Abstract
By merging electricity with sulfate, the Ritter-type amination of C(sp3)-H bonds is developed in an undivided cell under room temperature. This method features broad substrate generality (71 examples, up to 93% yields), high functional-group compatibility, facile scalability, excellent site-selectivity and mild conditions. Common alkanes and electron-deficient alkylbenzenes are viable substrates. It also provides a straightforward protocol for incorporating C-deuterated acetylamino group into C(sp3)-H sites. Application in the synthesis or modification of pharmaceuticals or their derivatives and gram-scale synthesis demonstrate the practicability of this method. Mechanistic experiments show that sulfate radical anion, formed by electrolysis of sulfate, served as hydrogen atom transfer agent to provide alkyl radical intermediate. This method paves a convenient and flexible pathway for realizing various synthetically useful transformations of C(sp3)-H bonds mediated by sulfate radical anion generated via electrochemistry.
Similar content being viewed by others
Introduction
Given the high prevalence of amines in natural products, agrochemicals, pharmaceuticals and organic materials1, tremendous progress has been achieved for the construction of C-N bonds, including reductive amination2, hydroamination3, and transition metal catalyzed cross coupling (e.g., Ullmann-Goldberg condensation, Chan–Lam coupling, Buchwald–Hartwig amination, etc)4,5,6. Among the various protocols reported, one of the most desirable strategies is the direct C-H amination in terms of high step- and atom-economy. Comparing with amination of C(sp2)-H bonds, direct incorporation of N-containing moieties into C(sp3)-H bonds is highly desirable but also challenging due to low reactivity and poor selectivity. To circumvent these limitations, many notable strategies were developed, including transition metal catalyzed C(sp3)-H activation7, C-H insertion catalysis8, and hydrogen atom transfer (HAT) process9. The functionalization of distinct C-H bonds via HAT process often proceeds in highly selective mode, thus avoiding the pre-incorporation of directing moiety into the substrate. Meanwhile, the selectivity and reactivity of HAT process can be rationally tuned by varying hydrogen acceptor and reaction additive. For instance, highly selective amination of C(sp3)-H bonds has been achieved by employing N-fluorobenzenesulfonimide10,11, N-hydroxyimide derivatives12,13,14 and di-tert-butyl peroxide15,16,17 as HAT agents. Besides, 1,5-HAT enabled by nitrogen-centered radicals has also served as a robust and practical strategy in selective amination of remote unactivated C(sp3)-H bonds, especially the classic Hofmann–Löffler–Freytag reaction18,19,20.
In recent years, the flourishing development of visible light induced photoredox catalysis enables a myriad of elegant C(sp3)-H functionalizations via HAT process under mild conditions. In particular, the decatungstate anion (W; [W10O32]4−)21,22, in excited state, nitrogen radical23,24,25, oxygen radical26,27,28,29, and bromine radical30 generated via photoredox have been employed as the efficient HAT agents for the selective amination of C(sp3)-H bonds. In addition, electrochemistry, which utilizes electron as oxidant or reductant, has accelerated clean, efficient and scalable transformation of C-H bonds into C-N bonds31,32,33,34,35,36. Generally, these processes based on the direct or indirect anodic oxidation of substrates were highly limited to the relatively active C(sp3)-H bonds, including C(sp3)-H bonds adjacent to heteroatoms37,38,39,40, carbonyl41, allylic42, and benzylic groups43,44,45,46,47,48,49,50.
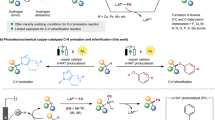
However, C-H amination through HAT process enabled by electrochemistry and electrophotocatalysis51,52,53,54 could enlarge the substrate scope to unactivated alkanes. For instance, the remote inert C(sp3)-H bonds amination was developed to construct pyrrolidines through 1,5-HAT process, which was initiated by the electrochemical generated N radical55,56,57. Using DDQ and Mn(IV) diazide intermediate as HAT agents, Lei group58 and Ackermann group59 independently reported manganese-catalyzed photoelectrochemical and electrochemical oxidative azidation of C(sp3)-H bonds, respectively (Fig. 1a).
a Examples of the azide reaction of unactivated alkanes via (photo)electrocatalysis. b Examples of Ritter-type C-H amination via Selectfluor, hypervalent iodine reagent and TAC electrophotocatalyst. c Examples of intermolecular amidation of unactivated alkanes via tBuOOtBu and Cu cat. d This work: Ritter-Type amination of C(sp3)-H bonds enabled by the electrochemistry with SO42−.
