Abstract
DNA G-quadruplexes (G4s) spontaneously arise in guanine-rich regions such as telomeres. Although G4s regulate essential biological process, their persistence can interfere with DNA metabolism and induce replication stress (RS). Helicases such as BLM and FANCJ play key roles in resolving G4 structures to maintain genome stability. Our investigation delves into how BLM and FANCJ respond to telomeric RS induced by the telomeric G4 ligand, RHPS4. To examine their individual and combined functions, we employed CRISPR/Cas9 to deplete BLM and siRNA to silence FANCJ in U251MG glioblastoma cells. Our results revealed a functional interplay between the two helicases: RHPS4 treatment led to increased FANCJ protein levels and telomeric recruitment in both wild-type and BLM-/- cells. Notably, FANCJ depletion resulted in heightened sensitivity to RHPS4, accompanied by increased telomere dysfunction and DNA damage, particularly in the absence of BLM. These findings underscore a compensatory role for FANCJ in maintaining telomere integrity under RS and highlight its potential as a therapeutic target for enhancing the efficacy of G4-stabilizing agents in cancer treatment.
Similar content being viewed by others
Introduction
Since the early 1960s, it has been known that DNA sequences rich in guanines have an intrinsic propensity to self-associate, giving rise to four-stranded helical secondary structures known as G-quadruplexes (G4)1. In vitro experiments have shown that the highly repetitive hexameric TTAGGG motif of mammalian telomeres can arrange into G4 structures2,3. Since then, it has been recognized that G4s can spontaneously occur in both DNA and RNA4,5. In addition to telomeres, G4s have been found in proximity to copy number alterations of cancer cells6, rDNA gene clusters7, subtelomeric regions8, gene promoters9, and 5′-UTR of more than 3000 encoding mRNAs10. This suggests a regulatory role in processes such as gene expression and translation repression8,9,11,12. Surprisingly, 90% of human DNA replication origins contain G4 motifs13,14,15, and therefore G4 structures may arise during replication when DNA molecules are transiently single-stranded16,17,18. In principle, their presence represents an obstacle to the progression of the replication machinery, and if not resolved, G4s can potentially threaten genome stability19. Therefore, it is not surprising that evolution has provided living organisms with enzymes capable of removing a wide range of DNA secondary structures, including G4s, which can potentially impede faithful DNA replication. In humans, over a hundred helicases involved in every aspect of DNA metabolism have been identified, capable of binding with different affinities and unwinding a variety of structural DNA substrates formed during DNA replication, recombination, and repair.
Based on the observation that some human genetic diseases associated with helicase mutations share similar clinical and cellular phenotypes, it is plausible that these helicases may have overlapping functions in preserving genome stability. This appears to be the case for the helicases encoded by the human BLM and FANCJ genes, for which crosstalk has been recognized in double-strand breaks (DSBs) repair, replication stress response, and G4 metabolism20,21. These two gene products interact physically and functionally in living cells, and BLM protein stability is dependent on FANCJ, whereas the converse is not true. Indeed, a significant reduction of BLM protein through degradation via a proteasome-mediated pathway has been observed in FANCJ-deficient cells21.
BLM encodes a member of the 3′–5′ RecQ-like helicase family, which is involved in maintaining genome integrity, homologous recombination (HR), and suppression of illegitimate recombination during DSB repair22,23. Patients affected by Bloom syndrome (BS), a rare autosomal recessive disorder caused by mutations in the BLM gene, are characterized by increased baseline frequency of sister chromatid exchanges (SCEs)24,25. They also exhibit chromosome and chromatid-type aberrations and are hypersensitive to DSB-inducing agents such as ionizing radiation26 and interstrand cross-linking agents like mitomycin C27. Interestingly, the role of the ATP-dependent 3′–5′ helicase activity of the BLM protein is not restricted to the unwinding of canonical Watson-Crick B-DNA in vitro28, but it shows much higher affinity for stable G4 structures29,30. Failure to properly manage G-rich sequences may therefore contribute to the increase in genetic exchanges characteristic of BS. Telomeric sequences are enriched in guanine and are therefore prone to forming G4 structures, which may interfere with proper telomeric replication. It has been shown that BLM plays an important role in telomere metabolism. Specifically, BLM localizes to telomeres31 and physically interacts with shelterin proteins. Both POT1 and TRF232,33,34 affect BLM activity, probably supporting BLM in tha unwinding of telomeric G4 structures during replication. The observeddecrease in the replication speed of the G-rich telomeric strand under BLMdeficient conditions is supported by several studies35,36,37,38. Molecules that trap and stabilize G4 structures cause a delay in telomeric replication39,40. Defective BLM function is accompanied by both telomere fragility41 and telomere shortening31,42. In addition to telomeres, BLM is involved in resolving G4 structures during replication at other genomic loci. Lansdorp and colleagues43 showed that SCEs in BLM defective cells are not randomly distributed but rather overlap with genes rich in G4 motifs. This suggests that G4 motif-rich strands have a higher propensity to form G4 structures during transcription44,45.
Biallelic mutations in the FANCJ/BRIP1 gene are present in a small subset of patients affected by the autosomal recessive cancer-predisposing Fanconi Anemia syndrome, characterized by a hypersensitivity to inter-strand cross-link-inducing agents, spontaneous chromosome instability and SCEs, albeit at a lesser frequency compared to individuals with BS21. Additionally, mutations in FANCJ are frequently observed in breast and ovarian cancer46,47,48, indicating its role as a tumor suppressor gene49,50,51 that is also supported by its physical interaction with BRCA148. FANCJ is unique among FA proteins due to the presence of an enzymatic conserved N-terminal helicase domain, which is absent in the others52,53. In biochemical in vitro assays, FANCJ acts as an iron–sulfur (Fe–S) cluster-binding 5′–3′ ATP-dependent DNA helicase with a preference for duplex DNA47. Of note, FANCJ not only binds to unwind DNA structures but also resolves G4s in the processing of replication intermediates53,54,55. The link between genome maintenance and helicase activity of FANCJ is supported by the observation that FANCJ mutations found in FA patients are localized within the helicase core domain. Additionally, cells mutated in FANCJ show hypersensitivity to replication inhibitors21,56. Moreover, mutations associated with cancer are often located in the Fe–S domain of FANCJ57. Both the cluster and the intact helicase domain are essential for the proper processing of G4 structures53,57. FANCJ-deficient cells display sensitivity towards G4-stabilizing agents such as CX-5461, pyridostatin (PDS), and telomestatin (TMS)53,57,58, supporting a model in which the helicase function of the protein is necessary for resolving G4 secondary structures during DNA replication57,59.
In this manuscript, we investigate the roles of BLM and FANCJ helicases in resolving G4 structures at telomeres under replicative stress conditions induced by the telomeric G4-stabilizing agent, RHPS4. Our study focuses on the impact of RHPS4-induced replicative stress on telomeric and genomic replication in cells depleted of either BLM, FANCJ, or both proteins.
Results
Characterization of U251MG CRISPR/Cas9 edited clones
To evaluate the contribution of BLM to the response to replicative stress induced by the telomeric G4 ligand RHPS4, we generated BLM knockout (KO) genome edited U251MG cell lines using the CRISPR/Cas9 technique (Supplementary Fig. 1). The level of BLM protein in the clones obtained through genome editing (herein designated as A2, A9, A12 and C2), was evaluated by western blot (WB) (Supplementary Fig. 2a, b). The analysis revealed decreased BLM protein levels in all clones, whereas complete knockout of the helicase was observed only in clone A9 (Supplementary Fig. 2a). We further evaluated residual BLM expression in all clones by immunofluorescence, confirming the complete absence of BLM protein signal in the A9 clone (Supplementary Fig. 2c–g). To verify the lack of functionality of the BLM protein, we conducted the genomic-Sister Chromatid Exchange (G-SCE) assay, which is a well-known marker of BLM-deficient cells (Supplementary Fig. 3a–e represent wild type (WT), cl. A2, cl. A9, cl. A12, and cl. C2, respectively). The assay confirmed the absence of a functional BLM protein in three out of four analyzed clones, as evidenced by the significantly higher frequency of G-SCE events compared to WT cells. Clone A9 exhibited the highest frequency of G-SCE, which was approximately 16-fold higher than observed in U251MG WT, consistent with the complete KO of the protein (Supplementary Fig. 3f). Additionally, Chromosome Orientation (CO)-FISH staining (Supplementary Fig. 3g) confirmed the G-SCE frequency. Using CO-FISH, cis configurations (indicating even exchanges) were used to estimate SCE frequency, as previously reported60 (Supplementary Fig. 3h). Based on these results, the A9 clone, hereafter referred to as BLM−/−, was chosen from among the analyzed clones and utilized in all the experiments conducted throughout this study.
