Abstract
An effective vaccine against hepatitis C virus (HCV) should elicit both humoral and cellular immune responses. Previously, we characterized a bivalent vaccine candidate against hepatitis B (HBV) and HCV using chimeric HBV-HCV virus-like particles (VLP), in which the highly conserved epitope of HCV E2 glycoprotein (residues 412–425) was inserted into the hydrophilic loop of HBV small surface antigen (sHBsAg). While sHBsAg_412–425 elicited cross-neutralizing antibodies, it did not trigger a T-cell response against HCV. Thus, this study aimed to develop a vaccine candidate engaging both arms of adaptive immune response, potentially offering stronger protection against HCV. We evaluated the immunogenicity of minicircle (MC) DNA vaccines encoding sHBsAg_412–425 and HCV nonstructural (NS) proteins in BALB/c mice. Co-administration of sHBsAg_412–425 and NS induced a potent T-cell response, especially against NS3 and high titers of antibodies specific to HCV E2. Additionally, these antibodies recognized native HCV envelope glycoprotein heterodimers (E1E2) across multiple HCV genotypes and showed binding profiles to E1E2 alanine mutants comparable to the broadly neutralizing AP33 antibody. Overall, the findings demonstrate that MC DNA vaccine incorporating both sHBsAg_412–425 and HCV NS protein sequences induces robust, T-cell and AP33-like antibody responses, highlighting its potential as pan-genotypic prophylactic vaccine against HCV.
Similar content being viewed by others
Introduction
Hepatitis C virus (HCV) continues to be a significant global health problem, contributing to an increased risk of chronic liver disease and hepatocellular carcinoma. Despite the discovery of highly effective antivirals, there are still about one million new HCV infections each year, posing a major public health challenge. Consequently, the World Health Organization (WHO) has implemented a global hepatitis strategy aiming to reduce new HCV infections by 90% and deaths by 65% by 20301. However, those goals seem unachievable, particularly as in many countries the rate of new infections continues to exceed the number of patients treated. Thus, developing an effective prophylactic vaccine is essential for the global elimination of the HCV.
Numerous HCV vaccine candidates have been evaluated for their immunogenicity in animal models; many of these have focused either on the envelope glycoproteins (E1E2) to generate broad neutralizing antibodies (bnAb) or on the nonstructural proteins (NS) that induce strong T-cell responses2. However, E1E2-based vaccine failed to induce strong bnAb in most healthy volunteers3. Additionally, recent phase I and II clinical trials using adenoviral vector-based vaccines that express only HCV NS proteins have not prevented chronic HCV infection, indicating no protective efficacy4. This outcome highlights the need for novel vaccine strategies to induce simultaneously both potent T-cell and bnAb responses.
The development of an HCV vaccine is challenged by the virus’s high genetic diversity, with eight major genotypes and over 90 subtypes that exhibit up to 30–40% differences at the nucleotide sequence level5. Therefore, targeting the highly conserved proteins or epitopes across all genotypes appears to be a promising strategy in the design of a pan-genotype HCV vaccine.
In our previous studies, we developed a bivalent vaccine candidate against hepatitis B virus (HBV) and HCV using chimeric HBV-HCV virus-like particles (VLP) where the highly conserved epitope 412–425 derived from HCV envelope glycoprotein 2 (E2) was inserted into the hydrophilic loop of the HBV small surface antigen (sHBsAg). The sHBsAg_412–425 construct was expressed in Leishmania tarentolae cells, and the resulting VLPs were capable of eliciting a cross-neutralizing antibody response against HCV6,7. Here, we propose a DNA variant of the vaccine based on minicircle (MC) technology as an antigen delivery method alternative to conventional plasmid vectors. MC, which are small DNA vectors derived from parental plasmids (PP) by eliminating bacterial backbone sequences, improve safety and enable higher levels of transgene expression8. Therefore, this study aims to examine the immunogenicity of MC-based vaccines encoding sHBsAg_412–425, HCV NS3/4A protease, and NS5B polymerase in a mouse model, analyzing combined immunization strategies to engage both arms of the adaptive immune response against HCV.
Results
MC production and characterization
DNA vaccines, encoding sHBsAg_412–425, NS3/4A and NS5B, were constructed in parental plasmid (PP) pMC.CMV-MCS-SV40polyA (Fig. 1A). Induction of MC formation was performed in ZYCY10P3S2T E. coli strain which produces integrase and endonuclease I-SceI simultaneously upon the addition of L-Arabinose to the overnight bacteria culture. The integrase joins the attB and attP sites and MC is formed, while the endonuclease I-SceI cleaves the bacterial backbone, leading to its degradation. To confirm induction of MC, purified plasmids from induced ([+] L-Arabinose) and non-induced ([−] L-Arabinose) bacteria cultures were digested with KpnI restriction enzyme. In MC, one of the two KpnI restriction sites is removed with the PP bacterial backbone, thus properly induced MC is linearized with KpnI. On the other hand, in non-induced PP a DNA fragment of 760 base pairs can be observed on the agarose gel (Fig. 1B).
