Abstract
Background
SGLT2 inhibitors (SGLTi) confer significant cardiometabolic benefits in diabetes, but are associated with increased risk of diabetic ketoacidosis (DKA). We hypothesized that supporting anaplerosis with glycerol could mitigate SGLTi-induced ketosis, enhance insulin-mediated resolution of severe diabetic ketosis, and lower the risk of DKA when used as a dietary supplement.
Methods
Insulin-dependent diabetes was induced in yearling sheep using alloxan. The animals subsequently developed severe hyperglycemia (glucose > 500 mg/dL) and hyperketonemia (β-hydroxybutyrate [BHB] > 5 mM), requiring daily insulin therapy for metabolic control and survival. Ketosis was induced by partial insulin withdrawal and with the administration of 10 mg of the SGLT2 inhibitor empagliflozin. Animals were randomly assigned in a cross-over trial to evaluate the effects of intravenous 5% glycerol infusions for the treatment of moderate diabetic ketosis (1.5 mM < BHB < 3.0 mM), and as an insulin-adjunct therapy in cases of severe diabetic ketosis (BHB > 3.0 mM). Longer-term effects of dietary glycerol were assessed by supplementing 5% glycerol in the drinking water of the diabetic sheep under empagliflozin treatment. Data were analyzed using repeated measures ANOVA with a standard least squares fit model in JMP.
Results
Intravenous glycerol administration decreased moderate ketosis by 28.5% (P = 0.03), increased glucose by 63.3% (P = 0.04), and enhanced insulin-mediated resolution of severe diabetic ketosis (P = 0.001). Nightly dietary glycerol intake suppressed empagliflozin-induced morning elevations in blood BHB (P = 0.002).
Conclusions
Intravenous glycerol reduced empagliflozin-induced ketosis and provided protection against hypoglycemia. When used as an insulin adjunct, it enhanced insulin resolution of severe diabetic ketosis induced by empagliflozin. Dietary glycerol supplementation improved the energetic state and reduced the risk of ketosis in diabetic sheep undergoing SGLTi therapy. These findings may have clinical relevance for DKA management and nutritional prevention, particularly but not necessarily under SGLTi therapy.
Similar content being viewed by others
Introduction
Individuals with type 1 diabetes (T1D) rely on insulin administration for survival. However, adverse effects of insulin therapy such as weight gain and hypoglycemia often hinder the achievement of glycemic and metabolic targets [1, 2]. Conversely, poor adherence to insulin therapy, strenuous physical activity, intercurrent illnesses, and the use of drugs that affect carbohydrate metabolism may precipitate diabetic ketoacidosis (DKA). This life-threatening metabolic crisis is characterized by the metabolic triad of hyperglycemia (glucose ≥ 200 mg/dL), ketosis (β-hydroxybutyrate ≥ 3.0 mM), and anion-gap acidosis (pH ≤ 7.3) [3]. If not promptly treated, its consequences can be fatal, making DKA a leading cause of mortality in young patients with T1D [4, 5]. The pathophysiology of DKA stems from the extreme catabolic milieu induced by insulin deficiency that shifts the body’s mode of energy production from hepatic carbohydrates to fat catabolism into ketone bodies (KB) [6]. While the synthesis of KB as alternative energy metabolites is part of the normal adaptation to glucose shortage under starvation physiology, upon export to the bloodstream, KB contribute equimolar hydrogen ions [7]. Therefore, high concentrations of blood KB (ketosis or hyperketonemia) increase the anion gap and promote metabolic acidosis [8], which in the case of uncontrolled diabetes can precipitate DKA. Notably, ketosis with or without acidosis is considered to be an acute complication of poorly controlled or newly diagnosed type I or type II diabetes mellitus, and higher levels of oxidative stress and inflammation were reported in patients with hyperketonemic diabetes [9]. Accordingly, diabetic ketosis by itself serves as a medically important target regardless of its role in potentiating DKA.
By inhibiting renal reabsorption of urinary glucose, inhibitors of sodium-glucose cotransporters (SGLTi), such as gliflozins, represent a relatively new class of attractive antidiabetic agents that lower blood glucose independently of insulin [10,11,12,13]. Indeed, SGLTi therapy has shown promise in improving glycemic control and reducing insulin doses required to meet glycemic targets, thereby decreasing the risk of developing insulin resistance and conferring cardiometabolic benefits [14]. However, SGLTi therapy has a significant drawback: it predisposes to DKA [15,16,17], often at near-normal glucose levels (euglycemia) [17,18,19]. Euglycemic DKA can delay the diagnosis and management of this emergency condition [19,20,21]. Therefore, despite the recognized metabolic merits of SGLTi therapy, the US Food and Drug Administration (FDA) has approved its use only for Type 2 diabetes (T2D), not for patients with T1D who are more prone to develop DKA [22, 23]. Yet, SGLTi therapy remains a concern for DKA development in T2D as well [24].
The mechanism of SGLTi predisposition to DKA has not been fully established [5, 25]. However, several explanations have been proposed. These include decreased urinary excretion of KB, as well as factors associated with the reduction in daily insulin doses, such as decreased suppression of lipolysis and ketogenesis, and increased glucagon levels [5, 25].
We have reasoned that prolonged urinary excretion of blood glucose by SGLTi therapy might result in a harmful deficiency of intracellular carbohydrates, significantly affecting liver energy metabolism. In such circumstances, metabolites derived from carbohydrate-based structures, particularly the TCA-cycle intermediates that are interconnected with cellular glucose levels via the cataplerotic or anaplerotic flow of mitochondrial oxaloacetate, may substantially decrease. Depletion of TCA-cycle intermediate metabolites reduces the hepatic oxidative capacity [26, 27] and, consequently, the oxidation of acetyl coenzyme A (acetyl-CoA), the accumulation of which could enhance ketogenesis by substrate push mechanism [28, 29]. Following this logic, the intracellular glucose deficiency and elevated KB resulting from insufficient insulin or impaired insulin signaling (insulin resistance) in diabetes might be further exacerbated by SGLTi therapy, leading to an increased risk of euglycemic DKA development [5, 19, 25, 30, 31].
