Abstract
Actinobacteria from the unique intertidal ecosystem of the mangroves are known to produce novel, bioactive secondary metabolites. A novel strain known as MUSC 136T (=DSM 100712T = MCCC 1K01246T) which was isolated from Malaysian mangrove forest soil has proven to be no exception. Assessed by a polyphasic approach, its taxonomy showed a range of phylogenetic and chemotaxonomic properties consistent with the genus of Streptomyces. Phylogenetically, highest similarity was to Streptomyces misionensis NBRC 13063T (99.6%) along with two other strains (>98.9% sequence similarities). The DNA–DNA relatedness between MUSC 136T and these type strains ranged from 22.7 ± 0.5% to 46.5 ± 0.2%. Overall, polyphasic approach studies indicated this strain represents a novel species, for which the name Streptomyces malaysiense sp. nov. is proposed. The potential bioactivities of this strain were explored by means of antioxidant and cytotoxic assays. Intriguingly, MUSC 136T exhibited strong antioxidative activities as evaluated by a panel of antioxidant assays. It was also found to possess high cytotoxic effect against HCT-116 cells, which probably mediated through altering p53 protein and intracellular glutathione levels. Chemical analysis of the extract using GC-MS further affirms that the strain produces chemopreventive related metabolites.
Similar content being viewed by others
Introduction
There is an enormous need for novel chemotherapeutic agents in the ongoing battle against cancer, the global burden of which is steadily increasing1,2. Natural compounds are fast gaining interest as a potential source of new cancer treatments; it may serve as an alternative to overcome some of the current problems faced including debilitating side effects caused by drugs lacking in specificity3,4,5,6. Microorganisms are a prolific source of structurally diverse bioactive metabolites; some of which have been approved to be used as cancer chemotherapeutic agents7,8. Among microbes, the genus Streptomyces has garnered much interest from the scientific community due to its unrivaled capacity for the production of bioactive metabolites7. The genus Streptomyces was initially proposed by Waksman and Henrici9 in 1943; around the same time as the discovery of actinomycin from Actinomyces antibioticus (now Streptomyces antibioticus)8. Since the recognition of actinomycin D as an anticancer agent, many anticancer compounds have been isolated from Streptomyces species, including anthracyclines, bleomycin and mitosanes7,8.
In recent years, there has been increasing research investigating the biosynthetic potential of Streptomyces from underexplored areas, including mangrove forests10,11,12. The mangrove-derived microbial resources could potentially be an important source of compounds with pharmaceutical value, given that the changes in salinity and tidal gradient in the mangrove ecosystem may have resulted in the evolution of metabolic pathway producing valuable metabolites11,12,13. Numerous studies have discovered novel Streptomyces from mangrove environments, as demonstrated by the recent isolation of Streptomyces sanyensis14, Streptomyces pluripotens15, Streptomyces gilvigriseus16 and Streptomyces mangrovisoli17.
In this study, soil samples from the Tanjung Lumpur mangrove forest located in the east coast of Peninsular Malaysia was screened to identify novel strains exhibiting cytotoxic and antioxidant activities. Polyphasic approach revealed that MUSC 136T strain represents a novel species of Streptomyces genus, for which the name Streptomyces malaysiense sp. nov. is proposed. The extract from this novel strain was demonstrated to have cytotoxic and antioxidant activities which was further characterized using gas chromatography-mass spectrometry (GC-MS). The findings of this research are anticipated to provide a strong basis for further in-depth molecular studies on the chemopreventive properties of the novel strain MUSC 136T.
Results
Phenotypic analyses of strain Streptomyces malaysiense MUSC 136T
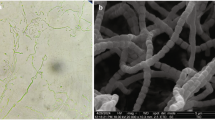
Strain MUSC 136T grew well on ISP 2, ISP 5, ISP 6, ISP 7 agar, actinomycetes isolation agar, nutrient agar and starch casein agar after 7–14 days at 28 °C, moderately on Streptomyces agar and ISP 3 agar, and not at all on ISP 4 agar. The colors of the aerial and substrate mycelium were media-dependent (See Supplementary Table S1). The morphological observation of a 15-day-old culture grown on ISP 2 agar revealed abundant growth of both aerial and vegetative hyphae, which were well developed and not fragmented. These morphological features are consistent with assignment of the strain to the genus Streptomyces18 (Fig. 1, Table 1). Growth was found to occur at pH 6.0–7.0 (optimum pH 7.0), with 0–6% NaCl tolerance (optimum 0–2%) and at 26–40 °C (optimum 28–32 °C). Cells were found to be positive for catalase but negative for hemolytic activity and melanoid pigment production. Hydrolysis of soluble starch, tributyrin (lipase) and casein were found to be positive, but negative for hydrolysis of carboxymethylcellulose, chitin and xylan. Strain MUSC 136T can be differentiated from closely related members of the genus Streptomyces using a range of phenotypic properties (Table 1). In chemical sensitivity assays, cells are resistant to aztreonam, fusidic acid, guanine HCl, lincomycin, lithium chloride, minocycline, nalidixic acid, niaproof 4, potassium tellurite, rifamycin RV, sodium bromate, sodium butyrate, 1% sodium lactate, tetrazolium blue, tetrazolium violet, troleandomycin and vancomycin.
Phylogenetic and genomic analyses
The nearly complete 16S rRNA gene sequence was obtained for strain MUSC 136T (1489 bp; GenBank/EMBL/DDBJ accession number KJ632663) and phylogenetic trees were reconstructed to determine the phylogenetic position of this strain (Fig. 2 & See Supplementary Fig. S1). Phylogenetic analysis revealed that strain MUSC 136T is closely related to Streptomyces phaeoluteichromatogenes NRRL 5799T (99.6% sequence similarity) and Streptomyces misionensis NBRC 13063T (99.6%), as they formed a distinct clade (Fig. 2). The 16S rRNA gene sequence analysis of strain MUSC 136T showed the highest similarity to that of Streptomyces misionensis NBRC 13063T (99.6%), followed by Streptomyces phaeoluteichromatogenes NRRL 5799T (99.6%) and Streptomyces rutgersensis NBRC 12819T (98.9%); sequences similarities of less than 98.9% were obtained with the type strains of other species of the genus Streptomyces. The DNA–DNA relatedness values between strain MUSC 136T and Streptomyces misionensis NBRC 13063T (46.5 ± 0.2%), S. phaeoluteichromatogenes DSM 41898T (44.5 ± 0.4%) and Streptomyces rutgersensis NBRC 12819T (22.7 ± 0.5%) were significantly below 70%, the threshold value for the delineation of bacterial species19.
