Abstract
Glucocorticoid-induced osteonecrosis of the femoral head is a joint dysfunction disease. Impaired local angiogenesis and reduced perfusion are early pathological features of glucocorticoid-induced osteonecrosis of the femoral head, resulting from vascular endothelial cell damage and suppressed angiogenesis caused by prolonged glucocorticoid exposure. This study focuses on the role of angiogenesis in glucocorticoid-induced osteonecrosis of the femoral head, particularly the potential of GDF11 in promoting angiogenesis. Our findings indicate that GDF11 expression diminished in patients with osteonecrosis of the femoral head compared to those with femoral neck fractures. We establish a male Sprague-Dawley rat model of glucocorticoid-induced osteonecrosis of the femoral head and utilize human umbilical vein endothelial cells to explore the role of GDF11 on osteogenesis and angiogenesis. A series of in vivo and in vitro experiments are conducted, the result shows that GDF11 could reverse the damaged angiogenic and osteogenic ability caused by methylprednisolone. We perform RNA sequencing in human umbilical vein endothelial cells and reveal that GDF11 promotes the expression of angiogenic factors and migration of endothelial cells through activating the PI3K/AKT/eNOS pathway, thereby promoting angiogenesis. Our study clarifies the role and mechanism of GDF11 in regulating local angiogenesis in glucocorticoid-induced osteonecrosis of the femoral head.

Similar content being viewed by others
Introduction
Glucocorticoid (GC)-induced osteonecrosis of the femoral head (ONFH) represents a prevalent disorder affecting joint function causing disability and severely affecting patients’ quality of life. GC-induced ONFH is predominantly caused by the prolonged and high-dose use of GC1. Over the years, GC-induced ONFH has become the major etiologic type among nontraumatic femoral head necrosis due to the wide clinical application of GCs2. GC-induced ONFH initially manifests as necrosis and cystic degeneration in the femoral head, if timely treatment is not received, the femoral head of 80% patients collapse within 4 years and usually with complication of hip joint osteoarthritis, which requires total hip arthroplasty (THA)3,4. Currently, as the pathogenesis of GC-induced ONFH is not clearly understood, effective treatment in the early stage is still lacking. Thus, exploring the pathogenesis and new therapeutic targets is of great importance for the diagnosis and treatment of GC-induced ONFH.
Neovascularization is an important part of the homeostatic renewal of bone, it involves a coupling between angiogenesis and osteogenesis through a complicated signaling network. Decreased local vascularization of the femoral head and inadequate blood flow are important early pathological changes in GC-induced ONFH5,6. Evidence indicates impaired vascular endothelial cells and angiogenesis inhibition in femoral head of patients with high-dose GC7. In addition, GC-mediated inhibition of angiogenesis lead to a hypercoagulable state of blood related to a direct reduction of blood flow in femoral head and lead to enhanced vasoconstriction of peripheral tissues, which contribute to the development of GC-induced ONFH1. Human umbilical vein endothelial cells (HUVECs) is widely used to explore the in vitro angiogenesis ability of endothelial cells in cardiovascular disease, oncology, tissue engineering, and is also routinely used in ONFH and other vascular-related bone diseases to study angiogenesis in bone8,9,10,11,12. Studies have verified the ability of GC to inhibit angiogenesis by suppressing the proliferation of endothelial cells13, inhibiting the formation of vessel structure14, and decreasing the expression of pro-angiogenic factors15,16. Sirtuin 1, growth differentiation factor 11 (GDF11), growth differentiation factor 15 (GDF15) and klotho are potential biomarkers of aging17, some of which have been reported to exert multiple effects in various diseases.
GDF11, a member of the transforming growth factor beta (TGF-β) superfamily, was predominantly expressed in mammals in various organs such as bone and kidney, it has a critical role as a key regulator of patterning and formation of several tissues in embryonic development18,19,20,21. GDF11 signals through a heteromeric complex of type II (ACVR2A, ACVR2B) and type I (ACVR1B, ACVR1C, TGFBR1) activin receptors22. It regulates development, repair and metabolism, with non-endothelial-restricted expression but critical endothelial modulatory roles in angiogenesis and regeneration23,24. Recently, accumulating evidence has indicated the ability of GDF11 to rejuvenate angiogenesis25,26. In the treatment of ischemic diseases, GDF11 was shown to positively regulate the progression of angiogenesis as well as proangiogenic activities of stem cells27. GDF11 can improve the cerebral vasculature and enhance neurogenesis18. Furthermore, it has been found that supplementation with exogenous GDF11 significantly enhanced re-endothelialization of endothelial progenitor cells (EPC), which increases angiogenesis, improve blood flow in diabetic rats with hindlimb ischemia, treatment of EPCs with GDF11 alleviated the functional abnormalities and apoptosis of EPCs28. There is growing evidence regarding the diverse effects of GDF11, it remains unclear whether GDF11 can mitigate GC-induced ONFH, including its specific mechanism of action.
ERK-MAPK, PI3K-AKT, BMP-SMAD, and JAK-STAT are important pathways in ONFH and are involved in angiogenesis and osteogenesis within the skeletal system29,30,31,32. Mechanistic studies reveal that Ginkgo biloba extract elicits vasoprotection by activating the PI3K/AKT/eNOS axis. This pathway upregulates key angiogenic and anti-apoptotic factors, driving functional angiogenesis and restoring hemodynamic perfusion in femoral heads33. PI3K/ATK pathway plays a critical role in many biological processes in vivo through phosphorylation and dephosphorylation, especially in angiogenesis34. Cao’s research demonstrated that inhibitors targeting PI3K and eNOS could selectively induce apoptosis and compromise angiogenic capacity in endothelial progenitor cells33, Lin et al. elucidated the involvement of the PI3K/AKT/eNOS pathway in protection against inflammation in HUVECs35. To delineate GDF11’s mechanistic contributions to GC-induced ONFH, we developed a rat GC-induced ONFH model, conducted RNA sequencing analysis of GDF11-treated HUVECs, and validated findings using PI3K-AKT-eNOS pathway inhibitors.
In this study, we found GDF11 levels were significantly reduced in patients and rat model of GC-induced, Administration of exogenous GDF11 rescued the angiogenesis impairment caused by GC. Utilizing both HUVECs and a GC-induced ONFH rat model, we discovered that GDF11 promotes angiogenesis through phosphorylation of PI3K/AKT/eNOS pathway. Our results could illuminate novel therapeutic approach against GC-induced ONFH, supported by histological, cellular, and molecular evidence.
Results
Reduction of GDF11 is associated with impaired angiogenesis in GC-induced ONFH
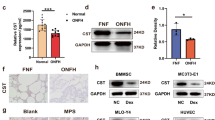
To investigate the changes in angiogenesis ability of overdose of GC, we selected HUVECs to investigate the effects of methylprednisolone (MP) on endothelial cell injury. HUVECs were cultured under 100 μM of MP, the concentration was determined by the Cell Counting Kit-8 (CCK-8) assay (Fig. S1A). RNA sequencing was performed in control group and MP treatment group. The results indicated a decrease of GDF11 and angiogenesis biomarkers in MP treatment group compared with the control group (Fig. 1A). Gene Set Enrichment Analysis (GSEA) results revealed downregulation of genes involved in cell migration, circulatory system development, blood vessel morphogenesis, circulatory system development and vasculature development following MP treatment (Fig. 1B, Fig. S1B). Gene Ontology (GO) enrichment analysis showed that MP affected angiogenesis (Fig. 1C). Then, we collected femoral head samples from patients undergoing THA (Fig. 1D), detailed patient information was shown in Table S1. Hematoxylin and eosin (H&E) and Safranin O staining demonstrated extensive destruction of trabecular architecture and marrow necrosis in femoral head tissues from GC-induced ONFH patients, markedly exceeding the pathological changes observed in non-ONFH controls (Fig. 1E, F). In GC-induced ONFH, radiographic and MRI examinations demonstrate significant femoral head collapse, accompanied by bone marrow edema and necrotic areas (Fig. 1G). Immunofluorescence detection of vascular endothelial growth factor A (VEGFA) was performed on femoral head sections from both non-ONFH patients and ONFH patients. Results demonstrated significantly reduced VEGFA levels in GC-induced ONFH specimens compared to non-ONFH tissues, suggesting diminished vascular density in ONFH (Fig. S1C, D). Additionally, immunofluorescence staining demonstrated diminished GDF11 expression in femoral-head tissues obtained from patients with GC-induced ONFH (Fig. 1H, I). Moreover, Western blotting analysis of human femoral head tissues revealed significantly reduced expression of GDF11 and angiogenesis markers (CD31, VEGFA) in GC-induced ONFH patients compared to non-ONFH controls (Fig. 1J-M). Staining for tartrate resistant acid phosphatase (TRAP) revealed an abnormal increase in the number of osteoclasts within the subchondral bone of the femoral head in patients with GC-induced ONFH. Masson trichrome staining demonstrated obliteration of the stratified structure in GC-induced ONFH patients versus non-ONFH controls, characterized by chaotic intermingling of blue-stained collagen and red-stained cellular elements. This pattern signifies fracture of the calcified cartilage layer and osteochondral dissociation (Fig. S1E, F).
