Abstract
Human cerebral organoids hold promise for studying neurodevelopment, modelling disease, and drug screening. Electrophysiology is a key functional property for these studies; yet, performing high-throughput electrophysiological studies with organoids remains a critical bottleneck. Current state-of-the-art recording technologies, including 2D and 3D microelectrode arrays (MEAs), are low-throughput, expensive to fabricate and purchase, and often incompatible with routine organoid culture. These limitations restrict their adoption, and many studies report electrophysiological activity from insufficient sample sizes to accurately capture the widely accepted biological variability inherent to organoid models. Here, we present a scalable, low-cost, plug-and-play platform that integrates a new class of carbon nanotube-based 3D microelectrode arrays into standard cell culture plates. This system enables high-throughput extracellular recordings from many organoids without specialised workflows. Using this system, we record electrophysiological signals from 74 human cortical organoids, the largest scale reported in organoid electrophysiology studies to the best of our knowledge. The measurements involve capturing electrophysiological phenotypes across neurotypical and Angelman Syndrome organoids. We also show that the use of carbon nanotubes in place of conventional gold electrodes achieves superior electrical, electrochemical, and electromechanical properties at a fraction of the cost while enabling a new scalable manufacturing technique. This technology establishes a standardised and accessible route to large-scale electrophysiological measurements in organoids.
Similar content being viewed by others
Introduction
Human cerebral organoids represent a promising class of in vitro models for disease modelling, drug discovery, and developmental biology1,2,3,4. By recapitulating human-specific cellular compositions and electrophysiological function, they offer an opportunity to complement the limitations of pre-clinical animal models. A key challenge is their well-documented experimental variability, which necessitates large sample sizes for meaningful studies5,6. Many molecular profiling methods, including imaging and transcriptomic approaches, are now able to scale sufficiently to meet this challenge; however, electrophysiological measurement platforms, including patch clamp and microelectrode arrays (MEA), trail substantially.
Cerebral organoids present the additional challenge of measuring organoid electrophysiology in three dimensions (3D)7,8,9. Commercially available MEA platforms are typically associated with high capital and recurring costs due to proprietary hardware and consumables, and are predominantly optimised for planar, 2D sensing. Emerging 3D MEAs can overcome this constraint but remain low-throughput and difficult to integrate into standardised organoid culture workflows10,11,12,13,14,15,16,17,18. Their fabrication relies on highly specialised cleanroom-intensive processes10,13,15,16,17,18 and expensive materials, such as gold (Au)10,12,15,17,18 and platinum13, and they also often require modifications to organoid culture protocols17. Additionally, surface coatings are required to improve the electrical performance of these noble metal microelectrodes11,12,15,18. These limitations reduce the number of organoids that can be practically assayed per experiment, contributing to the small sample sizes commonly reported in published studies. Cost, access, and complexity hinder the adoption and scientific impact of organoid electrophysiological measurements.
Here, we introduce a scalable, low-cost, yet high-performance 3D platform for organoid electrophysiology. The device, termed CAMEO (Conformal Array for Monitoring Electrophysiology of Organoids), transforms from a 2D cartwheel layout into a mechanically robust, basket-like structure that conforms around organoids. Several CAMEOs can be integrated within standard culture plates. We demonstrate a 6-well prototype in this work. CAMEO is fabricated using inexpensive, scalable manufacturing methods and leverages single-walled carbon nanotubes (SWCNTs) as the electrode material. They achieve high electrical performance without relying on costly materials such as Au, additional surface coatings, or cleanroom processing. This platform integrates seamlessly with commercial acquisition systems, requires minimal user intervention, and enables parallel fabrication of multiple devices at a fraction of the cost of conventional MEAs. We describe its electrochemical and mechanical properties and demonstrate its scalable application by performing recordings from a set of 74 human neurotypical and Angelman Syndrome cortical organoids.
Results
CAMEOs are designed for high-throughput applications
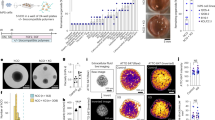
Each CAMEO is composed of twelve SWCNT microelectrodes, each with an active area of 200 µm × 500 µm. This electrode area is selected to capture population-level extracellular activity from ensembles of neurons in close proximity, providing a spatially averaged signal that is robust to the inherent heterogeneity of cerebral organoids. A two-dimensional (2D) architecture resembling a twelve-spoke cartwheel serves as a precursor to the 3D CAMEO. The SWCNT microelectrodes act as the spokes of this cartwheel. Figure 1a presents a schematic illustration of the key components of the cartwheel and the resulting CAMEO, while Fig. 1b shows optical images. The 2D structure consists of three stacked layers: a 20 µm thick bottom layer of polystyrene-block-polyisoprene-block-polystyrene (SIS), which functions as the substrate; a 10 µm thick middle layer of pristine SWCNTs serving as the electrode material; and a 20 µm thick top layer of SIS that defines the active electrode areas and contact pads. The inset of Fig. 1b provides a zoomed-in view of the twelve-electrode array, with a dotted red box marking the active area. A thin assembly ring secures the final 3D shape (Fig. 1a, b).
a Exploded schematic of the 2D cartwheel design, highlighting the constituent layers and transformation into a 3D CAMEO. b Photograph of a fabricated cartwheel with magnified view of the central region showing 12 independent SWCNT microelectrodes (outlined in red), alongside a fully assembled CAMEO. c Complete platform with CAMEOs suspended from a standard 6-well plate lid. d Enlarged top-view of a suspended CAMEO, showing contact pads routed through a transparent flexible printed circuit board. e Photograph illustrating organoid loading via pipetting through an opening on the plate lid. f Image showing all CAMEOs conformally wrapped around organoids in a 6 well platform. g Close-up view of a single CAMEO conformally wrapped around an organoid, with microelectrodes visible on the organoid surface. Scale bars, 1 cm unless otherwise indicated.
The dimensions of CAMEO are optimised for easy integration with standard culture plates (Fig. 1c). A thin, transparent printed circuit board bonded to the plate’s lid facilitates electrical interfacing with external readout systems (Fig. 1d). Laser-patterned openings in the lid allow for facile loading of organoids into CAMEOs using standard pipettes (Fig. 1e). The CAMEOs are fixed to the lid such that a single CAMEO is centrally located in each well of the plate, enabling simultaneous and independent monitoring of organoids across all wells and rapid media changes simply by moving the lid from one plate to another (Fig. 1f). A schematic of the cross section of a single well of the modified culture plate appears in Supplementary Fig. S1.
