Abstract
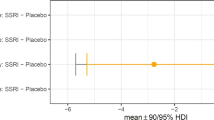
Serotonin fosters cognitive flexibility, but how, exactly, remains unclear. We developed a computational theory that proposes that serotonin reduces belief stickiness: the tendency to get ‘stuck’ in a belief about the state of the world despite incoming contradicting evidence. We tested this theory in a randomized, double-blind, placebo-controlled study using a single dose of the selective serotonin reuptake inhibitor escitalopram. In the escitalopram group, higher escitalopram plasma levels reduced belief stickiness more, resulting in better inference about the state of the world. Moreover, participants with sufficiently high escitalopram plasma levels had less belief stickiness, and therefore better state inference, than participants on placebo. We also propose that obsessions may result from excessive belief stickiness. Indeed, participants with more obsessions had greater belief stickiness, and therefore worse state inference. The opposite relations of escitalopram and obsessions with belief stickiness may explain the therapeutic effect of selective serotonin reuptake inhibitors in obsessive–compulsive disorder.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$79.00 per year
only $6.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
All data analyzed in this article are available via Zenodo at https://zenodo.org/records/10066829 (ref. 138).
Code availability
The code implementing all computational models and analyses in this article is available via GitHub at https://github.com/vaconceicao/serotonin-reduces-belief-stickiness.
References
Clarke, H. F., Dalley, J. W., Crofts, H. S., Robbins, T. W. & Roberts, A. C. Cognitive inflexibility after prefrontal serotonin depletion. Science 304, 878–880 (2004).
Clarke, H. F., Walker, S., Dalley, J., Robbins, T. & Roberts, A. Cognitive inflexibility after prefrontal serotonin depletion is behaviorally and neurochemically specific. Cereb. Cortex 17, 18–27 (2007).
Nilsson, S. R. O., Phillips, B. U., Axelsson, S. F. A. & Alsiö, J. in The Serotonin System (eds Tricklebank, M. D. & Daly, E.) 133–154 (Elsevier, 2019).
Maia, T. V. Reinforcement learning, conditioning, and the brain: successes and challenges. Cogn. Affect. Behav. Neurosci. 9, 343–364 (2009).
Yin, H. H. & Knowlton, B. J. The role of the basal ganglia in habit formation. Nat. Rev. Neurosci. 7, 464–476 (2006).
Collins, A. G. E. & Frank, M. J. Opponent actor learning (OpAL): modeling interactive effects of striatal dopamine on reinforcement learning and choice incentive. Psychol. Rev. 121, 337–366 (2014).
Sutton, R. S. & Barto, A. G. Reinforcement Learning: An Introduction (MIT Press, 1998).
Behrens, T. E. J., Woolrich, M. W., Walton, M. E. & Rushworth, M. F. S. Learning the value of information in an uncertain world. Nat. Neurosci. 10, 1214–1221 (2007).
Grossman, C. D., Bari, B. A. & Cohen, J. Y. Serotonin neurons modulate learning rate through uncertainty. Curr. Biol. 32, 586–599.e7 (2022).
Gershman, S. J., Blei, D. M. & Niv, Y. Context, learning, and extinction. Psychol. Rev. 117, 197–209 (2010).
Niv, Y. Learning task-state representations. Nat. Neurosci. 22, 1544–1553 (2019).
Redish, A. D., Jensen, S., Johnson, A. & Kurth-Nelson, Z. Reconciling reinforcement learning models with behavioral extinction and renewal: implications for addiction, relapse, and problem gambling. Psychol. Rev. 114, 784–805 (2007).
Wilson, R. C., Takahashi, Y. K., Schoenbaum, G. & Niv, Y. Orbitofrontal cortex as a cognitive map of task space. Neuron 81, 267–279 (2014).
Hornak, J. et al. Reward-related reversal learning after surgical excisions in orbito-frontal or dorsolateral prefrontal cortex in humans. J. Cogn. Neurosci. 16, 463–478 (2004).
Izquierdo, A., Brigman, J. L., Radke, A. K., Rudebeck, P. H. & Holmes, A. The neural basis of reversal learning: an updated perspective. Neuroscience 345, 12–26 (2017).
Remijnse, P. L. et al. Reduced orbitofrontal-striatal activity on a reversal learning task in obsessive-compulsive disorder. Arch. Gen. Psychiatry 63, 1225–1236 (2006).
