Abstract
Under predicted future ocean conditions, corals will experience frequent and intense thermal stress events while simultaneously being exposed to chronic ocean acidification. Yet, some corals will likely be more resistant and/or resilient to these predicted conditions than others and may be critical to reef persistence in the future. Following natural thermal stress in two consecutive years (2014 and 2015), we evaluated the effects of feeding and simulated ocean acidification on the physiological recovery of Montipora capitata and Porites compressa sourced from Kāneʻohe Bay and Waimānalo Bay, Hawaiʻi. Following the 2014 thermal stress event, simulated ocean acidification did not slow recovery of the holobiont and feeding enhanced recovery. However, feeding did not decrease susceptibility to the 2015 thermal stress event, and simulated ocean acidification did not increase susceptibility. Recovery strategies employed between species and between sites clearly differed, highlighting that coral reef restoration and management should consider species-level and site-specific vulnerabilities. Overall, our findings call attention to the immediate threat which ocean warming presents, the lack of additional stress to the holobiont from ocean acidification, the importance of heterotrophy in coral resilience, and the potential significance of additional local biotic stressors (i.e., predator outbreaks) for coral resiliency under annual thermal stress.
Similar content being viewed by others
Introduction
Global climate change is leading to increasing sea surface temperatures and ocean acidification (OA)1, threatening coral reefs worldwide e.g., refs. 2,3,4,5. Mean global ocean temperature is expected to increase by 0.2–3.5 °C by 2100, relative to 1995–2014, depending on the emissions scenario1, but will most likely reach 1–2 °C above present-day temperatures (or 2–3 °C above the pre-industrial) given recent global commitments6. As corals already live close to their upper thermal tolerance limits7, this baseline temperature shift is increasing the frequency and severity of mass coral bleaching events e.g., refs. 4,8,9 resulting in decreased coral health and growth, and increased mortality e.g., refs. 9,10,11. Bleaching temperature thresholds vary regionally e.g., refs. 12,13,14 while susceptibility, physiological response, and recovery rate (i.e., resilience) varies among species, within species, and among individuals e.g., refs. 14,15,16,17. With some locations now experiencing thermal stress events in two or more consecutive years, it is expected that temperatures that result in bleaching among present-day communities will occur annually in regions like the Caribbean by 2030, and globally by 205018,19.
Concurrently, ocean surface pH is expected to decrease by up to 0.37 pH units by the end of this century, relative to 1995–2014, depending on the emissions scenario, with the most likely change being a decrease of 0.1–0.2 pH units relative to present-day (or 0.2–0.3 pH units relative to the pre-industrial) given recent global commitments6. Such changes in the carbonate chemistry will result in a decrease in aragonite saturation state. Under OA, coral skeletal growth (i.e., calcification) appears to be energetically costly20, resulting in decreased calcification rates in many e.g., refs. 21,22,23, but not all corals e.g., refs. 23,24,25,26, and slowing reef accretion e.g., refs. 27,28,29,30. Previous studies on the combined effects of elevated temperature and OA on coral calcification have produced varying results among species e.g., refs. 5,14,23,31,32 and may depend on the experiment duration. Studies of two-months duration or less suggest that OA can increase bleaching susceptibility in some corals e.g., refs. 23,33,34,35 while studies of longer duration do not e.g., refs. 25,26,31,36,37. However, it is unknown if OA prolongs coral recovery following bleaching, or if OA increases the bleaching susceptibility of corals experiencing thermal stress in consecutive years.
Heterotrophic feeding can mitigate many of the negative effects of thermal and/or OA stress on corals, including mitigating declines in the carbon budget e.g., refs. 33,38,39,40,41,42,43,44. Following bleaching, heterotrophically derived carbon is the primary fixed carbon source for tissue repair and lipid synthesis45,46,47. Corals with high feeding rates and/or the capacity to increase their feeding rate or those with large lipid reserves to catabolize during thermal stress, bleaching, and recovery, will likely be more resilient e.g., refs. 23,42,43,48,49. Therefore, providing corals with zooplankton post-bleaching could supplement the coral’s nutritional needs, promote the retention and/or synthesis of new lipid reserves, minimize mortality, accelerate recovery rates, and may also mitigate any negative effects of OA on corals.
The aim of this study was to experimentally evaluate the potential effects of simulated OA and zooplankton availability (termed “feeding”) on two dominant Hawaiian coral species50 (Montipora capitata and Porites compressa) sourced from two locations around Oʻahu, Hawaiʻi (Moku o Loʻe within Kāneʻohe Bay, and Waimānalo Bay; Fig. 1A) following the 2014 thermal stress event. Coincidentally, our experiment also captured a second, unexpected thermal stress event in 2015. To the best of our knowledge, this is the first study to capture the impact of natural thermal stress in two consecutive years (2014 and 2015) and to evaluate the effects of simulated OA and feeding on long-term coral recovery following bleaching. Following the 2014 heat-stress event, we hypothesized that OA prolonged recovery of corals, but that heterotrophic feeding on zooplankton mitigated the negative effects of OA on recovery from bleaching. Following the unexpected 2015 heat-stress event, a posteriori we hypothesized that OA increased the bleaching susceptibility of corals, but that feeding mitigated the negative effect of OA-induced bleaching susceptibility. Following both heat-stress events, we hypothesized that the coral responses were species- and site-specific.
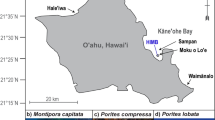
A Map of the two collection sites around the island of Oʻahu; Moku o Loʻe reef (21°26’06” N, 157°47’12” W) in Kāneʻohe Bay, and Waimānalo Bay (21°19’36” N, 157°40’54” W). B Environmental conditions in situ on the reef and in the experimental tanks. Mean weekly sea surface temperature (SST) for Moku o Loʻe reef in Kāneʻohe Bay (dark gray line; data sourced from NOAA National Ocean Service Station MOKH1) and Waimānalo Bay (light gray line; data sourced from PacIOOS Station 51202), showing seasonal temperature variability in situ between August 2014 and December 2015. The Coral Reef Watch Bleaching Watch (yellow shading), Bleaching Warning (green shading), and the 2014 and 2015 bleaching events (Alert Levels 1 and 2; red shading) are also shown (https://coralreefwatch.noaa.gov/product/5km/index_5km_baa-max-7d.php). The bleaching threshold sea surface temperature (SST) for the Hawaiian Archipelago is 28 °C (horizontal dashed orange line). Average weekly experimental tanks temperature (black line) and pHT in situ (Control and Fed treatments = solid blue line; OA and OA+Fed treatments = solid red line) throughout the experiment. All means are plotted ±1 standard deviation. Tank temperature SDs are smaller than the line and not visible. pH in the ambient and simulated OA tanks was significantly different over the course of the experiment (chi-squared = 297.17, df = 1, p < 2.2e−16). C Experimental design schematic: twelve ramets were collected from each of six initially non-bleached and six bleached parent colonies of Montipora capitata and Porites compressa from Kāneʻohe Bay and Waimānalo Bay (HI, USA) in November 2014. These ramets were distributed among all eight experimental tanks (two tanks per treatment). Treatments included: 1) Control (ambient seawater of pH 7.96 and unfed), 2) Fed (ambient seawater of pH 7.96 and fed), 3) OA (acidified seawater of pH 7.73 and unfed), and 4) OA+Fed (acidified seawater pH 7.73 and fed). Tanks are illustrated in ordered blocks for clarity, but in reality they were randomly positioned. One ramet from each parent colony of each bleaching status, species, site, and treatment were randomly removed from the experiment in December 2014, June 2015, and November 2015 for non-destructive and destructive physiological analysis. In total, 96 ramets (i.e., 1 ramet × 6 parent colonies × 2 health status states × 2 sites × 4 treatments) were assigned per time-point per species.
Results
Over the duration of the experiment, the mean pH (±SD; reported on the total hydrogen ion scale) of the Control and Fed treatment tanks was pH 7.96 ± 0.04 and the mean pH of the OA and OA+Fed treatment tanks was pH 7.73 ± 0.10 (Fig. 1B; Table 1). Average ambient and simulated OA seawater pH significantly differed over the duration of the study (Fig. 1B, Table 1, chi-squared = 297.17, df = 1, p < 2.2e−16). This difference in pH was maintained when tanks were measured daily during the physiological measurements (Supplementary Fig. 1; December 2014, chi-squared = 41.393, df = 1, p = 1.245e−10; June 2015, chi-squared = 57.912, df = 1, p = 2.74e−14; November 2015, chi-squared = 33.802, df = 1, p = 6.103e−09) and over the course of three 24-h measurement periods (Supplementary Fig. 2; December 2014, chi-squared = 47.262, df = 1, p = 6.212e−12; March 2015, chi-squared = 47.262, df = 1, p = 6.212e−12; June 2015, chi-squared = 47.263, df = 1, p = 6.208e−12). The average pH difference between the ambient and simulated OA treatments was 0.2 (Table 1).