Numbers at nodes indicate percentages of 1000 bootstrap re-samplings, only values above 50% are shown. Bar, 0.005 substitutions per site. Asterisks indicate that the corresponding nodes were also recovered using the maximum-likelihood tree-making algorithm. Streptacidiphilus albus NBRC 100918T was used as an outgroup.
The BOX-PCR results indicated that strain MUSC 136T yielded a unique BOX-PCR fingerprint compared with the closely related type strains (See Supplementary Fig. S2). These results are in agreement with results of DNA-DNA hybridizations, which indicate that strain MUSC 136T represents a novel species.
Chemotaxonomic analyses
The fatty acid profile of strain MUSC 136T and closely related type strains are shown in Table 2. The major cellular fatty acids in MUSC 136T were identified as anteiso-C15: 0 (35.3%), iso-C16: 0 (12.4%), iso-C15: 0 (12.2%) and anteiso-C17: 0 (11.8%). The fatty acid profile of MUSC 136T is consistent with those of closely related phylogenetic neighbours such as S. misionensis NBRC 13063T, S. phaeoluteichromatogenes DSM 41898T and S. rutgersensis NBRC 12819T, which contain anteiso-C15: 0 (32.2–40.1%), iso-C16: 0 (10.3–17.7%), iso-C15: 0 (5.2–12.3%) and anteiso-C17: 0 (11.8–19.3%) as their major fatty acids (Table 2). Nevertheless, the fatty acid profile of MUSC 136T was quantitatively different from those of these type strains; for example, although anteiso-C15: 0 (35.3%) was found to be predominant in strain MUSC 136T, the amount of anteiso-C15: 0 was much more (40.1%) in S. misionensis NBRC 13063T (Table 2). The polar lipid analysis showed the presence of aminolipid, diphosphatidylglycerol, glycolipid, phospholipid, phosphatidylinositol, phosphatidylethanolamine, phosphoglycolipid and lipid. Differences in polar lipid profiles indicated that MUSC 136T is different from related type strains; for instance, type strain Streptomyces misionensis NBRC 13063T (See Supplementary Fig. S3b) was found to contain phosphatidylglycerol, lipids that were not detected in MUSC 136T (See Supplementary Fig. S3a).
The cell wall of strain MUSC 136T is of cell-wall type I as it contains LL-diaminopimelic20. The presence of LL-diaminopimelic has been observed in many other species of the genus Streptomyces15,16,17. The predominant menaquinones of strain MUSC 136T were identified as MK-9(H8) (57%) and MK-9(H6) (20%). The detection of these predominant menaquinones is in agreement with report of Kim et al.21. The cell wall sugars detected were glucose and ribose. The G+C content of strain MUSC 136T was determined to be 72.3 mol%; which is within the range of 67.0–78.0 mol% described for species of the genus Streptomyces22.
Antioxidant activity of MUSC 136T extract
The results show that MUSC 136T extract demonstrated significant free radical scavenging activity (Table 3). At the highest tested concentration (2 mg/mL), the extract was able to scavenge 27.24 ± 1.91% of DPPH radicals and 27.87 ± 2.19% ABTS radicals. MUSC 136T exhibited superoxide dismutase (SOD)-like activity as high as 68.27 ± 3.67% when tested at 2 mg/mL. Furthermore, the metal chelating activity (which ranged from 10.22–37.01% depending on concentration of extract) exhibited by MUSC 136T further emphasizes its antioxidative potential by means of preventing transition metals from enhancing the generation of reactive oxygen species.
Cytotoxic activity of MUSC 136T extract
The cytotoxic potential of MUSC 136T extract was tested against several human derived cancer cells (HCT-116, HT-29, Ca Ski and A549). The extract was found to be most toxic against HCT-116 with cell viability at 48.8 ± 4.1% when tested at 400 μg/mL (Fig. 3a). Additionally, a dose dependent effect was also observed when it was tested against HCT-116 cells. Second to HCT-116 cells, the cervical cancer cell line, Ca Ski showed decreased in cell viability to 55.6 ± 1.2% after treatment with MUSC 136T extract (Fig. 3b). On the other hand, lung cancer cell line, A549 was found to be least sensitive to the extract treatment with cell viability at 67.1 ± 0.6% (Fig. 3c). Interestingly, differing levels of activity were seen among the colon cancer cells (HCT-116, Fig. 3a and HT-29, Fig. 3d) with a higher cytotoxic effect against HCT-116 cells.
The measurement of cell viability was done using MTT assay. The graphs show cytotoxic effect of MUSC 136T extract against (a) HCT-116, (b) Ca Ski, (c) A549, (d) HT-29. All data are expressed as mean ± standard deviation (n = 4) and analyzed using one-way analysis of variance (ANOVA). A difference was considered statistically significant when p ≤ 0.05. (e) Morphological studies show that after treatment with the extract, cells shrunk to smaller rounder cells and detached from the surface.
Morphological changes associated with apoptosis induced by MUSC 136T extract
In order to visualize morphological changes in response to MUSC 136T extract, HCT-116 cells were observed using phase contrast microscopy. Figure 3(e) shows features of control (untreated) cells which mostly consists of elongated attached cells. In contrast, cells treated with MUSC 136T extract exhibited morphological alterations, including shrunken cell and detached from the surface.
Measurement of intracellular glutathione (GSH) level and p53 protein expression following MUSC 136T treatment on HCT-116 cells
MUSC 136T extract has caused a significant increase in number of cells experiencing in intracellular GSH depletion as seen in Fig. 4(a). The treatment of extract at the concentration of 400 μg/mL resulted in higher proportion of cells (44.11 ± 6.21%) undergoing GSH depletion as compared to control.
