Abstract
Background
Bempedoic acid is an oral cholesterol-lowering medication used to reduce low-density lipoprotein cholesterol (LDL-C), particularly in patients who require additional lipid lowering. Clinical trials have shown that bempedoic acid increases serum uric acid levels, but the implications of this effect in real-world clinical practice, including the need for anti-gout treatment, are not well defined.
Methods
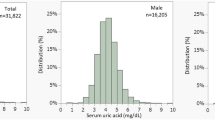
We conducted a retrospective cohort study using a large, multi-institutional U.S. electronic health record database. Adult patients with hyperlipidemia who have newly initiated bempedoic acid were compared with those initiating ezetimibe. Propensity score matching was used to balance baseline characteristics, yielding 7676 patients in each group. The primary outcome was incident hyperuricemia, defined as serum uric acid >7.0 mg/dL. A secondary outcome was initiation of anti-gout therapy.
Results
During 12 months of follow-up, hyperuricemia occurs more frequently in patients treated with bempedoic acid than in those receiving ezetimibe (hazard ratio [HR] 1.94; 95% confidence interval [CI] 1.58–2.37; p = 0.008). In contrast, initiation of anti-gout therapy does not differ between groups (HR 1.06; 95% CI 0.86–1.29; p = 0.59). Longitudinal changes in lipid parameters, inflammatory markers, and glycemic measures are generally similar between groups.
Conclusions
In real-world clinical practice, bempedoic acid use is associated with a higher incidence of laboratory-defined hyperuricemia compared with ezetimibe, but this increase does not translate into greater use of anti-gout medications. These findings support the continued use of bempedoic acid for additional LDL-C lowering, with awareness of its effect on serum uric acid.
Plain language summary
High cholesterol is a major cause of heart disease, and for some people current medicines aren’t sufficient to lower their cholesterol. Bempedoic acid is a newer drug that can help reduce certain kinds of cholesterol, but it may raise levels of a compound called uric acid, which can lead to a condition known as gout. We studied over 15,000 patients in U.S. healthcare systems to see whether this medicine increases the risk of hyperuricemia or gout compared with ezetimibe. We found that bempedoic acid caused higher uric acid levels, but most patients did not need gout treatment. This means that while uric acid may increase, serious symptoms are uncommon. These findings provide real-world information on how bempedoic acid affects uric acid levels and the need for gout treatment in clinical practice.
Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the TriNetX Research Network (TriNetX LLC), but restrictions apply to the availability of these data, which were used under license for the current study and are not publicly available. Researchers may obtain access to the TriNetX platform through institutional subscription and approval by TriNetX.
References
Nelson, R. H. Hyperlipidemia as a Risk Factor for Cardiovascular Disease. Prim. Care: Clin. Off. Pract. 40, 195–211 (2013).
Rosenblit, P. D. Extreme Atherosclerotic Cardiovascular Disease (ASCVD) Risk Recognition. Curr. Diab Rep. 19, 61 (2019).
Wilson, P. W. F. et al. Systematic Review for the 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the Management of Blood Cholesterol: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation 139, (2019).
Liu, P. Y. Reappraisal of the New Clinical Pathway National Consensus on Lipid Profile in Taiwan 2025: Where Do We Stand Now? Acta Cardiol Sin. 41, 166–168 (2025).
Stroes, E. S. et al. Statin-associated muscle symptoms: impact on statin therapy—European Atherosclerosis Society Consensus Panel Statement on Assessment, Aetiology and Management. Eur. Heart J. 36, 1012–1022 (2015).
Di Minno, A. et al. Efficacy and Safety of Bempedoic Acid in Patients With Hypercholesterolemia: Systematic Review and Meta-Analysis of Randomized Controlled Trials. J. Am. Heart Assoc. 9, e016262 (2020).
Mach, F. et al. ESC/EAS Scientific Document Group. 2025 Focused Update of the 2019 ESC/EAS Guidelines for the management of dyslipidaemias. Eur. Heart J. 46, 4359–4378 (2025).
Ray, K. K. et al. Safety and Efficacy of Bempedoic Acid to Reduce LDL Cholesterol. N. Engl. J. Med. 380, 1022–1032 (2019).
Yarrarapu, S. N. S. et al. Comprehensive review of statin-intolerance and the practical application of Bempedoic Acid. J. Cardiol. 84, 22–29 (2024).
Nissen, S. E. et al. Bempedoic Acid and Cardiovascular Outcomes in Statin-Intolerant Patients. N. Engl. J. Med. 388, 1353–1364 (2023).
