Abstract
Aging involves the systemic deterioration of all known cell types in most eukaryotes. Several recently discovered compounds that extend the healthspan and lifespan of model organisms decelerate pathways that govern the aging process. Among these geroprotectors, spermidine, a natural polyamine ubiquitously found in organisms from all kingdoms, prolongs the lifespan of fungi, nematodes, insects and rodents. In mice, it also postpones the manifestation of various age-associated disorders such as cardiovascular disease and neurodegeneration. The specific features of spermidine, including its presence in common food items, make it an interesting candidate for translational aging research. Here, we review novel insights into the geroprotective mode of action of spermidine at the molecular level, as we discuss strategies for elucidating its clinical potential.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Fontana, L. The scientific basis of caloric restriction leading to longer life. Curr. Opin. Gastroenterol. 25, 144–150 (2009).
Lee, C. & Longo, V. Dietary restriction with and without caloric restriction for healthy aging. F1000Res 5, F1000 (2016).
Green, C. L., Lamming, D. W. & Fontana, L. Molecular mechanisms of dietary restriction promoting health and longevity. Nat. Rev. Mol. Cell. Biol. https://doi.org/10.1038/s41580-021-00411-4 (2021).
Longo, V. D., Di Tano, M., Mattson, M. P. & Guidi, N. Intermittent and periodic fasting, longevity and disease. Nat. Aging 1, 47–59 (2021).
Hofer, S. J., Carmona-Gutierrez, D., Mueller, M. I. & Madeo, F. The ups and downs of caloric restriction and fasting: from molecular effects to clinical application. EMBO Mol. Med. https://doi.org/10.15252/emmm.202114418 (2021).
Hofer, S. J., Davinelli, S., Bergmann, M., Scapagnini, G. & Madeo, F. Caloric restriction mimetics in nutrition and clinical trials. Front. Nutr. 8, 717343 (2021).
Ingram, D. K. & Roth, G. S. Glycolytic inhibition as a strategy for developing calorie restriction mimetics. Exp. Gerontol. 46, 148–154 (2011).
Ingram, D. K. et al. Calorie restriction mimetics: an emerging research field. Aging Cell 5, 97–108 (2006).
Madeo, F., Carmona-Gutierrez, D., Hofer, S. J. & Kroemer, G. Caloric restriction mimetics against age-associated disease: targets, mechanisms, and therapeutic potential. Cell Metab. 29, 592–610 (2019).
Madeo, F., Eisenberg, T., Pietrocola, F. & Kroemer, G. Spermidine in health and disease. Science 359, eaan2788 (2018).
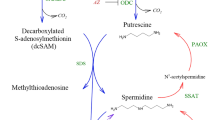
Pegg, A. E. Mammalian polyamine metabolism and function. IUBMB Life 61, 880–894 (2009).
Teixeira, D., Santaolaria, M. L., Meneu, V. & Alonso, E. Dietary arginine slightly and variably affects tissue polyamine levels in male swiss albino mice. J. Nutr. 132, 3715–3720 (2002).
López-Otín, C., Blasco, M. A., Partridge, L., Serrano, M. & Kroemer, G. The hallmarks of aging. Cell 153, 1194–1217 (2013).
Eisenberg, T. et al. Induction of autophagy by spermidine promotes longevity. Nat. Cell Biol. 11, 1305–1314 (2009).
Gupta, V. K. et al. Restoring polyamines protects from age-induced memory impairment in an autophagy-dependent manner. Nat. Neurosci. 16, 1453–1460 (2013).
Nishimura, K., Shiina, R., Kashiwagi, K. & Igarashi, K. Decrease in polyamines with aging and their ingestion from food and drink. J. Biochem. 139, 81–90 (2006).
Jänne, J., Raina, A. & Siimes, M. Spermidine and spermine in rat tissues at different ages. Acta Physiol. Scand. 62, 352–358 (1964).
Ferioli, M. E. & Comolli, R. Changes of liver and kidney polyamine levels during ageing. Exp. Gerontol. 10, 13–15 (1975).
Das, R. & Kanungo, M. S. Activity and modulation of ornithine decarboxylase and concentrations of polyamines in various tissues of rats as a function of age. Exp. Gerontol. 17, 95–103 (1982).
Ferioli, M. E., Sessa, A., Tunici, P., Pinotti, O. & Perin, A. Aging and polyamine acetylation in rat kidney. Biochim. Biophys. Acta 1317, 15–18 (1996).
Liu, P., Gupta, N., Jing, Y. & Zhang, H. Age-related changes in polyamines in memory-associated brain structures in rats. Neuroscience 155, 789–796 (2008).
Zwighaft, Z. et al. Circadian clock control by polyamine levels through a mechanism that declines with age. Cell Metab. 22, 874–885 (2015).
Pekar, T. et al. Spermidine in dementia: relation to age and memory performance. Wien. Klin. Wochenschr. 132, 42–46 (2020).
Pucciarelli, S. et al. Spermidine and spermine are enriched in whole blood of nona/centenarians. Rejuvenation Res. 15, 590–595 (2012).
Zhang, H. et al. Polyamines control eIF5A hypusination, TFEB translation and autophagy to reverse B cell senescence. Mol. Cell 76, 110–125 (2019).
Alsaleh, G. et al. Autophagy in T cells from aged donors is maintained by spermidine and correlates with function and vaccine responses. eLife 9, e57950 (2020).
Elworthy, P. & Hitchcock, E. Polyamine levels in red blood cells from patient groups of different sex and age. Biochim Biophys. Acta 993, 212–216 (1989).
Soda, K., Uemura, T., Sanayama, H., Igarashi, K. & Fukui, T. Polyamine-rich diet elevates blood spermine levels and inhibits pro-inflammatory status: an interventional study. Med. Sci. 9, 22 (2021).
Uemura, T., Akasaka, Y. & Ikegaya, H. Correlation of polyamines, acrolein-conjugated lysine and polyamine metabolic enzyme levels with age in human liver. Heliyon 6, e05031 (2020).