The response of the BLM−/− cell line to replicative stress-inducing drugs
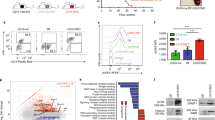
To evaluate the recruitment of BLM to telomeres after RHPS4 treatment (as schematically represented in the experimental set-up reported in Fig. 1a), we performed a co-immunofluorescence (Co-IF) assay using an anti-BLM antibody combined with anti-TRF1 or anti-TRF2 antibodies in the parental U251MG cell line (Fig. 1b). Data obtained showed that RHPS4 induced a concentration-dependent increase in BLM localization to telomeres, as evidenced by the colocalization of BLM with TRF1/2. The recruitment of the BLM protein to telomeric regions was significantly increased following a 96-h RHPS4 0.5 μM treatment (about a 3-fold increase for both TRF1 and TRF2) (Fig. 1c). To analyze the antiproliferative effect of RHPS4 on BLM−/− cells, a dose-response curve was performed. The evaluation was conducted at 96 h after treatment with RHPS4 or with MMC, a well-known replicative stress-inducing agent used as a positive control (Fig. 1d, e). Data confirmed that BLM−/− cells were more sensitive to MMC than the parental cell line, specifically within the range of 0.1–3 μg/ml. However, no differences in proliferation rates were observed upon RHPS4 treatment. Interestingly, HU and CPT treatmnents were also ineffective in BLM−/− U251MG cells (Supplementary Fig. 4a, b), which is consistent with previous research61,62,63. Results obtained from RHPS4 dose-response curve experiments performed 96 h after treatments showed no difference in sensitivity between the parental U251MG and the BLM−/− cell line, indicating no antiproliferative effect of the G4 ligand in the BLM-depleted cell model. To further support this result, both short- (96 h) and long-term (21 days) growth curves were performed (Fig. 1f, g). Overall, the data obtained did not demonstrate an increased sensitivity of BLM-depleted cells to the G4 ligand. Specifically, the short-term growth curves revealed that RHPS4 inhibited cell proliferation to a similar extent in both the parental and BLM−/− cell lines. This observation was statistically supported, with significant results showing a p value lower than 0.0001 in the parental cell line and a p value of 0.018 in the BLM-depleted cell line, as observed 96 h after treatment (Fig. 1f). Furthermore, the long-term growth curves (Fig. 1g) confirmed that the depletion of BLM did not affect cell proliferation in response to RHPS4.
a The diagram shows the workflow on a timeline: on the day after cell seeding, cells were treated with RHPS4 (0.2 and/or 0.5 μM). At 48 h after treatment, cells were again treated with RHPS4 (0.2 and/or 0.5 μM) or harvested and counted. At 72 h after the first treatment, the cell viability was checked, and finally at 96 h after the first treatment, the cells were harvested and processed. b Representative images showing U251MG cells stained by immunofluorescence using anti-BLM (red signals), and anti-TRF1 or anti-TRF2 (green signals) antibodies. Merged images allow visualization of colocalizing dots (yellow signals). Yellow arrows indicate BLM and TRF1-TRF2 colocalizations. c Quantification of the colocalizations between BLM and TRF1 or TRF2 proteins in U251MG cell line upon RHPS4 treatment (0.2 and 0.5 μM). d, e Sensitivity of U251MG and BLM−/− cell lines to RHPS4 concentrations ranging from 0.01 to 2 μM (0.01; 0.125; 0.25; 0.5; 1; 2 μM), evaluated 96 h after the first treatment. Mitomycin C (MMC) was used as a positive control at concentrations of 0.1; 0.5; 1; 2; 5 μg/ml. The Sulforhodamine B (SRB) cytotoxicity assay showed that RHPS4 sensitivity was unchanged in U251MG and BLM−/− (IC50 was 0.56 μM in both cell lines). f Short-term cell proliferation in untreated cells, U251MG and BLM−/−, and in RHPS4-treated cells (0.5 μM) as evaluated 48 and 96 h after the first treatment. g Long-term cell proliferation in U251MG and BLM−/− untreated and RHPS4-treated cells (0.5 μM). Scale bars represent 5 μm. *p < 0.05, **p < 0.01 (two-way ANOVA; n = 3). Error bars denote the standard deviation of the mean.
FANCJ silencing impacts cell proliferation in U251MG WT and BLM−/− cell lines
The depletion of BLM did not result in an increased sensitivity to RHPS4. To investigate the contribution of FANCJ in G4 unfolding, we employed small interfering RNA (siRNA) to transiently silence the gene encoding FANCJ in both U251MG and BLM−/− cell lines (Fig. 2a). This allowed us to assess the individual role of FANCJ in G4 unfolding as well as its combined effect with BLM helicase depletion. The FANCJ silencing was assessed by WB experiments (Fig. 2b, f), which confirmed a significant reduction in protein levels in both U251MG (about an 80% reduction) and BLM−/− cells (about a 90% reduction) (Fig. 2c, g). Short-term growth curves (48 and 96 h after treatment) were performed to determine whether FANCJ silencing affected the response of U251MG and BLM−/− cells to RHPS4 in terms of cell proliferation (Fig. 2d, h). As demonstrated in previously published works64, a concentration of 0.5 mM RHPS4 reduced the proliferation of U251MG cells by 50% (Fig. 2d). BLM−/− cells did not exhibit increased sensitivity to RHPS4, showing a 50% reduction in proliferation, similar to the parental line. In contrast to BLM depletion, FANCJ silencing affected cell proliferation per se (50% reduction when compared to control cells), and under this condition, RHPS4 treatment completely inhibited cell growth (Fig. 2d). Conversely, cells lacking both BLM and FANCJ were unable to proliferate, and therefore no significant difference was observed after treatment with RHPS4 (Fig. 2h). Overall, the effect of RHPS4 treatment on cell proliferation was more pronounced in U251MG-FANCJ-silenced cells (Fig. 2e), indicating a more relevant role of FANCJ than BLM in resolving RHPS4-induced G4 stabilization (Fig. 2i). Similar results were also obtained using PDS as an alternative G4 ligand (Supplementary Fig. 5a, b).
a The diagram shows the workflow on a timeline: immediately after cells seeding, siFANCJ (25 nM) was added for 4 h. The day after, siFANCJ (25 nM) was newly added to adhesion cells and then treated with RHPS4 (0.5 μM). Twenty-four hours after, the cell viability was checked, and 48 h after RHPS4 treatment, siFANCJ (25 nM) was re-added, and the cells were re-treated with RHPS4 (0.5 μM) or harvested and counted. Seventy-two hours after the first treatment, the cell viability was checked again, and finally at 96 h after the first treatment, the cells were harvested, processed, and/or counted. b, f Western blot representative image of FANCJ protein level in control, siSCR, and siFANCJ U251MG and BLM−/− cells, respectively. c, g Western blot analysis of the FANCJ protein in U251MG and BLM−/−, respectively. c, g Quantification of protein levels showed a strong decrease in FANCJ protein expression in both U251MG-siFANCJ and BLM−/−-siFANCJ cell lines. No significant variations of FANCJ protein levels were observed in U251MG-siSCR and BLM−/−-siSCR cells. The graph shows the proliferation curve in U251MG and siFANCJ untreated (blue and burgundy, respectively), and RHPS4-treated cells (light blue and orange, respectively) (d), and in BLM−/− and BLM−/−-siFANCJ untreated (red and purple, respectively) and RHPS4-treated cells (light rose and fuchsia, respectively) (h). e The graph shows the treated/untreated ratio of U251MG (light blue) and siFANCJ (orange) cells. i The graph shows the treated/untreated ratio of BLM−/− (light rose) and BLM−/−-siFANCJ (fuchsia). *p < 0.05, **p < 0.01, ***p < 0.001 (Unpaired t-test; n = 3). Error bars denote the standard deviation of the mean.
FANCJ–BLM functional crosstalk in untreated and RHPS4-treated U251MG cells
Previous studies have shown that FANCJ depletion negatively impacts BLM protein stability21. To confirm this in our experimental system, we performed immunofluorescence (IF) and WB analyses in U251MG cells following FANCJ knockdown (siFANCJ). As shown in Fig. 3a, b, BLM nuclear foci were markedly reduced in siFANCJ cells compared to both parental U251MG and siSCR controls. Consistently, WB analysis revealed a strong decrease in BLM protein levels upon FANCJ silencing (Fig. 3c). To investigate the reciprocal effect, we analyzed FANCJ expression in BLM−/− cells by WB. Interestingly, unlike the reduction in BLM levels observed upon FANCJ depletion, loss of BLM led to an approximately fourfold increase in FANCJ protein levels (Fig. 3d). This suggests an asymmetric compensatory mechanism between the two helicases, which, to our knowledge, has not been previously reported. We next examined the basal interaction between FANCJ and BLM in U251MG cells and how it is affected by RHPS4 treatment. Proximity ligation assay (PLA) (Fig. 3e) revealed detectable basal interaction in untreated U251MG cells, which was absent in BLM−/− cells used as a negative control (Fig. 3f). Notably, RHPS4 treatment significantly enhanced the FANCJ–BLM interaction, supporting their coordinated function under G4-induced replication stress (Fig. 3f).
a Representative images of U251MG and siFANCJ cells immunostained using anti-BLM and anti-FANCJ antibodies (red and green signals, respectively). b The graph shows the frequency of BLM (red circles), FANCJ (green circles), and colocalization (orange circles) foci in untreated and RHPS4-treated U251MG, siSCR, and siFANCJ cells. *p < 0.05, ****p < 0.0001 (ordinary one-way ANOVA; n = 3). c Western blot showing BLM protein levels in U251MG and siFANCJ cells. d Western blot representative image and densitometric analysis of FANCJ protein level in U251MG and BLM−/− cells. *p < 0.05 (Unpaired t-test) (n = 3). e Representative PLA images of BLM−/−, U251MG, and RHPS4-treated U251MG cells. BLM−/− cells were used as controls. f Quantification of the PLA signal was significantly higher in RHPS4-treated than in untreated cells, indicating that the treatment induced an increase in FANCJ–BLM interaction. *p < 0.05, ****p < 0.0001 (ordinary one-way ANOVA, Dunnett’s post-test; n = 3). Error bars denote the standard deviation of the mean.
