Abstract
Intelligent blood pressure monitoring is transforming hypertension management. In this review, we introduce a novel classification based on measurement principles, categorizing devices into mechanical and non-mechanical methods. We offer fresh insights into the non-mechanical pipeline: advanced sensors, data preprocessing, modeling algorithms, and calibration strategies. We analyze clinical validation standards and limitations, highlighting the pressing need for updated frameworks tailored to next-generation cuffless systems blood pressure, non-mechanical monitoring, deep learning.
Similar content being viewed by others
Introduction
Hypertension is a major global concern and a major risk factor for cardiovascular diseases. According to the World Health Organization (WHO), an estimated 1.4 billion people aged 30–79 years were living with hypertension in 20241. Hypertension notably elevates the risk of severe complications, including stroke, coronary artery disease, heart failure, and end-stage renal disease. However, nearly 46% are unaware of their condition, and over half are not receiving the treatment they need2. The alarming contrast between the high prevalence of hypertension and the low rate of awareness underscores a critical gap in global health management. A key step in addressing this challenge lies in ensuring accurate and accessible blood pressure (BP) measurement methods3.
As shown in the timeline in Fig. 1, the history of BP monitoring reflects a continuous evolution driven by the need of better accessibility and comfort. Invasive BP measurement, typically performed at the radial or femoral artery, was first performed in the 18th century and remains the clinical reference method due to its accuracy4. However, it is confined to surgical and critical care settings due to the need for intra-arterial cannulation. To enable routine monitoring, the auscultatory method introduced in 1905 by detecting Korotkoff sounds through a compressed brachial artery. Although it remains the gold standard for non-invasive measurement, it requires trained personnel and is prone to subjective interpretation. The oscillometric method developed in 1976 automated this process by analyzing the pressure oscillation envelope at the upper arm or wrist to estimate BP5. While these cuff-based methods are invaluable, they share critical drawbacks. First, they require high-pressure cuff inflation, which can cause discomfort and disturbance, limiting the use for frequent monitoring6. Second, they can only provide readings under static conditions, making them unsuitable to capture dynamic BP fluctuations induced by daily physical or psychological activity7.
Key milestones in the evolution of blood pressure monitoring. The timeline highlights representative devices and time points. Images are adapted with permission from Springer Nature50,65,175,177,178, AAAS179,180, and ACM14. The Invasive Catheter, Mercury Manometer, and Automated Arm Cuff images are courtesy of Red Roan, Michael V Hayes, and Jacek Halicki, respectively, used under CC BY-SA licenses.
In the last few years, BP measurement technology has been accelerating rapidly, driven by the advance in consumer electronics and new sensors. In particular, BP measurement has been integrated into different kinds of wearable devices, such as patches8, smart watches9, rings10, in-ear devices11, and neckbands12 so as to enable ubiquitous BP tracking. Beyond wearable devices, there is a growing trend towards fully contact-less solutions. Recent work has demonstrated the feasibility of estimating BP by contactless sensors, such as remote photoplethysmography (rPPG)13 and millimeter-wave (mmWave) sensors14,15. To navigate the wide range of BP measurement methods, this paper presents a systematic review from the new perspective of measurement principles, with a particular focus on the rapidly emerging approaches for convenient and continuous BP monitoring. Unlike existing review works, our work differs in two key aspects:
New perspective on categorizing existing works and the consequent new insights
Previous reviews typically classified BP technologies by device form factors—such as wearable or cuff-less designs—which reflect usage scenarios16,17,18,19,20,21. In contrast, we propose a more fundamental classification based on whether the final BP values are obtained through direct pressure measurement or indirect estimation from physiological surrogates. This divides into mechanical and non-mechanical. Mechanical methods rely on force transduction through physical application (occlusion or applanation). The measurement directly correlates with physical pressure, and the output is expressed in the standard unit of mmHg. While non-mechanical methods estimate BP from measured physiological signals (e.g., pulse waveform, vascular radius). These signals serve as surrogates, and the final BP value is derived via a computational model. This categorization offers new insights, as the two types differ significantly in sensing, modeling, and signal interpretation. We focus on non-mechanical methods and provide an in-depth analysis of their full processing pipeline: signal acquisition, preprocessing, modeling, and calibration.
Analysis on clinical validation
Despite the promising potential of non-mechanical technologies, significant challenges remain in translating current technologies into clinical applications. To bridge this gap, we conduct a comprehensive investigation into clinical validation, an often overlooked aspect in previous reviews. We summarize international validation standards, outlining their limitations and applicability to underscore the need for revised frameworks tailored to next-generation BP monitoring systems. We also examine both academic prototypes and commercial devices that have undergone clinical validation, highlighting the extent of their compliance with criteria, and common shortcomings.
Measurement Principle
Based on the fundamental principle of BP derivation, the emerging intelligent BP monitoring has evolved into two conceptual categories: Mechanical Methods and Non-mechanical Methods, as visually summarized in Fig. 1.
Mechanical Methods
This category, depicted by the blue arrow in Fig. 1, encompasses representative techniques whose unifying principle is the active application of external mechanical pressure to the artery. These methods rely on force transduction, where the measurement directly correlates with the physical pressure. Historically, this category includes Invasive catheter22, classic cuff devices, such as the Mercury Manometer23, and Automated Arm Cuff devices24. More recently, this classification extends to continuous methods based on applanation tonometry25 and the vascular unloading principle 26,27. It is worth noting that the necessity of implementing external pressure defines the classification. Although methods like Vascular Unloading and emerging Finger-press Oscillometry28 utilize advanced sensors (like PPG) for system control or oscillation detection, their core function requires the active application of external counter-pressure for measurement.
Non-mechanical Methods
In contrast, the Non-mechanical category, shown by the green arrow, infers BP values by measuring related physiological parameters that serve as surrogates. These signals are then processed and modeled to derive the final BP value. They basically rely on the principles of the following three methods:
Vessel Wall Tracking
During the cardiac cycle, heart contraction leads to the increased BP, resulting in the expansion of blood vessels, while relaxation leads to a decrease in blood flow and subsequent vessel contraction. Thus, by tracking the changes in the vessel wall diameter and transforming them into the corresponding BP waveform, BP values can be obtained29. This relationship is locally dependent and is often applied to skin regions capable of clearly detecting the target artery’s pulse, such as the forearm, brachium, neck, chest, abdomen30. The mathematical transformation can be represented by:
where \(p(t)\) and \(A(t)\) represent the BP values and arterial cross-section at time \(t\) at the measured site, respectively, while \({p}_{D}\) is DBP and \({A}_{D}\) is the diastolic arterial cross-section at the measured site. The coefficient \(\alpha\) represents the vessel rigidity, and can be calculated as:
where \({p}_{S}\) is SBP and \({A}_{S}\) is systolic arterial cross-section. \(A(t)\) can then be calculated as:
where \(d(t)\) is the radius waveform of the measured artery.
Pulse Wave Velocity
Pulse Wave Velocity (PWV) is defined as the velocity of pulse wave propagation in the artery. It serves as an important clinical indicator for assessing vascular function and predicting the risk of cardiovascular events31,32. Extensive experiments have demonstrated that higher PWV is associated with a greater risk of hypertension33. Besides, the variation of PWV can be utilized to correlate the absolute BP change individually34. PWV is typically represented by the Moens-Korteweg (MK) equation35:
where \(E\) is the modulus of elasticity, \(h\) is the thickness of arterial wall, \(\rho\) is the blood density and \({d}_{D}\) is the diameter of the artery at the end of diastole. As a more accessible approach to BP estimation, Pulse Transit Time (PTT) refers to the time it takes for the pulse wave to travel through a segment of the arterial tree, typically measured as the delay between the foot points of pulse waveforms obtained at two distinct anatomical sites36. A related and often more practical metric is Pulse Arrival Time (PAT)37. PAT is defined as the interval from the cardiac electrical activation (e.g. ECG R-wave on the chest) to the arrival of the pulse at a peripheral artery, such as the finger32. Additional variants also can characterize pulse wave timing within the vasculature. For example, Reflection Wave Transit Time (RWTT) measures the interval or amplitude difference between the forward and reflected pulse waves at the same anatomical site38,39.
Pulse Wave Analysis
With the development of machine learning and non-mechanical devices, Pulse Wave Analysis (PWA) has become a focal area for estimating BP values by analyzing the shape and characteristics of the pulse waveform. Analysis involves methods ranging from extracting explicit temporal and morphological features40,41 to leveraging deep learning for end-to-end signal analysis42. While PWA differs from the above methods in that it relies less on a single explicit bio-mechanical equation, extensive data-driven evidence demonstrates that waveform morphology inherently and robustly encodes critical hemodynamic status, including cardiovascular characteristics, such as arterial elasticity and stiffness43.