Morrison, L. D., Becker, L., Ang, L. C. & Kish, S. J. Polyamines in human brain: regional distribution and influence of aging. J. Neurochemistry 65, 636–642 (1995).
Igarashi, K. & Kashiwagi, K. Use of polyamine metabolites as markers for stroke and renal failure. in Polyamines (eds. A. E. Pegg & R. A. Casero) vol. 720, 395–408 (Humana Press, 2011).
Cheng, M.-L. et al. Metabolic disturbances identified in plasma are associated with outcomes in patients with heart failure: diagnostic and prognostic value of metabolomics. J. Am. Coll. Cardiol. 65, 1509–1520 (2015).
Pan, X. et al. Alzheimer’s disease-like pathology has transient effects on the brain and blood metabolome. Neurobiol. Aging 38, 151–163 (2016).
Graham, S. F. et al. Untargeted metabolomic analysis of human plasma indicates differentially affected polyamine and l-arginine metabolism in mild cognitive impairment subjects converting to Alzheimer’s disease. PLoS ONE 10, e0119452 (2015).
Schuller, A. P., Wu, C. C., Dever, T. E., Buskirk, A. R. & Green, R. eIF5A functions globally in translation elongation and termination. Mol. Cell 66, 194–205 (2017).
Lubas, M. et al. eIF5A is required for autophagy by mediating ATG3 translation. EMBO Rep. 19, e46072 (2018).
Frankel, L. B. EIF5A mediates autophagy via translation of ATG3. Autophagy 14, 1288–1289 (2018).
Liang, Y. et al. eIF5A hypusination, boosted by dietary spermidine, protects from premature brain aging and mitochondrial dysfunction. Cell Rep. 35, 108941 (2021).
Schroeder, S. et al. Dietary spermidine improves cognitive function. Cell Rep. 35, 108985 (2021).
Beyer, H. S., Ellefson, M., Sherman, R. & Zieve, L. Aging alters ornithine decarboxylase and decreases polyamines in regenerating rat liver but putrescine replacement has no effect. J. Lab. Clin. Med. 119, 38–47 (1992).
Wang, W. et al. Exercise training preserves ischemic preconditioning in aged rat hearts by restoring the myocardial polyamine pool. Oxid. Med. Cell. Longev. 2014, 457429 (2014).
Wang, J. et al. Spermidine alleviates cardiac aging by improving mitochondrial biogenesis and function. Aging 12, 650–671 (2020).
Yang, D., Oike, H., Furuse, M. & Yasuo, S. Spermidine resets circadian clock phase in NIH3T3 cells. Biomed. Res 42, 221–227 (2021).
Madeo, F. et al. Nutritional aspects of spermidine. Annu. Rev. Nutr. 40, 135–159 (2020).
Yin, Z., Pascual, C. & Klionsky, D. J. Autophagy: machinery and regulation. Micro. Cell 3, 588–596 (2016).
Klionsky, D. J. et al. Autophagy in major human diseases. EMBO J. 40, e108863 (2021).
Aman, Y. et al. Autophagy in healthy aging and disease. Nat. Aging 1, 634–650 (2021).
Pyo, J.-O. et al. Overexpression of Atg5 in mice activates autophagy and extends lifespan. Nat. Commun. 4, 2300 (2013).
Bjedov, I. et al. Fine-tuning autophagy maximises lifespan and is associated with changes in mitochondrial gene expression in Drosophila. PLoS Genet 16, e1009083 (2020).
Schinaman, J. M., Rana, A., Ja, W. W., Clark, R. I. & Walker, D. W. Rapamycin modulates tissue aging and lifespan independently of the gut microbiota in Drosophila. Sci. Rep. 9, 7824 (2019).
Bjedov, I. et al. Mechanisms of lifespan extension by rapamycin in the fruit fly Drosophila melanogaster. Cell Metab. 11, 35–46 (2010).
Yue, F. et al. Spermidine prolongs lifespan and prevents liver fibrosis and hepatocellular carcinoma by activating MAP1S-mediated autophagy. Cancer Res. 77, 2938–2951 (2017).
Hansen, M., Rubinsztein, D. C. & Walker, D. W. Autophagy as a promoter of longevity: insights from model organisms. Nat. Rev. Mol. Cell Biol. 19, 579–593 (2018).
Holbert, C. E. et al. Autophagy induction by exogenous polyamines is an artifact of bovine serum amine oxidase activity in culture serum. J. Biol. Chem. 295, 9061–9068 (2020).
Wang, L. et al. Oxidative degradation of polyamines by serum supplement causes cytotoxicity on cultured cells. Sci. Rep. 8, 10384 (2018).
Pietrocola, F. et al. Spermidine induces autophagy by inhibiting the acetyltransferase EP300. Cell Death Differ. 22, 509–516 (2015).
Morselli, E. et al. Spermidine and resveratrol induce autophagy by distinct pathways converging on the acetylproteome. J. Cell Biol. 192, 615–629 (2011).
Liu, X. et al. Spermidine inhibits vascular calcification in chronic kidney disease through modulation of SIRT1 signaling pathway. Aging Cell 20, e13377 (2021).
Liu, L., McKeehan, W. L., Wang, F. & Xie, R. MAP1S enhances autophagy to suppress tumorigenesis. Autophagy 8, 278–280 (2012).
Park, I.-H. & Kim, M.-M. Spermidine inhibits MMP-2 via modulation of histone acetyltransferase and histone deacetylase in HDFs. Int. J. Biol. Macromol. 51, 1003–1007 (2012).
Hai, Y., Shinsky, S. A., Porter, N. J. & Christianson, D. W. Histone deacetylase 10 structure and molecular function as a polyamine deacetylase. Nat. Commun. 8, 15368 (2017).
