Abstract
Background
Hepatitis C infection (HCV) is a leading cause of liver disease and mortality. Despite curative treatment options, HCV elimination remains elusive. Although the US has national screening and treatment recommendations, HCV remains under-screened and under-diagnosed. We utilized two national surveys to estimate trends in overall HCV care utilization and testing in the US.
Methods
Data from the National Ambulatory Medical Care Survey and the National Hospital Ambulatory Medical Care Survey from 2010 to 2019 were analyzed using inverse probability weighting to generate national estimates of visits and testing. Weighted chi-square and logistic regression analyses adjusted for demographics, payor type, and time assessed the primary outcome of ambulatory care utilization and the secondary outcome of office-based hepatitis C screening.
Results
Between 2010–2019, 23,469,344 HCV ambulatory visits are identified with higher overall adjusted visit rates for men (OR 1.54), people born 1945–1965 (OR 4.00), and insured by Medicare (OR 1.98) with increased utilization by White, privately insured patients in the office since 2016. HCV cases with Medicaid (OR 6.05) or have associated substance use disorder (SUD) (OR 3.30) are more likely to utilize the ER than office care. Screening rates are low in initial (2%) and overall (1%) primary care health visits.
Conclusions
In a nationally representative study, we find increasing rates of HCV visits, largely in White, privately insured patients seen in office. Low screening rates and disproportionate ER utilization among rural, racial/ethnic minorities, Medicaid insured, and patients with SUD highlight the importance of policy and practice guideline updates to improve identification and care linkage for HCV.
Plain language summary
Hepatitis C infection remains a leading cause of liver disease and mortality in the US despite the presence of a national screening recommendation and curative treatment options. Here, we utilized two national surveys to estimate trends in hepatitis C testing and care utilization in the US. We demonstrated that White, privately insured patients are more likely to obtain care in an office setting, whereas patients with Medicaid and those with substance use disorder are more likely to obtain care from an emergency room. It is critical to ensure that patients with hepatitis C who seek care from the emergency room receive adequate linkage to care and continuity of care as we work towards viral hepatitis elimination.
Similar content being viewed by others
Introduction
Hepatitis C virus (HCV) infection is among the leading causes of liver disease and mortality, with over 70 million people infected worldwide. The prevalence of patients with chronic hepatitis C (CHC) infection is between 1–2% in the United States, making it the most common bloodborne infection in the nation1,2. CHC is a major cause of mortality, and if untreated, patients with CHC can develop cirrhosis and hepatocellular carcinoma3,4.
The approval of direct-acting antivirals has revolutionized the treatment for patients with HCV, with estimated cure rates of 97–99%5. However, despite advances in medical therapy, HCV remains a leading cause of liver-related death and transplantation and is rising in incidence in younger individuals. One major factor in the persistent burden of CHC is under-screening, with studies estimating that less than half of those with CHC are aware of their positive status6. In order to be treated, the disease must be identified, which generally requires an infected individual to interface with the healthcare system. Most medical care occurs in the ambulatory or emergency setting, marking these as the most likely places for individuals with hepatitis C to seek care, and thus be screened and linked to care.
In 2012, the Centers for Disease Control (CDC) recommended one-time screening for HCV in all individuals born between 1945 and 1965, hereafter “baby boomers”, due to the high prevalence of CHC in this cohort in addition to risk factor based screening at any age7. Recently, studies found marked increases in HCV infection in those aged 19–39 as well as those with a pattern of injection drug use, coinciding with the ongoing opioid epidemic. In response to such trends, the CDC and United States Preventive Services Task Force in 2020 released revised recommendations suggesting that all adults receive one-time screening for HCV regardless of risk factors. These efforts are in accordance with national and international goal to eliminate HCV as a public health threat by 20308. Despite national efforts, significant racial, socioeconomic, and geographic disparities in HCV screening and treatment remain in the United States9,10.
One critical aspect driving disparities in the CHC diagnosis and care continuum is inadequate access to care. One study found that patients without a regular source of care are 19 times more likely to be unaware of their CHC status11. As we continue to strive towards HCV elimination goals set forth by the White House as well as the World Health Organization, it is critical to better understand care utilization, especially for patients with CHC. The goal of this study is to understand national demographic trends in hepatitis C care utilization during a critical era of change in the landscape of hepatitis C diagnosis and management.
To assess rates of utilization, we utilize two national databases to estimate overall rates of use among those with identified HCV in the United States and to identify disparities in access and utilization of HCV screening. We find that overall screening rates remain low in both initial and subsequent primary care office visits. Men, people born between 1945 and 1965, and insured by Medicare are more likely to have ambulatory visits associated with HCV. White, privately insured patients are more likely to obtain care in an office setting whereas those insured with Medicaid or have associated substance use disorder are more likely to obtain care from the emergency room rather than in the office setting. These findings highlight the need for policy and practice updates to improve patient access and linkage to care, particularly for those who are more likely to experience fragmented care.