Data Acquisition
As illustrated in Fig. 2, the typical process flow for non-mechanical BP estimation encompasses four key phases: (i) acquiring biosignals using sensors, (ii) preprocessing the biosignals, (iii) modeling the relationship between the biosignals and BP, and (iv) calibration. Each of these components will be addressed in detail. The first step is to collect effective BP-related biosignals that reflect cardiovascular activity. We will introduce the existing novel non-mechanical sensors and the principle of how they obtain biosignals in Section 3.1. Then, we introduce the available large-scale datasets and their characteristics in Section 3.2.
The process involves four critical stages: sensors to capture biosignals, preprocessing to enhance signal quality, estimation modeling to derive BP values, and calibration to ensure accuracy across individuals and conditions.
Advances in Biosignal Acquisition
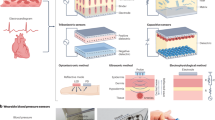
With the advancement of technology, a growing diversity of techniques for obtaining biosignals has emerged. These methods can be categorized into two types based on the requirement of physical contact with the skin: Contact-Based methods require direct physical coupling between the sensor and the skin, generally offering high signal fidelity but potentially compromising long-term comfort or adherence. Conversely, contactless methods operate remotely, significantly enhancing user comfort and enabling passive, long-term monitoring. However, these methods often require complex signal processing to mitigate ambient noise and interference. The following sections detail the acquisition principles and representative examples for each type of monitoring. The vast array of data modalities relevant to these two categories is visually summarized in Fig. 3. This figure provides an overview of the key biosignals used for non-mechanical BP estimation, demonstrating the distinct morphology and temporal synchronization of signals obtained via both contact-based and contactless acquisition methods44,45,46,47,48,49.
rPPG images adapted from13 (Wolters Kluwer Health).
Contact-based Devices
Ultrasound operates by generating high-frequency sound waves that can penetrate the body and reflect off internal structures. As a widely used diagnostic tool in medical imaging, ultrasound can also be applied to assess BP values by measuring the dynamic changes in arterial diameter. To achieve this, the ultrasound probe or patch is placed over specific arterial sites, typically the carotid artery (CA), brachial artery (BA), and radial artery (RA). High-frequency sound waves are then emitted and travel through tissues, reflected back to the transducer upon encountering interfaces including the vessel walls. By continuously recording the time taken for the echo to return, the device can locate the dynamic change of anterior and posterior vessel walls and estimate BP values according to vessel wall tracking method as mentioned in Section 2.2.1. The recent emergence of wearable ultrasound patches50,51 adopts ultra-thin flexible circuits and stretchable sensor arrays, enabling continuous and non-invasive monitoring. However, the cost of single use for such wearable solutions remains relatively high.
Photoplethysmography (PPG) is one of the most extensively explored technologies for BP measurement52,53. It uses light to probe blood volume changes beneath the skin of the ear canal, fingers, and wrist54,55. As light passes through tissue, its absorption varies with pulsatile blood flow. During systole, increased arterial blood volume leads to greater absorption; during diastole, absorption decreases accordingly56. By analyzing these signal fluctuations, signals regarding cardiovascular health and hemodynamic parameters can be extracted. However, variations in skin tone and thickness can affect the quality of PPG signals, as darker skin tones and thicker skin layers tend to absorb more light and reduce signal strength57. The pressure applied to PPG sensor can also impact the amplitude and shifts of signals58. A potential solution is to integrate a sensor capable of measuring the applied force, which allows for standardizing PPG measurements across different users and conditions59.
Seismocardiography (SCG) and ballistocardiography (BCG) detect the subtle mechanical vibrations generated by the heart’s activity, including contractions and blood flow within each heartbeat. SCG typically uses an accelerometer placed on the chest to capture the mechanical vibrations from the chest wall due to heart activity60,61. Devices for BCG measurement typically employ highly sensitive sensors, such as electromechanical62, accelerometric63, piezoelectric64, fiber-optic65,66 and hydraulic sensors67. These BCG sensors are positioned unobtrusively beneath the user on surfaces, such as mattresses, chairs, or insoles, capturing pressure changes in the body at the wrist and feet, as well as in sitting and lying postures, without requiring direct contact with user. Due to its high sensitivity to external movements and vibrations, SCG and BCG are prone to noise and artifacts, which can interfere with the clarity of signal68,69.
Phonocardiogram (PCG) measures the acoustic manifestations of the cardiac cycle, i.e. the heart sounds (\(\text{S}1\) and \(\text{S}2\)), which are generated by the mechanical action of valve closure and blood flow turbulence70. PCG is acquired using highly sensitive microphones or acoustic sensors placed on the chest or neck. It can provide precise timing markers (e.g., \(\text{S}1\)) for the accurate decomposition of \(\text{PAT}\) when combined with other sensors71. In addition, the \(\text{PCG}\) waveform itself is utilized in end-to-end deep learning models for BP prediction72. However, heart sound signals are extremely weak (usually between 20-200 Hz) and are easily drowned out by external sounds.
Deformable wearable mechanoelectric sensors convert mechanical stimuli from arterial deformation or vibration into electrical signals using various transduction mechanisms. They are typically placed on the radial artery or brachial artery to detect pulse waves. Common types include piezoelectric (stress-induced charge generation)73,74, triboelectric (contact-induced charge transfer)75, piezoresistive (pressure-induced resistance change)76,77, and capacitive (deformation-driven capacitance change) sensors78. While these patch-based sensors offer high sensitivity, ensuring stable, conformal contact on moving skin and long-term durability remains a key research challenge.
Bioimpedance (Bio-Z) is typically performed using a four-electrode configuration to enhance measurement accuracy. Two outer electrodes inject a small alternating current, while two inner electrodes measure the resulting voltage to derive the impedance value. When these four electrodes are aligned with the objective artery, they measure the underlying tissue bioimpedance, which typically ranges from \(10-100\Omega\). The fundamental principle is that the change in impedance is inversely related to the pulsatile change in blood volume, generating a pulse bioimpedance waveform that reflects the pressure pulse wave79,80.
Electrocardiogram (ECG) captures heart activity by detecting electrical impulses, serving as a highly precise timing reference in cardiovascular measurements. Electrodes are accurately placed on the skin of the chest and limbs to measure the potential difference. The distinct \(\text{R}\)-wave in the \(\text{ECG}\), corresponding to ventricular depolarization, is utilized as the reliable fiducial point for starting time-interval measurements, such as PAT81,82. In wearable applications, simplified single-lead configurations, often taken across the chest or wrist-to-wrist, are typically employed in conjunction with other sensors to calculate \(\text{PAT}\).
Contactless Devices
RF signals, including Ultra-Wideband (UWB), and millimeter-wave (mmWave) radar, have gained popularity in BP estimation. The radar system emits electromagnetic waves that can penetrate clothing, with some of the energy being absorbed by the body and a portion reflected back to the receiving antenna83. By analyzing the phase changes and signal strength of the reflected signals, the system precisely tracks the minor displacement of the chest wall or the arterial skin surface. UWB signals offer high temporal resolution and have been integrated into existing smart devices84,85. mmWave radar (typically in the 60 GHz, or 77 GHz bands in BP estimation) provides even finer spatial and temporal resolution thanks to its short wavelength and wide bandwidth14,86,87. These RF-based approaches enable unobtrusive BP monitoring without direct skin contact, though challenges remain in susceptibility to environmental interference and precise sensor positioning.
Remote photoplethysmography (rPPG) operates on principles similar to contact-based PPG, which employs an RGB camera and a light source to capture color video of the skin region, typically the face or palm. The system identifies regions of interest within each frame and extracts raw signals from different color channels. By analyzing the fluctuations of signals over time, rPPG can derive physiological parameters, such as heart rate and BP values88,89. However, challenges remain in ensuring consistent performance under varying environmental conditions and skin tones, which can affect signal quality and accuracy90.
Public Dataset
In addition to building custom systems using the aforementioned sensing methods, there are also publicly available biosignal-BP datasets that can facilitate research. Table 1 summarizes commonly used datasets that are invaluable for developing reliable BP estimation models. In experimental and testing processes, accounting for factors such as sample size, demographic diversity, and cardiovascular disease risk among subjects can significantly advance the applicability of intelligent BP measurement systems to clinical settings.
Data Preprocessing
After obtaining biosignals, conducting data preprocessing is crucial for enhancing the reliability and robustness of BP measurement systems. In section 4.1, we introduce methods for denoising to remove environmental and physiological noise. Section 4.2 covers data augmentation techniques used to expand and diversify training datasets for more robust model performance, while Section 4.3 focuses on aligning multimodal data streams accurately across devices.