The material properties and dimensions of the individual spokes and the central hub allow CAMEOs to conformally wrap around organoids. Figure 1g shows this for a cortical organoid (diameter: 2 mm), which is further supported by the predictions from finite element analysis (FEA) simulations (Supplementary Fig. S2). Supplementary Fig. S2a compares a representative optical image with the simulated strain distribution of the deformed CAMEO configuration before organoid deposition. After deposition, both experimental observations and simulations show that the electrodes bend inward, forming a conformal net around the organoid. FEA quantifies this mechanical response by showing uniform inward bending of the spokes and a principal strain distribution below ~1% across most regions of the CAMEO and the deformable organoid, except at the contact locations (Supplementary Fig. S2b). These strain levels remain well below the experimentally measured yield strain of ~2% for SWCNT sheets (Supplementary Fig. S2c). The simulations reveal a distinct strain gradient between the SWCNT and SIS layers (1.6 vs. 0.5%). This strain difference arises from a thickness- and stiffness-mismatch between the two layers, indicating a bending-dominated deformation. Such mechanically guided deformation enables designing CAMEOs with individual electrodes precisely positioned for 3D spatial monitoring of organoids of given dimensions, as shown in Fig. 1g. Parametric simulations of an organoid with a radius of 1.25 mm across different CAMEO positions reveal the evolution of strain during deposition and entrapment (Supplementary Fig. S2d). Maximum strains of approximately 2.5% occur during the initial deposition step (i), localised to regions undergoing the largest curvature changes near the top of the device. As deposition progresses, these high-strain regions propagate sequentially from steps (ii–v) toward the bottom of the CAMEO. To evaluate the effect of organoid radii on CAMEO mechanics, Supplementary Fig. S2e shows the deformed configurations of devices interacting with an organoid of radii 0.75 mm, 1.25 mm, 1.75 mm where increasing organoid size progressively alters the spoke curvature to achieve conformal wrapping and the principal strain distribution remains below ~0.5% across the region surrounding the organoids except at the CAMEO-organoid contact interfaces, where localised strain hotspots emerge. These contact-induced strains reach maximum values of approximately 0.5%, 1.6%, and 1.8% for organoid radii of 0.75, 1.25, and 1.75 mm, respectively. Thickness-weighted average density calculations for the CAMEO, based on values in Supplementary Table ST1, indicate negligible net body forces per unit volume (~1.6 kN m⁻³), such that immersion does not alter the static bending response. Although the CAMEO conforms around the organoid to establish reliable electrical contact, this interaction is fully reversible and does not permanently deform, embed, or trap the tissue. Organoids can be easily removed by submerging the CAMEOs in culture media and using a wide cut pipette tip to capture and extract the organoid from the CAMEO. A schematic of the insertion and removal process is shown in Supplementary Fig. S3.
Unique materials and fabrication processes enable low-cost and scalable manufacturing of CAMEOs
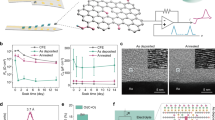
SWCNTs and SIS serve as electrode and encapsulation materials for CAMEOs, respectively. They offer unusual characteristics that combine high performance with low cost and ease of bulk processing, enabling a high-throughput manufacturing framework compared to conventional materials, such as noble metals (e.g., Au, platinum) and flexible plastics (e.g., polyimide). However, while pristine SWCNTs possess inherently high electrical conductivity, electrochemical properties, and mechanical flexibility, their conventional, low-cost mass production methods typically rely on casting or printing SWCNT solutions prepared by sonicating SWCNTs in a solvent containing surfactants or polymers as stabilisers19. This sonication process fractures the SWCNTs, which significantly diminishes their electrical and mechanical properties19. Furthermore, encapsulation of the SWCNTs by stabilisers negatively affects these properties, resulting in a final product with substantially reduced performance compared to pristine SWCNTs20. Therefore, we developed a method for bulk processing of SWCNTs that yields flexible, free-standing, centimetre-scale SWCNT films (Fig. 2a). This process involves sonication- and stabiliser-free dispersion of pristine SWCNT powder in chlorosulfonic acid (CSA) followed by the self-assembly of the SWCNTs at the air-solvent interface. CSA, a superacid, protonates the SWCNT sidewalls, which disrupts the strong, inter-SWCNT van der Waals forces allowing for a stable, liquid-crystalline dispersion of individual or small bundles of pristine SWCNTs without the need for destructive sonication or performance-degrading stabilisers21. Exposure of the dispersion to ambient air results in gradual degradation of CSA into sulphuric acid and vapours of hydrochloric acid. This process de-protonates the SWCNTs causing destabilization of the dispersion and slow re-aggregation of the SWCNTs via van der Waals forces to form self-assembled, free-standing SWCNT films that float on the surface of the solvent22. Because the films consist of pristine, unbroken SWCNTs, they retain their remarkable electrical and mechanical properties (~2% yield strain, 500 MPa Elastic Modulus, ~8.3 × 10-6 N·mm2 Bending Stiffness) at a fraction of the cost (approximately 15× lower) of Au.
a Schematic and photographs illustrating formation of single-walled carbon nanotube (SWCNT) films via self-assembly in chlorosulfonic acid (CSA). Partly created in BioRender, Mishra, N. (2026) https://BioRender.com/p4goa9x. b Patterning of SWCNT films on tattoo paper using a picosecond laser to define 2D cartwheel structures, followed by assembly within a 3D-printed mould. c Scalable fabrication of six CAMEOs through stamping and 2D-to-3D transformation, with subsequent integration into a 6-well plate. Insets show magnified views of individual devices on the assembly platform. Scale bars represent 1 cm. Partly created in BioRender. Mishra, N. (2026) https://BioRender.com/66vxdvz.
We use laser cutting rather than conventional, low-throughput, and expensive lithography methods to fabricate the cartwheel structures in a scalable, cost-effective manner. SWCNT and SIS films are laminated onto a sacrificial temporary tattoo paper to facilitate handling during fabrication. The SWCNT film/tattoo paper and SIS/tattoo paper are separately laser-cut (Fig. 2b) and then stacked and heat-bonded to simultaneously produce multiple cartwheels. Following fabrication, the fully assembled cartwheels (comprising the following layers: top SIS/SWCNT/bottom SIS/tattoo paper) are placed into the open trenches of a custom-designed 3D printed mould. The position, orientation, and number of these trenches are optimised to ensure scalable, symmetrical assembly of the CAMEOs across all wells of a given culture plate. For example, Fig. 2b illustrates the mould designed for integrating CAMEOs into a standard 6-well plate, and Supplementary Fig. S4 provides a close-up view of a cartwheel positioned within one of these trenches. A custom-printed assembly platform transforms the cartwheels into CAMEOs (Fig. 2c). The platform consists of an array of pillars, with an assembly ring symmetrically placed around each pillar. The position of these pillars precisely matches the centre of the trenches in the mould (Fig. 2b). The assembly rings are composed of a doughnut-shaped adhesive tape laminated with patterned cellulose paper, where the openings in the paper match the contact pads of the CAMEOs. Holes patterned into the adhesive tape by a laser function as via holes for the electrical connection of the contact pads of the CAMEOs to a breakout board (Supplementary Fig. S5).
A thin ring of double-sided tape provides temporary adhesion to hold the assembly ring in place. The tops of each pillar are coated with a thin layer of tacky silicone, which enables the easy lifting of the cartwheels from the mould’s trenches. Applying water to the tattoo paper dissolves the water-soluble adhesive layer, softening it and allowing the cartwheel spokes to bend easily without rigidity-induced mechanical fracture and to align with their corresponding slots on the assembly ring. The contact pads are then securely bonded to the assembly ring using the tape's adhesive, with the slot geometry ensuring deterministic placement of each pad. Simulation results shown in Fig. 2c reveal maximum strain localisation at the electrode base, the region with the highest curvature, reaching 1.8% during assembly. This simulated mechanical response captures the strain evolution during assembly, where sequential fabrication steps guide the CAMEO into a basket-like configuration capable of conformally wrapping an organoid. After this bonding, applying additional water removes the tattoo paper, yielding the completed CAMEOs. The assembly platform, with the fully assembled CAMEOs, is then pressed into a culture plate with laser-patterned openings in its lid. A thin layer of epoxy is pre-patterned around each opening on the lid. The epoxy provides a permanent bond that is substantially stronger than the temporary adhesion provided by silicone and double-sided tape during assembly, enabling straightforward transfer of the CAMEOs to the plate lid. Following epoxy curing, the assembly stand is lifted away, and the temporary adhesion is overcome without disrupting the bonded CAMEOs. A schematic illustrating the adhesion-guided transfer process and sequential use of temporary and permanent adhesives appears in Supplementary Fig. S6. Finally, a custom-designed transparent breakout board is bonded to the top side of the lid, completing the fabrication of the high-throughput monitoring system.
