Abstract
The Social Responsiveness Scale (SRS) is an established tool for screening autism. An increasing number of studies have utilized the SRS in the general population as an outcome measure to gain insight into the etiology of autism spectrum disorder (ASD). However, SRS scores have not been well characterized in large pediatric cohorts, particularly in relation to their demographic, genetic, neuroimaging, and comorbidity profiles, or how these patterns compare to those observed in clinically diagnosed ASD. This study included 9788 non-ASD children and 182 autistic children aged 9–11 years from the Adolescent Brain Cognitive Development Study. Generalized linear mixed-effect models were applied to evaluate the associations of short social responsiveness scale (SSRS) with a spectrum of demographic, genetic, neuroimaging, and behavioral characteristics. We estimated the heritability of SSRS using a subsample of twin and sibling data. Our finding revealed that children with higher SSRS exhibited a higher male-to-female ratio. SSRS had a high heritability of 0.52 (95% CI, 0.45–0.63), and higher SSRS scores were correlated with increased polygenic risk for ASD (P < 0.001). Neuroimaging analyses identified both overlapping and unique neurobiological underpinnings, with sex-specific variations in structural and functional connectivity similar to those observed in ASD. Higher SSRS scores were linked to lower fluid intelligence, more behavioral problems, more sleep problems, and more psychotic-like symptoms. These findings highlight both the overlap and distinction between patterns reflected in SSRS scores and those observed in clinical ASD, highlighting the need for caution when interpreting findings only utilizing SRS as the outcome for autistic-like trait.
Similar content being viewed by others
Introduction
The Social Responsiveness Scale (SRS) is an instrument designed to assess social behaviors and screen autism spectrum disorder (ASD), mostly completed by caregivers and teachers [1]. In addition to its demonstrated efficacy in autism screening, SRS has been commonly used a quantitative outcome measure for autistic-like traits in epidemiological studies investigating risk factors and etiological pathways of ASD among the general population [2,3,4]. Although the quantitative traits offer promising insights into autism, the traits may lack specificity due to overlap with features of other neurodevelopmental and psychiatric conditions. It remains unclear whether this overlap represents the confounding of distinct psychiatric dimensions or reflects the well-documented comorbidity patterns between autism and other conditions such as anxiety and Attention-Deficit/Hyperactivity Disorder (ADHD) [5]. Despite the broad application of SRS, comprehensive characterization of SRS in large pediatric cohorts remains limited, leaving a gap in understanding the extent to which the SRS reflects an autism-related dimension [6,7,8,9,10]. A more systematic investigation of its genetic, neuroimaging, and behavioral profiles is therefore required.
To address this research gap, our study leveraged the extensive dataset of the Adolescent Brain Cognitive Development (ABCD) study, which includes a shortened version of SRS (short SRS, SSRS) and comprehensive phenotypic data from over 10,000 children across the United States. We primarily investigated correlates of SSRS among typically developing children. Meanwhile, we utilized a subset diagnosed with ASD as positive controls, since numerous studies and ongoing projects have referred to the SRS as indicative of ‘autistic-like traits’, a dimension present across the general population and potentially sharing etiology with ASD [11]. Given this presumption, we formulated four primary hypotheses. First, children with higher SSRS scores are likely to exhibit demographic profiles similar to those with ASD, such as a higher male-to-female ratio. Second, the traits may show a high degree of heritability and a correlation with the polygenic risk score (PRS) for ASD, an acknowledged genetic factor that explains 40–76% of the etiology [11,12,13]. Third, the traits may share similar neural correlates with ASD, such as atypical development of the frontal and temporal lobes, reduced gray and white matter volumes, and functional or structural alterations in the default mode network (DMN) [14,15,16,17]. Lastly, children with higher SSRS scores are anticipated to display a spectrum of physical and mental health comorbidities commonly associated with ASD. Through systematic examination of these hypotheses, our study aims to provide a comprehensive characterization of the SRS phenotype in children.
Methods
Study design
The ABCD Study enrolled a total of 11,876 children between 9 and 11 years of age through school systems at 21 research sites across the United States between 2016 and 2018 [18]. This cohort is comprised of over one-fifth of the population within this age range in the United States [18, 19]. This study utilized the ABCD Data Release 4.0. In this study, we excluded children who had no or invalid total score of SSRS (N = 1906), which was of our primary interest. All participants and their guardians provided informed consent [18].
This study examined the relationship between SSRS scores and various characteristics known to be associated with ASD. These characteristics include the proportion of males [20], PRS for ASD [21], structural brain alterations [22], white matter integrity [7], brain connectivity [23], cognitive function [24], behavioral problems [25], sleep disturbances [26], and psychotic-like symptoms [27].
Measures
Diagnosis of ASD
By design, the ABCD study excluded children diagnosed with “moderate or severe” ASD, intellectual disability, and major neurological conditions [18]. During recruitment, trained interviewers conducted comprehensive face-to-face assessment with the children and their guardians [28]. Diagnosis of ASD was based on a parent-reported response to the question, “Has your child been diagnosed with autism spectrum disorder?”. Additionally, parents reported whether their children with ASD were attending regular classes at school, which might reflect their disease severity.
SSRS
The SSRS is an 11-item parent-reported instrument (scored from 0–3 per item, with a total score ranging from 0–33) derived from the 65-item Social Responsiveness Scale (SRS). SSRS data were collected at one-year follow-up assessment. The SRS is a validated screening tool for ASD and could reflect disease severity [10, 12, 29, 30]. To address the right-skewed distribution of SSRS scores and explore the potential non-linear associations with outcomes, non-ASD children were categorized into four groups based on the 33rd, 66th, and 95th percentiles of their SSRS scores, referred to as Q1, Q2, Q3, and the top 5% (Figure S1). A higher score indicated more pronounced autistic-like traits. Children with ASD were classified as a distinct group.
PRS for ASD
Genotype data from the ABCD Study were sourced from saliva or blood specimens using the Affymetrix NIDA SmokeScreen Array [31]. A subset of 5807 individuals of European descent were selected based on genetic lineage. Quality control and imputation were conducted using PLINK v1.90 [32], Michigan Imputation Server [33], and Eagle v2.4, resulting in 4673 samples for analysis. To construct PRS for ASD, we incorporated data from the iPSYCH-2017 dataset including 18,382 ASD cases and 27,969 controls of European ancestry [34], using a continuous shrinkage with a global shrinkage prior of 0.01 [35]. The first ten ancestry principal components were calculated and used as covariates in the PRS-related analyses.
Neuroimage data
The ABCD teams conducted standardized preprocessing pipelines to the structural, diffusion, and resting-state functional magnetic resonance imaging (MRI) data [36], including manual quality control, reconstruction, and subcortical segmentation using the FreeSurfer v5.3 software (http://surfer.nmr.mgh.harvard.edu/) [37]. The quality control was performed following the recommended image inclusion criteria of the ABCD 4.0 (see Supplement).
For structural MRI (sMRI), high resolution T1- and T2-weighted structural MR images (1 mm isotropic, prospective motion correction) were collected and processed [36]. The regions of interested included 68 cortical thicknesses, 68 cortical areas, 68 cortical volumes, and 40 subcortical volumes defined by the Destrieux atlas [38, 39].
