Abstract
Bone fractures represent a significant global healthcare burden. Although fractures typically heal on their own, some fail to regenerate properly, leading to nonunion, a condition that causes prolonged disability, morbidity, and mortality. The challenge of treating nonunion fractures is further complicated in patients with underlying bone disorders where systemic and local factors impair bone healing. Traditional treatment approaches, including autografts, allografts, xenografts, and synthetic biomaterials, face limitations such as donor site pain, immune rejection, and insufficient mechanical strength, underscoring the need for alternative strategies. Biologic therapies have emerged as promising tools to enhance bone regeneration by leveraging the body’s natural healing processes. This review explores the critical role of conventional and emerging biologics in fracture healing. We categorize biologic therapies into protein-based treatments, gene and transcript therapies, small molecules, peptides, and cell-based therapies, highlighting their mechanisms of action, advantages, and clinical relevance. Finally, we examine the potential applications of biologics in treating fractures associated with bone disorders such as osteoporosis, osteogenesis imperfecta, rickets, osteomalacia, Paget’s disease, and bone tumors. By integrating biologic therapies with existing biomaterial-based strategies, these innovative approaches have the potential to transform clinical management and improve outcomes for patients with difficult-to-heal fractures.

Similar content being viewed by others
Introduction
Bone fractures are a prevalent medical condition, with an estimated 178 million new fractures occurring globally each year.1 Despite advances in medical treatment, 5%–10% of fractures result in nonunion, a condition where the bone fails to heal properly.2 The FDA defines a nonunion as a fracture that shows no visible signs of healing at least nine months post-trauma.3 This condition can lead to prolonged disability and significantly impact patients’ quality of life.
The risk of nonunion formation is influenced by factors such as fracture size, mechanical instability, shear stress, poor vascularization, and infections.4 In patients with bone disorders, this risk is further increased due to a diminished pool of osteogenic precursors with impaired proliferative capacity, altered bone mineral matrix composition, and local or systemic disruptions in hormonal and growth factor signaling.5,6,7,8
Managing nonunion fractures remains challenging and costly, with treatment expenses ranging from €8 000 to €90 000 per patient until complete healing is achieved.9,10 The gold standard remains autologous bone grafting, which uses the patient’s own bone—typically harvested from the iliac crest—due to its high osteogenic potential and lack of immune rejection.11 However, this procedure is limited by donor site morbidity, including chronic pain, infection, and nerve or muscle injury, as well as constraints in graft availability and shape.12,13
To address bone deficits, allografts and xenografts have been explored as alternatives. Allografts, sourced from living donors or cadavers, offer immediate availability in various sizes and shapes,14,15 but have reduced osteogenic potential compared to autografts.16 Similarly, xenografts, typically derived from bovine, porcine, or coral sources, offer osteoconductive properties at a lower cost. However, bovine and porcine xenografts carry risks of immune rejection and disease transmission,17,18 while coral-derived xenografts are limited by poor resorption rates.19
Synthetic biomaterials, such as calcium phosphate ceramics and cements, offer further options for bone defect treatment due to their composition, which mimics the native bone matrix and supports osteoconduction.20,21 While effective for small defect reconstruction, these materials are often unsuitable for larger defects due to limited mechanical strength and insufficient neovascularization.4,22
Biologic therapies have emerged as a promising approach to enhancing bone healing by leveraging biological molecules that support the body’s natural regeneration mechanisms. Among these, growth factors and cell-based therapies have shown encouraging clinical results. However, challenges such as limited efficacy, immune responses, and high production costs necessitate further advancements in the field.
Gene and transcript therapies, which are DNA or RNA based, respectively, represent a novel frontier in regenerative medicine for bone repair.23 Gene therapy involves the introduction, alteration, or removal of genes to treat or prevent a disease, while transcript therapy focuses on the delivery of RNA transcripts to modulate gene expression.24,25 By directly delivering genetic material or its transcribed mRNA into the fracture site, these therapies enable the localized production of therapeutic proteins, promoting and accelerating osteogenesis and vascularization.25 Currently, most transcript-based strategies rely on chemically modified mRNA, owing to its enhanced translational efficiency, reduced immunogenicity, and tunable protein expression, with an improved safety profile and affordable costs.
Recent breakthroughs in gene delivery platforms, including lipid- and polymer-based vectors as well as lipid nanoparticles, have enhanced the stability, bioavailability, and cellular uptake of nucleic acids, overcoming key barriers to clinical translation.26,27,28 Furthermore, biomaterial scaffolds engineered to carry gene or transcript therapies have improved their targeted application, ensuring optimal integration into the bone microenvironment.29
This review provides a comprehensive overview of the current landscape of biologic therapies for nonunion fractures. We begin by outlining the key stages of fracture healing, highlighting the main cell types and growth factors involved in the repair process. Next, we categorize biologic therapies that are either under investigation or in clinical use, including protein therapy, gene therapy, transcript therapy, cell therapy, peptides, and small molecules. Each therapeutic category is explored in terms of its distinct mechanism of action, therapeutic advantages, limitations, and clinical relevance.
Furthermore, we discuss the potential applications of these biologic therapies across diverse patient populations, with a focus on individuals affected by osteoporosis, osteogenesis imperfecta, rickets, osteomalacia, bone tumors, and Paget’s disease. By evaluating the efficacy and safety profiles of these therapies in various pathological contexts, we aim to underscore their transformative potential in addressing the unmet needs of bone regeneration and fracture healing.
The fracture healing process: key cellular and growth factor contributors
Fracture healing can be divided into four phases, each characterized by the presence of different cells, extracellular matrix (ECM) components and growth factors (Fig. 1). These sequential phases include the inflammatory phase, soft callus formation, ossification, and remodeling.30 Physiological growth factors are primarily stored in bone ECM and are actively released upon injury to initiate the regeneration process.31 These growth factors exert their effects by binding to transmembrane receptors, triggering intracellular signaling cascades that activate specific transcription factors and gene expression programs.32
Expression profile of osteogenic growth factors during fracture healing. This schematic illustrates the four phases of bone healing: inflammatory phase, soft callus formation, ossification, and remodeling. At each stage, specific cell populations locally and focally express osteogenic growth factors. The asterisks and intensity of each arrow represent the expression level of a given factor by a particular cell type. OB osteoblasts, MSC mesenchymal stem cells, CH chondrocytes, PMSC periosteal mesenchymal stem cells, ICH immature chondrocytes, MP macrophages, PL platelets, MO monocytes, EC endothelial cells, HCH hypertrophic chondrocytes, OC osteoclasts, BMP bone morphogenetic protein, PDGF platelet-derived growth factor, VEGF vascular endothelial growth factor, FGF fibroblast growth factor, TGF-β1 transforming growth factor-β1, IGF insulin-like growth factors, NGF nerve growth factor
In the inflammatory phase, vascular disruption leads to hematoma formation containing bone fragments and immune cells.33 Peaking within 48 h and subsiding by one week, this phase immobilizes the fracture through pain-induced restriction and swelling.34 The acidic microenvironment triggers platelet degranulation, coagulation, and growth factor release. Concurrently, immune cells secrete cytokines that promote osteogenesis, angiogenesis, and initiate tissue repair.35
The soft callus formation phase lasts for several weeks. During this time, mesenchymal stem cells (MSCs) differentiate into fibroblasts, chondroblasts, and osteoblasts.36 Fibroblasts produce ECM proteins like type I and II collagen, while chondroblasts and osteoblasts form a cartilaginous callus that stabilizes the fracture, providing initial mechanical support.37,38 As local pH shifts from acidic to slightly alkaline, the enzyme alkaline phosphatase (ALP) facilitates mineral deposition and bone formation.39
During the ossification phase, bone forms through intramembranous and endochondral ossification. Intramembranous ossification primarily occurs in the periosteal region of the fracture, specifically at the fracture periphery.40 It involves direct osteoblast-mediated ECM secretion, primarily composed of type I collagen and osteocalcin, and calcium phosphate deposition, leading to osteoblast differentiation to osteocytes.41 Endochondral ossification, occurring predominantly in the central regions of the fracture, relies on chondrocyte-produced collagen type II matrices, which are later replaced by bone tissue predominantly containing type I collagen.42
Finally, the remodeling phase converts immature, weak woven bone into organized lamellar bone.43 In this process, osteoblasts, osteocytes, and osteoclasts work together to restore bone structure and mechanical strength, a process that can last months to years.44
Given that these signaling pathways regulate multiple genes simultaneously, individual growth factors can exert pleiotropic effects within and across different cell types.45 Biologic therapies leverage these endogenous mechanisms by delivering specific growth factors at the site of injury to stimulate and accelerate healing.46,47 Key growth factors for bone regeneration are shown in Table 1, and its specific role in the fracture healing process is explained in the next sections.
Bone morphogenetic proteins (BMPs)
BMPs are acknowledged as the most efficient growth factors to support the healing of large bone defects.48 During fracture healing, BMP-2, BMP-4, BMP-7, and BMP-9 are strongly expressed in undifferentiated MSCs during the inflammatory phase.49 These BMPs are also highly expressed in proliferating osteoblasts during intramembranous ossification50 and in proliferating chondrocytes during chondrogenesis and endochondral ossification. Their expression decreases in mature chondrocytes but remains high in osteoblasts near the endochondral ossification front.51 Additionally, BMPs regulate the expression of other critical growth factors, including insulin-like growth factors (IGFs) and transforming growth factors (TGFs), thereby orchestrating bone formation. Beyond osteogenesis, BMPs also contribute to angiogenesis, particularly BMP-4, which enhances vascularization through interactions with type I and IV collagen and heparin.30
Fibroblast growth factors (FGFs)
FGFs constitute a large family of signaling proteins that play crucial roles in tissue development, metabolism, and bone repair. Among them, FGF-1 and FGF-2 are the most extensively studied in the context of bone metabolism. During the inflammatory phase of fracture healing, FGF-1 is expressed in macrophages and periosteal cells. As healing progresses, it is detected in osteoblasts during intramembranous ossification and in immature chondrocytes during chondrogenesis.30 In endochondral ossification, its expression is limited to osteoblasts. In contrast, FGF-2 shows consistent expression throughout regeneration, found in macrophages, osteoblasts, and chondrocytes, during the ossification phase.30,52 FGFs also regulate angiogenesis by upregulating VEGF and enhance bone formation by interacting with BMP-2.53 FGF-2 deficiency impairs BMP-2 function, while higher BMP-2 levels enhance FGF-2 osteogenic potential.54
Platelet-derived growth factors (PDGFs)
PDGF is a signaling molecule with receptor tyrosine kinase activity that plays a critical role in angiogenesis and the proliferation of MSCs.55,56 Initially, PDGF expression is weak during the inflammatory phase but increases and remains stable throughout the repair process, contributing to both cartilage formation and intramembranous bone development.52 PDGF acts as a potent chemoattractant for pericytes, facilitating their detachment from the vessel wall, allowing them to transition into MSCs. These MSCs, in turn, are stimulated by PDGF to differentiate into osteoblastic progenitors, thereby enhancing bone regeneration.57
Despite its well-established involvement in the fracture healing cascade, the therapeutic potential of PDGF in bone repair remains controversial. For instance, even when combined with osteoconductive materials, PDGF has not consistently demonstrated significant enhancement of bone regeneration in clinical or preclinical models.58,59 Furthermore, emerging evidence suggests that PDGF may antagonize the osteoinductive activity of BMP-2.60 These findings underscore the complexity of PDGF’s role in bone healing and highlight the need for a more nuanced understanding of its mechanisms of action.
Transforming growth factor-β1 (TGF-β1)
TGF-β1 is one of the most abundant cytokines in the bone matrix, existing in an inactive precursor form until it is cleaved and activated by acidic conditions or osteoblast-mediated processing.50 Once active, TGF-β1 functions as a chemotactic agent to recruit local MSCs for osteogenic differentiation.61 At the fracture site, TGF-β1 is produced by various cell types, including platelets, macrophages, osteoblasts, osteoclasts, and chondrocytes.52,62 Extracellularly, it is present in the hematoma immediately after injury, playing an essential role in early bone healing.63 During intramembranous ossification, TGF-β1 is highly expressed in proliferating osteoblasts, while during chondrogenesis and endochondral ossification, its expression peaks in proliferating chondrocytes.50 Additionally, TGF-β1 enhances the production of key bone and cartilage extracellular matrix components, including various collagen types, fibronectin, osteopontin, osteonectin, thrombospondin, proteoglycans, and ALP.64
Insulin-like growth factors (IGFs)
IGFs, also known as somatomedins or sulfation factors, function as key autocrine and paracrine regulators in bone metabolism. IGFs produced by bone tissue become sequestered within the bone matrix and are later released during bone resorption or fracture, where they stimulate osteoblast precursor proliferation via activation of the mTOR signaling pathway.65 Outside the liver, bone is the richest source of IGF-1 and IGF-2 in mammals, where these locally produced IGFs play a key role in skeletal development and remodeling. In particular, IGF-1 is essential for creating the osteogenic microenvironment required for MSCs to differentiate into osteoblasts.65 Unlike other growth factors involved in fracture healing, IGF-1 mRNA is not detected during the inflammatory phase. Instead, its expression is observed in osteoblasts and prehypertrophic chondrocytes during intramembranous ossification.66 IGF-2 mRNA, is expressed in certain osteoclasts and osteoblasts during the remodeling phase, though most osteoblasts remain negative for IGF-2 expression.66 Both IGF-1 and IGF-2 contribute to bone formation by enhancing collagen synthesis and reducing collagen degradation.67
Vascular endothelial growth factor (VEGF)
Fractures often disrupt the local blood supply, necessitating rapid revascularization to deliver oxygen and nutrients. VEGF plays a pivotal role in angiogenesis during fracture healing.68 Its expression is initiated during the inflammatory phase, driven by hypoxic conditions and the fragmented bone matrix within the hematoma, which stimulates its release from platelets and infiltrating immune cells.69,70 Beyond its angiogenic function, VEGF also regulates inflammatory responses, contributing to a balanced and controlled repair environment.71 Although cartilage is inherently avascular, studies have demonstrated that hypertrophic chondrocytes actively express and secrete VEGF, facilitating the transition from cartilage to bone.38 VEGF further acts synergistically with BMPs to enhance preosteoblast differentiation and metabolism. Additionally, during the remodeling phase, VEGF promotes osteoclast recruitment, survival, and activity, all of which are essential for bone resorption and structural refinement.72
Nerve growth factor (NGF)
Mature bone is a highly innervated tissue, and accumulating evidence suggests that nerve-derived signals play a crucial role in bone regeneration.73,74 Several studies have reported a significant upregulation of NGF immediately after bone injury, highlighting its importance in the fracture healing process.75,76 Experimental nerve resection via nonspecific surgical methods has been shown to impair fracture healing in mouse models, further underscoring NGF’s role in bone regeneration.77,78 Beyond its well-established function in promoting neuronal growth and extension, NGF enhances osteogenic activity, potentially expediting fracture repair.79 It has been shown to stimulate osteoblast mitosis and actively participate in skeletal development and ossification. NGF is expressed in MSCs,80 osteoclasts, osteocytes, chondrocytes, and synovial fibroblasts.81,82
Categories of biologic therapies
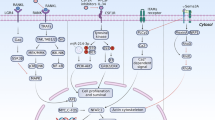
Advancements in biologic therapies are continually enhancing the ability to regulate fracture healing pathways, providing innovative treatment options for bone regeneration. According to the FDA, biologics encompass a diverse range of therapeutic products, including recombinant proteins, gene and transcript-based therapies, small molecules, peptides, and cell-based treatments, and blood-derived products (Fig. 2). The following sections will explore each category in the context of bone healing, highlighting their mechanisms, benefits, and challenges (Fig. 3).
