Abstract
The bile acid isoallolithocholic acid (isoalloLCA) has been observed to be reduced in patients with inflammatory bowel diseases (IBD). However, its role in the pathogenesis of pediatric IBD remains poorly understood. Here we show evidence that isoalloLCA treatment decreases lipopolysaccharide (LPS)-induced tumor necrosis factor (TNF) production in blood cells from children diagnosed with IBD. In experimental models of IBD, isoalloLCA alleviates acute intestinal inflammation caused by LPS or dextran sulfate sodium (DSS) and shows therapeutic efficacy in a chronic colitis model using Il10 knockout (Il10−/−) mice. Within the mucosa of these murine models, isoalloLCA enhances the expression of the regulatory T cell transcription factor Forkhead box P3 (Foxp3), while simultaneously inhibiting ETS2, a critical regulator of inflammatory macrophages in IBD. In bone marrow-derived macrophages (BMDMs), isoalloLCA mitigates LPS-induced inflammation, potentially through the enhancement of mitochondrial reactive oxygen species (mitoROS) production and inhibition of the ETS2-HIF1A/PFKFB3 signaling pathway. Simultaneously, isoalloLCA metabolically reprograms macrophages by enhancing oxidative phosphorylation (OXPHOS) that is linked to anti-inflammatory effects. Our research indicates that metabolic modulation of macrophages amplifies the anti-inflammatory properties of isoalloLCA, thereby revealing a promising therapeutic avenue for addressing pediatric IBD.

Similar content being viewed by others
Introduction
The increasing prevalence of autoimmune and inflammatory disorders, such as inflammatory bowel disease (IBD), affects ~5% of the global population and poses a health threat. IBD has become a pressing global health concern1. The incidence of IBD in developing and newly industrialized nations is relatively low, at 1 to 10 cases per 100,000 individuals, but is rising. In contrast, Western countries have a higher stable incidence ranging from 25 to 40 cases per 100,000 individuals2. IBD primarily affects individuals under 40 and has a low mortality rate; however, its incidence in Western nations is growing at an annual rate of about 5%. By 2028, it is projected that around 1% of these populations will be affected3,4. While noticeable progress has been made in IBD treatments, these options are still suboptimal and require innovative solutions. Mononuclear phagocytes, including monocytes, macrophages, and dendritic cells, are among the most abundant leukocytes in the gastrointestinal tract5. Notably, macrophages form a complex network crucial for intestinal homeostasis. Dysregulated macrophage activity plays an important role in IBD pathogenesis6. Given their essential role in orchestrating inflammation resolution and facilitating tissue repair, macrophages are regarded as highly promising therapeutic targets within the context of IBD7. Consequently, there is strong interest in understanding the intricate immunobiology of macrophages and the subtle changes in their functions during disease.
Bile acids, which are derived from the oxidative transformation of cholesterol, play a crucial role in modulating nutrient absorption8. Similar to many other steroids originating from cholesterol, bile acids have been identified as key regulatory molecules within the immune system9,10,11,12,13,14. The bacterial bile acid metabolite isoallolithocholic acid (isoalloLCA) has been shown to intricately modulate adaptive immunity by enhancing the differentiation of anti-inflammatory regulatory T cells (Treg cells)11,12. IsoalloLCA plays a pivotal role in promoting the differentiation of naive T cells into Treg cells by enhancing acetylation at the promoter region of Forkhead box protein P3 (Foxp3)11,12. Nevertheless, it remains uncertain whether isoalloLCA exerts any influence on macrophages. IsoalloLCA is synthesized by gut microbiota, and a state of dysbiosis may catalyze its depletion12,15. The Odoribacteraceae family plays an indispensable role in the intricate metabolism of bile acids within the gastrointestinal tract, thereby facilitating the production of isoalloLCA12,15. The administration of Odoribacteraceae augmented the induction of immunosuppressive Kupffer cells within murine livers, concomitantly accompanied by elevated levels of isoalloLCA16. This observation suggests that isoalloLCA may partially orchestrate its anti-inflammatory effects through intricate reprogramming of macrophages.
In the present study, we demonstrate that levels of isoalloLCA are evidently reduced in children suffering from inflammatory bowel disease (IBD). The administration of isoalloLCA attenuates lipopolysaccharide (LPS)-induced tumor necrosis factor (TNF) production in whole blood and peripheral blood mononuclear cells (PBMCs) obtained from pediatric IBD patients. Treatment with isoalloLCA alleviates both acute and chronic intestinal inflammation in murine models potentially through downregulating a key regulator of inflammatory macrophages, ETS2, and simultaneously enhancing the expression of Foxp3. IsoalloLCA induces metabolic reprogramming in bone marrow-derived macrophage (BMDM) cells by promoting oxidative phosphorylation (OXPHOS), and potentially disrupting the ETS2-HIF1A/PFKFB3 signaling pathway. Our findings indicate that modulation of macrophage metabolism enhances the anti-inflammatory effects of isoalloLCA, underscoring its potential as a therapeutic agent for IBD.
Results
Reduced isoalloLCA is linked to the progression of intestinal inflammation
We initially investigated the fluctuations in isoalloLCA levels throughout human development, utilizing data sourced from the Metabolomics Workbench (https://www.metabolomicsworkbench.org, Project ID PR001435). As illustrated in Fig. 1, fecal concentrations of isoalloLCA exhibited a progressive increase from birth through to the age of two years (Fig. 1a). In pediatric patients with ulcerative colitis (UC)17, the concentrations of isoalloLCA in stool samples were diminished during active disease states compared to those observed in periods of remission (Fig. 1b). In addition, the levels of fecal isoalloLCA exhibited a marked reduction in individuals with moderate to severe disease when compared to those presenting with mild disease (Fig. 1b). In alignment with prior research12, the levels of fecal isoalloLCA were reduced in both adult patients suffering from Crohn’s disease (CD) and those afflicted with UC, when juxtaposed against their healthy counterparts (Fig. 1c). Given that bacteria predominantly synthesize isoalloLCA12,15, the analyzed data18 revealed the abundance of Parabacteroides merdae, Parabacteroides distasonis, and Parabacteroides gordonii in stool was much lower in both CD and UC than their controls (Fig. 1d). Meanwhile, we meticulously examined lymphocyte-specific transcription factors, namely TBX21 (Th1), FOXP3 (Treg), RORγt (Th17), CD68+ macrophages, and ETS2, the regulator of inflammatory macrophages in IBD19, through the application of immunohistochemical (IHC) staining. As illustrated in Fig. 2, there was a notable reduction in FOXP3-positive cells within the inflamed mucosal layers of both UC and CD when compared to their corresponding uninflamed ones (Fig. 2a, b). While the populations of TBX21−, RORγt−, CD68−, and ETS2− positive cells exhibited a notable increase in inflamed mucosa of both UC and CD when compared to uninflamed subjects, (Fig. 2a, b).
a The stool isoalloLCA abundance was increased during infants growing (samples number: Birth, 0 month, n = 29; 2 months, n = 177; 6 months = 194; 10 months, n = 214; 15 months, n = 214; 18 months, n = 218; 24 months, n = 212). b IsoalloLCA was negative with pediatric ulcerative colitis (UC) severity (F1, collection Week 0, mild, n = 12; F2, collection Week 0, moderate/severe, n = 24; F3, collection Week 4, inactive, n = 14; F4, collection Week 4, active, n = 11). c Stool isoalloLCA reduced in adult UC and Crohn’s disease (CD) patients (controls samples, n = 56; CD samples, n = 88; UC samples, n = 76). d Bacteria Parabacteroides abundance in feces was reduced in adult UC and CD subjects (controls samples, n = 56; CD samples, n = 88; UC samples, n = 76). Zero values are plotted toward the y-axis minima and enumerated per-group as in percent. Representative data (a–d) expressed as the mean ± standard error (SEM).
a Representative immunohistochemistry (IHC) images of FOXP3 (Treg), RORγt (Th17), TBX21 (Th1), ETS2 (macrophage), and CD68+ macrophages in mucosal layers of pediatric patients with CD and UC. b Quantification of IHC staining (each group, n = 5) in (a). Representative data shown in (b) expressed as the mean ± standard error (SEM). Unpaired two-tailed Student’s t test with or without Welch’s correction analysis for (b). Statistical significance: *p < 0.05, ** p < 0.01, ***p < 0.001, **** p < 0.0001. CD Crohn’s disease, UC ulcerative colitis.
