Abstract
Background
Hepatectomy is considered advisable for some patients with intermediate or advanced hepatocellular carcinoma (HCC). The efficacy and safety of neoadjuvant transarterial chemoembolization with tyrosine kinase and immune checkpoint inhibitors (neoadjuvant triple therapy) for these patients remain unclear.
Methods
583 patients who met the resectable criteria and were assigned to receive neoadjuvant triple therapy (n = 205) or direct hepatectomy (n = 378) at 20 Chinese medical centers (2019–2023) were retrospectively compared in terms of overall survival (OS), event-free survival (EFS), adverse events, and postoperative complications. The subgroup stratified includes 106 patients who underwent neoadjuvant triple therapy followed by hepatectomy or 99 patients who received it without subsequent hepatectomy.
Results
Compared to patients who undergo direct hepatectomy, those who receive neoadjuvant triple therapy show significantly higher OS (hazard ratio [HR] 0.70, 95%CI 0.53–0.92) and significantly longer median EFS (19.7 vs 10.9 months). Similar results are obtained after propensity score matching (PSM). Among patients who undergo hepatectomy, those with prior neoadjuvant triple therapy have significantly better OS (HR 0.45, 95%CI 0.31–0.66) and EFS (HR 0.49, 95%CI 0.38–0.63) than those with direct hepatectomy. Similar results are obtained after PSM. Among patients who receive neoadjuvant triple therapy, OS is significantly better among those who subsequently underwent hepatectomy (HR 0.40, 95%CI 0.24-0.67). neoadjuvant triple therapy results in a complete pathologic response rate of 34.0%. However, the regimen is associated with high rates of serious adverse events and postoperative complications, including hepatic insufficiency, bile leakage, and ascites.
Conclusions
Neoadjuvant triple therapy offers OS and EFS benefits for patients with resectable intermediate or advanced HCC, but is associated with an increased risk of adverse events and postoperative complications.
Plain language summary
Intermediate- or advanced-stage hepatocellular carcinoma (HCC) is a cancer of the liver in which multiple tumors are present. People with HCC have a poor prognosis. One treatment is the delivery of chemotherapy directly to the tumor whilst blocking the blood supply to the tumor (transarterial chemoembolization) combined with other anti-cancer drugs (tyrosine kinase inhibitors and immune checkpoint inhibitors) prior to surgery to remove the cancer. This treatment, termed neoadjuvant triple therapy, followed by the surgery gives improved survival compared to surgery prior to treatment with drugs. However, this treatment is also associated with a higher incidence of severe postoperative complications. These findings suggest that neoadjuvant triple therapy may represent a viable treatment option for some people with intermediate- or advanced-stage HCC, but its application requires careful consideration of the survival benefits relative to the risks of treatment-related adverse events and death.
Similar content being viewed by others
Introduction
At the time of presentation, approximately 70% of patients with hepatocellular carcinoma (HCC) are found to be in intermediate or advanced stages1,2. According to Chinese HCC guidelines, hepatectomy is appropriate for selected patients in intermediate or advanced stages3,4. However, despite the absence of a universally accepted definition for resectable intermediate or advanced HCC in international guidelines5,6, hepatectomy for such patients classified as “resectable” has demonstrated survival benefits, albeit with high postoperative recurrence rates7,8,9,10. The American Association for the Study of Liver Diseases (AASLD) and the Japan Society of Hepatology extend the indication for hepatectomy to these groups of HCC patients in their latest consensus guidelines6,11. To reduce the risk of recurrence, many medical centers deliver neoadjuvant therapy, which may be local and/or systemic, in order to shrink tumors and eradicate micrometastatic disease before hepatectomy. EASL guidelines recommend transarterial chemoembolization (TACE) as the standard treatment for HCC patients in intermediate or certain advanced stages5,12. Previous studies found that neoadjuvant TACE did not reduce the risk of postoperative recurrence in resectable HCC13,14. While two clinical trials have demonstrated the survival benefit of neoadjuvant cemiplimab or neoadjuvant nivolumab plus ipilimumab for patients with resectable HCC15,16, neoadjuvant therapy carries the risk of adverse events, tumor progression, or difficulties in subsequent hepatectomy17,18. In the LEAP-012 and EMERALD-1 studies, TACE combined with immune checkpoint inhibitors (ICIs) and tyrosine kinase inhibitors (TKIs), or TACE combined with ICIs and anti-VEGF monoclonal antibody, for unresectable HCC achieved significantly improved overall survival and progression-free survival rates compared with TACE alone19,20. Our recent large retrospective study also supports the efficacy of TACE combined with ICIs and TKIs for unresectable HCC21. However, in patients with resectable intermediate or advanced HCC, the efficacy of neoadjuvant TACE combined with ICIs and TKIs (neoadjuvant triple therapy) in terms of improving prognosis and reducing the risk of postoperative recurrence remains uncertain.
This multicenter study confirms that neoadjuvant triple therapy serves as an effective conversion therapy for initially resectable intermediate/advanced HCC. It achieves a successful downstaging rate in a substantial proportion of patients, enabling curative resection. Furthermore, the therapy offers sustained disease control with a tolerable safety profile in both surgical and non-surgical cohorts, establishing its dual role in the management of HCC.
Methods
The use of data from this registry for the present study was reviewed and approved by the Ethics Committee of Guangxi Medical University Cancer Hospital (approval KY2024386), and conducted in accordance with the Declaration of Helsinki22. The committee granted a waiver of individual informed consent for this specific analysis because patients, at the time of treatment, consented to their anonymized medical data being analyzed and published for research purposes. The data for this study were extracted from the regional registry “Guangxi Liver Cancer Clinical Study Alliance” (GUIDANCE)21,23,24, curated by the Liver Cancer Committee of the Guangxi Anticancer Association, and the study was registered on ClinicalTrials.gov (NCT06405321). The database was developed by the corresponding author’s institution and was intended for internal use exclusively by the GUIDANCE investigators.
