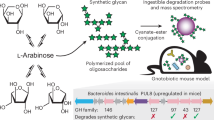
Abstract
Commensal bacteria affect host health by producing various metabolites from dietary carbohydrates via bacterial glycometabolism; however, the underlying mechanism of action remains unclear. Here, we identified Streptococcus salivarius as a unique anti-obesity commensal bacterium. We found that S. salivarius may prevent host obesity caused by excess sucrose intake via the exopolysaccharide (EPS) –short-chain fatty acid (SCFA) –carbohydrate metabolic axis in male mice. Healthy human donor-derived S. salivarius produced high EPS levels from sucrose but not from other sugars. S. salivarius abundance was significantly decreased in human donors with obesity compared with that in healthy donors, and the EPS–SCFA bacterial carbohydrate metabolic process was attenuated. Our findings reveal an important mechanism by which host–commensal interactions in glycometabolism affect energy regulation, suggesting an approach for preventing lifestyle-related diseases via prebiotics and probiotics by targeting bacteria and EPS metabolites.
Similar content being viewed by others
Introduction
Although diet is the most important factor for daily nutrient acquisition, the dysregulation of energy homeostasis due to excessive dietary intake, particularly high fat and sugar intake, leads to obesity1,2. Sucrose (table sugar), glucose (dextrose), and fructose (fruit sugar) are simple saccharides. Of the sugars consumed daily3,4, sucrose and glucose are the most common. As sucrose intake increases in Western countries, sucrose-rich diets may be associated with rising health problems such as obesity and diabetes.
Although bacteria also utilise these sugars as an energy source, their metabolic pathway differs from that of humans5,6. After glycolysis, anaerobic bacteria convert pyruvate to lactate and other organic acids such as short-chain fatty acids (SCFAs; acetate, propionate, and butyrate). Gut microbes produce these end products through sugar metabolism in environments where oxygen is limited5,6. They produce SCFAs from fermentable fibres, which are indigestible polysaccharides that are not absorbed by the small intestine because host enzymes cannot digest them7,8. SCFAs act as energy sources for the host and as signalling factors via host G protein-coupled receptors GPR41 and GPR43, improving host homeostasis by acting on the endocrine systems9,10,11. GPR41 influences host metabolic functions, enhances sympathetic activity, and promotes gut hormone secretion11,12,13, whereas GPR43 suppresses fat accumulation and promotes gut hormone secretion11,12,14.
Sugars are anabolized into polysaccharides during glycometabolism. Storage polysaccharides such as glycogen and starch are carbohydrates used to store and provide energy. In animals, glycogen is primarily stored in the liver and muscles3, resulting in sudden increases in energy requirements. Additionally, starch, found in grains, potatoes, and legumes, is the main form of sugar stored in plants5. These carbohydrates serve as energy sources and are broken down into glucose during digestion. Bacteria also produce different types of storage polysaccharides, such as levans and dextrans15,16,17, depending on the type of bacteria and environmental conditions. We recently reported that prebiotics associated with the exopolysaccharide (EPS) produced by Leuconostoc mesenteroides provide substantial metabolic benefits to the host18,19. This polysaccharide is indigestible because its glycosidic linkages cannot be cleaved by host amylase. Furthermore, fermented foods, such as pickles, kimchi, and sauerkraut, are produced by the fermentative action of L. mesenteroides, a lactic acid bacterium20,21.
These findings suggest that some gut microbes may produce indigestible polysaccharides from sugars and contribute to host metabolic benefits. Therefore, in this study, we sought high-EPS-producing gut microbes in humans and investigated the relationship between host sugar intake and the prebiotic effects of gut microbe-produced EPS, as well as the molecular mechanism underlying the effect of microbial metabolites on host health.
Results
Isolation of EPS-producing human commensal bacteria S. salivarius
We first screened EPS-producing bacteria using ropy bacterial colonies as an indicator22 in human faeces (472 donors) (Supplementary Fig. 1). These samples were cultured on de Man, Rogosa, and Sharpe (MRS) agar with different sugar sources (Fig. 1a). Ropy bacterial colonies (47 donors) were then observed in the cultures of human faeces with sucrose alone, excluding other sugar sources. However, such colonies were absent from the cultures of mouse faeces with sucrose (Fig. 1a). The bacterial colonies were identified as Weissella cibaria (19 donors), L. mesenteroides (14 donors), Streptococcus salivarius (six donors), W. confusa (five donors), and L. lactis (three donors) (Fig. 1b). These are known as lactic acid bacteria commonly found in humans and fermented foods23,24. Furthermore, we investigated the relationship between the presence of bacteria in human faeces and the body mass index (BMI) of donors (Fig. 1c). Remarkably, only S. salivarius was sufficiently detected in all donors, and its occupancy showed an inverse correlation with obesity (BMI ≥ 30). Therefore, we focused on S. salivarius.
a High-EPS-producing bacteria using ropy bacterial colonies as an indicator in human and mouse faeces cultured in MRS medium containing 15% fructose, galactose, glucose, lactose, maltose, and sucrose. Red arrowheads indicate EPS production. b Types of EPS-producing bacteria isolated from 47 human faecal samples. c Correlation between high-EPS-producing bacterial abundance in human faeces and donor body mass index (BMI). (n = 132, 48 independent experiments). Two-tailed Mann–Whitney U test was used for the statistical analysis. d Structural characterisation was performed using proton nuclear magnetic resonance (1H NMR) spectroscopy. e Growth curves of EPS biosynthesis and optical density at 600 nm (OD600) (n = 3 independent experiments). f, g The KEGG pathway enrichment of EPS synthesis pathways and expression of putative levansucrase and glycosyltransferases mRNAs in MRS medium containing sucrose or glucose during bacterial culture for 10 h were determined using RNA-seq (n = 4 independent experiments). h Expression of putative levansucrase and glycosyltransferases mRNAs in MRS medium containing sucrose or glucose during bacterial culture for 10 h was measured using RT-qPCR (n = 4 independent experiments). Two-tailed Mann–Whitney U test was used for the statistical analysis. Results are presented as the mean ± standard error of the mean (SE). Source data are provided as a Source Data file.
The isolated S. salivarius (a Gram-positive, spherical, facultative anaerobe) produced EPS on MRS agar with sucrose (Supplementary Fig. 2a). S. salivarius-produced EPS (SsEPS) was purified by ethanol precipitation/dialysis and analysed using 1H NMR spectroscopy. The purified SsEPS consisted of levan (fructan, with linear structures of fructose linked by β-2,6-glycosidic bonds) and dextran (α-glucan, with main-chain glucose monomers linked by α-1,6-glycosidic bonds and branched by α-1,3-glycosidic side chains) (Fig. 1d and Supplementary Fig. 2b–i). In MRS broth containing 15% sucrose, S. salivarius produced large amounts of EPS (13 mg/mL). However, when the MRS medium contained 15% glucose, 15% fructose, or 7.5% glucose +7.5% fructose, EPS production failed despite bacterial proliferation (Fig. 1e). Upon RNA sequencing, Kyoto Encyclopedia of Genes and Genome (KEGG) Orthology analysis showed that the EPS synthesis pathway was enriched in sucrose-supplemented S. salivarius cultures (Fig. 1f and Supplementary Fig. 3). Furthermore, 14 putative glycosyltransferase-encoding genes were extracted by comparing their mRNA expression between sucrose- and glucose-supplemented cultures (Fig. 1g). The mRNA expression of the five putative glycosyltransferase genes significantly increased along with the SsEPS yield in the medium containing 15% sucrose, but not in the medium containing glucose (Fig. 1h). Additionally, levansucrase and glycosyltransferase, RS02300 and RS07295, respectively were highly expressed after sucrose supplementation. Therefore, SsEPS may be synthesised by the putative levansucrase and glycosyltransferase genes. Thus, the human commensal bacterium S. salivarius produces large amounts of EPS in the form of levans and dextrans.
We investigated the gut microbial composition between donors with and without obesity (Supplementary Fig. 4). Besides the occupancy of S. salivarius, the levels of SCFAs and EPS in the faeces of lean donors were also significantly higher than those in donors with obesity (Fig. 2a). Shotgun metagenomic sequencing data showed that obesity was associated with the carbohydrate metabolic pathway. Correlation analysis revealed a strong correlation between EPS hydrolase, glycolysis, and SCFA production (Fig. 2b). Moreover, glycolytic pathway analysis showed that obesity attenuated EPS and glycolysis pathways (Fig. 2c). Therefore, human obesity is associated with a decrease in S. salivarius and the attenuation of EPS and SCFA synthesis cascades.
a Spearman’s rank correlation between faecal levels of total short-chain fatty acids (SCFAs) such as acetate, propionate, and butyrate, exopolysaccharides (EPS) and donor body mass index (BMI) in human faeces (n = 132, 48 independent experiments). Two-tailed Mann–Whitney U test was used for the statistical analysis. b Results of correlation analysis among EPS hydrolase, glycolysis, and SCFA production between lean donors and donors with obesity (n = 26 independent experiments). c Comparison of polysaccharide synthesis, polysaccharide decomposition, glycolytic pathway enrichment, and SCFA synthesis between lean donors and donors with obesity in shotgun metagenomic sequencing analysis (n = 26 independent experiments). ***P < 0.001, **P < 0.01, *P < 0.05 Two-tailed Mann–Whitney U test was used for the statistical analysis. Results are presented as the mean ± standard error of the mean (SE). Source data are provided as a Source Data file.