Methods
Data sources
Data from the National Ambulatory Medical Care Survey (NAMCS) and the National Hospital Ambulatory Medical Care Survey (NHAMCS), two publicly available databases, were extracted. NAMCS tracks care utilization in the outpatient, or ambulatory, setting, while NHAMCS uses similar sampling techniques to track care visits in the emergency room setting. We utilized data between 2010–2019. Due to changes in the sampling technique, the NAMCS 2017 data remain unreleased and were therefore excluded. NAMCS/NHAMCS are publicly funded survey-based datasets that include annual, national probability samples of ambulatory care visits and are designed to estimate national utilization of ambulatory health services. The basic sampling unit is the patient visit, with the overall raw sample size varying by year according to the number of hospitals/physician offices sampled and the response rate. Only visits by adults at least 18 years old at time of visit were included.
Surveys include patient demographics, reason for visit, visit diagnoses, procedures performed, diagnostic tests ordered, and medications prescribed12. Medical coding and adjudication are provided by the National Center for Health Statistics. Weights are provided for each observation in the data sets to allow researchers to extrapolate and produce national estimates. A model-based single imputation is used to fill in missing data.
Data usage
Survey data were analyzed using inverse probability weighting for each sample visit or each physician response, as appropriate. Sample weights were adjusted for the nonresponse rate at certain times of year, by geographic region, urban/rural location, and ownership designations to create unbiased national estimates. Due to concern over bias from overweighting of individual samples, NAMCS/NHAMCS do not recommend making conclusions based on weighted data with fewer than 30 unweighted instances. To increase the number of unweighted instances, NAMCS/NHAMCS datasets were combined as performed in prior studies and as recommended in documentation provided by NAMCS/NHAMCS13,14. As the survey design can vary slightly by annual iteration, coded variables may change and were accounted for according to the accompanying public use documentation.
Identification of variables of interest
Study outcomes
The primary variable of interest in this study was a care visit for a patient with identified CHC during which HCV-specific care was provided. Presence of CHC was identified either according to International Classification of Diseases (ICD)-9-CM (2010–2015) or ICD-10-CM coding (2016–2019) in one of the 5 visit diagnoses, or by direct survey-level checkbox for current hepatitis C infection, available since 2016. Our secondary outcome measure was performance of a hepatitis C screening test in an ambulatory care setting (NAMCS), which was also available in survey checkbox format under “testing performed” for all years after 2013.
Additional variables of interest
To assess trends in utilization and testing, we collected demographic data, including sex, birth year, self-identified race/ethnicity, expected payment method for visit, and metropolitan statistical area (MSA) status. Of note, in the 2018 NAMCS survey, the total number of responses was considered too low to accurately weight based on region. Therefore, the region (Northeast, South, Midwest, West) variable was not published and is therefore unavailable for observations from 2018. Acceptance of private insurance as a payment type was not collected in 2010 for NAMCS. Concomitant presence of substance abuse or substance use disorder was identified using ICD-9/10 codes as well as survey direct identification through the “does the patient now have opioid abuse or dependence” questions. A complete list of variables of interest, including ICD codes utilized by year and dataset, can be found in Supplementary Table 1.
Primary outcome analysis
To assess rates of utilization among those with identified CHC, after extraction of variables of interest, descriptive statistics were computed using nationally weighted estimates with corresponding 95% confidence intervals (CIs). To account for the complex survey design, a single-unit certainty ultimate cluster method was used for variance estimation in strata containing only a single primary sampling unit (PSU). Differences in categorical variables were assessed using chi-square tests, implemented via the “svychisq” function in the R “survey” package.
Differences in characteristics associated with HCV-related visits were analyzed using both univariable and multivariable logistic regression models (adjusted for significant variables identified in univariable analysis), adjusting for the complex survey design. To assess temporal trends, data from two consecutive years were combined when necessary to ensure adequate raw sample sizes. Significant differences in utilization over 2-year periods were assessed by modeling time as a linear variable in weighted logistic regression. The total number of visits and visit rates were estimated by aggregating all available data from NAMCS and NHAMCS over the study period.
Secondary outcome analysis
To assess rates of hepatitis C testing among groups at risk for hepatitis C, we included only ambulatory visits considered “at risk” for HCV screening. Visits prior to 2014, when HCV testing data were not collected, as well as visits where the patient already had a diagnosis of hepatitis B or C, likely cirrhosis, or was actively pregnant, were excluded. To better describe the utilization of HCV testing in routine clinical settings, further analysis was conducted, excluding visits to surgical subspecialists, visits for acute problems, flares of chronic conditions, pre- or post-surgical management, or visits where the provider indicated they were not the patient’s primary care provider. Subgroup analyses were performed to evaluate testing patterns among individuals who would have been eligible for screening under the nationwide guidelines, which in 2012 were expanded to include all baby boomers, those with end-stage renal disease, and those with substance use disorder.