IsoalloLCA attenuates LPS-induced inflammation in the blood of pediatric patients with IBD
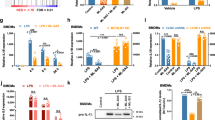
To assess the potential of isoalloLCA in mitigating inflammation among patients with IBD, a series of assays were meticulously conducted utilizing both whole blood and peripheral blood mononuclear cells (PBMCs), as depicted in Fig. 3. Whole blood samples from pediatric IBD patients (CD, n = 5; UC, n = 2) onset, and healthy controls (5 samples) were stimulated with lipopolysaccharide (LPS, 2 μg/ml), alone or together with isoalloLCA (20 μM) for 24 hours. Subsequently, contents of tumor necrosis factor alpha (TNF-α) in blood were determined using enzyme-linked immunosorbent assay (ELISA). As shown in Fig. 3b, the whole-blood assay revealed that LPS administration precipitated a pronounced upregulation of TNF-α production when juxtaposed against non-stimulated counterparts. Notably, exogenous isoalloLCA attenuated LPS-induced TNF-α synthesis in the blood of pediatric IBD patients with disease onset (Fig. 3b). In parallel assessments involving PBMCs, isolated PBMCs derived from pediatric CD patients underwent stimulation with LPS alone or alongside isoalloLCA (20 μM) for an equivalent period of 24 hours (Fig. 3c). The ELISA results obtained from supernatants demonstrated that isoalloLCA effectively suppressed TNF-α production synthesized by PBMCs (Fig. 3c). Furthermore, quantitative reverse transcription polymerase chain reaction (qRT-PCR) analysis elucidated that isoalloLCA inhibited the expression of key inflammatory genes such as IL23A, NOS2, ITGAM, TNFA and IL17A, but ITGAM or TNFA did not reach significant levels (Fig. 3d). In alignment with findings observed in naive CD4+ T cells11, we observed that isoalloLCA enhanced mitochondrial respiratory metabolism within PBMCs sourced from CD patients, which featured with increasing oxygen consumption rates measurement (OCR) (Supplementary Fig. 1a) and decreasing extracellular acidification rates measurement (ECAR) (Supplementary Fig. 1b). In order to elucidate the potential mechanisms underlying these anti-inflammatory effects of isoalloLCA, we conducted a comprehensive analysis of the available dataset (GSE163349)12. Differential expression analysis revealed that isoalloLCA predominantly upregulated the regulatory T cell signature gene Foxp3, while concurrently downregulating several inflammatory genes, including Nos2, Ccl3, and Ccl4 (Supplementary Fig. 2a). Furthermore, enrichment analysis through Gene Ontology (GO) indicated that these differentially expressed genes affected by isoalloLCA were primarily involved in pathways associated with the negative regulation of immune system responses (Supplementary Fig. 2b).
a Schematic of the isoalloLCA-treated whole blood and peripheral blood mononuclear cells (PBMCs) from pediatric IBD patients and their match healthy controls. For whole blood assay, lipopolysaccharide (LPS) was added at a concentration of 2 μg/mL either individually or in conjunction with isoalloLCA (20 μM), and incubated for a period of 24 hours under conditions of temperature maintained at constant level of 37 °C along with an atmosphere containing 5% CO2. After completion of the incubation process, the supernatants were subjected to performed with enzyme-linked immunosorbent assay (ELISA) of tumor necrosis factor alpha (TNF-α). For analysis in PBMCs, 5 × 10 6 PBMCs each well were cultured in RPMI 1640 medium with or without 2 μg/ml LPS or/and 20 μM isoalloLCA for 24 hours. The supernatant was collected for ELSIA and the cells were used to extract RNA for quantitative real-time polymerase chain reaction (qRT-PCR). b Enzyme-linked immunosorbent assay (ELISA) assay for tumor necrosis factor alpha (TNF-α) expression in whole blood from healthy controls (n = 5) and IBD patients with onset (CD, n = 5, UC, n = 2). b ELISA assay for TNF-αsupernatants of peripheral blood mononuclear cells (PBMCs) from Crohn’s disease (CD) patients (n = 4). c Quantitative real-time polymerase chain reaction (qRT-PCR) analysis was performed on the PBMCs and determined the expression of interleukin 23 subunit alpha (IL23A), nitric oxide synthase 2 (NOS2), integrin subunit alpha M (ITGAM), tumor necrosis factor (TNFA), and interleukin 17 A (IL17A). The genes were calculated against hypoxanthine-phosphoribosyltransferase 1 (HPRT1). Representative data (b–d) expressed as the mean ± standard error (SEM). Ordinary one-way ANOVA analysis for (b–d). ns, not significant (p ≥ 0.05), * p < 0.05, ** p < 0.01, *** p < 0.001, **** p < 0.0001.
IsoalloLCA ameliorates intestinal inflammation in mice induced by DSS or LPS
To assess effect of isoalloLCA on colitis, an acute colitis mouse model was established using 3% DSS, as outlined in Fig. 4a. DSS-induced body weight loss and shortened colonic tissue were ameliorated by isoalloLCA treatment (Fig. 4b, c). Mice subjected to DSS exhibited an increased pathological scores in colon and was attenuated by given isoalloLCA (Fig. 4d). The immunochemistry (IHC) staining revealed that isoalloLCA treatment reduced the DSS-induced CD45+ and CD68+ cells infiltration, but reduced the ki-67-positive cells in mucosa (Fig. 4e). Additionally, to investigate the anti-inflammatory effects of isoalloLCA on colonic tissue damage, quantitative Real-time PCR (qRT-PCR), western blot (WB) and immunochemistry (IHC) analysis were conducted. qRT-PCR analysis showed that isoalloLCA treatment reduced the elevated mRNA levels of proinflammatory genes like Il1b, Tnf, and Il17 in colon tissues, while increasing the anti-inflammatory genes Il10. However, these changes were not statistically significant (Supplementary Fig. 3). Interestingly, DSS induced the dual oxidase 2 (Duox2) involved in hydrogen peroxide generation, which was reversed by isoalloLCA (Supplementary Fig. 3). IsoalloLCA slightly reduced mRNA levels of intestinal stem cell markers like Lgr5 (Supplementary Fig. 3) and inhibited the growth of murine intestinal organoids. (Supplementary Fig. 4a, b). Moreover, isoalloLCA was found to enhance Foxp3 gene expression (not statistically significant) while decreasing transcription factor genes such as Ets2, RORγt (Rorc), and Tbx21 (Fig. 5a). In line with qRT-PCR findings, WB analyses showed that isoalloLCA treatment increased FOXP3 protein levels while reducing the inflammatory macrophage marker ETS2 (Fig. 5b–d). IHC assays revealed an increase in FOXP3-positive cells in DSS isoalloLCA mice compared to DSS mice (Fig. 5e). Notably, isoalloLCA did not affect the protein levels of RORγt, TBX21, phosphorylated AMPK, and phosphorylated mTOR in the colonic mucosa during DSS exposure (Fig. 5b, e).
a Schematic of isoalloLCA treated the DSS murine model. The colitis was induced with 3% DSS for 7 days and mice were a dose of isoallo-LCA (5 mg/kg, body weight, intraperitoneal injection, i.p) or equal volumes of DMSO every day. Four groups: DMSO control (female, n = 5; male, n = 5), isoallo-LCA mice (female, n = 4; male, n = 6), DSS DMSO mice (female, n = 5; male, n = 5), and DSS isoalloLCA (female, n = 5; male, n = 5). b The alterations of body weight in these mice with treatments above. c Quantification of the colons’ length among these mice. d Representative images of hematoxylin and eosin (H&E) staining and Alcian Blue/Periodic Acid Schiff (AB-PAS) staining in the colons of treated mice. Quantification of historical scores and goblet cells number (each group, n = 4). e Representative images of immunochemistry stain (IHC) stain for CD45, CD68, and Ki67 in colons of treated mice. Qualification of CD45, CD68, and Ki67 IHC (each group, n = 4). Representative data shown in (b–e) expressed as the mean ± standard error (SEM). Ordinary One-way ANOVA analysis for (c–e). ns, not significant (p ≥ 0.05), * p < 0.05, ** p < 0.01, *** p < 0.001, **** p < 0.0001.
a The quantitative real-time PCR (qRT-PCR) analyses the changes of Foxp3, Ets2, Rorc, and Tbx21 in the colonic mucosa of DMSO control, isoalloLCA, DSS DMSO, and DSS isoalloLCA mice (each group, n = 4–6). The genes were calculated against Actb. b Western blot (WB) analysis for FOXP3, RORγt, phosphorylated mTOR (p-mTOR), TBX21, phosphorylated AMPK (p-AMPK), and ETS2 in colonic mucosa of DMSO control, isoalloLCA, DSS DMSO, and DSS isoalloLCA mice (each group, n = 3). c Quantification of proteins levels in (b). d Schematic for potential targets of isoalloLCA. e Representative IHC images of FOXP3 (Treg) in DMSO control, isoalloLCA mice, DSS DMSO mice, and DSS isoalloLCA. Quantification of FOXP3 immunohistochemistry (IHC) staining (each group, n = 4). Representative data shown in (a, c, and e) expressed as the mean ± standard error (SEM). Ordinary One-way ANOVA analysis for (a, c, and e). The proteins levels were calculated against β-actin. ns, not significant (p ≥ 0.05), * p < 0.05, ** p < 0.01, *** p < 0.001, **** p < 0.0001.
