Abstract
The development of an HIV cure is a global health priority, with the target product profile (TPP) for an HIV cure guiding research efforts. Using a mathematical model calibrated to data from men who have sex with men (MSM) in the Netherlands, we assessed whether an effective cure could help end the HIV epidemic. Following the TPP, we evaluated two scenarios: (i) HIV remission, where the virus is suppressed in an individual without ongoing antiretroviral therapy (ART) but may rebound, and (ii) HIV eradication, which aims to completely remove the virus from the individual. Here, we show that sustained HIV remission (without rebound) or HIV eradication could reduce new HIV infections compared to a scenario without a cure. In contrast, transient HIV remission with a risk of rebound could increase new infections if rebounds are not closely monitored, potentially undermining HIV control efforts. Our findings emphasize the critical role of cure characteristics in maximizing cure benefits for public health and highlight the need to align HIV cure research with public health objectives to end the HIV epidemic.
Similar content being viewed by others
Introduction
Over the past decades, significant progress has been made toward achieving the United Nations Sustainable Development Goal of ending the HIV epidemic by 20301. Successes in reducing HIV transmission are particularly notable among men who have sex with men (MSM) in several Western European countries, including the Netherlands2,3,4. Since 2008, annual HIV diagnoses and HIV incidence among MSM in the Netherlands have declined by approximately 70%2, largely due to public health interventions such as pre-exposure prophylaxis (PrEP)5 and treatment as prevention6.
Despite these advances, developing an HIV cure is a global health priority7,8,9, as millions of people worldwide depend on lifelong antiretroviral treatment (ART) for viral suppression and a preventive vaccine is not yet available. People with HIV, including those in key populations such as MSM, experience impaired health-related quality of life despite effective ART10,11,12,13. Compared to individuals without HIV, people with HIV have worse physical and mental health due to chronic comorbidities and stigma10,12. Emerging biomedical technologies, such as an HIV cure, have the potential to improve health and well-being of all people with HIV including MSM across various life domains, similar to how ART revolutionized HIV treatment in the past7,14,15.
Biomedical research and human clinical trials for curative HIV interventions are advancing rapidly16,17. To date, several patients have been cured of HIV through HIV-resistant stem cell transplants18, but a scalable cure has yet to be developed and may take considerable time to become widely available. The consensus is that an acceptable and scalable cure for HIV will most likely require a combination of strategies targeting different aspects of HIV infection7,9,19. HIV eradication—aiming for the complete removal of HIV from the body—represents the ultimate goal for people with HIV14,20. However, HIV remission, where the virus remains suppressed below transmissible levels without ongoing ART, could be a more attainable target for researchers. Strategies that are currently being developed may contribute to both HIV remission and HIV eradication7,19,21,22,23,24.
The target product profile (TPP) for an HIV cure, a tool commonly used to guide drug development, has been devised by the International AIDS Society to align stakeholders on essential attributes for potential HIV cure interventions9. The TPP specifies key characteristics of a cure, including the target population, clinical efficacy, protection from re-infection, time to viral rebound, and monitoring requirements (Table 1). According to the TPP’s minimum and optimum criteria, interventions resulting in either HIV remission or HIV eradication could be considered curative. Following the TPP framework, we refer to each of these cure scenarios as ‘a cure’ and to individuals who achieve it as ‘cured’.
The characteristics of a cure intervention could have major consequences not only for the well-being of people with HIV but also for virus transmission at the population level. It is often hypothesized that an effective cure could aid in controlling the HIV epidemic7,8,24. However, there is limited research investigating whether and how a cure would affect HIV transmission dynamics. For example, in an HIV remission scenario with a risk of viral rebound, transmission of the virus could occur. In an HIV eradication scenario, if a cure does not confer immunity, cured individuals may still be susceptible to re-infection. These considerations underscore the need for further investigation into the potential impact of curative HIV interventions on transmission dynamics. The early analyses would help identify which cure characteristics mentioned in the TPP are important and provide an initial understanding of which cures may align with the current public health objectives of ending the HIV epidemic.
From the early days of the HIV epidemic, mathematical modeling played an important role in advancing our understanding of HIV epidemiology, evaluating the impact of interventions, and shaping public health strategies25,26,27. For example, the seminal modeling study by Granich et al.28 investigated the impact of test-and-treat on reducing HIV transmission, significantly influencing the international policy to end the HIV epidemic. Understanding how emerging HIV technologies, such as a potential cure, may affect the course of the epidemic is key to their successful implementation at the population level.
The impact of a cure intervention in a specific context may depend on several factors, such as key populations, the state of the HIV epidemic, standards of HIV care and prevention, and sexual behaviors relevant to HIV transmission. Previous modeling studies investigated the potential impact of a cure on generalized epidemics in heterosexual populations in Africa29,30. These studies suggested that a curative intervention could reduce HIV incidence in settings with less controlled epidemics. However, their focus on generalized epidemics differs from the concentrated HIV epidemic among MSM in the Netherlands, where substantial progress has been made toward HIV control under the current standard of care and prevention, leading to low HIV incidence and high ART coverage. Furthermore, they did not explicitly incorporate the full range of cure characteristics and possible cures outlined in the TPP. In this study, we used a mathematical model calibrated to epidemiological and sexual behavior data to evaluate the potential impact of curative interventions in this distinct population. We modeled how different cure scenarios, based on the full range of acceptable criteria established in the TPP, would affect HIV transmission dynamics. Finally, we explored the conditions under which these interventions may increase or decrease HIV transmission among MSM in the Netherlands and discussed the implications for the population-level cure implementation.
Results
Dynamics without cure
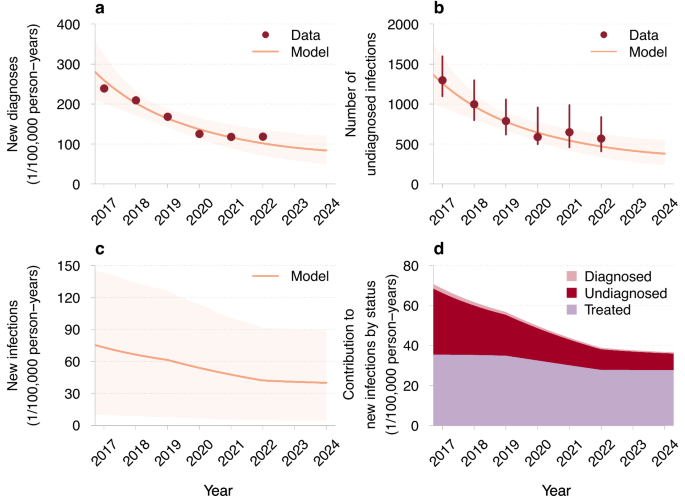
The model was fitted to and reproduced well the annual number of new HIV diagnoses and the estimated number of undiagnosed HIV infections among MSM from 2017 till 2022, reported by the Dutch HIV Monitoring Foundation (Stichting hiv monitoring, SHM) (Fig. 1)2. The number of new HIV infections per 100,000 persons per year acquired in the country was estimated to decrease from 73 (95%CrI 10–143) in 2017 to 42 (95%CrI 5–91) in 2022. The estimated mean time to diagnosis was 28 months (95%CrI 24–32), consistent with the SHM data2. The model predicted that, among all new HIV diagnoses, the proportions of diagnoses within 6 months, between 6 to 12 months, and more than 12 months since HIV infection were 22% (95%CrI 20–24%), 14% (95%CrI 13–16%), and 63% (95%CrI 60–66%), respectively, also aligning with the SHM data2. In 2024, treated individuals accounted for 75% (95%CrI 23–94%) of new infections acquired in the country, undiagnosed individuals for 23% (95%CrI 6%–71%), and diagnosed but untreated individuals for only 2% (95%CrI 0–6%), compared to the respective contributions of these groups of 52% (95%CrI 7–85%), 45% (95%CrI 14–85%), and 3% (95%CrI 1–6%) in 2017. Note that in the model, the category of treated individuals has a very low, but non-negligible infectivity, as it includes not only virally suppressed individuals but also those who are not virally suppressed due to recent treatment initiation, imperfect treatment adherence, treatment failure, or dropout. The estimated HIV prevalence was almost constant, around 6–7% throughout the considered period, which is in the range observed for MSM in Western Europe31. The model was additionally validated using independent data, not included in the model fitting, on the annual number of MSM on PrEP, the annual number of MSM on ART immigrating to the Netherlands from abroad, and the ART coverage among all MSM with HIV from 2017 till 2022 (Fig. S1).
a Incidence of new HIV diagnoses per 100,000 persons per year, b estimated total number of undiagnosed HIV infections, including importations from abroad, c incidence of new HIV infections acquired in the country per 100,000 persons per year, and d contribution to the mean new HIV infections by status of the source of HIV infection. The red dots and the error bars correspond to the mean estimates and the 95% confidence intervals reported by SHM2. The mean trajectories estimated from the model are shown as orange lines. The shaded regions correspond to 95% credible intervals based on 100 samples from the joint posterior parameter distribution.
