Abstract
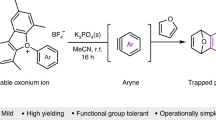
Despite numerous studies of trialkyloxonium ions in the literature, investigations into the chemistry of allylic, benzylic, propargylic and allenylic oxonium ions are rare. Existing reports on well-characterized allylic and benzylic oxonium ions invariably construct these species based on constrained tricyclic oxatriquinane or oxatriquinacene scaffolds, with only limited studies reported on unconstrained benzylic oxonium ions. Here we report an investigation on a collection of allylic, benzylic and hitherto unknown propargylic and allenylic oxonium ions prepared on unconstrained scaffolds by a general, modular and unified strategy. Permutation of the substitution pattern of these oxonium ions allowed the extension of the strategy for the syntheses of various doubly substituted oxonium ions. Most of these oxonium ions could be characterized at room temperature by NMR spectroscopy, and a series of unexpected reactions and chemical behaviours pertinent to these species are briefly described.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
The data (experimental procedures, characterization data, NMR spectra, and computational methods and results for oxonium ion characterization) supporting the findings of this study are available within the Article and its Supplementary Information.
References
Meerwein, H., Hinz, G., Hofmann, P., Kroning, E. & Pfeil, E. Über Tertiäre Oxoniumsalze, I. J. Prakt. Chem. 147, 257–285 (1937).
Gargiulo, R. J. & Tarbell, D. S. Cyclic oxonium ions and related structures. Proc. Natl Acad. Sci. USA 62, 52–55 (1969).
Lambert, J. B. & Johnson, D. H. The stereochemistry and inversion of trivalent oxygen. J. Am. Chem. Soc. 90, 1349–1350 (1968).
Klages, F., Gordon, J. E. & Jung, H. A. NMR- und IR-Spektren von Oxoniumsalzen. Chem. Ber. 98, 3748–3756 (1965).
Meerwein, H., Battenberg, E. & Gold, H. Über Tertiäre Oxoniumsalze, II. J. Prakt. Chem. 154, 83–156 (1939).
Etzkorn, M. et al. 1-Oxoniaadamantane. Eur. J. Org. Chem. 2008, 4555–4558 (2008).
Olah, G. A., Doggweiler, H., Felberg, J. D. & Frohlich, S. Onium ions. 33. (Trimethylsilyl)- and [(trimethylsilyl)methyl]oxonium and halonium ions. J. Org. Chem. 50, 4847–4851 (1985).
Klages, F., Jung, H. A. & Wagner, H. Synthese und Eigenschaften eines bicyclischen Oxoniumsalzes. Chem. Ber. 98, 3757–3764 (1965).
Smith, S., Schultz, W. J. & Newmark, R. A. in Ring-Opening Polymerization, ACS Symposium Series Vol. 59 (eds Saegusa, T. & Goethals, E.) 13–23 (American Chemical Society, 1977).
Mascal, M., Hafezi, N., Meher, N. K. & Fettinger, J. C. Oxatriquinane and oxatriquinacene: extraordinary oxonium ions. J. Am. Chem. Soc. 130, 13532–13533 (2008).
Stoyanov, E. S. et al. The R3O+···H+ hydrogen bond: toward a tetracoordinate oxadionium(2+) ion. J. Am. Chem. Soc. 134, 707–714 (2012).
Gunbas, G. et al. Extreme oxatriquinanes and a record C–O bond length. Nat. Chem. 4, 1018–1023 (2012).
Gunbas, G., Sheppard, W. L., Fettinger, J. C., Olmstead, M. M. & Mascal, M. Extreme oxatriquinanes: structural characterization of alpha-oxyoxonium species with extraordinarily long carbon–oxygen bonds. J. Am. Chem. Soc. 135, 8173–8176 (2013).
Suzuki, H. & Muratake, H. Functionalized oxatriquinanes and their structural equilibrium in protic solvent. Chem. Pharm. Bull. 62, 921–926 (2014).
