Abstract
Background
This study aimed to evaluate the distinct prognostic value of [18F]FDG and [68Ga]Ga-PSMA-11 PET imaging in advanced hormone-sensitive prostate cancer, highlighting their complementary roles at both patient and lesion levels.
Methods
This study retrospectively included 298 patients with newly diagnosed prostate cancer who underwent baseline dual-tracer PET imaging. Quantitative PET parameters for both [18F]FDG and [68Ga]Ga-PSMA-11 were extracted. Clinical outcomes, including progression-free survival, biochemical response, and radiographic response, were collected during follow-up. Lesion-based analysis was performed in 267 measurable lesions to evaluate the association between tracer uptake and local radiographic response, as determined by changes in lesion diameter following systemic therapy.
Results
Here we show that higher whole-body [18F]FDG uptake, including total lesion glycolysis (HR = 3.525, p < 0.001) and metabolic tumor volume (HR = 2.757, p < 0.001), is significantly associated with shorter progression-free survival. In contrast, only PSMA-derived tumor volume (HR = 2.019, p = 0.018), but not total lesion PSMA uptake (HR = 1.438, p = 0.221), demonstrates prognostic value. Moreover, patients with higher whole-body [18F]FDG burden receive greater benefit from chemotherapy (HR = 2.936, p = 0.004). At the lesion level, higher [68Ga]Ga-PSMA-11 uptake is significantly correlated with more favorable radiographic response (p < 0.001), while lesion-level [18F]FDG uptake does not demonstrate predictive value.
Conclusions
[18F]FDG PET and [68Ga]Ga-PSMA-11 PET provide distinct but complementary prognostic value in advanced hormone-sensitive prostate cancer. Dual-tracer PET imaging enhances prognostic accuracy and provides comprehensive guidance for individualized treatment strategies.
Plain language summary
Positron emission tomography (PET) uses two radioactive substances called FDG and PSMA-11 to detect tumor lesions in advanced prostate cancer. However, it remains unclear whether these PET signals can help predict how well patients will respond to hormone therapy. Here, we include 298 patients with newly diagnosed advanced prostate cancer who had PET before treatment. We collect the clinical characteristics, imaging results, and follow-up data for further analysis. Results show that higher whole-body FDG signal predicts shorter survival, while higher PSMA-11 signal in individual lesion indicates better tumor shrinkage after treatment. These results suggest that FDG and PSMA-11 provide different but complementary information, helping doctors offer more personalized disease management for people with advanced prostate cancer.
Similar content being viewed by others
Introduction
Over the past decade, prostate cancer has been one of the most common malignancies among men worldwide, with significant morbidity and mortality1,2. Hormone-sensitive prostate cancer represents the initial state of prostate adenocarcinoma, characterized by active androgen receptor signaling pathway and high sensitivity to androgen-deprivation therapy (ADT)3,4. For patients with locally advanced or metastatic hormone-sensitive prostate cancer, ADT remains the standard first-line treatment, often in combination with androgen receptor pathway inhibitors (ARPIs) and/or chemotherapy to enhance disease control5,6,7. Despite initial therapeutic efficacy, a subset of patients demonstrates heterogeneous treatment responses, with biochemical or radiographic recurrence after a relatively short period following initial treatment8.
Prostate-specific membrane antigen (PSMA) serves as a prominent target for molecular imaging and targeted radiotherapy in prostate cancer9,10. Targeting PSMA has greatly improved lesion detection rates in prostate cancer, increasing sensitivity from less than 70% with conventional fluorine-18 fluorodeoxyglucose ([18F]FDG) PET to approximately 95%11,12,13. Consequently, PSMA-PET has been endorsed by international guidelines as the standard imaging modality for hormone-sensitive prostate cancer14,15,16. However, recent evidence indicates considerable heterogeneity in PSMA expression, even at the hormone-sensitive stage17,18,19. Our previous clinical observations revealed that approximately 7–10% of histologically confirmed prostate cancer lesions exhibited low PSMA avidity (miPSMA score ≤ 1)20. These lesions with low PSMA uptake may reflect distinct biological subtypes with reduced androgen receptor signaling activity and decreased sensitivity to ADT + ARPIs, thereby contributing to heterogeneous survival outcomes21,22.
Meanwhile, previous studies have demonstrated that elevated [18F]FDG uptake correlates with more aggressive disease phenotypes and worse prognosis in prostate cancer, especially in locally advanced stages23,24,25. These findings support the use of dual-tracer PET imaging with [18F]FDG and [68Ga]Ga-PSMA-11, which may provide complementary molecular information by capturing both glycolytic activity and PSMA expression at both patient and lesion levels26,27. Given these considerations, we hypothesize that lesions with low PSMA avidity may exhibit poorer response to ADT-based systemic therapies and be associated with inferior outcomes. Integrating lesion-level [68Ga]Ga-PSMA-11 metrics with whole-body [18F]FDG burden may refine the prediction of therapeutic responses and help guide personalized treatment strategies in hormone-sensitive prostate cancer28.
In this context, we have conducted a retrospective study involving 298 newly diagnosed patients who underwent baseline [18F]FDG and [68Ga]Ga-PSMA-11 dual-tracer PET scan before receiving ADT-based systemic therapy. Our study evaluates both patient-level and lesion-level imaging parameters to investigate their associations with clinical outcomes, including progression-free survival (PFS), prostate-specific antigen (PSA) response, and lesion-level radiographic response. Here we show that higher whole-body [18F]FDG uptake is associated with shorter survival and identifies patients who benefit more from chemotherapy, whereas [68Ga]Ga-PSMA-11-derived tumor volume offers limited prognostic value at the whole-body level. At the lesion level, higher [68Ga]Ga-PSMA-11 uptake aligns with better short-term radiographic response, while [18F]FDG uptake does not predict tumor shrinkage. Our findings indicate that dual-tracer PET provides complementary prognostic information and guides individualized treatment planning for patients with advanced hormone-sensitive prostate cancer.
Methods
Ethics Approval and Consent to Participate
This retrospective study was performed in accordance with the principles outlined in the Declaration of Helsinki and its later amendments. Ethical approval was obtained from the Ethics Committee of Ren Ji Hospital, Shanghai Jiao Tong University School of Medicine (Approval number: RA-2025-897). Given the retrospective study design and the use of de-identified clinical data, the requirement of informed consent was waived by the Ethics Committee.