Patients
Data were retrospectively analyzed for patients who were (1) treated with neoadjuvant TACE combined with ICIs and TKIs (neoadjuvant triple therapy) or hepatectomy directly at one of the 20 participating medical centers in China between January 1, 2019 and June 31, 2023; (2) 18–75 years old on admission; (3) had at least one measurable intrahepatic lesion according to Response Evaluation Criteria in Solid Tumors version 1.1(RECIST 1.1)25, and (4) diagnosed with “resectable” (see hepatectomy below) primary HCC in stage B (“intermediate”) or C (“advanced”) of the Barcelona Clinic Liver Cancer (BCLC) staging system12 based on biopsy or noninvasive assessment according to consensus guidelines26. Patients were excluded (1) if there was a presence of extrahepatic metastasis; (2) If there is a presence of cancer thrombus involving the main portal vein or the portal vein branches on the lobe opposite to the primary focus, or involving the inferior vena cava; (3) if there was diffuse HCC; (4) if they had been diagnosed with any other malignancy in the previous five years; (5) if they were followed up for shorter than three months after the first study treatment; (6) or if their data on clinicodemographic characteristics, laboratory tests and outcomes were incomplete.
Neoadjuvant triple therapy
Neoadjuvant triple therapy consists of neoadjuvant TACE, ICIs and TKIs. TACE was performed as follows: under digital subtraction angiography guidance, clinicians performed superselective microcatheter cannulation into the arterial branches supplying tumor nodules. Epirubicin (Pfizer Pharmaceutical, Wuxi, China) was slowly injected in iodized oil until tumor saturation, followed by gelatin sponge particles as needed until microcatheter tip blood flow ceased. All tumors required embolization, and the tumor with the largest diameter was designated as the target lesion.
In addition to TACE, patients in the “neoadjuvant triple therapy” group also received the following TKIs plus ICIs (Supplemental Table 2). Different combination therapies with TKIs plus ICIs were permitted across the study population to reflect real-world practice more accurately, as these combinations showed similar overall response rate27. The following ICIs were used: tislelizumab (BGB-A317, BeiGene, Beijing, China), 200 mg; atezolizumab (Tecentriq, Roche, Basel, Switzerland), 1200 mg; sintilimab (IBI308, Innovent Biologics, Suzhou, China), 200 mg; camrelizumab (SHR-1210, Jiangsu HengRui Medicine), 200 mg; toripalimab (triprizumab or JS001, Shanghai Junshi Biosciences, Shanghai, China), 240 mg; and pembrolizumab (Keytruda, MSD Ireland, Carlow, Ireland), 200 mg. The following TKIs were used: lenvatinib (Eisai, Woodcliff Lake, NJ, USA), orally once a day at a dose of 12 mg or, for individuals <60 kg, 8 mg; donafenib (Zepsun®, Suzhou Zejing Biopharmaceutical, Suzhou, China), orally twice a day at a daily dose of 0.2 g; apatinib (Jiangsu HengRui Medicine, Jiangsu, China), orally once a day at a dose of 250 mg; and bevacizumab (Avastin, Shanghai Roche Pharmaceutical, Shanghai, China), intravenously at a dose of 15 mg/kg once every three weeks.
In order to ensure patient safety, hepatectomy was conducted at least four weeks after the last cycle of TACE28. TKIs were discontinued 1–2 weeks before hepatectomy; ICIs, 2–4 weeks before; and bevacizumab, 6 weeks before28.
At 3–4 weeks after neoadjuvant triple therapy, tumor response was assessed using contrast-enhanced computed tomography or magnetic resonance imaging based on RECIST version 1.125 or its modified version29. Response was categorized as complete response, partial response, stable disease or progressive disease. The objective response rate was defined as the sum of the rates of complete and partial response, while the disease control rate was defined as the sum of the rates of objective response and stable disease. Response was determined independently by two experienced radiologists at each site, and discrepancies were resolved through discussion. Consistent with routine practice at the participating medical centers30, patients chronically infected with hepatitis B or C virus received antivirals or nucleoside/nucleotide analogs throughout the study.
Hepatectomy
BCLC B or C stage HCC had to meet all the following criteria3,31 to be considered resectable: (1) Eastern Cooperative Oncology Group performance status of 0 or 1; (2) non-diffuse HCC; (3) sufficient remnant functional liver volume after excision of all nodules, defined as a remnant liver volume ≥30% of the standard liver volume in patients without cirrhosis or ≥40% of the standard liver volume in patients with cirrhosis; (4) indocyanine green retention rate <15% within 15 min; (5) Child-Pugh class A liver function or, if fewer than three Couinaud liver segments had to be resected, Child-Pugh class B liver function; (6) no extrahepatic metastasis; (7) no cancer thrombus involving the main portal vein or the portal vein branches on the lobe opposite to the primary focus, and no cancer thrombus involving the inferior vena cava; (8) absence of other contraindications for hepatectomy. Patients with tumors localized to the same hepatic segment or ipsilateral hemiliver may undergo hepatectomy or intraoperative ablation of lesions outside the resection margin, and even if the tumor count exceeds three, surgical resection may be recommended following multidisciplinary team discussion (including hepatobiliary surgeons, radiologists, interventional radiologist, internists, and anesthesiologist)3,32. Physicians recommend either direct hepatectomy or neoadjuvant triple therapy as viable treatment options; however, the final decision regarding the treatment plan is made by the patient, their family, or their legal representative. All preoperative imaging was interpreted and evaluated for the resectability of the tumors by a senior radiologist at each participating center, as well as by one senior radiologist at the leading center, Guangxi Medical University Cancer Hospital. All interpreting radiologists had at least 10 years of professional experience. Surgical procedures have been described elsewhere3,6 and were conducted by surgeons with more than 10 years of experience and all participating centers followed the same surgical guidelines and standards26. Patients underwent hepatectomy following neoadjuvant triple therapy if the procedure was deemed appropriate by a multidisciplinary team and if patients consented to it. Curative resection was defined as the surgical removal of all tumor tissue, as well as the surrounding tissues and the regional lymph nodes that might have been invaded by the tumor, with the goal of achieving total cure. After hepatectomy, patients deemed to possess at least one characteristic placing them at high risk of recurrence received one cycle of adjuvant TACE; alternatively, they received adjuvant ICIs for up to 12 months, or until tumor recurrence occurred33. Adjuvant TKIs or radiotherapy were not normally recommended. All patients received active surveillance after hepatectomy34. Complete pathologic response was defined as the absence of residual viable tumor in successive pathology sections of all surgical resection specimens; major pathologic response, as a reduction of at least 70% in the number of tumor cells within the tumor bed; and non-major pathologic response, as a reduction of less than 70%28.
