Abstract
Highly efficient perovskite solar cells (PSCs) in the n-i-p structure have demonstrated limited operational lifetimes, primarily due to the layer-to-layer ion diffusion in the perovskite/doped hole-transport layer (HTL) heterojunction, leading to conductivity drop in HTL and component loss in perovskite. Herein, we introduce an ultrathin (~7 nm) p-type polymeric interlayer (D18) with excellent ion-blocking ability between perovskite and HTL to address these issues. The ultrathin D18 interlayer effectively inhibits the layer-to-layer diffusion of lithium, methylammonium, formamidium, and iodide ions. Additionally, D18 improves the energy-level alignment at the perovskite/HTL interface and facilitates efficient hole extraction. The resulting PSCs achieve efficiencies of 26.39 (certified 26.17) and 25.02% with aperture areas of 0.12 and 1.00 square centimeters, respectively. Remarkably, the devices retain 95.4% of the initial efficiency after 1100 hours of operation in maximum power point tracking, representing significant stability advancements for high-efficiency n-i-p PSCs.
Similar content being viewed by others
Introduction
Certified power conversion efficiencies (PCEs) exceeding 26% have been reported for both normal (n-i-p)1 and inverted (p-i-n)2 structure perovskite solar cells (PSCs). However, the long-term stability of the highly efficient n-i-p PSCs under light and heat stresses was significantly inferior to that of p-i-n ones3,4,5,6,7. The stability gap was primarily attributed to the difference in the adopted charge transport layers (CTL) and therefore the different perovskite/CTL interfaces, leading to divergent ion-diffusion behaviors within the CTL, perovskite layer, and across the perovskite/CTL interface in these two types of devices. Specifically, in the efficient p-i-n devices, the hole transport layer (HTL) made from self-assembled monolayers (SAMs) is uniquely thin (<2 nm) but still forms a uniform and complete barrier between the electrode and the perovskite layer8, and the thermal evaporated electron-transport layer (ETL) of C60 has additional corrosion resistance and ion blocking capabilities due to its good conformality and controllable thickness9,10,11. However, the most commonly used HTL in high-efficiency n-i-p PSCs, 2,2′,7,7′-tetrakis[N,N-di(4-methoxyphenyl)amino]-9,9′-spirobifluorene (Spiro-OMeTAD), is usually tousy and thick (200 ~ 370 nm)12,13, allowing ion diffusion stemming from dopants14, electrode elements15 or perovskite components16,17. Additionally, the electronic properties of Spiro-OMeTAD are sensitive to the escapable Li+ dopants18. Replacing Spiro-OMeTAD with more stable HTL or bi-layered stable HTL would produce more stable n-i-p devices19,20,21,22,23. However, the efficiency of the devices significantly declines (decreasing from over 25 to ~20%), which loses the advantage of the high efficiency of PSCs over other photovoltaic technologies. We note that for the development of n-i-p PSCs, stabilizing the conventional high-performance Spiro-OMeTAD-based devices is equally important to replacing Spiro-OMeTAD with more stable HTLs.
The perovskite side of the perovskite/HTL interface in n-i-p PSCs can serve as a reservoir of mobile ions. The components of perovskites, such as CH(NH2)2 (FA+), CH3NH3+ (MA+), Pb2+, I−, etc., are largely held together through ionic bonding24,25,26,27,28, resulting in a soft crystal lattice. This characteristic makes the perovskite film suffer from severe ion migration when exposed to heat, light, and electric fields29. Previous studies have proved that halogen ions can travel to corrode HTL and the metal electrode, gradually reducing their conductivity and damaging the stability of PSCs30,31,32. Therefore, the severe ion diffusion from perovskite to HTL leads to irreversible degradation of perovskite and the failure of HTL as well as the electrode, which is a key factor limiting the long-term stability of n-i-p PSCs33,34. Hence, designing internal barriers that block the layer-to-layer ion diffusion is critically important to improve the operation lifetime of n-i-p PSCs35,36,37. Previous studies have reported various interfacial materials including inorganic salts38, organic small molecules39,40,41, low-dimensional perovskites42,43,44, and polymers45,46 to inhabit ion inter-diffusion across the perovskite/HTL interface of n-i-p PSCs. However, the reported strategies have yet to significantly enhance the operational stability of n-i-p PCSs to a level comparable with the p-i-n PSCs. To fabricate efficient n-i-p PSCs with outstanding operational stability, we assume that the ion-blocking capability of the interlayers must be compatible with efficient hole extraction, which is usually a challenging trade-off.
Herein, we report the insertion of an ultrathin (~7 nm) p-type polymeric interlayer (D18) allows efficient hole transport but blocks ion diffusion between perovskite and Spiro-OMeTAD for the fabrication of efficient and stable n-i-p PSCs. D18, a widely adopted p-type donor material in organic solar cells, can form a dense polymeric membrane on the perovskite surface via hot casting, effectively blocking ion diffusion between the perovskite layer and the Spiro-OMeTAD layer. The comparison study showed that D18 possesses a much stronger ion-blocking ability than the commonly used polymeric HTLs of poly(3-hexylthiophene) (P3HT) and poly[bis(4-phenyl)(2,4,6-trimethylphenyl)amine] (PTAA) due to the much stronger π-π stacking interaction between the backbone of the D18 polymer chains. Accelerated thermal aging tests showed that D18 prevented perovskite film from degradation caused by component loss and protected Spiro-OMeTAD from ion corrosion. Moreover, the D18 hole-selective membrane improved the energy-level alignment at the perovskite/HTL interface, effectively reducing interfacial energy loss and enhancing the hole extraction. These synergetic effects resulted in n-i-p PSCs with a maximum efficiency of 26.39% (certified 26.17%) for an area of 0.12 cm2 and 25.02% for an area of 1 cm2. The PSCs maintained 95.4% of their initial efficiencies after 1100 h at maximum power point tracking under continuous 1-sun illumination at 50 °C, representing the most stable n-i-p PSCs with high efficiency.
Results
Ion-blocking effect of the D18 interlayer
The idea of incorporating a hole-selective interlayer in PSCs was inspired by proton exchange membrane (PEM) fuel cells, where the PEM serves as a proton conductor while blocking the diffusion of other chemical species. To achieve highly stable n-i-p PSCs with high efficiency, the inserted hole-selective interlayer is expected to efficiently transport photo-generated holes and inhibit ion diffusion. PDTBT2T-FTBDT (D18), an efficient donor polymer in organic solar cells47,48, was chosen on the basis of the following considerations. First, D18 has a fused-ring acceptor unit, dithieno[3′,2′:3,4;2'',3'':5,6]benzo[1,2-c][1,2,5]thiadiazole (DTBT), with a large molecular plane, which enhances the π–π stacking of the backbone, contributing to a tightly-packed and dense D18 film (Fig. 1a). Thick polymeric films are known to contact poor on the surface of perovskite by spin-coating method. Assuming an ultrathin polymeric D18 provides conformal coverage on the surface of perovskite due to the high fluidity of the diluted solution of D18, the dense and conformal D18 interlayer is expected to efficiently inhibit ion diffusion. Second, D18 features strong temperature-dependent aggregation in diluted solution and ordered packing in the condensed phase, the spin-coating of Spiro-OMeTAD at room temperature did not damage the integrity of D18. Third, D18 exhibits matched energy level alignment with adjacent perovskite and Spiro-OMeTAD (Supplementary Fig. 1) and possesses a high hole mobility of 3.8 × 10−2 cm2 V−1 s−1 (Supplementary Fig. 2), which makes it possible for D18 to act as a hole-selective interlayer.