Chan, H. S. S., Nguyen, Q. N. N., Paton, R. S. & Burton, J. W. Synthesis, characterization, and reactivity of complex tricyclic oxonium ions, proposed intermediates in natural product biosynthesis. J. Am. Chem. Soc. 141, 15951–15962 (2019).
Nesmeyanov, A. N., Makarova, L. G. & Tolstaya, T. P. Heterolytic decomposition of onium compounds (diphenyl halogenonium and triphenyloxonium salts). Tetrahedron 1, 145–157 (1957).
Nesmeyanov, A. N. & Tolstaya, T. P. Triphenyloxonium salts. Dokl. Akad. Nauk SSSR 117, 626–628 (1957).
Smith, O. et al. Harnessing triaryloxonium ions for aryne generation. Nat. Synth. 3, 58–66 (2023).
Chen, W. L. et al. Base-promoted sulfur arylation of sulfenamides to oxonium aryne precursors: chemoselective synthesis of sulfilimines and o-sulfanylanilines. J. Org. Chem. 90, 448–457 (2025).
Hellwinkel, D. & Seifert, H. Zur Frage des pentakoordinierten Stickstoffs: Reaktionen von (spiro)cyclischen Tetraarylammonium-Salzen mit Nucleophilen. Liebigs Ann. 762, 29–54 (1972).
Tolstaya, T. P., Tsariev, D. A. & Luzikov, Y. N. Reaction of triaryloxonium salts with bases via dehydroarenes. Tetrahedron Lett. 38, 4457–4458 (1997).
Smith, O. et al. Control of stereogenic oxygen in a helically chiral oxonium ion. Nature 615, 430–435 (2023).
Fleming, I. Molecular Orbitals and Organic Chemical Reactions (John Wiley, 2010).
Wu, C.-H. et al. Do π-conjugative effects facilitate SN2 reactions? J. Am. Chem. Soc. 136, 3118–3126 (2014).
Robiette, R., Trieu-Van, T., Aggarwal, V. K. & Harvey, J. N. Activation of the SN2 reaction by adjacent π systems: the critical role of electrostatic interactions and of dissociative character. J. Am. Chem. Soc. 138, 734–737 (2016).
Zhang, J., Liao, Z., Chen, L. & Zhu, S. Rapid access to oxa-bridged bicyclic skeletons through gold-catalyzed tandem rearrangement reaction. Chem. Eur. J. 25, 9405–9409 (2019).
Dean, A. C. et al. Alkene 1,3-difluorination via transient oxonium intermediates. Angew. Chem. Int. Ed. 63, e202404666 (2024).
Uemura, M., Watson, I. D. G., Katsukawa, M. & Toste, F. D. Gold(I)-catalyzed enantioselective synthesis of benzopyrans via rearrangement of allylic oxonium intermediates. J. Am. Chem. Soc. 131, 3464–3465 (2009).
Das, G. K., Mandal, S. C. & Mondal, N. Study on first reported (2,5) ene cyclisation: does it really follow a concerted pathway? J. Chem. Res. 2003, 588–590 (2003).
Liu, H. et al. An unexpected Lewis acid-catalyzed cascade during the synthesis of the DEF-benzoxocin ring system of nogalamycin and menogaril: mechanistic elucidation by intermediate trapping experiments and density functional theory studies. J. Org. Chem. 89, 5634–5649 (2024).
Okamoto, K., Kinoshita, T. & Shingu, H. The retentive solvolysis. VII. Structural effect of the leaving group on the steric course of the SN 1 phenolysis of 1-phenylethyl systems. Bull. Chem. Soc. Jpn 43, 1545–1553 (1970).
Słomkowski, S. & Penczek, S. The carbenium ⇄ oxonium ion equilibrium. Part I. Reversible reaction of non-paired triphenylmethyl cation with ethers and acetals. J. Chem. Soc. 2, 1718–1722 (1974).
Olah, G. A., Olah, J. A. & Ohyama, T. Friedel–Crafts alkylation of anisole and its comparison with toluene. Predominant ortho–para substitution under kinetic conditions and the effect of thermodynamic isomerizations. J. Am. Chem. Soc. 106, 5284–5290 (1984).
