Abstract
Background
Kidney volume, reflecting cumulative effects of many cysts, is an important prognostic biomarker for autosomal dominant polycystic kidney disease (ADPKD) but fails in many patients. Tracking individual cysts may more directly assess disease progression.
Methods
Individual cysts (n = 299) from 37 subjects were evaluated retrospectively over ≥ 8 years by serial MRI (mean follow-up = 11 years). Cysts were labeled on every available MRI scan, totaling 1654 contours (median timepoints per cyst = 5). Effects of cyst location, morphology, and growth pattern on kidney function decline were evaluated by univariate and multivariate analyses.
Results
Simple, T2-bright cysts follow logistic growth (median cyst growth rate = 11%/year). A subset (94/222, 42%) transitions over time to shrinking, to complex solid-fluid/fluid-fluid cysts, then to homogeneously T1-bright cysts and finally disappearing. By contrast, T1-bright complex cysts have no volume change (median cyst growth rate = 0%/year; p < 0.001). On multivariate analysis, faster kidney function decline is associated with simple cyst diameter > 2 cm on index scan (p = 0.007) and simple cyst transitions (p = 0.02). There is a trend towards faster kidney function decline with higher simple cyst growth rate (p = 0.16).
Conclusions
Profiling individual cysts on serial MRI to identify transitions as well as size and growth rate may improve predictions of ADPKD progression and treatment response.
Plain language summary
Polycystic kidney disease causes the growth of many fluid-filled sacs called cysts in the kidneys. Cysts can enlarge the kidneys and lead to kidney failure, requiring dialysis and/or kidney transplantation. It is important to be able to determine whether the disease is progressing so that patients may be treated appropriately. In many patients, doctors keep track of the disease by measuring kidney size using special images known as MRI scans. This doesn’t work for all patients. In our study, we analyzed MRI scans from different patients to see if we could use information from individual cysts, instead of measuring kidney size overall, to determine how the disease is advancing. We learned that specific changes in individual cysts may relate to kidney function. This could provide another tool for tracking disease progression in patients with polycystic kidney disease.
Similar content being viewed by others
Introduction
Autosomal dominant polycystic kidney disease (ADPKD) is the most prevalent inherited renal disorder, affecting 12 million people worldwide. It is characterized by renal and extrarenal manifestations, including end-stage kidney disease1. Total kidney volume (TKV) that reflects cumulative effect of many cysts is an established biomarker of risk of progressive chronic kidney disease and a predictor of therapeutic response to treatment with tolvaptan in ADPKD patients2,3,4. Although by combining TKV, height, and age, a risk classification (Mayo Image Classification, MIC) can be assigned for an individual patient, this approach fails in about 20–40% patients with typical ADPKD and cannot be applied to patients with atypical ADPKD2,5,6. With the typical ADPKD imaging phenotype, TKV is especially limited in early disease when the cyst volume is within the noise of TKV measurements. Therefore, there is a need for early prognostic markers to inform long-term decisions for ADPKD patients7,8. Gregory et al. found that total cyst number and total cyst parenchyma surface area more accurately predicted estimated glomerular filtration rate (eGFR) decline, yet this method was not widely adopted9,10. Since individual cysts are known to have a wide range of MRI appearances and a variable growth trajectory, tracking individual cysts could provide information about cyst growth and morphology directly impacting kidney function and thus might further improve accuracy of predicting eGFR decline.
Most renal cysts in ADPKD are simple cysts, which, by MRI, are homogeneously T2-bright and T1-dark, and typically fluid-filled with an imperceptibly thin rim. However, a subset of renal cysts in ADPKD are complex and appear on MRI with a darker T2 signal, brighter T1 signal and, occasionally, an increased signal on diffusion weighted imaging (DWI). These complex cysts are presumed to be filled with proteinaceous or hemorrhagic material, accounting for their increased T1 relaxation, and, compared with simple cysts, are associated with a more rapid decline in eGFR11,12.
Complex kidney cysts are often found incidentally during abdominal imaging but are generally evaluated in the context of infection or suspected neoplasm13,14. Despite the nearly universal occurrence of complex cysts in ADPKD, the incidence of cyst infection is rare, estimated to be 0.01 episodes per subject per year, usually as a complication of urinary tract infection15,16. Although papillary renal cell carcinoma is strongly associated with severe chronic kidney disease (stage 4 or worse) of any cause, a specific association with ADPKD, independent of CKD stage, has not been established17.