Denoising Technique
Physiological signals are often contaminated with various types of noise, including inherent sensor properties, such as power line interference, baseline wander, motion artifacts and ambient noise. These noises can obscure critical features of the signal, making accurate analysis and interpretation challenging. To prevent outliers from impacting subsequent processing, outlier detection and segment rejection are typically the first step to clean severely affected parts of the signal. Manual checks are used to exclude irregular and distorted segments, while the autocorrelation filters can discard corrupted segments85. The most common approach for further signal refinement involves traditional filtering techniques, which effectively suppress noise and enhance signal quality91. Tailored noise suppression strategies have also been developed for different sensing modalities. For example, for PPG, adaptive light intensity control is often used to balance baseline levels while preserving pulsatile components92. In mmWave sensing, multi-antenna phase compensation techniques can enhance pulse signal coherence93. Recent advances in deep learning have introduced data-driven denoising methods that learn to separate physiological signals from noise based on large training datasets94,95.
Data Augmentation
Due to the challenges in collecting extensive high-quality BP-signal data, data augmentation through variations of existing data is essential. One approach involves applying transformations to current data, ranging from traditional methods (such as flipping and cropping) to deep learning methods96. Another approach involves gathering diverse data details from a smaller subset and generalizing these variations across the dataset. By collecting signals at varying intensities, positions, and angles and utilize the different waveform patterns to unify the measurement in different situations61,97. In addition, BP datasets often suffer from imbalances. Certain BP ranges or personal information can restrict the output of model to the distribution of the training data98. Generating synthetic waveform data using deep learning can create more diverse and balanced dataset, enabling the model to learn a broader range of patterns and improving its robustness to real-world variations99,100.
Synchronization Between Devices
In BP estimation works, multiple devices are often used to capture physiological signals. However, the temporal misalignment of data could lead to unreliability of the estimation algorithms, especially for the PTT-based methods since it is extremely short, ranging from only tens to hundreds of milliseconds (\(\text{ms}\))101. Hence, accurate synchronization of multiple devices is crucial in BP estimation. A common approach involves generating an external stimulus that can be detected simultaneously by all sensors, enabling precise alignment and calibration of timing. For instance, interrupt signals, physical taps, speaker-generated beeps, or fixed-length data frames are used as stimuli to ensure synchronization across different sensors81.
Estimation Model
This section provides a comprehensive review of various models used for BP estimation, categorized into formula-based, machine learning, and deep learning methods.
Formula-based methods
Back in 1976, the theoretical foundation of pulse wave velocity (PWV) dependence on arterial pressure was established, with experimental evidence supporting the linear relationship between PWV and BP102. Based on the Moens-Korteweg (M-K) equation and vessel elasticity theory103, logarithmic and exponential dependencies were derived under the assumption of constant vessel thickness and radius104. To enhance individual accuracy, modeling has incorporated \(\text{PTT}\) alongside other physiological parameters, such as Heart Rate (\(\text{HR}\)) and morphological features105, providing the basis for the subsequent PTT-BP estimation algorithm.
Machine learning-based methods
Machine learning methods can offer greater flexibility in capturing the nonlinear relationships between pulse waveform features and BP. Traditional machine learning methods include decision trees (DT)106, random forest regression (RFR)107, adaptive boosting(AdaBoost)108, and K-nearest neighbors (KNN)109. Nisio et al.110 extracts 195 features from PPG pulse waveforms and evaluates the performance of various machine learning models. Among them, gaussian process regression (GPR) can capture complex nonlinear relationships and XGBoost excels in robust gradient boosting. Experiments indicate that their combination achieves the optimal trade-off between training time and accuracy.
Machine learning methods typically require careful feature engineering, which can introduce inaccuracies due to variations in signal quality and the absence of key features. Ideally, a pulse waveform contains the systolic peak, reflected wave, and dicrotic notch in a sequential pattern111. However, in risky groups, such as elderly individuals or hypertensive patients, the reflected wave may shift or even precede the systolic peak112.
Deep learning-based methods
Advanced deep learning methods utilize multi-layer neural networks to autonomously learn from large-scale datasets. This section discusses commonly used deep learning models in BP estimation algorithms, with representative studies, experimental scales, and results summarized in Table 2. Convolutional Neural Networks (CNN), originally designed for image processing, is highly effective in extracting hierarchical features from the pulse waveform. This process effectively replaces manual waveform feature extraction by finding optimal local feature maps across the pulse waveform113,114,115. Recurrent Neural Network (RNN) incorporates feedback connections to model temporal dependencies in sequential data116. Long Short-Term Memory (LSTM) network and the Gated Recurrent Unit (GRU) significantly improve upon RNN by using sophisticated gating mechanisms. They are particularly effective at modeling the long-term temporal dynamics and sequence-to-sequence relationships inherent in continuous pulse waveforms117,118,119. More advanced DL frameworks utilize specialized structures to refine sequential processing and fusion. The Encoder-Decoder framework processes the input signal sequence into a compressed latent representation, which serves as a compact summary of the underlying hemodynamic state, and then reconstructs the desired output or feature map120,121,122. The Attention mechanism is a crucial breakthrough. By allowing the decoder to selectively and dynamically focus on critical, BP-relevant feature points within the input sequence, it significantly enhances predictive performance123,124,125. Beyond traditional predictive modeling, Physics-Informed Neural Network (PINN) integrates theoretical rigor by incorporating known physical laws, such as the \(\text{Moens}-\text{Korteweg}\) equation, directly into the network’s loss function. By enforcing adherence to these physical constraints during training, \(\text{PINN}\) ensures that the resulting \(\text{BP}\) predictions are physically plausible, thereby enhancing the generalization ability and interpretability of the model126,127.
Although a single physiological modality provides valuable advancement, it inherently provides only a partial view of the complex cardiovascular system, and multimodal fusion has become increasingly popular. As Fig. 3 shows, each modality captures a distinct aspect of the cardiac cycle. For example, \(\text{ECG}\) provides precise electrical timing, \(\text{PPG}\) reflects peripheral volumetric changes, while \(\text{mmWave}\) quantifies vibration momentum. By leveraging the complementary physiological information from different sensor modalities, fusion strategies mitigate individual sensor limitations. In addition to improving accuracy, this enhances resilience by compensating for modality-specific biases (such as \(\text{PPG}\)’s sensitivity to skin tone), thereby achieving a more robust representation of the underlying hemodynamic state128,129,130.
Calibration
Unlike traditional oscilloscope devices that directly provide absolute BP values, cuff-less BP monitoring is particularly sensitive to individual physiological variations, such as posture, physical activity, emotional state, and long-term cardiovascular changes, as well as to environmental factors and sensor variability. Therefore, calibration is essential to assist in mapping the recorded signals to actual BP values. The calibration strategies can generally be divided into three categories:
Single-point calibration
These methods involve repeatedly measuring BP under the same physiological and environmental conditions and selecting part of these measurements from the same subject as the calibration standard and the rest serveing as verification. For instance, commercial solutions like Samsung Health Monitor131 and Somnotouch-NIBP132 mandate a rigorous calibration process before use. This process requires users to perform initial measurements against a medical-grade arm cuff (typically involving two to three comparison readings in a row) and necessitates periodic re-calibration (e.g., every 28 days for \(\text{Samsung}\)) to mitigate calibration drift. These methods may struggle to accurately track fluctuations in BP changes under varying conditions. Hence, they require frequent re-calibration to maintain accuracy, which poses challenges in ensuring reliability during long-term monitoring133.
Multi-point calibration
These methods introduce interventions that alter the environment or physiological state, allowing for a more comprehensive calibration by fitting multiple data points collected in different states. Huang et al.134 utilized a multi-point approach in laboratory settings, where the training data included signals collected during induced physiological changes (e.g., ice-water stimulation) to ensure a wide range of \(\text{BP}\) fluctuations is captured. For their long-term monitoring or \(\text{ICU}\) scenarios, this calibration is typically achieved by training the model on data collected during one distinct time segment and validating it on data from a later time segment, ensuring the model can adapt to temporal drift and maintain accuracy across different physiological states. Colburn et al. 135 presented a novel approach that leverages the hydrostatic pressure effect by adjusting the arm to different heights relative to heart level to induce pressure perturbations, thereby generating optimal PTT predictions using personalized calibration.
Model calibration
This approach leverages a pre-trained model from a large dataset and adapts it to the target individual’s data. The generalization capabilities enable it to capture common physiological patterns across a population. Nguyen et al.136 proposed a three-stage model specifically designed for BP prediction in pregnant women. It involves a baseline model trained from the general population, fine-tuning it for the pregnant population, and further personalizing the model to enhance accuracy for individual subjects. Fan et al.137 designed a few-shot transfer learning approach and achieved MAE of 6.68 mmHg for SBP and 3.91 mmHg for DBP with only ten pairs of personal data samples in cold pressor and HIIT tests. Hu et al.138 propose a Cross-Modality Knowledge Transfer (CMKT) strategy that leverages a teacher model pre-trained on the MIMIC dataset (ECG/PPG) to calibrate a millimeter-wave student network, embedding generalized cardiac knowledge. To further address individual physiological heterogeneity, they employ a subject-specific personalization scheme that fine-tunes the generalized model using approximately 20 minutes of target data, significantly mitigating distribution shifts and reducing estimation errors.