Finally, a more restrictive assessment of HCV screening rates was performed by including only visits in which the provider identified themselves as the patient’s primary care provider, the visit was for either routine chronic disease management or preventive care, and the patient was new to the provider. This strict definition was applied to reduce potential confounding from prior HCV testing, ensuring that only true screening visits were considered. However, the stringent inclusion criteria limited the number of raw visits available for analysis, restricting further subgroup analyses.
Statistics and reproducibility
All statistical analyses were conducted using R (Version 4.3.1 GUI 1.79 Big Sur ARM Build 8238). The “survey” package in R was used for weighting, variance estimation, chi-square testing, and logistic regression modeling to ensure accurate representation of national estimates. All statistical tests were two-sided, with p < 0.05 considered statistically significant.
Ethics statement
This research was a collaboration among investigators across expertise and backgrounds. Efforts were made to include both early-career and senior investigators; the investigators are committed to inclusion and the ethical conduct of research across their studies. No identifiable information is released in the public use data files by the National Center for Health Statistics. This study was determined to be exempt from IRB review under Weill Cornell Medicine Institutional Review Board policy because it uses publicly available, fully de-identified data.
Results
Utilization of HCV care
Between 2010 and 2019, a total of 9.7 billion care visits were identified. Of these, 23,469,344 visits (0.3%) were for patients with identified CHC, and 1.29 million visits were for patients with CHC and cirrhosis. A higher percentage of visits for those with already identified HCV occurred in the office than for the overall cohort without CHC (94.8% vs. 87.3%, p < 0.001). Across care locations, visits for patients with identified CHC were more common amongst men, baby boomers, people with private insurance, in the West region, and in MSAs, p < 0.001 for all. In contrast to office visits, ER visits were more frequent among individuals with CHC born after 1965, non-Hispanic Blacks, and insured by Medicaid (Table 1).
In weighted univariable logistic regression analysis, visit rates for CHC compared to no CHC were higher among men (OR 1.54, 95% CI [1.06, 2.22]), baby boomers (OR 4.00, 95% CI [1.57, 10.15]), and those insured by Medicare (OR 1.98, 95% CI [1.17, 3.35]). Younger age was inversely associated with HCV visits. Regional differences were also evident, with higher odds of HCV visits in the West region (OR 2.23, 95% CI [1.38, 3.60]), whereas living in a non-MSA was associated with reduced odds of HCV visits (OR 0.49, 95% CI [0.30, 0.80]) (Table 2). In weighted multivariable logistic regression analysis, odds of CHC visit were higher for male (OR 2.05, 95% CI [1.57, 2.69]), non-Hispanic Black (OR 1.71, 95% CI [1.19, 2.45]), baby boomer (OR 9.04, 95% CI [4.87, 16.82]), covered by Medicare (OR 2.19, 95% CI [1.50, 3.20]) or Medicaid (OR 2.48, 95% CI [1.71, 3.59]), and in the West region (OR 2.49, 95% CI [1.51, 4.12]) compared to those without CHC.
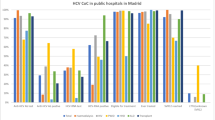
When comparing odds of care utilization for those with identified CHC by practice setting (ER vs. office), only Medicaid was significantly associated with ER usage (OR 6.05, 95% CI [2.88, 12.72]). The ER accounted for 16.5% of all CHC visits for those with Medicaid, compared to 2.1% for those with private insurance (Fig. 1). Among all visits to both clinician offices and ERs, non-Hispanic Black patients were more likely to have Medicaid as their primary payer type (29.7% vs. 10.5%, p < 0.001). This was particularly true for Black patients born after 1965, for whom Medicaid covered 44.8% of visits. Medicaid was also less likely to be accepted by primary care providers for new patient visits than Medicare or private insurance across all study periods (Fig. 2). In multivariable logistic regression analysis adjusted for age, sex, race/ethnicity, region, residing in an MSA, and care location across the entire study population to evaluate factors associated with Medicaid as the payment method for visits, non-Hispanic Black (OR 2.58, 95% CI [2.40, 2.77], p < 0.001), Hispanic (OR 3.31, 95% CI [2.99, 3.66], p < 0.001) or “Other” (OR 1.63, 95% CI [1.37, 1.94], p < 0.001) were associated with increased odds of having Medicaid as their primary payment type.
Bars show the survey-weighted mean proportion of visits within each setting (office, emergency department) by insurance type; error bars represent 95% confidence intervals based on survey-weighted variance estimation. Associations between visit setting and insurance type were examined using survey-weighted logistic regression models, with significance assessed by two-sided, design-adjusted Wald F tests of model coefficients. Patients with identified HCV seen in the office setting were more likely to have private insurance (OR 3.39, 95% CI [1.63, 7.05], p = 0.001), while those seen in the emergency department were more likely to be covered by Medicaid (OR 3.64, 95% CI [2.24, 5.90], p < 0.001). n denotes unweighted visit counts (office n = 568; emergency department n = 267).