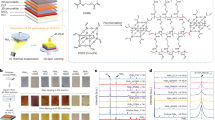
a Device architecture, the structure of D18 polymer, and the detailed ion blocking effect of D18 membrane. b Schematic for investigating the effectiveness of D18 on blocking ion diffusion. c XRD patterns of PbBr2, FAI, and films after spin-casting FAI on PbBr2 or D18-covered PbBr2. d HRTEM image of the enlarged PVSK/D18 interface for FTO/SnO2/PVSK/D18 structure, Spiro was first deposited on the top of FTO/SnO2/PVSK/D18 and then washed off with room-temperature chlorobenzene solvent. Au and Pt are protection layers deposited on the as-prepared film before FIB-cutting.
The cell architecture (glass/FTO/perovskite/D18/Spiro-OMeTAD/Ag) is presented in Fig. 1a. For convenience, perovskite and Spiro-OMeTAD are denoted as PVSK and Spiro, respectively. The interlayer was prepared by spin-coating a hot chlorobenzene (CB) solution of D18 on top of the PVSK film. D18 with different number-average molecular weight (Mn) has a great difference in solubility, which affects the properties of the formed membranes. We observed that the D18 with low Mn (22 kDa, D18-L) can readily dissolve in CB at room temperature, while that with moderate Mn (69 kDa, D18-M) and high Mn (98 kDa, D18-H) necessitate stirring at 90 °C for dissolution (Supplementary Fig. 3a, b). Consequently, D18-L is unsuitable for subsequent Spiro deposition as it compromises membrane integrity. Further analysis revealed that a hot solution of D18-M can uniformly cover the surface of PVSK, whereas D18-H formed a discontinuous layer (Supplementary Fig. 3c–e). Therefore, D18-M (abbreviated as D18) was selected as the preferred interfacial material. Additionally, we found that the quality of the D18 interlayer was influenced by the substrate temperature. Optimal results were achieved when a fresh hot solution of D18-M was spin-coated onto a hot PVSK film (Supplementary Fig. 4), ensuring the formation of a dense interlayer. SEM images demonstrated excellent coverage of D18 on the PVSK surface using thermal spin-coating technology (Supplementary Fig. 5a, b). However, when spin-coated onto a cool PVSK, D18 tends to aggregate (Supplementary Fig. 5c). These observations can be explained by the strong temperature-dependent aggregation of D18 in diluted solution and ordered packing in the condensed phase49. Analysis of the surface potential distribution of PVSK/D18 compared to pure PVSK (Supplementary Fig. 6) indicated that D18 achieved uniform contact with the PVSK surface, resulting in relatively uniform potential distribution.
A dense interlayer is expected to effectively block ion interdiffusion between the PVSK and Spiro layers, such as I−, FA+, MA+, and Li+. The ion-blocking effects of the D18 interlayer and the most commonly used polymer HTL (P3HT and PTAA), were assessed and compared based on the reaction between PbBr2 and FAI. When spin-coating FAI on PbBr2, FAPb(IxBr1−x)3 forms due to ion exchange, resulting in a color change from white to brown (Fig. 1b and Supplementary Fig. 7). However, when the PbBr2 substrate was covered with D18, white particles were observed on the surface of D18, indicating that the D18 membrane effectively blocked ion diffusion between PbBr2 and FAI. X-ray diffraction (XRD) measurements were performed to compare the ion-blocking effect of D18, P3HT, and PTAA. As shown in Fig. 1c, pristine PbBr2 film and FAPb(IxBr1−x)3 exhibit characteristic diffraction peaks at 18.48° and 14.2°, respectively. For PbBr2 film covered by D18 deposited with a concentration of 1 mg mL−1, characteristic peaks of FAPb(IxBr1−x)3 were observed, indicating that the resulting interlayer was not thick enough to prevent the diffusion of FA+ and I−. Increasing the concentration to 2 mg mL−1, instead of FAPb(IxBr1−x)3, diffraction peaks of PbBr2 and FAI were observed, indicating that the reaction was fully impeded. In contrast, for PbBr2 films covered by P3HT or PTAA, the necessitated concentrations of both P3HT and PTAA to completely inhibit the reaction were as high as 10 mg mL−1 (Supplementary Fig. 8). This result indicates that the effectiveness of inhabiting ion diffusion of D18 is superior to both P3HT and PTAA, which is primarily attributed to the strong π-π interaction of the DTBT unit in the D18 backbone, resulting in a compact polymer thin film (Fig. 1a, Supplementary Fig. 9 and Supplementary Note 1). We further investigate the time dependency of the reaction of D18-covered PbBr2 with FAI, CsBr, MABr, and MACl, the results show that the D18 layer has robust ion-blocking capability under thermal stress (Supplementary Fig. 10 and Supplementary Note 2).
To determine the minimum thickness of the D18 membrane screened by the abovementioned XRD experiments, we prepared a cross-sectional sample of FTO/SnO2/PVSK/D18 stack using 2 mg mL−1 D18 by focused ion beam (FIB), and the thickness was measured by high-resolution transmission electron microscopy (HRTEM). An ultrathin D18 layer with a thickness of around 7 nm could be discerned in the PVSK/D18 interface in Fig. 1d. Moreover, D18 is in close contact with the perovskite grain and grain boundary, providing conformal coverage (Supplementary Fig 11). Additionally, we compared the film integrality and thickness of the D18 interlayer before and after the deposition of Spiro by TEM. The cross-sectional sample of the device after the spin-coating of Spiro was prepared by first depositing the Spiro layer and then removing the Spiro film with room-temperature chlorobenzene solvent. The TEM images (Supplementary Fig 12) showed that the D18 interlayer for both samples was compact and the thickness was similar, indicating that the ultrathin D18 interlayer will not be damaged during the subsequent deposition of Spiro.