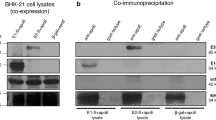
(A) Schematic diagram of the parental plasmids (PP) used in the study. DNA sequences encoding sHBsAg_412–425, NS3/4A and NS5B were cloned into pMC.CMV-MCS-SV40polyA, containing attB and attP recombination sites. KpnI restriction sites are marked with black lines. (B) Analysis of MC DNA. Lanes contain pMC.CMV-MCS-SV40polyA plasmid with NS3/4A, NS5B and sHBsAg_412–425 sequences. Plasmids were purified from bacteria cultures pre-induction ([−] L-Arabinose) and after induction ([+] L-Arabinose) and were digested using KpnI enzyme. M—Gene O’Ruler DNA ladder mix. (C) Transfection of HEK293T cells with MC. HEK 293 cells were transfected with 2 µg of NS3/4A, NS5B and sHBsAg_412–425 MCs. For the detection of NS3/4A, NS5B and sHBsAg_412–425 recombinant proteins, anti-NS3, anti-NS5 antibodies and AP33 monoclonal antibody binding 412–423 region of HCV E2 glycoprotein were used. (D) Western blot analysis of the proteins expressed in HEK293T cells after transfection with PP (parental plasmid) and MC (minicircle). Cell lysates were separated using SDS-PAGE and detected with the anti-HBsAg, anti-NS3 and anti-NS5 antibodies. The lysate from HEK293T cells was used as the negative control (NC). On the right, the results of transfection with empty MC (eMC) are shown. On the left, protein ladder, the molecular weight in kDa is given. The original blots are presented in Supplementary Fig. S1.
To confirm expression of NS3/4A, NS5B and sHBsAg_412–425 proteins in cell culture, HEK293T cells were transfected with equal amounts (2 µg) of MC and cell staining and western blot analysis of cell lysates with specific antibodies was performed (Fig. 1C,D). For all MC constructs recombinant proteins were detected at their expected molecular weights, with observed high transfection efficiency. The chimeric sHBsAg_412–425 VLP contains three N-glycosylation sites, one on sHBsAg platform and additional two derives from HCV E2 epitope 412–425. The additional bands on the first western blot with anti-sHBsAg Abs within the Fig. 1D correspond to the different N-glycosylation states of VLPs during the production process in HEK293T cells (result not shown—confirmed with the PNGase F assay).
Analysis of immune response
To evaluate the immunogenicity of the designed MC, six groups of BALB/c mice were immunized intravenously on day 0 and 28, using a polyethylenimine (PEI)-based transfection reagent. A two-dose system proved to be effective, partly due to the properties of minicircles, which exhibit a higher level of expression and persist longer in the cell due to their small size and the lack of bacterial backbone. Therefore, we lowered the second dose, to follow prime-boost regiment where boosting dose can often be reduced for safety and economic reasons. Additionally, we show that lowered second dose still significantly boosted Ab responses to 412–425 peptide (Supplementary Fig. S2).
Four weeks after the second immunization, splenocytes were isolated for IFN-γ ELISPOT and blood samples were collected to analyze the humoral responses against HCV and HBV (Fig. 2).
Mouse immunization schedule. Six groups of BALB/c mice were immunized intravenously on day 0 and 28, using a polyethylenimine (PEI)-based transfection reagent. Four weeks after the second immunization, splenocytes were isolated for IFN-γ ELISPOT and blood samples were collected to analyze the humoral responses against HCV and HBV. The figure was created in BioRender. Szewczyk, B. (2023) BioRender.com/x96o752.
For splenocyte stimulation, we used four pools of overlapping peptides spanning the entire sequences of NS3, NS4, NS5, and sHBsAg proteins. In all mouse groups, MC antigens stimulated IFN-γ response with the strongest responses observed for the NS3 antigen. Notably, in all groups immunized with NS3/4A we observed no IFN-γ response against NS4A. Furthermore, IFN-γ ELISPOT data indicated that response against carrier protein sHBsAg was significantly higher in mouse groups immunized with sHBsAg_412–425 in combination with NS3/4A or NS5B than in group immunized with sHBsAg_412–425 alone (Fig. 3).