The p53 protein in HCT-116 cells was measured with flow cytometry at four time points after treated with MUSC 136T extract (1.5, 3, 12, 24 hour); a significant increase was observed at 3 and 24 hour as compared to control (Fig. 4(b)). Overall, the p53 expression was found to be increasing 3 hour after MUSC 136T extract treatment up to 12 hour as no significant changes was observed later when measured at 24 hour.
GC-MC analysis of MUSC 136T extract
GC-MS analysis revealed nine constituents of MUSC 136T (Table 4) with the chemical structures (See Supplementary Fig. S4) Isomeric dihydro-methyl-furanone (1), 1-Pentadecene (2), Phenol, 2,5-bis (1,1-dimethylethyl)- (3), (3R,8aS)-3-methyl-1,2,3,4,6,7,8,8a-octahydropyrrolo[1,2-a]pyrazine-1,4-dione (4), 1,4-diaza-2,5-dioxobicyclo[4.3.0]nonane (5), Tetradecanoic acid, 12-methyl-, methyl ester (6), Pyrrolo[1,2-a]pyrazine-1,4-dione, hexahydro-3-(2-methylpropyl)- (7), Pentadecanoic acid, 14-methyl-, methyl ester (8) and Deferoxamine (9).
Discussion
Cancer initiation and progression has been linked to oxidative stress where elevated amounts of free radicals are observed22. The accumulation or uncontrolled production of free radicals causes negative effects on critical cellular macromolecules such as membrane lipids, proteins and DNA. Free radical induced DNA mutations are known to increase cancer risk. Antioxidants play an important role in preventing the deleterious effects of free radicals in biological systems. In fact, they are also considered as one of the most promising cytotoxic agents against various human cancers. Continuous efforts have been directed at the search for more effective antioxidants from natural resources which could be developed into new therapeutic drugs23. Streptomyces species are prolific producers of bioactive metabolites with various biological activities, representing one of the most extensively studied microbial genus for novel drugs9,15,17. In the current study, the polyphasic approach revealed that strain MUSC 136T merits assignment to a novel species in the genus Streptomyces. Thus, an attempt was made to explore the antioxidant potential of this novel strain utilizing DPPH, ABTS, metal chelating and superoxide anion scavenging assays. Our results revealed significant antioxidant potential of MUSC 136T extract, which implies that the strain may produce bioactive secondary metabolites that could potentially reduce cancer risk and form the basis for further development as chemopreventive drugs.
Carcinogenesis is a multi-step process, involving activation of oncogenes and inactivation of tumor suppressor genes along with other complex interactions between host and tumor cells. p53 as a tumor suppressor protein has been studied extensively as a promising target for cancer treatment due to its involvement in cancer initiation and progression24,25. It is known to be involved in inducing growth arrest or apoptosis upon activation; dysfunction in the gene responsible for this tumor suppressor protein might result in unregulated cell division – a scenario which is commonly seen in cancer cells with mutated p53 genes such as colon cancer cell line HT-29. The current results revealed differing susceptibility of two colon cell lines, HCT-116 and HT-29 towards MUSC 136T extract which could be due to the status of its tumor suppressor protein p5326,27. In view of this, the effect of MUSC 136T extract on p53 tumor suppressor protein in HCT-116 was examined. The experiment demonstrated that the treatment of MUSC 136T extract caused an increase in p53 protein expression in HCT-116 which may lead to the induction of apoptosis in this particular cell line. This prompted further analysis using morphological analysis. In brief, HCT-116 cells treated with MUSC 136T extract exhibited morphological alterations associated with cell death including presence of shrunken cells and detached from the surface. Apart from morphological studies, intracellular glutathione (GSH) content was also evaluated as this ubiquitous non-protein thiol is essential for cell survival. Interestingly, the HCT-116 cells treated with the extract resulted in a decrease in intracellular GSH content. Since a decline in this molecule is known to trigger apoptotic signaling cascades particularly through activation of p53 signaling pathway, we postulate that it might potentially trigger the activation of p53-dependent apoptosis pathways28. However, further mechanistic studies on p53 associated cell death pathway would be needed to improve the understanding of the action target of MUSC 136T extract.
Results from both antioxidant assays and in vitro cytotoxic screening suggest the presence of potent antioxidant(s) and anticancer agent(s) in the extract of MUSC 136T, thus chemical analysis using GC-MS was performed in order to provide clues as to their identities. This powerful analytical technique, which incorporates the separation power of gas chromatography and detection power of mass spectrometer has been widely used to identify the components of a mixture, including extracts of Streptomyces species29,30. In the current study, GC-MS detected nine compounds in the extract of MUSC 136T, with the majority of the compounds having been previously detected in marine-derived microorganisms including Streptomyces species7,17,31,32.
Pyrrolizidines, as natural occurring complex heterocyclic compounds, are known to exhibit a wide spectrum of bioactivity which includes antimicrobial, anticancer and antioxidant activity9,31. Two pyrrolizidines were identified in MUSC 136T extract, 1,4-diaza-2,5-dioxobicyclo[4.3.0]nonane (5) and pyrrolo[1,2-a]pyrazine-1,4-dione, hexahydro-3-(2-methylpropyl)- (7). Interestingly, compound (7), which was recently identified in Streptomyces cavouresis KU-V39 was associated with cytotoxic activity of human cervical cancer32.The compound pyrrolo[1,2-a]pyrazine-1,4-dione, hexahydro-3-(2-methylpropyl)- (7), either alone or in combination with other compounds may be responsible for the cytotoxic activity observed in MUSC 136T extract.
On the other hand, 1,4-diaza-2,5-dioxobicyclo[4.3.0]nonane (5) as a naturally occurring marine microbe-derived compound has been studied extensively for its antioxidant activity17,33. It was proven to be highly capable of scavenging or reducing the amount of free radicals when assessed with reducing power assay. It is postulated that the scavenging activity observed in MUSC 136T extract could be due to the presence of 1,4-diaza-2,5-dioxobicyclo[4.3.0]nonane (5). Based on the assumption that antioxidants may influence survival of cancer cells34, the same compound 1,4-diaza-2,5-dioxobicyclo[4.3.0]nonane may contribute to the observed cytotoxic effect exhibited by MUSC 136T extract.