Lines show the survey-weighted mean proportion of physicians accepting each coverage; error bars represent 95% confidence intervals derived from survey-weighted variance estimation. Differences in acceptance rates between insurance types were tested using two-sided, design-adjusted Wald F tests comparing weighted proportions. Across all years, Medicaid acceptance was significantly lower than Medicare and private insurance (all p < 0.01). n denotes unweighted physician observations per year (ranges: private n = 0–68,415, no data 2010; Medicare n = 7535–70,757; Medicaid n = 7500–70,500).
Trends in utilization of HCV care
Over the course of the study period, the percentage of total visits for identified HCV increased in both the office setting (OR 1.35, 95% CI [1.15, 1.60], p < 0.001) and the ER setting (OR 1.27, 95% CI [1.11, 1.45], p < 0.001). In a multivariable logistic regression controlling for age, gender, race, ethnicity, region, MSA status, and insurance coverage, the odds of an identified HCV visit were highest for the combined year period of 2016 and 2018 (OR 2.05, 95% CI [1.11, 3.78]). The largest increase in visit rate was observed between 2012–2014, from 0.22% to 0.43% or 98.8% increase. This is likely due to updated screening guidelines for baby boomers put forth by the CDC in 2012 (Fig. 3). However, when evaluating visits by race/ethnicity, the total number of visits by Black patients remained unchanged with an annual percentage change (APC) of −0.44% (95% CI [−10.46, 10.71], p < 0.001), compared to non-Hispanic White (APC 17.99%, 95% CI [10.28, 26.23], p = 0.041) and Hispanic (APC 27.71%, 95% CI [26.31, 31.15], p < 0.001) (Fig. 4).
Lines show the survey-weighted mean proportion of visits with identified HCV in each setting (office, emergency department); error bars represent 95% confidence intervals derived from survey-weighted variance estimation. Between-setting differences within each period were assessed using two-sided, design-adjusted Wald F tests comparing weighted proportions. The largest period-to-period increase occurred in the office setting from 2014/2015 to 2016/2018 (p = 0.02). n denotes unweighted visit counts (ranges: office n = 8250–131,203; emergency department n = 19,481–66,020).
Points show the survey-weighted totals of visits for each race/ethnicity across periods; error bars represent 95% confidence intervals derived from survey-weighted variance estimation. Changes over time were assessed within each race/ethnicity using two-sided, design-adjusted Wald F tests comparing pre- and post-periods. Total visits increased significantly after approval of direct-acting antivirals among White patients (p = 0.005) but not among Black (p = 0.220) or Hispanic/Latino patients (p = 0.08). n denotes unweighted visit counts (non-Hispanic White n = 32–166; non-Hispanic Black n = 9–58; Hispanic/Latino n = 10–32).
HCV testing
Between 2014–2019, a total of 3.7 billion office visits were made by non-pregnant adults without known hepatitis B, hepatitis C, or cirrhosis. During these visits, 36.6 million HCV tests were performed, for a testing rate of 1%. In univariable analysis, non-Hispanic Black patients had a higher likelihood of HCV testing, whereas males, individuals born before 1945, and those covered by Medicare had lower testing rates (Table 3). Estimates from multivariable logistic regression indicated that non-Hispanic Blacks patients had higher per-visit odds of HCV testing (OR 3.03, 95% CI [2.10, 4.37]) compared to patients of other races/ethnicities.
Testing rates were higher for visits during which the provider identified themselves as the patient’s primary care provider (1.7 vs. 0.5%, OR 3.23, 95% CI [2.93, 3.56], p < 0.001) as well as during visits to general practitioners when compared to medical or surgical specialists (1.7% vs. 0.5% vs. 0.2% respectively, OR 3.15 95% CI [3.14, 3.15], p < 0.001, OR 7.41 95% CI [7.40, 7.42], p < 0.001). Additionally, patients who presented for initial visits (1.6 vs. 0.9%, OR 1.92 95% CI [1.91, 1.93], p < 0.001) and preventative visits (2.7 vs. 0.7%, OR 3.91 95% CI [3.90, 3.92] p < 0.001) were more likely to undergo HCV testing when compared to follow-up visits and visits for acute medical problems, respectively.
