Abstract
This paper proposes a conceptual ethical framework to guide decision-making around blood-based biomarker (BBM) testing for Alzheimer’s disease in primary care, where patients often present with cognitive impairment and multiple chronic conditions. Grounded in current evidence and ethical principles, it addresses clinical benefit, patient values, risk, and justice. Developed by a multidisciplinary team, the framework is illustrated through real-world case scenarios and highlights future research needs to support ethical implementation.
Similar content being viewed by others
Introduction
The emergence of blood-based biomarkers (BBMs), alongside the availability of disease-modifying therapies (DMTs), is poised to transform the diagnostic and therapeutic landscape of Alzheimer’s disease (AD)1. Under recently updated diagnostic criteria, BBMs are expected to play a central role in identifying individuals who may benefit from treatment2,3,4. Compared to cerebrospinal fluid (CSF) and PET biomarkers, BBMs are more scalable and less invasive, enabling potential use beyond specialty centers, including in primary care3.
However, while clinical adoption of BBMs is advancing, a critical gap remains in ethical guidance. Current recommendations focus on BBM use in specialized care settings and emphasize analytical validity, performance thresholds, and appropriate diagnostic contexts2,5,6,7. BBMs are commercially available as clinician ordered Laboratory Developed Tests (LDTs) and, in some cases, through consumer-initiated pathways (e.g., Direct to Consumer Testing). Emerging reports suggest BBM is being selectively adopted in primary care and other nonspecialist settings, signaling a real-world shift that outpaces current guidance8,9,10. Recently, the first BBM testing, Elecsys pTau181, has been FDA cleared for use in primary‑care settings11,12. While BBM offers significant benefits for improving patient care and are central to accessing DMT, ethical guidance is needed to support responsible use of BBMs. This is particularly important for primary care, where most older adults with dementia are diagnosed7. In these settings, clinicians may be challenged to manage complex clinical scenarios that include multiple chronic conditions, prognostic uncertainty, and limited access to follow-up diagnostic resources7.
This manuscript introduces an ethical framework to guide decision-making around AD BBM testing, with a focus on primary care settings. It integrates anticipated benefits, patient-centered goals, potential harms/risks, and justice. Importantly, the framework considers medical futility as an ethical boundary for when testing is unlikely to offer meaningful benefit. By aligning testing decisions with clinical complexity, patient values, and evolving evidence, this framework aims to support ethically sound and person-centered use of BBMs that reflect the realities of clinical care.
Framework development
This ethical framework was developed within the Alzheimer’s Diagnosis in Older Adults with Chronic Conditions (ADACC) Network (NIH/NIA U24AG082930). The ADACC Ethics Working Group meets monthly to identify and evaluate ethical challenges associated with diagnosis, detection, and treatment of Alzheimer’s disease in the context of persons with co-morbidities and chronic conditions.
The Working Group identified a need for a framework to evaluate responsible use of BBMs in primary care settings. To develop the framework, the group conducted a literature review across four domains: (1) evidence on the clinical utility and performance of BBMs; (2) emerging data on stakeholder experiences and ethical concerns; (3) complexity of diagnosing cognitive impairment in real-world primary care; and (4) the concept of medical futility as applied to BBM testing. Drawing from this review, the group used an iterative, consensus-based process to define the framework’s core components.
To reinforce its clinical relevance, the framework was applied to case scenarios inspired by real-world dilemmas and refined through discussion with experienced dementia clinicians and scientists with expertise in dementia care, bioethics, and implementation research. These illustrative cases served to validate and shape the framework, ensuring it was conceptually sound, ethically grounded, and applicable to clinical practice.
Evidence of clinical utility and performance of BBM testing
AD BBMs are now clinically available in the U.S.13. Among BBMs, plasma p-tau217, used alone or in combination with the Aβ42/40 ratio, has demonstrated strong diagnostic performance1,6,12,13,14. It also shows high discriminative accuracy in differentiating AD from other neurodegenerative conditions, such as progressive supranuclear palsy15, supporting its potential clinical utility. In May 2025, the Lumipulse G p-tau217/β-amyloid 1-42 plasma ratio test became the first BBM to receive FDA 510(k) clearance for clinical use in AD diagnosis16.
