Abstract
We assessed pGF in 2497 AML patients undergoing HSCT from 8-10/10 HLA-matched UD with PTCy. pGF was defined as failure to achieve an ANC ≥ 0.5 × 109/L by day +30 after HSCT. The day +30 cumulative incidence of ANC was 92.6% (95%CI: 91.5–93.6), and the incidence of death without ANC recovery was 1.8% (95% CI: 1.3%–2.3%), corresponding to 141 (5.6%) patients not achieving an ANC ≥ 0.5 × 109/L by day +30. PB was the graft source in 89.4% of the patients, and 56% received reduced-intensity conditioning. 21 patients underwent a second HSCT (15 in the absence of ANC recovery and 6 after ANC recovery). 1-y NRM and RI post-pGF were 22.1% and 22.4%, respectively. 1-y LFS and OS post-pGF were 59% and 55.5%, respectively. ANC recovery evaluated as a time-dependent covariate, KPS ≥ 90, and being in CR at the time of HSCT were associated with improved OS. In conclusion, the incidence of pGF post-unrelated HSCT with PTCy was 5.6%. Of the patients who failed to engraft by day +30, 70.9% did so by day +60. A second transplant can save some of the patients with pGF.
Similar content being viewed by others
Introduction
Post-transplant cyclophosphamide (PTCy) based anti-graft-versus-host disease (GVHD) prophylaxis has proven to be highly effective in preventing GVHD and reducing rates of both GVHD and non-relapse mortality (NRM), leading to substantial improvement in GVHD-free, relapse-free survival, including in the unrelated setting [1,2,3,4,5,6,7]. Given these unprecedented results, PTCy is increasingly being used as GVHD prophylaxis post-unrelated donor allogeneic hematopoietic stem cell transplantation (UD-HSCT) for patients with acute myeloid leukemia (AML) [1,2,3,4,5,6,7]. PTCy is relatively safe, but it is not without cost; side effects and toxicities include a potentially increased risk of cardiac events, hemorrhagic cystitis, delayed immune reconstitution, and late infections [8,9,10,11]. As for engraftment, several studies indicate that this may be slower with PTCy compared to conventional anti-GVHD prophylaxis [1, 12,13,14,15,16].
Primary graft failure (pGF) is a rare but life-threatening complication of allogeneic HSCT [17]. It is usually categorized as lack of initial engraftment defined as absolute neutrophil count (ANC) <0.5 × 109/L in the absence of relapse and combined with the lack of donor chimerism by day +28 after transplantation from either mobilized peripheral blood (PB) stem cells or bone marrow (BM) grafts and by day +42 in the case of cord blood transplantation [17]. The incidence of pGF ranges from 0.8% to 20%, depending on the various transplant-related variables, including donor type, cell source, cell dose, primary disease, and conditioning intensity [18,19,20,21,22]. Known risk factors for pGF are human leukocyte antigen (HLA)-mismatching, cord blood transplantation, recipient’s donor-specific (anti-HLA) antibodies, non-myeloablative conditioning (NMA), low dose of infused hematopoietic stem cells, T-cell depletion, and ABO-mismatched transplants, among others [18, 19, 22,23,24,25,26,27]. Historically, pGF has been a major concern in the haploidentical setting, particularly when BM and reduced intensity conditioning (RIC) is used [28]. As for PTCy, limited data exist on incidence and risk factors for pGF in HSCT with PTCy. From a theoretical point of view, PTCy upregulates T regulatory cells, leading to long-term immune tolerance [10, 29, 30], and thus the incidence of pGF should be low despite the broad HLA disparity. In contrast, some recent data indicate higher early mixed donor chimerism with PTCy [31] and as mentioned above, delayed neutrophil engraftment and even increased red blood cells and platelet transfusion requirements during the first 30 days after transplantation [32]. Regarding haploidentical HSCT with PTCy, the initial study from Luznik et al. reported a graft failure rate of 13% with PTCy following BM infusion and NMA [33]. The Acute Leukemia Working Party (ALWP) previously analyzed risk factors for pGF in AML patients undergoing haploidentical HSCT with PTCy. The incidence of pGF was 6%, which is lower than that reported in T-cell-depleted haploidentical HSCT [28, 34]. Factors independently associated with the risk of non-engraftment were: secondary AML, RIC, and BM grafts [34]. However, the incidence of pGF in the setting of unrelated HSCT with PTCy may differ and is largely unknown. We thus assessed the incidence of pGF post 8–10/10 HLA-matched UD-HSCT with PTCy in patients with AML using the registry data of the ALWP of the European Society for Blood and Marrow Transplantation (EBMT).