In the collection of diffusion tensor imaging (DTI) data, high angular resolution diffusion imaging data were acquired at a resolution of 1.7 mm isotropic using multiband acquisition (factor = 3), comprising 96 diffusion directions and four distinct b-values [36, 37]. The ABCD team employed AtlasTrack, an automated segmentation method based on a probabilistic atlas, for the labeling of major white matter tracts [40]. In this study, we studied tract-average mean diffusivity (MD) and fractional anisotropy (FA) of 35 major white tracts.
Resting-state functional MRI (fMRI) scans in children were conducted using the high-resolution imaging with a multi-band technique [37]. The standardized preprocessing procedures included registration, distortion correction, and normalization [37]. Subsequently, connectivity measures were extracted within and between networks from the parcellated cortical ribbon, as delineated by the Gordon atlas [41]. The fMRI data obtained on Philips scanners were excluded due to problems in processing, in accordance with previous studies [42]. In line with this approach, we specifically examined 36 network-level RSFC averages encompassing both intra- and inter-task-control circuits that were reported to be related to ASD and social function [43,44,45], including cingulo-opercular (CO), cingulo-parietal (CP), dorsal attention network (DAN), fronto-parietal (FP), salience network (SN), ventral attention network (VAN), DMN, and auditory network (AN).
Cognitive functions
NIH Toolbox were utilized to assess the neurocognitive performance [46]. The NIH Toolbox Cognitive Function Battery comprises seven tasks that assess different dimensions of cognitive abilities. Cognitive performance was quantified using age-corrected T scores (mean = 100, SD = 15), which reflect a comprehensive measure of intelligence quotient (IQ). These scores were further analyzed to distinguish between crystallized and fluid intelligence components [47].
Behavioral problems
Behavioral problems were evaluated using the parent/guardian-reported child behavior checklist (CBCL) [48], a comprehensive tool to evaluate internalizing and externalizing symptoms across 113 items. The CBCL generates three composite scores: internalizing, externalizing, and total syndrome [49], with higher scores indicating greater severity of symptoms. Additionally, exploratory analyses were conducted on CBCL-derived measures, including anxiety-depress, withdraw-depress, somatic, social, thought, attention, rule-breaking, and aggressive problems.
Sleep problems
Sleep problems were evaluated using the Sleep Disturbance Scale for Children (SDSC) [50], reported by parents or guardians based on observations over the previous six months. The SDSC comprises a 26-item inventory rated on a 5-point Likert-type scale, resulting in six subscales that address various sleep disturbances, including disorders of initiating and maintaining sleep, sleep breathing disorders, disorder of arousal, sleep-wake transition disorders, disorders of excessive somnolence, and sleep hyperhidrosis. The overall sum score, which ranges from 26–130, indicates the severity of sleep problems, with higher scores reflecting poorer sleep quality.
Psychotic-like symptoms
Psychotic-like symptoms in adolescents were measured using the Prodromal Questionnaire - Brief Child version (PQ-BC) [51], which includes items querying the occurrence and distress level of psychotic experiences, rated on a 5-point Likert scale. The total PQ-BC score ranges from 0–105, with scores ≥ 2 standard deviations above the mean considered indicative of significant psychotic-like symptoms.
Covariates
We consider the following covariates as confounders: age, sex (male or female), race/ethnicity (White, Black, Hispanic, Asian, or other), and family annual income (<$35,000, $35,000 ~ $75,000, $75,000 ~ $100,000, or ≧$100,000) [52]. Body mass index (BMI; kg/m2, continuous) and pubertal score were also described, given their potential association with ASD [53, 54]. The pubertal score was the average of self- and parent-reported score on a scale from 1 (prepuberty) to 5 (post puberty) [55].
Statistical analysis
To confirm SSRS as a valid measure of autistic-like traits, we assessed the prediction performance of SSRS for ASD using the receiver operating characteristic (ROC) curve and reported the area under curve (AUC), which ranged from 0–1. An AUC close to 1 indicated that SSRS would be highly predictive for ASD.
To investigate the correlation between SSRS and demographic characteristics, we applied linear, logistic, and multinomial regressions, whichever appropriate, and tested the trend associated with a higher SSRS group with weights assigned according to the median of each category. The trend test was only performed among children with no ASD diagnosis. Also, we compared the demographic characteristics between children with ASD and children in the lowest SSRS tertile (Q1) for comparison, using t-test and chi-squared test, whichever appropriate.
Heritability of SSRS
The ABCD study is characterized by a substantial number of twins and siblings, permitting family-based design to estimate heritability. The ABCD team identified the monozygotic twins, dizygotic twins, and non-twin siblings based on genetic relatedness data [56]. To identify full siblings from non-twin siblings, we limited the sample to children whose both recorded guardians were their biological parents. In this subsample of twins and full siblings, we calculated the Spearman’s rank correlation coefficients for the SSRS scores within each kinship group (monozygotic twins, dizygotic twins, and full siblings). We applied the ACE model to decompose the variance in SSRS into proportions that could be explained by additive genetics (A), common environment (C), and unique environment (E), using structural equation modeling [57]. The ACE model assumed that the genetic relatedness was 1 between monozygotic twins and 0.5 between dizygotic twins and full siblings [25]. Correlation of shared environmental effect was assumed to be 1 between each pair of twins and full siblings. Unique environment involves nonshared environmental factors as well as measurement error. In an additional analysis, we excluded full siblings for comparison. SSRS scores were log-transformed to meet normality assumptions and regressed for age, sex, and study site prior to the ACE analyses [25]. The goodness-of-fit was measured and compared by log-likelihood, chi-square, Comparative Fit Index, Tucker-Lewis Index, and Root Mean Square Error of Approximation. The model was fitted with the R package lavaan (version 0.6–18) [58].
Associations with genetic, neural, and clinical indicators
We applied generalized linear mixed-effect models to estimate the associations between SSRS group and outcomes, accounting for the nested structure (family nested within site, or family nested within MRI scanner for neuroimaging analysis) of the ABCD data [59]. Specifically, linear regression was applied for normally distributed outcomes, including the PRS for ASD, neuroimaging measures, cognitive functions, and sleep problems. Poisson regression was applied to CBCL scores that were right-skewed and passed over-dispersion. Logistic regression was applied to the dichotomized outcome, psychotic-like symptoms. The association with PRS was investigated adjusting for the first ten principal component of the ABCD genotyping data. This adjustment was applied in both the regression models and correlation analyses. The associations with neuroimaging measures and mental health problems were investigated adjusting for sex, age, race, and family income [59]. In structural MRI analysis, we additionally adjusted for the child height, T1 image signal-to-noise, and intracranial volume. In DTI analyses, we additionally adjusted for mean frame-wise displacement. In RSFC analyses, we additionally adjusted for the number of frames retained after processing. In analyses on neuroimaging measures, due to the generally small effect size and the relatively small number of children in the top 5% group after quality control, risk estimates within this group were likely to be underpowered. Thus, we were interested in both alterations in the Q3 and the top 5% group compared to the Q1 group. For significant findings within the Q3 group, we then confirmed the directionality of the risk estimates in the top 5% group. Considering potential different etiology of autistic traits between male and female [8, 20], we tested the interaction term of SSRS × sex and stratified the analyses by sex when the interaction was significant. Trend test was also performed to investigate the trend of risk estimates associated with SSRS.