Stabilization effect of the D18 interlayer on perovskite and HTL
To investigate the stabilization effect of the D18 interlayer on PVSK under elevated temperatures, we measured the XRD and reflection spectra of the complete devices (Fig. 2a). We refer to the devices fabricated without and with D18 as the “control” and “D18” devices, respectively. The concentration of D18 used to deposit the membrane was 2 mg mL−1. From the XRD patterns, we found that the PVSK absorber in the control device degraded to PbI2 after 500 h of aging at 85 °C, whereas the D18 device maintained unchanged XRD peak intensity of PbI2 and PVSK (Fig. 2b). This implies that the D18 membrane could effectively protect PVSK from degradation under heat. From the reflection spectra (Fig. 2c), we found that the band edge of PVSK in the control device underwent a redshift, suggesting phase dissociation due to the escape of the volatile components. In contrast, in the D18 device, no obvious redshift was observed. The existence of the D18 interlayer may block the channels that allow the ion diffusion from PVSK to the HTL, preventing the loss of volatile components of PVSK, and therefore suppressing the degradation of PVSK. The cross-sectional SEM images of the aged devices in Fig. 2d demonstrate that there were many pinholes in the Spiro layer of the control device, which could provide channels for the escape of volatile species from the PVSK layer50, leading to severe morphological deformation of PVSK. Upon introducing the D18 membrane, the bulk PVSK maintained well-defined grains, indicating that D18 effectively blocked the escape of PVSK components by forming a dense membrane between the PVSK and Spiro layers.
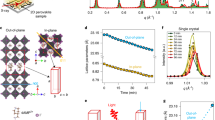
a Illustration of the XRD and reflection spectroscopy tests for the complete solar cell devices during accelerated thermal aging at 85 °C. XRD patterns (b) and reflection spectra (c) of the control and D18 devices before and after aging. d Cross-sectional SEM images of aged control and D18 devices. ToF-SIMS depth profile of I− and FA+ within the aged control (e) and D18 (f) devices. g Illustration of exposing the aged Spiro surface for KPFM measurement. KPFM images of Spiro of control (h) and D18 (i) devices before and after aging, respectively.
To elucidate the protection mechanism of the D18 membrane on PVSK under thermal stress, time-of-flight secondary ion mass spectroscopy (ToF-SIMS) measurements were conducted to investigate the element distribution within the control and D18 devices before and after thermal aging. We observed that D18 (indicated by the C4SH− signal) was indeed located at the interface between PVSK and Spiro in the D18 device (Supplementary Fig. 13). For the control device, the I− and FA+ signals in the Spiro layer of the aged device significantly increased compared to the fresh control device (Fig. 2e, f and Supplementary Fig. 13a, c), indicating the obvious diffusion of I− and FA+ from PVSK to Spiro under thermal stress, and further revealed the migration to the metal electrode through pinholes in the Spiro layer. In contrast, the aged D18 device showed almost unchanged I− and FA+ signals compared to the fresh sample (Fig. 2e, f and Supplementary Fig. 13b, d), verifying the blocked diffusion of I− and FA+ by inserting the D18 membrane. Therefore, we can conclude that the D18 membrane effectively protects PVSK degradation from heat by blocking ions diffusion from the PVSK to the Spiro layer.
Previous reports have demonstrated that diffused I− and MA+ can corrode the HTL, thereby reducing hole-transport capacity30,51. To elucidate the stabilization effect of the D18 membrane on Spiro and consequently improve the stability of the PVSK/HTL heterostructure, we measured the surface potential of Spiro layers using Kelvin probe force microscopy (KPFM) before and after aging. The devices were aged at 85 °C for 500 h and the Au electrode was removed before the measurement (Fig. 2g). As depicted in Fig. 2h, the surface potential of Spiro in the control device decreased from −207 to −609 mV after aging. In contrast, the surface potential of the D18 device exhibited a much smaller decrease from −156 to −293 mV (Fig. 2i). The work function (WF) of the Spiro layers was determined by calibrating the surface potential of the tip with an Au reference (WF = 5.20 eV) and is presented in Supplementary Table 1. The small difference in WF between fresh (~4.8 eV) and aged Spiro (~4.7 eV) in the D18 devices can be attributed to the impediment of I− and FA+ diffusion from PVSK to Spiro and the blocking of Li+ loss in the Spiro layer (Supplementary Fig. 13e, f). Conversely, the significantly decreased WF of the aged Spiro in the control device from ~4.8 to ~4.4 eV indicated much-reduced p-doping of Spiro. These results demonstrated that the D18 interlayer exhibited an astonishing stabilization effect on the PVSK/HTL heterostructure under thermal stress.
Charge carrier dynamics
To achieve highly efficient PSCs, the introduction of the interlayer should not impede charge transport between the PVSK and Spiro. To investigate the carrier dynamics within the PVSK/Spiro and PVSK/D18/Spiro stacks, we conducted steady-state photoluminescence (PL) and time-resolved photoluminescence (TRPL) measurements. As depicted in Fig. 3a, the PL intensity of the PVSK/D18 stack was significantly quenched compared to that of PVSK, and the PVSK/D18/Spiro stack showed the most efficient quenching effect, suggesting the excellent hole extraction efficiency of D18. TRPL measurements also revealed that PVSK/D18/Spiro displayed a faster TRPL decay than PVSK/Spiro (Fig. 3b), indicating improved hole transfer between PVSK and Spiro after inserting the D18 interlayer. Space charge limited current (SCLC) curves (Supplementary Fig. 14) of the hole-only devices demonstrated that the hole mobility for the ITO/PEDOT:PSS/PVSK/D18/Spiro (0.25 cm2 V−1 s−1) was higher than that of without D18 (0.22 cm2 V−1 s−1). Additionally, from the SCLC curves, the defect state density was calculated to be decreased from 3.69 × 1015 cm−3 for the control device to 2.49 × 1015 cm−3 for the D18 device. The decreased defect density may be related to the chemical interaction between D18 and the PVSK surface. Note that the PVSK surface was treated with OAI, thus D18 was deposited on the combined 3D/2D PVSK structure (Supplementary Fig. 15). Considering the inhomogeneity of the 2D passivation layer on the PVSK surface and the defects of the 2D PVSK itself (Supplementary Fig. 16), D18 may interact chemically with the defects on both the 3D and 2D PVSK surfaces. X-ray photoelectron spectroscopy (XPS) measurements for PVSK/D18 and 2D/D18 samples showed that the Pb 4f XPS peaks shifted to higher binding energies and the F 1s XPS peaks shifted to lower binding energies at the PVSK/D18 surface and a reduction of Pb0 defects at the 2D/D18 surface (Supplementary Fig. 17), indicating that F can interact with Pb2+ ions of 3D PVSK and Pb0 defects of 2D PVSK, respectively, providing additional passivation effect. In conclusion, the D18 interlayer does not hinder hole transport within the PVSK/HTL heterostructure but instead improves the hole extraction efficiency.
PL spectra (a) and TRPL decay (b) of PVSK, PVSK/D18, PVSK/Spiro, and PVSK/D18/Spiro stacks. c Energy level diagram of PVSK, PVSK/D18, and Spiro. d Measured VOC and QFLS of the control and D18 devices. e One sun J-V (lines) and pseudo J-V (symbols) curves of the control and D18 devices. f FF loss analysis of the control and D18 devices. The data points connected by the black dashed line are ideal FF from the Shockley-Queisser (SQ) limit. The data points connected by the red dashed line are the maximum FF without charge transport loss. The data points connected by the red solid line are the measured FF from the devices.