FANCJ silencing increases the frequency of chromosome and chromatid aberrations, independently of RHPS4 treatment
Given the significant impact of FANCJ silencing on cell proliferation, we aimed to investigate whether this decrease in cell growth correlated with any chromatid or chromosome damage. To accomplish this, we conducted chromosomal analysis using pancentromeric and telomeric FISH staining (Fig. 4a–d). We found a significant increase in chromatid breaks after FANCJ silencing in both U251MG and BLM−/− cell lines (Fig. 4e, f). Specifically, cells with FANCJ-silenced exhibited a tenfold increase in chromatid breaks in the parental cell line and an eighteenfold increase in BLM−/− cells. Additionally, we observed a significant increase in chromosome breaks in cells where FANCJ was silenced. The increase was fivefold in the parental cells and a fourfold increase in the BLM−/− cells (Fig. 4g, h). Interestingly, treatment with RHPS4 did not induce further increases in either chromatid or chromosome breaks, in both parental and BLM-depleted cells, regardless of the presence of the FANCJ protein. It is worth noting that BLM depletion alone did not have a significant impact on the frequency of chromatid and chromosome aberrations, which is consistent with previously reported cell proliferation rate experiments.
a–d Representative images of telomeric/pancentromeric FISH-stained metaphases in U251MG, BLM−/−, siFANCJ, and BLM−/−-siFANCJ cell line, respectively. Magnified images showing chromatid and chromosomal breaks (c, d). e The graphs show the frequency of chromatid breaks in U251MG and siFANCJ untreated and RHPS4-treated cells. f The frequency of chromatid breaks in BLM−/− and BLM−/−-siFANCJ untreated and RHPS4-treated cells. g Chromosomal breaks frequency in U251MG and siFANCJ untreated and RHPS4-treated cells. h Chromosomal breaks frequency in BLM−/− and BLM−/−-siFANCJ untreated and RHPS4-treated cells. Scale bars represent 10 μm. *p < 0.05, **p < 0.01, ***p < 0.001 (ordinary one-way ANOVA; n = 3). Error bars denote the standard deviation of the mean.
FANCJ localizes to pericentromeres, subtelomeres, and telomeres in response to RHPS4
Since the effect of RHPS4 on cell proliferation in siFANCJ cells was not mediated by the induction of chromatid or chromosome aberrations (Fig. 4), we investigated whether it could instead be due to defects in G4 resolution at specific highly repeated G-rich regions, such as subtelomeres/telomeres or pericentromeres. To explore this, we performed chromatin immunoprecipitation (ChIP) analysis to assess FANCJ localization in these repetitive and replication-challenging regions. The data indicated that, following RHPS4 treatment, FANCJ is recruited to subtelomeres but not to pericentromeres (Fig. 5a). Interestingly, in BLM−/− cells, we observed a high basal level of FANCJ at both subtelomeric and pericentromeric regions, suggesting enhanced recruitment of the protein in the absence of BLM. In contrast to U251MG WT cells, RHPS4 treatment in BLM−/− cells resulted in a significant reduction of FANCJ binding at both subtelomeric and pericentromeric regions (Fig. 5a). We next performed Co-IF experiments using FANCJ in combination with TRF1 and TRF2 antibodies to evaluate FANCJ recruitment to telomeric regions following RHPS4 treatment in both U251MG WT and BLM−/− cell lines (Fig. 5b). The recruitment of FANCJ to telomeres in the BLM−/− is approximately twice that observed in the parental cell line (Fig. 5c). RHPS4 treatment increases FANCJ recruitment to telomeres of U251MG cells, but not in BLM-depleted cells (Fig. 5c). These data suggest a role for FANCJ in telomeric G4 processing, which is further confirmed by immunofluorescence-FISH (IF-FISH) experiments using antibodies that recognize G4, pRPA and telomeric probes (Supplementary Fig. 6). The data confirmed that the absence of FANCJ, BLM, or their combined depletion led to an increased number of G4 signals per nucleus. Interestingly, colocalization between G4 structures and telomeric sequences was predominantly enriched in siFANCJ cells. In addition, although pRPA recruitment to telomeres was detected across all genetic backgrounds analyzed, it was more pronounced in siFANCJ cells (Supplementary Fig. 6). We next evaluated telomere dysfunction induced foci (TIF) upon RHPS4 treatment in the absence of BLM and FANCJ proteins (Fig. 5d, e). Data showed a significant increase in DNA damage in telomeric regions resulting from RHPS4 ligand treatment in both the U251MG WT and the BLM−/− cell lines (Fig. 5f, g). Furthermore, the absence of FANCJ-induced telomeric DNA damage supports the idea that FANCJ plays a role in telomeric stability. In this case, the highest level of telomeric DNA damage was observed in BLM-depleted cells lacking FANCJ. In double-depleted cells, treatment with the G4 ligand resulted in a slight but significant decrease in the frequency of telomeric DNA damage (TIFs), possibly due to the high mortality observed in these cells following treatment, which may selectively eliminate those with severe telomeric damage from the population. Notably similar data on telomere damage induction were also obtained using PDS (Supplementary Fig. 5c, d).
a ChIP-qPCR analysis shows FANCJ localization at subtelomeric and pericentromeric regions in untreated and RHPS4-treated U251MG and BLM−/− cells. b Representative images of U251MG and BLM−/− cells immunostained using anti-TRF1 and anti-FANCJ antibodies (red and green signals, respectively). Yellow arrows indicate TRF1 and FANCJ colocalizations (orange foci). c The graph shows the frequency of FANCJ-TRF1 colocalization in U251MG untreated and RHPS4-treated cells (blue) and in BLM−/− untreated and RHPS4-treated cells (red). *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001 (ordinary one-way ANOVA; n = 3). d, e Representative images of TIFs observed by immunofluorescence in untreated and RHPS4-treated U251MG, siFANCJ, BLM−/−, and BLM−/− siFANCJ cells. Single channels (TRF1 protein signals in red and γHA2X protein signals in green) and merged images are shown. White arrows indicate TRF1 and γHA2X colocalizations (orange foci). TRF1 and γHA2X colocalizations were indicated as telomere dysfunction-induced foci (TIFs). f The graph shows the frequency of TIFs in untreated and RHPS4-treated U251MG and siFANCJ cells. g The graph shows the frequency of TIFs in BLM−/− and BLM−/−-siFANCJ untreated and RHPS4-treated cells. Scale bars represent 5 μm. *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001 (Mann–Whitney test; n = 3). Error bars denote the standard deviation of the mean.
Both BLM and FANCJ depletion induce telomere abnormalities in the U251MG cell line
FISH experiments were conducted to analyze the frequency of telomere doublets and telomere loss. Telomere doublets are a well-known marker of telomeric replicative stress, while telomere loss may be generated by telomere trimming, especially in the absence of telomeric helicases (Fig. 6a–d). Data showed that FANCJ silencing resulted in both telomeric doublets and telomere loss in both WT and BLM−/− cells. In BLM WTcells (Fig. 6e, g), RHPS4 treatment further increased the frequency of telomeric doublets and telomere loss. This effect was less pronounced in BLM-deficient cells (Fig. 6f, h). Interestingly, in BLM-depleted cells, the overall frequency of telomere doublets was comparable to that observed in the WT cell line.
a–d Representative images of telomeric/pancentromeric FISH-stained metaphases in untreated and RHPS4-treated U251MG, BLM−/−, siFANCJ, and BLM−/−-siFANCJ cell lines, respectively. Detailed images showing telomeric doublets and telomere loss. e The graph shows the frequency of telomeric doublets in U251MG and siFANCJ untreated cells and RHPS4-treated cells. f The frequency of telomeric doublets in BLM−/− and BLM−/−-siFANCJ untreated and RHPS4-treated cells. g The graph shows the frequency of telomere loss in U251MG and siFANCJ untreated and RHPS4-treated cells. h The frequency of telomere loss in BLM−/− and BLM−/−-siFANCJ untreated and RHPS4-treated cells. Scale bars represent 10 μm. *p < 0.05, ***p < 0.001, ****p < 0.0001 (unpaired t-test; n = 3). Error bars denote the standard deviation of the mean.
Discussion
The present work investigates the role of BLM and FANCJ helicases in the response to telomeric replicative stress induced by G4 stabilization. Telomeres are known to behave as fragile sites and are particularly susceptible to replicative stress, requiring a complex interplay of proteins that act to avoid replication fork stalling/collapse and subsequent formation of DSBs in telomeric regions41,65. In this regard, BLM plays a significant role in resolving replicative stress at telomeres by aiding in the resolution of G4 structures and ensuring proper replicative fork progression31,38.
Given BLM’s ability to resolve G4 structures in vitro29,66,67, it has been suggested that BLM could facilitate telomere replication by eliminating these impediments from the lagging-strand ([TTAGGG]n) telomeric template. Supporting this notion, the depletion of BLM predominantly induces a fragile telomere phenotype at lagging-strand telomeres and enhances telomeric G4 signals68,69. This evidence is supported by the observation that telomeric G4 ligands, such as RHPS4, induce severe telomere abnormalities in BLM-silenced normal fibroblasts, suggesting that, under replicative stress conditions, telomeric replication is further hindered by the absence of BLM protein70.
To evaluate BLM’s role in responding to G4 stabilization, we used CRISPR/Cas9 genome editing in U251MG glioblastoma cells. Given the well-known role of BLM at telomeres upon induction of telomeric RS, we tested if its depletion increased sensitivity to the telomeric stressor RHPS4. Our data showed that although telomere G4 stabilization promotes BLM recruitment at telomeres, its absence did not impact cellular sensitivity to the G4 ligand. It is worth noting that BLM-depleted cells did not exhibit increased sensitivity to replicative stress-inducing agents, such as HU and CPT (Supplementary Fig. 4), in terms of proliferation inhibition, which is consistent with previously published papers61,63.
To explain this evidence, it is important to consider the acknowledged functional redundancy among helicases, suggesting that other helicases, such as WRN, may compensate for the absence of one with similar functions. In contrast, treatment with the crosslinking agent MMC reduces cell proliferation in BLM-depleted cells compared to controls. Previous studies71 have indicated that BLM is required to convert MMC-induced DNA damage, likely DNA ICL, into DSBs. These lesions are then repaired through DSB repair pathways, such as HR in replicating cells72. This means that cells lacking BLM have defects in the first step of HR-mediated repair of ICL that finally impact cell proliferation. Hence, BLM plays a crucial role in MMC DNA damage processing, while it appears to be dispensable for the response to telomeric G4 stabilization. This suggests that BLM plays a more relevant role in the response to overall genomic DNA damage than to telomeric damage and that other proteins take over BLM’s role at telomeres following G4 stabilization, resulting in no effect on proliferation.