A Microarray heatmap of HUVECs from the control group and MP group (n = 3 per group). B GSEA demonstrated negative enrichment in gene sets involved in cell migration, circulatory system process, and blood vessel morphogenesis after MP exposure. C GO enrichment analysis was performed on DEGs between the control group and the MP-treated group. D Gross morphology of non-ONFH and GC-induced ONFH femoral head. E, F H&E staining of femoral head tissue of non-ONFH patients and ONFH patients and histomorphometric analysis (n = 6 per group). Scale bar = 200 μm. G X-ray and MRI images of hip joints from patients. H Representative immunofluorescence images of femoral head tissues from non-ONFH patients and ONFH patients, GDF11 (green), nuclei counterstained with DAPI (blue). Scale bar = 200 μm. I Quantitative analysis of immunofluorescence staining of GDF11 (n = 6 per group). J Western blotting was performed to detect GDF11, CD31, and VEGFA protein expression in femoral head tissues from non-ONFH patients and ONFH patients (n = 6 per group). K–M Quantification of Western blotting analysis in different groups (n = 6 per group). Significant differences are indicated as follows: ns P > 0.05, *P < 0.05, **P < 0.01, ***P < 0.001 and ****P < 0.0001.
Supplementation with GDF11 attenuates GC-induced angiogenesis inhibition in HUVECs
Previous studies have highlighted the crucial role of angiogenesis in GC-induced ONFH, to determine whether GDF11 modulates the angiogenic capacity of endothelial cells exposed to high-dose MP in vitro, HUVECs were treated with MP alone or in combination with GDF11 for 24 h. The concentration of GDF11 was set at 1 ng/μl using CCK-8 assay (Fig. 2A). Real-time PCR analysis revealed that MP significantly downregulated GDF11 expression and reduced transcript levels of angiogenesis-related genes (VEGFA and CD31). GDF11 treatment reversed this MP-induced suppression of angiogenic gene expression (Fig. 2B), consistent with restored CD31 protein expression observed by immunofluorescence (Fig. 2C, D). The results of Western blotting showed that the addition of MP reduced the expression of VEGFA and CD31, the addition of GDF11 was able to reverse these trends (Fig. 2E–G). To examine the ability of GDF11 influences on cell migration and blood vessel formation, we performed transwell, tube formation and scratch assays. The transwell assay indicated that GDF11 mitigated the migration impairment of HUVECs caused by MP. Results from tube formation test indicated that GDF11 increased tube formation ability of HUVECs (Fig. 2H–J). The result of scratch assay indicated that MP markedly reduced the wound-healing capacity of HUVECs at 12, 24, and 48 h, which was counteracted by the addition of GDF11 (Fig. 2K, L). Together, the above findings suggest that the proangiogenic potential of GDF11 on HUVECs.
A CCK-8 assay of GDF11 in HUVECs (n = 3 per group), compared with control group (0 ng/μl). B The Real-time PCR was performed to detect GDF11, CD31 and VEGFA expression in control (untreated), MP-treated, and MP + GDF11-treated groups (n = 3 per group). C Representative immunofluorescence images of HUVECs in control (untreated), MP-treated, and MP + GDF11-treated groups, red (CD31), green (phalloidine), blue (nuclei). Scale bar = 20 μm. D Quantitative analysis of mean fluorescence in different groups (n = 3 per group). E Western blotting was performed to detect CD31 and VEGFA protein expression in control (untreated), MP-treated, and MP + GDF11-treated groups. F, G Quantification of Western blotting analysis in different groups (n = 3 per group). H Transwell assay and Tube formation assay in control (untreated), MP-treated, and MP + GDF11-treated groups. Scale bar = 200 μm (upper panel) and 100 μm (lower panel). I Quantification of migrated cells from Transwell assays in control (untreated), MP-treated, and MP + GDF11-treated groups (n = 3 per group). J Quantification of total branching points, total length and total meshes area in different groups (n = 3 per group). K, L Scratch assay for 0, 12, 24, and 48 h and quantitative analysis of different groups (n = 3 per group). Scale bar = 200 μm. Significant differences are indicated as follows: ns P > 0.05, *P < 0.05, **P < 0.01, ***P < 0.001 and ****P < 0.0001.
Effect of GDF11 on osteogenesis and blood perfusion in GC-induced ONFH
We established a rat model of GC-induced ONFH and applied adeno-associated virus overexpressing GDF11 (AAV-GDF11) to investigate the effect of GDF11 on GC-induced ONFH. Femoral head samples from the sham group, AAV-empty vector control group (AAV-NC), and AAV-GDF11 group were subjected to immunofluorescence co-staining for GDF11 and VEGFA. Results demonstrated significantly elevated GDF11 expression within VEGFA-high regions in the AAV-GDF11 group, indicating successful GDF11 overexpression in endothelial cells following AAV-GDF11 delivery (Fig. S2A, B). Micro computed tomography (micro-CT) scan was performed to measure morphological changes of GC-induced ONFH. From the coronal, cross-sectional views and three-dimensional (3D) reconstruction, it was observed that the MP group exhibited severe trabecular bone loss and collapse in the weight-bearing area. This condition was significantly improved after the injection of AAV-GDF11 (Fig. 3A). Based on micro-CT analysis of the region of interest (ROI) in the weight-bearing area, we observed that the mean bone mineral density (BMD) of the MP group (0.281 ± 0.043 g/cm³) was markedly reduced compared to the control group (0.841 ± 0.151 g/cm³). Intriguingly, we found that AAV-GDF11 improved BMD, bone volume/total volume fraction (BV/TV), trabecular number (Tb.N), and trabecular thickness (Tb.Th) compared with the model group, while reducing trabecular separation (Tb.Sp) (Fig. 3B–F).
A Micro-CT images (3D reconstructions, coronal planes) of the sham, MP, MP + AAV-NC, and MP + AAV-GDF11 groups. B Quantification of BMD in different groups (n = 6 per group). C Quantification of BV/TV in different groups (n = 6 per group). D Quantification of Tb.N in different groups (n = 6 per group). E Quantification of Tb.Th in different groups (n = 6 per group). F Quantification of Tb.Sp in different groups (n = 6 per group). G–I H&E staining and histomorphometric analysis of sham, MP, MP + AAV-NC groups, and MP + AAV-GDF11 groups (n = 6 per group). Scale bar = 1 mm (upper panel) and 50 mm (lower panel). J Representative images of double calcein labeling and Alizarin Red S staining in the sham, MP, MP + AAV-NC, and MP + AAV-GDF11 groups. Scale bar = 1 mm (upper panel) and 50 μm (lower panel). K Quantification of MAR in different groups (n = 6 per group). L Quantification of mineralizing surface/bone surface in different groups (n = 6 per group). Significant differences are indicated as follows: ns P > 0.05, *P < 0.05, **P < 0.01, ***P < 0.001 and ****P < 0.0001.