Tezcan, D., Tumkaya, S. & Bora, E. Reversal learning in patients with obsessive-compulsive disorder (OCD) and their unaffected relatives: is orbitofrontal dysfunction an endophenotype of OCD? Psychiatry Res. 252, 231–233 (2017).
Fellows, L. K. The role of orbitofrontal cortex in decision making. Ann. N. Y. Acad. Sci. 1121, 421–430 (2007).
Maia, T. V. & McClelland, J. L. The somatic marker hypothesis: still many questions but no answers. Trends Cogn. Sci. 9, 162–164 (2005).
Rudebeck, P. H., Saunders, R. C., Prescott, A. T., Chau, L. S. & Murray, E. A. Prefrontal mechanisms of behavioral flexibility, emotion regulation and value updating. Nat. Neurosci. 16, 1140–1145 (2013).
Bradfield, L. A., Dezfouli, A., van Holstein, M., Chieng, B. & Balleine, B. W. Medial orbitofrontal cortex mediates outcome retrieval in partially observable task situations. Neuron 88, 1268–1280 (2015).
Schuck, N. W., Cai, M. B., Wilson, R. C. & Niv, Y. Human orbitofrontal cortex represents a cognitive map of state space. Neuron 91, 1402–1412 (2016).
Schuck, N. W., Wilson, R. & Niv, Y. in Goal-Directed Decision Making: Computations and Neural Circuits (eds Morris, R. et al.) 259–278 (Academic Press, 2018).
Stalnaker, T. A., Cooch, N. K. & Schoenbaum, G. What the orbitofrontal cortex does not do. Nat. Neurosci. 18, 620–627 (2015).
Wikenheiser, A. M. & Schoenbaum, G. Over the river, through the woods: cognitive maps in the hippocampus and orbitofrontal cortex. Nat. Rev. Neurosci. 17, 513–523 (2016).
Clarke, H. F. et al. Prefrontal serotonin depletion affects reversal learning but not attentional set shifting. J. Neurosci. 25, 532–538 (2005).
Kanen, J. W. et al. Serotonin depletion impairs both Pavlovian and instrumental reversal learning in healthy humans. Mol. Psychiatry 26, 7200–7210 (2021).
Matias, S., Lottem, E., Dugué, G. P. & Mainen, Z. F. Activity patterns of serotonin neurons underlying cognitive flexibility. eLife 6, e20552 (2017).
Roberts, C., Sahakian, B. J. & Robbins, T. W. Psychological mechanisms and functions of 5-HT and SSRIs in potential therapeutic change: lessons from the serotonergic modulation of action selection, learning, affect, and social cognition. Neurosci. Biobehav. Rev. 119, 138–167 (2020).
Zhukovsky, P. et al. Perseveration in a spatial-discrimination serial reversal learning task is differentially affected by MAO-A and MAO-B inhibition and associated with reduced anxiety and peripheral serotonin levels. Psychopharmacology 234, 1557–1571 (2017).
Barlow, R. L. et al. Markers of serotonergic function in the orbitofrontal cortex and dorsal raphé nucleus predict individual variation in spatial-discrimination serial reversal learning. Neuropsychopharmacology 40, 1619–1630 (2015).
Groman, S. M. et al. Monoamine levels within the orbitofrontal cortex and putamen interact to predict reversal learning performance. Biol. Psychiatry 73, 756–762 (2013).
Maia, T. V. & Cano-Colino, M. The role of serotonin in orbitofrontal function and obsessive-compulsive disorder. Clin. Psychol. Sci. 3, 460–482 (2015).
Roberts, A. C. The importance of serotonin for orbitofrontal function. Biol. Psychiatry 69, 1185–1191 (2011).
Alsiö, J. et al. Serotonergic innervations of the orbitofrontal and medial-prefrontal cortices are differentially involved in visual discrimination and reversal learning in rats. Cereb. Cortex 31, 1090–1105 (2021).
Masaki, D. et al. Relationship between limbic and cortical 5-HT neurotransmission and acquisition and reversal learning in a go/no-go task in rats. Psychopharmacology 189, 249–258 (2006).
Chan, S. C. Y., Niv, Y. & Norman, K. A. A probability distribution over latent causes, in the orbitofrontal cortex. J. Neurosci. 36, 7817–7828 (2016).
Huys, Q. J. M. & Petzschner, F. H. Failure modes of the will: from goals to habits to compulsions? Am. J. Psychiatry 172, 216–218 (2015).