Jell, J. et al. Genetically altered expression of spermidine/spermine-N1-acetyltransferase affects fat metabolism in mice via Acetyl-CoA. J. Biol. Chem. 282, 8404–8413 (2007).
Kee, K. et al. Activated polyamine catabolism depletes acetyl-CoA pools and suppresses prostate tumor growth in TRAMP mice. J. Biol. Chem. 279, 40076–40083 (2004).
Pegg, A. E. Spermidine/spermine-N1-acetyltransferase: a key metabolic regulator. Am. J. Physiol. Endocrinol. Metab. 294, E995–1010 (2008).
Mariño, G. et al. Regulation of autophagy by cytosolic acetyl-coenzyme A. Mol. Cell 53, 710–725 (2014).
Puleston, D. J. et al. Polyamines and eIF5A hypusination modulate mitochondrial respiration and macrophage activation. Cell Metab. 30, 352–363 (2019).
Zhang, J. et al. Importance of TFEB acetylation in control of its transcriptional activity and lysosomal function in response to histone deacetylase inhibitors. Autophagy 14, 1043–1059 (2018).
Klier, H. et al. Isolation and structural characterization of different isoforms of the hypusine-containing protein eIF-5A from HeLa cells. Biochemistry 34, 14693–14702 (1995).
Tauc, M. et al. The eukaryotic initiation factor 5A (eIF5A1), the molecule, mechanisms and recent insights into the pathophysiological roles. Cell Biosci. 11, 219 (2021).
Ishfaq, M. et al. Acetylation regulates subcellular localization of eukaryotic translation initiation factor 5A (eIF5A). FEBS Lett. 586, 3236–3241 (2012).
Hofer, S. J. et al. Spermidine-induced hypusination preserves mitochondrial and cognitive function during aging. Autophagy 17, 2037–2039 (2021).
Padgett, L. R. et al. Deoxyhypusine synthase, an essential enzyme for hypusine biosynthesis, is required for proper exocrine pancreas development. FASEB J. 35, e21473 (2021).
Kar, R. K. et al. Neuron-specific ablation of eIF5A or deoxyhypusine synthase leads to impairments in growth, viability, neurodevelopment, and cognitive functions in mice. J. Biol. Chem. 297, 101333 (2021).
Barba-Aliaga, M. et al. Yeast translation elongation factor eIF5A expression is regulated by nutrient availability through different signalling pathways. Int. J. Mol. Sci. 22, 219 (2020).
Barba-Aliaga, M. & Alepuz, P. The activator/repressor Hap1 binds to the yeast eIF5A-encoding gene TIF51A to adapt its expression to the mitochondrial functional status. FEBS Lett. 596, 1809–1826 (2022).
Melis, N. et al. Targeting eIF5A hypusination prevents anoxic cell death through mitochondrial silencing and improves kidney transplant outcome. J. Am. Soc. Nephrol. 28, 811–822 (2017).
Cougnon, M. et al. Inhibition of eIF5A hypusination reprogrammes metabolism and glucose handling in mouse kidney. Cell Death Dis. 12, 283 (2021).
Karacay, C. et al. The effect of spermidine on autoimmunity and beta cell function in NOD mice. Sci. Rep. 12, 4502 (2022).
Carriche, G. M. et al. Regulating T cell differentiation through the polyamine spermidine. J. Allergy Clin. Immunol. 147, 335–348 (2021).
Sha, Y., Rao, L., Settembre, C., Ballabio, A. & Eissa, N. T. STUB1 regulates TFEB-induced autophagy–lysosome pathway. EMBO J. 36, 2544–2552 (2017).
Zhang, W., Li, X., Wang, S., Chen, Y. & Liu, H. Regulation of TFEB activity and its potential as a therapeutic target against kidney diseases. Cell Death Discov. 6, 32 (2020).
Zhao, E. & Czaja, M. J. TFEB: a central regulator of both the autophagosome and lysosome. Hepatol. 55, 1632–1634 (2012).
Liang, W. et al. A reciprocal regulation of spermidine and autophagy in podocytes maintains the filtration barrier. Kidney Int. 98, 1434–1448 (2020).
Dever, T. E. & Ivanov, I. P. Roles of polyamines in translation. J. Biol. Chem. 293, 18719–18729 (2018).
Igarashi, K. & Kashiwagi, K. Modulation of protein synthesis by polyamines. IUBMB Life 67, 160–169 (2015).
Mandal, S., Mandal, A., Johansson, H. E., Orjalo, A. V. & Park, M. H. Depletion of cellular polyamines, spermidine and spermine, causes a total arrest in translation and growth in mammalian cells. Proc. Natl Acad. Sci. USA 110, 2169–2174 (2013).
Stein, K. C., Morales-Polanco, F., van der Lienden, J., Rainbolt, T. K. & Frydman, J. Ageing exacerbates ribosome pausing to disrupt cotranslational proteostasis. Nature 601, 637–642 (2022).
Jing, Y.-H. et al. Spermidine ameliorates the neuronal aging by improving the mitochondrial function in vitro. Exp. Gerontol. 108, 77–86 (2018).
Yang, X. et al. Spermidine inhibits neurodegeneration and delays aging via the PINK1–PDR1-dependent mitophagy pathway in C. elegans. Aging 12, 16852–16866 (2020).
Eisenberg, T. et al. Cardioprotection and lifespan extension by the natural polyamine spermidine. Nat. Med. 22, 1428–1438 (2016).
Maglione, M. et al. Spermidine protects from age-related synaptic alterations at hippocampal mossy fiber-CA3 synapses. Sci. Rep. 9, 19616 (2019).
Chai, N. et al. Spermidine prevents heart injury in neonatal rats exposed to intrauterine hypoxia by inhibiting oxidative stress and mitochondrial fragmentation. Oxid. Med. Cell. Longev. 2019, 5406468 (2019).
Messerer, J. et al. Spermidine supplementation influences mitochondrial number and morphology in the heart of aged mice. J. Anat. https://doi.org/10.1111/joa.13618 (2021).