Current evidence and guidelines recommend BBM testing by specialists for patients with objectively confirmed cognitive impairment2,7. The guidelines distinguish the use of BBMs for “triage” versus “confirmatory” purposes2. The evidence establishes performance thresholds, approximately 90% sensitivity and specificity, for BBMs to be considered confirmatory tools and emphasizes the importance of interpreting results within the context of a comprehensive evaluation and high pretest probability of AD pathology2,5,6.
However, commercial tests such as C₂N’s PrecivityAD® and Quest AD-Detect™ are increasingly available as LDTs through clinician-ordered pathways, specific laboratories or networks, and are not subject to formal FDA oversight17. These tests are already being used selectively in non-specialist settings, including primary care, driven by their accessibility and appeal as less invasive diagnostic tools, particularly where CSF or PET-based diagnostics are impractical8,9,10. In addition, Elecsys pTau181 has recently been cleared for use in primary‑care settings to aid in assessment of Alzheimer’s‑related amyloid pathology15,16. This real-world expansion introduces ethical challenges not addressed by existing guidelines. These concerns are now increasingly reported by clinicians, patients, and caregivers with direct experience of BBM use.
Stakeholders’ experiences and ethical concerns
While emerging empirical studies highlight the perceived utility of BBMs across stakeholder groups, they also reveal several ethical concerns related to their clinical use. Caregivers and older adults report value of BBMs for facilitating health planning and early decision-making, but report concerns about psychological distress, privacy, and potential insurance discrimination18,19,20. Clinicians, particularly in primary care, echoed these concerns and expressed additional uncertainty about the interpretability of results, clinical utility in non-specialist settings, and the risk of unintended consequences such as overdiagnosis or financial strain on patients21. The Alzheimer’s Association’s 2025 guidelines similarly emphasize ethical challenges, including informed consent, result communication, and equitable access2. These findings underscore the need for ethical guidance that addresses the nuanced realities of BBM use, especially in cases complicated by multimorbidity, prognostic uncertainty, or diverse patient goals of care.
Complexity of diagnosing cognitive impairment in real-world primary care
Despite advances in AD biomarker development, BBMs should not be used in isolation. A positive result may suggest underlying AD pathology but must be interpreted within a comprehensive clinical evaluation that includes cognitive, neuropsychiatric, and functional assessment, and medical history7. This is especially critical in adults over 80, who often present with mixed or non-AD neuropathology, where a biomarker-positive result may not fully explain symptoms. In some cases, prioritizing interventions such as vascular risk management, sleep disorder treatment, depression care, or lifestyle modification may offer greater clinical value than BBM-driven diagnostics alone.
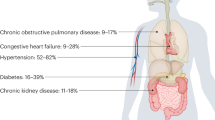
Patients in primary care are generally older, with an average diagnosis age of 8322, and typically present multiple chronic conditions. Over 95% of individuals with AD have at least one other chronic condition, and over 60% have three or more23,24,25. Common coexisting diagnoses, such as hypertension, heart disease, kidney disease, and diabetes, may influence cognitive decline or confound biomarker interpretation values26,27,28,29. Depression, alcohol use disorder, sleep-related issues, and polypharmacy may further impair cognition and obscure diagnostic clarity30,31,32,33. As a result, 50–70% of individuals with AD are undiagnosed or misdiagnosed in primary care3.
The concept of medical futility as applied to BBM testing
Medical futility offers a useful ethical boundary for identifying when BBM testing may be inappropriate, particularly in clinical contexts marked by multimorbidity, uncertainty, or limited benefit. Quantitative futility refers to situations in which a test is unlikely to yield a technically valid or actionable result, such as when comorbidities interfere with biomarker accuracy34,35. Qualitative futility applies when results would not improve quality of life, inform care decisions, or support a patient’s goals. For example, testing may be considered qualitatively futile for a patient with limited life expectancy or when testing would not meaningfully change diagnosis, management, or help achieve patient goals34,35.
Practical ethical framework for BBM use
The proposed framework (Table 1) is intended primarily for clinicians who care for older adults with cognitive impairment in primary care and other community-based settings, where access to specialists and advanced diagnostic tools may be limited. It may also inform efforts by health systems leaders, implementation researchers, and policymakers working to integrate BBMs ethically into routine care. By addressing ethical challenges that extend beyond specialty memory clinics, the framework supports more equitable, context-sensitive, and patient-centered decision-making.