Subjects and methods
Study design and data collection
This was a retrospective, multicenter analysis. Data were provided by the registry of the ALWP of the EBMT. The EBMT is a non-profit, scientific society representing more than 600 transplant centers, mainly located in Europe, which are required to report all consecutive stem cell transplantations and follow-ups once a year. Data are entered, managed, and maintained in a central database. Since 1990, all patients have provided informed consent authorizing the use of their personal information for research purposes. The validation and quality control program includes verification of the computer print-out of the entered data, cross-checking with the national registries, and on-site visits to selected teams. The study was approved by the ALWP of the EBMT institutional review board and conducted according to the Declaration of Helsinki and Good Clinical Practice guidelines.
Criteria for selection
To estimate the day 30 cumulative incidence of ANC recovery and associated impact factors, the eligibility criteria comprised adult patients ≥18 years of age with AML who underwent a first HSCT between 2010 and 2022 from an 8 to 10/10 HLA-matched UD with PTCy as GVHD prophylaxis. HSCTs from umbilical cord blood, siblings, and haploidentical donors were excluded. Pre-transplantation preparative regimens included both RIC and myeloablative conditioning (MAC). Patients had to have available information on neutrophil recovery. These inclusion criteria were met by 2497 patients.
For the main objective of this study, only patients with pGF (defined as ANC < 0.5 × 109/L within 30 days after HSCT and only patients alive by day +30) were included, corresponding to 141 patients.
Data collected included recipient and donor characteristics, including age, gender, cytomegalovirus serostatus, Karnofsky performance status (KPS), clinical characteristics, including disease type, disease status, cytogenetic risk (European LeukemiaNet [ELN] 2022 cytogenetics classification), year of transplant, type of conditioning regimen, stem cell source, and GVHD prophylaxis regimen. The latter was defined as MAC or RIC, based on reports from individual transplant centers as per previously established criteria [35], and regimens for GVHD prophylaxis were per institutional protocols. Grading of acute (a) GVHD was performed using established criteria [36]. Chronic (c) GVHD was classified as limited or extensive according to published criteria [37]. For this study, all necessary data were collected according to the EBMT guidelines, using the EBMT minimum essential data forms. The list of institutions contributing data to this study is provided in the Supplementary Appendix.
Statistical analysis
In a first step, the primary endpoint was neutrophil recovery, defined as achieving an ANC ≥ 0.5 × 109/L for 3 consecutive days on the full cohort of 2497 patients from HSCT. Cumulative incidence of day 30 ANC recovery was calculated, as well as the day 30 incidence of death without ANC recovery. Both outcomes were calculated using the cumulative incidence function and were mutually competing events. In order to evaluate the impact factor of day 30 ANC recovery and day 30 death without ANC recovery, these two outcomes were censored at day 30. In the second step, including the 141 patients with pGF and from day +31, the primary endpoint was ANC recovery as previously defined, and the secondary endpoints included the incidence of aGVHD, cGVHD, overall survival (OS), leukemia-free survival (LFS), relapse incidence (RI), and NRM.
Median follow-up was calculated by the reverse Kaplan–Meier method. OS was defined as the time to death from any cause. LFS was defined as survival with no evidence of relapse or progression. NRM was defined as death from any cause without previous relapse or progression. The probabilities of OS and LFS were calculated using the Kaplan–Meier method [38]. Neutrophil recovery, aGVHD, cGVHD, RI, and NRM were estimated using cumulative incidence curves in a competing risk setting. Death and second HSCT were considered as competing risks for neutrophil recovery. To estimate the cumulative incidence of aGVHD and cGVHD, relapse and death were considered as competing events. Univariate analyses were performed using the log-rank test for OS and LFS, and Gray’s test for cumulative incidence. Multivariate analyses were conducted using the Cox proportional-hazards regression model [38]. The impact of ANC recovery post-pGF was evaluated as a time-dependent covariate. Results were expressed as hazard ratios (HRs) with 95% confidence intervals (CIs). All p-values were two-sided, with a type I error rate of 0.05. Statistical analyses were performed using R 4.0.2 (R Core Team, 2020). R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL https://www.R-project.org/ [39].
Results
In total, 2497 patients met the study inclusion criteria, of whom 141 (5.6%) patients failed to achieve ANC ≥ 0.5 × 109/L by day +30 and were alive without second HSCT (Fig. 1). Table 1 shows the baseline demographic and clinical characteristics of these 141 patients. Median follow-up was 1.1 (95% CI 1–1.7) years post-pGF. The median year of the transplant was 2020 (IQR, 2017–2021). Median age was 56.4 (IQR, 46.2–64.3) years, and 61.7% were male. Disease status at HSCT was first or second complete remission in 65.7% and 8.6%, respectively, primary refractory or relapse in 21.4%, and “other” in 4.3% of the patients. Cytogenetic risk (ELN2022) was intermediate in 61.8%, adverse in 30.9%, and favorable in 7.3% of the patients, respectively, but data were missing for 31 (22%) of patients. Donors were 10/10 HLA-matched UD (50.1%) or 8-9/10 HLA-mismatched UD (36.8%), and 17 (12%) patients had missing data for this factor. KPS was ≥90 in 64.2% of the patients, and 80.4% were CMV seropositive. PB was the most frequently used stem cell source (89.4%). Female donor to male recipient was the combination used in 16.3% of patients. In vivo T cell depletion (TCD) with anti-thymocyte globulin was used in 14.9% of patients. Conditioning was RIC in 55.7% and MAC in 44.3% of patients (details of conditioning regimens are provided in Supplementary Table S1). The most frequent (29.8%) prophylaxis associated with PTCy was cyclosporine in combination with mycophenolate mofetil, followed by the latter in combination with tacrolimus (24.8%) of patients (Supplementary Table S2).
