Abstract
Contralateral prophylactic mastectomy (CPM) reduces contralateral breast cancer risk but improves survival only among young BRCA1/2 carriers. With expanded germline testing (GT) and increased use of oncologist-led and telehealth genetic services post-COVID, concerns remain about patients’ understanding of results and the impact on surgical decisions. We conducted a retrospective cohort study of 1,054 women with unilateral breast cancer who underwent GT at Columbia University Irving Medical Center from 2013–2022. Mean age was 51.2; 40.5% were non-Hispanic White (NHW), 28.4% Hispanic, 12.1% Black, and 10.3% Asian. Overall, 28% underwent CPM. Testing shifted from 99.7% in-person to 73.9% oncologist-led and 20.1% telehealth post-COVID. In multivariable analysis, CPM was associated with younger age, advanced stage, and pathogenic/likely pathogenic variants, but not with service delivery or VUS results. A trend toward higher CPM rates was observed among Hispanic versus NHW patients (OR = 1.34, 95% CI = 0.99-1.81, p = 0.055). Despite care delivery shifts, CPM rates remained stable.
Similar content being viewed by others
Introduction
In recent decades, contralateral prophylactic mastectomy (CPM) has been increasingly utilized by women with unilateral breast cancer, increasing from 3.4% in 2016 to 6.8% in 20191,2,3. While CPM reduces the risk of a contralateral breast cancer, current evidence is inconsistent with regard to the survival benefit for patients at average-risk for breast cancer4,5. However, for women with germline pathogenic variants in hereditary breast and ovarian cancer genes such as BRCA1/2, more recent data suggest CPM confers a significant improvement in overall survival, specifically for women <40 years, supporting consideration of CPM among this select, high-risk patient population6,7,8,9. Despite these findings, the growth in CPM rates do not appear to solely reflect improved identification of patients at high-risk for contralateral breast cancer and instead may be partially attributed to patient concerns about breast cancer recurrence or new primaries, or desire for symmetry with breast reconstruction for unilateral mastectomy10,11.
As genetic test results increasingly inform decisions around CPM, shifts in how these services are delivered may influence patient understanding, risk perception, and ultimately surgical choices. Traditionally, genetic counseling and testing have been provided by genetic counselors during in-person counseling sessions. However, evolving clinical practices, including expanded testing criteria, increased accessibility of germline testing, institutional adoption of streamlined or “mainstream” testing pathways and the necessity for remote telehealth services during the COVID pandemic, have accelerated the adoption of “mainstream” models, in which medical and surgical oncologists provide pretest counseling to patients with cancer, directly order germline testing, and return results12. While genetic counselors are ideally suited to provide patients with education to make informed, personalized decisions regarding genetic testing, a shortage of genetic counselors worldwide makes the demand to see more patients due to the broader criteria and larger panel testing difficult13. Telehealth-based genetic counseling has expanded access but may impact comprehension and shared decision-making14,15.
The clinical implications of this shift, particularly on surgical decision-making such as CPM, remain unclear. Although the shifts toward oncologist-led genetic testing and telehealth-delivered genetic counseling increase access to testing, these shifts could also lead to increased CPM, resulting in unnecessary procedures and costs, as patients might not fully understand how their genetic test results impact their cancer risk and should influence their surgical decisions. In this study, we investigated how the transition to oncologist-led and telehealth-based genetic services affected CPM uptake among a racially and ethnically diverse cohort of women with operable breast cancer at a large academic medical center.
Results
Baseline characteristics
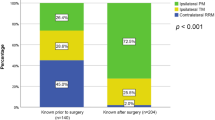
Among 1054 evaluable patients, the mean age at diagnosis was 51.3 years old (standard deviation [SD] = 12.4), with 40.5% non-Hispanic White, 28.4% Hispanic, 12.1% non-Hispanic Black, 10.3% Asian, and 8.8% other/unknown (Table 1). About two-thirds of the patients (68.4%) were diagnosed between 2013 and 2019, and 31.6% were diagnosed in 2020–2022. Over half (54.6%) of patients had a family history of breast cancer. Most patients (99.7%) diagnosed between 2013 and 2019 had in-person genetic counseling visits, compared to just 5.1% of patients diagnosed in 2020 or later (Supplementary Table 1). About 20% of patients diagnosed with breast cancer in 2020 or later had telehealth genetic counseling, and 74% had oncologist-led genetic counseling.