Improvement of host metabolic functions by SsEPS
Subsequently, we investigated the bacteria associated with SCFA production from SsEPS using in vitro gut microbe monoculture screening25. Among the 47 gut microbial strains tested, the Bacteroides species, B. ovatus and B. thetaiotaomicron and the Bacteroidales S24-7 group members Muribaculum intestinale, Paramuribaculum intestinale, and Duncaniella muris efficiently produced SCFAs after 0.3% SsEPS addition. In contrast, other gut microbes did not produce SCFAs (Fig. 3a). To determine whether SsEPS is an indigestible polysaccharide, we examined intestinal SCFA levels after SsEPS intake. The levels of faecal and plasma SCFAs (acetate, propionate, and butyrate) were significantly higher in mice fed a high-fat diet (HFD) supplemented with SsEPS than in those fed an HFD supplemented with cellulose (Fig. 3b). Thus, in the host intestine, the consumption of SsEPS promotes the production of SCFAs by gut microbes.
a Bacterial SCFA levels in the culture supernatants of each gut bacterium (n = 6 independent experiments). **P < 0.01. Two-tailed Mann–Whitney U test was used for the statistical analysis. b–e C57BL/6J and Gpr41Gpr43 double-deficient mice were fed an HFD supplemented with 10% cellulose or SsEPS for 12 weeks. Faecal and plasma SCFA levels were measured using GC-MS (b), changes in body and tissue weight (c) Epi, epididymal; peri, perirenal; sub, subcutaneous; WAT, white adipose tissue. (n = 9, 10 independent experiments). **P < 0.01, *P < 0.05. Two-tailed Mann–Whitney U test was used for the statistical analysis. Blood glucose, plasma non-esterified fatty acid (NEFAs) (d), GLP-1 (e), and PYY levels (f) were measured at the end of the experimental period (n = 9, 10 independent experiments). Two-tailed Mann–Whitney U test was used for the statistical analysis. NS, not significant. g Daily food intake at 12 weeks of age (n = 5 independent experiments). Two-tailed Mann–Whitney U test was used for the statistical analysis. h Expression of Ucp 1 mRNA (n = 10 independent experiments) was measured by RT-qPCR in subcutaneous WAT. Two-tailed Mann–Whitney U test was used for the statistical analysis. i, j Following 24 h of fasting, the mice were fed 0.2 g AIN-93G, containing 50% cellulose or 50% SsEPS, and an intraperitoneal glucose tolerance test was performed 1 h after feeding. Wild-type (n = 10 independent experiments), Gpr41Gpr43 double-deficient (n = 7, 8 independent experiments), ICR (n = 8, 9 independent experiments), and GF-ICR (n = 8 independent experiments) mice were used. **P < 0.01, *P < 0.05 (Mann–Whitney U test). Plasma insulin levels were measured 15 min after intraperitoneal glucose administration. Wild-type (n = 8, 9 independent experiments), Gpr41Gpr43 double-deficient (n = 7, 8 independent experiments), ICR and GF-ICR (n = 8 independent experiments) mice were used. Dunn’s post-hoc test was used for the statistical analysis. Results are presented as means ± standard error of the mean (SE). Source data are provided as a Source Data file.
We investigated the effects of SsEPS on host energy homeostasis in HFD-induced obese mice. Four-week-old mice were fed an HFD supplemented with either SsEPS or cellulose as non-fermented fibre for 12 weeks. The body weight of the mice fed with SsEPS was markedly lower than that of control mice fed with cellulose during growth (Fig. 3c). Furthermore, the fat mass of the white adipose tissue (WAT) of SsEPS-fed mice was significantly lower than that of the control mice at 16 weeks of age (Fig. 3c). Blood glucose, plasma triglyceride (TG), non-esterified fatty acid (NEFA), and total cholesterol levels in SsEPS-fed mice were significantly lower than those in the cellulose-fed control mice (Fig. 3d and Supplementary Fig. 5a). HFD-induced insulin resistance and impaired glucose tolerance, as assessed by an insulin tolerance test and glucose tolerance test (GTT), respectively, were significantly attenuated in SsEPS-fed mice compared to those in the cellulose-fed control mice (Supplementary Fig. 5b). Additionally, plasma insulin levels were significantly lower, and plasma glucagon like peptide-1 (GLP-1) and peptide YY (PYY) levels, as orexigenic gut hormones11, were significantly higher in SsEPS-fed mice than in cellulose-fed control mice (Fig. 3e, f and Supplementary Fig. 5c). Moreover, the resulting food intake was significantly lower in SsEPS-fed mice than in cellulose-fed control mice (Fig. 3g). GLP-1 was reported to promote brown adipose tissue thermogenesis and the browning of white adipocytes26. The expression of uncoupling protein 1 (UCP1), which is the main factor responsible for thermogenesis27, in subcutaneous WAT was significantly higher in SsEPS-fed mice than in cellulose-fed control mice (Fig. 3h). However, these SsEPS-induced effects, such as the suppression of body and fat weight gain (Fig. 3c), reduced hyperglycemia and hyperlipidemia (Fig. 3d and Supplementary Fig. 5a), increased plasma GLP-1 levels (Fig. 3e), and improved insulin sensitivity (Supplementary Fig. 5b), were sufficiently attenuated in Gpr41Gpr43 double-deficient mice. Dietary fibre-derived gut microbial SCFAs promote the secretion of gut hormone such as GLP-1 through GPR41 and GPR43, thereby maintaining energy homeostasis and glucose metabolism11. Therefore, continuous SsEPS intake improves energy homeostasis.
Additionally, we examined the effects of SsEPS-derived gut microbial SCFAs on glucose homeostasis in the host using the GTT. The administration of SsEPS significantly attenuated the increase in blood glucose levels after glucose administration compared to that in control mice; this effect was abolished in Gpr41Gpr43 double-deficient mice (Fig. 3i). Moreover, the plasma levels of insulin and incretin GLP-1 were higher in SsEPS-administered mice than in control mice after glucose administration. These effects were abolished in Gpr41Gpr43 double-deficient mice (Fig. 3i and Supplementary Fig. 5d). Furthermore, under germ-free (GF) conditions, the SsEPS-induced inhibition of blood glucose elevation and the increase in plasma insulin and GLP-1 levels were abolished (Fig. 3j and Supplementary Fig. 5d). Therefore, SsEPS supplementation improves glucose homeostasis in the host by producing gut microbial SCFAs.
Change in host gut microbiota by SsEPS
Continuous SsEPS intake markedly increased SCFA levels in the faeces and plasma (Fig. 3b). Given the pivotal role of SCFAs in the beneficial effects of SsEPS on the host, we investigated the changes in the gut microbial composition mediated by SsEPS and identified the gut microbes related to SCFA production. 16S rRNA amplicon sequencing showed that SsEPS supplementation altered the relative abundances of the major phyla in the gut microbiota (Fig. 4a). Notably, the abundances of Bacteroidota and Verrucomicrobiota were significantly increased, while that of Firmicutes was significantly decreased in SsEPS-fed mice (Fig. 4a). The effect of SsEPS on the gut microbiome was confirmed by the hierarchical clustering of individual families (Fig. 4b). These changes in the gut microbiota after SsEPS intake were associated with the abundance of several families of gut microbes (Fig. 4b). Subsequently, a correlation analysis between gut microbes and SCFAs performed by comparing these gut microbes at the genus level (Fig. 4c) showed high correlation coefficients for Bacteroidales S24-7, including Muribaculum, Paramuribaculum, Duncaniella, and Bacteroides (Fig. 4d). In addition, the abundance of five species that efficiently produced SCFAs during in vitro gut microbe monoculture (Fig. 3a) significantly increased after SsEPS supplementation (Fig. 4e). As shown by the shotgun metagenomic sequencing data, SsEPS uptake was associated with the carbohydrate metabolic pathway according to Gene Ontology (GO) enrichment analysis (Supplementary Fig. 6). Furthermore, glycolytic pathway analysis showed that SsEPS intake increased levan and glucan degradation and SCFA synthesis (Fig. 4f). Thus, SsEPS intake contributes to the production of SCFAs via the polysaccharide catabolic cascade of members of the Bacteroidales S24-7 group and the genus Bacteroides.