Mean tank temperature was 25.9 °C ± 1.8 °C over the course of the 12-month study, ranging from 20–30 °C (Fig. 1B, Table 1), and reflected both the diurnal and seasonal variability on the reef and the 2015 thermal stress event (Fig. 1B; Table 1). Weekly mean tank temperature did not statistically differ from the weekly mean reef temperature in Kāneʻohe Bay (at 18:00 h; t = 0.19, df = 106, p = 0.85). Salinity averaged 34.4 ± 0.5 ppt across tanks over the duration of the experiment (Table 1).
Effect of simulated OA and feeding on coral physiology following the 2014 thermal stress event
Ninety-eight percent of all experimental corals survived to December 2014 (Supplementary Table 1). While 98% of remaining M. capitata survived to June 2015 (Supplementary Table 1), predation by Phestilla sp. increased mortality such that only 68% of remaining P. compressa survived to that point (Supplementary Table 1).
Montipora capitata from Kāneʻohe Bay
In November 2014, one month after 2014 peak thermal stress, bleached parent colonies on the reef were visibly pale, had lower endosymbiont cell density, but similar tissue biomass and lipids compared to non-bleached parent colonies (Supplementary Fig. 3A–C; Supplementary Table 2). By December 2014, bleached and non-bleached parent colonies no longer differed from each other (Supplementary Fig. 3D–F; Supplementary Table 2).
In the experimental tanks, the physiological profiles of bleached and non-bleached ramets differed from each other in all treatments except the OA treatment by December 2014 (Table 2). Bleached ramets had lower endosymbiont cell density and calcification than non-bleached ramets, irrespective of OA and feeding, but did not differ in tissue biomass, CTAR, nor the underlying traits of CTAR (Fig. 2A–D and Supplementary Fig. 4A–E; Supplementary Tables 3 and 4). Total lipids were lower in bleached corals than in non-bleached corals when fed (Fig. 2E; Supplementary Table 3). By June 2015, after seven months in the experimental tanks, the physiological profiles of bleached and non-bleached ramets differed in all treatments except the OA+Fed treatment (Table 2). Bleached ramets in all treatments still had lower endosymbiont cell density compared to non-bleached ramets, coupled with higher lipids (Fig. 2F–J; Supplementary Table 3). While feeding capacity and CHAR increased in bleached ramets compared to non-bleached ramets in the Fed treatment, this was not the case in the OA+Fed treatment (Supplementary Fig. 4I–J; Supplementary Table 4). CTAR was, however, higher in the bleached ramets and in corals under OA, when fed (Fig. 2G; Supplementary Table 3).
Physiological measurements of Kāneʻohe Bay Montipora capitata experimental ramets in December 2014 (A–E) and June 2015 (F–J) for ramets that were originally non-bleached (solid gray bars) or bleached (open white bars) at the time of collection in November 2014. Mean (±1 SE) (A, F) endosymbiont cell density, (B, G) Contribution of Total carbon to Animal Respiration, CTAR, (C, H) calcification, and (D, I) tissue biomass, and (E, J) total lipids are shown for ramets that were kept in the Control (ambient seawater of pH 7.96 and unfed), Fed (ambient seawater of pH 7.96 and fed), OA (acidified seawater of pH 7.73 and unfed), and OA+Fed (acidified seawater pH 7.73 and fed) treatments. The dotted line in B and G indicates 100%. Sample sizes for each mean are indicated within each bar. Measurements were not made on ramets that were dead or nearly dead (i.e., <25% live tissue). Significant ANOVA main effects are listed in the top right corner of each panel. Letters indicate significant differences within each panel. Full ANOVA details are in Supplementary Table 3. The underlying data used to calculate CTAR (i.e., photosynthesis, respiration, feeding capacity, CZAR, and CHAR) are in Supplementary Fig. 4 and accompanying statistics are in Supplementary Table 4.
Montipora capitata from Waimānalo Bay
In November 2014, one month after 2014 peak thermal stress, bleached parent colonies on the reef were visibly pale though their endosymbiont cell density, tissue biomass, and total lipids did not differ from that of non-bleached parent colonies (Supplementary Fig. 5; Supplementary Table 5).
In the experimental tanks, the physiological profiles of bleached and non-bleached ramets did not differ from each other within any treatment in December 2014 and June 2015 (after one and seven months in the experimental tanks, respectively) (Table 2). While there was a trend (p = 0.002) of lower endosymbiont cell density in bleached compared to non-bleached ramets in December 2014, irrespective of OA and feeding (i.e., bleaching effect was significant, but the overall model was not) (Fig. 3A; Supplementary Table 6), there were no other significant differences between bleached and non-bleached ramet physiological traits (Fig. 3A–E and Supplementary Fig. 6A–E; Supplementary Tables 6 and 7). By June 2015, no significant effects were detected in any of the measured physiological traits (Fig. 3F–J and Supplementary Fig. 6F–J; Supplementary Tables 6 and 7).
Physiological measurements of Waimānalo Bay Montipora capitata experiment ramets in December 2014 (A–E) and June 2015 (F–J) for ramets that were originally non-bleached (solid gray bars) or bleached (solid white bars) at the time of collection in November 2014. Mean ( ± 1 SE) (A, F) endosymbiont cell density, (B, G) Contribution of Total carbon to Animal Respiration, CTAR, (C, H) calcification, and (D, I) tissue biomass, and (E, J) total lipids are shown for ramets that were kept in the Control (ambient seawater of pH 7.96 and unfed), Fed (ambient seawater of pH 7.96 and fed), OA (acidified seawater of pH 7.73 and unfed), and OA+Fed (acidified seawater pH 7.73 and fed) treatments. The dotted line in B and G indicates 100%. Sample sizes for each mean indicated within each bar. Full ANOVA details are in Supplementary Table 6. The underlying data used to calculate CTAR (i.e., photosynthesis, respiration, and feeding capacity, CZAR, and CHAR) are in Supplementary Fig. 6 and accompanying statistics are in Supplementary Table 7.
Porites compressa from Kāneʻohe Bay
In November 2014, one month after 2014 peak thermal stress, bleached parent colonies on the reef were visibly pale, had lower endosymbiont cell density, but similar tissue biomass and total lipids, compared to non-bleached parent colonies (Supplementary Fig. 7A–C; Supplementary Table 8). By December 2014, all measured physiological traits (endosymbiont cell density, tissue biomass, and total lipids) were significantly lower in the bleached compared to the non-bleached parent colonies (Supplementary Fig. 7E–H; Supplementary Table 8), but no longer differed by June 2015 (Supplementary Fig. 7I–L; Supplementary Table 8).
In the experimental tanks, the physiological profiles of the bleached ramets differed from the non-bleached ramets in the Fed and OA treatments, but not in the Control or OA+Fed treatments in December 2014 (Table 2). Across all treatments, endosymbiont cell densities, calcification, and total lipids were significantly lower in bleached than in non-bleached ramets (Fig. 4A–E, Supplementary Table 9). Calcification was also lower in fed ramets than in non-fed ramets (Fig. 4C; Supplementary Table 9). In the Fed treatment only, CTAR was higher in bleached compared to non-bleached ramets (Fig. 4B; Supplementary Table 9), due to higher CZAR (Supplementary Fig. 8C; Supplementary Table 10). Finally, tissue biomass was lower in bleached compared to non-bleached ramets in the Fed and OA treatments (Fig. 4D; Supplementary Table 9). By June 2015, after seven months in the experimental tanks, the physiological profiles of surviving experimental ramets no longer differed between bleached and non-bleached ramets (Table 2), although endosymbiont cell density was still lower in bleached compared to non-bleached ramets (Fig. 4F; Supplementary Table 9). No other measured physiological trait differed (Fig. 4G–J and Supplementary Fig. 8F–J; Supplementary Tables 9 and 10).
Physiological measurements of Kāneʻohe Bay Porites compressa experimental ramets in December 2014 (A–E) and June 2015 (F–J) for ramets that were originally non-bleached (solid gray bars) or bleached (solid white bars) at the time of collection in November 2014. Mean (±1 SE) (A) endosymbiont cell density, (B) Contribution of Total carbon to Animal Respiration, CTAR, (C) calcification, and (D) tissue biomass, and I total lipids are shown for ramets that were kept in the Control (ambient seawater of pH 7.96 and unfed), Fed (ambient seawater of pH 7.96 and fed), OA (acidified seawater of pH 7.73 and unfed), and OA+Fed (acidified seawater pH 7.73 and fed) treatments. The dotted line in B and G indicates 100%. Sample sizes for each mean indicated within each bar. Significant ANOVA main effects are listed in the top right corner of each panel. Letters indicate significant differences within each panel. Full ANOVA details are in Supplementary Table 9. The underlying data used to calculate CTAR (i.e., photosynthesis, respiration, and feeding capacity, CZAR, and CHAR) are in Supplementary Fig. 8 and accompanying statistics are in Supplementary Table 10.