Within the study population, 13.0% of women had P/LP variants and 21.3% had variants of unknown significance (VUS) results. Of patients who had P/LP, 62.8% underwent CPM. In comparison, 23.7% of patients with VUS, and 23.1% of patients with B/LB underwent CPM. Only 9.5% of patients with P/LP underwent oncologist-led counseling, compared to 26.3% among those with VUS and 25.4% among those with B/LB (Supplementary Table 2). Non-Hispanic Whites and Blacks had the highest frequency of P/LP variants, whereas VUS results were more common among Hispanics and Asians (Table 2).
In total, 492 women (46.7%) underwent lumpectomy, 263 women (25.0%) underwent unilateral mastectomy, and 299 women (28.4%) women underwent CPM. Of those who were diagnosed pre-COVID, 30.9% underwent CPM, compared to 22.8% of those diagnosed post-COVID.
Factors associated with CPM
On univariable analysis, patients who underwent oncologist-led genetic counseling were less likely to undergo CPM (OR = 0.51, 95% CI = 0.36–0.73, p < 0.001) (Table 3). In multivariable analysis, younger age at diagnosis and P/LP results on germline genetic testing were significantly associated with increased CPM rates. There was no significant association between the type of genetic counseling received and rates of CPM. On multivariable analysis, there was no significant difference in rates of CPM among patients with VUS compared to patients with B/LB (OR = 0.90, 95% CI = 0.61–1.34, p = 0.599).
Given collinearity of type of genetic counseling received and year of diagnosis (pre- vs. post-COVID), multivariable analyses only included type of genetic counseling received. Similar results were seen on multivariable analysis when year of diagnosis was used in place of type of genetic counseling received (Supplementary Table 3). Rates of CPM decreased over the study period, which was largely driven by decreasing use among patients with B/LB results (35.1% of patients undergoing CPM from 2013–2019, compared to 18.4% from 2020 to 2022, p = 0.003) (Supplementary Table 3). There was no significant difference in CPM rates over time among patients with VUS or P/LP.
There was a trend toward higher CPM rates among Hispanic women, compared to non-Hispanic Whites (OR = 1.31, 95% CI = 0.91–1.90, p = 0.152). Although racial and/or ethnic minorities are more likely to have VUS, there were no racial or ethnic differences in rates of CPM among patients who had VUS results on germline testing (Supplementary Table 4).
Discussion
In this racially and ethnically diverse cohort of women with operable unilateral breast cancer, we found that about one in three patients underwent CPM. Consistent with prior research, CPM was more common among women with pathogenic or likely pathogenic (P/LP) variants in breast cancer predisposition genes and younger age at diagnosis. We found that patients with VUS underwent similar CPM rates as those with B/LB results. We observed a trend among Hispanic women to undergo more CPM than non-Hispanic White women. Importantly, despite a notable transition to telehealth and oncologist-led genetic testing during the COVID pandemic, although we saw lower CPM rates among patients with B/LB results post-COVID, we did not observe a significant difference in CPM rates in the overall study population between the pre- and post-COVID eras after adjusting for clinical and demographic factors. Therefore, although the mode of delivery for genetic counseling and testing has shifted towards telehealth and oncologist-led testing, which has expanded access to genetic testing, there was no significant association with surgical decision-making for CPM. Prior studies have shown that telehealth is acceptable to patients, who report high rates of satisfaction with telehealth genetic testing14. However, to our knowledge, our study is the first to investigate the association between telehealth genetic counseling and surgical decision for CPM among patients with operable breast cancer.
Oncologist-led genetic testing models aim to increase access by allowing medical and surgical oncologists to initiate germline testing directly, particularly in environments where demand for genetic counselors exceeds capacity. However, the transition to oncologist-led testing, which may occur either at the initial or a subsequent visit, may affect patient understanding and interpretation of results, particularly in the case of patients with variants of uncertain significance (VUSs). In our study, Hispanic and Asian women were more likely to receive VUS results, consistent with previously documented disparities tied to underrepresentation of non-White populations in genomic databases16,17. Although we hypothesized that the trend towards higher CPM rates among Hispanic women in our population may reflect uncertainty or concern in the context of ambiguous genetic findings, our results did not show a difference in CPM rates by race/ethnicity among patients who had VUSs. There may be other factors influencing higher CPM rates among Hispanic patients which are not captured within our study, which warrant further investigation and emphasizing a need for improved risk communication and culturally responsive counseling. Overall, about a quarter of patients (28.4%) chose to have CPM, despite only 28.8% of those patients having a P/LP result. Although patient specific motivations for CPM were not captured in this study, prior literature suggests that decisions to pursue CPM among non-mutation carriers are often influenced by factors beyond genetic risk, including perceived contralateral breast cancer risk, cancer-related anxiety or fear of recurrence, desire for symmetry, and breast reconstruction considerations18,19. Another possible explanation for the trend towards higher CPM rates among Hispanic patients is a higher perceived risk for breast cancer recurrence. Prior studies have shown that among Hispanic patients undergoing screening mammography, 80% were found to overestimate their breast cancer risk20. Prior studies have also shown that Hispanic patients with non-metastatic breast cancer have increased worry about breast cancer recurrence compared to non-Hispanic White patients21. This increased worry may represent an area for improvement in care for Hispanic patients and highlights that further studies are needed to understand patient factors when deciding on CPM and to investigate methods for improving risk communication.