Several Ritter-type reactions have been disclosed by Baran60, Liu & Chen61, providing elegant methods for the amination of inert C(sp3)-H bonds (Fig. 1b). In these works, stoichiometric amounts of expensive oxidants (Selectfluor and hypervalent iodine) were employed to promote the conversion of C(sp3)-H to carbon cation. Lambert and coworkers developed the Ritter type C(sp3)-H amination of benzylic sites47 and the diamination of vicinal C–H bonds via Ritter-type step62 by virtue of trisaminocyclopropenium ion (TAC) as the notable electrophotocatalyst (Fig. 1b). König group29 and Hartwig group17 reported the reactions of alkanes with simple amides to form the corresponding N-alkyl products using tBuOOtBu as HAT agent (Fig. 1c). In order to enrich the application of Ritter-type C(sp3)-H amination reaction, we conceived to develop an economic manifold that avoids the use of expensive or unstable oxidants, through electrochemical approach.
Generally, thermal, photolysis, electrolysis and metal activated decomposition of peroxodisulfate, which is prepared by the electrolysis of sulfate in industrial process63,64, can provide a powerful HAT species, sulfate radical anion, thus initiating the C-H bonds functionalization65,66,67,68,69. As a consequence, we envisaged that S2O82- could be prepared by the anodic oxidation of H2SO4 and then would undergo cathodic reduction to yield sulfate radical anion, similar to the protocol reported by Xu group, in which chloride radical was generated by irradiating the anodically formed Cl2 from Cl- and served as robust HAT agent to form alkyl radical under photoelectrochemical systems70.
Here, we report a broadly applicable electrochemical strategy for Ritter type C(sp3)-H amination where aliphatic carbon cation is generated via sulfate radical anion mediated HAT followed by further oxidation under electrolysis (Fig. 1d).
Results and discussion
Reaction optimization
Since the product from direct C-H amination of 1,3-dimethyladamantane is used as prodrug for the treatment of Alzheimer’s disease, 1,3-dimethyladamantane was chosen as the model reactant to investigate the reaction conditions (Table 1). All the electrolysis reactions were conducted in undivided cells. Based on the conditions screening, the optimal conditions comprised Pt plates as anode and cathode, H2SO4 as pre-oxidant, nBu4NBF4 as electrolyte, CH3CN as solvent and nitrogen source. Under the standard conditions, 93% yield was obtained under 5 mA constant current for 20 h. The reaction did not proceed at room temperature in the absence of electricity (Table 1, entry 2) until the temperature was increased to 65 °C with a low yield of 14% (Table 1, entry 3). Among all the tested acids, H2SO4 gave the optimal yield. Decreasing the amount of H2SO4 was detrimental to the reaction yield (Table 1, entries 5–7). Performing the electrolysis without H2SO4 only gave trace amount of product, demonstrating the pivotal role of H2SO4 in the C(sp3)-H amination. Notably, the yield was also obviously diminished when the Pt anode was replaced by carbon materials (Table 1, entries 8–10). Either lower or higher current (e.g., 3.0 mA, 7 mA, 10 mA) was not conducive to this reaction (Table 1, entries 11–13). Replacing H2SO4 with equivalent amounts of K2S2O8 could also provide product 1 with 84% yield (Table 1, entry 14). Therefore, peroxodisulfate probably formed by electrolyzing sulfate assisted the rupture of C-H bonds. Besides, the yield could also reach 76% by using Na2SO4 as sulfate source (Table 1, entry 15). The methanesulfonic acid also showed good performance (Table 1, entry 16) due to the generation of permethanesulfonic acid by the anodic oxidation of methanesulfonic acid, which is similar to peroxydisulfate71. Other more detailed conditions were presented in the Supplementary Information (Supplementary Table 1).
Evaluation of substrate scope
With the optimized conditions in hand, we next evaluated the substrates scope of alkanes in this protocol (Fig. 2). First, tertiary alkanes were well tolerated and afforded amination products in 44–93% yields with excellent regioselectivity (Fig. 2, 1–8). Of note, the reaction with deuterated acetonitrile as nitrogen source gave C-deuterated acetamination products 2 and 6 with 85 and 78% yields, respectively. Generally, the amination of secondary C(sp3)-H bond is more challenging than that of tertiary C(sp3)-H bond, so 16.0 equivalents of H2SO4 is required. A variety of cyclic, secondary hydrocarbons were smoothly transformed into aliphatic amide products (Fig. 2, 9–12) in moderate to good yields, while the reaction with cyclododecane furnished the product 13 in 32% yield due to the poor solubility in acetonitrile. However, when using different types of linear alkanes, two isomers were obtained with different ratios. The electrolysis of n-hexane provided 1:1 ratio of isomers with 54% yield. For linear alkanes containing electron-deficient moieties, such as ester, bromine, and chlorine, the aminations were apt to take place at the secondary C-H bonds far away from these functional groups, leading to the mixture of acetamination products 17–19 with different ratios about 2.3:1, 1.5:1, and 1.3:1, respectively. To our delight, the amination of 1-bromopentane and 1-chloropentane occurred preferentially at C-H bond away from halogens with high regioselectivity and provided single products in good yields (Fig. 2, 15, 16).