Porites compressa from Waimānalo Bay
In November 2014, one month after 2014 peak thermal stress, bleached parent colonies on the reef were visibly pale, had lower endosymbiont cell densities and total lipid concentrations compared to non-bleached parent colonies, while tissue biomass did not differ (Supplementary Fig. 9; Supplementary Table 11).
In the experimental tanks, the physiological profiles of the bleached and non-bleached ramets differed within all treatments in December 2014 (Table 2). Endosymbiont cell density and total lipids were lower in bleached compared to non-bleached ramets (Fig. 5A, E; Supplementary Table 12). At the same time, CTAR was higher in bleached compared to non-bleached ramets (Fig. 5B; Supplementary Table 12) due to higher CZAR (Supplementary Fig. 10C; Supplementary Table 13). CZAR was also higher in ramets under OA treatments compared to those in ambient pH treatments (Supplementary Fig. 10C; Supplementary Table 13) and CHAR was higher in bleached ramets compared to non-bleached ramets (Supplementary Fig. 10E; Supplementary Table 13). Calcification was higher in the OA treatment than in all other treatments irrespective of bleaching status (Fig. 5C; Supplementary Table 12) and there was no difference in tissue biomass across treatments (Fig. 5D; Supplementary Table 12). By June 2015, after seven months in experimental tanks, physiological profiles no longer differed between bleached and non-bleached ramets in the fed treatments (Table 2), though there were insufficient survivors to determine if physiological profiles differed between bleached and non-bleached ramets in the unfed treatments. It was possible to conduct univariate analyses with the small sample sizes and calcification was higher in bleached ramets compared to non-bleached counterparts (Fig. 5H; Supplementary Table 12), while there was no difference in endosymbiont density, CTAR, biomass, or total lipids (Fig. 5A–B, I-J; Supplementary Table 12).
Physiological measurements of Waimānalo Bay Porites compressa experimental ramets in December 2014 (A–E) and June 2015 (F–J) for ramets that were originally non-bleached (solid gray bars) or bleached (solid white bars) at the time of collection in November 2014. Mean (±1 SE) (A, F) endosymbiont cell density, (B, G) Contribution of Total carbon to Animal Respiration, CTAR, (C, H) calcification, and (D, I) tissue biomass, and (E, J) total lipids are shown for ramets that were kept in the Control (ambient seawater of pH 7.96 and unfed), Fed (ambient seawater of pH 7.96 and fed), OA (acidified seawater of pH 7.73 and unfed), and OA+Fed (acidified seawater pH 7.73 and fed) treatments. The dotted line in B and G indicates 100%. Sample sizes for each mean indicated within each bar. Significant ANOVA main effects are listed in the top right corner of each panel. Letters indicate significant differences within each panel. Full ANOVA details are in Supplementary Table 12. The underlying data used to calculate CTAR (i.e., photosynthesis, respiration, and feeding capacity, CZAR, and CHAR) are in Supplementary Fig. 10 and accompanying statistics are in Supplementary Table 13.
Effect of simulated OA and feeding on M. capitata physiology after the 2015 thermal stress event
Parent colonies of M. capitata on the Kāneʻohe Bay reef that had bleached in 2014, bleached again in 2015 and the non-bleached colonies remained non-bleached in 2015 (Supplementary Fig. 11; Supplementary Table 14). Bleached colonies had lower endosymbiont cell densities but similar total lipid concentrations and tissue biomass as the non-bleached parent colonies (Supplementary Fig. 11A–C; Supplementary Table 14).
In the experimental tanks, 79% and 56% of remaining M. capitata ramets from Kāneʻohe Bay and Waimānalo Bay survived, respectively, until November 2015 (Supplementary Table 1). However, likely in part due to the Phestilla outbreak, survivorship of P. compressa was very low, with only 25% and 21% surviving from Kāneʻohe Bay and Waimānalo Bay, respectively (Supplementary Table 1), often with very little remaining live tissue, and no additional physiological analyses were performed. The physiological profiles of the surviving bleached and non-bleached ramets of M. capitata originally sourced from Kāneʻohe Bay did not differ from each other within any treatment (Table 2). However, bleached ramets had lower endosymbiont cell densities compared to non-bleached ramets (Fig. 6A; Supplementary Table 15), but did not differ in CTAR, calcification, tissue biomass, or total lipids (Fig. 6B–E; Supplementary Table 15). Under ambient pH, CTAR was lower in Control ramets compared to all other treatments irrespective of bleaching status, due to lower CZAR levels (Fig. 6B and Supplementary Fig. 12C; Supplementary Tables 15 and 16). The physiological profiles of the surviving bleached and non-bleached ramets of M. capitata originally sourced from Waimānalo Bay only differed within the Control treatment (Table 2). However, bleached ramets had lower endosymbiont cell density compared to non-bleached counterparts, irrespective of treatment (Fig. 6F; Supplementary Table 15) and higher calcification in OA treatments (Fig. 6H; Supplementary Table 15).
Physiological measurements of Montipora capitata experimental ramets from Kāneʻohe Bay (A–E) and Waimānalo Bay (F–J) in November 2015 following the second heat-stress event in this study, for ramets that were originally non-bleached (solid gray bars) or bleached (solid white bars) at the time of collection in November 2014. Mean (±1 SE) (A, F) endosymbiont cell density, (B, G) Contribution of Total carbon to Animal Respiration, CTAR, (C, H) calcification, and (D, I) tissue biomass, and (E, J) total lipids are shown for ramets that were kept in the Control (ambient seawater of pH 7.96 and unfed), Fed (ambient seawater of pH 7.96 and fed), OA (acidified seawater of pH 7.73 and unfed), and OA+Fed (acidified seawater pH 7.73 and fed) treatments. The dotted line in B and G indicates 100%. Sample sizes for each mean indicated within each bar. Significant ANOVA main effects are listed in the top right corner of each panel. Letters indicate significant differences within each panel. Full ANOVA details are in Supplementary Table 15. The underlying data used to calculate CTAR (i.e., photosynthesis, respiration, and feeding capacity, CZAR, and CHAR) are in Supplementary Fig. 12 and accompanying statistics are in Supplementary Table 16.
Discussion
Effect of simulated OA on bleaching recovery and susceptibility
Simulated OA did not prolong the recovery of overall coral holobiont physiology of either species from either site following the 2014 bleaching event (Table 2), as evidenced by the similarity between bleached ramets in the Control and OA treatments for almost every variable, species, and site in December 2014 and June 2015 (Figs. 2–5 and Supplementary Figs. 3–10; Table 2). Furthermore, simulated OA did not increase physiological susceptibility to annual bleaching in surviving M. capitata experimental ramets from both sites (Fig. 6). The absence of any systematic negative OA effects observed here supports a growing body of literature suggesting that temperature is of greater concern than OA for the persistence of coral reefs in the future e.g., refs. 5,14,23,25,31,36,37,51.
Despite this, some nuanced effects of OA were observed during the recovery of the bleached ramets for M. capitata from Kāneʻohe Bay. By June 2015 simulated OA depressed heterotrophic plasticity in bleached ramets (Supplementary Fig. 4I, J) and depressed CZAR and CTAR irrespective of bleaching status unless fed (Supplementary Figs. 4H and 2G respectively; Supplementary Tables 3 and 4). Elevated temperature52 and pCO253 alone decrease feeding rates in some coral species, though bleached M. capitata has been shown to increase feeding rate following thermal stress42. Our results suggest that the combination of elevated temperature and OA predicted in future oceans may ultimately affect the resiliency of this species and that feeding will be important for maintaining carbon budgets even when heterotrophic plasticity is affected.
It is particularly notable that simulated OA did not prolong recovery of calcification following temperature stress for either species within this study. Calcification may be energetically costly54,55 and therefore is often reduced under physiologically stressful conditions, presumably to conserve energy e.g., refs. 5,40,56,57,58. Here, while both species from Kāneʻohe Bay decreased calcification in response to bleaching, bleached corals fully recovered calcification in parallel with other measured physiological traits (CTAR, tissue biomass, and total lipids) within 8 months of initial bleaching, irrespective of treatment. This calcification recovery rate is consistent with previous findings for bleached M. capitata under ambient pH but is faster than that previously reported for bleached P. compressa57, supporting our finding that OA did not prolong recovery from bleaching. Our findings suggest that within this study, either: 1) corals were able to sustain sufficiently high rates of photosynthesis, the primary source of fixed carbon for calcification46, and offset any possible negative OA effects on calcification; 2) corals were able to take up additional dissolved or particulate organic carbon to sustain metabolic demand49,59 and support calcification under simulated OA conditions; or that 3) simulated OA was not physiologically stressful to these corals. Corals in this study sustained photosynthesis rates under OA (Supplementary Figs. 4, 6, 8, 10). While particulate organic carbon can be pivotal for meeting metabolic demand in bleached corals44, it was not measured here. In a 22-month experiment, calcification of surviving M. capitata and P. compressa was unaffected by OA26. Thus, it is likely that modest OA is not physiologically stressful, particularly when coral symbionts are photosynthesizing efficiently and/or corals have access to heterotrophic feeding.