Gut microbial composition was evaluated to perform principal coordinate analysis and determine the relative abundance at the phylum level (a), with a heatmap of the bacterial domain at the family level (b) and genus level (c) (n = 10 independent experiments). Two-tailed Mann–Whitney U test was used for the statistical analysis. d Spearman’s rank correlation between the levels of the main contributing bacterial genera and faecal short-chain fatty acids (SCFAs) in high-fat diet (HFD)-fed mice supplemented with cellulose versus HFD-fed mice supplemented with SsEPS. e SsEPS-utilising Bacteroides and Bacteroidales S24-7 group species were detected by qPCR (n = 10 independent experiments). Two-tailed Mann–Whitney U test was used for the statistical analysis. f EPS degradation, SCFA synthesis, and glycolysis pathways were compared between cellulose- and SsEPS-fed mice in shotgun metagenomic sequencing analysis (n = 5 independent experiments). Two-tailed Mann–Whitney U test was used for the statistical analysis. Results are presented as means ± standard error of the mean (SE). Source data are provided as a Source Data file.
Amelioration of sucrose-induced metabolic dysfunction by S. salivarius
S. salivarius and SsEPS were detected in human faeces but not in mouse faeces (Fig. 1a). While Bacteroides and Bacteroidales S24-7 groups are SCFA producers upon SsEPS intake in mice, the Bacteroidales S24-7 group is not dominant in humans28. Therefore, we generated an EPS-non-producing S. salivarius strain (Supplementary Fig 7a–f) and performed a co-transfer experiment with B. ovatus and B. thetaiotaomicron, confirming their intestinal colonisation (Supplementary Fig. 8a–c). We found that faecal EPS levels were sufficiently higher in S. salivarius-colonised and S. salivarius- and Bacteroides-co-colonised mice but not in Bacteroides-colonised mice after drinking 20% sucrose than in EPS-non-producing S. salivarius-colonised and GF mice (Fig. 5a and Supplementary Fig. 8d). Faecal acetate and propionate levels were markedly higher only in S. salivarius and Bacteroides-co-colonised mice. Conversely, butyrate levels were similar between these groups (Fig. 5b). Butyrate production by SsEPS intake (Fig. 3b) was hardly reflected in gnotobiotic experiments, possibly because of the interaction of other gut microbes besides Bacteroides. In contrast, these changes were not exhibited in mice colonised by EPS-non-producing S. salivarius (Supplementary Fig. 8d). Thus, S. salivarius produces EPS, and Bacteroides produces SCFAs from SsEPS in the gut.
Germ-free (GF) and colonised mice were generated and consumed sterilised water containing 20% sucrose. a Faecal EPS was measured by HPLC (n = 10, 10, 8, 10, 10, 8 independent experiments). Dunn’s post-hoc test was used statistical analysis. b Faecal SCFA levels were measured by GC/MS (n = 10, 9, 8, 10, 10, 8 independent experiments). Dunn’s post-hoc test was used for the statistical analysis. c After colonisation, an intraperitoneal GTT was performed (n = 8, 7, 10, 9, 6, 6 independent experiments). Dunn’s post-hoc test was used for the statistical analysis. d Plasma insulin (left; n = 8, 9, 10, 7, 9, 7, 7 independent experiments) and GLP-1 (right; n = 7, 8, 8, 7, 9, 7, 7 independent experiments) levels were measured 15 min after intraperitoneal glucose administration Dunn’s post-hoc test was used for the statistical analysis. e–k After colonisation, the mice were fed an AIN-93G diet or high-fat diet (HFD) for 9 weeks. e Experimental scheme for the gnotobiotic analysis. Changes in body and tissue weights (f) and blood glucose (g) (n = 10, 10, 8, 8, 8, 8 independent experiments) under an AIN-93G diet feeding with 20% sucrose drinking water. **P < 0.01, * P < 0.05, compared between Bo+Bt and Ss+Bo+Bt mice. ## P < 0.01, # P < 0.05, compared between Ss+Bo+Bt and Ss (Mut)+Bo+Bt mice. Dunn’s post-hoc test was used for the statistical analysis. h Plasma GLP-1 (n = 7, 7, 8, 8, 8, 8 independent experiments) levels were measured at the end of the experimental period. Dunn’s post-hoc test was used for the statistical analysis. Changes in body and tissue weights under HFD feeding (i), blood glucose ( j ) and plasma GLP-1 and insulin (k) levels were measured at the end of the experimental period (n = 5, 8, 8, 9, 9 independent experiments). Dunn’s post-hoc test was used for the statistical analysis. Results are presented as means ± standard error of the mean (SE). Source data are provided as a Source Data file.
Two weeks after colonisation, glucose clearance, as assessed using an intraperitoneal GTT (ipGTT) in S. salivarius- and Bacteroides-co-colonised mice, notably improved between co-colonised groups (Fig. 5c). Plasma insulin and GLP-1 levels after glucose administration were significantly higher in S. salivarius and Bacteroides-co-colonised mice than in S. salivarius or Bacteroides-colonised mice (Fig. 5d). Although plasma GLP-1 levels in GF mice (~10 pM)29,30 were elevated, GLP-1 levels under this GF condition (Fig. 5d) were even higher (~20 pM) than those in the general GF condition. Prolonged sucrose consumption may influence basal GLP-1 levels, and this may mean that differences in host sugar sensitivity also influence GLP-1 secretion independently from commensal bacteria, although the precise mechanism is still unclear. On the other hand, these effects were not observed in mice colonised by EPS-non-producing S. salivarius and Bacteroides-colonised mice (Fig. 5c, d). Moreover, we examined the effects of S. salivarius and Bacteroides co-colonisation on host energy homeostasis in a sucrose-treated or HFD-induced obese mouse model (Fig. 5e). The body weight of S. salivarius and Bacteroides-co-colonised mice was markedly lower than that of other colonised mice during growth (Fig. 5f). Additionally, the WAT fat mass was notably lower in S. salivarius and Bacteroides-co-colonised mice than in the EPS-non-producing S. salivarius and Bacteroides-co-colonised mice at 16 weeks of age (Fig. 5f). The blood glucose, plasma TG, and NEFA levels of S. salivarius and Bacteroides-co-colonised mice were markedly lower than those of EPS-non-producing S. salivarius and Bacteroides-co-colonised mice (Fig. 5g and Supplementary Fig. 8e). Furthermore, plasma GLP-1 and insulin levels were significantly higher in S. salivarius and Bacteroides-co-colonised mice than in EPS-non-producing S. salivarius and Bacteroides-co-colonised mice (Fig. 5h and Supplementary Fig. 8f). Additionally, under HFD treatment, both body weight and WAT fat mass, blood glucose levels, and plasma insulin levels were significantly lower, and plasma GLP-1 levels were significantly higher, in S. salivarius and Bacteroides-co-colonised mice than in EPS-non-producing S. salivarius and Bacteroides-co-colonised mice (Fig. 5i–k, and Supplementary Fig. 8g, h). Conversely, among the glucose- or fructose-treated mice, S. salivarius-colonised mice did not show an increase in faecal EPS levels (Supplementary Fig. 8i). Therefore, S. salivarius and Bacteroides-co-colonisation improved the metabolic state of the host upon sucrose intake.