Patients
In this study, we retrospectively screened all patients who underwent baseline [18F]FDG PET/MR and [68Ga]Ga-PSMA-11 PET/CT at the time of initial prostate cancer diagnosis at Ren Ji Hospital between January 2021 and December 2024. Among these patients, we identified 298 patients with newly diagnosed prostate adenocarcinoma who subsequently received ADT-based systemic therapy (Fig. 1A). In line with contemporary guidelines29,30, eligible candidates with de novo and/or high-volume metastatic disease and good ECOG performance status received docetaxel, which in this cohort was delivered exclusively within a triplet regimen (ADT + ARPI + docetaxel).
A Flow diagram of included and excluded patients in this retrospective study. B Schematic overview of patient-based analysis and lesion-based analysis in this retrospective study. Figure 1B is created with BioRender.com. Created in BioRender. Li, A. (2026) https://BioRender.com/mwjuznr (Agreement No. ZF290ATHTY).
Inclusion criteria were as follows: (1) Male patients aged ≥ 50 years; (2) Histologically confirmed diagnosis of prostate adenocarcinoma without prior treatment; (3) Underwent dual-tracer PET imaging with [18F]FDG and [68Ga]Ga-PSMA-11 within 60 days of pathological diagnosis and prior to the initiation of systemic therapy; (4) Received first-line ADT, with or without ARPIs and chemotherapy; (5) Availability of follow-up data including post-treatment PSA results.
Patients were excluded if they met any of the following criteria: (1) Missing essential baseline data, including serum PSA, PET imaging, or pathology; (2) More than 60 days between pathological diagnosis and baseline imaging; (3) Non-adenocarcinoma histology, including sarcoma, neuroendocrine tumor, or urothelial carcinoma involving the prostate; (4) Underwent radical prostatectomy or radical pelvic radiotherapy before receiving systemic therapies; (5) Incomplete follow-up records; (6) Prior history of other malignancies.
During treatment, serum PSA levels were regularly recorded to assess biochemical response. In addition, a subset of patients underwent pelvic MR imaging or a second PET/MR within the post-treatment period of 90–180 days in Ren Ji Hospital. These follow-up imaging data were also collected for subsequent lesion-based analysis. As depicted in Fig. 1B, patients’ baseline characteristics, imaging data, treatment details, and follow-up information were collected from electronic medical records and supplemented by telephone follow-up when necessary.
Dual-Tracer PET Scan
All enrolled patients underwent dual-tracer imaging with [18F]FDG PET/MR and [68Ga]Ga-PSMA-11 PET/CT. [18F]FDG and [68Ga]Ga-PSMA-11 were synthesized by the radiochemical laboratory within the Department of Nuclear Medicine at Ren Ji Hospital. Patients fasted for at least 6 h before [18F]FDG administration, and blood glucose levels were confirmed to be less than 140 mg/dL prior to radiotracer injection. The administered dose was 3.7 MBq/kg of body weight. After injection, patients rested quietly for 60 minutes before imaging. Integrated PET/MR scanning was conducted using a 3.0-T time-of-flight PET/MR scanner (uPMR790, United Imaging Healthcare, Shanghai, China). PET and MRI acquisitions were performed simultaneously, starting 50–60 min post-injection, with a scan duration of approximately 20 ± 3 min for pelvic imaging. Whole-body MR sequences were conducted for anatomical localization and attenuation correction.
[68Ga]Ga-PSMA-11 was administered intravenously at a dose of 1.85 MBq/kg of body weight. Patients underwent PET/CT scanning 50–60 min post-injection using a Biograph mCT PET/CT scanner (Siemens Healthcare, Erlangen, Germany). CT scanning parameters included an automatic tube current modulation at 120 kV and slice thickness of 3 mm, covering the region from the skull base down to the upper thigh. PET images were obtained with an acquisition duration of 3 min per bed position. To improve the evaluation of pelvic lesions, forced diuresis was performed with additional delayed imaging. Specifically, 20 mg of furosemide was administered intravenously, followed by oral hydration with at least 500 mL of water, and frequent voiding was encouraged. Delayed images were acquired 1.5 h after injection, covering two bed positions centered over the bladder region.
All PET scans were reviewed by experienced nuclear medicine physicians, and dual-tracer images were interpreted in accordance with established criteria to differentiate malignant from physiological uptake. Discrepancies were resolved by consensus. A lesion was considered PET-positive if its tracer uptake exceeded the mediastinal blood pool, after excluding known pitfalls and physiological uptake sites. Further validation was performed through longitudinal PSA trends, imaging follow-up, or biopsy when available.
Image Analysis
All PET images were independently reviewed by three nuclear medicine specialists, each of whom had over 5 years of clinical experience in prostate cancer imaging. The reviewers evaluated scans at different sessions, blinded to both clinical data and each other’s assessments. Any discrepancies between readers were resolved through consensus discussion to ensure accuracy and reproducibility. MRI imaging was analyzed separately using a dedicated software platform provided by the manufacturer (uWS-MR, United Imaging Healthcare, Shanghai, China).
For lesion identification and quantification, all PET-positive lesions were categorized based on five regions: prostate (primary lesion), pelvic lymph nodes, extra-pelvic lymph nodes, bone, and visceral organs. This classification corresponded to clinical TNM staging categories. For each identified lesion, a three-dimensional volume of interest (VOI) was manually delineated using a fixed threshold of 40% of the lesion’s maximum standardized uptake value (SUVmax). Quantitative metrics were extracted separately from [18F]FDG and [68Ga]Ga-PSMA-11 PET scans. For [18F]FDG PET, SUVmax, mean standardized uptake value (SUVmean), peak standardized uptake value (SUVpeak), total lesion glycolysis (TLG), and metabolic tumor volume (MTV) were measured. For [68Ga]Ga-PSMA-11 PET, SUVmax, SUVmean, SUVpeak, total lesion PSMA uptake (TL-PSMA), and PSMA-derived tumor volume (PSMA-TV) were measured. All analyses were performed using consistent VOI methodology and image reconstruction protocols to ensure reproducibility across time points.