The natural history of individual kidney cysts in ADPKD, apart from infection and neoplasm, has not been explored to the best of our knowledge. T1-bright and DWI-bright cysts are commonly dismissed as clinically insignificant findings18. In this study, we measure cyst size, appearance, rate of growth and shrinkage by tracking individual simple and complex cysts by serial MRI over eight to 20 years follow-up. Our data show simple, T2-bright cysts follow logistic growth with a subset transitioning over time to shrinking or to complex solid-fluid/fluid-fluid cysts, then to homogeneously T1-bright cysts and finally disappearing. Faster kidney function decline occurs with simple cyst diameter > 2 cm on index scans and simple cyst transitions. Thus, tracking individual cysts on serial MRI to identify transitions as well as size and growth rate may improve predictions of ADPKD progression and treatment response.
Methods
Subjects
This retrospective study of existing medical images of ADPKD subjects from the Rogosin PKD Data Repository (clinicaltrials.gov #NCT00792155) was conducted in accordance with the Declaration of Helsinki and was HIPAA-compliant and Weill Cornell Medicine institutional review board approved. All subjects provided written informed consent. The inclusion criteria for the study were: ADPKD diagnosis based upon Pei-Ravine criteria19; ≥ 4 abdominal MRI scans; ≥ 8-year interval between the first and last MRI.
The exclusion criteria were: treatments modifying kidney disease, such as aspiration, cyst fenestration or other kidney surgery or interventional procedures, kidney transplantation, dialysis, medication treatment to reduce kidney volume (e.g., tolvaptan, somatostatin analogs); unavailable T1-weighted images; or inability to provide informed consent.
Demographic and laboratory data
Information regarding demographic and laboratory values at the time of the first scan, and all available organ volumes and serum creatinine values were extracted from the electronic medical record. eGFR was calculated by the 2021 EPI-CKD equation20. The rate of eGFR decline was calculated by least squares linear fitting of eGFR vs. age, where the slope of the fit was utilized as the rate of eGFR decline. Images were obtained from the Picture Archival Computer System (PACS).
Image Analysis
Cyst Segmentation
One observer (ZH) labeled T2-bright cysts on each of the subject’s initial MRI scans (index scan). Three T2-bright cysts were labeled on left kidney and three were labeled on the right kidney, totaling six T2-bright cysts per subject. These cysts represented the largest lesions on the index scan and were clearly separated from the collecting system, liver, and bowel. They lacked septations and were visible on more than two contiguous slices, allowing confident longitudinal tracking. These cysts were then tracked on subsequent follow-up MRI scans, where observers located each cyst based on its relative location to other cysts in the kidney, and to other organs (Supplemental Fig. 1). Complex, T1-bright cysts (up to 4 per subject if present) were labeled in addition to the T2-bright cysts, Supplemental Table 1. Similarly, all DWI-bright cysts (ADC < 2 ⋅ 10-3 mm2/s) were segmented on high B value images, beginning with the first scan on which DWI was acquired.
Cyst volume
Individual cyst volume was calculated by multiplying the number of labeled pixels, pixel area, and slice spacing.
Cyst circularity
Circularity for each labeled cyst was calculated based on their two-dimensional projections onto the axial plane using Eq. 1,
where area A is the area and P is the perimeter of the cyst projection.
Apparent diffusion coefficient (ADC) of DWI-bright complex cysts
All DWI-bright complex cysts were first segmented on DWI images (high B value). The median ADC values were calculated based on cyst segmentations applied to the ADC maps generated from exponential fitting of all available diffusion images.
Cyst Classification
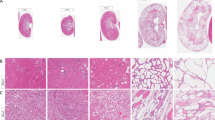
Based upon our experience reviewing more than 1000 abdominal MRI scans in over 400 ADPKD subjects, renal cysts were grouped into 5 categories (Fig. 1) as follows:
-
1.
Simple homogeneously T2-bright, T1-dark cysts with imperceptible rims (Fig. 1A)
-
2.
Complex cysts with T1-bright signal were subcategorized:
-
a.
T1-bright solid within a simple T2-bright, T1-dark cyst (Fig. 1B),
-
b.
Fluid-fluid level where T1-bright fluid separates from T2-bright, T1-dark simple cyst fluid at level (Fig. 1C),
-
c.
Homogeneously T1-bright cyst (Fig. 1D)
-
d.
Diffusion restricted cysts that were DWI-bright.