Finally, we investigated the effects of S. salivarius on host energy homeostasis using a mouse model with a gut microbiome culture-collection from human faeces. In this experiment, 7-week-old mice colonised with S. salivarius-dominant [Ss (+)] or S. salivarius-non-dominant human gut microbiota [Ss (-)] were fed HFDs containing a sugar type (sucrose, glucose, or fructose) for 9 weeks (Fig. 6a and Supplementary Fig. 9a–c). In cultured faecal microbiota transplantation (FMT) experiments, faecal SCFAs and EPS levels of [Ss (+)]-colonised mice with sucrose replacement were significantly higher than those of [Ss (-)]-colonised mice with sucrose replacement, and also higher than those of mice with glucose or fructose replacement (Fig. 6b, c). The number of S. salivarius, B. ovatus and B. thetaiotaomicron was also sufficiently increased in [Ss (+)]-colonised mice with sucrose replacement than in [Ss (-)]-colonised mice with sucrose, glucose, or fructose replacement (Fig. 6d). The body weight of [Ss (+)]-colonised mice treated with sucrose was markedly lower than that of [Ss (-)]-colonised mice treated with sucrose during growth, whereas it was similar between [Ss (+)]- and [Ss (-)]-colonised mice replaced with glucose or fructose (Fig. 6e). Additionally, WAT fat mass was significantly lower in [Ss (+)]-colonised mice with sucrose replacement than in [Ss (-)]-colonised mice with sucrose replacement, but not in those with glucose or fructose replacement, at 16 weeks of age (Fig. 6f). The blood glucose levels of [Ss (+)]-colonised mice with sucrose replacement were significantly lower than those of [Ss (-)]-colonised mice with sucrose replacement but not those of glucose- or fructose-treated colonised mice (Fig. 6g). Furthermore, plasma GLP-1 levels were significantly higher in [Ss (+)]-colonised mice treated with sucrose replacement than in [Ss (-)]-colonised mice treated with sucrose (Fig. 6h). Thus, S. salivarius-dominant human gut microbiota efficiently ameliorated sucrose-induced metabolic dysfunction.
a–h S. salivarius-dominant [Ss (+)] or -nondominant human gut microbiota culture colonised [Ss (-)] mice were generated from GF mice through transplantation with human faecal culture solution and fed a high-fat diet supplemented with sugars (sucrose, glucose, or fructose). a Experimental scheme for faecal microbiota transplantation experiment. Faecal total SCFAs (b) and EPS (c) were measured by GC/MS and HPLC. (n = 7, 7, 5, 7, 5, 7 independent experiments). Two-tailed Mann–Whitney U test was used for the statistical analysis. d Faecal S. salivarius, B. ovatus, and B. thetaiotaomicron were detected by qPCR (n = 7, 7, 5, 7, 5, 7 independent experiments). Two-tailed Mann–Whitney U test was used for the statistical analysis. e, f Changes in body and tissue weight (n = 7, 7, 5, 7, 5, 7 independent experiments). Two-tailed Mann–Whitney U test was used for the statistical analysis. Blood glucose (g) and plasma GLP-1 (h) levels were measured at the end of the experimental period (n = 7, 7, 5, 7, 5, 7 independent experiments). Two-tailed Mann–Whitney U test was used for the statistical analysis. Results are presented as means ± standard error of the mean (SE). Source data are provided as a Source Data file.
Discussion
Commensal bacteria affect host energy homeostasis by producing various metabolites from host carbohydrate intake via glycometabolism. However, its exact mechanism of action remains unclear. In this study, we found that gut microbes prevent host obesity through excess dietary sucrose intake via the exopolysaccharide (EPS)–SCFA–carbohydrate metabolism axis and identified S. salivarius as a unique anti-obesity commensal bacterium (Fig. 7).
Gut microbes prevent host obesity through excess dietary sucrose intake via the exopolysaccharide (EPS)–short-chain fatty acid (SCFA)–carbohydrate metabolism axis and identified S. salivarius as a unique anti-obesity commensal bacterium.
In the first screening, we identified commensal S. salivarius as a high-EPS-producing bacterium in human donors, as all other high-EPS-producing bacteria were mainly detected in fermented foods23,24. Additionally, while other human gut microbes may produce different EPS, S. salivarius, being the dominant EPS-producing bacterium in the gut microbiota, can produce large amounts of EPS from sucrose, suggesting that host sucrose intake affects the production of SCFAs via SsEPS. Although our study demonstrated that SsEPS was metabolised to SCFAs by gut microbes, thereby affecting host metabolic conditions, it is possible that EPS itself directly affects host physiological functions or that SCFAs affect the host in a receptor-independent manner, such as through de novo metabolic function via SCFA transporters and histone deacetylase inhibition31,32. Moreover, differences in S. salivarius strains may affect the EPS structure and production. Our findings indicate an inverse correlation between S. salivarius occupancy and obesity in a Japanese population. However, further research is necessary to clarify the contribution of S. salivarius to the human gut microbiota, considering that race, region, dietary habits, and lifestyle remarkably influence microbial composition. Continued investigation is required to clarify the relationship between human homeostasis and microbial EPS production, particularly the role of S. salivarius, which uniquely synthesises EPS from sucrose but not from glucose or fructose.
As a facultatively anaerobic lactic acid bacterium, S. salivarius preferentially inhabits the small intestine, in contrary to most other gut microbes, which inhabit the colon. This preference is because sucrose, a disaccharide composed of glucose and fructose, is digested by host sucrase into its constituent monosaccharides, which are absorbed in the small intestine. Therefore, S. salivarius competes with host sucrose digestion in the small intestine and inhibits the absorption of monosaccharides by the host. Furthermore, SsEPS, an indigestible polysaccharide produced from sucrose by S. salivarius, is not digested by host enzymes; Bacteroides species utilise it to produce SCFAs in the gut. The produced SCFAs contribute to the metabolic health of the host. Therefore, S. salivarius and EPS may partly contribute to metabolic improvement and high SCFA production by α-glucosidase inhibitors33,34. On the other hand, S. salivarius is also a commensal bacterium abundantly found in the oral cavity. Some reports indicate that the use of proton pump inhibitors may promote the intestinal colonisation of oral bacteria, including S. salivarius35, and this is associated with gut dysbiosis and metabolic disorders, particularly liver-related disease36,37 rather than leading to metabolic improvements. However, it is important to note that despite this finding, the presence of S. salivarius itself has not been definitively identified as the risk factor for liver disease. Additionally, it remains unclear whether the presence of S. salivarius is a cause or effect of such conditions. Thus, the metabolic benefits observed in our studies attributed to S. salivarius may still occur in this context. Nevertheless, regarding the use of S. salivarius as a probiotic, further investigation into its potential adverse effects, beyond EPS-mediated beneficial actions, is warranted.
S. salivarius may serve as a useful biomarker for resistance to obesity induced by high sugar (sucrose) intake, based on our findings, which have identified this commensal bacterium as possessing anti-obesity properties through the analysis of human donor samples. Our experimental results indicated a trend toward lower S. salivarius prevalence in donors with obesity. This finding suggests that individuals with lower levels of S. salivarius may be less resistant to sucrose-induced obesity. Alternatively, it is possible that environmental factors disrupt gut microbiota homeostasis, inhibiting the growth of S. salivarius and thereby contributing to obesity. Consequently, strategies to increase the proportion of S. salivarius in the gut—such as those using probiotics or prebiotics—may offer a new therapeutic approach to prevent sucrose-induced obesity.
Additionally, S. salivarius produces substantial amounts of EPS under high-sucrose conditions. This EPS production mediates the beneficial effects observed in obese mice fed a sucrose-replaced HFD and under sucrose imbibement, suggesting that S. salivarius acts as a probiotic indirectly through its prebiotic EPS production. However, our FMT experiments used a gut microbiome culture from human faeces that may not completely reflect the human gut microbiome. Additionally, our gnotobiotic experiments cannot partially deny the species-unspecified effect of host body weight gain reduction. Therefore, further studies are needed to clarify the relationship between EPS production and host metabolic contribution by gut microbiota. Although some reports demonstrate metabolic detriments of fructose intake38,39, such effects were not clearly observed in our sugar replacement experiments when compared with those of glucose and sucrose replacement. However, studies investigating fructose-induced metabolic dysregulation generally use high-sugar intake models. Some studies using normal fructose levels, including the present study report minimal effects of fructose40,41, suggesting that the moderate amount of this sugar used in our experiments, as opposed to the high levels used in other studies, might explain the lack of observed detriment effects of fructose intake on host metabolism. Additionally, under induced metabolic abnormalities through a high-sucrose diet, the metabolic dysfunctions typically observed with fructose might be mitigated. In particular, because the fructose component of sucrose is converted into indigestible polysaccharides, such as fructan, by S. salivarius, the detrimental metabolic effects of fructose produced from sucrose by host digestion may be bypassed. Consequently, S. salivarius may represent a novel synbiotic that combines both its probiotic and prebiotic properties, particularly in the context of typical human sucrose consumption. However, our research on the benefits of EPS has mainly been conducted in murine models, and further studies are needed to establish their relevance to human physiology.
In the present study, we demonstrated that commensal bacteria selectively confer obesity tolerance to their hosts through bacterial glycometabolism. Indigestible polysaccharides play a critical role in regulating the host gut environment and homeostasis by modulating gut microbiota5,7,10. These findings suggest a fundamental mechanism underlying the interplay among diet, host, and commensal bacteria for energy homeostasis via host and commensal glycometabolism and gut microbial EPS production. Additionally, they contribute to the development of breakthrough approaches to combat the effects of high-sugar intake in modern dietary habits and prevent lifestyle diseases such as obesity by promoting S. salivarius colonisation via probiotic functional foods or dietary supplements and tailoring the prebiotic use of EPS as a dietary fibre. EPS has diverse polysaccharide structures, monosaccharide components, main-chain lengths, and branching22,42. Moreover, constructing a whole-gut-microbiota metabolic pathway map to identify and analyse polysaccharide-synthesising enzymes beyond SsEPS, such as glycosyltransferases and levansucrase, may elucidate bacterial groups capable of producing several EPS species, which could serve as more robust biomarkers than S. salivarius alone. Furthermore, using metagenomic analysis to identify these groups under similar environmental conditions, we could pinpoint microbial clusters with specific co-occurring genes for specific EPS production. Analysing the association between the expression of these genes and EPS production would enable us to derive novel and robust biomarkers for myriad host disorders, promoting tailored approaches to lifestyle diseases.