Cure scenarios
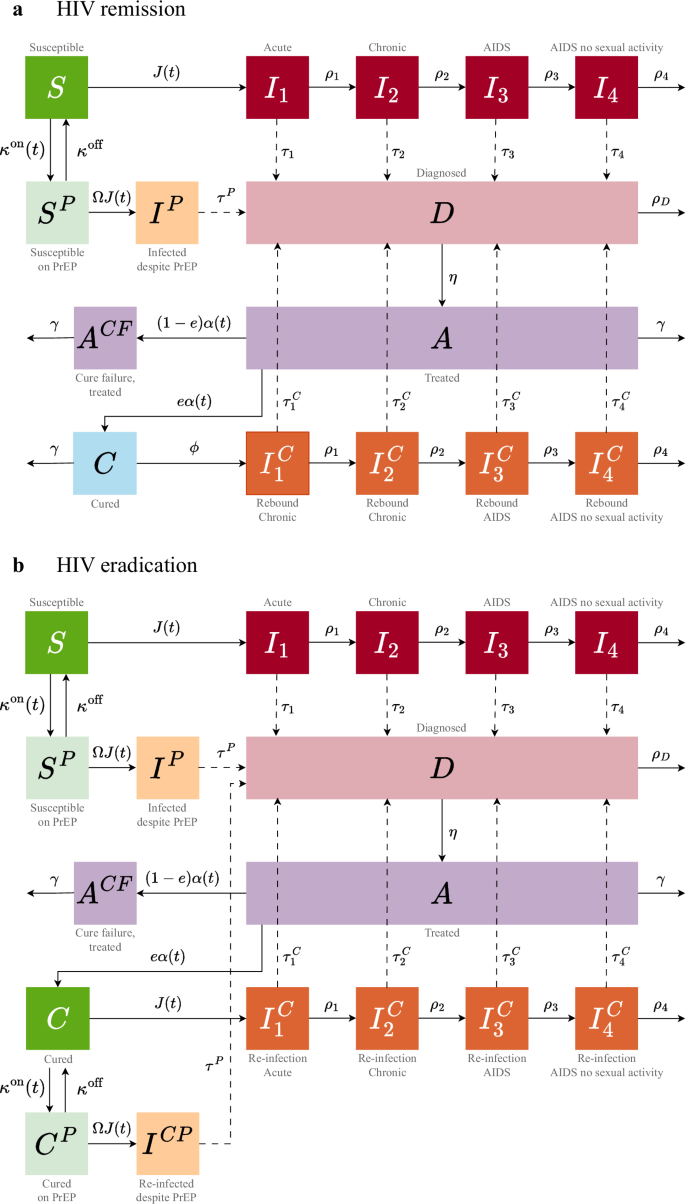
Guided by the TPP9, we developed a transmission model for the HIV remission and HIV eradication scenarios (see Fig. 2 and Methods). The relation of these cure scenarios to the minimum and optimum TPP9 is summarized in Table 1. In both scenarios, the cure was targeted at virologically suppressed individuals on ART (minimum TPP).
Transitions are shown for one risk group. Recruitment into and exit from the sexually active population are not shown. A detailed description of the model equations, parameters, and assumptions for a HIV remission and b HIV eradication is given in the Supplementary Material.
From an HIV dynamics perspective, the key difference between HIV remission and HIV eradication is that, in the remission scenario, the HIV reservoir within an individual is not completely removed. We defined ‘sustained’ HIV remission as a scenario where the virus remains below transmissible levels indefinitely (optimum TPP), whereas ‘transient’ HIV remission allows for possible viral rebound and potential onward transmission after a mean period of virus control of 2 years (minimum TPP). We assumed that individuals achieving HIV remission have full protection against re-infection (optimum TPP). In contrast, in the eradication scenario, all HIV including the intact and rebound-competent reservoir is fully removed. We further assumed that individuals achieving HIV eradication remain susceptible to re-infection (minimum TPP) and could start using PrEP if they have a high risk of HIV acquisition.
In both cure scenarios, we explored the entire range of acceptable values for cure characteristics outlined in the TPP, i.e., annual uptakes (proportion of eligible individuals receiving a cure intervention each year) of 10%, 50%, and 90%, and efficacy (proportion of individuals for whom the intervention was successful) of 20% (minimum TPP) and 90% (optimum TPP). In our model, the cure process is simplified by assuming that cure delivery and ART interruption are instantaneous. Individuals who did not take up a cure intervention or for whom the intervention was unsuccessful remained on ART. Moreover, we considered three monitoring strategies for individuals who may experience a viral rebound after HIV remission or re-infection after HIV eradication: (i) no monitoring (optimum TPP), resulting in a mean (across all HIV stages) diagnostic delay of 28 months, which aligns with the estimated delay of HIV infections under the current standard of care2; (ii) PrEP-like monitoring, with a mean diagnostic delay of 3 months, similar to the testing interval for PrEP users32; and (iii) frequent monitoring (minimum TPP) every two weeks, as used in analytical treatment interruptions studies33.
A cure intervention was assumed to be introduced in 2026, with maximum uptake reached within 3 years. We compared projections of HIV dynamics under the two cure scenarios and the no-cure scenario from 2026 to 2036. Changes in HIV incidence under the cure scenarios were always reported relative to the no-cure scenario. A detailed description of the model equations, parameters, and assumptions is provided in the Methods section, Fig. 2, and the Supplementary Material.
Dynamics for HIV remission scenario
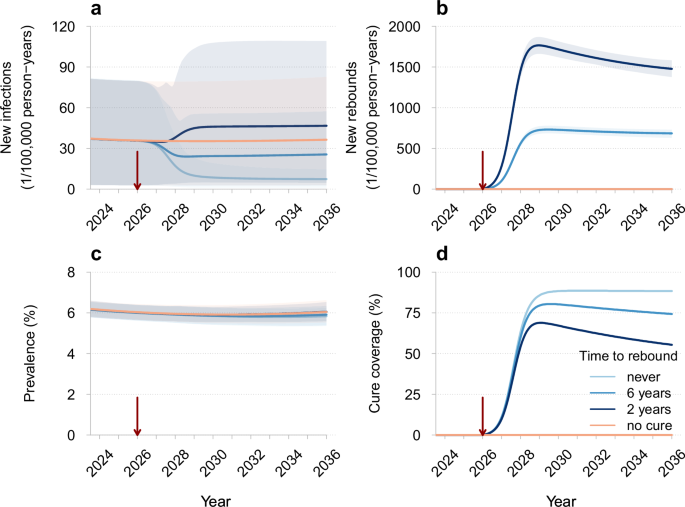
We first explored how the dynamics under the HIV remission scenario might unfold based on different assumptions about the time to viral rebound. We simulated the model with mean rebound times of 2 years and 6 years, as well as the possibility of sustained HIV remission (i.e., no rebound). We assumed 90% intervention efficacy and 90% annual uptake, along with a 3-month diagnostic delay of rebounds. Projections of the epidemic dynamics for these parameters are shown in Fig. 3. Sustained HIV remission, with no risk of rebound, resulted in the fewest new HIV infections compared to the no-cure scenario—7 (95%CrI 1–15) versus 36 (95%CrI 3–83) per 100,000 persons per year in 2036. If the time to viral rebound was 6 years, an estimated 26 (95%CrI 3–57) new HIV infections and 685 (95%CrI 630–730) new rebounds per 100,000 persons per year would occur in 2036. For a shorter rebound time of 2 years, new HIV infections and new rebounds increased to 47 (95%CrI 8–109) and 1,477 (95%CrI 1377–1582) per 100,000 persons per year in 2036. The HIV prevalence, including individuals in whom HIV remission was achieved but the viral reservoir was not fully removed, remained nearly constant regardless of the rebound time, while cure coverage (proportion of eligible individuals achieving HIV remission) was lower for shorter rebound times.