Each selected cyst (blue arrow) was labeled (pink for simple fluid and blue for T1-bright fluid or DWI-bright). Labeling was performed on both T1-weighted images (left column) and T2-weighted images (2nd column). Image insets show the selected cyst without the color label. A Simple cyst T1-dark, T2-bright. B Complex cyst with solid and fluid. C Complex cyst with fluid-fluid level. D Homogeneously T1-bright, T2-dark complex cyst.
Cysts were also classified based on location (left vs. right, interstitial vs. exophytic), where cysts with more than 50% of their volume outside of an imaginary renal boundary were designated exophytic cysts.
Organ volumes, total cyst volumes, TKV growth rate, cyst volume growth rate and MIC
Right and left kidney volumes were obtained by model-assisted contouring using a previously reported deep learning model and averaging the volumes obtained from all T1, T2 and SSFP sequences acquired with each MRI exam to reduce measurement variability21. TKV was calculated as the sum of right and left kidney volumes, and height-adjusted TKV (htTKV) was calculated as TKV divided by height. Based on a single htTKV measurement and age, MIC was calculated for the first and the last available MRI2. Kidney growth rate was calculated utilizing all available htTKV measurements paired with age using 2-parameter exponential fitting5. Total cyst volumes were calculated by multiplying cyst fractions indicated in radiology reports and TKV, and cyst volume growth rate was calculated using 2-parameter exponential fitting.
Cyst evolution
Fitting simple cyst volume evolution to growth/shrink model
Cyst volumes and ages for all available scans were fitted to a 2-parameter exponential model5 and a logistic model (Equation 3). To compare the goodness of fit, \(R^2\) for both methods was calculated. Growth rate measurement error was estimated to be 1%. Accordingly, cysts with growth rates less than −1%/year were categorized as shrinking.
Transitions
Each transition of a cyst from one category to another category was recorded between every two consecutive scans out of all available scans. The time between each event was calculated as the difference between the earliest scan date showing the cyst in the new category and the earliest scan date showing the cyst in the previous category.
Statistics and Reproducibility
Cyst characteristics
For each cyst characteristic (location, circularity, volume, occurrence of transitions, and individual cyst growth rate), ratios were calculated for categorical variables; means and standard deviations for normally distributed continuous variables; and medians with interquartile ranges for non-normally distributed continuous variables. Differences among subcategories were evaluated using Fisher’s exact test for categorical variables, Wilcoxon–Mann–Whitney U-tests for non-normally distributed continuous variables, and t-tests for normally distributed continuous variables. At the subject level, continuous parameters were aggregated as means, and categorical parameters as the proportion of cysts in each category (sum of the category divided by the total number of cysts tracked per subject). These aggregated cyst characteristics were then compared with subject-level variables, such as sex, MIC, kidney growth rate, and eGFR decline rate.
To assess reproducibility in cyst segmentation, a second observer (CL) independently labeled twelve cysts (the largest cyst in left and right kidney for six subjects (totaling 12 individual cysts tracked for all available scans) to calculate interobserver variability and Dice similarity coefficient.
Cyst location (exophytic, interstitial) and classification were determined independently by two observers experienced in analyzing ADPKD images for each time point (ZH, CL). For any disagreements, consensus was reached via discussion with a board-certified radiologist with 30+ years of experience imaging ADPKD (MRP).
Predicting kidney growth rates
Multivariate linear models relating cyst characteristics and the rate of eGFR decline were created. Cyst characteristics and demographic data were selected as parameters based on the univariate analysis (p < 0.05) and were aggregated to subject level to avoid clustering effects and overfitting. Per each subject, continuous parameters were aggregated based on the mean, and categorical parameters were aggregated based on the sum of a category divided by total number of cysts tracked per subject. The model with low Akaike Information Criterion value as well as low root mean squared error according to 5-fold cross validation was selected, where coefficient, standard error and 95% confidence interval, variance inflation factor (VIF) were calculated for each cyst characteristic parameter.