aIsolated yield; bElectrolysis performed on 10 mmol scale; cRegio isomeric ratios were determined by 1H NMR.
Next, the scope of alkylbenzenes was assessed (Fig. 3). The amination reaction of alkylbenzenes bearing different functional groups at aliphatic chain, such as carbonyl, alkenyl and cyano group, underwent smoothly to deliver 20–30 in 41–76% yields, with less amount of H2SO4 and shorter reaction times. Product 24 was afforded with excellent regioselectivity, illustrating that C(sp3)-H amination preferentially reacted at benzylic over tertiary C-H bond. Besides, the phenylpropene underwent isomerization to form 28 in 41% yield. Furthermore, derivatives of toluene and ethylbenzene containing various functional groups on the benzene ring, including halogen, cyano, ester, carbonyl, nitro, amino, methoxyl, and alkyl groups, were well accommodated (Fig. 3, 31–41). Mono-acetamination product were provided for the alkylbenzenes with multiple benzylic sites. Commonly, nitro group and iodine atom are prone to be reduced under electrolysis72,73,74,75, but remained intact under our reaction conditions. In particular, this reaction system was tolerated to unprotected amine groups, furnished products 39 and 41 in 25 and 54% yields, respectively, likely as a result of the acidification of amines. It is worth noting that previous electrochemical methods did not work for the amination of alkylbenzenes bearing strong electron-withdrawing groups43,44,45,46,48,49,50, which are viable reactants under our reaction systems. In addition, the α-amino acid precursors and β-amino acid precursors (Fig. 3, 58–62) were also obtained in good yields.
aIsolated yield; bElectrolysis performed on 10 mmol scale.
With the replacement of acetonitrile by ethyl cyanoacetate, butyronitrile, isobutyronitrile and adiponitrile, the reaction could also take place to yield products 63–68 in moderate to good yields. (Fig. 4a) To further explore the synthetic utility of the methodology, the modification of natural products and pharmaceuticals (Fig. 4b) and scaled-up experiments (see the Supplementary Methods for details) were carried out. The amination of esterified ibuprofen was site-selectively occured at the less hindered benzyl position in 77% yield. The natural deoxyfenisin and fragrance celestolide were funtionalized to give product 70 and 71, respectively. Next, we carried out gram-scale amplification experiments, in which aminated products (1, 12, 21) were obtained with satisfying yields.
a Products of other nitriles. b Products of natural products & drugs. Conditions: substrate (0.5 mmol), H2SO4 (54 μL, 1 mmol, 0.33 M), nBu4NBF4 (0.1 M), CH3CN (3 mL), 5 mA; aConditions: substrate (0.5 mmol), H2SO4 (108 μL, 2 mmol, 0.67 M), nBu4NBF4 (0.1 M), CH3CN (3 mL), 5 mA.
Compounds 1b and 3b are the precursors of the drug memantine hydrochloride for the treatment of Alzheimer’s disease and the antiviral drug amantadine hydrochloride, respectively. Therefore, further hydrolysis of compounds 1 and 3 were shown in Fig. 5. Our methodology could be applied for preparing the precursors of memantine hydrochloride 1b and amantadine hydrochloride 3b in two steps from simple materials, avoiding the use of bromine and the large amount of H2SO4 while maintaining high yields (79 and 57% total yields). The hydrolysis of 25 also yielded 25b, a key intermediate for the synthsis of rasagiline(see the Supplementary Methods for details).
a Conditions: Amide compound (3.0 mmol, 1 eq.), NaOH (24 mmol, 8 eq.), 1-butanol as solvent, 130–135 °C for 7 h. Then, the free base was added 5 M aq. HCl (3 mL) to give hydrochloride. b Conditions: 25 (3.0 mmol, 1 eq.), HCl (3 M, 10 eq.), reflux for 3 h.