Chan, H. S. S., Thompson, A. L., Christensen, K. E. & Burton, J. W. Forwards and backwards—synthesis of Laurencia natural products using a biomimetic and retrobiomimetic strategy incorporating structural reassignment of laurefurenynes C–F. Chem. Sci. 11, 11592–11600 (2020).
Krossing, I. The facile preparation of weakly coordinating anions: structure and characterisation of silverpolyfluoroalkoxyaluminates AgAl(ORF)4, calculation of the alkoxide ion affinity. Chem. Eur. J. 7, 490–502 (2001).
Allinger, N. L. & Coke, J. L. The relative stabilities of cis and trans isomers. VII. The hydrindanes. J. Am. Chem. Soc. 82, 2553–2556 (1960).
Farrant, R. D. et al. NMR quantification using an artificial signal. Magn. Reson. Chem. 48, 753–762 (2010).
Aue, D. H. in Dicoordinated Carbocations (eds Rappoport, Z. & Stang, P. J.) 126–129 (Wiley, 1997).
Aue, D. H. & Bowers, M. T. in Gas Phase Ion Chemistry (ed. Bowers, M. T.) 31–59 (Academic Press, 1979).
Anslyn, E. V. & Dougherty, D. A. Modern Physical Organic Chemistry (University Science Books, 2006).
Hansch, C., Leo, A. & Taft, R. W. A survey of Hammett substituent constants and resonance and field parameters. Chem. Rev. 91, 165–195 (1991).
Kimura, T., Takahashi, T., Nishiura, M. & Yamamura, K. Novel Metal-free hydrogenation of the carbon−carbon double bond in azulenoid enones by use of cycloheptatriene and protic acid. Org. Lett. 8, 3137–3139 (2006).
Eliel, E. L. & Wilen, S. H. Stereochemistry of Organic Compounds (John Wiley, 1994).
Brutiu, B. R., Iannelli, G., Riomet, M., Kaiser, D. & Maulide, N. Stereodivergent 1,3-difunctionalization of alkenes by charge relocation. Nature 626, 92–97 (2024).
G.-Simonian, N., Brutiu, B. R., Kaiser, D. & Maulide, N. Cationic, iodine(III)-mediated and directed diastereoselective oxidation of inert C–H bonds in cyclic hydrocarbons. Angew. Chem. Int. Ed. https://doi.org/10.1002/anie.202421872 (2025).
Bégué, J. P. & Bonnet-Delpon, D. Cyclic oxonium salts: 13C magnetic resonance spectroscopy. The correlation of chemical shifts with the calculated charge density. Org. Magn. Res. 14, 349–355 (1980).
Lubinskaya, O. V., Shashkov, A. S., Chertkov, V. A. & Smit, W. A. Facile synthesis of cyclic carboxonium salts by acylation of alkenes1. Synthesis 1976, 742–745 (1976).
Wang, M. Enantioselective intramolecular 1,5-hydride transfer/cyclization through the direct functionalization of C(sp3)–H bonds adjacent to a heteroatom: from nitrogen to oxygen. ChemCatChem 5, 1291–1293 (2013).
Wang, L. & Xiao, J. Advancement in cascade [1,n]-hydrogen transfer/cyclization: a method for direct functionalization of inactive C(sp3)–H bonds. Adv. Synth. Catal. 356, 1137–1171 (2014).
Haibach, M. C. & Seidel, D. C-H bond functionalization through intramolecular hydride transfer. Angew. Chem. Int. Ed. 53, 5010–5036 (2014).
Peng, B. & Maulide, N. The redox-neutral approach to C–H functionalization. Chem. Eur. J. 19, 13274–13287 (2013).
Pan, S. C. Organocatalytic C–H activation reactions. Beilstein J. Org. Chem. 8, 1374–1384 (2012).
Wang, B., Gandamana, D. A., Gagosz, F. & Chiba, S. Diastereoselective intramolecular hydride transfer under Bronsted acid catalysis. Org. Lett. 21, 2298–2301 (2019).