Accordingly, we hypothesized that providing energetic substrates to replenish the pool of TCA-cycle intermediates, either by enhancing the anaplerotic or reducing the cataplerotic flow, would reduce diabetic ketosis independently of insulin. Given that most potential fuel metabolites for this purpose are carbohydrate-based molecules, which should be avoided during such a diabetic crisis, we opted to investigate glycerol. This non-sugary yet efficient gluconeogenic and glycolytic substrate could serve as a potential alternative. As a three-carbon source for glucose or pyruvate synthesis, glycerol may contribute to the anaplerotic replenishment of the TCA-cycle intermediates via oxaloacetate. Additionally, it may reduce the cataplerotic flow of mitochondrial oxaloacetate and/or malate to the cytoplasm for gluconeogenesis.
Based on this reasoning, we have made the prediction that glycerol administration would reduce ketogenesis and ketosis in the diabetic context of SGLTi therapy. To test this hypothesis, we employed insulin-dependent diabetic sheep as a model system to investigate both short- and longer-term effects of glycerol treatments on diabetic ketosis induced by the sodium-glucose cotransporter-2 (SGLT2) inhibitor empagliflozin.
Materials and methods
Animal ethics (no human participants)
The animal experiments were conducted at the Volcani experimental farm (Rishon LeZion, Israel). All the procedures were approved by the Volcani Animal Care Committee (permit 885/20 IL), and the experiments were carried out by experienced personnel certified to work with sheep. This manuscript follows the ARRIVE reporting guidelines [32].
Handling of diabetic sheep
The choice of sheep as an experimental model for diabetes [33] was based on their similar body weight to that of humans, which enabled the utilization of similar drug doses and the collection of sufficient biological sample quantities for accurate metabolite measurements and analyses. For the same reasons, the results from this preclinical trial may be better translated to humans, and treatment doses of glycerol employed in this preclinical trial may serve as a good starting point to guide such future clinical trials. The selection of individual animals for the experiment was based on clinical veterinarian check-ups to ensure good health and physical status.
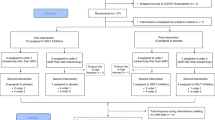
Six yearling ewes of the Afec-Assaf breed [30], approximately 11 months old with similar body weights ( ~ 54 kg), were selected from a cohort born in the same lambing period (around January). They were uniformly fed a maintenance diet to facilitate consistent administration of drug doses and to minimize the involvement of confounding factors. Unfortunately, one animal died a week after diabetes induction and was subsequently excluded from the final analysis. Insulin-dependent diabetes was induced by chemical destruction of pancreatic beta cells with alloxan, essentially as previously described [34,35,36]. Briefly, a filter-sterilized 5% alloxan-saline solution was administered intravenously as a single dose of 50 mg/kg body weight (BW) [36, 37]. As expected, the animals developed Type-1-like diabetes within 2-3 days, as confirmed by blood glucose values > 200 mg/dL [38] (Scheme 1), and their reliance on exogenous insulin administration to protect against hazardous metabolic values of blood glucose and β-hydroxybutyrate (BHB).
Timeline and experimental design for Experiments 1 and 2, for testing the capacity of glycerol, alone and as an insulin adjunct, to inhibit diabetic ketosis. Type-1-like diabetes was induced with alloxan infusions. Two days after confirming diabetes, the sheep were maintained on insulin therapy to manage the condition. The diabetic sheep were then randomized into GLY-SAL or SAL-GLY treatment sequences, and ketosis was induced through partial insulin withdrawal combined with empagliflozin administration. In Experiment 1, the treatments consisted of either 1 L saline (SAL) or 5% glycerol in saline (GLY). In Experiment 2, the treatments included 5 IU insulin supplemented with either 0.5 L SAL or GLY. Blood and plasma samples were collected repeatedly at indicated time points for up to 5 hours post-infusion. GLY: Glycerol. SAL: Saline. IV: intravenous.
If left untreated with insulin, these diabetic animals rapidly develop severe hyperglycemia and hyperketonemia within days (Fig. S1), closely resembling the presentation of DKA percipitation. Thus, strongly suggesting that virtually no residual endogenous insulin activity remains in the alloxan-treated sheep. These preliminary representative data from one such animal (as additional cases posed a high risk of lethality) indicate that the observed reduction in diabetic ketosis by glycerol (Fig. S1) is virtually independent of insulin.
As such, the animals were maintained on daily subcutaneous human insulin (Insulatard®) (Novo Nordisk A/S, Bagsvaerd, Denmark), according to the manufacturer’s instructions ( ~ 0.3–1.0 IU/Kg BW). The animals were fed a regular 100% maintenance diet based on the NRC guidelines [39] until the initiation of the experiments. Prior to the intravenous (IV) treatments, a single catheter (Delta Med s.p.a., Viadana, Italy) was installed into the jugular veins of each sheep, and as needed to prevent blood coagulation, the catheters were flushed with 3-5 mL of 20 IU/mL heparin (Merckle GmbH., Blaubeuren, Germany) in sterile saline (0.9% NaCl, Teva Medical Marketing Ltd., Ashdod, Israel).