Our study has several limitations. First, it is a single-institution retrospective analysis and may not be generalizable to other settings, and does not capture more nuanced decision-making, including patient beliefs/understanding around CPM and genetic testing that may have influenced surgical decision-making. Second, classification of the mode of genetic counseling (e.g., in-person vs. telehealth) was subject to documentation variability. Third, although the rate of VUS results was over 20%, our study may have been underpowered to detect differences in CPM rates by racial/ethnic backgrounds among patients with VUS.
Despite these limitations, our findings highlight the critical role for germline genetic testing in guiding surgical decisions for patients with breast cancer and underscore the need to ensure equitable, informed surgical decision-making. Future studies should focus on refining models of genetic service delivery to enhance comprehension, reduce health disparities, and support tailored decision-making.
Methods
Study design
We conducted a retrospective cohort study in a population of women who have been diagnosed with operable unilateral breast cancer and underwent germline genetic testing between 2013 and 2022 at Columbia University Irving Medical Center (CUIMC) in New York, NY. Patients with metastatic breast cancer at diagnosis, synchronous bilateral breast cancer, and male breast cancer were excluded from this study. The study was conducted in accordance with the ethical standards of the Declaration of Helsinki. Ethical approval was not required for this retrospective chart review, and the study was approved by the CUIMC Institutional Review Board (IRB #AAAL5871). Informed consent was waived, given the retrospective design of the study.
Data collection
We collected demographic characteristics from the electronic health record (EHR), including age at time of breast cancer diagnosis, race/ethnicity (non-Hispanic White, non-Hispanic Black, Hispanic, Asian, other/unknown), and marital status (married, unmarried, unknown). We also collected clinical data from the EHR, including year of diagnosis (stratified by pre-COVID-19 from 2013-2019 and post-COVID-19 from 2020-2022), breast cancer stage (0, I, II, III, unknown), tumor hormone receptor status (positive, negative, unknown), family history of breast cancer (yes, no/unknown), genetic test results (pathogenic/likely pathogenic [P/LP], variants of uncertain significance [VUS], and benign/likely benign [BLB]), and type of genetic counseling visit (in-person with genetic counselor, telehealth visit with genetic counselor, oncologist-led, unknown). Patients with both P/LP and VUS were documented as having P/LP. Genetic testing at CUIMC was performed according to NCCN guideline criteria.
Our primary outcome of interest was rates of CPM pre- and post-COVID-19. The extent of breast surgery (lumpectomy, unilateral mastectomy, and bilateral mastectomy) was ascertained from operative reports or outpatient clinic notes. Patients who underwent multiple breast surgeries were classified by their most advanced surgery (bilateral mastectomy > unilateral mastectomy > lumpectomy).
Statistical analysis
Baseline characteristics were stratified according to type of surgery received. We used the chi-squared test and Fisher’s exact test to evaluate for differences in the distribution of baseline characteristics. We conducted univariable and multivariable logistic regression analyses to estimate the odds ratio and 95% confidence interval associated with each variable and receipt of CPM. All statistical analysis was conducted using SAS OnDemand (SAS Institute, Cary, NC), and a p-value < 0.05 was considered statistically significant for the multivariable logistic regression.
Data availability
The dataset generated and analyzed for this study is not publicly available due to patient privacy restrictions, but access to the de-identified minimum dataset, which is necessary to interpret, verify, and extend the research are available from the corresponding author upon reasonable request and with approval from the CUIMC IRB.
References
Angelos, P. et al. Contralateral prophylactic mastectomy: challenging considerations for the surgeon. Ann. Surg. Oncol. 22, 3208–3212 (2015).
King, T. A. et al. Clinical management factors contribute to the decision for contralateral prophylactic mastectomy. J. Clin. Oncol. 29, 2158–2164 (2011).
Tuttle, T. M. et al. Increasing rates of contralateral prophylactic mastectomy among patients with ductal carcinoma in situ. J. Clin. Oncol. 27, 1362–1367 (2009).
