Abstract
Background
The impact of antiviral therapies, including Paxlovid, on post-acute sequelae of COVID-19 (PASC) remains inconclusive.
Methods
We analyzed data from 19,413 patients (age > 18) from a validated PASC research cohort in New England who experienced at least one COVID-19 infection episode between January 1, 2022, and June 7, 2022, totaling 22,094 episodes. Multivariable logistic regression with inverse probability weights was used to infer the causal effects of Paxlovid treatment during acute infection and the risk of PASC overall (primary outcome), stratified by age group and organ system.
Results
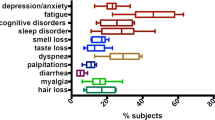
Across all age groups, Paxlovid shows no statistically significant effect in lowering overall PASC risk. Stratification by organ system reveals a 37% reduction in gastrointestinal PASC (OR: 0.63; 95% CI: [0.468, 0.850]; p < 0.05) but a 97.4% increase in the risk of eye and ear-related PASC (OR: 1.974; 95% CI: [1.048, 3.718]; p < 0.05). Among patients aged 65 to 75 years who were not hospitalized, Paxlovid is associated with a 16.8% reduction in PASC risk (OR: 0.832; 95% CI: [0.7, 0.989]; p < 0.05). No statistically significant effects is observed for other organ-specific outcomes.
Conclusions
Paxlovid demonstrates organ-specific effects on the risk of PASC, with a reduction in gastrointestinal symptoms and an increased risk of eye and ear-related symptoms. In older, non-hospitalized patients, Paxlovid modestly reduces overall PASC risk. These findings highlight the complexity of antiviral therapy’s long-term impact and underscore the need for further research to clarify the mechanisms underlying these outcomes.
Plain language summary
Long COVID refers to health problems that continue or appear weeks or months after a person recovers from COVID-19. In this study, we investigated whether taking the antiviral medication, Paxlovid, can reduce the risk of developing Long COVID. We analyzed medical records from over 19,000 adults treated at hospitals across New England. Overall, Paxlovid did not lower the total risk of developing Long COVID. However, it was linked with fewer digestive symptoms and showed modest benefit in older, non-hospitalized adults. At the same time, it was associated with a higher chance of eye or ear-related symptoms. These findings suggest that Paxlovid may help in specific situations, but more research is needed to understand who benefits most and why.
Similar content being viewed by others
Data availability
Due to privacy regulations and institutional review board approvals, patient-level data from this study cannot be shared, even in deidentified format. The combination of longitudinal clinical variables, rare diagnoses, and temporal information may pose a risk of re-identification. Access to the data is therefore restricted by institutional and legal requirements. Researchers who meet the criteria for access to confidential data may submit a request through Mass General Brigham Research & Innovation at: https://www.massgeneralbrigham.org/en/research-and-innovation/for-researchers-and-collaborators. Data requests will be reviewed in accordance with institutional policies, data use agreements, and applicable regulatory requirements.
Code availability
Statistical analyses were performed using R software, version 4.3.0 (R Foundation for Statistical Computing). The R packages Weightit38, cobalt39, and marginaleffects40 were used to compute entropy balancing weights, assess covariate balance, and estimate weighted odds ratios along with confidence intervals40,41. The computer codes can be accessed at https://github.com/clai-group/Long_covid_paxlovid_study?tab=readme-ov-file42.
References
Adjaye-Gbewonyo, D., Vahratian, A., Cria, G. P. & Bertolli, J. Long COVID in Adults: United States, 2022. (National Center for Health Statistics (U.S.), 2023). https://doi.org/10.15620/cdc:132417.
Hammond, J. et al. Oral nirmatrelvir for high-risk, nonhospitalized adults with Covid-19. N. Engl. J. Med. 386, 1397–1408 (2022).
Aggarwal, N. R. et al. Real-world use of nirmatrelvir-ritonavir in outpatients with COVID-19 during the era of omicron variants including BA.4 and BA.5 in Colorado, USA: a retrospective cohort study. Lancet Infect. Dis. 23, 696–705 (2023).
Xie, Y., Choi, T. & Al-Aly, Z. Association of treatment with nirmatrelvir and the risk of post-COVID-19 condition. JAMA Intern. Med. 183, 554–564 (2023).
Jiang, J. et al. Early use of oral antiviral drugs and the risk of post COVID-19 syndrome: a systematic review and network meta-analysis. J. Infect. 89, 106190 (2024).
Durstenfeld, M. S. et al. Association of nirmatrelvir for acute SARS-CoV-2 infection with subsequent Long COVID symptoms in an observational cohort study. J. Med. Virol. 96, e29333 (2024).