The proposed framework harnesses four key ethical norms, including anticipated benefits, patient-centered goals, potential harms or risks, and justice. We provide a flexible structure that encourages thoughtful clinical judgment and shared decision-making with patients, supported by current criteria and guidelines7. Within this structure, we highlight responsible use and a patient-centered approach to biomarker testing.
The first consideration is the potential benefit or value of BBM testing. The concept of “benefit” as a prima facia consideration is well established in ethical frameworks across domains and applications36,37,38. Broadly, BBMs may improve diagnostic accuracy and timeliness. However, benefits at an individual level may differ and depend on the potential uses or values of results for a patient. Clinicians may consider various factors, including:
-
The purpose of the biomarker testing (screening, confirmation, or to determine eligibility for treatment).
-
The availability of follow-up testing for the patient for this specific patient, including access to confirmatory testing.
-
Treatment options for the patient.
-
Non-treatment clinical benefits for this patient or personal utility39, including benefits of a definitive diagnosis or advanced care planning.
Patients’ goals of care and values may significantly inform the potential benefit that could be accomplished from testing (e.g. diagnostic clarity or planning for the future), even in the absence of treatment options2,34,40,41. Pre-test counseling is important to determine if an individual patient will benefit from testing. During counseling, if clinicians and patients do not identify a benefit, it is ethically justifiable to decline testing.
The second consideration involves patient-specific factors that affect the usefulness or reliability of BBM testing. Patient specific factors may range from clinical status (e.g., other chronic conditions), age, psychosocial factors, and patient values. A patient’s pre-existing clinical status, including co-morbidities and other chronic conditions, may affect the potential usefulness of biomarker testing results2. As highlighted above, some clinical patients may have chronic conditions associated with aging. The presence of other chronic conditions may cause cognitive symptoms unrelated to AD pathology (e.g., co-pathologies, multiple chronic conditions, polypharmacy, lifestyle or other factors). Underlying chronic conditions may also affect testing outcomes or treatment options. For example, impaired kidney function could affect test results and impede accurate interpretation42. This could undermine the value of testing, and it may be ethically justifiable to not offer BBM testing due to medical futility or to offer a different biomarker modality. Similarly, it may be justifiable to decline testing if a patient’s primary goal is to access DMT, but they are not a candidate due to other factors (e.g., a high number of microhemorrhages)43.
It is unethical to decline biomarker testing based on psychosocial factors (e.g., financial status). Yet, psychosocial factors may be relevant to broader decision-making (e.g., insurance status). If the patient’s primary goal of testing is to determine eligibility for DMT and their insurance coverage would not cover testing or treatment, clinicians should discuss the economic consequences of testing and barriers44. This should be a joint decision, and patient-financial factors are not determinative.
A third consideration is the potential for risks or harm to a patient following AD BBM testing and disclosure of the results. While physical risks of BBM testing are minimal, potential risks may include distress, discrimination, stigma, and financial distress45,46,47,48. These consequences could outweigh its potential value49. This may be particularly true for patients who remain employed45. BBM testing should not be performed if the patient does not wish to know their AD status given these risks. Additionally, testing may not be appropriate if patients or family members misinterpret the meaning of results. For example, if patients request testing to understand potential genetic or familial risk. Clinical counseling and education pre and post testing are important to avoid misinterpretation of the results. Identifying the potential risks relevant to an individual patient is critical to understanding whether the benefits of testing outweigh the risks.
A final consideration, justice, evaluates whether benefits or risks of testing could disproportionately affect sub-populations. This is particularly true if patient-level factors inadvertently serve as a discriminatory proxy for race, ethnicity, or socio-economic status. These factors could directly or indirectly increase barriers to testing and the benefits of testing to sub-populations. For example, some clinics may have limited access to DMTs, thereby reducing potential benefits of testing. Not performing BBM testing in this setting, with the rationale that treatments are not readily available, could exacerbate disparities in AD diagnosis and treatment.
This framework encourages clinicians to evaluate BBM testing through the intersecting lenses and centering patient values and goals relevant to testing. As an iterative process, the framework supports ongoing reflection to ensure that decisions remain responsive to evolving clinical circumstances and patient preferences. When such an assessment reveals that testing is unlikely to meaningfully inform care or align with what matters to the patient, it may be ethically inappropriate and constitute medical futility.