Mechanistic studies
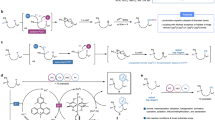
To obtain more insights into the Ritter-type amination reaction mechanism, a series of mechanistic studies were conducted. First, replacing sulfuric acid with sodium sulfate, potassium peroxodisulfate and methanesulfonic acid, which are unlikely to facilitate the Ritter-type step, 76–89% yields of desired product were provided (Fig. 6a). Besides, the formation of persulfate via the electrolysis of sulfuric acid under standard conditions was also proved by UV absorption spectra experiment (Supplementary Fig. 6). So, sulfuric acid was proved to promote the activation of C(sp3)-H rather than as an additive to facilitate the Ritter-type step. 65% yield of product 1 was delivered by electrolyzing 1a at anodic chamber under standard conditions, demonstrating the generation of sulfate radical cation through the direct anodic oxidation (Fig. 6a). By comparing the oxidation potentials of sulfate (Eo = 2.6 V vs NHE) and some alkanes in literatures76,77,78,79,80, sulfates are likely to be oxidized by anode in prior to alkanes. However, heating or irradiating K2S2O8 with 1a did not afford the product 1 (Fig. 6a). We envisaged that the conversion of carbon radical to carbon cation could be accomplished by anode oxidation rather than the single electron transfer of SO4-.. Radical clock experiment confirmed that radical intermediate 72b was generated and underwent ring opening and further oxidation, furnishing carbon cation intermediate (Fig. 6b).
a Control experiments. aFor the reaction, to the anodic chamber was added 1a (0.5 mmol), H2SO4 (108 μL, 2 mmol, 0.67 M), nBu4NBF4 (0.1 M) and CH3CN (3 mL), the cathodic chamber was added nBu4NBF4 (0.1 M) and CH3CN (3 mL). b Radical trapping experiments. c Proposed mechanism.
Based on the studies mentioned above, a possible mechanism for this reaction process was shown in Fig. 6c. Intially, sulfate anion SO42- underwent a single electron oxidation process at the anode to afford SO4-.. As a powerful HAT agent, SO4-. abstracted hydrogen atom from the substrate 1a to generate carbon radical 73, which was oxidized at the anode to the carbocation intermediate 74. Subsequently, nucleophilic attack of acetonitrile to 74 through classic Ritter reaction process provided final product 1. At the same time, the dimerization of SO4-. generated S2O82-, which was then transferred to the cathode area and reduced back to SO4-..
In summary, we have developed an electrochemical approach that can be applied for the Ritter-type amination reaction of aliphatic C(sp3)-H and benzylic C(sp3)-H under mild conditions. By electrolyzing sulfate, SO4-. was generated and abstracted hydrogen atom from C(sp3)-H bonds. This electrochemical method also exhibited excellent regioselectivity and functional groups tolerance, rendering the reaction simple, safe, widely applicable. It is anticipated that this method would provide a convenient and alternative entry for the synthesis of amines derivatives.
Methods
Representative procedure for amination
An oven-dried undivided cell was equipped with a stir bar, 1,3-dimethyladamantane (82 mg, 0.5 mmol, 1 eq.), 98% H2SO4 (108 μL, 2 mmol, 0.67 M, 4 eq.), nBu4NBF4 (98 mg, 0.3 mmol, 0.1 M), CH3CN (3 mL). Air has little effect on this reaction. The assembled electrodes were placed into the solution. The silica gel plug was sealed with film. The mixture was electrolyzed at a constant current of 5 mA until the tertiary alkane was completely consumed (See Supplementary Fig. 3). The Pt electrodes were washed by water, ethanol, and DCM in turn. After the reaction is over, drop NaHCO3 saturated solution into the reaction system slowly until no bubbles were observed. The aqueous layer was separated and extracted with EtOAc (3 × 10 mL), and the combined organic layers were washed with brine and dried over anhydrous Na2SO4. Following concentration in vacuo, the crude product was purified by column chromatography on silica gel to give pure product 1 (103 mg, 93%; see the Supplementary Methods for details).
Data availability
The authors declare that the data supporting the findings of this study are available within the article and its Supplementary Information file. For experimental details and compound characterization data see Supplementary Methods. For 1H NMR, 13C NMR, and 19F NMR spectra see Supplementary Figs. 7–82.
References
Ricci, A. Amino group chemistry, from synthesis to the life sciences. (Wiley-VCH: Weinheim. 2007).
Afanasyev, O. I., Kuchuk, E., Usanov, D. L. & Chusov, D. Reductive amination in the synthesis of pharmaceuticals. Chem. Rev. 119, 11857–11911 (2019).