Analysis of cellular response in BALB/c mice immunized with MC. Box plot of antigen-specific IFN-γ ELISPOT responses in MC groups. Splenocytes from six immunized BALB/c mice per group were stimulated with overlapping peptides spanning the sequences of sHBsAg, NS3, NS4 and NS5. Additionally for the mice immunized with sHBsAg_412–425 MC, sHBsAg peptides were divided into three groups (P1, P2, P3), corresponding to sHBsAg amino acid 1–99 (P1), 100–188 (P2) and 189–240 (P3). The splenocytes were collected 4 weeks after the last immunization. The box marks 25–75th percentile; the horizontal line indicates the median; whiskers extend to minimal and maximal values. Data was analyzed using two-way analysis of variance (ANOVA) (ns, P > 0.05; *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001).
To identify which regions of sHBsAg elicited the highest IFN-γ responses, we divided the sHBsAg peptide pool into three groups corresponding to amino acids 1–99 (P1), 100–188 (P2), and 189–240 (P3). The strongest T-cell responses to sHBsAg_412–425 were observed in pools P1 and P3, aligning with our previous studies6.
Regarding humoral response, we demonstrated strong and specific antibody responses against the HCV E2 epitope using a 412–425 peptide ELISA. Individual sera from mouse groups immunized with sHBsAg_412–425 alone, or in combination with NS antigens, reacted strongly to the biotinylated 412–425 peptide, with endpoint titers approaching 104 (Fig. 4A).
Analysis of antibodies elicited in mice following immunization with MC. (A) Analysis of binding of immune sera to 412–425 peptide. For analysis of binding of immune sera, streptavidin-coated microplates were coated with 5 µg/mL of biotinylated synthetic peptides covering HCV E2 epitope 412–425. Serum titer calculated as A450 value above cutoff value (three times the mean background value) is shown on axis y. Dot plot of the individual mouse serum titers against 412–425 peptide. Each dot represents a single mouse. Mean titer calculated for the group is marked with a solid line. Data were analyzed using unpaired t-test (ns, P > 0.05; *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001). (B) Analysis of immune sera binding to yeast-derived sHBsAg (yHBsAg). ELISA plates were coated with 5 µg/mL of purified sHBsAg protein derived from P. pastoris. The dilution factors of the mouse sera are shown on axis x. The mean A450 values are shown on axis y. The dashed horizontal line represents the cutoff value (three times the mean background value). Data were analyzed using two-way analysis of variance (ANOVA) Data were analyzed using two-way analysis of variance (ANOVA) (ns, P > 0.05; *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001). The data represent the results from two independent experiments performed in duplicate, and error bars indicate standard deviations. The background signal from the negative control mouse sera was subtracted from the obtained results.
Detailed serum characterization revealed that cross-reactivity with purified yeast-derived HBsAg protein (yHBsAg) was only observed in mice immunized with sHBsAg_412–425 in combination with HCV NS antigens. Surprisingly, no specific response against the carrier protein was observed in mice immunized with sHBsAg_412–425 alone (Fig. 4B).
Next, we tested the cross-reactivity of the immune sera against E1E2 heterodimers derived from HCV genotypes 1–4. All sera from groups immunized with sHBsAg_412–425 alone or in combination with NS antigens, recognized native E1E2 heterodimers derived from genotypes 1a, 1b, 2a, 3a, and 4a. Higher titers were observed for sera derived from mice immunized with a combination of MC carrying sHBsAg_412–425 and HCV NS sequences (Fig. 5).
Reactivity of immune sera to E1E2 complexes derived from different HCV genotypes. For the analysis of binding of immune sera to E1E2 heterodimers, GNA-coated ELISA plates were incubated with E1E2-containing cell lysates. Serum dilution factor is shown on axis x. The mean A450 values are shown on axis y. The background signal from the negative control mouse sera was subtracted from the obtained results. Data was analyzed using two-way analysis of variance (ANOVA) (ns, P > 0.05; *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001). The data represent the results from two independent experiments performed in duplicate, and error bars indicate standard deviations. The dashed horizontal line represents the cutoff value (three times the mean background value).
Finally, we evaluated binding profiles of antibodies elicited by sHBsAg_412–425 using a panel of E1E2 (genotype 1a H77C) mutants, where each residue across the E2 epitope located within amino acids 412–423 was individually replaced by alanine (Fig. 6). Similar to previous findings, the binding of antibodies elicited by sHBsAg_412–425 to E1E2 was abolished by alanine substitutions at L413, G418, or W420, and reduced by an alanine substitution at N4159. This result indicates that vaccination with sHBsAg_412–425 alone or in combination with HCV NS antigens, elicited antibodies with a binding profile comparable to bnAb AP33, as their binding to E1E2 is also abrogated by the same four mutations.
Reactivity of immune sera to a panel of E1E2 complex mutants in which residues across the 412–425 epitope were individually replaced by alanine. E1E2 heterodimer mutants were expressed in HEK293 cells, captured using GNA-coated ELISA plates and probed with immune sera (diluted 1:500) or AP33 mAb (diluted 1:2,500). The mean A450 values are shown on axis y. The substitution introduced to the E1E2 complex are annotated on axis x according to the amino acid occurring within the H77C sequence, and the amino acid position is relative to the H77C polyprotein chain. The data represent the results from two independent experiments performed in duplicate, and error bars indicate standard deviations.