After applying more stringent criteria excluding surgical specialties, visits where the physician indicated that they were not the patient’s primary care provider, and visits for an acute problem, a flare of a chronic problem, or a pre/post-operative evaluation, hepatitis testing was performed during 20 million of 887 million visits (2.2%). Black patients were tested at a significantly higher rate than non-Hispanic White and Hispanic and other (3.6 vs. 1.5 vs. 3.0 vs. 6.5%, p < 0.001. Regarding birth cohort, the highest testing rate was for those born between 1965 to 1985 (3.4%) compared to those born between 1945–1965 (2.1%), before 1945 (1.0%) and those born 1985–2005 (3.3%), p = 0.035. Among insurance categories, patients covered by Medicaid had the highest rate of hepatitis testing while those covered by Private insurance lower and by Medicare the lowest (4.9 vs. 2.4 vs. 1.4%, p = 0.023). In multivariable regression, age and race differences were retained while testing rates by primary insurance were no longer significantly different.
In subgroup analysis based on 2012 screening recommendations, there were 406 million visits for baby boomers, 33.2 million visits for people with substance use disorder (SUD) (as documented by the treating provider), and 1.5 million visits for patients with end stage renal disease. In total, 426 million visits occurred for those who met high-risk screening criteria, and hepatitis testing was performed during 2.1% of these visits, no higher than testing rates in those who did not meet screening criteria (2.4%). In contrast, a complete blood count was ordered during 22.7% of these visits, suggesting that the reason for not performing hepatitis testing was not lack of access to phlebotomy/laboratory evaluations.
Finally, when more stringent criteria was applied by only including those visits for which a self-identified primary care provider was seeing the patient for the first time for a preventive or routine care visit was applied, we estimated a testing rate of 9.8%. In comparison, complete blood count (40.0% of visits), complete metabolic panel (39.8% of visits) and glycosylated hemoglobin level (19.1% of visits) were ordered at significantly higher rates (p < 0.01 for all).
Care utilization in patients with substance use disorder
From 2010–2019, 69.6 million visits were identified for substance use disorder (SUD), and 1.3 million (1.8%) had identified HCV. Compared to those without SUD, those with SUD used the ER for a higher proportion of total visits (14.3 million of 69.6 million, 20.6% vs. 1.2 billion of 9.6 billion, 12.6%, OR 1.8 95% CI [1.4, 2.2], p < 0.001). Similarly, among patients with identified HCV, those with SUD were more likely to utilize care in the ER compared to those without (17.1% [of all HCV visits] vs. 4.5%, OR 4.4 95% CI [2.1, 9.0], p < 0.001). Only 3.6% of visits for people with SUD were of “baby boomer” age, compared to 19.3% of overall visits. In the baby boomer cohort with HCV, 0.1% of visits were for those with SUD compared to 0.5 vs. 1.5 vs. 1.2 in those born [before 1945 or 1965–1985 or 1985–2005] (Fig. 5). In the period 2010–2012, 0.2 million (4.5%) of visits for HCV were also associated with SUD compared to 0.6 million (4.8%) in 2016–2019. Medicaid was overrepresented as the primary payer type for those with SUD (33% of total). In multivariable regression analysis adjusting for age, gender, race, and primary insurance, there were increased odds of SUD among HCV-identified users of the ER (OR 3.48, 95% CI [1.23, 9.90], p = 0.02). Similarly, in adjusted multivariable analysis those with both identified HCV and SUD had higher odds of using the ER for care compared to those without SUD (OR 3.30 95% CI [1.23, 9.02], p = 0.02).
Bars show the percentage of visits with SUD within each cohort; error bars represent 95% confidence intervals derived from survey-weighted variance estimation. Higher SUD prevalence was observed among younger birth cohorts. n denotes unweighted visit counts (before 1945 n = 42; 1945–1965 n = 561; 1965–1985 n = 185; 1985–2005 n = 44).
Discussion
In this study, we utilized a public, nationally representative study of ambulatory care use to comprehensively evaluate hepatitis C testing and care utilization in emergency and office settings during a critical transition time in hepatitis C, between 2010–2019. In this period, an estimated 36.6 million HCV tests were captured in our analysis, and patients with HCV had 22 million office visits and 1.2 million ER visits. There are several key findings in this study. First, there were demographic differences in the most common care setting for those with HCV, namely the higher rates of ER use among Black patients, baby boomers, those in the West region, and those covered under Medicare and Medicaid. There was also disproportionate ER utilization among those with SUD, including those with already identified HCV. Second, there was an observed increase in visit proportions for those with hepatitis C, but primarily among White privately patients in the office setting. This may reflect access to office-based care as White patients were more likely to have private insurance. Third, HCV testing rates remained low. While on a per-visit basis Black patients were tested at a higher rate, this may be mitigated by lower overall access and visit rates. Taken together, these findings suggest that the current screening paradigm, which relies on office-based testing by primary care physicians, may have increasingly poor effectiveness in identifying hepatitis C and linking patients to treatment. Key strengths of this study include nationally representative nationwide estimates based on billions of setting-agnostic primary care visits, as well as low levels of missing data and consistency of data collection.