In addition, circular two-dimensional regions of interest were positioned within the descending aorta (blood pool), liver (right hepatic lobe), and parotid glands on [68Ga]Ga-PSMA-11 PET images. The SUVmax values from these reference tissues were recorded to establish background uptake levels, facilitating the calculation of miPSMA scores for individual lesions according to predefined criteria.
Follow-up and Outcomes
Follow-up data were retrospectively collected from electronic medical records and telephone interviews. The frequency and content of follow-up were determined by treating surgeons as part of routine clinical care. Serum PSA values were available in all patients, and PSA response was assessed according to predefined criteria.
PFS was defined based on both biochemical and imaging criteria, and calculated from the date of initial diagnosis to the first evidence of disease progression or death from any cause. PFS was primarily determined based on PSA dynamics, with different definitions applied to the patients with or without local therapies. For patients who did not undergo radical prostatectomy or definitive radiotherapy after systemic therapy, biochemical progression followed the Prostate Cancer Clinical Trials Working Group 3 (PCWG3) consensus and was determined based on post-treatment PSA kinetics31. Specifically, PSA progression was defined as a ≥ 25% increase from the PSA nadir, with an absolute rise of more than 2.0 ng/mL, confirmed by a repeated value ≥ 3 weeks later under castrate-level testosterone. For patients who subsequently received radical prostatectomy or definitive radiotherapy, biochemical progression was defined as two consecutive post-treatment PSA measurements > 0.2 ng/mL.
In addition to biochemical progression, radiographic response was assessed at the patient level using follow-up pelvic MRI or PET/MR performed 90–180 days after the initiation of systemic treatment. Based on the revised RECIST 1.1 guidelines, treatment response was evaluated by calculating the sum of longest diameters of measurable lesions32. To extend this standardized assessment, we additionally performed a lesion-based analysis, following the revised RECIST 1.1 criteria and established methodologies from prior tumor imaging studies33,34,35. Measurable lesions included primary or visceral metastases with a longest diameter ≥10 mm and lymph node metastases ≥15 mm. Bone metastases were excluded from lesion-based analyses due to non-conformity with measurable lesion criteria outlined in RECIST 1.1. Individual lesion responses were quantitatively classified as complete response (CR, disappearance), partial response (PR, ≥30% reduction), and minimal response (MR, <30% reduction) relative to baseline longest diameter.
Statistics and Reproducibility
All statistical analyses were conducted using SPSS software (Version 30.0, IBM SPSS Inc., USA). Survival was analyzed using the Kaplan-Meier method with two-sided log-rank tests. Cox proportional-hazards models were used for associations with PFS, and results are reported as hazard ratios (HRs) with 95% confidence intervals (CIs). For pre-specified dichotomization of PET-derived whole-body metrics (TLG, MTV, TL-PSMA, and PSMA-TV), we used the cohort median as the cutoff.
Continuous variables were first assessed for normality and homogeneity of variance. Comparisons were then performed, as appropriate, using independent-sample t tests or Mann-Whitney U tests (two groups), and one-way ANOVA or Kruskal-Wallis tests (three or more groups). When an overall multi-group test was significant, pairwise comparisons used t tests or Mann-Whitney U tests with multiplicity control. Categorical variables were compared using the chi-square test or Fisher’s exact test, as appropriate. Unless otherwise stated, all tests were two-sided with p < 0.05 considered statistically significant. Patient-level analyses included all eligible patients (N = 298). Lesion-based analyses treated each measurable lesion as an independent observational unit (N = 267). Response analyses based on follow-up imaging included patients with evaluable scans (N = 183). This retrospective study generated no technical replicates. Analyses were performed on available data. The number of observations contributing to each analysis is reported in the corresponding table or figure.
All graphical representations were generated using GraphPad Prism (Version 10.0, GraphPad Software Inc., USA). The schematic diagram (Fig. 1B) was created using biorender.com.
Results
Clinical Characteristics
In this retrospective study, we included a total of 298 patients and summarized their clinical characteristics in Table 1. The median follow-up duration for all patients was 372 days (IQR: 211–639 days). All the patients received ADT + ARPIs as the initial treatment. Among them, 51 patients (17.1%) received chemotherapy. Furthermore, 156 patients (52.4%) underwent radical prostatectomy or radical pelvic radiotherapy at least 90 days after the initiation of systemic treatment. The median interval between the start of systemic therapy and radical local therapy in these patients was 129 days (IQR: 103–160 days).
A total of 46 patients (15.4%) had experienced biochemical progression. Baseline clinical characteristics including cT stage (p = 0.005), cM stage (p < 0.001), and ADT-based systemic therapy (p = 0.015) differed significantly between patients with and without progression. The median PFS for the entire cohort was 305 days (IQR: 203–609 days).
Nuclear Medicine Characteristics
Baseline nuclear medicine characteristics were summarized in Supplementary Table 1. A total of 467 measurable lesions were identified in 298 patients, including 387 primary lesions (82.9%) and 80 metastatic lesions (17.1%). We excluded bone metastases due to the inability to reliably measure lesion size on PET/MR imaging.
All the lesions were measured on PET/MR imaging with a median longest diameter of 26.2 mm (IQR: 18.2–37.4 mm). For [18F]FDG PET parameters, the median SUVmax, SUVmean, SUVpeak were 5.34 (IQR: 3.51–8.42), 3.06 (IQR: 2.08–4.85), and 3.76 (IQR: 2.49–6.20), respectively. The median TLG and MTV were 10.46 (IQR: 4.45–27.32) and 3.14 (IQR: 1.60–7.18). For [68Ga]Ga-PSMA-11 PET parameters, the median SUVmax, SUVmean, SUVpeak were 21.08 (IQR: 10.86–35.09), 10.56 (IQR: 6.16–19.17), and 14.55 (IQR: 7.40–22.57), respectively. The median TL-PSMA and PSMA-TV were 40.27 (IQR: 13.49–113.16) and 3.93 (IQR: 1.79–7.81).