The natural trihydroxamate, deferoxamine (9) which has been reported in several members of the Streptomyces family35,36 was also detected in the extract of MUSC 136T. As a drug listed on the World Health Organization’s List of Essential Medicines37, deferoxamine or Desferal (prescription name) has been used extensively as an iron chelator particularly in protection against iron-induced oxidative stress38,39. Apart from its antioxidant activity, deferoxamine has been shown to be cytotoxic to a number of human cancer cells and tumors, mainly by altering signaling pathways which are crucial for cell proliferation40,41.
The presence of these biologically active compounds in MUSC 136T may account for the observed reducing power in antioxidant assays as well as its cytotoxic activity in the various cancer cells. Principally, by inhibiting oxidative stress induced mutagenesis and signaling cascades these compounds might prevent or treat cancer. The results obtained in the current study demonstrate that MUSC 136T extract has tremendous potential for development of cancer preventive agent, meriting further downstream mechanistic studies.
Description of Streptomyces malaysiense sp. nov
Streptomyces malaysiense (mal.ay.si.en’se. N.L. neur. adj. malaysiense belonging/pertaining to Malaysia, the source of the soil from which the organism was isolated).
Cells stain Gram-positive and form yellowish white aerial and pale yellow substrate mycelium on ISP 2 medium. The colors of the aerial and substrate mycelium are media-dependent (See Supplementary Table S1).
Grows well on ISP 2, ISP 5, ISP 6, ISP 7 agar, actinomycetes isolation agar, nutrient agar and starch casein agar after 7–14 days at 28 °C; grows moderately on Streptomyces agar and ISP 3 agar, and does not grow on ISP 4 agar. Grows at 26–40 °C (optimum 28–32 °C), pH 6.0–7.0 (optimum pH 7.0), with 0–6% NaCl tolerance (optimum 0–2%). Cells are positive for catalase but negative for hemolytic activity and melanoid pigment production. Soluble starch, tributyrin (lipase) and casein are hydrolysed but carboxymethylcellulose, chitin and xylan are not. The following compounds are utilized as sole carbon sources: acetic acid, acetoacetic acid, α-D-glucose, α-hydroxy-butyric acid, α-keto-butyric acid, α-keto-glutaric acid, β-hydroxyl-D,L-butyric acid, bromo-succinic acid, citric acid, D-arabitol, D-aspartic acid, D-cellobiose, dextrin, D-fructose, D-fructose-6-phosphate, D-fucose, D-galactose, D-galacturonic acid, D-glucose-6-phosphate, D-gluconic acid, D-glucuronic acid, D-lactic acid methyl ester, D-malic acid, D-maltose, D-mannitol, D-mannose, D-raffinose, D-saccharic acid, D-trehalose, D-turanose, formic acid, gelatin, gentiobiose, glucuronamide, glycerol, glycyl-L-proline, inosine, L-fucose, L-galactonic acid lactone, L-lactic acid, L-malic acid, L-rhamnose, methyl pyruvate, mucic acid, N-acetyl-β-D-mannosamine, N-acetyl-D-galactosamine, N-acetyl-D-glucosamine, N-acetyl-neuraminic acid, pectin, p-hydroxyl-phenylacetic acid, propionic acid, quinic acid, stachyose, sucrose, Tween 40 and γ-amino-butyric acid. The following compounds are not utilized as sole carbon sources: α-D-lactose, β-methyl-D-glucoside, D-melibiose, D-salicin, D-serine, D-sorbitol, 3-methyl glucose and myo-inositol. L-alanine, L-arginine, L-aspartic acid, L-glutamic acid, L-histidine, L-pyroglutamic acid and L-serine are utilized as sole nitrogen sources. Cells are sensitive to ampicillin sulbactam, cefotaxime, chloramphenicol, erythromycin, gentamicin, penicillin G, tetracycline and vancomycin; resistant to ampicillin and nalidixic acid. The cell wall peptidoglycan contains LL-diaminopimelic acid. The cell wall sugars are glucose and ribose. The predominant menaquinones are MK-9(H8) and MK-9(H6). The polar lipids consist of aminopilid, diphosphatidylglycerol, glycolipid, lipid, phosphatidylethanolamine, phosphatidylinositol, phospholipid and phosphoglycolipid. The major cellular fatty acids are anteiso-C15: 0, iso-C16: 0, iso-C15: 0 and anteiso-C17: 0.
The type strain is MUSC 136T (=DSM 100712T = MCCC 1K01246T), isolated from mangrove soil collected from the Tanjung Lumpur mangrove forest located in the state of Pahang, Peninsular Malaysia. The 16S rRNA gene sequence of strain MUSC 136T has been deposited in GenBank/EMBL/DDBJ under the accession number KJ632663. The G+C content of the genomic DNA of the type strain is 72.3 mol%.
Materials and Methods
Isolation and maintenance of isolate
Strain MUSC 136T was isolated from a soil sample collected at site MUSC-TLS3 (3° 48′ 11.1″ N 103 ° 26′ 6.9″ E), located in the mangrove forest of Tanjung Lumpur in the state of Pahang, Peninsular Malaysia in December 2012. Topsoil samples of the upper 20 cm layer (after removing the top 2–3 cm) were collected and sampled into sterile plastic bags using an aseptic metal trowel, and stored at −20 °C. Air-dried soil samples were ground with a mortar and pestle followed by selective pretreatment using wet heat in sterilized water16. The pretreated air-dried soil (5 g) in 45 mL sterilized water was spread onto the isolation medium ISP 2 supplemented with cycloheximide (25 μg/mL) and nystatin (10 μg/mL), and incubated at 28 °C for 14 days. Pure cultures of strain MUSC 136T were isolated and maintained on ISP 2 medium slants at 28 °C and preserved as glycerol suspensions (20%, v/v) at −20 °C.