Simmons, E. M. & Hartwig, J. F. On the interpretation of deuterium kinetic isotope effects in C–H bond functionalizations by transition-metal complexes. Angew. Chem. Int. Ed. 51, 3066–3072 (2012).
Overman, L. E. & Velthuisen, E. J. Scope and facial selectivity of the Prins–pinacol synthesis of attached rings. J. Org. Chem. 71, 1581–1587 (2006).
Overman, L. E. & Pennington, L. D. Strategic use of pinacol-terminated Prins cyclizations in target-oriented total synthesis. J. Org. Chem. 68, 7143–7157 (2003).
McCormack, A. C., More O’Ferrall, R. A., O’Donoghu, A. C. & Rao, S. N. Protonated benzofuran, anthracene, naphthalene, benzene, ethene, and ethyne: measurements and estimates of pKa and pKR. J. Am. Chem. Soc. 124, 8575–8583 (2002).
Acknowledgements
J.L.S. is grateful to the Royal Commission for the Exhibition of 1851 for an Industrial Fellowship and the generous support provided by that fellowship, and to the EPSRC Centre for Doctoral Training in Synthesis for Biology and Medicine (EP/L015838/1) for a studentship, generously supported by AstraZeneca, Diamond Light Source, Defence Science and Technology Laboratory, Evotec, GlaxoSmithKline, Janssen, Novartis, Pfizer, Syngenta, Takeda, UCB and Vertex. D.S.B. is grateful to the Biotechnology and Biological Sciences Research Council and Syngenta for the award of an iCASE (BB/P504890/1). R.S.P. acknowledges support from the National Science Foundation (NSF CHE2400056) and the Alpine HPC resource, which is jointly funded by the University of Colorado Boulder, the University of Colorado Anschutz and Colorado State University, and the Advanced Cyberinfrastructure Coordination Ecosystem: Services & Support (ACCESS) through allocation TG-CHE180056. We thank M. Mascal and N. Hafezi for productive discussions regarding NMR assignment of oxonium ions.
Author information
Authors and Affiliations
Contributions
J.W.B. and H.S.S.C. designed the project. H.S.S.C., J.L.S. and D.S.B. conducted the synthetic experiments. Y.L. and R.S.P. designed and performed the computational study. H.S.S.C., J.W.B. and R.S.P. wrote the paper with contributions from all the authors. All authors interpreted the results in the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Synthesis thanks the anonymous reviewers for their contribution to the peer review of this work. Primary Handling Editor: Thomas West, in collaboration with the Nature Synthesis team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Formation of Friedel–Crafts products 54 and 55 from oxonium ions 24m·SbF6 and 24l·SbF6.
Reagents and conditions: For 24m·SbF6: 1) CD2Cl2, r.t., 1.5 hrs. 2) 2.0 eq. Et3N, CD2Cl2, r.t., 61% over 2 steps. For 24l·SbF6: 1) CD2Cl2, −10 °C to r.t., 2 hrs. 2) 2.0 eq. Et3N, CD2Cl2, r.t. 68% over 2 steps.
Extended Data Fig. 2 Formation of 1,4-aryl transfer product 60 from oxonium ion 24l·Al(pftb)4.
Reagents and conditions: 1) CD2Cl2, 0 °C to r.t., 30 minutes. 2) Filter, then 5.0 eq. Tetrabutylammonium bromide, CD2Cl2, r.t. 60: 21% over 2 steps, 61: 42% over 2 steps (d.r. ~ 4:1).
Supplementary information
Supplementary Information (download PDF )
Experimental details, Supplementary Figs. 1–28, computational methods and Cartesian coordinates.
Supplementary Data 1 (download PDF )
Supplementary NMR spectra.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chan, H.S.S., Li, Y., Sutro, J.L. et al. Synthesis and properties of allylic, benzylic, propargylic and allenylic oxonium ions. Nat. Synth (2026). https://doi.org/10.1038/s44160-025-00964-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44160-025-00964-8