Whole-body burden metrics in each patient were derived by summing lesion-level TLG, MTV, TL-PSMA, and PSMA-TV values across all measurable lesions (Table 2). The median whole-body TLG and MTV were 19.55 (IQR: 9.13–41.01) and 5.55 (IQR: 3.10–11.19), respectively. The median whole-body TL-PSMA and PSMA-TV were 77.11 (IQR: 26.39–178.16) and 6.21 (IQR: 3.56–12.72), respectively. On diuretic-delayed scan, the median whole-body TL-PSMA increased to 115.48 (IQR: 37.05–257.12), and whole-body PSMA-TV was 6.16 (IQR: 3.50–12.73). Notably, patients who experienced disease progression had significantly higher whole-body dual-tracer metrics compared with patients who did not.
Survival Analysis and Cox-regression Analysis
We grouped patients according to the median value of the whole-body [18F]FDG and [68Ga]Ga-PSMA-11 metrics, and conducted Kaplan-Meier survival analysis. For whole-body [18F]FDG uptake (Fig. 2A, B), both higher TLG (HR = 3.525, 95%CI: 1.974–6.293, p < 0.001) and higher MTV (HR = 2.757, 95%CI: 1.539–4.939, p < 0.001) were significantly associated with shorter PFS. For whole-body [68Ga]Ga-PSMA-11 uptake (Fig. 2C, D; Supplementary Fig. 1A, B), higher PSMA-TV was significantly associated with shorter PFS (HR = 2.019, 95%CI: 1.130–3.609, p = 0.018), while no significant difference in PFS was observed between higher and lower TL-PSMA groups (HR = 1.438, 95%CI: 0.804–2.571, p = 0.193).
A Progression-free survival analysis stratified by whole-body TLG. B Progression-free survival analysis stratified by whole-body MTV. C Progression-free survival analysis stratified by whole-body TL-PSMA. D Progression-free survival analysis stratified by whole-body PSMA-TV. Analyses used Kaplan-Meier estimates with two-sided log-rank tests (no adjustment for multiple comparisons). Statistical significance was set at p < 0.05. All panels include 298 patients. Shaded error bands denote 95% confidence intervals. Risk tables show numbers at risk and numbers of events at each time point. Groups were split by the cohort median of each whole-body metric. Source data presented in this figure are available in Supplementary Data 1.
As demonstrated in Fig. 3, univariable Cox-regression analysis identified several risk factors for shorter PFS, including age ≥ 70 (HR = 1.880, 95%CI: 1.015–3.484, p = 0.045), clinical T4 stage (HR = 3.285, 95%CI: 1.232–8.763, p = 0.017), clinical M1 stage (HR = 2.929, 95%CI: 1.539–5.574, p = 0.001), whole-body TLG ≥ 70 (HR = 4.855, 95%CI: 2.171–10.856, p < 0.001), whole-body MTV (HR = 3.310, 95%CI: 1.596–6.866, p = 0.001), and whole-body PSMA-TV (HR = 2.136, 95%CI: 1.124–4.060, p = 0.021). Conversely, intensified androgen deprivation regimens emerged as protective factors. Combining gonadotropin-releasing hormone agonist (GnRHa) with first-generation ARPIs was associated with improved PFS (HR = 0.320, 95%CI: 0.120–0.859, p = 0.024), while the inclusion of second-generation ARPIs yielded even greater benefit (HR = 0.222, 95%CI: 0.091–0.539, p < 0.001). Similarly, the receipt of triplet regimen (ADT + ARPIs + chemotherapy), compared with ADT + ARPIs alone, was associated with a much longer PFS (HR = 0.320, 95%CI: 0.114–0.894, p = 0.030).
Forest plot of hazard ratios (HRs) with 95% CIs from univariable (left) and multivariable (right) Cox models in 298 patients. Points represent HRs with horizontal lines indicating 95% CIs. Covariates and reference categories are as shown in the figure. Tests are two-sided (Wald), and no multiple-comparison adjustment was applied. Statistical significance was set at p < 0.05. Source data presented in this figure are available in Supplementary Data 1.
In the multivariable analysis adjusting for relevant clinical and nuclear medicine predictors (excluding delayed imaging parameters due to collinearity), clinical M1 stage (HR = 3.443, 95%CI: 1.647–7.197, p = 0.001) and higher whole-body TLG (HR = 3.533, 95%CI: 1.227–10.177, p = 0.019) remained independent predictors of shorter PFS. In contrast, treatment with first-generation ARPIs (HR = 0.271, 95%CI: 0.089–0.823, p = 0.021), second-generation ARPIs (HR = 0.150, 95%CI: 0.051–0.434, p < 0.001), and chemotherapy (HR = 0.303, 95%CI: 0.106–0.863, p = 0.025) continued to demonstrate significant protective effects.
Subgroup Analysis for Chemotherapy
To further investigate the prognostic benefit of chemotherapy, survival outcomes were analyzed according to whole-body [18F]FDG and [68Ga]Ga-PSMA-11 uptake. In the entire cohort (Supplementary Fig. 2A), patients treated with ADT + ARPIs demonstrated significantly worse survival compared with those receiving the triplet regimen (HR = 2.247, 95%CI: 1.124–4.490, p = 0.022). As illustrated in Supplementary Fig. 2B, C, subgroup analyses according to whole-body [18F]FDG burden revealed that chemotherapy significantly improved PFS in patients with high TLG (HR = 2.936, 95%CI: 1.416–6.090, p = 0.004), but no significant benefit was observed in those with low TLG (HR = 0.597, 95%CI: 0.094–3.780, p = 0.373). These findings suggest that baseline whole-body [18F]FDG uptake may serve as a critical biomarker for identifying patients who benefit from chemotherapy in systemic treatment of advanced hormone-sensitive prostate cancer. However, as shown in Supplementary Fig. 2D, E, stratification based on TL-PSMA demonstrated no differential chemotherapy benefit (High TL-PSMA group: p = 0.0731; Low TL-PSMA group: p = 0.1214), indicating that whole-body [68Ga]Ga-PSMA-11 uptake was not predictive of chemotherapy efficacy.