Rigoux, L., Stephan, K. E. & Petzschner, F. H. Beliefs, compulsive behavior and reduced confidence in control. PLoS Comput. Biol. 20, e1012207 (2024).
Maia, T. V., Cooney, R. E. & Peterson, B. S. The neural bases of obsessive–compulsive disorder in children and adults. Dev. Psychopathol. 20, 1251 (2008).
Menzies, L. et al. Integrating evidence from neuroimaging and neuropsychological studies of obsessive-compulsive disorder: the orbitofronto-striatal model revisited. Neurosci. Biobehav. Rev. 32, 525–549 (2008).
Piras, F. et al. Widespread structural brain changes in OCD: a systematic review of voxel-based morphometry studies. Cortex 62, 89–108 (2015).
Rotge, J.-Y. et al. Provocation of obsessive-compulsive symptoms: a quantitative voxel-based meta-analysis of functional neuroimaging studies. J. Psychiatry Neurosci. 33, 405–412 (2008).
Rotge, J.-Y. et al. Meta-analysis of brain volume changes in obsessive-compulsive disorder. Biol. Psychiatry 65, 75–83 (2009).
Thorsen, A. L. et al. Emotional processing in obsessive-compulsive disorder: a systematic review and meta-analysis of 25 functional neuroimaging studies. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 3, 563–571 (2018).
Whiteside, S. P., Port, J. D. & Abramowitz, J. S. A meta-analysis of functional neuroimaging in obsessive-compulsive disorder. Psychiatry Res. 132, 69–79 (2004).
Maia, T. V. & McClelland, J. L. A neurocomputational approach to obsessive-compulsive disorder. Trends Cogn. Sci. 16, 14–15 (2012).
Rolls, E. T., Loh, M. & Deco, G. An attractor hypothesis of obsessive–compulsive disorder. Eur. J. Neurosci. 28, 782–793 (2008).
Chamberlain, S. R. et al. Orbitofrontal dysfunction in patients with obsessive-compulsive disorder and their unaffected relatives. Science 321, 421–422 (2008).
Freyer, T. et al. Frontostriatal activation in patients with obsessive-compulsive disorder before and after cognitive behavioral therapy. Psychol. Med. 41, 207–216 (2011).
Remijnse, P. L. et al. Differential frontal-striatal and paralimbic activity during reversal learning in major depressive disorder and obsessive-compulsive disorder. Psychol. Med. 39, 1503–1518 (2009).
Apergis-Schoute, A. M. et al. Perseveration and shifting in obsessive-compulsive disorder as a function of uncertainty, punishment, and serotonergic medication. Biol. Psychiatry Glob. Open Sci. 4, 326–335 (2024).
Hirschtritt, M. E., Bloch, M. H. & Mathews, C. A. Obsessive-compulsive disorder: advances in diagnosis and treatment. JAMA 317, 1358–1367 (2017).
Sanchez, C., Reines, E. H. & Montgomery, S. A. A comparative review of escitalopram, paroxetine, and sertraline: are they all alike? Int. Clin. Psychopharmacol. 29, 185–196 (2014).
Saxena, S. & Rauch, S. L. Functional neuroimaging and the neuroanatomy of obsessive-compulsive disorder. Psychiatr. Clin. North Am. 23, 563–586 (2000).
Foa, E. B. et al. The Obsessive-Compulsive Inventory: development and validation of a short version. Psychol. Assess. 14, 485–496 (2002).
Clark, D. A. et al. A question of perspective: the association between intrusive thoughts and obsessionality in 11 countries. J. Obsessive Compuls. Relat. Disord. 3, 292–299 (2014).
Fullana, M. A. et al. Obsessions and compulsions in the community: prevalence, interference, help-seeking, developmental stability, and co-occurring psychiatric conditions. Am. J. Psychiatry 166, 329–336 (2009).
Abramowitz, J. S. et al. The relevance of analogue studies for understanding obsessions and compulsions. Clin. Psychol. Rev. 34, 206–217 (2014).
Draper, C. F. et al. Menstrual cycle rhythmicity: metabolic patterns in healthy women. Sci. Rep. 8, 1–15 (2018).
Jovanovic, H. et al. A PET study of 5-HT1A receptors at different phases of the menstrual cycle in women with premenstrual dysphoria. Psychiatry Res. 148, 185–193 (2006).