Genomic and phylogenetic analyses
DNA extraction for PCR was performed as described previously12 while 16S rRNA gene was amplified as described by Lee et al.15. The 16S rRNA gene sequence of strain MUSC 136T was aligned with representative sequences of related type strains of the genus Streptomyces retrieved from the GenBank/EMBL/DDBJ databases. Phylogenetic trees were constructed with the neighbour-joining (Fig. 2) and maximum-likelihood algorithms (See Supplementary Fig. S1). Evolutionary distances for the neighbour-joining algorithm were computed using Kimura’s two-parameter model42. The EzTaxon-e server (http://eztaxon-e.ezbiocloud.net/) was used for calculations of sequence similarity43.
BOX-PCR fingerprint analysis and DDH were performed as previously described44,45. The extraction of genomic DNA for DNA-DNA hybridization of strain MUSC 136T, Streptomyces misionensis NBRC 13063T, Streptomyces phaeoluteichromatogenes DSM 41898T and Streptomyces rutgersensis NBRC 12819T were carried out by the Identification Service of the DSMZ, Braunschweig, Germany. The G+C content of strain MUSC 136T was determined by HPLC46.
Phenotypic characteristics
Cultural characteristics of strain MUSC 136T were determined following growth on ISP 2, ISP 3, ISP 4, ISP 5, ISP 6 and ISP 7 agar, actinomycetes isolation agar (AIA), Streptomyces agar (SA), starch casein agar (SCA) and nutrient agar for 14 days at 28 °C. Light microscopy (80i, Nikon) and scanning electron microscopy (JEOL-JSM 6400) were used to observe the morphology of the strain after incubation on ISP 2 medium at 28 °C for 7–14 days. The designation of colony color was determined using the ISCC-NBS color charts while gram staining performed by standard Gram reaction and confirmed by KOH lysis15,16,17. The effects of pH and salinity on growth of MUSC 136T were evaluated in tryptic soy broth (TSB), while effects of temperature were studied in ISP 2 agar15. Production of melanoid pigments were examined using ISP 7 agar while its hemolytic activity was assessed on blood agar medium containing 5% (w/v) peptone, 3% (w/v) yeast extract, 5% (w/v) NaCl and 5% (v/v) horse blood after incubation at 32 °C for 7–14 days16. Amylolytic, cellulase, chitinase, lipase, protease, xylanase and catalase activities were determined on ISP 2 following protocols described by Lee et al.47. Antibiotic susceptibility tests were performed by the disc diffusion method as described by Shieh et al.48 Carbon-source utilization and chemical sensitivity assays were determined using Biolog GenIII MicroPlates (Biolog, USA) according to the manufacturer’s instructions.
All phenotypic assays mentioned above were performed concurrently for strains MUSC 136T, Streptomyces misionensis NBRC 13063T, Streptomyces phaeoluteichromatogenes DSM 41898T and Streptomyces rutgersensis NBRC 12819T.
Chemotaxonomic characteristics
The analyses of peptidoglycan amino acid composition and sugars of strain MUSC 136T were carried out by the Identification Service of the DSMZ15,16,17. Analysis of respiratory quinones, polar lipids and fatty acids were carried out by the Identification Service of the DSMZ. Major diagnostic cell wall sugars of strain MUSC 136T were obtained as described by Whiton et al.49 and analyzed by TLC on cellulose plates50.
Extract preparation of MUSC 136T
MUSC 136T was grown in TSB as seed medium for 14 days prior to fermentation process. The fermentation medium, FM3 was autoclaved at 121 °C for 15 min prior to experiment12,51. Fermentation was carried out in 200 mL FM3, shaking at 200 rpm for 7–10 days at 28 °C, inoculated with 200 μL seed media16,17. The resulting FM3 medium was recovered by centrifugation at 12000 g for 15 min. The supernatant was filtered and subjected to freeze drying process. The freeze-dried sample was extracted with methanol and the final extract concentrated by rotary evaporator at 40 °C. The final concentrate was suspended in DMSO prior to bioactivity screening assays.
Evaluation of antioxidant activity using different assays
2,2-diphenyl-1-picrylhydrazyl (DPPH) was used to examine antioxidant activity by measuring its hydrogen donating or radical scavenging ability. Scavenging activity against DPPH free radicals by MUSC 136T extract was determined using previously described protocol17,52. The reduction in radical content was measured by the decrease in the absorbance at 515 nm. 2,2′-azino-bis (3-ethylbenzothiazoline-6-sulphonic acid) (ABTS) assay was performed as previously described in published literature with some modifications53. ABTS radical cation (ABTS∙) was generated by reacting ABTS stock solution (7 mM) and potassium persulphate (2.45 mM) for 24 hour prior to assay. The change in radical amount was indicated by decrease in absorbance at 743 nm. Superoxide anion scavenging activity was determined using commercially available colorimetric microtiter plate method (19160 SOD Assay Kit-WST, Sigma Aldrich) according to manufacturer’s protocol. Metal chelating activity was determined by measuring formation of Fe2+-ferrozine complex as previously described by Manivasagan et al. with slight modification54. 2 mM of FeSO4 was added to extract and the reaction was initiated by adding 5 mM of ferrozine before measuring at 562 nm using spectrophotometer.
Cell lines maintenance and growth condition
All human derived cancer cell lines used in the study were maintained in RPMi 1640 supplemented with 10% FBS in a humidified incubator (5% CO2 in air at 37 °C)55. The cancer cell lines included were cervical cancer cell, Ca Ski; colon cancer cell, HCT-116 and HT-29; lung carcinoma cell line, A549.
Cytotoxicity activity determination using 3-(4,5-dimethylthazol-2yl)-2,5-diphenyl tetrazolium-bromide (MTT) assay
The cytotoxic activity of MUSC 136T extract was investigated using 3-(4,5-dimethylthazol-2yl)-2,5-diphenyl tetrazolium-bromide (MTT) assay according to previously established method18. The determination of cell viability was performed using a microplate reader at 570 nm (with 650 nm as reference wavelength).
Measurement of intracellular GSH content
Intracellular GSH content was measured using CellMetrix™ Intracellular GSH Assay Kit according to manufacturer’s protocol. Cells were seeded in 6-well dishes at density of 3 × 105 and allowed to adhere overnight. Subsequently, the cells were treated with MUSC 136T extract. After the incubation period, cells were harvested and stained prior to analysis using BD FACSVerseTM flow cytometer.