The flow of the 141 patients that failed to achieve absolute neutrophil count (ANC) > 0.5 × 109/L by day +30. ANC absolute neutrophil count, d day, HSCT hematopoietic stem cell transplantation, Med median, min minimum, max maximum, Q quarter, GF graft failure, CNS central nervous system, GVHD graft-versus-host disease, VOD Veno-occlusive disease.
Transplantation outcomes
Myeloid engraftment (ANC ≥ 0.5 × 109/L) by day +30 post HSCT was 92.6% (95% CI, 62.6–77.7%), incidence of death within 30 days without ANC recovery was 1.8% (95%CI: 1.3–2.3%). In total, 141 patients (5.6%) did not achieve ANC recovery by day +30 and were alive and free of second HSCT. Of these, 109 (77.3%) recovered within the 30 consecutive days (Fig. 2). Twenty-one patients (15.2%) underwent a second HSCT as a rescue procedure (Figs. 1 and 3), 15 without ANC recovery and 6 with (Supplementary Table S3). Incidence of aGVHD grades II–IV and III-IV at day +100 post pGF were 15.5% (95% CI, 10–22%) and 3.6% (95% CI, 1.3–7.7%), respectively, while those of 1–year post pGF all grades and extensive cGVHD were 11.7% (95% CI, 6.1–19.1%) and 5.0% (95% CI, 1.8–10.6%), respectively (Table 2; Fig. 3). One-year NRM post pGF was 22.1% (95% CI, 15.1%–13.1%). A total of 53 patients died during the study period, including 14 in the absence of ANC recovery and subsequent HSCT (Table 3, Fig. 1). The original disease was the main cause of death in 41.2% of those who died, followed by GF in 21.5% (in whom about two-thirds had an accompanying contributing factor). Infection was the third most common cause of death (17.6%), and GVHD accounted for 5.9% of deaths. Other causes were infrequent (Table 3).
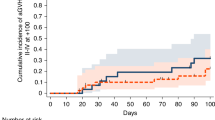
Cumulative incidence of ANC recovery by day (d).
Cumulative incidence of polymorphonuclear neutrophils (PMN) recovery, Cumulative incidence of second allogeneic transplantation (Subsequent HSCT), Overall survival (OS), leukemia-free survival (LFS); Relapse incidence (RI), Non-relapse mortality (NRM). PMN polymorphonuclear, HSCT hematopoietic stem cell transplantation, OS overall survival, LFS leukemia free survival, RI relapse incidence, NRM non relapse mortality.
One-year RI post-pGF was 22.4% (95% CI, 15.2–30.3%). One-year LFS and OS post-pGF were 59% (95% CI, 45.8–64.1%) and 55.5% (95% CI, 49.1–67.5%), respectively (Table 2; Fig. 2). Table 4 summarizes the results of the univariate analysis. At 1 year, age ≥56 years (median age at HSCT), KPS < 90, female gender, and mismatched UD were significantly associated with an increased NRM. The only factor significantly associated with increased 1-year RI was not being in CR at the time of transplantation, which was also significantly associated with inferior 1-year LFS and OS. A KPS < 90 was also associated with inferior 1-year LFS, OS, and NRM, and RIC was significantly associated with inferior OS. PB grafts were associated with better day +30 ANC recovery post-pGF (Table 4).
Multivariate results are shown in Table 5. ANC recovery post pGF and KPS ≥ 90 was associated with improved 1-year OS (HR 0.46, 95% CI: 0.26–0.84, p = 0.01) and (HR 0.37, 95% CI: 0.21–0.65, p < 0.001), respectively, while not being in CR at the time of HSCT significantly decreased OS (HR 2.66, 95% CI: 1.50–4.71, p < 0.001).
