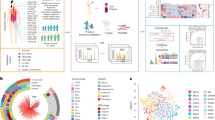
Abstract
Background
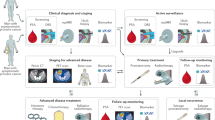
Radiotherapy induces systemic changes beyond the targeted tumour site, yet the biological mechanisms driving these effects remain poorly understood. This study aims to longitudinally profile acute systemic plasma proteomic responses to radiotherapy in patients with prostate, bladder, and head and neck cancers, providing insight into shared and tumour-specific effects and identifying biomarkers predictive of treatment-related toxicities.
Methods
We apply our previously developed Nano-proteomics workflow to comprehensively analyse longitudinal weekly plasma samples collected before and during radiotherapy. We perform mass spectrometry-based differential protein analysis to identify acute changes of the plasma proteome and enriched biological pathways involved. In the prostate cohort, we associate plasma proteomics with clinical toxicity outcomes.
Results
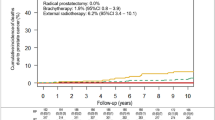
Our data indicate that the most significant systemic proteomic changes occur within the first two weeks of radiotherapy, highlighting a critical period for biomarker identification. Across all patient cohorts, we observe common biological responses: rapid activation of inflammatory and immune pathways, followed by structural reorganisation and immune resolution, regardless of tumour type or concurrent treatments. Despite this shared acute response, distinct protein mediators are found to be dysregulated in a tumour-specific manner. In the prostate cancer cohort, plasma profiling at baseline, one week after the initiation of radiotherapy, and at the end of radiotherapy, results in the identification of 28, 29, and 20 proteins, respectively, that are associated with subsequent bowel and urinary toxicities.
Conclusions
This study underscores the value of longitudinal proteomics in uncovering systemic effects of radiotherapy and supports the potential of plasma proteomic biomarkers to identify patients at increased risk of radiotherapy-induced toxicity, paving the way for personalised treatment strategies.
Plain language summary
Radiotherapy targets tumour cells but also triggers systemic effects beyond the irradiated site. To characterise these responses, we analysed blood samples from patients with prostate, bladder, and head and neck cancers collected before and during treatment. Using a nanoparticle-enabled proteomics workflow, we profiled weekly changes in circulating proteins and observed the strongest systemic alterations within the first two weeks of radiotherapy. While many systemic responses were shared across cancer types, tumour-specific patterns also emerged. In the prostate cancer cohort, longitudinal plasma profiles revealed proteins linked to later bowel and urinary toxicities, suggesting potential predictive biomarkers. Overall, our findings underscore the systemic nature of radiotherapy and highlight longitudinal plasma proteomics as a valuable tool for biomarker discovery and personalised treatment.
Similar content being viewed by others
Data availability
The mass spectrometry proteomics data have been deposited to the ProteomeXchange Consortium via the PRIDE [1] partner repository with the dataset identifier PXD071492 and 10.6019/PXD071492. The source data for Fig. 3(a-b), Fig. 4g–I and for the supporting Figures S1b, S2b, S3a–c, and S5a are provided in Supplementary Data set 2.
Code availability
The underlying code for this study is available at https://github.com/assisalam/nanoOmic/ [github.com]. Computational method summary table is provided in Supplementary Data 5.
References
Jelonek, K., Pietrowska, M. & Widlak, P. Systemic effects of ionizing radiation at the proteome and metabolome levels in the blood of cancer patients treated with radiotherapy: the influence of inflammation and radiation toxicity. Int. J. Radiat. Biol. 93, 683–696 (2017).
Balázs, K. et al. Radiotherapy-induced changes in the systemic immune and inflammation parameters of head and neck cancer patients. Cancers 11, 1324 (2019).
Chen, H. H. W. & Kuo, M. T. Improving radiotherapy in cancer treatment: promises and challenges. Oncotarget 8, 62742–62758 (2017).