Follow-up and further treatment
All patients underwent rigorous follow-up, beginning with visits every 6–8 weeks after the first cycle of neoadjuvant therapy. After hepatectomy, patients were followed up 1 month later, then once every 3–4 months during the first 2 years, then once every 6 months thereafter3. Outcomes including survival, tumor recurrence and tumor progression were assessed at outpatient visits, after readmission or through telephone follow-up. Each follow-up comprised physical examinations, α-fetoprotein testing, and vein contrast-enhanced computed tomography or magnetic resonance imaging. Assessments were conducted during outpatient visits or after readmission; when necessary, data on survival were obtained via telephone. The last follow-up in this study occurred on June 15, 2024. HCC recurrence or metastasis during follow-up were treated through TACE, repeat hepatectomy or local ablation35 depending on the patient’s liver function and overall condition, as well as on the number, size and location of tumors.
Outcomes
The primary endpoint was overall survival, defined as the time from the date of the initial treatment to the date of death from any cause or the last follow-up. The secondary endpoint was event-free survival, defined as the interval from when a patient underwent initial treatment (direct hepatectomy or first neoadjuvant triple therapy) until tumor progression in patients who did not undergo hepatectomy, tumor recurrence in those who did, or any-cause death from any cause occurring before tumor progression or recurrence. If patients did not experience tumor progression, recurrence or death, event-free survival was defined as the time from the start of the first study treatment until the last follow-up.
Postoperative complications were assessed using the Clavien-Dindo classification36. Liver failure was diagnosed on postoperative day 5 according to the “50–50 criteria”37.
Statistics and reproducibility
Data were analyzed statistically using SPSS 27.0 (IBM, Chicago, IL, USA), GraphPad Prism 8.0 (GraphPad Software, San Diego, CA, USA) and R 4.3.2 (R Project, Vienna, Austria) Results associated with a two-tailed p < 0.05 were considered significant. Continuous data were reported as mean ± standard deviation if normally distributed, or as median (interquartile range, IQR) if skewed. Categorical data were expressed as n (%). Inter-group differences in continuous variables were assessed using Student’s t test or the Mann–Whitney U test, while differences in categorical variables were evaluated using Pearson’s chi-squared test or Fisher’s exact test.
Overall and event-free survival were calculated using Kaplan-Meier curves, which were compared between groups using the log-rank test. A univariable Cox regression analysis was conducted to identify factors influencing poor prognosis, including first study treatment, age, gender, hepatitis B virus surface antigen status, liver cirrhosis, alanine aminotransferase levels, prothrombin time, platelet count, Child-Pugh liver function score, α-fetoprotein levels, maximum tumor diameter, tumor number, and macrovascular invasion. Variables associated with poor survival in univariable Cox regression (p < 0.05) were included in a multivariable Cox proportional-hazards model. The independent influence of each such variable on risk was quantified in terms of hazard ratios (HRs) and corresponding 95% confidence intervals (CIs). Logistic regression analysis was conducted to detect predictors of the loss of resection opportunity resulting from tumor progression after neoadjuvant triple therapy, with predictors significant in the univariate analysis (p < 0.05) being included in the multivariable analysis.
Patients were stratified into subgroups based on risk factors, including age, sex, hepatitis B virus surface antigen status, liver cirrhosis, Child-Pugh liver function, α-fetoprotein levels, maximum tumor diameter, tumor number, macrovascular invasion, and BCLC stage. To minimize confounding from clinical and demographic differences at baseline, some analyses were repeated using 1:1 or 1:2 matched subsets of patients from the two groups, based on propensity score matching (PSM) with a caliper width of 0.2 standard deviations. A standardized mean difference (SMD) < 0.1 between matched groups indicated good balance38.
Ethics statement
This study was approved by the Ethics Committee of Guangxi Medical University Cancer Hospital (approval KY2024386), registered on ClinicalTrials.gov (NCT06405321) and conducted in accordance with the Declaration of Helsinki. The data for this study were extracted from the database of the regional registry “Guangxi Liver Cancer Clinical Study Alliance (GUIDANCE)” curated by the Liver Cancer Committee of the Guangxi Anticancer Association.
Results
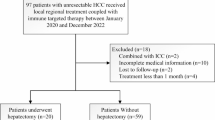
Of the 2239 patients screened for enrollment, 583 were included in the final analysis (Fig. 1), of whom 378 (64.8%) underwent hepatectomy directly and 205 (35.2%) received neoadjuvant triple therapy. In patients after neoadjuvant triple therapy, 106 (51.7%) underwent hepatectomy, and 99 (48.3%) did not receive hepatectomy, either because the patient refused surgery and opted for local and/or systemic therapy despite resectability (n = 83, 83.8%) or because their tumors had progressed to an unresectable condition (n = 16, 16.2%). Median follow-up was 30.2 months for patients who received direct hepatectomy and 26.1 months for those who received neoadjuvant triple therapy.
HCC hepatocellular carcinoma, TACE transarterial chemoembolization, HAIC hepatic arterial infusion chemotherapy.
Compared to patients who received neoadjuvant triple therapy, those who proceeded directly to hepatectomy were significantly more likely to have smaller tumors, or stage C disease according to the BCLC system; they showed significantly higher level of total bilirubin; and they showed significantly lower albumin and platelet count level (Table 1). No significant difference in baseline features existed when subsets of 167 pairs of patients from neoadjuvant triple therapy and direct hepatectomy group (all SMD < 0.1). The probability density curves pre- and post-PSM are presented in Supplementary Fig. 1.
Compared to patients who underwent hepatectomy directly, those who received neoadjuvant triple therapy first and then underwent hepatectomy were older, less likely to have smaller tumors, a single tumor, stage C disease; they showed significantly higher level of total bilirubin; and they had significantly lower albumin and platelet count levels (Supplementary Table 1). None of these differences persisted when subsets of 96 patients from each group were matched using PSM. Compared to patients who underwent direct hepatectomy, those who received neoadjuvant triple therapy without subsequent hepatectomy were older, less likely to have stage C disease, and showed significantly lower total bilirubin levels (Supplementary Table 2). After PSM, 88 patients who received neoadjuvant triple therapy without subsequent hepatectomy were compared to 176 patients who underwent hepatectomy directly, and the SMDs for all baseline characteristics were smaller than 0.1, indicating good balance.