We next to evaluate the impact of isoalloLCA on LPS-induced inflammatory responses within the small intestine using mice that were subjected to intraperitoneal injections of LPS at a dosage of 5 mg/kg, as delineated in Fig. 6a. Twenty hours after LPS administration, treatment with isoalloLCA reduced LPS-induced mucosal inflammation and injury in the small intestines (Fig. 6b). Consistent with findings in DSS-treated mice, qRT-PCR analysis showed that isoalloLCA treatment reduced mRNA levels of proinflammatory genes such as Il1b, Il6, Il23a, Tnf, Il17, and Ets2 in small intestinal tissues while increasing anti-inflammatory genes Il10 and Foxp3. Neither were these changes statistically significant (Supplementary Fig. 5). We subsequently isolated crypts from the small bowel of mice and subjected them to stimulation with LPS, both in the presence and absence of isoalloLCA treatments. Remarkably, qRT-PCR analysis revealed that isoalloLCA exerted an inhibitory effect on the expression of pro-inflammatory genes, including Il1b, Tnf, Infg, Il6, Il15, Il22, and Il23a, as well as chemokine genes such as Ccl2 and Ccl28, alongside pivotal genes involved in the Tlr4 signaling pathway (Supplementary Fig. 6). IHC analyses revealed an increase in FOXP3-positive cells in the proximal small bowel of isoalloLCA-treated mice compared to DMSO controls, both untreated and after LPS injection (Fig. 6c). WB analysis confirmed that isoalloLCA elevated FOXP3 protein levels in the small intestinal mucosa (Fig. 6d, e). The WB analysis further demonstrated that the administration of isoalloLCA resulted in a decrease in the protein levels of ETS2 and inhibited the phosphorylation of extracellular signal-regulated kinase 1/2 (ERK1/2) (Fig. 6d, e). Notably, isoalloLCA exhibited no discernible effects on TBX21, RORγt proteins, or phosphorylation of AMPK and mTOR (Fig. 6d, e).
a Schematic of isoalloLCA treated the LPS-induced intestinal inflammation in mice. A dose of isoallo-LCA (5 mg/kg, body weight, intraperitoneal injection, i.p) prior 24-hour to lipopolysaccharide (LPS) (intraperitoneally, i.p.; 5 mg/kg) treated mice, another isoalloLCA was given at the same time of LPS-treatment. The mice were sacrificed after 20-hour LPS-given. Four groups: DMSO control, isoallo-LCA mice, LPS DMSO mice, and LPS isoalloLCA (each group, female, n = 3; male, n = 3). b Quantification of pathological scores and representative images of hematoxylin and eosin (H&E) staining for proximal small intestine from DMSO control, isoallo-LCA mice, LPS DMSO mice, and LPS isoalloLCA mice (each group, n = 4). c Quantification of FOXP3 immunohistochemistry (IHC) staining (each group, n = 4) and representative IHC) images of FOXP3 in proximal small intestine from DMSO control, isoallo-LCA mice, LPS DMSO mice, and LPS isoalloLCA mice. d Western blot (WB) analysis for FOXP3, RORγt, ETS2, phosphorylated mTOR (p-mTOR), TBX21, phosphorylated AMPK (p-AMPK), and phosphorylated ERK1/2 in proximal small intestine from DMSO control, isoallo-LCA mice, LPS DMSO mice, and LPS isoalloLCA mice. (each group, n = 3). e Quantification of proteins levels in panel (d). Representative data shown in (b, c, and e) expressed as the mean ± standard error (SEM). Ordinary one-way ANOVA analysis for (b, c, and e). The proteins levels were calculated against β-actin. ns, not significant (p ≥ 0.05), *p < 0.05, **p < 0.01, ****p < 0.0001.
IsoalloLCA alleviates enterocolitis in Il10-deficient mice
To evaluate the impact of isoalloLCA on chronic enterocolitis, a murine model was meticulously established utilizing Il10 knockout (Il10−/−) mice, as delineated in Fig. 7a. IsoalloLCA-treated Il10−/− mice exhibited much lower pathological scores in the colon compared to their counterparts that did not receive isoalloLCA (Fig. 7b). IHC analysis revealed that administration of isoalloLCA markedly diminished the infiltration of CD68+ macrophages within the submucosa and mucosa of Il10 −/− mice (Fig. 7c). WB analysis initially confirmed the completely knockout of the Il10 gene in these mice (Fig. 7d, e). In alignment with observations made during acute intestinal inflammation, isoalloLCA effectively mitigated chronic enterocolitis by reducing levels of EST2, while simultaneously elevating the FOXP3 levels in Il10−/− mice (Fig. 7d, e). In contrary to findings in acute inflammation mice models, the levels of proteins TBX21and RORγt increased during the isaolloLCA given 4 weeks (Fig. 7d, e). Furthermore, we also observed that isoalloLCA administration enhanced the expression of MARCO, a scavenger receptor for damage-associated molecular patterns20, and GATA2/3, which is essential to development of T helper type 2 (Th2) cells21 (Fig. 7d, e). In Il10−/− mice, isoalloLCA neither altered the AMPK phosphorylation or mTOR phosphorylation (Fig. 7d, e).
a Schematic of isoalloLCA treated the chronic enterocolitis in Il10 knockout (Il10−/−) mice. A dose of isoallo-LCA (5 mg/kg, body weight, intraperitoneal injection, i.p) was given to wild-type (Wt) or Il10−/− mice 3-time weekly and lasted 4 weeks, DMSO was severed as controls. Four groups: DMSO Wt, isoallo-LCA Wt, DMSO Il10−/−, and isoallo-LCA Il10−/− (each group, n = 6–10). b Quantification of pathological scores and representative images of hematoxylin and eosin (H&E) staining for colons from DMSO Wt, isoallo-LCA Wt, DMSO Il10−/−, and isoallo-LCA Il10−/− mice (each group, n = 4). c IsoalloLCA alleviates macrophages infiltrations in Il10−/− mice. Representative immunohistochemistry (IHC) images of CD68+ macrophages in colons from DMSO Wt, isoallo-LCA Wt, DMSO Il10−/−, and isoallo-LCA Il10−/− (n = 4). d Western blot (WB) analysis for IL10, ETS2, MACRO, GATA2/3, FOXP3, RORγt, phosphorylated mTOR (p-mTOR), TBX21, and phosphorylated AMPK (p-AMPK) in colonic mucosa of DMSO Wt, isoallo-LCA Wt, DMSO Il10−/−, and isoallo-LCA Il10−/− mice (each group, n = 3). e Quantification of proteins levels in (d). The proteins levels were calculated against β-actin. Representative data shown in (b, c, and e) expressed as the mean ± standard error (SEM). Ordinary one-way ANOVA analysis for (b, c, and e). ns, not significant (p ≥ 0.05), * p < 0.05, ** p < 0.01, *** p < 0.001, **** p < 0.0001.
IsoalloLCA enhances aerobic respiration in macrophages
In a concerted effort to elucidate the anti-inflammatory mechanisms of isoalloLCA within the cells of the innate immune system, we conducted an extracellular metabolic flux analysis on bone marrow-derived macrophages (BMDMs) harvested from both Wt and Il10−/− mice, under conditions devoid of and in response to LPS. Our initial findings revealed that treatment with isoalloLCA effectively counteracted the LPS-induced inhibition of mitochondrial respiration, as evidenced by a notable increase in oxygen consumption rate (OCR)-a key indicator of oxidative phosphorylation (OXPHOS)-12 h post-treatment with isoalloLCA and LPS (Supplementary Fig. 7a). Furthermore, we observed that isoalloLCA suppressed LPS-induced aerobic glycolysis in BMDMs, as assessed through extracellular acidification rate (ECAR) analysis at the 12-hour mark—serving as a measure for glycolytic activity (Supplementary Fig. 7b). Consistently, after 24 hours of treatment, isoalloLCA was found to enhance OXPHOS while concurrently inhibiting glycolysis in BMDMs derived from Wt mice (Fig. 8a, b). Subsequently, we demonstrated that LPS stimulation led to elevated basal ECAR but diminished OCR in BMDMs (Fig. 8a, b). The compromised OXPHOS coupled with heightened glycolysis observed in Il10−/− BMDMs was ameliorated upon administration of exogenous isoalloLCA (Fig. 8a, b), resulting in altered profiles for ECAR and OCR akin to those seen in Wt BMDMs. We then proceeded to evaluate the functional characteristics of mitochondria by monitoring real-time fluctuations in OCR during sequential treatments involving oligomycin [an inhibitor of adenosine triphosphate (ATP) synthase], cyanide p-trifluoromethoxyphenyl-hydrazone (FCCP; an H+ ionophore), and rotenone [an inhibitor targeting the electron transport chain] (Fig. 8a, b). Notably, Il10−/− BMDMs exhibited reduced levels across several parameters: basal mitochondrial respiration (BASAL), maximal respiratory capacity (MRC), mitochondrial ATP production, and spare respiratory capacity (SRC), particularly when compared with their Wt counterparts (Fig. 8a, b). While LPS stimulation resulted in diminished BASAL, MRC, ATP production, and SRC across Wt BMDMs, but they were attenuated by isoalloLCA (Fig. 8a, b). Aerobic respiration and OXPHOS are linked to anti-inflammatory phenotypes in macrophages22. ETS2 drives macrophage inflammation via two metabolic targets, PFKFB3, a rate-limiting enzyme of glycolysis and HIF1α, a key factor contributing to inflammatory responses reprogramming19. Consistent with the observed metabolic changes, we observed isoalloLCA treatment suppressed ETS2 and its targets PFKFB3 and HIF1α (Fig. 8c, d). Consistent with previous findings that demonstrated isoalloLCA could induce mitochondrial reactive oxygen species (mtROS) production in regulatory T cells11,12, our results indicated that isoalloLCA also augmented mtROS generation within BMDMs (Supplementary Fig. 8).