Previous reports showed correlations between SRS and other psychiatric conditions [51]. In sensitivity analysis, we excluded non-ASD children that had diagnoses of other psychiatric conditions reported by parents, including attention deficit hyperactivity disorder, depression, bipolar disorder, anxiety, phobias, schizophrenia, alcohol or substance use disorder, and other psychological or psychiatric diagnoses. We performed all analyses using R software version 4.2.1. Mixed models were fitted with the package lme4 (version 1.1–31) [60]. Missing values were imputed by chained equations with package mice (version 3.15.0). In the neuroimaging analyses, to address the issue of multiple comparisons, false discovery rate (FDR) correction was applied.
Results
Of the 9970 eligible children in the ABCD study aged 9–11, 182 (1.8%) were reported to be diagnosed with ASD (87% male; Figure S1). Consistent with the eligible criteria of the ABCD study, these children with ASD showed normal cognitive function (mean NIH Toolbox total score, 99.0 [SD, 20.1], compared to 101.5 [SD, 17.7] in children with no diagnosis of ASD). The majority (N = 177 [97.3%]] were able to attend regular school. Among non-ASD children, 275 (2.8%) had NIH Toolbox total cognitive scores below 70, while among children with ASD, 10 (5.5%) scored below 70.
The SSRS had a satisfactory predictive accuracy for ASD (AUC, 0.928; Figure S2A). We categorized children with no ASD according to the 33rd, 66th, and 95th percentiles of their SSRS score (Q1, 0–1, N = 4502 [46.0%]; Q2, 2–3, N = 2409 [24.6%]; Q3, 4–11, N = 2460 [25.1%]; top 5%, 12 or higher, N = 417 [4.3%]; Figure S2B). Compared with children in the Q1 group, children with higher SSRS, as well as the children with ASD, were more likely to be male (P for trend, <0.001), had higher BMIs (P for trend, <0.001), and had parents with lower income levels (P for trend, <0.001; Table 1).
The heritability of SSRS
We hypothesized that SSRS should exhibit a comparable level of heritability to ASD, which is known to be 50–80% [61]. We identified 343 pairs of monozygotic twins, 552 pairs of dizygotic twins, and 485 pairs of full siblings. The Spearman’s ρ for SSRS score were 0.66, 0.22, and 0.24 among these groups, respectively. In the ACE model, we found no evidence of common environmental influences (C) contributing to SSRS (the estimated coefficient for the C paths was <0.001). After dropping the C paths, we yielded a model with good fit (Table S10) and estimated a genetic contribution of 52% (A, 0.52, 95% CI, 0.42–0.54), with the remaining 48% (E, 0.48, 95% CI, 0.45–0.60) of the variation in SSRS score accounted for by unique environmental factors. In the additional analysis restricted on twins only, the path estimate for A was consistent (A, 0.54, 95% CI, 0.45–0.63).
SSRS and genetic predisposition for ASD
We hypothesized that SSRS should share, at least partially, some of the polygenic risk associated with ASD. In a subsample of 4288 white children, we validated that those children with ASD had a higher average PRS for ASD compared to the Q1 group (P = 0.026; Fig. 1A). The top 5% group also showed a higher average PRS for ASD than the Q1 group (P = 0.006; Fig. 1A). An additional analysis considering SSRS score as a continuous variable showed consistent results of positive correlation with the PRS for ASD. This correlation remained significant after adjusting for the first ten ancestry principal components (Spearman’s rho = 0.059, p < 0.001; Pearson’s r = 0.008, p < 0.001; Fig. 1B).
A Distribution of PRS for ASD by SSRS categories. The non-ASD children were categorized into four groups according to the 33, 66, and 95 percentiles of their SSRS score (herein referred to as Q1, Q2, Q3, and the top 5%), where higher score indicated more prominent autistic-like traits. Children with ASD were classified as a distinct group. B The scatter plot between SSRS and PRS for ASD. A solid line indicates the fitting correlation between PRS and SSRS. SSRS was log-transformed to account for its right-skewed distribution. Group difference: ns p > 0.05, *p < 0.05, **p < 0.01. ASD autism spectrum disorder, SSRS short social responsiveness scale, ABCD adolescent brain cognitive development, PRS polygenic risk scores.
SSRS and grey matter structure, functional connectivity in the brain, and white matter integrity
We hypothesized that SSRS should exhibit similar neural correlates with ASD. Thus, we examined the neuroimaging correlates of SSRS with the sMRI, RSFC, and DTI features.
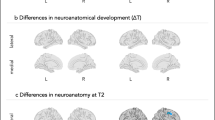
On structural features, boys with ASD showed an increased volume of the left parahippocampal cortex (t (4544) = 4.056, P < 0.001, FDR = 0.014) compared with Q1, whereas girls with ASD exhibited a decreased volume of this region (t (4132) = −2.841, P = 0.01, FDR = 0.360; P for interaction, 0.002; Fig. 2A, C). This finding was not observed in non-ASD children with higher SSRS scores. In addition, boys in Q3 exhibited lower volume of cerebral white matter in both hemispheres, but this was not observed in ASD boys, while the risk estimate for males in the top 5% group was in the opposite direction (Fig. 2A), which did not support a consistent pattern. No other differences in cortical thickness, area, volume, and subcortical volume passed the FDR correction (Table S1).
A Sex-specific associations between SSRS and structural MRI. B Sex-specific Associations between SSRS and resting-state functional connectivity. C The position of the Left Parahippocampal Cortex. Below the brain image, a color scale ranging from yellow to red indicates a gradient corresponding to the estimate of the association between Autistic Traits and Structural MRI in the highlighted region. D Indication of the 6 white matter tracts with higher MD in both children with ASD and the Q3 group measured by DTI: left superior longitudinal fasciculus, left temporal superior longitudinal fasciculus, left parietal superior longitudinal fasciculus, right superior corticostriate tract, right superior corticostriate tract - frontal cortex and right superior corticostriate tract - parietal cortex. Each cross-section is marked with different color areas, indicating regions of abnormalities within white matter integrity measured by DTI. The color bar with a gradient from white to red represented estimates of the associations between autistic traits and DTI. The non-ASD children were categorized into four groups according to the 33, 66, and 95 percentiles of their SSRS score (herein referred to as Q1, Q2, Q3, and the top 5%), where higher score indicated more prominent autistic-like traits. Children with ASD were classified as a distinct group. Bold indicates p-values that have passed FDR correction. sMRI structural magnetic resonance imaging, lh-parahippocampal left parahippocampal, RSFC resting-state functional connectivity, CP cingulo-parietal network, DMN default mode network, DTI diffusion tensor imaging, MD mean diffusivity, AN auditory network, ASD autism spectrum disorder, SSRS short social responsiveness scale.
Regarding RSFC, compared with Q1, both boys within the Q3 and those diagnosed with ASD exhibited an enhanced positive AN-CP connectivity (Q3, t (3238) = 3.930, P < 0.001, FDR = 0.02; ASD, t (3238) = 2.720, P = 0.007, FDR = 0.19; P for trend, <0.001; Fig. 2B). Also, boys with ASD showed the enhanced negative within-DMN connectivity as compared with Q1 (t (3238) = −4.595, P < 0.001, FDR = 0.004). Compared with Q1, boys in the Q2, Q3, the top 5% group showed a consistent negative pattern in within-DMN connectivity (P for trend, <0.001; Fig. 2B), though the 95% CIs for some risk estimates crossed unity. These findings were not observed in girls (Fig. 2B; Table S2).