Biochemical and Radiographic Response
In addition to survival analysis, we aimed to provide a comprehensive assessment of treatment response by evaluating changes in both biochemical kinetics and radiographic response. Supplementary Fig. 3A-D depict short-term biochemical responses stratified by whole-body [18F]FDG and [68Ga]Ga-PSMA-11 uptake. To describe biochemical response, we used PSA-90 response, which was defined as a ≥ 90% decline in serum PSA from baseline. By 30 days post-treatment, patients with high whole-body TLG demonstrated a significantly greater PSA-90 response rate compared with low TLG patients (73.2% vs 53.1%, p = 0.009). Likewise, high whole-body [68Ga]Ga-PSMA-11 uptake was associated with improved 30-day PSA-90 rates (68.8% vs 55.4%, p = 0.036). In contrast, by 90 days after treatment, neither [18F]FDG (95.5% vs 96.4%, p = 0.098) nor [68Ga]Ga-PSMA-11 (97.4% vs 94.4%, p = 0.090) stratification predicted PSA-90 response, indicating that early metabolic burden may be more relevant to immediate biochemical effects than later PSA dynamics.
We evaluated radiographic response to further characterize short-term efficacy. Among the 298 patients, 183 underwent follow-up pelvic MRI or PET/MR within 90-180 days after initial systemic treatment, enabling quantitative assessment of treatment response based on the sum of longest diameters of measurable lesions. Lesion measurability and response classification followed the revised RECIST 1.1 guidelines. As depicted in Supplementary Fig. 3E, patients who achieved partial response (PR) exhibited significantly higher whole-body [18F]FDG TLG than those with complete response (CR) (p = 0.032), whereas no significant difference in TLG was observed between PR and stable disease (SD) groups. Conversely, as shown in Supplementary Fig. 3F, whole-body [68Ga]Ga-PSMA-11 TL-PSMA did not significantly differ between patients in CR and PR groups, but was significantly lower in the SD group compared with PR group (p < 0.001). We next assessed clinical downstaging base on radiographic T and N classifications. As illustrated in Supplementary Fig. 3G, H, whole-body TLG failed to distinguish downstaging rates in either T stage (70.8% vs 71.3%, p > 0.999) or N stage (46.7% vs 57.9%, p = 0.322). However, when stratified by whole-body [68Ga]Ga-PSMA-11 uptake, patients with higher TL-PSMA demonstrated significantly higher rates of T-stage downstaging than those with lower uptake (84.5% vs 61.6%, p < 0.001), while N-stage downstaging showed no significance between groups (56.4% vs 54.3%, p > 0.999).
Lesion-Based Analysis
To further investigate the discrepancy between long-term survival and short-term radiographic responses, we performed a lesion-based analysis. Based on the previously mentioned 183 patients with available imaging follow-up for quantitative radiographic response assessment, a total of 267 measurable lesions were included for lesion-level analysis. Among these, we included 215 primary or visceral metastatic lesions measuring ≥ 10 mm in longest diameter and 52 lymph node metastatic lesions measuring ≥ 15 mm in longest diameter.
Lesion responses were quantitatively classified into complete response (CR), partial response (PR), and minimal response (MR) categories based on diameter changes following systemic therapy, as following the revised RECIST 1.1 criteria. Among the 267 lesions analyzed, 67 lesions (25.1%) achieved CR, 157 lesions (58.8%) achieved PR, and 43 lesions (16.1%) had MR. As Fig. 4A and Supplementary Fig. 4 depicted, baseline [68Ga]Ga-PSMA-11 SUVmax significantly differed among these groups, with higher SUVmax observed in CR group than PR group lesions (p = 0.027), and in PR group compared to MR group lesions (p < 0.001). Similar trends were observed in [68Ga]Ga-PSMA-11 SUVmean (CR vs PR: p = 0.017, PR vs MR: p < 0.001) and SUVpeak (CR vs PR: p = 0.028, PR vs MR: p < 0.001). Neither of them provides additional predictive value beyond SUVmax. In contrast, Fig. 4B demonstrated that no significant differences in baseline [18F]FDG uptake parameters were detected across lesion response categories.
A, B Comparison of lesion-based baseline [68Ga]Ga-PSMA-11 (A) and [18F]FDG (B) PET parameters (SUVmax, SUVmean, and SUVpeak) across radiographic response categories (CR, PR, MR) in 267 measurable lesions. Group comparisons in Fig. 4A, B used Kruskal-Wallis test; when the overall test was significant (p < 0.05), pairwise Mann-Whitney U tests were performed (two-sided; no multiple-comparison adjustment). C Lesion-based analysis of treatment response stratified by miPSMA score. D Lesion-based analysis of treatment response across combined [18F]FDG and [68Ga]Ga-PSMA-11 groups. Group comparisons in Fig. 4C, D used two-sided chi-square or Fisher’s exact tests (significance p < 0.05). Source data presented in this figure are available in Supplementary Data 1.
We next categorized these lesions using the miPSMA score to account for inter-patient heterogeneity in [68Ga]Ga-PSMA-11 uptake. Among the 267 lesions, 29 lesions (10.9%) were classified as low PSMA avidity (miPSMA=1), 66 lesions (24.7%) as intermediate (miPSMA=2), and 172 lesions (64.4%) as high PSMA avidity (miPSMA=3). Notably, as Fig. 4C depicted, lesions with miPSMA 1-2 display a higher proportion of minimal response and a lower proportion of complete response compared with miPSMA 3 (p < 0.001), indicating that reduced [68Ga]Ga-PSMA-11 uptake may predicted decreased lesion sensitivity to systemic therapy and higher recurrence potential. To examine FDG-PSMA overlap or discrepancy, we further stratify lesions by [68Ga]Ga-PSMA-11 level (PSMA-low: miPSMA 1-2; PSMA-high: miPSMA 3) and, within each stratum of PSMA level, dichotomize [18F]FDG uptake using the median lesion-level [18F]FDG SUVmax. As Fig. 4D depicted, within PSMA-low lesions, radiographic response does not differ between FDG-low and FDG-high groups. The same is observed within PSMA-high lesions. In contrast, within FDG-low or FDG-high lesion, PSMA-high lesions show more favorable responses than PSMA-low lesions (FDG-low: p < 0.001; FDG-high: p = 0.002). Together, these analyses show that lesion-level [68Ga]Ga-PSMA-11 avidity predicts short-term radiographic response, and this holds irrespective of [18F]FDG high/low uptake status.