Huang, L., Arndt, M., Gooßen, K., Heydt, H. & Gooßen, L. J. Late transition metal-catalyzed hydroamination and hydroamidation. Chem. Rev. 115, 2596–2697 (2015).
Khan, F., Dlugosch, M., Liu, X. & Banwell, M. G. The palladium-catalyzed Ullmann cross-coupling reaction: a modern variant on a time-honored process. Acc. Chem. Res. 51, 1784–1795 (2018).
Chen, J.-Q., Li, J.-H. & Dong, Z.-B. A review on the latest progress of Chan-Lam coupling reaction. Adv. Synth. Catal. 362, 3311–3331 (2020).
Forero-Cortés, P. A. & Haydl, A. M. The 25th anniversary of the Buchwald-Hartwig amination: development, applications, and outlook. Org. Process Res. Dev. 23, 1478–1483 (2019).
Timsina, Y. N., Gupton, B. F. & Ellis, K. C. Palladium-catalyzed C-H amination of C(sp2) and C(sp3)-H bonds: mechanism and scope for N-based molecule synthesis. ACS Catal. 8, 5732–5776 (2018).
Shin, K., Kim, H. & Chang, S. Transition-metal-catalyzed C-N bond forming reactions using organic azides as the nitrogen source: a journey for the mild and versatile C-H amination. Acc. Chem. Res. 48, 1040–1052 (2015).
Capaldo, L., Ravelli, D. & Fagnoni, M. Direct photocatalyzed hydrogen atom transfer (HAT) for aliphatic C-H bonds elaboration. Chem. Rev. 122, 1875–1924 (2022).
Suh, S.-E. et al. Site-selective copper-catalyzed azidation of benzylic C-H bonds. J. Am. Chem. Soc. 142, 11388–11393 (2020).
Muñoz-Molina, J. M., Belderrain, T. R. & Pérez, P. J. Recent advances in copper-catalyzed radical C-H bond activation using N-F reagents. Synthesis 53, 51–64 (2021).
Kato, T. & Maruoka, K. Design of bowl-shaped N-hydroxyimide derivatives as new organoradical catalysts for site-selective C(sp3)-H bond functionalization reactions. Angew. Chem. Int. Ed. 59, 14261–14264 (2020).
Shibuya, M., Orihashi, T., Li, Y. & Yamamoto, Y. N-hydroxyphthalimide-catalyzed chemoselective intermolecular benzylic C-H amination of unprotected arylalkanols. Chem. Commun. 57, 8742–8745 (2021).
Kiyokawa, K., Takemoto, K. & Minakata, S. Ritter-type amination of C-H bonds at tertiary carbon centers using iodic acid as an oxidant. Chem. Commun. 52, 13082–13085 (2016).
Chikkade, P. K., Kuninobu, Y. & Kanai, M. Copper-catalyzed intermolecular C(sp3)-H bond functionalization towards the synthesis of tertiary carbamates. Chem. Sci. 6, 3195–3200 (2015).
Teng, F., Sun, S., Jiang, Y., Yu, J.-T. & Cheng, J. Copper-catalyzed oxidative C(sp3)-H/N-H coupling of sulfoximines and amides with simple alkanes via a radical process. Chem. Commun. 51, 5902–5905 (2015).
Tran, B. L., Li, B., Driess, M. & Hartwig, J. F. Copper-catalyzed intermolecular amidation and imidation of unactivated alkanes. J. Am. Chem. Soc. 136, 2555–2563 (2014).
Wappes, E. A., Nakafuku, K. M. & Nagib, D. A. Directed β C-H amination of alcohols via radical relay chaperones. J. Am. Chem. Soc. 139, 10204–10207 (2017).
Bao, X., Wang, Q. & Zhu, J. Copper-catalyzed remote C(sp3)-H azidation and oxidative trifluoromethylation of benzohydrazides. Nat. Commun. 10, 769–775 (2019).
Nakafuku, K. M. et al. Enantioselective radical C-H amination for the synthesis of β-amino alcohols. Nat. Chem. 12, 697–704 (2020).
Wan, T. et al. Decatungstate-mediated C(sp3)-H heteroarylation via radical-polar crossover in batch and flow. Angew. Chem. Int. Ed. 60, 17893–17897 (2021).
Capaldo, L., Quadri, L. L., Merli, D. & Ravelli, D. Photoelectrochemical cross-dehydrogenative coupling of benzothiazoles with strong aliphatic C–H bonds. Chem. Commun. 57, 4424–4427 (2021).