a New HIV infections, b new rebounds in individuals achieving HIV remission, c HIV prevalence (proportion of individuals with HIV), and d cure coverage (proportion of eligible individuals achieving HIV remission) for different times until viral rebound. The legend for different curves shown in d corresponds to all panels. The red vertical arrows indicate the cure introduction. The mean trajectories from the model are shown as solid lines. The shaded regions correspond to 95% credible intervals based on 100 samples from the joint posterior parameter distribution. Different shades of blue correspond to different times until viral rebound. The projections of the model without a cure are shown in orange. Parameters: efficacy of 90% (proportion of individuals for whom the intervention was successful), annual uptake of 90% (proportion of eligible individuals receiving the intervention each year), and a 3-month diagnostic delay of rebounds in individuals who achieved HIV remission.
The contribution of different groups to new HIV infections occurring in the country varied depending on the time to rebound (Fig. S2). For sustained HIV remission, undiagnosed and treated individuals remained the primary sources of new infections. Specifically, in 2036, these groups accounted for 51% (95%CrI 10%–88%) and 45% (95%CrI 5%–89%) of new infections, respectively, compared to 20% (95%CrI 5–67%) and 79% (95%CrI 27–95%) in the no-cure scenario. This suggests that the main effect of sustained HIV remission is a reduction in the contribution to HIV incidence of the population on ART, associated with its reduced relative weight. In the case of transient HIV remission, the contribution of the population on ART to incidence decreased too; however, individuals who were diagnosed but not yet treated, as well as those who experienced viral rebound, became substantial sources of new HIV infections. In 2036, diagnosed but untreated individuals and those with viral rebound accounted for 20% (95%CrI 11–28%) and 27% (95%CrI 14–39%) of new infections for a mean rebound time of 6 years, with even higher proportions, 23% (95%CrI 13–32%) and 32% (95%CrI 18–44%), for a rebound time of 2 years.
The cumulative person-years on ART averted over ten years, relative to the no-cure scenario, also depended on the time to rebound (Fig. S3a). In the sustained HIV remission scenario, ART use was reduced by 73% (95%CrI 72–74%) compared to the no-cure scenario. When the rebound time was 6 years and 2 years, ART use was reduced by 66% (95%CrI 65%–67%) and 56% (95%CrI 54–58%), respectively.
Impact of HIV remission under varied intervention characteristics
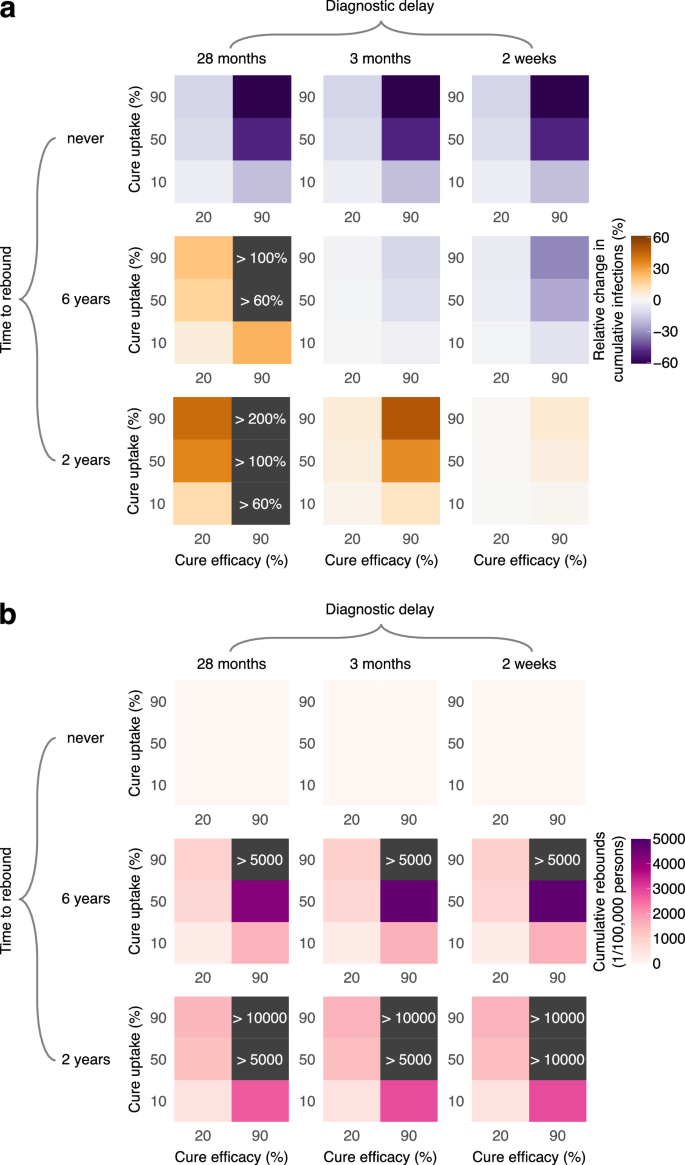
We further systematically compared the impact of HIV remission under varied efficacy, uptake, time to rebound, and diagnostic delay from its introduction in 2026 to 2036 (Fig. 4). Sustained HIV remission consistently resulted in fewer HIV infections, regardless of diagnostic delay (Fig. 4, top rows). Mean reductions in cumulative HIV infections over this period, compared to the no-cure scenario, ranged from 60% (95%CrI 21–72%) for 90% uptake and 90% efficacy to 4% (95%CrI 1–6%) for 10% uptake and 20% efficacy. Intermediate reductions in HIV infections were predicted for all other parameter combinations.
The color bar corresponds to a mean change in cumulative HIV infections relative to the no-cure scenario and b mean cumulative rebounds from the introduction of HIV remission scenario in 2026 to the end of the simulation in 2036.
The impact of transient HIV remission on cumulative HIV infections from 2026 to 2036, compared to the no-cure scenario, depended on diagnostic delay and rebound time (Fig. 4, middle and bottom rows). Without a dedicated monitoring strategy for diagnosing rebounds—resulting in a 28-month diagnostic delay, as observed under the current standard of care—this intervention consistently led to an increase in HIV infections. The extent of this increase was greater for shorter rebound times and for higher cure uptake and efficacy. This rise can be attributed to individuals experiencing viral rebound, whose contribution to incidence grows over time, reaching up to 72% (95%CrI 65–84%) in 2036 in the worst-case scenario of a 2-year rebound time, no additional monitoring, and 90% uptake and efficacy (Fig. S4).
In contrast, frequent monitoring of individuals in HIV remission (i.e., a 2-week diagnostic delay of rebounds) led to a reduction in cumulative HIV infections compared to the no-cure scenario for a 6-year rebound time. However, this came with an estimated maximum of 6132 (95%CrI 5770–6484) cumulative rebounds per 100,000 persons. For a shorter 2-year rebound time, even with frequent monitoring, cumulative HIV infections slightly exceeded those in the no-cure scenario, with an estimated maximum of 14,518 (95%CrI 13,611–15,430) cumulative rebounds per 100,000 persons.