Application of the framework: case scenarios
The framework is not intended as a rigid algorithm but as a dynamic process that supports ethical deliberation. Clinicians are encouraged to iteratively consider the relevant ethical standards as they reassess decisions over time in response to changing patient needs, values, and prognoses. To support structured clinical reasoning, we synthesize these considerations into a proposed conceptual decision pathway that illustrates how empirical constraints, contextual factors, and ethical considerations interact to determine when blood-based biomarker testing is ethically justified or futile (Fig. 1). The following case scenarios illustrate its application across common primary care contexts.
This proposed conceptual framework illustrates a structured decision pathway designed to support clinicians in synthesizing empirical evidence, clinical context, and ethical considerations when evaluating the use of Alzheimer’s disease blood-based biomarkers in primary care. The pathway integrates assessment of clinical appropriateness, test interpretability, intended context of use (screening versus diagnostic confirmation), availability of confirmatory testing, implications for disease-modifying therapy eligibility and reasonable accessibility, potential risks and harms, equity considerations, and patient-centered values. Within this framework, determinations of clinical or ethical futility emerge as the outcome of sequential empirical and contextual assessments rather than as a priori value judgments. The pathway is intended as a decision-support model rather than a prescriptive algorithm and is designed to complement, not replace, clinical judgment and shared decision-making. * This includes cognitive symptoms or evaluation context consistent with current appropriate-use recommendations; patient-specific factors (such as chronic conditions, polypharmacy, age) do not impede testing interpretation. † consider another more appropriate biomarker modality (e.g., CSF, PET). Ψ Reasonable accessibility refers to the plausibility of timely and equitable access to disease-modifying therapy and required downstream care in a given clinical context. ‡ including insurance coverage, access to confirmatory testing and downstream care.
Patient one: a 72-year-old man with very mild but increasing forgetfulness over the past 18 months and multiple morbidities who wants to know whether he has AD. His stated goals are to access AD DMTs and use biomarkers for disease monitoring
Biomarker testing is ethically justifiable in a cognitively impaired patient with a strong desire to learn whether AD is the likely etiology of their symptoms. However, additional considerations warrant exploration, including whether the patient would be eligible for DMT. First, it is ethically important to evaluate the impact of the patient’s comorbidities. Co-morbidities may be a source of cognitive impairment, impede accurate biomarker testing results, or affect DMT eligibility. Therefore, co-morbidities may affect whether the BBMs would be beneficial to this patient. While not determinative, it is important to verify insurance coverage for BBM testing and treatment options. If coverage gaps exist, these should be communicated along with other risks. If any of these factors negatively affect the potential benefits of testing, it is important to reassess whether testing is appropriate.
The patient’s stated goal relevant to disease monitoring may not be sufficient to warrant testing. Despite other potential benefits associated with BBMs, existing data does not provide sufficient evidence that serial testing (e.g., annual BBM testing) offers clinical value to individual patients for monitoring purposes2,50. Additionally, in a clinical trial of an anti-amyloid antibody that used amyloid PET to monitor amyloid clearance, there were only modest correlations between individual rates of change in BBM levels and amyloid PET values51. This suggests that BBM tests cannot currently monitor anti-amyloid treatment effects for individuals. Accordingly, if the patient is found ineligible for treatment, testing may be discouraged due to misalignment with the patient’s goal. Alternative monitoring approaches should be explored.
Patient two: a frail, 91-year-old woman accompanied by her caregiver, who is concerned about her declining cognition and functional status over the past three years
The ethical justification for BBM testing in a nonagenarian with cognitive impairment depends on whether it offers meaningful benefit (e.g., diagnostic value, treatment or non-treatment implications, or personal utility). Given the high pre-test probability of AD pathology in the oldest-old, testing would mainly reinforce clinical suspicion. However, the prevalence of comorbid conditions, polypharmacy, and mixed neuropathology (e.g., vascular pathology, TDP-43) increases the likelihood multifactorial cognitive impairment. Consequently, if amyloid pathology is present, it may not be the primary cause. Relying solely on BBM results may underappreciate other contributing conditions, including treatable polypharmacy. In poor health, limited diagnostic value may be further reduced by short life expectancy, as BBM utility in this group remains unclear2.