However, there remain significant challenges in the practical application of calibration. Firstly, only when the reference values are accurate, can the predicted BP be reliable. Yet, home-use wrist and arm-based oscillometric devices are often prone to overestimation, and their accuracy can degrade further over time if not regularly re-calibrated139. Mehta et al.140 pointed out that several studies have employed short time-scale data for BP estimation, where the actual values tend to remain relatively stable. As a result, even if a model predicts constant values, it can still achieve deceptively high accuracy, giving the false impression of model effectiveness. Additionally, the accuracy levels of using discrete BP data points as reference values and continuous BP waveforms are different141, but using continuous waveforms is impractical in typical home-use monitoring devices. Secondly, there is limited systematica research conducted on the available frequency of calibration, and yet no standardized guidelines exist for determining how often calibration should be performed142. Han et al.143 investigated the accuracy of user-driven re-calibration, revealing a pre-post calibration absolute \(\text{SBP}\) difference of \(6.8\pm 5.6\) mmHg (with a range up to \(33.8\text{mmHg}\)). Crucially, this significant drift was not due to intrinsic device instability (found to be \(\approx 0.02\text{mmHg}/\text{day}\)), but was primarily influenced by changes in the participant’s average physiological \(\text{SBP}\) level prior to re-calibration. The 2023 ESH consensus highlights that the lack of accuracy in verification reinforces the necessity for frequent calibration. Fundamentally, these cuffless devices track changes relative to a calibration point or attempt to predict BP using demographic and machine learning techniques rather than measure BP. We advocate for more research to validate the effectiveness of methods through long-term monitoring application, specifically investigating the impact of calibration frequency and methods.
Validation
Clinical validation ensures that the device performs reliably under practical conditions, accounting for physiological variability, environmental noise, and diverse patient populations—factors often absent in controlled lab experiments. However, most research remains in the laboratory validation stage to date, with only a few studies having conducted clinical validation of their devices to demonstrate their accuracy in specific clinical environments. Among them, studies 144,145,146,147,148,149 have undergone clinical validation and reported BP measurement results, though they did not align with international standards. Research and commercial devices including FreeScan150, LiveOne151, Lifelight152, CardioWatch153 have assessed accuracy based on the AAMI/ESH/ISO universal standard154. However, some studies have only met the error requirements, without fulling other essential criteria, such as including a minimum of 85 subjects or ensuring a sufficient representation of cases with abnormal BP values.
Recognizing the unique characteristics of cuffless devices in measurement principles, sensing modalities, functionalities, and calibration154, emphasizes the need for distinct validation criteria. The IEEE has released the standard protocol IEEE 1708-2014155 and it has been adopted in various studies for validation65. The most comprehensive recent effort comes from the European Society of Hypertension (\(\text{ESH}\)) Working Group (2023)156. The \(\text{ESH}\) explicitly recommends six distinct validation tests for cuffless \(\text{BP}\) devices, moving validation beyond simple absolute accuracy: These tests include the static test (absolute \(\text{BP}\) accuracy), device position test (hydrostatic pressure effect robustness), treatment test (\(\text{BP}\) decrease accuracy), awake/asleep test (\(\text{BP}\) change accuracy), exercise test (\(\text{BP}\) increase accuracy), and the re-calibration test (stability over time). Successful completion of all required tests, depending on the device type, is necessary for clinical recommendation. Despite these significant advances, a universally accepted clinical protocol for all intelligent \(\text{BP}\) monitoring devices has not yet been achieved8.
Future Direction
Despite significant progress in laboratory conditions, the clinical adoption of cuffless BP measurement has remained cautious due to the lack of full validation of its reliability. Improving technical methods, enhancing real-world accessibility, refining evaluation standards, and establishing clinical relevance are key directions to build clinical trust in the work of new researchers and ensure its application in mHealth.
-
Integration with Large-Scale Foundation Models for Diagnostics. Existing work has already demonstrated the success trail of large models in complex medical tasks157,158,159. For \(\text{non}-\text{mechanical BP monitoring}\), we envision leveraging the capacity of Large Language Models (LLM) to integrate structured biosignals with rich clinical narratives and patient history. By processing these heterogeneous data types concurrently, the \(\text{LLM}\) could establish a context-aware unified representation of the patient’s hemodynamic state. This approach promises truly personalized, context-aware \(\text{BP}\) prediction, thus significantly enhancing the robustness and clinical utility of cuffless devices.
-
Enhancing usability in diverse daily scenarios. Despite the portability of wearable devices, there remains room for improving 24 h comfort and achieving truly imperceptible designs. For contactless systems, future development should aim to reduce sensitivity to body posture and alignment without requiring active user adjustment. In addition, charging frequency for sensors requiring high sampling rates or continuous wireless transmission remains a critical limitation.
-
Exploring new sensing modalities. New sensing technologies hold significant potential for improving the quality of pulse detection and BP measurement. For example, terahertz (THz) technology160, with its high resolution and sensitivity, can help detect more detailed variations in pulse signals. Photoacoustic imaging161 offers a more interpretable approach to observing pulse dynamics.
-
Improving evaluation criteria. Considering the characteristics of non-mechanical BP monitoring, a standardized validation protocol should incorporate the following requirements: 1) Reference Devices: Given the trend towards continuous monitoring, it is essential to determine whether a clinical catheter-based device should be used as reference2) Sensor-Specific Validation Criteria: Each sensor type requires tailored validation to ensure accuracy, such as the skin tone sensitivity of PPG. 3) Location Requirements: Specify the allowable range of positions for sensor placement, as non-mechanical sensors are sensitive to the physical posture and activity of the subjects. 4) Dataset Separation: To ensure generalization, training and testing datasets should be separated by distinct populations reasonably in deep learning model. 5) Calibration efficiency: Assess its stability over time and across different activities and environmental conditions.
-
Investigating the relationship with diseases. Different monitoring technologies may be better suited for specific sub-types of hypertension patients and populations. For instance, contactless systems are particularly advantageous for long-duration nocturnal monitoring, wearable sensors with high sampling rates and motion tolerance are more appropriate for tracking BP dynamics of orthostatic hypertension162. In specific populations, such as pregnant women, adolescents, and cardiovascular patients, monitoring priorities vary significantly in terms of required accuracy, frequency, and safety. Additionally, exploring the relationship between BP and CVDs is likely to become a key focus of future research.
Conclusion
Accurate, continuous, and user-friendly BP monitoring is essential for addressing the global burden of hypertension. As conventional cuff-based methods face limitations in comfort and dynamic tracking, intelligent BP measurement systems have emerged as promising alternatives, driven by advances in sensing and computing technologies. This review aims to serve as a roadmap for researchers, clinicians, and device developers working toward the goal to bridge the gap between technical innovation and clinical application. In this review, we re-frame the landscape of BP monitoring through a new classification based on measurement principles in mechanical and non-mechanical methods. We provide a comprehensive analysis of non-mechanical methods, highlighting innovations across the full pipeline from signal acquisition to calibration. Moreover, we emphasize the critical yet under-explored role of clinical validation. By reviewing current standards and evaluating existing devices, we identify key gaps and call for updated validation frameworks that align with the unique characteristics of next-generation cuffless systems.
Data availability
No datasets were generated or analyzed during the current study.
References
Kario, K., Okura, A., Hoshide, S. & Mogi, M. The WHO global report 2023 on hypertension warning the emerging hypertension burden in globe and its treatment strategy. Hypertension Res. 47, 1099–1102 (2024).
Kario, K., Okura, A., Hoshide, S. & Mogi, M. The WHO global report 2023 on hypertension warning the emerging hypertension burden in globe and its treatment strategy. Hypertension Research 1–4 (WHO, 2024).
Members, W. C. et al. 2025 AHA/ACC/AANP/AAPA/ABC/ACCP/ACPM/AGS/AMA/ASPC/NMA/PCNA/SGIM guideline for the prevention, detection, evaluation and management of high blood pressure in adults: A report of the american college of cardiology/american heart association joint committee on clinical practice guidelines. Circulation 152, e114–e218 (2025).
Hales, S. & Cournand, A. Statical essays: containing haemastaticks. (No Title) (Austrian National Library, 1733).
Geddes, L., Voelz, M., Combs, C., Reiner, D. & Babbs, C. F. Characterization of the oscillometric method for measuring indirect blood pressure. Ann. Biomed. Eng. 10, 271–280 (1982).
Maxwell, M., Schroth, P., Waks, A., Karam, M. & Dornfeld, L. Error in blood-pressure measurement due to incorrect cuff size in obese patients. Lancet 320, 33–36 (1982).
Stergiou, G. S. et al. Cuffless blood pressure measuring devices: review and statement by the european society of hypertension working group on blood pressure monitoring and cardiovascular variability. J. Hypertension 40, 1449–1460 (2022).