Our findings complement those of previous studies examining demographic shifts in hepatitis C in the US. Since 2010, a significant increase in HCV incidence in the United States has been reported, with new cases nearly quadrupling between 2011 and 201915. This dramatic increase in HCV has largely been attributed to the nation’s opioid epidemic, with some studies estimating that up to one-third of persons who inject drugs (PWID) will develop HCV within the first year16,17. Patients with SUD, along with Black patients and those covered by Medicaid and Medicare were more likely to utilize care from the ER rather than from an office visit. These findings are consistent with prior studies that found that Black patients and those with Medicaid are less likely to utilize primary care as their usual source of care and are more likely to visit the ER for non-urgent care18,19,20,21. One major barrier to improved HCV screening is the lack of insurance coverage for HCV testing in the ER. ER-based HCV screening has been shown to be cost-effective and has the opportunity to significantly reduce future complications, particularly for vulnerable patients22.
Furthermore, while the ER can play a crucial role in caring for patients with SUD and identifying HCV cases, SUD and HCV treatment often require longitudinal outpatient management. As the prevalence of CHC decreases in baby boomers and those with higher socioeconomic status, case identification may increasingly rely on emergency care. Prior studies have indicated significant patient attrition along the HCV care cascade, highlighting the need for structured linkage-to-care programs, particularly for patients who primarily obtain care from the ER23,24. Ensuring effective follow-up for these patients is essential for improving treatment uptake and outcomes. Innovative approaches, such as integrating patient navigators and expanding telemedicine-based follow-up, may enhance care continuity and treatment initiation25. One recent study has shown high levels of satisfaction with telemedicine and high levels of HCV treatment completion amongst those enrolled in opioid treatment programs26. Additionally, telemedicine may help mitigate non-financial barriers faced by underserved populations21,27. It is critical to continue to implement strategies to mitigate access barriers despite financial challenges that may be faced by healthcare providers.
There has also been a dramatic demographic shift among individuals with hepatitis C, largely driven by the opioid epidemic as well as more successful screening and elimination in the baby boomer population. However, it is concerning that the HCV testing rates may not reflect this demographic shift. In 2021, the CDC estimated that rural areas had significantly higher rates of HCV incidence (58 per 100,000) when compared to urban areas (36 per 100,000)28. The same report found White patients and male patients had higher rates of HCV. In this study, we found similar rates of HCV testing in metropolitan and non-metropolitan areas. This may be the result of persistent geographic disparities in access to care and patients often need to travel further to obtain care27, as many specialists who treat patients with CHC are more likely to be in urban areas29,30,31. Additionally, we found a significantly higher testing rate for Black patients (2.0%) when compared to White patients (0.6%). While the exact reason for this is unclear, it is possible that a larger proportion of White patients live in rural areas that have lower geographic access to care. However, as noted earlier, Black patients were more likely to obtain care at the ER, which may fragment care and reduce linkage to outpatient treatment, even when identified as a case21.
Screening is a critical first step towards our goal of HCV elimination, and there are many barriers that must be addressed as efforts continue towards achieving this goal. One such barrier is access to care. For example, as mentioned above, there are significant geographic disparities to access care, especially in areas that have been most impacted by HCV in recent years. While not assessed in our study, others have found that point of care (POC) HCV testing has shown promise in other countries and was recently approved by the FDA32. The implementation of POC HCV screening at community centers, supervised consumption services, and non-clinical justice settings could improve HCV screening rates, especially in at-risk areas23,33,34. This study has several limitations. First, due to the cross-sectional nature of NAMCS/NHAMCS, our study lacked longitudinal information. This could lead to inaccurate estimations of testing as we cannot account for individuals who previously underwent screening; any bias in utilization would be expected to trend towards the null in our analysis. However, to our knowledge, there are no nationally representative surveys that provide longitudinal data on this population. Additionally, the lack of longitudinal data prevents us from better understanding the impact of different care utilization practices on clinical outcomes. An additional limitation of this study was that NAMCS/NHAMCS data estimate healthcare utilization rather than true HCV prevalence. As such, visits by patients with HCV may be under-identified due to reliance on ICD-9 and ICD-10 codes. Previous validation studies suggest these codes have moderate sensitivity (50–70%) but high specificity, meaning some undiagnosed or undocumented cases were likely missed. Likewise, NHAMCS does not capture visits to freestanding urgent care centers that are not affiliated with hospitals. As such, we may underestimate care utilization, particularly among patients who seek low-cost, walk-in care outside of traditional emergency or office-based settings. However, cases were unlikely to have been systematically missing due to specific patient characteristics or visit type, mitigating concerns of potential bias. This limitation underscores the need for future studies using biomarker-based databases or electronic health records with longitudinal follow-up. Finally, a critical transition in hepatitis C screening and management took place during this study period, including widespread adoption of screening in baby boomers. Regardless, our study serves to detail demographic shifts in usage rates, including among younger individuals and those in rural areas or with SUD who are less likely to seek care in commonly researched settings. Lastly, our most recent data were from 2019, a year prior to CDC’s updated recommendation of universal screening for HCV. The onset of the COVID-19 pandemic has led to a halt in data collection and the subsequent decision to discontinue NHAMCS permanently. The future lack of public, nationally representative and comparable data between care settings, however, adds to the importance of this contemporary analysis of HCV care in the US. Moving forward, it is critical to have updated HCV screening rates and to evaluate the effect of universal HCV screening put forth in 2020.