Sass, F. et al. TFEB deficiency attenuates mitochondrial degradation upon brown adipose tissue whitening at thermoneutrality. Mol. Metab. 47, 101173 (2021).
Mansueto, G. et al. Transcription factor EB controls metabolic flexibility during exercise. Cell Metab. 25, 182–196 (2017).
Wang, S. et al. Emerging role of transcription factor EB in mitochondrial quality control. Biomed. Pharmacother. 128, 110272 (2020).
Yan, J. et al. Spermidine-enhanced autophagic flux improves cardiac dysfunction following myocardial infarction by targeting the AMPK/mTOR signalling pathway. Br. J. Pharmacol. 176, 3126–3142 (2019).
Gomes, A. P., Schild, T. & Blenis, J. Adding polyamine metabolism to the mTORC1 toolkit in cell growth and cancer. Dev. Cell 42, 112–114 (2017).
Zabala-Letona, A. et al. mTORC1-dependent AMD1 regulation sustains polyamine metabolism in prostate cancer. Nature 547, 109–113 (2017).
Martina, J. A., Chen, Y., Gucek, M. & Puertollano, R. MTORC1 functions as a transcriptional regulator of autophagy by preventing nuclear transport of TFEB. Autophagy 8, 903–914 (2012).
Napolitano, G. et al. mTOR-dependent phosphorylation controls TFEB nuclear export. Nat. Commun. 9, 3312 (2018).
Liao, C.-Y. et al. The autophagy inducer spermidine protects against metabolic dysfunction during overnutrition. J. Gerontol. A Biol. Sci. Med Sci. 76, 1714–1725 (2021).
Yang, Y. et al. Induction of autophagy by spermidine is neuroprotective via inhibition of caspase 3-mediated Beclin 1 cleavage. Cell Death Dis. 8, e2738 (2017).
Li, G. et al. Spermidine suppresses Inflammatory DC function by activating the FOXO3 pathway and counteracts autoimmunity. iScience 23, 100807 (2020).
Freitag, K. et al. Spermidine reduces neuroinflammation and soluble amyloid beta in an Alzheimer’s disease mouse model. J. Neuroinflammation 19, 172 (2022).
Ding, Y. et al. SOX15 transcriptionally increases the function of AOC1 to modulate ferroptosis and progression in prostate cancer. Cell Death Dis. 13, 673 (2022).
Jeong, J.-W. et al. Spermidine protects against oxidative stress in inflammation models using macrophages and zebrafish. Biomol. Ther. 26, 146–156 (2018).
Liu, S. et al. Spermidine suppresses development of experimental abdominal aortic aneurysms. J. Am. Heart Assoc. 9, e014757 (2020).
Sutter, B. M., Wu, X., Laxman, S. & Tu, B. P. Methionine inhibits autophagy and promotes growth by inducing the SAM-responsive methylation of PP2A. Cell 154, 403–415 (2013).
Ouyang, Y., Wu, Q., Li, J., Sun, S. & Sun, S. S-adenosylmethionine: a metabolite critical to the regulation of autophagy. Cell Prolif. 53, e12891 (2020).
LaRocca, T. J., Gioscia-Ryan, R. A., Hearon, C. M. & Seals, D. R. The autophagy enhancer spermidine reverses arterial aging. Mech. Ageing Dev. 134, 314–320 (2013).
Knott, A. B. & Bossy-Wetzel, E. Impact of nitric oxide on metabolism in health and age-related disease. Diabetes Obes. Metab. 12, 126–133 (2010).
Valerio, A. & Nisoli, E. Nitric oxide, interorganelle communication, and energy flow: a novel route to slow aging. Front Cell Dev. Biol. 3, 6 (2015).
Ni, Y.-Q. & Liu, Y.-S. New insights into the roles and mechanisms of spermidine in aging and age-related diseases. Aging Dis. 12, 16 (2021).
Pillai, S. P. & Shankel, D. M. Polyamines and their potential to be antimutagens. Mutat. Res. 377, 217–224 (1997).
Lee, C. -Y. et al. Promotion of homology-directed DNA repair by polyamines. Nat. Commun. 10, 65 (2019).
Cooke, M. S., Evans, M. D., Dizdaroglu, M. & Lunec, J. Oxidative DNA damage: mechanisms, mutation, and disease. FASEB J. 17, 1195–1214 (2003).
Ha, H. C. et al. The natural polyamine spermine functions directly as a free radical scavenger. Proc. Natl Acad. Sci. USA 95, 11140–11145 (1998).
Khan, A. U., Mei, Y. H. & Wilson, T. A proposed function for spermine and spermidine: protection of replicating DNA against damage by singlet oxygen. Proc. Natl Acad. Sci. USA 89, 11426–11427 (1992).
Vujcic, S., Diegelman, P., Bacchi, C. J., Kramer, D. L. & Porter, C. W. Identification and characterization of a novel flavin-containing spermine oxidase of mammalian cell origin. Biochemical J. 367, 665–675 (2002).
Vujcic, S., Liang, P., Diegelman, P., Kramer, D. L. & Porter, C. W. Genomic identification and biochemical characterization of the mammalian polyamine oxidase involved in polyamine back-conversion. Biochemical J. 370, 19–28 (2003).
Wang, Y. et al. Cloning and characterization of a human polyamine oxidase that is inducible by polyamine analogue cxposure. Cancer Res. 61, 5370–5373 (2001).
Murray Stewart, T., Dunston, T. T., Woster, P. M. & Casero, R. A. Polyamine catabolism and oxidative damage. J. Biol. Chem. 293, 18736–18745 (2018).
Pegg, A. E. Functions of polyamines in mammals. J. Biol. Chem. 291, 14904–14912 (2016).
Wirth, A. et al. Novel aspects of age-protection by spermidine supplementation are associated with preserved telomere length. GeroScience 43, 673–690 (2021).