SWCNTs are electrochemically superior to conventionally used gold
We next compare the electrochemical performance of SWCNT-based electrodes to widely used Au on polyimide (PI) electrodes (Fig. 3a, b). Cyclic voltammetry (CV) experiments are conducted in a traditional three-electrode system comprising a SWCNT- or Au- based working electrode (electrode radius: 2 mm), platinum wire-based counter electrode, and silver/silver chloride (Ag/AgCl)-based reference electrode immersed in 1X phosphate-buffered saline (pH 7) containing 100 mM potassium ferricyanide as the redox mediator (Fig. 3a). SWCNT-based electrodes exhibit peak height (0.38 ± 0.01 mA) comparable to those obtained for Au-based electrodes (0.31 ± 0.02 mA). However, pre-treating the SWCNT-based electrodes by subjecting the electrodes to +1 V (versus Ag/AgCl electrode) for 10 min in 1X phosphate-buffered saline (pH 7) results in a near 60% increase in peak height (0.51 ± 0.03 mA) compared to Au-based electrodes. The enhanced electrochemical performance following oxidative activation can be attributed to the introduction of oxygen-containing functionalities that increase surface wettability, alongside the exposure of new edge-plane defects and the opening of previously inaccessible sites within the SWCNT network. Together, these effects facilitate improved ionic penetration and reduced charge-transfer resistance23,24. Further examination of the electrodes sheds additional information on the electrochemical features of the different electrode materials (Fig. 3a). The anodic-to-cathodic peak current ratio (ipa/ipc) remains close to unity for all electrode types (Au: 1.10 ± 0.01; SWCNT: 0.93 ± 0.02; activated SWCNT: 1.02 ± 0.01), indicating favourable redox reversibility under all conditions. However, the peak-to-peak separation (ΔE) reveals differences in electron-transfer kinetics between these electrodes. For example, Au-based electrodes exhibit ΔE of 0.27 ± 0.03 V while pristine SWCNTs and activated SWCNT-based electrodes offer lower ΔE of 0.12 ± 0.01 V and 0.17 ± 0.02 V, respectively. These results indicate sluggish electron transfer across the Au electrode-electrolyte interface compared to SWCNTs.
a Comparison of Au, SWCNT, and electrochemically activated SWCNT electrodes via cyclic voltammetry. Shaded error bands represent N = 3 electrodes. b Bode plots comparing Au, SWCNT, and electrochemically activated SWCNT electrodes via electrochemical impedance spectroscopy. Shaded error bands represent N = 3 electrodes. c Comparison of 2D cartwheels and 3D CAMEOs via cyclic voltammetry. Shaded error bands represent N = 3 replicates. d Bode plots comparing 2D cartwheels and 3D CAMEOs via electrochemical impedance spectroscopy. Shaded error bands represent N = 3 electrodes. e Benchtop validation of CAMEOs using a waveform generator to simulate neuronal action potential-like signals. Schematic created in BioRender. Mishra, N. (2026) https://BioRender.com/ycteceq. All data are presented as mean ± SD.
Bode plots derived from electrochemical impedance spectroscopy (EIS) for the three electrode types appear in Fig. 3b. The plots reveal that the total impedance (Ztot) of the three electrode types is similar (Au: 105.8 ± 9.3 Ω, SWCNT: 105.2 ± 2.9 Ω, Activated SWCNT: 88.5 ± 13.4 Ω) between 104-105 Hz. However, a distinct divergence in Ztot for the electrodes is observed for frequencies below 104 Hz. Ztot rapidly rises and reaches approximately 5.5 ± 1.4 kΩ at 0.1 Hz for Au-based electrodes. On the other hand, Ztot is frequency independent for pristine and activated SWCNT-based electrodes up to 10 Hz, below which Ztot rapidly rises and reaches 2.8 ± 0.3 kΩ and 2.3 ± 1.1 kΩ at 0.1 Hz for pristine and activated electrodes, respectively. At 1 kHz, the frequency most relevant to neural recordings, SWCNT electrodes (111.6 ± 4.3 Ω) and activated SWCNT electrodes (89.8 ± 13.7 Ω) show significantly lower impedance than Au electrodes (308.5 ± 38.4 Ω). The lower impedance reflects improved conductivity and reduced charge-transfer resistance, properties that facilitate more reliable neural signal detection than Au. The phase angle (Φ) gives additional information about the complex impedimetric characteristics of the three electrode types. The Φ for Au-based electrodes exhibit a high dependence on frequency, starting approximately at 10° at 105 Hz and rapidly increasing and peaking at approximately 50° at 103 Hz before falling and then again rising to around 40° at 0.1 Hz. In contrast, Φ for both pristine and activated SWCNT-based electrodes follow similar trajectory across the studied frequency range with Φ starting at approximately at 10° at 105 Hz and then dropping to around 5° in the frequency range of 104 to 105 Hz. The Φ for the two SWCNT electrodes then increases to approximately 30° at 10 Hz before rapidly rising and stabilising at around 60° and 70° for pristine and activated SWCNTs, respectively.
The Bode plots for the electrodes collectively reveal that the electrode-electrolyte interface in the case of SWCNT-based electrodes behaves as a near-ideal resistor in the frequency range of 10 to 105 Hz, while this interface shows a capacitive nature at frequencies below 10 Hz. In contrast, the Au-based electrode exhibits a more complex behaviour with simultaneous display of resistive and capacitive features throughout the entire frequency range. Together, these results demonstrate that SWCNT electrodes exhibit more efficient charge transfer and reduced phase distortion than Au electrodes at recording-relevant frequencies, enabling high-fidelity neural signal acquisition with preserved timing and waveform integrity25,26. At lower frequencies, their pronounced capacitive phase response further enhances safety during electrical stimulation by minimising irreversible faradaic reactions27,28,29.
EIS analysis using the Randles circuit model underscores the superior electrochemical characteristics of SWCNT-based electrodes for neural recording applications. Au electrodes exhibit the highest charge transfer resistance (1516.7 ± 363.5 Ω), which can contribute to signal attenuation and reduced sensitivity to weak neural spikes. In contrast, SWCNT electrodes (234.4 ± 51.5 Ω) and activated SWCNT electrodes (81.8 ± 28.2 Ω) offer substantially lower resistance values, reflecting faster charge transfer kinetics at the electrode–electrolyte interface. Such reductions in interfacial resistance directly translate to lower baseline noise and improved fidelity in capturing high-frequency neural activity. Furthermore, the SWCNT electrodes demonstrate markedly higher double-layer capacitance (37.8 ± 5.4 µF for SWCNT; 59.3 ± 9.9 µF for activated SWCNT) compared to Au (5.47 ± 0.82 µF), arising from their nanostructured morphology and increased electrochemically active surface area. This elevated capacitance enhances charge storage capacity and supports stable electrode performance during continuous neural recordings. Together, these attributes establish SWCNT-based electrodes as a more suitable platform than conventional Au electrodes for reliable, high-sensitivity organoid electrophysiology.