Several studies have explored the interaction between FANCJ and BLM helicases, demonstrating that they act synergistically to unwind damaged DNA substrates or G4 structures21. It is widely accepted that these helicases have common or partially overlapping functions in preserving genomic stability20. The physical interaction between BLM and FANCJ may enhance the collective functioning of the dual helicase motor, allowing it to operate more efficiently in its unwinding capacity, especially in the presence of damaged DNA or specific DNA structures (such as G4). The coordinated action of a 3′ to 5′ DNA helicase (BLM) alongside a 5′ to 3′ helicase (FANCJ) presents a distinctive mechanism for unwinding structured nucleic acids73. Moreover, it was also shown that FANCJ stabilizes BLM protein and, as a consequence, FANCJ silencing leads to BLM protein decrease21.
Notably, FANCJ has a specific function, and mutations in FANCJ result in cellular deficiencies that extend beyond a diminished capacity for ICL repair. These deficiencies include a broader range of challenges associated with various forms of DNA structural impediments to replication53. To evaluate whether combined depletion of BLM and FANCJ increases sensitivity to stabilized G4 structures, FANCJ was transiently silenced in both U251MG and BLM−/− cells. The results indicate that FANCJ and BLM are not epistatic, as evidenced by the observation that double-depleted cells exhibited a markedly severe basal growth inhibition, greater than that observed in either single depletion. Similar results were obtained with PDS, another well-known G4 ligand capable of inducing replication stress.
In vitro and in vivo studies have demonstrated a physical and functional interaction between BLM and FANCJ21,53,54. Importantly, FANCJ has been shown to work in tandem with the BLM helicase to preserve the epigenetic stability of DNA sequences that form G4 structures74. Our experiments confirmed that FANCJ silencing negatively affects BLM protein levels, reducing BLM availability at RS loci. Interestingly, we observed that the reverse is not true as BLM depletion resulted an induction of FANCJ expression, suggesting a compensatory mechanism. Another notable finding was that RHPS4 treatment significantly enhanced the interaction between FANCJ and BLM, supporting the hypothesis that both helicases cooperate to manage G4-induced replicative stress. In light of these data, it is not surprising that U251MG-siFANCJ cells appear to be more sensitive than siSCR cells and BLM−/− to RHPS4 treatment. This finding is consistent with previously published data indicating that FANCJ-depleted cells are sensitive to the telomeric G4 ligand TMS75 and CX-546176,77 and that FANCJ unwinds telomeric G4 structures in vitro53. The interaction and functional interplay between FANCJ and BLM have been well documented in the literature20. To investigate whether cell proliferation inhibition could be attributed to chromosomal damage, we analyzed the induction of chromosomal aberrations in helicase-depleted cells. Data showed that BLM depletion did not significantly induce chromatid or chromosome breaks in either the control or the RHPS4-treated samples. However, a significant increase in chromatid and chromosome breaks was observed in FANCJ-silenced samples, regardless of RHPS4 treatment. These data explain the more severe reduction of cell proliferation observed in FANCJ knockdown cells when compared to BLM−/− cells and suggests that the decreased cell proliferation in U251MG was not due to genome-wide induction of DNA damage. Indeed, FANCJ depletion, more than BLM or double depletion, led to a significant increase in telomeric G4 stabilization and RPA recruitment to telomeres, as observed in IF-FISH experiments. These findings confirm the relevance of RPA/FANCJ and RPA/BLM interplay and suggest that RPA loading occurs independently of helicase recruitment78,79. We therefore investigated whether FANCJ may have a specific role in G-rich, hard-to-replicate genome regions such as subtelomeres/telomeres or pericentromeres. It is worth noting that, although FANCJ’s telomeric role is not well characterized (with the exception of some evidence observed in alternative lengthening of telomeres (ALT)-positive cells80), FANCJ depletion has been associated with a high sensitivity to the telomeric ligand TMS in vitro and in vivo53,75.
Notably, in our experiments, RHPS4 treatment promoted the recruitment of FANCJ to subtelomeric and telomeric regions—but not to pericentromeres—in U251MG cells. This finding suggests that, independently of BLM function, FANCJ localizes to telomeric and subtelomeric regions in response to replicative stress occurring at these sites. Interestingly, in BLM−/− cells, FANCJ was constitutively enriched at both subtelomeric/telomeric and pericentromeric regions; however, this enrichment unexpectedly decreased following RHPS4 treatment. These results suggest that in the presence of BLM, FANCJ is specifically recruited to subtelomeres/telomeres experiencing replicative stress due to stabilized G4 structures. In contrast, BLM depletion leads to increased FANCJ recruitment at hard-to-replicate regions, regardless of RHPS4 treatment. The RHPS4-mediated reduction in FANCJ localization at these regions in BLM−/− cells remains unclear. However, we hypothesize that it may reflect a redistribution of FANCJ from constitutive difficult-to-replicate regions to newly emerging sites of genome-wide replication stress induced by RHPS4.
Telomere dysfunction was assessed in cells lacking one and/or both BLM and FANCJ helicases, both in the presence and absence of RHPS4-mediated telomeric replicative stress. Intriguingly, depletion of either FANCJ or BLM led to a significant increase in TIFs compared to U251MG parental cells. The highest induction of TIFs occurred in double-depleted cells (p < 0.0001), confirming distinct roles for the two helicases at telomeres. As expected, RHPS4 significantly induced TIFs in various genetic backgrounds analyzed, except in double-depleted cells, where TIFs slightly decreased following RHPS4 treatment. This decrease may be due to the induction of cell death, as suggested by the observed inhibition of cell proliferation in RHPS4-treated double-depleted cells.
In addition to FANCJ, BLM and WRN have also been detected at telomeres in ALT-positive cells80, thus, it is possible that BLM and FANCJ work together to eliminate G4 structures from the telomere leading G-rich strands. Alternatively, to bypass G4 structures on the leading strand, BLM and WRN can employ a fork regression/restoration mechanism instead of directly unwinding the impediment, as shown by their ability to reverse replicative forks in vitro36,37. However, this mechanism may result in the loss of telomeric DNA38. Interestingly, although this model proposes that the two helicases collaborate in resolving G4 structures in telomeric regions, counteracting replicative stress, our data support the notion that FANCJ maintains essential BLM-independent functions in counteracting telomeric replicative stress.
Finally, we examined the occurrence of telomeric doublets and the induction of telomere loss as measures of replicative stress or fragile telomeres and telomeric DNA breakage, respectively. Consistent with the TIFs analysis, depletion of both FANCJ and BLM resulted in the induction of telomere loss, indicating that the absence of helicases leads to a replication defect in telomeric regions, causing DNA damage and loss of blocks of telomeric repeats. RHPS4 also produced the same effect in helicase-proficient cells, indicating that resolving G4 structures is a significant challenge in telomere replication.
Analysis of telomeric doublets in siFANCJ cells revealed a significantly higher frequency compared to controls, a pattern not observed in BLM-depleted cells. This finding contrasts with previous reports in the literature, which demonstrated an increased frequency of doublets in BLM-deficient cells41,69. However, this apparent discrepancy may be explained by the elevated rates of telomere loss observed in BLM−/− cells, another telomeric anomaly associated with BLM deficiency, reflecting results previously reported in BLM-deficient human fibroblasts31.
These findings further support the hypothesis that FANCJ and BLM helicases have only partially overlapped roles at telomeres and emphasize the significant role of FANCJ, even in telomerase-positive (ALT-negative) cells like U251MG. In siFANCJ cells, the frequency of telomeric doublets increased even more following RHPS4 treatment, reaching far higher levels than in other samples, confirming FANCJ’s crucial function in resolving telomeric G4 structures. Based on our findings, we conclude that there is partial redundancy between BLM and FANCJ helicases in resolving telomeric replicative stress induced by G4 stabilization. Notably, the impact of FANCJ silencing on overall genome stability was greater than that of BLM. As suggested in the literature21 and confirmed in our cellular model, this may be due to FANCJ silencing reducing BLM protein levels, resulting in decreased levels of both helicases, whereas BLM depletion activates compensation mechanisms leading to increased protein levels of FANCJ. Notably, the absence of FANCJ also had a greater effect on telomeric stability, leading to an increased number of telomeric defects. This unexpected finding reveals a previously unrecognized role for FANCJ in resolving replicative stress within the telomeric regions of non-ALT cells.
It is noteworthy that recent observations have shown reduced poly (ADP-ribose) polymerase 1 (PARP1) activity during the S phase in FANCJ-deficient cells, as PARP1 becomes “sequestered” in the chromatin, deviating from the typical chromatin trapping of PARP181. It is also known that inhibition of PARP1 affects the recruitment of certain helicases, including BLM and WRN, to TRF1-containing complexes during the S phase, leading to replication-associated DNA damage and telomere fragility82. PARP1 is also enriched in telomeric chromatin during G4 stabilization to resolve replication-induced damage70,83 and therefore has been recently proposed as “surveillant” of telomere replication82. In line with this model, FANCJ-deficient cells exhibit an increased accumulation of G4 structures, which are PARP1 substrates84,85 resulting in greater PARP1 sequestration and diminished activity. In addition to the direct telomeric role of FANCJ depletion, this observation offers an alternative explanation for our findings and suggests that PARP1 may contribute to the telomeric effects observed upon FANCJ silencing.