Scherz-Shouval, R. & Elazar, Z. Regulation of autophagy by ROS: physiology and pathology. Trends Biochemical Sci. 36, 30–38 (2011).
Bose, R. & Kanungo, M. S. Polyamines modulate phosphorylation and acetylation of non-histone chromosomal proteins of the cerebral cortex of rats of various ages. Arch. Gerontol. Geriatrics 1, 339–348 (1982).
Oehme, I. et al. Histone deacetylase 10 promotes autophagy-mediated cell survival. Proc. Natl Acad. Sci. USA 110, E2592–2601 (2013).
Burgio, G., Corona, D. F. V., Nicotra, C. M. A., Carruba, G. & Taibi, G. P/CAF-mediated spermidine acetylation regulates histone acetyltransferase activity. J. Enzyme Inhib. Med. Chem. 31, 75–82 (2016).
Hougaard, D. M., Nielsen, J. H. & Larsson, L. I. Localization and biosynthesis of polyamines in insulin-producing cells. Biochem J. 238, 43–47 (1986).
Welsh, N. & Sjöholm, A. Polyamines and insulin production in isolated mouse pancreatic islets. Biochem J. 252, 701–707 (1988).
Sjoholm, A. Role of polyamines in the regulation of proliferation and hormone production by insulin-secreting cells. Am. J. Physiol. 264, C501–C518 (1993).
Ma, L. et al. Spermidine improves gut barrier integrity and gut microbiota function in diet-induced obese mice. Gut Microbes 12, 1832857 (2020).
Wang, J. -Y. et al. Effect of spermidine on ameliorating spermatogenic disorders in diabetic mice via regulating glycolysis pathway. Reprod. Biol. Endocrinol. 20, 45 (2022).
Tain, L. S. et al. Longevity in response to lowered insulin signaling requires glycine N-methyltransferase-dependent spermidine production. Aging Cell 19, e13043 (2020).
Rajeeve, V., Pearce, W., Cascante, M., Vanhaesebroeck, B. & Cutillas, P. R. Polyamine production is downstream and upstream of oncogenic PI3K signalling and contributes to tumour cell growth. Biochemical J. 450, 619–628 (2013).
Chrisam, M. et al. Reactivation of autophagy by spermidine ameliorates the myopathic defects of collagen VI-null mice. Autophagy 11, 2142–2152 (2015).
Baek, A. R. et al. Spermidine attenuates bleomycin-induced lung fibrosis by inducing autophagy and inhibiting endoplasmic reticulum stress-induced cell death in mice. Exp. Mol. Med 52, 2034–2045 (2020).
Yuan, X. et al. Spermidine induces cytoprotective autophagy of female germline stem cells in vitro and ameliorates aging caused by oxidative stress through upregulated sequestosome-1/p62 expression. Cell Biosci. 11, 107 (2021).
Xu, T.-T. et al. Spermidine and spermine delay brain aging by inducing autophagy in SAMP8 mice. Aging 12, 6401–6414 (2020).
Green, D. R., Galluzzi, L. & Kroemer, G. Mitochondria and the autophagy-inflammation-cell death axis in organismal aging. Science 333, 1109–1112 (2011).
Qi, Y., Qiu, Q., Gu, X., Tian, Y. & Zhang, Y. ATM mediates spermidine-induced mitophagy via PINK1 and Parkin regulation in human fibroblasts. Sci. Rep. 6, 24700 (2016).
Han, X. et al. Spermidine regulates mitochondrial function by enhancing eIF5A hypusination and contributes to reactive oxygen species production and ganoderic acids biosynthesis in Ganoderma lucidum. Appl. Environ. Microbiol. https://doi.org/10.1128/AEM.02037-21 (2022).
Rayess, H., Wang, M. B. & Srivatsan, E. S. Cellular senescence and tumor suppressor gene p16. Int. J. Cancer 130, 1715–1725 (2012).
Che, H. et al. Rebalance of the polyamine metabolism suppresses oxidative stress and delays senescence in nucleus pulposus cells. Oxid. Med. Cell. Longev. 2022, 8033353 (2022).
García-Prat, L. et al. Autophagy maintains stemness by preventing senescence. Nature 529, 37–42 (2016).
Balnis, J. et al. Deaccelerated myogenesis and autophagy in genetically induced pulmonary emphysema. Am. J. Respir. Cell Mol. Biol. 66, 623–637 (2022).
Ramot, Y. et al. Polyamines and hair: a couple in search of perfection. Exp. Dermatol. 19, 784–790 (2010).
Ramot, Y. et al. Spermidine promotes human hair growth and is a novel modulator of human epithelial stem cell functions. PLoS ONE 6, e22564 (2011).
Zhang, D. et al. AMD1 is essential for ESC self-renewal and is translationally down-regulated on differentiation to neural precursor cells. Genes Dev. 26, 461–473 (2012).
Chen, T. et al. Rapamycin and other longevity-promoting compounds enhance the generation of mouse induced pluripotent stem cells. Aging Cell 10, 908–911 (2011).
Ma, L. et al. Spermidine ameliorates high-fat diet-induced hepatic steatosis and adipose tissue inflammation in preexisting obese mice. Life Sci. 265, 118739 (2021).
Gassen, N. C. et al. SARS-CoV-2-mediated dysregulation of metabolism and autophagy uncovers host-targeting antivirals. Nat. Commun. 12, 3818 (2021).
Wagner, A. et al. Metabolic modeling of single TH17 cells reveals regulators of autoimmunity. Cell 184, 4168–4185 (2021).
Pietrocola, F., Bravo-San Pedro, J. M., Galluzzi, L. & Kroemer, G. Autophagy in natural and therapy-driven anticancer immunosurveillance. Autophagy 13, 2163–2170 (2017).
Pietrocola, F. et al. Caloric restriction mimetics enhance anticancer immunosurveillance. Cancer Cell 30, 147–160 (2016).