If the goal of testing is to determine eligibility for DMTs, other eligibility factors must be considered. A 91-year-old patient is likely to have comorbidities that would be contradictions for DMT treatment. For example, multiple previous strokes would indicate that the individual is ineligible for current treatments43. A patient with significant frailty (e.g., functional dependence, weight loss, or limited physiological reserve) may be less able to tolerate the burden of treatment (e.g., regular infusions and multiple MRI scans)52. Finally, it is unknown whether treatments have similar safety and efficacy in nonagenarians.
If the BBM test is unlikely to impact diagnosis, guide further evaluation, or treatment and management plan, it may be deemed futile. In this case, testing may impose additional burdens on patients and their caregivers without benefits and therefore, would be ethically inappropriate.
Non-treatment benefits, such as life planning, must also be considered. Testing may aid advance planning, legal and financial decision-making, or caregiver preparation. However, if these have already been addressed, AD diagnosis may not change care. Depending on the patient’s health status, priorities may center on symptom management and quality of life, including emotional and social engagement. If, for example, the patient’s main goal is maintaining a positive mood and family connection, testing decisions must align accordingly.
Patient three: a 78-year-old woman with moderate dementia who lives in an assisted living facility, is seen due to worsening delusions and agitation
In a patient with more advanced dementia, determining the etiology may not change the diagnosis, testing, or the management plan. The patient would not be a candidate for DMTs, and other considerations are less likely to be relevant (e.g., advance care planning). Biomarker testing is unlikely to meet the patient’s current needs and is likely to be deemed futile. It is ethically justifiable to decline BBM testing given a lack of a clear benefit to the patient. Testing to provide information not directly valuable to the patient should be avoided. This includes testing requested by family members to infer their own risks. This indicates a significant misinterpretation of the meaning of individual biomarker results, which do not provide information on genetic risk for disease. Accordingly, BBM testing is not likely ethically justified.
Conclusion
The advancements associated with BBM testing in recent years are laudable with significant benefits that will advance clinical care of patients affected by AD. However, ethical clarity and guidance are increasingly needed to support the responsible use of BBMs as they move beyond specialist environments. This manuscript introduces an ethical framework for BBM testing. Although conceptually grounded, the framework is designed to be practical and responsive to the complexities of the primary care context. Its application to case scenarios drawn from real-world dementia practice, refined with input from interdisciplinary experts, demonstrates its relevance to common dilemmas in BBM decision-making. We also incorporated insights from emerging empirical literature on stakeholder concerns, including psychological, financial, and interpretive challenges identified by patients, caregivers, and primary care clinicians. While the framework is not yet empirically tested, it offers an actionable tool for guiding BBM use in ethically complex cases. Future work should assess its feasibility, acceptability, and outcomes through implementation studies in diverse care environments, particularly in resource-constrained and underserved settings. Anticipated implementation challenges include variability in clinician preparedness, lack of clear decision algorithms, and unequal access to follow-up testing and treatment in primary care. To address these barriers, implementation efforts should include the development of decision aids and training toolkits to support ethical, evidence-informed use of BBMs. Additionally, policy action is needed to address access and affordability concerns, ensuring that AD BBM testing and subsequent testing (e.g., PET) and treatments do not leave underserved groups behind. This framework presents an initial step toward bridging ethical gaps, supporting alignment between emerging diagnostic innovations and the complexities of everyday clinical practice.
Data availability
No datasets were generated or analysed during the current study.
References
Palmqvist, S. et al. Blood biomarkers to detect Alzheimer disease in primary care and secondary care. JAMA 332, 1245–1257 (2024).
Palmqvist, S. et al. Alzheimer’s Association Clinical Practice Guideline on the use of blood-based biomarkers in the diagnostic workup of suspected Alzheimer’s disease within specialized care settings. Alzheimers Dement. 21, e70535 (2025).
Hansson, O. Biomarkers for neurodegenerative diseases. Nat. Med. 27, 954–963 (2021).
Mielke, M. M. et al. Considerations for widespread implementation of blood-based biomarkers of Alzheimer’s disease. Alzheimers Dement. 20, 8209–8215 (2024).
Schindler, S. E. et al. Acceptable performance of blood biomarker tests of amyloid pathology — recommendations from the Global CEO Initiative on Alzheimer’s Disease. Nat. Rev. Neurol. 20, 426–439 (2024).