In conclusion, it is interesting to highlight that FANCJ and double-depleted cells are more sensitive to G4 stabilizer treatment, as also reported in the literature for TMS treatment. This finding could have positive implications for the development of new therapeutic anti-cancer strategies in the clinical setting, potentially paving the way for the use of helicase inhibitors in combination with G4-stabilizing compounds.
Materials and methods
Cell lines and culture conditions, and siRNA transfection
Media, supplements, and plasticware for cell culture were purchased from Euroclone (Euroclone, Pero, MI, Italy). U251MG cells (grade IV human glioblastoma) from ECACC – IRCCS-IST, Genova ITA were routinely maintained in minimum essential medium (MEM) with Earle’s balanced salt solution (EBSS) supplemented with 10% fetal bovine serum, 2 mM L-glutamine, 1 mM sodium Pyruvate, 1% non-essential amino acids (NEAA) (Sigma-Aldrich, Co., St. Louis, MO, USA), 100 units mL−1 penicillin, active against Gram+ bacteria and 100 μg mL−1 streptomycin, active against Gram− bacteria. U251MG was proven to be telomerase positive based on previously published results86. U251MG-BLM KO cell lines were obtained through the genomic editing technique CRISPR/Cas9 on the BLM gene (details on CRISPR/Cas9 gene targeting were included in Supplementary Materials and Methods, and characterization of the cell clones was reported in Supplementary Figs. 1–3). BLM KO cell lines were maintained in culture and treated with the same conditions as the U251MG WT cell line. All cell lines were maintained under sterile conditions at 37 °C in a 5% CO2 and 95% air atmosphere and were divided whenever they reached subconfluence (about two times a week). Their doubling time, DT, under these conditions is about 32 ± 2 h.
Silencing of the FANCJ gene in U251MG and BLM−/− cells was achieved via transient transfection of FANCJ#1 GUACAGUACCCCACCUUAUUU50, FANCJ#2 AAACAGCAAGCAACAUUGUUU87 (Sigma-Aldrich) oligos (see Table 1) at a final concentration of 25 nM and the Lipofectamine RNAiMAX transfection reagent (Thermo Fisher Scientific, Waltham, MA, USA), according to the manufacturer’s instructions. Cells silenced for FANCJ were subjected to triple transfection: “reverse” (cells in suspension) for 4 h on day 1, “forward” (adherent cells) on day 2, and again in adhesion on day 4. The cells were then processed and analyzed 120 h after the first transfection. Control cells were transfected with all the reagents except for the oligos.
Chemical compounds and treatments
The pentacyclic acridine, 3,11-difluoro-6,8,13-trimethyl-8Hquino [4,3,2-kl] acridinium methosulfate (RHPS4) (Tocris, Bristol, UK), was dissolved in dimethyl sulfoxide (DMSO) as well as Mitomycin C (MMC) (Sigma-Aldrich) to a final concentration of 10 mM and 1 mM, respectively. Drugs were prepared from fresh frozen aliquots for each set of experiments.
If not differently stated, for each experiment involving RHPS4 treatment, cells were seeded in complete growth medium and treated at 0.5 μM (IC50 as calculated in SRB assay 96 h after treatment) and then fixed or harvested 48 and/or 96 h after treatment.
Hydroxyurea (HU) (Sigma-Aldrich), Camptothecin (CPT) (Sigma-Aldrich), and Pyridostatin (PDS) (SelleckChem, Houston, TX, USA) were dissolved in dimethyl sulfoxide (DMSO) to a final concentration of 1 M, 10 mM, and 1.4 mM, respectively. Drugs were prepared from fresh frozen aliquots for each set of experiments.
HU and CPT treatments in Sulforhodamine B (SRB) assay were performed using the following concentrations: 0.25; 0.5; 1; 2; 4 mM (HU); 31.25; 62.5; 125; 250; 500; 1000 nM (CPT) (Supplementary Fig. 4).
For all experiments involving PDS, cells were seeded and treated with the IC50 (2 μM) calculated from a 96-h proliferation dose-response assay (Supplementary Fig. 7). Cells were either harvested and counted at 48 and 96 h post-treatment for growth curve analysis or fixed at 96 h for TIF experiments (Supplementary Fig. 5).
Immunofluorescence
Cells were seeded at a density of 15,000 cells/cm2 on sterilized 22 × 22 mm slides in 35-mm Petri dishes. The slides were washed in phosphate-buffered saline 1X (PBS) and fixed in 4% paraformaldehyde (PFA) (Sigma-Aldrich) for 15 min on ice. After three washes in 1X PBS, cells were permeabilized in 0.2% Triton X-100 in 1X PBS for 5 min on ice and then washed three times in 1X PBS. The slides were then incubated in blocking buffer (1% BSA dissolved in 1X PBS [w/v]) and then incubated overnight (ON) at 4 °C with the primary antibodies (rabbit polyclonal anti-BLM (#A300-110A, Bethyl) (1:100); mouse monoclonal anti-TRF1 (4E4 clone, GTX70304, GeneTex) (1:20) or mouse monoclonal anti-TRF2 (9F10 clone, sc-47693, Santa Cruz Biotechnology) (1:100)). The day after, the slides were washed three times in 1% BSA and then incubated with secondary antibodies (Alexa Fluor 488 goat anti-mouse (Invitrogen) and Alexa Fluor 546 goat anti-rabbit (Invitrogen)) diluted in blocking buffer. Finally, slides were washed in 1% BSA, then once in 1X PBS and counterstained with 4′,6-diamidino-2-phenylindole (DAPI) 1 μg/mL (Sigma-Aldrich) in antifade solution Vectashield (Vector Laboratories, Burlingame, CA, USA). The images were acquired with an Axio Imager.M1 fluorescence microscope (Carl Zeiss, Germany) equipped with a FITC filter (λ excitation: 494; λ emission: 523) and a Cy3 filter (λ excitation: 544; λ emission: 566) and analyzed using ISIS software (Metasystems, Milano, Italy).
The same protocol was applied for the primary antibodies mouse monoclonal anti-BLM (B-4 clone, sc-365753, Santa Cruz Biotechnology) (1:100) in combination with a rabbit polyclonal anti-FANCJ (#B1310, Sigma-Aldrich) (1:50) which were used for the BLM-FANCJ co-immunofluorescence; rabbit polyclonal anti-FANCJ (#B1310, Sigma-Aldrich) (1:50) in combination with mouse monoclonal anti-TRF1 (4E4 clone, GTX70304, GeneTex) (1:20) which were used for the FANCJ-TRF1 co-immunofluorescence; rabbit polyclonal anti-TRF1 (H-242 clone, sc-5596, Santa Cruz Biotechnology) (1:100) in combination with a mouse monoclonal anti-γH2AX (JBW301 clone, #05-636, Millipore) (1:100) to perform TIF (telomere dysfunction-induced foci) co-immunostaining both in U251MG and BLM−/− cell lines. The frequency of BLM-FANCJ or TRF1-2/BLM-FANCJ or TRF1/γH2AX colocalization per cell was scored in 100 nuclei in three independent experiments.
Sulforhodamine B (SRB) assay
Exponentially growing U251MG and BLM−/− cells were harvested, counted, and seeded in a 96-well plate (1500 cells/well). Optimal seeding density was experimentally determined to ensure exponential growth during a 5-day assay (120 h). Treatments were performed using the following concentrations: 0.01; 0.1; 0.125; 0.2; 0.25; 0.5; 1; 2 μM (RHPS4); 0.1; 0.5; 1; 2; 5 μg/ml (MMC).
The SRB assay was performed as previously described88, with minor modifications. Cells were fixed in 10% cold trichloroacetic acid (TCA), incubated at 4 °C for 1 h, and then washed with deionized water. Cells were stained with 200 µl/well of 0.1% SRB (Sigma-Aldrich) for 30 min and washed four times with 1% acetic acid. Plates were air-dried at room temperature (RT), and stained proteins were solubilized with 200 µl/well of 10 mM unbuffered Tris base (tris(hydroxymethyl) aminomethane) (Sigma-Aldrich). Optical density was read at 530 nm with a Victor plate reader (VICTOR X3 Multilabel plate reader, PerkinElmer, Waltham, MA, USA). Experiments were repeated five times.
Short- and long-term proliferation assessment
To assess short-term proliferation rate, 15,000 cells/cm2 were seeded in 35-mm Petri dishes and exposed to RHPS4 IC50 (0.5 μM). Two (48 h) and four (96 h) days following treatment, cells were washed two times with 1X PBS, detached by trypsin, and counted in a Burker hemocytometer counting chamber. To determine the long-term proliferation rate, 100,000 cells of U251MG and BLM−/− cell lines were seeded and grown for 21 days with two intermediate passages after 7 and 14 days of culture. After harvesting, cells were counted using the Burker hemocytometer counting chamber and reseeded (100,000 cells in 10-mm Petri dishes). Each time the cells were reseeded, they were exposed to RHPS4 IC50 (0.5 μM). The cumulative population doubling level (cPDL) after 7, 14, and 21 days was calculated as follows: cPDL = log 2(Nf /N0), where Nf is the final cell number, and N0 is the initial number of seeded cells. The experiments were repeated three times.
Western blot
Cells were lysed in 10 mM Tris-HCl, pH 7.4, 100 mM NaCl, 1 mM EDTA, 1% Triton-100X, 0.1% SDS, 0.5% deoxycholate, and protease inhibitors. Protein extracts (15 μg) were loaded on an SDS-PAGE and transferred onto a polyvinylidene fluoride (PVDF) membrane (pore size 0.45 μm; Immobilion-P, Millipore, Massachusetts, MA, USA). Filters were blocked with 3% BSA dissolved in Tris-buffered saline (TBS) with 0.05% Tween 20 (TBS-T) for 40 min at RT. Membranes were then incubated at 4 °C ON with the following primary antibodies: rabbit polyclonal anti-BLM (#A300-110A, Bethyl) (1:500); rabbit polyclonal anti-FANCJ (#B1310, Sigma-Aldrich) (1:1000), and mouse monoclonal anti-Vinculin (#V9131, Sigma-Aldrich) (1:1000). Finally, membranes were incubated 1 h at RT with the appropriate HRP-conjugated secondary antibody (Bio-Rad, Hercules, CA, USA). Proteins were visualized by the ChemiDoc enhanced chemiluminescence detection system (Bio-Rad). Experiments were repeated at least three times. The images were analyzed with Image Lab (Bio-Rad).