Measurement of p53 protein
The status of p53 protein was investigated with flow cytometry system as described by Goh et al. with slight modification24. Cells were seeded in 6-well dishes at density of 3 × 105 and allowed to adhere overnight before treated with MUSC 136T extract. After the incubation period, cells were then washed twice with PBS, fixed with 4% paraformaldehyde with 0.5% sodium azide before permeabilized with Perm/Wash™ buffer (BD Biosciences). For detection of p53, the cells were incubated with 100 μL of Perm/Wash™ buffer containing mouse anti-human p53 monoclonal IgG1 or IgG1 isotype control. After washing procedure, the cells were further incubated with FITC-conjugated goat anti-mouse IgG1 at 4 °C in the dark for 30 min. Finally, the cells were then washed with Perm/Wash™ buffer before analysis by using BD FACSVerseTM flow cytometer.
Gas chromatography-mass spectrometry (GC-MS) analysis
GC-MS analysis was performed as previously described17. Analysis of MUSC 136T extract was performed on an Agilent Technologies 6980N (GC) equipped with 5979 Mass Selective Detector (MS) using a HP-5MS (5% phenyl methyl siloxane) capillary column of dimensions 30.0 m × 250 μm × 0.25 μm and helium as carrier gas at 1 mL/min. The column temperature was programmed initially at 40 °C for 10 min, followed by an increase of 3 °C/min to 250 °C and was kept isothermally for 5 min. The MS was operating at 70 eV. The constituents were identified by comparison of their mass spectral data with those from NIST 05 Spectral Library.
Statistical analysis
Experiments to investigate antioxidant and cytotoxic activities were done in quadruplicate while measurement of p53 and GSH using flow cytometry in triplicate. Data analysis was performed with SPSS statistical analysis software and the results were expressed as mean ± standard deviation (SD). One-way analysis of variance (ANOVA) followed by the appropriate post hoc test (Tukey) was used to establish whether significant differences existed between groups. A difference was considered statistically significant when p ≤ 0.05.
Additional Information
How to cite this article: Ser, H.-L. et al. Streptomyces malaysiense sp. nov.: A novel Malaysian mangrove soil actinobacterium with antioxidative activity and cytotoxic potential against human cancer cell lines. Sci. Rep. 6, 24247; doi: 10.1038/srep24247 (2016).
References
Jemal, A. et al. Global cancer statistics. CA Cancer J. Clin. 61, 69–90, 10.3322/caac.20107 (2011).
Jemal, A., Center, M. M., DeSantis, C. & Ward, E. M. Global patterns of cancer incidence and mortality rates and trends. Cancer Epidemiol. Biomarkers Prev. 19, 1893–1907, 10.1158/1055-9965.EPI-10-0437 (2010).
Chin, Y.-W., Balunas, M. J., Chai, H. B. & Kinghorn, A. D. Drug discovery from natural sources. AAPS J. 8, E239–E253. 10.1007/BF02854894 (2006).
Lalla, R. V., Saunders, D. P. & Peterson, D. E. Chemotherapy or radiation-induced oral mucositis. Dent. Clin. North Am. 58, 341–349, 10.1016/j.cden.2013.12.005 (2014).
Robles-Fernandez, I. et al. Antitumor properties of natural compounds and related molecules. Recent Pat. Anticancer Drug Discov. 8, 203–215 (2013).
Newman, D. J. & Cragg, G. M. Marine-sourced anti-cancer and cancer pain control agents in clinical and late preclinical development. Mar. Drugs 12, 255–278, 10.3390/md12010255 (2014).
Berdy, J. Bioactive microbial metabolites. J. Antibiotics 58, 1–26, 10.1038/ja.2005.1 (2005).
Demain, A. L. & Sanchez, S. Microbial drug discovery: 80 years of progress. J. Antibiotics 62, 5–16, 10.1038/ja.2008.16 (2009).
Waksman, S. A. & Henrici, A. T. The nomenclature and classification of the actinomycetes. J. Bacteriol. 46, 337 (1943).
Alongi, D. M. Mangrove forests: resilience, protection from tsunamis, and responses to global climate change. Estuar.Coast. Shelf Sci. 76, 1–13, 10.1016/j.ecss.2007.08.024 (2008).
Lee, L. H. et al. Diversity and antimicrobial activities of actinobacteria isolated from tropical mangrove sediments in Malaysia. Scientific World J. 2014, 698178, 10.1155/2014/698178 (2014).
Hong, K. et al. Actinomycetes for marine drug discovery isolated from mangrove soils and plants in China. Mar. drugs 7, 24–44, 10.3390/md7010024 (2009).
Azman, A. S., Othman, I., Velu, S. S., Chan, K. G. & Lee, L. H. Mangrove rare actinobacteria: taxonomy, natural compound, and discovery of bioactivity. Front. Microbiol. 6, 856, 10.3389/fmicb.2015.00856 (2015).
Sui, J.-L. et al. Streptomyces sanyensis sp. nov., isolated from mangrove sediment. Int. J. Syst. Evol. Microbiol. 61, 1632–1637, 10.1099/ijs.0.023515-0 (2011).
Lee, L. H. et al. Streptomyces pluripotens sp. nov., a bacteriocin-producing streptomycete that inhibits meticillin-resistant Staphylococcus aureus . Int. J. Syst. Evol. Microbiol. 64, 3297–3306, 10.1099/ijs.0.065045-0 (2014).
Ser, H.-L. et al. Streptomyces gilvigriseus sp. nov., a novel actinobacterium isolated from mangrove forest soil. Antonie van Leeuwenhoek 107, 1369–1378, 10.1007/s10482-015-0431-5 (2015).
Ser, H.-L. et al. Presence of antioxidative agent, Pyrrolo[1,2-a]pyrazine-1,4-dione, hexahydro- in newly isolated Streptomyces mangrovisoli sp. nov. Front. Microbiol. 6, 854, 10.3389/fmicb.2015.00854 (2015).
Williams, S. T., Goodfellow, M. & Alderson, G. Genus Streptomyces Waksman and Henrici 1943, 339AL . In Bergey’s Manual of Systematic Bacteriology, Volume 4. (eds Williams, S. T. et al. ), 2452–2492, (Williams & Wilkins, 1989).