Results
ADPKD subjects (n = 37) meeting the inclusion/exclusion criteria, Table 1 and Supplemental Fig. 2, had a median htTKV of 463 mL/m, eGFR of 102 mL/min/1.73 m², and mean age of 40 years at the time of index scan. Nineteen (51%) of all subjects were determined to be at high risk of rapid progression based upon their Mayo Image Classification (MIC). These subjects had a median of 5 scans over an average of 11 years of imaging follow-up. Subjects meeting the inclusion/exclusion criteria had a higher eGFR at index scan, younger age, and lower htTKV compared to subjects from the Repository that did not meet the inclusion/exclusion criteria (Supplemental Table 2), reflecting the requirement for long term (> 8 years) imaging follow-up without intervention. We tracked 299 individual cysts including 222 simple T2-bright cysts (six simple cysts per subject) and 77 complex cysts identified from the first (index) MRI scan. This included 66 T1-bright complex cysts (see Supplemental Table 1 for number of T1-bright complex cysts tracked per patient) and 11 DWI-bright (complex) cysts. Together with 13 cysts that transitioned from T2-bright simple cysts on index MRI to T1-bright complex cysts on later scans, a total of 90 complex cysts were tracked and analyzed. For the 299 cysts, 1654 cyst contours were obtained from 203 MRI exams of 37 ADPKD subjects.
Interobserver variability
For the largest cyst on each kidney for six randomly selected subjects (n = 12 in total) the intraclass correlation coefficient (ICC) for largest cyst volume was 0.99 (p < 0.001). The mean Dice similarity coefficient was 0.93, 95% confidence interval [0.92, 0.94] comparing the largest cyst label between the two observers. For these 12 largest cysts, the two observers had 100% agreement in identifying cysts transitions.
For all 299 cysts, the cyst location was assessed by the same two observers for every cyst. There was 98% agreement (293 out of 299 cysts). All disagreements were resolved by consensus for further analysis.
Simple Cysts
Table 2 shows simple cysts (n = 222) individual cyst characteristics including cyst location and subject characteristics vary by cyst size, and growth rate, and Table 3 shows simple cyst characteristics aggregated per subject vary by sex and MIC.
Simple Cysts Follow Logistic Growth and Shrinkage
We observed that simple cysts grow fastest initially when they are relatively small, and their growth rate slows as they become larger (Fig. 2) following a logistic model (Eq. 1):
where V(t) is the volume of an individual cyst as a function of time (t), Vmax is the predicted maximum volume that cyst can reach in its lifetime based upon local constraints, rcyst is the rate of growth, and \({t}_{0}\) is the age of the subject when cyst volume reached Vmax/2. The logistic model was found to be a superior fit for cyst growth (median R2 = 0.88 [IQR 0.72, 0.98]) compared to the exponential model (median R2 = 0.78 [IQR 0.55, 0.94], p < 0.001).
A T2-weighted MRI shows a right upper pole simple cyst (pink) of a 33-year-old male ADPKD subject in 2011 measuring 3 mL. B This cyst reached Vmax = 54 mL in 2022. C All available scans, show the cyst in (A) and (B) followed logistic growth (rcyst = 88% per year, t0 = 2016, R2 = 0.99). D T2-weighted MRI shows a right lower pole simple cyst (pink) of a 62-year-old male ADPKD subject in 2014 measuring 5 mL. E This cyst reached Vmax = 75 mL in 2023. F All available scans show the cyst in (D) and (E) followed logistic growth (rcyst = 56% per year, t0 = 2017, R2 = 0.99).
Among the 222 simple cysts tracked in 37 subjects, the median annual individual simple cyst growth rate was 11%, IQR: [0%, 29%], calculated using the logistic model (Table 2). Simple cysts were dichotomized based on the median annual simple cyst growth rate: simple cysts with rapid growth (\({r}_{{\mathrm{cyst}}}\) > 10%/year) and simple cysts with slow growth (\({r}_{{\mathrm{cyst}}}\) < 10%/year). Simple cysts with rapid growth (8% shrinking) were found to be less likely to transition to shrinking compared to cysts with slow growth (50% shrinking, p < 0.001).
Effect of simple cyst size
The median initial diameter of all simple cysts tracked was 2 cm [IQR 1.7, 3.8] (median volume = 8 mL [IQR 3, 28]; Table 2). Accordingly, simple cysts were dichotomized into large simple cysts (d > 2 cm) and small simple cysts (d < 2 cm) on the index scan. It was more likely for cysts with d < 2 cm to eventually disappear than for cysts with d > 2 cm (disappearance rate = 26% vs 11% for initially small and initially large simple cyst respectively, p = 0.004). As expected with logistic growth, rapidly growing simple cysts (growth rate > 10%/year) were smaller in size on the index scan (median cyst diameter = 22 mm [IQR 15, 33]) compared to simple cysts with growth rate <10% (median cyst diameter = 30 mm [IQR 20, 44], p < 0.001). Simple cysts with diameter greater than 2 cm were more likely to be exophytic than interstitial (p = 0.02). Nevertheless, exophytic cysts and interstitial cysts had similar individual cyst growth rate (interstitial: median 11%/year [IQR 0, 26], exophytic: median 11%/year [IQR 0, 34], p = 0.91).