Regarding DTI, girls with ASD had lower FA in 9 tracts and the higher MD in 28 tracts (all FDR < 0.05; Table S3, Table S4). We observed consistent findings of MD among girls in the Q3 group among 6 tracts: left superior longitudinal fasciculus, left temporal superior longitudinal fasciculus, left parietal superior longitudinal fasciculus, right superior corticostriate tract, right superior corticostriate tract - frontal cortex and right superior corticostriate tract - parietal cortex (Fig. 2D, Table S3). Point estimates of associations with these tracts among the top 5% group showed a consistent direction, while the 95% CIs crossed unity. No difference in FA or MD was observed in autistic male compared to the Q1 group (all FDR > 0.05).
SSRS and cognitive ability, behavioral problems, sleep problem, and psychotic-like symptoms
To assess whether sex-specific analyses were warranted, we tested for interaction between sex and SSRS on each outcome of interest. Significant sex-by-SSRS interactions were observed only for behavioral problems, whereas no significant interactions were found for cognitive ability, sleep problems, or psychotic-like symptoms (Table S5).
Children with higher SSRS exhibited slightly lower cognitive ability as measured by the NIH Toolbox (mean ± SD for Q1, Q2, Q3, and the top 5% group, 102.8 ± 17.1, 102.4 ± 18.3, 99.0 ± 17.9, 97.7 ± 18.0, respectively; mean ± SD for children with ASD, 99.0 ± 20.1). Adjusting for age, sex, race, and family income, we still observed lower cognitive ability in children in the Q3 group (β, −2.29; 95% CI, −3.09 to −1.48), top 5% group (β, −3.56; 95% CI, −5.18 to −1.95), and in children with ASD (β, −2.28; 95% CI −4.72–0.15; P for trend among the non-ASD children, <0.001; Fig. 3A). Specifically, this trend was consistently observed in fluid intelligence, while children with ASD showed comparable crystallized intelligence with Q1 (Fig. 3A; Figure S3A).
Forest plot depicting results from regression modelling of the association between the SSRS and (A) cognition ability, (B) sleep problem, (C) psychotic-like symptoms, and (D) behavioral problems. All analyses were adjusted for age, sex, race, and family income. P for trend shows results within the non-ASD groups. The non-ASD children were categorized into four groups according to the 33, 66, and 95 percentiles of their SSRS score (herein referred to as Q1, Q2, Q3, and the top 5%), where higher score indicated more prominent autistic-like traits. Children with ASD were classified as a distinct group. CBCL child behavior checklist, SDSC sleep disturbance scale for children, PQ-BC prodromal questionnaire - brief child version, ASD autism spectrum disorder, SSRS short social responsiveness scale, N number, SD standard deviation, IQR interquartile range, OR odds ratio.
Behavioral problems were more prevalent in children with higher SSRS score. We found interaction between SSRS and sex in this association (P for interaction, 0.037; Table S5). Compared to children in the lowest SSRS tertile, children with higher SSRS and children with ASD had higher mean total CBCL scores in both girls (mean ratio [95% CI] for Q2, Q3, top 5%, and children with ASD, 1.45 [1.38–1.52], 2.32 [2.20–2.43], 3.53, [3.20–3.90], and 3.13 [2.71–3.61], respectively; P for trend, <0.001; Fig. 3D) and boys (mean ratio [95% CI] for Q2, Q3, top 5%, and children with ASD, 1.50 [1.43–1.56], 2.17 [2.08–2.27], 3.26, [3.04–3.50], and 2.64 [2.42–2.88], respectively; P for trend, <0.001; Fig. 3D). The associations were consistent in both internalizing and externalizing syndrome scales (Fig. 3D). Specifically, we found that the association between externalizing syndrome and SSRS appealed to be more prominent in male children (Fig. 3D). Consistently, higher SSRS was associated with increased mean scores among all eight syndrome subscales of CBCL (Figure S3B).
Children with higher SSRS score and children diagnosed with ASD exhibited more sleep problems, measured by the SDSC scale, compared to children in the lowest SSRS tertile (β [95% CI] for Q2, Q3, top 5%, and children with ASD, 0.05 [0.04–0.06], 0.11 [0.10–0.12], 0.21 [0.20–0.23], and 0.14 [0.11–0.16], respectively; P for trend, <0.001; Fig. 3B). Specifically, this trend was consistently observed in all six subscales of SDSC (Figure S3C).
Children with higher SSRS score and children diagnosed with ASD were more likely to have psychotic-like symptoms, measured by the PQ-BC scale, compared to children in the lowest SSRS tertile (OR [95% CI] for Q2, Q3, top 5%, and children with ASD, 1.24 [0.97–1.58], 1.87 [1.50–2.35], 2.45 [1.68–3.58], and 4.46 [2.72–7.31], respectively; P for trend, <0.001; Fig. 3C).
Sensitivity analyses
After excluding children with any psychiatric diagnosis from the non-ASD group, the associations of SSRS scores problems were generally consistent, while the associations with neuroimaging measures attenuated. (Figure S4; Table S6–9).
Discussion
This study systematically characterized the SSRS across demographic, genetic, neuroimaging, and behavioral domains in a pediatric cohort. Our findings revealed several key patterns. First, our data reveal a consistent male-to-female disparity with higher SSRS scores more likely to be in males. Second, the heritability of SSRS was estimated at approximately 52%. Higher SSRS was correlated with increased polygenic risks for ASD. Third, neuroimaging analyses identified both overlapping and unique neurobiological underpinnings between high SSRS score and ASD, with sex-specific variations in structural and functional connectivity mirroring those seen in ASD. Fourth, children with higher SSRS exhibited a broad spectrum of neurodevelopmental and mental health problems. These findings showed that while correlates of SSRS showed a similar pattern to ASD, the complex non-ASD-specific cognitive and behavioral symptoms reflected in SSRS suggest caution when interpreting SRS as a continuous measure of autistic-like traits in general population.
ASD is more frequently diagnosed in males, a pattern that is consistent to the increasing male-to-female ratio we observed with rising SSRS scores. This consistent sex disparity suggests possible differences in the pathogenesis of ASD across sexes. Moreover, there is growing evidence that this disparity may be partially due to the underdiagnosis of ASD in females, attributed to camouflaging behaviors. Camouflaging behaviors refer to behavioral coping strategies used by individuals, particularly females, to conceal autistic traits in social situations, making these traits less noticeable to others. Examples include imitating social gestures, suppressing autistic-related behaviors, or actively rehearsing social interactions [62]. Such behaviors can lead to misdiagnosis or delayed diagnosis in females, thereby contributing to the observed sex disparities in ASD prevalence. The similar patterns of sex disparity linked with higher SSRS scores in our study may also reflect underlying, sex-specific expressions. Also, we could not rule out that SSRS might socialization of a typically masculine nature, which could be more pronounced among males. This, in turn, might explain why a higher proportion of males exhibit elevated SSRS scores.