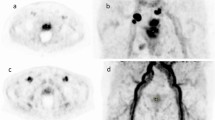
Figure 5A, B illustrated two representative cases in our cohort and further exemplified the association between baseline [68Ga]Ga-PSMA-11 uptake and lesion-level treatment responses. In patient 1 (Fig. 5A), a primary lesion with high [68Ga]Ga-PSMA-11 uptake (SUVmax = 48.82, miPSMA = 3) showed a marked response following systemic therapy, with lesion diameter decreasing from 41.1 to 12.2 mm, consistent with a PR outcome. In contrast, patient 2 (Fig. 5B) had a lesion with low [68Ga]Ga-PSMA-11 uptake (SUVmax = 6.71, miPSMA = 1), which demonstrated a minimal response despite receiving a similar treatment. The lesion diameter decreased from 27.6 to 23.4 mm, indicating only a 15.2% shrinkage.
A Representative patient #1: A 78-year-old male with newly diagnosed prostate cancer underwent baseline PET/MR imaging in June 2023. The primary lesion exhibited high [68Ga]Ga-PSMA-11 uptake (SUVmax = 48.82, miPSMA = 3), with a longest diameter of ~ 41.1 mm on axial pelvic MR. After 134 days of androgen deprivation therapy (Leuprorelin plus Rezvilutamide), follow-up pelvic MR in October 2023 demonstrated significant treatment response (longest diameter ~ 12.2 mm). B Representative patient #2: A 69-year-old male with newly diagnosed prostate cancer underwent baseline PET/MR imaging in September 2024. The primary lesion exhibited low [68Ga]Ga-PSMA-11 uptake (SUVmax = 6.71, miPSMA = 1), with a baseline diameter of ~ 27.6 mm on axial pelvic MR. After 138 days of androgen deprivation therapy (Triptorelin plus Rezvilutamide), follow-up pelvic MR in January 2025 showed limited treatment response (longest diameter ~ 23.4 mm).
Discussion
In this retrospective study, we evaluated the prognostic roles of dual-tracer PET imaging using [18F]FDG and [68Ga]Ga-PSMA-11 in patients with advanced hormone-sensitive prostate cancer. At the patient level, elevated whole-body [18F]FDG uptake parameters, particularly TLG and MTV, were significantly associated with inferior PFS. In contrast, among whole-body PSMA-derived metrics, only PSMA-TV demonstrated prognostic relevance, while TL-PSMA showed limited predictive value. Notably, lesion-based analysis revealed that individual lesions with higher PSMA avidity exhibited improved radiographic sensitivity to ADT-based systemic therapy, highlighting the complementary prognostic value of PSMA expression at the lesion level in predicting therapeutic responses.
These findings align with previous studies conducted in prostate cancer, which demonstrated that higher [18F]FDG parameters, such as TLG and MTV, were associated with inferior survival outcomes23,25. Conversely, existing literature indicates no difference in survival outcomes between high and low [68Ga]Ga-PSMA-11 SUVmax in patients with hormone-sensitive prostate cancer36,37. In our cohort, we observed distinct prognostic implications between whole-body TL-PSMA and PSMA-TV. The differential prognostic relevance between these two parameters suggested that high tumor volume, rather than higher PSMA expression, may be the critical determinant of poor PFS in patients who received ADT-based systemic therapy.
Our lesion-based analysis further supported these observations. Specifically, lesions with higher [68Ga]Ga-PSMA-11 avidity demonstrated better radiographic responses following ADT-based systemic therapy, indicating that patients with high PSMA burden were more likely to achieve prolonged PFS. In contrast, lesions with lower [68Ga]Ga-PSMA-11 avidity demonstrated inferior treatment responses, suggesting that these PSMA-low lesions could serve as potential sources for disease recurrence. These findings highlight the importance of lesion-level [68Ga]Ga-PSMA-11 uptake as a valuable biomarker for predicting lesion-level treatment responses and subsequently influencing PFS outcomes.
Current clinical consensus favors PSMA-targeted imaging as the primary PET modality for hormone-sensitive prostate cancer, given its high tissue specificity and diagnostic sensitivity. Notably, our study demonstrated the complementary roles of [18F]FDG PET and [68Ga]Ga-PSMA-11 PET, as the combined imaging approach may improve risk stratification and guide individualized treatment strategies. In particular, the optimal use of chemotherapy in addition to ADT + ARPIs for patients remains contentious. Our findings emphasized the value of baseline whole-body [18F]FDG burden as a potential biomarker to refine chemotherapy decision-making, supporting a more individualized and effective treatment strategy.
This study has several limitations. First, our lesion-based analysis excluded bone metastatic lesions due to imaging and measurement restrictions, potentially introducing selection bias. In addition, given the retrospective study design, variations in follow-up durations among patients enrolled from 2021 to 2024 could introduce temporal bias, impacting survival outcomes. Second, tumor cell purity varies across prostate cancer lesions, reflecting biological heterogeneity. The purity denotes the proportion of malignant cells within a lesion relative to non-malignant components (stroma, benign glands, inflammatory/immune cells, hemorrhage, necrosis)38,39. Lower purity can dilute tumor-specific signal and thereby attenuate [68Ga]Ga-PSMA-11 uptake, potentially influencing lesion-level quantitative metrics. To address this concern, we conducted a preliminary analysis of biopsy samples collected before systemic treatment, observing no significant differences in tumor cell purity between lesions with high versus low [68Ga]Ga-PSMA-11 avidity. However, we also noticed that lesions with higher [68Ga]Ga-PSMA-11 avidity appeared to be more aggressive, as indicated by higher Gleason scores. Future studies integrating detailed pathological analyses are necessary to confirm these preliminary findings and enhance clinical applicability.
Conclusions
In this retrospective study, we conducted comprehensive patient- and lesion-based analyses to explore the distinct prognostic value of [18F]FDG and [68Ga]Ga-PSMA-11 PET imaging in advanced hormone-sensitive prostate cancer. Patient-based survival analysis and multivariable Cox-regression analysis identified higher whole-body [18F]FDG burden, including TLG and MTV, as significant predictors of shorter PFS. In contrast, lesion-based analysis revealed that higher [68Ga]Ga-PSMA-11 uptake at the lesion level was associated with better radiographic response following ADT-based systemic therapy. Collectively, these findings support the utility of dual-tracer PET imaging in guiding risk stratification and personalizing treatment strategies for patients with prostate cancer.