Ioannou, G. N. et al. Effectiveness of nirmatrelvir-ritonavir against the development of post-COVID-19 conditions among U.S. veterans: a target trial emulation. Ann. Intern. Med. 176, 1486–1497 (2023).
Bajema, K. L. et al. Effectiveness of COVID-19 treatment with nirmatrelvir-ritonavir or molnupiravir among U.S. veterans: target trial emulation studies with one-month and six-month outcomes. Ann. Intern. Med. 176, 807–816 (2023).
Fung, K. W., Baye, F., Baik, S. H. & McDonald, C. J. Nirmatrelvir and molnupiravir and post-COVID-19 condition in older patients. JAMA Intern. Med. 183, 1404–1406 (2023).
Wang, H. et al. Association of nirmatrelvir-ritonavir with post-acute sequelae and mortality in patients admitted to hospital with COVID-19: a retrospective cohort study. Lancet Infect. Dis. 24, 1130–1140 (2024).
Geng, L. N. et al. Nirmatrelvir-ritonavir and symptoms in adults with postacute sequelae of SARS-CoV-2 infection: the STOP-PASC randomized clinical trial: the STOP-PASC randomized clinical trial. JAMA Intern. Med. 184, 1024–1034 (2024).
Chuang, M. H. et al. Efficacy of nirmatrelvir and ritonavir for post-acute COVID-19 sequelae beyond 3 months of SARS-CoV-2 infection. J. Med. Virol. 95, e28750 (2023).
Wang, F. et al. Real-World Effectiveness of Nirmatrelvir in Protecting Long COVID for Outpatient Adult Patients-A Large-Scale Observational Cohort Study from the RECOVER Initiative. Res sq https://doi.org/10.21203/rs.3.rs-4536807/v1 (2024).
Shen Y. et al. Assessment of Nirmatrelvir-Ritonavir Effects on Acute and Post-Acute COVID-19 Illness in US Adolescents: Target Trial Emulation. MedRXiv https://doi.org/10.1101/2025.04.02.25324861 (2025).
Sharif-Askari, F. S. et al. Nirmatrelvir plus ritonavir reduces COVID-19 hospitalization and prevents long COVID in adult outpatients. Sci. Rep. 14, 25901 (2024).
Sk Abd Razak, R. et al. Post-COVID syndrome prevalence: a systematic review and meta-analysis. BMC Public Health 24, 1785 (2024).
Azhir, A. et al. Precision phenotyping for curating research cohorts of patients with unexplained post-acute sequelae of COVID-19. Med https://doi.org/10.1016/j.medj.2024.10.009 (2024).
WHO. Coronavirus disease (COVID-19): Post COVID-19 condition. Accessed 2023. https://www.who.int/news-room/questions-and-answers/item/coronavirus-disease-(covid-19)-post-covid-19-condition.
Ely, E. W., Brown, L. M. & Fineberg, H. V. National academies of sciences, engineering, and medicine committee on examining the working definition for long covid. Long Covid defined. N. Engl. J. Med. 391, 1746–1753 (2024).
Strasser, Z. H., Greifer, N., Hadavand, A., Murphy, S. N. & Estiri, H. Estimates of SARS-CoV-2 Omicron BA.2 subvariant severity in New England. JAMA Netw. Open 5, e2238354 (2022).
Yahav, D. et al. Definitions for coronavirus disease 2019 reinfection, relapse and PCR re-positivity. Clin. Microbiol. Infect. 27, 315–318 (2021).
Soriano, J. B., Murthy, S., Marshall, J. C., Relan, P. & Diaz, J. V. WHO clinical case definition working group on post-COVID-19 condition. A clinical case definition of post-COVID-19 condition by a Delphi consensus. Lancet Infect. Dis. 22, e102–e107 (2022).
Gasparini, A. comorbidity: An R package for computing comorbidity scores. J. Open Source Softw. 3, 648 (2018).
Hainmueller, J. Entropy balancing for causal effects: a multivariate reweighting method to produce balanced samples in observational studies. Polit. Anal. 20, 25–46 (2012).
Xu, E., Xie, Y. & Al-Aly, Z. Long-term gastrointestinal outcomes of COVID-19. Nat. Commun. 14, 983 (2023).
Zuo, W. et al. The persistence of SARS-CoV-2 in tissues and its association with long COVID symptoms: a cross-sectional cohort study in China. Lancet Infect. Dis. 24, 845–855 (2024).