Methods
Human faecal sample collection
Study participants were recruited between 2017 and 2022, all experimental procedures involving human faecal sample collection were performed according to the protocols approved by the Ethics Committee of Kyoto University (permit number: R2875-4), Keio University (permit number: 20210021), Kobe University (permit number: B210124), Kyoto Medical Center (permit number: 20-074), Tokyo University of Agriculture and Technology (permit number: 210704-2846) and Fukujuji Hospital (permit number: 21016) and were conducted in accordance with their guidelines. The volunteers included both male and female Japanese individuals aged 20–80 years. The exclusion criteria were as follows: participants with a BMI below 18.5 or above 60 kg m–2; those who regularly took proton pump inhibitor medications; those with diabetes or hyperlipidemia; those who used antibiotics within 2 weeks; and those who consumed probiotic supplements, including milk, yogurt, and fermented food before sample collection. All the participants involved in this study provided written informed consent. The participants collected their stool sample without any restrictions, such as fasting, and submitted within 3 days. Physical measurements, including body weight, height and disease information were obtained through health research survey and medical records. Faecal samples were collected using a stool collection tube and stored at −80 °C until preparation and analysis.
Faecal sample culture conditions
Human and wild-type mouse faecal samples were cultured on MRS agar (Difco Laboratories Inc., Detroit, MI, USA) or MRS agar containing 15% fructose, galactose, glucose, lactose, maltose, and sucrose at 30 °C for 48 h under anaerobic conditions. EPS-producing ropy colonies on MRS agar containing 15% sucrose were selected and underwent 16S ribosomal RNA (rRNA) gene amplification using the primers 27 F (5′-AGAGTTTGATCCTGGCTCAG-3′) and 1492 R (5′-GGTTACCTTGTTACGACTT-3′). The PCR products were purified using an UltraClean PCR Clean-Up Kit (MO BIO Laboratories, San Diego, CA, USA) and directly sequenced using a Big Dye Terminator Cycle Sequencing Kit ver. 3.1 (Applied Biosystems, Foster City, CA, USA) and an ABI 3730xl DNA analyser system (Applied Biosystems). The isolated strains shared more than 98% similarity in their 16S rRNA gene sequences.
Bacterial culture
The cultivation of S. salivarius isolated human faecal sample in MRS medium containing 15% sucrose, 15% glucose, 15% fructose, and 7.5% glucose +7.5% fructose was monitored for 24 h. For SCFA measurement in vitro screening, the dominant gut bacteria were selected using a human gut microbial gene catalog25 obtained from the Japan Collection of Microorganisms (Ibaraki, Japan). Bacteria were recovered according to the manufacturer’s instructions as previously described18. Intestinal bacteria were collected in nutrient broth (Difco Laboratories Inc.) containing 10% glycerol and stored at −80 °C. The obtained bacteria are listed in Supplementary Table 1.
Characterisation of S. salivarius-produced EPS
S. salivarius was cultured on MRS agar alone at 37 °C or MRS agar containing 15% sucrose at 30 °C for 48 h under anaerobic conditions and imaged using scanning electron microscopy (JSM-7500F; HUSRI, Aichi, Japan). SsEPS was collected from the agar plate and purified using ethanol precipitation, as previously described18 or dialysis membranes with a molecular cutoff of 3500 Da (Snake Skin Dialysis Tubing, Thermo Fisher Scientific, Waltham, MA, USA). The precipitated SsEPS was dried over calcium chloride for 24–48 h. To determine its monosaccharide composition, SsEPS was extracted as described previously18, with certain modifications. Briefly, SsEPS was hydrolysed by the addition of trifluoroacetic acid (0.5 M) and incubated at 120 °C for 0.5–2 h. After incubation, the supernatant was filtered through a 0.45 μm filter. The monosaccharide composition was analysed by ligand exchange chromatography using an 8.0 × 300 mm SUGAR SC1011 column (Shodex, Tokyo, Japan). Detection was performed using an RID-20A instrument (Shimadzu, Kyoto, Japan), with D-glucose and D-fructose (Nacalai Tesque, Kyoto, Japan) as standards. The average molecular weight of SsEPS was determined by size exclusion chromatography using an 8.0 × 300 mm OHpak SB-800 HQ series column (Shodex). Standards for purchased pullulans (Shodex) and dextrans (Sigma-Aldrich, St. Louis, MO, USA) with average molecular weights of 5900–1,600,000 and 1,500,000–2,800,000 Da, respectively, were established using calibration curves.
Structure of S. salivarius-produced EPS
The structure of the SsEPS was confirmed using 1H and 13C NMR spectroscopy. SsEPS was dissolved in 750 μL of D2O containing 0.1% 3-(trimethylsilyl) propionic-2,2,3,3-d4 acid sodium salt (TMSP). After allowing the solution to stand for 12 h, the 1H NMR 13C NMR spectra were recorded using a JEOL ECA-500 spectrometer with a frequency of 500 and 125 MHz, respectively at 25 °C. Chemical shifts are reported in δ (ppm) relative to TMSP as the chemical shift internal standard. The infrared (IR) spectra were recorded using a JASCO FT/IR-4100 spectrometer.
S. salivarius-produced levan: IR (neat cm-1): 3415 (OH); 1H NMR (500 MHz, D2O): δ 4.20 (d, J = 8.0 Hz, 1H), 4.14–4.08 (m, 1H), 3.98–3.87 (m, 2H), 3.78 (d, J = 12.0 Hz, 1H), 3.69 (d, J = 12.0 Hz, 1H), 3.59–3.55 (m, 1H); 13C{1H} NMR (125 MHz, D2O): δ 107.1, 83.2, 79.2, 78.1, 66.2, 62.8.
S. salivarius-produced glucan: IR (neat cm-1): 3375 (OH); 1H NMR (500 MHz, D2O): δ 4.99 (d, J = 2.9 Hz, 1H), 4.04–3.92 (m, 2H), 3.80–3.70 (m, 2H), 3.59 (dd, J = 9.7, 2.9 Hz, 1H); 13C{1H} NMR (125 MHz, D2O): δ 100.5, 76.2, 74.3, 73.0, 72.4, 68.4.
RNA isolation and quantitative reverse transcription polymerase chain reaction (RT-qPCR)
S. salivarius was cultured in MRS medium containing 15% sucrose or 15% glucose at 30 °C for 10 h under anaerobic conditions. Total RNA from each S. salivarius cultivation was extracted using the NucleoSpin RNA kit (Takara Bio, Shiga, Japan). Total RNA from subcutaneous white adipose tissue was extracted using an RNAiso Plus reagent (Takara Bio). cDNA was transcribed using RNA as templates and Moloney murine leukemia virus reverse transcriptase (Thermo Fisher Scientific). SYBR Premix Ex Taq II (Takara Bio) and StepOnePlus real-time PCR system (Applied Biosystems) were used for RT-qPCR analysis, as previously described18. SsEPS-synthesised enzyme primer sequences are listed in Supplementary Table 2. Primer sequences in vivo were as follows: Ucp1, 5′-GGCATTCAGAGGCAAATCAG-3′(forward) and 5′-AAGCATTGTAGGTCCCCGTG-3′ (reverse); and 18S, 5′-ACGCTGAGCCAGTCAGTGTA-3′ (forward) and 5′-CTTAGAGGGACAAGTGGCG-3′ (reverse).
RNA-sequencing data analysis
The extracted total RNA from S. salivarius was used and sequencing libraries were constructed using an NEBNext rRNA Depletion Kit (New England Biolabs, Inc., Ipswich, MA, USA) and a TruSeq Stranded mRNA Library Prep Kit (Illumina, CA, USA) according to the manufacturer’s protocols. The libraries were sequenced on an Illumina HiSeq 2500 platform with 100 bp paired-end reads. On average, 1.2 million read pairs per sample were sequenced across eight samples (four glucose and four sucrose samples). RNA-Seq data were analysed using the CLC Genomics Workbench (Qiagen Bioinformatics, Venlo, Netherlands) to identify differentially expressed genes. To obtain clean reads, low-quality reads were removed by trimming, whereas high-quality reads were aligned to the S. salivarius NCTC 7366 genome retrieved from the NCBI database. The parameters were set as follows: minimum length fraction = 0.8 and minimum similarity fraction = 0.8. Expression values were established as transcripts per million reads. A gene set enrichment analysis was performed using the KEGG database (http://www.genome.jp/kegg/) from the GhostKOALA result of expressed genes. The enriched pathways was identified using a Welch’s t-test with a false discovery rate correction (q < 0.01) using the R software environment (v4. 2. 1).