Categories of biologic therapies for bone regeneration. Schematic overview of representative examples across distinct classes of biologic therapies for bone regeneration, including protein therapy, gene therapy, transcript therapy, cell therapy, small molecules, and peptides. Some therapies, such as recombinant BMP-2 and BMP-7, are FDA-approved, while others—like VEGF pDNA gene therapy—are currently in clinical trials. Emerging approaches such as transcript therapy (e.g., cmRNA) remain in the preclinical stage. BMP bone morphogenetic protein, PDGF platelet-derived growth factor, VEGF vascular endothelial growth factor, pDNA plasmid DNA, HA hydroxyapatite, BMA bone marrow aspirate, PRP platelet-rich plasma, cmRNA chemically modified mRNA, HTS high-throughput screening, RGD arginine-glycine-aspartic acid tripeptide
Comparative overview of the advantages and limitations of various biologic strategies for bone regeneration, including protein, gene, transcript, cell, peptide, and small molecule therapies. PTM post-translational modifications, BMAs bone marrow aspirates
Protein therapy
Recombinant protein therapy involves the direct administration of purified proteins to promote bone regeneration.83 These proteins are produced by inserting the gene encoding the desired protein into host cells—such as bacteria, yeast, or mammalian cells—which are then cultured in bioreactors under controlled conditions. The resulting protein is harvested, purified through multiple chromatography steps, and subjected to rigorous quality control assessments to ensure therapeutic efficacy and safety.
Numerous clinical studies have explored the use of recombinant proteins for fracture healing, with BMP-2 and BMP-7 among the most extensively studied and being among the few FDA-approved osteoinductive growth factors for bone regeneration. Commercially available products such as INFUSE® (rhBMP-2, Medtronic) are approved for indications like open tibial fractures and spinal fusions, though they are also used off-label for treating nonunions and in select dental procedures.84,85,86 Other growth factors, such as FGF-2,87,88 PDGF,89,90,91 and IGF-192,93 have also been investigated for their roles in stimulating bone repair or improving bone mineral density.94
Despite their comparable efficacy to autografts, recombinant protein therapies often require supraphysiological doses to achieve therapeutic effects. In clinical settings, these doses can exceed natural levels by several orders of magnitude, which increases efficacy but also elevates the risk of adverse effects. BMP-2 and BMP-7, for example, have been associated with ectopic bone formation, inflammation, and nerve compression.84,85,86
In addition to growth factors, parathyroid hormone (PTH) and its synthetic analogs, such as teriparatide (PTH(1–34), Forteo®) and abaloparatide (a PTHrP analog), have also been used as anabolic agents in bone regeneration.95,96 Intermittent PTH administration transiently activates PTH1 receptors on osteoblasts and osteocytes, triggering intracellular signaling cascades (e.g., cAMP/PKA, PLC/PKC) that promote osteoblast survival, differentiation, and bone-forming activity.97 Clinical trials investigating PTH-based strategies for fracture repair and nonunions have produced mixed results. Some studies have reported enhanced healing rates and reduced time to union in specific fracture types (e.g., vertebral, sternal, tibial, and femoral fractures),98,99,100,101 while others have shown no significant advantage in long bone nonunions compared to placebo or conventional care.102,103 In clinical practice, its application in nonunions is often off-label, particularly in patients with metabolic bone disorders or delayed healing, where an anabolic boost is desired.104
The Wnt/β-catenin signaling pathway also represents a highly promising target for bone regenerative therapies. Wnt proteins bind to Frizzled receptors and LRP5/6 co-receptors on the surface of osteoprogenitor cells, thereby inhibiting the β-catenin destruction complex. This results in β-catenin stabilization, nuclear translocation, and activation of osteogenic gene expression.105 Activation of this pathway promotes osteoblast differentiation and bone formation.106 In a study by Minear et al., liposomal Wnt3a was delivered to skeletal defects in mice, stimulating progenitor cell proliferation and accelerating osteogenic differentiation, ultimately enhancing bone regeneration.107 Given the central role of this pathway, multiple therapeutic strategies targeting various components of Wnt/β-catenin signaling have been developed—these are further explored in the following section.
Monoclonal antibodies in bone regeneration
Monoclonal antibodies (mAbs) represent another class of protein-based therapeutics that differ from conventional recombinant proteins, such as BMPs, in their mechanism of action. Instead of replacing or supplementing missing proteins, mAbs function by binding to specific molecular targets involved in bone remodeling, thereby modulating cellular pathways with high specificity. For example, sclerostin, a glycoprotein secreted by osteocytes, is a negative regulator of bone formation by inhibiting the Wnt/β-catenin signaling pathway. Anti-sclerostin antibodies neutralize sclerostin, thereby disinhibiting Wnt signaling and promoting osteoblast proliferation, differentiation, and bone formation, while also reducing bone resorption.
One advantage of monoclonal antibodies is their ability to be administered systemically, allowing them to reach fracture sites through circulation and exert their effects on bone cells throughout the skeleton. For example, Pui Kit Suen et al. investigated the effects of systemic administration (subcutaneous injection, 25 mg/kg, two times per week) of sclerostin antibody (SclAb) on fracture repair in a rat femoral osteotomy model.108 SclAb treatment significantly increased bone mineral density (BMD) and enhanced neovascularization. Mechanical testing at weeks 6 and 9 demonstrated significantly higher ultimate load in the SclAb-treated group, indicating improved bone strength.108
Clinically, monoclonal antibody therapies have shown promise in difficult-to-heal fractures. A case report by Takuya Uemura et al. described a 61-year-old heavy smoker with a distal radius nonunion who achieved successful bone healing through a combination of distraction plate fixation, graft substitutes, and systemic administration of romosozumab—a monoclonal antibody targeting sclerostin.109 The study concluded that combining β-tricalcium phosphate bone grafting with bridge plating and systemic romosozumab can be an effective treatment option for complex nonunion cases, particularly in patients with risk factors such as smoking, diabetes, and bone fragility.109
However, the clinical use of romosozumab has raised safety concerns due to an observed increase in serious cardiovascular events, including myocardial infarction and stroke. In clinical trials, patients receiving romosozumab exhibited a higher incidence of these adverse effects compared to those treated with alendronate.110 As a result, this therapy should be used with caution in patients with a history of cardiovascular disease or other risk factors. Moreover, the therapeutic potential of romosozumab in fracture healing remains debated. A phase 2 double-blind, randomized clinical trial in patients with acute unilateral tibial diaphyseal fractures found no significant improvement in healing time, regardless of the dosage administered.111 Similar results were reported in a second phase 2 dose-finding study involving patients with hip fractures, where romosozumab also failed to accelerate fracture healing.112
While anti-sclerostin antibodies remain the most clinically advanced class of anabolic agents for bone, with potential applications in fracture healing and nonunion, other therapeutic targets—such as Dickkopf-1 (DKK1)—are under active investigation. DKK1 is a secreted glycoprotein that inhibits the canonical Wnt/β-catenin signaling pathway by binding to LRP5/6 co-receptors, thereby preventing Wnt ligand binding and suppressing osteoblast development and activity.113,114 Anti-DKK1 antibodies counteract this inhibition, allowing Wnt signaling to proceed and stimulating bone formation.115 Although still in the preclinical stage, primarily in rodent models, studies have shown that anti-DKK1 antibodies enhance callus formation, improve mechanical strength, and accelerate endochondral ossification in bone fracture models.116
Another promising therapeutic is denosumab, a monoclonal antibody that targets RANK Ligand (RANKL)—a key regulator of osteoclast differentiation and activity.117 By blocking RANKL-RANK interactions, denosumab effectively inhibits osteoclast-mediated bone resorption.118 Although primarily used in the treatment of osteoporosis,119 its application in nonunion cases is gaining attention, particularly where excessive or dysregulated bone resorption impairs healing. In a prospective study involving 43 patients with aseptic tibial shaft nonunions following intramedullary nailing, the addition of subcutaneous denosumab (60 mg) postoperatively to plate augmentation and autologous bone grafting significantly reduced the average time to union from 14.82 weeks to 9.45 weeks, with no major complications reported.120 While denosumab appears to enhance early callus consolidation, its inhibitory effect on bone resorption may result in delayed remodeling, with woven bone persisting longer before transitioning to mature lamellar bone. Further studies are needed to fully understand the long-term impact of denosumab on bone remodeling and its overall therapeutic value in nonunion management.
Challenges and limitations of protein therapy
Beyond their side effects, the high cost of recombinant proteins and monoclonal antibody therapies remains a major barrier to widespread clinical adoption.121 The price of treatment varies depending on the specific protein, fracture type, dosage, and healthcare setting. For instance, the costs of the commercial BMP-7 product Osigraft® were between €15 000 and €18 000 per patient for tibial shaft nonunions before its production was discontinued.122
Therapeutic proteins are often produced in non-human systems (e.g., bacteria or yeast) that lack the necessary cellular machinery for human-specific post-translational modifications, such as glycosylation, phosphorylation, or acetylation—modifications essential for proper protein function.123 Another major challenge is maintaining effective protein levels at the fracture site, as their short half-life often necessitates repeated administrations.124 Recombinant proteins are highly susceptible to denaturation during production, storage, and delivery, leading to misfolding or partial unfolding, which significantly reduces their biological activity.125 These limitations further hinder their feasibility for routine clinical use, highlighting the need for advanced delivery strategies that enhance protein stability, bioavailability, and retention at the injury site.
Gene therapy
Gene therapy aims to introduce genes encoding therapeutic proteins into target cells, enabling them to endogenously produce one or more desired proteins at the site of injury with native conformation and proper post-translational modifications.23 This approach has the potential to overcome the limitations of recombinant protein therapy by ensuring sustained, physiologically relevant levels of therapeutic proteins directly at the fracture site.126 Additionally, since a single copy of plasmid DNA (pDNA) can give rise to multiple copies of the therapeutic protein, gene therapy offers an efficient way to amplify protein production. While this enhanced endogenous protein production is certainly an advantage, a challenge of this approach compared to protein therapy, where precise dosing is possible and controlled protein delivery is feasible, is that the exact amount of protein copies that a DNA molecule produces is unknown, which could be a regulatory challenge when it comes to describing the immediate bioavailability of the active agent.
Gene delivery can be achieved using viral (transduction) or non-viral (transfection) vectors, through either an in vivo or ex vivo gene-transfer strategy.126 Both methods have demonstrated promising results in preclinical models.127 Among all growth factors, BMP genes have been extensively studied for bone healing, with gene therapy-based delivery showing encouraging outcomes in animal studies.128,129,130 Adenoviral vectors, in particular, have been used to achieve prolonged gene expression at the fracture site.131 However, gene therapy for bone regeneration has seen limited clinical application, with only four trials conducted to date, all in the Russian Federation.132 The first trial (NCT02293031), initiated in 2014, evaluated a gene-activated bone substitute incorporating a collagen–hydroxyapatite scaffold loaded with a VEGFA-encoding plasmid.133 Results showed improved bone density within three months and no reported adverse effects. However, the trial was withdrawn due to financial constraints.134
A subsequent trial (NCT03076138) by the same research team investigated an octacalcium phosphate (OCP) scaffold carrying a VEGFA plasmid for maxillofacial bone regeneration. The study, involving 20 patients, demonstrated successful bone formation with no complications.132 These promising outcomes led to regulatory approval in Russia in 2019, making this technology available for clinical use in oral and maxillofacial surgery. Another trial (NCT04705857) investigated a gene-activated matrix combining a demineralized bone allograft with a dual cassette vector expressing VEGFA and BMP-2 for treating ulnar pseudarthrosis.135 Results showed enhanced bone regeneration without adverse effects. The most recent ongoing trial (NCT04511689) is evaluating gene-activated bone substitutes containing OCP and VEGFA-expressing DNA against a xenogeneic deproteinized bone matrix for alveolar ridge augmentation before dental implantation. The trial is still in progress, with results yet to be published.
Despite its promise, gene therapy remains expensive and faces considerable regulatory hurdles.126 Treatments can cost an estimated $1 million or more, primarily due to the high expenses associated with viral vector production.25 Viral vectors are frequently used to deliver DNA because they more efficiently cross the nuclear membrane—especially in non-dividing cells—than non-viral alternatives. However, regulatory concerns persist regarding the use of viral vectors, particularly the risk of transgene integration into the host genome, which can lead to potentially dangerous insertional mutagenesis effects.127 Consequently, further research is needed to establish the long-term safety and efficacy of gene therapy for bone healing.
Transcript therapy
While protein replacement therapy and gene therapy hold promise for bone regeneration, their clinical translation remains challenging due to financial, safety, and regulatory hurdles.136 This is reflected in the limited number of FDA-approved recombinant proteins for bone repair137 and the scarcity of approved gene therapy protocols targeting musculoskeletal disorders—currently, only two gene therapies are approved for osteoarthritis and disc degeneration.138 To address these challenges, a newer category of gene therapy, transcript therapy, has emerged. Instead of using pDNA, this approach delivers messenger RNA (mRNA) encoding the therapeutic protein. Transcript therapy offers several advantages, including reduced risk of genomic integration and potentially lower manufacturing costs, making it a promising alternative for safer and more affordable gene-based treatments for bone fractures.139
Transcript therapy relies on chemically modified mRNA (cmRNA) to enhance protein production. Structural modifications to mRNA, including adjustments to the 5′ cap, untranslated regions (UTRs), poly-A tail, and modified nucleotides, enhance stability, increase translation efficiency, and reduce immunogenicity.140 This approach enables prolonged protein synthesis (lasting 1–2 weeks) while ensuring proper native conformation and post-translational modifications—a key advantage over recombinant protein therapies.141 Additionally, cmRNA mitigates safety concerns associated with viral vectors in DNA-based gene therapy.