Holbert, C. E., Cullen, M. T., Casero, R. A. & Stewart, T. M. Polyamines in cancer: integrating organismal metabolism and antitumour immunity. Nat. Rev. Cancer 22, 467–480 (2022).
Alexander, E. T., Minton, A., Peters, M. C., Phanstiel, O. & Gilmour, S. K. A novel polyamine blockade therapy activates an anti-tumor immune response. Oncotarget 8, 84140–84152 (2017).
Hayes, C. S. et al. Polyamine-blocking therapy reverses immunosuppression in the tumor microenvironment. Cancer Immunol. Res. 2, 274–285 (2014).
Fan, Y. & Pedersen, O. Gut microbiota in human metabolic health and disease. Nat. Rev. Microbiol. 19, 55–71 (2021).
Ramos-Molina, B., Queipo-Ortuño, M. I., Lambertos, A., Tinahones, F. J. & Peñafiel, R. Dietary and gut microbiota polyamines in obesity- and age-related diseases. Front. Nutr. 6, 24 (2019).
Wang, D. et al. Oral spermidine targets brown fat and skeletal muscle to mitigate diet-induced obesity and metabolic disorders. Mol. Nutr. Food Res. 65, 2100315 (2021).
Bui, T. I., Gill, A. L., Mooney, R. A. & Gill, S. R. Modulation of gut microbiota metabolism in obesity-related type 2 diabetes reduces osteomyelitis severity. Microbiol. Spectr. 10, e0017022 (2022).
Liu, S. et al. Effects of spermidine on gut microbiota modulation in experimental abdominal aortic aneurysm mice. Nutrients 14, 3349 (2022).
Reggiori, F. & Klionsky, D. J. Autophagic processes in yeast: mechanism, machinery and regulation. Genetics 194, 341–361 (2013).
Zimmermann, A. et al. Autophagy: one more Nobel Prize for yeast. Microb. Cell 3, 579–581 (2016).
Carmona-Gutierrez, D. et al. Guidelines and recommendations on yeast cell death nomenclature. Microb. Cell 5, 4–31 (2018).
Yamaguchi, O. & Otsu, K. Role of autophagy in aging. J. Cardiovasc Pharmacol. 60, 242–247 (2012).
Büttner, S. et al. Spermidine protects against α-synuclein neurotoxicity. Cell Cycle 13, 3903–3908 (2014).
Bito, T. et al. Involvement of spermidine in the reduced lifespan of Caenorhabditis elegans during vitamin B12 deficiency. Metabolites 9, 192 (2019).
Ao, Y. et al. Lamin A buffers CK2 kinase activity to modulate aging in a progeria mouse model. Sci. Adv. https://doi.org/10.1126/sciadv.aav5078 (2019).
Soda, K., Dobashi, Y., Kano, Y., Tsujinaka, S. & Konishi, F. Polyamine-rich food decreases age-associated pathology and mortality in aged mice. Exp. Gerontol. 44, 727–732 (2009).
Matsumoto, M., Kurihara, S., Kibe, R., Ashida, H. & Benno, Y. Longevity in mice is promoted by probiotic-induced suppression of colonic senescence dependent on upregulation of gut bacterial polyamine production. PLoS ONE 6, e23652 (2011).
Kibe, R. et al. Upregulation of colonic luminal polyamines produced by intestinal microbiota delays senescence in mice. Sci. Rep. 4, 4548 (2014).
Filfan, M. et al. Long-term treatment with spermidine increases healthspan of middle-aged Sprague-Dawley male rats. GeroScience 42, 937–949 (2020).
Fernández, Á. F. et al. Autophagy couteracts weight gain, lipotoxicity and pancreatic β-cell death upon hypercaloric pro-diabetic regimens. Cell Death Dis. 8, e2970 (2017).
Partridge, L., Fuentealba, M. & Kennedy, B. K. The quest to slow ageing through drug discovery. Nat. Rev. Drug Discov. 19, 513–532 (2020).
Janssens, G. E. & Houtkooper, R. H. Identification of longevity compounds with minimized probabilities of side effects. Biogerontology 21, 709–719 (2020).
Schwarz, C. et al. Safety and tolerability of spermidine supplementation in mice and older adults with subjective cognitive decline. Aging 10, 19–33 (2018).
Moskalev, A. et al. Developing criteria for evaluation of geroprotectors as a key stage toward translation to the clinic. Aging Cell 15, 407 (2016).
Binh, P. N. T., Soda, K., Maruyama, C. & Kawakami, M. Relationship between food polyamines and gross domestic product in association with longevity in Asian countries. Health 2, 1390–1396 (2010).
Kiechl, S. et al. Higher spermidine intake is linked to lower mortality: a prospective population-based study. Am. J. Clin. Nutr. 108, 371–380 (2018).
Soda, K., Kano, Y. & Chiba, F. Food polyamine and cardiovascular disease—an epidemiological study. Glob. J. Health Sci. 4, 170–178 (2012).
Zoumas-Morse, C. et al. Development of a polyamine database for assessing dietary intake. J. Am. Diet. Assoc. 107, 1024–1027 (2007).
Muñoz-Esparza, N. C. et al. Polyamines in Food. Front. Nutr. 6, 108 (2019).
Buyukuslu, N., Hizli, H., Esin, K. & Garipagaoglu, M. A cross-sectional study: nutritional polyamines in frequently consumed foods of the Turkish population. Foods 3, 541–557 (2014).
Binh, P. N. T., Soda, K. & Kawakami, M. Mediterranean diet and polyamine intake: possible contribution of increased polyamine intake to inhibition of age-associated disease. NDS 3, 1–7 (2010).
Bardócz, S. Polyamines in food and their consequences for food quality and human health. Trends Food Sci. Technol. 6, 341–346 (1995).
Ali, M. A., Poortvliet, E., Strömberg, R. & Yngve, A. Polyamines: total daily intake in adolescents compared to the intake estimated from the Swedish Nutrition Recommendations Objectified (SNO). Food Nutr. Res. 55 (2011).