Jack, J. C. liffordR. et al. Revised criteria for diagnosis and staging of Alzheimer’s disease: Alzheimer’s Association Workgroup. Alzheimers Dement. 20, 5143 (2024).
Atri, A. et al. Alzheimer’s Association clinical practice guideline for the Diagnostic Evaluation, Testing, Counseling, and Disclosure of Suspected Alzheimer’s Disease and Related Disorders (DETeCD-ADRD): Executive summary of recommendations for primary care. Alzheimers Dement. 21, e14333 (2025).
“C₂N Diagnostics Releases the PrecivityAD2TM Blood Test for Clinical Care, A Robust Assay with High Concordance to Amyloid PET and CSF,” C2N Diagnostics. Accessed: Aug. 15, 2025. [Online]. Available: https://c2n.com/news-releases/cnnbspdiagnostics-releases-the-precivityad2-blood-test-for-clinical-care.
“Quest Introduces First-To-Market Consumer-Initiated Blood Test for Alzheimer’s Disease Risk Assessment on questhealth.com,” Quest Diagnostics Newsroom. Accessed: Aug. 15, 2025. [Online]. Available: https://newsroom.questdiagnostics.com/2023-07-31-Quest-Introduces-First-To-Market-Consumer-Initiated-Blood-Test-for-Alzheimers-Disease-Risk-Assessment-on-questhealth-com.
“JAMA Publishes Landmark Clinical Study Showing C2N Diagnostics’ PrecivityAD2TM Blood Test Markedly Improves Accuracy of AD Diagnosis in Both Primary Care and Specialized Memory Care,” PrecivityAD®. Accessed: Aug. 15, 2025. [Online]. Available: https://precivityad.com/news/jama-publishes-landmark-clinical-study-showing-c2n-diagnostics-precivityad2-blood-test-markedly-improves-accuracy-of-ad-diagnosis-in-both-primary-care-and-specialized-memory-care.
“Roche’s Elecsys® pTau181 becomes the only FDA-cleared blood test for use in primary care to rule out Alzheimer’s-related amyloid pathology,” Diagnostics. Accessed: Oct. 27, 2025. [Online]. Available: https://diagnostics.roche.com/us/en/news-listing/2025/fda-cleared-ptau181-alzheimers-blood-test.html.
“Alzheimer’s Association Welcomes FDA Clearance of First Blood Test for Use in Primary Care to Rule Out Alzheimer’s-Related Amyloid Pathology,” Alzheimer’s Association. Accessed: Oct. 27, 2025. [Online]. Available: https://www.alz.org/news/2025/fda-clearance-blood-test-primary-care-rule-out-alzheimers-related-amyloid-pathology.
Schindler, S. E. et al. Head-to-head comparison of leading blood tests for Alzheimer’s disease pathology. Alzheimers Dement. 20, 8074–8096 (2024).
Barthélemy, N. R. et al. Highly accurate blood test for Alzheimer’s disease is similar or superior to clinical cerebrospinal fluid tests. Nat. Med. 30, 1085–1095 (2024).
Palmqvist, S. et al. Discriminative accuracy of plasma phospho-tau217 for Alzheimer disease vs other neurodegenerative disorders. JAMA 324, 1 (2020).
“FDA Clears First Blood Test Used in Diagnosing Alzheimer’s Disease | FDA.” Accessed: Oct. 27, 2025. [Online]. Available: https://www.fda.gov/news-events/press-announcements/fda-clears-first-blood-test-used-diagnosing-alzheimers-disease?utm_source=chatgpt.com.
C. for D. and R. Health, “Laboratory Developed Tests,” FDA. Accessed: Mar. 04, 2025. [Online]. Available: https://www.fda.gov/medical-devices/in-vitro-diagnostics/laboratory-developed-tests.
Bolsewig, K. et al. Caregivers’ attitudes toward blood-based biomarker testing for Alzheimer’s disease. Alzheimers Dement. Amst. Neth. 16, e12549 (2024).
Cox, C. G. et al. Older US adults’ experiences with and views about cognitive screening and blood biomarker testing for Alzheimer’s disease. Alzheimers Dement. Amst. Neth. 17, e70067 (2025).