Patients who received neoadjuvant triple therapy received a median of 2 TACE cycles (IQR, 1 to 3), 3 cycles of ICIs (IQR, 2 to 4), and TKIs for a median duration of 3 months (IQR, 2.0 to 6.0). The combinations of ICIs and TKIs used in this study are shown in Supplementary Table 3. Neither overall survival nor event-free survival varied significantly with the type of ICI used in neoadjuvant triple therapy (Supplementary Fig. 2A, B). Overall survival varied significantly according to the type of TKI used in neoadjuvant triple therapy, whereas event-free survival remained similar across subgroups (Supplementary Fig. 2C, D).
Efficacy
During follow-up, 170 (45.0%) of patients who underwent direct hepatectomy died, compared to 18 (17.0%) of patients who underwent hepatectomy after neoadjuvant triple therapy and 38 (38.4%) of patients who received neoadjuvant triple therapy but did not subsequently undergo hepatectomy. Patients who underwent neoadjuvant triple therapy were associated with significantly a higher rate of overall survival at 1 year (88.7 vs 78.7%), 2 years (65.9 vs 59.5%), and 3 years (52.9 vs 49.3%) than those who underwent hepatectomy directly without prior neoadjuvant therapy (HR 0.70, 95%CI 0.53–0.92, p = 0.017; Fig. 2a). This protective effect was also observed in the subsets of patients matched through PSM (Fig. 2c) and in subgroups stratified by various clinicodemographic variables, except those who were older than 60 years, female, had an absence of cirrhosis, were seropositive for hepatitis B surface antigen, had α-fetoprotein levels <400 ng/ml, had Child-Pugh liver function grade B, had a maximum tumor diameter <10 cm, had a single tumor, had macrovascular invasion, or were in BCLC stage B (Supplementary Fig. 3a). One of the three events (death, disease progression, or recurrence) occurred during follow-up in 97 (47.3%) of patients who received neoadjuvant triple therapy and 267 (70.6%) of patients who underwent direct hepatectomy. Neoadjuvant triple therapy was associated with significantly longer median event-free survival [19.7 months (95%CI 16.9 to 22.5) vs 10.0 months (95%CI, 6.7 to 13.3); p < 0.001] (Fig. 2b). This survival benefit persisted after PSM (Fig. 2d) and after stratifying patients by various clinicodemographic variables, except those who were female and presence of cirrhosis (Supplementary Fig. 3b).
a overall survival in the entire study sample; b event-free survival in the entire study sample; c overall survival in propensity score-matched subgroups; d event-free survival in propensity score-matched subgroups. CI confidence interval; neoadjuvant triple therapy, neoadjuvant transarterial chemoembolization combined with immune checkpoint inhibitors and tyrosine kinase inhibitors.
Patients who underwent hepatectomy with prior neoadjuvant triple therapy were associated with a significantly higher rate of overall survival at 1 year (95.0 vs 79.2%), 2 years (73.7 vs 62.3%) and 3 years (68.4 vs 52.1%) than those who underwent hepatectomy directly (HR 0.45, 95%CI 0.31–0.66, p = 0.001; Fig. 3a). Similar results were observed after PSM (Fig. 3c). Patients who underwent hepatectomy with prior neoadjuvant triple therapy were associated with significantly longer event-free survival [19.7 months (95%CI 16.6–22.8) vs 10.0 months (95%CI, 6.7–13.3); p < 0.001] (Fig. 3b). This survival benefit persisted after PSM (Fig. 3d). Patients received neoadjuvant triple therapy without subsequent hepatectomy demonstrated significantly longer event-free survival than those underwent direct hepatectomy (HR 0.65, 95% CI 0.50–0.84; Supplementary Fig. 4A, B), but similar overall survival (HR 0.99, 95% CI 0.70–1.42). After PSM, no significant differences in event-free survival (HR 0.73, 95% CI 0.54-1.02) or overall survival (HR 0.92, 95% CI 0.68–1.50; Supplementary Fig. 4C, D) were observed between the two groups. In patients with BCLC stage C HCC, those who underwent hepatectomy following prior neoadjuvant triple therapy demonstrated significantly improved overall survival (HR 0.33, 95%CI 0.20–0.56) and event-free survival (HR 0.39, 95%CI 0.26–0.57) compared to those who underwent direct hepatectomy (Supplementary Fig. 5), with no significant differences in baseline features between the two groups (Supplementary Table 4). Among patients who received neoadjuvant therapy, overall survival was significantly higher when it was followed by hepatectomy (HR 0.40, 95%CI 0.24-0.67, p < 0.001; Supplementary Fig. 6a). However, event-free survival was similar regardless of whether it was followed by hepatectomy or not (HR 0.73, 95%CI 0.49–1.09; Supplementary Fig. 6b). After excluding 16 patients who lost the opportunity for surgery due to tumor progression, patients who underwent hepatectomy after neoadjuvant triple therapy exhibited significantly higher overall survival (HR 0.41 95%CI 0.23–0.74) but similar event-free survival (HR 0.92 95%CI 0.60–1.42) compared to those who refused to undergo hepatectomy after neoadjuvant triple therapy (Fig. 4). The proportion of a single tumor at baseline was significantly higher in patients with complete or partial response after neoadjuvant triple therapy than in those with stable or progressive disease (p = 0.014; Supplementary Table 5).
a Overall survival in the entire study sample; b event-free survival in the entire study sample; c overall survival in propensity score-matched subgroups; d event-free survival in propensity score-matched subgroups. CI confidence interval; neoadjuvant triple therapy, neoadjuvant transarterial chemoembolization combined with immune checkpoint inhibitors and tyrosine kinase inhibitors.
a overall survival; b event-free survival. CI confidence interval; neoadjuvant triple therapy, neoadjuvant transarterial chemoembolization combined with immune checkpoint inhibitors and tyrosine kinase inhibitors.
Predictors of worse survival
In the overall population, clinicodemographic variables significantly associated with worse overall or event-free survival were entered into multivariable analysis, which identified the following independent predictors of worse survival (Supplementary Table 6): direct hepatectomy instead of neoadjuvant triple therapy, absence of cirrhosis, α-fetoprotein ≥400 ng/ml, and macrovascular invasion. Child-Pugh liver function grade B was a predictor of poor event-free survival, but not poor overall survival. In patients who underwent hepatectomy whether they had prior neoadjuvant triple therapy or not, clinicodemographic variables significantly associated with worse overall or event-free survival were entered into multivariable analysis, which identified the following independent predictors of worse survival (Supplementary Table 7): no prior neoadjuvant triple therapy, liver cirrhosis, maximum tumor diameter ≥10 cm, and macrovascular invasion. Child-Pugh liver function grade B, α-fetoprotein ≥400 ng/ml, and tumor differentiation of type III/IV were predictors of poor event-free survival, but not poor overall survival. Hepatic insufficiency was a predictor of poor overall survival, but not poor event-free survival.