The heritability of ASD is estimated at 50–80% [21, 61], reflecting a significant genetic contribution to its etiology. This aligns with earlier research that attributes high heritability to autistic-like traits, ranging from 0.40–0.76 [11, 12], one measured by SRS and the other by another screening questionnaire for ASD (Autism-Tics, Attention-Deficit/Hyperactivity Disorder, and Other Comorbidities Inventory). Our study corroborates these findings, showing that the heritability of SSRS is approximately 52%. We observed a statistically significant, though modest, positive correlation. This correlation suggests that genetic loci implicated in ASD may also contribute to individual differences in social responsiveness, particularly in those with elevated SSRS scores. Although the effect size of this association was modest, such small yet significant associations are common in polygenic risk studies of dimensional behavioral traits and reflect the complex, polygenic architecture of social responsiveness [63, 64]. This genetic overlap supports the notion of a continuum in social responsiveness traits, ranging from typical variation to clinical ASD, and suggests that shared biological pathways may underlie both clinical and subclinical manifestations. Such findings contribute to a dimensional perspective on autistic-like traits.
Neuroimaging studies focusing on the “social brain” have identified structural and functional alterations associated with ASD [24, 65], particularly in the insula, anterior cingulate cortex/medial prefrontal cortex, and DMN, as supported by recent meta-analyses [17]. Similarly, investigations into autistic-like traits using various behavioral and cognitive assessment scales have revealed neuroanatomical changes, such as reductions in global brain volume, cerebellar size, and cortical metrics including gyrification, thickness, and surface area [7, 66]. However, these studies have typically been limited by small sample sizes, often involving fewer than 50 participants. In this study, our large-cohort study identified specific structural changes in males, such as decreased cerebral white matter volume in boys with higher SSRS scores and increased volume of the left parahippocampal gyrus in boys with ASD, both linked to challenges in social interaction and paralleled in neuroimaging profiles of individuals with ASD [67,68,69]. Other findings further refine the understanding of structural connectivity in ASD. One case-control study noted a decrease in FA of the frontal aslant tract across typical development, ASD-sibling, and ASD groups [70]. In this study, we observed distinct alterations in MD of six fiber tracts exclusively in females with higher SSRS scores, which align with changes seen in the ASD group. Notably, despite previous reports suggesting the involvement of corpus callosum connectivity in ASD pathogenesis [22, 43, 71, 72], our study did not observe a significant association between corpus callosum measures and SSRS scores. Regarding functional connectivity, prior research has been inconsistent, likely due to factors such as small sample sizes, variable ages at measurement, and ASD heterogeneity [23, 73,74,75,76,77,78]. Our study contributes to this domain by demonstrating that both the group with higher SSRS scores and the ASD group exhibit enhanced positive connectivity within the AN-CP connectivity and enhanced negative connectivity within the DMN connectivity, exclusively in males. These findings underscore potential shared and unique neuroimaging biomarkers between SSRS phenotype and ASD, suggesting that underlying neural mechanisms may differ by sex. In the sensitivity analysis excluding children with other neurodevelopmental disorders, the associations with neuroimaging measures were attenuated, likely due to the reduced sample size in the analysis. This highlights the need for cautious interpretation, as the observed associations could also potentially be influenced by comorbid mental health issues associated with SSRS scores. This complexity points to the necessity of larger, more nuanced studies to disentangle these interactions and further our understanding of ASD and related conditions.
In line with previous reports, our findings revealed that SSRS scores are associated a broad spectrum of mental health problems frequently observed in autistic individuals. These include variations in cognitive performance [79], behavioral problems [80], sleep problems [81], and psychotic-like experiences [51]. Prior studies suggested overlaps of both phenotypic and genetic, between autistic-like traits and these mental health issues [79, 82]. A particular observation from our data is the association of higher SSRS scores with reduced fluid intelligence, whereas no consistent pattern emerged with crystallized intelligence. This divergence may suggest that individuals with high SSRS scores might leverage their verbal abilities as a compensatory strategy for deficits in cognitive flexibility [83]. In addition, it is noteworthy that the SSRS not only reflects a dimension of sociability but also other mental health conditions. Studies using SSRS as an outcome to gain insights into ASD should be cautious, as the outcome may lack specificity. Adjustments for other comorbid mental health issues would therefore be recommended.
Strengths and limitations
This study leverages the extensive dataset of the ABCD study, notable for its large sample size and comprehensive multimodal data drawn from a demographically diverse cohort. These attributes enable a detailed characterization of SRS phenotype during early adolescence. Additionally, including autistic individuals from the same cohort as a positive control enriches our comparative analysis between children with high SSRS scores in the general population and those with a clinical diagnosis. Furthermore, our exploration of sex differences across various traits offers novel insights, suggesting that the underlying neurobiological mechanisms might differ between high SSRS scores and ASD, potentially informing sex-specific approaches in future research and interventions.
However, several limitations must be considered when interpreting our findings. First, the ABCD study’s exclusion of children with intellectual disabilities may introduce a selection bias, as the enrolled autistic participants generally did not have intellectual disabilities. This exclusion might also explain why the median SSRS scores among children with ASD were lower than those in the top 5% of the general population without an ASD diagnosis. Secondly, reliance on parent-reported ASD diagnoses could lead to misclassification; yet, the reported ASD incidence of 1.8%—considering the exclusion of severe cases—aligns with the general population incidence of 1–2% [84]. Thirdly, the SSRS’s association with other psychiatric conditions such as attention deficit hyperactivity disorder could suggest overlapping patterns that might not be exclusive to autistic-like traits. This overlap was controlled for in further analyses, which excluded diagnosed psychiatric problems, yielding consistent findings. Additionally, the study’s cross-sectional design limits causal inference. While sex-stratified analyses revealed important insights, they were constrained by the imbalanced distribution of high SSRS scores and ASD diagnoses across sexes. The reduced sample size within each sex group, particularly among females, may have further limited the statistical power to detect small or moderate effects, especially for neuroimaging outcomes. Future studies with larger samples are warranted to confirm and extend these findings. Finally, it is important to emphasize that the primary focus of this study was on the SSRS as a dimensional measure of social function in the general pediatric population. While SSRS scores are often interpreted as proxies for autistic-like traits, the scale captures a broader spectrum of social function that may not be specific to ASD. As a shortened version of the full SRS, the SSRS may also reduce measurement granularity, and the implications of its results should be interpreted with caution.
Conclusion
This study, using the extensive dataset from the ABCD study, explored demographic, genetic, neural, and behavioral correlates of the SRS phenotype within the general population. Commonly used as a measure of autistic-like traits, the SRS phenotype exhibited both overlapping and distinct features compared to ASD, highlighting the need for caution when interpreting findings only utilizing SRS as the outcome for autistic-like trait.
Data availability
The data that support the findings of this study are openly available in the ABCD Dataset Data Release 4.0 at https://nda.nih.gov/abcd. The scripts used for these analyses will be made available upon publication at the following https://doi.org/10.15154/rbtk-xg97.
References
Constantino JN, Przybeck T, Friesen D, Todd RD. Reciprocal social behavior in children with and without pervasive developmental disorders. J Dev Behav Pediatr. 2000;21:2–11.
Bölte S, Poustka F, Constantino JN. Assessing autistic traits: cross-cultural validation of the social responsiveness scale (SRS). Autism Res. 2008;1:354–63.