In this study using two large, nationally representative databases, we estimated ambulatory care utilization for individuals with HCV. Our study showed greater ER utilization among patients who were younger, Black, and covered by Medicaid. Additionally, we found that HCV screening practices did not address current patient demographic trends for those with HCV. Policy and practice updates must be implemented to enhance providers awareness of the current state of HCV infection in the United States and to improve patient access to adequate screening and linkage to care. As efforts continue toward the goal of HCV elimination, it is crucial for future research and further initiatives to focus on at risk populations.
Data availability
The data analyzed in this study are publicly accessible from the National Center for Health Statistics. Specifically, the National Ambulatory Medical Care Survey (NAMCS) and the National Hospital Ambulatory Medical Care Survey (NHAMCS) datasets (2010–2019) can be obtained through the NCHS public-use data portal. Metadata, processed variables, and all numerical source data underlying the figures in this paper are available at Zenodo [https://zenodo.org/records/15333357]35.
Code availability
The R scripts used for curation, analysis, and visualization, along with documentation, are openly available at Zenodo [https://zenodo.org/records/15333357]35.
References
Hall, E. W. et al. Estimating hepatitis C prevalence in the United States, 2017–2020. Hepatology 81, 625–636 (2025).
Blach, S. et al. Global prevalence and genotype distribution of hepatitis C virus infection in 2015: a modelling study. Lancet Gastroenterol. Hepatol. 2, 161–176 (2017).
Ly, K. N. et al. Deaths associated with hepatitis C virus infection among residents in 50 states and the District of Columbia, 2016-2017. Clin. Infect. Dis. 71, 1149–1160 (2020).
Westbrook, R. H. & Dusheiko, G. Natural history of hepatitis C. J. Hepatol. 61, S58–S68 (2014).
Afdhal, N. et al. Ledipasvir and sofosbuvir for untreated HCV genotype 1 infection. N. Engl. J. Med. 370, 1889–1898 (2014).
Ha, S. & Timmerman, K. Awareness and knowledge of hepatitis C among health care providers and the public: a scoping review. Can. Commun. Dis. Rep. 44, 157–165 (2018).
Smith, B. D. et al. Recommendations for the identification of chronic hepatitis C virus infection among persons born during 1945-1965. MMWR Recomm. Rep. 61, 1–32 (2012).
Schillie, S., Wester, C., Osborne, M., Wesolowski, L. & Ryerson, A. B. CDC recommendations for hepatitis C screening among adults—United States, 2020. MMWR Recomm. Rep. 69, 1–17 (2020).
Biondi, B. E. et al. Racial and ethnic disparities in hepatitis C care in reproductive-aged women with opioid use disorder. Clin. Infect. Dis. 79, 1428–1436 (2024).
Nili, M., Luo, L., Feng, X., Chang, J. & Tan, X. Disparities in hepatitis C virus infection screening among Baby Boomers in the United States. Am. J. Infect. Control 46, 1341–1347 (2018).
Artenie, A. A., Bruneau, J., Lévesque, A. & Wansuanganyi, J.-M. B. Role of primary care providers in hepatitis C prevention and care: one step away from evidence-based practice. Can. Fam. Physician 60, 881–2, e468-70 (2014).
McCaig, L. F. & McLemore, T. Plan and operation of the National Hospital Ambulatory Medical Survey. Series 1: programs and collection procedures. Vital Health Stat. 1 34, 1–78 (1994).
Fleming-Dutra, K. E. et al. Prevalence of inappropriate antibiotic prescriptions among US ambulatory care visits, 2010-2011. JAMA 315, 1864 (2016).
Center for Disease Control and Prevention. NAMCS and NHAMCS survey methods and analytic guidelines. https://archive.cdc.gov/www_cdc_gov/nchs/ahcd/survey-methods-archived.htm (2019).
Nguyen, I., Moussa, K. & Gutierrez, J. Hepatitis C virus elimination in the United States: challenges, progress, and future steps. Gastroenterol. Hepatol. 19, 700–707 (2023).
Gonzalez, S. A. & Trotter, J. F. The rise of the opioid epidemic and hepatitis C–positive organs. Hepatology 67, 1600–1608 (2018).
Hagan, H., Pouget, E. R., Des Jarlais, D. C. & Lelutiu-Weinberger, C. Meta-regression of hepatitis C virus infection in relation to time since onset of illicit drug injection: the influence of time and place. Am. J. Epidemiol. 168, 1099–1109 (2008).