Predictors of the loss of resection opportunity resulting from tumor progression
31 patients developed tumor progression after neoadjuvant triple therapy, and 16 of these patients missed the opportunity for surgery due to tumor progression. Univariate logistic regression identified several baseline clinical factors associated with the loss of resection opportunity due to tumor progression after neoadjuvant triple therapy. Among these, α-fetoprotein ≥400 ng/ml and macrovascular invasion were independently identified as predictors of increased likelihood of tumor progression and subsequent loss of resection opportunity (Supplementary Table 8).
Safety, pathologic characteristics, and adjuvant treatments
One in three of the 205 patients who underwent neoadjuvant triple therapy experienced a grade 3 or 4 adverse event, and more than 10% of patients experienced at least one of the following events: vomiting, hypertension, abdominal pain, hypoalbuminemia, rash, ascites, decreased appetite, anemia, fever, fatigue, hand–foot skin reaction, or nausea (Supplementary Table 9). Neoadjuvant triple therapy was associated with an objective response rate of 67.3% and a disease control rate of 84.9% based on modified RECIST criteria39, as well as with a complete pathologic response rate of 34.0% and major pathologic response rate of 43.4%. Compared to patients who underwent hepatectomy directly, those who underwent hepatectomy after neoadjuvant triple therapy were significantly more likely to experience postoperative complications, such as hepatic insufficiency (grade III/IV), bile leakage (grade I/II), and ascites (grade I/II) (Supplementary Table 10). Patients who underwent hepatectomy after prior neoadjuvant triple therapy had significantly higher rates of absence of microvascular invasion and greater intraoperative blood loss compared to those who did not undergo prior neoadjuvant triple therapy; however, they had similar rates of tumor differentiation (type III/IV), adjuvant therapy, and treatment outcomes after recurrence or metastasis (Supplementary Table 11).
Discussion
Our multi-site study with a relatively large population of intermediate or advanced stages HCC patients suggests that neoadjuvant triple therapy prior to hepatectomy significantly prolongs overall survival or event-free survival, and results in a good major or complete pathologic response. However, this therapy may significantly increase the risk of adverse events, including serious ones. Our analyses suggest that, on balance, neoadjuvant triple therapy provides greater survival benefits than direct hepatectomy in this population, but it should be administered after careful consideration of adverse events and postoperative complications.
In this study, neoadjuvant triple therapy significantly extended overall survival and reduced postoperative recurrence while delaying tumor progression compared with direct hepatectomy. Currently, neither our findings nor those of other studies can definitively identify whether TACE, TKIs, or ICIs are the primary drivers of therapeutic efficacy in triple therapy, a critical challenge in both clinical practice and translational research. However, emerging evidence from the LEAP-012 and EMERALD-1 trials robustly demonstrated that triple conversion therapy was associated with longer median OS and PFS than TACE alone in patients with unresectable HCC40,41. In the present study, the 3-year overall survival rate for neoadjuvant triple therapy in resectable HCC was 52.9%, higher than the 40.4% reported for neoadjuvant TACE in a previous study13. Similarly, the 3-year event-free survival rate for neoadjuvant triple therapy was 29.4%, compared to 25.5% for neoadjuvant TACE13. In the trial of neoadjuvant three-dimensional conformal radiotherapy for resectable HCC, the interleukin-6 may serve as a potential biomarker for predicting treatment response to radiation42. Specifically, in this study, the presence of a single tumor at baseline was associated with a better treatment response to neoadjuvant triple therapy. However, further research is needed to clarify the optimal patient population most likely to benefit from this treatment approach. Among our patients who received neoadjuvant triple therapy, those who subsequently underwent hepatectomy showed significantly better overall survival than those who did not, consistent with previous work43. One disadvantage of neoadjuvant triple therapy in this cohort is that it meant postponing hepatectomy, which led to tumor progression in 16 initially resectable patients, rendering them ineligible for hepatectomy, representing a key trade-off between disease control and the preservation of surgical eligibility. The tumor progression to unresectable disease during neoadjuvant therapy may be related to tumor biology and early advancement driven by tumor heterogeneity and insensitivity to therapy. Logistic regression identified α-fetoprotein ≥400 ng/ml and macrovascular invasion as independent predictors of increased likelihood of tumor progression and subsequent loss of resection opportunity. Close monitoring of tumor response to neoadjuvant triple therapy, particularly via imaging and α-fetoprotein level changes, is recommended for patients with the aforementioned risk factors. Prompt termination of therapy and transition to surgical intervention should be considered if the anticipated tumor shrinkage is not achieved. This result mirrors the findings of the previous trial on neoadjuvant radiotherapy in resectable HCC42. This result highlights the potential risks of neoadjuvant therapy (e.g., disease progression) and the complexity of treatment timing, directly underscoring the “double-edged sword” nature of neoadjuvant therapy: on one hand, it may create surgical opportunities in the short term by reducing tumor burden (e.g., downstaging); on the other hand, disease progression during treatment can lead to irreversible loss of resectability (e.g., tumor dissemination or increased irresectability), thereby potentially compromising long-term survival outcomes. This trade-off must be carefully considered in designing individualized treatment strategies, emphasizing the importance of closely monitoring responses to neoadjuvant therapy. A multidisciplinary team consultation involving hepatobiliary surgeons, interventional radiologists, diagnostic radiologists, oncologists, hepatologists, anesthesiologists, and other relevant specialists will be conducted prior to hepatectomy to comprehensively evaluate the resectability of HCC, optimize perioperative care and long-term management of HCC patients, minimize adverse events, proactively manage postoperative complications, and ultimately enhance individualized treatment outcomes, including prognosis and quality of life3. This process integrates patient-specific factors (such as liver function reserve, comorbidities, and physical condition), tumor characteristics (such as the depth of response to neoadjuvant therapy, aggressiveness), and treatment goals to determine the optimal surgical timing. We look forward to future studies exploring potential biomarkers through blood sample analysis and cancer tissue analysis to guide the identification of patients most likely to benefit from neoadjuvant triple therapy.