Effect of sex on simple cysts
The overall annual kidney growth rate in male subjects was higher than in female subjects (mean 5.4 ± 2.5% vs. 3.1 ± 1.3%, p = 0.002; Table 3). However, aggregated individual simple cyst growth rates did not differ significantly by sex. This may reflect a higher cyst fraction in males since male subjects had larger kidney cysts (mean aggregated diameter, \(\bar{d}\) = 3.3 cm for male subjects vs. \(\bar{d}\) = 2.5 cm females subjects, p = 0.002).
Effect of Mayo Image Classification on simple cysts
Cysts were characterized based on the MIC of subjects on their index scan into two groups: rapid progressors (MIC 1C, 1D, 1E) and mild progressors (MIC 1 A, 1B). The size of cysts in rapid progressors on index scan (median diameter = 3.5 cm [IQR 2.7, 4.2]) was larger than the size of cysts in mild progressors (2.3 cm [IQR 1.8, 2.4], p < 0.001). Moreover, there was a trend toward more transition-free cysts for mild progressors (aggregated % of cysts without transitioning = 65% ± 21%) than for rapid progressors (50 ± 27%, p = 0.07).
Multivariate Analysis Relating Simple Cyst Characteristics and Demographics to Rate of eGFR Decline
Based on the univariate analysis, multivariate linear models that relate aggregated cyst characteristics per subject (n = 37) to the rate of eGFR decline were constructed. The model with the best fit (Supplemental Table 3) indicated that given an individual cyst, presence of transitions was associated with more rapid eGFR decline (p = 0.007). Similarly, a cyst diameter greater than 2 cm was also associated with more rapid eGFR decline (p = 0.02). There was a trend suggesting that higher individual simple cyst growth rate was related to more rapid eGFR decline (p = 0.16).
Although on univariate analysis, rapid progression (MIC 1 C, 1D, 1E) vs. mild progression (MIC 1 A, 1B) had a significant difference in the rate of eGFR decline, this classification lost significance in aggregated multivariate models shown in Supplemental Table 3 including cyst diameter, individual simple cyst growth rate and cyst transitions. Height-adjusted TKV on index scan, TKV growth rate, height-adjusted total cyst volume on index scan, and total cyst volume growth rate were not statistically significant at predicting rate of eGFR decline compared to aggregated cyst diameter category and aggregated cyst no transition rate in the multivariate analysis (Supplemental Table 3).
Complex Cysts
We followed 77 complex cysts on index scan plus 13 cysts transitioned from T2-bright simple cysts for a total of complex cysts including 59 homogeneously T1-bright cysts, 10 solid-fluid cysts, 10 fluid-fluid cysts, and 11 DWI-bright cysts (median ADC = 0.9 · 10-3 mm2/s [IQR 0.7, 1.4]). Their features, including cyst size, circularity, location, and transition rates are shown in Supplemental Table 4. Of the 20 fluid-fluid and solid-fluid cysts tracked, 10 (50%) were interstitial and 10 (50%) were exophytic. By contrast, 68% of homogeneously T1-bright cysts (40 of 59; p = 0.02) and 100% of DWI-bright cysts (11 of 11; p = 0.005) were interstitial.
Since complex cysts frequently underwent transitions and disappeared, individual complex cyst growth rate could only be calculated for 21 homogeneously T1-bright cysts with sufficient imaging timepoints as homogeneously T1-bright cyst. These complex renal cysts had a median growth rate of 0.0% ± 0.1% per year, significantly less than simple cysts (median cyst growth rate = 8%/year, p < 0.001).
Cyst Evolution
DWI-bright cysts (n = 11) had a median follow-up of 3 years since DWI was not included in the MRI protocol until 2017. Three (27%) DWI-bright cysts disappeared during this follow-up interval. Excluding those 11 DWI-bright cysts, the remaining 288 had 134 (47%) cysts that underwent transitions during the 8-to-20-year follow-up period. Fifteen cysts underwent two transitions, and four cysts underwent three transitions for a total of 157 transitions (Table 4). Among the 222 T2-bright simple cysts, 65 (29%) decreased in size (Shrink), 36 of 222 (16%) resolved completely (Disappearance), 6 of 222 (3%) developed a complex solid-fluid or fluid-fluid interface, and 7 of 222 (3%) developed a homogeneously T1-bright appearance (see Fig. 3 and Supplemental Fig. 3 for examples).