We also computed the minimum monitoring frequency required for the HIV remission cure intervention to provide a net benefit in terms of cumulative new infections, using the values of efficacy, uptake, and time to rebound explored in Fig. 4 (see Table S1). The minimum monitoring frequency is approximately every 4.5 to 5 months for all combinations of cure uptake and efficacy, if the mean time to rebound is 6 years. However, if the mean time to rebound is only 2 years, even monitoring as frequently as every two weeks would be insufficient to mitigate the increase in new infections.
Dynamics for HIV eradication scenario
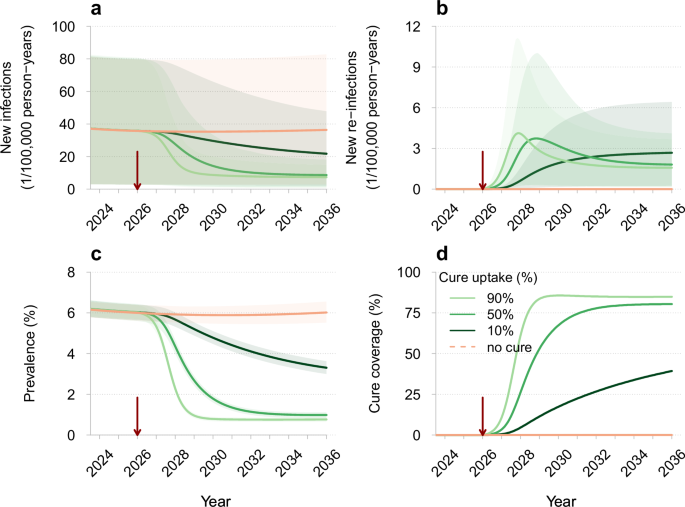
Similar to HIV remission, we first assessed transmission dynamics under the HIV eradication scenario, considering 90% efficacy and a 3-month diagnostic delay for re-infections. The introduction of HIV eradication led to a consistent reduction in the number of new HIV infections in naive individuals, with values varying based on cure uptake (Fig. 5). For 90% uptake, the estimated number of new HIV infections decreased to 7 (95%CrI 1–15) per 100,000 persons per year in 2036, which is comparable to the outcome of sustained HIV remission. For lower uptakes, new HIV infections also declined, albeit less markedly, reaching 9 (95%CrI 1–18) and 22 (95%CrI 2–48) per 100,000 persons per year for 50% and 10% uptake, respectively. Notably, across the entire range of uptakes considered, the estimated number of re-infections in cured individuals remained low, approaching only about 2 re-infections per 100,000 persons per year in 2036. Furthermore, unlike in the HIV remission scenario, HIV prevalence markedly dropped after the introduction of HIV eradication, reaching 3.31% (95%CrI 3.00–3.63%) for 10% uptake and falling to less than 1% for uptakes above 50%. The model also indicated that for lower uptakes, equilibrium in HIV dynamics had not yet been reached, with new HIV infections projected to decline further after 2036 (e.g., falling below 22 per 100,000 persons for 10% uptake).
a New HIV infections (primary infections in naive individuals), b new HIV re-infections (secondary infections in cured individuals), c HIV prevalence (proportion of individuals with HIV), and d cure coverage (proportion of cured individuals among all eligible) for different cure uptakes. The legend for different curves shown in d corresponds to all panels. The red vertical arrows indicate the cure introduction. The mean trajectories from the model are shown as solid lines. The shaded regions correspond to 95% credible intervals based on 100 samples from the joint posterior parameter distribution. Different shades of green correspond to different cure uptakes. The projections of the model without a cure are shown in orange. Parameters: efficacy of 90% (proportion of individuals for whom the intervention was successful) and a 3-month diagnostic delay of re-infections in individuals who achieved HIV eradication.
Similarly to sustained HIV remission, the HIV eradication intervention substantially reduced the contribution of individuals on ART to new HIV infections (Fig. S5). In 2036, this contribution declined from 79% (95%CrI 27–95%) in the no-cure scenario to 44% (95%CrI 5–89%) with a cure uptake of 90%. The contribution of re-infections was negligible. HIV eradication reduced ART use by 73% (95%CrI 72–75%) for a cure uptake of 90% (Fig. S3b), mirroring the effect of sustained HIV remission.
Impact of HIV eradication under varied intervention characteristics
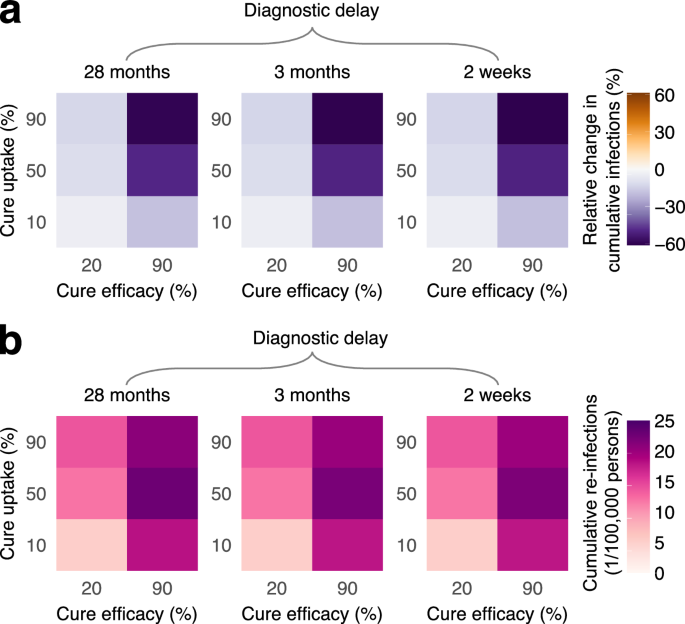
A systematic comparison of the impact of HIV eradication under varied efficacy, uptake, and diagnostic delay of re-infections is shown in Fig. 6. Similar to sustained HIV remission, HIV eradication consistently reduced HIV infections in naive individuals, regardless of diagnostic delay. Like before, the largest and smallest reductions in cumulative HIV infections over the 2026–2036 period were observed at the extreme values of uptakes and efficacy, with the reduction ranging from 4% (95%CrI 1–6%) to 60% (95%CrI 21–72%) for all parameter combinations explored. For all diagnostic delays, the estimated number of cumulative re-infections remained low, not exceeding 23 per 100,000 persons over 10 years.
The color bar corresponds to a mean change in cumulative HIV infections (primary infections in naive individuals) relative to the no-cure scenario and b mean cumulative HIV re-infections (secondary infections in cured individuals) from the introduction of HIV eradication scenario in 2026 to the end of the simulation in 2036. The color bar scale in (a) is the same as that in Fig. 4a for direct comparison.
Robustness and sensitivity analyses
The qualitative trends in projected HIV dynamics from the main analysis hold under alternative assumptions about importation of infected individuals, including a scenario without importation and one where the proportion of undiagnosed imported cases matches that of undiagnosed individuals in the infected population within the country (Figs. S6-S11). Furthermore, our results remained robust across different years of cure introduction (Figs. S12-S13) and when assessed over longer post-cure time frames (Figs. S14-S15). A potential increase in sexual risk behavior among MSM in response to a cure based on survey data did not qualitatively affect the overall outcomes (Fig. S16). Specifically, ten years post-cure, the number of new HIV infections was always lower for HIV eradication compared to the no-cure scenario, but this was not the case for transient HIV remission. Sensitivity analyses regarding the infectivity of individuals after viral rebound indicated that our projections for HIV remission were the most optimistic (Fig. S17). In contrast, the results for HIV eradication were consistent across varying assumptions about the infectivity of re-infections (Fig. S18). In an HIV remission scenario including both rebounds and re-infections (Fig.S19), the results remained consistent with those in Fig. 3, showing an increase in new HIV infections when the time to rebound is two years. The findings in Fig. 3 also hold for a scenario in which HIV remission is modeled with a rebound time following an Erlang distribution instead of an exponential distribution (Fig. S20). Whether an HIV remission cure prolonged survival compared to ART had minimal impact on our results. The qualitative insights into the impact of cure scenarios relative to the no-cure scenario were also robust to potential changes in the underlying trend in HIV incidence, particularly when new HIV infections started to increase from 2022 onward (Fig. S21). Further details of these and additional sensitivity analyses are given in the Supplementary Material.