Bone marrow-derived macrophages (BMDMs) were isolated from both wild-type (Wt) and Il10 knockout (Il10−/−) mice and induced for 9 days with GM-CSF. BMDMs were treated isoalloLCA (20 µM) with or without LPS (100 ng/mL) for 24 hours. a, b OCR data from a mitochondrial stress test and ECAR data from a glycolysis stress test of BMDMs. OCR oxygen consumption rate, ECAR extracellular acidification rate, FCCP phenylhydrazone, 2-DG 2-deoxyglucose, BASAL basal mitochondrial respiration, MRC maximal respiratory capacity, SRC mitochondrial ATP production, and spare respiratory capacity. c Western blot (WB) analysis for ETS2, HIF 1α and PFKFB3 in Wt and Il10−/− BMDMs. d Quantification of proteins levels in panel (c). The proteins levels were calculated against β-actin. Representative data shown in (a, b, and d) expressed as the mean ± standard error (SEM). Ordinary one-way ANOVA analysis for (d). ns, not significant (p ≥ 0.05), * p < 0.05, ** p < 0.01, *** p < 0.001, **** p < 0.0001.
IsoalloLCA metabolically reprograms the macrophages
We then sought to elucidate whether this metabolic reprogramming induced by isoalloLCA was associated with its anti-inflammatory properties in LPS-stimulated BMDMs. Notably, treatment with isoalloLCA inhibited the expression of inflammatory transcription factors such as phosphorylated STAT3 while simultaneously upregulating the anti-inflammatory macrophage marker ARG1 within BMDMs (Fig. 9a, b). Moreover, isoalloLCA increased expression of the scavenger receptors MARCO, along with nucleotide-binding oligomerization domain-containing protein 2 (NOD2) in LPS BMDMs (Fig. 9a, b). Subsequently, performed phagocytosis effects of isoalloLCA in BMDMs, as illustrated in Supplementary Fig. 9, administration of isoalloLCA resulted in a notable increase in the number of intracellular beads and an elevated level of intracellular fluorescence intensity, thereby enhancing the phagocytic activity of BMDMs (Supplementary Fig. 9). Furthermore, it was observed that the energy-sensing pathway involving AMPK-mTOR was slightly attenuated following administration of isoalloLCA (Fig. 9a, b). As illustrated in Fig. 10, Il10−/− BMDMs exhibited heightened inflammatory characteristics characterized by increased protein levels phosphorylated STAT3 compared to Wt BMDMs, and these effects were mitigated by treatment with isoalloLCA (Fig. 10a, b). Additionally, Il10−/− BMDMs displayed diminished levels of phagocytosis-associated proteins MARCO and NOD2 relative to Wt counterparts-a reduction that was restored upon exposure to isoalloLCA treatment (Fig. 10a, b). Moreover, phosphorylation levels of mTOR and AMPK were found to be reduced in Il10−/− BMDMs when contrasted with Wt BMDM. Intriguingly, these alterations were not restored upon treatment with isoalloLCA (Fig. 10a, b). Similar to the findings observed in LPS Wt BMDMs, we also found that isoalloLCA disrupted the ETS2-HIF1A/PFKFB3 pro-inflammatory signaling pathway and attenuated the expression of proinflammatory cytokine IL-1β in Il10−/− BMDMs under LPS stimulation conditions (Supplementary Fig. 10). Additionally, isoalloLCA increased the anti-inflammatory macrophage marker CD206 while decreasing the pro-inflammatory marker CD86 (Supplementary Fig. 10).
Bone marrow-derived macrophages (BMDMs) were treated with 20 µM isoalloLCA overnight and stimulated with 1 µg/mL lipopolysaccharide (LPS) for 2 hours. a Western blot (WB) analysis for EMR1, ARG1, MACRO, NOD2, phosphorylated GSK3β (p-GSK3β), GAGA2/3, phosphorylated AKT (p-AKT), phosphorylated mTOR (p-mTOR), phosphorylated AMPK (p-AMPK), phosphorylated S670 (p-S670), TBX21, RORγt, and phosphorylated STAT3 (p-STAT3) in BMDMs (each group, n = 3). b Quantification of proteins levels in panel (a). The proteins levels were calculated against β-actin. Representative data shown in (b) expressed as the mean ± standard error (SEM). Ordinary one-way ANOVA analysis for (b). ns not significant (p ≥ 0.05), *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001.
a Bone marrow-derived macrophages (BMDMs) were treated with 20 µM isoalloLCA overnight. a Western blot (WB) analysis for EMR1, ARG1, MACRO, NOD2, phosphorylated GSK3β (p-GSK3β), GAGA2/3, phosphorylated AKT (p-AKT), phosphorylated mTOR (p-mTOR), phosphorylated AMPK (p-AMPK), phosphorylated S670 (p-S670), TBX21, RORγt, and phosphorylated STAT3 (p-STAT3) in Wt and Il10−/− BMDMs (each group, n = 3). b Quantification of proteins levels in panel (a). The proteins levels were calculated againstβ-actin. Representative data shown in (b) expressed as the mean ± standard error (SEM). Ordinary One-way ANOVA analysis for (b). ns, not significant (p ≥ 0.05), *p < 0.05, ***p < 0.001, ****p < 0.0001.
Discussion
The global prevalence of inflammatory bowel disease (IBD) is rising annually, affecting patients’ quality of life and imposing a substantial economic burden on governments. There is an urgent need for effective therapeutic interventions. In this study, we demonstrate that isoalloLCA levels are lower in individuals with IBD. IsoalloLCA effectively inhibits LPS-induced inflammation in the blood of pediatric IBD patients and reduces acute intestinal inflammation caused by LPS or DSS. It also alleviates chronic colitis in Il10 knockout (Il10−/−) mice, where FOXP3 levels increased while ETS2 decreased in the intestinal mucosa. IsoalloLCA enhances oxidative phosphorylation (OXPHOS) in LPS-activated bone marrow-derived macrophages (BMDMs). Furthermore, it inhibits the transition to pro-inflammatory macrophages by targeting ETS2 and its downstream targets HIF1A and PFKFB3. Our findings suggest that metabolic regulation of macrophages boosts the anti-inflammatory effects of isoalloLCA, highlighting its potential as a promising therapeutic agent for IBD.
Bile acids play a pivotal role in modulating gut-associated inflammation, underscoring their remarkable potential to orchestrate the regulation of mucosal immune cells within the complex landscape of the gastrointestinal tract23,24. The bacterial bile acids metabolites are highly regarded for their prominent contributions across diverse biological contexts, particularly in the modulation of host metabolic and immune responses. Notably, there is a strong emphasis on the regulation of T cells9,11,12,14,25, and the prevention of intestinal pathogen proliferation15,26,27. Notably, lithocholic acid (LCA), deoxycholic acid (DCA), and isolithocholic acid (isoLCA) have been shown to inhibit the growth of Clostridium difficile15,26,27. Moreover, additional investigations have unveiled a notable attenuation in the differentiation of Th17 cells, elicited by both isoLCA and lithocholic acid-3-sulfate (LCA-3S)25,28. The oral administration of Bacteroides uniformis in murine models increased bile acid production, including isoLCA and isoDCA, influencing Th17 cell differentiation29. Our research has elucidated that the isoalloLCA is depleted in pediatric patients suffering from IBD. Given that these bacteria primarily synthesize isoalloLCA12,15, we conducted a re-analysis of the dataset. Our findings indicated that abundance of Parabacteroides merdae, Parabacteroides distasonis, and Parabacteroides gordonii in stool was much lower in both CD and UC than their controls18. In the study, it was demonstrated that the administration of isoalloLCA in blood and PBMCs from IBD patients could inhibit LPS-stimulated TNF-α production. Furthermore, we subsequently established that treatment with isoalloLCA could markedly alleviate both acute and chronic intestinal inflammation in murine models. We reaffirmed the pivotal role of isoalloLCA in enhancing the differentiation of anti-inflammatory Treg cells through the upregulation of FOXP3 expression. It has also been observed that isoalloLCA inhibits inflammatory macrophages in the context of IBD by regulating ETS2 in these murine models. However, it remains unclear how isoalloLCA exerts its influence on macrophages.