Impact factors for day 30 absolute neutrophil count recovery
The impact factors for day 30 ANC recovery were evaluated (Supplementary Tables S4–6). Day 30 ANC recovery was positively affected by PB vs BM (1.60 (1.32–1.94), p < 0.001) and the use of in vivo TCD (HR 1.34,95% CI: 1.20–1.50, p < 0.001), while negatively affected by not being in CR at HSCT (HR 0.82,95% CI: 0.73–0.93, p = 0.002) and positive patient CMV serology (HR 0.91,95% CI: 0.83–0.99, p = 0.0047) (Supplementary Table S6). Death by day 30 in the absence of ANC recovery was negatively impacted by KPS < 90 (HR 2.02,95% CI: 1.08–3.79, p = 0.003) and not being in CR at HSCT (HR 3.35,95% CI: 1.79–6.27, p < 0.001) (Supplementary Table S6).
Discussion
In this registry-based, real-life study assessing the incidence of pGF in a large cohort of AML patients who underwent UD transplantation with PTCy as GVHD prophylaxis, we demonstrated a rather low incidence (5.6%) of pGF; only 141 did not achieve ANC recovery by day +30. Of note, although 70.6% of the 141 patients engrafted by day +30, most of them recovered their myeloid counts by day +60. This observed incidence of pGF seems to be lower than the incidence previously reported for UD HSCT with calcineurin-based anti-GVHD prophylaxis [19,20,21, 26, 40,41,42]. The lower incidence may be due to improvement in HLA typing and donor selections, transplantation techniques and platforms, and pre-transplantation conditioning protocols, as well as due to novel anti-bacterial, viral, and fungal prophylaxis, GVHD prophylaxis, and better supportive care [43,44,45]. However, it is conceivable that the low incidence of pGF including in the mismatched UD HSCT cohort is due to the PTCy mechanism of action, impairing the proliferation of alloreactive T-cells, downregulating of pro-inflammatory cytokines, upregulating T regulatory cells, inducing T cell hypo-responsiveness and tolerance, combined with transcriptional exhaustion phenotype [10, 29, 30, 46,47,48,49].The low pGF we observed with PTCy is of paramount clinical importance as pGF is a lethal complication with a high mortality rate, higher than any other transplant-related complication [40, 50]. Of interest, the Center for International Blood and Marrow Transplant Research (CIBMTR) recently assessed the risk of late GF in adult patients with AML, acute lymphatic leukemia, or myelodysplastic syndrome (MDS) undergoing RIC haploidentical versus 8/8 UD-HSCT with PTCy, reporting similar 2-year incidences of 6.5% and 5.9%, respectively, indicating (as for the haploidentical HSCT) that the initial intense bidirectional alloreactivity between donor and recipient in haploidentical HSCT results in greater dysfunction and early engraftment failure but does not affect the graft long term [51]. However, the mortality rate was higher in the haploidentical HSCT (HR, 1.46; p = 0.007), with recurrent disease being the most common cause of death in both groups. Notably, none of the patients died of graft failure in the matched UD group, compared to 2% in the haploidentical group [51]. Recurrent disease was also the most common cause of death in our study, with GF being the second most frequent cause. GF is associated with a high mortality rate; we recently analyzed 243 patients with acute leukemia undergoing HSCT from various donors (~40% UD) who were complicated by pGF. We observed a 5-year NRM of 52% with infections being the main cause of mortality, in agreement with previous publications. pGF can lead to severe infections and hemorrhagic complications and is the most aggressive post-transplant problem, with extremely high overall mortality [27, 40, 41]. In the current study, infection was the third most frequent cause of death.
Our study is one of the first to focus on pGF in a homogeneous group of UD-HSCT recipients with PTCy in AML. One of the key findings is that about 77% of the patients who did not engraft by day +30 did so by day +60. This accords with the unique biology of PTCy-based anti-GVHD prophylaxis and the slower engraftment reported with PTCy, which is rather encouraging, and it differs from the kinetics seen with conventional anti-GVHD prophylaxis [12,13,14,15,16, 18,19,20,21,22,23]. This finding has major clinical implications for the timing of necessary salvage intervention and mainly salvage rescue transplantation, including from alternative donors [40, 52, 53]. Indeed, only 21 patients (15.2%) in our current study underwent a second HSCT.
Risk factors for pGF in HSCT with PTCy may differ from those previously reported with conventional-based GVHD prophylaxis. In our study, we observed better ANC recovery post-pGF with PB grafts, and an association of ANC recovery with OS in agreement with previous literature [18,19,20,21, 34, 40]. Other factors associated with improved OS were KPS and disease status at HSCT, as previously reported [40]. Recently, Mata et al. assessed pGF in 958 patients with various hematological malignancies receiving NMA HSCT with PTCy-based GVHD prophylaxis. Observed risk factors for pGF were age ≥65 years, an underlying diagnosis of MDS or myeloproliferative disorder (MPD), post-transplant viremia with human herpes virus-6, and low CD34+ and total nucleated cell dose (in those receiving BM grafts). Notably, pGF was not associated with HLA disparity [54]. The incidence of pGF was 3.8% and the 3-year NRM was 59% [54], both higher than we observed in a more homogeneous cohort of AML patients undergoing UD-HSCT, the majority (~90%) with PB grafts. The difference is most probably due to the many variances between the two studies, including factors known to affect the incidence of pGF, such as basic disease (higher incidence in MDS/MPD), type of donor (higher incidence with haploidentical donors), conditioning (higher incidence with NMA), and more. Concerning the risk of pGF in haploidentical-HSCT compared to UD-HSCT and the role of conditioning intensity, Gooptu et al. recently showed a significantly higher incidence of GF (11% vs. 3%, p < 0.001) after transplantation from haploidentical versus UDs in patients with acute leukemia or MDS when using PTCy and RIC in both groups, while no differences in GF were observed with MAC [55].