Pandey, G. & Laha, R. Visible-light-catalyzed direct benzylic C(sp3)-H amination reaction by cross-dehydrogenative coupling. Angew. Chem. Int. Ed. 54, 14875–14879 (2015).
Wang, H., Zhang, D. & Bolm, C. Sulfoximidations of benzylic C-H bonds by photocatalysis. Angew. Chem. Int. Ed. 57, 5863–5866 (2018).
Wu, F. et al. Halogen-bond-induced consecutive Csp3-H aminations via hydrogen atom transfer relay strategy. Org. Lett. 22, 2135–2140 (2020).
Margrey, K. A., Czaplyski, W. L., Nicewicz, D. A. & Alexanian, E. J. A general strategy for aliphatic C-H functionalization enabled by organic photoredox catalysis. J. Am. Chem. Soc. 140, 4213–4217 (2018).
Hu, A., Guo, J.-J., Pan, H. & Zuo, Z. Selective functionalization of methane, ethane, and higher alkanes by cerium photocatalysis. Science 361, 668–672 (2018).
An, Q. et al. Cerium-catalyzed C-H functionalizations of alkanes utilizing alcohols as hydrogen atom transfer agents. J. Am. Chem. Soc. 142, 6216–6226 (2020).
Zheng, Y.-W., Narobe, R., Donabauer, K., Yakubov, S. & König, B. Copper(II)-photocatalyzed N−H alkylation with alkanes. ACS Catal. 10, 8582–8589 (2020).
Jia, P. et al. Light-promoted bromine-radical-mediated selective alkylation and amination of unactivated C(sp3)-H bonds. Chem 6, 1766–1776 (2020).
Jiang, Y., Xu, K. & Zeng, C. Use of electrochemistry in the synthesis of heterocyclic structures. Chem. Rev. 118, 4485–4540 (2018).
Kärkäs, M. D. Electrochemical strategies for C-H functionalization and C-N bond formation. Chem. Soc. Rev. 47, 5786–5865 (2018).
Xiong, P. & Xu, H.-C. Chemistry with electrochemically generated N-centered radicals. Acc. Chem. Res. 52, 3339–3350 (2019).
Dagar, N., Sen, P. P. & Roy, S. R. Electrifying sustainability on transition metal-free modes: an eco-friendly approach for the formation of C-N bonds. ChemSusChem 14, 1229–1257 (2021).
Bewick, A., Edwards, G. J., Jones, S. R. & Mellor, J. M. Electrochemical difunctionalisation of adamantane and further oxidation of substituted adamantanes. J. Chem. Soc., Perkin Trans. I. 16, 1831–1834 (1977).
Bewick, A., Edwards, G. J., Jones, S. R. & Mellor, J. M. The electrochemical difunctionalisation of saturated hydrocarbons. Tetrahedron Lett. 17, 631–634 (1976).
Wu, J., Zhou, Y., Zhou, Y., Chiang, C.-W. & Lei, A. Electro-oxidative C(sp3)-H amination of azoles via intermolecular oxidative C(sp3)-H/N-H cross-coupling. ACS Catal. 7, 8320–8323 (2017).
Lian, F., Sun, C., Xu, K. & Zeng, C. Electrochemical dehydrogenative imidation of N-methyl-substituted benzylamines with phthalimides for the direct synthesis of phthalimide-protected gem-diamines. Org. Lett. 21, 156–159 (2019).
Wan, Z. et al. Electrochemical oxidative C(sp3)-H azolation of lactams under mild conditions. Green. Chem. 22, 3742–3747 (2020).
Huang, H., Strater, Z. M. & Lambert, T. H. Electrophotocatalytic C-H functionalization of ethers with high regioselectivity. J. Am. Chem. Soc. 142, 1698–1703 (2020).
Wang, H. et al. Electrochemical synthesis of trans-2,3-disubstituted aziridines via oxidative dehydrogenative intramolecular C(sp3)-H amination. Org. Lett. 21, 9430–9433 (2019).
Liu, S. et al. An electrochemical cinnamyl C-H amination reaction using carbonyl sulfamate. Chin. J. Chem. 37, 570–574 (2019).
Morofuji, T., Shimizu, A. & Yoshida, J. Direct C-N coupling of imidazoles with aromatic and benzylic compounds via electrooxidative C-H functionalization. J. Am. Chem. Soc. 136, 4496–4499 (2014).
Lin, M.-Y. et al. Intermolecular electrochemical C(sp3)-H/N-H cross-coupling of xanthenes with N-alkoxyamides: radical pathway mediated by ferrocene as a redox catalyst. Adv. Synth. Catal. 360, 1665–1672 (2018).
