Abstract
Background
Alzheimer’s disease (AD) carries a high societal burden inequitably distributed across demographic groups. Using real-world electronic health record (EHR) data with accurate population identification, we examine demographic differences and potentially modifiable drivers of AD decline.
Methods
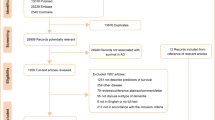
Leveraging EHR data (1994–2022) from two large independent healthcare systems, we applied an unsupervised phenotyping algorithm to predict AD diagnosis and validated using gold-standard chart-reviewed and registry-derived diagnosis labels. Among patients with ≥24 months of EHR data not living in nursing homes pre-AD diagnosis, we estimated the time-to-decline (nursing home admission, death) in healthcare system-specific covariate-adjusted competing risk survival analyses stratified by demographic groups. We then performed covariate-adjusted fixed-effects meta-analyses using inverse variance weighting.
Results
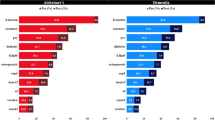
The algorithm demonstrates robust performance in identifying AD populations across healthcare systems and demographic groups (AUROC score range: 0.835-0.923). Of the 29,262 AD patients in both healthcare systems (61% women, 90% non-Hispanic White, 79.52 ± 9.39 years of age at AD diagnosis), 49% transition to nursing homes and 52% die during follow-up. In covariate-adjusted fixed-effects meta-analysis, women have higher nursing home admission risk (HR [95% CI] = 1.061 [1.024-1.100], p = 1.203×10-3) but lower death risk (HR [95% CI] = 0.856 [0.811-0.904], p = 2.434×10-8) than men. Non-Hispanic White individuals have similar nursing home risk (HR [95% CI] = 1.006 [0.952-1.063], p = 8.306×10-1) but higher death risk (HR [95% CI] = 1.376 [1.245-1.521], p = 4.084×10-10) than racial and ethnic minorities. Older age at AD diagnosis and greater comorbidity burden increase both nursing home admission and death risk.
Conclusions
We provide real-world evidence of drivers of demographic differences in AD decline that could inform individual clinical management and public health policies.
Plain language summary
People with Alzheimer’s disease have memory loss and behavior changes. They experience varying rates of decline, with some facing higher risks of entering nursing homes or dying. In this study, we examined which individuals were at greater risk of decline by analyzing medical records. We identified 29,262 people with Alzheimer’s disease using an accurate patient identification tool. We found that women were more likely to enter nursing homes, while men and non-Hispanic White individuals were more likely to die. Patients diagnosed at an older age and those with additional health conditions faced increased risks of both poor outcomes. These insights from real-world clinical data may help clinicians tailor individualized care and promote more equitable Alzheimer’s disease management.
Similar content being viewed by others
Data availability
Anonymous summary-level registry data and EHR data will be made available upon reasonable request to the corresponding author. The rationale for not sharing patient-level data is that patient-level clinical data (either de-identified information or limited protected health information containing dates of clinical events or even if anonymous due to concern for re-identification) are universally subject to the rules and regulation of each healthcare system, which may only be affiliated with but are not the same as the primary academic institutions of the study investigators. Sharing of de-identified EHR data with qualified external researchers by each of the study performance sites may be permissible only after the approval of the respective Institutional Review Boards (IRBs), regulatory oversight agents of the healthcare systems (that own the clinical data) as well as the appropriate Data Usage Agreements (DUA) between institutions.
Code availability
All statistical analyses were conducted using R (version 4.4.1). Codes for KOMAP and project analysis are publicly available on Github46,70. Data harmonization procedures, covariate definitions, and validation of outcomes are described in the Methods to enable replication in other healthcare systems with EHR data.
References
Scheltens, P. et al. Alzheimer’s disease. Lancet 397, 1577–1590 (2021).
Jack, C. R. et al. Revised criteria for diagnosis and staging of Alzheimer’s disease: Alzheimer’s Association Workgroup. Alzheimer’s. Dement. 20, 5143–5169 (2024).
Alzheimer's Association Report. 2025 Alzheimer’s disease facts and figures. Alzheimer’s Dement. 21, e70235 (2025).
Gracner, T. et al. Advanced cognitive impairment among older nursing home residents. BMC Geriatr. 21, 382 (2021).
Lusk, J. B. et al. Sex differences in mortality and health care utilization after dementia diagnosis. JAMA Neurol. 82, 1048–1056 (2025).
Rivera-Hernandez, M., Kumar, A., Epstein-Lubow, G. & Thomas, K. S. Disparities in nursing home use and quality among African American, Hispanic, and White medicare residents with Alzheimer’s disease and related dementias. J. Aging Heal 31, 1259–1277 (2019).