While not being the main objective of the study, the impact factors for day 30 ANC recovery were evaluated as well. Day 30 ANC recovery was positively affected by the graft source (PB vs BM) graft manipulation (the use of in vivo TCD), while negatively affected by not being in CR at the time of HSCT and positive patient CMV serology in agreement with previous publications [18, 19, 21, 26]. Moreover, as death in the absence of ANC recovery is the competing event of ANC recovery, it is important to mention that death by day 30 in the absence of ANC recovery was negatively impacted by KPS < 90 and not being in CR at the time of HSCT. As for additional risk factors for pGF, due to being a retrospective registry-based study, missing data such as cell dose, donor-specific antibody levels, early viral reactivation (e.g., HHV-6), and heterogeneity in some variables such as conditioning regimen, disease status, and HLA disparities, we could not define risk factors in our study. Other limitations of our study, being a retrospective registry design study, are the possibility that unavailable data, such as frontline therapies, molecular markers, measurable residual disease, or comorbidities, and center-level heterogeneity, might have influenced the outcomes.
However, in this real-life study, we observed a low incidence of pGF, which seems to be lower than that previously reported for UD HSCT with calcineurin-based anti-GVHD prophylaxis. Further studies aiming at improving engraftment, reducing the infection rate, and transplantation-related toxicity are warranted in an attempt to improve outcomes for leukemic patients with post-transplantation GF in the setting of PTCy.
Data availability
AN JEG, FC, and MM had full access to all study data (available upon data-specific request).
References
Bolaños-Meade J, Hamadani M, Wu J, Al Malki MM, Martens MJ, Runaas L, et al. Post-transplantation cyclophosphamide-based graft-versus-host disease prophylaxis. N Engl J Med. 2023;388:2338–48.
Broers AEC, de Jong CN, Bakunina K, Hazenberg MD, van Marwijk Kooy M, et al. Posttransplant cyclophosphamide for prevention of graft-versus-host disease: results of the prospective randomized HOVON-96 trial. Blood Adv. 2022;6:3378–85.
Battipaglia G, Labopin M, Kröger N, Vitek A, Afanasyev B, Hilgendorf I, et al. Posttransplant cyclophosphamide vs antithymocyte globulin in HLA-mismatched unrelated donor transplantation. Blood. 2019;134:892–9.
Brissot E, Labopin M, Moiseev I, Cornelissen JJ, Meijer E, Van Gorkom G, et al. Post-transplant cyclophosphamide versus antithymocyte globulin in patients with acute myeloid leukemia in first complete remission undergoing allogeneic stem cell transplantation from 10/10 HLA-matched unrelated donors. J Hematol Oncol. 2020;13:87.
Nagler A, Labopin M, Swoboda R, Schroeder T, Hamladji RM, Griskevicius L, et al. Post-transplant cyclophosphamide, calcineurin inhibitor, and mycophenolate mofetil compared to anti-thymocyte globulin, calcineurin inhibitor, and methotrexate combinations as graft-versus-host disease prophylaxis after allogeneic stem cell transplantation. Bone Marrow Transpl. 2024;59:1012–21.
Penack O, Abouqateb M, Peczynski C, Boreland W, Gülbas Z, Gedde-Dahl T, et al. PTCy versus ATG as graft-versus-host disease prophylaxis in mismatched unrelated stem cell transplantation. Blood Cancer J. 2024;14:45.
Penack O, Abouqateb M, Peczynski C, Boreland W, Kröger N, Stelljes M, et al. ATG or post-transplant cyclophosphamide to prevent GVHD in matched unrelated stem cell transplantation? Leukemia. 2024;38:1156–63
Ngo D, Samuels D, Chen J, Koller PB, Al Malki MM. A clinical review of the different strategies to minimize hemorrhagic cystitis associated with the use of post-transplantation cyclophosphamide in an allogeneic transplant. Transplant Cell Ther. 2022;28:349–54.
Duléry R, Mohty R, Labopin M, Sestili S, Malard F, Brissot E, et al. Early cardiac toxicity associated with post-transplant cyclophosphamide in allogeneic stem cell transplantation. JACC Cardio Oncol. 2021;3:250–9.