Macrophages function as essential effector cells, intricately interwoven into the intricate tapestry of both innate and adaptive immunity. They bear the crucial responsibility of eradicating necrotic cells and pathogens from the organism during states of homeostasis, thereby playing a pivotal role in mitigating inflammation7,30. Macrophages play a crucial role in maintaining intestinal homeostasis and are involved in the pathogenesis of in IBD. In individuals suffering from immune disorders, the excessive activation and infiltration of macrophages can cause considerable damage to the intestinal environment. The inflammatory phenotype is believed to be closely linked to the onset of IBD. Furthermore, anti-TNFα therapy has shown effectiveness in improving this condition by promoting the polarization of macrophages towards the more reparative anti-inflammatory phenotype31,32. The metabolic characteristics of inflammatory and anti-inflammatory macrophages unveil striking distinctions. Aerobic glycolysis plays a pivotal role in the polarization of inflammatory macrophages, while oxidative phosphorylation emerges as a fundamental component in the polarization of anti-inflammatory macrophages. Furthermore, alterations in metabolic pathways exert disparate influences on various macrophage phenotypes, with particularly pronounced effects observed within the phenotype of inflammatory macrophages33,34. Over the past decade, it has become increasingly evident that cellular metabolism plays a crucial and irreplaceable role in orchestrating macrophage activation35. Indeed, the aberrant metabolism of macrophages has been intricately associated with tumorigenesis, atherosclerosis, and chronic inflammatory conditions such as rheumatoid arthritis36. We have elucidated that isoalloLCA alleviates LPS-induced inflammation in bone marrow-derived macrophages (BMDMs) by augmenting oxidative phosphorylation (OXPHOS) while simultaneously curtailing glycolysis. In concordance with previous findings11,12, the effects of isoalloLCA are mediated through the induction of mitochondrial reactive oxygen species (mtROS), which play a pivotal role in facilitating Treg differentiation. Our investigations reveal that isoalloLCA elevates mtROS levels within BMDMs, potentially culminating in enhanced mitochondrial OXPHOS among macrophages. ETS2, a distinguished transcription factor that is part of the illustrious ETS family and recognized as a proto-oncogene, plays an essential role in orchestrating the intricate regulation of human inflammatory macrophages along with their associated inflammatory responses19. Notably, the disruption of ETS2 has been shown to provoke anti-inflammatory phenotypes by intricately modulating aerobic respiration and OXPHOS within macrophages. This modulation transpires through the regulation of crucial target genes that play a noteworthy role in this intricate process, including hypoxia-inducible factor 1-alpha (HIF1A) and the glycolytic enzyme 6-phosphofructo-2-kinase/fructose-2,6-bisphosphatase 3 (PFKFB3)19,37. Our findings elucidate that the administration of isoalloLCA engenders a pronounced diminution in the expression levels of ETS2 and its downstream targets, HIF1A and PFKFB3, within macrophages. This observation intimates that the anti-inflammatory effects mediated by isoalloLCA may be intricately orchestrated through the modulation of the ETS2 signaling pathway.
In conclusion, our findings enhance the existing body of evidence regarding the anti-inflammatory properties of isoalloLCA, which are intricately linked to the reprogramming of mitochondrial metabolism within macrophages. This complex process culminates in a marked attenuation of signaling through both ETS2 pathways, ultimately leading to a pronounced suppression of the inflammatory response. These compelling results indicate that isoalloLCA holds considerable therapeutic potential for patients suffering from IBD.
Materials and methods
Data availability and analysis
This data of isoalloLCA abundance in infants with different ages and in UC patients with different stages are available at the NIH Common Fund’s National Metabolomics Data Repository (NMDR) website, the Metabolomics Workbench, https://www.metabolomicsworkbench.org. The data about isoalloLCA abundance in infants has been assigned Project ID PR001435, which can be accessed directly via Project https://doi.org/10.21228/M87D7V. The data about isoalloLCA abundance in UC patients has been assigned ID PR00159617, which can be accessed directly via Project https://doi.org/10.21228/M8D701.This work is supported by Metabolomics Workbench/National Metabolomics Data Repository (NMDR) (grant# U2C-DK119886), Common Fund Data Ecosystem (CFDE) (grant# 3OT2OD030544) and Metabolomics Consortium Coordinating Center (M3C) (grant# 1U2C-DK119889). Statistical analysis of isoalloLCA levels and microbial data in human IBD cohorts are available at Prospective Registry in IBD Study at MGH (PRISM). Metabolomics profiles and microbial data from the PRISM were obtained from the supporting information of a publication18.
Human subjects and analysis
Pediatric patients confirmed as IBD were recruited from the Division of Pediatric Gastroenterology and Nutrition in Xinhua Hospital affiliated to Shanghai Jiao Tong University School of Medicine. The blood as well as normal intestinal biopsies were collected from 41 CD pediatric patients and 8 UC pediatric patients during their endoscopy visit (Supplementary Table 1). Pediatric patients (age <18 years) were diagnosed according to Revised Porto Criteria38. All methods in this study were carried out in accordance with the relevant guidelines. All patients’ guardians provided written informed consent. The institutional ethics committee of Xinhua Hospital affiliated to Shanghai Jiao Tong University School of Medicine approved this study (XHEC-D-2022-030). Whole blood samples from both patients (CD, n = 5; UC, n = 2) and controls (n = 5) were transferred to a sterile 96-well plate using pipettes. RPMI 1640 medium without any additives served as the negative control. The final dilution ratio for whole blood was set at 1:5. To stimulate immune response, LPS was added at a concentration of 2 μg/mL either individually or in conjunction with isoalloLCA (20 μM). The plate was then incubated for a period of twenty-four hours under conditions of temperature maintained at constant level of 37 °C along with an atmosphere containing 5% CO2. After completion of the incubation process, the supernatants were taken and subjected to Enzyme-linked immunosorbent assay (ELISA) analysis of tumor necrosis factor alpha (TNF-α). The peripheral blood mononuclear cells (PBMCs) were isolated from CD patients using Ficoll’s method as described previously39. Briefly, about 5 mL of whole blood was mixed with Dulbecco’s Phosphate Buffered Saline (DPBS) (1:1) and carefully added on top of Ficoll density gradient (Ficoll® Paque Plus, Merck). The samples were centrifuged for 30-min at 400 × g with no braking. The PBMC layer was then collected and washed twice in 40 mL DPBS for 10-min at 80 × g without applying braking and then resuspended in 1 mL DPBS for immediate use or stored at −80 °C for later analysis. Total PBMCs were counted using an Invitrogen Countess® Automated Cell Counter. A total of 5 × 10 6 PBMCs each well were cultured in RPMI 1640 medium with or without 2 μg/ml LPS or/and 20 μM isoalloLCA for 24 hours. After centrifugation, the supernatant was collected for TNF-αELSIA and the cells were used to extract RNA using the RNA isolation kit (TIANGEN, Shanghai, China). The isolated PBMCs from CD patients (n = 16) were also used to analyze Oxygen consumption rates measurement (OCR) and Extracellular acidification rates measurement (ECAR) as described following. The mucosal biopsies (CD, n = 5; UC, n = 5) taken from inflamed tissues and corresponding noninflammed mucosa adjacent to the inflamed during their endoscopy were used to immunochemistry (IHC) staining.
Enzyme-linked immunosorbent assay (ELISA) for TNF-α
The levels of TNF-α in whole blood and supernatants of PBMCs were quantified using commercially available ELISA kits (Proteintech#KE00154) following the manufacturer’s instructions. Briefly, samples and standards were added to a 96-well plate and incubated at 37 °C for 2 hours. Subsequently, the plates were washed, and a detection antibody was added followed by an incubation period of 1 hour at 37 °C. After another round of washing, HRP-labeled secondary antibodies were added to each well and incubated for 40 minutes at 37 °C. Following a final wash step, a color-developing reagent was introduced into each well. After an incubation period of 15–25 minutes at 37 °C, stop solution was added, and the optical densities were measured at wavelengths of both 450 nm with wavelength correction at 630 nm. All experiments were conducted in triplicate.