Kim, H., McConnell, K. J. & Sun, B. C. Comparing emergency department use among medicaid and commercial patients using all-payer all-claims data. Popul Health Manag. 20, 271–277 (2017).
Doty, M. M. & Holmgren, A. L. Health care disconnect: gaps in coverage and care for minority adults. Findings from the Commonwealth Fund Biennial Health Insurance Survey (2005). Issue Brief. Common. Fund. 21, 1–12 (2006).
Parast, L. et al. Racial/ethnic differences in emergency department utilization and experience. J. Gen. Intern. Med. 37, 49–56 (2022).
Ying, X. et al. Racial disparities in cost and non-cost barriers to care: an analysis of the All of Us Survey. J. Gen. Intern. Med. 39, 2875–2877 (2024).
Choi, S. A. et al. Cost-effectiveness analysis of emergency department-based hepatitis C screening and linkage-to-care program. BMC Health Serv. Res. 24, 1–11 (2024).
Trickey, A., Fajardo, E., Alemu, D., Artenie, A. A. & Easterbrook, P. Impact of hepatitis C virus point-of-care RNA viral load testing compared with laboratory-based testing on uptake of RNA testing and treatment, and turnaround times: a systematic review and meta-analysis. Lancet Gastroenterol. Hepatol. 8, 253–270 (2023).
Tran, L., Jung, J., Feldman, R. & Riley, T. Disparities in the quality of care for chronic hepatitis C among Medicare beneficiaries. PLoS ONE 17, e0263913 (2022).
Hyde, Z. et al. Evaluation of a pilot emergency department linkage to care program for patients previously diagnosed with Hepatitis C. J. Viral Hepat. 30, 129–137 (2023).
Talal, A. H. et al. High satisfaction with patient-centered telemedicine for hepatitis C virus delivered to substance users: a mixed-methods study. Telemed. e-Health 29, 395–407 (2023).
Reed, M. E. et al. Patient characteristics associated with choosing a telemedicine visit vs office visit with the same primary care clinicians. JAMA Netw. Open 3, e205873 (2020).
Centers for Disease Control and Prevention. Viral Hepatitis Surveillance Report—United States, 2021. https://Www.Cdc.Gov/Hepatitis-Surveillance-2021/about/Index.Html (2023).
Estadt, A. T. et al. Differences in hepatitis C virus (HCV) testing and treatment by opioid, stimulant, and polysubstance use among people who use drugs in rural U.S. communities. Harm Reduct. J. 21, 214 (2024).
Zhang, D. et al. Assessment of changes in rural and urban primary care workforce in the United States from 2009 to 2017. JAMA Netw. Open 3, e2022914 (2020).
Ying, X., Yao, L., Mathis, W. S., Congly, S. E. & Jesudian, A. B. Geographic disparities in access to gastroenterologists in the United States. Gastroenterology https://doi.org/10.1053/j.gastro.2025.01.232 (2025).
Food and Drug Administration. FDA permits marketing of first point-of-care hepatitis C RNA test. (2024).
Harvey, L. et al. Feasibility and performance of a point-of-care hepatitis C RNA assay in a community supervision cohort. JAMA Netw. Open 7, e2438222 (2024).
Lettner, B. et al. Rapid hepatitis C virus point-of-care RNA testing and treatment at an integrated supervised consumption service in Toronto, Canada: a prospective, observational cohort study. Lancet Reg. Health Am. 22, 100490 (2023).
Buckholz, A. et al. Sociodemographic disparities in hepatitis C care utilization and testing in the United States: a nationwide survey analysis. Zenodo. (2025).
Acknowledgements
This study was supported by funding from the National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases, fund number 2T32DK116970-06. Data from the National Ambulatory Medical Care Survey (NAMCS), conducted by the National Center for Health Statistics (NCHS), were used for this study. The interpretations, conclusions, and opinions expressed in this study are solely those of the study team and do not represent the official findings, policies, or recommendations of the National Center for Health Statistics, the Centers for Disease Control and Prevention, or the U.S. Government.
Author information
Authors and Affiliations
Contributions
Writing and original draft preparation: X.Y., Y.L., H.M., and A.P.B.; Conceptualization: X.Y., R.S.B., and M.U.; Methodology: A.P.B., H.M., and M.U.; Data curation: X.Y., B.M., O.B., and A.P.B.; Project supervision and funding acquisition: M.U. and R.S.B. All authors interpreted the data, reviewed the manuscript, and approved the final version.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Medicine thanks the anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Buckholz, A.P., Ying, X., Liu, Y. et al. Sociodemographic disparities in Hepatitis C care utilization and testing in the United States. Commun Med 6, 155 (2026). https://doi.org/10.1038/s43856-025-01352-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s43856-025-01352-1