Herold, S., Bafaluy, D. & Muñiz, K. Anodic benzylic C(sp3)-H amination: unified access to pyrrolidines and piperidines. Green. Chem. 20, 3191–3196 (2018).
Yang, Y.-Z., Song, R.-J. & Li, J.-H. Intermolecular anodic oxidative cross-dehydrogenative C(sp3)-N bond-coupling reactions of xanthenes with azoles. Org. Lett. 21, 3228–3231 (2019).
Shen, T. & Lambert, T. H. C-H amination via electrophotocatalytic Ritter-type reaction. J. Am. Chem. Soc. 143, 8597–8602 (2021).
Ruan, Z. et al. Late-stage azolation of benzylic C-H bonds enabled by electrooxidation. Sci. China Chem. 64, 800–807 (2021).
Hou, Z.-W., Li, L. & Wang, L. Organocatalytic electrochemical amination of benzylic C-H bonds. Org. Chem. Front. 8, 4700–4705 (2021).
Hou, Z.-W. et al. Site-selective electrochemical benzylic C-H amination. Angew. Chem. Int. Ed. 60, 2943–2947 (2021).
Barham, J. P. & König, B. Synthetic photoelectrochemistry. Angew. Chem. Int. Ed. 59, 11732–11747 (2020).
Yu, Y., Guo, P., Zhong, J.-S., Yuan, Y. & Ye, K.-Y. Merging photochemistry with electrochemistry in organic synthesis. Org. Chem. Front. 7, 131–135 (2020).
Novaes, L. F. T. et al. Electrocatalysis as an enabling technology for organic synthesis. Chem. Soc. Rev. 50, 7941–8002 (2021).
Wu, S., Kaur, J., Karl, T. A., Tian, X. & Barham, J. P. Synthetic molecular photoelectrochemistry: new frontiers in synthetic applications, mechanistic insights and scalability. Angew. Chem. Int. Ed. 61, 1–27 (2022).
Hu, X., Zhang, G., Bu, F., Nie, L. & Lei, A. Electrochemical-oxidation-induced site-selective intramolecular C(sp3)-H amination. ACS Catal. 8, 9370–9375 (2018).
Nikolaienko, P., Jentsch, M., Kale, A. P., Cai, Y. & Rueping, M. Electrochemical and scalable dehydrogenative C(sp3)-H amination via remote hydrogen atom transfer in batch and continuous flow. Chem. Eur. J. 25, 7177–7184 (2019).
Wang, F. & Stahl, S. S. Merging photochemistry with electrochemistry: functional-group tolerant electrochemical amination of C(sp3)-H bonds. Angew. Chem. Int. Ed. 58, 6385–6390 (2019).
Niu, L. et al. Manganese-catalyzed oxidative azidation of C(sp3)-H bonds under electrophotocatalytic conditions. J. Am. Chem. Soc. 142, 17693–17702 (2020).
Meyer, T. H., Samanta, R. C., Vecchio, A. D. & Ackermann, L. Mangana (iii/iv) electro-catalyzed C(sp3)-H azidation. Chem. Sci. 12, 2890–2897 (2021).
Michaudel, Q., Thevenet, D. & Baran, P. S. Intermolecular Ritter-Type C-H amination of unactivated sp3 carbons. J. Am. Chem. Soc. 134, 2547–2550 (2012).
Li, G.-X. et al. A unified photoredox-catalysis strategy for C(sp3)-H hydroxylation and amidation using hypervalent iodine. Chem. Sci. 8, 7180–7185 (2017).
Shen, T. & Lambert, T. H. Electrophotocatalytic diamination of vicinal C–H bonds. Science 371, 620–626 (2021).
Thomas, L. & Patrick, S. Process for the production of alkali metal and ammonium peroxodisulfate. European patent 1148155 A2 (2001).
Woollins, J. D. The preparation of potassium peroxodisulfate, K2S2O8 in Inorganic Experiments (3rd Edition) (eds, Zdirad, Z. & Antonin, R.) 89-91. (Wiley-VCH, Weinheim, 2010).
Jin, J. & MacMillan, D. W. C. Direct α-arylation of ethers through the combination of photoredox-mediated C-H functionalization and the Minisci reaction. Angew. Chem. Int. Ed. 54, 1565–1569 (2015).
Zhang, Y., Teuscher, K. B. & Ji, H. Direct α-heteroarylation of amides (α to nitrogen) and ethers through a benzaldehyde-mediated photoredox reaction. Chem. Sci. 7, 2111–2118 (2016).