Bölte S, Westerwald E, Holtmann M, Freitag C, Poustka F. Autistic traits and autism spectrum disorders: The clinical validity of two measures presuming a continuum of social communication skills. J Autism Dev Disord. 2011;41:66–72.
Reiersen AM, Constantino JN, Volk HE, Todd RD. Autistic traits in a population-based ADHD twin sample. J Child Psychol Psychiatry. 2007;48:464–72.
Lyall K. What are quantitative traits and how can they be used in autism research?. Autism Res. 2023;16:1289–98.
Sharp TH, Elsabbagh M, Pickles A, Bedford R. The subcortical correlates of autistic traits in school-age children: a Population-Based Neuroimaging study. Mol Autism. 2023;14:6.
Chien YL, Chen YJ, Hsu YC, Tseng WI, Gau SS. Altered white-matter integrity in unaffected siblings of probands with autism spectrum disorders. Hum Brain Mapp. 2017;38:6053–67.
Stroth S, Tauscher J, Wolff N, Küpper C, Poustka L, Roepke S, et al. Phenotypic differences between female and male individuals with suspicion of autism spectrum disorder. Mol Autism. 2022;13:11.
Hyseni F, Blanken LME, Muetzel R, Verhulst FC, Tiemeier H, White T. Autistic traits and neuropsychological performance in 6–10-year-old children: a Population-Based study. Child Neuropsychol. 2019;25:352–69.
Hus V, Bishop S, Gotham K, Huerta M, Lord C. Factors influencing scores on the social responsiveness scale. J Child Psychol Psychiatry. 2013;54:216–24.
Taylor MJ, Rosenqvist MA, Larsson H, Gillberg C, D’Onofrio BM, Lichtenstein P, et al. Etiology of autism spectrum disorders and autistic traits over time. JAMA Psychiatry. 2020;77:936–43.
Constantino JN, Todd RD. Autistic traits in the general population: a Twin study. Arch Gen Psychiatry. 2003;60:524–30.
de Wit MM, Morgan MJ, Libedinsky I, Austerberry C, Begeer S, Abdellaoui A, et al. A systematic review and meta-analysis: research using the autism polygenic score. medRxiv [Preprint]. 2024. Available from: https://doi.org/10.1101/2024.03.08.24303918.
Hashem S, Nisar S, Bhat AA, Yadav SK, Azeem MW, Bagga P, et al. Genetics of structural and functional brain changes in autism spectrum disorder. Transl Psychiatry. 2020;10:229.
Durkut M, Blok E, Suleri A, White T. The longitudinal bidirectional relationship between autistic traits and brain morphology from childhood to adolescence: a Population-Based Cohort study. Mol Autism. 2022;13:31.
Nickl-Jockschat T, Habel U, Michel TM, Manning J, Laird AR, Fox PT, et al. Brain structure anomalies in autism spectrum disorder–a meta-analysis of VBM studies using anatomic likelihood estimation. Hum Brain Mapp. 2012;33:1470–89.
Guo Z, Tang X, Xiao S, Yan H, Sun S, Yang Z, et al. Systematic review and meta-analysis: multimodal functional and anatomical neural alterations in autism spectrum disorder. Mol Autism. 2024;15:16.
Garavan H, Bartsch H, Conway K, Decastro A, Goldstein RZ, Heeringa S, et al. Recruiting the ABCD sample: design considerations and procedures. Dev Cogn Neurosci. 2018;32:16–22.
Volkow ND, Koob GF, Croyle RT, Bianchi DW, Gordon JA, Koroshetz WJ, et al. The conception of the ABCD study: from substance use to a broad NIH collaboration. Dev Cogn Neurosci. 2018;32:4–7.
Calderoni S. Sex/gender differences in children with autism spectrum disorder: a brief overview on epidemiology, symptom profile, and neuroanatomy. J Neurosci Res. 2023;101:739–50.
Sandin S, Lichtenstein P, Kuja-Halkola R, Larsson H, Hultman CM, Reichenberg A. The familial risk of autism. Jama. 2014;311:1770–7.
Prigge MBD, Lange N, Bigler ED, King JB, Dean DC 3rd, et al. A 16-year study of longitudinal volumetric brain development in males with autism. Neuroimage. 2021;236:118067.
Long Z, Duan X, Mantini D, Chen H. Alteration of functional connectivity in autism spectrum disorder: effect of age and anatomical distance. Sci Rep. 2016;6:26527.
Pelphrey K, Adolphs R, Morris JP. Neuroanatomical substrates of social cognition dysfunction in autism. Ment Retard Dev Disabil Res Rev. 2004;10:259–71.
Assary E, Oginni OA, Morneau-Vaillancourt G, Krebs G, Peel AJ, Palaiologou E, et al. Genetics of environmental sensitivity and its association with variations in emotional problems, autistic traits, and wellbeing. Mol Psychiatry. 2024;29:2438–46.
Cohen S, Conduit R, Lockley SW, Rajaratnam SM, Cornish KM. The relationship between sleep and behavior in Autism Spectrum Disorder (ASD): a review. J Neurodev Disord. 2014;6:1–10.
Kiyono T, Morita M, Morishima R, Fujikawa S, Yamasaki S, Nishida A, et al. The prevalence of psychotic experiences in autism spectrum disorder and autistic traits: a systematic review and meta-analysis. Schizophr Bull Open. 2020;1:sgaa046.
Barch DM, Albaugh MD, Avenevoli S, Chang L, Clark DB, Glantz MD, et al. Demographic, physical and mental health assessments in the Adolescent Brain and Cognitive Development study: rationale and description. Dev Cogn Neurosci. 2018;32:55–66.
Schwarzlose RF, Tillman R, Hoyniak CP, Luby JL, Barch DM. Sensory over-responsivity: a feature of childhood psychiatric illness associated with altered functional connectivity of sensory networks. Biol Psychiatry. 2023;93:92–101.
Cholemkery H, Kitzerow J, Rohrmann S, Freitag CM. Validity of the social responsiveness scale to differentiate between autism spectrum disorders and disruptive behaviour disorders. Eur Child Adolesc Psychiatry. 2014;23:81–93.
Baurley JW, Edlund CK, Pardamean CI, Conti DV, Bergen AW. Smokescreen: a targeted genotyping array for addiction research. BMC Genom. 2016;17:145.
Purcell S, Neale B, Todd-Brown K, Thomas L, Ferreira MA, Bender D, et al. PLINK: a tool set for whole-genome association and population-based linkage analyses. Am J Hum Genet. 2007;81:559–75.
Das S, Forer L, Schönherr S, Sidore C, Locke AE, Kwong A, et al. Next-generation genotype imputation service and methods. Nat Genet. 2016;48:1284–7.
Grove J, Ripke S, Als TD, Mattheisen M, Walters RK, Won H, et al. Identification of common genetic risk variants for autism spectrum disorder. Nat Genet. 2019;51:431–44.
Ge T, Chen CY, Ni Y, Feng YA, Smoller JW. Polygenic prediction via Bayesian regression and continuous shrinkage priors. Nat Commun. 2019;10:1776.