Effect of heterotrophic feeding on bleaching recovery and susceptibility
While no systematic negative effects of simulated OA on coral bleaching recovery were observed within this study, our results support the growing body of evidence that heterotrophic feeding is important for coral recovery from bleaching e.g., refs. 15,42,45,52,60,61,62,63. For example, when compared to their non-bleached counterparts, surviving bleached P. compressa sourced from Kāneʻohe Bay in the Fed treatment exhibited enhanced CTAR in December 2014 (Fig. 4B), bleached M. capitata sourced from Kāneʻohe Bay in the Fed treatment had double the feeding rate in June 2015 (Supplementary Fig. 4I), and bleached P. compressa sourced from Waimānalo Bay in the OA+Fed treatment had double the calcification in June 2015 (Fig. 5H). Both M. capitata and P. compressa are known to rely heavily on heterotrophy to meet metabolic demand following bleaching15,43, for rebuilding tissues and total lipids for at least 11 months post-bleaching45,46,47, and to net-translocate heterotrophically fixed carbon from the host to the endosymbiotic algae when bleached46. Our findings suggest that the importance of heterotrophy also includes enhancing calcification in some populations of bleached corals.
In Kāneʻohe Bay, in situ bleached parent colonies recovered all measured physiological parameters (i.e., endosymbiont cell density, tissue biomass, and total lipids) prior to their experimental counterparts in the Fed treatments (note, data was not available for the in-situ parent colonies from Waimānalo Bay). Bleached M. capitata and P. compressa parent colonies on the reef in Kāneʻohe Bay recovered all measured parameters within 2 months and 8 months, respectively, while their experimental counterparts in the Fed treatments recovered their tissue biomass and total lipids but not their endosymbiont density within 8 months. This suggests that the number of zooplankton provided to the Fed corals in this study may have been insufficient compared to the zooplankton availability on the reef. Thus, our estimates of the impact of feeding on coral physiological responses to single and annual bleaching are likely conservative and it is possible that the impact of feeding on coral physiology is much greater in situ than what is reported from this study. We acknowledge that recovery rate of corals in the field could also be due to parameters not evaluated (i.e., presence of grazers) or measured (i.e., contribution of particulate organic carbon) within the experiment.
Feeding, however, did not decrease the susceptibility of M. capitata to annual bleaching. Genets that had bleached in 2014 bleached again in 2015, irrespective of feeding treatment or site of origin (Fig. 6A, F; Supplementary Table 15). However, feeding stimulated CZAR, which bolstered CTAR in repeat bleached M. capitata from Kāneʻohe Bay (Fig. 6B, Supplementary Fig. 12C), and may optimize recovery from annual bleaching in M. capitata. Any boost to CZAR was not reflected in CHAR as feeding rates did not increase in response to repeat bleaching in the Fed treatments (Supplementary Fig. 12D, E, I, J). A loss of heterotrophic plasticity following repeat bleaching has been previously reported for three species of Caribbean corals44, making it harder to predict resistance to annual bleaching events15.
Resilience varies by species and location
The physiological profiles of each species-site combination responded differently throughout the experiment (Tables 2, 3, and Supplementary Table 17), with each employing a different recovery strategy (Figs. 2–6). Following the 2014 heat-stress event, M. capitata appeared more resilient than P. compressa. By June 2015, M. capitata displayed four distinct advantages over P. compressa. First, while both species catabolized lipid reserves when bleached, non-bleached and bleached M. capitata had 76% and 236% more total lipids than did P. compressa, respectively (Figs. 2J and 3J compared to Figs. 4J and 5J), which is consistent with previous studies on these species in Hawaiʻi48,64. Higher lipids are known to be associated with higher resilience in corals48,65,66. Second, bleached M. capitata from Kāneʻohe Bay increased feeding in response to bleaching at ambient pH (Supplementary Fig. 4I). Montipora capitata switches between autotrophy and heterotrophy and is thus more heterotrophically plastic42,43. Both high baseline feeding and heterotrophic plasticity in response to bleaching are associated with coral resilience e.g., refs. 42,44. Third, endosymbiotic algae associated with M. capitata include both Cladocopium (C31 and C21) and the thermally tolerant Durisdinium (D1 and D4), whereas P. compressa associates almost exclusively with Cladocopium (C15)67,68,69,70,71. Following heat-stress, the proportionate contribution of Durisdinium (D4 and D1) in M. capitata from Kāneʻohe Bay increases significantly among colonies that were less susceptible to bleaching16,71, which may increase the probability of persistence of this species under climate change conditions. At the same time, the endosymbiont community in P. compressa remains virtually unchanged following heat-stress, which may increase the sensitivity of this species to future climate change conditions71. Finally, M. capitata is not susceptible to predation by nudibranchs of the genus Phestilla that are specialists on Porites corals72,73. Phestilla infestation is at least partially responsible for the disproportionate mortality of P. compressa (Supplementary Table 1). We acknowledge, however, that in the field, P. compressa would likely have fared better as Phestilla infestations are minimized in the presence of fish and crustacean predators74.
While the physiological recovery rates of survivors were faster in corals of both species from Waimānalo Bay compared to Kāneʻohe Bay (Fig. 2 vs 3, and 4 vs 5), the benefits of these faster recovery rates are overshadowed by the higher survivorship among corals from Kāneʻohe Bay, especially following the 2015 heat-stress event (Supplementary Table 1). The cumulative negative effects of repeated bleaching have also been observed in Caribbean and Hawaiian corals15,51 and annual bleaching can lead to selection for thermally tolerant corals75. Indeed, coral resilience can be a function of acclimation26, or selection for more stress tolerant individuals leading to thermal adaptation76,77. In Kāneʻohe Bay, summer seawater temperatures are naturally 1–2 °C higher14,78,79 and pH is 0.1–0.2 units lower14,79,80 than in Waimānalo Bay. Thus, corals from Kāneʻohe Bay are preconditioned to withstand higher temperatures and lower pH conditions than conspecifics from Waimānalo Bay14,26,51,81, which may in part explain these survivorship findings.
Previous studies have also found M. capitata to be more resilient than P. compressa to heat-stress e.g., refs. 42,48,57,82 and future climate change scenarios31. However, this contrasts with results of a field survey of the corals in Kāneʻohe Bay following the 2015 bleaching event showing that P. compressa recovered more quickly83,84 and a long-term experimental study showing that M. capitata suffered higher mortality, though these corals had been deprived of zooplankton26. Therefore, location and coral species composition will likely affect the outcomes of annual bleaching on reefs, globally.
Conclusions
Overall, we find that OA does not prolong coral holobiont recovery following thermal stress nor does it increase susceptibility to annual bleaching. Our results clearly support previous findings that increasing temperatures are more stressful to coral holobiont physiology than OA and suggest that while bleaching resistant phenotypes of M. capitata and P. compressa can be found in Kāneʻohe Bay and Waimānalo Bay16,17,78, M. capitata is more likely to persist on Hawaiian reefs than P. compressa under predicted future ocean conditions. We were also able to demonstrate the advantages of heterotrophy under OA conditions, even when heterotrophic plasticity is negatively impacted, particularly for enhancing calcification and CTAR in some cases, further supporting previous findings40. It is clear from our results, and those of previous studies e.g., ref. 15, that the conditions resulting in one species, or population of a species, performing physiologically better than another following thermal stress depends on very nuanced circumstances (e.g., local biotic stressor, environmental pre-conditioning), and that this is even more so the case under annual thermal stress. Therefore, we suggest that coral managers create recovery plans on a species-specific and site basis, and that land-based nurseries should provide corals with supplemental zooplankton during bleaching and recovery to promote survival. Finally, while substantial mortality occurs during such long-term, multiple stressor experiment, it behooves us to run such experiments even with these risks, as that it what the future holds for coral reefs. With the frequency and intensity of heat-stress events predicted to increase over the coming decades, understanding the long-term responses of corals to chronic and punctuated stressors is critical.