To comprehend the enhanced hole extraction of the PVSK/D18/Spiro stack, ultraviolet photoelectron spectroscopy (UPS) was performed to determine the interfacial energy level of PVSK and PVSK/D18 (Supplementary Fig. 18a, b). The optical bandgap of the PVSK was determined to be 1.52 eV by differentiating the external quantum efficiency (EQE) of the complete device (Supplementary Fig. 18c). The schematic energy level diagram of PVSK, PVSK/D18, and Spiro is presented in Fig. 3c, where Spiro shows a large misalignment of 450 meV between its HOMO and the valence band maximum (VBM) of PVSK, whereas the corresponding misalignment between Spiro and the PVSK/D18 is only 20 meV. The VBM up-shift of the PVSK/D18 combination may be related to the matched energy level alignment of D18 with adjacent PVSK and Spiro. Previous studies have shown that introducing interfacial molecules with graded energy levels between PVSK and Spiro forms a cascade energy level structure, making the energy levels between PVSK and Spiro more matched and facilitating hole extraction52,53. Therefore, the improved band alignment by introducing the D18 interlayer contributed to the enhanced hole extraction between PVSK and Spiro.
To quantitatively investigate the effect of the well-matched band alignment of PVSK/D18/Spiro stack on minimizing the interfacial energy loss and enhancing charge transfer, photoluminescence quantum yield (PLQY) measurements and quasi-Fermi level splitting (QFLS) analysis were conducted. The glass/PVSK/D18 stack showed a decreased PLQY value of 6.04% compared with the glass/PVSK stack of 10.83%, consistent with the PL results (Supplementary Fig. 19). The decreased PLQY of the glass/PVSK/D18 stack indicates that charge extraction predominates at the PVSK/D18 interface rather than chemical passivation. However, in the complete stack with SnO2, the D18 device exhibited a higher PLQY than the control device, indicating that introducing D18 reduced across-interface recombination rather than directly passivating the perovskite surface. We propose that the reduced interfacial losses upon D18 incorporation originated from the well-matched band alignment of the PVSK/D18/Spiro stack (Fig. 3c). The decreased valence band misalignment between Spiro and the PVSK/D18 will enable a higher VOC since the electron and hole quasi-Fermi levels within the illuminated PVSK absorber need to align with the Fermi levels of the charge-selective contacts to produce an optimal built-in voltage54. We summarized the open-circuit voltage (VOC) and QLFS gains of the D18 device compared with the control device in Fig. 3d. The details of the QLFS calculation were present in the method. The D18 device showed a higher QFLS than the control, which agreed with the measured VOC. We note that the QFLS value of the D18 device increased by 20 meV, whereas the measured VOC of the device increased by 33 mV. Thus, the VOC increase was not only caused by a reduction of non-radiative recombination loss but also an improved interfacial band alignment.
Moreover, the JSC-VOC technique55 was conducted to determine the series resistance (Rs) inside the device (Fig. 3e), which is closely related to interfacial charger transfer. Rs was calculated to be 1.11 Ω cm2 for the D18 device, which was smaller than that of the control device (1.70 Ω cm2). This reduced Rs can only come from the introduction of the D18 interlayer since all other layers of the devices that contribute to the series resistance were the same. The PVSK/D18 stack has a 20 meV deeper valence band edge than the HOMO of Spiro, so good hole transport from PVSK to Spiro is expected, contributing to a smaller Rs. The decreased Rs also contributed to a 3.55% fill factor (FF) gain in the D18 device, as shown in Fig. 3f.
Device performance and stability
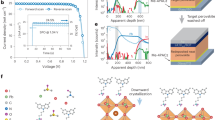
We further evaluated the effect of the D18 interlayer on the photovoltaic performance of PSCs. The optimal concentration of D18 was determined to be 2 mg mL−1 (Supplementary Fig. 20). The representative current density-voltage (J-V) curves are presented in Fig. 4a, and the statistical results of 67 cells are shown in Fig. 4b and Supplementary Fig. 21. The best D18 device achieved a PCE of 26.39% in the reverse scan and 26.12% in the forward scan. One of the best-performing devices was sent for third-party certification, and the certified PCE was 26.17% (Supplementary Fig. 22). When the D18 interlayer was inserted between PVSK and Spiro, the J-V curves in Fig. 4a showed an FF increase from 80.37 to 83.92% and a VOC increase from 1.152 to 1.185 V. We attribute the improved device performance to a more favorable band alignment and suppression of non-radiative recombination.
a Reverse and forward J-V scan of the control and D18 devices (0.12 cm2). –R indicates reverse scan and –F indicates forward scan. b Statistical PCE of the control and D18 devices. c Reverse and forward J-V scan of large-area (1 cm2) control and D18 devices. d Operational stability of the control and D18 devices. e Thermal stability of the control and D18 devices under 85 °C. f Dark shelf stability of the control and D18 devices. All stability tests were based on devices without encapsulation.
The external quantum efficiency (EQE) spectrum of the D18 device showed an integrated short-circuit current density of 25.86 mA cm−2 (Supplementary Fig. 23), which was in agreement with the J-V curves. The steady-state power output (SPO) efficiency of the device reached a stabilized efficiency of 25.64% after 200 s (Supplementary Fig. 24). We further fabricated PSCs with larger areas (1 cm2), and the best-performing device exhibited a forward PCE of 25.02% and a reverse PCE of 24.80% (Fig. 4c), which is one of the highest PCEs ever reported for a device area of 1 cm2. We also fabricated devices based on pure D18 as HTL. The device showed a PCE of 23.27% when the used concentration of D18 was 6 mg mL−1 with a thickness of 70 nm (Supplementary Figs. 25 and 26a). However, the cross-sectional SEM image in Supplementary Fig. 26b showed that the thick D18 polymer HTL has poor contact with the PVSK surface, forming obvious pinholes at the PVSK/D18 interface, which would cause severe recombination. This result indicates that the interface quality at the PVSK/HTL interface highly depends on the thickness of the polymer, and the ultrathin polymeric D18 interlayer shows superior performance.
Transient photovoltage (TPV) and transient photocurrent (TPC) measurements were conducted to check and compare charge recombination and transport. In the TPV test, the decay time increased from 97 μs for the control device to 564 μs for the D18 devices (Supplementary Fig. 27a), indicating significantly suppressed charge recombination. In the TPC test, the decay time decreased from 3.15 μs for the control device to 1.84 μs for the D18 devices (Supplementary Fig. 27b), confirming improved charge transfer for the D18 device. We measured the VOC-light intensity dependency (Supplementary Fig. 28) and calculated the ideality factor for the control device to be 1.49, which was reduced to 1.39 for the D18 devices, indicating decreased non-radiative recombination loss. Additionally, we studied the electroluminescence performance of the devices working as light-emitting diodes. The D18 device exhibited an external quantum efficiency of 8.76%, compared to 4.01% of the control device (Supplementary Fig. 29), consistent with the trend of VOC increase. We conclude that the increase in VOC and FF was mainly caused by suppressed interfacial non-radiative recombination and improved charge transport.