Frank, M. J., Moustafa, A. A., Haughey, H. M., Curran, T. & Hutchison, K. E. Genetic triple dissociation reveals multiple roles for dopamine in reinforcement learning. Proc. Natl Acad. Sci. USA 104, 16311–16316 (2007).
Guitart-Masip, M. et al. Go and no-go learning in reward and punishment: interactions between affect and effect. NeuroImage 62, 154–166 (2012).
Conceição, V. A. & Maia, T. V. in Computational Psychiatry: A Primer (ed. Seriès, P.) 205–246 (MIT Press, 2020).
Collins, A. G. E. & Frank, M. J. Cognitive control over learning: creating, clustering, and generalizing task-set structure. Psychol. Rev. 120, 190–229 (2013).
Collins, A. G. E. & Frank, M. J. Neural signature of hierarchically structured expectations predicts clustering and transfer of rule sets in reinforcement learning. Cognition 152, 160–169 (2016).
Babayan, B. M., Uchida, N. & Gershman, S. J. Belief state representation in the dopamine system. Nat. Commun. 9, 1891 (2018).
Gershman, S. J. & Uchida, N. Believing in dopamine. Nat. Rev. Neurosci. 20, 703–714 (2019).
Starkweather, C. K., Babayan, B. M., Uchida, N. & Gershman, S. J. Dopamine reward prediction errors reflect hidden-state inference across time. Nat. Neurosci. 20, 581–589 (2017).
Lartillot, N. & Philippe, H. Computing Bayes factors using thermodynamic integration. Syst. Biol. 55, 195–207 (2006).
Sengupta, B., Friston, K. J. & Penny, W. D. Gradient-free MCMC methods for dynamic causal modelling. NeuroImage 112, 375–381 (2015).
Daunizeau, J., Adam, V. & Rigoux, L. VBA: a probabilistic treatment of nonlinear models for neurobiological and behavioural data. PLoS Comput. Biol. 10, e1003441 (2014).
Hampton, A. N., Bossaerts, P. & O’Doherty, J. P. The role of the ventromedial prefrontal cortex in abstract state-based inference during decision making in humans. J. Neurosci. 26, 8360–8367 (2006).
Schlagenhauf, F. et al. Striatal dysfunction during reversal learning in unmedicated schizophrenia patients. NeuroImage 89, 171–180 (2014).
Kanen, J. W., Ersche, K. D., Fineberg, N. A., Robbins, T. W. & Cardinal, R. N. Computational modelling reveals contrasting effects on reinforcement learning and cognitive flexibility in stimulant use disorder and obsessive-compulsive disorder: remediating effects of dopaminergic D2/3 receptor agents. Psychopharmacology 236, 2337–2358 (2019).
Marzuki, A. A. et al. Association of environmental uncertainty with altered decision-making and learning mechanisms in youths with obsessive-compulsive disorder. JAMA Netw. Open 4, e2136195 (2021).
Rygula, R. et al. Role of central serotonin in anticipation of rewarding and punishing outcomes: effects of selective amygdala or orbitofrontal 5-HT depletion. Cereb. Cortex 25, 3064–3076 (2015).
Luo, Q. et al. Comparable roles for serotonin in rats and humans for computations underlying flexible decision-making. Neuropsychopharmacology 49, 600–608 (2024).
Robbins, T. W. & Cardinal, R. N. Computational psychopharmacology: a translational and pragmatic approach. Psychopharmacology 236, 2295–2305 (2019).
Langley, C. et al. Chronic escitalopram in healthy volunteers has specific effects on reinforcement sensitivity: a double-blind, placebo-controlled semi-randomised study. Neuropsychopharmacology 48, 664–670 (2023).
Gardier, A., Malagié, I., Trillat, A., Jacquot, C. & Artigas, F. Role of 5-HT1A autoreceptors in the mechanism of action of serotoninergic antidepressant drugs: recent findings from in vivo microdialysis studies. Fundam. Clin. Pharmacol. 10, 16–27 (1996).
Altman, D. G. & Bland, J. M. Statistics notes: absence of evidence is not evidence of absence. BMJ 311, 485 (1995).
Cohen, J. & Nee, J. C. M. Estimators for two measures of association for set correlation. Educ. Psychol. Meas. 44, 907–917 (1984).
Guitart-Masip, M. et al. Differential, but not opponent, effects of l-DOPA and citalopram on action learning with reward and punishment. Psychopharmacology 231, 955–966 (2014).
