Abstract
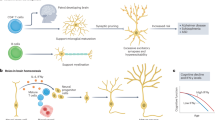
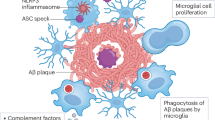
The cautious optimism following recent anti-amyloid therapeutic trials for Alzheimer’s disease (AD) provides a glimmer of hope after years of disappointment. Although these encouraging results represent discernible progress, they also highlight the need to enhance further the still modest clinical efficacy of current disease-modifying immunotherapies. Here, we highlight crucial milestones essential for advancing precision medicine in AD. These include reevaluating the choice of therapeutic targets by considering the key role of both central neuroinflammation and peripheral immunity in disease pathogenesis, refining patient stratification by further defining the inflammatory component within the forthcoming ATN(I) (amyloid, tau and neurodegeneration (and inflammation)) classification of AD biomarkers and defining more accurate clinical outcomes and prognostic biomarkers that better reflect disease heterogeneity. Next-generation immunotherapies will need to go beyond the current antibody-only approach by simultaneously targeting pathological proteins together with innate neuroinflammation and/or peripheral–central immune crosstalk. Such innovative immunomodulatory combination therapy approaches should be evaluated in appropriately redesigned clinical therapeutic trials, which must carefully integrate the neuroimmune component.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Salloway, S. et al. Two phase 3 trials of bapineuzumab in mild-to-moderate Alzheimer’s disease. N. Engl. J. Med. 370, 322–333 (2014).
Doody, R. S. et al. Phase 3 trials of solanezumab for mild-to-moderate Alzheimer’s disease. N. Engl. J. Med. 370, 311–321 (2014).
Honig, L. S. et al. Trial of solanezumab for mild dementia due to Alzheimer’s disease. N. Engl. J. Med. 378, 321–330 (2018).
Ostrowitzki, S. et al. A phase III randomized trial of gantenerumab in prodromal Alzheimer’s disease. Alzheimers Res. Ther. 9, 95 (2017).
Egan, M. F. et al. Randomized trial of verubecestat for prodromal Alzheimer’s disease. N. Engl. J. Med. 380, 1408–1420 (2019).
Henley, D. et al. Preliminary results of a trial of atabecestat in preclinical Alzheimer’s disease. N. Engl. J. Med. 380, 1483–1485 (2019).
Siemers, E. R. et al. Phase 3 solanezumab trials: secondary outcomes in mild Alzheimer’s disease patients. Alzheimers Dement. 12, 110–120 (2016).
Fillit, H. & Green, A. Aducanumab and the FDA—where are we now? Nat. Rev. Neurol. 17, 129–130 (2021).
van Dyck, C. H. et al. Lecanemab in early Alzheimer’s disease. N. Engl. J. Med. 388, 9–21 (2023).
Sims, J. R. et al. Donanemab in early symptomatic Alzheimer disease: the TRAILBLAZER-ALZ 2 randomized clinical trial. JAMA 330, 512–527 (2023).
Jun, H., Cho, S. K., Yoong, J. & Mattke, S. Is Korea prepared for an Alzheimer’s disease-modifying therapy? Assessing the Korean healthcare system infrastructure and the effect of blood-based biomarker tests. Ann. Geriatr. Med. Res. 25, 33–38 (2021).
Mattke, S. et al. Preparedness of the Brazilian health-care system to provide access to a disease-modifying Alzheimer’s disease treatment. Alzheimers Dement. 19, 375–381 (2023).
Leng, F. & Edison, P. Neuroinflammation and microglial activation in Alzheimer disease: where do we go from here? Nat. Rev. Neurol. 17, 157–172 (2021).
Bettcher, B. M., Tansey, M. G., Dorothée, G. & Heneka, M. T. Peripheral and central immune system crosstalk in Alzheimer disease—a research prospectus. Nat. Rev. Neurol. 17, 689–701 (2021).
Imbimbo, B. P., Watling, M., Imbimbo, C. & Nisticò, R. Plasma ATN(I) classification and precision pharmacology in Alzheimer’s disease. Alzheimers Dement. 19, 4729–4734 (2023).
Sarazin, M., Dorothée, G., de Souza, L. C. & Aucouturier, P. Immunotherapy in Alzheimer’s disease: do we have all the pieces of the puzzle? Biol. Psychiatry 74, 329–332 (2013).
Holstege, H. et al. Exome sequencing identifies rare damaging variants in ATP8B4 and ABCA1 as risk factors for Alzheimer’s disease. Nat. Genet. 54, 1786–1794 (2022).
Bellenguez, C. et al. New insights into the genetic etiology of Alzheimer’s disease and related dementias. Nat. Genet. 54, 412–436 (2022).