Hepatectomy should be performed if appropriate to ensure survival benefit. The data support the usefulness of hepatectomy for appropriately selected patients with intermediate or advanced HCC, as first suggested by numerous retrospective studies7,8,9,44 and reflected in the expanded indications for hepatectomy in the latest guidelines from the AASLD6. The patients in this study who underwent direct hepatectomy without prior neoadjuvant triple therapy achieved a 3-year overall survival rate of 52.1%, but experienced a high recurrence rate. Patients who received neoadjuvant triple therapy followed by hepatectomy achieved a 3-year overall survival rate of 68.4% and median event-free survival of 19.7 months, significantly better than those who underwent direct hepatectomy. We caution that direct comparisons of outcomes between patients underwent hepatectomy direct and those received neoadjuvant therapy followed by hepatectomy may be confounded by potential selection bias, as the latter group might exhibit more favorable tumor biology, demonstrate superior treatment response to neoadjuvant therapy, and achieve tumor downstaging. This indicates that a good major or complete pathologic response and a lower M0 microvascular invasion rate after neoadjuvant triple therapy translates to better overall or event-free survival. Similar results were observed in the BCLC stage C subset. This study included a large number of BCLC stage C patients, for whom resectability assessment is challenging and may introduce selection bias. It is recommend that aggressive surgical resection of intermediate or advanced HCC be performed only at tertiary care centers with sufficient experience and after multidisciplinary evaluation, in accordance with the latest AASLD and Chinese guidelines3,6. Such assessments should incorporate factors including extrahepatic spread, tumor burden and invasion, tumor location, α-fetoprotein level, liver dysfunction, size of future liver remnant, age, performance status, and comorbidities.
The subgroup analysis showed that most clinical characteristics benefit from neoadjuvant triple therapy before hepatectomy. However, subgroup analysis identified certain patient characteristics that may not necessitate neoadjuvant triple therapy but instead allow for direct hepatectomy, which are associated with better survival outcomes. These characteristics include being female, the absence of cirrhosis, the presence of a single tumors, and the absence of macrovascular invasion. Consistent with previous work43, this study identified α-fetoprotein level, liver cirrhosis, and macrovascular invasion as independent risk predictors of overall survival in patients with initially resectable HCC in intermediate or advanced stages. Future studies should be sure to consider these factors as potential selection criteria for neoadjuvant therapy.
Over one-third of the patients who received neoadjuvant triple therapy experienced grade 3 or 4 adverse events, and 83.9% had any adverse events. The rates of bile leakage, hepatic insufficiency (grade III/IV), ascites and intraoperative blood loss were relatively high among patients who underwent neoadjuvant triple therapy followed by hepatectomy. Moreover, long-term systemic therapy and local interventional therapy increase healthcare costs and psychological pressure. Previous studies have demonstrated that preoperative TACE may increase intraoperative difficulty and perioperative complications45. A subset of patients may experience tumor necrosis with adhesion to surrounding tissues after neoadjuvant therapy, necessitating surgeons with advanced technical expertise to manage these treatment-induced adhesions. However, existing literature suggests that the adverse effects of preoperative TACE on surgical outcomes can be mitigated if an adequate interval is maintained between the final cycle of TACE or systemic therapy and surgical resection46,47. Therefore, it is imperative to establish a multidisciplinary team for preoperative evaluation of surgical feasibility, procedural complexity, and the necessity of alternative therapeutic strategies. This approach ensures informed decision-making. It also optimizes patient outcomes in the context of neoadjuvant therapy. The study suggested that neoadjuvant triple therapy in patients with initially resectable intermediate or advanced stages HCC can lead to a high proportion of adverse events and postoperative complications. These complications affected quality of life and reduced patients’ willingness to complete the therapy, which must be taken into account by surgeons when making medical decisions. After curative hepatectomy, patients who received neoadjuvant therapy were no longer on the preoperative systemic therapy regimens. The rate of adjuvant therapy was similar between patients who underwent hepatectomy with or without prior neoadjuvant triple therapy48. Adjuvant therapy, including TACE and/or ICIs, was administered to nearly one-third of patients. The included cohort represented a highly selected group, and the increased perioperative complications associated with preoperative therapies necessitated careful risk-benefit evaluation. Future studies should focus on identifying patients most likely to benefit from this approach, particularly those who are otherwise unresectable patients at high risk of recurrence.
The study acknowledges some limitations. First, the retrospective design may have introduced selection bias, but recruitment from 20 medical centers captured natural clinical heterogeneity. Although we employed PSM and multivariate regression analysis to minimize confounding, potential selection bias may still have systematically influenced our analyses. Second, the combinations of ICIs and TKIs were treated as an aggregate variable, and this heterogeneity was not adjusted. Third, despite standardized surgical protocols and experienced surgeons, variations in surgical techniques and quality were unavoidable. Fourth, although hepatectomy is not recommended in some Western guidelines for intermediate or advanced stages HCC, patients in this real-world study chose it due to financial burden, personal preference, and surgical expertise. Fifth, the limited case volume per center in the neoadjuvant triple therapy group may compromise the robustness of the results. Sixth, due to the inherently limited number of BCLC B or C stage HCC patients and eligible for surgical resection, particularly those undergoing neoadjuvant therapy, the overall sample size across 20 centers over four years remained constrained. Future work should validate these findings with prospective, randomized trials with larger cohorts.
Despite its limitations, this multicenter retrospective study provided the first evidence that neoadjuvant triple therapy achieved better overall and event-free survival than direct hepatectomy for resectable intermediate or advanced stages HCC. However, the survival benefits must be carefully balanced against the associated risks. Further validation through a trial-level analysis is required.
Data availability
The source data underlying the present analyses are available in Supplementary Data 1-5. The data for individual graphs and tables can be found as follows: Supplementary Data 1-2 (for Fig. 2 and Table 1), Supplementary Data 3-4 (for Fig. 3), and Supplementary Data 5 (for Fig. 4). The datasets used and/or analyzed during the current study are available from the corresponding author, Jian-Hong Zhong (email: zhongjianhong66@163.com), upon reasonable request.