Discussion
Although various HCV antigens have been proposed as potential vaccine candidates, no vaccine has yet been approved for human use. The E1E2 heterodimer localized on the surface of the viral particle, is the primary target for host-neutralizing antibodies and has been proposed as a promising vaccine antigen3,10,11. However, in phase I clinical trials, only a small fraction of immunized patients exhibited potent bnAb responses when vaccinated with the E1E2 heterodimer combined with the MF59 adjuvant12,13,14. Limited cross-reactivity is caused by the high genetic variability of the E1E2 heterodimer, especially in the immunodominant hypervariable regions of the E2 glycoprotein. Therefore, future vaccines should aim to preferentially elicit bnAb targeting the highly conserved occluded regions of the E1E2.
HCV NS are considered prime targets for eliciting cross-reactive T-cell responses15,16. In phase I and II clinical trials the adenoviral vector carrying HCV NS elicited strong T-cell responses, that unfortunately did not confer protection against chronic HCV infection4. Additionally, a phase I trial of a therapeutic DNA vaccine encoding HCV NS proteins demonstrated no notable antiviral effects in individuals already infected with HCV17. Thus, the data from clinical trials suggest that protective immune responses against HCV involve both bnAb against HCV envelope and potent cytotoxic responses. Recently, this have been supported by HCV-related rodent hepacivirus (RHV) study showing that synergy between antibody and T-cell responses is required for RHV clearance18.
Here we propose two-pronged approach to elicit both nAb against occluded, conserved HCV E2 epitope and potent cellular responses against HCV NS proteins. To elicit strong T-cell responses we designed DNA-based vaccines coding two HCV NS proteins—NS3 protease and its cofactor NS4A and NS5B polymerase. Both have been previously described as strong HCV antigens and contain the most potent and conserved epitopes for cell-mediated immunity19. It has been shown that the addition of the NS3 protease cofactor, NS4A, further enhances the cytotoxic response against NS3 peptides20. For humoral responses we used previously described sHBsAg_412–425 sequence containing one of the most important and conserved E2 epitopes—epitope I, located within amino acids 412–423 of the E2 glycoprotein which have been shown to be targeted by bnAb such as AP33 and HCV121,22. However, the immunogenicity of the 412–423 epitope in natural infection, has been reported to be low23. Therefore, efficient presentation of the epitope I to the immune system should be an important goal in HCV vaccine design. We have previously shown that exposing of amino acids 412–425 on the sHBsAg-based VLP elicited high levels of bnAb against HCV, thus making sHBsAg_412–425 VLP a viable candidate for an effective HCV vaccine7,9.
DNA-based vaccines have demonstrated safety and the ability to elicit both humoral and cellular immune responses against various infectious diseases, including HCV24,25. Recent studies using DNA encoding the HCV E1E2 glycoprotein complex or NS proteins have highlighted the potential of these candidates in human trials26,27. Although the first generation of DNA vaccines showed lower immunogenicity in humans and larger animals compared to mice, recent advancements have significantly enhanced their efficiency24. Recently, MC DNA has gained attention as a promising gene delivery method that offers better biosafety and higher expression rates due to its small size and lack of bacterial backbone28,29. MC technology have been employed in various applications and have shown promising results providing new insights into gene therapy and vaccine design30,31,32,33. Here, we show that combined MC immunization of mice with sHBsAg_412–425 and either NS3/4A protease or NS5B polymerase induced a potent and specific T-cell response against both HBV sHBsAg and HCV NS proteins. Notably, the IFN-γ response against sHBsAg was significantly higher in combined groups than in mice immunized by sHBsAg_412–425 alone as well as previously reported immunization with protein variant of the sHBsAg_412–425 VLP vaccine6. Regarding humoral response, we observed a strong and specific antibody response against 412–425 HCV epitope with endpoint titers approaching 104 in groups immunized with sHBsAg_412–425. Furthermore, we also confirmed that vaccine consists of sHBsAg_412–425 induces AP33-like antibodies recognizing not only 412–425 peptide, but also native E1E2 HCV glycoprotein heterodimers derived from genotypes 1a, 1b, 3a and 4a. This suggests that antibodies elicited by sHBsAg_412–425 MC could show similar cell culture HCV (HCVcc) neutralizing activity, as we have previously demonstrated for sHBsAg_412–425 VLP-based antigen7,9. In