Lifesciences, Intra-cellular Therapies, Jazz Pharmaceuticals, Praxis Precision Medicine, Scion, and Teva. He has served on the speaking bureau for AbbVie, Amneal pharmaceuticals, Biogen, Merz, and Supernus. He has also received grant support from the HDSA. Author BW has served as a consultant in the last 12 months for Abbott, Boston Scientific, Medtronic, Supurnus, Mitsubishi Tanabe Pharma, Teva, and the Parkinson’s Foundation. Additionally, he has received grant support from the Parkinson’s Foundation and has received royalties from Springer. Author CW has reported consulting fees from Abbott, Boston Scientific, BrainLab, Medtronic, Nevro, NeuroOne, and Renishaw. He is also a shareholder for ZetaSurgical, and has received research support from the NIH (5U01NS113198) and Dixi Medical. Author CL is a consultant for Medtronic, Boston Scientific, and AbbVie. Author DW has received travel grants, speaker's honoraria, and research funding from Abbott, Boston Scientific, and Medtronic. Authors CAS and DN are Abbott employees. This study was funded by Abbott. Abbott provided technical support and participated in the study design and data analysis and aided in drafting the manuscript in collaboration with the academic authors.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Gharabaghi, A., Groppa, S., Casas, E. et al. Real-world multicenter assessment of sustained clinical outcomes after digital deep brain stimulation. npj Digit. Med. 9, 133 (2026). https://doi.org/10.1038/s41746-025-02315-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41746-025-02315-5