Proximity ligation assay (PLA)
U251MG and BLM−/− cells were seeded on coverslips and treated with RHPS4 as described above or left untreated. Then, cells were fixed in 2% formaldehyde and permeabilized in 0.25% Triton X-100 in PBS for 5 min at RT. Successively, samples were processed for immunolabeling with mouse monoclonal anti-BLM (B-4 clone, sc-365753, Santa Cruz Biotechnology) and rabbit polyclonal anti-FANCJ (#B1310, Sigma-Aldrich) antibodies. PLA was performed by using the DUOLINK® In situ detection reagents Red (Sigma-Aldrich) following the manufacturer’s instructions. Finally, coverslips were counterstained with DAPI, and images were acquired with a CrestOptics V3 confocal spinning disk mounted on a Nikon Ti2-E Inverted microscope with an integrated camera and a laser source (Lumencor). PLA signals within each nucleus were quantified by Cell Profiler software. Experiments were repeated at least three times.
Collection of chromosome spreads
Chromosome spreads were obtained following 4 h incubation in 5 × 10–6 M colchicine (Sigma-Aldrich). Spreads of chromosomes were prepared following a standard procedure consisting of treatment with a hypotonic solution (75 mM KCl) for 20 min at 37 °C, followed by fixation in freshly prepared Carnoy solution (3:1 v/v methanol/acetic acid). Cells were then dropped onto clean slides, air-dried, and utilized for cytogenetic analysis.
Pancentromeric and telomeric FISH
Pancentromeric and telomeric FISH staining was performed as previously described89,90,91. Briefly, 48 h after the seeding, slides were rinsed with PBS pH 7.5 and fixed in 4% PFA for 2 min. After two rinses in PBS, slides were incubated in acidified pepsin solution for 10 min, rinsed, and dehydrated through graded alcohols. Slides and probes were co-denatured at 80 °C for 3 min and hybridized for 2 h at RT in a humidified chamber (probes were reported in Table 2). After hybridization, slides were washed twice for 15 min in 70% formamide, 10 mM Tris-HCl at pH 7.2, and 0.1% BSA, followed by three 5-min washes in TBS/Tween 20 0.08%. Slides were then dehydrated with an ethanol series and air-dried. Finally, samples were counterstained with DAPI in Vectashield (Vector Laboratories, Burlingame, CA, USA). Images were captured at 63× magnification with an Axio Imager Z2 (Carl Zeiss, Germany) equipped with a charge-coupled device camera, and the telomere was analyzed with ISIS software (MetaSystems, Milano, Italy)86,92. For each sample, at least 100 metaphases have been analyzed in three independent experiments.
Chromatin immuno-precipitation (ChIP)
U251MG and BLM−/− cells were seeded and treated with RHPS4 0.5 μM. At the end of treatments, cells were crosslinked by adding formaldehyde 1% directly to culture medium for 10 min at RT. Cell pellets were lysed in nuclei isolation buffer (5 mM PIPES, pH 8, 85 mM KCl, 0.5% NP-40), then nuclei were pelleted by centrifugation and lysed in nuclei lysis buffer (50 mM Tris-HCl, pH 8, 10 mM EDTA, 1% SDS), both supplemented with protease inhibitors. Sonicated chromatin (60 μg/sample, sonicated at 7 × 1 min 50% in easy mode Bioruptor Pico Sonication device Diagenode) was immunoprecipitated (IP) at 4 °C ON with 4 μg of the rabbit polyclonal anti-FANCJ (#B1310, Sigma-Aldrich) or Rabbit IgG (Santa Cruz Biotechnologies) antibodies. 1% of chromatin was used as an Input sample. Crosslink was then reversed with NaCl 5 M, and DNA was extracted with the phenol-chloroform method. ChIP quantification was performed by qPCR assay using Luna SYBR Green Master Mix (New England Biolabs) in Quant Studio 3 (Thermo Fisher Scientific) (primers were reported in Table 3). The analysis was performed using the percent Input method. Experiments were repeated three times.
CRISPR/Cas9 gene editing
Human BLM KO cells were generated using CRISPR/Cas9 gene targeting of U251MG cells via NHEJ and HR-mediated repair. sgRNA was cloned into pCDNA5-H1-sgRNA (Addgene, Cambridge, MA, USA) and co-transfected with the plasmid pSpCas9(BB)-2A-Puro (PX459) (Addgene) encoding the Cas9-nuclease from S. pyogenes. Cells efficiently edited were selected with puromycin. Targeting of BLM in U251MG cells was carried out with the following sgRNA: GGGGACTGTTTACTGACTAC (Exon 7). To introduce stop codons in U251MG cells by HR-mediated repair, a donor cassette was used with the following sequence: GTCGGATCCTTTAAACCTTAATTAAGCTGTTGTAG. Clones derived from single cell lines were genotyped to determine successful targeting. DNA sequencing confirmed gene disruption in the A9 clone. BLM KO in single clones was also confirmed by quantification of protein levels by Western blotting. Kit used for the sequencing of the clones: pGEM®-T Easy Vector System I.
Primers for sequencing:
FW 5′-AATAAAAGTGATGCCAGTCTTCTTG-3′
REV 5′-CAGCAGTGCTTGTGAGAACATTT-3′
Sister Chromatid Exchange (SCE) assay
For each U251MG clone, 400,000 cells were seeded in 10-mm Petri dishes and, after 24 h, they were treated with BrdU (5′-bromo-2′-deoxyuridine, Sigma-Aldrich) at a final concentration of 15 μM for about 48 h, allowing them to replicate their DNA twice. Afterward, cells were collected, and chromosome spreads were prepared as described above. After squashing of metaphases spreads, 1 ml of Acridine Orange (AO) 0.01% was added to each slide and incubated for 5 min. AO was then removed, and 2 ml of Sørensen at pH 6.8 was added to each slide and left to acidify for 5 min. Subsequently, the slides were washed under running water, and finally, they were mounted using coverslips and analyzed under a fluorescence microscope using the ISIS - MetaSystem software (Metasystems, Milano, Italy).
Chromosome orientation fluorescence in situ hybridization (CO-FISH) analysis
U251MG clones were subcultured in the presence of BrdU (Sigma-Aldrich) at a final concentration of 2.6 × 10–5 M and were allowed to replicate their DNA once at 37 °C (about 24 h). Cells were then collected, and chromosome spreads were prepared as described above. CO-FISH was performed as previously described93 using a (TTAGGG)3 probe labeled with FITC and a (CCCTAA)3 probe labeled with Cy3 (Panagene, Yuseong-gu, Korea). Images were captured with an Axio Imager.M1 equipped with a CCD camera. SCEs were evaluated by scoring the frequency of trans and cis CO-FISH signals configuration as proposed by Cornforth and Eberle60. To correct cis frequencies for multiple crossovers, we used the following formula60,93:
which is equivalent to
in which the frequency of SCEs that would be necessary to account for the number of cis events represents the mean (μ) of a Poisson distribution, whose sum of odd-numbered terms equals the observed cis frequency (γ). The latter equation was used to convert the observed cis frequencies into estimates of the true crossover frequency, also referred to as the SCE equivalent frequency. For this purpose, 4000 chromosome ends were scored in three independent experiments.
IF/FISH
At the end of treatments, cells were fixed in 2% formaldehyde and permeabilized in 0.25% Triton X-100 in PBS for 5 min at RT. Then, samples were processed for immunolabeling with mouse monoclonal anti-G4 (BG4 clone, Ab00174-1.1, Absolute Antibody) or rabbit polyclonal anti-pRPA32 Ser33 (#A300-246A, Bethyl) antibodies, followed by the anti-mouse IgG or anti-rabbit IgG Alexa fluor 488 secondary antibodies (Cell Signaling). Then, samples were re-fixed in 2% formaldehyde, dehydrated with ethanol series (70, 90, 100%), air-dried, co-denatured for 3 min at 80 °C with a Cy3-labeled PNA probe, specific for telomere sequences (TelC-Cy3, Panagene, Daejon, South Korea), and incubated for 2 h in a humidified chamber at RT in the dark. After hybridization, slides were washed with 70% formamide, 10 mM Tris-HCl, pH 7.2, BSA 0.1%, and then rinsed in TBS/Tween 0.08%, dehydrated with ethanol series, and finally counterstained with DAPI (0.5 μg/ml, Sigma-Aldrich) and mounted on slides in mounting medium (Gelvatol Moviol, Sigma-Aldrich). Fluorescence signals were acquired with a CrestOptics V3 confocal spinning disk mounted on a Nikon Ti2-E Inverted microscope with an integrated camera and a laser source (Lumencor).
Statistical analysis and reproducibility
GraphPad Prism software (version 10.5.0; GraphPad Software Inc.) was used to perform statistical analyses. Data obtained from a minimum of three independent experiments are reported as the mean ± standard deviation. The significance of differences between the two groups was determined with a two-tailed unpaired Student’s t-test (Figs. 2, 3d, 5f, e and 6). For multigroup comparisons, one-way analysis of variance (ANOVA) with Dunnett’s test (Figs. 3b, f, 4 and 5a, c) or two-way ANOVA with Tukey’s test (Fig. 1c) was employed. For all analyses, a probability (p) value of <0.05 was considered statistically significant.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
All data supporting the findings of this study are available within the paper and its Supplementary Information. Uncropped western blots are included in the supplementary information file (Supplementary Figs. 8–10). All source data can be found in the supplementary data file.