Experimental design
To efficiently study the effects of the treatments, i.e., glycerol vs. control (saline or water), a 2 × 2 cross-over design was employed to minimize between-animal variation and confounding factors. For a complete randomized design, we estimated based on previous work [40], that 8 sheep would suffice to detect a difference of 20% in blood KB at a statistical power of 85% and a significance level of 0.05. However, we expected the cross-over design to be significantly more sensitive than a parallel trial and theoretically require half the number of animals to achieve the same statistical power. Therefore, five sheep (N = 5) were randomized, a priori, into two treatment sequences: [1] glycerol-control or [2] control-glycerol, where the individual animals served as the experimental units. Randomization was performed using the RAND function in Excel (Microsoft Office 2019). Therefore, each animal was treated with both interventions (glycerol and control) sequentially at a random order, allowing 12 h of “Washout” time (equating at least twice the time of the measured effects) between interventions to minimize carryover effects, as depicted in Scheme 1. Noteworthy, the carry-over effects, if any exist, are expected to be negligible for a treatment with energy substrates that are relatively rapidly metabolized, particularly as measured previously for glycerol [40].
Three (3) experiments were conducted with the diabetic sheep, each addressing a specific objective.
Experiment 1 aimed to study short-term effects of bolus glycerol treatments on diabetic ketosis, categorically defined as 1.5 mM < BHB < 3 mM. Ketosis was induced by partial insulin withdrawal ( ~ half the maintenance dose) and subcutaneous administration of 10 mg empagliflozin (dissolved in 4 ml of 50% ethanol) as a single dose per day. The IV-infused interventions consisted of 1 L filter-sterilized solution of saline (SAL) as a control, or of 5% glycerol (GLY) (Glycerin USP grade, 99.5% pure, C.S. Chemicals Ltd., Haifa, Israel) dissolved in saline; both administered sequentially to each animal at a random order (Scheme 1). The interventions were initiated only once sub-clinical ketosis was confirmed (typically ~ 8 h after empagliflozin injection). For each intervention, blood samples of 6 mL were collected into heparin vacutainers (BD., Plymouth, UK), immediately before the IV interventions, and at regular intervals for 5 hours post infusion (30, 60, 120, 180, 240 and 300 min, Scheme 1).
Experiment 2 aimed to evaluate the benefit of a combined treatment of insulin and glycerol on the resolution of severe diabetic ketosis, categorically defined as BHB > 3.0 mM, as typified in DKA. Current urgent care practice to resolve DKA employs initial treatment with insulin only, and, as needed, glucose may be supplemented a few hours later to prevent hypoglycemia. Therefore, Experiment 2 aims to shed light on the potential advantage of a combined hormonal-and-energetic therapy provided at once to target both DKA and hypoglycemia right from the start, for potentially safer and more efficient DKA resolution. The animals were IV treated sequentially with 5 IU of insulin supplemented with either 0.5 L of sterile saline only (SAL) as the control or 0.5 L of filter-sterilized 5% glycerol dissolved in saline (GLY), using the same timeline and diabetic ketosis induction procedures detailed for Experiment 1 (Scheme 1). For Experiments 1 and 2, the animals were housed as a group and fed a regular maintenance diet for ~2 weeks. To minimize diet-induced energetic fluctuations, a day before the initiation of the interventions and during the 5 h of monitoring time, the sheep were deprived of the concentrate (high energy feed) and served with straw only.
Experiment 3 aimed to investigate longer-term effects of dietary glycerol consumption on diabetic ketosis to explore the potential benefit in reducing the risk for DKA under SGLTi therapy. Diabetes was controlled with human insulin, as described above. For this experiment, insulin was administered mainly in the morning, whereas at around 5 pm, the animals were pretreated with subcutaneous 10 mg empagliflozin to induce moderate ketosis overnight. The diabetic sheep were housed individually to ensure precise control over their glycerol intake in the drinking water and their feed consumption, which consisted of a maintenance diet ration. The drinking interventions were provided immediately after empagliflozin administration by serving each sheep with 5 L of either water (control) or 5% glycerol in water (GLY) for overnight consumption. Each animal was treated with GLY or regular water for 7 consecutive nights, followed by one “Washout” day and a treatment swap for another week. Blood samples were collected daily at 8 am, before the morning feeding, for biochemical analysis (Scheme 2).
Timeline and experimental design for the nutritional study (Experiment 3). The diabetic sheep received daily insulin injections each morning and with empagliflozin each evening. Before the dietary interventions, the animals were randomized into one of two treatment sequences: GLY-WATER or WATER-GLY. Each intervention consisted of providing 5 L of 5% glycerol (GLY) or water) daily at approximately 5 pm for overnight consumption over 7 consecutive nights per period. Blood and plasma samples were collected each morning at around 8 am to measure BHB, glucose, and NEFA concentrations.
Blood biochemical analysis
Blood BHB and glucose concentrations were determined in real-time using the Freestyle Optium Neo glucometers (Abbot Diabetes Care Ltd., Oxfordshire, UK). The heparinized blood was centrifuged at 2000 × g at 4 °C for 15 min to isolate plasma samples, which were immediately stored at −80 °C until further biochemical analyses. Concentrations of plasma non-esterified fatty acids (NEFA) were determined using a NEFA analysis kit (Wako Chemicals, GmbH, Neuss, Germany).
Statistical analysis
All the statistical analyses were performed in JMP (VERSION 15.1.0, SAS Institute Inc., Cary, NC, USA). Continuous dependent variables (glucose, BHB, and NEFA), measured repeatedly over time, were analyzed using the JMP mixed-model approach via repeated measures ANOVA. The model included: Treatment (GLY vs. SAL) as a within-subject fixed factor, Sequence (GLY-SAL and SAL-GLY) as a between-subject fixed factor, Time as a within-subject nominal fixed factor nested within Treatment, Treatment x Sequence Interaction, and Individual Animal as a random factor nested within Sequence. Access to the raw data may be provided upon request.