To improve cmRNA efficacy, researchers have focused on both sequence engineering and delivery strategies. One common approach to enhance translation is pyrimidine substitution, where uridine and cytidine are partially replaced with for instance 2-thiouridine and 5-methylcytidine.47 This modification reduces Toll-like receptor (TLR) and RIG-I recognition, minimizing immune activation and inflammation.142 Another optimization involves minimizing the 5′-UTR, as excessive secondary structures can hinder ribosome binding and slow translation. For example, Zhang et al. demonstrated that incorporating a translation initiator of short untranslated regions (TISU) into BMP-2 cmRNA significantly improved mRNA stability and transfection efficiency, leading to robust BMP-2 expression.143
Like all nucleic acid-based therapies, cmRNA delivery remains a major challenge due to its highly anionic nature and susceptibility to RNase degradation. To achieve efficient cellular uptake, cmRNA must be encapsulated in carriers with strong cell-penetrating properties. Among the various delivery systems, cationic lipids have shown the most promise as non-viral vectors for mRNA transport. Lipid nanoparticles (LNPs) have been successfully utilized to deliver BMP-2 cmRNA for bone regeneration.144
Beyond lipid-based vectors, researchers have explored cationic polymers—such as polyamines, block copolymers, and polypeptides—for mRNA delivery. However, few polymeric systems have been applied to bone regeneration. Interestingly, one study found that cmRNA transfection of human MSCs, fibroblasts, and osteoblasts using the commercial polymeric vector TransIT-X2 resulted in significantly lower protein expression compared to lipid-based vectors.145 In contrast, pDNA transfection with the same polymeric vector resulted in higher protein expression, highlighting the need for further optimization of polymeric carriers for efficient cmRNA delivery in bone repair applications. In this context, studies have demonstrated that bone tissue formation can be induced by delivering BMP-2/NS1 mRNAs loaded into lipopolyplexes. These are hybrid delivery alternatives, consisting of polycations that condense nucleic acids and lipid components that coat the polyplex to enhance membrane interaction and cellular uptake.146
Previous studies have leveraged cmRNA encoding BMP-2 in combination with biomaterials to create transcript-activated matrices (TAMs) for osteogenic differentiation. For instance, researchers have combined BMP-2 cmRNA with fibrin gel and micro-macro biphasic calcium phosphate (MBCP) granules to construct TAMs.147 These materials facilitated sustained release of hBMP-2 cmRNA for 7 days, with a faster release observed from MBCP granules compared to fibrin gels. To further demonstrate the potential of biomaterial-assisted cmRNA delivery for bone regeneration, another study explored cmRNA-based BMP-2 coatings on titanium implants, assessing different coating strategies and biomaterial incorporation techniques.148 Researchers applied cmRNA coatings using three biocompatible materials: poly(d,l-lactic acid) (PDLLA), fibrin, and fibrinogen. These coatings significantly improved transfection efficiency by delaying cmRNA release.
Several studies have demonstrated the effectiveness of cmRNA encoding BMP-2, BMP-7, and BMP-9 in promoting bone healing across in vitro and in vivo models.47,149,150 Among these, BMP-2 cmRNA has been the most widely explored. A recent study by De La Vega et al. examined the use of optimized BMP-2 cmRNA for healing large segmental osseous defects in rats.150 The cmRNA remained localized at the defect site, generating sustained BMP-2 expression for several days. Notably, bone defects healed with doses as low as 25 μg of BMP-2 cmRNA, and 50 μg treatments successfully bridged defects within 4 weeks—without the excessive callus formation observed with recombinant human BMP-2. This study highlighted cmRNA’s ability to accelerate fracture healing while promoting natural bone remodeling compared to recombinant proteins.
An innovative approach also explored the co-delivery of cmRNA encoding VEGF and Runx2. When applied to rat mandibular defects, this combination significantly enhanced vascularization and bone formation.151 This strategy highlights the potential of multi-gene cmRNA therapies and extends beyond traditional growth factor delivery by incorporating transcription factors, offering new alternatives that can further be explored to enhance bone formation.
Gene therapy has historically been viewed as a high-risk and costly intervention, subject to strict regulatory scrutiny, particularly in non-lethal conditions such as musculoskeletal injuries.152 The lengthy and expensive approval process has contributed to its limited adoption.153 However, cmRNA therapy has the potential to shift this perception by offering a safer and more affordable alternative, particularly in bone regeneration, where cost-effective solutions are crucial to improving the lives of millions affected by fractures.
Despite its promise, cmRNA therapy still faces significant challenges. Comprehensive pharmacokinetic, toxicological, and biodistribution studies will be essential before clinical translation. Additionally, demonstrating efficacy in large-animal models will likely be a prerequisite for advancing to human clinical trials.
microRNA for fracture healing
MicroRNAs (miRNAs) are a class of small, non-coding RNA molecules, approximately 22 nucleotides in length, that play a pivotal role in post-transcriptional gene regulation.154 They influence a wide range of cellular processes—including differentiation, proliferation, and apoptosis155—and have emerged as critical regulators of osteogenesis by modulating the expression of genes involved in osteoblast differentiation and function.156,157 Numerous miRNAs have been identified as either promoters or inhibitors of bone formation, acting through various molecular targets and signaling pathways, for example (Table 2).
To deliver therapeutic miRNAs, both viral and non-viral vector systems have been explored. Viral vectors, such as lentiviruses, offer high transfection efficiency and stable gene expression, but carry risks including insertional mutagenesis and immunogenicity, which limit their clinical application.158 In contrast, non-viral vectors—such as nanoparticles, liposomes, and exosomes—provide a safer alternative with tunable properties for targeted and controlled miRNA delivery.159,160
Additionally, miRNA-functionalized scaffolds made from biomaterials like collagen, chitosan, or tricalcium phosphate have been engineered to promote localized and sustained osteogenic effects, showing promising results in preclinical models of bone regeneration.160,161,162,163
Despite these advances, challenges such as susceptibility to enzymatic degradation, off-target effects, and the development of efficient, tissue-specific delivery systems must be addressed before miRNA-based therapies can be translated into routine clinical practice.
Peptides
Peptides are short amino acid oligomers derived from the active domains of proteins, which often lack a stable 3D structure. They can be derived from larger proteins or designed de novo. They present a cost-effective and easily synthesizable alternative to protein therapy.164 These peptides typically function by binding to high-affinity receptors on target cells, enhancing bioavailability and targeted delivery to fracture sites.
Several peptides have been engineered to enhance osteogenic responses, many of which are derived from growth factors and bone-related proteins. Lactoferrin-derived peptides and calcitonin gene-related peptides, for example, have been reported to promote fracture healing and enhance bone formation in vivo.164,165 Another interesting peptide is thrombin peptide 508 (TP508), or Chrysalin, which maintains thrombin’s regenerative properties without its clotting effect, enhancing osteoblast proliferation, differentiation, and chemotaxis.166,167
Peptides serve various functions in bone regeneration, such as promoting cell binding, enhancing osteoblast adhesion, and facilitating scaffold integration.168 P-15, a 15-amino acid sequence identical to the cell-binding region of collagen type I, improves cell attachment to bone substitutes and stimulates extracellular matrix production.169 RGD-containing peptides support osteoblast adhesion, proliferation, and osteogenic marker expression on scaffolds and graft materials.170,171 Meanwhile, self-assembling peptides, often described as “molecular Lego”, feature alternating hydrophilic and hydrophobic amino acids, enabling the formation of nanofiber matrices that enhance cell adhesion, osteoblast differentiation, and bone volume.172,173
For a comprehensive review of clinical and experimental studies on peptides in bone regeneration, the literature article by Puntos et al. provides an in-depth analysis of their therapeutic potential.168
Small molecules
Small molecules with bone regenerative potential have gained attention due to their efficacy, stability, and cost-effectiveness.174,175 Their small size (<1 000 Da) reduces the likelihood of triggering immune responses,176 and unlike protein therapies that depend on maintaining a specific three-dimensional structure for bioactivity, small molecules are typically less susceptible to structural changes and retain their function under a wider range of conditions.177,178 Over the last years, advances in high-throughput screening have led to the discovery of numerous osteoinductive small molecules.179,180,181
Among the most studied is simvastatin, which has demonstrated increased body weight, bone mineral content, and mechanical strength in osteoporotic animal models.182,183 The osteogenic oxysterol Oxy133 has shown comparable efficacy to recombinant human BMP-2 in spinal fusion while avoiding its adverse effects.184 Interestingly, FTY720, a sphingosine-1-phosphate analog, has shown contradictory results in fracture healing studies. While some research suggests that local delivery of FTY720 enhances both bone formation and microvascular development in a rat cranial defect model,185,186 another study reported that systemic administration of FTY720 did not significantly improve bone regeneration in a murine fracture model.187 These findings highlight the importance of delivery strategy, as the therapeutic efficacy of small molecules like FTY720 may depend heavily on their mode and site of administration.
Another notable molecule, FK506 (tacrolimus), functions as an immunosuppressant by inhibiting calcineurin and blocking T-cell activation in transplant recipients.188 A study by De la Vega et al. investigated FK506 in rat femoral critical-size defects using both unmodified and adenovirally transduced MSCs expressing BMP-2.189 Interestingly, defects did not heal in the presence or absence of FK506 when unmodified cells were used. However, when combined with BMP-2-expressing cells, FK506 led to complete defect healing, resulting in a slimmer callus, improved cortication, and advanced marrow reconstitution. These findings suggest that FK506 alone may not significantly influence intrinsic bone healing but can enhance the regenerative response when paired with BMP-2-expressing MSCs.
Additionally, other small molecules have also demonstrated significant osteoinductive potential. For instance, the small-molecule BMP activator SVAK-12 markedly accelerated fracture healing in a rat femoral fracture model without requiring exogenous rhBMPs.190 Moreover, some small compounds have been evaluated in large animal models, further supporting their translational potential. One such example is calcium phosphate-coated implants releasing bisphosphonates, which enhanced periprosthetic bone density and implant integration in an osteoporotic sheep model, underscoring their promise in orthopedic applications.191 Beyond these, numerous small molecules have been investigated for their osteogenic properties. Zhang et al. reviewed various small-molecule amides, detailing their biochemical properties and regulatory mechanisms in bone regeneration.192
These findings underscore the potential of small molecules as scalable, stable, and cost-effective alternatives to protein-based therapies, offering innovative strategies for bone repair and regeneration.
Cell therapy and platelet-based strategies
Cell therapy
Cell therapies involve transplanting stem cells, progenitor cells, or differentiated cells, which can come from processed host tissue, culture-expanded stem cells, purified populations of stem cells, or genetically modified stem cells.193 These therapies are appealing for treating impaired bone healing because they are typically autologous, meaning they come from the patient’s own body, thus minimizing the risk of rejection.194
Stem cells have the capacity to proliferate and differentiate into distinct cell lineages, including osteoprogenitors. The most abundant sources of stem cells are adipose tissue and bone marrow, however they can be found in other tissues in smaller numbers (peripheral blood, muscle, dermis, etc.).195,196 Several studies have demonstrated the efficiency and safety of MSCs in bone healing in clinical settings. In a pilot clinical study, Marcacci and colleagues treated four patients with diaphyseal bone defects using autologous bone marrow-derived osteoprogenitor. Complete healing was achieved in all patients (two with ulna defects, one with a humerus defect, and one with a tibial defect) within six months, with no reported complications over a 6–7-year follow-up period.197 Quarto et al. successfully treated large bone defects in three patients using autologous bone marrow-derived MSCs on a ceramic scaffold. Radiographs and computed tomography scans showed significant callus formation and good integration with the host bone by the second month after surgery.198 Additionally, in a multicenter clinical trial, 64 patients with delayed long bone fracture healing were treated with autologous cultured osteoblasts or no treatment. The osteoblast group showed a significant increase in radiographic callus formation at two months compared to controls, highlighting the potential of cellular therapies in bone repair.199
A common option to deliver stem cells is via bone marrow aspirates (BMAs). These aspirates contain a heterogeneous population of cells, including stem cells, progenitor cells, and hematopoietic cells. Thus, BMAs contain cells capable of differentiating into osteoblasts when exposed to osteoinductive signals and have been employed to treat delayed unions and nonunions.200,201 While the iliac crest has traditionally been the primary site for bone marrow aspiration, other sites such as the vertebral body, proximal humerus, proximal and distal tibia, calcaneus, and fibula have also been used.
Despite their promise, cell therapies face significant challenges. Isolating and expanding MSCs can be labor-intensive, particularly in older patients, where stem cell numbers and function decline.202 Ensuring consistent differentiation into osteogenic lineages remains a hurdle, as factors such as the donor’s age, health status, and genetic background can influence the behavior and regenerative potential of the cells.203 This variability can lead to inconsistent outcomes that influence cell fate. Moreover, the high costs of cell isolation, expansion, and regulatory compliance limit widespread clinical application. There is also a potential risk of unwanted differentiation or tumorigenesis, especially with induced pluripotent stem cells (iPSCs).204 Nonetheless, advances in cell engineering, biomaterials, and bioreactors continue to improve the feasibility of cell-based approaches for bone regeneration.
Interestingly, most current clinical trials for nonunions, registered in ClinicalTrials.gov, are exploring cell-based strategies. Cellular mechanisms with their multifaceted regenerative capabilities— including secreting growth factors, modulating inflammation, and directly contributing to tissue formation—offer a comprehensive approach to tackling the biological complexities of nonunion. Table 3 provides a detailed overview of various clinical trials currently registered for bone regeneration in nonunions. While challenges related to standardization, scalability, and long-term safety persist, ongoing research and the increasing number of clinical investigations highlight cell therapy as a cornerstone of future nonunion treatment strategies.
Platelet-based strategies
A relatively novel approach in bone regeneration involves platelet-rich plasma (PRP), a blood-based therapy utilizing platelets rich in growth factors and cytokines that play a crucial role in the early phases of bone regeneration.205 However, PRP research is still in its early stages, and its efficacy as a standalone treatment remains unclear, as it is often used in combination with other adjuvants.