In addition, we prepared rat femoral head sections, histological staining further underscored the impact of AAV-GDF11 on mitigating bone formation inhibition induced by MP. Specifically, H&E staining revealed significantly higher numbers of empty lacunae and more pronounced trabecular collapse (defined by the presence of at least one of the following: focal angulation >30°, microfracture, focal compression with >50% trabecular thinning, or fragmentation) in the MP group. These pathological changes were notably reversed after AAV-GDF11 administration (Fig. 3G–I). Safranin O staining was performed to assess the condition of cartilage, AAV-GDF11 administration ameliorated MP-induced histopathological changes, including reductions in calcified cartilage zones, tidemark disruption, and trabecular fragmentation (Fig. S2C). AAV-GDF11 injection significantly enhanced osteogenic activity, as evidenced by elevated mineral apposition rate (MAR) and mineralized surface/bone surface (MS/BS) in double calcein labeling and alizarin red S staining assays in rat femoral head sections. Quantifications were performed at the endocortical surface of the ROI (Fig. 3J–L). Besides, we investigated the effect of GDF11 on osteogenesis in Rats-derived bone mesenchymal stem cells (BMSC) by western blotting assay. Except for the control group, BMSC were treated with either 100 μM MP alone or 100 μM MP combined with 1 ng/μl GDF11. The results showed that GDF11 could promote the expression of collagen I (COL1) and runt-related transcription factor 2 (RUNX2) protein in vitro (Fig. S2D, E). Additionally, alkaline phosphatase (ALP) and alizarin red S (ARS) staining was performed on cells cultured in osteogenic medium for at least 14 days. The results demonstrated that GDF11 promotes osteogenic differentiation of BMSC, increases ALP activity, and enhances calcification in the extracellular matrix (Fig. S2F, G).
Blood supply is also an important aspect of GC-induced ONFH. To explore whether GDF11 could promote angiogenesis on established rat model, we evaluated the levels of femoral-head vascularization by angiography. We generated 3D reconstructions of the vascular network within the femoral head and measured the vascular volume (Fig. 4A). Our analysis revealed that MP treatment significantly compromised both the perfusion area and volume of the femoral head vasculature. However, overexpression of GDF11 effectively mitigated these adverse effects, restoring vascular integrity (Fig. 4B, Fig. S3A). Then, we collected femoral heads from rats underwent angiography mentioned above, and cross-section slices of the specimens were observed using H&E and Masson staining (Fig. 4C). The number of Microfil particles on the slides represents the density of blood vessels within the femoral head (Fig. 4D). Immunofluorescence staining of femoral heads revealed that the expression pattern and distribution of the vascular marker CD31 closely matched those of the osteogenic marker COL1. Expression levels of both COL1 and CD31 were reduced in the MP group compared to the control group and the GDF11 overexpression group (Fig. 4E–G). Histological staining, immunohistochemical (IHC) staining for CD31 were performed to further confirm the blood supply status. Both the expression of CD31 and the number of Microfil particles were significantly decreased in the MP group; however, GDF11 overexpression reversed these changes (Fig. 4H, I).
A, B Angiography of femoral heads and subsequent vessel volume quantification in the experimental groups: sham, MP, MP + AAV-NC, MP + AAV-GDF11 (n = 6 per group). C Representative transverse sections of H&E and Masson staining from the sham, MP, MP + AAV-NC, and MP + AAV-GDF11 groups are shown. Scale bar = 500 μm. D Quantification of angiography particles number in different groups (n = 6 per group). E Representative immunofluorescence images of the sham, MP, MP + AAV-NC, and MP + AAV-GDF11 groups showing CD31 (green), COL1 (red), and DAPI-stained nuclei (blue). Scale bar = 100 μm. F, G Quantitative analysis of mean fluorescence in different groups (n = 6 per group). H, I IHC staining for CD31 and quantitative analysis of CD31-positive cells in the sham, MP, MP + AAV-NC, and MP + AAV-GDF11 groups (n = 6 per group). Scale bar = 100 μm (upper panel) and 50 μm (lower panel). Significant differences are indicated as follows: ns P > 0.05, *P < 0.05, **P < 0.01, ***P < 0.001 and ****P < 0.0001.
GDF11 exerts vasculogenic function through the PI3K-AKT-eNOS pathway
GDF11 shows anti-inflammation, anti-oxidation, and anti-aging effects as a key regulator. PI3K-AKT-eNOS pathway was proven to protect GC-induced ONFH by promoting angiogenesis. We performed RNA-sequencing in MP group and MP + GDF11 treatment group, the result showed the expression level of NOS3 and angiogenesis biomarkers was increased in GDF11 supplementation group (Fig. 5A, Fig. S3B). GO and Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment analysis indicated that GDF11 supplementation enhanced angiogenesis and cell migration, promoted protein phosphorylation, and activated the PI3K-AKT signaling pathway (Fig. 5B, C). GSEA results revealed upregulation of genes involved in cell migration, vasculature development, blood vessel morphogenesis and circulatory system development following GDF11 treatment (Fig. 5D, Fig. S3C).
A Microarray heatmap of HUVECs from the MP groups with or without GDF11 treatment (n = 3 per group). B Bubble plots of differentiated pathways in indicated groups. C GO enrichment analysis of DEGs was conducted comparing MP groups with versus without GDF11 supplementation. D GSEA demonstrated significant positive enrichment in gene sets involved in cell migration, circulatory system process, and blood vessel morphogenesis after GDF11 treatment. E, F Immunofluorescence staining of p-PI3K and p-eNOS in HUVECs, with quantitative analysis of mean fluorescence intensity in control (untreated), MP-treated, and MP + GDF11-treated groups (n = 3 per group). Scale bar = 20 μm. G Western blotting was performed to detect PI3K, AKT, eNOS, p-PI3K, p-AKT, and p-eNOS protein expression in control (untreated), MP-treated, and MP + GDF11-treated groups. H Quantification of Western blotting analysis in different groups (n = 3 per group). I NO levels in control (untreated), MP-treated, and MP + GDF11-treated groups (n = 3 per group). Significant differences are indicated as follows: ns P > 0.05, *P < 0.05, **P < 0.01, ***P < 0.001 and ****P < 0.0001.
In the results of immunofluorescence staining of HUVECs, we found that MP could inhibit the phosphorylation of PI3K and eNOS, while the addition of GDF11 could reverse this trend (Fig. 5E, F), suggesting GDF11 might exert its effect via PI3K and eNOS. The result of GSEA also showed upregulation of genes involved in PI3K-AKT signaling pathway following GDF11 treatment (Fig. S3D). Then Western blotting of HUVECs revealed that MP inhibited the phosphorylation of PI3K, AKT and eNOS, which was antagonized with GDF11 supplementation (Fig. 5G, H). The nitrite levels of different groups were examined through Griess method, indicating that intracellular nitric oxide (NO) generation was promoted with GDF11 supplementation (Fig. 5I). We also conducted immunofluorescence staining on rat tissue sections and found that GDF11 can alleviate the inhibition of PI3K and eNOS phosphorylation caused by MP (Fig. S4A, B).
To further investigate the effect of GDF11 on GC-induced angiogenesis suppression, we treated HUVECs with GDF11, in the presence of the PI3K inhibitor LY294002 (10 μM) or the eNOS inhibitor N omega-Nitro-L-arginine methyl ester hydrochloride (L-NAME, 100 μM) for 24 h. The concentrations of LY294002 and L-NAME were predetermined by CCK-8 assays (Fig. S3E, F). The results of Western blotting showed that the inhibitor could counteract the promoting effect of GDF11 on the phosphorylation of the PI3K-AKT-eNOS signaling pathway (Fig. 6A, B). The levels of NO in the inhibitor groups were significantly lower than those in the GDF11-added groups, indicating that the addition of LY294002 and L-NAME could inhibit the effect of GDF11 in promoting NO production (Fig. 6C). Furthermore, we used immunofluorescence to detect the effects of adding inhibitors on VEGFA. The results showed that the addition of inhibitors could significantly inhibit the promoting effect of GDF11 on VEGFA (Fig. 6D, E). The results of Western blotting also showed that the addition of inhibitors could inhibit the promoting effect of GDF11 on the expression of VEGFA and CD31 proteins (Fig. 6F, G). The migration of HUVECs is crucial for angiogenesis. Scratch assay results demonstrated that, at 12, 24, and 48 h, the wound healing capacity of the GDF11-treated group was significantly enhanced compared to the MP group. However, this effect was attenuated by the inhibitors LY294002 and L-NAME (Fig. 6H, I). Similarly, the transwell migration assay revealed that GDF11 supplementation robustly restored the impaired migratory activity observed in the MP group, an effect that was counteracted by PI3K and eNOS inhibitors (Fig. 6J, K). Tube formation assays, evaluating the in vitro angiogenic potential of HUVECs, demonstrated that GDF11 treatment significantly enhanced tubulogenesis relative to the MP group. However, this pro-angiogenic effect was abolished by co-treatment with either LY294002 or L-NAME (Fig. 6L). Quantitative assessment further demonstrated that GDF11 treatment enhanced the total meshed area, total length, and branching point count, with these effects being nullified by the inhibitors (Fig. 6M–O).