To further quantify the effects of the Treatment factors on the response variables, in a manner that is independent of the sampling time, two additional statistics were employed - Area Under the Curve (AUC) and “Delta”. AUC was computed, using the trapezium rule, as the total area between the response curve and the linear line that connects the first and the last measured data points. The Delta statistic represents the difference between the baseline and the absolute maximum value obtained, and is expressed as a percentage of the baseline value. Statistical analyses of the treatment effects on the AUC and Delta statistics were performed using the standard least squares fit model in JMP at a significance level of 0.05. The model included: Treatment (GLY vs. SAL), Sequence (GLY-SAL and SAL-GLY), and Treatment x Sequence interaction as fixed factors, as well as Individual Animal as a random factor nested within Sequence.
Results
Intravenous glycerol inhibited the progression of empagliflozin-induced ketosis
To study the effect of glycerol as an anaplerotic substance on diabetic ketosis under SGLTi therapy, the diabetic sheep were pretreated with empagliflozin and partial insulin withdrawal (Materials & Methods; Experiment 1). As previously reported for patients with diabetes on SGLTi therapy, these sheep experienced euglycemic ketosis, as manifested by the baseline blood BHB and glucose concentrations of 2.4 mM/L and 167.4 mg/dL, respectively (Table 1). The IV GLY infusion lowered blood BHB and increased blood glucose levels significantly better than the control SAL infusion (Fig. 1). As expected for a bolus treatment with a fuel substrate, the effect on blood BHB varied substantially with the sampling time (P < 0.0001, Table 1), yielding a bell-shaped response that can be suitably quantified by analyzing the AUC and the maximal response (Delta) statistics. Based on the AUC analysis, GLY reduced ketosis significantly greater than SAL (P = 0.006, Table 2), decreasing the blood BHB substantially by 28.5% compared to only 11.5% by SAL, as shown by the Delta analysis (P = 0.03, Table 2). The initial subtle decrease in BHB values in response to SAL (Fig. 1A) can be expected as a result of blood dilution and subsequent loss of blood BHB to the urine.
Diabetic sheep were pre-induced into moderate ketosis via empagliflozin and insulin withdrawal. Bolus IV infusions of 1 L 5% glycerol (solid line) or saline (dotted line) were administered, with blood BHB (A) and glucose (B) levels monitored repeatedly over 5 h. The reduction in ketosis (BHB) peaked approximately 1 h post-treatment, while the increase in blood glucose peaked within around 4 h. The statistical analyses are presented in Table 1.
As expected for a non-energetic substance, the saline infusion did not increase blood glucose levels (Fig. 1B). Glycerol, on the other hand, increased blood glucose concentrations significantly. Yet the rise was gradual, with a peak value at 4 h post-infusion, indicative of the glycerol utilization in gluconeogenesis. These findings suggest that glycerol administration can protect against hypoglycemia without spiking the glycemic index. Notably, BHB levels declined rapidly (peaking at ~1 h), while glucose rose more gradually (peaking at ~4 h), suggesting the anti-ketotic effect of glycerol is not driven by glucose-induced insulin. Moreover, the persistence of hyperglycemia despite the in-parallel reduction in ketosis following the GLY treatment strongly indicates the absence of endogenous insulin activity.
Glycerol improved insulin resolution of severe diabetic ketosis induced by empagliflozin
Owing to the capacity of glycerol to inhibit the progression of subclinical ketosis (Fig. 1A), we next evaluated its potential benefit as an insulin-adjunctive agent in improving the resolution of severe clinical ketosis as manifested in DKA. To study this, we induced severe ketosis (BHB > 3.0 mM) in the diabetic sheep via partial insulin withdrawal and empagliflozin administration (Experiment 2). The sheep were then treated with 5 IU of insulin, infused together with 0.5 L of either saline as a control or 5% glycerol in saline. The blood BHB, glucose, and plasma NEFA responses to these treatments are graphed in Fig. 2.
Diabetic sheep were pre-induced into severe ketosis by empagliflozin administration and insulin withdrawal. Insulin (5 IU) was infused intravenously along with either 0.5 L of 5% glycerol (solid lines) or saline (dotted lines). Blood BHB (A), glucose (B), and plasma NEFA (C) levels were monitored over 5 h. Statistical analyses are detailed in Table 1.
The insulin-GLY infusion reduced blood BHB levels substantially better than the insulin-SAL infusion (P = 0.0001, Fig. 2A), as also reflected by the Delta analysis showing a respective reduction in blood BHB of 48% compared to 24% (P = 0.001, Table 2). Likewise, the AUC analysis shows a greater BHB-reducing effect for insulin-GLY compared to insulin-SAL infusion (P = 0.03, Table 2).
Similarly, the insulin-GLY infusion improved the glycemic status, substantially better than the insulin-SAL infusion (P < 0.0001, Fig. 2B), as also indicated by the AUC and Delta analysis (P = 0.004, Table 2). Notably, the addition of glycerol to insulin countered the immediate glucose-reducing effect of insulin (Fig. 2B), resulting in a gradual increase in blood glucose concentration, starting ~30 min post-infusion and peaking ~3 after. In the absence of glycerol, insulin reduced blood glucose levels much faster and for an extended duration (Fig. 2B), thereby increasing the risk of hypoglycemia.
Unlike the significantly different effects of the insulin-GLY vs. the insulin-SAL treatment on blood BHB and glucose levels, the effects on the plasma NEFA values (Fig. 2C) were not significantly different between the treatments (P = 0.2, Table 1). In fact, within one hour of the infusions, both treatments reduced the NEFA levels substantially from their basal values by (67%) and (60%), respectively, yet the effects were not significantly different between the treatments (P = 0.1, Table 2).
Glycerol did not enhance the effect of insulin on plasma NEFA levels (P = 0.2, Fig. 2C), suggesting that its effect on blood BHB (Fig. 2B) is independent of insulin.