Discussion
In light of rapidly advancing HIV cure research19, anticipating the potential population-level impact of cure implementation is essential before effective interventions become available. From a public health perspective, an effective cure should align with the United Nations Sustainable Development Goal of ending the HIV epidemic1. Indeed, there is a prevailing assumption that a cure will contribute to epidemic control7,8,24. In Western European countries where HIV epidemics are concentrated among MSM and HIV incidence is already low, this assumption implies that the implementation of a cure should not disrupt the progress made in HIV control under the current prevention and treatment standards.
We found that introducing either cure scenario could alter the course of the HIV epidemic among MSM in the Netherlands. The impact differed between sustained HIV remission or HIV eradication, which both have the potential to further reduce HIV transmission, and transient HIV remission, which could increase HIV transmission. This divergence in the potential outcomes of cure scenarios underscores the importance of evaluating their public health impacts, which, for now, can only be done through mathematical modeling34. Specifically, the model projections indicated that while sustained HIV remission consistently resulted in a decrease in cumulative HIV incidence over ten years (by up to 60% for optimum TPP), transient HIV remission could increase new infections. Notably, the increase in incidence was observed for plausible parameter combinations that might be achievable in a real-world setting (e.g., a 3-month diagnostic delay, which matches the testing interval for PrEP users32, and the TPP’s minimum requirement of 2 years before relapse9). This effect remained unmitigated even with frequent monitoring (e.g., every 2 weeks), which is unlikely to be feasible in a real-world setting. HIV incidence under transient HIV remission increased in the model because individuals who experienced viral rebound could transmit the virus before being diagnosed and after diagnosis until treatment initiation. This led to a counterintuitive effect where higher incidence was observed for higher cure uptakes and efficacies. The more individuals achieved HIV remission, the more rebounds occurred, leading to an increase in new infections. This effect could be mitigated by developing HIV remission strategies with longer times to rebound or through stricter monitoring. However, even in scenarios where transient HIV remission reduces HIV incidence, managing numerous rebound cases may pose a challenge in the real world and lead to adverse health outcomes for patients. Lifelong monitoring has been shown to negatively impact acceptance of a potential cure among people with HIV in the Netherlands, though it is not their primary concern compared to other cure characteristics35. However, its potential mental burden on cured individuals remains to be investigated. People with HIV in the Netherlands and Australia have also raised concerns about transient HIV remission because of fear of transmitting the virus to their partners and stigma14,20. Frequent rebound episodes would require ongoing efforts for rapid viral load monitoring and timely diagnosis of rebounds to maximize the public health benefits of any HIV remission strategy. The development of a simple point-of-care or at-home diagnostic for detecting rebounds may be a potential solution mentioned by experts who developed the TPP for an HIV cure9.
In contrast, the HIV eradication scenario presents a more optimistic outlook for HIV dynamics. Our findings suggest that this scenario would always reduce HIV incidence among naive individuals, without the requirement of additional monitoring, with projected cumulative new infections decreasing similarly to the sustained HIV remission scenario (by up to 60% for optimum TPP) within ten years. Importantly, the HIV eradication scenario would maintain a low number of re-infections, which is relevant for those MSM who are particularly vulnerable due to their sexual risk behavior36. The main reason for few re-infections under the HIV eradication scenario and a potentially large number of rebounds under the transient HIV remission scenario is that the rate of re-infections depends on the population with HIV, while rebounds occur at a constant rate among individuals who achieved HIV remission. This could also explain why, in high-incidence settings like South Africa, the potential for re-infection might offset the decline in incidence expected from an eradication cure30.
It is recognized that mathematical modeling can help advance HIV cure research34. However, few studies have modeled the impact of cure strategies at the population level. To our knowledge, this is the first study to assess the potential impact of an HIV cure on an epidemic among MSM in a Western European country with low HIV incidence. We ensured the reliability of our model to project HIV dynamics without a cure by inferring key model parameters and conducting model validation on multiple datasets. Unlike other studies assessing interventions for HIV control in low-incidence settings37,38,39, our model accounted for the openness of the MSM population by incorporating immigration of individuals to the Netherlands who acquired HIV abroad. The strength of our modeling approach lies in its ability to project HIV dynamics without requiring precise information on the biological mechanisms underlying a cure. Instead, our analyses were guided by the TPP, using the full range of acceptable values for several cure characteristics9. Our findings indicate that certain cure scenarios meeting the TPP criteria may lead to unintended increases in transmission. This suggests that further refinement of the TPP may be necessary to ensure that curative interventions align optimally with public health goals, particularly in concentrated epidemics such as among MSM in the Netherlands.
There is concern that the introduction of a cure could shift perceptions of HIV risk, severity, and prevention, as observed when ART became widely available40. Our study is the first to incorporate actual survey data on potential behavioral changes among MSM following the introduction of a cure into sensitivity analyses. Our findings suggest that risk compensation could further reduce the effectiveness of the transient HIV remission scenario while having minimal impact on the outcomes of the HIV eradication scenario. Therefore, real-world cure interventions may need to be accompanied by additional prevention strategies to address the potential change in sexual behavior.
While our findings are particularly relevant for similar HIV epidemics among MSM in Western European countries, where HIV incidence is low, they exhibit qualitative similarities with two previous modeling studies for a generalized epidemic in heterosexual populations in Africa29,30. The qualitative comparison with these studies is possible because they also explored scenarios with more optimistic epidemic projections and relatively high ART coverage, similar to the Dutch setting. Beacroft and Hallett30 demonstrated that in a well-controlled epidemic, both HIV eradication and HIV remission with 100% efficacy and a time to rebound of 8 years would reduce the number of new infections, aligning with our findings. While Dimitrov et al.29 concluded that HIV eradication strategies targeting treated individuals have minimal impact on incidence in low-ART settings, their results in high-ART settings qualitatively agree with our findings for the Netherlands, showing substantial reductions in new infections. However, our results differ from ref. 30, as we found that the timing of cure introduction did not substantially alter the outcomes of cure scenarios. This discrepancy may be explained by the fact that the HIV epidemic in the Netherlands was estimated to be close to reaching a low stable level of incidence, unlike the more dynamic epidemic in South Africa.
Our study has several limitations. First, we used a classical deterministic compartmental model, which assumed an exponential distribution for the time until viral rebound. Although this distribution may not perfectly capture relapse timing, it provides a way to compare cure scenarios conceptually, given the limited empirical data available. To test this assumption, we conducted a sensitivity analysis using an Erlang distribution for the time to rebound, which is more peaked but preserves the same mean. This modification did not alter the overall long-term trends observed in the HIV remission scenario. While a stochastic model might yield different quantitative outcomes, our primary conclusions would likely remain robust. Second, self-reported sexual behavior data may be biased, potentially affecting the estimated per-partnership transmission probability. However, because this parameter primarily scales the relative contributions of different risk groups to overall transmissibility, a uniform underestimation of contact rates across risk groups would likely have minimal impact on transmission dynamics. If the underestimation varied across risk groups, its effects on model estimates could be more complex and merit further investigation. Third, our projections are not intended as precise forecasts, given that future changes in the national PrEP program, the transition from daily ART regimens to long-acting injectable ART formulations, sexual behavior, or migration trends could influence the quantitative predictions for the no-cure scenario, potentially leading to an increase in HIV incidence. Nevertheless, our sensitivity analyses showed that the qualitative insights regarding the impact of cure scenarios relative to the no-cure scenario are expected to hold despite such changes. Fourth, despite the formulation of the TPP, uncertainty remains around several biological parameters for cured individuals. Based on data from ART interruption studies, we assumed that the infectivity of individuals following viral rebound would be similar to that observed during the chronic stage of HIV infection41,42. Conversely, we assumed the infectivity of individuals re-infected after HIV eradication would resemble the acute stage of HIV infection, similar to what occurs with superinfection by a different HIV subtype43 or re-infection with hepatitis C virus36,44. While these assumptions are biologically plausible, they remain hypothetical. Our sensitivity analyses demonstrated that high infectivity could undermine the effectiveness of the transient HIV remission scenario and should be carefully considered when developing a cure. Moreover, in line with the call for equitable HIV cure solutions45,46,47,48, our analysis did not focus on targeting cure strategies to specific population groups based on behavior or HIV status. Instead, in agreement with the inclusion criteria for many HIV cure trials49 and the minimum requirement in the TPP9, our model assumes that a cure is administered to individuals on ART. Since most MSM with HIV in the Netherlands are diagnosed and receive effective treatment promptly upon diagnosis2,50, this assumption likely has minimal impact on our findings. Lastly, to assess whether certain cure scenarios would bring the epidemic closer to or further from elimination, we used HIV incidence as the primary outcome. While we also examined how cure interventions could reduce the burden on the healthcare system by lowering the number of people on ART, we did not assess their cost-effectiveness or the disability-adjusted life years they could prevent. A health economic evaluation of cure interventions—relative to the standard of care with lifelong daily ART regimens, as well as forthcoming long-acting injectable ART formulations—should be a focus of future research.