Zhou, S. et al. Clinical validation of a wearable ultrasound sensor of blood pressure. Nat. Biomed. Eng. 9, 865–881 (2025).
Pan, H.-Y. et al. The role of wearable home blood pressure monitoring in detecting out-of-office control status. Hypertension Res. 47, 1033–1041 (2024).
Zhu, G. et al. RingBP: Towards continuous, comfortable, and generalized blood pressure monitoring using a smart ring. Proc. ACM Interact. Mob. Wearable Ubiquitous Technol. 9, 1–24 (2025).
Zhao, Z. et al. HearBP: Hear your blood pressure via in-ear acoustic sensing based on heart sounds. in IEEE INFOCOM 2024-IEEE conference on computer communications 991–1000 (IEEE, 2024).
Kim, T.-H. et al. Neckband-type earphone for continuous monitoring of cardiovascular symptoms via self-powered box knot pulse-wave sensor. npj Biomed. Innov. 2, 4 (2025).
Luo, H. et al. Smartphone-based blood pressure measurement using transdermal optical imaging technology. Circul. Cardiovasc. Imaging 12, e008857 (2019).
Liang, Y. et al. airBP: monitor your blood pressure with millimeter-wave in the air. ACM Trans. Internet Things 4, 1–32 (2023).
Shi, Z., Gu, T., Zhang, Y. & Zhang, X. mmBP: Contact-free millimetre-wave radar based approach to blood pressure measurement. in Proceedings of the 20th ACM Conference on Embedded Networked Sensor Systems 667–681 (ACM, 2022).
Min, S. et al. Wearable blood pressure sensors for cardiovascular monitoring and machine learning algorithms for blood pressure estimation. Nat. Rev. Cardiol. 22, 629–648 (2025).
Panula, T., Sirkiä, J.-P., Wong, D. & Kaisti, M. Advances in non-invasive blood pressure measurement techniques. IEEE Rev. Biomed. Eng. 16, 424–438 (2022).
Mukkamala, R., Stergiou, G. S. & Avolio, A. P. Cuffless blood pressure measurement. Annu. Rev. Biomed. Eng. 24, 203–230 (2022).
Chen, S. et al. Flexible wearable sensors for cardiovascular health monitoring. Adv. Healthc. Mater. 10, 2100116 (2021).
Lin, J. et al. Wearable sensors and devices for real-time cardiovascular disease monitoring. Cell Rep. Phys. Sci. 2, 100541 (2021).
Yi, Z., Zhang, W. & Yang, B. Piezoelectric approaches for wearable continuous blood pressure monitoring: a review. J. Micromech. Microeng. 32, 103003 (2022).
CODAN Companies. The intelligent system for blood pressure monitoring. URL: https://www.codancompanies.com/products/invasive-blood-pressure-monitoring-systems/ (Accessed: 2025-11-20).
A&D Company Ltd. UM-102A Mercury Free Sphygmomanometers. URL: https://medical.andprecision.com/product/um-102a-mercury-free-sphygmomanometers/ (Accessed: 2025-11-20).
OMRON Healthcare, Inc. Gold Wireless Wrist Blood Pressure Monitor. URL: https://omronhealthcare.com/products/gold-wireless-wrist-blood-pressure-monitor-bp4350/ (Accessed: 2025-11-20).
Cardiex. SphygmoCor XCEL. URL: https://cardiex.com/products/sphygmocor-xcel/ (Accessed: 2025-11-20).
Ilies, C. et al. Comparison of a continuous noninvasive arterial pressure device with invasive measurements in cardiovascular postsurgical intensive care patients: a prospective observational study. Eur. J. Anaesthesiol. EJA 32, 20–28 (2015).
CNSystems Medizintechnik GmbH. CNAP TECHNOLOGY: Continuous Non-invasive Arterial Pressure. URL: https://www.cnsystems.com/technology/cnap-technology/ (Accessed: 2025-11-20).
Chandrasekhar, A., Natarajan, K., Yavarimanesh, M. & Mukkamala, R. An iPhone application for blood pressure monitoring via the oscillometric finger pressing method. Sci. Rep. 8, 13136 (2018).
Arndt, J. O., Klauske, J. & Mersch, F. The diameter of the intact carotid artery in man and its change with pulse pressure. Pflüger.’s. Arch. f.ür. die Gesamt. Physiologie des. Menschen und der Tiere 301, 230–240 (1968).
Lin, M. et al. A fully integrated wearable ultrasound system to monitor deep tissues in moving subjects. Nat. Biotechnol. 42, 448–457 (2024).
Blacher, J., Asmar, R., Djane, S., London, G. M. & Safar, M. E. Aortic pulse wave velocity as a marker of cardiovascular risk in hypertensive patients. Hypertension 33, 1111–1117 (1999).
Ohashi, N. et al. Nighttime systolic blood pressure determined by pulse transit time predicts cardiac events in patients with heart failure. J. Am. Heart Assoc. 14, e039917 (2025).
Koivistoinen, T. et al. Pulse wave velocity predicts the progression of blood pressure and development of hypertension in young adults. Hypertension 71, 451–456 (2018).
Ma, Y. et al. Relation between blood pressure and pulse wave velocity for human arteries. Proc. Natl. Acad. Sci. 115, 11144–11149 (2018).
Bramwell, J. C. & Hill, A. V. The velocity of pulse wave in man. Proc. R. Soc. Lond. Ser. B Containing Pap. a Biol. Character 93, 298–306 (1922).
Wu, Y. et al. Ubicon-BP: towards ubiquitous, contactless blood pressure detection using smartphone. IEEE Trans. Mobile Comput. 24, 7680–7692 (2025).
Newlin, D. & Levenson, R. Pre-ejection period: measuring beta-adrenergic influences upon the heart. Psychophysiology 16, 546–553 (1979).
Liao, C.-F., Cheng, H.-M., Sung, S.-H., Yu, W.-C. & Chen, C.-H. Determinants of pressure wave reflection: characterization by the transit time-independent reflected wave amplitude. J. Hum. Hypertension 25, 665–671 (2011).
Cao, Y., Chen, H., Li, F. & Wang, Y. Crisp-BP: continuous wrist PPG-based blood pressure measurement. in Proc. 27th annual International Conference on Mobile Computing and Networking 378–391 (ACM, 2021).
Turki, A. F. Assessing the efficacy of various machine learning algorithms in predicting blood pressure using pulse transit time. Diagnostics 15, 261 (2025).
Kanoga, S. et al. Comparison of seven shallow and deep regressors in continuous blood pressure and heart rate estimation using single-channel photoplethysmograms under three evaluation cases. Biomed. Signal Process. Control 85, 105029 (2023).
Pan, J. et al. Robust modelling of arterial blood pressure reconstruction from photoplethysmography. Sci. Rep. 14, 30333 (2024).
Ferizoli, R., Karimpour, P., May, J. M. & Kyriacou, P. A. Arterial stiffness assessment using PPG feature extraction and significance testing in an in vitro cardiovascular system. Sci. Rep. 14, 2024 (2024).
Yu, Z., Li, X. & Zhao, G. Facial-video-based physiological signal measurement: Recent advances and affective applications. IEEE Signal Process. Mag. 38, 50–58 (2021).
Mehrgardt, P., Khushi, M., Poon, S. & Withana, A. Pulse transit time PPG dataset (version 1.1.0). https://doi.org/10.13026/jpan-6n92 (2022).
Carlson, C. et al. Bed-based ballistocardiography dataset. IEEE Dataport https://doi.org/10.21227/77hc-py84 (2020).
Parlato, S. et al. A forcecardiography dataset with simultaneous SCG, heart sounds, ECG, and respiratory signals. Sci. Data 12, 1370 (2025).
Yang, B. et al. BrainZ-BP: a noninvasive cuff-less blood pressure estimation approach leveraging brain bio-impedance and electrocardiogram. IEEE Trans. Instrum. Meas. 73, 1–13 (2024).
Poh, M.-Z., McDuff, D. J. & Picard, R. W. Advancements in noncontact, multiparameter physiological measurements using a webcam. IEEE Trans. Biomed. Eng. 58, 7–11 (2011).
Wang, C. et al. Monitoring of the central blood pressure waveform via a conformal ultrasonic device. Nat. Biomed. Eng. 2, 687–695 (2018).
Hu, H. et al. A wearable cardiac ultrasound imager. Nature 613, 667–675 (2023).
Elgendi, M. et al. The use of photoplethysmography for assessing hypertension. npj Digit. Med. 2, 60 (2019).
Scardulla, F. et al. Photoplethysmograhic sensors, potential and limitations: is it time for regulation? A comprehensive review. Measurement 218, 113150 (2023).
Park, J.-W. et al. Wearable hyperspectral photoplethysmography allows continuous monitoring of exercise-induced hypertension. Adv. Sci. 12, 2417625 (2025).