A Simple cyst (pink) of a female ADPKD subject, 56-year-old at index scan in 2006. B The cyst eventually transitioned into homogeneously T1-bright complex cyst (cyan) in 2024. C All available scans show the cyst was first growing at rcyst = 5% per year (R2 = 0.83), transitioned in 2012 to shrinking (rcyst = −10% per year, R2 = 0.90), eventually further transitioning to homogeneously T1-bright complex cyst.
Complex cysts always transitioned to another complex cyst type or to disappearance, never reverting back to a simple cyst state (see Supplemental Fig. 3 for examples of complex cysts transitioning). Of the complex cysts with a solid-fluid or fluid-fluid level found at the initial scan, 11 of 20 (55%) transitioned to a homogeneously T1-bright appearance and 4 of 20 (20%) transitioned to disappearance. Homogeneously T1-bright cysts only transitioned to disappearance (34 of 69, 49%).
Discussion
This study tracked 299 individual kidney cysts in 37 ADPKD subjects over an average of 11 years by serial MRI to investigate the natural history of simple and complex cysts. These data support a model (Fig. 4) with logistic growth of simple cysts, and transitions to rupture, hemorrhage and/or disappearance in nearly half of these cysts. The initial transition appears to be a break in the simple cyst wall leading to leaking, shrinking and eventual disappearance or hemorrhage with T1-bright internal contents. These complex T1-bright solid-fluid or fluid-fluid level cysts then transition to homogeneously T1-bright cysts that sometimes disappear. Based on this model, individual cyst diameter, growth rate, and transitions may help predict the rate of eGFR decline.
ADPKD simple renal cysts after transitioning from logistic growth to shrinking were observed to follow two pathways: 1. Disappear; 2. Hemorrhage with fluid-fluid level or fluid with blood clots (T1-bright solids), which leads to further shrinking, homogeneously T1-bright appearance, and eventually disappearing. Black = T1-bright; Blue = T2-bright.
The exponential growth model is commonly used to estimate ADPKD kidney growth2,5,22. It assumes that there is no limit to cyst or kidney volumes. However, for an individual cyst, there are neighboring cysts, parenchyma of varying density and interstitial pressure, and adjacent organs that can constrain cyst growth as the cyst enlarges. These factors limit the assumption of exponential growth. Presumably, individual cysts reach a maximum volume related to local constraints. Thus, the logistic growth model more accurately describes our observations of individual simple cyst growth trajectories. In the growth phase, cysts increase from a negligible volume toward a maximum volume, Vmax, restricted by several factors that are incompletely defined. In the shrink phase, there is net fluid loss from cysts, either due to leakage or secretion of fluid from the cyst, or a decrease or cessation of fluid secretion into it. Consequently, once shrinking begins, cyst volumes decrease asymptotically from Vmax to zero.
A subset of individual simple cysts transitioned to shrinking, disappearance or complex T1-bright cysts with minimal further growth. These transitions were associated with more rapid eGFR decline even though complex cysts were not growing. This is consistent with prior observations of the number of complex, T1-bright cysts which may approximate the number of transitions being associated with an increased rate of eGFR decline11. This suggests that transitions contribute to renal parenchymal injury in ADPKD or are related to loss of nephrons.
We found that relatively large cysts, defined by a diameter > 2 cm at index scan, were associated with more rapid eGFR decline. This finding is consistent with Gregory et al. reporting that high total cyst parenchyma surface area, which is directly related to cyst diameter, was associated with more rapid eGFR decline10. A composite phenotype that includes cyst diameter at index scan, cyst transitions and cyst growth rate may be useful for predicting ADPKD progression. The creation of this composite phenotype may also lead to new directions in developing ADPKD treatments as well as evaluating therapeutic outcomes. There have been many efforts focusing on shrinking cyst size and lowering cyst growth rate23,24,25,26,27. However, as suggested by the strong association of cyst transitions with rapid kidney function decline, future therapy design may also focus on preventing cyst transitions.
We also observed that exophytic cysts encountered fewer transitions compared to interstitial cysts, which may explain Bae et al. finding that exophytic cysts affect kidney function to a less than interstitial cysts28. In our data, there was also a trend toward slower eGFR decline with exophytic cysts.