References
Zhong, J. H. et al. Tumor stage and primary treatment of hepatocellular carcinoma at a large tertiary hospital in China: a real-world study. Oncotarget 8, 18296–18302 (2017).
Park, J. W. et al. Global patterns of hepatocellular carcinoma management from diagnosis to death: the BRIDGE Study. Liver Int. 35, 2155–2166 (2015).
Zhou, J. et al. Guidelines for the diagnosis and treatment of primary liver cancer (2022 Edition). Liver Cancer 12, 405–444 (2023).
Zhong, J. H. et al. Intermediate-stage HCC–upfront resection can be feasible. Nat. Rev. Clin. Oncol. 12, 295 (2015).
EASL Clinical Practice Guidelines on the management of hepatocellular carcinoma. J. Hepatol. 82, 315–374 (2025).
Singal, A. G. et al. AASLD Practice Guidance on prevention, diagnosis, and treatment of hepatocellular carcinoma. Hepatology 78, 1922–1965 (2023).
Zhong, J. H., Ke, Y., Wang, Y. Y. & Li, L. Q. Liver resection for patients with hepatocellular carcinoma and macrovascular invasion, multiple tumours, or portal hypertension. Gut 64, 520–521 (2015).
Zhong, J. H. et al. Hepatic resection associated with good survival for selected patients with intermediate and advanced-stage hepatocellular carcinoma. Ann. Surg. 260, 329–340 (2014).
Kokudo, T. et al. Survival benefit of liver resection for hepatocellular carcinoma associated with portal vein invasion. J. Hepatol. 65, 938–943 (2016).
Govalan, R. et al. Comparison of surgical resection and systemic treatment for hepatocellular carcinoma with vascular invasion: National Cancer Database Analysis. Liver Cancer 10, 407–418 (2021).
Hasegawa, K. et al. Clinical practice guidelines for hepatocellular carcinoma: The Japan Society of Hepatology 2021 version (5th JSH-HCC Guidelines). Hepatol. Res. 53, 383–390 (2023).
Reig, M. et al. BCLC strategy for prognosis prediction and treatment recommendation: the 2022 update. J. Hepatol. 76, 681–693 (2022).
Zhou, W. P. et al. A prospective, randomized, controlled trial of preoperative transarterial chemoembolization for resectable large hepatocellular carcinoma. Ann. Surg. 249, 195–202 (2009).
Chua, T. C. et al. Systematic review of neoadjuvant transarterial chemoembolization for resectable hepatocellular carcinoma. Liver Int. 30, 166–174 (2010).
Marron, T. U. et al. Neoadjuvant cemiplimab for resectable hepatocellular carcinoma: a single-arm, open-label, phase 2 trial. Lancet Gastroenterol. Hepatol. 7, 219–229 (2022).
Kaseb, A. O. et al. Perioperative nivolumab monotherapy versus nivolumab plus ipilimumab in resectable hepatocellular carcinoma: a randomised, open-label, phase 2 trial. Lancet Gastroenterol. Hepatol. 7, 208–218 (2022).
Llovet, J. M. et al. Adjuvant and neoadjuvant immunotherapies in hepatocellular carcinoma. Nat. Rev. Clin. Oncol. 21, 294–311 (2024).
Vogel, A. et al. Adjuvant and neoadjuvant therapies for hepatocellular carcinoma. Hepatology 82, 777–793 (2025).
Kudo, M. et al. Safety analysis by treatment periods from EMERALD-1: A phase 3, randomized, placebo-controlled study of transarterial chemoembolization with durvalumab with/without bevacizumab in participants with embolization-eligible unresectable hepatocellular carcinoma. J. Clin. Oncol. 42, 4122 (2024).
Llovet, J. et al. LBA3 Transarterial chemoembolization (TACE) with or without lenvatinib (len) + pembrolizumab (pembro) for intermediate-stage hepatocellular carcinoma (HCC): Phase III LEAP-012 study. Ann. Oncol. 35, S1229 (2024).
Yang, D. L. et al. Multicenter, retrospective GUIDANCE001 study comparing transarterial chemoembolization with or without tyrosine kinase and immune checkpoint inhibitors as conversion therapy to treat unresectable hepatocellular carcinoma: Survival benefit in intermediate or advanced, but not early, stages. Hepatology 82, 357–369 (2025).
World Medical Association Declaration of Helsinki ethical principles for medical research involving human subjects. Jama 310, 2191–2194 (2013).
Yang, D. L. et al. Survival benefit of hepatectomy after complete or partial response to conversion therapy in unresectable hepatocellular carcinoma (GUIDANCE003): a multicenter study. Liver Cancer 14, 687–703 (2025).
Yang, D. L. et al. Prognostic value of radiological and pathological complete response following immune-based conversion therapy in patients with unresectable hepatocellular carcinoma (GUIDANCE004). JHEP Rep. 7, 101587 (2025).
Eisenhauer, E. A. et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur. J. Cancer 45, 228–247 (2009).
Zhou, J. et al. Guidelines for diagnosis and treatment of primary liver cancer in China (2017 Edition). Liver Cancer 7, 235–260 (2018).
Huang, C. et al. Organ specific responses to first-line lenvatinib plus anti-PD-1 antibodies in patients with unresectable hepatocellular carcinoma: a retrospective analysis. Biomark. Res. 9, 19 (2021).
Sun, H. C. et al. Chinese expert consensus on conversion therapy for hepatocellular carcinoma (2021 edition). Hepatobil. Surg. Nutr. 11, 227–252 (2022).
Llovet, J. M. & Lencioni, R. mRECIST for HCC: performance and novel refinements. J. Hepatol. 72, 288–306 (2020).
Pan, L. X. et al. Entecavir versus tenofovir for prevention of hepatitis B virus-associated hepatocellular carcinoma after curative resection: study protocol for a randomized, open-label trial. Trials 25, 25 (2024).
Akahoshi, K. et al. Oncological resectability criteria for hepatocellular carcinoma in the era of novel systemic therapies: The Japan Liver Cancer Association and Japanese Society of Hepato-Biliary-Pancreatic Surgery Expert Consensus Statement 2023. Liver Cancer 13, 0–10 (2024).
Tsilimigras, D. I. et al. Overall tumor burden dictates outcomes for patients undergoing resection of multinodular hepatocellular carcinoma beyond the Milan criteria. Ann. Surg. 272, 574–581 (2020).