In summary, our study suggests that both HIV eradication and sustained HIV remission have the potential to reduce new HIV infections, contributing to the United Nations Sustainable Development Goal of ending the HIV epidemic, while transient HIV remission could increase infections if rebounds are not promptly monitored. These findings emphasize the need for further research to ensure strategic development and effective implementation of cure interventions.
Methods
Overview
A classical deterministic compartmental model was calibrated using sexual behavior and HIV surveillance data for MSM in the Netherlands. The model was validated against independent data not used in the fitting process. This calibrated model was then applied to explore the potential impact of an HIV cure on epidemic dynamics at the population level.
Data
Sexual behavior data
We used sexual behavior data from a cross-sectional survey conducted from October 2021 to June 202251. The survey aimed to assess the anticipated impact of an HIV cure on quality of life, sexual satisfaction, stigma, and sexual and preventive behaviors among people with HIV and key populations in the Netherlands. From this survey, we extracted and analyzed responses from n = 529 MSM participants who provided information on the number of sexual partners in the last six months and condom use, and hypothetical changes in these variables following the introduction of HIV remission and HIV eradication scenarios. Additional details on the use of behavioral data are provided in the Model Calibration section and Supplementary Material.
HIV surveillance data
In the Netherlands, care for people with HIV is provided by 24 designated HIV treatment centers. The Dutch HIV Monitoring Foundation (Stichting hiv monitoring, SHM) was appointed by the Ministry of Health, Welfare and Sport to continuously monitor the HIV epidemic in the Netherlands and report on all aspects of HIV care. Since 1998, SHM has collected data in the ATHENA cohort from over 98% of all people with HIV who are receiving care in the Netherlands52. SHM annually reports on the number of new HIV diagnoses and publishes estimates of the number of newly acquired HIV infections and the undiagnosed population. We used the SHM data for MSM from 2015 to 2022 on the annual number of new HIV diagnoses, the annual number of individuals on ART immigrating to the Netherlands from abroad, and ART coverage2. We also used the SHM estimates of undiagnosed HIV infections, calculated using the European Centre for Disease Prevention and Control HIV Platform tool2,53. Data on the number of PrEP users in the national PrEP program from the national sexually transmitted infections surveillance database54, available from June 2019 to April 2022, were taken from ref. 37.
Transmission model
Model without cure
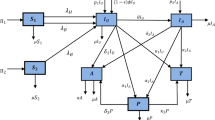
The model described the sexual transmission of HIV among MSM in the Netherlands. The population was stratified into four risk groups based on the average number of new sexual partners per year, cl38. We denoted risk groups with subscript l = 1, 2, 3, 4, where l = 1 represented the group with the lowest number of partners and l = 4 with the highest. Individuals did not change risk groups. The population in risk group l was further stratified by disease status into different compartments as shown in Fig. 2.
Susceptible individuals were recruited into the sexually active population at an overall rate of βN0 and were assigned to risk group l with a probability ql, where N0 is the initial population size, β is the rate of entry into the sexually active population, and ql is the initial fraction of individuals in risk group l. Cessation of sexual activity occurred at rate μ. In addition, we modeled the immigration of individuals to the Netherlands from abroad who were undiagnosed or already on ART and assigned them to risk group l with a probability Ql. Imported undiagnosed infections were assigned to HIV stage k, where k = 1, 2, 3, 4, with a probability pk.
In each risk group l, susceptible individuals acquired HIV at a time-dependent rate Jl(t) (i.e., the force of infection) through sexual contacts with the infectious individuals. Upon infection, they progressed through four stages: acute (k = 1), chronic (k = 2), AIDS with severe symptoms (k = 3), and AIDS without sexual activity (k = 4). These stages were characterized by different progression rates ρk and infectivities. Individuals in the last stage died from HIV at rate ρ4. Infected individuals in stage k were diagnosed at rate τk. Once diagnosed, they either began ART at rate η or died from HIV at rate ρD. The average lifespan in the infected undiagnosed and diagnosed untreated compartments was assumed to be equal (i.e., 1/ρD = \(\mathop{\sum }_{k=1}^{4}1/{\rho }_{k}\), see Table S2). The infectivity of diagnosed individuals was computed as a weighted average of the infectivity of undiagnosed individuals, with weights proportional to the duration of the respective infectious stages. Individuals on ART left the population at rate γ due to a shorter lifespan compared to susceptible individuals. Note that the category of individuals on ART includes not only virally suppressed individuals but also those who are not virally suppressed due to recent treatment initiation, imperfect treatment adherence, treatment failure, or dropout. In the Netherlands, the rate of discontinuation of ART among MSM is low, and a variety of next-line ART regimens are available in the event of ART failure. To reflect this, we did not explicitly model ART discontinuation or failure, considering instead that individuals on ART had very low, but non-negligible infectivity. Susceptible individuals in risk groups l = 3, 4 started using PrEP at rates \({k}_{l}^{\,{\mbox{on}}\,}(t)\) and discontinued it at rate koff. PrEP reduced the risk of infection with an effectiveness of (1 − Ω). Individuals who acquired HIV while on PrEP had lower infectivity and were diagnosed more rapidly (at rate τP) than individuals who did not use PrEP due to 3-monthly HIV testing.
Cure scenarios
The schematics of the model for HIV remission and HIV eradication are shown in Fig. 2. In both cure scenarios, all individuals on ART, regardless of their risk group, were eligible for a cure. The rollout of the cure was modeled as a time-dependent uptake rate, α(t), which grew logistically from the start of the rollout in 2026 until it reached a maximum uptake rate, \({\alpha }^{\max }\), within 3 years. The efficacy of the cure, e, was defined as the proportion of individuals on ART for whom a cure intervention was successful (i.e., who achieved HIV remission or HIV eradication). Individuals who did not achieve a cure or did not take up a cure intervention remained on ART.
Individuals who achieved HIV remission (Fig. 2a) experienced a viral rebound at a constant rate ϕ. We assumed that, upon rebound, individuals entered the chronic HIV stage, similar to what occurs during analytical treatment interruption studies41,42, and continued through disease progression at rates ρk. They were monitored and diagnosed at rates \({\tau }_{k}^{C}\), depending on the specific stage k, or died from HIV at rate ρ4. Since the viral reservoir was not completely removed by the intervention, we assumed that individuals in HIV remission died at the same rate as individuals on ART. After achieving HIV eradication (Fig. 2b), individuals were assumed to become fully susceptible and acquired HIV at the same rate Jl(t) as naive susceptible individuals. Cured individuals in risk groups l = 3, 4 began using PrEP at rates \({k}_{l}^{\,{\mbox{on}}\,}(t)\) and discontinued it at rate koff. Upon re-infection, these individuals entered the acute stage, similar to what occurs with superinfection by a different subtype43, and followed the same disease progression and diagnosis patterns as those in the HIV remission scenario.