We further investigated the operational stability of the control and D18 devices by MPP tracking under 1-sun illumination at around 50 °C in a nitrogen atmosphere. As shown in Fig. 4d, the PCE of the control device decreased to 80% of the initial value after 278 h, whereas the D18 device maintained 95.4% of its initial efficiency after 1100 h. We further studied their thermal stability at 85 °C (Fig. 4e). After aging for 800 h, the control device showed a decline to 28.26% of its initial PCE. However, the D18 device retained 84.1% of its initial PCE. The enhanced heat resistance might be explained by the reduced ions diffusion in the D18 device. To further verify the effectiveness of D18 in ensuring ion-blocking, we replaced the Spiro with PTAA, fabricated the corresponding PSCs, and evaluated the devices’ thermal stability. As shown in Supplementary Fig. 30, both the PCE and thermal stability were improved after incorporating D18. The improvement of PCE may related to that D18 serves as a buffer layer between the PVSK and PTAA films and avoids the direct contact between perovskite and electrode caused by the thin PTAA layer. Although PTAA is stable up to high temperatures, previous studies have shown that PTAA was not a good barrier against ionic diffusion56. The enhanced heat resistance of the D18/PTAA device verifies the effectiveness of D18 in ion-blocking. Additionally, we also investigated the operational and thermal stability of the solar cells utilizing pure D18 as HTL, however, they didn’t show better operational and thermal stability than D18/Spiro devices (Supplementary Fig. 31). This could be attributed to the above-mentioned thickness-dependent interface quality between PVSK and D18. The thick D18 polymer HTL has poor contact with the PVSK surface, forming obvious pinholes at the PVSK/D18 interface, which is not conducive to the operational and thermal stability of the devices. These results indicated that the higher stability of HTL materials can only transform into stable solar cells if the interface quality of the PVSK/polymer is comparable with that of the PVSK/small molecules. Finally, the shelf lifetime of the unencapsulated devices was studied. The D18 device maintained 95.5% of the PCE after being stored at 25 °C and around 10% relative humidity for 5088 h, whereas the control devices only retained 58.2% of the PCE (Fig. 4f).
Discussion
In summary, we have shown that the introduction of the polymeric D18 interlayer can effectively block layer-to-layer ion diffusion within PSCs while maintaining highly efficient hole transport, leading to significantly improved stability of the n-i-p PSCs with a certified PCE of over 26%. Particularly, the hole-selective interlayer inhibited ion diffusion from PVSK to HTL under thermal stress, effectively preventing the ion-corrosion-induced degradation of HTL. Furthermore, an improved interfacial band alignment between PVSK/D18 and Spiro was achieved, minimizing interfacial recombination loss for higher VOC and minimizing transport losses for higher FF. Ultimately, we achieved n-i-p PSCs with a maximum efficiency of 26.39% (certified 26.17%) for a device area of 0.12 cm2 and 25.02% for 1 cm2. The devices exhibited a T95 operational lifetime of over 1100 h, representing the significant stability advancement in highly efficient n-i-p PSCs (Supplementary Table 2). We believe that incorporating a polymeric charge-selective interlayer is a promising strategy to enhance the operational lifetime of perovskite solar cells and solar modules, paving the way for their commercial application.
Methods
Materials
Tin (II) chloride dihydrate (SnCl2·2H2O, 99.99%) was purchased from Aladdin. Lead iodide (PbI2, 99.99%) and guanidinium iodide (GAI, 97%) were purchased from TCI. Formamidinium iodide (FAI, 99.99%) and methylammonium iodide (MAI, 98.0%) were purchased from GreatcellSolar. Methylammonium chloride (MACl, 99.5%), n-octylammonium iodide (OAI, 99.5%), and poly[bis(4-phenyl)(2,4,6-trimethylphenyl)amine] (PTAA) were purchased from Xi’an Polymer Light Technology in China. Polymer donor D18 was synthesized according to the literature48. Poly(3-hexylthiophene) (P3HT) was purchased from 1-Material. 2,2′,7,7′-tetrakis [N,N-p-di(methoxy-phenyl)amino]-9,9′-spirobifluorene (Spiro-OMeTAD, 99.8%) was purchased from Feiming Technology Corporation. Lithium bis(trifluoromethanesulfonyl)imide (Li-TFSI, 99.95%), urea (98%), thioglycolic acid (TGA, 98%), N,N-dimethylformamide (DMF, 99.8%), dimethyl sulfoxide (DMSO, 99.9%), isopropanol (IPA, 99.5%), chlorobenzene (CB, 99.9%), 4-tert-butylpyridine (4-TBP, 98%) and acetonitrile (99.8%) were purchased from Sigma-Aldrich. Poly(3,4-ethylenedioxythiophene):poly(styrenesulfonate) (PEDOT:PSS, Clevios PVP AI4083) was purchased from Heraeus. Hydrochloric acid (HCl, 37 wt. % in water) was purchased from Sinopharm Chemical Reagent Co., Ltd. All the materials were used as received without any purification.
Device fabrication
The FTO substrates were cleaned by a sequential sonication treatment in a 2% Hellmanex solution, DI water, isopropanol, and ethanol for 20 min, respectively. The SnO2 layer was fabricated using a chemical bath deposition (CBD) method, similar to the previous report57, and annealed at 170 °C for 60 min in the air. Before perovskite deposition, the FTO/SnO2 substrate was treated with O2 plasma for 10 min. The perovskite film was fabricated by a two-step solution process: the PbI2 solution (1.5 M, dissolved in DMF/DMSO (volume ratio 9:1)) mixed with 5% CsI was spin-coated on SnO2 substrate at 2000 r.p.m. for 30 s and then annealed at 70 °C for 1 min. Organic salt solution (FAI 0.55 M; MACl 0.12 M; MAI 0.02 M; GAI 0.01 M, dissolved in IPA) was spin-coated on PbI2 film at 1700 r.p.m. for 30 s and then annealed at 150 °C for 15 min in air with 40% RH. OAI (3 mg mL−1, dissolved in IPA) was spin-coated onto the perovskite films at 5000 r.p.m. for 30 s and annealed at 100 °C for 3 min. Both the control and D18 devices were passivated by OAI. For D18-covered perovskite, D18 solution (2 mg mL−1, dissolved in CB and stirred at 90 °C for 2 h) was spin-coated onto the passivated perovskite films at 3000 r.p.m. for 30 s. The hole transport layer was prepared by spin-coating Spiro-OMeTAD solution (90 mg mL−1, dissolved in CB) doped with 36.1 μL of TBP and 21.9 μL of Li-TFSI (520 mg mL−1, dissolved in acetonitrile) at 3,000 r.p.m. for 30 s. Finally, a 100 nm Au or Ag electrode was thermally evaporated on the hole transport layer to complete the device.