References
Gellert, M., Lipsett, M. N. & Davies, D. R. Helix formation by guanylic acid. Proc. Natl. Acad. Sci. USA 48, 2013–2018 (1962).
Lipps, H. J., Gruissem, W. & Prescott, D. M. Higher order DNA structure in macronuclear chromatin of the hypotrichous ciliate Oxytricha nova. Proc. Natl. Acad. Sci. USA 79, 2495–2499 (1982).
Sundquist, W. I. & Klug, A. Telomeric DNA dimerizes by formation of guanine tetrads between hairpin loops. Nature 342, 825–829 (1989).
Sen, D. & Gilbert, W. Formation of parallel four-stranded complexes by guanine-rich motifs in DNA and its implications for meiosis. Nature 334, 364–366 (1988).
Fay, M. M., Lyons, S. M. & Ivanov, P. RNA G-quadruplexes in biology: principles and molecular mechanisms. J. Mol. Biol. 429, 2127–2147 (2017).
De, S. & Michor, F. DNA secondary structures and epigenetic determinants of cancer genome evolution. Nat. Struct. Mol. Biol. 18, 950–955 (2011).
Hanakahi, L. A., Sun, H. & Maizels, N. High affinity interactions of nucleolin with G-G-paired rDNA. J. Biol. Chem. 274, 15908–15912 (1999).
Lam, E. Y. N., Beraldi, D., Tannahill, D. & Balasubramanian, S. G-quadruplex structures are stable and detectable in human genomic DNA. Nat. Commun. 4, 1796 (2013).
Huppert, J. L. & Balasubramanian, S. G-quadruplexes in promoters throughout the human genome. Nucleic Acids Res. 35, 406–413 (2007).
Kumari, S., Bugaut, A., Huppert, J. L. & Balasubramanian, S. An RNA G-quadruplex in the 5′ UTR of the NRAS proto-oncogene modulates translation. Nat. Chem. Biol. 3, 218–221 (2007).
Bugaut, A. & Balasubramanian, S. 5’-UTR RNA G-quadruplexes: translation regulation and targeting. Nucleic Acids Res. 40, 4727–4741 (2012).
Beaudoin, J.-D. & Perreault, J.-P. Exploring mRNA 3′-UTR G-quadruplexes: evidence of roles in both alternative polyadenylation and mRNA shortening. Nucleic Acids Res. 41, 5898–5911 (2013).
Cayrou, C. et al. Genome-scale analysis of metazoan replication origins reveals their organization in specific but flexible sites defined by conserved features. Genome Res. 21, 1438–1449 (2011).
Cayrou, C., Grégoire, D., Coulombe, P., Danis, E. & Méchali, M. Genome-scale identification of active DNA replication origins. Methods 57, 158–164 (2012).
Besnard, E. et al. Unraveling cell type–specific and reprogrammable human replication origin signatures associated with G-quadruplex consensus motifs. Nat. Struct. Mol. Biol. 19, 837–844 (2012).
Hardin, C. C., Watson, T., Corregan, M. & Bailey, C. Cation-dependent transition between the quadruplex and Watson-Crick hairpin forms of d(CGCG3GCG). Biochemistry 31, 833–841 (1992).
Li, W., Wu, P., Ohmichi, T. & Sugimoto, N. Characterization and thermodynamic properties of quadruplex/duplex competition. FEBS Lett. 526, 77–81 (2002).
Biffi, G., Tannahill, D., McCafferty, J. & Balasubramanian, S. Quantitative visualization of DNA G-quadruplex structures in human cells. Nat. Chem. 5, 182–186 (2013).
Bochman, M. L., Paeschke, K. & Zakian, V. A. DNA secondary structures: stability and function of G-quadruplex structures. Nat. Rev. Genet. 13, 770–780 (2012).
Suhasini, A. N. & Brosh, R. M. Fanconi anemia and Bloom’s syndrome crosstalk through FANCJ–BLM helicase interaction. Trends Genet. 28, 7–13 (2012).
Suhasini, A. N. et al. Interaction between the helicases genetically linked to Fanconi anemia group J and Bloom’s syndrome. EMBO J. 30, 692–705 (2011).
Wu, L. & Hickson, I. D. The Bloom’s syndrome helicase suppresses crossing over during homologous recombination. Nature 426, 870–874 (2003).
Patel, D. S., Misenko, S. M., Her, J. & Bunting, S. F. BLM helicase regulates DNA repair by counteracting RAD51 loading at DNA double-strand break sites. J. Cell Biol. 216, 3521–3534 (2017).
Chaganti, R. S. K., Schonberg, S. & German, J. A manyfold increase in sister chromatid exchanges in Bloom’s syndrome lymphocytes. Proc. Natl. Acad. Sci. USA 71, 4508–4512 (1974).
Ray, J. H. & German, J. Bloom’s syndrome and EM9 cells in BrdU-containing medium exhibit similarly elevated frequencies of Sister Chromatid Exchange but dissimilar amounts of cellular proliferation and chromosome disruption. Chromosoma 90, 383–388 (1984).
Aurias, A., Antoine, J.-L., Assathiany, R., Odievre, M. & Dutrillaux, B. Radiation sensitivity of Bloom’s syndrome lymphocytes during S and G2 phases. Cancer Genet. Cytogenet. 16, 131–136 (1985).
Hook, G. J., Kwok, E. & Heddle, J. A. Sensitivity of Bloom syndrome fibroblasts to mitomycin C. Mutat. Res./DNA Repair Rep. 131, 223–230 (1984).
Chu, W. K. & Hickson, I. D. RecQ helicases: multifunctional genome caretakers. Nat. Rev. Cancer 9, 644–654 (2009).
Sun, H., Karow, J. K., Hickson, I. D. & Maizels, N. The Bloom’s syndrome helicase unwinds G4 DNA. J. Biol. Chem. 273, 27587–27592 (1998).
Sato, K. & Knipscheer, P. G-quadruplex resolution: from molecular mechanisms to physiological relevance. DNA Repair 130, 103552 (2023).
Barefield, C. & Karlseder, J. The BLM helicase contributes to telomere maintenance through processing of late-replicating intermediate structures. Nucleic Acids Res. 40, 7358–7367 (2012).
Lillard-Wetherell, K. et al. Association and regulation of the BLM helicase by the telomere proteins TRF1 and TRF2. Hum. Mol. Genet. 13, 1919–1932 (2004).
Opresko, P. L. et al. Telomere-binding protein TRF2 binds to and stimulates the Werner and Bloom syndrome helicases. J. Biol. Chem. 277, 41110–41119 (2002).
Opresko, P. L. et al. POT1 stimulates RecQ helicases WRN and BLM to unwind telomeric DNA substrates. J. Biol. Chem. 280, 32069–32080 (2005).
Cheok, C. F. The Bloom’s syndrome helicase promotes the annealing of complementary single-stranded DNA. Nucleic Acids Res. 33, 3932–3941 (2005).
Machwe, A., Xiao, L., Groden, J. & Orren, D. K. The Werner and Bloom syndrome proteins catalyze regression of a model replication fork. Biochemistry 45, 13939–13946 (2006).
Ralf, C., Hickson, I. D. & Wu, L. The Bloom’s syndrome helicase can promote the regression of a model replication fork. J. Biol. Chem. 281, 22839–22846 (2006).
Drosopoulos, W. C., Kosiyatrakul, S. T. & Schildkraut, C. L. BLM helicase facilitates telomere replication during leading strand synthesis of telomeres. J. Cell Biol. 210, 191–208 (2015).
Balasubramanian, S. & Neidle, S. G-quadruplex nucleic acids as therapeutic targets. Curr. Opin. Chem. Biol. 13, 345–353 (2009).
McLuckie, K. I. E. et al. G-quadruplex DNA as a molecular target for induced synthetic lethality in cancer cells. J. Am. Chem. Soc. 135, 9640–9643 (2013).
Sfeir, A. et al. Mammalian telomeres resemble fragile sites and require TRF1 for efficient replication. Cell 138, 90–103 (2009).
Stavropoulos, D. J. The Bloom syndrome helicase BLM interacts with TRF2 in ALT cells and promotes telomeric DNA synthesis. Hum. Mol. Genet. 11, 3135–3144 (2002).
van Wietmarschen, N. et al. BLM helicase suppresses recombination at G-quadruplex motifs in transcribed genes. Nat. Commun. 9, 271 (2018).
Kim, N. The interplay between G-quadruplex and transcription. Curr. Med. Chem. 26, 2898–2917 (2019).
Spiegel, J. et al. G-quadruplexes are transcription factor binding hubs in human chromatin. Genome Biol. 22, 117 (2021).
Cantor, S. B. & Guillemette, S. Hereditary breast cancer and the BRCA1-associated FANCJ/BACH1/BRIP1. Future Oncol. 7, 253–261 (2011).
Cantor, S. et al. The BRCA1-associated protein BACH1 is a DNA helicase targeted by clinically relevant inactivating mutations. Proc. Natl. Acad. Sci. USA 101, 2357–2362 (2004).
Cantor, S. B. et al. BACH1, a novel helicase-like protein, interacts directly with BRCA1 and contributes to its DNA repair function. Cell 105, 149–160 (2001).
Levitus, M. et al. The DNA helicase BRIP1 is defective in Fanconi anemia complementation group J. Nat. Genet. 37, 934–935 (2005).
Litman, R. et al. BACH1 is critical for homologous recombination and appears to be the Fanconi anemia gene product FANCJ. Cancer Cell 8, 255–265 (2005).