Temkin-Greener, H., Yan, D., Wang, S. & Cai, S. Racial disparity in end-of-life hospitalizations among nursing home residents with dementia. J. Am. Geriatr. Soc. 69, 1877–1886 (2021).
Mayeda, E. R. et al. Survival after dementia diagnosis in five racial/ethnic groups. Alzheimer’s. Dement. 13, 761–769 (2017).
Chen, Y., Crimmins, E., Ferido, P. & Zissimopoulos, J. M. Racial/ethnic disparities in length of life after dementia diagnosis: an 18-year follow-up study of medicare beneficiaries. Lancet Reg. Heal - Am. 8, 100179 (2022).
Khan, A. H. et al. Analysis of Alzheimer’s disease-related mortality rates among the elderly populations across the United States: an analysis of demographic and regional disparities from 1999 to 2020. Curr. Alzheimer Res. 21, 384–394 (2024).
Yen, T. Y. et al. Medical comorbidities and association with mortality risk in Alzheimer’s disease: population-based study of 132,405 geriatric inpatients. Cureus 12, e8203 (2020).
Taudorf, L., Nørgaard, A., Brodaty, H., Laursen, T. M. & Waldemar, G. Dementia increases mortality beyond effects of comorbid conditions: a national registry-based cohort study. Eur. J. Neurol. 28, 2174–2184 (2021).
Rajamaki, B., Hartikainen, S. & Tolppanen, A. M. The effect of comorbidities on survival in persons with Alzheimer’s disease: a matched cohort study. BMC Geriatr. 21, 173 (2021).
Armstrong, M. J., Song, S., Kurasz, A. M. & Li, Z. Predictors of mortality in individuals with dementia in the National Alzheimer’s Coordinating Center. J. Alzheimer’s. Dis. 86, 1935–1946 (2022).
Lanctôt, K. L. et al. Burden of illness in people with Alzheimer’s disease: a systematic review of epidemiology, comorbidities and mortality. J. Prev. Alzheimer’s. Dis. 11, 97–107 (2024).
Zheng, X., Wang, S., Huang, J., Li, C. & Shang, H. Predictors for survival in patients with Alzheimer’s disease: a large comprehensive meta-analysis. Transl. Psychiatry 14, 184 (2024).
Grodstein, F. et al. Identification of dementia in recent medicare claims data, compared with rigorous clinical assessments. J. Gerontol: Ser. A 77, 1272–1278 (2021).
Bhattacharyya, J. et al. Evaluating linked ICD-10 medicare claims data as a method of dementia case ascertainment in research settings. Alzheimer’s. Dement. 21, e70200 (2025).
McCarthy, E. P. et al. Validation of claims algorithms to identify Alzheimer’s disease and related dementias. J. Gerontol: Ser. A 77, 1261–1271 (2021).
Haye, S. et al. Estimates of diagnosed dementia prevalence and incidence among diverse beneficiaries in traditional medicare and medicare advantage. Alzheimer’s. Dement.: Diagn. Assess. Dis. Monit. 15, e12472 (2023).
Gianattasio, K. Z. et al. Case definition for diagnosed Alzheimer disease and related dementias in medicare. JAMA Netw. Open 7, e2427610 (2024).
Phung, T. K. T. et al. Validity of dementia diagnoses in the Danish Hospital registers. Dement. Geriatr. Cogn. Disord. 24, 220–228 (2007).
Salem, L. C. et al. Overdiagnosis of dementia in young patients—a nationwide register-based study. Dement Geriatr. Cogn. Disord. 34, 292–299 (2013).
Rizzuto, D. et al. Detection of dementia cases in two Swedish health registers: a validation study. J. Alzheimer’s. Dis. 61, 1301–1310 (2018).
Tjandra, D., Migrino, R. Q., Giordani, B. & Wiens, J. Cohort discovery and risk stratification for Alzheimer’s disease: an electronic health record-based approach. Alzheimer’s. Dement.: Transl. Res Clin. Inter. 6, e12035 (2020).
Walling, A. M., Pevnick, J., Bennett, A. V., Vydiswaran, V. G. V. & Ritchie, C. S. Dementia and electronic health record phenotypes: a scoping review of available phenotypes and opportunities for future research. J. Am. Méd. Inf. Assoc. 30, 1333–1348 (2023).
Oh, I. Y. et al. Extraction of clinical phenotypes for Alzheimer’s disease dementia from clinical notes using natural language processing. JAMIA Open 6, ooad014 (2023).
Estiri, H. et al. Temporal characterization of Alzheimer’s disease with sequences of clinical records. eBioMedicine 92, 104629 (2023).
Chen, Z. et al. Predicting the risk of Alzheimer’s disease and related dementia in patients with mild cognitive impairment using a semi-competing risk approach. Informatics 10, 46 (2023).