CAMEOs maintain electrochemical function under bending
CAMEOs experience bending stresses during two key stages: first, during fabrication, when 2D cartwheels are shaped into 3D CAMEOs; and second, during operation, as the CAMEOs conformally interface with curvilinear organoids. Supplementary Fig. S7 studies the performance of SWCNT/SIS as a structural material for developing CAMEOs by investigating the effect of repeated (1000 cycles) flexural stressors on its electrical resistance. For comparison, control experiments with Au/PI are also reported. Both materials begin with comparable baseline resistances (~50 Ω). The overall drift in resistance across the full set of cycles remains below 1% for both electrode types, confirming that neither undergoes catastrophic degradation under repeated strain. However, analysis of intra-cycle resistance modulation reveals a striking difference. Au electrodes exhibit a pronounced average peak-to-peak resistance change (ΔR) of 0.23 ± 0.02 Ω, reflecting measurable fluctuations in conductivity with each bend–release motion. By contrast, SWCNT electrodes maintain essentially invariant resistance, with an average ΔR of only 1.1 ± 0.4 µΩ, nearly three orders of magnitude smaller. This pronounced stability underscores the mechanical compliance and robust conductive pathways of SWCNT networks, which remain unaffected by localised strain that otherwise perturbs Au films. For neural recording applications, such intra-cycle stability is critical, as resistance fluctuations directly contribute to baseline noise and signal distortion. The negligible ΔR of SWCNT electrodes ensures consistent impedance characteristics throughout mechanical deformation, thereby preserving signal fidelity. These findings provide quantitative evidence that SWCNT electrodes are mechanically superior to conventional Au electrodes, validating their suitability for long-term operation as mechanically dynamic bioelectronic interfaces.
CV and EIS studies on SWCNT/SIS-based cartwheel and CAMEO further investigate the effects of bending on the electrochemical performance of SWCNT electrodes. Figure 3c shows comparable peak heights (148 ± 23 µA and 141.6 ± 7.1 µA) and ipa/ipc (1.03 and 0.99) for cartwheel and CAMEO electrodes, respectively. Figure 3d reveals that the Ztot are nearly overlapping with subtle differences in the phase angle for the two systems, with the impedance at 1 kHz being 185.6 ± 15.4 Ω for the cartwheel and 159.8 ± 14.9 Ω for the CAMEO. The phase-angle deviation between CAMEO and cartwheel is investigated to elucidate the effect of bending stresses on the electrochemical performance of SWCNT-based electrodes. We employ EIS to evaluate microcrack formation during the transformation of SWCNT electrodes from a planar cartwheel to the 3D CAMEO configuration, by analysing the shift in impedance phase across frequency (Supplementary Fig. S8). Plotting the differential phase angle (ΔPhase = Phasecartwheel – PhaseCAMEO) across frequency reveals subtle deviations at low frequencies (<100 Hz) consistent with localised microstructural perturbations, most likely minor crack formation within the SWCNT network during bending. These cracks alter the capacitive behaviour of the electrode, which is most prominent in the low-frequency regime, as also described in prior literature on stretchable electrode systems30,31. Importantly, ΔPhase remains nearly flat in the 103-105 Hz range, the bandwidth relevant for action potential detection in neural recordings, indicating that the integrity of high-frequency charge transport pathways is preserved. Complementary CV measurements show only a minor increase in peak-to-peak separation (ΔE value) for electrodes in CAMEO compared to those in cartwheel, with ΔE of 0.2 ± 0.06 V for the former and 0.14 ± 0.03 V for the latter, suggesting a slight reduction in redox reversibility after bending. This modest increase in ΔE supports the inference of partial microcrack formation from EIS and also confirms that such defects do not substantially hinder electron-transfer kinetics. Notably, the preservation of near-reversible CV profiles and consistent high-frequency impedance behaviour indicates that the 3D electrodes retain electrochemical functionality comparable to their planar counterparts. Together, these results demonstrate that although mechanical deformation induces subtle microstructural changes, the 3D SWCNT electrodes maintain their functional performance for electrophysiological applications. The mechanical flexibility required for organoid interfacing is achieved without compromising the high-fidelity signal transduction needed for neural spike acquisition, validating the robustness of our 3D CAMEO architecture.
CAMEOs reliably capture and transmit low-amplitude signals
Having established the electrochemical properties of CAMEO devices, we next validate their functionality, especially in biologically relevant parameter ranges, and their ability to function in a complete acquisition system. Low-amplitude signals are particularly relevant to biological systems. To validate CAMEO’s ability to detect such signals, we use a waveform generator as a positive control to deliver fully defined signals typically expected from cerebral and cardiac tissues. A waveform generator interfaced to a voltage divider circuit supplies a signal of 500 μV at 35 Hz to the solution in which the CAMEO is suspended. Figure 3e reflects the input signal via the waveform generator captured on an oscilloscope (570 μV peak to peak) and the output signal captured by the CAMEO (103.9 μV peak to peak) transmitted to and extracted from a neural data acquisition system (Blackrock CerePlex Direct, Blackrock Neurotech, Salt Lake City, Utah). Notably, the stimulation wires are immersed in the solution at a distance >1 cm from the CAMEO, unlike the intended application where organoids establish direct conformal contact with the sensing surface. This spatial separation likely contributes to the observed signal loss. Nevertheless, the preservation of the waveform shape validates the ability of CAMEO to capture low-amplitude bioelectrical signals reliably.
CAMEOs probe organoid electrophysiology
We finally verify the ability of CAMEOs to record electrophysiological signals from human cerebral organoids and, in particular, from many organoids. We culture cortical organoids using a protocol adapted from Velasco et al.32 (Fig. 4a). While DMEM/F12 serves as a basal medium for organoid generation, it is known to impair neuronal excitability, including spontaneous and evoked action potentials33. To promote neuronal function while maintaining cost-efficiency, we transition organoids to BrainPhys Neuronal Medium8,9,18 two weeks prior to recording, a timeline validated by previous reports34. On the days of recording, the CAMEOs are sterilised under UV light for 30 min. Organoids are pipetted into them (Fig. 1e) and incubated for 1 h to promote conformal electrode-tissue contact and stabilisation of the organoids in the new environment. The CAMEOs are directly connected to a neural data acquisition system (Blackrock CerePlex Direct) through a custom printed circuit board, ensuring standardised and reproducible acquisition across experiments.
a Schematic of the experimental protocol and recording timeline. Schematic created in BioRender. Mishra, N. (2026) https://BioRender.com/7mcqta8. b Representative filtered voltage traces from individual CAMEO channels and overlaid spike waveforms. Scale bars: 100 s (horizontal) and 100 µV (vertical) for traces; 1 ms (horizontal) and 20 µV (vertical) for waveforms. c Pharmacological modulation of activity with 20 µM glutamate. Left: raster plot representation from the same organoid. Right: comparison between spontaneous and glutamate-stimulated firing rates (n = 48 channels from 4 organoids). Statistical significance was assessed using a Mann–Whitney U test (P = 1.43 × 10⁻⁶). Scale bar: 100 s. Data are presented as mean ± SD. d Schematic comparison of neurotypical and Angelman syndrome (AS) chromosomes showing paternal (P) and maternal (M) alleles, with corresponding spike amplitude distributions in neurotypical controls and AS iPSC-derived organoids (Imprinting Centre Defect [ICD] and Uniparental Disomy [UPD] genotypes). Recordings performed at day 170 (34 organoids in experiment). Statistical significance was assessed using a Mann–Whitney U test (P = 0.0261). Schematic created in BioRender. Mishra, N. (2026) https://BioRender.com/lba01cf. e Scatter plot of spike amplitudes across all organoids measured (74 organoids).
Recordings from organoids consistently show extracellular spikes with amplitudes of 10–100 μV and durations of 0.5–1 ms (Fig. 4b), characteristic of neuronal activity, and no signal without organoids (Supplementary Fig. S9). Channels exhibiting low-noise, stable baselines but no detectable spikes reflect spatially quiescent regions of the organoid, consistent with prior reports of electrophysiological heterogeneity in cerebral organoids10,17,18,34 (Supplementary Fig. S10). We validate the physiological origin of these signals through pharmacological stimulation. Glutamate application (20 μM) significantly increases spiking activity, as evidenced by raster plots and a tenfold rise in average spike rate (Fig. 4c). Similarly, potassium chloride (80 mM) elicits an immediate increase in spiking (Supplementary Fig. S11a). While this study focuses on basic spike detection and amplitude measurements to establish proof-of-concept, our recordings also capture additional features, including network bursts (Supplementary Fig. S11b).