Romero-Molina, C., Garretti, F., Andrews, S. J., Marcora, E. & Goate, A. M. Microglial efferocytosis: diving into the Alzheimer’s disease gene pool. Neuron 110, 3513–3533 (2022).
Hamelin, L. et al. Early and protective microglial activation in Alzheimer’s disease: a prospective study using 18F-DPA-714 PET imaging. Brain 139, 1252–1264 (2016).
Hamelin, L. et al. Distinct dynamic profiles of microglial activation are associated with progression of Alzheimer’s disease. Brain 141, 1855–1870 (2018).
Kunkle, B. W. et al. Genetic meta-analysis of diagnosed Alzheimer’s disease identifies new risk loci and implicates Aβ, tau, immunity and lipid processing. Nat. Genet. 51, 414–430 (2019).
Wightman, D. P. et al. A genome-wide association study with 1,126,563 individuals identifies new risk loci for Alzheimer’s disease. Nat. Genet. 53, 1276–1282 (2021).
Paolicelli, R. C. et al. Microglia states and nomenclature: a field at its crossroads. Neuron 110, 3458–3483 (2022).
Escartin, C. et al. Reactive astrocyte nomenclature, definitions, and future directions. Nat. Neurosci. 24, 312–325 (2021).
Ulland, T. K. & Colonna, M. TREM2—a key player in microglial biology and Alzheimer disease. Nat. Rev. Neurol. 14, 667–675 (2018).
Schlepckow, K., Morenas-Rodríguez, E., Hong, S. & Haass, C. Stimulation of TREM2 with agonistic antibodies—an emerging therapeutic option for Alzheimer’s disease. Lancet Neurol. 22, 1048–1060 (2023).
Chen, X. & Holtzman, D. M. Emerging roles of innate and adaptive immunity in Alzheimer’s disease. Immunity 55, 2236–2254 (2022).
Hartnell, I. J., Blum, D., Nicoll, J. A. R., Dorothee, G. & Boche, D. Glial cells and adaptive immunity in frontotemporal dementia with tau pathology. Brain 144, 724–745 (2021).
Laurent, C. et al. Hippocampal T cell infiltration promotes neuroinflammation and cognitive decline in a mouse model of tauopathy. Brain 140, 184–200 (2017).
Chen, X. et al. Microglia-mediated T cell infiltration drives neurodegeneration in tauopathy. Nature 615, 668–677 (2023).
Hartnell, I. J. et al. Glial reactivity and T cell infiltration in frontotemporal lobar degeneration with tau pathology. Brain https://doi.org/10.1093/brain/awad309 (2024).
Dansokho, C. et al. Regulatory T cells delay disease progression in Alzheimer-like pathology. Brain 139, 1237–1251 (2016).
Alves, S. et al. Interleukin-2 improves amyloid pathology, synaptic failure and memory in Alzheimer’s disease mice. Brain 140, 826–842 (2017).
Yuan, L. et al. Low-dose IL-2 treatment rescues cognitive deficits by repairing the imbalance between Treg and Th17 cells at the middle Alzheimer’s disease stage. J. Neuroimmune Pharmacol. https://doi.org/10.1007/s11481-023-10090-x (2023).
Stym-Popper, G. et al. Regulatory T cells decrease C3-positive reactive astrocytes in Alzheimer-like pathology. J. Neuroinflammation 20, 64 (2023).
Baruch, K. et al. Breaking immune tolerance by targeting Foxp3+ regulatory T cells mitigates Alzheimer’s disease pathology. Nat. Commun. 6, 7967 (2015).
Da Mesquita, S. et al. Aging-associated deficit in CCR7 is linked to worsened glymphatic function, cognition, neuroinflammation, and β-amyloid pathology. Sci. Adv. 7, eabe4601 (2021).
Elyahu, Y. et al. Aging promotes reorganization of the CD4 T cell landscape toward extreme regulatory and effector phenotypes. Sci. Adv. 5, eaaw8330 (2019).
Piehl, N. et al. Cerebrospinal fluid immune dysregulation during healthy brain aging and cognitive impairment. Cell 185, 5028–5039 (2022).
Yuan, Y. et al. Therapeutic potential of interleukin-2 in autoimmune diseases. Trends Mol. Med. 28, 596–612 (2022).
Zhang, H. et al. In vivo expansion of regulatory T cells with IL-2/IL-2 antibody complex protects against transient ischemic stroke. J. Neurosci. 38, 10168–10179 (2018).
Webster, K. E. et al. In vivo expansion of T reg cells with IL-2–mAb complexes: induction of resistance to EAE and long-term acceptance of islet allografts without immunosuppression. J. Exp. Med. 206, 751–760 (2009).
Gao, W. et al. IL-2/anti-IL-2 complex attenuates inflammation and BBB disruption in mice subjected to traumatic brain injury. Front. Neurol. 8, 281 (2017).