Cools, R., Nakamura, K. & Daw, N. D. Serotonin and dopamine: unifying affective, activational, and decision functions. Neuropsychopharmacology 36, 98–113 (2011).
Daw, N. D., Kakade, S. & Dayan, P. Opponent interactions between serotonin and dopamine. Neural Netw. 15, 603–616 (2002).
Dayan, P. & Huys, Q. J. M. Serotonin in affective control. Annu. Rev. Neurosci. 32, 95–126 (2009).
Bloch, M. H., McGuire, J., Landeros-Weisenberger, A., Leckman, J. F. & Pittenger, C. Meta-analysis of the dose-response relationship of SSRI in obsessive-compulsive disorder. Mol. Psychiatry 15, 850–855 (2010).
Pittenger, C. & Bloch, M. H. Pharmacological treatment of obsessive-compulsive disorder. Psychiatr. Clin. North Am. 37, 375–391 (2014).
Furr, A., Lapiz-Bluhm, M. D. & Morilak, D. A. 5-HT2A receptors in the orbitofrontal cortex facilitate reversal learning and contribute to the beneficial cognitive effects of chronic citalopram treatment in rats. Int. J. Neuropsychopharmacol. 15, 1295–1305 (2012).
Walker, S. C., Robbins, T. W. & Roberts, A. C. Differential contributions of dopamine and serotonin to orbitofrontal cortex function in the marmoset. Cereb. Cortex 19, 889–898 (2009).
Bari, A. et al. Serotonin modulates sensitivity to reward and negative feedback in a probabilistic reversal learning task in rats. Neuropsychopharmacology 35, 1290–1301 (2010).
Blier, P., Pineyro, G., El Mansari, M., Bergeron, R. & Montigny, C. Role of somatodendritic 5-HT autoreceptors in modulating 5-HT neurotransmission. Ann. N. Y. Acad. Sci. 861, 204–216 (1998).
Hjorth, S. Serotonin 5-HT1A autoreceptor blockade potentiates the ability of the 5-HT reuptake inhibitor citalopram to increase nerve terminal output of 5-HT in vivo: a microdialysis study. J. Neurochem. 60, 776–779 (1993).
Beyer, C. E. & Cremers, T. I. F. H. Do selective serotonin reuptake inhibitors acutely increase frontal cortex levels of serotonin? Eur. J. Pharmacol. 580, 350–354 (2008).
Nord, M., Finnema, S. J., Halldin, C. & Farde, L. Effect of a single dose of escitalopram on serotonin concentration in the non-human and human primate brain. Int. J. Neuropsychopharmacol. 16, 1577–1586 (2013).
Invernizzi, R., Belli, S. & Samanin, R. Citalopram’s ability to increase the extracellular concentrations of serotonin in the dorsal raphe prevents the drug’s effect in the frontal cortex. Brain Res. 584, 322–324 (1992).
Hirano, K. et al. Relationship between brain serotonin transporter binding, plasma concentration and behavioural effect of selective serotonin reuptake inhibitors. Br. J. Pharmacol. 144, 695–702 (2005).
Meyer, J. H. et al. Serotonin transporter occupancy of five selective serotonin reuptake inhibitors at different doses: an [11C]DASB positron emission tomography study. Am. J. Psychiatry 161, 826–835 (2004).
Chamberlain, S. R. et al. Neurochemical modulation of response inhibition and probabilistic learning in humans. Science 311, 861–863 (2006).
Skandali, N. et al. Dissociable effects of acute SSRI (escitalopram) on executive, learning and emotional functions in healthy humans. Neuropsychopharmacology 43, 2645 (2018).
Fritze, S., Spanagel, R. & Noori, H. R. Adaptive dynamics of the 5-HT systems following chronic administration of selective serotonin reuptake inhibitors: a meta-analysis. J. Neurochem. 142, 747–755 (2017).
Cools, R., Roberts, A. C. & Robbins, T. W. Serotoninergic regulation of emotional and behavioural control processes. Trends Cogn. Sci. 12, 31–40 (2008).
Boureau, Y.-L. & Dayan, P. Opponency revisited: competition and cooperation between dopamine and serotonin. Neuropsychopharmacology 36, 74–97 (2011).
Michely, J., Eldar, E., Erdman, A., Martin, I. M. & Dolan, R. J. Serotonin modulates asymmetric learning from reward and punishment in healthy human volunteers. Commun. Biol. 5, 812 (2022).