Angelman Syndrome (AS) is a genetic disorder caused by loss of UBE3A protein in neurons leading to neurodevelopmental delays35,36,37. Despite the likely role of UBE3A in regulating neuronal activity, as it is known to modulate synaptic protein levels, it remains unclear whether and how their electrophysiological properties are altered. A major challenge is the potential subtlety of phenotypes masked by the variability of cell culture systems. We therefore apply the CAMEO platform to organoids generated from human induced pluripotent stem cells(hiPSCs) derived from AS individuals and compared them to human neurotypical organoids.
In neurotypical individuals, UBE3A expression is restricted to the maternal allele, with the paternal allele silenced in neurons. AS individuals with Uniparental Disomy (UPD) and Imprinting Centre Defect (ICD) epigenotypes aberrantly silence UBE3A on both chromosomes, resulting in a lack of UBE3A protein in neurons (Fig. 4d). UPD and ICD together account for approximately 3–5% of AS cases and their mechanisms and phenotypes are therefore understudied38. Leveraging the high-throughput feature of the CAMEO platform, we record thirty-four 170-day-old organoids derived from patient iPSCs (UPD and ICD) and neurotypical controls. These recordings reveal a significant reduction in spike amplitudes in 18 AS organoids (16.06 ± 4.74 µV) compared to 16 controls (25.56 ± 17.14 µV) (Fig. 4d). We age-match and culture AS and control organoids in parallel, maintaining them on separate plates under identical conditions. Each organoid represents an independent biological sample and is recorded independently on the CAMEO platform. Our results align with previous electrophysiological studies in human iPSC-derived AS neuronal models. Fink et al.36 report extensive patch-clamp and calcium-imaging analyses of AS patient-derived neurons and reported impaired electrophysiological maturation relative to controls. At later developmental stages, similar to our experimental timelines, AS neurons exhibit significantly reduced action potential amplitudes and broader spike waveforms, along with a higher fraction of neurons failing to generate mature firing patterns. These intrinsic deficits persist with neuronal maturation and are accompanied by reduced spontaneous calcium activity at advanced time points. Therefore, our observation of reduced extracellular spike amplitudes in AS UPD and ICD organoids is consistent with previously reported intrinsic electrophysiological deficits in another AS human model. While establishing confidence in the new recording platform, these measurements also extend our understanding of electrophysiological investigation via MEAs to intact 3D patient-derived AS organoids for the first time, to the best of our knowledge, including the first recordings from human UPD and ICD AS genotypes, specifically.
To further demonstrate the scalability and reproducibility of the CAMEO system, we record spontaneous activity from forty more cortical organoids (Fig. 4e). This dataset highlights the ability to handle large experimental cohorts efficiently, overcoming a key limitation of prior organoid electrophysiology studies that typically report only a small number of samples due to technical throughput limitations despite the known biological variability of these models. Because these organoids are pooled across multiple experiments, developmental stages, and genetic backgrounds, including healthy H9 and genetically modified lines, the data are presented to demonstrate platform scalability rather than to support specific biological conclusions.
Discussion
This work addresses a major multifaceted limitation of current organoid electrophysiology platforms: scalability. Multiple barriers must be addressed to achieve practical, high-throughput measurements including cost, mechanical integrity, electrochemical robustness, facile assembly and manufacturing, and compatibility with standard organoid culture protocols. CAMEO directly addresses these barriers. Its use of SWCNTs reduces fabrication costs while providing superior electrical properties, including low impedance and high charge-transfer capability. The fabrication process is cleanroom-independent, enhancing access to labs that may not have the infrastructure to support resource-intensive lithography and deposition techniques. A key innovation is the system's scalable manufacturing. The fabrication process has the potential to produce centimetre-scale films by tailoring the initial SWCNT concentration in the dispersion, the dispersion volume, and the size of the assembly container. Furthermore, the CAMEO fabrication process can be adapted to extend the batch yield dramatically, using a laser or die cutting machine with a conveyor belt. The manufacturing setup used in this work supports the simultaneous fabrication of 48 cartwheels in a single batch. Our system also reliably records neural signals without needing to coat electrodes with extracellular matrix proteins or to chemically modify the surface, streamlining the workflow and reducing experimental variability.
We also describe an even lower cost manufacturing route using a CO₂ laser cutter (Epilog, Golden, CO) (Supplementary Fig. S12a, b). While this manufacturing tool results in larger-sized CAMEOs (active area 500 µm × 1000 µm), the resulting devices are still capable of real-time electrophysiological monitoring. This alternative fabrication approach enables the platform to be deployed in basic laboratory settings without sacrificing performance. Beyond fabrication, we also address the cost barrier associated with commercial neural data acquisition systems, which often limit the adoption of electrophysiological studies in organoid research. As demonstrated in Supplementary Fig. S12c, our device can be directly interfaced with a low-cost, handheld bio amplifier (Neuron Spiker Box, Backyard Brains, Ann Arbour, Michigan)39 to record high-fidelity neural activity. This plug-and-play compatibility ensures that the platform can be implemented even in resource-limited environments, expanding its potential user base and enabling broader adoption.
Organoid electrophysiology remains challenging due to the sporadic and heterogeneous nature of organoid activity, which is influenced by factors including differentiation state, culture conditions, and experimental setup. To advance the field, it is essential to establish standardised, scalable technologies that enable comparable data generation across laboratories. Our work represents a significant step toward this goal by delivering an accessible yet high-performance multi-organoid screening platform. By lowering the barriers to adoption while ensuring robust signal acquisition, this technology has the potential to accelerate discoveries in neurodevelopmental disease modelling, drug screening, and large-scale functional phenotyping. Eventually, longitudinal electrophysiological monitoring of organoids will be essential for capturing developmental trajectories and long-term network dynamics. As a preliminary assessment of CAMEO’s suitability for such applications, accelerated ageing studies show that the platform remains electrically functional after incubation in phosphate-buffered saline at 60 °C for seven days, with impedance at 1 kHz exhibiting a maximum absolute deviation of approximately 26% relative to the initial measurement (Supplementary Fig. S13a, b). Building on this foundation, future work will involve further development of the CAMEO platform to support longitudinal electrophysiological recordings under physiological culture conditions, alongside the development of advanced data analysis pipelines capable of processing large-scale neural datasets and extracting complex network parameters such as burst synchrony, oscillatory patterns, and connectivity metrics.
Methods
Fabrication of the 3D CAMEO platform
Fabrication began by dispersing carbon nanotube (SWCNT) powder (Tuball, OCSiAl, Leudelange, Luxembourg) in CA (Sigma Aldrich, St. Louis, MO) at a concentration of 1 mg/mL, followed by vortexing for 3 min. Pouring the dispersion into a glass petri dish initiated self-assembly of the SWCNT sheet. Washing with distilled water and transferring the film onto tattoo paper (BubblePop!, Amazon, Seattle, WA) prepared it for subsequent processing. The sheet was allowed to air dry under ambient conditions.
A 30% (w/v) polystyrene-block-polyisoprene-block-polystyrene (SIS) polymer (Sigma Aldrich, St. Louis, MO) solution in Toluene (Sigma Aldrich, St. Louis, MO) was screen-printed onto tattoo paper to form the insulating substrates. Designs for all three cartwheel layers were created using a computer-aided-design software (AutoCAD, Autodesk, San Francisco, CA). A picosecond laser system (ProtoLaser R4, LPKF, Garbsen, Germany) defined the electrode geometry by cutting the SWCNT layer, which was then laminated onto the SIS-coated tattoo paper and heat bonded at 140 °C for 10 s. Applying water removed the tattoo paper backing, exposing the pristine SWCNT surface.