Seal, S. et al. Truncating mutations in the Fanconi anemia J gene BRIP1 are low-penetrance breast cancer susceptibility alleles. Nat. Genet. 38, 1239–1241 (2006).
Wu, Y. & Brosh, R. Jr. FANCJ helicase operates in the Fanconi anemia DNA repair pathway and the response to replicational stress. Curr. Mol. Med. 9, 470–482 (2009).
Wu, Y., Shin-ya, K. & Brosh, R. M. FANCJ helicase defective in Fanconia anemia and breast cancer unwinds G-quadruplex DNA to defend genomic stability. Mol. Cell Biol. 28, 4116–4128 (2008).
London, T. B. C. et al. FANCJ is a structure-specific DNA helicase associated with the maintenance of genomic G/C tracts. J. Biol. Chem. 283, 36132–36139 (2008).
Brosh, R. M. & Wu, Y. An emerging picture of FANCJ’s role in G4 resolution to facilitate DNA replication. NAR Cancer 3, zcab034 (2021).
Hiom, K. FANCJ: Solving problems in DNA replication. DNA Repair 9, 250–256 (2010).
Odermatt, D. C. et al. Cancer-associated mutations in the iron-sulfur domain of FANCJ affect G-quadruplex metabolism. PLoS Genet. 16, e1008740 (2020).
Schwab, R. A., Nieminuszczy, J., Shin-ya, K. & Niedzwiedz, W. FANCJ couples replication past natural fork barriers with maintenance of chromatin structure. J. Cell Biol. 201, 33–48 (2013).
Castillo Bosch, P. et al. FANCJ promotes DNA synthesis through G‐quadruplex structures. EMBO J. 33, 2521–2533 (2014).
Cornforth, M. N. Termini of human chromosomes display elevated rates of mitotic recombination. Mutagenesis 16, 85–89 (2001).
Mao, F. J., Sidorova, J. M., Lauper, J. M., Emond, M. J. & Monnat, R. J. The human WRN and BLM RecQ helicases differentially regulate cell proliferation and survival after chemotherapeutic DNA damage. Cancer Res. 70, 6548–6555 (2010).
Ababou, M., Dumaire, V., Lécluse, Y. & Amor-Guéret, M. Cleavage of BLM and sensitivity of Bloom’s syndrome cells to hydroxurea and UV-C radiation. Cell Cycle 1, 262–266 (2002).
Lahkim Bennani-Belhaj, K. et al. The Bloom syndrome protein limits the lethality associated with RAD51 deficiency. Mol. Cancer Res. 8, 385–394 (2010).
Berardinelli, F. et al. The G-quadruplex-stabilising agent RHPS4 induces telomeric dysfunction and enhances radiosensitivity in glioblastoma cells. DNA Repair 25, 104–115 (2015).
Martínez, P. et al. Increased telomere fragility and fusions resulting from TRF1 deficiency lead to degenerative pathologies and increased cancer in mice. Genes Dev. 23, 2060–2075 (2009).
Mohaghegh, P. The Bloom’s and Werner’s syndrome proteins are DNA structure-specific helicases. Nucleic Acids Res. 29, 2843–2849 (2001).
Huber, M. D. G4 DNA unwinding by BLM and Sgs1p: substrate specificity and substrate-specific inhibition. Nucleic Acids Res. 30, 3954–3961 (2002).
Zimmermann, M., Kibe, T., Kabir, S. & de Lange, T. TRF1 negotiates TTAGGG repeat-associated replication problems by recruiting the BLM helicase and the TPP1/POT1 repressor of ATR signaling. Genes Dev. 28, 2477–2491 (2014).
Yang, G. et al. Poly(ADP-ribosyl)ation mediates early phase histone eviction at DNA lesions. Nucleic Acids Res. 48, 3001–3013 (2020).
Rizzo, A. et al. Stabilization of quadruplex DNA perturbs telomere replication leading to the activation of an ATR-dependent ATM signaling pathway. Nucleic Acids Res. 37, 5353–5364 (2009).
Chu, W. K., Hanada, K., Kanaar, R. & Hickson, I. D. BLM has early and late functions in homologous recombination repair in mouse embryonic stem cells. Oncogene 29, 4705–4714 (2010).
Nguyen, G. H. et al. Regulation of gene expression by the BLM helicase correlates with the presence of G-quadruplex DNA motifs. Proc. Natl. Acad. Sci. USA 111, 9905–9910 (2014).
Suhasini, A. N. & Brosh, R. M. Disease-causing missense mutations in human DNA helicase disorders. Mutat. Res./Rev. Mutat. Res. 752, 138–152 (2013).
Sarkies, P. et al. FANCJ coordinates two pathways that maintain epigenetic stability at G-quadruplex DNA. Nucleic Acids Res. 40, 1485–1498 (2012).
Henderson, A. et al. Detection of G-quadruplex DNA in mammalian cells. Nucleic Acids Res. 42, 860–869 (2014).
Li, G. et al. Alternative splicing of human telomerase reverse transcriptase in gliomas and its modulation mediated by CX-5461. J. Exp. Clin. Cancer Res. 37, 78 (2018).
Sullivan, H.-J., Chen, B. & Wu, C. Molecular dynamics study on the binding of an anticancer DNA G-quadruplex stabilizer, CX-5461, to human telomeric, c-KIT1, and c-Myc G-quadruplexes and a DNA duplex. J. Chem. Inf. Model. 60, 5203–5224 (2020).
Lee, W. T. C. et al. Single-molecule imaging reveals replication fork coupled formation of G-quadruplex structures hinders local replication stress signaling. Nat. Commun. 12, 2525 (2021).
Awate, S. & Brosh, R. Jr. Interactive roles of DNA helicases and translocases with the single-stranded DNA binding protein RPA in nucleic acid metabolism. Int. J. Mol. Sci. 18, 1233 (2017).
Déjardin, J. & Kingston, R. E. Purification of proteins associated with specific genomic loci. Cell 136, 175–186 (2009).
Cong, K. et al. FANCJ promotes PARP1 activity during DNA replication that is essential in BRCA1 deficient cells. Nat. Commun. 15, 2599 (2024).
Maresca, C. et al. PARP1 allows proper telomere replication through TRF1 poly (ADP-ribosyl)ation and helicase recruitment. Commun. Biol. 6, 234 (2023).
Salvati, E. et al. PARP1 is activated at telomeres upon G4 stabilization: possible target for telomere-based therapy. Oncogene 29, 6280–6293 (2010).
Soldatenkov, V. A., Vetcher, A. A., Duka, T. & Ladame, S. First evidence of a functional interaction between DNA quadruplexes and poly(ADP-ribose) polymerase-1. ACS Chem. Biol. 3, 214–219 (2008).
Edwards, A. D., Marecki, J. C., Byrd, A. K., Gao, J. & Raney, K. D. G-Quadruplex loops regulate PARP-1 enzymatic activation. Nucleic Acids Res. 49, 416–431 (2021).
Berardinelli, F. et al. G-quadruplex ligand RHPS4 radiosensitizes glioblastoma xenograft in vivo through a differential targeting of bulky differentiated- and stem-cancer cells. J. Exp. Clin. Cancer Res. 38, 311 (2019).
Kumaraswamy, E. & Shiekhattar, R. Activation of BRCA1/BRCA2-associated helicase BACH1 is required for timely progression through S phase. Mol. Cell Biol. 27, 6733–6741 (2007).
Vichai, V. & Kirtikara, K. Sulforhodamine B colorimetric assay for cytotoxicity screening. Nat. Protoc. 1, 1112–1116 (2006).
Berardinelli, F. et al. Telomere alterations and genomic instability in long-term cultures of normal human fibroblasts irradiated with X rays and protons. Radiat. Prot. Dosim. 143, 274–278 (2011).
Berardinelli, F. et al. The role of telomere length modulation in delayed chromosome instability induced by ionizing radiation in human primary fibroblasts. Environ. Mol. Mutagen. 54, 172–179 (2013).
Surace, C. et al. Telomere shortening and telomere position effect in mild ring 17 syndrome. Epigenetics Chromatin 7, 1 (2014).
Perner, S. et al. Quantifying telomere lengths of human individual chromosome arms by centromere-calibrated fluorescence in situ hybridization and digital imaging. Am. J. Pathol. 163, 1751–1756 (2003).
Berardinelli, F. et al. Transient activation of the ALT pathway in human primary fibroblasts exposed to high-LET radiation. Radiat. Res. 174, 539–549 (2010).
Acknowledgement
L.B. is a BMCA PhD fellow at the Department of Science, Roma Tre University. This study was also supported by grants from Ateneo Roma Tre to F.B. and A.A. The Grant of Excellence Departments, MIUR (ARTICOLO 1, COMMI 314–337 LEGGE 232/2016) to the Department of Science, University Roma Tre is gratefully acknowledged.
Author information
Authors and Affiliations
Contributions
D.M. and E.F. designed and performed CRISPR/CAS9 experiments and analyzed data. L.B. and F.B. designed and performed siRNA experiments and analyzed the data. L.B. and F.B. designed and performed cytogenetic and immunofluorescence experiments and analyzed data. E.S., A.P., and L.B. designed and performed IF-FISH, PLA, ChIP, and WB experiments and analyzed the data. F.B. and A.A. conceived, initiated, and coordinated the project, designed and performed the experiments, analyzed data, and wrote the manuscript. E.F. and E.S. contributed to writing and revising the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Biology thanks the anonymous reviewers for their contribution to the peer review of this work. Primary Handling Editors: Valeria Naim and Mengtan Xing.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Bonanni, L., Muoio, D., Fouquerel, E. et al. BLM and FANCJ role in the response to G-quadruplex-dependent telomeric replicative stress. Commun Biol 9, 100 (2026). https://doi.org/10.1038/s42003-025-09367-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s42003-025-09367-z