Huang, C.-Y., Sung, Y.-L., Ni, S.-H. & Wang, T.-W. A skin tone insensitive, fully integrated smartwatch for continuous blood pressure measurement. IEEE Trans. Instrum. Meas. 74, 4000318 (2024).
Reisner, A. et al. Utility of the photoplethysmogram in circulatory monitoring. J. Am. Soc. Anesthesiol. 108, 950–958 (2008).
Philip, K. E., Tidswell, R. & McFadyen, C. Racial bias in pulse oximetry: more statistical detail may help tackle the problem. bmj 372, 298 (2021).
Teng, X.-F. & Zhang, Y.-T. Theoretical study on the effect of sensor contact force on pulse transit time. IEEE Trans. Biomed. Eng. 54, 1490–1498 (2007).
Chandrasekhar, A., Yavarimanesh, M., Natarajan, K., Hahn, J.-O. & Mukkamala, R. PPG sensor contact pressure should be taken into account for cuff-less blood pressure measurement. IEEE Trans. Biomed. Eng. 67, 3134–3140 (2020).
Wang, E. J. et al. Seismo: Blood pressure monitoring using built-in smartphone accelerometer and camera. in Proc. CHI conference on human factors in computing systems 1–9 (CHI, 2018).
Wang, L. et al. Accurate blood pressure measurement using smartphone’s built-in accelerometer. Proc. ACM Interact. Mob. Wearable Ubiquitous Technol. 8, 1–28 (2024).
Carlson, C. et al. Bed-based ballistocardiography: dataset and ability to track cardiovascular parameters. Sensors 21, 156 (2020).
Shin, S. et al. Posture-dependent variability in wrist ballistocardiogram-photoplethysmogram pulse transit time: implication to cuff-less blood pressure tracking. IEEE Trans. Biomed. Eng. 69, 347–355 (2021).
Xing, X., Li, H., Chen, Q., Jiang, C. & Dong, W. Blood pressure monitoring with piezoelectric bed sensor systems. Biomed. Signal Process. Control 87, 105479 (2024).
Huang, Y. et al. AI-driven system for non-contact continuous nocturnal blood pressure monitoring using fiber optic ballistocardiography. Commun. Eng. 3, 183 (2024).
Zhan, J. et al. Non-contact assessment of cardiac physiology using FO-MVSS-based ballistocardiography: a promising approach for heart failure evaluation. Sci. Rep. 14, 3269 (2024).
Liu, F. et al. Unobtrusive mattress-based identification of hypertension by integrating classification and association rule mining. Sensors 19, 1489 (2019).
Nikbakht, M., Chan, M., Lin, D. J., Gazi, A. H. & Inan, O. T. A residual u-net neural network for seismocardiogram denoising and analysis during physical activity. IEEE J. Biomed. Health Inform. 28, 3942–3952 (2024).
Inan, O. T. et al. Adaptive cancellation of floor vibrations in standing ballistocardiogram measurements using a seismic sensor as a noise reference. IEEE Trans. Biomed. Eng. 57, 722–727 (2009).
Ahmad, R.uS. et al. Advancements in wearable heart sounds devices for the monitoring of cardiovascular diseases. SmartMat 6, e1311 (2025).
Li, Y. et al. Exploring multi-source information fusion for cuff-less blood pressure measurement based on wearable watch-type device. Biomed. Signal Process. Control 112, 108695 (2026).
Jiang, Y., Chen, Y. & Tang, H. Real-time intraoperative blood pressure monitoring via heart sound. Biomed. Signal Process. Control 112, 108922 (2026).
Li, J. et al. Thin, soft, wearable system for continuous wireless monitoring of artery blood pressure. Nat. Commun. 14, 5009 (2023).
Tian, G. et al. Hierarchical piezoelectric composites for noninvasive continuous cardiovascular monitoring. Adv. Mater. 36, 2313612 (2024).
Zhang, T. et al. Nanopores-templated CNT/PDMS microcolumn substrate for the fabrication of wearable triboelectric nanogenerator sensors to monitor human pulse and blood pressure. Adv. Mater. Technol. 10, 2400749 (2025).
Xia, Y. et al. The piezoresistive pressure sensors with elastic graphene-coated silicone spheres. J. Mater. Sci. Mater. Electron. 35, 1235 (2024).
Park, Y. et al. Soft, full wheatstone bridge 3D pressure sensors for cardiovascular monitoring. npj Flex. Electron. 8, 6 (2024).
Liu, C. et al. Highly sensitive flexible capacitive pressure sensor based on a multicross-linked dual-network ionic hydrogel for blood pressure monitoring applications. ACS Appl. Mater. Interfaces 16, 34042–34056 (2024).
Namkoong, M. et al. Add-on soft electronic interfaces for continuous cuffless blood pressure monitoring. Adv. Mater. Technol. 8, 2300158 (2023).
Ni, R., Li, X., Zhao, H. & Ji, Z. A wireless portable system with few electrodes using thoracic electrical signals for long-term and real-time hemodynamic monitoring. IEEE Trans. Instrum. Meas. 74, 4007513 (2025).
Hesar, M. E. et al. AI-enabled epidermal electronic system to automatically monitor a prognostic parameter for hypertension with a smartphone. Biosens. Bioelectron. 241, 115693 (2023).
Yang, S., Zhang, Y., Cho, S.-Y., Correia, R. & Morgan, S. P. Non-invasive cuff-less blood pressure estimation using a hybrid deep learning model. Opt. Quantum Electron. 53, 1–20 (2021).
Zhang, B. et al. Pi-vimo: physiology-inspired robust vital sign monitoring using mmwave radars. ACM Trans. Internet Things 4, 1–27 (2023).
Wang, Z. et al. RF-AE: Single-site arterial elasticity estimation using UWB signals. Proc. ACM Interact., Mob., Wearable Ubiquitous Technol. 9, 1–26 (2025).
Wang, Z., Jin, B., Zhang, F., Li, S. & Ma, J. Uwb-enabled sensing for fast and effortless blood pressure monitoring. Proc. ACM Interact. Mob. Wearable Ubiquitous Technol. 8, 1–26 (2024).
Geng, F. et al. Non-contact stable arterial pulse measurement using mmWave array radar. Bioengineering 11, 1203 (2024).
Cao, Y., Zhang, S., Li, F., Chen, Z. & Luo, J. hBP-fi: Contactless blood pressure monitoring via deep-analyzed hemodynamics. in IEEE INFOCOM 2024-IEEE Conference on Computer Communications 1211–1220 (IEEE, 2024).
Slapničar, G., Wang, W. & Luštrek, M. Feasibility of remote blood pressure estimation via narrow-band multi-wavelength pulse transit time. ACM Trans. Sens. Netw. 20, 1–21 (2024).
Lian, C. et al. Robust and remote photoplethysmography based on smartphone imaging of the human palm. IEEE Trans. Instrum. Meas. 72, 1–11 (2023).
Wang, W. & Shan, C. Impact of makeup on remote-ppg monitoring. Biomed. Phys. Eng. Express 6, 035004 (2020).
Kamanditya, B., Fuadah, Y. N., Mahardika T, N. Q. & Lim, K. M. Continuous blood pressure prediction system using conv-LSTM network on hybrid latent features of photoplethysmogram (PPG) and electrocardiogram (ECG) signals. Sci. Rep. 14, 16450 (2024).
Li, H. et al. Wearable skin-like optoelectronic systems with suppression of motion artifacts for cuff-less continuous blood pressure monitor. Natl. Sci. Rev. 7, 849–862 (2020).
Gong, H. et al. Enabling orientation-free mmwave-based vital sign sensing with multi-domain signal analysis. in ICASSP 2024-2024 IEEE International Conference on Acoustics, Speech and Signal Processing (ICASSP) 8751–8755 (IEEE, 2024).
Gautam, Y. & Jebelli, H. Autoencoder-based photoplethysmography (PPG) signal reliability enhancement in construction health monitoring. Autom. Constr. 165, 105537 (2024).
Afandizadeh Zargari, A. H., Aqajari, S. A. H., Khodabandeh, H., Rahmani, A. & Kurdahi, F. An accurate non-accelerometer-based ppg motion artifact removal technique using cyclegan. ACM Trans. Comput. Healthc. 4, 1–14 (2023).
Mou, H. et al. Using data augmentation to improve the accuracy of blood pressure measurement based on photoplethysmography. Electronics 13, 1599 (2024).
Jeong, J.-H. et al. Cardiovascular hardware simulator and artificial aorta-generated central blood pressure waveform database according to various vascular ages for cardiovascular health monitoring applications. Computers Biol. Med. 172, 108224 (2024).
Liu, Q. et al. Photoplethysmography-based non-invasive blood pressure monitoring via ensemble model and imbalanced dataset processing. Phys. Eng. Sci. Med. 47, 1307–1321 (2024).