Laufs, U. et al. Efficacy and Safety of Bempedoic Acid in Patients With Hypercholesterolemia and Statin Intolerance. J. Am. Heart Assoc. 8, e011662 (2019).
Biolo, G. et al. Mechanism of action and therapeutic use of bempedoic acid in atherosclerosis and metabolic syndrome. Front. Cardiovasc. Med. 9, 1028355 (2022).
Richette, P. et al. 2016 updated EULAR evidence-based recommendations for the management of gout. Ann. Rheum. Dis. 76, 29–42 (2017).
Zhang, S. et al. Hyperuricemia and Cardiovascular Disease. Curr. Pharm. Des. 25, 700–709 (2019).
D’Agostino, R. B., Kannel, W. B., Stepanians, M. N. & D’Agostino, L. C. A comparison between lovastatin and gemfibrozil in the treatment of primary hypercholesterolemia. Am. J. Cardiol. 69, 28–34 (1992).
Davidson, M. H. & Toth, P. P. Comparative effects of lipid-lowering therapies. Prog. Cardiovasc Dis. 47, 73–104 (2004).
Leung, N., Yip, K., Pillinger, M. H. & Toprover, M. Lowering and Raising Serum Urate Levels: Off-Label Effects of Commonly Used Medications. Mayo Clin. Proc. 97, 1345–1362 (2022).
Ballantyne, C. M. et al. Role of Bempedoic Acid in Clinical Practice. Cardiovasc Drugs Ther. 35, 853–864 (2021).
De Filippo, O. et al. Safety and efficacy of bempedoic acid: a systematic review and meta-analysis of randomised controlled trials. Cardiovasc Diabetol. 22, 324 (2023).
Khan, M. U., Khan, M. Z., Munir, M. B., Balla, S. & Khan, S. U. Meta-analysis of the Safety and Efficacy of Bempedoic Acid. Am. J. Cardiol. 131, 130–132 (2020).
FitzGerald, J. D. et al. 2020 American College of Rheumatology Guideline for the Management of Gout. Arthritis Care Res (Hoboken) 72, 744–760 (2020).
Ray, K. K. et al. Association of Uric Acid-Lowering Therapies on Gout Frequency with Bempedoic Acid. Clin. Insights CLEAR Outcomes JACC Adv. 4, 102207 (2025).
Wang, X. et al. Novel insights into post-marketing AEs associated with bempedoic acid: a comprehensive analysis utilizing the FAERS database. Expert Opin. Drug Saf. 1–8. https://doi.org/10.1080/14740338.2025.2468868. Online ahead of print. (2025).
Wang, J., Tsai, K. W., Lu, C. L. & Lu, K. C. Analyzing clinical laboratory data outcomes in retrospective cohort studies using TriNetX. Biochem. Med. (Zagreb) 35, 030502 (2025).
Acknowledgements
The grant assistance from the National Cheng Kung University Hospital, Tainan, Taiwan, Republic of China is gratefully acknowledged. This study was supported in part by grants from the National Science and Technology Council of Taiwan (NSTC 114-2628-B-006-005-; NSTC 114-2314-B-006-089-MY3), the National Health Research Institutes of Taiwan (NHRI-14A1-CG-CO-04-2225-1) to ECCL and NCKUH-11504005 from National Cheng Kung University Hospital to PYL. Additionally, this research was supported in part by Higher Education Sprout Project, Ministry of Education to the Headquarters of University Advancement at National Cheng Kung University (NCKU). The funders had no role in considering the study design or in the collection, analysis, interpretation of data, writing of the report, or decision to submit the article for publication.
Author information
Authors and Affiliations
Contributions
Po-Hsueh Su: Conceptualization, methodology, formal analysis, writing - original draft, and writing - review & editing. Daniel Hsiang-Te Tsai: Methodology, software, investigation, and formal analysis. Miyuki Hsing-Chun Hsieh: Methodology, software, investigation, and formal analysis. Tharmaraj Vairaperumal: Writing - review & editing. Edward Chia-Cheng Lai: Supervision and acquisition of funding. Ping-Yen Liu: Conceptualization, supervision, and funding acquisition.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Consent for publication
All the authors listed consent to the submission.
Peer review
Peer review information
Communications Medicine thanks the anonymous reviewers for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Su, PH., Tsai, D.HT., Hsieh, M.HC. et al. Hyperuricemia risk in bempedoic acid-treated hyperlipidemic patients. Commun Med (2026). https://doi.org/10.1038/s43856-026-01545-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s43856-026-01545-2