Dietary glycerol consumption inhibited empagliflozin-induced ketosis and lipolysis
To explore the potential benefit of dietary glycerol consumption on reducing the risk for DKA under SGLTi therapy, we investigated the effect of regular supplementation of glycerol in the drinking water on ketosis in diabetic sheep maintained on a daily 10 mg dose of empagliflozin. The sheep were kept in individual pens and fed during the day only. In the evenings for a whole week, each animal was served with either 5% glycerol or water as a control for overnight drinking, then a treatment swap was implemented for each animal (Scheme 2). To assess their ketotic and energetic status, blood samples were collected daily every morning for pre-prandial BHB, glucose, and plasma NEFA measurements.
The baseline blood BHB levels were indifferent between the treatments (P = 0.2, Table 1); however, as can be seen in Fig. 3A, morning BHB values were remarkably lower in response to glycerol in the drinking water compared to regular water (P = 0.002, Table 1). Blood glucose levels were also significantly higher for the glycerol treatment (P < 0.0001, Fig. 3B).
Each diabetic sheep received overnight treatment for 7 consecutive days with either 10 L of 5% glycerol (solid black) or water (gray), followed by a crossover treatment swap for an additional 7 days. Blood samples were collected each morning, and average values of BHB (A), glucose (B), and plasma NEFA (C) were plotted. Statistical analyses by repeated-measures ANOVA revealed significant treatment effects on all three variables (Table 1).
Consistent with the low BHB levels with the glycerol consumption, the plasma NEFA concentrations were also reduced by 10% relative to the control (P = 0.009, Table 1, Fig. 3C), indicative of reduced adipose lipolysis likely due to the improved energetic status.
Discussion
DKA remains a major metabolic complication of diabetes, affecting particularly patients with poorly managed T1D or acute T2D illness with increased insulin needs. While beneficial in improving glycemic control, the implementation of SGLTi therapy for the management of diabetes requires close monitoring and careful consideration due to the increased risk for euglycemic DKA development. We postulated that prolonged urinary glucose loss by SGLTi therapy may not only lead to a deceptive need to lower insulin doses, but also deplete intracellular glucose to levels that restricts TCA-cycle oxidative capacity, both of which can enhance ketogenesis and predispose to DKA.
Blood BHB levels directly reflect the hepatic rate of ketogenesis [41] and the contribution of equimolar hydrogen ions upon release into the circulation [7]. Ketosis, therefore, is widely recognized as the direct cause of the anion-gap metabolic acidosis characteristic of DKA. Investigating the regulation of ketosis, as done here in the diabetic context, may therefore offer valuable insight into the pathophysiology and management of DKA.
Interestingly, diabetic ketosis exhibits significant metabolic resemblance to starvation ketosis [15], a condition in which we previously identified glycerol as an effective and safe therapeutic agent for ketotic sheep [40]. In the liver, phosphorylation of glycerol by glycerol kinase enables its metabolic utilization in either gluconeogenesis or glycolysis [42,43,44]. Through the glycolytic pathway, glycerol is converted to pyruvate, thereby contributing to anaplerosis through its subsequent conversion to oxaloacetate by mitochondrial pyruvate carboxylase [27, 29, 45]. By feeding the gluconeogenic pathway, glycerol not only raises blood glucose levels but also spares mitochondrial oxaloacetate and malate from being diverted toward gluconeogenic flux [39, 53–55], thereby extenuating cataplerosis [27, 46, 47]. Collectively, the enhanced anaplerotic input of oxaloacetate, coupled with reduced cataplerosis, supports greater TCA-cycle capacity for acetyl-CoA oxidation, ultimately limiting ketogenesis and the progression of ketosis [29, 45]. Notably, the absence of glycerol kinase in pancreatic β-cells precludes glycerol metabolism, resulting in virtually no direct insulin-secretagogue activity, in contrast to glucose [48, 49].
In this study, we employed a type-1-like diabetic sheep model to investigate the potential therapeutic benefits of glycerol in the context of diabetes-associated ketosis under SGLTi therapy. We first assessed the independent capacity of IV glycerol in mitigating diabetic ketosis. Upon establishing this effect, we evaluated glycerol’s potential as an insulin-adjunctive agent to enhance the resolution of severe diabetic ketosis, as manifested in DKA. Finally, we examined the feasibility of dietary glycerol supplementation as a preventive nutritional strategy to reduce predisposition to ketosis during SGLTi treatment.
We found that IV administration of glycerol effectively countered the progression of empagliflozin-induced euglycemic diabetic ketosis, as evidenced by a significant reduction in blood BHB (Fig. 1A), consistent with observations from the study on starvation ketosis [40]. As anticipated for a gluconeogenic substrate, glycerol also increased blood glucose levels; however, in a gradual manner, peaking approximately four hours post-infusion at a rate dictated by the animals’ metabolic capacity (Fig. 1B). Collectively, these findings indicate that in the diabetic ketotic state, glycerol is efficiently utilized for both pyruvate and glucose synthesis. As such, glycerol supports the reduction of ketosis by replenishing the TCA cycle. In contrast to the glycemic spiking induced by direct glucose administration [50], glycerol leads to a moderate elevation in blood glucose that is controlled by the animal’s endogenous rate of gluconeogenesis.
As an insulin-adjunctive therapy, we found that the combined insulin-glycerol infusion in diabetic sheep with empagliflozin-induced euglycemic ketosis resulted in a twofold reduction in blood BHB levels and a more rapid resolution of ketosis compared to insulin alone (Fig. 2A). Additionally, glycerol improved the glycemic profile by countering the immediate glucose-lowering effect of insulin, followed by a gradual rise in blood glucose levels (Fig. 2B).
