Abstract
Resistance to 5-fluorouracil (5-FU) remains a major challenge in the treatment of colorectal cancer (CRC). Here, we identify ETS variant transcription factor 7 (ETV7) as significantly upregulated in CRC tissues and cell lines, with elevated expression associated with poor clinical prognosis. Functional assays demonstrate that ETV7 enhances CRC cell proliferation, invasion, and resistance to 5-FU. Mechanistically, ETV7 transcriptionally upregulates CXCL1, leading to increased neutrophil recruitment and enhanced formation of neutrophil extracellular traps (NETs). The resulting NETs-enriched tumor microenvironment promotes tumor aggressiveness and chemoresistance. Pharmacological inhibition of CXCL1 or degradation of NETs effectively attenuates ETV7-driven malignant phenotypes in vitro and in vivo. Collectively, these findings establish an ETV7–CXCL1–NETs axis that contributes to 5-FU resistance in CRC and suggest that targeting this pathway may improve chemotherapy response.
Similar content being viewed by others
Introduction
Colorectal cancer (CRC) remains a major global health burden, with high incidence and mortality largely attributable to tumor recurrence and resistance to systemic therapies1. 5-Fluorouracil (5-FU)-based chemotherapy continues to serve as a cornerstone in CRC treatment2. However, its clinical benefits are frequently limited by the emergence of drug resistance2. Despite extensive efforts, the mechanisms driving chemoresistance in CRC are highly heterogeneous and incompletely understood, particularly those involving tumor–microenvironment interactions rather than tumor-intrinsic alterations alone. This gap highlights the need to explore non–cell-autonomous regulatory mechanisms that contribute to therapeutic failure.
ETS variant transcription factor 7 (ETV7), also known as TEL2 or TELB, is a member of the E26 transformation-specific (ETS) family of transcription factors3,4,5. ETS family members regulate the expression of genes involved in tumor initiation and progression, thereby influencing key biological processes such as cell proliferation, migration, cell cycle control, and angiogenesis6,7,8. Accumulating evidence indicates that ETV7 is aberrantly overexpressed in multiple malignancies5,7, including hematologic cancers5,as well as solid tumors such as breast cancer9, bladder cancer10, and melanoma8. In recent years, increasing attention has been directed toward the role of ETV7 in cancer progression and therapeutic resistance6. For instance, in breast cancer, ETV7 has been shown to maintain tumor cell stemness, thereby reducing cellular sensitivity to 5-FU and radiotherapy4. Moreover, emerging studies suggest that ETV7 participates in the regulation of the tumor immune microenvironment9,11. Notably, ETV7 has been reported to impair the antiviral and antitumor functions of CD8⁺ T cells by promoting their differentiation toward an exhausted phenotype8. In colorectal cancer, however, investigations into the biological function of ETV7 remain limited7. Collectively, current evidence links ETV7 to malignant progression and treatment resistance across multiple tumor types, although the precise molecular mechanisms underlying its effects require further elucidation.
Neutrophils are increasingly recognized as critical regulators of tumor progression12. One of their key functional outputs is the formation of neutrophil extracellular traps (NETs)13, which consist of extracellular DNA fibers associated with histones and granular proteins14. Originally characterized as an antimicrobial defense mechanism, NETs are now understood to participate in non-infectious pathological processes, including cancer progression15,16. Rather than acting solely as passive byproducts of inflammation, NETs can actively remodel the tumor microenvironment by influencing stromal interactions, extracellular matrix dynamics, and immune cell behavior13,15,16,17. Recent findings suggest that NETs may also interfere with therapeutic responses by creating a protective niche that supports tumor cell survival under cytotoxic stress12,18,19. However, the upstream signals derived from tumor cells that trigger NETs formation during chemotherapy remain largely undefined.
CXCL1, a member of the CXC chemokine family, plays an important role in neutrophil recruitment through activation of the CXCR2 receptor20,21,22. Beyond its well-established chemotactic function, CXCL1 has recently been implicated in modulating neutrophil activation states, although its involvement in NETs formation appears to be context-dependent21,22,23,24. In tumor settings, aberrant CXCL1 secretion may contribute not only to neutrophil accumulation but also to functional reprogramming of these cells within the tumor microenvironment25,26. Whether tumor-derived CXCL1 directly links oncogenic signaling to NETs-mediated chemoresistance remains an open question.
Although ETV7 and CXCL1 have been implicated in tumor resistance and immune regulation within the tumor microenvironment, respectively, whether a functional association exists between them remains unclear. To date, no direct evidence has demonstrated that ETV7 modulates the neutrophil/NETs axis in the tumor microenvironment to influence chemotherapy response. In this study, we propose a model in which tumor-intrinsic transcriptional regulation intersects with microenvironmental immune responses to drive chemoresistance. Specifically, we hypothesized that ETV7 enhances CXCL1 transcription in CRC cells, thereby promoting neutrophil activation and NETs formation, which in turn supports tumor progression and reduces sensitivity to 5-FU treatment. By integrating tumor biology with immune microenvironment dynamics, our work aims to uncover a previously unrecognized regulatory axis that contributes to chemotherapy resistance in colorectal cancer.
Results
ETV7 upregulation correlates with adverse clinical outcomes in colorectal cancer
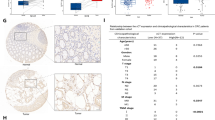
Public database analysis revealed a significant elevation of ETV7 expression in CRC tissues compared with normal controls (Supplementary Fig. 1). This observation was further validated in our clinical samples using RT-qPCR, Western blotting, and immunohistochemistry, consistently demonstrating increased ETV7 levels in tumor tissues (Fig. 1A–C). Importantly, survival analyses indicated that patients with higher ETV7 expression exhibited reduced overall survival (OS) and disease-free survival (DFS), suggesting a potential prognostic value of ETV7 in CRC (Fig. 1D, E).