A Western blotting was performed to detect expression of PI3K, AKT, eNOS, p-PI3K, p-AKT, and p-eNOS in the following groups: MP, MP + GDF11-treated, MP + GDF11 + LY294002-treated, and MP + GDF11 + L-NAME-treated cells. B Quantification of Western blotting analysis in different groups (n = 3 per group). C NO levels in MP, MP + GDF11-treated, MP + GDF11 + LY294002-treated, and MP + GDF11 + L-NAME-treated groups (n = 3 per group). D Representative immunofluorescence images of MP, MP + GDF11-treated, MP + GDF11 + LY294002-treated, and MP + GDF11 + L-NAME-treated groups showing red (CD31), green (phalloidine), blue (nuclei). Scale bar = 20 μm. E Quantitative analysis of mean fluorescence in different groups (n = 3 per group). F Western blotting was performed to detect expression of CD31 and VEGFA in the following groups: MP, MP + GDF11-treated, MP + GDF11 + LY294002-treated, and MP + GDF11 + L-NAME-treated cells. G Quantification of Western blotting analysis in different groups (n = 3 per group). H, I Scratch assay for 0, 12, 24, and 48 h and quantitative analysis in different groups (n = 3 per group). Scale bar = 200 μm. J Transwell assays were performed in four groups: MP, MP + GDF11-treated, MP + GDF11 + LY294002-treated, and MP + GDF11 + L-NAME-treated cells. Scale bar = 100 μm. K Quantification of migration cells of Transwell assay (n = 3 per group). L Tube formation assays were performed in four groups: MP, MP + GDF11-treated, MP + GDF11 + LY294002-treated, and MP + GDF11 + L-NAME-treated cells. Scale bar = 200 μm. M–O Quantification of Tube formation assay (n = 3 per group). Significant differences are indicated as follows: ns P > 0.05, *P < 0.05, **P < 0.01, ***P < 0.001 and ****P < 0.0001.
We further administered LY294002 and L-NAME to rat models of GC-induced ONFH to elucidate the angiogenic effects of GDF11 in vivo, PI3K-antagonist group was treated with 0.5 mg/kg LY294002 via intraperitoneal (i.p.) injection, eNOS-antagonist group was treated with 20 mg/kg L-NAME, groups were administered daily for 3 consecutive weeks from the second day after MP injection. Micro-CT scan was performed on different groups to determine the radiological manifestations of GC-induced ONFH after PI3K and eNOS were inhibited, the result showed that LY294002 and L-NAME treatment significantly impaired the protective effect of GDF11 on GC-induced ONFH (Fig. 7A), We observed that the introduction of inhibitors led to reductions in BMD, BV/TV, Tb.N, and Tb.Th when compared to the group receiving GDF11 supplementation, while simultaneously enhancing Tb.Sp (Fig. 7B–F). In H&E staining, more trabecular collapse was observed after LY294002 and L-NAME treatment compared with GDF11 supplementation group (Fig. 7G, H). Safranin O and Masson staining were performed for the assessment of bone and cartilage conditions (Fig. S4C). We also found that the expression level of CD31 was reduced in LY294002 and L-NAME treatment group compared with GDF11 supplementation group in immunofluorescence staining of femoral head tissue (Fig. 7I, J). To visually reflect the blood supply of the femoral head, we performed angiography in inhibitors addition groups, we discovered that the blood supply to the femoral head significantly decreased after the addition of the inhibitor (Fig. 7K–M). Besides, we collected the liver, heart, spleen, lung, and kidney from rats in each group for H&E staining and found no significant differences (Fig. S4D). Together, the above findings suggest that the protective effects of GDF11 are associated with the stimulation of angiogenic responses through PI3K-AKT-eNOS pathway.
A Micro-CT images (3D reconstructions, coronal planes) of the MP + AAV-NC, MP + AAV-GDF11, MP + AAV-GDF11 + LY294002, and MP + AAV-GDF11 + L-NAME groups. B Quantification of BMD in different groups (n = 6 per group). C Quantification of BV/TV in different groups (n = 6 per group). D Quantification of Tb.N in different groups (n = 6 per group). E Quantification of Tb.Th in different groups (n = 6 per group). F Quantification of Tb.Sp in different groups (n = 6 per group). G, H H&E staining and histomorphometric analysis of the MP + AAV-NC, MP + AAV-GDF11, MP + AAV-GDF11 + LY294002, and MP + AAV-GDF11 + L-NAME groups (n = 6 per group). Scale bar = 1 mm (upper panel) and 50 mm (lower panel). I Representative immunofluorescence images of MP + AAV-NC, MP + AAV-GDF11, MP + AAV-GDF11 + LY294002, and MP + AAV-GDF11 + L-NAME groups showing green (CD31) and blue (nuclei). Scale bar = 100 μm. J Quantitative analysis of mean fluorescence in different groups (n = 6 per group). K–M Representative angiography images of femoral heads and quantification of vessel volume and vessel volume fraction in MP + AAV-NC, MP + AAV-GDF11, MP + AAV-GDF11 + LY294002, and MP + AAV-GDF11 + L-NAME groups (n = 6 per group). Significant differences are indicated as follows: ns P > 0.05, *P < 0.05, **P < 0.01, ***P < 0.001 and ****P < 0.0001.
Discussion
Reduced vascularization of the femoral head and disruption of its blood supply are key pathological changes in early GC-induced ONFH. In adults, the primary blood supply to the femoral head originates from branches of the profunda femoris artery. These branches anastomose on the anterior and posterior femur, forming an arterial ring. Unlike traumatic ONFH, GC-induced ONFH typically involves vascular damage distal to these major arteries—specifically, within the micro-arteries and specialized vessels of the intraosseous bone tissue. Recent studies have found that osteogenesis-angiogenesis coupling plays an important role in protecting the osteogenesis in ONFH5. In the process of bone metabolism, neovascularization is an important link, which involves the complex coupling between angiogenesis and osteogenesis through various cells, factors, and signaling pathways36. However, there is still a very limited understanding of the specific mechanisms, and also a lack of effective interventions and protective methods targeting the local blood supply of the femoral head37.
A substantial amount of research has confirmed that high doses of GC can directly cause endothelial cell injury and inhibit local angiogenesis38,39. Furthermore, the inhibition of angiogenesis mediated by GC not only directly reduces local blood supply within bone tissue but may also lead to a hypercoagulable state and enhance peripheral tissue vasoconstriction, which are high-risk factors for the development of ONFH40. In our study, we established a rat model of GC-induced ONFH by intramuscular injection of MP into the gluteus maximus, which has been proved to be an ideal preclinical animal model for GC-induced ONFH41. We demonstrated MP’s inhibitory effect on angiogenic function in vivo using Micro-CT and histological staining. This effect was further confirmed in vitro by treating HUVECs with exogenous MP, HUVECs are a well-established in vitro model for studying fundamental endothelial cell behaviors due to their conserved expression of key markers (e.g., CD31, vWF) and functional pathways across vascular beds. While they do not fully replicate the specialized heterogeneity of bone marrow endothelial cells (BMECs), they provide a reproducible platform for investigating core mechanisms relevant to vascular biology in skeletal tissues. Angiography offers distinct benefits in visualizing the blood supply and internal vasculature of the femoral head. Previous studies that performed angiography mainly analyzed the coronal angiograms of the femoral head after 3D reconstruction39. In our angiography analysis, we innovatively extracted the femoral heads from rats that had undergone angiography, performed slicing and staining, and discovered that the contrast agent residues could be observed and were correlated with the blood supply of the femoral head. This method of ours can provide important references for subsequent research.