the related study, Yato group used Japanese encephalitis virus (JEV) subviral particles to display E2 epitope I (aa412–423) and investigated whether a DNA construct could induce functional antibodies compared to its protein variant. Although the study did not target T-cell responses, it successfully produced specific anti-E2 antibodies, as measured by a 412–423 peptide ELISA, with endpoint titers comparable to those observed in our study. However, direct comparisons are challenging due to differences in immunization schedules, antigen delivery, and doses. Furthermore, chimeric DNA constructs that elicited higher titers of anti-HCV epitope antibodies were capable of neutralizing HCV but not JEV particles34. Interestingly, here, an antibody response against the carrier protein was observed only after combined DNA immunization with sHBsAg_412–425 and NS antigens, suggesting that the addition of NS antigens to the vaccine formulation might enhance the humoral response against the carrier protein by increasing overall immunogenicity through innate, non-specific mechanisms (e.g. inflammation) or by specifically modulating the antigenicity of the sHBsAg. This is further supported by increased IFN-γ responses and E1E2 heterodimers binding in the groups immunized with a combination of sHBsAg_412–425 and HCV NS. Co-administration of HCV antigens with genetic adjuvants has been shown to enhance or prolong their immunogenicity25. Recent strategies using conventional plasmid-based DNA vaccines coding for HCV sequences have reported that addition of IL-28B or truncated mouse perforin sequence to the vaccine formulation enhance immunological response against HCV antigens17,26,35. Another approach utilized DNA vaccine encoding secreted forms of HCV envelope glycoproteins with oligomerization domain of the C4b-binding protein was found to be most effective in eliciting both humoral and cellular immunity against HCV. The end-point titers for anti-E1E2 specific antibodies reached 103 and were higher in groups of mice immunized with DNA constructs encoding oligomerization domain27.
In our previous study, sHBsAg_412–425 VLP, a recombinant protein variant of the vaccine, elicited strong antibody response against sHBsAg carrier6. In context to the bivalent HBV/HCV vaccine, our study suggests that for MC setting, combined immunizations with NS antigens are required to elicit anti-sHBsAg antibodies. Alternatively, a boost with recombinant VLP sHBsAg_412–425, would be beneficial to enhance antibody titer against sHBsAg.
To conclude, this study represents the first attempt to utilize MC as an HCV vaccine candidate. Our findings suggest that MCs are a promising tool for HCV vaccine design providing an easy platform to combine multiple antigens into one vaccine formulation. The landscape of HCV vaccine research is dynamic, with ongoing efforts to optimize vaccine design and enhance immune responses. In fact, vaccine based on only one epitope has some limitations due to conformational flexibility of 412–423 epitope. However, sHBsAg_412–425 construct can be used as an additional vaccine component to enhance pan-genotypic antibody response in the next approaches.
Methods
DNA plasmid construction
The sequences coding for HCV proteins NS3/4A and (nucleotides (nt) 3411–5465) and NS5B (nt 7590–9365 [del 8536–8544—GDD motif]) were obtained from HCV genotype 1B (GenBank: M58335), construct sHBsAg_412–425 has been described before6. The region of HCV E2 glycoprotein, spanning the 412–425 residues (isolate H77c, GenBank accession no. AF011751) was inserted within HBV subtype adw2 sHBsAg (GenBank accession no. AF397207.1). All sequences were synthetized using H. sapiens-adapted codons (GenArt, Thermo Scientific) and cloned into pMC.CMV-MCS-SV40polyA (System Biosciences) using XbaI and NheI cloning sites. Prepared plasmids were used to transform ZYCY10P3S2T cells (System Biosciences).
Generation and purification of MC
MC were generated using MC-Easy™ Minicircle DNA production kit (System Biosciences), following manufacturer’s guidelines. Briefly, 150 mL of an overnight bacterial culture (OD600 6–8, pH = approx. 7), cultivated in growth media (System Biosciences), was supplemented with 150 mL of L-Arabinose-containing induction media (System Biosciences). Subsequently, bacterial cultures were incubated at 30 °C for 3 h, followed by an additional 1-h incubation at 37 °C. The resultant bacterial cultures were harvested through centrifugation and subsequently frozen.
To extract MC from the bacterial pellet, purification was executed using the EndoFree Plasmid Maxi Kit (Qiagen). For the assessment of MC production, the isolated MC were treated with KpnI restriction enzyme (Thermo Scientific). As a control, plasmids were purified from bacterial cultures cultivated without L-Arabinose.