Li, Q. et al. Early prediction of Alzheimer’s disease and related dementias using real-world electronic health records. Alzheimer’s Dement. 19, 3506–3518 (2023).
Zhang, Q., Coury, R. & Tang, W. Prediction of conversion from mild cognitive impairment to Alzheimer’s disease and simultaneous feature selection and grouping using Medicaid claim data. Alzheimer’s. Res. Ther. 16, 54 (2024).
He, X. et al. Develop and validate a computable phenotype for the identification of Alzheimer’s disease patients using electronic health record data. Alzheimer’s. Dement.: Diagn. Assess. Dis. Monit. 16, e12613 (2024).
Zhu, W. et al. Predicting risk of Alzheimer’s diseases and related dementias with AI foundation model on electronic health records. Preprint at medRxiv https://doi.org/10.1101/2024.04.26.24306180 (2024).
Tang, A. S. et al. Leveraging electronic health records and knowledge networks for Alzheimer’s disease prediction and sex-specific biological insights. Nat. Aging 4, 379–395 (2024).
Han, E., Kharrazi, H. & Shi, L. Identifying predictors of nursing home admission by using electronic health records and administrative data: scoping review. JMIR Aging 6, e42437 (2023).
Mielke, M. M. et al. Consideration of sex and gender in Alzheimer’s disease and related disorders from a global perspective. Alzheimer’s. Dement 18, 2707–2724 (2022).
Shaaban, C. E. & Rosso, A. L. Current topics in behavioral neurosciences, Curr.Topics Behav.Neurosci., racial, ethnic, and geographic diversity in population neuroscience. Curr. Top Behav. Neurosci. 68, 67–85 (2024).
Nelson, S. J., Zeng, K., Kilbourne, J., Powell, T. & Moore, R. Normalized names for clinical drugs: RxNorm at 6 years. J. Am. Méd. Inf. Assoc. 18, 441–448 (2011).
McDonald, C. J. et al. LOINC, a universal standard for identifying laboratory observations: a 5-year update. Clin. Chem. 49, 624–633 (2003).
Liao, K. P. et al. High-throughput multimodal automated phenotyping (MAP) with application to PheWAS. J. Am. Med Inf. Assoc. 26, 1255–1262 (2019).
Zhang, Y. et al. High-throughput phenotyping with electronic medical record data using a common semi-supervised approach (PheCAP). Nat. Protoc. 14, 3426–3444 (2019).
Yu, S., Cai, T. & Cai, T. NILE: fast natural language processing for electronic health records. Preprint at https://arxiv.org/abs/1311.6063 (2013).
Bodenreider, O. The Unified Medical Language System (UMLS): integrating biomedical terminology. Nucleic Acids Res. 32, D267–D270 (2004).
Buuren van, S. & Groothuis-Oudshoorn, K. Mice: multivariate imputation by chained equations in R. J. Stat. Softw. 45, 1–67 (2011).
Zhang, G. et al. Multiple imputation of missing race and ethnicity in CDC COVID-19 case-level surveillance data. Int. J. Stat. Méd. Res. 11, 1–11 (2022).
Xiong, X. et al. Knowledge-driven online multimodal automated phenotyping system. Preprint at medRxiv https://doi.org/10.1101/2023.09.29.23296239 (2023).
Wang, L. et al. Stratification of Alzheimer’s disease patients using knowledge-guided unsupervised latent factor clustering with electronic health record data. medRxiv https://doi.org/10.1101/2024.12.23.24319588 (2024).
McKhann, G. M. et al. The diagnosis of dementia due to Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimer’s. Dement. 7, 263–269 (2011).
Pinker, E. Reporting accuracy of rare event classifiers. NPJ Digit Med. 1, 56 (2018).
Hou, J. et al. Comparison of dimethyl fumarate vs fingolimod and rituximab vs natalizumab for treatment of multiple sclerosis. JAMA Netw. Open 4, e2134627 (2021).
Elixhauser, A., Steiner, C., Harris, D. R. & Coffey, R. M. Comorbidity measures for use with administrative data. Méd. Care. 36, 8–27 (1998).
Gasparini, A. comorbidity: an R package for computing comorbidity scores. J. Open Source Softw. 3, 648 (2018).
Walraven, C. et al. A modification of the elixhauser comorbidity measures into a point system for hospital death using administrative data. Méd. Care 47, 626–633 (2009).
Yan, B. W. et al. Widening gender gap in life expectancy in the US, 2010-2021. JAMA Intern. Med. 184, 108–110 (2024).
Rentería, M. A. et al. Representativeness of samples enrolled in Alzheimer’s disease research centers. Alzheimer’s. Dement.: Diagn. Assess. Dis. Monit. 15, e12450 (2023).