Data availability
Source data utilized in the analysis are provided in Supplementary Data 1. This retrospective study did not generate genetic or sequencing datasets. Therefore, mandatory deposition requirements for sequence/omics data do not apply. Due to patient privacy considerations, individual-level clinical records and raw imaging data are not publicly shared. However, de-identified data can be obtained from the corresponding author on reasonable request, subject to approval by the Ren Ji Hospital Ethics Committee and execution of a data use agreement. Please contact wqi@sjtu.edu.cn for the above requests. We will acknowledge receipt and respond to data access requests within 15 business days. If approved, de-identified data may be used for cross-cohort meta-analysis, independent replication, and publication, subject to the terms of the data use agreement. The data use agreement will prohibit any attempt to re-identify patients, onward sharing of the data to third parties, and use of the data beyond the approved research purpose.
References
Siegel, R. L., Giaquinto, A. N. & Jemal, A. Cancer statistics, 2024. CA Cancer J. Clin. 74, 12–49 (2024).
Sung, H. et al. Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 71, 209–249 (2021).
Fujita, K. & Nonomura, N. Role of androgen receptor in prostate cancer: a review. World J. Mens. Health 37, 288–295 (2019).
Wala, J., Nguyen, P. & Pomerantz, M. Early treatment intensification in metastatic hormone-sensitive prostate cancer. J. Clin. Oncol. 41, 3584–3590 (2023).
Logothetis, C. J. Improved outcomes in men with advanced prostate cancer. N. Engl. J. Med. 377, 388–390 (2017).
Fizazi, K. et al. Abiraterone plus prednisone in metastatic, castration-sensitive prostate cancer. N. Engl. J. Med. 377, 352–360 (2017).
Klotz, L. Apalutamide for metastatic, hormone-responsive prostate cancer. N. Engl. J. Med. 381, 84–86 (2019).
Nakazawa, M., Paller, C. & Kyprianou, N. Mechanisms of therapeutic resistance in prostate cancer. Curr. Oncol. Rep. 19, 13 (2017).
Heesterman B. L. et al. Increased incidence of primary metastatic prostate cancer in the era of PSMA PET/CT: a population-based analysis. Eur. J. Nucl. Med. Mol. Imaging https://doi.org/10.1007/s00259-025-07431-8 (2025).
Capasso, G., Stefanucci, A. & Tolomeo, A. A systematic review on the current status of PSMA-targeted imaging and radioligand therapy. Eur. J. Med. Chem. 263, 115966 (2024).
Roberts, M. J. et al. A prospective, multi-centre trial of PSMA-PET compared to FDG-PET for staging of newly diagnosed high risk prostate cancer. EJNMMI Res. 15, 92 (2025).
Ferraro, D. A. et al. Immunohistochemical PSMA expression patterns of primary prostate cancer tissue are associated with the detection rate of biochemical recurrence with 68Ga-PSMA-11-PET. Theranostics 10, 6082–6094 (2020).
Gossili, F. et al. The diagnostic accuracy of radiolabeled PSMA-ligand PET for tumour staging in newly diagnosed prostate cancer patients compared to histopathology: a systematic review and meta-analysis. Eur. J. Nucl. Med. Mol. Imaging 51, 281–294 (2023).
Fendler, W. P. et al. PSMA PET/CT: joint EANM procedure guideline/SNMMI procedure standard for prostate cancer imaging 2.0. Eur. J. Nucl. Med. Mol. Imaging 50, 1466–1486 (2023).
Cornford et al. EAU-EANM-ESTRO-ESUR-ISUP-SIOG Guidelines on Prostate Cancer—2024 Update. Part I: screening, diagnosis, and local treatment with curative intent. Eur. Urol. Elsevier 86, 148–163 (2024).
Gómez Rivas, J. et al. Prostate-specific membrane antigen positron emission tomography (PSMA-PET) in initial staging of prostate cancer patients: the beginning of a new era. Medicina 61, 924 (2025).
Mannweiler, S. et al. Heterogeneity of prostate-specific membrane antigen (PSMA) expression in prostate carcinoma with distant metastasis. Pathol. Oncol. Res. 15, 167–172 (2009).
Mulati, Y. et al. Characterizing PSMA heterogeneity in prostate cancer and identifying clinically actionable tumor associated antigens in PSMA negative cases. Sci. Rep. 15, 23902 (2025).
Bakht, M. K. & Beltran, H. Biological determinants of PSMA expression, regulation and heterogeneity in prostate cancer. Nat. Rev. Urol. 22, 26–45 (2025).
Dong, L. et al. Prospective evaluation of 68Ga-PSMA-11 PET/CT in Chinese men with biochemical recurrence after radical prostatectomy for prostate cancer: relationships between location of recurrence, time after prostatectomy, and serum PSA level. Med. Oncol. 37, 89 (2020).
Weiner, A. B. et al. Molecular hallmarks of prostate-specific membrane antigen in treatment-naïve prostate cancer. Eur. Urol. 86, 579–587 (2024).
Roy, J. et al. Monitoring PSMA responses to ADT in prostate cancer patient-derived xenograft mouse models using [18F]DCFPyL PET imaging. Mol. Imaging Biol. 23, 745–755 (2021).
Li, A. et al. Prognostic value of [18F]FDG PET/CT in metastatic hormone-sensitive prostate cancer at initial diagnosis: a retrospective cohort study. Ann. Med 56, 2411017 (2024).
Bauckneht, M. et al. The prognostic power of 18F-FDG PET/CT extends to estimating systemic treatment response duration in metastatic castration-resistant prostate cancer (mCRPC) patients. Prostate Cancer Prostatic Dis. Nat. Publ. Group 24, 1198–1207 (2021).
Jadvar, H. et al. Prediction of time to hormonal treatment failure in metastatic castration-sensitive prostate cancer with 18F-FDG PET/CT. J. Nucl. Med. 60, 1524–1530 (2019).