Nikolich-Žugich, J. The twilight of immunity: emerging concepts in aging of the immune system. Nat. Immunol. 19, 10–19 (2018).
Liu, Z. et al. Immunosenescence: molecular mechanisms and diseases. Sig. Transduct. Target. Ther. 8, 200 (2023).
Santoro, A., Bientinesi, E. & Monti, D. Immunosenescence and inflammaging in the aging process: age-related diseases or longevity? Ageing Res. Rev. 71, 101422 (2021).
Sun, G., Lin, K., Ai, J. & Zhang, W. The efficacy of antivirals, corticosteroids, and mAbs as acute COVID treatments in reducing the incidence of long COVID’-Author’s reply. Clin. Microbiol. Infect. 30, 1618–1619 (2024).
Berry, M. et al. Risk of long COVID in hospitalized individuals treated with remdesivir for acute COVID-19. Sci. Rep. 15, 27441 (2025).
Islam, M. S., Wang, Z., Abdel-Mohsen, M., Chen, X. & Montaner, L. J. Tissue injury and leukocyte changes in post-acute sequelae of SARS-CoV-2: review of 2833 post-acute patient outcomes per immune dysregulation and microbial translocation in long COVID. J. Leukoc. Biol. 113, 236–254 (2023).
Greenhalgh, T., Sivan, M., Perlowski, A. & Nikolich, J. Ž Long COVID: a clinical update. Lancet 404, 707–724 (2024).
Sherif, Z. A., Gomez, C. R., Connors, T. J., Henrich, T. J. & Reeves, W. B. Pathogenic mechanisms of post-acute sequelae of SARS-CoV-2 infection (PASC). Elife 12, e86002 (2023).
Mantovani, A. et al. Covid-19 Commission of the Accademia Nazionale dei Lincei Cappuccinelli Pietro 13 Fitzgerald Garrett 14 Bacci Massimo Livi 15 Melino Gennaro 16 Parisi Giorgio 17 Rappuoli Rino 18 19 Rezza Giovanni 20 Vineis Paolo 21. Long Covid: where we stand and challenges ahead. Cell Death Differ. 29, 1891–1900 (2022).
Frontera, J. A. et al. Post-acute sequelae of COVID-19 symptom phenotypes and therapeutic strategies: a prospective, observational study. Plos ONE 17, e0275274 (2022).
Sawano, M. et al. Nirmatrelvir–ritonavir versus placebo–ritonavir in individuals with long COVID in the USA (PAX LC): a double-blind, randomised, placebo-controlled, phase 2, decentralised trial. Lancet Infect. Dis. 25, 936–946 (2025).
Greifer, N. Weighting for covariate balance in observational studies [R package WeightIt version 0.13.1]. Published online 2022. Accessed July 7, 2022. https://CRAN.R-project.org/package=WeightIt.
Greifer, N. Covariate balance tables and plots: a guide to the cobalt package. 2024. Accessed May 31, 2024. https://cloud.r-project.org/web/packages/cobalt/vignettes/cobalt.html.
Arel-Bundock, V., Greifer, N. & Bacher, E. Marginaleffects: Predictions, Comparisons, Slopes, Marginal Means, and Hypothesis Tests.; 2024. Accessed May 31, 2024. https://CRAN.R-project.org/package=marginaleffects.
Survey: analysis of complex survey samples. Comprehensive R Archive Network (CRAN). Accessed 2022. https://cran.r-project.org/web/packages/survey/index.html.
Azhir, A., Cheng, J. & Estiri, H. Paxlovid shows organ-specific and age-specific impacts on risk of developing post-acute sequelae of COVID-19. Version 1.0.1. Zenodo https://doi.org/10.5281/zenodo.18749575 (2026).
Acknowledgements
This study has been supported by grants from the National Institute of Allergy and Infectious Diseases (NIAID) R01AI165535.
Author information
Authors and Affiliations
Contributions
A.A., H.E., and J.C. designed the study. A.A. and J.C. developed the computational code. H.E., S.M., and A.A. created the initial statistical model. H.E., S.M., and J.T. provided the source data needed for the analysis. H.E. and S.M. oversaw the completion of the project. J.C. generated all figures. All authors contributed to writing the manuscript, revisions, and final submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Medicine thanks Yishan Shen and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Azhir, A., Cheng, J., Tian, J. et al. Paxlovid shows organ-specific and age-specific impacts on risk of developing post-acute sequelae of COVID-19. Commun Med (2026). https://doi.org/10.1038/s43856-026-01535-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s43856-026-01535-4