Vozenin, M.-C., Bourhis, J. & Durante, M. Towards clinical translation of FLASH radiotherapy. Nat. Rev. Clin. Oncol. 19, 791–803 (2022).
Ouerhani, A., Chiappetta, G., Souiai, O., Mahjoubi, H. & Vinh, J. Investigation of serum proteome homeostasis during radiation therapy by a quantitative proteomics approach. Biosci. Rep. 39, 1–13 (2019).
Carvalho, H., de, A. & Villar, R. C. Radiotherapy and immune response: the systemic effects of a local treatment. Clinics 73, e557s (2018).
Guckenberger, M. Systemic effects of local radiotherapy: first clinical evidence. Strahlentherapie und Onkol. 197, 458–460 (2021).
Candéias, S. M. & Testard, I. The many interactions between the innate immune system and the response to radiation. Cancer Lett. 368, 173–178 (2015).
Lumniczky, K. & Sáfrány, G. The impact of radiation therapy on the antitumor immunity: Local effects and systemic consequences. Cancer Lett. 356, 114–125 (2015).
De Ruysscher, D. et al. Blood-based biomarkers for precision medicine in lung cancer: Precision radiation therapy. Transl. Lung Cancer Res. 6, 661–669 (2017).
Ajdari, A. et al. Toward Personalized Radiation Therapy of Liver Metastasis: Importance of Serial Blood Biomarkers. JCO Clin. Cancer Informatics 315–325, https://doi.org/10.1200/cci.20.00118 (2021).
Cai, X. W. et al. Baseline plasma proteomic analysis to identify biomarkers that predict radiation-induced lung toxicity in patients receiving radiation for non-small cell lung cancer. J. Thorac. Oncol. 6, 1073–1078 (2011).
Zebene, E. D. et al. Serum-based proteomic approach to identify clinical biomarkers of radiation exposure. Cancers 17, 1–21 (2025).
Widlak, P. et al. Serum proteome signature of radiation response: upregulation of inflammation-related factors and downregulation of apolipoproteins and coagulation factors in cancer patients treated with radiation therapy—a pilot study. Int. J. Radiat. Oncol. Biol. Phys. 92, 1108–1115 (2015).
Cai, X. W. et al. Plasma proteomic analysis may identify new markers for radiation-induced lung toxicity in patients with non-small-cell lung cancer. Int. J. Radiat. Oncol. Biol. Phys. 77, 867–876 (2010).
Gkikoudi, A. et al. Molecular biomarkers for predicting cancer patient radiosensitivity and radiotoxicity in clinical practice. Appl. Sci. 13, (2023).
Bleaney, C. W. et al. Clinical biomarkers of tumour radiosensitivity and predicting benefit from radiotherapy: a systematic review. Cancers 16, 1942 (2024).
WidŁak, P. et al. Radiation-related changes in serum proteome profiles detected by mass spectrometry in blood of patients treated with radiotherapy due to larynx cancer. J. Radiat. Res. 52, 575–581 (2011).
Widłak, P. et al. Radiotherapy-related changes in serum proteome patterns of head and neck cancer patients; the effect of low and medium doses of radiation delivered to large volumes of normal tissue. J. Transl. Med. 11, 1–9 (2013).
Truscott, T. G. & Sinclair, R. S. Advances in Experimental Medicine and Biology. International Journal of Radiation Biology vol. 44 (1983).
Rifai, N., Gillette, M. A. & Carr, S. A. Protein biomarker discovery and validation: The long and uncertain path to clinical utility. Nat. Biotechnol. 24, 971–983 (2006).
Christensen, E. et al. Longitudinal cytokine expression during IMRT for prostate cancer and acute treatment toxicity. Clin. Cancer Res. 15, 5576–5583 (2009).
Rübe, C. E. et al. Cytokine plasma levels: reliable predictors for radiation pneumonitis? PLoS One 3, e2898 (2008).