GDF11, as a member of the TGF-β superfamily, has shown broad application prospects in the study of GC-induced ONFH. GDF11 has antioxidant and cell-protective effects42. Studies have found that GDF11, by activating autophagy, can significantly reduce the oxidative stress-induced senescence of endothelial progenitor cells43 and GDF11 also plays an important role in maintaining bone volume and the number of osteoblasts44. Prior research has established the presence of GDF11 in various tissues including bone, blood vessels, spleen, intestines, and brain45,46,47,48,49,50. Our findings revealed that GDF11 is expressed in the femoral head, and its expression levels are reduced in patients with GC-induced ONFH. Additionally, we observed that overexpression of GDF11 enhances angiogenesis during MP treatment in a rat model of GC-induced ONFH, consistent with previous studies demonstrating that GDF11 promotes angiogenesis28,44. GDF11 treatment enhanced cell migration and tube formation in vitro, key processes for vascular regeneration following vascular damage caused by GC. Moreover, GDF11 upregulated the expression of angiogenic markers such as CD31 and VEGFA both in vivo and in vitro. Furthermore, we found that GDF11 counteracted the inhibitory effect of GC on the phosphorylation of the PI3K-AKT-eNOS signaling pathway, which is implicated in GC-induced ONFH33.
In our study, GC decreased the phosphorylation level of PI3K-AKT-eNOS signaling pathway, however, upregulated GDF11 reversed the expression of p-PI3K, p-AKT and p-eNOS. The PI3K-AKT-eNOS signaling pathway is believed to play a significant role in angiogenesis in ONFH, which is consistent with our experimental results33. Based on the antibodies we used, we speculated that the action site of PI3K is located at p85 regulatory subunit, which has also been confirmed to be associated with angiogenesis in previous studies51. Also, we reversely verified the results on the PI3K-AKT-eNOS signaling pathway with the inhibitor LY294002 and L-NAME. However, the upstream signals that regulate the PI3K-AKT-eNOS signaling pathway remain unclear and require further investigation.
The current research was subject to several limitations. Firstly, this study did not examine the upstream and downstream regulation of the observed effects. However, based on existing literature, receptors such as TGF-β type I receptors may mediate the angiogenic effects of GDF1152,53. Additionally, inflammation and osteoclast activity are known to contribute to bone resorption, and GDF11’s anti-inflammatory properties have been previously documented. Therefore, exploring other relevant signaling pathways in future studies could provide further insights. Moreover, while this study primarily focused on GDF11’s pro-angiogenic role, its osteogenic effects were not thoroughly investigated and should be examined in subsequent studies. Besides, due to the species differences between rats and humans, the preventive effect and mechanisms of GDF11 on human need to be further verified.
In summary, our findings suggest that GDF11 exerts a protective effect against GC-induced ONFH by enhancing the secretion of angiogenic cytokines, boosting cell migration, and stimulating tube formation. Moreover, we noted a significant improvement in blood circulation within the femoral head of rats overexpressing GDF11. These actions may be linked to the activation of the PI3K-AKT-eNOS signaling pathway through phosphorylation. Collectively, these results indicate that GDF11 mitigates GC-induced vascular damage and could represent a promising therapeutic target for the treatment of GC-induced ONFH in the future.
Methods and materials
Human femoral head sample
All clinical femoral head specimens were obtained from patients undergoing THA at Qilu Hospital of Shandong University, with informed consent and ethical approval (KYLL-2022(ZM)-1220). Specimens from the osteonecrosis group and non-osteonecrosis control group were derived from patients with comparable baseline characteristics including sex and surgical time. All specimens in the osteonecrosis group were definitively diagnosed as GC-induced ONFH, while control specimens were radiologically intact femoral heads retrieved during surgery for femoral neck fractures. All ethical regulations relevant to human research participants were followed. Detailed patient information for both groups is presented in Table S1. Patient-derived femoral head specimens were sectioned into regular 1-cm³ cubes including both cortical and cancellous bone, harvested from the subchondral region. The samples were fixed in 10% formalin for 48 h, followed by decalcification in 10% ethylenediaminetetraacetic acid (EDTA, 25102-12-9, Sigma-Aldrich) for 4 weeks. Then, paraffin-embedded sections were cut into 6μm-thick slices for subsequent histological and immunofluorescence staining.
AAV
The AAV serotype 9 vectors used in this study were purchased from Shanghai Genechem Co., Ltd. The recombinant AAV9 vectors carried the rat Gdf11 (rGdf11) coding sequence under the control of the CMV promoter (designated AAV9-CMV-rGdf11, abbreviated as AAV-GDF11). The control vector contained a non-targeting empty sequence driven by the same promoter (AAV9-CMV-NC, abbreviated as AAV-NC).
Animals
The experimental animals utilized were male Sprague–Dawley (SD) rats (10 weeks old, 300 ± 30 g), sourced from the Beijing Vital River Experimental Animal Center. These rats were maintained in a controlled environment with a temperature of 20 ± 2°C and humidity levels between 50 and 65%, under a 12-h light/dark cycle, and were given unrestricted access to food and water. A total of 192 rats were allocated across six groups: Sham-operated (n = 18), MP-treated (n = 18), MP + AAV-NC (n = 36), MP + AAV-GDF11 (n = 36), MP + AAV-GDF11 + LY294002 (n = 18), and MP + AAV-GDF11 + L-NAME (n = 18). Group allocation (initial and subgrouping) utilized simple randomization via an Excel-generated number table. To minimize bias, animals were identified by unique numerical codes and personnel involved in animal care, data recording, and outcome assessment were blinded to group assignments whenever feasible. For each group, samples were randomly assigned to histological staining, angiography, or calcein and alizarin red s staining. The order of all the measurements and treatments were randomized to minimize confounding effects. General health was monitored regularly. Since no animals died or were otherwise excluded, the final sample size for analysis was 6 animals per group. The sample size (n = 6 per final group) was based on previous studies54,55 for sufficient statistical power.
Model setup and drug treatment
According to the protocol prepared before the study, GC-induced ONFH models were constructed as the following steps. Daily i.p. injection of lipopolysaccharides (LPS, 20 μg/kg, 93572-42-0, Sigma-Aldrich) was given for the first 3 days, pre-treatment with LPS creates a pro-inflammatory, pro-thrombotic, and hyperlipidemic state that significantly lowers the threshold for high-dose steroids to induce the vascular compromise and cellular damage characteristic of osteonecrosis in the rat femoral head. Then an intramuscular (i.m.) dose of MP (60 mg/kg, Pfizer) was administered each day for the next 4 days. MP was injected into the left and right gluteus muscles alternately. Rats in the AAV-GDF11 group were treated with AAV9 serotype vectors carrying the rat Gdf11 coding sequence under the CMV promoter (AAV9-CMV-Gdf11, 1.0 × 10¹² vg/day, GeneChem). Controls received equivalent doses of non-targeting empty control vectors. PI3K-antagonist group was treated with LY294002 (0.5 mg/kg/day, A10547, Adooq, China) via i.p. injection, eNOS-antagonist group was treated with L-NAME (20 mg/kg/day, HY-18729A, MCE, USA), groups were administered daily for 3 consecutive weeks from the second day after MP injection. The control group received physiological saline. Four weeks after MP administration, animals were euthanized via CO₂.
Micro-CT
To assess the imaging changes, the femoral heads of rats were scanned and analyzed using a high-resolution micro-CT Quantum GX2 (PerkinElmer, Japan). The scanning protocol included an isometric resolution of 15 μm, with X-ray energy settings of 70 kV and 200 μA. The ROI for quantitative analysis was the weight bearing area of the femoral head under the articular cortical bone. 3D image reconstruction was created and morphometric parameters were evaluated, including BV/TV, BMD, Tb.N, Tb.Sp, and Tb.Th.