Model implementation
The model was implemented in R with the RStudio interface (version 2022.12.0+353) using a system of ordinary differential equations for the number of individuals in different compartments. The equations are provided in the Supplementary Material, along with a table describing all model parameters (Table S2) and sensitivity analyses related to key model assumptions.
Model calibration
The model was calibrated in three steps: (i) sexual partner change rates were estimated from the sexual behavior survey data; (ii) diagnosis rates were estimated from the SHM data on the proportions of diagnoses by time since acquiring HIV; (iii) other parameters were estimated from fitting the model to the SHM data on the annual number of HIV diagnoses and the estimated number of undiagnosed HIV infections from 2017 to 2022. While undiagnosed infections are not directly observed, including estimates of this data stream in the model fit is essential for parameter identifiability. Other observed data, such as ART coverage, not included in the fitting process, were used for model validation (Supplementary Fig. S1).
Sexual partner change rates
The average number of new sexual partners per year in risk group l, cl, where l = 1, 2, 3, 4, was estimated by fitting a probability density function of a Weibull distribution to the sexual behavior data using the maximum likelihood method, as described in detail in ref. 55. A full explanation of the estimation procedure is reported in the Supplementary Material. The estimated sexual partner change rates are given Table S2 and Fig. S22.
In the main analyses presented in Figs. 1–6, we assumed that sexual partner change rates remained the same after the introduction of the cure and reflected survey data for the no-cure scenario. In the sensitivity analyses, we relaxed this assumption and considered an increase in sexual risk behavior, as estimated from survey data.
Diagnosis rates
We fixed the rates of diagnosis in AIDS stages, τ3 and τ4, so that the average time until an AIDS diagnosis was 1 month. The rates of diagnosis in the acute and chronic stages, τ1 and τ2, were estimated prior to fitting the model to the epidemiological data. To this end, we used a simple stochastic model that simulated the dynamics of disease progression and diagnosis, aiming to reproduce the proportions of diagnoses, among all new HIV diagnoses, within 6 months, between 6 to 12 months, and more than 12 months since acquiring HIV, based on the SHM data2 (Fig. S23). The detailed estimation procedure is described in the Supplementary Material. The estimated diagnosis rates are given in Table S2 and Fig. S24.
Other estimated parameters
The estimated parameters were the probability of transmission per sexual partner, λ; the parameter describing mixing between risk groups, ω; the relative infectivity of diagnosed and treated individuals, ε; the number of undiagnosed individuals immigrating to the Netherlands from abroad per year MI; the initial number of undiagnosed individuals for model burn-in, U0; and the probability of initial and imported HIV infections being assigned to risk group l, Ql, where l = 1, …, 4. These parameters were estimated by fitting the model to the SHM data on new HIV diagnoses and the estimated number of undiagnosed HIV infections from 2017 to 2022, using an Approximate Bayesian Computation approach based on Latin Hypercube Sampling. The detailed procedure for obtaining the posterior distributions of all estimated parameters is presented in the Supplementary Material. The estimated parameters are given in Table S2 and Fig. S25. The remaining model parameters were fixed based on the literature (Table S2).
Varied cure parameters
The parameters varied in both cure scenarios were efficacy, e, and maximum annual uptake percentage, \({\bar{\alpha }}^{\max }\). In the HIV remission scenario, we also varied the average time until viral rebound, 1/ϕ. The maximum annual uptake percentage was calculated as \({\bar{\alpha }}^{\max }=\left[1-\exp (-{\alpha }^{\max }\times 1\,{\mbox{year}})\right]100\%\), where \({\alpha }^{\max }\) is the maximum uptake rate per year used in the model equations.
Monitoring strategies were defined by varying the diagnostic delay for rebounds/re-infections in infection stage k, \(1/{\tau }_{k}^{C}\): (i) No monitoring, a diagnostic delay as estimated under the current standard of care: \(1/{\tau }_{k}^{C}=1/{\tau }_{k}\) for all k with τk values taken from the estimated posterior distribution, resulting in an average (across all HIV stages) diagnostic delay of 28 months. (ii) PrEP-like monitoring: \(1/{\tau }_{1,2}^{C}=1/4\) year (acute and chronic stages) and \(1/{\tau }_{3,4}^{C}=1/12\) year (AIDS stages), resulting in an average diagnostic delay of 3 months, recommended for PrEP users in the Netherlands. (iii) Frequent monitoring: \(1/{\tau }_{k}^{C}=1/24\) for all k, yielding an average diagnostic delay of 0.5 months.
Model outcomes
The model outcomes were annual new HIV infections (primary infections in naive individuals), the change in cumulative HIV infections compared to the no-cure scenario from 2026 to 2036, the contribution of different groups to new HIV infections, and the cumulative person-years on ART averted over 10 years relative to the no-cure scenario. Estimates of HIV incidence presented in this study referred to infections acquired in the country, excluding importations from abroad. For HIV remission, we estimated annual and cumulative viral rebounds; for HIV eradication, we estimated annual and cumulative re-infections (secondary infections in cured individuals) over the same period. For HIV remission, HIV prevalence was calculated as the ratio of the sum of individuals who are infected, diagnosed, treated, and in remission and the total population size. Individuals in remission were included in the prevalence calculation because their HIV reservoir is not completely removed. For HIV eradication, the compartments considered for HIV prevalence calculation were infected, diagnosed, and treated. Cure coverage was calculated as the proportion of cured individuals among all eligible (all treated and cured compartments). All model projections were reported as mean values with 95% credible intervals based on 100 samples from the joint posterior parameter distribution.
Sensitivity analyses
In the sensitivity analyses provided in the Supplementary Material, we explored the impact on model projections of (i) importation of infected individuals, (ii) the year of cure introduction, (iii) time horizon of scenario simulations, (iv) infectivity of individuals after rebound and re-infection, (v) risk compensation following cure introduction, (vi) the underlying trend in HIV incidence, (vii) the distribution of the time to rebound, (viii) susceptibility to re-infection of individuals achieving HIV remission and HIV eradication, and (ix) duration of survival of individuals achieving HIV remission.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The study that collected survey data received ethical approval from the University Medical Center Utrecht (UMC Utrecht) ethics committee (20-546/C). All datasets analyzed and generated in this study are available at https://github.com/alfredodebellis/HIVcure_rev56.
Code availability
The codes reproducing the results of this study are available at https://github.com/alfredodebellis/HIVcure_rev56.
References
The Joint United Nations Programme on HIV/AIDS (UNAIDS). Understanding fast-track accelerating action to end the AIDS epidemic by 2030 https://www.unaids.org/sites/default/files/media_asset/201506_JC2743_Understanding_FastTrack_en.pdf (2015).
van Sighem, A. et al. Monitoring Report 2023. Human Immunodeficiency Virus (HIV) infection in the Netherlands. Amsterdam: Stichting HIV monitoring https://www.hiv-monitoring.nl (2023).
Brizzi, F. et al. Tracking elimination of HIV transmission in men who have sex with men in England: a modelling study. Lancet HIV 8, e440–e448 (2021).
Palk, L., Gerstoft, J., Obel, N. & Blower, S. A modeling study of the Danish HIV epidemic in men who have sex with men: travel, pre-exposure prophylaxis and elimination. Sci. Rep. 8, 16003 (2018).
McCormack, S. et al. Pre-exposure prophylaxis to prevent the acquisition of HIV-1 infection (PROUD): effectiveness results from the pilot phase of a pragmatic open-label randomised trial. Lancet 387, 53–60 (2016).
Cohen, M. S. et al. Prevention of HIV-1 infection with early antiretroviral therapy. N. Engl. J. Med 365, 493–505 (2011).
Ndung’u, T., McCune, J. M. & Deeks, S. G. Why and where an HIV cure is needed and how it might be achieved. Nature 576, 397–405 (2019).
Dybul, M. et al. The case for an HIV cure and how to get there. Lancet HIV 8, e51–e58 (2021).
Lewin, S. R. et al. Multi-stakeholder consensus on a target product profile for an HIV cure. Lancet HIV 8, e42–e50 (2021).