Laser cutting the top SIS layer defined the electrode window. Alignment and lamination onto the CNT film, followed by a second round of heat bonding and water application, completed encapsulation. Cutting the bottom geometry released the full cartwheel layout. Six cartwheels were simultaneously loaded into a 3D-printed stencil for further assembly.
Assembly began with 3D printing a six-pillar stand. Coating the pillar tips with tacky silicone (Ecoflex GEL, Smooth-on, Macungie, PA) and curing them generated adhesive sites for cartwheel pickup. An assembly ring with twelve contact pad slots was laser-cut from paper and bonded to a second ring of tape (Gorilla Tape, The Gorilla Glue Company, Cincinnati, OH) with laser-cut via holes. This assembly was secured to the stand using double-sided tape (Scotch, 3 M, St. Paul, MN).
Inverting the stand and bringing it into contact with the stencil allowed the adhesive-coated pillars to pick up the cartwheels at their centres. Lifting the stand transferred all six cartwheels. Water was applied to the spokes of the cartwheel, and each contact pad was aligned and pressed into the corresponding slot on the assembly ring using fine forceps. Applying water dissolved the water-soluble adhesive layer of the tattoo paper, exposing the transformed CAMEO.
Preparation of the tissue culture plate involved laser-cutting 6 mm holes into the centre of each well in a six-well plate lid (Corning) using a CO₂ laser (Epilog, Golden, CO). A thin ring of epoxy (JB Weld, Sulphur Springs, TX) was patterned onto the lid to match the assembly ring geometry. Inverting the loaded stand and placing it onto the lid inserted the 3D CAMEOs through the holes, where the gel secured them in place. Removing the stand completed the assembly. A custom PCB breakout board (PCBWay, Hangzhou, Zhejiang, China) was mounted onto the lid to establish electrical interfacing, finalising the fabrication of the 6-well CAMEO platform.
Electrochemical characterisation of SWCNT and gold electrodes
Electrochemical comparisons between carbon nanotube (SWCNT) and gold electrodes took place using identically structured sensors. Fabrication began with laser-cutting circular electrodes (2 mm diameter) from SWCNT and gold films (200 nm Au with an underlying layer of 20 nm titanium sputtered on 75 µm polyimide film) followed by affixing the electrodes onto polyimide (PI) substrates via an epoxy glue for mechanical support. Conductive silver epoxy (MG Chemicals, Burlington, Ontario, Canada) established electrical connections between electrodes and hook-up wires, and marine epoxy (JB Weld) insulated exposed conductive regions. A potentiostat (PalmSens4, PalmSens BV, Houten, Netherlands) interfaced with PS Trace software (PalmSens BV) was used to perform CV at a scan rate of 50 mV/s over a potential window of −0.8 V to +0.8 V and Electrochemical impedance spectroscopy (EIS) across a frequency range of 0.1–105 Hz. Ag/AgCl served as the reference electrode and a platinum wire acted as the counter electrode. All measurements were carried out in 1X potassium ferricyanide (Sigma Aldrich, St. Louis, MO) prepared in phosphate-buffered saline. Data was exported to a data analysis and graphing software (Origin, OriginLabs, Northampton, MA). An open-source Python package impedance.py40 allowed fitting the impedance data to a Randles circuit.
Electrochemical assessment of CAMEO performance under mechanical bending
The 2D cartwheels for this study were fabricated by halting fabrication after the laser cutting stage. Attaching wires to the contact pads using silver epoxy (MG Chemicals, Burlington, Ontario, Canada) established electrical connections, followed by insulation with marine epoxy (JB Weld, Sulphur Springs, TX). CV was performed over a potential window of -0.3 V to 0.8 V at a 50 mV/s scan rate and electrochemical impedance spectroscopy (EIS) across a frequency range of 105–0.1 Hz on both 2D cartwheels and 3D CAMEOs in a 100 mM potassium ferricyanide solution prepared in phosphate-buffer saline. Ag/AgCl and a platinum wire served as the reference electrode and counter electrode respectively. Data was exported to a data analysis and graphing software (Origin) for further analysis.
Validation of the recording ability of CAMEOs using simulated waveforms
A waveform generator (Agilent, Santa Clara, CA) was used to supply a 500 mV signal at 35 Hz via a voltage divider circuit into a well of a 6-well plate containing phosphate-buffered saline using two wires. The same well contained the CAMEO suspended from the lid. Input voltage from the waveform generator was measured on an oscilloscope (Keysight, Santa Rosa, CA), and the CAMEO was interfaced with the neural data acquisition system (Blackrock CerePlex Direct, Blackrock Neurotech, Salt Lake City, UT) via a custom printed circuit board (PCBWay, Hangzhou, Zhejiang, China) to record the signals captured by it. The input and output voltage traces were compared in MATLAB.
Mechanical modelling of the 3D CAMEOs
Three-dimensional (3D) quasi-static finite element (FE) simulations were performed in an FEA software (ABAQUS 2023, Dassault Systèmes, Vélizy-Villacoublay, France) to model the mechanical response of the CAMEO system, with the goal of capturing the deformation behaviour of MEAs under experimental loading conditions and identifying strain localisation in regions critical for structural integrity and functional performance. The modelled cartwheel structure features twelve symmetric legs extending radially from the centre to the bonding sites with an overall diameter of 36.6 mm. Boundary conditions were defined to replicate the assembly process of the device. Each electrode was prescribed a displacement of 14.15 mm in the radial direction and 14.00 mm in the vertical direction toward the bottom assembly ring. The central region in contact with the cylindrical pillar was fixed in all degrees of freedom to represent the mechanical constraint imposed during assembly. Each leg (width = 0.3 mm) ended in a square contact pad that was constrained in-plane for conformal adhesion to the assembly ring and to guarantee the zero-rotation boundary conditions at the leg boundaries. The cartwheel layers consisted of a thin 10 µm carbon nanotube (SWCNT) film embedded between two polystyrene-block-polyisoprene-block-polystyrene encapsulation layers, each 20 µm thick. The total structural thickness, including the metal traces, was approximately 50 µm. In the simulations, the yield strain of the SWCNT layer was defined as 2.0%, as determined from experimental measurements. Self-contact was enforced for each electrode surface to eliminate nonphysical penetration during deformation. Following device assembly, a prescribed spherical displacement was imposed on the central cradle region to replicate the confinement/wrapping produced by the organoid. This boundary condition enabled a parametric study of organoid radius, varied from 0.3 mm to 3.5 mm, to evaluate its effect on local strain distributions in the legs and overall deformation behaviour. The device was discretized using four-node reduced-integration shell elements (S4R), while the support cylinder and organoid were modelled as hyperelastic Mooney Rivlin solids (C10 = 0.201 kPa, C01 = 0.051 kPa, and D1 = 0.08 kPa-1) with first-order, eight-node hybrid solid elements (C3D8H). The final mesh contained approximately 160,000 elements, which ensured computational accuracy and convergence. Each electrode leg was meshed with at least twelve elements across the width to capture strain gradients. The stress-strain curves of SWCNT and SIS substrates were obtained using a tensile testing machine (Instron 5943, Instron, Norwood, MA). The elastic modulus and Poisson’s ratio were obtained from fits with the experimental measurements.
Cyclic Bending Test
Bending tests utilised three rectangular electrodes (20 mm×5 mm) each for Au/PI and SWCNT/SIS substrates. Mounting the substrates on metal strips attached to a motorised tension / compression test stand (ESM303, Mark-10, Copiague, NY) allowed for continuous uniaxial bending of the substrates for 1000 cycles at the rate of 0.17 Hz. A Data Acquisition System (DAQ970A, Keysight, Santa Rosa, CA) recorded resistances of the substrates at a rate of 1 Hz via wires attached to both the contact pads of the substrates. Recorded resistances were exported to a data analysis and graphing software (Origin) for analysis and interpretation.