A, B RT-qPCR (A) (n = 8 biologically independent samples) and Western blot (B) (n = 12 biologically independent samples) experiments were used to detect ETV7 in fresh CRC and normal tissues. C Immunohistochemical detection of ETV7 expression in paired colorectal cancer tissues (n = 80 biologically independent samples, scale bar 50 μm). The right panel shows quantitative analysis. Statistical analysis of overall survival (D) and disease-free survival (E) using the Kaplan-Meier method (n = 80 biologically independent samples).
ETV7 enhances malignant phenotypes and contributes to 5-FU resistance in a microenvironment-dependent manner
ETV7 enhances malignant phenotypes and contributes to 5-FU resistance in a microenvironment-dependent manner, we first assessed its expression in CRC cell lines and the normal intestinal epithelial cell line NCM460 using RT-qPCR and Western blotting (Supplementary Fig. 2A, B). Based on endogenous expression levels, RKO and DLD1 cells (low ETV7 expression) were selected to generate stable ETV7-overexpressing cell lines, whereas HCT116 and SW620 cells (high ETV7 expression) were used to establish stable ETV7 knockdown models (Supplementary Fig. 2C–F). Silencing ETV7 significantly impaired cell proliferation, migration, and invasion (Supplementary Fig. 2G–J), whereas ectopic expression of ETV7 produced the opposite effects, indicating its pro-tumorigenic role in vitro (Fig. 2A–D). Interestingly, modulation of ETV7 did not significantly affect cellular sensitivity to 5-FU under standard in vitro conditions (Fig. 2E, supplementary Fig. 2K). However, in vivo experiments using xenograft models revealed that tumors with elevated ETV7 expression exhibited enhanced growth despite 5-FU treatment (Fig. 2F–J). This discrepancy between in vitro and in vivo findings suggests that the contribution of ETV7 to chemoresistance is not solely dependent on tumor-intrinsic mechanisms but may require interactions with the tumor microenvironment.
A CCK8 assay to detect changes in proliferation of CRC cells RKO (top) and DLD1 (bottom) after ETV7 overexpression (n = 3 independent experiments). B Colony formation assay to detect representative images of colonies formed by colorectal cancer cells overexpressing ETV7. The bottom panel shows quantitative analysis (n = 3 independent experiments). C Transwell invasion assay showing representative images of the invasive ability of colorectal cancer cells overexpressing ETV7 (n = 3 independent experiments, scale bar 50 μm). The lower panel shows quantitative analysis. D Wound healing assay showing representative images of the migratory ability of colorectal cancer cells overexpressing ETV7 (n = 3 independent experiments, scale bar 100 μm). The right panel shows quantitative analysis. E Cell viability assay to assess cell viability in RKO cells after ETV7 overexpression, with IC50 curve plotted. F–J Establishment of a mouse subcutaneous tumor model overexpressing ETV7 (20 days post-injection, n = 6 animals). F Photograph of the tumor at the end of the experiment. Tumor volume (G) and weight (H) at the end of the experiment. I Representative images of HE staining and Ki-67 immunohistochemistry of the tumors (scale bar 50 μm). J Quantitative analysis of Ki-67.
ETV7 is associated with neutrophil infiltration and promotes NETs formation in the tumor microenvironment
Given the observed context-dependent effects, we next investigated whether ETV7 influences the tumor microenvironment. Bioinformatic analysis indicated a positive correlation between ETV7 expression and neutrophil infiltration in colorectal cancer (Supplementary Fig. 3). Consistently, increased neutrophil accumulation was observed in tumor tissues derived from ETV7-overexpressing cells (Fig. 3A). Functional co-culture experiments further showed that the presence of neutrophils significantly amplified the proliferative (Fig. 3B, C), invasive (Fig. 3D, F), and drug-resistant phenotypes (Fig. 3E, G) of CRC cells with high ETV7 expression.
(A) Immunofluorescence analysis of neutrophil infiltration in subcutaneous tumors of mice overexpressing ETV7 (scale bar 20 μm, n = 6 animals). Neutrophils in mice were labeled with Ly-6G. The lower panel shows the quantitative analysis of neutrophils. In vitro functional experiments were conducted by co-culturing CRC cells overexpressing (B, D, E) or knocking down (C, F, G) ETV7 with neutrophils. B, C CCK8 assay to detect CRC cell proliferation (n = 3 independent experiments). D, F Transwell invasion assay to assess CRC cell invasion capacity (scale bar 50 μm, n = 3 independent experiments). The right panel shows quantitative results. E, G Cell viability assay to assess CRC cell sensitivity to 5-FU treatment and plot IC50 curves.
KEGG pathway enrichment analysis of TCGA-CRC data indicated that genes associated with high ETV7 expression were significantly enriched in pathways related to neutrophil extracellular traps (NETs) formation (Fig. 4A). Further analyses demonstrated elevated levels of NETs-associated markers in tumors with high ETV7 expression (Fig. 4B, C). Correlation analysis revealed a positive association between ETV7 expression and NETs levels (Supplementary Fig. 4A). In vitro assays confirmed that conditioned media from ETV7-overexpressing CRC cells enhanced NETs release from neutrophils, whereas ETV7 silencing attenuated this effect (Fig. 4D–G, supplementary Fig. 4B–E). These findings indicate that ETV7 contributes to tumor progression, at least in part, by modulating neutrophil activity and promoting NETs formation within the tumor microenvironment.
A KEGG enrichment analysis of TCGA-CRC data shows that ETV7 is enriched in pathways associated with colorectal cancer. B Immunofluorescence analysis of NETs in subcutaneous tumors of mice overexpressing ETV7 (scale bar 20 μm, n = 6 animals). The lower panel shows quantitative analysis. C Immunofluorescence analysis of NETs formation in human colorectal cancer tissues with low ETV7 expression and high ETV7 expression (scale bar 20 μm, n = 80 biologically independent samples). The right panel shows quantitative analysis. NETs formation in conditioned medium from ETV7-overexpressing colorectal cancer cells was analyzed using Sytox immunofluorescence (D, F) and MPO-DNA ELISA (E, G) to label NETs (scale bar 10 μm, n = 3 independent experiments). NETs were labeled using Sytox immunofluorescence (H) and MPO-DNA ELISA (I) to analyze the effect of DNase I on NETs formation in neutrophils cultured in conditioned medium from ETV7-overexpressing CRC cells (scale bar 10 μm, n = 3 independent experiments). Analysis of the effects of DNase I on the proliferation, invasion capacity, and 5-FU treatment efficacy of CRC cells co-cultured with neutrophils in ETV7-overexpressing CRC cells, using CCK8 proliferation assays (J) (n = 3 independent experiments), Transwell invasion assays (K, J) (n = 3 independent experiments), and cell viability assays (L) (scale bar 50 μm).