Casey BJ, Cannonier T, Conley MI, Cohen AO, Barch DM, Heitzeg MM, et al. The Adolescent Brain Cognitive Development (ABCD) study: imaging acquisition across 21 sites. Dev Cogn Neurosci. 2018;32:43–54.
Hagler DJ Jr, Hatton S, Cornejo MD, Makowski C, Fair DA, Dick AS, et al. Image processing and analysis methods for the Adolescent Brain Cognitive Development study. Neuroimage. 2019;202:116091.
Dale AM, Fischl B, Sereno MI. Cortical surface-based analysis. I. Segmentation and surface reconstruction. Neuroimage. 1999;9:179–94.
Destrieux C, Fischl B, Dale A, Halgren E. Automatic parcellation of human cortical gyri and sulci using standard anatomical nomenclature. Neuroimage. 2010;53:1–15.
Hagler DJ Jr, Ahmadi ME, Kuperman J, Holland D, McDonald CR, Halgren E, et al. Automated white-matter tractography using a probabilistic diffusion tensor atlas: application to temporal lobe epilepsy. Hum Brain Mapp. 2009;30:1535–47.
Gordon EM, Laumann TO, Adeyemo B, Huckins JF, Kelley WM, Petersen SE. Generation and evaluation of a cortical area parcellation from resting-state correlations. Cereb Cortex. 2016;26:288–303.
Pagliaccio D, Durham K, Fitzgerald KD, Marsh R. Obsessive-compulsive symptoms among children in the Adolescent Brain and Cognitive Development study: clinical, cognitive, and brain connectivity correlates. Biol Psychiatry Cogn Neurosci Neuroimaging. 2021;6:399–409.
Li Q, Becker B, Jiang X, Zhao Z, Zhang Q, Yao S, et al. Decreased interhemispheric functional connectivity rather than corpus callosum volume as a potential biomarker for autism spectrum disorder. Cortex. 2019;119:258–66.
Wang J, Fu K, Chen L, Duan X, Guo X, Chen H, et al. Increased gray matter volume and resting-state functional connectivity in somatosensory cortex and their relationship with Autistic symptoms in young boys with autism spectrum disorder. Front Physiol. 2017;8:588.
Setton R, Mwilambwe-Tshilobo L, Sheldon S, Turner GR, Spreng RN. Hippocampus and temporal pole functional connectivity is associated with age and individual differences in autobiographical memory. Proc Natl Acad Sci USA. 2022;119:e2203039119.
Weintraub S, Dikmen SS, Heaton RK, Tulsky DS, Zelazo PD, Bauer PJ, et al. Cognition assessment using the NIH toolbox. Neurology. 2013;80:S54–64.
Akshoomoff N, Beaumont JL, Bauer PJ, Dikmen SS, Gershon RC, Mungas D, et al. VIII. NIH toolbox Cognition Battery (CB): composite scores of crystallized, fluid, and overall cognition. Monogr Soc Res Child Dev. 2013;78:119–32.
Achenbach TM, Rescorla LA Manual for ASEBA school-age forms & profiles. Burlington, VT: University of Vermont Research Center for Children, Youth, & Families; 2001.
Mansolf M, Blackwell CK, Cummings P, Choi S, Cella D. Linking the child behavior checklist to the Strengths and Difficulties Questionnaire. Psychol Assess. 2022;34:233–46.
Bruni O, Ottaviano S, Guidetti V, Romoli M, Innocenzi M, Cortesi F, et al. The Sleep Disturbance Scale for Children (SDSC). Construction and validation of an instrument to evaluate sleep disturbances in childhood and adolescence. J Sleep Res. 1996;5:251–61.
Jutla A, Donohue MR, Veenstra-VanderWeele J, Foss-Feig JH. Reported autism diagnosis is associated with psychotic-like symptoms in the Adolescent Brain Cognitive Development cohort. Eur Child Adolesc Psychiatry. 2022;31:1–10.
Petersen AC, Crockett L, Richards M, Boxer A. A self-report measure of pubertal status: reliability, validity, and initial norms. J Youth Adolesc. 1988;17:117–33.
Corbett BA, Muscatello RA, Horrocks BK, Klemencic ME, Tanguturi Y. Differences in body mass index (BMI) in early adolescents with autism spectrum disorder compared to youth with typical development. J Autism Dev Disord. 2021;51:2790–9.
Muscatello RA, Corbett BA. Comparing the effects of age, pubertal development, and symptom profile on cortisol rhythm in children and adolescents with autism spectrum disorder. Autism Res. 2018;11:110–20.
MacSweeney N, Allardyce J, Edmondson-Stait A, Shen X, Casey H, Chan SW, et al. The role of brain structure in the association between pubertal timing and depression risk in an early adolescent sample (the ABCD study®): a registered report. Dev Cogn Neurosci. 2023;60:101223.
Fan CC, Loughnan R, Wilson S, Hewitt JK, Agrawal A, Dowling G, et al. Genotype data and derived genetic instruments of Adolescent Brain Cognitive Development study® for better understanding of human brain development. Behav Genet. 2023;53:159–68.
Yuan K-H, Bentler PM. 10 structural equation modeling. Handb Stat. 2006;26:297–358.
Marsh H, Alamer A. When and how to use set-exploratory structural equation modelling to test structural models: a tutorial using the R package lavaan. Br J Math Stat Psychol. 2024;77:459–76.
Pagliaccio D, Alqueza KL, Marsh R, Auerbach RP. Brain volume abnormalities in youth at high risk for depression: Adolescent Brain and Cognitive Development study. J Am Acad Child Adolesc Psychiatry. 2020;59:1178–88.
Bates D, Mächler M, Bolker B, Walker S. Fitting linear mixed-effects models using lme4. J Stat Softw. 2015;67:1–48.
Bai D, Yip BHK, Windham GC, Sourander A, Francis R, Yoffe R, et al. Association of genetic and environmental factors with autism in a 5-country cohort. JAMA Psychiatry. 2019;76:1035–43.
Green RM, Travers AM, Howe Y, McDougle CJ. Women and autism spectrum disorder: diagnosis and implications for treatment of adolescents and adults. Curr Psychiatry Rep. 2019;21:22.
Rodrigue AL, Mathias SR, Knowles EEM, Mollon J, Almasy L, Schultz L, et al. Specificity of psychiatric polygenic risk scores and their effects on associated risk phenotypes. Biol Psychiatry Glob Open Sci. 2023;3:519–29.
Shen X, Howard DM, Adams MJ, Hill WD, Clarke TK, Deary IJ, et al. A phenome-wide association and Mendelian Randomisation study of polygenic risk for depression in UK Biobank. Nat Commun. 2020;11:2301.
Brothers L. The social brain: a project for integrating primate behavior and neuropsychology in a new domain. Concepts Neurosci. 1990;1:27–51.
Shiota Y, Soma D, Hirosawa T, Yoshimura Y, Tanaka S, Hasegawa C, et al. Alterations in brain networks in children with sub-threshold autism spectrum disorder: a Magnetoencephalography study. Front Psychiatry. 2022;13:959763.
Xu MX, Ju XD. Abnormal brain structure is associated with social and communication deficits in children with autism spectrum disorder: a voxel-based morphometry analysis. Brain Sci. 2023;13:779.
Geng F, Xu W, Riggins T. Interactions between the hippocampus and fronto-parietal regions during memory encoding in early childhood. Hippocampus. 2022;32:108–20.