Angiography
Rats were fixed on the operating table after anesthesia. The skin, muscle, and peritoneum were incised at the abdominal midline to expose the abdominal aorta and arterial catheter indwelling, systemic blood was heparinized and fixed by injecting heparin saline and formalin under appropriate pressure. The proximal end of the abdominal aorta was ligated above the left renal artery, and the inferior vena cava was ligated proximally. Then 20 ml Microfil (MV-112, Flow Tech, Inc., Carver, MA, USA) was injected slowly at a uniform speed using a computer-controlled pneumatic pumps (Harvard PHD 2000) to ensure pressure ≤180 mmHg, and rats were sacrificed during this process. The femoral heads were removed after 12 h storage at 4 °C, and decalcified at 37 °C for 2 months. Then, the femoral head was scanned and reconstructed with the above-mentioned micro-CT, and a 3D model of femoral-head angiography was created.
IHC, and immunofluorescence staining
The femoral heads were fixed in 10% formalin for 48 h, followed by decalcification in 10% ethylenediaminetetraacetic acid (EDTA, 25102-12-9, Sigma-Aldrich) for 4 weeks. Then, paraffin-embedded sections were cut into 6μm-thick slices, the morphological features of the samples were observed using H&E staining, trabecular collapse was defined by the presence of at least one of the following: focal angulation >30°, microfracture, focal compression with >50% trabecular thinning, or fragmentation. Safranin O and fast green staining (G1371, Salorbio, China) was performed to determine changes in proteoglycans. Masson’s trichrome staining (G1340, Salorbio, China) was used to measure the total collagen content. Section images were acquired using a VS120 Research Slide Scanner (Olympus, Japan). IHC staining was conducted to delineate the expression of markers associated with osteogenesis, vascularization, and signaling pathways. Briefly, sections were dewaxed and gradient hydrated to retrieve antigen. Then antibodies including eNOS (27120-1-AP, Proteintech), p-eNOS (PC3447, Abmart), PI3K (T40115, Abmart), p-PI3K (TA3242, Abmart), CD31 (ab182981, Abcam) and corresponding secondary antibodies (ab150077, Abcam) were incubated. The chromogenic reaction was induced by a DAB Kit (ZLI-9018, ZSGB-BIO). Immunofluorescence staining was performed to identify the expression level and localization of certain targets. Primary antibodies like COL1 (#72026, Cell Signaling Technology), VEGFA (ab52917, Abcam), CD31 (ab182981, Abcam), p-PI3K (TA3242, Abmart), p-eNOS (PC3447, Abmart) and corresponding fluorescent secondary antibodies (ab150079 and ab150077, Abcam) were used. The tissue sections were observed with a fluorescence microscope. IHC-staining-positive cells and areas were measured using a light microscope, and counted by two independent observers. For comparative analyses, tissue sections within the same experimental group were employed for colorimetric and fluorescent staining to ensure sample uniformity.
TRAP staining
TRAP staining was performed to detect the changes in proteoglycans with a TRAP staining kit (G1492, SolarBio) according to the manufacture’s recommended procedure.
Safranin O staining
Safranin O staining was performed to detect the changes in proteoglycans with a Safranin O staining kit (G1371, SolarBio) according to the manufacturer’s recommended procedure.
Masson’s trichrome staining
Masson’s trichrome staining was performed to detect the changes in total collagen content with a Masson’s trichrome staining kit (G1340, SolarBio) according to the manufacturer’s recommended procedure.
Double calcein labeling and alizarin red s staining
Rats were injected intraperitoneally with calcein (10 mg/kg, C0875, Sigma-Aldrich) and alizarin red s (20 mg/kg, A5533, Sigma-Aldrich) at 10 and 3 days before euthanasia. Then, undecalcified femoral heads were sectioned after fixation and dehydration. Images were observed by a fluorescence microscope; parameters including inter-label width and MAR were measured. We selected the cortical bone closest to the ROI for measurement.
Cell culture and proliferation assay
HUVECs were obtained from the Cell Bank of the Chinese Academy of Sciences (Shanghai, China). These cells were cultured in Endothelial Cell Medium (#1001, Sciencell, United States) supplemented with 5% fetal bovine serum (FBS, 164210, Gibco), 100 μg/ml streptomycin, and 100 U/ml penicillin (C100C5, New Cell & Molecular Biotech). The cells were maintained in a humidified incubator at 37 °C with 5% CO₂. Cells were treated for 24 h with medium containing MP, or combined with GDF11. Cytotoxicity was assessed using the CCK-8 (C0037, Beyotime, China), MP (HY-B0260, MedChemExpress) environment was established at 100 μM, while GDF11 (120-11, Pepro Tech) was used at a concentration of 1 ng/μl. BMSC were extracted from the tibia and femur of the SD rats and cultured in α-MEM medium (C12571, Gibco). The medium consisted of 10% FBS and 1% penicillin/streptomycin, for osteogenic differentiation, dexamethasone (Y063999, Beyotime, China), vitamin C (ST1434, Beyotime, China), and β-glycerophosphate (G8100, Solarbio) were contained. The cells were cultivated at 37 °C in 5% CO2, and the culture medium was replaced every 2 days to remove non-adherent cells. Third-passage (P3) cells were used for experiments.
Blocking of the PI3K/AKT/eNOS pathway
HUVECs were treated with MP (100 μM) or MP (100 μM) + GDF11 (10 ng/ml) in the presence or absence of the PI3K inhibitor LY294002 (10 μM, A10547, Adooq, China) or eNOS inhibitor L-NAME (100 μM, HY-18729A, MCE, USA). The control was treated with only the media.
Western blotting
Proteins in HUVECs were obtained by RIPA Lysis Buffer (G2002, Servicebio Corporation, Nanjing. China). The BCA Protein Assay Kit (P0012, Beyotime, China) was used to quantify protein concentration. Equal amounts of the extracted proteins were electrophoresed on SDS-PAGE gels and transmitted to a polyvinylidene fluoride membrane (ISEQ00010, Millipore), membrane was blocked (G2052, Servicebio) and incubated with primary antibodies overnight at 4°C, including GAPDH (1:1000, 10494-1-AP, Proteintech), β-tubulin (1:1000, 10094-1-AP, Proteintech), β-actin (1:1000, ab8227, Abcam), GDF11 (MAB19625, Abnova), RUNX2 (1:1000, ab192256, Abcam), CD31 (1:500, GB11063-2, Servicebio), VEGFA (1:500, GB11034B, Servicebio), COL1 (1:500, ab279711, Abcam), AKT (1:500, #9272, Cell Signaling Technology), p-AKT (1:500, #4060, Cell Signaling Technology), PI3K (1:500, T40115, Abmart), p-PI3K (1:500, TA3242, Abmart), eNOS (1:1000, 27120-1-AP, Proteintech) and p-eNOE (1:500, PC3447, Abmart). After incubation with corresponding secondary antibody, the protein bands were captured by enhanced chemiluminescence (MAO186-1, Meilunbio). Protein quantification was performed using ImageJ software (1.4.3.67, National Institutes of Health [NIH], USA).
Real-time quantitative PCR
The total RNA of HUVECs was extracted by TRIzol (15596026CN, ThermoFisher). and cDNA was obtained by reverse transcription using PrimeScript RT Master Mix cDNA Synthesis Kit (FSQ-101, Toyobo), and Real-time PCR was carried out with SYBR Green PCR Master Mix (OPK-201, Toyobo). We calculated the fold change in RNA expression compared to that of the control using the ΔΔCt method.
Cell immunofluorescence staining
Treated HUVECs on coverslips were fixed with 4% paraformaldehyde for 30 min. Then, 0.2% Triton X-100 (P0096, Beyotime, China) was added for cell permeabilization. After incubation with bovine serum albumin (B2064, Sigma-Aldrich) at 37 °C for 1 h, cells were incubated with primary antibody including CD31 (ab182981, Abcam), VEGFA (66828-1-Ig, Proteintech), p-PI3K (TA3242, Abmart), p-eNOS (PC3447, Abmart) and secondary antibody (ab150079 and ab150077, Abcam), then counterstained with DAPI for 15 min. The results were observed using a fluorescence microscope (Zeiss), and fluorescence intensity was assessed using ImageJ software (1.4.3.67, National Institutes of Health [NIH], USA).