Loosen, A. M. & Hauser, T. U. Towards a computational psychiatry of juvenile obsessive-compulsive disorder. Neurosci. Biobehav. Rev. 118, 631–642 (2020).
Fradkin, I., Adams, R. A., Parr, T., Roiser, J. P. & Huppert, J. D. Searching for an anchor in an unpredictable world: a computational model of obsessive compulsive disorder. Psychol. Rev. 127, 672–699 (2020).
Fradkin, I., Ludwig, C., Eldar, E. & Huppert, J. D. Doubting what you already know: uncertainty regarding state transitions is associated with obsessive compulsive symptoms. PLoS Comput. Biol. 16, e1007634 (2020).
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders: DSM-5 (American Psychiatric Association, 2013).
Foa, E. B. et al. DSM-IV field trial: obsessive-compulsive disorder. Am. J. Psychiatry 152, 90–96 (1995).
Jakubovski, E. et al. Dimensional correlates of poor insight in obsessive-compulsive disorder. Prog. Neuropsychopharmacol. Biol. Psychiatry 35, 1677–1681 (2011).
Vaghi, M. M. et al. Compulsivity reveals a novel dissociation between action and confidence. Neuron 96, 348–354.e4 (2017).
White, J., Tannenbaum, C., Klinge, I., Schiebinger, L. & Clayton, J. The integration of sex and gender considerations into biomedical research: lessons from international funding agencies. J. Clin. Endocrinol. Metab. 106, 3034–3048 (2021).
Hall, E. & Steiner, M. Serotonin and female psychopathology. Women's Health 9, 85–97 (2013).
Thorne, B. N., Ellenbroek, B. A. & Day, D. J. Sex bias in the serotonin transporter knockout model: implications for neuropsychiatric disorder research. Neurosci. Biobehav. Rev. 134, 104547 (2022).
Recommendations for the Conduct, Reporting, Editing, and Publication of Scholarly Work in Medical Journals (International Committee of Medical Journal Editors (ICMJE), 2025); https://www.icmje.org/icmje-recommendations.pdf
Gönner, S., Leonhart, R. & Ecker, W. The Obsessive–Compulsive Inventory-Revised (OCI-R): validation of the German version in a sample of patients with OCD, anxiety disorders, and depressive disorders. J. Anxiety Disord. 22, 734–749 (2008).
Watkins, C. J. C. H. Learning from Delayed Rewards. PhD thesis, Univ. Cambridge (1989).
Maia, T. V. & Conceição, V. A. The roles of phasic and tonic dopamine in tic learning and expression. Biol. Psychiatry 82, 401–412 (2017).
Clarke, H. F. et al. Orbitofrontal dopamine depletion upregulates caudate dopamine and alters behavior via changes in reinforcement sensitivity. J. Neurosci. 34, 7663–7676 (2014).
Daw, N. D. in Decision Making, Affect, and Learning: Attention and Performance XXIII (eds Delgado, M. R., Phelps, E. A. & Robbins, T. W.) 3–38 (Oxford Univesity Press, 2011).
Robert, C. & Casella, G. Monte Carlo Statistical Methods (Springer, 2013).
Penny, W. D. Comparing dynamic causal models using AIC, BIC and free energy. NeuroImage 59, 319–330 (2012).
Frässle, S. et al. TAPAS: an open-source software package for translational neuromodeling and computational psychiatry. Front. Psychiatry 12, 680811 (2021).
Aponte, E. A. et al. mpdcm: a toolbox for massively parallel dynamic causal modeling. J. Neurosci. Methods 257, 7–16 (2016).
Aponte, E. A., Schöbi, D., Stephan, K. E. & Heinzle, J. The Stochastic Early Reaction, Inhibition, and late Action (SERIA) model for antisaccades. PLoS Comput. Biol. 13, e1005692 (2017).
Calderhead, B. & Girolami, M. Estimating Bayes factors via thermodynamic integration and population MCMC. Comput. Stat. Data Anal. 53, 4028–4045 (2009).
Gelman, A., Carlin, J. B., Stern, H. S. & Rubin, D. B. Bayesian Data Analysis Vol. 2 (Chapman & Hall/CRC, 2014).
Rigoux, L., Stephan, K. E., Friston, K. J. & Daunizeau, J. Bayesian model selection for group studies—revisited. NeuroImage 84, 971–985 (2014).