Walker, M. J. et al. Discovery and validation of predictive biomarkers of survival for non-small cell lung cancer patients undergoing radical radiotherapy: two proteins with predictive value. EBioMedicine 2, 841–850 (2015).
Cohen, I. R., Lajtha, A., Lambris, J. D. & Paoletti, R. Radiation Proteomics, Chapter 4, Serum and Plasma Proteomics and Its Possible Use as Detector and Predictor of Radiation Diseases. Proteomics vol. 13 (2013).
Hadjidemetriou, M., Al-ahmady, Z., Buggio, M., Swift, J. & Kostarelos, K. A novel scavenging tool for cancer biomarker discovery based on the blood-circulating nanoparticle protein corona. Biomaterials 188, 118–129 (2019).
Hadjidemetriou, M. et al. The human in vivo biomolecule corona onto pegylated liposomes: a proof-of-concept clinical study. Adv. Mater. 31, e1803335 (2019).
Hadjidemetriou, M. et al. Nano-scavengers for blood biomarker discovery in ovarian carcinoma. Nano Today 34, 100901 (2020).
Hadjidemetriou, M., Mahmoudi, M. & Kostarelos, K. In vivo biomolecule corona and the transformation of a foe into an ally for nanomedicine. Nat. Rev. Mater.2024 94 9, 219–222 (2024).
Liu, X. et al. Plasma-to-tumour tissue integrated proteomics using nano-omics for biomarker discovery in glioblastoma. Nat. Commun. 16, 3412 (2025).
Silva, J. C., Gorenstein, M. V., Li, G. Z., Vissers, J. P. C. & Geromanos, S. J. Absolute quantification of proteins by LCMSE: a virtue of parallel MS acquisition. Mol. Cell. Proteom. 5, 144–156 (2006).
Argelaguet, R. et al. Multi-omics factor analysis—a framework for unsupervised integration of multi-omics data sets. Mol. Syst. Biol. 14, 1–13 (2018).
Byrnes, H., Maxim, H. H. & Norris, J. M. Identifying temporal and spatial patterns of variation from multi_modal data using MEFISTO. Mod. Lang. J. 94, 1–202 (2010).
Papafilippou, L., Claxton, A., Dark, P., Kostarelos, K. & Hadjidemetriou, M. Protein corona fingerprinting to differentiate sepsis from non-infectious systemic inflammation. Nanoscale 12, 10240–10253 (2020).
Gardner, L., Kostarelos, K., Mallick, P., Dive, C. & Hadjidemetriou, M. Nano-omics: nanotechnology-based multidimensional harvesting of the blood-circulating cancerome. Nat. Rev. Clin. Oncol. 19, 551–561 (2022).
Hadjidemetriou, M. et al. In vivo biomolecule corona around blood-circulating, clinically used and antibody-targeted lipid bilayer nanoscale vesicles. ACS Nano 9, 8142–8156 (2015).
Hadjidemetriou, M., Al-Ahmady, Z. & Kostarelos, K. Time-evolution of in vivo protein corona onto blood-circulating PEGylated liposomal doxorubicin (DOXIL) nanoparticles. Nanoscale 8, 6948–6957 (2016).
Ward, B. et al. Deep plasma proteomics with data-independent acquisition: clinical study protocol optimization with a COVID-19 cohort. J. Proteome Res. 23, 3806–3822 (2024).
Kyle W., Joel E. Tepper, M. Radiation therapy- associated toxicity: etiology, management, and prevention. at (2021).
Chugh, R., Bisht, Y. S., Nautiyal, V. & Jindal, R. Factors influencing the severity of acute radiation-induced skin and mucosal toxicity in head and neck cancer. Cureus 13, e18147 (2021).
Wijerathne, H. et al. Mechanisms of radiation-induced endothelium damage: emerging models and technologies Harshani. Radiother. Oncol. 158, 21–32 (2022).