NETs formation is required for ETV7-mediated tumor-promoting and chemoresistant phenotypes
To determine whether NETs are functionally involved in ETV7-driven tumor progression, rescue experiments were performed by modulating NETs formation. Degradation of NETs using DNase I, a NETs-degrading enzyme, significantly reduced the enhanced proliferation, invasion, and 5-FU resistance observed in CRC cells with high ETV7 expression in the presence of neutrophils (Fig. 4H–L). Conversely, pharmacological induction of NETs restored these malignant phenotypes even when ETV7 expression was suppressed (Supplementary Fig. 5A–E). These results demonstrate that NETs formation is a critical downstream event required for the tumor-promoting and chemoresistance-associated effects of ETV7.
ETV7 transcriptionally upregulates CXCL1 expression in colorectal cancer cells
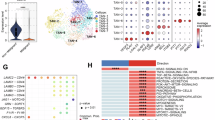
To identify the molecular link between ETV7 and neutrophil activation, transcriptomic analyses were performed, revealing enrichment of Cytokines and Inflammatory Response pathways in tumors with high ETV7 expression (Fig. 5A). Among candidate mediators, CXCL1 was found to be strongly associated with ETV7 expression (Fig. 5B). Experimental validation confirmed that ETV7 positively regulates CXCL1 at both mRNA and protein levels (Fig. 5C, D). Mechanistically, chromatin immunoprecipitation and luciferase reporter assays demonstrated that ETV7 directly binds to the promoter region of CXCL1 and enhances its transcriptional activity (Fig. 5E, F). These findings identify CXCL1 as a downstream target of ETV7 and a potential mediator linking tumor cells to neutrophil responses.
A GSEA enrichment analysis of TCGA-CRC data visualizes the CYTOKINES AND INFLAMMATORY RESPONSE pathway. B Analysis of the correlation between ETV7 expression and CXCL1 expression in colorectal cancer using the GEPIA database. C RT-qPCR experiments analyzed the effects of ETV7 overexpression and knockdown on CXCL1 mRNA (n = 3 independent experiments). D ELISA experiments analyzed the effects of ETV7 overexpression and knockdown on CXCL1 protein (n = 3 independent experiments). E ChIP-qPCR detected ETV7 binding in the CXCL1 promoter region (n = 3 independent experiments). F Double luciferase reporter gene assay to detect the relative luciferase activity of the CXCL1 promoter in HEK293T cells (n = 3 independent experiments).
CXCL1 mediates ETV7-driven NETs formation and contributes to tumor progression and 5-FU resistance
To further clarify the role of CXCL1, rescue experiments were conducted using combined modulation of ETV7 and CXCL1 expression. Silencing CXCL1 significantly attenuated NETs formation induced by conditioned media from ETV7-overexpressing CRC cells (Fig. 6A, B), while reintroduction of CXCL1 restored NETs production in ETV7-deficient conditions (Supplementary Fig. 6A, B). These results indicate that CXCL1 is required for ETV7-mediated activation of neutrophils. In CRC cells cultured alone, CXCL1 silencing moderately reduced the proliferation and invasion enhanced by ETV7 overexpression (Fig. 6D, E), but did not significantly affect 5-FU sensitivity (Fig. 6C). However, in the neutrophil co-culture system, CXCL1 knockdown markedly reversed the increased 5-FU resistance, proliferation, and invasion driven by ETV7 overexpression (Fig. 6C–E). Importantly, PMA treatment restored these malignant phenotypes despite CXCL1 silencing, supporting a NETs-dependent mechanism (Fig. 6C–E). Similarly, CXCL1 overexpression partially rescued the reduced proliferation and invasion observed in ETV7-silenced CRC cells (Supplementary Fig. 6C, D). In the presence of neutrophils, CXCL1 overexpression reversed the inhibitory effects of ETV7 knockdown on CRC cell malignancy, whereas DNase I treatment significantly abrogated these effects (Supplementary Fig. 6C, D), further confirming the functional involvement of NETs formation. In the subcutaneous xenograft mouse model under 5-FU treatment, DNase I administration, CXCL1 silencing, or anti-CXCL1 antibody treatment each significantly suppressed the enhanced tumor growth induced by ETV7 overexpression (Fig. 6F–I). Notably, combined treatment with DNase I and anti-CXCL1 antibody exerted a stronger inhibitory effect than either intervention alone (Fig. 6F–I). Collectively, these findings demonstrate that ETV7 promotes colorectal cancer progression and chemoresistance primarily through a CXCL1-dependent activation of neutrophils and subsequent NETs formation within the tumor microenvironment.
NETs formation was analyzed by Sytox immunofluorescence (A) and MPO-DNA ELISA (B) in neutrophils incubated with CRC cell culture supernatants from cells overexpressing ETV7 and simultaneously silencing CXCL1(n = 3 independent experiments, scale bar 10 μm). Analysis of changes in CRC cell 5-FU sensitivity, proliferation, and invasion capacity after co-culturing CRC cells overexpressing ETV7 while silencing CXCL1 with neutrophils (with or without PMA) using cell viability assays (C), CCK8 proliferation assays (D), and Transwell invasion assays (E) (Scale bar: 50 μm) (n = 3 independent experiments). F–I By establishing an in vivo mouse subcutaneous tumor model, under 5-FU treatment conditions, mice were administered DNase I, CXCL1 silencing, Anti-CXCL1, or a combination of DNase I and Anti-CXCL1 (n = 4 animals). F Photographs of tumors at the end of the experiment. Tumor volume (G) and weight (H) at the end of the experiment. I Representative images of HE-stained tumors (scale bar 50 μm).