Dextran sodium sulfate (DSS)-induced colitis in mice
All animal experiments were carried out in accordance with China’s guidelines and laws for the use and care of experimental animals. Male/Female C57BL/6 J wild-type (Wt) mice (20 ± 2 g, 7 to 8 weeks of age) were purchased from GemPharmatech Co., Ltd. (Nanjing, China), and housed in specific pathogen-free conditions (temperature, 22 ± 2 °C; relative humidity, 55–60%; and a regular 12/12 h light/dark cycle). All mice were acclimated to the environment for 1 week before the formal experiment, and then randomly divided into groups. The DMSO control group (DSMO) was treated via the administration of distilled water and intraperitoneally DMSO each day for 7 days (female, n = 8; male, n = 10). The isoalloLCA group was intraperitoneally (i.p.; 5 mg/kg body weight, BW) every day for 7 days (female, n = 6; male, n = 8). The model group (DSS) was treated with DSS solution to induce experimental colitis for 7 days and given them DMSO each day (female, n = 8; male, n = 8). The DSS plus isoalloLCA group was given DSS and isoalloLCA (5 mg/kg body weight, BW) each day for 7 days (female, n = 8; male, n = 9). DSS was added to the drinking water (3%) from day 1 to 7 of the experimental colitis mice model. Body weights were recorded once daily. The mice were sacrificed prior to being anesthetized with an injection of Pentobarbital Sodium (P3761, Sigma-Aldrich) at a dosage of 50 mg/kg. All organs were collected at day 8 and stored at −80 °C for further analysis. All animal experiments were approved by the Experimental Animal Protection and Use Committee of Xinhua Hospital (No. XHEC-C-F-2022-010). We have complied with all relevant ethical regulations for animal use.
Lipopolysaccharide (LPS)–induced acute inflammation in mice
The model was established by injecting C57BL/6 mice ~6 weeks old with lipopolysaccharide (LPS) intraperitoneally (i.p.; #G5032; Wuhan Servicebio Technology Co., Ltd., Wuhan, China) at a dose of 5 mg/kg for 20 hours, and then randomly divided into groups. The day before the LPS injection, the control group (Control; female, n = 5; male, n = 5) was given DMSO. The isoalloLCA group was intraperitoneally (5 mg/kg body weight, BW; female, n = 5; male, n = 5) two times. The LPS group (female, n = 10; male, n = 12) was given LPS and plus DMSO. The LPS plus isoalloLCA group was given the isoalloLCA at a day before LPS injection and the time injection (5 mg/kg body weight, BW; female, n = 8; male, n = 7). The mice were sacrificed prior to being anesthetized with an injection of Pentobarbital Sodium (P3761, Sigma-Aldrich) at a dosage of 50 mg/kg. All organs were collected after 20 hours of LPS administration and stored at −80 °C for further analysis.
Il10 knockout (Il10 −/−)-induced inflammation in mice
Knockout mice (Il10−/−) were obtained from GemPharmatech (Nanjing, China) and cultured in animal center of Xinhua Hospital under normal condition. Wt and Il10−/− mice were raised to about 8-week old and given them DMSO or isoalloLCA every 3-time in a week, respectively. The groups including Wt DMSO (female, n = 8; male, n = 10), Il10−/− DMSO (female, n = 6; male, n = 6), Wt isoalloLCA (5 mg/kg body weight, BW; female, n = 8; male, n = 8) and Il10−/− isoalloLCA (5 mg/kg body weight, BW; female, n = 5; male, n = 7). After 4 weeks of treatments, the mice were humanely euthanized prior to being anesthetized with an injection of Pentobarbital Sodium (P3761, Sigma-Aldrich) administered at a dosage of 50 mg/kg. All organs were collected and stored at -80 °C for further analysis.
Intestinal characterization and histological score
Intestinal tissues from mice were fixed in 4% paraformaldehyde (PFA) for 24 hours and subsequently sectioned into 4 μm slices for hematoxylin and eosin (H&E) staining. Villus height and crypt depth were measured using the National Institutes of Health (NIH) Image software (NIH, Bethesda, MD, USA) in conjunction with a microscope (Nikon, Tokyo, Japan). Villus height was assessed from five well-oriented villi on each slide, with five fields analyzed per section. Goblet cells were counted and mucous secretions quantified utilizing Alcian blue/periodic acid–Schiff (AB/PAS) staining. For histological score, histological alterations in the intestinal mucosa were assessed as previously described40,41. In brief, histological scores were determined in a blinded manner based on the cumulative scores of epithelial loss and infiltration. The scoring for epithelial changes was as follows: 0 = normal; 1 = localized loss of goblet cells; 2 = extensive loss of goblet cells; 3 = localized crypt loss; and 4 = extensive crypt loss. The infiltration scores were categorized as follows: 0 = normal; 1 = infiltrate surrounding the crypt base; 2 = moderate infiltrate extending to the muscularis mucosae; 3 = significant infiltration reaching the muscularis mucosae; and 4 = infiltration into the submucosa.
Immunohistochemistry (IHC) staining
The inflamed mucosal biopsies (CD, n = 5; UC, n = 5) and corresponding noninflammed mucosa from IBD patients, as well as mice intestinal tissues from mice inflamed models were used to perform immunohistochemistry (IHC) staining. IHC staining was performed according to a previously published protocol. Briefly, 3 μm thick paraffin-embedded sections were dried at 70 °C for 30 minutes and then dewaxed. Endogenous peroxidases were blocked using 3% H2O2 for 25 minutes at room temperature (RT), followed by sealing the sections with 3% bovine serum albumin for 30 minutes. The antibodies were applied overnight at their optimal concentration in a humidified chamber at 4 °C. Subsequently, the slides were rinsed with phosphate-buffered saline (PBS) and incubated with the appropriate secondary antibody for one hour at RT. Tissue sections were visualized using diaminobenzidine substrate chromogen, followed by counterstaining with hematoxylin. Images were captured using a microscope and analyzed using ImageJ Software. Five randomly selected microscopic high-power fields per sample were used to determine average optical density, which was further averaged for statistical analysis. IHC staining was performed utilizing antibodies listed in Supplementary Table 2.
Western blot (WB) analysis
The total protein was extracted from mice intestinal tissues or BMDMs using the radioimmune precipitation assay buffer (RIPA) (#G2002, Servicebio) containing proteases and phosphatase inhibitors. Protein concentration was measured using a BCA protein assay kit (#P0012, Beyotime). An equal amount of protein in each panel was isolated on SurePAGE™ 4-20% Bis-Tris gels (#M00657, Genscript) and then transferred onto polyvinylidene fluoride (PVDF, #ISEQ00010, Cellpro) membranes. Subsequently, the membranes were sealed using Protein Free Rapid Blocking Buffer (#PS108P, Epizyme) for 10 minutes at room temperature. Next, the membranes were treated with primary antibodies at 4°C overnight. The next day, the membranes were added with the secondary antibody for 60 minutes at room temperature. The protein bands were visualized by an integrated chemiluminescence instrument ChemiDocXRS+ (Bio-Rad), and the band intensities were quantified using Image J software. The primary antibodies used were listed in Supplementary Table 2.
Quantitative real-time polymerase chain reaction (qRT-PCR)
Total RNA was extracted from human PBMCs, mice intestinal mucosal tissues and mice small bowel crypts using an RNeasy kit (QIAGEN) according to the manufacturer’s protocol. For isolation of small intestinal crypts, the isolated methods were conducted as previously described42. The isolated small bowel crypts were given isoalloLCA (20 µM) 1 hour prior to treated them with LPS (1 µg/mL) for 2 hours. The quantity of RNA was assessed with a NanoDrop spectrophotometer (Applied Biosystems). For reverse transcription, we utilized a High Capacity Complementary DNA (cDNA) Reverse Transcription Kit (Applied Biosystems), employing 2 μg of RNA. Subsequently, real-time PCR reactions were conducted using a ViiA 7 Real-Time PCR System in conjunction with the PowerUp SYBR Green Master Mix Kit (both from Applied Biosystems). The PCR reactions were incubated in a 384-well plate at 95 °C for 10 minutes, followed by 40 cycles consisting of denaturation at 95 °C for 15 seconds and annealing/extension at 60 °C for one minute. All samples were analyzed in triplicate, and data normalization was performed against the endogenous control β-actin or hypoxanthine-phosphoribosyltransferase 1 (HPRT1). Relative RNA expression levels were calculated utilizing the ΔΔCt method. Primers, which were adapted from previous studies43,44,45 and synthesized by Invitrogen (Shanghai, China), are detailed in Supplementary Table 3.
Isolation and treatments of mice bone marrow-derived macrophages (BMDMs)
The preparation of BMDMs was performed as the protocol described46,47,48. Briefly, the mice were killed with cervical dislocation and sterilized with 75% alcohol. The femur and tibia tissues were isolated under aseptic conditions. Muscle tissues on the bone surface were removed, and both ends were carefully cut using sterile scissors. The bone marrow cells were collected by flushing the bone marrow cavity with DMEM medium until it turned white. The rinsing solution was centrifuged at 1200 rpm for 5 min, and then the supernatant was discarded. BMDMs were generated by bone marrow cells cultured in DMEM supplemented with 10% fetal bovine serum, 1% penicillin-streptomycin, and 10 ng/mL GM-CSF (#315-03-20UG, Preprotech) for 9 days. On day 3, an equal amount of fresh medium containing GM-CSF was added. On day 7, BMDMs strongly adhered to the surface of the dish were visible, and the culture medium with unattached cells was replaced with fresh media, at which time the cells could be treated. For cell treatments, BMDMs were treated with 20 µM isoalloLCA for overnight and then stimulated with 1 µg/mL lipopolysaccharide (LPS) for 2 hours. DMSO treatment served as the vehicle control. The cells were then collected for further experiments.