Constanzo, J., Faget, J., Ursino, C., Badie, C. & Pouget, J. P. Radiation-induced immunity and toxicities: the versatility of the cGAS-STING pathway. Front. Immunol. 12, 1–17 (2021).
Venkatesulu, B. P. et al. Radiation-induced endothelial vascular injury: a review of possible mechanisms. JACC Basic Transl. Sci. 3, 563–572 (2018).
Koch, A., Reinhardt, P., Elicin, O., Aebersold, D. M. & Schanne, D. H. Predictive biomarkers of radiotherapy- related dermatitis, xerostomia, mucositis and dysphagia in head and neck cancer: a systematic review. Radiother. Oncol. 203, 110689 (2025).
Azria, D. et al. Data-based radiation oncology: Design of clinical trials in the toxicity biomarkers era. Front. Oncol. 7, 1–11 (2017).
Ree, A. H. & Redalen, K. R. Personalized radiotherapy: concepts, biomarkers and trial design. Br. J. Radiol. 88, 20150009 (2015).
Lacombe, J., Azria, D., Mange, A. & Solassol, J. Proteomic approaches to identify biomarkers predictive of radiotherapy outcomes. Expert Rev. Proteom. 10, 33–42 (2013).
Nasser, N. J., Klein, J. & Agbarya, A. Markers of toxicity and response to radiation therapy in patients with prostate cancer. Adv. Radiat. Oncol. 6, 100603 (2021).
Kovacs, C. J. et al. Cytokine profiles in patients receiving wide-field + prostate boost radiotherapy (xRT) for adenocarcinoma of the prostate. Cytokine 23, 151–163 (2003).
Hille, A. et al. Faecal calprotectin and lactoferrin values during irradiation of prostate cancer correlate with chronic radiation proctitis: results of a prospective study. Scand. J. Gastroenterol. 44, 939–946 (2009).
Onal, C. et al. Plasma citrulline levels predict intestinal toxicity in patients treated with pelvic radiotherapy. Acta Oncol. 50, 1167–1174 (2011).
Meehan, J. et al. Precision medicine and the role of biomarkers of radiotherapy response in breast cancer. Front. Oncol. 10, 1–16 (2020).
Acknowledgements
The clinical research was carried out at the National Institute for Health and Care Research (NIHR) Manchester Biomedical Research Centre (BRC) (NIHR203308). The proteomic analyses and DCW were supported by Cancer Research UK RadNet Manchester [C1994/A28701]. We thank the Faculty of Biology, Medicine and Health, University of Manchester Biological Mass Spectrometry Facility (Bio-MS) staff for their support. We thank the Manchester Cancer Research Centre biobank (MCRC biobank). E.C., CGQ & DCW are funded by the NIHR Biomedical Research Centre (NIHR203308). A.C. and C.W. are supported by the NIHR Manchester Biomedical Research Centre. We would like to thank the research nurse team at the Christie Hospital.
Author information
Authors and Affiliations
Contributions
H.Ab. initiated and performed the proteomic experiments, analysed the data, and wrote the manuscript. S.A., X.L., and C.G. contributed to the proteomics data analysis. E.Ch. and T.L. provided clinical samples and clinical information. C.W. designed and was Chief Investigator of the clinical study. A.C. was involved in patient recruitment. K.J.W., E.C., K.K., A.C., D.W., and C.W. supervised the work and contributed to the design of the study, interpretation of the data and the revising of the manuscript. M.H. contributed to the conceptualization of the study, designed the experiments, supervised the execution of the proteomics experimental work and data analysis, contributed to the interpretation of the data, and prepared and reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Medicine thanks Karol Jelonek, Javier F. Torres-Roca and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Abumanhal-Masarweh, H., Assi, S.A., Liu, X. et al. Longitudinal plasma nano-proteomics reveals acute systemic responses to radiotherapy and predictive biomarkers of late toxicity. Commun Med (2026). https://doi.org/10.1038/s43856-026-01552-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s43856-026-01552-3