Film fabrication
For the investigation of the effectiveness of different polymers at inhibiting ion diffusion, the PbBr2 films were deposited by spin coating PbBr2 solution (0.4 M) on glass substrates at 2000 r.p.m. for 30 s and then annealed at 100 °C for 5 min. The polymer layers were deposited by spin coating D18, P3HT, and PTAA solutions (2, 4, 6, 10 mg mL−1) on PbBr2 films at 3000 r.p.m. for 30 s, and then the FAI solution (0.4 M) was spin-coated on PbBr2/D18, PbBr2/P3HT, and PbBr2/PTAA films at 1700 r.p.m. for 30 s and then annealed at 100 °C for 10 min.
Device characterization
Current density-voltage characterizations were measured using a digital source meter (Keithley 2400) under AM 1.5 G conditions (EnliTech, AAA solar simulator). The J-V measurements were performed in an N2-filled glove box at room temperature. The scan rate was 250 mV s−1 (voltage step of 10 mV and delay time of 40 ms). The light intensity was calibrated using an NREL-calibrated Si solar cell equipped with an infrared cut-off filter (KG-5). The active area of PSCs was 0.2 cm2. A black shadow mask with an effective area of 0.12 cm2 or 1.00 cm2 was used to reduce the light scattering. Device certification was conducted by FUJIAN METROLOGY INSTITUTE, National PV Industry Measurement and Testing Center. The device has an active area of 0.1182 cm2. External quantum efficiencies were carried out on an EQE system (EnliTech, QER666) without any bias light. Unless otherwise stated, the J-V curves, certified efficiency, and EQE spectra were obtained from the devices with anti-reflective coating. The operational stability was monitored by a solar cell stability test system, which consisted of a white LED lamp with a power density of 100 mW cm−2 as the light source. For the device operating as a LED, the EQEEL was determined by measuring the absolute radiation flux using a source meter (Keithley 2400) and a commercialized system (XPQY-EQE, Guangzhou Xi Pu Optoelectronics Technology Co., Ltd.) that was equipped with an integrated sphere (GPS-4P-SL, Labsphere) and a QE-Pro spectrometer (Ocean Optics). Transient photovoltage and transient photocurrent were carried out on a Zahner electrochemical workstation equipped with a transient electrochemical measurement unit (Fast CIMPS).
Morphology characterization
Scanning electron microscopy (SEM) was carried out on a field-emission scanning electron microscope (JEOL JSM-7610F). Top-view SEM morphologies of perovskite films and cross-sectional morphologies of PSCs were operated at voltages of 3 and 5 kV, respectively. The cross-sectional transmission electron microscope (TEM) samples were prepared with a focused ion beam (FEI Scios). To protect the samples, Au and Pt layers were deposited on top of samples before FIB-cutting. The TEM images were characterized using JEOL F200 TEM.
XRD measurement
X-ray diffraction (XRD) was performed on a SmartLab X-ray diffractometer (Rigaku Corporation) using a Cu Kα radiation source operating at a voltage of 40 kV and a current of 30 mA.
Reflection spectra
Reflection spectra were measured using a Perkin Elmer UV-Vis spectrophotometer (Lambda 1050) in a wavelength range between 400 and 1000 nm.
PL, TRPL, and PLQY measurements
Steady-state photoluminescence (PL) spectra were acquired on an instrument supplied by Xipu Electronics equipped with an integration sphere in the glove box, using a 365 nm excitation laser. Time-resolved photoluminescence (TRPL) was measured using time-correlated single photon counting (TCSPC) equipment (FLS1000, Edinburgh Instruments Ltd) with a 375 nm pulsed laser (EPL-375). Photoluminescence quantum yield (PLQY) was measured using an integrating sphere (FLS1000, Edinburgh Instruments Ltd), and the excitation wavelength was set as 500 nm.
UPS and KPFM measurement
Ultraviolet photoelectron spectroscopy (UPS) spectra were collected using a Thermo Fisher Scientific ESCALAB 250XI at a bias of −5 V using a He-Iα (21.22 eV) UV light source. The Kelvin probe force microscopy (KPFM) measurements were carried out in air conditions (Bruker Multimode 8) using frequency-modulation KPFM (FM-KPFM) to probe the contact potential difference (CPD) between the perovskite surface and the tip.
XPS and ToF-SIMS measurements
X-ray photoelectron spectroscopy (XPS) was collected on Thermo Fisher Scientific K-alpha+ using an Al Kα X-ray (1486.68 eV). The time-of-flight secondary ion mass spectrometry (ToF-SIMS) measurements for the complete device were performed on an ION-ToF GmbH instrument (ToF-SIMS 5). A pulsed 30 keV Bi1+ primary ion beam was utilized to generate the secondary ions. The beam current of the Bi1+ primary ion beam was 1 pA. All the ToF-SIMS results were obtained from a 100 μm × 100 μm area on the sample surface. During the sputtering, a 1 keV oxygen sputter beam was applied to remove the surface material. The oxygen sputter beam was applied on a large 300 × 300 μm area with a beam current of 100 nA. The ejected secondary ions were accelerated by a sample bias of 3 kV.
Hole mobility measurement
The hole mobility was measured using an organic thin film transistor (OTFT). We fabricated bottom-gate/bottom-contact (BGBC) OTFT based on D18 organic active layer and adopted the octadecyltrichlorosilane (OTS)-treated Cr/Au as the source-drain electrodes. The field-effect mobility in saturation (VGS − VT < VDS) is calculated from the following equation:
where µ is hole mobility, L is channel length (80 µm), W is channel width (1000 µm), Cj is dielectric capacitance per unit area, IDS is source-drain current, VGS is gate voltage, VDS is drain voltage, and VT is the threshold voltage. The VT can be calculated from the x-axis intercept of the square root of IDS versus VGS line. From Supplementary Fig. 2, the µ was calculated to be 3.8 × 10−2 cm2 V−1 s−1.
Calculation of QFLS
The quasi-Fermi level splitting (QFLS) was calculated based on the PLQY measurements and the following equation58:
where J0,rad is the radiative thermal recombination current density in the dark, kB is the Boltzmann constant, T is the temperature, and JG is the generation current density. The generation current density JG hereby was approximated with the short-circuit current density of the complete solar cell. Similarly, the J0,rad was estimated by integrating the overlap of the external quantum efficiency of the device (EQE) with the black body spectrum ΦBB at 300 K over the energy (Eq. 3).
This resulted in J0,rad values of 3.48 × 10−20 mA cm−2 and 3.62 × 10−20 mA cm−2 for the control and D18 device, respectively.