Macrophage phagocytosis
To assess the phagocytic capacity of isoalloLCA, BMDMs were treated with 20 µM isoalloLCA for overnight. On the subsequent day, silica beads functionalized with carboxyl (Sigma#L4655) were suspended in the cellular growth medium and incubated with AF488 (10 μg/ml) at 37°C for 30 minutes. After being washed six times with PBS, the beads were resuspended in fresh medium. BMDMs were stimulated with or without 1 µg/mL lipopolysaccharide (LPS) for 2 hours. Subsequently, macrophages were exposed to AF488-labeled silica beads and incubated at 37°C for an additional 2 hours. The culture medium was then washed to remove extracellular beads. Finally, the cells were washed for three times, and the optical densities were measured at an excitation/emission wavelength of 490/520 nm and the fluorescence microscope was used to photograph.
Mitochondrial reactive oxygen species (mitoROS) in BMDMs
For cell treatments, BMDMs were treated with 20 µM isoalloLCA for overnight and then stimulated with 1 µg/mL lipopolysaccharide (LPS) for 2 hours. The 50 µg MitoSOX Red Mitochondrial Superoxide Indicator (#40778ES50, Yeasen) was combined with 13 µl of DMSO to create a 5 mM storage solution. The working solution of the probe was prepared by diluting the Z5 mM storage solution to a final concentration of 5 µM using HBSS (which includes Ca2+ and Mg2+). The cells were thoroughly covered with the probe working solution and incubated at 37 °C for 10 minutes in the absence of light. Following incubation, the cells were gently washed three times with preheated HBSS buffer. Fluorescence intensity was measured, utilizing an excitation/emission wavelength of 356/411 nm for the probe itself, while oxidized MitoSOX exhibited an excitation/emission wavelength of 396/580 nm. A fluorescence microscope was employed to capture fluorescence images.
Oxygen consumption rates measurement (OCR)
The mitochondrial oxygen consumption rate measurement (OCR) was measured by the XF96 Extracellular Flux analyzer, following the protocol as previously described49,50. The mouse bone marrow cells were seeded in the Seahorse XF Cell Culture Microplate with a density of about 20,000 cells, and cultured in DMEM medium containing 10 ng/mL GM-CSF for 9 days to harvest BMDMs. On day 9, BMDMs were treated with isoalloLCA (20 µM) with or without LPS (100 ng/mL) for 12 or 24 hours. The OCR assay in PBMCs, the isolated PBMCs (2 * 104/well) isoalloLCA (20 µM) with or without LPS (100 ng/mL) for 16 hours. Cells were treated PBS served as the vehicle control. After treatments, BMDMs were washed three times with Seahorse XF medium (DMEM without phenol red supplemented with 1 mM pyruvate, 2 mM glutamine, and 10 mM glucose), and the Seahorse Mito Stress protocol was run. Oxygen consumption was assessed by the sequential addition of 1.5 µM oligomycin A, 1 µM FCCP, and 0.5 µM/0.5 µM rotenone-antimycin A. ATP-linked OCR was calculated by subtracting OCR values after oligomycin A treatment from basal OCR values. Each time point was calculated as an average from at least 3 replicates.
Extracellular acidification rates measurement (ECAR)
The extracellular acidification rates measurement (ECAR) was assessed in BMDMs using the XF96 Extracellular Flux analyzer, following the manufacturer’s instructions51. The mouse bone marrow cells were seeded in the Seahorse XF Cell Culture Microplate with a density of 20,000 cells, and cultured in DMEM medium containing 10 ng/mL GM-CSF for 9 days to harvest BMDMs. On day 9, BMDMs were treated with isoalloLCA (20 µM) with or without LPS (100 ng/mL) for 12 or 24 hours. The ECAR assay in PBMCs, the isolated PBMCs (2 * 104/well) isoalloLCA (20 µM) with or without LPS (100 ng/mL) for 16 hours. PBS treatment served as the vehicle control. The next day, BMDMs were washed three times with Seahorse XF medium (DMEM without phenol red supplemented with 2 mM glutamine). ECAR measurements were conducted using the XF96 Extracellular Flux Analyzer under basal conditions, and subsequent measurements were taken after the addition of specific compounds, including 10 mM glucose, 1.5 mM oligomycin, and 50 mM the glucose analog, 2-deoxyglucose, 2DG.
Mice intestinal organoids culture
The isolation of intestinal crypts and the culture of organoids were conducted as previously described42. Briefly, following the euthanasia of mice via CO2 and the extraction of the small intestine, the organ was longitudinally opened and rinsed with cold phosphate-buffered saline (PBS). The small intestine was then cut into approximately 2 mm segments and incubated in 10 mM EDTA with PBS for 20 minutes at 4 °C. After the EDTA solution was removed, the tissue pieces were suspended with 10 ml of cold PBS, discarding the supernatant. The sediment was then vigorously resuspended in PBS, the supernatant was enriched for crypts. This fraction was filtered through a 70-μm cell strainer to eliminate impurities. The isolated crypts were centrifuged at 150 × g for 10 minutes to separate them from single cells. For the preparation of mouse organoids, 1000 crypts per well were embedded in Matrigel (#356255, Corning) within 24-well plates. The Matrigel was polymerized for 10 minutes at 37 °C and then immersed with the IntestiCulture™ organoid growth medium (#6005, STEMCELL), which contained 10% FBS and 1% penicillin-streptomycin. The organoids were treated with 10 µM isoalloLCA. Daily photographs of the organoids were taken, and the medium was refreshed every 2–3 days.
Statistics and reproducibility
Detailed sample size (n) and number of replicates are provided in Methods and Figure legends. Statistical tests were performed using GraphPad Prism 8 Software (GraphPad, San Diego, CA). Statistical analysis was performed by using an unpaired Student’s t test or one-way analysis of variance for comparisons of different groups. Non-normally distributed data were determined by using the Mann–Whitney U test or the Kruskal–Wallis test. Each mouse was assessed as an individual sample. Representative data shown and expressed as the mean ± standard error (SEM). p values < 0.05 were considered statistically significant. Significance levels were split further as to **p < 0.01, ***p < 0.001, ****p < 0.0001.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The data generated or analyzed during this study are available from the corresponding author upon reasonable request. Source data, as well as statistical analysis for all graphs, are provided in the Excel file Supplementary Data 1.This data of isoalloLCA abundance in Fig. 1a, b are available at the NIH Common Fund’s National Metabolomics Data Repository (NMDR) website, the Metabolomics Workbench (https://www.metabolomicsworkbench.org)17. Statistical analysis of isoalloLCA levels and microbial data in human IBD cohorts in Fig. 1c, d are available at Prospective Registry in IBD Study at MGH (PRISM), and the PRISM data were obtained from the supporting information of a publication18. Supplementary Fig. 2 was reanalyzed using the dataset (GSE163349)12. Source images for representative western blots shown in figures are provided in Supplementary Fig. 11.
References
Miller, F. W. The increasing prevalence of autoimmunity and autoimmune diseases: an urgent call to action for improved understanding, diagnosis, treatment, and prevention. Curr. Opin. Immunol. 80, 102266 (2023).
Ng, S. C. et al. Worldwide incidence and prevalence of inflammatory bowel disease in the 21st century: a systematic review of population-based studies. Lancet 390, 2769–2778 (2017).
Coward, S. et al. Past and future burden of inflammatory bowel diseases based on modeling of population-based data. Gastroenterology 156, 1345–1353 e1344 (2019).
Jones, G. R. et al. IBD prevalence in Lothian, Scotland, derived by capture-recapture methodology. Gut 68, 1953–1960 (2019).
Mowat, A. M. & Agace, W. W. Regional specialization within the intestinal immune system. Nat. Rev. Immunol. 14, 667–685 (2014).
Martin, J. C. et al. Single-cell analysis of Crohn’s disease lesions identifies a pathogenic cellular module associated with resistance to anti-TNF therapy. Cell 178, 1493–1508 e1420 (2019).
Hegarty, L. M., Jones, G. R. & Bain, C. C. Macrophages in intestinal homeostasis and inflammatory bowel disease. Nat. Rev. Gastroenterol. Hepatol. 20, 538–553 (2023).
Mohanty, I. et al. The changing metabolic landscape of bile acids–keys to metabolism and immune regulation. Nat. Rev. Gastroenterol. Hepatol. 21, 493–516 (2024).
Campbell, C. et al. Bacterial metabolism of bile acids promotes generation of peripheral regulatory T cells. Nature 581, 475–479 (2020).
Guo, C. et al. Bile acids control inflammation and metabolic disorder through inhibition of NLRP3 inflammasome. Immunity 45, 802–816 (2016).