Yshii, L. et al. Astrocyte-targeted gene delivery of interleukin 2 specifically increases brain-resident regulatory T cell numbers and protects against pathological neuroinflammation. Nat. Immunol. 23, 878–891 (2022).
Lopes, J. R. et al. Nasal administration of anti-CD3 monoclonal antibody ameliorates disease in a mouse model of Alzheimer’s disease. Proc. Natl Acad. Sci. USA 120, e2309221120 (2023).
Faridar, A. et al. A phase 1 open-label pilot study of low-dose interleukine-2 immunotherapy in patients with Alzheimer’s disease. Transl. Neurodegener. 12, 54 (2023).
Baek, H. et al. Neuroprotective effects of CD4+CD25+Foxp3+ regulatory T cells in a 3xTg-AD Alzheimer’s disease model. Oncotarget 7, 69347–69357 (2016).
Faridar, A. et al. Restoring regulatory T-cell dysfunction in Alzheimer’s disease through ex vivo expansion. Brain Commun. 2, fcaa112 (2020).
Yang, H. et al. Adoptive therapy with amyloid-β specific regulatory T cells alleviates Alzheimer’s disease. Theranostics 12, 7668–7680 (2022).
Yeapuri, P. et al. Amyloid-β specific regulatory T cells attenuate Alzheimer’s disease pathobiology in APP/PS1 mice. Mol. Neurodegener. 18, 97 (2023).
Saetzler, V. et al. Development of β-amyloid-specific CAR-Tregs for the treatment of Alzheimer’s disease. Cells 12, 2115 (2023).
Baruch, K. et al. PD-1 immune checkpoint blockade reduces pathology and improves memory in mouse models of Alzheimer’s disease. Nat. Med. 22, 135–137 (2016).
Rosenzweig, N. et al. PD-1/PD-L1 checkpoint blockade harnesses monocyte-derived macrophages to combat cognitive impairment in a tauopathy mouse model. Nat. Commun. 10, 465 (2019).
Latta-Mahieu, M. et al. Systemic immune-checkpoint blockade with anti-PD1 antibodies does not alter cerebral amyloid-β burden in several amyloid transgenic mouse models. Glia 66, 492–504 (2018).
Kummer, M. P. et al. Microglial PD-1 stimulation by astrocytic PD-L1 suppresses neuroinflammation and Alzheimer’s disease pathology. EMBO J. 40, e108662 (2021).
Kim, K. et al. Therapeutic B-cell depletion reverses progression of Alzheimer’s disease. Nat. Commun. 12, 2185 (2021).
Wolf, S. D., Dittel, B. N., Hardardottir, F. & Janeway, C. A. Experimental autoimmune encephalomyelitis induction in genetically B cell-deficient mice. J. Exp. Med. 184, 2271–2278 (1996).
Jack, C. R. et al. NIA-AA Research Framework: toward a biological definition of Alzheimer’s disease. Alzheimers Dement. 14, 535–562 (2018).
Jutten, R. J. et al. Finding treatment effects in Alzheimer trials in the face of disease progression heterogeneity. Neurology 96, e2673–e2684 (2021).
Bernick, C., Cummings, J., Raman, R., Sun, X. & Aisen, P. Age and rate of cognitive decline in Alzheimer disease: implications for clinical trials. Arch. Neurol. 69, 901–905 (2012).
Olivieri, P. et al. Characterization of the initial complaint and care pathways prior to diagnosis in very young sporadic Alzheimer’s disease. Alzheimers Res. Ther. 13, 90 (2021).
Lagarde, J. et al. Tau-PET imaging predicts cognitive decline and brain atrophy progression in early Alzheimer’s disease. J. Neurol. Neurosurg. Psychiatry 93, 459–467 (2022).
Mintun, M. A. et al. Donanemab in early Alzheimer’s disease. N. Engl. J. Med. 384, 1691–1704 (2021).
Groot, C. et al. Diagnostic and prognostic performance to detect Alzheimer’s disease and clinical progression of a novel assay for plasma p-tau217. Alzheimers Res. Ther. 14, 67 (2022).
Dujardin, S. et al. Tau molecular diversity contributes to clinical heterogeneity in Alzheimer’s disease. Nat. Med. 26, 1256–1263 (2020).
Dong, Y. et al. Neutrophil hyperactivation correlates with Alzheimer’s disease progression. Ann. Neurol. 83, 387–405 (2018).
Manly, J. J. & Deters, K. D. Donanemab for Alzheimer disease—who benefits and who is harmed? JAMA 330, 510–511 (2023).
Livingston, G. et al. Dementia prevention, intervention, and care: 2020 report of the Lancet Commission. Lancet 396, 413–446 (2020).
Liu, K. Y. et al. Evaluation of clinical benefits of treatments for Alzheimer’s disease. Lancet Healthy Longev. 4, e645–e651 (2023).