Methods
2014 and 2015 Hawaiian Archipelago thermal stress events
Thermal stress events in two consecutive years, 2014 and 2015, were the largest ever recorded for the Hawaiian Archipelago and resulted in mass coral bleaching throughout the region51,85. In 2014, the Coral Bleaching Alert for the Hawaiian Archipelago reached Alert Level 1 between 11–25 October, while in 2015 the Hawaiian Archipelago again reached Alert Level 1 on 4 September 2015, increased to Alert Level 2 on 19 September 2015, and returned to a Watch on 29 October 2015 (Fig. 1B86). These sustained increases above mean monthly maximum temperature resulted in 4.5- and 12.5-Degree Heating Weeks in 2014 and 2015, respectively87, with mass coral bleaching across the entire length of the Hawaiian Archipelago in both years9. At our collection sites, Degree Heating Weeks were 2.6 and 3.7 in Kāneʻohe Bay, 0.2 and 2.9 in Waimānalo Bay (Fig. 1A) in 2014 and 2015 respectively. Sea surface temperature (SST) at Moku o Loʻe reef in Kāneʻohe Bay was obtained from Station MOKH1, owned and maintained by the National Oceanographic and Atmospheric Administration’s (NOAA) National Ocean Service, while SST for Waimānalo Bay was obtained from Station 51202, owned and maintained by the Pacific Islands Ocean Observing System (PacIOOS).
Sample collection
Between 10–12 November 2014, three weeks following the peak of the 2014 thermal stress event (Fig. 1B), six visibly non-bleached and six visibly bleached parent colonies of M. capitata and P. compressa were collected [SAP 2015-48, SAP 2016-69] from 1–3 m depth from the fringing reefs surrounding Moku o Loʻe in Kāneʻohe Bay (centered at 21°26’6” N, 157°47’12” W) and in Waimānalo Bay (centered at 21°19’36” N, 157°40’54” W) using a hammer and chisel. In Kāneʻohe Bay, bleached and non-bleached parent colonies were paired (<2 m apart), whereas in Waimānalo Bay the colonies were more dispersed due to the less-even distribution of corals at this site. Selected parent colonies were tagged with numbers on waterproof paper attached with zip-ties to enable repeated sampling periodically throughout the experiment (i.e., in situ reef controls). Unfortunately, the tags in Waimānalo Bay went missing shortly after initial collection. Thus, we were only able to track Kāneʻohe Bay parent colonies for both species. One ramet from each colony was immediately placed in a −20 °C freezer to evaluate the initial physiology of the parent colonies prior to experimental manipulation. An additional twelve ramets were collected from each parent colony of each species from each location (totaling 576 fragments) and mounted on pre-labeled plastic tiles for use in the manipulative experiment. The designation of bleached or non-bleached refers to the visual status of the parent colony, determined qualitatively by expert opinion at the time of the ramet collections in November 2014. We maintain this nomenclature throughout the text, even when the ramets that were originally bleached regained pigmentation during recovery so that the original status on the reef in November 2014 is preserved.
Experimental design
All experimental ramets were returned to the Hawaiʻi Institute of Marine Biology (HIMB, Moku o Loʻe in Kāneʻohe Bay; Fig. 1A) and allowed to recover under ambient seawater conditions in outdoor, unfiltered, flow-through seawater tanks for three days. Natural light levels were reduced using 40% neutral density shade cloth such that photosynthetically active radiation levels mimicked that at collection depth as estimated from previous work e.g., ref. 88.
On 15 November 2014, ramets were equally divided among eight outdoor, 38 L experimental tanks supplied with unfiltered, flow-through seawater. Though the incoming seawater was unfiltered, virtually no zooplankton is delivered to experimental tanks at HIMB due to scouring of the inflowing seawater by in-line invertebrates26. Two experimental tanks were randomly assigned to each of the following four treatments: 1) Control (ambient pH and unfed), 2) Fed (ambient pH and fed), 3) OA (simulated OA and unfed), and 4) OA+Fed (simulated OA and fed) (Fig. 1C; Table 1). Three ramets from three bleached and three non-bleached colonies of each species (M. capitata and P. compressa) from each site (Kāneʻohe Bay and Waimānalo Bay) were placed within each tank (72 ramets per tank).
Target pH range for simulated OA was 0.2 pH units lower than ambient pH as this represents the most likely change in pH, relative to present day, given recent global commitments6. The seawater pH was reduced in the simulated OA treatments by bubbling CO2 gas into the tanks. To ensure tanks remained within target pH range, all tanks were spot-checked with a calibrated pH probe every 1–2 days throughout the experiment. This data was recorded during periods when physiological measurements were being made (Supplementary Fig. 1). Small adjustments to the CO2 bubbling rate were made as needed to maintain the desired 0.2 pH offset between ambient and simulated OA treatments. Weekly, pH and total alkalinity measurements were made on seawater samples collected from each tank at 18:00 h. Total alkalinity was measured using a modified Gran titration and checked for accuracy and precision (~5 µmol kg−1) using certified reference materials obtained from A. Dickson (Scripps Institution of Oceanography)89. pH was measured spectrophotometrically at 25 °C with m-cresol purple (precision ~0.002 pH units)89. These parameters were then entered into CO2Sys90 to calculate pH in situ (Fig. 1b). The CO2 bubbling rate was then adjusted as necessary, pH was measured again using a calibrated pH probe, and any further adjustments were made until the desired pH was consistently read. In December 2014, March 2015, and June 2015, diel cycle chemistry was measured over a 24-hour period across all tanks using the same method (Supplementary Fig. 2).
In addition, air was bubbled into all experimental tanks to provide intra-tank water flow speeds of ~5 cm s−1, to assist with oxygenation, and generate water motion within the tanks. Tank in-flow rate was adjusted to 1.5 ± 0.5 L minute−1 twice per week, and 100% tank turnover occurred approximately every 25 min. Temperature was not manipulated, and tanks temperatures reflected reef seawater temperatures throughout the experiment, including natural diurnal and seasonal temperature and the thermal stress event of 2015 (Fig. 1B). As the experimental tanks were outdoors, light levels also varied diurnally and seasonally. Tank temperature and salinity were measured twice per week with a YSI multi-meter at 18:00 h. Nutrient concentrations were not measured. However, the flow-through seawater source was the same for all tanks providing confidence that nutrient concentrations of the inflowing seawater was the same in all tanks.
Corals in the Fed and OA+Fed treatments were provided fresh-caught zooplankton for one hour twice a week. Zooplankton were caught by suspending a 50 µm mesh plankton net, fitted with a dive torch to attract zooplankton, at 1.5 m depth near the adjacent reef for 15 minutes, one hour after local sunset. After capture, zooplankton were re-suspended in 1 L seawater, a 40 ml subsample was collected and preserved in Formalin for plankton concentration determination, and the remainder was evenly divided among fed treatment tanks. This resulted in zooplankton densities of 0.47 ± 0.38 zooplankton ml−1 in the tanks (Supplementary Table 18). While this is much higher than natural zooplankton densities on the reef e.g., refs. 43,91, corals on the reef can feed for several hours per night every night whereas corals within the experiment could only feed for the single hour twice per week. Experimental tank seawater in-flow and CO2-dosing was suspended during the one hour of feeding to maximize zooplankton retention in the tanks, whereas air bubbling was continued to maintain water circulation.
Tanks and ramet tiles were cleaned twice weekly at which time ramet positions within each tank were adjusted randomly to minimize positional effects within the tanks. Ramets which were 100% dead were removed and discarded throughout the experiment (Supplementary Table 4). Prior to measurements conducted in June 2015, a parasitic nudibranch (Phestilla sp.) outbreak in the experimental tanks in March 2015 ultimately resulted in the mortality of 10% (28) non-bleached and 7% (19) bleached P. compressa ramets over the course of the experiment.
Coral physiology
Coral holobiont physiology was assessed for parent colonies (i.e., in situ reef controls) and a subset of experimental corals in December 2014 (2 months after 2014 peak thermal stress; 1 month in experimental tanks), June 2015 (8 months after 2014 peak thermal stress; 7 months in experimental tanks), and November 2015 (one month after 2015 peak thermal stress; 13 months in experimental tanks), according to methods described below. Each species-site combination initially had 36 ramets within each treatment (three from each of 12 parent colonies per site), with the plan to remove, measure, and sacrifice 192 ramets at each of the three time-points. Twelve ramets (each from a different parent colony) were removed from the experiment at each time-point for physiological trait measurements (n = 6 that were initially bleached and n = 6 that were non-bleached in November 2014). However, if the selected ramet had <50% live tissue, it was not possible to make accurate photosynthesis, respiration, and zooplankton feeding capacity measurements due to the interference of filamentous algae. At the first time point (December 2014), physiological trait measurements in the field were only conducted on five of the six sampled ramets (n = 5) due to time constraints. Additionally, due to the nature of the experiment, severe tissue loss and/or mortality occurred over time resulting in smaller sample sizes at the later time points.
Assessments of the parent colonies served to document coral in situ responses to both thermal stress events. Ramets were collected from the parent colonies of both species on the reef from Waimānalo Bay at the time of collection, and from Kāneʻohe Bay at all time points, and frozen at −20 °C prior to shipment on dry ice to The Ohio State University. On arrival, they were stored at −80 °C for endosymbiont cell density, tissue biomass, and total lipids analysis as described below.