Gianattasio, K. Z. et al. Generalizability of findings from a clinical sample to a community-based sample: a comparison of ADNI and ARIC. Alzheimer’s. Dement. 17, 1265–1276 (2021).
Ferretti, M. T. et al. Sex differences in Alzheimer disease—the gateway to precision medicine. Nat. Rev. Neurol. 14, 457–469 (2018).
Mjørud, M. et al. Time from dementia diagnosis to nursing-home admission and death among persons with dementia: a multistate survival analysis. PLoS ONE 15, e0243513 (2020).
Manton, K. G., Poss, S. S. & Wing, S. The Black/White mortality crossover: investigation from the perspective of the components of aging. Gerontology 19, 291–300 (1979).
Hinton, L., Tran, D., Peak, K., Meyer, O. L. & Quiñones, A. R. Mapping racial and ethnic healthcare disparities for persons living with dementia: a scoping review. Alzheimer’s. Dement. 20, 3000–3020 (2024).
National Academies of Sciences, Engineering, and Medicine. Rethinking Race and Ethnicity in Biomedical Research. https://doi.org/10.17226/27913 (2024).
Sharma, S. et al. Leveraging multi-site electronic health data for characterization of subtypes: a pilot study of dementia in the N3C Clinical Tenant. JAMIA Open 7, ooae076 (2024).
Miller, E. A., Schneider, L. S. & Rosenheck, R. A. Predictors of nursing home admission among Alzheimer’s disease patients with psychosis and/or agitation. Int. Psychogeriatr. 23, 44–53 (2011).
Forns, J. et al. Clinical outcomes and treatment patterns of older adults with dementia-related psychosis by dementia type in the United States. BMC Geriatr. 22, 784 (2022).
Knapp, M. et al. Predictors of care home and hospital admissions and their costs for older people with Alzheimer’s disease: findings from a large London case register. BMJ Open 6, e013591 (2016).
Joling, K. J. et al. Time from diagnosis to institutionalization and death in people with dementia. Alzheimer’s. Dement. 16, 662–671 (2020).
Kosar, C. M., Mor, V., Werner, R. M. & Rahman, M. Risk of discharge to lower-quality nursing homes among hospitalized older adults with Alzheimer disease and related dementias. JAMA Netw. Open 6, e2255134 (2023).
Maserejian, N., Krzywy, H., Eaton, S. & Galvin, J. E. Cognitive measures lacking in EHR prior to dementia or Alzheimer’s disease diagnosis. Alzheimer’s. Dement. 17, 1231–1243 (2021).
Al-Sahab, B., Leviton, A., Loddenkemper, T., Paneth, N. & Zhang, B. Biases in electronic health records data for generating real-world evidence: an overview. J. Health. Inf. Res. 8, 121–139 (2024).
Venkatesh, S. et al. Leveraging electronic health records to examine differential clinical outcomes in people with Alzheimer’s Disease. medRxiv [Preprint]. https://doi.org/10.1101/2025.04.22.25326230. (2025).
Venkatesh, S. Figure 1A. Created in BioRender. https://www.biorender.com/xwiof9h (2025).
Venkatesh, S. Figure 2. Created in BioRender. https://BioRender.com/58sbkky (2025).
Acknowledgements
We would like to thank the patients whose data contributed to the research findings. This study was supported by the National Institutes of Health under award numbers R01 NS098023 and R01 NS124882 from the National Institute of Neurological Disorders and Stroke. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Author information
Authors and Affiliations
Contributions
Shruthi Venkatesh, Linshanshan Wang, Michele Morris, Mohammed Moro, Ratnam Srivastava, Yunqing Han, Riddhi Patira, Sarah Berman, Oscar Lopez, Shyam Visweswaran, Tianrun Cai, Tianxi Cai, and Zongqi Xia contributed to the design and conceptualization of the study. Shruthi Venkatesh and Linshanshan Wang contributed equally as co-first authors to data analysis and manuscript writing. Michele Morris, Mohammed Moro, Ratnam Srivastava, Yunqing Han, Riddhi Patira, Sarah Berman, Oscar Lopez, Shyam Visweswaran, Tianrun Cai, Tianxi Cai, and Zongqi Xia contributed to data acquisition and manuscript writing. Tianxi Cai and Zongqi Xia contributed equally as co-senior authors and jointly supervised this work. All authors have reviewed and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Medicine thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Venkatesh, S., Wang, L., Morris, M. et al. Leveraging electronic health records to examine differential clinical outcomes in people with Alzheimer’s disease. Commun Med (2026). https://doi.org/10.1038/s43856-026-01443-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s43856-026-01443-7