Wayne, L. G. et al. Report of the Ad Hoc Committee on Reconciliation of Approaches to Bacterial Systematics. Int. J. Syst. Evol. Microbiol. 37, 463–464, 10.1099/00207713-37-4-463 (1987).
Lechevalier, M. P. & Lechevalier, H. Chemical composition as a criterion in the classification of aerobic actinomycetes. Int. J. Syst. Bacteriol. 20, 435–443, 10.1099/00207713-20-4-435 (1970).
Kim, S. B., Lonsdale, J., Seong, C. N. & Goodfellow, M. Streptacidiphilus gen. nov., acidophilic actinomycetes with wall chemotype I and emendation of the family Streptomycetaceae (Waksman and Henrici (1943)AL) emend. Rainey et al. 1997. Antonie van Leeuwenhoek 83, 107–116 (2003).
Reuter, S., Gupta, S. C., Chaturvedi, M. M. & Aggarwal, B. B. Oxidative stress, inflammation, and cancer: how are they linked? Free Rad. Biol. Med. 49, 1603–1616, 10.1016/j.freeradbiomed.2010.09.006 (2010).
Kawanishi, S., Oikawa, S. & Murata, M. Evaluation for safety of antioxidant chemopreventive agents. Antioxid. Redox Signal 7, 1728–1739, 10.1089/ars.2005.7.1728 (2005).
Goh, B. H., Chan, C. K., Kamarudin, M. N. & Abdul Kadir, H. Swietenia macrophylla King induces mitochondrial-mediated apoptosis through p53 upregulation in HCT116 colorectal carcinoma cells. J. Ethnopharmacol. 153, 375–385, 10.1016/j.jep.2014.02.036 (2014).
Schneider-Stock, R. et al. 5-Aza-cytidine is a potent inhibitor of DNA methyltransferase 3a and induces apoptosis in HCT-116 colon cancer cells via Gadd45- and p53-dependent mechanisms. J. Pharmacol. Exp. Ther. 312, 525–536, 10.1124/jpet.104.074195 (2005).
Makizumi, R., Yang, W.-L., Owen, R. P., Sharma, R. R. & Ravikumar, T. S. Alteration of drug sensitivity in human colon cancer cells after exposure to heat: implications for liver metastasis therapy using RFA and chemotherapy. Int. J. Clin. Exp. Med. 1, 117–129 (2008).
Lin-Lee, Y.-C., Tatebe, S., Savaraj, N., Ishikawa, T. & Tien Kuo, M. Differential sensitivities of the MRP gene family and γ-glutamylcysteine synthetase to prooxidants in human colorectal carcinoma cell lines with different p53 status. Biochem. Pharmacol. 61, 555–563, 10.1016/S0006-2952(00)00592-X (2001).
Shukla, S. & Gupta, S. Apigenin-induced prostate cancer cell death is initiated by reactive oxygen species and p53 activation. Free Rad. Biol. Med. 44, 1833–1845, 10.1016/j.freeradbiomed.2008.02.007 (2008).
Jog, R., Pandya, M., Nareshkumar, G. & Rajkumar, S. Mechanism of phosphate solubilization and antifungal activity of Streptomyces spp. isolated from wheat roots and rhizosphere and their application in improving plant growth. Microbiol. 160, 778–788, 10.1099/mic.0.074146-0 (2014).
Olano, C., Mendez, C. & Salas, J. A. Antitumor compounds from actinomycetes: from gene clusters to new derivatives by combinatorial biosynthesis. Nat. Prod. Rep. 26, 628–660, 10.1039/b822528a (2009).
Wang, C. et al. Antifungal activity of volatile organic compounds from Streptomyces alboflavus TD-1. FEMS Microbiol. Lett. 341, 45–51, 10.1111/1574-6968.12088 (2013).
Narendhran, S., Rajiv, P., Vanathi, P. & Sivaraj, R. Spectroscopic analysis of bioactive compounds from Streptomyces cavouresis kuv39: Evaluation of antioxidant and cytotoxicity activity. Int. J. Pharm. Pharmaceut. Sci. 6, 319–322 (2014).
Gopi, M., Dhayanithi, N. B., Devi, K. N. & Kumar, T. T. A. Marine natural product, Pyrrolo [1,2-a] pyrazine-1,4-dione, hexahydro-(C7H10N2O2) of antioxidant properties from Bacillus species at Lakshadweep archipelago. J. Coast. Life Med. 2, 632–637, 10.12980/JCLM.2.201414J40 (2014).
Wojcik, M., Burzynska-Pedziwiatr, I. & Wozniak, L. A. A review of natural and synthetic antioxidants important for health and longevity. Curr. Med. Chem. 17, 3262–3288, 10.2174/092986710792231950 (2010).
Barona-Gomez, F., Wong, U., Giannakopulos, A. E., Derrick, P. J. & Challis, G. L. Identification of a cluster of genes that directs desferrioxamine biosynthesis in Streptomyces coelicolor M145. J. Am. Chem. Soc. 126, 16282–16283, 10.1021/ja045774k (2004).
Yamanaka, K. et al. Desferrioxamine E produced by Streptomyces griseus stimulates growth and development of Streptomyces tanashiensis . Microbiol. 151, 2899–2905, 10.1099/mic.0.28139-0 (2005).
WHO. WHO model list of essential medicines: 19thlist (updated) April 2015. (2015). Available at: http://www.who.int/medicines/publications/essentialmedicines/en/. (Accessed: 19th August 2015).
van der Kraaij, A. M., Mostert, L. J., van Eijk, H. G. & Koster, J. F. Iron-load increases the susceptibility of rat hearts to oxygen reperfusion damage. Protection by the antioxidant (+)-cyanidanol-3 and deferoxamine. Circulation 78, 442–449, 10.1161/01.CIR.78.2.442 (1988).
Lan, J. & Jiang, D. H. Desferrioxamine and vitamin E protect against iron and MPTP-induced neurodegeneration in mice. J. Neural Transm. 104, 469–481, 10.1007/BF01277665 (1997).
Tomoyasu, S. et al. Suppression of HL-60 cell proliferation by deferoxamine: changes in c-myc expression. Anticancer Res. 13, 407–410 (1992).