Live physiological trait measurements (calcification, photosynthesis, respiration, zooplankton feeding rate capacity) were conducted on a subset of the experimental ramets over a 10-day period at each time point. In brief, calcification was measured using the buoyant weight technique92, photosynthesis and respiration rate were measured according to methods modified from Rodrigues and Grottoli48, and zooplankton feeding rate capacity was measured according to methods modified from Ferrier-Pages et al.93 and Dobson et al.41. Photosynthesis, day respiration, and night respiration rates were used to calculate the Contribution of Zooxanthellae (Symbiodiniaceae) to Animal Respiration (CZAR)94. Zooplankton feeding capacity was used to calculate Contribution of Heterotrophy to Animal Respiration (CHAR) from feeding on Artemia nauplii according to methods modified from Grottoli et al.15. To characterize the total carbon budget, CZAR and CHAR were summed to yield Contribution of Total fixed carbon to Animal Respiration (CTAR)15. To minimize the risk of overestimating CTAR, the calculated CTAR values for corals in Fed treatments are only representative of days on which they were fed. Additional details of the live measurements and subsequent calculations can be found in the Supplementary Methods.
Upon completion of the live physiological measurements, the same experimental ramets were frozen at −20 °C, transported on dry ice to The Ohio State University, and stored at −80 °C awaiting further analyses (endosymbiont cell density, tissue biomass, and total lipids). Live measurements were not conducted on any ramets in the subset which were partially dead (>50%), but these ramets were still frozen and transported for further analyses and the dead portions cut off with a wet-cutting circular saw. In some cases, there was not enough sample to complete all analyses (resulting in smaller samples size in some of the Figures). Surface area of each frozen coral ramet was measured using the aluminum foil method95. Each ramet was then individually homogenized using a mortar and pestle, and sub-sampled for endosymbiont cell density, tissue biomass, and total lipids. Endosymbiont cell density was measured according to methods modified from Rodrigues and Grottoli48 and standardized to surface area (cells cm−2). Tissue biomass was quantified according to methods in McLachlan et al.96. Total lipids were extracted according to methods in McLachlan et al.97, converted to Joules98, and normalized to tissue biomass (g g−1 ash free dry weight).
Statistical analyses
Kruskal–Wallice chi-squared tests were run to determine if pH values significantly differed between ambient and simulated OA tanks over the course of the experiment.
Differences in the physiological profiles (including endosymbiont cell density, CTAR, calcification, tissue biomass, and total lipids) between species, sites, and sites within species was assessed with a multivariate analysis of similarity (ANOSIM). Since all multivariate comparisons resulted in significant differences (Supplementary Table 3), downstream analyses were conducted separately on each species-site combination (see details below). To evaluate the effect of bleaching status on in situ parent colonies of both species from both sites in November 2014, univariate analyses of variance (ANOVA) were performed for each physiological trait. The same was done for Kāneʻohe Bay parent colonies of both species in December 2014 and June 2015, and just M. capitata in November 2015.
To test the first hypothesis, the physiological profiles of the experimental ramets in December 2014 and June 2015 were evaluated using both multivariate and univariate analyses. The responses of the bleached and non-bleached holobionts to OA and feeding was assessed on the physiological profile for each species-site combination at these time-points using ANOSIM. Next, three-way Type III univariate ANOVA were run (bleaching status, OA, feeding) and used in interpretation of the data.
Due to almost complete loss of all P. compressa ramets following the 2015 thermal stress event, the second hypothesis could only be evaluated for M. capitata in December 2015. Again, the responses of the bleached and non-bleached holobionts to OA and feeding was assessed on the physiological profile for each species-site combination using ANOSIM, and ANOVA were used to evaluate the effect of bleaching status, OA, and feeding on each physiological trait of each species-site combination.
All multivariate analyses were conducted using the software package Primer v699. All univariate parametric statistics were generated using SAS software, Version 9.3 of the SAS System for Windows. Additional details for all statistics performed are described in the Supplementary Methods.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
All metadata and data that support the results of this study are available at BCO-DMO under accession number https://doi.org/10.26008/1912/bco-dmo.914498.1.
References
Pörtner, H.-O. et al. IPCC, 2022: Summary for Policymakers. In Climate Change 2022: Impacts, Adaptation and Vulnerability. Contribution of Working Group II to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change (eds. Pörtner, H.-O. et al.) (Cambridge University Press, Cambridge, 2022). https://doi.org/10.1017/9781009325844.001.
Brown, B. E. Coral bleaching: Causes and consequences. Coral Reefs 16, S129–S138 (1997).
Orr, J. C. et al. Anthropogenic ocean acidification over the twenty-first century and its impact on calcifying organisms. Nature 437, 681–686 (2005).
Hughes, T. P. et al. Global warming and recurrent mass bleaching of corals. Nature 543, 373–377 (2017).
van der Zande, R. M. et al. Paradise lost: End-of-century warming and acidification under business-as-usual emissions have severe consequences for symbiotic corals. Glob. Chang Biol. 26, 2203–2219 (2020).
Rogelj, J. et al. Paris Agreement climate proposals need a boost to keep warming well below 2 °C. Nature 534, 631–639 (2016).
Coles, S. L., Jokiel, P. L. & Lewis, C. R. Thermal tolerance in tropical versus subtropical Pacific reef corals. Pac. Sci. 30, 159–166 (1976).
Hughes, T. P. et al. Spatial and temporal patterns of mass bleaching of corals in the Anthropocene. Science 359, 80–83 (2018).
Eakin, C. M., Sweatman, H. P. A. & Brainard, R. E. The 2014–2017 global-scale coral bleaching event: insights and impacts. Coral Reefs 38, 539–545 (2019).
Magel, J. M. T., Burns, J. H. R., Gates, R. D. & Baum, J. K. Effects of bleaching-associated mass coral mortality on reef structural complexity across a gradient of local disturbance. Sci. Rep. 9, 2512 (2019).
Souter, D. et al. Status of Coral Reefs of the World: 2020 Report, https://doi.org/10.59387/WOTJ9184 (2021).
Fitt, W. K., Brown, B. E., Warner, M. E. & Dunne, R. P. Coral bleaching: Interpretation of thermal tolerance limits and thermal thresholds in tropical corals. Coral Reefs 20, 51–65 (2001).
Teneva, L. et al. Predicting coral bleaching hotspots: The role of regional variability in thermal stress and potential adaptation rates. Coral Reefs 31, 1–12 (2012).
Jury, C. P. & Toonen, R. J. Adaptive responses and local stressor mitigation drive coral resilience in warmer, more acidic oceans. Proc. R. Soc. B Biol. Sci. 286, 20190614 (2019).
Grottoli, A. G. et al. The cumulative impact of annual coral bleaching can turn some coral species winners into losers. Glob. Chang Biol. 20, 3823–3833 (2014).
Cunning, R., Ritson-Williams, R. & Gates, R. D. Patterns of bleaching and recovery of Montipora capitata in Kāneʻohe Bay, Hawaiʻi, USA. Mar. Ecol. Prog. Ser. 551, 131–139 (2016).
Wall, C. B., Ritson-Williams, R., Popp, B. N. & Gates, R. D. Spatial variation in the biochemical and isotopic composition of corals during bleaching and recovery. Limnol. Oceanogr. 64, 2011–2028 (2019).
Kwiatkowski, L., Cox, P., Halloran, P. R., Mumby, P. J. & Wiltshire, A. J. Coral bleaching under unconventional scenarios of climate warming and ocean acidification. Nat. Clim. Chang 5, 777–781 (2015).
van Hooidonk, R., Maynard, J. A., Liu, Y. & Lee, S. K. Downscaled projections of Caribbean coral bleaching that can inform conservation planning. Glob. Chang Biol. 21, 3389–3401 (2015).
Cohen, A. L. & Holcomb, M. Why corals care about ocean acidification: Uncovering the mechanism. Oceanography 22, 118–127 (2009).
Marubini, F., Ferrier-Pages, C. & Cuif, J. P. Suppression of skeletal growth in scleractinian corals by decreasing ambient carbonate-ion concentration: A cross-family comparison. Proc. R. Soc. B Biol. Sci. 270, 179–184 (2003).
Jokiel, P. L. The reef coral two compartment proton flux model: A new approach relating tissue-level physiological processes to gross corallum morphology. J. Exp. Mar. Biol. Ecol. 409, 1–12 (2011).
Schoepf, V. et al. Coral energy reserves and calcification in a high-CO2 world at two temperatures. PLoS One 8, e108082 (2013).