Generation of cortical organoids
WiCell (Madison, WI) originally provided feeder-independent H9 human embryonic stem cells (hESCs) (WA09). Dr. Yong-Hui Jiang at the Yale University School of Medicine, New Haven, CT provided control hiPSC lines along with AS UPD and ICD hiPSC lines. Cells were maintained in tissue culture-treated six well plates (Fisher Scientific, Waltham, MA) coated with growth factor reduced Matrigel (Corning, Corning, NY) at 37 °C and 5% CO2 until ready for organoid culture in mTeSR Plus medium (Stemcell Technologies, Vancouver, Canada). Human cortical human cerebral organoids were generated using a protocol adapted from Velasco et al.32. Briefly, culture began with lifting 70-80% confluent cells from the plate using 0.5 mM EDTA (Invitrogen, Thermofisher Scientific, Waltham, MA) solution in DPBS without calcium or magnesium (Gibco, Thermofisher Scientific, Waltham, MA) and dissociated into single cells using Accutase (BioLegend, San Diego, CA). This was followed by resuspending the cells in cortical differentiation medium I (CDM I) containing G-MEM (Gibco), KnockOut Serum Replacement (Gibco), Non Essential Amino Acids (NEAA) (Hyclone, Cytiva, Marlborough, MA), Sodium Pyruvate (Gibco), 2-mercaptoethanol (Amresco, Solon, OH) and Penicillin/Streptomycin (GenClone, Genesee Scientific, El Cajon, CA). Seeding 9000–10,000 cells into each well of a 96-well V bottom plate (S-BIO) containing CDM I, ROCK inhibitor (MedChemExpress, Monmouth Junction, NJ), IWR1 (Millipore Sigma, Burlington, MA) and SB 431542 (Stemcell Technologies) completed the initial formation of embryoid bodies. The media was changed every 3 days until day 18. ROCK inhibitor was only included in the first 6 days following seeding. The organoids were transferred to 100-mm dishes containing CDM II (DMEM/F12 (Gibco), GlutaMAX Supplement (Gibco), N-2 supplement (Gibco), Chemically Defined Lipid Concentrate (Gibco), Fungizone/Amphotericin B (Gibco) and Penicillin/Streptomycin (GenClone) on day 18 and placed on a shaker in a tissue culture incubator (70 rpm). Day 35 onwards, the organoids were cultured in 100 mm dishes containing CDM III (DMEM/F12 (Gibco), Foetal Bovine Serum (FBS) (Seradigm, Avantor, Radnor, PA), chemically defined lipid concentrate (Gibco), GlutaMAX supplement (Gibco), N-2 supplement (Gibco), heparin (Sigma-Aldrich), fungizone/amphotericin B (Gibco), penicillin/streptomycin (GenClone) and geltrex (Gibco). Day 70 onwards, medium was switched to CDM IV (DMEM/F12 (Gibco), FBS Seradigm, Avantor), Chemically Defined Lipid Concentrate (Gibco), GlutaMAX supplement (Gibco), N-2 supplement (Gibco), Heparin (Sigma-Aldrich), Fungizone/Amphotericin B (Gibco), Penicillin/Streptomycin (GenClone), B-27 supplement (Gibco) and Geltrex (Gibco). Organoids were maintained on a shaker in a humid incubator at 37 °C with 5% CO2 until the day of experiments. Two weeks prior to recording, organoids were transferred to BrainPhys Neuronal Medium (Stemcell Technologies). For the first week, half media changes were done with 50% CDM IV and 50% BrainPhys to adapt the organoids to the change followed by full media changes with Brainphys the second week. Organoids were maintained in BrainPhys henceforth, for the full duration of experiments.
Insertion of Organoids into the CAMEOs
Sterilising the modified 6-well plate and the lid with suspended CAMEOs under UV light for 30 min prepared it for insertion of organoids. Cutting the ends of 1 mL pipette tips to widen the opening allowed gentle handling of organoids. Pipetting the organoids into the CAMEOs through the holes on the lid and closing the lid on the plate with filled media completed the process. The plate was incubated at 37 °C and 5% CO₂ for 1 h prior to recording. Performing all organoid handling and transfer steps within a biosafety cabinet using sterile culture media and pipette tips maintained sterility.
Electrophysiological Data Acquisition and processing
Blackrock CerePlex Direct in conjunction with a 32-channel Blackrock μ headstage (Blackrock Neurotech, Salt Lake City, UT) served as the data acquisition system for electrophysiological recordings. Copper wires served as reference and ground electrodes, placed in the media at a distance from the CAMEOs. The data sampling rate was 30 kHz and recording duration was 20 min. Covering the openings of the CAMEOs with sterile tape during electrophysiological recordings minimised environmental exposure and reduced the risk of contamination.
Data analysis began with importing raw.ns6 files from the acquisition system into a software that supports scripts provided by Blackrock Neurotech (MATLAB, MathWorks, Natick, MA). We developed a custom MATLAB code, partially adapted from Floch et al.17 for further analysis. The code applied a 4th-order Butterworth high-pass filter (300 Hz) and identified spikes using a threshold set at -6 times the standard deviation of the filtered signal. For each electrode, it calculated the spike count and average amplitude. Test recordings from the Blackrock digital neural signal simulator validated the accuracy of our code. Statistical tests were carried out in MATLAB.
Data availability
The research findings presented in this study are supported by data included in the main text and in the Supplementary Information. Due to large size, raw datasets are available from the corresponding author on reasonable request. Code Availability Statement: The code used for neural data analysis in this study is available from the corresponding authors on reasonable request.
Code availability
The code used for neural data analysis in this study is available from the corresponding authors on reasonable request.
References
Rubio, A. D. et al. A comprehensive review on utilizing human brain organoids to study neuroinflammation in neurological disorders. J. Neuroimmune Pharmacol. 20, 23 (2025).
Pașca, S. The rise of three-dimensional human brain cultures. Nature 553, 437–445 (2018).
Ajongbolo, A. O. & Langhans, S. A. Brain organoids and assembloids-from disease modeling to drug discovery. Cells 14, 842 (2025).
Lancaster, M. A. et al. Cerebral organoids model human brain development and microcephaly. Nature 501, 373–379 (2013).
Sandoval, S. O. et al. Rigor and reproducibility in human brain organoid research: where we are and where we need to go. Stem Cell Rep. 19, 796–816 (2024).
Urrestizala-Arenaza, N., Cerchio, S., Cavaliere, F. & Magliaro, C. Limitations of human brain organoids to study neurodegenerative diseases: a manual to survive. Front. Cell Neurosci. 18, 1419526 (2024).
Trujillo, C. A. et al. Complex oscillatory waves emerging from cortical organoids model early human brain network development. Cell Stem Cell 25, 558–569 (2019).
Fair, S. R. et al. Electrophysiological maturation of cerebral organoids correlates with dynamic morphological and cellular development. Stem Cell Rep. 15, 855–868 (2020).
Sharf, T. et al. Functional neuronal circuitry and oscillatory dynamics in human brain organoids. Nat. Commun. 13, 4403 (2022).
Yang, X. et al. Kirigami electronics for long-term electrophysiological recording of human neural organoids and assembloids. Nat. Biotechnol. 42, 1836–1843 (2024).
Wu, Y. et al. Three-dimensional liquid metal-based neuro-interfaces for human hippocampal organoids. Nat. Commun. 15, 4047 (2024).