Inactivation of levansucrase in S. salivarius
Levansucrase was inactivated via homologous recombination using a single crossover event. A suicide plasmid was constructed as follows: pIB184 (EM), an E. coli- Streptococci shuttle vector obtained from Addgene (Watertown, MA, USA), was digested with Bpu10I (New England Biolabs) and PciI (New England Biolabs). A 2.7-kb DNA fragment, containing a pUC ori replicon and an erythromycin resistance gene was ligated to a 529-bp PCR-amplified internal homologous region and digested by restriction enzymes using Ligation high Ver.2 (Toyobo, Osaka, Japan). S. salivarius, prepared for transformation, were grown for 12 h in a Gifu anaerobic medium (GAM) (Nissui, Tokyo, Japan) containing 1% glycine, collected, and suspended in ice-cold transformation buffer (50 mM sucrose and 1 mM magnesium chloride). Plasmid electroporation was performed under the following conditions: 200 Ω; 12.5 kV/cm; and 25 μF using a Gene Pulser Xcell™ Electroporation system (Bio-Rad, Hercules, CA, USA). The accurate insertion of the plasmid was confirmed by genome PCR and sequencing analysis.
Shotgun metagenomic sequencing data analysis
The DNA extracted from human and mouse faecal samples was quantitated using Qubit fluorometric quantitation (Thermo Fisher Scientific) and qualified by DNA size profiling on a fragment analyser (Agilent, Santa Clara, CA, USA). High molecular weight DNA (>10 kbp; 3 μg) was used to build the library. DNA was sheared into fragments of approximately 150 bp using an ultrasonicator (Covaris, Woburn, MA, USA), and the DNA fragment library was constructed using the Ion Plus Fragment Library and Ion Xpress Barcode Adapters kits (Thermo Fisher Scientific). Purified and amplified DNA fragment libraries were sequenced using DNBSEQ-G400 (MGI Tech) with a minimum of 20 million high-quality reads of 150 bp (on average) generated per library. The paired-end sequences were merged using BBmaps (v38.84-0)43, and preprocessed using Kneaddata (v0.12.0) to remove the host genome based on the human (hg37 dec_v0.1) and mouse (C57BL_6NJ) genome databases. The Whole genome sequence based taxonomy profile was generated by MetaPhlAn (v4.0.4)44. Microbial gene families and metabolic pathways were assessed using HUMAnN3 (v3.8)45 based on the UniRef90 EC filtered database (uniref90_201901). MaAsLin2 was used to identify significant pathway from HUMAnN3 outputs46. The Gene Ontology terms of biological process and molecular function were considered. All computational scripts are available on GitHub [https://github.com/petadimensionlab/EPS] and Zenodo (https://doi.org/10.5281/zenodo.14550033).
SCFAs measurement
SCFA levels in human faeces, mouse faeces, and mouse plasma were measured following a previously described modified protocol47. Ether layers containing SCFAs were collected and pooled for gas chromatography-mass spectrometry (GC-MS) using a GCMS-QP2010 Ultra GC mass spectrometer (Shimadzu). The SCFA concentration was evaluated over a specified concentration range.
EPS measurement
Human faeces and mouse jejunum, ileum, caecum and faeces were immediately mixed with five volumes of sterile distilled water containing 2% 5-sulfosalicylic acid and vortexed. The mixture was then centrifuged 14,000 × g for 15 min at 4 °C, and the supernatant containing the EPS was collected. Two volumes of hexane were added to the supernatant, which was then vortexed for 5 min. After centrifugation of the samples at 10,000 × g for 15 min, the water layers containing EPS were collected and subjected to high-performance liquid chromatography (HPLC) analysis using an RID-20A instrument (Shimadzu) and an 8.0 × 300 mm OHpak SB-800 HQ series column (Shodex).
Animal study
C57BL/6J (wild-type), Gpr41Gpr43 double-deficient, and ICR mice were housed under a 12-h light–dark cycle and fed normal chow (CE-2; CLEA, Tokyo, Japan). C57BL/6J male mice were purchased from Japan SLC (Shizuoka, Japan). Germ-free (GF)-ICR male mice were housed in vinyl isolators under a 12-h light–dark cycle and fed normal chow (CL-2, 50kGy irradiated; CLEA). Gpr41Gpr43 double-deficient mice were generated as described previously12. Male mice were used in all experiments. All experimental procedures involving mice were performed according to the protocols approved by the Committee on the Ethics of Animal Experiments of the Kyoto University Animal Experimentation Committee (Lif-K21020) and Tokyo University of Agriculture and Technology (permit number: R05-47 and R05-48). All mice were sacrificed under deep isoflurane-induced anesthesia. All efforts were made to minimise suffering. Sample sizes were selected based on pilot experiments or based on other published work that gave reliable statistical results.
Four-week-old C57BL/6J and Gpr41Gpr43 double-deficient mice were fed a modified D12492 diet (60% kcal fat; Research Diets, New Brunswick, NJ, USA) for 12 weeks in HFD studies. The composition of the modified diet is shown in Supplementary Table 3. For the oral GTT, 16-h-fasted obese mice were administered glucose (2 g/kg of body weight) via oral gavage. For the insulin tolerance test, 3-h-fasted obese mice were intraperitoneally injected with human insulin (0.75 mU/g; Sigma-Aldrich). Plasma glucose concentration was monitored before and 15, 30, 60, 90, and 120 min after injection.
After fasting 24 h, 7-week-old C57BL/6J, Gpr41Gpr43 double-deficient, conventional ICR, and GF-ICR mice were fed 0.2 g AIN-93G (Research Diets), containing 50% cellulose or 50% SsEPS. After 1 h, glucose (2 g/kg body weight) was intraperitoneally administered to each mouse. Blood glucose levels in the tail vein were measured using a OneTouch UltraVue glucometer (LifeScan, Milpitas, CA, USA) and an LFS Quick Sensor (LifeScan) before and at 15, 30, 60, 90, and 120 min after injection. Plasma samples were collected from the inferior vena cava at 15 min after glucose administration for insulin and GLP-1 measurement18,48. To prevent the degradation of active GLP-1, plasma samples were treated with a dipeptidyl peptidase IV inhibitor (Merck Millipore, Burlington, MA, USA).
GF and Gnotobiotic experiments
GF mice were maintained in vinyl isolators under a 12-h light–dark cycle and routinely assessed for sterility by culturing under aerobic and anaerobic conditions, as well as by qPCR analysis (16S rRNA gene) using fresh faecal samples49,50. GF mice fed a chow diet (50 kGy irradiated; CL-2, CLEA) and drank an autoclaved water ad libitum.
GF mice were colonised by gavage of each bacterial strain (S. salivarius, B. ovatus, and B. thetaiotaomicron). After colonisation, each gnotobiotic mouse was housed in an independent vinyl isolator51. Gut microbial composition following colonisation was monitored by culturing under aerobic and anaerobic conditions. Five-week-old GF-ICR mice were fed an AIN-93G diet (50kGy irradiated) for 4 weeks. After 2 weeks, S. salivarius, B. ovatus and B. thetaiotaomicron (1 × 109 CFU/mouse) were administered via oral gavage three times per week. Sterilised water containing 20% sucrose, glucose, or fructose was administered for 2 weeks. After 16 h, for GTT, mice were administered glucose (2 g/kg of body weight) intraperitoneally, because oral glucose administration directly stimulates GLP-1 and other incretin secretions in gastrointestinal tract and may compete with imbibed sugar. The blood glucose concentration in the tail vein was monitored before glucose injection and at 15-, 30-, 60-, 90-, and 120-min after injection. For long-term treatment, 7-week-old GF-ICR mice were fed an AIN-93G diet, D12492 diet, or modified D12492 diet (50 kGy irradiated) for 9 weeks. Each bacterial strain (1 × 109 CFU/mouse) was administered via oral gavage three times a week at 7 and 11 weeks of age. The composition of the modified D12492 diet is shown in Supplementary Table 4.