Salloway, S. et al. Amyloid-related imaging abnormalities in 2 phase 3 studies evaluating aducanumab in patients with early Alzheimer disease. JAMA Neurol. 79, 13–21 (2022).
Budd Haeberlein, S. et al. Two randomized phase 3 studies of aducanumab in early Alzheimer’s disease. J. Prev. Alzheimers Dis. 9, 197–210 (2022).
Cummings, J. et al. Lecanemab: appropriate use recommendations. J. Prev. Alzheimers Dis. 10, 362–377 (2023).
Chantran, Y. et al. Letter to the editor: serum anti-Aβ antibodies in cerebral amyloid angiopathy. Autoimmun. Rev. 20, 102870 (2021).
Alves, F., Kalinowski, P. & Ayton, S. Accelerated brain volume loss caused by anti-β-amyloid drugs: a systematic review and meta-analysis. Neurology 100, e2114–e2124 (2023).
Alexander, G. C., Emerson, S. & Kesselheim, A. S. Evaluation of aducanumab for Alzheimer disease: scientific evidence and regulatory review involving efficacy, safety, and futility. JAMA 325, 1717–1718 (2021).
Liu, K. Y. & Howard, R. Can we learn lessons from the FDA’s approval of aducanumab? Nat. Rev. Neurol. 17, 715–722 (2021).
Alexander, G. C. et al. Revisiting FDA approval of aducanumab. N. Engl. J. Med. 385, 769–771 (2021).
de Souza, L. C. et al. Similar amyloid-β burden in posterior cortical atrophy and Alzheimer’s disease. Brain 134, 2036–2043 (2011).
Rabinovici, G. D. et al. Increased metabolic vulnerability in early-onset Alzheimer’s disease is not related to amyloid burden. Brain 133, 512–528 (2010).
Ikonomovic, M. D. et al. Post-mortem correlates of in vivo PiB-PET amyloid imaging in a typical case of Alzheimer’s disease. Brain 131, 1630–1645 (2008).
Moloney, C. M., Lowe, V. J. & Murray, M. E. Visualization of neurofibrillary tangle maturity in Alzheimer’s disease: a clinicopathologic perspective for biomarker research. Alzheimers Dement. 17, 1554–1574 (2021).
Lagarde, J. et al. Could tau-PET imaging contribute to a better understanding of the different patterns of clinical progression in Alzheimer’s disease? A 2-year longitudinal study. Alzheimers Res. Ther. 15, 91 (2023).
Acknowledgements
G.D. is funded by the Agence Nationale de la Recherche (grant ANR-21-CE17-0054-01), the Fondation Alzheimer, the Fondation pour la Recherche Médicale and the Fondation Pierre Deniker. G.D. and B.B. are funded by the Association France Alzheimer, the Fondation Vaincre Alzheimer and Sorbonne University. M.S., J.L. and M.B. are funded by the Fondation Alzheimer, the Fondation Recherche Alzheimer, the Association France Alzheimer, the Fondation pour la Recherche Médicale and the French Ministry of Health (grants PHRC-2010-0054-N and PHRC-2013-0919).
Author information
Authors and Affiliations
Contributions
All authors contributed to researching data and literature related to the article, discussing its content, and writing, reviewing and/or editing the manuscript.
Corresponding authors
Ethics declarations
Competing interests
G.D. is listed as an inventor on one patent and one patent application, both on Treg-based immunomodulatory treatments in AD and other tauopathies. G.D. served on the scientific advisory board of Coya Therapeutics and received lecturer fees from Eisai. G.D. and M.S. received research support from Institut Roche. L.C.d.S. participated in the advisory board for Biogen and as speaker in symposia sponsored by Biogen. The other authors declare no competing interests.
Peer review
Peer review information
Nature Aging thanks Marie-Victoire Guillot-Sestier, Alon Monsonego and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sarazin, M., Lagarde, J., El Haddad, I. et al. The path to next-generation disease-modifying immunomodulatory combination therapies in Alzheimer’s disease. Nat Aging 4, 761–770 (2024). https://doi.org/10.1038/s43587-024-00630-2
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s43587-024-00630-2
This article is cited by
-
The aging gut–glia–immune axis in alzheimer’s disease: microbiome-derived mediators of neuroinflammation and therapeutic innovation
GeroScience (2026)
-
Antagonizing Il10 and Il4 signaling via intracerebral decoy receptor expression attenuates Aβ accumulation
Acta Neuropathologica Communications (2025)
-
Viruses and neurodegeneration: a growing concern
Journal of Translational Medicine (2025)
-
Exploring immunotherapeutic strategies for neurodegenerative diseases: a focus on Huntington’s disease and Prion diseases
Acta Pharmacologica Sinica (2025)
-
The immune system offers a window into aging
Nature Aging (2025)