J SC-V OC technique
JSC-VOC technique is a predecessor of the Suns-VOC technique that was introduced by Wolf and Rauschenbach in 196359. Sinton Suns–VOC tool is a well-established technology to measure the series resistance at MPP of silicon solar cells. The Suns–VOC tool was used to measure the VOC under a series of illumination levels and construct a pseudo-J-V curve that is free of series resistance, as no current flows during the measurement. The series resistance can be derived by comparing the pseudo-J-V curve with the one-sun J-V curve, specifically, by taking the voltage difference (∆V) divided by the current density at MPP condition (Eq. 5).
Using the JSC-VOC technique, we measured the J-V curve at different light intensities to obtain the JSC and VOC pairs of PSCs. These JSC-VOC pairs were then converted into a pseudo-J-V curve using Eq. 644, which reassigns a current value, J, to each measured \({J}_{{{{\rm{sc}}}}}^{{{{\rm{filtered}}}}}\) value in accordance with the principle of superposition and the one-sun JSC of the cell, \({J}_{{{{\rm{sc}}}}}^{1-{{{\rm{sun}}}}}\). Rs at MPP was determined from the voltage shift at JMPP between the one sun J-V curve and pseudo-J-V curve (Fig. 3e).
Fill factor (FF) loss analysis
The FF losses in high-performing PSCs are determined by two main factors, non-radiative recombination and series resistance60. The maximum FF (FFmax) can be empirically calculated according to the following equation:
where n is the ideality factor, KB is the Boltzmann constant, T is temperature, and q is the elementary charge. The ideality factors were extracted from the VOC-light intensity dependent (Supplementary Fig. 28) and calculated to be 1.49 and 1.39 for the control and D18 devices, respectively.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The data that support the findings of this study are available in the following repository: https://doi.org/10.6084/m9.figshare.27264276.
References
Zhou, J. J. et al. Highly efficient and stable perovskite solar cells via a multifunctional hole transporting material. Joule 8, 1691–1706 (2024).
Chen, H. et al. Improved charge extraction in inverted perovskite solar cells with dual-site-binding ligands. Science 384, 189–193 (2024).
Luo, C. et al. Engineering the buried interface in perovskite solar cells via lattice-matched electron transport layer. Nat. Photonics 17, 856–864 (2023).
Tang, H. et al. Reinforcing self-assembly of hole transport molecules for stable inverted perovskite solar cells. Science 383, 1236–1240 (2024).
Shen, Z. C. et al. Efficient and stable perovskite solar cells with regulated depletion region. Nat. Photonics 18, 450–457 (2024).
Azmi, R. et al. Damp heat-stable perovskite solar cells with tailored-dimensionality 2D/3D heterojunctions. Science 376, 73–77 (2022).
Li, C. W. et al. Rational design of lewis base molecules for stable and efficient inverted perovskite solar cells. Science 379, 690–694 (2023).
Magomedov, A. et al. Self‐assembled hole transporting monolayer for highly efficient perovskite solar cells. Adv. Energy Mater. 8, 1801892 (2018).
Shao, Y. H., Xiao, Z. G., Bi, C., Yuan, Y. B. & Huang, J. S. Origin and elimination of photocurrent hysteresis by fullerene passivation in CH3NH3PbI3 planar heterojunction solar cells. Nat. Commun. 5, 5784 (2014).
Bi, E. et al. Diffusion engineering of ions and charge carriers for stable efficient perovskite solar cells. Nat. Commun. 8, 15330 (2017).
Huang, H.-H. et al. Robust unencapsulated perovskite solar cells protected by a fluorinated fullerene electron transporting layer. ACS Energy Lett. 6, 3376–3385 (2021).
Kim, G.-W., Shinde, D. V. & Park, T. Thickness of the hole transport layer in perovskite solar cells: performance versus reproducibility. RSC Adv. 5, 99356–99360 (2015).
Grill, I. et al. Charge transport limitations in perovskite solar cells: the effect of charge extraction layers. ACS Appl. Mater. Interfaces 9, 37655–37661 (2017).
Li, Z. et al. Extrinsic ion migration in perovskite solar cells. Energy Environ. Sci. 10, 1234–1242 (2017).
Guarnera, S. et al. Improving the long-term stability of perovskite solar cells with a porous Al2O3 buffer layer. J. Phys. Chem. Lett. 6, 432–437 (2015).
Kato, Y. et al. Silver iodide formation in methyl ammonium lead iodide perovskite solar cells with silver top electrodes. Adv. Mater. Interfaces 2, 1500195 (2015).
Wang, S., Jiang, Y., Juarez-Perez, EmilioJ., Ono, LuisK. & Qi, Y. Accelerated degradation of methylammonium lead iodide perovskites induced by exposure to iodine vapour. Nat. Energy 2, 16195 (2016).
Shen, Y., Deng, K. M., Chen, Q. H., Gao, G. & Li, L. Crowning lithium ions in hole-transport layer toward stable perovskite solar cells. Adv. Mater. 34, 2200978 (2022).
Zhao, Y. et al. A bilayer conducting polymer structure for planar perovskite solar cells with over 1400 h operational stability at elevated temperatures. Nat. Energy 7, 144–152 (2022).
Du, T. et al. Efficient, stable, and fully printed carbon-electrode perovskite solar cells enabled by hole-transporting bilayers. Joule 7, 1920–1937 (2023).
Yuan, L. et al. A conformally bonded molecular interface retarded iodine migration for durable perovskite solar cells. Energy Environ. Sci. 16, 1597–1609 (2023).
Zheng, K. H. et al. Approaching the fill factor limit in dopant-free hole transporting layer-based all-inorganic perovskite solar cells. ACS Appl. Mater. Interfaces 15, 14748–14759 (2023).
Meng, L. et al. Tailored phase conversion under conjugated polymer enables thermally stable perovskite solar cells with efficiency exceeding 21. J. Am. Chem. Soc. 140, 17255–17262 (2018).
Wang, Y. et al. Density functional theory analysis of structural and electronic properties of orthorhombic perovskite CH3NH3PbI3. Phys. Chem. Chem. Phys. 16, 1424–1429 (2014).
Yin, W.-J., Shi, T. & Yan, Y. Unusual defect physics in CH3NH3PbI3 perovskite solar cell absorber. Appl. Phys. Lett. 104, 063903 (2014).
Agiorgousis, M. L., Sun, Y.-Y., Zeng, H. & Zhang, S. Strong covalency-induced recombination centers in perovskite solar cell material CH3NH3PbI3. J. Am. Chem. Soc. 136, 14570–14575 (2014).
Amat, A. et al. Cation-induced band-gap tuning in organohalide perovskites: interplay of spin-orbit coupling and octahedra tilting. Nano Lett. 14, 3608–3616 (2014).
Diaz, J. J. et al. Analysis of the bonding’s energy in metal-halide perovskites and brief evaluation of meta-GGA functionals TPSS and revTPSS. J. Mater. Sci. 59, 2361–2374 (2024).
Zhong, Y. et al. Inhibition of ion migration for highly efficient and stable perovskite solar cells. Adv. Mater. 35, e2302552 (2023).