RNA sequencing and transcription factors enrichment analysis
HUVECs were stimulated as indicated. After 24 h, cells were harvested by a cell brush and then centrifuged. Then cell precipitates were collected, quick-frozen in liquid nitrogen, and transported on dry ice. RNA sequencing was performed by Genepioneer Biotechnology Co., Ltd. Differentially expressed gene (DEG) analysis was conducted using the DEGSeq method, with a threshold of P < 0.05 and |Fold Change | >2. GO (http://www.geneontology.org/) and KEGG were used to perform enrichment analysis of DEGs to explore their potential role in phenotypic changes. GSEA was used to analyze the enriched biological pathways of the DEGs.
NO Generation
NO generation of HUVECs was characterized by testing the nitrite levels through Griess method. The culture medium was extracted for further NO testing using a NO assay kit after 24 h of treatment (S0021S, Beyotime, China).
Transwell migration assay
Preconditioned HUVECs were plated into the upper chambers of a transwell plate (14341, Corning). Complete culture medium was placed in the lower chamber and used as a chemoattractant. Twenty-four hours later, the membranes were fixed with ethanol and then stained with crystal violet (C0121, Beyotime, China). Then, the membranes were mounted and observed under a light microscope (Leica DMI3000B).
Scratch assay
HUVECs were seeded into 6-well plates and cultured until a monolayer was formed. Two separate wounds were scratched using a 200-μL pipet tip. The cells were gently washed three times with phosphate-buffered saline (PBS) to remove cellular debris and any other non-adherent material. The cells were subsequently washed with medium devoid of serum. At 0, 12, or 24 h after scratching, pictures were taken under the light microscope.
Tube formation
Matrigel (356234, Corning, USA) was applied to 96-well plates 50 μL/well at 4 °C and solidify at 37 °C for 40 min before use. HUVECs were seeded at 1 × 10⁴ cells/well and allowed to adhere for 24 h. Cells were then treated for 24 h with medium containing MP (100 μM), or combined with GDF11 (1 ng/mL). Then, tube formation was observed and the images were obtained. The tube length and branch points were quantified using Image-Pro Plus (IPP) software.
ALP and ARS staining
ALP staining was performed on cells cultured in osteogenic medium for 14 days, BCIP/NBT (P0321S, Beyotime, China) working solution was used to incubate. ARS staining was performed on cells cultured in osteogenic medium for 21 days, using ARS staining solution (G1038, Servicebio). Pictures of well plates and images captured by microscope were preserved as quantitative basis.
Statistics and reproducibility
Cell experiments were performed with at least three independent replicates, and human femoral head explants samples and animal experiments were conducted with at least six independent replicates. Data were presented as mean ± standard error (SE). IBM SPSS Statistics (version 20.0 for Windows, USA) and GraphPad Prism (version 10.1.2 for Windows, USA) were utilized for statistical analyses. Prior to parametric analyses, normality was verified using the Shapiro-Wilk test, and homogeneity of variances was assessed via Levene’s test. Group comparisons were conducted using unpaired t-tests or ANOVA as appropriate. Welch’s correction was applied when variances were unequal (Levene’s test p < 0.05). All tests were two-tailed, with p < 0.05 as statistically significant.
Ethics statement
All animal experiments described in this study were performed in accordance with institutional guidelines and approved by the Ethical Management Committee of Qilu Hospital, Shandong University (KYLL-2022(ZM)-1220). Ethical regulations concerning work with human participants were strictly followed, and the study received approval from the Ethical Management Committee of Qilu Hospital, Shandong University (KYLL-2022(ZM)-1220).
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
References
Kerachian, M. A., Séguin, C. & Harvey, E. J. Glucocorticoids in osteonecrosis of the femoral head: a new understanding of the mechanisms of action. J. Steroid Biochem Mol. Biol. 114, 121–128 (2009).
Chang, C., Greenspan, A. & Gershwin, M. E. The pathogenesis, diagnosis and clinical manifestations of steroid-induced osteonecrosis. J. Autoimmun. 110, 102460 (2020).
Mont, M. A., Salem, H. S., Piuzzi, N. S., Goodman, S. B. & Jones, L. C. Nontraumatic osteonecrosis of the femoral head: where do we stand today? A 5-year update. J. Bone Jt. Surg. Am. 102, 1084–1099 (2020).
Petrigliano, F. A. & Lieberman, J. R. Osteonecrosis of the hip: novel approaches to evaluation and treatment. Clin. Orthop. Relat. Res. 465, 53–62 (2007).
Kusumbe, A. P., Ramasamy, S. K. & Adams, R. H. Coupling of angiogenesis and osteogenesis by a specific vessel subtype in bone. Nature 507, 323–328 (2014).
Sojo, K., Sawaki, Y., Hattori, H., Mizutani, H. & Ueda, M. Immunohistochemical study of vascular endothelial growth factor (VEGF) and bone morphogenetic protein-2, -4 (BMP-2, -4) on lengthened rat femurs. J. Craniomaxillofac Surg. 33, 238–245 (2005).
Zuo, W., Guo, W.-S., Yu, H.-C., Liu, P. & Zhang, Q.-D. Role of junction-mediating and regulatory protein in the pathogenesis of glucocorticoid-induced endothelial cell lesions. Orthop. Surg. 12, 964–973 (2020).
Thapa, D. et al. Novel hexahydrocannabinol analogs as potential anti-cancer agents inhibit cell proliferation and tumor angiogenesis. Eur. J. Pharm. 650, 64–71 (2011).
Zhou, X. et al. Spatiotemporal regulation of angiogenesis/osteogenesis emulating natural bone healing cascade for vascularized bone formation. J. Nanobiotechnol. 19, 420 (2021).
Nan, K. et al. Exosomes from miRNA-378-modified adipose-derived stem cells prevent glucocorticoid-induced osteonecrosis of the femoral head by enhancing angiogenesis and osteogenesis via targeting miR-378 negatively regulated suppressor of fused (Sufu). Stem Cell Res. Ther. 12, 331 (2021).
Shen, J. et al. EGFL6 regulates angiogenesis and osteogenesis in distraction osteogenesis via Wnt/β-catenin signaling. Stem Cell Res. Ther. 12, 415 (2021).
Zhang, X. et al. Bevacizumab promotes active biological behaviors of human umbilical vein endothelial cells by activating TGFβ1 pathways via off-VEGF signaling. Cancer Biol. Med. 17, 418–432 (2020).
Hasan, Q., Tan, S. T., Gush, J., Peters, S. G. & Davis, P. F. Steroid therapy of a proliferating hemangioma: histochemical and molecular changes. Pediatrics 105, 117–120 (2000).
Logie, J. J. et al. Glucocorticoid-mediated inhibition of angiogenic changes in human endothelial cells is not caused by reductions in cell proliferation or migration. PLoS ONE 5, e14476 (2010).
Yano, A., Fujii, Y., Iwai, A., Kageyama, Y. & Kihara, K. Glucocorticoids suppress tumor angiogenesis and in vivo growth of prostate cancer cells. Clin. Cancer Res. 12, 3003–3009 (2006).
Hadoke, P. W. F., Iqbal, J. & Walker, B. R. Therapeutic manipulation of glucocorticoid metabolism in cardiovascular disease. Br. J. Pharm. 156, 689–712 (2009).
Borsky, P. et al. Evaluation of potential aging biomarkers in healthy individuals: telomerase, AGEs, GDF11/15, sirtuin 1, NAD+, NLRP3, DNA/RNA damage, and klotho. Biogerontology 24, 937–955 (2023).
Katsimpardi, L. et al. Vascular and neurogenic rejuvenation of the aging mouse brain by young systemic factors. Science 344, 630–634 (2014).
Egerman, M. A. et al. GDF11 increases with age and inhibits skeletal muscle regeneration. Cell Metab. 22, 164–174 (2015).
McPherron, A. C., Lawler, A. M. & Lee, S. J. Regulation of anterior/posterior patterning of the axial skeleton by growth/differentiation factor 11. Nat. Genet 22, 260–264 (1999).