Schwarz, C. et al. Spermidine intake is associated with cortical thickness and hippocampal volume in older adults. NeuroImage 221, 117132 (2020).
Atiya Ali, M., Poortvliet, E., Strömberg, R. & Yngve, A. Polyamines in foods: development of a food database. Food Nutr. Res. 55 (2011).
Wirth, M. et al. The effect of spermidine on memory performance in older adults at risk for dementia: a randomized controlled trial. Cortex 109, 181–188 (2018).
Wirth, M. et al. Effects of spermidine supplementation on cognition and biomarkers in older adults with subjective cognitive decline (SmartAge)-study protocol for a randomized controlled trial. Alzheimers Res Ther. 11, 36 (2019).
Schwarz, C. et al. Effects of spermidine supplementation on cognition and biomarkers in older adults with subjective cognitive decline: a randomized clinical trial. JAMA Netw. Open 5, e2213875 (2022).
Witkowska, A. M. Soluble ICAM-1: a marker of vascular inflammation and lifestyle. Cytokine 31, 127–134 (2005).
Pekar, T. et al. The positive effect of spermidine in older adults suffering from dementia: first results of a 3-month trial. Wien. Klin. Wochenschr. 133, 484–491 (2021).
Huang, J. et al. Spermidine exhibits protective effects against traumatic brain injury. Cell Mol. Neurobiol. 40, 927–937 (2020).
Guerra, G. P., Rubin, M. A. & Mello, C. F. Modulation of learning and memory by natural polyamines. Pharm. Res. 112, 99–118 (2016).
Matsumoto, M. & Benno, Y. Consumption of Bifidobacterium lactis LKM512 yogurt reduces gut mutagenicity by increasing gut polyamine contents in healthy adult subjects. Mutat. Res. 568, 147–153 (2004).
Matsumoto, M., Kitada, Y. & Naito, Y. Endothelial function is improved by inducing microbial polyamine production in the gut: a randomized placebo-controlled trial. Nutrients 11, 1188 (2019).
Rinaldi, F., Marzani, B., Pinto, D. & Ramot, Y. A spermidine-based nutritional supplement prolongs the anagen phase of hair follicles in humans: a randomized, placebo-controlled, double-blind study. Dermatol. Pract. Concept. 7, 17–21 (2017).
Chai, M. et al. Stimulation of hair growth by small molecules that activate autophagy. Cell Rep. 27, 3413–3421 (2019).
Vargas, A. J. et al. Dietary polyamine intake and risk of colorectal adenomatous polyps. Am. J. Clin. Nutr. 96, 133–141 (2012).
Vargas, A. J. et al. Dietary polyamine intake and colorectal cancer risk in postmenopausal women. Am. J. Clin. Nutr. 102, 411–419 (2015).
Pietrocola, F. et al. Spermidine reduces cancer-related mortality in humans. Autophagy 15, 362–365 (2019).
Gobert, A. P. et al. Protective role of spermidine in colitis and colon carcinogenesis. Gastroenterology 162, 813–827 (2022).
Chin, A., Bieberich, C. J., Stewart, T. M. & Casero, R. A. Polyamine depletion strategies in cancer: remodeling the tumor immune microenvironment to enhance anti-tumor responses. Med. Sci. 10, 31 (2022).
Faundes, V. et al. Impaired eIF5A function causes a Mendelian disorder that is partially rescued in model systems by spermidine. Nat. Commun. 12, 833 (2021).
Murray-Stewart, T., Dunworth, M., Foley, J. R., Schwartz, C. E. & Casero, R. A. Polyamine homeostasis in Snyder-Robinson syndrome. Med. Sci. 6, 112 (2018).
Larcher, L. et al. The complete loss of function of the SMS gene results in a severe form of Snyder-Robinson syndrome. Eur. J. Med. Genet. 63, 103777 (2020).
Lauren Cason, A. et al. X-linked spermine synthase gene (SMS) defect: the first polyamine deficiency syndrome. Eur. J. Hum. Genet 11, 937–944 (2003).
Li, C. et al. Spermine synthase deficiency causes lysosomal dysfunction and oxidative stress in models of Snyder-Robinson syndrome. Nat. Commun. 8, 1257 (2017).
Murray Stewart, T. et al. (R,R)-1,12-Dimethylspermine can mitigate abnormal spermidine accumulation in Snyder-Robinson syndrome. J. Biol. Chem. 295, 3247–3256 (2020).
Kapur, M. & Ackerman, S. L. mRNA translation gone awry: translation fidelity and neurological disease. Trends Genet. 34, 218–231 (2018).
Schultz, C. R., Bupp, C. P., Rajasekaran, S. & Bachmann, A. S. Biochemical features of primary cells from a pediatric patient with a gain-of-function ODC1 genetic mutation. Biochemical J. 476, 2047–2057 (2019).
VanSickle, E. A. et al. Expanding the phenotype: four new cases and hope for treatment in Bachmann-Bupp syndrome. Am. J. Med. Genet. A 185, 3485–3493 (2021).
Bupp, C. P., Schultz, C. R., Uhl, K. L., Rajasekaran, S. & Bachmann, A. S. Novel de novo pathogenic variant in the ODC1 gene in a girl with developmental delay, alopecia, and dysmorphic features. Am. J. Med. Genet. A 176, 2548–2553 (2018).
Rodan, L. H. et al. Gain-of-function variants in the ODC1 gene cause a syndromic neurodevelopmental disorder associated with macrocephaly, alopecia, dysmorphic features, and neuroimaging abnormalities. Am. J. Med. Genet. A 176, 2554–2560 (2018).
Rajasekaran, S. et al. Repurposing eflornithine to treat a patient with a rare ODC1 gain-of-function variant disease. Elife 10, e67097 (2021).
Mayeux, R. Biomarkers: potential uses and limitations. NeuroRx 1, 182–188 (2004).