Kopic, I. et al. Inkjet-printed 3D electrode arrays for recording signals from cortical organoids. Adv. Mater. Technol. 9, 2400645 (2024).
Zhang, H., Huang, N., Bian, S. & Sawan, M. Platinum wire-embedded culturing device for interior signal recording from lollipop-shaped neural spheroids. Cyborg Bionic Syst. 6, 0220 (2025).
Kim, E. et al. Magnetically reshapable 3D multi-electrode arrays of liquid metals for electrophysiological analysis of brain organoids. Nat. Commun. 16, 2011 (2025).
Huang, Q. et al. Shell microelectrode arrays (MEAs) for brain organoids. Sci. Adv. 8, 5031 (2022).
McDonald, M. et al. A mesh microelectrode array for non-invasive electrophysiology within neural organoids. Biosens. Bioelectron. 228, 115223 (2023).
Le Floch, P. et al. Stretchable mesh nanoelectronics for 3D single-cell chronic electrophysiology from developing brain organoids. Adv. Mater. 34, 2106829 (2022).
Park, Y. et al. Three-dimensional, multifunctional neural interfaces for cortical spheroids and engineered assembloids. Sci. Adv. 7, abf9153 (2021).
Siljander, S. et al. Effect of surfactant type and sonication energy on the electrical conductivity properties of nanocellulose-CNT nanocomposite films. Int. J. Mol. Sci. 19, 1819 (2018).
Jiang, Y., Song, H. & Xu, R. Research on the dispersion of carbon nanotubes by ultrasonic oscillation, surfactant and centrifugation respectively and fiscal policies for its industrial development. Ultrason. Sonochem. 48, 30–38 (2018).
Ramesh, S. et al. Dissolution of pristine single walled carbon nanotubes in superacids by direct protonation. J. Phys. Chem. B. 108, 8794–8798 (2004).
Davis, V. A. et al. True solutions of single-walled carbon nanotubes for assembly into macroscopic materials. Nat. Nanotechnol. 4, 830–834 (2009).
Musameh, M., Lawrence, N. S. & Wang, J. Electrochemical activation of carbon nanotubes. Electrochem. Commun. 7, 14–18 (2005).
Komarova, N. S., Krivenko, A. G., Ryabenko, A. G. & Naumkin, A. V. Active forms of oxygen as agents for electrochemical functionalization of SWCNTs. Carbon 53, 188–196 (2013).
Nelson, M. J., Pouget, P., Nilsen, E. A., Patten, C. D. & Schall, J. D. Review of signal distortion through metal microelectrode recording circuits and filters. J. Neurosci. Methods 169, 141–157 (2008).
Lewis, C. M. et al. Recording quality is systematically related to electrode impedance. Adv. Healthc. Mater. 13, 2303401 (2024).
Vitale, F., Summerson, S. R., Aazhang, B., Kemere, C. & Pasquali, M. Neural stimulation and recording with bidirectional, soft carbon nanotube fiber microelectrodes. ACS Nano 9, 4465–4474 (2015).
Balamur, R. et al. Capacitive and efficient near-infrared stimulation of neurons via an ultrathin AgBiS2 nanocrystal layer. ACS Appl. Mater. Interfaces 16, 29610–29620 (2024).
Kadan-Jamal, K. et al. Electrical stimulation of cells: drivers, technology, and effects. Chem. Rev. 125, 6874–6905 (2025).
Bosch, R. W. Electrochemical impedance spectroscopy for the detection of stress corrosion cracks in aqueous corrosion systems at ambient and high temperature. Corros. Sci. 47, 125–143 (2005).
Bandodkar, A. J., Nuñez-Flores, R., Jia, W. & Wang, J. All-printed stretchable electrochemical devices. Adv. Mater. 27, 3060–3065 (2015).
Velasco, S., Paulsen, B. & Arlotta, P. Highly reproducible human brain organoids recapitulate cerebral cortex cellular diversity. Protoc. Exch. https://doi.org/10.21203/rs.2.9542/v1 (2019).
Bardy, C. et al. Neuronal medium that supports basic synaptic functions and activity of human neurons in vitro. Proc. Natl. Acad. Sci. USA 112, E2725–E2734 (2015).
Oliva, M. K. et al. Standardizing a method for functional assessment of neural networks in brain organoids. J. Neurosci. Methods 409, 110178 (2024).
Xuyang Sun, A. et al. Potassium channel dysfunction in human neuronal models of Angelman Syndrome. Science 366, 1486–1492 (2019).
Fink, J. J. et al. Disrupted neuronal maturation in Angelman syndrome-derived induced pluripotent stem cells. Nat. Commun. 8, 15038 (2017).
Sen, D., Voulgaropoulos, A., Drobna, Z. & Keung, A. J. Human cerebral organoids reveal early spatiotemporal dynamics and pharmacological responses of UBE3A. Stem Cell Rep. 15, 845–854 (2020).
Eggermann, T. et al. Imprinting disorders: a group of congenital disorders with overlapping patterns of molecular changes affecting imprinted loci. Clin. Epigenet. 7, 1–18 (2015).
Marzullo, T. C. & Gage, G. J. The SpikerBox: a low cost, open-source bioamplifier for increasing public participation in neuroscience inquiry. PLoS One 7, e30837 (2012).
Murbach, M. D., Gerwe, B., Dawson-Elli, N. & Tsui, L. Impedance.py: a Python package for electrochemical impedance analysis. J. Open Source Softw. 5, 2349 (2020).
Acknowledgements
This study was funded by the Foundation for AS Therapeutics (FAST), Austin, TX (Award Number FT2022-02). The funder played no role in study design, data collection, analysis and interpretation of data, or the writing of this manuscript. A.J.B. acknowledges funding from the National Science Foundation (Award Number ECCS-2216312) for purchasing the picosecond laser (LPKF R4) that was used for fabricating the CAMEOs. This work was performed in part at the NCSU Nanofabrication Facility (NNF), a member of the North Carolina Research Triangle Nanotechnology Network (RTNN), which is supported by the National Science Foundation (Award Number ECCS-2025064) as part of the National Nanotechnology Coordinated Infrastructure (NNCI). We thank Dr. Yong-Hui Jiang (Yale University School of Medicine, New Haven, CT) for graciously providing us the control hiPSC lines along with AS UPD and ICD hiPSC lines.
Author information
Authors and Affiliations
Contributions
N.M., A.J.K., and A.J.B. conceptualised the project and designed experiments. N.M. performed experiments. N.M. and R.K. designed, fabricated, and photographed the devices and generated schematics embedded in the manuscript. P.L. and R.A. performed mechanical simulations and wrote the corresponding part of the manuscript. N.M., B.E.U., and A.M. designed PCBs. N.M., A.M., and A.T. wrote the code for neural data analysis and analysed the corresponding data. N.M. analysed and interpreted data for all other studies. N.M., Z.B.Y., and T.J. cultured cells. N.M., B.E.U., and P.A. performed benchtop electrical simulation experiments. N.M., A.J.K., and A.J.B. wrote the manuscript. Q.W., Y.Z., J.Y.K., M.D.D., P.B., S.M., P.A., A.B., and S.V. provided access and training on laboratory instruments for studies.
Corresponding authors
Ethics declarations
Competing interests
N.M., A.J.K., and A.J.B. have filed a disclosure on the CAMEO technology from the North Carolina State University, NC, USA. The other authors do not have a competing interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mishra, N., Kaveti, R., Liu, P. et al. Carbon nanotube microelectrode arrays enable scalable and accessible electrophysiological recordings of cerebral organoids. npj Biosensing 3, 20 (2026). https://doi.org/10.1038/s44328-026-00088-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44328-026-00088-9