Strengths of this study include the relatively long duration of follow-up (8+ years) by MRI including T1- and T2-weighted pulse sequences at all timepoints. The population overall was well characterized, as it was derived from the Rogosin PKD Data Repository, a prospective study that includes biennial protocol MRI scans.
Limitations of this study include a relatively small cohort that reflects the relatively long duration (8+ years) of imaging follow-up. Since DWI-bright cysts were rare and DWI sequences were included in routine ADPKD MRI exams relatively late compared to T1 and T2 images, there was less long-term imaging follow-up of DWI-bright cysts. Consequently, these DWI-bright cysts were not as well characterized. Besides DWI, advanced MRI techniques including quantitative susceptibility mapping may further aid characterization of complex cysts29. There were no infected cysts or known cystic neoplasms included in this study, so those lesions were not evaluated. Due to the resolution limits of MRI images, the observers could not label cysts with diameter smaller than 1 cm. Thus, microcysts were not tracked which may limit the potential of this approach for predicting disease progression. Improving MRI resolution using methods such as super-resolution reconstruction may help to track smaller cysts and offer clearer explanations regarding the correlation of cyst transitions with eGFR decline30. For routine cyst tracking, deep learning methods may offer an opportunity to automate the process. There is also potential using total number of cysts, number of complex cysts or cysts with transition for predicting eGFR trajectory in ADPKD, which we plan to explore in future investigations.
In summary, this study shows the natural history of individual renal cysts in ADPKD on MRI. These simple cysts, initially following logistic growth, can transition to leaking or hemorrhaging, shrinking and disappearing. Cyst transitions, cyst diameter as well as cyst growth rate predict the rate of eGFR decline. Thus, individual cyst profiling improves our understanding of ADPKD progression and may lead to better prognostic ADPKD biomarkers.
Supplementary Materials: Supplemental Table 1. Number of trackable T1-bright complex cysts in 37 subjects with ADPKD; Supplemental Table 2. Demographical Information of Excluded Subjects; Supplemental Table 3. Multivariate linear model candidates for predicting rate of eGFR decline based on aggregated cyst characteristics and demographics; Supplemental Table 4. Complex cyst characteristics by cyst type; Supplemental Fig. 1. An example of tracking a simple, T2-bright ADPKD renal cyst. Supplemental Fig. 2. Flow chart. Supplemental Fig. 3. Examples of cyst transitions without significant cyst volume change. Supplementary Source Data.
Data availability
The source data for all tables is provided in Supplementary Source Data. Subject imaging data can be shared upon signing a Data Sharing Agreement with Weill Cornell Medicine.
References
Grantham, J. J. Clinical practice. Autosomal dominant polycystic kidney disease. N. Engl. J. Med 359, 1477–1485 (2008).
Irazabal, M. V. et al. Imaging classification of autosomal dominant polycystic kidney disease: a simple model for selecting patients for clinical trials. J. Am. Soc. Nephrol. 26, 160–172 (2015).
Grantham, J. J. et al. Volume progression in polycystic kidney disease. N. Engl. J. Med. 354, 2122–2130 (2006).
Yu, A. S. L. et al. Long-term trajectory of kidney function in autosomal-dominant polycystic kidney disease. Kidney Int 95, 1253–1261 (2019).
Hu, Z. et al. Improved predictions of total kidney volume growth rate in ADPKD using two-parameter least squares fitting. Sci. Rep. 14, 13794 (2024).
Borrego Utiel, F. J. & Espinosa Hernandez, M. How to estimate kidney growth in patients with autosomal dominant polycystic kidney disease. J. Am. Soc. Nephrol. 34, 944–950 (2023).
Lanktree, M. B., Kline, T. & Pei, Y. Assessing the risk of progression to kidney failure in patients with autosomal dominant polycystic kidney disease. Adv. Kidney Dis. Health 30, 407–416 (2023).
Cho, Y. et al. Establishing a core outcome set for autosomal dominant polycystic kidney disease: report of the standardized outcomes in nephrology-polycystic kidney disease (SONG-PKD) consensus workshop. Am. J. Kidney Dis. 77, 255–263 (2021).
Gregory, A. V. et al. Semantic instance segmentation of kidney cysts in MR images: a fully automated 3D approach developed through active learning. J. Digit Imaging 34, 773–787 (2021).
Gregory, A. V. et al. Utility of new image-derived biomarkers for autosomal dominant polycystic kidney disease prognosis using automated instance cyst segmentation. Kidney Int. 104, 334–342 (2023).