Zhao, Y. et al. Mobile-ion-induced degradation of organic hole-selective layers in perovskite solar cells. J. Phys. Chem. C. 121, 14517–14523 (2017).
Besleaga, C. et al. Iodine migration and degradation of perovskite solar cells enhanced by metallic electrodes. J. Phys. Chem. Lett. 7, 5168–5175 (2016).
Wang, T. et al. Transporting holes stably under iodide invasion in efficient perovskite solar cells. Science 377, 1227–1231 (2022).
Yuan, Y. & Huang, J. Ion migration in organometal trihalide perovskite and its impact on photovoltaic efficiency and stability. Acc. Chem. Res. 49, 286–293 (2016).
Li, N., Niu, X., Chen, Q. & Zhou, H. Towards commercialization: the operational stability of perovskite solar cells. Chem. Soc. Rev. 49, 8235–8286 (2020).
Cheng, Y. & Ding, L. Pushing commercialization of perovskite solar cells by improving their intrinsic stability. Energy Environ. Sci. 14, 3233–3255 (2021).
Zai, H., Ma, Y., Chen, Q. & Zhou, H. Ion migration in halide perovskite solar cells: Mechanism, characterization, impact and suppression. J. Energy Chem. 63, 528–549 (2021).
Koushik, D. et al. High-efficiency humidity-stable planar perovskite solar cells based on atomic layer architecture. Energy Environ. Sci. 10, 91–100 (2017).
Zhao, Y. et al. In Situ, cesium modification at interface enhances the stability of perovskite solar cells. ACS Appl. Mater. Interfaces 10, 33205–33213 (2018).
Wang, B. et al. Robust molecular dipole-enabled defect passivation and control of energy-level alignment for high-efficiency perovskite solar cells. Angew. Chem. Int. Ed. 60, 17664–17670 (2021).
Zhang, Z. et al. Surface‐anchored acetylcholine regulates band‐edge states and suppresses ion migration in a 21%‐efficient quadruple‐cation perovskite solar cell. Small 18, 2105184 (2021).
Zai, H. et al. Sandwiched electrode buffer for efficient and stable perovskite solar cells with dual back surface fields. Joule 5, 2148–2163 (2021).
Sutanto, A. A. et al. Dynamical evolution of the 2D/3D interface: a hidden driver behind perovskite solar cell instability. J. Mater. Chem. A 8, 2343–2348 (2020).
Ma, K. et al. Multifunctional conjugated ligand engineering for stable and efficient perovskite solar cells. Adv. Mater. 33, 2100791 (2021).
Yang, G. et al. Stable and low-photovoltage-loss perovskite solar cells by multifunctional passivation. Nat. Photonics 15, 681–689 (2021).
Bi, E. et al. Efficient perovskite solar cell modules with high stability enabled by iodide diffusion barriers. Joule 3, 2748–2760 (2019).
Li, Z. et al. Undoped hole transport layer toward efficient and stable inorganic perovskite solar cells. Adv. Funct. Mater. 33, 2214562 (2023).
Jiang, K. et al. Suppressed recombination loss in organic photovoltaics adopting a planar–mixed heterojunction architecture. Nat. Energy 7, 1076–1086 (2022).
Liu, Q. et al. 18% Efficiency organic solar cells. Sci. Bull. 65, 272–275 (2020).
Wang, Z. et al. Thermodynamic properties and molecular packing explain performance and processing procedures of three D18:NFA organic solar cells. Adv. Mater. 32, e2005386 (2020).
You, S. et al. Radical polymeric p-doping and grain modulation for stable, efficient perovskite solar modules. Science 379, 288–294 (2023).
Kim, S. et al. Relationship between ion migration and interfacial degradation of CH3NH3PbI3 perovskite solar cells under thermal conditions. Sci. Rep. 7, 1200 (2017).
Tan, F. et al. In Situ back-contact passivation improves photovoltage and fill factor in perovskite solar cells. Adv. Mater. 31, 1807435 (2019).
Sun, J. et al. Hybrid block copolymer/perovskite heterointerfaces for efficient solar cells. Adv. Mater. 35, 2206047 (2023).
Wolff, C. M., Caprioglio, P., Stolterfoht, M. & Neher, D. Nonradiative recombination in perovskite solar cells: the role of interfaces. Adv. Mater. 31, 1902762 (2019).
Mundhaas, N. et al. Series resistance measurements of perovskite solar cells using jsc–voc measurements. Sol. RRL 3, 1800378 (2019).
Rombach, F. M., Haque, S. A. & Macdonald, T. J. Lessons learned from spiro-OMeTAD and PTAA in perovskite solar cells. Energy Environ. Sci. 14, 5161–5190 (2021).
Yoo, J. J. et al. Efficient perovskite solar cells via improved carrier management. Nature 590, 587–593 (2021).
Caprioglio, P. et al. On the relation between the open‐circuit voltage and quasi‐fermi level splitting in efficient perovskite solar cells. Adv. Energy Mater. 9, 1901631 (2019).
Wolf, M. & Rauschenbach, H. Series resistance effects on solar cell measurements. Adv. Energy Convers. 3, 455–479 (1963).
Green, M. A. Accuracy of analytical expressions for solar cell fill factors. Sol. Cells 7, 337–340 (1982).
Acknowledgements
We would like to thank the Instrumental Analysis Center of Huaqiao University for providing the various tests. This work was financially supported by the following funding: National Natural Science Foundation of China (22179042, L.X.), National Natural Science Foundation of China (U21A2078, Z.W.), and Natural Science Foundation of Fujian Province (2023J06034, L.X.).
Author information
Authors and Affiliations
Contributions
Z.W. and L.X. supervised the work. Z.W., L.X., and L.S. conceived the idea. K.J. synthesized the D18 polymers and measured the hole mobility of D18. A.K.,-Y.J. supervised the synthesis of D18 polymers. L.S. and P.S. fabricated and characterized the devices. L.Z. participated in device fabrication and certification. J.Q. performed the time-of-flight secondary ion mass spectrometry (ToF-SIMS) measurements. Y.H., F.L., C.T., and J.Y. contributed to improving the devices’ performance. L.S. drafted the manuscript. L.X., Z.W., L.S., and P.S. revised the manuscript. All authors were involved in discussions and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks MOHAMMAD KHAJA Nazeeruddin, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Shen, L., Song, P., Jiang, K. et al. Ultrathin polymer membrane for improved hole extraction and ion blocking in perovskite solar cells. Nat Commun 15, 10908 (2024). https://doi.org/10.1038/s41467-024-55329-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-024-55329-0
This article is cited by
-
Synchronously modulating the strength of chemical and electric field-induced passivation for robust and efficient perovskite photovoltaics
Science China Chemistry (2026)
-
Efficiency improvement in silicon and perovskite solar cells through nanofluid cooling using citrate and PVP stabilized silver nanoparticles
Scientific Reports (2025)