Discussion
This study delineates the role of ETV7 in colorectal cancer (CRC) progression and 5-FU resistance. Mechanistically, we demonstrate that ETV7 transcriptionally upregulates CXCL1 in CRC cells, thereby promoting neutrophil extracellular trap (NETs) formation and enhancing tumor aggressiveness and chemoresistance. Importantly, ETV7-mediated CXCL1 upregulation partially augments malignant phenotypes in CRC cells independent of NETs formation; however, it does not directly increase 5-FU resistance under tumor cell–intrinsic conditions. In contrast, CXCL1-driven NETs formation markedly enhances CRC cell proliferation, invasion, and resistance to 5-FU (Supplementary Fig. 7), highlighting the critical contribution of the tumor microenvironment in mediating ETV7-associated chemoresistance.
This study highlights the pivotal role of the transcription factor ETV7 in CRC progression and 5-FU resistance. Clinical analyses demonstrated that ETV7 is significantly overexpressed in CRC and is associated with unfavorable patient outcomes. Consistently, we observed elevated ETV7 expression in CRC tissues and cell lines compared with normal controls. Functional assays further confirmed that ETV7 enhances CRC cell proliferation, migration, and invasion. Collectively, these findings support the notion that ETV7 functions as a pro-tumorigenic transcription factor in CRC, consistent with its reported oncogenic roles in other malignancies, including breast cancer4.
Our findings further demonstrate that ETV7 contributes to chemotherapy resistance in CRC. In vitro drug sensitivity assays revealed that ETV7-overexpressing CRC cells exhibited significantly increased resistance to 5-FU when co-cultured with neutrophils, whereas ETV7 silencing enhanced cellular sensitivity to the drug. In contrast, under tumor cell–intrinsic conditions without neutrophil co-culture, modulation of ETV7 expression did not significantly alter 5-FU sensitivity. These observations indicate that the effect of ETV7 on 5-FU resistance is largely dependent on the tumor microenvironment. This context-dependent role is consistent with previous reports implicating ETV7 in chemotherapy resistance4. As a transcription factor, ETV7 may regulate multiple resistance-related pathways. For example, ETV7 has been reported to repress DNAJC15 expression, thereby promoting treatment resistance6. Collectively, these findings suggest that elevated ETV7 expression enhances tumor cell survival capacity, contributing to reduced responsiveness to chemotherapy, including 5-FU, in CRC.
More importantly, this study provides mechanistic insight into how ETV7 shapes the tumor immune microenvironment in CRC. Our data indicate that ETV7 promotes tumor progression and chemoresistance by enhancing neutrophil extracellular trap (NETs) formation. Previous studies have suggested that ETV7 can influence tumor behavior through microenvironmental modulation8. Consistently, our bioinformatics analyses revealed a positive association between ETV7 expression and neutrophil infiltration. Emerging evidence has established NETs formation as an active contributor to tumor progression and therapeutic resistance12,13,20,27,28. NETs are web-like DNA–protein complexes released by activated neutrophils and were initially characterized as antimicrobial structures14. However, they are now recognized as key regulators of tumor development and immune remodeling13. In our study, elevated ETV7 expression was associated with increased NETs accumulation within the tumor microenvironment. Mechanistically, this effect appears to be mediated by ETV7-driven chemokine signaling. We demonstrate that ETV7 transcriptionally upregulates CXCL1 in CRC cells. CXCL1 is a potent neutrophil chemoattractant that promotes neutrophil recruitment and activation at tumor sites20,22,23. High neutrophil infiltration has been correlated with poor chemotherapy response12,28, and CXCL1 has been implicated in tumor resistance, particularly within metastatic niches12,20,21,29. Notably, Acharyya et al. identified a CXCL1/2-mediated paracrine network in breast cancer linking chemotherapy resistance with metastatic progression20. Our findings suggest that ETV7 functions as an upstream regulator of a CXCL1-driven tumor–immune interaction axis. Through CXCL1-mediated neutrophil recruitment and NETs formation, elevated ETV7 expression connects tumor-intrinsic transcriptional programs with immune microenvironmental remodeling, thereby establishing a permissive niche that favors tumor survival. Consistently, inhibition of CXCL1 signaling or degradation of NETs partially reversed the invasive and chemoresistant phenotypes induced by ETV7 overexpression, supporting the conclusion that ETV7 promotes CRC progression by reprogramming the tumor immune microenvironment.
Accumulating evidence indicates that NETs exert multifaceted pro-tumor effects in tumor progression and chemotherapy resistance12,30,31. Mechanistically, NETs promote malignant phenotypes through several complementary pathways. First, NETs-associated proteases and cytokines can induce epithelial–mesenchymal transition (EMT), thereby enhancing invasive and drug-resistant characteristics in tumor cells12,32. For example, NETs have been reported to capture TGF-β via integrin α_vβ_1 and facilitate its activation through MMP9, driving the acquisition of a mesenchymal phenotype associated with increased invasiveness and chemoresistance12. Second, NETs remodel the extracellular matrix within primary tumors and metastatic niches, facilitating tumor dissemination and colonization19,33. NETs-derived DNA and proteases directly modify matrix components; cathepsin-mediated degradation of laminin, for instance, can expose cryptic epitopes in the basement membrane and reactivate dormant tumor cells17,19. Third, NETs contribute to immune evasion. The DNA–protein scaffold of NETs acts as both a physical and biochemical barrier, impairing the function of antitumor immune effector cells22,34,35. In addition, NETs can shield tumor cells from immune-mediated clearance19,21,22. Finally, NETs may directly reduce chemotherapy efficacy12,30. The DNA backbone of NETs can bind and sequester small-molecule agents, while chronic NETs-rich inflammatory environments activate DNA damage responses and pro-survival signaling pathways, thereby promoting resistance to agents such as 5-FU12. Taken together, these observations position NETs as active facilitators of tumor progression and therapeutic resistance. The ETV7/CXCL1/NETs axis identified in our study represents a specific mechanistic framework through which tumor cells exploit neutrophil-mediated immune remodeling to support their growth and evade treatment. Our findings further highlight how tumor-intrinsic transcriptional alterations—such as ETV7 upregulation—can reshape the tumor microenvironment via CXCL1-dependent inflammatory signaling, ultimately influencing disease progression and treatment outcomes.