Rambaldi B, Kim HT, Reynolds C, Chamling Rai S, Arihara Y, et al. Impaired T- and NK-cell reconstitution after haploidentical HCT with posttransplant cyclophosphamide. Blood Adv. 2021;5:352–5364.
Irene GC, Albert E, Anna BV, Rahinatu A, Silvana N, Silvana S, et al. Patterns of infection and infection-related mortality in patients receiving post-transplant high-dose cyclophosphamide as graft-versus-host-disease prophylaxis: impact of HLA donor matching. Bone Marrow Transpl. 2021;56:818–27.
Mehta RS, Saliba RM, Chen J, Rondon G, Hammerstrom AE, Alousi A, et al. Post-transplantation cyclophosphamide versus conventional graft-versus-host disease prophylaxis in mismatched unrelated donor haematopoietic cell transplantation. Br J Haematol. 2016;173:444–55.
Salas MQ, Eikema DJ, Koster L, Maertens J, Passweg J, Finke J, et al. Impact of post-transplant cyclophosphamide (PTCy)-based prophylaxis in matched sibling donor allogeneic haematopoietic cell transplantation for patients with myelodysplastic syndrome: a retrospective study on behalf of the Chronic Malignancies Working Party of the EBMT. Bone Marrow Transpl. 2024;59:479–88.
Battipaglia G, Labopin M, Hamladji RM, Blaise D, Chevallier P, Brissot E, et al. Post-transplantation cyclophosphamide versus antithymocyte globulin in patients with acute myeloid leukemia undergoing allogeneic stem cell transplantation from HLA-identical sibling donors: a retrospective analysis from the Acute Leukemia Working Party of the European Society for Blood and Marrow Transplantation. Cancer. 2021;127:209–18
Modi D, Kondrat K, Kim S, Deol A, Ayash L, Ratanatharathorn V, et al. Post-transplant cyclophosphamide versus thymoglobulin in HLA-mismatched unrelated donor transplant for acute myelogenous leukemia and myelodysplastic syndrome. Transplant Cell Ther. 2021;27:760–7.
Nagler A, Ngoya M, Galimard JE, Labopin M, Blau IW, Kröger N, et al. Comparable relapse incidence after unrelated allogeneic stem cell transplantation with post-transplant cyclophosphamide versus conventional anti-graft versus host disease prophylaxis in patients with acute myeloid leukemia: a study on behalf of the Acute Leukemia Working Party of the European Society for Blood and Marrow Transplantation. Am J Hematol. 2024;99:1732–45.
Kharfan-Dabaja MA, Kumar A, Ayala E, Aljurf M, Nishihori T, Marsh R, et al. Standardizing definitions of hematopoietic recovery, graft rejection, graft failure, poor graft function, and donor chimerism in allogeneic hematopoietic cell transplantation: a Report on behalf of the American Society for Transplantation and Cellular Therapy. Transplant Cell Ther. 2021;27:642–9.
Olsson R, Remberger M, Schaffer M, Berggren DM, Svahn BM, Mattsson J, et al. Graft failure in the modern era of allogeneic hematopoietic SCT. Bone Marrow Transpl. 2013;48:537–43.
Chen J, Pang A, Zhao Y, Liu L, Ma R, Wei J, et al. Primary graft failure following allogeneic hematopoietic stem cell transplantation: risk factors, treatment, and outcomes. Hematology. 2022;27:293–9.
Park JH, Lee JH, Lee JH, Park HS, Choi EJ, Kang YA, et al. Incidence, management, and prognosis of graft failure and autologous reconstitution after allogeneic hematopoietic stem cell transplantation. J Korean Med Sci. 2021;36:e151.
Davies SM, Kollman C, Anasetti C, Antin JH, Gajewski J, Casper JT, et al. Engraftment and survival after unrelated-donor bone marrow transplantation: a report from the National Marrow Donor Program. Blood. 2000;96:4096–102.
Rubinstein P, Carrier C, Scaradavou A, Kurtzberg J, Adamson J, Migliaccio AR, et al. Outcomes among 562 recipients of placental-blood transplants from unrelated donors. N Engl J Med. 1998;339:1565–77.
Chakrabarty JH, Glover J, Schmidt S, Phan M, Bycko M, Duong Q, et al. Incidence and risk factors for graft failure in the modern era of cord blood transplantation. Vox Sang. 2022;117:1405–10.
Ciurea SO, Cao K, Fernandez-Vina M, Kongtim P, Malki MA, Fuchs E, et al. The European Society for Blood and Marrow Transplantation (EBMT) consensus guidelines for the detection and treatment of donor-specific anti-HLA antibodies (DSA) in haploidentical hematopoietic cell transplantation. Bone Marrow Transpl. 2018;53:521–34.