PRP is typically obtained by drawing patient’s blood into a tube containing an anticoagulant, followed by centrifugation. In some cases, it is further treated with calcium chloride and bovine thrombin to induce platelet activation and form a gel-like matrix for direct application. This activation triggers platelet degranulation, leading to the sustained release of growth factors, while the gel serves as a biological carrier that prolongs their availability at the treatment site.206 Since PRP is autologous, it is generally safe and non-immunogenic. However, the use of bovine thrombin carries a risk of autoantibody formation and coagulopathies.207
In vitro studies have shown that PRP promotes MSCs proliferation and osteogenic differentiation, though excessive concentrations may have an inhibitory effect.208 Clinical trials have produced mixed results. For instance, one study comparing PRP-enhanced bone grafts to standard bone grafts in spinal fusion found no difference in nonunion rates, leading the authors to advise against PRP use.209 Conversely, another study on tibial osteotomies reported faster union in the PRP-treated group, yet no long-term functional benefits were observed.210
Currently, PRP remains an exploratory therapy for bone healing. Due to inconsistent clinical outcomes and uncertainties regarding optimal concentration, it cannot yet be recommended for routine fracture treatment.
Synergistic potential with advanced biomaterials
The therapeutic efficacy of biologics for bone regeneration can be significantly amplified by their combination with biomaterials. Beyond simply serving as inert delivery vehicles, biomaterial scaffolds are designed to provide crucial structural support for defect filling, offering a temporary template for new bone formation.211 More importantly, these materials can be engineered to precisely control the spatial and temporal release kinetics of incorporated biologics, ensuring sustained and localized concentrations at the repair site.212,213 This controlled delivery minimizes systemic exposure, reduces off-target effects, and optimizes the therapeutic window for cellular responses.
Furthermore, biomaterials can exert intrinsic biological cues that actively participate in the regenerative process.214 In particular, scaffolds composed of bioactive ceramics, polymers, or composites can provide critical structural support in bone defects, mimicking the native extracellular matrix and creating a permissive environment for cell infiltration, attachment, and differentiation.215,216,217 Their surface properties, pore architecture, and mechanical characteristics can be tailored to influence cell adhesion, proliferation, differentiation, and migration, thus guiding the host’s endogenous regenerative machinery.147,218 When integrated with biologics, such as growth factors or cells, these interactive scaffolds create a microenvironment that not only presents the therapeutic agent but also provides the necessary physical and chemical signals to profoundly enhance osteoinduction, osteoconduction, and overall tissue integration, thereby maximizing the potential for successful bone regeneration.219,220
Potential clinical applications
The use of biologics is not limited to patients suffering from severe traumatic injuries that result in nonunion fractures. Biologic therapies are also highly relevant for individuals with degenerative and genetic bone diseases, which can predispose them to non-self-healing fractures (Fig. 4). In the following sections, we discuss key clinical applications where biologics have already shown, or are expected to have, a significant impact in enhancing fracture healing.
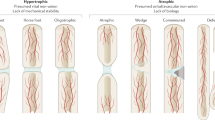
Pathological conditions associated with difficult-to-heal bone fractures. Several bone diseases, including osteoporosis, Paget’s disease of bone, rickets, osteomalacia, osteogenesis imperfecta, and bone tumors, predispose patients to recurrent fractures with delayed or failed healing. This schematic highlights the key characteristics of each condition. Patients affected by these disorders present deficiencies in bone metabolism, structural integrity, or cellular function; therefore, they may particularly benefit from biologic therapies designed to stimulate bone regeneration
Osteoporosis
Osteoporosis is characterized by low bone mineral density and deteriorated bone microarchitecture, leading to increased bone fragility, particularly among the elderly and postmenopausal women.221 Individuals with osteoporosis face a high risk of fractures, which can be physically debilitating and contribute to a decline in overall health and quality of life.222 The most common form, primary osteoporosis, occurs naturally with aging, whereas secondary osteoporosis results from underlying diseases or long-term medication use.223
Osteoporotic fractures, particularly those affecting the spine, pelvis, or hip, are associated with increased morbidity and mortality.224 Most epidemiological studies have focused on hip fractures, which have the most severe sequelae and are the easiest to study since most patients undergo hospitalization. Contrarily, vertebral fractures are harder to track as they can be asymptomatic.224
Biologic therapies such as denosumab, a monoclonal antibody that inhibits osteoclast activity, have been shown to reduce fracture risk in postmenopausal osteoporosis.225 Other anti-resorptive therapies include bisphosphonates, which increase bone mineral density and reduce fracture incidence.223 Additionally, biologics like romosozumab (Evenity®), which target sclerostin, exhibit both anabolic and anti-resorptive effects. This dual mechanism makes romosozumab particularly effective for treating osteoporosis by simultaneously promoting bone formation and reducing bone resorption.226,227,228
Emerging gene therapy strategies for osteoporosis have shown promise in preclinical studies, where engineered cells overexpressing BMP-2 or type I collagen successfully increased bone mass and accelerated fracture healing in osteoporotic animal models, including rats and sheep.229,230,231 Beyond conventional DNA-based gene therapy, cmRNA approaches are also being explored for their therapeutic potential in osteoporosis. For instance, a study by Liu et al. demonstrated that systemic delivery of 7-methylguanosine (m⁷G)-modified Runx2 mRNA using bone-targeted lipid nanoparticles effectively enhanced bone formation in senile osteoporotic mice, highlighting cmRNA as a promising and translatable strategy for treating osteoporotic-related fractures.232
Rickets and osteomalacia
Two rather uncommon diseases prone to end up in fracture are rickets and osteomalacia, affecting children and adults, respectively. They are caused by defective bone mineralization due to vitamin D deficiency or genetic mutations affecting calcium and phosphate metabolism.233 Rickets results in skeletal deformities, such as bowed legs, due to impaired calcium and phosphate deposition during bone growth.234 In contrast, osteomalacia, which occurs in adults after bone growth has ceased, does not cause deformities but increases fracture risk, particularly in weight-bearing bones such as the pelvis, hip, femur and tibia.235,236
These conditions may arise from environmental factors, such as inadequate sun exposure, or genetic mutations affecting vitamin D metabolism.237 Vitamin D is synthesized in the skin upon sunlight exposure, so individuals living in high latitudes or those with chronic illnesses that limit outdoor activity are at increased risk.233 Additionally, gastrointestinal diseases like gastrectomy and malabsorption syndromes can impair vitamin D absorption, further contributing to these disorders.224
X-linked hypophosphatemic rickets (XLH) is the most common inherited form of rickets, caused by mutations in the PHEX gene that lead to excessive production of fibroblast growth factor 23 (FGF23).238 This hormone reduces phosphate reabsorption in the kidneys, resulting in chronic hypophosphatemia and impaired bone mineralization. Treatment with burosumab (Crysvita), a fully human monoclonal antibody that inhibits FGF23, has been shown to increase serum phosphate levels and enhance skeletal mineralization.239 As such, burosumab represents a promising biologic therapy to support fracture healing in patients with XLH. Similarly, children with hypophosphatasia (HPP)—a rare genetic disorder caused by deficient tissue-nonspecific alkaline phosphatase (TNSALP)—may benefit from enzyme replacement therapy with asfotase alfa, a recombinant form of ALP.240 By hydrolyzing inorganic pyrophosphate (PPi), a natural inhibitor of mineralization, asfotase alfa restores proper bone mineral deposition, promoting improved healing of fractures and pseudofractures in patients with HPP.241
Overall, studies specifically targeting fracture healing in the context of rickets and osteomalacia remain largely unexplored, but cmRNAs offer a promising approach. These can be engineered to transiently express osteogenic factors such as Runx2, Osterix, or BMPs to promote bone formation, while simultaneously addressing the underlying metabolic deficiencies. For example, in osteomalacia, cmRNAs encoding functional 1α-hydroxylase (CYP27B1) could enhance the local conversion of inactive to active vitamin D (1,25(OH)₂D₃), boosting calcium absorption and osteoblast activity.242 In hereditary hypophosphatemic rickets, cmRNAs could be designed to encode FGF23 antagonists or regulators of phosphate reabsorption in the kidney, restoring phosphate balance and improving bone mineralization.243
Paget’s disease of bone
Paget’s disease of bone is a progressive disorder of bone remodeling, characterized by excessive osteoclast activity, leading to rapid and disorganized bone formation.244 It commonly affects the spine, pelvis, legs, and skull, increasing susceptibility to bone pain, deformity, and fractures.224 The prevalence of Paget’s disease is approximately 1.3% among individuals aged 45–74, but many cases remain undiagnosed due to the asymptomatic nature of early-stage disease.244 Although the exact cause remains unclear, possible contributors include slow viral infections, environmental toxins such as arsenic and lead, and genetic predisposition, as familial aggregation is observed in 40% of cases.245,246
The incidence of Paget’s disease has declined in recent years due to the widespread use of effective anti-resorptive therapies such as bisphosphonates and calcitonin. Nonetheless, severe cases and untreated individuals remain at high risk of pathological fractures, particularly transverse femoral fractures, which are prone to delayed healing or nonunion.247 These clinical challenges arise from the hallmark features of Paget’s disease: aberrant bone remodeling characterized by excessive osteoclast-mediated resorption followed by disorganized and mechanically compromised bone formation. Interestingly, Dkk1 RNA and protein levels have been reported to increase in pagetic osteoblast and stromal cells,248 as well as in the circulating serum of affected patients,248 suggesting that Dkk1 may be a viable therapeutic target for modulating dysregulated bone formation.
While cell-based therapies remain underexplored in the context of Paget’s disease–related fractures, they present a compelling theoretical approach due to their capacity to both supply functional osteoprogenitors and modulate the local bone remodeling environment. For instance, delivering exogenous MSCs could augment the pool of functional osteoprogenitors at the fracture site, which might be critical given the dysregulated endogenous bone cell behavior in Paget’s disease.
Novel approaches using microRNA therapeutics offer promising avenues to enhance local bone regeneration and correct remodeling defects. For instance, inhibiting miRNAs like miR-223, which promotes osteoclast differentiation, may help suppress excessive bone resorption.249 Delivering these therapeutics locally via nanoparticles or scaffold-based systems could enable targeted, site-specific action while minimizing systemic side effects. Importantly, miRNA therapies are non-integrative, reversible, and can be tailored or combined with other biologics, making them a versatile and personalized strategy for treating fractures associated with Paget’s disease.
Osteogenesis imperfecta
Osteogenesis imperfecta (OI) is one of the most common genetic bone disorders, characterized by mutations affecting type I collagen production, resulting in bones that fracture easily.250 The severity of OI varies across subtypes; while mild cases may be asymptomatic, more severe forms are associated with frequent fractures, short stature or growth retardation, hearing loss, dental abnormalities, and ligament laxity.251 The most harsh cases generally lead to early death as the mutations are incompatible with life.224
Approximately 75% of OI-related fractures occur in childhood, but fractures in adulthood present additional challenges due to bone deformities and prior surgeries.251 Delayed unions and nonunion are often encountered and in such cases, implanting a bone graft is required with complex surgeries and frequent post-operative problems.252 Given the genetic basis of OI, personalized treatment strategies are essential. Some OI patients exhibit high bone mineralization, making their bones more brittle, rather than simply lacking bone mass.253 Gene and stem cell therapies are under investigation, with efforts focused on delivering wild-type collagen genes while silencing mutant collagen alleles using plasmid DNA and antisense gene therapy.254,255
Like its application in osteoporosis, therapies targeting sclerostin inhibition have shown promise in enhancing osteoblastic bone formation in OI. In a murine model of type III OI, treatment with an anti-sclerostin antibody significantly improved BMD, tibial cortical thickness, ultimate load, and stiffness.256 Furthermore, an open-label phase 2 clinical trial evaluated the pharmacodynamics and safety of BPS804, an anti-sclerostin antibody, in adults with moderate OI. Patients received three escalating doses (5, 10, and 20 mg/kg) at two-week intervals, while a reference group received no treatment. The therapy was well tolerated and resulted in increased bone formation, reduced bone resorption, and improved lumbar spine BMD.257
Bone tumors and other malignancies
Bone tumors are abnormal growths within bone tissue that can be classified as either benign (non-cancerous) or malignant (cancerous). Malignant bone tumors are further categorized into primary tumors, which originate in the bone, and secondary tumors, which result from metastasis of cancers such as breast, prostate, and pancreatic cancer.224 Multiple myeloma is a common example of a secondary bone tumor, leading to bone destruction in over 80% of affected patients.258 Metastatic bone disease often affects the spine, pelvis, hip, femur, and skull, leading to severe pain, pathological fractures, and hypercalcemia.259,260
Fractures associated with bone tumors, whether primary or metastatic, pose significant clinical challenges due to persistent tumor-induced osteolysis and a compromised bone healing environment.260 In addition, cancer therapies—including hormone treatments, chemotherapy (particularly glucocorticoid-based regimens), and radiation—further reduce bone mineral density and accelerate bone loss. Addressing these complications requires a multidisciplinary strategy that not only promotes bone regeneration but also targets the underlying malignancy.261
mRNA therapeutics offer a particularly promising avenue. Beyond delivering cmRNAs encoding osteoinductive factors, cmRNAs can be engineered to express osteoprotegerin (OPG) or RANKL inhibitors, thereby limiting osteoclast-mediated bone resorption and restoring remodeling balance. Moreover, multi-targeted platforms could be designed to co-deliver anti-tumor mRNAs—such as those encoding immunostimulatory cytokines or apoptosis-inducing proteins—together with bone-regenerative cues, enabling localized and synergistic treatment of both the tumor and the fracture.262
Complementary to this molecular approach, bioactive scaffolds have shown strong clinical potential. For example, a study by Wang et al. introduced an innovative approach for reconstructing bone defects following the surgical excision of benign tumors in the lower extremities.263 The authors employed bioactive scaffolds combined with a proprietary stem cell rapid screening–enrichment–composite technology. When applied in clinical settings, these scaffolds seeded with enriched BMSCs significantly enhanced bone integration and healing, offering a promising and translational solution for improving postoperative outcomes in patients with tumor-related bone defects.263
Conclusions
Biologic therapies represent a promising frontier in the treatment of nonunions. While protein and gene therapies have been explored for years, their clinical translation has been slow and limited. Emerging strategies, such as transcript therapy using chemically modified mRNA, along with advances in microRNA-based regulation, bioactive peptides, and small molecule modulators, offer a new wave of precise, controllable, and safer regenerative interventions.
As our understanding of bone regeneration at the molecular and cellular mechanisms deepens, we can develop more precise, biology-driven regenerative therapies. Future strategies should not only target osteogenesis but also incorporate key aspects of bone healing, including innervation, angiogenesis, and controlled inflammation, to promote the formation of fully functional bone tissue.