Transfection of HEK293T cells
HEK293T cells were cultured in a 6-well plate in Dulbecco modified Eagle’s medium (DMEM, Thermo Fisher Scientific) supplemented with 1% penicillin–streptomycin (Thermo Fisher Scientific) and 10% fetal bovine serum (Thermo Fisher Scientific) at 37 °C in 5% CO2. Cells were transfected with 2 µg of NS3/4A, NS5B and sHBsAg_412–425 MC and incubated at 37 °C for 48 h. For immunostaining, cells were rinsed with PBS, fixed with a 4% paraformaldehyde and permeabilized with 0.02% Triton X-100 in PBS. For the detection of NS3/4A and NS5B recombinant proteins, anti-NS3 and anti-NS5 antibodies (Santa Cruz Biotechnology; cat no. sc-69938 and sc-58146 respectively) were used (dilution 1:200). For the detection of sHBsAg_412–425 recombinant protein, AP33 mAb binding 412–423 region of HCV E2 glycoprotein was used in dilution 1:500. For detection, secondary goat anti-mouse antibodies conjugated with HRP were used (dilution 1:1,000). Immunostaining was developed using NovaRED substrate (Vector Laboratories).
SDS-PAGE and western blot
Analysis of the protein expression was carried out by SDS-PAGE using 4–12% gradient Bis–Tris gels in MES or MOPS SDS running buffer. After electrophoresis, proteins were transferred onto PVDF membrane using electroblotting, and subsequently the membranes were blocked overnight at 4 °C with 3% nonfat milk in TBST (TBS buffer, 0.1% (v/v) Tween-20). Following blocking, membranes were incubated for 1 h at RT with primary anti-HBsAg (dilution 1:1,000) (Origene; cat no. BP2029P), anti-NS3 (dilution 1:500) and anti-NS5 (dilution 1:200) (Santa Cruz Biotechnology; cat no. sc-69938 and sc-58146 respectively) antibodies diluted in nonfat milk in TBST, washed with TBST, and then incubated with goat anti-rabbit or goat anti-mouse antibodies conjugated with HRP (dilution 1:1,000) (Santa Cruz Biotechnology). The results were obtained through development in the substrate for chemiluminescence (Thermo Scientific). The original blots are presented in Supplementary Fig. S1.
Immunization of mice
Groups of 6 female BALB/c mice (Tri-City University Animal House—Research Service Centre at the Medical University of Gdańsk, n = 6 per group), 6–8 weeks of age, were immunized intravenously (i.v.) with two doses on day 0 and 28 with polyethylenimine (PEI)-based transfection reagent (in vivo-jetPEI, Polyplus) in the presence of 5% glucose. For NS3/4A 10 μg/dose, for NS5B 10 μg/dose 1 and 5 μg/dose 2, for sHBsAg_412–425 20 μg/dose 1 and 10 μg/dose 2 were used. Four weeks after the second immunization, animals were subjected to general anesthesia using isoflurane and total blood and spleens were collected. All experiments on animals were conducted by certified animal facility at the Medical University of Gdańsk (Tri-City Academic Laboratory Animal Centre, Medical University of Gdańsk), in accordance with the ARRIVE-10 questionnaire and national Polish guidelines for animal experimentation. The protocols were approved by the Local Committee on the Ethics of Animal Experiments of the University of Science and Technology in Bydgoszcz (Permit Number: 8/2021).
ELISPOT assay
T-cell response was analyzed by ELISPOT assay for detection of IFN-γ according to the manufacturer’s instructions (BD Biosciences). Briefly, ELISPOT 96-well plates were coated with purified anti-mouse IFN-γ antibody and blocked using RPMI Medium 1640 (Thermo Fisher Scientific) supplemented with 10% fetal bovine serum, 100 μg/mL penicillin, and 2 mM L-glutamine (Thermo Fisher Scientific). Splenocytes were isolated from the mice 28 days after last immunization by passage through 70 μm strainers followed by treatment with ACK lysing buffer (Thermo Fisher Scientific). After washing, splenocytes were seeded at 5 × 105 cells/well and stimulated overnight at 37 °C with peptides. Peptide pools for NS3, NS4 and NS5 were derived from HCV genotype 1B (Miltenyi Biotec). Peptides for sHBsAg were pooled into 3 groups spanning the sHBsAg amino acid sequence: P1—aa 1–99, P2—aa 100–188 and P3—aa 189–240 (JPT Peptide Technologies). All peptides were 15-mer sequences with 11 amino acids overlap and were used at the concentration of 20 μg/mL. Concanavalin A was used as positive control. The mean number of spots from duplicate wells were calculated for each animal and adjusted to represent the mean number of spots per 106 splenocytes.
Characterization of antibody responses
The antibody response against HCV E2 412–425 epitope was measured by direct solid-phase ELISA. Preblocked streptavidin-coated plates (Thermo Fisher Scientific) were incubated with biotinylated 412–425 peptide at 5 µg/mL (GenScript) for 16 h at 4 °C. After coating and washing, serially diluted mouse sera were added to the wells and incubated for 1 h at RT. Goat anti-mouse secondary HRP-conjugated antibodies (Santa Cruz Biotechnology) and TMB substrate were used for detection. Similarly, the antibody response against sHBsAg protein was evaluated using ELISA plates coated with P. pastoris-derived sHBsAg protein (yHBsAg) at 5 µg/mL (OriGene). The plates were then blocked for 2 h with 3% (w/v) BSA in PBST (PBS buffer, 0.05% (v/v) Tween-20), and diluted mouse sera were added to the wells as above.