Taken together, these observations suggest that a glycerol-based insulin adjunctive therapy enhances the insulin resolution of empagliflozin-induced severe ketosis, offering metabolic benefits of clinical relevance for DKA management. In contrast to the conventional ‘hormonal first’ approach – where insulin is administered initially and glucose is introduced significantly later to counter hypoglycemia (51), the combined ‘hormonal and energetic’ approach employed in this study addresses both ketosis and the insulin-induced hypoglycemia right from the outset. This integrated approach may improve the efficiency of DKA resolution while reducing the risk of hypoglycemic complications.
Despite the apparent improved energetic status under insulin-glycerol treatment (Fig. 2B), plasma NEFA levels remained unchanged during the few hours of the experimental timeframe (Fig. 2C). This likely reflects the differential spatiotemporal effects of glycerol. As the primary site for energy metabolism and gluconeogenesis, the liver efficiently metabolizes glycerol, accounting for the prompt changes in blood glucose and BHB levels (Figs. 1 and 2). In contrast, adipose tissue has a lower metabolic rate and limited glycerol kinase activity [51, 52], restricting the direct utilization of glycerol. Thus, glycerol’s impact on adipose tissue is likely indirect, i.e., mediated by hepatic metabolism and systemic hormonal shifts. Given that lipolysis is primarily regulated by insulin, and insulin levels were comparable across treatments, a longer duration of intervention may be necessary to observe any effects on adipose lipolysis by the broader hormonal milieu.
To shed light on the longer-term potential use of dietary glycerol to reduce the risk of DKA development, we next investigated the effects of glycerol consumption in drinking water on the energetic state of diabetic sheep induced for ketosis via empagliflozin. We found that the nightly glycerol drinking consumption substantially lowered morning blood BHB and increased glucose levels. Although ruminal digestion can metabolize glycerol [44, 53], the findings of this experiment (Fig. 3A, B) are consistent with outcomes of the IV administration (Fig. 1), as well as with other studies suggesting a substantial absorption of intact glycerol from the ruminant digestive system into the blood [43, 54].
In this longer-term intervention, the NEFA levels indeed decreased significantly with regular glycerol consumption (Fig. 3C), indicating reduced adipose lipolysis, in line with starvation ketosis studies [40, 54,55,56]. In healthy individuals, the ingestion of energy substrates, particularly carbohydrates, suppresses adipose lipolysis primarily through glucose-stimulated insulin action on adipocytes [57, 58]. However, under conditions of insulin deficiency, as in our type-I-like diabetic model, glycerol would not be expected to markedly inhibit adipose lipolysis due to the absence of insulin signaling [38]. Nonetheless, the observed effect may be attributed to a reduction in glucagon secretion via insulin-independent pathways [59, 60], leading to decreased glucagon-madiated lipolysis.
Conclusion
This study demonstrates the potential of glycerol as an anaplerotic substrate for mitigating both mild and severe diabetic ketosis independently of insulin, under SGLTi therapy in a type-1-like diabetic sheep model. Intravenous glycerol effectively reduced empagliflozin-induced diabetic ketosis. When used as an adjunct to insulin, it enhanced the resolution of severe diabetic ketosis while lowering the risk for hypoglycemia induced by insulin therapy. Furthermore, regular consumption of glycerol via drinking water provided sustained protection against SGLTi-induced ketosis, highlighting potential implications of dietary glycerol in mitigating euglycemic DKA, and in nutritional management for individuals with T1D or T2D at risk for DKA. The outcomes of this preclinical study support further investigation of glycerol in clinical and nutritional studies in the diabetic contexts of ketosis, hypoglycemia, and SGLTi therapy.
Data availability
All data supporting the findings of this study are available within the article and its supplementary materials.
References
Cryer PE. Glycemic goals in diabetes: Trade-off between glycemic control and iatrogenic hypoglycemia. Diabetes. 2014. https://doi.org/10.2337/db14-0059.
Wright LA, Hirsch IB. Non-insulin treatments for Type 1 diabetes: critical appraisal of the available evidence and insight into future directions. Diabet Med. 2019;36:665–78.
Umpierrez GE, Davis GM, Elsayed NA, Fadini GP, Galindo RJ, Hirsch IB, et al. Hyperglycemic crises in adults with diabetes: a consensus report. Diabetes Care. 2024;47:1257–75.
Weinstock RS, Xing D, Maahs DM, Michels A, Rickels MR, Peters AL et al. Severe hypoglycemia and diabetic ketoacidosis in adults with type 1 diabetes: Results from the T1D exchange clinic registry. J Clin Endocrinol Metab. 2013. https://doi.org/10.1210/jc.2013-1589.
Nyenwe EA, Kitabchi AE. The evolution of diabetic ketoacidosis: an update of its etiology, pathogenesis and management. Metabolism. 2016;65:507–21.
McGarry JD, Foster DW. Regulation of hepatic fatty acid oxidation and ketone body production. Annu Rev Biochem. 1980;49:395–420.
Halperin ML, Bear RA, Hannaford MC, Goldstein MB. Selected aspects of the pathophysiology of metabolic acidosis in diabetes mellitus. Diabetes. 1981;30:781–7.
Puchalska P, Crawford PA. Multi-dimensional Roles of Ketone Bodies in Fuel Metabolism, Signaling, and Therapeutics. Cell Metab. 2017. https://doi.org/10.1016/j.cmet.2016.12.022.
Kanikarla-Marie P, Jain SK. Hyperketonemia and ketosis increase the risk of complications in type 1 diabetes. Free Radic Biol Med. 2016;95:268–77.
Rossetti L, Smith D, Shulman GI, Papachristou D, DeFronzo RA. Correction of hyperglycemia with phlorizin normalizes tissues sensitivity to insulin in diabetic rats. J Clin Invest. 1987;79:1510–5.