The ETV7/CXCL1/NETs axis identified in this study carries both biological and potential translational significance. ETV7 may serve as a candidate biomarker for aggressive CRC and chemotherapy response. Elevated ETV7 expression was associated with increased neutrophil infiltration and NETs enrichment, features indicative of an immunosuppressive tumor microenvironment and potentially linked to higher risks of recurrence, metastasis, and reduced responsiveness to 5-FU. These observations suggest that patients with high ETV7 expression may benefit from therapeutic strategies targeting the tumor immune microenvironment in combination with conventional chemotherapy. In particular, interventions aimed at inhibiting CXCL1 signaling or NETs formation could represent rational adjunct approaches to mitigate resistance associated with activation of the ETV7/CXCL1/NETs axis. Further preclinical and clinical studies are warranted to evaluate the feasibility of such combination strategies. Several limitations should be acknowledged. Because ETV7 is absent in mice, our in vivo experiments relied on xenograft models rather than fully immunocompetent systems, which restricts comprehensive assessment of adaptive immune responses. Nevertheless, our primary objective was to clarify how ETV7 regulates neutrophil extracellular trap formation within the tumor microenvironment. In this context, our findings substantially advance understanding of how tumor-intrinsic transcriptional alterations can remodel immune landscapes to influence disease progression and chemotherapy response. In conclusion, we demonstrate that ETV7 promotes CRC progression and 5-FU resistance through transcriptional activation of CXCL1 and subsequent NETs formation. These findings provide mechanistic insight into tumor–immune interactions in colorectal cancer and offer a conceptual framework for targeting microenvironment-driven chemoresistance.
Materials and methods
Cell culture
The normal human colon epithelial cell line FHC and seven colorectal cancer (CRC) cell lines including HCT116, SW620, DLD1, SW480, RKO, CACO2 and HT29 were obtained from the Cell Bank of the Chinese Academy of Sciences (Shanghai, China). Cell line authentication was performed by short tandem repeat (STR) profiling, and all experiments were conducted within 6 months after cell recovery. All CRC cell lines were maintained in RPMI-1640 medium (Gibco, Gaithersburg, MD, USA) supplemented with 10% fetal bovine serum (FBS; HyClone, Logan, UT, USA) and 100 U/mL penicillin-streptomycin (Gibco). Cells were cultured in a humidified incubator at 37 °C with 5% CO₂36.
CCK8, colony formation assay, cell wound healing assay, and transwell invasion assay
Cell proliferation was evaluated using the Cell Counting Kit-8 (CCK-8; Dojindo, China). Approximately 1 × 10³ cells were seeded into 96-well plates and cultured for 24 h, followed by incubation with CCK-8 reagent for 2 h. Absorbance was measured at 450 nm using a microplate reader (Bio-Rad, Hercules, CA, USA). Experiments were performed in triplicate.
For colony formation assays, cells were plated in six-well plates (200 cells per well) and cultured for about 2 weeks. Colonies were fixed with 4% paraformaldehyde and stained with 1% Giemsa. Colonies containing more than 50 cells were counted.
For cell wound-healing assays, 1.2 × 10⁶ cells were seeded into six-well plates and grown to confluence. A scratch was generated using a 10-μL pipette tip. After washing with PBS, cells were cultured in serum-free RPMI-1640 medium. Images were captured at 0 h and 48 h, and migration ability was evaluated by measuring wound width in three random fields (200×).
Cell invasion assays were conducted using transwell chambers (8-μm pore size; BD Biosciences, San Jose, CA, USA) coated with Matrigel. Cells (2 × 10⁵) suspended in serum-free medium were added to the upper chamber, while medium containing 10% FBS was placed in the lower chamber. After 48 h incubation, invaded cells were fixed, stained with 0.5% crystal violet, and counted in five randomly selected fields (200×)36. When performing the above experiments with colorectal cancer cells co-cultured with human neutrophils, 1 × 10⁵ cells were added. CCK8 and transwell invasion assays were performed under different conditions (25 ng/ml PMA, 0.25 U/ml DNase I).
Cell viability assay
Colorectal cancer cells were cultured in vitro and treated with 5-FU at gradient concentrations. After 72 h, CCK8 was used to assess CRC cell viability and evaluate CRC cell sensitivity to 5-FU. In brief, CRC cells were seeded into 96-well plates and allowed to grow to the logarithmic phase. CCK-8 solution (Dojindo, China) was added to each well and incubated at 37 °C for 2 h. Absorbance was measured at 450 nm using a spectrophotometer, and cell viability was quantified.
Immunohistochemistry
Immunohistochemical (IHC) staining was performed to assess ETV7 expression in 80 paired CRC tissues and adjacent normal tissues. Paraffin sections were heated, deparaffinized, rehydrated, and subjected to antigen retrieval using sodium citrate buffer (pH 6.0). Endogenous peroxidase activity was blocked with 3% hydrogen peroxide, followed by blocking with 10% fetal bovine serum. Sections were incubated overnight at 4 °C with rabbit anti-ETV7 antibody (HPA029033, 1:1000, Sigma), followed by incubation with secondary antibody (anti-rabbit IgG, 1:2000; #7074, Cell Signaling Technology) for 40 min at 37 °C. Signals were visualized using DAB and counterstained with hematoxylin36.
IHC scoring was independently performed by two pathologists blinded to clinical data. Staining intensity was graded as follows: 0 (negative), 1 (weak), 2 (moderate), and 3 (strong). The proportion of positive tumor cells was scored as 0 (0%), 1 (1–25%), 2 (26–50%), 3 (51–75%), and 4 (76–100%)36. The final score (0–15) was calculated by multiplying intensity and proportion scores. Scores å 7 were defined as high expression and ≤7 as low expression.