Yamasaki, T., Terai, S. & Sakaida, I. Deferoxamine for advanced hepatocellular carcinoma. New Eng. J. Med. 365, 576–578, 10.1056/NEJMc1105726 (2011).
Kimura, M. A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J. Mol. Evol. 16, 111–120 (1980).
Kim, O. S. et al. Introducing EzTaxon-e: a prokaryotic 16S rRNA gene sequence database with phylotypes that represent uncultured species. Int. J. Syst. Evol. Microbiol. 62, 716–721, 10.1099/ijs.0.038075-0 (2012).
Lee, L. H. et al. Novosphingobium malaysiense sp. nov. isolated from mangrove sediment. Int. J. Syst. Evol. Microbiol. 64, 1194–1201, 10.1099/ijs.0.059014-0 (2014).
Lee, L.-H. et al. Microbacterium mangrovi sp. nov., an amylolytic actinobacterium isolated from mangrove forest soil. Int. J. Syst. Evol. Microbiol. 64, 3513–3519, 10.1099/ijs.0.059014-0 (2014).
Mesbah, M., Premachandran, U. & Whitman, W. B. Precise measurement of the G+C content of deoxyribonucleic acid by high-performance liquid chromatography. Int. J. Syst. Bacteriol. 39, 159–167, 10.1099/00207713-39-2-159 (1989).
Lee, L. H. et al. Mumia flava gen. nov., sp. nov., an actinobacterium of the family Nocardioidaceae. Int. J. Syst. Evol. Microbiol. 64, 1461–1467, 10.1099/ijs.0.058701-0 (2014).
Shieh, W. Y., Chen, Y. W., Chaw, S. M. & Chiu, H. H. Vibrio ruber sp. nov., a red, facultatively anaerobic, marine bacterium isolated from sea water. Int. J. Syst. Evol. Microbiol. 53, 479–484, doi: 10.1099/ijs.0.02307-0 (2003).
Whiton, R. S., Lau, P., Morgan, S. L., Gilbart, J. & Fox, A. Modifications in the alditol acetate method for analysis of muramic acid and other neutral and amino sugars by capillary gas chromatography-mass spectrometry with selected ion monitoring. J. Chromatogr. 347, 109–120, 10.1016/S0021-9673(01)95474-3 (1985).
Staneck, J. L. & Roberts, G. D. Simplified approach to identification of aerobic actinomycetes by thin-layer chromatography. Appl. Microbiol. 28, 226–231 (1974).
Lee, L.-H. et al. Molecular characterization of Antarctic actinobacteria and screening for antimicrobial metabolite production. World J. Microbiol. Biotech. 28, 2125–2137, 10.1007/s11274-012-1018-1 (2012).
Al-Henhena, N. et al. Chemopreventive effects of Strobilanthes crispus leaf extract on azoxymethane-induced aberrant crypt foci in rat colon. Sci. Rep. 5, 10.1038/srep13312 (2015).
Miser-Salihoglu, E., Akaydin, G., Caliskan-Can, E. & Yardim-Akaydin, S. Evalution of antioxidant activity of various herbal folk medicines. J. Nutri. Food Sci. 3, 3–5, 10.4172/2155-9600.1000222 (2013).
Manivasagan, P., Venkatesan, J., Sivakumar, K. & Kim, S. K. Production, characterization and antioxidant potential of protease from Streptomyces sp. MAB18 using poultry wastes. BioMed Res. Int. 2013, 496586–496586, 10.1155/2013/496586 (2013).
Tan, L. T. H. et al. Investigation of antioxidative and anticancer potentials of Streptomyces sp. MUM256 isolated from Malaysia mangrove soil. Front. Microbiol. 6, 1316, 10.3389/fmicb.2015.01316 (2015).
Acknowledgements
This work was inspired by Monash PhD Research Training Module which entitled “Bioprospective of microbes with biopharmaceutical potential with bioinformatics and drug discovery platforms” and financially supported by a University of Malaya for High Impact Research Grant (UM-MOHE HIR Nature Microbiome Grant No. H-50001-A000027 and No. A000001-50001) awarded to K.-G. C., MOSTI eScience Funds (06-02-10-SF0300) awarded to L.-H. L. and (02-02-10-SF0215) awarded to B.-H.G., External Industry Grant (Biotek Abadi - Vote No. GBA-808138 and GBA-808813) awarded to L.-H.L. The authors are thankful to Professor Bernhard Schink for the support in the Latin etymology of the new species name.
Author information
Authors and Affiliations
Contributions
Conceived and designed the experiments: H.-L.S., B.-H.G. and L.-H.L. Performed the experiments: H.-L.S., B.-H.G. and L.-H.L. Analyzed the data: H.-L.S., U.D.P., K.-G.C., B.-H.G. and L.-H.L. Contributed reagents/materials/analysis tools: H.-L.S., U.D.P., K.-G.C., W.-F.Y., B.-H.G. and L.-H.L. Wrote the paper: H.-L.S., B.-H.G. and L.-H.L.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Ser, HL., Palanisamy, U., Yin, WF. et al. Streptomyces malaysiense sp. nov.: A novel Malaysian mangrove soil actinobacterium with antioxidative activity and cytotoxic potential against human cancer cell lines. Sci Rep 6, 24247 (2016). https://doi.org/10.1038/srep24247
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep24247
This article is cited by
-
Antioxidant and chemoprotective potential of Streptomyces levis strain isolated from human gut
AMB Express (2023)
-
A pilot survey on antimicrobial activity and diversity of soil-derived actinobacteria from different depths in Gudzhirganskoe saline lake in Eastern Siberia
Journal of Oceanology and Limnology (2023)
-
Genetic Diversity and Anti-Oxidative Potential of Streptomyces spp. Isolated from Unexplored Niches of Meghalaya, India
Current Microbiology (2022)
-
Evaluation of the anticarcinogenic potential of the endophyte, Streptomyces sp. LRE541 isolated from Lilium davidii var. unicolor (Hoog) Cotton
Microbial Cell Factories (2021)
-
Pharmacological properties of some mangrove sediment-associated bacillus isolates
Archives of Microbiology (2021)