Stephan, K. E., Penny, W. D., Daunizeau, J., Moran, R. J. & Friston, K. J. Bayesian model selection for group studies. NeuroImage 46, 1004–1017 (2009).
Winkler, A. M., Renaud, O., Smith, S. M. & Nichols, T. E. Permutation inference for canonical correlation analysis. NeuroImage 220, 117065 (2020).
McGarigal, K., Stafford, S. & Cushman, S. Multivariate Statistics for Wildlife and Ecology Research (Springer, 2000).
Sherry, A. & Henson, R. K. Conducting and interpreting canonical correlation analysis in personality research: a user-friendly primer. J. Pers. Assess. 84, 37–48 (2005).
RStudio Team. RStudio: integrated development environment for R. http://www.rstudio.com/ (2020).
R Core Team R: A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, 2021).
The jamovi project. jamovi (version 1.6.23) [Computer Software]. https://www.jamovi.org (2020).
Hohl, K. & Dolcos, S. Measuring cognitive flexibility: a brief review of neuropsychological, self-report, and neuroscientific approaches. Front. Hum. Neurosci. 18, 1331960 (2024).
Translational Neuromodeling Unit (TNU), Institute for Biomedical Engineering, University of Zurich and ETH Zurich. Dataset from a probabilistic Go/NoGo reversal-learning task. Zenodo https://doi.org/10.5281/zenodo.10066829 (2024).
Acknowledgements
We are very grateful to K. Stephan for his generous guidance and support of this project. We gratefully acknowledge support by the Clinical Research Priority Program ‘Molecular Imaging’ at UZH, Fundação para a Ciência e a Tecnologia, Portugal (PhD fellowship PD/BD/105852/2014 to V.A.C.), the Tourette Association of America (T.V.M.) and the Brainstorm Program at the Robert J. & Nancy D. Carney Institute for Brain Science (F.H.P.). V.A.C. and T.V.M. also acknowledge their participation in a twinning project (SynaNet) from the European Union Horizon 2020 Programme (project number 692340). We also acknowledge the valued study support of our colleagues J. Heinzle, T. Baumgartner, K. Treiber, C. Schnyder, A. Diaconescu, S. Iglesias, H. Haker and Q. Huys. Finally, we thank A. Winkler (University of Texas Rio Grande Valley) for guidance concerning the permutation tests used to obtain P values for the cross-loadings in the canonical correlation analysis.
Author information
Authors and Affiliations
Contributions
The study was conceptualized by T.V.M., F.H.P., V.A.C. and D.M.C. The behavioral task was designed by T.V.M., V.A.C. and F.H.P., and implemented and piloted by F.H.P. The data were collected by D.M.C., K.V.W. and F.H.P. The computational models were designed by V.A.C., F.H.P., S.R. and T.V.M., and implemented by V.A.C. and S.R. The statistical analyses were designed by V.A.C., F.H.P. and T.V.M., and implemented by V.A.C. and F.H.P. Escitalopram plasma levels were measured by D.M. The article was written by V.A.C., F.H.P. and T.V.M.
Corresponding authors
Ethics declarations
Competing interests
S.R. is currently at Novartis, but all his work on this article was performed while he was at the Translational Neuromodeling Unit. The other authors declare no competing interests.
Peer review
Peer review information
Nature Mental Health thanks Praveen Suthaharan, Hang Yang and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Behavior and model fit for an example participant.