RNA extraction, RT-qPCR, and Western blot analysis
Total RNA from cells and fresh CRC tissues was extracted using TRIzol reagent (Invitrogen, Carlsbad, CA, USA). Complementary DNA (cDNA) was synthesized with the PrimeScript RT kit (Promega, Madison, WI, USA). RT-qPCR was performed using SYBR Premix Ex Taq (Takara, Dalian, China) on an ABI 7500 system (Applied Biosystems, Foster City, CA, USA). Relative gene expression was calculated using the 2−ΔΔCt method, with GAPDH as the internal control36. The primer sequences for amplifying ETV7 were 5′-GTGCACGTGGGTGCCTAGGGG-3′ (forward) and 5′-GCTTGCACGTGGGTGCCTAG-3′ (reverse). The primer sequences for amplifying CXCL1 were 5′-CTCGAGGCCCCTGGGGCAGAAGCCTC-3′ (forward) and 5′-GATATCGGGGCTCAGCAGGCGGGTCT-3′ (reverse).
Proteins extracted from cells or tissues were separated by SDS-polyacrylamide gel electrophoresis (SDS-PAGE) and subsequently transferred onto polyvinylidene fluoride (PVDF) membranes (Pall Corp., Port Washington, NY, USA). Membranes were blocked with 5% non-fat milk and then incubated overnight at 4 °C with primary antibodies. The following primary antibodies were used: anti-ETV7 (1:1000, Sigma) and α-Tubulin (1:5000, Abmart). After incubation with HRP-conjugated secondary antibodies (anti-rabbit IgG, 1:3000; Cell Signaling Technology), protein bands were visualized using an enhanced chemiluminescence detection system (Pierce, Rockford, IL, USA)36.
Lentiviral Transfection
ETV7 overexpression and silencing lentiviruses were constructed by WZ Biosciences Inc (Shandong, China). Transfections were performed using POLO Deliver 3000 reagent (Remos Biotechnology, Shanghai, China). After transduction, cells were selected with 0.6 mg/mL puromycin for 14 days. Successful overexpression or knockdown was confirmed by RT-qPCR and Western blot36.
Mouse subcutaneous tumor formation model
BALB/C-nu/nu nude female mice (3 weeks old) were purchased from Guangdong Yaokang Biotechnology Co., Ltd. in China and were kept at 18–23 °C, 22–50% humidity, and 24 h (12/12 h) light/dark cycle. For quantitative analysis, at least three independent experiments were conducted for statistical analysis. RKO cells (4 × 10⁶) from each group (NC/ETV7) were subcutaneously injected into mice. Tumor volume (length × width² × 0.5) was measured every 4 days. 5-FU (30 mg/kg, i.p., twice weekly, Selleck, China) was administered concurrently with the following reagents. For the effect of shCXCL1, RKO cells transfected with ETV7 and shCXCL1 were injected subcutaneously into mice. To study the effect of DNase I, mice were administered DNase I (2.5 mg/kg, i.p., Roche) once daily after injection of RKO ETV7 cells, while the control group received PBS. For the effect of Anti-CXCL1, after injecting RKO ETV7 cells subcutaneously into mice, mice were administered Anti-CXCL1 (1 μg/kg, i.p., PeproTech) twice weekly, while the control group received isotype control IgG. The tumor burden was less than the maximum diameter (15 mm) approved by the Animal Experiment Ethics Committee of Sun Yat-sen University. Measure tumor volume using electronic calipers every 4 days along three perpendicular axes (volume = (length × width × height)/2). The mice were killed on the 20th day after surgery, and all mice were euthanized by carbon dioxide inhalation. Perform dissection, photograph, and weigh the tumor to determine its size. Tumor tissue was excised and fixed with 10% neutral formaldehyde, followed by paraffin embedding, dehydration, serial tissue sectioning, and HE staining. HE sections were observed under a light microscope. Ki-67 (Proteintech) immunohistochemical staining was performed. Immunofluorescence was used to detect neutrophil infiltration (anti-Ly-6G antibody, Abcam) and NETs formation (MPO, cit-H3) in tumor tissues. Three co-experimenters were responsible for grouping, treatment, and data collection using a blinded and randomized method. All experimental procedures were strictly conducted in accordance with the recommendations of the National Institutes of Health Guide for the Care and Use of Laboratory Animals. This protocol was approved by the Animal Experiment Ethics Committee of Sun Yat-sen University. We have complied with all relevant ethical regulations for animal use.
Enzyme-linked immunosorbent assay (ELISA)
Each assay was performed according to the manufacturer’s instructions. The human CXCL1 kit was purchased from Proteintech (China). For measuring MPO-DNA complexes, a capture ELISA was used to quantify NETs by identifying MPO-DNA complexes. A 96-well microtiter plate was coated with anti-MPO monoclonal antibody (Proteintech, 22225-1-AP) as the capture antibody (75 μl per well) and incubated overnight at 4 °C. After blocking with 1% BSA (125 μl per well), add 40 μl of sample and peroxidase-labeled anti-DNA monoclonal antibody (component 2 of the cell death detection ELISA kit, Roche, 11774425001), and incubate at room temperature for 2 h. Add the peroxidase substrate (ABTS) (Roche, 11774425001). After incubating at 37 °C in the dark for 40 min, measure the optical density at 405 nm using a microplate reader.
Human neutrophil separation
Neutrophils were isolated from human peripheral blood using dextran-Ficoll-Paque Premium (GE Healthcare) via density gradient separation. Detailed methods for isolating peripheral blood neutrophils are described in the section “Peripheral Blood Neutrophil Isolation: Isolation and Purification of Peripheral Blood Neutrophils” within the reference book (https://link.springer.com/book/10.1007/978-1-4939-8570-8#publish-with-us). The purity of the isolated neutrophils was confirmed to be >95% by immunofluorescence analysis of neutrophil markers. Unless otherwise specified, neutrophils were cultured in RPMI 1640 medium containing 20% FBS.