Mandal, S., Bera, T., Dubey, G., Saha, J. & Laha, J. K. Uses of K2S2O8 in metal-catalyzed and metal-free oxidative transformations. ACS Catal. 8, 5085–5144 (2018).
Kaur, J., Shahin, A. & Barham, J. P. Photocatalyst-free, visible-light-mediated C(sp3)-H arylation of amides via a solvent-caged EDA complex. Org. Lett. 23, 2002–2006 (2021).
Ueda, M. et al. Site-selective alkenylation of unactivated C(sp3)- H bonds mediated by compact sulfate radical. Angew. Chem. Int. Ed. 60, 3545–3550 (2021).
Xu, P., Chen, P.-Y. & Xu, H.-C. Scalable photoelectrochemical dehydrogenative cross-coupling of heteroarenes with aliphatic C-H Bonds. Angew. Chem. Int. Ed. 59, 14275–14280 (2020).
Börgel, J., Tanwar, L., Berger, F. & Ritter, T. Late-stage aromatic C-H oxygenation. J. Am. Chem. Soc. 140, 16026–16031 (2018).
Kulakova, E. V. Electrocatalytic hydrogenation of 5-, 8-nitro-, and 6,8-dinitroquinolines. Russ. J. Electrochem. 47, 1186–1189 (2011).
Liu, Y.-Z., Zhang, A.-J., Wang, H., Zhang, L. & Lu, J.-X. Synthesis of 1,5-diaminonaphthalene by electrochemical reduction. Chin. J. Org. Chem. 28, 804–809 (2008).
Waldvogel, S. R. & Trosiena, S. Oxidative transformation of aryls using molybdenum pentachloride. Chem. Commun. 48, 9109–9119 (2012).
Morofuji, T., Shimizu, A. & Yoshida, J. Electrochemical C-H amination: synthesis of aromatic primary amines via N-arylpyridinium ions. J. Am. Chem. Soc. 135, 5000–5003 (2013).
Eberson, L. Electron-transfer reactions in organic chemistry. Adv. Phys. Org. Chem. 18, 79–185 (1982).
Połczyński, P., Jurczakowski, R. & Grochala, W. Stabilization and strong oxidizing properties of Ag(ii) in a fluorine-free solvent. Chem. Commun. 49, 7480–7482 (2013).
Akhmedov, M. A. & Khidirov, S. S. Anodic processes at smooth platinum electrode in concentrated solution of methanesulfonic acid. Russ. J. Electrochem. 55, 579–589 (2019).
Tajima, T., Kishi, Y. & Nakajima, A. Anodic acyloxylation based on the acid–base reactions between acetic acid or trifluoroacetic acid and solid-supported bases. Electrochim. Acta 54, 5959–5963 (2009).
Fleischmann, M. & Pletcher, D. The electrochemical oxidation of some aliphatic hydrocarbons in acetonitrile. Tetrahedron Lett. 9, 6255–6258 (1968).
Acknowledgements
This work was partially sponsored by the National Natural Science Foundation of China (22071056, J.Y.; 22001070, Y.M.), and Shanghai Sailing Program (20YF1410400, Y.M.).
Author information
Authors and Affiliations
Contributions
L.Z. and Y.F. contributed equally to this work. L.Z., Y.F., and Y.S. performed the experiments, analyzed the data. C.L. and M.S. assisted the purification of compounds and analysis of data. R.C., W.Z., and X.Q. contributed to mechanistic studies. L.Z., Y.F., Y.M., and J.Y. wrote the manuscript, supplementary methods, and related materials. Y.M. and J.Y. designed and directed the project. L.Z., Y.F., R.C., W.Z., X.Q., Y.M., and J.Y. revised the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhang, L., Fu, Y., Shen, Y. et al. Ritter-type amination of C(sp3)-H bonds enabled by electrochemistry with SO42−. Nat Commun 13, 4138 (2022). https://doi.org/10.1038/s41467-022-31813-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-022-31813-3
This article is cited by
-
Electro-oxidative amination of benzylic C(sp³)–C(sp³) bonds in aromatic hydrocarbons
Nature Communications (2025)
-
Undiscovered roles of aminopolycarboxylates as reversible masking agents for the DMPO-OH artifact in persulfate oxidation systems
Research on Chemical Intermediates (2025)
-
Electrochemical radical-polar crossover: a radical approach to polar chemistry
Science China Chemistry (2024)
-
Electrochemical oxidative C(sp3)–H cross-coupling with hydrogen evolution
Nature Synthesis (2023)