Quian Quiroga R. How are memories stored in the human hippocampus?. Trends Cogn Sci. 2021;25:425–6.
Lo YC, Chen YJ, Hsu YC, Chien YL, Gau SS, Tseng WI. Altered frontal aslant tracts as a heritable neural basis of social communication deficits in autism spectrum disorder: a sibling study using tract-based automatic analysis. Autism Res. 2019;12:225–38.
Paul LK, Corsello C, Kennedy DP, Adolphs R. Agenesis of the corpus callosum and autism: a comprehensive comparison. Brain. 2014;137:1813–29.
Minnigulova A, Davydova E, Pereverzeva D, Sorokin A, Tyushkevich S, Mamokhina U, et al. Corpus callosum organization and its implication to core and co-occurring symptoms of autism spectrum disorder. Brain Struct Funct. 2023;228:775–85.
Woodward ND, Giraldo-Chica M, Rogers B, Cascio CJ. Thalamocortical dysconnectivity in autism spectrum disorder: an analysis of the autism brain imaging data exchange. Biol Psychiatry Cogn Neurosci Neuroimaging. 2017;2:76–84.
Lee Y, Park BY, James O, Kim SG, Park H. Autism spectrum disorder related functional connectivity changes in the language network in children, adolescents and adults. Front Hum Neurosci. 2017;11:418.
Farrant K, Uddin LQ. Atypical developmental of dorsal and ventral attention networks in Autism. Dev Sci. 2016;19:550–63.
Dajani DR, Uddin LQ. Local brain connectivity across development in autism spectrum disorder: a cross-sectional investigation. Autism Res. 2016;9:43–54.
Burrows CA, Laird AR, Uddin LQ. Functional connectivity of brain regions for self- and other-evaluation in children, adolescents and adults with autism. Dev Sci. 2016;19:564–80.
Lawrence KE, Hernandez LM, Bookheimer SY, Dapretto M. Atypical longitudinal development of functional connectivity in adolescents with autism spectrum disorder. Autism Res. 2019;12:53–65.
Skjegstad CL, Trevor C, Swanborough H, Roswandowitz C, Mokros A, Habermeyer E, et al. Psychopathic and autistic traits differentially influence the neural mechanisms of social cognition from communication signals. Transl Psychiatry. 2022;12:494.
Arias AA, Rea MM, Adler EJ, Haendel AD, Van Hecke AV. Utilizing the Child Behavior Checklist (CBCL) as an autism spectrum disorder preliminary screener and outcome measure for the PEERS® intervention for autistic adolescents. J Autism Dev Disord. 2022;52:2061–74.
Chen H, Yang T, Chen J, Chen L, Dai Y, Zhang J, et al. Sleep problems in children with autism spectrum disorder: a multicenter survey. BMC Psychiatry. 2021;21:406.
Hope S, Shadrin AA, Lin A, Bahrami S, Rødevand L, Frei O, et al. Bidirectional genetic overlap between autism spectrum disorder and cognitive traits. Transl Psychiatry. 2023;13:295.
Solomon M, Gordon A, Iosif AM, Geddert R, Krug MK, Mundy P, et al. Using the NIH toolbox to assess cognition in adolescents and young adults with autism spectrum disorders. Autism Res. 2021;14:500–11.
Salari N, Rasoulpoor S, Rasoulpoor S, Shohaimi S, Jafarpour S, Abdoli N, et al. The global prevalence of autism spectrum disorder: a comprehensive systematic review and meta-analysis. Ital J Pediatr. 2022;48:112.
Acknowledgements
Data used in the preparation of this article were obtained from the Adolescent Brain Cognitive Development (ABCD) Study (https://abcdstudy.org), held in the NIMH Data Archive (NDA). This is a multisite, longitudinal study designed to recruit more than 10,000 children age 9–10 and follow them over 10 years into early adulthood. We appreciate the children and their families who have participated in the study. The ABCD Study is supported by the National Institutes of Health and additional federal partners under award numbers U01DA041048,U01DA050989,U01DA051016,U01DA041022,U01DA051018,U01DA051037,U01DA050987,U01DA041174,U01DA041106,U01DA041117,U01DA041028,U01DA041134,U01DA050988,U01DA051039,U01DA041156,U01DA041025,U01DA041120,U01DA051038,U01DA041148,U01DA041093,U01DA041089,U24DA041123,U24DA041147.A full list of supporters is available at https://abcdstudy.org/federal-partners.html. A listing of participating sites and a complete listing of the study investigators can be found at https://abcdstudy.org/consortium_members/. ABCD consortium investigators designed and implemented the study and/or provided data but did not necessarily participate in the analysis or writing of this report. This manuscript reflects the views of the authors and may not reflect the opinions or views of the NIH or ABCD consortium investigators. The ABCD data repository grows and changes over time. The ABCD data used in this report came from [NIMH Data Archive Digital Object Identifier (DOI)]. DOIs can be found at https://doi.org/10.15154/rbtk-xg97. The computations in this paper were run on the Siyuan-1 cluster supported by the Center for High Performance Computing at Shanghai Jiao Tong University.
Funding
This study was supported by grants from the National Key Research and Development Program of China (No. 2023YFE0109700), the China Brain Initiative Grant (STI2030-Major Projects 2021ZD0200800), the National Natural Science Foundation of China (82125032, 81930095, 81761128035, and 82204048), the Science and Technology Commission of Shanghai Municipality (23Y21900500, 19410713500, and 2018SHZDZX01), the Shanghai Municipal Commission of Health and Family Planning (GWV-11.1–34, 2020CXJQ01, and 2018YJRC03), the Shanghai Clinical Key Subject Construction Project (shslczdzk02902), Innovative research team of high-level local universities in Shanghai (SHSMU-ZDCX20211100), and the Guangdong Key Project (2018B030335001).
Author information
Authors and Affiliations
Contributions
LKH, QL, TR and FL designed the study. LKH, RQH, and TR conducted data analysis. LKH, RQH, and GHS drafted the manuscript. LKH, RQH, GHS, WCD, LZ, QL, TR and FL contributed to the interpretation of the data and critically and substantively revised the manuscript. QL, TR and FL contributed equally to the work as senior investigators. They designed the study, supervised the data analyses, and supervised the drafting and revising of the manuscript. The corresponding author attests that all the listed authors meet authorship criteria and that no others meeting the criteria have been omitted. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
All methods were performed in accordance with the relevant guidelines and regulations. All procedures involving human subjects were approved by a central Institutional Review Board (IRB) at the University of California, San Diego (IRB approval number 160091), and in some cases by individual site IRBs (e.g., Washington University in St. Louis) (https://www.sciencedirect.com/science/article/pii/S1878929317300622). Written informed consent was obtained from all parents or guardians.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
41398_2025_3648_MOESM5_ESM.jpg (download JPG )
Supplementary Figure 4. Association between SSRS and mental health problems after excluding children with any psychiatric diagnoses from the non-ASD group.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Huang, L., Huang, R., Sui, G. et al. Demographic, genetic, neuroimaging, and behavioral correlates of short social responsiveness scale in a large pediatric cohort. Transl Psychiatry 15, 396 (2025). https://doi.org/10.1038/s41398-025-03648-1
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41398-025-03648-1