Bolton, C. J. et al. Disclosure of Alzheimer’s disease blood-based biomarker results in a primary care setting: opportunities and challenges. J. Prev. Alzheimers Dis. 12, 100310 (2025).
Claessen, T. et al. General practitioners’ perspectives on blood biomarkers for Alzheimer’s disease. Alzheimers Dement. Diagn. Assess. Dis. Monit. 17, e70186 (2025).
Plassman, B. L. et al. Incidence of dementia and cognitive impairment, not dementia in the United States. Ann. Neurol. 70, 418–426 (2011).
Schubert, C. C. et al. Comorbidity profile of dementia patients in primary care: are they sicker?. J. Am. Geriatr. Soc. 54, 104–109 (2006).
Kulshreshtha, A. et al. Prevalence of unrecognized cognitive impairment in federally qualified health centers. JAMA Netw. Open 7, e2440411 (2024).
Sanderson, M. et al. Co-morbidity associated with dementia. Am. J. Alzheimers Dis. Other Demen. 17, 73–78 (2002).
2022 Alzheimer’s disease facts and figures. Alzheimers Dement. 18, 700–789 (2022).
Wallace, L. M. K. et al. Investigation of frailty as a moderator of the relationship between neuropathology and dementia in Alzheimer’s disease: findings from the Rush Memory and Aging Project cohort study. Lancet Neurol. 18, 177–184 (2019).
Calvin, C. M., Conroy, M. C., Moore, S. F., Kuźma, E. & Littlejohns, T. J. Association of multimorbidity, disease clusters, and modification by genetic factors with risk of dementia. JAMA Netw. Open 5, e2232124 (2022).
Ben Hassen, C. et al. Association between age at onset of multimorbidity and incidence of dementia: 30 year follow-up in Whitehall II prospective cohort study. BMJ 376, e068005 (2022).
Ismail, Z. et al. Prevalence of depression in patients with mild cognitive impairment: a systematic review and meta-analysis. JAMA Psychiatry 74, 58–67 (2017).
Alattar, M., Harrington, J. J., Mitchell, C. M. & Sloane, P. Sleep problems in primary care: a north carolina family practice research network (NC-FP-RN) study. J. Am. Board Fam. Med. 20, 365–374 (2007).
Steffens, D. C., Wang, L., Manning, K. J. & Holzhauer, C. G. Alcohol use disorder in older adults: a review of recent literature on epidemiology, cognitive outcomes, and neuroimaging findings and treatment. Am. J. Geriatr. Psychiatry Open Sci. Educ. Pract. 1, 39–51 (2024).
Growdon, M. E., Gan, S., Yaffe, K. & Steinman, M. A. Polypharmacy among older adults with dementia compared to those without dementia in the US. J. Am. Geriatr. Soc. 69, 2464–2475 (2021).
Kopar, P. K., Visani, A., Squirrell, K. & Brown, D. E. Addressing futility: a practical approach. Crit. Care Explor. 4, e0706 (2022).
Schneiderman, L. J. Medical futility: its meaning and ethical implications. Ann. Intern. Med. 112, 949 (1990).
Kass, N. E. An ethics framework for public health. Am. J. Public Health 91, 1776–1782 (2001).
Marckmann, G., Schmidt, H., Sofaer, N. & Strech, D. Putting public health ethics into practice: a systematic framework. Front. Public Health 3, 23 (2015).
O. for H. R. Protections (OHRP), “Read the Belmont Report.” Accessed: Jul. 15, 2025. [Online]. Available: https://www.hhs.gov/ohrp/regulations-and-policy/belmont-report/read-the-belmont-report/index.html.
Kohler, J. N., Turbitt, E., Biesecker, B. B., Turbitt, E. & Biesecker, B. B. Personal utility in genomic testing: a systematic literature review. Eur. J. Hum. Genet. 25, Art. no. 6 (2017).
Maddocks, I. Futility and utility. Med. J. Aust. 204, 289–289 (2016).
Marco, C. A. & Larkin, G. L. Ethics seminars: case studies in ‘futility’—challenges for academic emergency medicine. Acad. Emerg. Med. 7, 1147–1151 (2000).
Arslan, B. et al. The impact of kidney function on Alzheimer’s disease blood biomarkers: implications for predicting amyloid-β positivity. Alzheimers Res. Ther. 17, 48 (2025).