Agarwal, S., Pappas, L., Matsen, C. B. & Agarwal, J. P. Second primary breast cancer after unilateral mastectomy alone or with contralateral prophylactic mastectomy. Cancer Med. 9, 8043–8052 (2020).
Portschy, P. R., Kuntz, K. M. & Tuttle, T. M. Survival outcomes after contralateral prophylactic mastectomy: a decision analysis. J. Natl. Cancer Inst. 106, dju160 (2014).
Blondeaux, E. et al. Association between risk-reducing surgeries and survival in young BRCA carriers with breast cancer: an international cohort study. Lancet Oncol. 26, 759–770 (2025).
Kostov, A. M. et al. Timely germline BRCA testing after invasive breast cancer promotes contralateral risk-reducing mastectomy and improves survival: an observational retrospective study. Breast Cancer Res. Treat. 212, 309–323 (2025).
Metcalfe, K. et al. Contralateral mastectomy and survival after breast cancer in carriers of BRCA1 and BRCA2 mutations: retrospective analysis. BMJ 348, g226 (2014).
Heemskerk-Gerritsen, B. A. M. et al. Improved overall survival after contralateral risk-reducing mastectomy in BRCA1/2 mutation carriers with a history of unilateral breast cancer: a prospective analysis. Int. J. Cancer 136, 668–677 (2015).
Ager, B. et al. Contralateral prophylactic mastectomy (CPM): a systematic review of patient reported factors and psychological predictors influencing choice and satisfaction. Breast Edinb. Scotl. 28, 107–120 (2016).
Hawley, S. T. et al. Social and clinical determinants of contralateral prophylactic mastectomy. JAMA Surg. 149, 582–589 (2014).
Bokkers, K. et al. The feasibility of implementing mainstream germline genetic testing in routine cancer care-A systematic review. Cancers 14, 1059 (2022).
Stoll, K., Kubendran, S. & Cohen, S. A. The past, present and future of service delivery in genetic counseling: Keeping up in the era of precision medicine. Am. J. Med. Genet. C. Semin. Med. Genet. 178, 24–37 (2018).
Danylchuk, N. R. et al. Telehealth for genetic counseling: a systematic evidence review. J. Genet. Couns. 30, 1361–1378 (2021).
Vaynrub, A. et al. The breast cancer genetic testing experience: probing the potential utility of an online decision aid in risk perception and decision making. BMC Cancer 25, 19 (2025).
Sirugo, G., Williams, S. M. & Tishkoff, S. A. The missing diversity in human genetic studies. Cell 177, 26–31 (2019).
Ro, V. et al. Association between genetic testing for hereditary breast cancer and contralateral prophylactic mastectomy among multiethnic women diagnosed with early-stage breast cancer. JCO Oncol. Pract. 18, e472–e483 (2022).
Srethbhakdi, A., Brennan, M. E., Hamid, G. & Flitcroft, K. Contralateral prophylactic mastectomy for unilateral breast cancer in women at average risk: systematic review of patient reported outcomes. Psychooncology 29, 960–973 (2020).
Jagsi, R. et al. Contralateral prophylactic mastectomy decisions in a population-based sample of patients with early-stage breast cancer. JAMA Surg. 152, 274–282 (2017).
Graves, K. D. et al. Perceived risk of breast cancer among Latinas attending community clinics: risk comprehension and relationship with mammography adherence. Cancer Causes Control CCC 19, 1373–1382 (2008).
Janz, N. K. et al. Correlates of worry about recurrence in a multiethnic population-based sample of women with breast cancer. Cancer 117, 1827–1836 (2011).
Acknowledgements
This work was supported by the National Institutes of Health, National Cancer Institute ELISABETH grant (UG3CA297490) and CCSG grant (P30CA013696). The funder had no role in study design, data collection, data analysis, data interpretation, or writing of the manuscript.
Author information
Authors and Affiliations
Contributions
V.R., A.M., and K.L. completed the data analysis. A.M. and V.R. wrote the main manuscript text. K.D.C, M.S.T., R.R., E.L., C.K., and R.K. contributed to the drafting of the manuscript. K.D.C. supervised the data analysis and writing of this manuscript. K.D.C. obtained funding. All authors contributed to the critical review of the manuscript for important intellectual content.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Michel, A., Ro, V., Luo, K. et al. Impact of telehealth genetic counseling on contralateral prophylactic mastectomy rates among multi-ethnic breast cancer patients. npj Womens Health 4, 17 (2026). https://doi.org/10.1038/s44294-026-00137-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44294-026-00137-8