Li, L. et al. Adjuvant immune checkpoint inhibitors associated with higher recurrence-free survival in postoperative hepatocellular carcinoma (PREVENT): a prospective, multicentric cohort study. J. Gastroenterol. 58, 1043–1054 (2023).
Li, L. et al. Adjuvant therapy for hepatocellular carcinoma after curative treatment: several unanswered questions. J. Clin. Transl. Hepatol. 12, 525–533 (2024).
Zhong, J. H. et al. Repeat hepatic resection versus radiofrequency ablation for recurrent hepatocellular carcinoma: retrospective multicentre study. Br. J. Surg. 109, 71–78 (2021).
Dindo, D., Demartines, N. & Clavien, P. A. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann. Surg. 240, 205–213 (2004).
Balzan, S. et al. The “50-50 criteria” on postoperative day 5: an accurate predictor of liver failure and death after hepatectomy. Ann. Surg. 242, 824–828 (2005).
Austin, P. C. Balance diagnostics for comparing the distribution of baseline covariates between treatment groups in propensity-score matched samples. Stat. Med. 28, 3083–3107 (2009).
Scheiner, B. et al. Outcome and management of patients with hepatocellular carcinoma who achieved a complete response to immunotherapy-based systemic therapy. Hepatology 81, 1714–1727 (2025).
Kudo, M. et al. Transarterial chemoembolisation combined with lenvatinib plus pembrolizumab versus dual placebo for unresectable, non-metastatic hepatocellular carcinoma (LEAP-012): a multicentre, randomised, double-blind, phase 3 study. Lancet 405, 203–215 (2025).
Sangro, B. et al. Durvalumab with or without bevacizumab with transarterial chemoembolisation in hepatocellular carcinoma (EMERALD-1): a multiregional, randomised, double-blind, placebo-controlled, phase 3 study. Lancet 405, 216–232 (2025).
Wei, X. et al. Neoadjuvant three-dimensional conformal radiotherapy for resectable hepatocellular carcinoma with portal vein tumor thrombus: a randomized, open-label, multicenter controlled study. J. Clin. Oncol. 37, 2141–2151 (2019).
Zhu, X. D. et al. Hepatectomy after conversion therapy using tyrosine kinase inhibitors plus anti-PD-1 antibody therapy for patients with unresectable hepatocellular carcinoma. Ann. Surg. Oncol. 30, 2782–2790 (2023).
Zhang, X. P. et al. An Eastern Hepatobiliary Surgery Hospital/portal vein tumor thrombus scoring system as an aid to decision making on hepatectomy for hepatocellular carcinoma patients with portal vein tumor thrombus: a multicenter study. Hepatology 69, 2076–2090 (2019).
Sasaki, A. et al. Preoperative transcatheter arterial chemoembolization reduces long-term survival rate after hepatic resection for resectable hepatocellular carcinoma. Eur. J. Surg. Oncol. 32, 773–779 (2006).
Li, C. et al. Preoperative transcatheter arterial chemoembolization for surgical resection of huge hepatocellular carcinoma (≥10 cm): a multicenter propensity matching analysis. Hepatol. Int 13, 736–747 (2019).
Badwei, N. HCC case management-related challenges: think outside the box!. iLiver 2, 143–145 (2023).
Xu, J., Gu, L. & Yang, T. Optimising postoperative surveillance for hepatocellular carcinoma: beyond histological predictors. iLiver 3, 100110 (2024).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (82260569), the Guangxi Key Research and Development Plan (GuiKe AB24010082, GuiKe AB25069099), and the First-class Discipline Innovation-driven Talent Program of Guangxi Medical University, and the Guangxi Undergraduate Training Program for Innovation and Entrepreneurship (202410598051).
Author information
Authors and Affiliations
Consortia
Contributions
Da-Long Yang, Chuang Qin: contributed equally to this work; Da-Long Yang and Jian-Hong Zhong: conceptualization; Da-Long Yang, Chuang Qin, Ning Peng, Shao-Ping Liu, Ya-Qun Yu, Jun-Liang Nong, Fan-Jian Zeng, Ze Su, Yong-Yu Yang, Fu-Quan Yang, Mian-Jing Li, Ming-Song Wu, Xue-Yao Wang, Yong-Rong Liang, Fu-Xin Li, Shu-Chang Chen, Yong-Cheng Lai, Qing-Qing Pang, Teng-Meng Zhong, Yi-He Yan: methodology; Da-Long Yang, Fan-Jian Zeng, Shao-Ping Liu, Xue-Yao Wang, Hong-Bing Yao, and Chuang Qin: formal analysis; Da-Long Yang, Teng-Meng Zhong, Qing-Qing Pang, Min Luo, Wen-Feng Li, Kang Chen, Fu-Quan Yang, Yong-Cheng Lai, Ming-Song Wu, Yong-Yu Yang, Ning Peng, Jun-Liang Nong, Ze Su, Lin Ye, Fan-Jian Zeng, Shao-Ping Liu, Xue-Yao Wang, Chuang Qin, Mian-Jing Li, Yong-Rong Liang, Fu-Xin Li, Shu-Chang Chen: investigation and resources; Liang Ma, Da-Long Yang, and Jian-Hong Zhong: writing, review, original draft preparation, and editing; Da-Long Yang, Jian-Hong Zhong, Jun-Jie Ou 16, Yao-Zhi Chen, Guo-Dong Wang, Kang Chen, Zhu-Jian Deng, Jia-Yong Su, Guo-Gao Qiu, Zhi-Dong Liu, Jin-Min Wang, Yi Zhang, Xiu-Mei Liang, Bang-De Xiang, Liang Ma, Jian-Hong Zhong: visualization; Jian-Hong Zhong: supervision; Da-Long Yang and Jian-Hong Zhong: validation and project administration; All authors have read and agreed to the published version of this manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Consent to participate
This study was exempted by the Ethics Committee of Guangxi Medical University Cancer Hospital from the requirement of written informed consent.
Peer review
Peer review information
Communications Medicine thanks Luigi Lupo and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Yang, DL., Qin, C., Peng, N. et al. Neoadjuvant transarterial chemoembolization with tyrosine kinase and immune checkpoint inhibitors improves survival from resectable hepatocellular carcinoma in a multicenter, retrospective study (GUIDANCE002). Commun Med 6, 139 (2026). https://doi.org/10.1038/s43856-025-01303-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s43856-025-01303-w