Iacovitti, C. M. et al. Diagnostic value of dual-tracer PET/CT with [18F]FDG and PSMA ligands in prostate cancer: an updated systematic review. Front. Med. 12, 1607227 (2025).
Bian, L. et al. Dual-Tracer 18 F-FDG and 68 Ga-PSMA PET/CT Imaging of Heterogeneous Phenotypes of Metastatic Castration-Resistant Prostate Cancer for Predicting Response to Novel Hormone Therapy. Clin. Nucl. Med 50, 143–149 (2025).
Kim, J. et al. Combination of [18F]FDG and [18F]PSMA-1007 PET/CT predicts tumour aggressiveness at staging and biochemical failure postoperatively in patients with prostate cancer. Eur. J. Nucl. Med. Mol. Imaging 51, 1763–1772 (2024).
Virgo, K. S., Rumble, R. B. & Talcott, J. Initial management of noncastrate advanced, recurrent, or metastatic prostate cancer: ASCO guideline update. J. Clin. Oncol. 41, 3652–3656 (2023).
Spratt, D. E. et al. Prostate Cancer, Version 3.2026, NCCN clinical practice guidelines in oncology. J. Natl. Compr. Canc Netw. 23, 469–493 (2025).
Hi S. et al. Trial design and objectives for castration-resistant prostate cancer: updated recommendations from the prostate cancer clinical trials working Group 3. Journal of clinical oncology: official journal of the American Society of Clinical Oncology [Internet]. J Clin Oncol; 2016 [cited 2025 Nov 18];34. https://doi.org/10.1200/JCO.2015.64.2702
Eisenhauer, E. A. et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur. J. Cancer 45, 228–247 (2009).
Schwarzenböck, S. M. et al. Prospective evaluation of [11C]Choline PET/CT in therapy response assessment of standardized docetaxel first-line chemotherapy in patients with advanced castration refractory prostate cancer. Eur. J. Nucl. Med. Mol. Imaging 43, 2105–2113 (2016).
Cheng, G., Alavi, A., Werner, T. J., Del Bello, C. V. & Akers, S. R. Serial changes of FDG uptake and diagnosis of suspected lung malignancy: a lesion-based analysis. Clin. Nucl. Med. 39, 147–155 (2014).
Kubota, K. et al. Lesion-based analysis of 18F-FDG uptake and 111In-Pentetreotide uptake by neuroendocrine tumors. Ann. Nucl. Med 28, 1004–1010 (2014).
Henríquez, I. et al. PSMA PET/CT SUVmax as a prognostic biomarker in patients with metachronous metastatic hormone-sensitive prostate cancer (mHSPC). Clin. Transl. Oncol. 27, 706–715 (2025).
Marchetti, A. et al. The prognostic role of 68GA-PSMA-PET/CT in metastatic hormone-sensitive prostate cancer: a preliminary analysis. Clinical Genitourinary Cancer [Internet]. Elsevier; 2025 [cited 2025 Aug 2];0. https://doi.org/10.1016/j.clgc.2025.102406
Aran, D., Sirota, M. & Butte, A. J. Systematic pan-cancer analysis of tumour purity. Nat. Commun. Nat. Publ. Group 6, 8971 (2015).
Oner, M. U. et al. Obtaining spatially resolved tumor purity maps using deep multiple instance learning in a pan-cancer study. Patterns 3, 100399 (2022).
Acknowledgements
This work was supported by National Natural Science Foundation of China (82373358), Shanghai Municipal Education Commission Grant Support (2023ZKZD23), Ren Ji Hospital (LYZXHXKT220845, PNO-0106, RJLJHZ-02), China Postdoctoral Science Foundation (2025M772209). We thank the staff of the Department of Nuclear Medicine, Ren Ji Hospital for assistance with radiochemistry and PET imaging. Figure 1B was created with BioRender.com (Agreement No. ZF290ATHTY).
Author information
Authors and Affiliations
Contributions
Contributions: A.L.: Writing–original draft, Data curation, Formal analysis, Investigation, Methodology, Visualization. H.W.: Writing - original draft, Formal analysis, Investigation, Methodology, Software, Validation. X.Z.: Formal analysis, Investigation, Methodology, Software, Visualization. Y.Z: Formal analysis, Validation. W.Z.: Formal analysis, Validation. K.S.: Validation, Funding acquisition. L.T.: Data curation. J.Y.: Data curation. B.L.: Data curation. R.S.: Investigation. X.L.: Investigation. X.C.: Visualization. Q.W.: Conceptualization, Writing - review & editing, Supervision. J.P.: Conceptualization, Writing - review & editing, Supervision, Resources, Funding acquisition. W.X.: Conceptualization, Supervision, Project administration, Funding acquisition.
Corresponding authors
Ethics declarations
Competing interests
The author declares no competing interests.
Peer review
Peer review information
Communications Medicine thanks Hina Shah and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Li, A., Wu, H., Zhou, X. et al. Distinct prognostic value of [18F]FDG PET and [68Ga]Ga-PSMA-11 PET in advanced hormone-sensitive prostate cancer. Commun Med 6, 164 (2026). https://doi.org/10.1038/s43856-026-01444-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s43856-026-01444-6



![Fig. 2: Kaplan-Meier curves of progression-free survival stratified by whole-body [18F]FDG and [68Ga]Ga-PSMA-11 uptake.](http://media.springernature.com/lw685/springer-static/image/art%3A10.1038%2Fs43856-026-01444-6/MediaObjects/43856_2026_1444_Fig2_HTML.png)

![Fig. 4: Correlation between lesion-level [18F]FDG and [68Ga]Ga-PSMA-11 uptake and radiographic response.](http://media.springernature.com/lw685/springer-static/image/art%3A10.1038%2Fs43856-026-01444-6/MediaObjects/43856_2026_1444_Fig4_HTML.png)
![Fig. 5: Representative cases showing the correlation between [68Ga]Ga-PSMA-11 uptake and short-term response to ADT + ARPIs.](http://media.springernature.com/lw685/springer-static/image/art%3A10.1038%2Fs43856-026-01444-6/MediaObjects/43856_2026_1444_Fig5_HTML.png)