Cefalo CMA, Cinti F, Moffa S, Impronta F, Sorice GP, Mezza T, et al. Sotagliflozin, the first dual SGLT inhibitor: current outlook and perspectives. Cardiovasc Diabetol. 2019;18:1–14.
Fala L. Jardiance (Empagliflozin), an SGLT2 inhibitor, receives FDA approval for the treatment of patients with type 2 diabetes. Am Heal drug benefits. 2015;8:92–5.
Santos LL, De Lima FJC, De Sousa-Rodrigues CF, Barbosa FT. Use of SGLT-2 inhibitors in the treatment of type 2 diabetes mellitus. Rev Assoc Med Bras. 2017;63:636–41.
McCrimmon RJ, Henry RR. SGLT inhibitor adjunct therapy in type 1 diabetes. Diabetologia. 2018;61:2126–33.
Danne T, Garg S, Peters AL, Buse JB, Mathieu C, Pettus JH, et al. International consensus on risk management of diabetic ketoacidosis in patients with type 1 diabetes treated with sodium-glucose cotransporter (SGLT) inhibitors. Diabetes Care. 2019;42:1147–54.
Rosenstock J, Marquard J, Laffel LM, Neubacher D, Kaspers S, Cherney DZ, et al. Empagliflozin as adjunctive to insulin therapyin type 1 diabetes: The EASE trials. Diabetes Care. 2018;41:2560–9.
Candelario N, Wykretowicz J. The DKA that wasn’t: a case of euglycemic diabetic ketoacidosis due to empagliflozin. Oxford Med Case Rep. 2016;2016:144–6.
Roach P, Skierczynski P. Euglycemic diabetic ketoacidosis in a patient with type 2 diabetes after treatment with empagliflozin. Diabetes Care. 2016;39:e3.
Modi A, Agrawal A, Morgan F. Euglycemic diabetic ketoacidosis: a review. Curr Diabetes Rev. 2016;13:315–21.
Peters AL, Buschur EO, Buse JB, Cohan P, Diner JC, Hirsch IB. Euglycemic diabetic ketoacidosis: A potential complication of treatment with sodium-glucose cotransporter 2 inhibition. Diabetes Care. 2015;38:1687–93.
Research C for DE and.. FDA revises labels of SGLT2 inhibitors for diabetes to include warnings about too much acid in the blood and serious urinary tract infections. Fda. 2020;2:1–8.
Food and Drug Administration US. FDA Drug Safety Communication: FDA revises labels of SGLT2 inhibitors for diabetes to include warnings about too much acid in the blood and serious urinary tract infections. FDA Drug Saf Commun. 2015. http://www.fda.gov/Drugs/DrugSafety/ucm475463.htm.
Fadini GP, Bonora BM, Avogaro A. SGLT2 inhibitors and diabetic ketoacidosis: data from the FDA Adverse Event Reporting System. Diabetologia. 2017;60:1385–9.
Marilly E, Cottin J, Cabrera N, Cornu C, Boussageon R, Moulin P, et al. SGLT2 inhibitors in type 2 diabetes: a systematic review and meta-analysis of cardiovascular outcome trials balancing their risks and benefits. Diabetologia. 2022;65:2000–10.
Taylor SI, Blau JE, Rother KI. SGLT2 inhibitors may predispose to ketoacidosis. J Clin Endocrinol Metab. 2015;100:2849–52.
BACH SJ, HIBBITT KG. Biochemical aspects of bovine ketosis. Biochem J. 1959;72:87–92.
White H. The role of TCA cycle anaplerosis in ketosis and fatty liver in periparturient dairy cows. Animals. 2015;5:793–802.
Laffel L. Ketone bodies: a review of physiology, pathophysiology and application of monitoring to diabetes. Diabetes Metab Res Rev. 1999;15:412–26.
Cappel DA, Deja S, Duarte JAG, Kucejova B, Iñigo M, Fletcher JA, et al. Pyruvate-carboxylase-mediated anaplerosis promotes antioxidant capacity by sustaining TCA cycle and redox metabolism in liver. Cell Metab. 2019;29:1291–.e8.
Ogawa W, Sakaguchi K. Euglycemic diabetic ketoacidosis induced by SGLT2 inhibitors: Possible mechanism and contributing factors. J Diabetes Investig. 2016;7:135–8.
Umpierrez G, Korytkowski M. Diabetic emergencies-ketoacidosis, hyperglycaemic hyperosmolar state and hypoglycaemia. Nat Rev Endocrinol. 2016;12:222–32.
du Sert NP, Hurst V, Ahluwalia A, Alam S, Avey MT, Baker M, et al. The arrive guidelines 2.0: updated guidelines for reporting animal research. PLoS Biol. 2020. https://doi.org/10.1371/journal.pbio.3000410.
del Valle HF, Lascano EC, Negroni JA. Ischemic preconditioning protection against stunning in conscious diabetic sheep: role of glucose, insulin, sarcolemmal and mitochondrial KATP channels. Cardiovasc Res. 2002;55:642–59.
Henderson GD, Read LC, Snoswell AM. Studies of liver lipids in normal, alloxan-diabetic and pregnancy-toxaemic sheep. Biochim Biophys Acta (BBA)/Lipids Lipid Metab. 1982;710:236–41.
Kazemi-Darabadi S, Sarrafzadeh-Rezaei F, Farshid AA, Baradar-Jalili R. Healing of excisional wound in alloxan induced diabetic sheep: a planimetric and histopathologic study. Vet Res Forum. 2013;4:149–55.
Leenanuruksa D, Mc Dowell GH. Experimental diabetes in lactating sheep: Effects of alloxan on plasma insulin, glucose, glucose kinetics and milk characteristics. Aust J Biol Sci. 1988. https://doi.org/10.1071/BI9880223.
McCandless EL, Woodward BA, DYE JA. Alloxan diabetes in sheep under fasting and non-fasting conditions. Am J Physiol. 1948;154:94–106.