Dobson, R. et al. Eligibility for antiamyloid treatment: preparing for disease-modifying therapies for Alzheimer’s disease. J. Neurol. Neurosurg. Psychiatry 95, 796–803 (2024).
Riggs, K. R. & Ubel, P. A. Overcoming barriers to discussing out-of-pocket costs with patients. JAMA Intern. Med. 174, 849–850 (2014).
Arias, J. J. & Karlawish, J. Confidentiality in preclinical Alzheimer disease studies: when research and medical records meet. Neurology 82, 725–729 (2014).
Arias, J. J., Tyler, A. M., Oster, B. J. & Karlawish, J. The proactive patient: long-term care insurance discrimination risks of Alzheimer’s disease biomarkers. J. Law Med. Ethics J. Am. Soc. Law Med. Ethics 46, 485–498 (2018).
Stites, S. D., Rubright, J. D. & Karlawish, J. What features of stigma do the public most commonly attribute to Alzheimer’s disease dementia? Results of a survey of the U.S. general public. Alzheimers Dement. 14, 925–932 (2018).
Largent, E. A., Stites, S. D., Harkins, K. & Karlawish, J. That would be dreadful: The ethical, legal, and social challenges of sharing your Alzheimer’s disease biomarker and genetic testing results with others. J. Law Biosci. 1 (2021).
White, B. et al. What does ‘futility’ mean? An empirical study of doctors’ perceptions. Med. J. Aust. 204, 318–318 (2016).
Brum, W. S. et al. The diurnal variation of Alzheimer’s disease plasma biomarkers: a 25h study with hourly sampling. Alzheimers Dement. 19, e079719 (2023).
Pontecorvo, M. J. et al. Association of donanemab treatment with exploratory plasma biomarkers in early symptomatic Alzheimer disease: a secondary analysis of the TRAILBLAZER-ALZ randomized clinical trial. JAMA Neurol. 79, 1250–1259 (2022).
Sadlon, A., Ott, M. & Kressig, R. W. Patients with geriatric syndromes and anti-amyloid therapies: lack of consideration? An exploratory analysis of the literature. Front. Aging Neurosci. 15, 1256968 (2023).
Acknowledgements
This work was supported by the National Institutes of Health/National Institute on Aging (U24AG082930).
Author information
Authors and Affiliations
Contributions
C.E.A.S. and J.J.A. conceptualized the manuscript. C.E.A.S. conducted the literature synthesis and drafted the initial version. J.J.A. provided critical guidance on the ethical framework and supervised the project. M.M.M., A.C.R., and S.E.S. contributed to the clinical and scientific interpretation of biomarker evidence and critically reviewed the manuscript. All authors reviewed and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
C.E.A.S. receives fellowship grant support from the Alzheimer’s Association International Research Grant Program (25AARF-1411192). A.C.R. receives funding from the National Institute of Mental Health (MH129799), National Institute on Aging (AG066515) and the Mental Illness Research, Education, and Clinical Centers/Department of Veterans Affairs. M.M.M. has served on scientific advisory boards and/or has consulted for Althira, Biogen, Cognito Therapeutics, Eisai, LabCorp, Lilly, Merck, Novo Nordisk, Roche, Siemens Healthineers; received honorariums speaking from Novo Nordisk, PeerView Institute, and Roche; and receives grant support from the NIH, Department of Defense, Alzheimer’s Association, and Davos Alzheimer’s Collaborative. S.E.S. has served scientific advisory boards on biomarker testing and clinical care pathways for Eisai and Novo Nordisk and has received speaking fees for presentations on biomarker testing from Eisai, Eli Lilly, and Novo Nordisk. J.J.A. receives funding from the NIH, Alzheimer’s Association, and the Alzheimer’s Disease Data Initiative, she serves on the Executive Committee for the National Centralized Repository for Alzheimer’s disease and Related Dementias (NCRAD) and is a member of the Medial Advisory Council for the Association for Frontotemporal Degeneration.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Scipion, C.E.A., Mielke, M.M., Rosen, A.C. et al. An ethical framework for the clinical use of Alzheimer’s disease biomarker testing. npj Dement. 2, 15 (2026). https://doi.org/10.1038/s44400-026-00060-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44400-026-00060-4