Personalized medicine is essential for optimizing biologic treatments, considering factors like age, sex, fracture type, and underlying bone pathologies. Advanced strategies could involve delivering a cocktail of transcription factors, growth factors, or ECM proteins delivered via transcript therapy with spatial and temporal gradients, along with miRNA mimics or inhibitors, mimicking natural bone healing, enabling precise control while maintaining low therapeutic doses. Additionally, hybrid approaches combining stem cell therapies may offer more effective and accelerated healing solutions.
To ensure widespread accessibility, cost-effective production methods and advanced delivery systems must be developed. Finally, robust clinical trials are necessary to validate the long-term safety and efficacy of these therapies across diverse patient populations and fracture types.
References
Collaborators, G. M. D Global, regional, and national burden of 12 mental disorders in 204 countries and territories, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet Psychiatry 9, 137–150 (2022).
Calori, G., Mazza, E., Colombo, M., Ripamonti, C. & Tagliabue, L. Treatment of long bone non-unions with polytherapy: indications and clinical results. Injury 42, 587–590 (2011).
Frölke, J. P. M. & Patka, P. Definition and classification of fracture non-unions. Injury 38, S19–S22 (2007).
Santolini, E., West, R. & Giannoudis, P. V. Risk factors for long bone fracture non-union: a stratification approach based on the level of the existing scientific evidence. Injury 46, S8–S19 (2015).
Mathieu, M. et al. Decreased pool of mesenchymal stem cells is associated with altered chemokines serum levels in atrophic nonunion fractures. Bone 53, 391–398 (2013).
Heaney, R. P., Recker, R. R. & Saville, P. D. Menopausal changes in bone remodeling. J. Lab. Clin. Med. 92, 964–970 (1978).
Coipeau, P. et al. Impaired differentiation potential of human trabecular bone mesenchymal stromal cells from elderly patients. Cytotherapy 11, 584–594 (2009).
Eriksen, E. F. et al. Cancellous bone remodeling in type I (postmenopausal) osteoporosis: quantitative assessment of rates of formation, resorption, and bone loss at tissue and cellular levels. J. Bone Miner. Res. 5, 311–319 (1990).
Mills, L. A., Aitken, S. A. & Simpson, A. H. R. The risk of non-union per fracture: current myths and revised figures from a population of over 4 million adults. Acta Orthop. 88, 434–439 (2017).
Dahabreh, Z., Calori, G., Kanakaris, N., Nikolaou, V. & Giannoudis, P. A cost analysis of treatment of tibial fracture nonunion by bone grafting or bone morphogenetic protein-7. Int. Orthop. 33, 1407–1414 (2009).
Athanasiou, V. T. et al. Histological comparison of autograft, allograft-DBM, xenograft, and synthetic grafts in a trabecular bone defect: an experimental study in rabbits. Med. Sci. Monit. 16, BR24–BR31 (2009).
Pereira, H. F., Cengiz, I. F., Silva, F. S., Reis, R. L. & Oliveira, J. M. Scaffolds and coatings for bone regeneration. J. Mater. Sci. Mater. Med 31, 27 (2020).
Dahabreh, Z., Dimitriou, R. & Giannoudis, P. V. Health economics: a cost analysis of treatment of persistent fracture non-unions using bone morphogenetic protein-7. Injury 38, 371–377 (2007).
Paul, L. et al. Selection of massive bone allografts using shape-matching 3-dimensional registration. Acta Orthop. 81, 250–255 (2010).
Putzer, D., Coraça-Huber, D., Wurm, A., Schmoelz, W. & Nogler, M. Optimizing the grain size distribution of allografts in bone impaction grafting. J. Orthop. Res. 32, 1024–1029 (2014).
Coquelin, L. et al. In vivo and in vitro comparison of three different allografts vitalized with human mesenchymal stromal cells. Tissue Eng. Part A 18, 1921–1931 (2012).
Aro, H. T. & Aho, A. J. Clinical use of bone allografts. Ann. Med. 25, 403–412 (1993).
Kim, Y., Nowzari, H. & Rich, S. K. Risk of prion disease transmission through bovine-derived bone substitutes: a systematic review. Clin. Implant Dent. Relat. Res. 15, 645–653 (2013).
Pountos, I. & Giannoudis, P. V. Is there a role of coral bone substitutes in bone repair?. Injury 47, 2606–2613 (2016).
Bertol, L. S., Schabbach, R. & dos Santos, L. A. L. Dimensional evaluation of patient-specific 3D printing using calcium phosphate cement for craniofacial bone reconstruction. J. Biomater. Appl. 31, 799–806 (2017).
Lodoso-Torrecilla, I. et al. Multimodal pore formation in calcium phosphate cements. J. Biomed. Mater. Res. A 106, 500–509 (2018).
Stanovici, J. et al. Bone regeneration strategies with bone marrow stromal cells in orthopaedic surgery. Curr. Res. Transl. Med. 64, 83–90 (2016).
Balmayor, E. R. & Van Griensven, M. Gene therapy for bone engineering. Front. Bioeng. Biotechnol. 3, 9 (2015).
Eftekhari, Z. et al. Advancements and challenges in mRNA and ribonucleoprotein-based therapies: from delivery systems to clinical applications. Mol. Ther.-Nucleic Acids 35, 102313 (2024).
De la Vega, R. E. et al. Gene therapy for bone healing: lessons learned and new approaches. Transl. Res. 236, 1–16 (2021).
Kowalski, P. S., Rudra, A., Miao, L. & Anderson, D. G. Delivering the messenger: advances in technologies for therapeutic mRNA delivery. Mol. Ther. 27, 710–728 (2019).
Yin, J. & Luan, S. Opportunities and challenges for the development of polymer-based biomaterials and medical devices. Regen. Biomater. 3, 129–135 (2016).
Li, W. & Szoka, F. C. Lipid-based nanoparticles for nucleic acid delivery. Pharm. Res. 24, 438–449 (2007).
Ghassemi, T. et al. Current concepts in scaffolding for bone tissue engineering. Arch. bone Jt. Surg. 6, 90 (2018).
Sfeir, C., Ho, L., Doll, B. A., Azari, K. & Hollinger, J. O. in Bone Regeneration and Repair 21–44 (Springer, 2005).
Devescovi, V., Leonardi, E., Ciapetti, G. & Cenni, E. Growth factors in bone repair. La Chirurgia Degli Organ. di Mov. 92, 161–168 (2008).
Katagiri, T., Tsukamoto, S., Osawa, K. & Kokabu, S. in A Tissue Regeneration Approach to Bone and Cartilage Repair 1–15 (Springer, 2015).
Ozaki, A., Tsunoda, M., Kinoshita, S. & Saura, R. Role of fracture hematoma and periosteum during fracture healing in rats: interaction of fracture hematoma and the periosteum in the initial step of the healing process. J. Orthop. Sci. 5, 64–70 (2000).
Schmidt-Bleek, K. et al. Inflammatory phase of bone healing initiates the regenerative healing cascade. Cell tissue Res. 347, 567–573 (2012).
Einhorn, T. A., Majeska, R. J., Rush, E. B., Levine, P. M. & Horowitz, M. C. The expression of cytokine activity by fracture callus. J. Bone Miner. Res. 10, 1272–1281 (1995).
Granero-Moltó, F. et al. Regenerative effects of transplanted mesenchymal stem cells in fracture healing. Stem cells 27, 1887–1898 (2009).
Zhang, H. et al. Acceleration of fracture healing by overexpression of basic fibroblast growth factor in the mesenchymal stromal cells. Stem Cells Transl. Med. 6, 1880–1893 (2017).
Kodama, J., Wilkinson, K. J., Iwamoto, M., Otsuru, S. & Enomoto-Iwamoto, M. The role of hypertrophic chondrocytes in regulation of the cartilage-to-bone transition in fracture healing. Bone Rep. 17, 101616 (2022).
Vimalraj, S. Alkaline phosphatase: structure, expression and its function in bone mineralization. Gene 754, 144855 (2020).
Ghimire, S. et al. The investigation of bone fracture healing under intramembranous and endochondral ossification. Bone Rep. 14, 100740 (2021).
Zhu, S., Chen, W., Masson, A. & Li, Y.-P. Cell signaling and transcriptional regulation of osteoblast lineage commitment, differentiation, bone formation, and homeostasis. Cell Discov. 10, 71 (2024).
Ford, J. L., Robinson, D. E. & Scammell, B. E. The fate of soft callus chondrocytes during long bone fracture repair. J. Orthop. Res. 21, 54–61 (2003).
Buckwalter, J. Healing of the musculoskeletal tissues. Rockwood and Green’s fractures in adults (1991).
Einhorn, T. A. The cell and molecular biology of fracture healing. Clin. Orthop. Relat. Res.® 355, S7–S21 (1998).
Moucha, C. S. & Einhorn, T. A. in Bone Regeneration and Repair 169–194 (Springer, 2005).
Schlickewei, C. W. et al. Current and future concepts for the treatment of impaired fracture healing. Int. J. Mol. Sci. 20, 5805 (2019).
Balmayor, E. R. et al. Chemically modified RNA induces osteogenesis of stem cells and human tissue explants as well as accelerates bone healing in rats. Biomaterials 87, 131–146 (2016).
Perez, J. R. et al. Tissue engineering and cell-based therapies for fractures and bone defects. Front. Bioeng. Biotechnol. 6, 105 (2018).
Cheng, H. et al. Osteogenic activity of the fourteen types of human bone morphogenetic proteins (BMPs). JBJS 85, 1544–1552 (2003).
Liu, Z., Luyten, F., Lammens, J. & Dequeker, J. Molecular signaling in bone fracture healing and distraction osteogenesis. Histol. Histopathol. 14, 587–595 (1999).
Onishi, T. et al. Distinct and overlapping patterns of localization of bone morphogenetic protein (BMP) family members and a BMP type II receptor during fracture healing in rats. Bone 22, 605–612 (1998).
Bolander, M. E. Regulation of fracture repair by growth factors. Proc. Soc. Exp. Biol. Med. 200, 165–170 (1992).
Murakami, M. et al. FGF-dependent regulation of VEGF receptor 2 expression in mice. J. Clin. Invest. 121, 2668–2678 (2011).
Naganawa, T. et al. Reduced expression and function of bone morphogenetic protein-2 in bones of Fgf2 null mice. J. Cell. Biochem. 103, 1975–1988 (2008).
Xie, H. et al. PDGF-BB secreted by preosteoclasts induces angiogenesis during coupling with osteogenesis. Nat. Med. 20, 1270–1278 (2014).
Wang, F. et al. PDGFR in PDGF-BB/PDGFR signaling pathway does orchestrates osteogenesis in a temporal manner. Research 6, 0086 (2023).
Caplan, A. I. & Correa, D. PDGF in bone formation and regeneration: new insights into a novel mechanism involving MSCs. J. Orthop. Res. 29, 1795–1803 (2011).
de Oliveira, J. C. S. et al. Evaluation of the osteoinductive effect of PDGF-BB associated with different carriers in bone regeneration in bone surgical defects in rats. Implant Dent. 26, 559–566 (2017).
Kim, H. J. et al. In ovariectomy-induced osteoporotic rat models, BMP-2 substantially reversed an impaired alveolar bone regeneration whereas PDGF-BB failed. Clin. Oral. Investig. 25, 6159–6170 (2021).
Del Rosario, C., Rodriguez-Evora, M., Reyes, R., Delgado, A. & Evora, C. BMP-2, PDGF-BB, and bone marrow mesenchymal cells in a macroporous β-TCP scaffold for critical-size bone defect repair in rats. Biomed. Mater. 10, 045008 (2015).
Wu, M., Chen, G. & Li, Y.-P. TGF-β and BMP signaling in osteoblast, skeletal development, and bone formation, homeostasis and disease. Bone Res. 4, 16009 (2016).
Steinbrech, D. S. et al. Gene expression of TGF-β, TGF-β receptor, and extracellular matrix proteins during membranous bone healing in rats. Plast. Reconstr. Surg. 105, 2028–2038 (2000).
Sarahrudi, K. et al. Elevated transforming growth factor-beta 1 (TGF-β1) levels in human fracture healing. Injury 42, 833–837 (2011).
Massague, J. The transforming growth factor-beta family. Annu. Rev. Cell Biol. 6, 597–641 (1990).
Xian, L. et al. Matrix IGF-1 maintains bone mass by activation of mTOR in mesenchymal stem cells. Nat. Med. 18, 1095 (2012).
Andrew, J., Hoyland, J., Freemont, A. & Marsh, D. Insulinlike growth factor gene expression in human fracture callus. Calcif. tissue Int. 53, 97–102 (1993).
Yakar, S., Werner, H. & Rosen, C. J. 40 YEARS OF IGF1: Insulin-like growth factors: actions on the skeleton. J. Mol. Endocrinol. 61, T115–T137 (2018).
Street, J. et al. Is human fracture hematoma inherently angiogenic? Clin. Orthop. Relat. Res. 378, 224–237 (2000).
Ancelin, M. et al. Vascular endothelial growth factor VEGF189 induces human neutrophil chemotaxis in extravascular tissue via an autocrine amplification mechanism. Lab. Invest. 84, 502–512 (2004).
Martino, M. M., Briquez, P. S., Ranga, A., Lutolf, M. P. & Hubbell, J. A. Heparin-binding domain of fibrin (ogen) binds growth factors and promotes tissue repair when incorporated within a synthetic matrix. Proc. Natl. Acad. Sci. 110, 4563–4568 (2013).
Hu, K. & Olsen, B. R. The roles of vascular endothelial growth factor in bone repair and regeneration. Bone 91, 30–38 (2016).
Beamer, B., Hettrich, C. & Lane, J. Vascular endothelial growth factor: an essential component of angiogenesis and fracture healing. HSS J. 6, 85–94 (2010).
Grills, B. L. & Schuijers, J. A. Immunohistochemical localization of nerve growth factor in fractured and unfractured rat bone. Acta Orthop. Scand. 69, 415–419 (1998).
Hukkanen, M. et al. Rapid proliferation of calcitonin gene-related peptide-immunoreactive nerves during healing of rat tibial fracture suggests neural involvement in bone growth and remodelling. Neuroscience 54, 969–979 (1993).
Li, Z. et al. Fracture repair requires TrkA signaling by skeletal sensory nerves. J. Clin. Invest. 129, 5137–5150 (2019).
Parker, R. S. et al. Role of the neurologic system in fracture healing: an extensive review. Curr. Osteoporos. Rep. 22, 205–216 (2024).