Guru Murthy GS, Logan BR, Bo-Subait S, Beitinjaneh A, Devine S, Farhadfar N, et al. Association of ABO mismatch with the outcomes of allogeneic hematopoietic cell transplantation for acute leukemia. Am J Hematol. 2023;98:608–19.
Cluzeau T, Lambert J, Raus N, Dessaux K, Absi L, Delbos F, et al. Risk factors and outcome of graft failure after HLA-matched and mismatched unrelated donor hematopoietic stem cell transplantation: a study on behalf of SFGM-TC and SFHI. Bone Marrow Transpl. 2016;51:687–91.
Ferrà C, Sanz J, Díaz-Pérez MA, Morgades M, Gayoso J, Cabrera JR, et al. Outcome of graft failure after allogeneic stem cell transplant: study of 89 patients. Leuk Lymphoma. 2015;56:656–62.
Aversa F, Tabilio A, Velardi A, Cunningham I, Terenzi A, Falzetti F, et al. Treatment of high-risk acute leukemia with T-cell-depleted stem cells from related donors with one fully mismatched HLA haplotype. N Engl J Med. 1998;339:1186–93.
Mayumi H, Umesue M, Nomoto K. Cyclophosphamide-induced immunological tolerance: an overview. Immunobiology. 1996;195:129–39.
Aversa F, Lustig EB, Geva NO, Prezioso L, Bonomini S, Manfra I, et al. Immune tolerance induction by nonmyeloablative haploidentical HSCT combining T-cell depletion and posttransplant cyclophosphamide. Blood Adv. 2017;1:2166–75.
Hoff FW, Chung SS, Patel PA, Permnath N, Khatib J, Ovcina MT, et al. Post-transplant cyclophosphamide and early mixed donor Chimerism in myeloid malignancies: a single-center experience. Transpl Immunol. 2023;77:101808.
Marco- Ayala J, Sanz JM, Seguí IG, Balaguer-Rosello A, Montoro J, Guerreiro M, et al. Impact of post-transplantation cyclophosphamide on transfusion requirements in HLA-matched sibling peripheral blood stem cell transplantation. Transplant Cell Ther. 2023;29:313.e1–313.e.
Luznik L, O’Donnell PV, Symons HJ, Chen AR, Leffell MS, Zahurak M, et al. HLA-haploidentical bone marrow transplantation for hematologic malignancies using nonmyeloablative conditioning and high-dose, post-transplantation cyclophosphamide. Biol Blood Marrow Transpl. 2008;14:641–50.
Ruggeri A, Labopin M, Angelucci E, Blaise D, Ciceri F, Koc Y, et al. Prognostic factors for neutrophil engraftment after haploidentical cell transplantation with PT-Cy in patients with acute myeloid leukemia in complete remission, on behalf of the ALWP-EBMT. Bone Marrow Transpl. 2021;56:1842–9.
Bacigalupo A, Ballen K, Rizzo D, Giralt S, Lazarus H, Ho V, et al. Defining the intensity of conditioning regimens: working definitions. Biol Blood Marrow Transpl. 2009;15:1628–33.
Przepiorka D, Weisdorf D, Martin P, Klingemann HG, Beatty P, Hows J, et al. 1994 consensus conference on acute GVHD grading. Bone Marrow Transpl. 1995;15:825–8.
Shulman HM, Sullivan KM, Weiden PL, McDonald GB, Striker GE, Sale GE, et al. Chronic graft-versus-host syndrome in man. A long-term clinicopathologic study of 20 Seattle patients. Am J Med. 1980;69:204–17.
Kanate AS, Nagler A, Savani B. Summary of scientific and statistical methods, study endpoints, and definitions for observational and registry-based studies in hematopoietic cell transplantation. Clin Hematol Int. 2019;2:2–4.
R Core Team. R: a language and environment for statistical computing. R Foundation for Statistical Computing; 2020.https://www.R-project.org/
Nagler A, Labopin M, Swoboda R, Kulagin A, Velardi A, Sanz J, et al. Long-term outcome of second allogeneic hematopoietic stem cell transplantation (HSCT2) for primary graft failure in patients with acute leukemia in remission: A study on behalf of the Acute Leukemia Working Party of the European Society for Blood and Marrow Transplantation. Bone Marrow Transpl. 2023;58:1008–16.
Schriber J, Agovi MA, Ho V, Ballen KK, Bacigalupo A, Lazarus HM, et al. Second unrelated donor hematopoietic cell transplantation for primary graft failure. Biol Blood Marrow Transpl. 2010;16:1099–106.
Shouval R, Fein JA, Labopin M, Kröger N, Duarte RF, Bader P, et al. Outcomes of allogeneic haematopoietic stem cell transplantation from HLA-matched and alternative donors: a European Society for Blood and Marrow Transplantation registry retrospective analysis. Lancet Haematol. 2019;6:e573–584.