Sytox Green dye staining
For visualization, neutrophils cultured in conditioned medium (CM) from different CRC cells were seeded into 96-well plates for incubation, and the impermeable DNA dye SytoxGreen (Thermo Fisher Scientific, 1:10,000) and the permeable DNA dye Hoechst33342 (Thermo Fisher Scientific, 1:1000) were added to the incubation system. At the end of incubation, the plate was directly transferred to a fluorescence microscope to observe NETs formation. For each assay, three independent experiments were performed, each using neutrophils from different donors.
Immunofluorescence for NETs formation
For paraffin-embedded tissue samples, after dewaxing, antigen retrieval was performed using citric acid solution in a microwave oven (95 °C, 30 min). The samples were incubated overnight at 4 °C in a mixture of two primary antibodies. The primary antibodies used included anti-rabbit antibody cit-H3 (Abcam) and anti-mouse antibody MPO. The next day, after washing with cold PBS, the samples were incubated for 1 h at room temperature in the dark with a mixture of two different species-specific secondary antibodies. The secondary antibodies used included Alexa Fluor 488-labeled anti-rabbit antibody and Alexa Fluor 594-labeled anti-mouse antibody. Cells were re-stained with 4′,6-diamino-2-phenylpyridine (DAPI) (Sigma-Aldrich) to visualize cell nuclei. Slides were sealed with a mounting medium containing an anti-fluorescence quencher. Each sample was observed and photographed under a fluorescence microscope. NETs formation was determined by calculating the percentage of citrullinated histone 3 (cit-H3) positive cells within the field of view.
ChIP-qPCR experiment
ChIP was performed according to the instructions of the chromatin immunoprecipitation kit (Abcam, UK). Anti-ETV7 antibody and IgG were used as controls. Purified DNA was used for RT-qPCR, and primers specific for the CXCL1 promoter were designed. qPCR analysis was performed to measure fold enrichment by comparing the Ct values of the ETV7 immunoprecipitation fraction with the IgG isotype fraction, and normalization was performed using the ΔCt formula.
Dual luciferase reporter gene assay
All experiments were performed according to the kit instructions (Yeasen, China). Cells were seeded in 96-well plates, cell lysis buffer was transferred to black microplates, and firefly luciferase reaction solution was added, followed by measurement of firefly luciferase activity. After adding sea squirt luciferase reaction solution, activity was measured.
Human specimens
All experimental specimens involving patients were obtained from the First Affiliated Hospital of Sun Yat-sen University. These included fresh cancerous tissue and adjacent normal tissue from 12 colorectal cancer patients who underwent intestinal resection due to colorectal cancer. Paraffin-fixed colorectal cancer and adjacent normal tissue were obtained from 80 colorectal cancer patients who underwent intestinal resection at our hospital. All samples were obtained in accordance with the Declaration of Helsinki. The use of anonymized samples was approved by the First Affiliated Hospital of Sun Yat-sen University, and informed consent was waived. All ethical regulations relevant to human research participants were followed.
Statistical analysis and reproducibility
Error bars represent the mean ± SD. Statistical analysis was performed using Prism9 (GraphPad Software), ImageJ software, or SPSS Statistics 22 (IBM Corp.) software. All data followed a normal distribution with homogenous variance. When comparing two groups, P values were calculated using a two-tailed Student’s t-test. When comparing more than two groups, P values were calculated using analysis of variance (ANOVA). Survival curves were plotted using the Kaplan-Meier method. Correlation analysis was performed using Correlation of Ozone correlations. P < 0.05 was considered statistically significant (*,P < 0.05; **,P < 0.01; ***,P < 0.001; ****,P < 0.0001; ns indicates no statistical difference). Each experiment was independently repeated at least three times.
Ethics statement
All experiments involving patients were approved by the Ethics Committee of the First Affiliated Hospital of Sun Yat-sen University (approval number [2025] No. 630) and complied with the Declaration of Helsinki. All animal experiments were conducted under the approval of the Animal Ethics Committee of Sun Yat-sen University, ensuring ethical and humane treatment (approval number 2024002055).
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
References
Keum, N. & Giovannucci, E. Global burden of colorectal cancer: emerging trends, risk factors and prevention strategies. Nat. Rev. Gastroenterol. Hepatol. 16, 713–732 (2019).
Peeters, M. et al. Randomized phase III study of panitumumab with fluorouracil, leucovorin, and irinotecan (FOLFIRI) compared with FOLFIRI alone as second-line treatment in patients with metastatic colorectal cancer. J. Clin. Oncol. 28, 4706–4713 (2010).
Bartel, F. O., Higuchi, T. & Spyropoulos, D. D. Mouse models in the study of the Ets family of transcription factors. Oncogene 19, 6443–6454 (2000).
Pezzè, L. et al. ETV7 regulates breast cancer stem-like cell features by repressing IFN-response genes. Cell Death Dis. 12, 742 (2021).
Carella, C. et al. The ETS factor TEL2 is a hematopoietic oncoprotein. Blood 107, 1124–1132 (2006).
Alessandrini, F., Pezzè, L., Menendez, D., Resnick, M. A. & Ciribilli, Y. ETV7-mediated DNAJC15 repression leads to doxorubicin resistance in breast cancer cells. Neoplasia 20, 857–870 (2018).
Chai, B. et al. ETV7 promotes colorectal cancer progression through upregulation of IFIT3. Funct. Integr. Genom. 24, 8 (2024).
Cheng, J. et al. ETV7 limits the antiviral and antitumor efficacy of CD8(+) T cells by diverting their fate toward exhaustion. Nat. cancer 6, 338–356 (2025).
Meškytė, E. M. et al. ETV7 reduces inflammatory responses in breast cancer cells by repressing the TNFR1/NF-κB axis. Cell Death Dis. 14, 263 (2023).
Li, D. et al. DARS2 promotes bladder cancer progression by enhancing PINK1-mediated mitophagy. Int. J. Biol. Sci. 21, 1530–1544 (2025).
Qu, H. et al. Integrated analysis of the ETS family in melanoma reveals a regulatory role of ETV7 in the immune microenvironment. Front. Immunol. 11, 612784 (2020).