Madsen, J. E. et al. Fracture healing and callus innervation after peripheral nerve resection in rats. Clin. Orthop. Relat. Res. 351, 230–240 (1998).
Nordsletten, L. et al. The neuronal regulation of fracture healing: effects of sciatic nerve resection in rat tibia. Acta Orthop. Scand. 65, 299–304 (1994).
Taghi, G. M., Maryam, G. K., Taghi, L., Leili, H. & Leyla, M. Characterization of in vitro cultured bone marrow and adipose tissue-derived mesenchymal stem cells and their ability to express neurotrophic factors. Cell Biol. Int. 36, 1239–1249 (2012).
Nakanishi, T. et al. Expression of nerve growth factor family neurotrophins in a mouse osteoblastic cell line. Biochem. Biophys. Res. Commun. 198, 891–897 (1994).
Stapledon, C. J. et al. Human osteocyte expression of nerve growth factor: the effect of pentosan polysulphate sodium (PPS) and implications for pain associated with knee osteoarthritis. PLoS One 14, e0222602 (2019).
Takano, S. et al. Nerve growth factor regulation by TNF-α and IL-1β in synovial macrophages and fibroblasts in osteoarthritic mice. J. Immunol. Res. 2016, 5706359 (2016).
Li, R., Stewart, D. J., von Schroeder, H. P., Mackinnon, E. S. & Schemitsch, E. H. Effect of cell-based VEGF gene therapy on healing of a segmental bone defect. J. Orthop. Res. 27, 8–14 (2009).
Vukicevic, S., Sampath, K. T., Grgurevic, L., Cohen, C. & Opperman, H. (Google Patents, 2019).
Cahill, K. S., McCormick, P. C. & Levi, A. D. A comprehensive assessment of the risk of bone morphogenetic protein use in spinal fusion surgery and postoperative cancer diagnosis. J. Neurosurg. Spine 23, 86–93 (2015).
James, A. W. et al. A review of the clinical side effects of bone morphogenetic protein-2. Tissue Eng. B Rev. 22, 284–297 (2016).
Kuroda, Y. et al. Recombinant human FGF-2 for the treatment of early-stage osteonecrosis of the femoral head: TRION, a single-arm, multicenter, Phase II trial. Regen. Med. 16, 535–548 (2021).
Kawaguchi, H. et al. Local application of recombinant human fibroblast growth factor-2 on bone repair: a dose–escalation prospective trial on patients with osteotomy. J. Orthop. Res. 25, 480–487 (2007).
Nevins, M., Camelo, M., Nevins, M. L., Schenk, R. K. & Lynch, S. E. Periodontal regeneration in humans using recombinant human platelet-derived growth factor-BB (rhPDGF-BB) and allogenic bone. J. Periodontol. 74, 1282–1292 (2003).
Younger, A., Penner, M. & Montijo, H. E. Vancouver experience of recombinant human platelet-derived growth factor. Foot Ankle Clin. 21, 771–776 (2016).
DiGiovanni, C. W. et al. The importance of sufficient graft material in achieving foot or ankle fusion. JBJS 98, 1260–1267 (2016).
Haines, M. S. et al. Sequential therapy with recombinant human IGF-1 followed by risedronate increases spine bone mineral density in women with anorexia nervosa: a randomized, placebo-controlled trial. J. Bone Miner. Res. 36, 2116–2126 (2021).
Howell, T. H. et al. A phase I/II clinical trial to evaluate a combination of recombinant human platelet-derived growth factor-BB and recombinant human insulin-like growth factor-I in patients with periodontal disease. J. Periodontol. 68, 1186–1193 (1997).
Nevins, M. et al. Platelet-derived growth factor stimulates bone fill and rate of attachment level gain: results of a large multicenter randomized controlled trial. J. Periodontol. 76, 2205–2215 (2005).
Saag, K. G. et al. Effects of teriparatide versus alendronate for treating glucocorticoid-induced osteoporosis: thirty-six–month results of a randomized, double-blind, controlled trial. Arthritis Rheumatism 60, 3346–3355 (2009).
Neer, R. M. et al. Effect of parathyroid hormone (1-34) on fractures and bone mineral density in postmenopausal women with osteoporosis. N. Engl. J. Med. 344, 1434–1441 (2001).
Komatsubara, S. et al. Human parathyroid hormone (1–34) accelerates the fracture healing process of woven to lamellar bone replacement and new cortical shell formation in rat femora. Bone 36, 678–687 (2005).
Cosman, F. et al. Parathyroid hormone added to established hormone therapy: effects on vertebral fracture and maintenance of bone mass after parathyroid hormone withdrawal. J. Bone Miner. Res. 16, 925–931 (2001).
Mancilla, E. E., Brodsky, J. L., Mehta, S., Pignolo, R. J. & Levine, M. A. Teriparatide as a systemic treatment for lower extremity nonunion fractures: a case series. Endocr. Pract. 21, 136–142 (2015).
Chintamaneni, S., Finzel, K. & Gruber, B. Successful treatment of sternal fracture nonunion with teriparatide. Osteoporos. Int. 21, 1059–1063 (2010).
Kastirr, I. et al. Therapy of aseptic nonunions with parathyroid hormone. Eur. J. Orthop. Surg. Traumatol. 29, 169–173 (2019).
Johansson, T. PTH 1-34 (teriparatide) may not improve healing in proximal humerus fractures: a randomized, controlled study of 40 patients. Acta Orthop. 87, 79–82 (2016).
Bhandari, M. et al. Does teriparatide improve femoral neck fracture healing: results from a randomized placebo-controlled trial. Clin. Orthop. Relat. Res. 474, 1234–1244 (2016).
Ciurlia, E., Leali, P. T. & Doria, C. Use of teriparatide off-label: our experience and review of literature. Clin. Cases Miner. Bone Metab. 14, 28 (2017).
Kim, J. B. et al. Bone regeneration is regulated by Wnt signaling. J. Bone Miner. Res. 22, 1913–1923 (2007).
Hadjiargyrou, M., Kotsiopriftis, M., Lauzier, D., Hamdy, R. C. & Kloen, P. Activation of Wnt signaling in human fracture callus and nonunion tissues. Bone Rep. 22, 101780 (2024).
Minear, S. et al. Wnt proteins promote bone regeneration. Sci. Transl. Med. 2, 29ra30–29ra30 (2010).
Suen, P. K. et al. Sclerostin monoclonal antibody enhanced bone fracture healing in an open osteotomy model in rats. J. Orthop. Res. 32, 997–1005 (2014).
Uemura, T. et al. Bone healing of distal radius nonunion treated with bridge plating with bone graft substitutes in combination with systemic romosozumab administration: a case report. Jt. Dis. Relat. Surg. 32, 526 (2021).
Shakeri, A. & Adanty, C. Romosozumab (sclerostin monoclonal antibody) for the treatment of osteoporosis in postmenopausal women: a review. J. Popul. Ther. 27, e25–e31 (2020).
Bhandari, M. et al. Romosozumab in skeletally mature adults with a fresh unilateral tibial diaphyseal fracture: a randomized phase-2 study. JBJS 102, 1416–1426 (2020).
Schemitsch, E. H. et al. A randomized, placebo-controlled study of romosozumab for the treatment of hip fractures. JBJS 102, 693–702 (2020).
Glinka, A. et al. Dickkopf-1 is a member of a new family of secreted proteins and functions in head induction. Nature 391, 357–362 (1998).
Butler, J. S. et al. Silencing Dkk1 expression rescues dexamethasone-induced suppression of primary human osteoblast differentiation. BMC Musculoskelet. Disord. 11, 1–10 (2010).
Agholme, F., Isaksson, H., Kuhstoss, S. & Aspenberg, P. The effects of Dickkopf-1 antibody on metaphyseal bone and implant fixation under different loading conditions. Bone 48, 988–996 (2011).
Jin, H. et al. Anti-DKK1 antibody promotes bone fracture healing through activation of β-catenin signaling. Bone 71, 63–75 (2015).
Lacey, D. et al. Osteoprotegerin ligand is a cytokine that regulates osteoclast differentiation and activation. Cell 93, 165–176 (1998).
Törring, O. Effects of denosumab on bone density, mass and strength in women with postmenopausal osteoporosis. Ther. Adv. Musculoskelet. Dis. 7, 88–102 (2015).
Papapoulos, S. et al. The effect of 8 or 5 years of denosumab treatment in postmenopausal women with osteoporosis: results from the FREEDOM Extension study. Osteoporos. Int. 26, 2773–2783 (2015).
Shiva, P. Effect of denosumab injection in early union and consolidation of callus in aseptic tibial shaft non-union. J. Pharm. Res. 23, 35 (2024).
Garrison, K. R. et al. Clinical effectiveness and cost-effectiveness of bone morphogenetic proteins in the non-healing of fractures and spinal fusion: a systematic review. Health Technology Assessment (Winchester, England) 11, 1–150, iii (2007).
Kanakaris, N. & Giannoudis, P. V. The health economics of the treatment of long-bone non-unions. Injury 38, S77–S84 (2007).
Kamionka, M. Engineering of therapeutic proteins production in Escherichia coli. Curr. Pharm. Biotechnol. 12, 268–274 (2011).
Zaman, R. et al. Current strategies in extending half-lives of therapeutic proteins. J. Control. Release 301, 176–189 (2019).
Beygmoradi, A., Homaei, A., Hemmati, R. & Fernandes, P. Recombinant protein expression: challenges in production and folding related matters. Int. J. Biol. Macromol. 233, 123407 (2023).
Evans, C. H., Ghivizzani, S. C. & Robbins, P. D. Orthopaedic gene therapy: twenty-five years on. JBJS Rev. 9, e20 (2021).
Rao, G. A. & Buethe, D. D. Plasmid based gene delivery for orthopedic disorders: a brief review. J. Drug Target. 12, 341–345 (2004).
Park, J. et al. Bone regeneration in critical size defects by cell-mediated BMP-2 gene transfer: a comparison of adenoviral vectors and liposomes. Gene Ther. 10, 1089–1098 (2003).
Osawa, K., Okubo, Y., Nakao, K., Koyama, N. & Bessho, K. Osteoinduction by microbubble-enhanced transcutaneous sonoporation of human bone morphogenetic protein-2. J. Gene Med. 11, 633–641 (2009).
Wang, J. C. et al. Effect of regional gene therapy with bone morphogenetic protein-2-producing bone marrow cells on spinal fusion in rats. JBJS 85, 905–911 (2003).
Van Griensven, M. et al. Adenoviral gene transfer in a rat fracture model. Lab. Anim. 36, 455–461 (2002).
Wilkinson, P. et al. Systematic review of the preclinical technology readiness of orthopedic gene therapy and outlook for clinical translation. Front. Bioeng. Biotechnol. 9, 626315 (2021).
Bozo, I., Deev, R., Drobyshev, A., Isaev, A. & Eremin, I. World’s first clinical case of gene-activated bone substitute application. Case Rep. Dent. 2016, 8648949 (2016).
Ranjbarnejad, F., Khazaei, M., Shahryari, A., Khazaei, F. & Rezakhani, L. Recent advances in gene therapy for bone tissue engineering. J. Tissue Eng. Regen. Med. 16, 1121–1137 (2022).
Masgutov, R. et al. Use of gene-activated demineralized bone allograft in the therapy of ulnar pseudarthrosis. Case report. BioNanoScience 7, 194–198 (2017).
Evans, C. H., Ghivizzani, S. & Robbins, P. D. Orthopedic gene therapy—lost in translation?. J. Cell. Physiol. 227, 416–420 (2012).
Argintar, E., Edwards, S. & Delahay, J. Bone morphogenetic proteins in orthopaedic trauma surgery. Injury 42, 730–734 (2011).
Evans, C. H. The vicissitudes of gene therapy. Bone Jt. Res. 8, 469–471 (2019).
Weissman, D. mRNA transcript therapy. Expert Rev. Vaccines 14, 265–281 (2015).
Wang, Y.-S. et al. mRNA-based vaccines and therapeutics: an in-depth survey of current and upcoming clinical applications. J. Biomed. Sci. 30, 84 (2023).
Huysmans, H. et al. Expression kinetics and innate immune response after electroporation and LNP-mediated delivery of a self-amplifying mRNA in the skin. Mol. Ther. -Nucleic Acids 17, 867–878 (2019).
Kormann, M. S. et al. Expression of therapeutic proteins after delivery of chemically modified mRNA in mice. Nat. Biotechnol. 29, 154–157 (2011).
Zhang, W. et al. An improved, chemically modified RNA encoding BMP-2 enhances osteogenesis in vitro and in vivo. Tissue Eng. Part A 25, 131–144 (2019).
Andrée, L., Yang, F., Brock, R. & Leeuwenburgh, S. Designing biomaterials for the delivery of RNA therapeutics to stimulate bone healing. Mater. Today Bio 10, 100105 (2021).
Runzer, C. D. T. et al. Cellular uptake of modified mRNA occurs via caveolae-mediated endocytosis, yielding high protein expression in slow-dividing cells. Mol. Ther. -Nucleic Acids 32, 960–979 (2023).
Wang, P. et al. Co-delivery of NS1 and BMP2 mRNAs to murine pluripotent stem cells leads to enhanced BMP-2 expression and osteogenic differentiation. Acta Biomater. 108, 337–346 (2020).
Balmayor, E. R. et al. Modified mRNA for BMP-2 in combination with biomaterials serves as a transcript-activated matrix for effectively inducing osteogenic pathways in stem cells. Stem Cells Dev. 26, 25–34 (2017).
Fayed, O., van Griensven, M., Tahmasebi Birgani, Z., Plank, C. & Balmayor, E. R. Transcript-activated coatings on titanium mediate cellular osteogenesis for enhanced osteointegration. Mol. Pharm. 18, 1121–1137 (2021).
Khorsand, B. et al. A comparative study of the bone regenerative effect of chemically modified RNA encoding BMP-2 or BMP-9. AAPS J. 19, 438–446 (2017).
De La Vega, R. E. et al. Efficient healing of large osseous segmental defects using optimized chemically modified messenger RNA encoding BMP-2. Sci. Adv. 8, eabl6242 (2022).
Zhang, M. et al. Enhancement of bone regeneration by coadministration of angiogenic and osteogenic factors using messenger RNA. Inflamm. Regen. 43, 32 (2023).
Halioua-Haubold, C.-L. et al. Focus: genome editing: regulatory considerations for gene therapy products in the US, EU, and Japan. Yale J. Biol. Med. 90, 683 (2017).