Jury, C. P., Whitehead, R. F. & Szmant, A. M. Effects of variations in carbonate chemistry on the calcification rates of Madracis auretenra (= Madracis mirabilis sensu Wells, 1973): bicarbonate concentrations best predict calcification rates. Glob. Chang Biol. 16, 1632–1644 (2010).
Bahr, K. D., Jokiel, P. L. & Rodgers, K. S. Seasonal and annual calcification rates of the Hawaiian reef coral, Montipora capitata, under present and future climate change scenarios. ICES J. Mar. Sci. 74, 1083–1091 (2017).
McLachlan, R. H. et al. Physiological acclimatization in Hawaiian corals following a 22-month shift in baseline seawater temperature and pH. Sci. Rep. 12, 3712 (2022).
Leclercq, N., Gattuso, J. P. & Jaubert, J. CO2 partial pressure controls the calcification rate of a coral community. Glob. Chang Biol. 6, 329–334 (2000).
Hoegh-Guldberg, O. Low coral cover in a high-CO2 world. J. Geophys. Res. Oceans 110, C09S06 (2005).
Veron, J. E. N. et al. The coral reef crisis: The critical importance of <350 ppm CO2. Mar. Pollut. Bull. 58, 1428–1436 (2009).
Albright, R. et al. Carbon dioxide addition to coral reef waters suppresses net community calcification. Nature 555, 516–519 (2018).
Bahr, K. D., Jokiel, P. L. & Rodgers, K. S. Relative sensitivity of five Hawaiian coral species to high temperature under high-pCO2 conditions. Coral Reefs 35, 729–738 (2016).
Dobson, K. L. et al. Moderate nutrient concentrations are not detrimental to corals under future ocean conditions. Mar. Biol. 168, 98 (2021).
Anthony, K. R. N., Kline, D. I., Diaz-Pulido, G., Dove, S. & Hoegh-Guldberg, O. Ocean acidification causes bleaching and productivity loss in coral reef builders. Proc. Natl Acad. Sci. USA 105, 17442–17446 (2008).
Edmunds, P. J., Brown, D. & Moriarty, V. Interactive effects of ocean acidification and temperature on two scleractinian corals from Mo’orea, French Polynesia. Glob. Chang Biol. 18, 2173–2183 (2012).
Hoadley, K. D. et al. Physiological response to elevated temperature and pCO2 varies across four Pacific coral species: Understanding the unique host+symbiont response. Sci. Rep. 5, 18371 (2015).
Noonan, S. H. C. & Fabricius, K. E. Ocean acidification affects productivity but not the severity of thermal bleaching in some tropical corals. ICES J. Mar. Sci. 73, 715–726 (2016).
Davies, S. W., Ries, J. B., Marchetti, A. & Castillo, K. D. Symbiodinium functional diversity in the coral Siderastrea siderea is influenced by thermal stress and reef environment, but not ocean acidification. Front. Mar. Sci. 5, 150 (2018).
Houlbrèque, F. & Ferrier-Pagès, C. Heterotrophy in tropical scleractinian corals. Biol. Rev. 84, 1–17 (2009).
Edmunds, P. J. Zooplanktivory ameliorates the effects of ocean acidification on the reef coral Porites spp. Limnol. Oceanogr. 56, 2402–2410 (2011).
Towle, E. K., Enochs, I. C. & Langdon, C. Threatened Caribbean coral is able to mitigate the adverse effects of ocean acidification on calcification by increasing feeding rate. PLoS One 10, e0139398 (2015).
Dobson, K. L., Ferrier-Pagès, C., Saup, C. M. & Grottoli, A. G. The effects of temperature, light, and feeding on the physiology of Pocillopora damicornis, Stylophora pistillata, and Turbinaria reniformis corals. Water 13, 2048 (2021).
Grottoli, A. G., Rodrigues, L. J. & Palardy, J. E. Heterotrophic plasticity and resilience in bleached corals. Nature 440, 1186–1189 (2006).
Palardy, J. E., Rodrigues, L. J. & Grottoli, A. G. The importance of zooplankton to the daily metabolic carbon requirements of healthy and bleached corals at two depths. J. Exp. Mar. Biol. Ecol. 367, 180–188 (2008).
Levas, S. et al. Can heterotrophic uptake of dissolved organic carbon and zooplankton mitigate carbon budget deficits in annually bleached corals? Coral Reefs 35, 495–506 (2016).
Hughes, A. D. & Grottoli, A. G. Heterotrophic compensation: A possible mechanism for resilience of coral reefs to global warming or a sign of prolonged stress? PLoS One 8, e81172 (2013).
Hughes, A. D., Grottoli, A. G., Pease, T. K. & Matsui, Y. Acquisition and assimilation of carbon in non-bleached and bleached corals. Mar. Ecol. Prog. Ser. 420, 91–101 (2010).
Baumann, J., Grottoli, A. G., Hughes, A. D. & Matsui, Y. Photoautotrophic and heterotrophic carbon in bleached and non-bleached coral lipid acquisition and storage. J. Exp. Mar. Biol. Ecol. 461, 469–478 (2014).
Rodrigues, L. J. & Grottoli, A. G. Energy reserves and metabolism as indicators of coral recovery from bleaching. Limnol. Oceanogr. 52, 1874–1882 (2007).
Levas, S. J., Grottoli, A. G., Hughes, A., Osburn, C. L. & Matsui, Y. Physiological and biogeochemical traits of bleaching and recovery in the mounding species of coral Porites lobata: Implications for resilience in mounding corals. PLoS One 8, e63267 (2013).
Franklin, E. C., Jokiel, P. L. & Donahue, M. J. Predictive modeling of coral distribution and abundance in the Hawaiian Islands. Mar. Ecol. Prog. Ser. 481, 121–132 (2013).
Bahr, K. D., Rodgers, K. S. & Jokiel, P. L. Impact of three bleaching events on the reef resiliency of Kāneʻohe Bay, Hawaiʻi. Front Mar. Sci. 4, 398 (2017).
Ferrier-Pagès, C., Rottier, C., Beraud, E. & Levy, O. Experimental assessment of the feeding effort of three scleractinian coral species during a thermal stress: Effect on the rates of photosynthesis. J. Exp. Mar. Biol. Ecol. 390, 118–124 (2010).
Houlbréque, F. et al. Ocean acidification reduces feeding rates in the scleractinian coral Stylophora pistillata. Limnol. Oceanogr. 60, 89–99 (2015).
Allemand, D., Tambutté, É., Zoccola, D. & Tambutté, S. Coral calcification, cells to reefs. Coral Reefs: Ecosyst. Transit. https://doi.org/10.1007/978-94-007-0114-4_9 (2011).
Cohen, A. L. & Holcomb, M. Why corals care about ocean acidification uncovering the mechanism. Oceanography 22, 118–127 (2009).
Marubini, F. & Atkinson, M. J. Effects of lowered pH and elevated nitrate on coral calcification. Mar. Ecol. Prog. Ser. 188, 117–121 (1999).
Rodrigues, L. J. & Grottoli, A. G. Calcification rate and the stable carbon, oxygen, and nitrogen isotopes in the skeleton, host tissue, and zooxanthellae of bleached and recovering Hawaiian corals. Geochim Cosmochim. Acta 70, 2781–2789 (2006).
Holcomb, M., McCorkle, D. C. & Cohen, A. L. Long-term effects of nutrient and CO2 enrichment on the temperate coral Astrangia poculata (Ellis and Solander, 1786). J. Exp. Mar. Biol. Ecol. 386, 27–33 (2010).
Levas, S. et al. Organic carbon fluxes mediated by corals at elevated pCO2 and temperature. Mar. Ecol. Prog. Ser. 519, 153–164 (2015).
Borell, E. M. & Bischof, K. Feeding sustains photosynthetic quantum yield of a scleractinian coral during thermal stress. Oecologia 157, 593–601 (2008).
Tolosa, I., Treignier, C., Grover, R. & Ferrier-Pagès, C. Impact of feeding and short-term temperature stress on the content and isotopic signature of fatty acids, sterols, and alcohols in the scleractinian coral Turbinaria reniformis. Coral Reefs 30, 763–774 (2011).
Connolly, S. R., Lopez-Yglesias, M. A. & Anthony, K. R. N. Food availability promotes rapid recovery from thermal stress in a scleractinian coral. Coral Reefs 31, 951–960 (2012).
Tremblay, P., Gori, A., Maguer, J. F., Hoogenboom, M. & Ferrier-Pagès, C. Heterotrophy promotes the re-establishment of photosynthate translocation in a symbiotic coral after heat stress. Sci. Rep. 6, 38112 (2016).
Grottoli, A. G. & Rodrigues, L. J. Bleached Porites compressa and Montipora capitata corals catabolize δ13C-enriched lipids. Coral Reefs 30, 687–692 (2011).
Anthony, K. R. N., Hoogenboom, M. O., Maynard, J. A., Grottoli, A. G. & Middlebrook, R. Energetics approach to predicting mortality risk from environmental stress: A case study of coral bleaching. Funct. Ecol. 23, 539–550 (2009).