Cultured faecal transplantation in animal experiment
The faecal samples from two women, [Ss (+)] (aged 43 with a BMI of 30.0 kg m-2) and Ss (-) (aged 45 with a BMI of 40.5 kg m-2) were diluted in 1.0% saline solution and filtered through a membrane paper. The filtered faecal samples were suspended in equal volumes of nutrient broth (Difco Laboratories Inc.) containing 10% glycerol and stored at −80 °C until use. The thawed frozen samples were cultured anaerobically at 37 °C for 24 h in GAM, filtered through a membrane paper. Mice were administered faecal culture solutions (approximately 250 μL per mouse) via oral gavage two times per week until 16 weeks of age52,53.
Biochemical analyses
Blood glucose levels were measured using a OneTouch UltraVue glucometer (LifeScan) and an LFS Quick Sensor (LifeScan). The levels of plasma non-esterified fatty acids (LabAssayTM NEFA; Wako Pure Chemical Co. Ltd., Osaka, Japan), triglycerides (LabAssayTM Triglyceride; Wako Pure Chemical Co. Ltd.), total cholesterol (LabAssayTM Cholesterol; Wako Pure Chemical Co. Ltd.), insulin (Mouse Insulin enzyme-linked immunosorbent assay [ELISA]; Shibayagi, Gunma, Japan), active glucagon like peptide-1 (GLP-1) (GLP-1 [Active] ELISA; Merck Millipore, Billerica, MA, USA), and PYY (Mouse/Rat PYY ELISA Kit; Wako Pure Chemical Co. Ltd.) were measured according to the manufacturer’s instructions.
DNA extraction and gut microbial composition
DNA was extracted from human and mouse faecal samples using the FastDNA SPIN kit (MP Biomedicals, Irvine, CA, USA) as described previously18. Partial 16S rRNA gene sequences were amplified by targeting the hypervariable regions v4 using the primers 515 F; 5′-TCGTCGGCAGCGTCAGATGTGTATAAGAGACAGGTGYCAGCMGCCGCGGTAA-3′ and 806 R; 5′-GTCTCGTGGGCTCGGAGATGTGTATAAGAGACAGGGACTACHVGGGTWTCTAAT-3′. Amplicons generated from each sample were purified using AMPure XP Beads (Beckman Coulter, Brea, CA, USA), and appended with Nextera XT index kit (Illumina, San Diego, CA, USA). Amplicons were sequenced using a MiSeq sequencer (Illumina) and MiSeq Reagent kit (version 3.0; 600 cycles). The 16S rRNA sequence data were then processed using the Quantitative Insights Into Microbial Ecology 2 (QIIME 2) pipeline, and analysed using the MiSeq Reporter software with the SILVA database (Illumina). Diversity was analysed using QIIME script core_diversity_analyses.py. Permutational multivariate analysis of variance (QIIME script compare_categories.py) was used to assess the statistical significance of sample groupings. Gut microbial composition was evaluated to perform principal coordinate analysis using the R software environment (v4. 2. 1). For quantitative PCR, SYBR Premix Ex Taq II (Takara Bio) and a StepOnePlus real-time PCR system (Applied Biosystems) were used. The bacterial primer sequences are listed in Supplementary Tables 5 and 6.
Statistical analysis
The mean ± standard error of the mean is presented for all values. We assessed the normality of the data using the Shapiro–Wilk test (normal distribution was defined at p ≥ 0.05). To determine the statistical significance between two groups with normal distribution, we used Student’s t test. For groups with non-normal distribution, the Mann–Whitney U test was used for comparison. One-way analysis of variance was used to compare data from multiple groups (three or more). For normally distributed sample sets, Dunnett’s post-hoc test was used, whereas the Kruskal–Wallis test paired with Dunn’s post-hoc test was used for non-normally distributed sample sets. Statistical significance was set at p < 0.05. Additionally, The Benjamini–Hochberg procedure was used to estimate the false discovery rates (Q-values) of the 16S rRNA gene sequencing data. The correlations between microbiota and gut environmental factors were analysed. To calculate correlations, we used Spearman’s rank correlation coefficients for the abundance of bacterial genera, including Muribaculum, Paramuribaculum, Duncaniella, Bacteroides, Akkermansia, Faecalitalea, Desulfovibrio, Streptococcus, Blautia, and Ruminococcus and faecal SCFAs, such as acetate, propionate, and n-butyrate. We selected only correlations with an absolute value above 0.6 and a Q-value below 0.05. Outliers were evaluated using the Smirnov–Grubbs test.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The 16S rRNA sequencing data generated in this study have been deposited in the DNA Data Bank of Japan (DDBJ) under accession Nos. DRA017528, DRA017529, and DRA017628. The shotgun metagenomic sequencing data are available under restricted accession Nos. DRA017626 and DRA017627. RNA sequencing data are accessible via accession Nos. DRA017530 and E-GEAD-664. The draft genome sequencing data of S. salivarius are available under restricted accession Nos. DRA018992 and BAAFPP010000000. The Figs. 1–6, Supplementary Figs. 1–9 data generated in this study are provided in the Supplementary Information/Source Data file. The Figs. 1–6, Supplementary Figs. 1–9, 16S rRNA sequencing, shotgun metagenomic sequencing, and RNA sequencing data used in this study are available in the Dryad repository under accession code (https://doi.org/10.5061/dryad.6djh9w17p). Source data are provided with this paper.
References
Kahn, S. E., Hull, R. L. & Utzschneider, K. M. Mechanisms linking obesity to insulin resistance and type 2 diabetes. Nature 444, 840–846 (2006).
Zimmet, P., Alberti, K. G. & Shaw, J. Global and societal implications of the diabetes epidemic. Nature 414, 782–787 (2001).
Cori, C. F. Mammalian carbohydrate metabolism. Physiol. Rev. 11, 144–275 (1931).
Tappy, L. & Lê, K. A. Metabolic effects of fructose and the worldwide increase in obesity. Physiol. Rev. 90, 23–46 (2010).
Flint, H. J., Bayer, E. A., Rincon, M. T., Lamed, R. & White, B. A. Polysaccharide utilization by gut bacteria: potential for new insights from genomic analysis. Nat. Rev. Microbiol. 6, 121–131 (2008).
Lee, J. Y., Tsolis, R. M. & Bäumler, A. J. The microbiome and gut homeostasis. Science 377, eabp9960 (2022).
Sonnenburg, J. L. & Bäckhed, F. Diet-microbiota interactions as moderators of human metabolism. Nature 535, 56–64 (2016).
Litvak, Y., Byndloss, M. X. & Bäumler, A. J. Colonocyte metabolism shapes the gut microbiota. Science 362, eaat9076 (2018).
Ikeda, T., Nishida, A., Yamano, M. & Kimura, I. Short-chain fatty acid receptors and gut microbiota as therapeutic targets in metabolic, immune, and neurological diseases. Pharmacol. Ther. 239, 108273 (2022).
Koh, A., De Vadder, F., Kovatcheva-Datchary, P. & Bäckhed, F. From dietary fibre to host physiology: short-chain fatty acids as key bacterial metabolites. Cell 165, 1332–1345 (2016).
Kimura, I., Ichimura, A., Ohue-Kitano, R. & Igarashi, M. Free fatty acid receptors in health and disease. Physiol. Rev. 100, 171–210 (2020).
Kimura, I. et al. Maternal gut microbiota in pregnancy influences offspring metabolic phenotype in mice. Science 367, eaaw8429 (2020).
Kimura, I. et al. Short-chain fatty acids and ketones directly regulate sympathetic nervous system via G protein-coupled receptor 41 (GPR41). Proc. Natl Acad. Sci. USA 108, 8030–8035 (2011).
Kimura, I. et al. The gut microbiota suppresses insulin-mediated fat accumulation via the short-chain fatty acid receptor GPR43. Nat. Commun. 4, 1829 (2013).
Moradali, M. F. & Rehm, B. H. A. Bacterial biopolymers: from pathogenesis to advanced materials. Nat. Rev. Microbiol. 18, 195–210 (2020).
Santos, M., Teixeira, J. & Rodrigues, A. Production of dextransucrase, dextran and fructose from sucrose using Leuconostoc mesenteroides NRRL B512(f). Biochem. Eng. J. 4, 177–188 (2000).
Dols, M., Chraibi, W., Remaud-Simeon, M., Lindley, N. D. & Monsan, P. F. Growth and energetics of Leuconostoc mesenteroides NRRL B-1299 during metabolism of various sugars and their consequences for dextransucrase production. Appl. Environ. Microbiol. 63, 2159–2165 (1997).
Miyamoto, J. et al. Host metabolic benefits of prebiotic exopolysaccharides produced by Leuconostoc mesenteroides. Gut Microbes 15, 2161271 (2023).
Matsuzaki, C. et al. Enzymatically synthesized exopolysaccharide of a probiotic strain Leuconostoc mesenteroides NTM048 shows adjuvant activity to promote IgA antibody responses. Gut Microbes 13, 1949097 (2021).