Evans, C. H. & Huard, J. Gene therapy approaches to regenerating the musculoskeletal system. Nat. Rev. Rheumatol. 11, 234–242 (2015).
Li, B. MicroRNA regulation in osteogenic and adipogenic differentiation of bone mesenchymal stem cells and its application in bone regeneration. Curr. Stem cell Res. Ther. 13, 26–30 (2018).
Nakasa, T., Yoshizuka, M., Andry Usman, M., Elbadry Mahmoud, E. & Ochi, M. MicroRNAs and bone regeneration. Curr. Genomics 16, 441–452 (2015).
Groven, R. V. et al. Early total care and damage control orthopaedics result in partially contrasting patterns of microRNA expression at the fracture site and in the systemic circulation: an animal study. Bone Jt. J. 107, 193–203 (2025).
Fang, S., Deng, Y., Gu, P. & Fan, X. MicroRNAs regulate bone development and regeneration. Int. J. Mol. Sci. 16, 8227–8253 (2015).
Liu, Z. et al. Lentivirus-mediated microRNA-26a overexpression in bone mesenchymal stem cells facilitates bone regeneration in bone defects of calvaria in mice. Mol. Med. Rep. 18, 5317–5326 (2018).
Hosseinpour, S. et al. A comparative study of mesoporous silica and mesoporous bioactive glass nanoparticles as non-viral microRNA vectors for osteogenesis. Pharmaceutics 14, 2302 (2022).
Kuang, H. et al. Integrated osteoinductive Factors─ Exosome@ MicroRNA-26a hydrogel enhances bone regeneration. ACS Appl. Mater. Interfaces 15, 22805–22816 (2023).
Zhang, X., Li, Y., Chen, Y. E., Chen, J. & Ma, P. X. Cell-free 3D scaffold with two-stage delivery of miRNA-26a to regenerate critical-sized bone defects. Nat. Commun. 7, 10376 (2016).
Remy, M. T. et al. Rat calvarial bone regeneration by 3D-printed β-tricalcium phosphate incorporating microRNA-200c. ACS Biomater. Sci. Eng. 7, 4521–4534 (2021).
Sadowska, J. M. et al. A multifunctional scaffold for bone infection treatment by delivery of microRNA therapeutics combined with antimicrobial nanoparticles. Adv. Mater. 36, 2307639 (2024).
Tian, M. et al. The role of lactoferrin in bone remodeling: evaluation of its potential in targeted delivery and treatment of metabolic bone diseases and orthopedic conditions. Front. Endocrinol. 14, 1218148 (2023).
Sample, S. J., Hao, Z., Wilson, A. P. & Muir, P. Role of calcitonin gene-related peptide in bone repair after cyclic fatigue loading. PLoS One 6, e20386 (2011).
Sheller, M. R. et al. Repair of rabbit segmental defects with the thrombin peptide, TP508. J. Orthop. Res. 22, 1094–1099 (2004).
Hedberg, E. L. et al. Effect of varied release kinetics of the osteogenic thrombin peptide TP508 from biodegradable, polymeric scaffolds on bone formation in vivo. J. Biomed. Mater. Res. A. 72, 343–353 (2005).
Pountos, I. et al. The role of peptides in bone healing and regeneration: a systematic review. BMC Med. 14, 1–15 (2016).
Nguyen, H., Qian, J. J., Bhatnagar, R. S. & Li, S. Enhanced cell attachment and osteoblastic activity by P-15 peptide-coated matrix in hydrogels. Biochem. Biophys. Res. Commun. 311, 179–186 (2003).
Durrieu, M.-C. et al. Grafting RGD containing peptides onto hydroxyapatite to promote osteoblastic cells adhesion. J. Mater. Sci. Mater. Med. 15, 779–786 (2004).
Kantlehner, M. et al. Surface coating with cyclic RGD peptides stimulates osteoblast adhesion and proliferation as well as bone formation. Chembiochem 1, 107–114 (2000).
Hou, T. et al. A composite demineralized bone matrix–self assembling peptide scaffold for enhancing cell and growth factor activity in bone marrow. Biomaterials 35, 5689–5699 (2014).
Zhang, S. Emerging biological materials through molecular self-assembly. Biotechnol. Adv. 20, 321–339 (2002).
Han, Q.-Q., Du, Y. & Yang, P.-S. The role of small molecules in bone regeneration. Future Med. Chem. 5, 1671–1684 (2013).
Laurencin, C. T., Ashe, K. M., Henry, N., Kan, H. M. & Lo, K. W.-H. Delivery of small molecules for bone regenerative engineering: preclinical studies and potential clinical applications. Drug Discov. Today 19, 794–800 (2014).
Blaich, G., Janssen, B., Roth, G. & Salfeld, J. Overview: differentiating issues in the development of macromolecules compared with small molecules. Handbook Pharm. Biotechnol. 89–123 (2007).
WH Lo, K., D. Ulery, B., Deng, M., M. Ashe, K. & T. Laurencin, C. Current patents on osteoinductive molecules for bone tissue engineering. Recent Pat. Biomed. Eng. 4, 153–167 (2011).
Lo, K. W., Ashe, K. M., Kan, H. M. & Laurencin, C. T. The role of small molecules in musculoskeletal regeneration. Regen. Med. 7, 535–549 (2012).
Hojo, H. et al. Development of high-throughput screening system for osteogenic drugs using a cell-based sensor. Biochem. Biophys. Res. Commun. 376, 375–379 (2008).
Brey, D. M. et al. High-throughput screening of a small molecule library for promoters and inhibitors of mesenchymal stem cell osteogenic differentiation. Biotechnol. Bioeng. 108, 163–174 (2011).
Vrijens, K. et al. Identification of small molecule activators of BMP signaling. PLoS One 8, e59045 (2013).
Du, Z., Chen, J., Yan, F. & Xiao, Y. Effects of Simvastatin on bone healing around titanium implants in osteoporotic rats. Clin. Oral. implants Res. 20, 145–150 (2009).
Yang, G. et al. Bone responses to simvastatin-loaded porous implant surfaces in an ovariectomized model. Int. J. Oral Maxillofac. Implants 27, 369−374 (2012).
Montgomery, S. R. et al. A novel osteogenic oxysterol compound for therapeutic development to promote bone growth: activation of hedgehog signaling and osteogenesis through smoothened binding. J. Bone Miner. Res. 29, 1872–1885 (2014).
Aronin, C. E. P. et al. The enhancement of bone allograft incorporation by the local delivery of the sphingosine 1-phosphate receptor targeted drug FTY720. Biomaterials 31, 6417–6424 (2010).
Huang, C. et al. Local delivery of FTY720 accelerates cranial allograft incorporation and bone formation. Cell Tissue Res. 347, 553–566 (2012).
Heilmann, A. et al. Systemic treatment with the sphingosine-1-phosphate analog FTY720 does not improve fracture healing in mice. J. Orthop. Res. 31, 1845–1850 (2013).
Thomson, A., Bonham, C. & Zeevi, A. Mode of action of tacrolimus (FK506): molecular and cellular mechanisms. Ther. Drug Monit. 17, 584–591 (1995).
De la Vega, R. et al. Effects of FK506 on the healing of diaphyseal, critical size defects in the rat femur. Eur. Cells Mater. 40, 160 (2020).
Wong, E. et al. A novel low-molecular-weight compound enhances ectopic bone formation and fracture repair. JBJS 95, 454–461 (2013).
Stadelmann, V. A., Terrier, A., Gauthier, O., Bouler, J.-M. & Pioletti, D. P. Implants delivering bisphosphonate locally increase periprosthetic bone density in an osteoporotic sheep model. A pilot study. Eur. Cells Mater. 16, 10–16 (2008).
Zhang, Q. et al. Small-molecule amines: a big role in the regulation of bone homeostasis. Bone Res. 11, 40 (2023).
Patterson, T. E., Kumagai, K., Griffith, L. & Muschler, G. F. Cellular strategies for enhancement of fracture repair. JBJS 90, 111–119 (2008).
Virk, M. S. & Lieberman, J. R. Biologic adjuvants for fracture healing. Arthritis Res. Ther. 14, 1–13 (2012).
Giordano, A., Galderisi, U. & Marino, I. R. From the laboratory bench to the patient’s bedside: an update on clinical trials with mesenchymal stem cells. J. Cell. Physiol. 211, 27–35 (2007).
Tsuji, W., Rubin, J. P. & Marra, K. G. Adipose-derived stem cells: implications in tissue regeneration. World J. Stem Cells 6, 312 (2014).
Marcacci, M. et al. Stem cells associated with macroporous bioceramics for long bone repair: 6-to 7-year outcome of a pilot clinical study. Tissue Eng. 13, 947–955 (2007).
Quarto, R. et al. Repair of large bone defects with the use of autologous bone marrow stromal cells. N. Engl. J. Med. 344, 385–386 (2001).
Kim, S.-J. et al. A multi-center, randomized, clinical study to compare the effect and safety of autologous cultured osteoblast (Ossron™) injection to treat fractures. BMC Musculoskelet. Disord. 10, 1–9 (2009).
Goel, A., Sangwan, S., Siwach, R. & Ali, A. M. Percutaneous bone marrow grafting for the treatment of tibial non-union. Injury 36, 203–206 (2005).
Kettunen, J., Mäkelä, E., Turunen, V., Suomalainen, O. & Partanen, K. Percutaneous bone grafting in the treatment of the delayed union and non-union of tibial fractures. Injury 33, 239–245 (2002).
Chen, H., Liu, O., Chen, S. & Zhou, Y. Aging and mesenchymal stem cells: therapeutic opportunities and challenges in the older group. Gerontology 68, 339–352 (2022).
Widholz, B., Tsitlakidis, S., Reible, B., Moghaddam, A. & Westhauser, F. Pooling of patient-derived mesenchymal stromal cells reduces inter-individual confounder-associated variation without negative impact on cell viability, proliferation and osteogenic differentiation. Cells 8, 633 (2019).
Sato, Y. et al. Tumorigenicity assessment of cell therapy products: the need for global consensus and points to consider. Cytotherapy 21, 1095–1111 (2019).
Alsousou, J., Thompson, M., Hulley, P., Noble, A. & Willett, K. The biology of platelet-rich plasma and its application in trauma and orthopaedic surgery: a review of the literature. J. Bone Jt. Surg. Br. 91, 987–996 (2009).
Sánchez, M. et al. Comparison of surgically repaired Achilles tendon tears using platelet-rich fibrin matrices. Am. J. sports Med. 35, 245–251 (2007).
Sánchez, A. R., Sheridan, P. J. & Kupp, L. I. Is platelet-rich plasma the perfect enhancement factor? A current review. Int. J. Oral Maxillofac. Implants 18, 93−103 (2003).
Oryan, A., Alidadi, S. & Moshiri, A. Platelet-rich plasma for bone healing and regeneration. Expert Opin. Biol. Ther. 16, 213–232 (2016).
Sys, J., Weyler, J., Van Der Zijden, T., Parizel, P. & Michielsen, J. Platelet-rich plasma in mono-segmental posterior lumbar interbody fusion. Eur. Spine J. 20, 1650–1657 (2011).
Peerbooms, J. C. et al. No positive bone healing after using platelet rich plasma in a skeletal defect. An observational prospective cohort study. Int. Orthop. 36, 2113–2119 (2012).
Abagnale, G. et al. Surface topography enhances differentiation of mesenchymal stem cells towards osteogenic and adipogenic lineages. Biomaterials 61, 316–326 (2015).
Sadowska, J. M. et al. Development of miR-26a-activated scaffold to promote healing of critical-sized bone defects through angiogenic and osteogenic mechanisms. Biomaterials 303, 122398 (2023).
Liu, Y. et al. Sustained delivery of a heterodimer bone morphogenetic protein-2/7 via a collagen hydroxyapatite scaffold accelerates and improves critical femoral defect healing. Acta Biomater. 162, 164–181 (2023).
Kim, S. et al. Sequential delivery of BMP-2 and IGF-1 using a chitosan gel with gelatin microspheres enhances early osteoblastic differentiation. Acta Biomater. 8, 1768–1777 (2012).
Xin, T. et al. Programmed sustained release of recombinant human bone morphogenetic protein-2 and inorganic ion composite hydrogel as artificial periosteum. ACS Appl. Mater. interfaces 12, 6840–6851 (2020).
Zhao, C. et al. The role of the micro-pattern and nano-topography of hydroxyapatite bioceramics on stimulating osteogenic differentiation of mesenchymal stem cells. Acta Biomater. 73, 509–521 (2018).
Kang, Y., Kim, S., Khademhosseini, A. & Yang, Y. Creation of bony microenvironment with CaP and cell-derived ECM to enhance human bone-marrow MSC behavior and delivery of BMP-2. Biomaterials 32, 6119–6130 (2011).
Chandrakar, A. et al. Melt electrowriting of hydrophilic/hydrophobic multiblock copolymers for bone tissue regeneration. Biomater. Adv. 169, 214167 (2025).
Sanchez, A. A. et al. Enhanced osteogenic differentiation in hyaluronic acid methacrylate (HAMA) matrix: a comparative study of hPDC and hBMSC spheroids for bone tissue engineering. Biofabrication 17, 025013 (2025).
van der Heide, D. et al. implementing bmp-7 chemically modified RNA for bone regeneration with 3D printable hyaluronic acid-collagen granular gels. Adv. Healthcare Mater. 14, 2405047 (2025).
Alejandro, P. & Constantinescu, F. A review of osteoporosis in the older adult. Clin. Geriatr. Med. 33, 27–40 (2017).
Center, J. R. Outcomes following osteoporotic fractures. In Osteoporosis (fourth edition) 841–852 (2013).
Langdahl, B. L. Overview of treatment approaches to osteoporosis. Br. J. Pharm. 178, 1891–1906 (2020).
Office of the Surgeon General (US). Bone Health and Osteoporosis: A Report of the Surgeon General. The Burden of Bone Disease. (Rockville (MD), 2004).
Cummings, S. R. et al. Denosumab for prevention of fractures in postmenopausal women with osteoporosis. N. Engl. J. Med. 361, 756–765 (2009).
Lyu, Q. Therapeutic potential of nucleic acid aptamers against sclerostin in the treatment of osteoporosis, Hong Kong Baptist University, (2017).
Laine, C. M. et al. WNT1 mutations in early-onset osteoporosis and osteogenesis imperfecta. N. Engl. J. Med. 368, 1809–1816 (2013).
Houschyar, K. S. et al. Wnt pathway in bone repair and regeneration–what do we know so far. Front. Cell Dev. Biol. 6, 170 (2019).