Canaani J, Beohou E, Labopin M, Ghavamzadeh A, Beelen D, Hamladji RM, et al. Trends in patient outcome over the past two decades following allogeneic stem cell transplantation for acute myeloid leukaemia: an ALWP/EBMT analysis. J Intern Med. 2019;285:407–18.
Penack O, Peczynski C, Mohty M, Yakoub-Agha I, Styczynski J, Montoto S, et al. How much has allogeneic stem cell transplant-related mortality improved since the 1980s? A retrospective analysis from the EBMT. Blood Adv. 2020;4:6283–90.
Schmälter AK, Ngoya M, Galimard JE, Bazarbachi A, Finke J, Kröger N, et al. Continuously improving outcome over time after second allogeneic stem cell transplantation in relapsed acute myeloid leukemia: an EBMT registry analysis of 1540 patients. Blood Cancer J. 2024;14:76.
Wachsmuth LP, Patterson MT, Eckhaus MA, Venzon DJ, Gress RE, Kanakry CG. Post-transplantation cyclophosphamide prevents graft-versus-host disease by inducing alloreactive T-cell dysfunction and suppression. J Clin Invest. 2019;129:2357–73.
Kanakry CG, Ganguly S, Zahurak M, Bolaños-Meade J, Thoburn C, Perkins B, et al. Aldehyde dehydrogenase expression drives human regulatory T cell resistance to post-transplantation cyclophosphamide. Sci Transl Med. 2013;5:211ra157.
McCurdy SR, Radojcic V, Tsai HL, Vulic A, Thompson E, Ivcevic S, et al. Signatures of GVHD and relapse after posttransplant cyclophosphamide revealed by immune profiling and machine learning. Blood. 2022;139:608–23.
Zhao C, Bartock M, Jia B, Shah N, Claxton DF, Wirk B, et al. Post-transplant cyclophosphamide alters immune signatures and leads to impaired T cell reconstitution in allogeneic hematopoietic stem cell transplant. J Hematol Oncol. 2022;15:64.
Sawyer J, Elliott T, Orton L, Sowell H, Gatwood K, Shultes K. Prevention and management of acute toxicities from conditioning regimens during hematopoietic stem cell transplantation. Clin Hematol Int. 2024;6:1–10.
Hickey CL, Zhang MJ, Allbee-Johnson M, Romee R, Majhail NS, Malki MMA, et al. Donor type does not impact late graft failure following reduced-intensity allogeneic hematopoietic cell transplantation with post-transplant cyclophosphamide-based graft-versus-host disease prophylaxis. Transplant Cell Ther. 2025;31:174.e1–12.
Giammarco S, Raiola AM, Di Grazia C, Bregante S, Gualandi F, Varaldo R, et al. Second haploidentical stem cell transplantation for primary graft failure. Bone Marrow Transpl. 2021;56:1291–6.
Harada K, Fuji S, Seo S, Kanda J, Ueki T, Kimura F, et al. Comparison of the outcomes after haploidentical and cord blood salvage transplantations for graft failure following allogeneic hematopoietic stem cell transplantation. Bone Marrow Transpl. 2020;55:1784–95.
Mata JR, Zahurak M, Rosen N, DeZern AE, Jones RJ, Ambinder AJ. Graft failure incidence, risk factors, and outcomes in patients undergoing non-myeloablative allogeneic hematopoietic cell transplantation using post-transplant cyclophosphamide. Transplant Cell Ther. 2024;30:588–96.
Gooptu M, Romee R, St Martin A, Arora M, Al Malki M, Antin JH, et al. HLA-haploidentical vs matched unrelated donor transplants with posttransplant cyclophosphamide-based prophylaxis. Blood. 2021;138:273–82.
Funding
Open access funding provided by Tel Aviv University.
Author information
Authors and Affiliations
Contributions
AN wrote the manuscript, designed the study, and interpreted the data. SK helped edit the first draft of the manuscript and reviewed the manuscript. JEG, MM, and FC designed the study, performed the statistical analyses, interpreted the data, and edited the manuscript. AK, DB, EP, AECB, MIR, GC, RMB, SS, MR, RVDB, AB, and SP reviewed the manuscript and provided clinical data. All authors approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
The scientific boards of the ALWP of the EBMT approved this study. All methods were performed in accordance with the relevant guidelines and regulations. Informed consent was obtained from all subjects.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Nagler, A., Galimard, JE., Kayser, S. et al. Outcomes of primary graft failure in acute myeloid leukemia patients following unrelated transplantation with post-transplant cyclophosphamide: a study from the ALWP/EBMT. Bone Marrow Transplant 61, 51–58 (2026). https://doi.org/10.1038/s41409-025-02726-8
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41409-025-02726-8