Hang, S. et al. Bile acid metabolites control T(H)17 and T(reg) cell differentiation. Nature 576, 143–148 (2019).
Li, W. et al. A bacterial bile acid metabolite modulates T(reg) activity through the nuclear hormone receptor NR4A1. Cell Host Microbe 29, 1366–1377 e1369 (2021).
Ma, C. et al. Gut microbiome-mediated bile acid metabolism regulates liver cancer via NKT cells. Science 360, eaan5931 (2018).
Song, X. et al. Microbial bile acid metabolites modulate gut RORgamma(+) regulatory T cell homeostasis. Nature 577, 410–415 (2020).
Sato, Y. et al. Novel bile acid biosynthetic pathways are enriched in the microbiome of centenarians. Nature 599, 458–464 (2021).
Miyamoto, Y. et al. Periportal macrophages protect against commensal-driven liver inflammation. Nature 629, 901–909 (2024).
Schirmer, M. et al. Linking microbial genes to plasma and stool metabolites uncovers host-microbial interactions underlying ulcerative colitis disease course. Cell Host Microbe 32, 209–226 e207 (2024).
Lloyd-Price, J. et al. Multi-omics of the gut microbial ecosystem in inflammatory bowel diseases. Nature 569, 655–662 (2019).
Stankey, C. T. et al. A disease-associated gene desert directs macrophage inflammation through ETS2. Nature 630, 447–456 (2024).
Ojala, J. R., Pikkarainen, T., Tuuttila, A., Sandalova, T. & Tryggvason, K. Crystal structure of the cysteine-rich domain of scavenger receptor MARCO reveals the presence of a basic and an acidic cluster that both contribute to ligand recognition. J. Biol. Chem. 282, 16654–16666 (2007).
Hirasawa, R. et al. Essential and instructive roles of GATA factors in eosinophil development. J. Exp. Med. 195, 1379–1386 (2002).
Kelly, B. & O’Neill, L. A. Metabolic reprogramming in macrophages and dendritic cells in innate immunity. Cell Res. 25, 771–784 (2015).
Duboc, H. et al. Connecting dysbiosis, bile-acid dysmetabolism and gut inflammation in inflammatory bowel diseases. Gut 62, 531–539 (2013).
Martinez-Moya, P. et al. Dose-dependent antiinflammatory effect of ursodeoxycholic acid in experimental colitis. Int. Immunopharmacol. 15, 372–380 (2013).
Paik, D. et al. Human gut bacteria produce Tau(Eta)17-modulating bile acid metabolites. Nature 603, 907–912 (2022).
Buffie, C. G. et al. Precision microbiome reconstitution restores bile acid mediated resistance to Clostridium difficile. Nature 517, 205–208 (2015).
Thanissery, R., Winston, J. A. & Theriot, C. M. Inhibition of spore germination, growth, and toxin activity of clinically relevant C. difficile strains by gut microbiota derived secondary bile acids. Anaerobe 45, 86–100 (2017).
Xiao, R. P. et al. Synthesis and identification of lithocholic acid 3-sulfate as RORγt ligand to inhibit Th17 cell differentiation. J. Leukoc. Biol. 112, 835–843 (2022).
Yan, Y. et al. Bacteroides uniformis-induced perturbations in colonic microbiota and bile acid levels inhibit TH17 differentiation and ameliorate colitis developments. NPJ Biofilms Microbiomes 9, 56 (2023).
Zhang, K., Guo, J., Yan, W. & Xu, L. Macrophage polarization in inflammatory bowel disease. Cell Commun. Signal 21, 367 (2023).
Bloemendaal, F. M. et al. Anti-tumor necrosis factor with a glyco-engineered Fc-region has increased efficacy in mice with colitis. Gastroenterology 153, 1351–1362 e1354 (2017).
Vos, A. C. et al. Anti-tumor necrosis factor-alpha antibodies induce regulatory macrophages in an Fc region-dependent manner. Gastroenterology 140, 221–230 (2011).
Wang, S. et al. Isosteviol sodium exerts anti-colitic effects on BALB/c mice with dextran sodium sulfate-induced colitis through metabolic reprogramming and immune response modulation. J. Inflamm. Res. 14, 7107–7130 (2021).
Thibaut, R., Orliaguet, L., Ejlalmanesh, T., Venteclef, N. & Alzaid, F. Perspective on direction of control: Cellular metabolism and macrophage polarization. Front. Immunol. 13, 918747 (2022).
O’Neill, L. A., Kishton, R. J. & Rathmell, J. A guide to immunometabolism for immunologists. Nat. Rev. Immunol. 16, 553–565 (2016).
Tannahill, G. M. et al. Succinate is an inflammatory signal that induces IL-1beta through HIF-1alpha. Nature 496, 238–242 (2013).
Ray, K. ETS2 regulates human inflammatory macrophages in IBD. Nat. Rev. Gastroenterol. Hepatol. 21, 534 (2024).
Levine, A. et al. ESPGHAN revised porto criteria for the diagnosis of inflammatory bowel disease in children and adolescents. J. Pediatr. Gastroenterol. Nutr. 58, 795–806 (2014).
Riedhammer, C., Halbritter, D. & Weissert, R. Peripheral blood mononuclear cells: isolation, freezing, thawing, and culture. Methods Mol. Biol. 1304, 53–61 (2016).
Xiao, Y. T., Yan, W. H., Cao, Y., Yan, J. K. & Cai, W. Neutralization of IL-6 and TNF-alpha ameliorates intestinal permeability in DSS-induced colitis. Cytokine 83, 189–192 (2016).
Obermeier, F. et al. Interferon-gamma (IFN-gamma)- and tumour necrosis factor (TNF)-induced nitric oxide as toxic effector molecule in chronic dextran sulphate sodium (DSS)-induced colitis in mice. Clin. Exp. Immunol. 116, 238–245 (1999).
Sato, T. et al. Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature 459, 262–265 (2009).
Okumura, R. et al. Lypd8 promotes the segregation of flagellated microbiota and colonic epithelia. Nature 532, 117–121 (2016).
Ranjan, K., Hedl, M., Sinha, S., Zhang, X. & Abraham, C. Ubiquitination of ATF6 by disease-associated RNF186 promotes the innate receptor-induced unfolded protein response. J. Clin. Invest. 131, e145472 (2021).
Wang, Z., Huang, J., Yang, S. P. & Weaver, D. F. Anti-inflammatory anthranilate analogue enhances autophagy through mTOR and promotes ER-turnover through TEX264 during Alzheimer-associated neuroinflammation. ACS Chem. Neurosci. 13, 406–422 (2022).
Dong, Y. et al. Generation and identification of GM-CSF derived alveolar-like macrophages and dendritic cells from mouse bone marrow. J. Vis. Exp. 25, 54194 (2016).
Luu, A. M., Shepardson, K. M. & Rynda-Apple, A. A comprehensive protocol for the collection, differentiation, cryopreservation, and resuscitation of primary murine bone marrow derived macrophages (BMDM). Immunol. Invest. 53, 1001–1012 (2024).
Lee, S. W. et al. IDH2 regulates macrophage polarization and tumorigenesis by modulating mitochondrial metabolism in macrophages. Mol. Med. 30, 143 (2024).
Samson, N. et al. HSDL2 links nutritional cues to bile acid and cholesterol homeostasis. Sci. Adv. 10, eadk9681 (2024).
Shum, M. et al. ABCB10 exports mitochondrial biliverdin, driving metabolic maladaptation in obesity. Sci. Transl. Med. 13, eabd1869 (2021).
Li, Y. et al. The mitochondrial TSPO ligand Atriol mitigates metabolic-associated steatohepatitis by downregulating CXCL1. Metabolism 159, 155942 (2024).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (82270537, 82570601), National Key R&D Program of China (2023YFC2706505), Natural Science Foundation of Shanghai Municipality (22ZR1480600), Shanghai Key Laboratory of Pediatric Gastroenterology and Nutrition (17DZ2272000). We appreciated the individuals who (Haoyun Mao and Jun Du) participated in the study.
Author information
Authors and Affiliations
Contributions
Wang Y., Yan W.H., Zhao H.X., Lu Y., and Xiao Y.T.: investigation, methodology, visualization. Li Y.H. and PENG S.H.: software and validation. Wang Y. and Xiao Y.T: conceptualization, data curation, project administration, resources, funding acquisition, supervision, writing—original draft, writing–review & editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Biology thanks Yu Miyamoto and the other, anonymous, reviewers for their contribution to the peer review of this work. Primary Handling Editors: Si Ming Man and Ophelia Bu.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wang, Y., Yan, W., Zhao, H. et al. Isoallolithocholic acid ameliorates intestinal inflammation via metabolically reprogrammed macrophages. Commun Biol 8, 1718 (2025). https://doi.org/10.1038/s42003-025-09123-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s42003-025-09123-3