Egermann, M. et al. Direct adenoviral transfer of bone morphogenetic protein-2 cDNA enhances fracture healing in osteoporotic sheep. Hum. Gene Ther. 17, 507–517 (2006).
Zeng, Y. et al. Gene therapy with Tetracycline-regulated human recombinant COLIA1 cDNA direct adenoviral delivery enhances fracture healing in osteoporotic rats. Hum. Gene Ther. 29, 902–915 (2018).
Tang, Y. et al. Combination of bone tissue engineering and BMP-2 gene transfection promotes bone healing in osteoporotic rats. Cell Biol. Int. 32, 1150–1157 (2008).
Liu, J. et al. Delivery of m7G methylated Runx2 mRNA by bone-targeted lipid nanoparticle promotes osteoblastic bone formation in senile osteoporosis. Nano Today 54, 102074 (2024).
Michigami, T. Rickets/Osteomalacia. Consensus on vitamin D deficiency and insufficiency in children. Clin. Calcium 28, 1307–1311 (2018).
Sahay, M. & Sahay, R. Rickets–vitamin D deficiency and dependency. Indian J. Endocrinol. Metab. 16, 164 (2012).
Francis, R. M. & Selby, P. L. Osteomalacia. Bailliere’s. Clin. Endocrinol. Metab. 11, 145–163 (1997).
Land, C. & Schoenau, E. Fetal and postnatal bone development: reviewing the role of mechanical stimuli and nutrition. Best. Pract. Res. Clin. Endocrinol. Metab. 22, 107–118 (2008).
Acar, S., Demir, K. & Shi, Y. Genetic causes of rickets. J. Clin. Res. Pediatr. Endocrinol. 9, 88 (2017).
Beck-Nielsen, S. S. et al. FGF23 and its role in X-linked hypophosphatemia-related morbidity. Orphanet J. Rare Dis. 14, 1–25 (2019).
Bai, X., Levental, M. & Karaplis, A. C. Burosumab treatment for autosomal recessive hypophosphatemic rickets type 1 (ARHR1). J. Clin. Endocrinol. Metab. 107, 2777–2783 (2022).
Mornet, E. Hypophosphatasia. Metabolism 82, 142–155 (2018).
Kang, M., Wu, M. & Crane, J. L. Asfotase alfa improved skeletal mineralization and fracture healing in a child with MCAHS. Bone 172, 116778 (2023).
Dardenne, O., Prud’homme, J. E., Arabian, A., Glorieux, F. H. & St-Arnaud, R. Targeted inactivation of the 25-hydroxyvitamin D3-1α-hydroxylase gene (CYP27B1) creates an animal model of pseudovitamin D-deficiency rickets. Endocrinology 142, 3135–3141 (2001).
Fukumoto, S. FGF23-related hypophosphatemic rickets/osteomalacia: diagnosis and new treatment. J. Mol. Endocrinol. 66, R57–R65 (2021).
Siris, E. S. Paget’s disease of bone. J. Bone Miner. Res. 13, 1061–1065 (1998).
Friedrichs, W. E. et al. Sequence analysis of measles virus nucleocapsid transcripts in patients with Paget’s disease. J. Bone Miner. Res. 17, 145–151 (2002).
Singer, F. R. Paget’s disease of bone—genetic and environmental factors. Nat. Rev. Endocrinol. 11, 662 (2015).
Singer, F., Schiller, A. & Pyle, E. Paget’s disease of bone. Metabolic Bone Disease 2, 489−575 (1978).
Naot, D. et al. Differential gene expression in cultured osteoblasts and bone marrow stromal cells from patients with Paget’s disease of bone. J. Bone Miner. Res. 22, 298–309 (2007).
Xia, Z., Chen, C., Chen, P., Xie, H. & Luo, X. MicroRNAs and their roles in osteoclast differentiation. Front. Med. 5, 414–419 (2011).
Forlino, A. & Marini, J. C. Osteogenesis imperfecta. Lancet 387, 1657–1671 (2016).
Ralston, S. H. & Gaston, M. S. Management of Osteogenesis Imperfecta. Front Endocrinol 10, 924 (2019).
Agarwal, V. & Joseph, B. Non-union in osteogenesis imperfecta. J. Pediatr. Orthop. B 14, 451–455 (2005).
Roschger, P. et al. Evidence that abnormal high bone mineralization in growing children with osteogenesis imperfecta is not associated with specific collagen mutations. Calcif. Tissue Int. 82, 263–270 (2008).
Niyibizi, C., Wang, S., Mi, Z. & Robbins, P. Gene therapy approaches for osteogenesis imperfecta. Gene Ther. 11, 408–416 (2004).
Horwitz, E. M. et al. Isolated allogeneic bone marrow-derived mesenchymal cells engraft and stimulate growth in children with osteogenesis imperfecta: implications for cell therapy of bone. Proc. Natl. Acad. Sci. 99, 8932–8937 (2002).
Cardinal, M. et al. Sclerostin antibody reduces long bone fractures in the oim/oim model of osteogenesis imperfecta. Bone 124, 137–147 (2019).
Glorieux, F. H. et al. BPS804 anti-sclerostin antibody in adults with moderate osteogenesis imperfecta: results of a randomized phase 2a trial. J. Bone Miner. Res. 32, 1496–1504 (2017).
Melton, L. J. III, Kyle, R. A., Achenbach, S. J., Oberg, A. L. & Rajkumar, S. V. Fracture risk with multiple myeloma: a population-based study. J. Bone Miner. Res. 20, 487–493 (2005).
Bryson, D. J., Wicks, L. & Ashford, R. U. The investigation and management of suspected malignant pathological fractures: a review for the general orthopaedic surgeon. Injury 46, 1891–1899 (2015).
Tsuzuki, S., Park, S. H., Eber, M. R., Peters, C. M. & Shiozawa, Y. Skeletal complications in cancer patients with bone metastases. Int. J. Urol. 23, 825–832 (2016).
Castañeda, S. et al. Bone loss induced by cancer treatments in breast and prostate cancer patients. Clin. Transl. Oncol. 24, 2090–2106 (2022).
Hamouda, A. E. et al. Intratumoral delivery of lipid nanoparticle-formulated mRNA encoding IL-21, IL-7, and 4-1BBL induces systemic anti-tumor immunity. Nat. Commun. 15, 1–20 (2024).
Wang, L. et al. A clinical study on bone defect reconstruction and functional recovery in benign bone tumors of the lower extremity, treated by bone marrow mesenchymal stem cell rapid screening–enrichment–composite system. World J. Surg. Oncol. 19, 1–7 (2021).
Bostrom, M. P. et al. Immunolocalization and expression of bone morphogenetic proteins 2 and 4 in fracture healing. J. Orthop. Res. 13, 357–367 (1995).
Gillman, C. E. & Jayasuriya, A. C. FDA-approved bone grafts and bone graft substitute devices in bone regeneration. Mater. Sci. Eng. C. 130, 112466 (2021).
Eppley, B. L., Doucet, M., Connolly, D. T. & Feder, J. Enhancement of angiogenesis by bFGF in mandibular bone graft healing in the rabbit. J. oral. Maxillofac. Surg. 46, 391–398 (1988).
Kawaguchi, H. et al. A local application of recombinant human fibroblast growth factor 2 for tibial shaft fractures: A randomized, placebo-controlled trial. J. Bone Miner. Res. 25, 2735–2743 (2010).
Daniels, T. R. et al. Prospective randomized controlled trial of hindfoot and ankle fusions treated with rhPDGF-BB in combination with a β-TCP-collagen matrix. Foot Ankle Int. 36, 739–748 (2015).
Joyce, M. E., Roberts, A. B., Sporn, M. B. & Bolander, M. E. Transforming growth factor-beta and the initiation of chondrogenesis and osteogenesis in the rat femur. J. Cell Biol. 110, 2195–2207 (1990).
Xu, J. et al. High-dose TGF-β1 impairs mesenchymal stem cell–mediated bone regeneration via Bmp2 inhibition. J. Bone Miner. Res. 35, 167–180 (2020).
Boonen, S. et al. Musculoskeletal effects of the recombinant human IGF-I/IGF binding protein-3 complex in osteoporotic patients with proximal femoral fracture: a double-blind, placebo-controlled pilot study. J. Clin. Endocrinol. Metab. 87, 1593–1599 (2002).
Street, J. et al. Vascular endothelial growth factor stimulates bone repair by promoting angiogenesis and bone turnover. Proc. Natl. Acad. Sci. 99, 9656–9661 (2002).
Peng, H. et al. Synergistic enhancement of bone formation and healing by stem cell–expressed VEGF and bone morphogenetic protein-4. J. Clin. Invest. 110, 751–759 (2002).
Hu, K. & Olsen, B. R. Osteoblast-derived VEGF regulates osteoblast differentiation and bone formation during bone repair. J. Clin. Invest. 126, 509–526 (2016).
Yang, P. et al. rhVEGF165 delivered in a porous β-tricalcium phosphate scaffold accelerates bridging of critical-sized defects in rabbit radii. J. Biomed. Mater. Res. A 92, 626–640 (2010).
Zhang, L. et al. Improvement in angiogenesis and osteogenesis with modified cannulated screws combined with VEGF/PLGA/fibrin glue in femoral neck fractures. J. Mater. Sci.: Mater. Med. 25, 1165–1172 (2014).
Quinlan, E. et al. Controlled release of vascular endothelial growth factor from spray-dried alginate microparticles in collagen–hydroxyapatite scaffolds for promoting vascularization and bone repair. J. tissue Eng. Regen. Med. 11, 1097–1109 (2017).
Rivera, K. O. et al. Local injections of β-NGF accelerates endochondral fracture repair by promoting cartilage to bone conversion. Sci. Rep. 10, 22241 (2020).
Wang, Z. et al. A regulatory loop containing miR-26a, GSK3β and C/EBPα regulates the osteogenesis of human adipose-derived mesenchymal stem cells. Sci. Rep. 5, 15280 (2015).
Yang, C. et al. miRNA-21 promotes osteogenesis via the PTEN/PI3K/Akt/HIF-1α pathway and enhances bone regeneration in critical size defects. Stem Cell Res. Ther. 10, 1–11 (2019).
Krist, B., Florczyk, U., Pietraszek-Gremplewicz, K., Józkowicz, A. & Dulak, J. The role of miR-378a in metabolism, angiogenesis, and muscle biology. Int. J. Endocrinol. 2015, 281756 (2015).
You, L. et al. MiR-378 overexpression attenuates high glucose-suppressed osteogenic differentiation through targeting CASP3 and activating PI3K/Akt signaling pathway. Int. J. Clin. Exp. Pathol. 7, 7249 (2014).
Zhang, B. et al. MicroRNA-378 promotes osteogenesis-angiogenesis coupling in BMMSCs for potential bone regeneration. Anal. Cell. Pathol. 2018, 8402390 (2018).
Tian, Z., Zhou, H., Xu, Y. & Bai, J. MicroRNA-495 inhibits new bone regeneration via targeting high mobility group AT-Hook 2 (HMGA2). Med. Sci. Monit. Int. Med. J. Exp. Clin. Res. 23, 4689 (2017).
Minamizaki, T. et al. The matrix vesicle cargo miR-125b accumulates in the bone matrix, inhibiting bone resorption in mice. Commun. Biol. 3, 30 (2020).
Joris, V., Balmayor, E. R. & van Griensven, M. miR-125b differentially impacts mineralization in dexamethasone and calcium-treated human mesenchymal stem cells. Mol. Ther. Nucleic Acids 36, 102446 (2025).
Ito, S. et al. Overexpression of miR-125b in osteoblasts improves age-related changes in bone mass and quality through suppression of osteoclast formation. Int. J. Mol. Sci. 22, 6745 (2021).
Zhang, W. et al. miRNA-133a-5p inhibits the expression of osteoblast differentiation-associated markers by targeting the 3′ UTR of RUNX2. DNA Cell Biol. 37, 199–209 (2018).
Miscianinov, V. et al. MicroRNA-148b targets the TGF-β pathway to regulate angiogenesis and endothelial-to-mesenchymal transition during skin wound healing. Mol. Ther. 26, 1996–2007 (2018).
Moncal, K. K. et al. Collagen-infilled 3D printed scaffolds loaded with miR-148b-transfected bone marrow stem cells improve calvarial bone regeneration in rats. Mater. Sci. Eng. C. 105, 110128 (2019).
Mollazadeh, S. et al. Overexpression of MicroRNA-148b-3p stimulates osteogenesis of human bone marrow-derived mesenchymal stem cells: the role of MicroRNA-148b-3p in osteogenesis. BMC Med. Genet. 20, 1–10 (2019).
Acknowledgements
This work has been performed as part of the cmRNAbone project funded by the European Union’s Horizon 2020 research and innovation program under the Grant Agreement No 874790.
Author information
Authors and Affiliations
Contributions
Cd.T.R.: literature search, writing first draft, preparing figures, approval of final version. E.R.B.: conceptualization, analyzing chosen literature, revising draft and input for revising figures, approval of final version. Mv.G.: conceptualization, supervision, analyzing chosen literature, revising draft and input for revising figures, approval of final version.
Corresponding author
Ethics declarations
Competing interests
CdTR: Salary paid from the cmRNAbone project granted by the European Union’s Horizon 2020 research and innovation program under the Grant Agreement No 874790. ERB: Is inventor on a patent regarding therapeutic RNA for osteogenesis: Induction of osteogenesis by delivering BMP encoding RNA, BALMAYOR, Elizabeth, RUDOLPH, Carsten, PLANK, Christian. Publication Date: 19.05.2016. Publication No.: WO/2016/075154. ERB’s work on osteogenesis and RNA therapeutics has been partially funded by the NIH (R01 AR074395 from NIAMS). Member of the executive council of TERMIS-EU. MvG: MvG’s work on osteogenesis has been partially funded by the cmRNAbone project granted by the European Union’s Horizon 2020 research and innovation program under the Grant Agreement No 874790. President of the Section Research of the German Society for Orthopedics and Trauma Surgery (unpaid). Member of the scientific advisory board for CIBER-BBN in Spain (unpaid). Has received compensation as a member of the scientific advisory board for Centre for Doctoral Training in Advanced Biomedical Materials (Manchester/Sheffield, UK).
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Del Toro Runzer, C., Balmayor, E.R. & van Griensven, M. Biologics for bone regeneration: advances in cell, protein, gene, and mRNA therapies. Bone Res 14, 5 (2026). https://doi.org/10.1038/s41413-025-00487-0
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41413-025-00487-0