Grottoli, A. G., Tchernov, D. & Winters, G. Physiological and biogeochemical responses of super-corals to thermal stress from the northern gulf of Aqaba, Red Sea. Front Mar. Sci. 4, 215 (2017).
LaJeunesse, T. C. et al. High diversity and host specificity observed among symbiotic dinoflagellates in reef coral communities from Hawaiʻi. Coral Reefs 23, 596–603 (2004).
Magalon, H., Flot, J. F. & Baudry, E. Molecular identification of symbiotic dinoflagellates in Pacific corals in the genus Pocillopora. Coral Reefs 26, 551–558 (2007).
Pochon, X. et al. Comparison of endosymbiotic and free-living Symbiodinium (Dinophyceae) diversity in a Hawaiian reef environment. J. Phycol. 46, 53–65 (2010).
LaJeunesse, T. C. et al. Systematic revision of Symbiodiniaceae highlights the antiquity and diversity of coral endosymbionts. Curr. Biol. 28, 2570–2580 (2018).
De Souza, M. R. et al. Community composition of coral-associated Symbiodiniaceae differs across fine-scale environmental gradients in Kāne’ohe Bay. R. Soc. Open Sci. 9, 212042 (2022).
Ritson-Williams, R., Shjegstad, S. & Paul, V. Host specificity of four corallivorous Phestilla nudibranchs (Gastropoda: Opisthobranchia). Mar. Ecol. Prog. Ser. 255, 207–218 (2003).
Faucci, A., Toonen, R. J. & Hadfield, M. G. Host shift and speciation in a coral-feeding nudibranch. Proc. R. Soc. B Biol. Sci. 274, 20063685 (2007).
Gochfeld, D. J. & Aeby, G. S. Control of populations of the coral-feeding nudibranch Phestilla sibogae by fish and crustacean predators. Mar. Biol. 130, 63–69 (1997).
Gintert, B. E. et al. Marked annual coral bleaching resilience of an inshore patch reef in the Florida Keys: A nugget of hope, aberrance, or last man standing? Coral Reefs 37, 533–547 (2018).
Coles, S. L. et al. Evidence of acclimatization or adaptation in Hawaiian corals to higher ocean temperatures. PeerJ 6, e5347 (2018).
Bairos-Novak, K. R., Hoogenboom, M. O., van Oppen, M. J. H. & Connolly, S. R. Coral adaptation to climate change: Meta-analysis reveals high heritability across multiple traits. Glob. Chang Biol. 27, 5694–5710 (2021).
McLachlan, R. H. et al. Environmental gradients drive physiological variation in Hawaiian corals. Coral Reefs 40, 1505–1523 (2021).
Price, J. T. et al. Effect of species, provenance, and coral physiology on the composition of Hawaiian coral-associated microbial communities. Coral Reefs 40, 1537–1548 (2021).
Jury, C. P., Thomas, F. I. M., Atkinson, M. J. & Toonen, R. J. Buffer capacity, ecosystem feedbacks, and seawater chemistry under global change. Water 5, 1303–1325 (2013).
Schoepf, V., Jury, C. P., Toonen, R. J. & McCulloch, M. T. Coral calcification mechanisms facilitate adaptive responses to ocean acidification. Proc. R. Soc. B Biol. Sci. 284, 20172117 (2017).
Bahr, K. D., Jokiel, P. L. & Rodgers, K. S. The 2014 coral bleaching and freshwater flood events in Kāneʻohe Bay, Hawaiʻi. PeerJ 3, e1136 (2015).
Matsuda, S. B. et al. Coral bleaching susceptibility is predictive of subsequent mortality within but not between coral species. Front Ecol. Evol. 8, 178 (2020).
Ritson-Williams, R. & Gates, R. D. Coral community resilience to successive years of bleaching in Kāneʻohe Bay, Hawaiʻi. Coral Reefs 39, 757–769 (2020).
Couch, C. S. et al. Mass coral bleaching due to unprecedented marine heatwave in Papahānaumokuākea Marine National Monument (Northwestern Hawaiian Islands). PLoS One 12, e0185121 (2017).
NOAA Coral Reef Watch. Main Hawaiian Islands 5 km Regional Bleaching Heat Stress Maps and Gauges (v3.1), Aug 2014–Dec 2015. Updated daily, https://coralreefwatch.noaa.gov/product/vs/gauges/hawaii.php (2000).
NOAA Coral Reef Watch. Polynesia 5 km Regional Virtual Station Time Series Graphs (v3.1): Main Hawaiian Islands, 2014–2015. https://coralreefwatch.noaa.gov/product/vs/timeseries/polynesia.php#hawaii (2019).
Dunne, R. P. & Brown, B. E. Penetration of solar UVB radiation in shallow tropical waters and its potential biological effects on coral reefs; results from the Central Indian Ocean and Andaman Sea. Mar. Ecol. Prog. Ser. 144, 109–118 (1996).
Dickson, A. G., Sabine, C. L. & Christian, J. R. Guide to Best Practice for Ocean CO2 Measurements, vol. 3. (PICES Special Publication, 2007).
Lewis, E. R. & Wallace, D. W. R. Program Developed for CO2 System Calculation (Oak Ridge National Laboratory, United States Department of Energy, 1998).
Heidelberg, K. B., Sebens, K. P. & Purcell, J. E. Composition and sources of near reef zooplankton on a Jamaican forereef along with implications for coral feeding. Coral Reefs 23, 263–276 (2004).
Jokiel, P. L., Maragos, J. & Franzisket, L. Coral growth: buoyant weight technique. In Coral reefs: research methods (eds. Stoddart, D. R. & Johannes, R. E.) (UNESCO, 1978).
Ferrier-Pagès, C. et al. In situ assessment of the daily primary production of the temperate symbiotic coral Cladocora caespitosa. Limnol. Oceanogr. 58, 1409–1418 (2013).
Muscatine, L., McCloskey, L. R. & Marian, R. E. Estimating the daily contribution of carbon from zooxanthellae to coral animal respiration. Limnol. Oceanogr. 26, 601–611 (1981).
Marsh, J. A. Primary productivity of reef-building calcareous red algae. Ecology 51, 255–263 (1970).
McLachlan, R., Dobson, K. & Grottoli, A. G. Quantification of total biomass in ground coral samples (Protocols.io, 2020).
McLachlan, R. H., Muñoz-Garcia, A. & Grottoli, A. G. Extraction of total soluble lipid from ground coral samples (Protocols.io, 2020).
Gnaiger, E. & Bitterlich, G. Proximate biochemical composition and caloric content calculated from elemental CHN analysis: a stoichiometric concept. Oecologia 62, 289–298 (1984).
Clarke K. & Gorley R. Primer V6. (PRIMER-E Ltd. Plymouth Marine Laboratory UK, 2005).
Acknowledgements
A.G.G. and R.J.T. obtained primary funding for this research from the National Science Foundation (OCE#1514859) to A.G.G. (OCE#1838667 and 1459536). Additional support was provided to A.G.G. by the H.W.H. Hoover Foundation, to R.J.T. and C.P.J. by UH Sea Grant, and to K.L.D. by The Ohio State University’s School of Earth Sciences Friends of Orton Hall (FOH) fund and Sigma Xi–The Scientific Honor Society (National Chapter). We thank Paul Jokiel, Ku’ulei Rodgers, and all the staff at the Hawaiʻi Institute of Marine Biology for providing necessary tools, equipment, and workspace required for this experiment. We also thank Natasha Griswold, Claire Juraka, Annce Kadri, Casey McKenzie, Alec Moore, Margaret Otto, Jamie Price, Kristen Rockwell, Casey Saup, Katiri Snyder, and Andrea Wertz for field and laboratory analysis assistance. The study was conducted under Special Activity Permits from the State of Hawaiʻi Division of Aquatic Resources (SAP 2015-48, SAP 2016-69).
Author information
Authors and Affiliations
Contributions
A.G.G. and R.J.T. designed the study and secured the primary funding. K.L.D., A.G.G., R.H.M., and J.C.W. participated in fieldwork. C.P.J. maintained the experiment for its duration. K.L.D. and C.P.J. carried out lab analyses. K.L.D. drafted the manuscript; all authors contributed to revising the manuscript and gave final approval for publication. All authors have read and agreed to the published version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Earth & Environment thanks Erica Towle and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editors: Christopher Cornwall, Joe Aslin, Clare Davis and Heike Langenberg. A peer review file is available
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Dobson, K.L., Jury, C.P., Toonen, R.J. et al. Ocean acidification does not prolong recovery of coral holobionts from natural thermal stress in two consecutive years. Commun Earth Environ 5, 515 (2024). https://doi.org/10.1038/s43247-024-01672-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s43247-024-01672-5