Shin, H. Signal completion using generative adversarial networks for enhanced photoplethysmography measurement accuracy. Computers Biol. Med. 180, 108952 (2024).
Kang, J. et al. Few-shot PPG signal generation via guided diffusion models. IEEE Sens. J. 24, 32792–32800 (2024).
Eleveld, N., Harmsen, M., Elting, J. W. J. & Maurits, N. M. Haemosync: a synchronisation algorithm for multimodal haemodynamic signals. Computer Methods Prog. Biomed. 254, 108298 (2024).
Gribbin, B., Steptoe, A. & Sleight, P. Pulse wave velocity as a measure of blood pressure change. Psychophysiology 13, 86–90 (1976).
Nichols, W. W., O’Rourke, M., Edelman, E. R. & Vlachopoulos, C. McDonald’s Blood Flow in Arteries: Theoretical, Experimental and Clinical Principles. (CRC Press, 2022).
Guo, C.-Y., Huang, C.-H., Chang, C.-C., Wang, K.-J. & Hsieh, T.-L. Combining local PWV and quantified arterial changes for calibration-free cuffless blood pressure estimation: a clinical validation. IEEE Sens. J. 23, 658–668 (2022).
Kachuee, M., Kiani, M. M., Mohammadzade, H. & Shabany, M. Cuff-less high-accuracy calibration-free blood pressure estimation using pulse transit time. in 2015 IEEE international symposium on circuits and systems (ISCAS) 1006–1009 (IEEE, 2015).
Li, X. et al. Continuous blood pressure monitoring and hypertension risk screening using smart watch. in 2024 46th annual international conference of the IEEE engineering in medicine and biology society (EMBC) 1–6 (IEEE, 2024).
Sevakula, R. K. et al. Machine learning based, subject-specific, gender and race independent, non-invasive estimation of the arterial blood pressure. npj Cardiovasc. Health 2, 41 (2025).
Sel, K. et al. Continuous cuffless blood pressure monitoring with a wearable ring bioimpedance device. npj Digit. Med. 6, 59 (2023).
Gupta, K., Bajaj, V. & Ansari, I. A. A support system for automatic classification of hypertension using BCG signals. Expert Syst. Appl. 214, 119058 (2023).
Di Nisio, A., De Palma, L., Ragolia, M. A., Lanzolla, A. M. L. & Attivissimo, F. Performance comparison of machine learning algorithms for the estimation of blood pressure using photoplethysmography. Biomed. Signal Process. Control 99, 106838 (2025).
Elgendi, M. et al. Photoplethysmography features correlated with blood pressure changes. Diagnostics 14, 2309 (2024).
Sun, H. et al. Wave reflection quantification analysis and personalized flow wave estimation based on the central aortic pressure waveform. Front. Physiol. 14, 1097879 (2023).
Joung, J. et al. Continuous cuffless blood pressure monitoring using photoplethysmography-based PPG2BP-net for high intrasubject blood pressure variations. Sci. Rep. 13, 8605 (2023).
Koparır, H. M. & Arslan, Ö Cuffless blood pressure estimation from photoplethysmography using deep convolutional neural network and transfer learning. Biomed. Signal Process. Control 93, 106194 (2024).
Jalan, I., et al. ThermalBP: A unified thermal imaging model to estimate blood pressure. in 2024 46th annual international conference of the IEEE engineering in medicine and biology society (EMBC) 1–4 (IEEE, 2024).
Kandhasamy, A. & Senthamaraikkannan, V. Intelligent system to predict renovascular hypertension using ECG sensor with RNNs. OPSEARCH 1–14 (Springer, 2025).
Mahardika T, N. Q., Fuadah, Y. N., Jeong, D. U. & Lim, K. M. PPG signals-based blood-pressure estimation using grid search in hyperparameter optimization of CNN. Lstm. Diagnostics 13, 2566 (2023).
Xiang, T. et al. Dynamic beat-to-beat measurements of blood pressure using multimodal physiological signals and a hybrid CNN-LSTM model. IEEE J. Biomed. health Inform. 29, 5438–5451 (2025).
Rizal, S. & Rahma, Y. A. GRU-based fusion models for enhanced blood pressure estimation from PPG signals. IEEE Access 12, 80317–80326 (2024).
Ma, C. et al. PPG-based continuous BP waveform estimation using polarized attention-guided conditional adversarial learning model. IEEE J. Biomed. Health Inform. 29, 3918–3929 (2023).
Liu, Z.-D. et al. HGCTNet: handcrafted feature-guided CNN and transformer network for wearable cuffless blood pressure measurement. IEEE J. Biomed. Health Inform. 28, 3882–3894 (2024).
Huang, P. et al. Continuous blood pressure monitoring based on improved multi-scale u-net assisted with piezoelectric polymer nanocomposite sensors. Biomed. Signal Process. Control 105, 107598 (2025).
Zheng, Z. et al. BP3: Improving cuff-less blood pressure monitoring performance by fusing mmWave pulse wave sensing and physiological factors. in Proceedings of the 22nd ACM conference on embedded networked sensor systems 730–743 (ACM, 2024).
Qiu, Y. et al. Non-contact blood pressure estimation from radar signals by a stacked deformable convolution network. IEEE J. Biomed. Health Inform. 28, 4553–4564 (2024).
Jamil, Z., Lui, L. T. & Chan, R. H. Blood pressure estimation using self-attention mechanism built-in ResUNet on PulseDB: demographic fairness, and generalization. IEEE Sens. J. 25, 1694–1705 (2024).
Wang, R., Qi, M., Shao, Y., Zhou, A. & Ma, H. PITN: physics-informed temporal networks for cuffless blood pressure estimation. IEEE Transactions on Mobile Computing (IEEE, 2025).
Sel, K., Mohammadi, A., Pettigrew, R. I. & Jafari, R. Physics-informed neural networks for modeling physiological time series for cuffless blood pressure estimation. npj Digital Med. 6, 110 (2023).
Chen, J. et al. rU-net, multi-scale feature fusion and transfer learning: unlocking the potential of cuffless blood pressure monitoring with PPG and ECG. IEEE J. Biomed. Health Inform. 29, 166–176 (2025).
Long, W. & Wang, X. BPNet: A multi-modal fusion neural network for blood pressure estimation using ECG and PPG. Biomed. Signal Process. Control 86, 105287 (2023).
Liu, S.-H., Sun, Y., Wu, B.-Y., Chen, W. & Zhu, X. Using machine learning models for cuffless blood pressure estimation with ballistocardiogram and impedance plethysmogram. Front. Digit. Health 7, 1511667 (2025).
SAMSUNG. SAMSUNG Galaxy Watch8. URL: https://www.samsung.com/us/watches/galaxy-watch8/ (Accessed: 2025-11-20).
SOMNOmedics. Somnotouch NIBP. URL: https://somnomedics.de/enus/somnomedics-diagnostic-devices/research/somnotouch-nibp-2/ (Accessed: 2025-11-20).
Derendinger, F. C. et al. Ability of a 24-h ambulatory cuffless blood pressure monitoring device to track blood pressure changes in clinical practice. J. Hypertension 42, 662–671 (2024).
Huang, Y. et al. Camera-based blood pressure monitoring based on multi-site and multi-wavelength pulse transit time features. IEEE Trans. Instrum. Meas. 73, 1–14 (2024).
Colburn, D. A. et al. A method for blood pressure hydrostatic pressure correction using wearable inertial sensors and deep learning. npj Biosens. 2, 5 (2025).
Nguyen, D. H. et al. Predicting blood pressures for pregnant women by PPG and personalized deep learning. IEEE J. Biomed. Health Inform. 29, 5–16 (2024).
Fan, F., Gu, Y., Shen, J., Dong, F. & Chen, Y. FewShotBP: towards personalized ubiquitous continuous blood pressure measurement. Proc. ACM Interact. Mob. Wearable Ubiquitous Technol. 7, 1–39 (2023).
Hu, Q. et al. Contactless arterial blood pressure waveform monitoring with mmWave radar. Proc. ACM Interact. Mob. Wearable Ubiquitous Technol. 8, 1–29 (2024).
Sharman, J. E. et al. Automated ‘oscillometric’blood pressure measuring devices: how they work and what they measure. J. Hum. hypertension 37, 93–100 (2023).
Mehta, S., Kwatra, N., Jain, M. & McDuff, D. Examining the challenges of blood pressure estimation via photoplethysmogram. Sci. Rep. 14, 18318 (2024).
Mukkamala, R. et al. The microsoft research aurora project: important findings on cuffless blood pressure measurement. Hypertension 80, 534–540 (2023).
Mukkamala, R., Shroff, S. G., Kyriakoulis, K. G., Avolio, A. P. & Stergiou, G. S. Cuffless blood pressure measurement: where do we actually stand? Hypertension 82, 957–970 (2025).