Strimbu, K. & Tavel, J. A. What are biomarkers? Curr. Opin. HIV AIDS 5, 463–466 (2010).
Amin, M. et al. Polyamine biomarkers as indicators of human disease. Biomarkers 26, 77–94 (2021).
Maksymiuk, A. W. et al. Spermidine/spermine-N1-acetyltransferase-1 as a diagnostic biomarker in human cancer. Future Sci. OA 4, FSO345 (2018).
Morrison, L. D. & Kish, S. J. Brain polyamine levels are altered in Alzheimer’s disease. Neurosci. Lett. 197, 5–8 (1995).
Saiki, S. et al. A metabolic profile of polyamines in Parkinson disease: a promising biomarker. Ann. Neurol. 86, 251–263 (2019).
Sternberg, Z. et al. Elevated spermidine serum levels in mild cognitive impairment, a potential biomarker of progression to Alzheimer dementia, a pilot study. J. Clin. Neurosci. 100, 169–174 (2022).
Xu, J. et al. Non-linear association between serum spermidine and mild cognitive impairment: results from a cross-sectional and longitudinal study. Front. Aging Neurosci. 14, 924984 (2022).
Polis, B., Karasik, D. & Samson, A. O. Alzheimer’s disease as a chronic maladaptive polyamine stress response. Aging 13, 10770–10795 (2021).
Zheng, L. et al. Serum spermidine in relation to risk of stroke: a multilevel study. Front Nutr. 9, 843616 (2022).
Yu, Z. et al. Effect of serum spermidine on the prognosis in patients with acute myocardial infarction: a cohort study. Nutrients 14, 1394 (2022).
Omar, E. M., Omar, R. S., Shoela, M. S. & El Sayed, N. S. A study of the cardioprotective effect of spermidine: a novel inducer of autophagy. Chin. J. Physiol. 64, 281–288 (2021).
Magnes, C. et al. Polyamines in biological samples: rapid and robust quantification by solid-phase extraction online-coupled to liquid chromatography–tandem mass spectrometry. J. Chromatogr. A 1331, 44–51 (2014).
Srivastava, V. et al. Distinct designer diamines promote mitophagy, and thereby enhance healthspan in C. elegans and protect human cells against oxidative damage. Autophagy 0, 1–31 (2022).
Acknowledgements
All figures were initially created with BioRender.com. S.J.H. acknowledges support from a Longevity Impetus Grant from Norn Group. F.M. is grateful to the Austrian Science Fund FWF (F3012, W1226, P29203, P29262, P27893, P31727) and the Austrian Federal Ministry of Education, Science and Research as well as the University of Graz for grants ‘Unkonventionelle Forschung-InterFast and Fast4Health’ and ‘flysleep’ (BMWFW-80.109/0001-WF/V/3b/2015). T.E. acknowledges support from Austrian Science Fund FWF (P 33957 and TAI 602 1000). We acknowledge the support of the Field of Excellence BioHealth of NAWI Graz and the BioTechMed-Graz flagship project ‘EPIAge’. G.K. is supported by the Ligue contre le Cancer (équipe labellisée); Agence National de la Recherche (ANR)–Projets blancs; AMMICa US23/CNRS UMS3655; Association pour la recherche sur le cancer (ARC); Cancéropôle Ile-de-France; Fondation pour la Recherche Médicale (FRM); a donation by Elior; Equipex Onco-Pheno-Screen; European Joint Programme on Rare Diseases (EJPRD); Gustave Roussy Odyssea, the European Union Horizon 2020 Projects Oncobiome and Crimson; Fondation Carrefour; Institut National du Cancer (INCa); Institut Universitaire de France; LabEx Immuno-Oncology (ANR-18-IDEX-0001); a Cancer Research ASPIRE Award from the Mark Foundation; the RHU Immunolife; Seerave Foundation; SIRIC Stratified Oncology Cell DNA Repair and Tumor Immune Elimination (SOCRATE); and SIRIC Cancer Research and Personalized Medicine (CARPEM). This study contributes to the IdEx Université de Paris ANR-18-IDEX-0001.
Author information
Authors and Affiliations
Contributions
S.J.H. wrote the first draft. S.J.H., G.K. and F.M. conceptualized the review. M.B. and S.J.H. collected information for the tables. All authors proofread, edited and contributed substantially to the final version.
Corresponding authors
Ethics declarations
Competing interests
F.M. and G.K. are scientific cofounders of Samsara Therapeutics, a company that develops new pharmacological autophagy inducers. F.M. and T.E. have equity interests in and are advisors of The Longevity Labs (TLL). A.K.S. is consulting for TLL and Oxford Healthspan. G.K. is a scientific cofounder of everImmune, Osasuna Therapeutics and Therafast Bio. G.K. is holding research contracts with Daiichi Sankyo, Eleor, Osasuna Therapeutics, Samsara Therapeutics, Sanofi, Tollys and Vascage. S.J.H. and M.B. declare no competing interests.
Peer review
Peer review information
Nature Aging thanks Luigi Fontana, Tracy Murray-Stewart and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hofer, S.J., Simon, A.K., Bergmann, M. et al. Mechanisms of spermidine-induced autophagy and geroprotection. Nat Aging 2, 1112–1129 (2022). https://doi.org/10.1038/s43587-022-00322-9
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s43587-022-00322-9
This article is cited by
-
SS-31 improves the quality of maternally aged oocytes by ameliorating mitochondrial function and metabolism
Journal of Ovarian Research (2026)
-
The role of spermidine in plants and humans: a pathway from climate change adaptation to health benefits
npj Science of Food (2026)
-
Potential dietary geroprotectors and their impact on key mechanisms of aging
Biogerontology (2026)
-
The role of mitochondrial dysfunction in the pathogenesis of Alzheimer’s disease and future strategies for targeted therapy
European Journal of Medical Research (2025)
-
Analysis of spermidine’s effect on coronary heart disease risk using bidirectional Mendelian randomization and LC-MS/MS
Hereditas (2025)