Mousset, A. et al. Neutrophil extracellular traps formed during chemotherapy confer treatment resistance via TGF-β activation. Cancer Cell 41, 757–775.e710 (2023).
Cristinziano, L. et al. Neutrophil extracellular traps in cancer. Semin. Cancer Biol. 79, 91–104 (2022).
Brinkmann, V. et al. Neutrophil extracellular traps kill bacteria. Science 303, 1532–1535 (2004).
Masucci, M. T., Minopoli, M., Del Vecchio, S. & Carriero, M. V. The emerging role of neutrophil extracellular traps (NETs) in tumor progression and metastasis. Front. Immunol. 11, 1749 (2020).
Adrover, J. M., McDowell, S. A. C., He, X. Y., Quail, D. F. & Egeblad, M. NETworking with cancer: the bidirectional interplay between cancer and neutrophil extracellular traps. Cancer cell 41, 505–526 (2023).
Albrengues, J. et al. Neutrophil extracellular traps produced during inflammation awaken dormant cancer cells in mice. Science 361, https://doi.org/10.1126/science.aao4227 (2018).
Shahzad, M. H. et al. Neutrophil extracellular traps in cancer therapy resistance. Cancers 14, https://doi.org/10.3390/cancers14051359 (2022).
Hu, W. et al. Neutrophil extracellular traps facilitate cancer metastasis: cellular mechanisms and therapeutic strategies. J. Cancer Res. Clin. Oncol. 149, 2191–2210 (2023).
Acharyya, S. et al. A CXCL1 paracrine network links cancer chemoresistance and metastasis. Cell 150, 165–178 (2012).
Teijeira, Á et al. CXCR1 and CXCR2 chemokine receptor agonists produced by tumors induce neutrophil extracellular traps that interfere with immune cytotoxicity. Immunity 52, 856–871.e858 (2020).
Xiong, G. et al. CD276 regulates the immune escape of esophageal squamous cell carcinoma through CXCL1-CXCR2 induced NETs. J. Immunother. Cancer 12, https://doi.org/10.1136/jitc-2023-008662 (2024).
Chen, H. et al. METTL3 inhibits antitumor immunity by targeting m(6)A-BHLHE41-CXCL1/CXCR2 axis to promote colorectal cancer. Gastroenterology 163, 891–907 (2022).
Xie, S. Z. et al. Targeting SPP1-orchestrated neutrophil extracellular traps-dominant pre-metastatic niche reduced HCC lung metastasis. Exp. Hematol. Oncol. 13, 111 (2024).
Demers, M. et al. Priming of neutrophils toward NETosis promotes tumor growth. Oncoimmunology 5, e1134073 (2016).
Nie, M. et al. Neutrophil extracellular traps induced by IL8 promote diffuse large B-cell lymphoma progression via the TLR9 signaling. Clin. Cancer Res. 25, 1867–1879 (2019).
Chen, L. et al. The lipid-metabolism enzyme ECI2 reduces neutrophil extracellular traps formation for colorectal cancer suppression. Nat. Commun. 15, 7184 (2024).
Németh, T., Sperandio, M. & Mócsai, A. Neutrophils as emerging therapeutic targets. Nat. Rev. Drug Discov. 19, 253–275 (2020).
Jaillon, S. et al. Neutrophil diversity and plasticity in tumour progression and therapy. Nat. Rev. Cancer 20, 485–503 (2020).
He, D. et al. Chemotherapy awakens dormant cancer cells in lung by inducing neutrophil extracellular traps. Cancer Cell. https://doi.org/10.1016/j.ccell.2025.06.007 (2025).
Cools-Lartigue, J. et al. Neutrophil extracellular traps sequester circulating tumor cells and promote metastasis. J. Clin. Investig. 123, 3446–3458 (2013).
Zhan, X. et al. Elevated neutrophil extracellular traps by HBV-mediated S100A9-TLR4/RAGE-ROS cascade facilitate the growth and metastasis of hepatocellular carcinoma. Cancer Commun. 43, 225–245 (2023).
Xiao, Y. et al. Cathepsin C promotes breast cancer lung metastasis by modulating neutrophil infiltration and neutrophil extracellular trap formation. Cancer Cell 39, 423–437.e427 (2021).
He, X. Y. et al. Chronic stress increases metastasis via neutrophil-mediated changes to the microenvironment. Cancer Cell 42, 474–486.e412 (2024).
Tan, H. et al. Cryoablation-induced neutrophil Ca(2+) elevation and NET formation exacerbate immune escape in colorectal cancer liver metastasis. J. Exp. Clin. Cancer Res. 43, 319 (2024).
Chen, L. et al. CSRP2 suppresses colorectal cancer progression via p130Cas/Rac1 axis-meditated ERK, PAK, and HIPPO signaling pathways. Theranostics 10, 11063–11079 (2020).
Acknowledgements
We appreciate the effort of the physicians in enrolling patients and thank all the patients involved for allowing us to analyze their clinical data. This work was supported by the National Natural Science Foundation of China (Grant Nos. 82403041, 82103504) and the China Postdoctoral Science Foundation (Grant No. 2025M771958).
Author information
Authors and Affiliations
Contributions
S.M., P.X., and Y.R.L. performed the experiments, analyzed the data, and drafted the manuscript. L.L. and L.C. collected CRC tissue and performed IHC. S.J.H. and H.B.G. assisted with the animal experiments. S.M. contributed to the data analysis. J.Q.W., A.J.H., and L.X.C. designed the experiments and revised the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Biology thanks Rakesh Rachamadugu and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editors: Prof. Toshiro Moroishi and Dr. Nilanjan Banerjee & Dr. Christina Karlsson Rosenthal.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Mo, S., Xia, P., Lv, Y. et al. ETV7 promotes 5-FU resistance and malignant progression through CXCL1-induced NETs formation in colorectal cancer. Commun Biol 9, 472 (2026). https://doi.org/10.1038/s42003-026-09976-2
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s42003-026-09976-2