Chun, B. H., Kim, K. H., Jeon, H. H., Lee, S. H. & Jeon, C. O. Pan-genomic and transcriptomic analyses of Leuconostoc mesenteroides provide insights into its genomic and metabolic features and roles in kimchi fermentation. Sci Rep. 7, 11504 (2017).
Masuda, Y. et al. Identification and characterization of leucocyclicin Q, a novel cyclic bacteriocin produced by Leuconostoc mesenteroides TK41401. Appl. Environ. Microbiol. 77, 8164–8170 (2011).
Rühmann, B., Schmid, J. & Sieber, V. Methods to identify the unexplored diversity of microbial exopolysaccharides. Front. Microbiol. 6, 565 (2015).
Kim, E., Yang, S. M. & Kim, H. Y. Weissella and the two Janus faces of the genus. Appl. Environ. Microbiol. 107, 1119–1127 (2023).
D’Angelo, L. et al. Leuconostoc strains isolated from dairy products: Response against food stress conditions. Food Microbiol. 66, 28–39 (2017).
Hirano, R. et al. Next-generation prebiotic promotes selective growth of bifidobacteria, suppressing Clostridioides difficile. Gut Microbes 13, 1973835 (2021).
Beiroa, D. et al. GLP-1 agonism stimulates brown adipose tissue thermogenesis and browning through hypothalamic AMPK. Diabetes 63, 3346–3358 (2014).
Sidossis, L. & Kajimura, S. Brown and beige fat in humans: thermogenic adipocytes that control energy and glucose homeostasis. J. Clin. Investig. 125, 478–486 (2015).
Nagpal, R. et al. Comparative Microbiome signatures and short-chain fatty acids in mouse, rat, non-human primate, and human faeces. Front. Microbiol. 9, 2897 (2018).
Wichmann, A. et al. Microbial modulation of energy availability in the colon regulates intestinal transit. Cell Host Microbe 14, 582–590 (2013).
Miyamoto, J. et al. Barley β-glucan improves metabolic condition via short-chain fatty acids produced by gut microbial fermentation in high fat diet fed mice. PLoS ONE 13, e0196579 (2018).
Furusawa, Y. et al. Commensal microbe-derived butyrate induces the differentiation of colonic regulatory T cells. Nature 504, 446–450 (2013).
Salazar, N. et al. Functional effects of EPS-Producing. Front. Microbiol. 10, 1809 (2019).
Smith, B. J. et al. Changes in the gut microbiome and fermentation products concurrent with enhanced longevity in acarbose-treated mice. BMC Microbiol. 19, 130 (2019).
Zhang, X. et al. Effects of acarbose on the gut microbiota of prediabetic patients: a randomized, double-blind, controlled crossover trial. Diabetes Ther. 8, 293–307 (2017).
Horvath, A. et al. Biomarkers for oralization during long-term proton pump inhibitor therapy predict survival in cirrhosis. Sci. Rep. 9, 12000 (2019).
Imai, J. et al. The pathogenic oral-gut-liver axis: new understandings and clinical implications. Expert Rev. Clin. Immunol. 17, 727–736 (2021).
Yukawa, M. Y. et al. Distinct responsiveness to rifaximin in patients with hepatic encephalopathy depends on functional gut microbial species. Hepatol. Commun. 6, 2090–2104 (2022).
Zhao, S. et al. Dietary fructose feeds hepatic lipogenesis via microbiota-derived acetate. Nature 579, 586–591 (2020).
Ishimoto, T. et al. Opposing effects of fructokinase C and A isoforms on fructose-induced metabolic syndrome in mice. Proc. Natl Acad. Sci. USA 109, 4320–4325 (2012).
Softic, S. et al. Divergent effects of glucose and fructose on hepatic lipogenesis and insulin signaling. J. Clin. Investig. 127, 4059–4074 (2017).
Wali, A. J. et al. Determining the metabolic effects of dietary fat, sugars and fat-sugar interaction using nutritional geometry in a dietary challenge study with male mice. Nat. Commun. 14, 4409 (2023).
Poli, A., Anzelmo, G. & Nicolaus, B. Bacterial exopolysaccharides from extreme marine habitats: production, characterization and biological activities. Mar. Drugs. 8, 1779–1802 (2010).
Bushnell, B., Rood, J. & Singer, E. BBMerge - Accurate paired shotgun read merging via overlap. PLoS ONE 12, e0185056 (2017).
Blanco-Míguez, A. et al. Extending and improving metagenomic taxonomic profiling with uncharacterized species using MetaPhlAn 4. Nat. Biotechnol. 41, 1633–1644 (2023).
Beghini, F. et al. Integrating taxonomic, functional, and strain-level profiling of diverse microbial communities with bioBakery 3. Elife 10, e65088 (2021).
Mallick, H. et al. Multivariable association discovery in population-scale meta-omics studies. PLoS Comput. Biol. 17, e1009442 (2021).
Miyamoto, J. et al. Ketone body receptor GPR43 regulates lipid metabolism under ketogenic conditions. Proc. Natl Acad. Sci. USA 116, 23813–23821 (2019).
Nishida, A., Miyamoto, J., Shimizu, H. & Kimura, I. Gut microbial short-chain fatty acids-mediated olfactory receptor 78 stimulation promotes anorexigenic gut hormone peptide YY secretion in mice. Biochem. Biophys. Res. Commun. 557, 48–54 (2021).
Kasahara, K. et al. Gut bacterial metabolism contributes to host global purine homeostasis. Cell Host Microbe 31, 1038–1053 (2023).
Sjöland, W. et al. Absence of gut microbiota reduces neonatal survival and exacerbates liver disease in Cyp2c70-deficient mice with a human-like bile acid composition. Clin. Sci. 137, 995–1011 (2023).
Muller, P. A. et al. Microbiota modulate sympathetic neurons via a gut-brain circuit. Nature 583, 441–446 (2020).
Choi, B. et al. Feeding diversified protein sources exacerbates hepatic insulin resistance via increased gut microbial branched-chain fatty acids and mTORC1 signaling in obese mice. Nat. Commun. 12, 3377 (2021).
Olsson, L. M. et al. Gut microbiota of obese subjects with Prader-Willi syndrome is linked to metabolic health. Gut 69, 1229–1238 (2020).
Acknowledgements
This works was supported by research grants from the AMED (JP17gm1010007 and JP23gm1510011 to I.K.), JSPS KAKENHI (JP21H04862 to I.K., JP22K17771 to J.M., and JP21K19813 to S.Nakaoka.), JST-OPERA (JPMJOP1833 to I.K.), JST-MOONSHOOT (JPMJMS2023 to I.K. and S.Nakaoka.), and Noster Inc (to I.K.).
Author information
Authors and Affiliations
Contributions
H.S. performed the experiments and wrote the paper; J.M. performed the experiments and wrote the paper; K.H. performed the experiments and interpreted the data; R.O.K. performed the experiments and interpreted the data; H.Takada performed the experiments and interpreted the data; M.Y. performed the experiments; A.N. performed the experiments; D.S. performed the experiments and interpreted the data; Y.M. performed the experiments; K.W. performed the experiments; S.Nishikawa performed the experiments; S.T. performed the experiments; T.I. performed the experiments; Y.N. performed the experiments; N.Y. performed the experiments; C.M. performed the experiments and interpreted the data; T.Kageyama performed the experiments; I.H. performed the experiments; A.M. performed the experiments and interpreted the data; R.A. performed the experiments and interpreted the data; S.K. performed the experiments; M.U. performed the experiments; T.M. performed the experiments and interpreted the data; S.I. performed the experiments and interpreted the data; J.I. performed the experiments and interpreted data; N.S.A. performed the experiments and interpreted the data; H.Toju performed the experiments and interpreted the data; H.M. performed the experiments and interpreted the data; S.Nakaoka performed the experiments and interpreted the data; T.Y. performed the experiments and interpreted the data; A.T. performed the experiments and interpreted the data; K.Y. performed the experiments and interpreted the data; H.O. performed the experiments and interpreted the data; T.Katayama performed the experiments and interpreted the data; H.I. performed the experiments and interpreted the data; I.K. supervised the project, interpreted the data, and wrote the paper; I.K. had primary responsibility for the final content. All authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
H.S., K.H., and D.S. are employees of Noster Inc. The other authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Changtao Jiang, Fernando Anhe and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Shimizu, H., Miyamoto, J., Hisa, K. et al. Sucrose-preferring gut microbes prevent host obesity by producing exopolysaccharides. Nat Commun 16, 1145 (2025). https://doi.org/10.1038/s41467-025-56470-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-56470-0