Riyahi, S. et al. Hemorrhagic cysts and other MR biomarkers for predicting renal dysfunction progression in autosomal dominant polycystic kidney disease. J. Magn. Reson Imaging 53, 564–576 (2021).
Suwabe, T. et al. Acute renal intracystic hemorrhage in patients with autosomal dominant polycystic kidney disease. J. Nephrol. 36, 999–1010 (2023).
Silverman, S. G. et al. Bosniak classification of cystic renal masses, version 2019: an update proposal and needs assessment. Radiology 292, 475–488 (2019).
Suwabe, T. et al. Clinical features of cyst infection and hemorrhage in ADPKD: new diagnostic criteria. Clin. Exp. Nephrol. 16, 892–902 (2012).
Sallee, M. et al. Cyst infections in patients with autosomal dominant polycystic kidney disease. Clin. J. Am. Soc. Nephrol. 4, 1183–1189 (2009).
Suwabe, T. Cyst infection in autosomal dominant polycystic kidney disease: our experience at Toranomon Hospital and future issues. Clin. Exp. Nephrol. 24, 748–761 (2020).
Zhang, W. et al. Papillary renal cell carcinoma with a somatic mutation in MET in a patient with autosomal dominant polycystic kidney disease. Cancer Genet 209, 11–20 (2016).
Kidney Disease: Improving Global Outcomes AWG. KDIGO 2025 clinical practice guideline for the evaluation, management, and treatment of autosomal dominant polycystic kidney disease (ADPKD). Kidney Int. 107, S1–S239 (2025).
Pei, Y. et al. Unified criteria for ultrasonographic diagnosis of ADPKD. J. Am. Soc. Nephrol. 20, 205–212 (2009).
Inker, L. A. et al. New creatinine- and cystatin C-based equations to estimate GFR without race. N. Engl. J. Med 385, 1737–1749 (2021).
He X. et al. Test retest reproducibility of organ volume measurements in ADPKD using 3D multimodality deep learning. Acad. Radiol. 31, 889–899 (2023).
Higashihara, E. et al. Age- and height-adjusted total kidney volume growth rate in autosomal dominant polycystic kidney diseases. Clin. Exp. Nephrol. 23, 100–111 (2019).
Shillingford, J. M., Piontek, K. B., Germino, G. G. & Weimbs, T. Rapamycin ameliorates PKD resulting from conditional inactivation of Pkd1. J. Am. Soc. Nephrol. 21, 489–497 (2010).
Lee, E. C. et al. Discovery and preclinical evaluation of anti-miR-17 oligonucleotide RGLS4326 for the treatment of polycystic kidney disease. Nat. Commun. 10, 4148 (2019).
Lakhia, R. et al. PKD1 and PKD2 mRNA cis-inhibition drives polycystic kidney disease progression. Nat. Commun. 13, 4765 (2022).
Cabrita, I. et al. Cyst growth in ADPKD is prevented by pharmacological and genetic inhibition of TMEM16A in vivo. Nat. Commun. 11, 4320 (2020).
Torres, V. E. et al. Tolvaptan in patients with autosomal dominant polycystic kidney disease. N. Engl. J. Med 367, 2407–2418 (2012).
Kim, Y., Bu, S., Tao, C., Bae, K. T. & Group, H. P. K. D. S. Deep learning-based automated imaging classification of ADPKD. Kidney Int Rep. 9, 1802–1809 (2024).
Schumacher, K. et al. Quantitative susceptibility mapping for detection of kidney stones, hemorrhage differentiation, and cyst classification in ADPKD. Abdom. Radio. 49, 2285–2295 (2024).
Safari M., Eidex Z., Chang C. W., Qiu R. L. J., Yang X. Advancing MRI reconstruction: a systematic review of deep learning and compressed sensing integration. ArXiv, (2025).
Acknowledgements
This work was supported by NIH NIDDK grant #1U54DK144866-01 and Shaw Family Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Dr. Blumenfeld reports research support for an unrelated project from Vertex. The other authors have no conflicts of interest to disclose.
Peer review
Peer review information
Communications Medicine thanks Anna Caroli and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Hu, Z., Li, C., Blumenfeld, J.D. et al. Natural history of simple and complex cysts in autosomal dominant polycystic kidney disease on MRI. Commun Med 6, 62 (2026). https://doi.org/10.1038/s43856-025-01318-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s43856-025-01318-3