Care D, Suppl SS. Classification and diagnosis of diabetes: standards of medical care in diabetes. Diabetes Care. 2018;41:S13–S27.
Council NR Nutrient Requirements of Small Ruminants. National Academies Press: Washington, D.C., 2007 https://doi.org/10.17226/11654.
Kalyesubula M, Rosov A, Alon T, Moallem U, Dvir H. Intravenous infusions of glycerol versus propylene glycol for the regulation of negative energy balance in sheep: a randomized trial. Animals. 2019;9. https://doi.org/10.3390/ani9100731.
Miles JM, Gerich JE. Glucose and ketone body kinetics in diabetic ketoacidosis. Clin Endocrinol Metab. 1983;12:303–19.
Lin ECC. Glycerol utilization and its regulation in mammals. Annu Rev Biochem. 1977;46:765–95.
Osman MA, Allen PS, Mehyar NA, Bobe G, Coetzee JF, Koehler KJ, et al. Acute metabolic responses of postpartal dairy cows to subcutaneous glucagon injections, oral glycerol, or both. J Dairy Sci. 2008;91:3311–22.
Werner Omazic A, Kronqvist C, Zhongyan L, Martens H, Holtenius K. The fate of glycerol entering the rumen of dairy cows and sheep. J Anim Physiol Anim Nutr (Berl). 2015;99:258–64.
Burgess N, Inigo M, Deja S, Burgess SC. Ins and outs of the TCA cycle: the central role of anaplerosis. 2021;41:19–47.
Williamson JR, Scholz R, Browning ET. Control mechanisms of gluconeogenesis and ketogenesis. II. Interactions between fatty acid oxidation and the citric acid cycle in perfused rat liver. J Biol Chem. 1969;244:4617–27.
Rui L. Energy metabolism in the liver. Compr Physiol. 2014;4:177–97.
Noel RJ, Antinozzi PA, McGarry JD, Newgard CB. Engineering of glycerol-stimulated insulin secretion in islet beta cells. J Biol Chem. 1997;272:18621–7.
Skelly RH, Wicksteed B, Antinozzi PA, Rhodes CJ. Glycerol-stimulated proinsulin biosynthesis in isolated pancreatic rat islets via adenoviral-induced expression of glycerol kinase is mediated via mitochondrial metabolism. Diabetes. 2001;50:1791–8.
Khetarpal R, Chatrath V, Kaur J, Bala A, Singh H. Impact of different intravenous fluids on blood glucose levels in nondiabetic patients undergoing elective major noncardiac surgeries. Anesth essays Res. 2016;10:425–31.
Koschinsky T, Gries FA, Herberg L. Regulation of glycerol kinase by insulin in isolated fat cells and liver of Bar Harbor Obese Mice. Diabetologia. 1971. https://doi.org/10.1007/BF01219464.
Robinson J, Newsholme EA. Glycerol kinase activities in rat heart and adipose tissue. Biochem J. 1967;104:2C–4C.
Kupczyński R, Szumny A, Wujcikowska K, Pachura N. Metabolism, ketosis treatment and milk production after using glycerol in dairy cows: A review. Animals. 2020;10:1–17.
Polizel DM, Susin I, Gentil RS, Ferreira EM, de Souza RA, Freire APA, et al. Crude glycerin decreases nonesterified fatty acid concentration in ewes during late gestation and early lactation. J Anim Sci. 2017;95:875–83.
Ferraro SM, Mendoza GD, Miranda LA, Gutiérrez CG. In vitro ruminal fermentation of glycerol, propylene glycol and molasses combined with forages and their effect on glucose and insulin blood plasma concentrations after an oral drench in sheep. Anim Feed Sci Technol. 2016;213:74–80.
Alon T, Rosov A, Lifshitz L, Dvir H, Gootwine E, Moallem U. The distinctive short-term response of late-pregnant prolific ewes to propylene glycol or glycerol drenching. J Dairy Sci. 2020;103:10245–57.
Zhao J, Wu Y, Rong X, Zheng C, Guo J. Anti-lipolysis induced by insulin in diverse pathophysiologic conditions of adipose tissue. Diabetes, Metab Syndr Obes Targets Ther. 2020;13:1575–85.
Duncan RE, Ahmadian M, Jaworski K, Sarkadi-Nagy E, Sul HS. Regulation of lipolysis in adipocytes. Annu Rev Nutr. 2007;27:79–101.
Schade DS, Woodside W, Eaton RP. The role of glucagon in the regulation of plasma lipids. Metabolism. 1979;28:874–86.
Scott RV, Bloom S. Problem or solution: the strange story of glucagon. Peptides. 2018;100:36–41.
Acknowledgements
This study was supported by the Israel Science Foundation (grant no. 1004/22).
Author information
Authors and Affiliations
Contributions
Conceptualization: HD; Data curation, JA and HD.; Formal analysis, JA and HD; Investigation, JA, MK MR, AR and HD; Resources, HD; Supervision, HD; Writing – original draft, JA and HD; Writing – review & editing, JA, MK, BS and HD.
Corresponding author
Ethics declarations
Competing interests
HD filed for a US patent application entitled “Methods for preventing or treating diabetic and non-diabetic ketoacidosis”. PCT/IL2019/051207.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
41387_2025_398_MOESM1_ESM.docx (download DOCX )
Supplementary Figure S1. Representative time course of hyperglycemia and hyperketonemia following alloxan infusion in sheep.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Asiku, J., Kalyesubula, M., Ross, M. et al. Non-carbohydrate anaplerotic therapy counters empagliflozin-induced diabetic ketosis. Nutr. Diabetes 15, 42 (2025). https://doi.org/10.1038/s41387-025-00398-3
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41387-025-00398-3