To test sera reactivity to E1E2 complexes, HEK293T cells were transfected with plasmids expressing wild-type E1E2 glycoproteins derived from genotypes: 1a(H77C); 1b(12.6); 2a(2.4); 2b(1.1); 3a(1.9); 4a(21.16). For the evaluation of sera binding to E1E2 alanine-scanning mutants, plasmids encoding alanine substitution mutants of genotype 1a strain H77C E1E2 were used to transfect HEK293 cells. Seventy-two hours after transfection, the cells were washed with PBS buffer and lysed in lysis buffer (PBS buffer, 0.5% Triton X-100). The clarified cell lysates were normalized against each other using mouse ALP98 monoclonal antibody. Next, the cell lysates were transferred to ELISA plates precoated with G. nivalis lectin (GNA) and incubated for 16 h at 4 °C. Next, the plates were blocked in PBST with 3% BSA and either used immediately or stored at − 20 °C. The pooled mouse sera were diluted in PBST containing 0.3% BSA. Finally, the binding of the antibodies to the recombinant proteins was detected by goat anti-mouse HRP-conjugated secondary antibodies diluted to 1:2500 (Santa Cruz Biotechnology) and TMB substrate.
Statistical analysis
All the graphs, calculations, and statistical analyses were performed using GraphPad Prism software version 7.05 (GraphPad Software, USA).
Data availability
All data generated or analyzed during this study are included in this article.
References
Global hepatitis report 2024: action for access in low- and middle-income countries. 242 https://www.who.int/publications/i/item/9789240091672 (2024).
Hartlage, A. S. & Kapoor, A. Hepatitis C virus vaccine research: time to put up or shut up. Viruses 13, 1596 (2021).
Bailey, J. R., Barnes, E. & Cox, A. L. Approaches, progress, and challenges to hepatitis C vaccine development. Gastroenterology 156, 418–430 (2019).
Page, K. et al. Randomized trial of a vaccine regimen to prevent chronic HCV infection. N. Engl. J. Med. 384, 541–549 (2021).
Bukh, J. The history of hepatitis C virus (HCV): Basic research reveals unique features in phylogeny, evolution and the viral life cycle with new perspectives for epidemic control. J. Hepatol. 65, S2–S21 (2016).
Czarnota, A. et al. Immunogenicity of Leishmania-derived hepatitis B small surface antigen particles exposing highly conserved E2 epitope of hepatitis C virus. Microb. Cell Fact. 15, 62 (2016).
Czarnota, A. et al. Specific antibodies induced by immunization with hepatitis B virus-like particles carrying hepatitis C virus envelope glycoprotein 2 epitopes show differential neutralization efficiency. Vaccines (Basel) 8, 294 (2020).
Almeida, A. M., Queiroz, J. A., Sousa, F. & Sousa, Â. Minicircle DNA: the future for DNA-based vectors?. Trends Biotechnol. 38, 1047–1051 (2020).
Czarnota, A. et al. Effect of glycan shift on antibodies against hepatitis C virus E2 412–425 epitope elicited by chimeric sHBsAg-based virus-like particles. Microbiol. Spectr. 11 (2023).
Gomez-Escobar, E., Roingeard, P. & Beaumont, E. Current hepatitis C vaccine candidates based on the induction of neutralizing antibodies. Viruses 15, 1151 (2023).
Offersgaard, A., Bukh, J. & Gottwein, J. M. Toward a vaccine against hepatitis C virus. Science 1979(380), 37–38 (2023).
Frey, S. E. et al. Safety and immunogenicity of HCV E1E2 vaccine adjuvanted with MF59 administered to healthy adults. Vaccine 28, 6367–6373 (2010).
Chen, F. et al. Antibody responses to immunization with HCV envelope glycoproteins as a baseline for B-cell-based vaccine development. Gastroenterology 158, 1058-1071.e6 (2020).
Law, J. L. M. et al. A hepatitis C virus (HCV) vaccine comprising envelope glycoproteins gpE1/gpE2 derived from a single isolate elicits broad cross-genotype neutralizing antibodies in humans. PLoS One 8, e59776 (2013).
Thimme, R. T cell immunity to hepatitis C virus: Lessons for a prophylactic vaccine. J. Hepatol. 74, 220–229 (2021).
Shoukry, N. H. Hepatitis C vaccines, antibodies, and T cells. Front. Immunol. 9 (2018).
Jacobson, J. M. et al. Phase I trial of a therapeutic DNA vaccine for preventing hepatocellular carcinoma from chronic hepatitis C virus (HCV) infection. Cancer Prev. Res. 16, 163–173 (2023).