Kim, J. et al. GDF11 controls the timing of progenitor cell competence in developing retina. Science 308, 1927–1930 (2005).
Walker, R. G. et al. Structural basis for potency differences between GDF8 and GDF11. BMC Biol. 15, 19 (2017).
Sako, D. et al. Characterization of the ligand binding functionality of the extracellular domain of activin receptor type IIb. J. Biol. Chem. 285, 21037–21048 (2010).
Zhang, Y. et al. GDF11 promotes wound healing in diabetic mice via stimulating HIF-1ɑ-VEGF/SDF-1ɑ-mediated endothelial progenitor cell mobilization and neovascularization. Acta Pharmacol. Sin. 44, 999–1013 (2023).
Kato, T. & Lee, R. T. Gdf-11 as a potential cardiac pro-angiogenic factor. JACC Basic Transl. Sci. 8, 636–637 (2023).
Yu, X. et al. Growth Differentiation Factor 11 promotes abnormal proliferation and angiogenesis of pulmonary artery endothelial cells. Hypertension 71, 729–741 (2018).
Du, G.-Q. et al. Targeted myocardial delivery of GDF11 gene rejuvenates the aged mouse heart and enhances myocardial regeneration after ischemia-reperfusion injury. Basic Res. Cardiol. 112, 7 (2017).
Zhang, J. et al. GDF11 improves angiogenic function of EPCs in Diabetic Limb Ischemia. Diabetes 67, 2084–2095 (2018).
Zeng, W., Yan, Y., Zhang, F., Zhang, C. & Liang, W. Chrysin promotes osteogenic differentiation via ERK/MAPK activation. Protein Cell 4, 539–547 (2013).
Liu, Z. et al. SIRT1 activation promotes bone repair by enhancing the coupling of type H vessel formation and osteogenesis. Cell Prolif. 57, e13596 (2024).
Liu, S. et al. Tissue engineering of JAK inhibitor-loaded hierarchically biomimetic nanostructural scaffold targeting cellular senescence for aged bone defect repair and bone remolding. Adv. Health. Mater. 12, e2301798 (2023).
Huang, X. et al. miR-122-5p targets GREM2 to protect against glucocorticoid-induced endothelial damage through the BMP signaling pathway. Mol. Cell Endocrinol. 544, 111541 (2022).
Cao, F. et al. Ginkgo biloba L. extract prevents steroid-induced necrosis of the femoral head by rescuing apoptosis and dysfunction in vascular endothelial cells via the PI3K/AKT/eNOS pathway. J. Ethnopharmacol. 296, 115476 (2022).
Hoxhaj, G. & Manning, B. D. The PI3K-AKT network at the interface of oncogenic signalling and cancer metabolism. Nat. Rev. Cancer 20, 74–88 (2020).
Lin, F. et al. Hydrogen sulfide protects against high glucose-induced human umbilical vein endothelial cell injury through activating PI3K/Akt/eNOS pathway. Drug Des. Dev. Ther. 14, 621–633 (2020).
Qin, Q. et al. Neurovascular coupling in bone regeneration. Exp. Mol. Med. 54, 1844–1849 (2022).
Tomaru, Y. et al. Mid-term results of concentrated autologous bone marrow aspirate transplantation for corticosteroid-associated osteonecrosis of the femoral head in systemic lupus erythematosus. Int. Orthop. 42, 1623–1630 (2018).
Liu, X. et al. Osteoclasts protect bone blood vessels against senescence through the angiogenin/plexin-B2 axis. Nat. Commun. 12, 1832 (2021).
Zuo, R. et al. Exosomes derived from human CD34+ stem cells transfected with miR-26a prevent glucocorticoid-induced osteonecrosis of the femoral head by promoting angiogenesis and osteogenesis. Stem Cell Res. Ther. 10, 321 (2019).
Boss, J. H. & Misselevich, I. Osteonecrosis of the femoral head of laboratory animals: the lessons learned from a comparative study of osteonecrosis in man and experimental animals. Vet. Pathol. 40, 345–354 (2003).
Zheng, L.-Z. et al. Steroid-associated osteonecrosis animal model in rats. J. Orthop. Transl. 13, 13–24 (2018).
Zhang, P. et al. GDF11 protects against mitochondrial-dysfunction-dependent NLRP3 inflammasome activation to attenuate osteoarthritis. J. Adv. Res. 73, 501–515 (2025).
Tao, P. et al. Growth differentiation factor 11 alleviates oxidative stress-induced senescence of endothelial progenitor cells via activating autophagy. Stem Cell Res. Ther. 15, 370 (2024).
Suh, J. et al. GDF11 promotes osteogenesis as opposed to MSTN, and follistatin, a MSTN/GDF11 inhibitor, increases muscle mass but weakens bone. Proc. Natl. Acad. Sci. USA 117, 4910–4920 (2020).
Egerman, M. A. & Glass, D. J. The role of GDF11 in aging and skeletal muscle, cardiac and bone homeostasis. Crit. Rev. Biochem. Mol. Biol. 54, 174–183 (2019).
Zhang, W. et al. GDF11 rejuvenates cerebrovascular structure and function in an animal model of Alzheimer’s disease. J. Alzheimers Dis. 62, 807–819 (2018).
Ma, J. et al. Transcutaneous auricular vagus nerve stimulation regulates expression of growth differentiation factor 11 and activin-like kinase 5 in cerebral ischemia/reperfusion rats. J. Neurol. Sci. 369, 27–35 (2016).
Wang, L. et al. Growth differentiation factor 11 ameliorates experimental colitis by inhibiting NLRP3 inflammasome activation. Am. J. Physiol. Gastrointest. Liver Physiol. 315, G909–G920 (2018).
Wang, D.-X. et al. GDF11 slows excitatory neuronal senescence and brain ageing by repressing p21. Nat. Commun. 14, 7476 (2023).
Sinha, M. et al. Restoring systemic GDF11 levels reverses age-related dysfunction in mouse skeletal muscle. Science 344, 649–652 (2014).
Ma, Y.-S. et al. KDM5A silencing transcriptionally suppresses the FXYD3-PI3K/AKT axis to inhibit angiogenesis in hepatocellular cancer via miR-433 up-regulation. J. Cell Mol. Med. 25, 4040–4052 (2021).
Zhao, Y. et al. GDF11 enhances therapeutic efficacy of mesenchymal stem cells for myocardial infarction via YME1L-mediated OPA1 processing. Stem Cells Transl. Med. 9, 1257–1271 (2020).
Finkenzeller, G., Stark, G. B. & Strassburg, S. Growth differentiation factor 11 supports migration and sprouting of endothelial progenitor cells. J. Surg. Res. 198, 50–56 (2015).
Serdar, C. C., Cihan, M., Yücel, D. & Serdar, M. A. Sample size, power and effect size revisited: simplified and practical approaches in pre-clinical, clinical and laboratory studies. Biochem. Med. 31, 010502 (2021).
Festing, M. F. On determining sample size in experiments involving laboratory animals. Lab. Anim. 52, 341–350 (2018).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (82302699, 82272548), China Postdoctoral Science Foundation (2023M732099), and Shandong Provincial Natural Science Foundation (ZR2023QH303).
Author information
Authors and Affiliations
Contributions
L. Cheng, P. Liu, and H. Sun contributed to the study conception and design. In vivo experiments were performed by Z. Liu and K. Liu. In vitro experiments were performed by Z. Liu and L. Yu. Data collection and analysis were performed by Y. Zhang and H. Zhai. X, Li maintained the rats. The first draft of the manuscript was written by X. Xu, Q. Lu, S. Li, J. Yang and Z. Li. M. Si edited the manuscript and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Biology thanks the anonymous reviewers for their contribution to the peer review of this work. Primary Handling Editors: Martina Rauner and Mengtan Xing.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Liu, Z., Yu, L., Zhang, Y. et al. GDF11 alleviates glucocorticoid-induced osteonecrosis of the femoral head by regulating angiogenesis via the PI3K-AKT-eNOS pathway. Commun Biol 8, 1682 (2025). https://doi.org/10.1038/s42003-025-09078-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s42003-025-09078-5