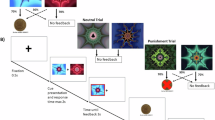
Each plot represents a shell. Points at the bottom and top of each plot indicate the participant’s trial-by-trial responses: black points at the bottom represent NoGo responses; colored points at the top represent Go responses, with the color representing the reinforcement received (blue, reward; yellow, neutral; red, punishment). a, Go probability, P(Go), predicted by the winning model (S–S–R–8; solid black line) and its corresponding S–R model (S–R–8; dashed gray line). Horizontal color-coded bars indicate the shell’s season (blue, rewarding; yellow, neutral; red, punishing). Model S–S–R–8 tracked the participant’s behavior better, especially when seasons recurred (last two seasons for each shell). b, Belief trajectory for model S–S–R–8. Horizontal color-coded bars on top indicate the season inferred by the model; horizontal color-coded bars immediately below indicate the true season. The lines depict the posterior probability distribution of the beliefs about the shell season (state), P(State), on each trial: the beliefs that the shell is in rewarding, neutral and punishing states are represented in blue, yellow and red, respectively. In the model, each state corresponds to an outcome distribution; states are not labeled a priori as rewarding, neutral or punishing. To represent the inferred seasons in the top horizontal bars and the beliefs in the plotted lines, we assigned rewarding, neutral and punishing labels to states whose final distribution consisted mostly of reward, neutral or punishment outcomes, respectively. Given that the task included three season types, the model assumed that there could be up to three hidden states (see ‘S–S–R models’ in ‘Computational models’ in the Supplementary Methods). In all plots except the top-left plot, the model only inferred up to two states; a state that was never inferred does not have outcomes associated, so we represent it in black. Some lines overlap and therefore are not visible. The line markers indicate the state that the model believes applies after each trial; usually, this corresponds to the state with the largest posterior probability, but sometimes it does not because the model has a (parameter-dependent) tendency to stick with the prior belief (see ‘S–S–R models’ in ‘Computational models’ in the Supplementary Methods).
Extended Data Fig. 2 Model S–R–8, the S–R equivalent of winning model S–S–R–8, replicates some of the results concerning participants’ (n = 44) group-level behavior (panels a and b) but not the result that requires state inference (panel c).
This figure is equivalent to Fig. 4 but for model S–R–8 rather than model S–S–R–8. a, Average (±s.e.m.) Go probability, P(Go), for each season type: rewarding (R), neutral (N) and punishing (P). We analyzed the model fits using the approach described in the caption of Fig. 4a. Like for participants (see caption of Fig. 4a), P(Go) for the fits of model S–R–8 was largest for rewarding, intermediate for neutral and smallest for punishing seasons (main effect of season type: \({\chi }_{2}^{2}=971.71\), P < 10−16; repeated contrasts: rewarding versus neutral, b = 0.68, z = 8.79, two-sided P < 10−16, 95% CI (0.53, 0.83), OR = 1.97; neutral versus punishing, b = 1.39, z = 23.41, two-sided P < 10−16, 95% CI (1.27, 1.50), OR = 4.00). b, Average (±s.e.m.) P(Go) as a function of the shell phase, for each shell. The fits from model S–R–8 tracked the changes in participants’ choices as the seasons changed. c, Average (±s.e.m.) probability of a correct response, P(Correct), in the phases following the first and second identical state transitions: phases 2 and 4, respectively. As in Fig. 4c, P(Correct) was calculated using only R and P seasons, as there was no correct response for N seasons. We analyzed P(Correct) using a rmANOVA with independent variables phase (2 versus 4) and data type (participants versus fits of the S–R–8 model). As expected, the interaction was significant (F1,43 = 11.19, P = 0.002, \({\eta }_{p}^{2}=0.21\)). Planned contrasts confirmed that P(Correct) was significantly greater in phase 4 than in phase 2 for participants (mean difference = 0.05, t43 = 2.61, two-sided P = 0.012, 95% CI (0.01, 0.10), Cohen’s d = 0.39), like in Fig. 4c, but not for fits of the S–R–8 model (mean difference = 0.00, t43 = 0.01, two-sided P = 0.994, 95% CI (–0.02, 0.02), Cohen’s d = 0.00). Thus, unlike participants and model S–S–R–8 (Fig. 4c), model S–R–8 did not show better performance in phase 4 than in phase 2. NS: not significant; *P < 0.05; ***P < 0.001.
Extended Data Fig. 3 Behavior was better explained by a model that extended S–R learning with state inference than by models that implemented alternative hypotheses.
The winning state-inference model (model S–S–R–8) explained behavior better than models that implemented learning rates that could vary across the task (S–Rα(T) models) or models that implemented stimulus stickiness (S–RStimStick models; see ‘Alternative model families’ in ‘Computational models’ in the Methods and in the Supplementary Methods). a, Model frequencies. b, PEPs. The horizontal red line indicates the threshold for confident selection of a model. State-inference model S–S–R–8 was confidently selected as the best model.
Supplementary information
Supplementary Information (download PDF )
Supplementary Methods, Supplementary Results, Tables 1–4 and Figs. 1–8.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Conceição, V.A., Petzschner, F.H., Cole, D.M. et al. Serotonin reduces belief stickiness. Nat. Mental Health (2026). https://doi.org/10.1038/s44220-026-00621-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44220-026-00621-9