Gridley, J. et al. Concerted synergy between viral-specific IgG and CD8+ T cells is critical for clearance of an HCV-related rodent hepacivirus. Hepatology https://doi.org/10.1097/HEP.0000000000000753 (2024).
Gededzha, M. P., Mphahlele, M. J. & Selabe, S. G. Prediction of T-cell epitopes of hepatitis C virus genotype 5a. Virol. J. 11, 187 (2014).
Frelin, L. et al. Low dose and gene gun immunization with a hepatitis C virus nonstructural (NS) 3 DNA-based vaccine containing NS4A inhibit NS3/4A-expressing tumors in vivo. Gene Ther. 10, 686–699 (2003).
Broering, T. J. et al. Identification and characterization of broadly neutralizing human monoclonal antibodies directed against the E2 envelope glycoprotein of hepatitis C virus. J. Virol. 83, 12473–12482 (2009).
Owsianka, A. M. et al. Broadly neutralizing human monoclonal antibodies to the hepatitis C virus E2 glycoprotein. J. Gen. Virol. 89, 653–659 (2008).
Tarr, A. W. et al. Determination of the human antibody response to the epitope defined by the hepatitis C virus-neutralizing monoclonal antibody AP33. J. Gen. Virol. 88, 2991–3001 (2007).
Kutzler, M. A. & Weiner, D. B. DNA vaccines: ready for prime time?. Nat. Rev. Genet. 9, 776–788 (2008).
Shayeghpour, A. et al. Hepatitis C virus DNA vaccines: a systematic review. Virol. J. 18, 248 (2021).
Gummow, J. et al. Safety profile of a multi-antigenic DNA vaccine against hepatitis C virus. Vaccines (Basel) 8, 53 (2020).
Masavuli, M. G. et al. A hepatitis C virus DNA vaccine encoding a secreted, oligomerized form of envelope proteins is highly immunogenic and elicits neutralizing antibodies in vaccinated mice. Front. Immunol. 10 (2019).
Shafaati, M. et al. A brief review on DNA vaccines in the era of COVID-19. Future Virol. 17, 49–66 (2022).
Arévalo-Soliz, L. M. et al. Improving therapeutic potential of non-viral minimized DNA vectors. Cell Gene Ther. Insights 6, 1489–1505 (2020).
Rim, Y. A., Nam, Y., Park, N. & Ju, J. H. Minicircles for investigating and treating arthritic diseases. Pharmaceutics 13, 736 (2021).
Ventura, C. et al. Maximization of the minicircle DNA vaccine production expressing SARS-CoV-2 RBD. Biomedicines 10, 990 (2022).
Shaabani, E. et al. Gene therapy to enhance angiogenesis in chronic wounds. Mol. Ther. Nucleic Acids 29, 871–899 (2022).
Eusébio, D. et al. The performance of minicircle DNA versus parental plasmid in p53 gene delivery into HPV-18-infected cervical cancer cells. Nucleic Acid Ther. 31, 82–91 (2021).
Yato, K. et al. Induction of neutralizing antibodies against hepatitis C virus by a subviral particle-based DNA vaccine. Antiviral Res. 199, 105266 (2022).
Lee, H. et al. Preclinical evaluation of multi antigenic HCV DNA vaccine for the prevention of hepatitis C virus infection. Sci. Rep. 7, 43531 (2017).
Acknowledgements
We would like to thank Arvind Patel for the kind gift of alanine E1E2 mutant plasmids and AP33 mAb. We are also grateful to Grażyna Peszyńska-Sularz, Anna Żyłko, Klaudia Konieczna-Wolska, Magdalena Węsierska (Tri-City Academic Laboratory Animal Centre, Medical University of Gdańsk) for animal care and Alicja Chmielewska and Maksymilian Chmielewski for experimental support. This study was supported by The National Science Centre, Poland grant no. 2017/27/B/NZ6/01403 (K.G.).
Author information
Authors and Affiliations
Contributions
Conceptualization: A.C. and K.G.; Methodology: A.C. and K.G.; Validation: A.C. and K.G.; Formal analysis: A.C. and K.G.; Investigation: A.C., A.R., A.S., K.G.; Resources: K.B.S.; Data curation: A.C. and K.G.; Writing—original draft preparation: A.C. and K.G.; Writing—review and editing: A.C., A.S. and K.G.; Supervision: K.G.; Project administration: K.G.; Funding acquisition: K.G.; All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Czarnota, A., Raszplewicz, A., Sławińska, A. et al. Minicircle-based vaccine induces potent T-cell and antibody responses against hepatitis C virus. Sci Rep 14, 26698 (2024). https://doi.org/10.1038/s41598-024-78049-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-78049-3