Han, M. et al. Feasibility and measurement stability of smartwatch-based cuffless blood pressure monitoring: a real-world prospective observational study. Hypertension Res. 46, 922–931 (2023).
Liu, Z.-D. et al. Cuffless blood pressure measurement using smartwatches: a large-scale validation study. IEEE J. Biomed. Health Inform. 27, 4216–4227 (2023).
Almeida, T. P. et al. Aktiia cuffless blood pressure monitor yields equivalent daytime blood pressure measurements compared to a 24-h ambulatory blood pressure monitor: preliminary results from a prospective single-center study. Hypertension Res. 46, 1456–1461 (2023).
Min, S. et al. Clinical validation of a wearable piezoelectric blood-pressure sensor for continuous health monitoring. Adv. Mater. 35, 2301627 (2023).
Tan, I. et al. Evaluation of the ability of a commercially available cuffless wearable device to track blood pressure changes. J. Hypertension 41, 1003–1010 (2023).
Zhou, S. et al. Clinical validation of a wearable ultrasound sensor of blood pressure. Nat. Biomed. Eng. 9, 865–881 (2024).
Hofmann, G. et al. A novel smartphone app for blood pressure measurement: a proof-of-concept study against an arterial catheter. J. Clin. Monit. Comput. 37, 249–259 (2023).
Kargarandehkordi, A. et al. Personalized AI-driven real-time models to predict stress-induced blood pressure spikes using wearable devices: Proposal for a prospective cohort study. JMIR Res. Protoc. 13, e55615 (2024).
Sayer, G. et al. Continuous monitoring of blood pressure using a wrist-worn cuffless device. Am. J. Hypertension 35, 407–413 (2022).
Kapoor, M., Holman, B. & Cohen, C. Contactless and calibration-free blood pressure and pulse rate monitor for screening and monitoring of hypertension: Cross-sectional validation study. JMIR cardio 8, e57241 (2024).
Vliet et al. Evaluation of a novel cuffless photoplethysmography-based wristband for measuring blood pressure according to the regulatory standards. Eur. Heart J.-Digit. Health 5, 335–343 (2024).
Stergiou, G. S. et al. A universal standard for the validation of blood pressure measuring devices: association for the advancement of medical instrumentation/european society of hypertension/international organization for standardization (AAMI/ESH/ISO) collaboration statement. Hypertension 71, 368–374 (2018).
Association, I. S. et al. IEEE standard for wearable, cuffless blood pressure measuring devices–amendment 1 (amendment to IEEE std 1708–2014). IEEE Std. 1708a-2019 19, 1–35 (2019).
Stergiou, G. S. et al. European society of hypertension recommendations for the validation of cuffless blood pressure measuring devices: European society of hypertension working group on blood pressure monitoring and cardiovascular variability. J. Hypertension 41, 2074–2087 (2023).
Tian, D. et al. ECG-doctor: An interpretable multimodal ECG diagnosis framework based on large language models. in Proceedings of the 34th ACM International Conference on Information and Knowledge Management 2863–2873 (ACM, 2025).
Liu, L., Cui, G., Wan, C., Wu, D. & Li, Y. ECG-LLM: Leveraging large language models for low-quality ECG signal restoration. in 2024 IEEE international conference on bioinformatics and biomedicine (BIBM) 3537–3542 https://doi.org/10.1109/BIBM62325.2024.10822461 (2024).
Jiang, X. et al. TC–RAG: Turing–complete RAG’s case study on medical LLM systems. in Proceedings of the 63rd annual meeting of the association for computational linguistics (volume 1: Long papers) 11400–11426 (ACL, 2025).
Rong, Y., Theofanopoulos, P. C., Trichopoulos, G. C. & Bliss, D. W. A new principle of pulse detection based on terahertz wave plethysmography. Sci. Rep. 12, 6347 (2022).
Zhang, Y. et al. Ultrafast longitudinal imaging of haemodynamics via single-shot volumetric photoacoustic tomography with a single-element detector. Nat. Biomed. Eng. 8, 712–725 (2024).
Liu, J. et al. Asian expert consensus on nocturnal hypertension management. Hypertension 82, 945–956 (2025).
Johnson, A. E. et al. MIMIC-IV, a freely accessible electronic health record dataset. Sci. Data 10, 1 (2023).
Johnson, A. E. et al. MIMIC-III, a freely accessible critical care database. Sci. data 3, 1–9 (2016).
Saeed, M., Lieu, C., Raber, G. & Mark, R. G. MIMIC II: a massive temporal ICU patient database to support research in intelligent patient monitoring. in Computers in cardiology 641–644 (IEEE, 2002).
Lee, H.-C. et al. VitalDB, a high-fidelity multi-parameter vital signs database in surgical patients. Sci. Data 9, 279 (2022).
Mieloszyk, R. et al. A comparison of wearable tonometry, photoplethysmography, and electrocardiography for cuffless measurement of blood pressure in an ambulatory setting. IEEE J. Biomed. Health Inform. 26, 2864–2875 (2022).
Schumann, A. & Bär, K.-J. Autonomic aging–a dataset to quantify changes of cardiovascular autonomic function during healthy aging. Sci. Data 9, 95 (2022).
Goldberger, A. L. et al. PhysioBank, physiotoolkit, and physionet: components of a new research resource for complex physiologic signals. Circulation 101, e215–e220 (2000).
Liang, Y., Chen, Z., Liu, G. & Elgendi, M. A new, short-recorded photoplethysmogram dataset for blood pressure monitoring in China. Sci. Data 5, 1–7 (2018).
Gomes, E., Liao, C., Shay, O. & Bikhchandani, N. A dataset of synchronized signals from wearable cardiovascular monitoring sensors. IEEE Dataport https://doi.org/10.21227/3yte-wz05 (2021).
Speth, J. et al. Mspm: A multisite physiological monitoring dataset for remote pulse, respiration, and blood pressure estimation. IEEE Trans. Instrum. Meas. 73, 1–14 (2024).
Baek, S., Jang, J. & Yoon, S. End-to-end blood pressure prediction via fully convolutional networks. IEEE Access 7, 185458–185468 (2019).
Miao, F. et al. Continuous blood pressure measurement from one-channel electrocardiogram signal using deep-learning techniques. Artif. Intell. Med. 108, 101919 (2020).
Kireev, D. et al. Continuous cuffless monitoring of arterial blood pressure via graphene bioimpedance tattoos. Nat. Nanotechnol. 17, 864–870 (2022).
Ibrahim, B. & Jafari, R. Cuffless blood pressure monitoring from a wristband with calibration-free algorithms for sensing location based on bio-impedance sensor array and autoencoder. Sci. Rep. 12, 319 (2022).
Hoog Antink, C. et al. Accuracy of heart rate variability estimated with reflective wrist-PPG in elderly vascular patients. Sci. Rep. 11, 8123 (2021).
Roth, S. et al. Noninvasive pulse contour analysis for determination of cardiac output in patients with chronic heart failure. Clin. Res. Cardiol. 107, 395–404 (2018).
Chandrasekhar, A. et al. Smartphone-based blood pressure monitoring via the oscillometric finger-pressing method. Sci. Transl. Med. 10, eaap8674 (2018).
Li, S. et al. Monitoring blood pressure and cardiac function without positioning via a deep learning–assisted strain sensor array. Sci. Adv. 9, eadh0615 (2023).
Acknowledgements
The study was supported by the National Major Scientific Instruments and Equipments Development Project of National Natural Science Foundation of China (62427810), Beijing Natural Science Foundation(L223002), the 111 Project (B18008), the Youth Top Talent Support Program. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
A.Z., C.S., J.L., Y.Z., H.M., and Z.Z. conceived the study, made substantial contribution to discussion of content, screened and edited manuscript before submission. Z.Z. and H.H. wrote the paper. Z.Z., H.H., Y.H., and R.L. researched data for article. All authors approved the final paper.
Corresponding authors
Ethics declarations
Competing interests
A.Z. and H.M. are inventors on granted patents (CN114642409B, CN114818910B); A.Z., R.L., H.H., Z.Z., and Y.H. are inventors on a pending patent application (CN119405292A). These applications were submitted by Beijing University of Posts and Telecommunications regarding contactless BP monitoring and signal processing algorithms. Additionally, C.S., A.Z., H.M., Z.Z., and R.L. are inventors on a granted patent (CN118078230B) submitted by Peking University Third Hospital and Beijing University of Posts and Telecommunications regarding cardiovascular disease risk prediction methods. J.L. is an Editorial Board Member of npj Cardiovascular Health and was not involved in the editorial review or the decision to publish this article. The remaining authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zheng, Z., Hao, H., Huang, Y. et al. Advances in principles and technologies of non-mechanical blood pressure monitoring. npj Cardiovasc Health 3, 7 (2026). https://doi.org/10.1038/s44325-025-00102-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44325-025-00102-5