Langebeek, N. et al. Impact of comorbidity and ageing on health-related quality of life in HIV-positive and HIV-negative individuals. AIDS 31, 1471–1481 (2017).
Zeluf-Andersson, G. et al. Beyond viral suppression: the quality of life of people living with HIV in Sweden. AIDS Care 31, 403–412 (2019).
van Bilsen, W. P. H., Zimmermann, H. M. L., Boyd, A. & Davidovich, U. Burden of living with HIV among men who have sex with men: a mixed-methods study. Lancet HIV 7, e835–e843 (2020).
de los Rios, P. et al. Physical, emotional, and psychosocial challenges associated with daily dosing of HIV medications and their impact on indicators of quality of life: findings from the positive perspectives study. AIDS Behav 25, 961–972 (2021).
Romijnders, K. A. et al. The perceived impact of an HIV cure by people living with HIV and key populations vulnerable to HIV in the Netherlands: a qualitative study. J. Virus Erad. 8, 100066 (2022).
Romijnders, K. A. G. J. et al. The experienced positive and negative influence of HIV on quality of life of people with HIV and vulnerable to HIV in the Netherlands. Sci. Rep. 12, 21887 (2022).
Schou, M. D., Søgaard, O. S. & Rasmussen, T. A. Clinical trials aimed at HIV cure or remission: new pathways and lessons learned. Expert Rev. Anti-infective Ther 21, 1227–1243 (2023).
Treatment Action Group. Research toward a cure trials www.treatmentactiongroup.org/cure/trials (2024).
Khan, A., Paneerselvam, N. & Lawson, B. R. Antiretrovirals to CCR5 CRISPR/Cas9 gene editing-A paradigm shift chasing an HIV cure. Clin. Immunol. 255, 109741 (2023).
Deeks, S. G. et al. Research priorities for an HIV cure: International AIDS society global scientific strategy 2021. Nat. Med. 27, 2085–2098 (2021).
Power, J. et al. The significance and expectations of HIV cure research among people living with HIV in Australia. PLoS One 15, 1–15 (2020).
Pitman, M. C., Lau, J. S. Y., McMahon, J. H. & Lewin, S. R. Barriers and strategies to achieve a cure for HIV. Lancet HIV 5, e317–e328 (2018).
Vansant, G., Bruggemans, A., Janssens, J. & Debyser, Z. Block-and-lock strategies to cure HIV infection. Viruses 12, 84 (2020).
Bailon, L., Mothe, B., Berman, L. & Brander, C. Novel approaches towards a functional cure of HIV/AIDS. Drugs 80, 859–868 (2020).
Landovitz, R. J., Scott, H. & Deeks, S. G. Prevention, treatment and cure of HIV infection. Nat. Rev. Microbiol. 21, 657–670 (2023).
Garnett, G. P. An introduction to mathematical models in sexually transmitted disease epidemiology. Sex. Transm. Infect. 78, 7–12 (2002).
Giddings, R. et al. Infectious disease modelling of HIV prevention interventions: a systematic review and narrative synthesis of compartmental models. PharmacoEconomics 41, 693–707 (2023).
Hallett, T. B. et al. Using modeling to inform international guidelines for antiretroviral treatment. AIDS 28, S1–S4 (2014).
Granich, R. M., Gilks, C. F., Dye, C., De Cock, K. M. & Williams, B. G. Universal voluntary HIV testing with immediate antiretroviral therapy as a strategy for elimination of HIV transmission: a mathematical model. Lancet 373, 48–57 (2008).
Dimitrov, D. T., Kiem, H.-P., Jerome, K. R., Johnston, C. & Schiffer, J. T. A curative regimen would decrease HIV prevalence but not HIV incidence unless targeted to an ART-naíve population. Sci. Rep. 6, 22183 (2016).
Beacroft, L. & Hallett, T. B. The potential impact of a “curative intervention” for HIV: a modelling study. Glob. Health Res. Policy 4, 1–8 (2019).
The Joint United Nations Programme on HIV/AIDS (UNAIDS). UNAIDS Key Population Atlas https://kpatlas.unaids.org/dashboard (2024).
Nederlandse multidisciplinaire richtlijn. Pre-expositie profylaxe (PrEP) ter preventie van HIV (2022). https://www.soaaids.nl/files/2022-07/20220711-PrEP-richtlijn-Nederland-versie-3-update-2022.pdf. Accessed August 18, 2024.
Stecher, M. et al. Systematic review and meta-analysis of treatment interruptions in human immunodeficiency virus (HIV) type 1–infected patients receiving antiretroviral therapy: implications for future HIV cure trials. Clin. Infect. Dis. 70, 1406–1417 (2020).
Freedberg, K. A. et al. The HIV cure research agenda: the role of mathematical modelling and costeffectiveness analysis. J. Virus Erad. 1, 245–249 (2015).
Noorman, M. A. J. et al. Towards inclusive HIV cure development: Identifying important and acceptable HIV cure attributes and strategies among people with HIV in The Netherlands. Submitted (2025).
Newsum, A. M. et al. Incidence of HCV reinfection among HIV-positive MSM and its association with sexual risk behavior: a longitudinal analysis. Clin. Infect. Dis. 73, 460–467 (2020).
Reitsema, M. et al. Impact of varying pre-exposure prophylaxis programs on HIV and Neisseria gonorrhoeae transmission among MSM in the Netherlands: a modeling study. JAIDS J. Acquired Immune Defic. Syndromes 97, 325–333 (2024).
Rozhnova, G. et al. Elimination prospects of the dutch HIV epidemic among men who have sex with men in the era of preexposure prophylaxis. AIDS 32, 2615–2623 (2018).
Nichols, B. E., Boucher, C. A., van der Valk, M., Rijnders, B. J. & van de Vijver, D. A. Cost-effectiveness analysis of pre-exposure prophylaxis for HIV-1 prevention in the Netherlands: a mathematical modelling study. Lancet Infect. Dis. 16, 1423–1429 (2016).
Jansen, I. A. et al. Ongoing HIV-1 transmission among men who have sex with men in Amsterdam: a 25-year prospective cohort study. AIDS 25, 493-501 (2011).
Pilcher, C. D. et al. Amplified transmission of HIV-1: comparison of HIV-1 concentrations in semen and blood during acute and chronic infection. AIDS 21, 1723–1730 (2007).
Castagna, A. et al. Analytical treatment interruption in chronic HIV-1 infection: time and magnitude of viral rebound in adults with 10 years of undetectable viral load and low HIV-DNA (APACHE study). J. Antimicrobial Chemother 74, 2039–2046 (2019).
van der Kuyl, A. C. & Cornelissen, M. Identifying HIV-1 dual infections. Retrovirology 4, 1–12 (2007).
Lambers, F. A. et al. Alarming incidence of hepatitis C virus re-infection after treatment of sexually acquired acute hepatitis C virus infection in HIV-infected MSM. AIDS 25, (2011).
Lo, B., Grady, C. & on Ethics of the International AIDS Society, W. G. et al. Ethical considerations in HIV cure research: points to consider. Curr. Opin. HIV AIDS 8, 243–249 (2013).
Dubé, K. et al. Research on HIV cure: mapping the ethics landscape. PLoS Med 14, e1002470 (2017).
Dubé, K. et al. Ethics of HIV cure research: an unfinished agenda. BMC Med. Ethics 22, 1–14 (2021).
Grossman, C. I. et al. Towards multidisciplinary HIV-cure research: integrating social science with biomedical research. Trends Microbiol 24, 5–11 (2016).
Ismail, S. D. et al. Addressing an HIV cure in LMIC. Retrovirology 18, 21 (2021).
Dijkstra, M. et al. Decreased time to viral suppression after implementation of targeted testing and immediate initiation of treatment of acute human immunodeficiency virus infection among men who have sex with men in Amsterdam. Clin. Infect. Dis. 72, 1952–1960 (2021).
Romijnders, K. et al. The expected impact of HIV post-treatment control and HIV elimination on quality of life, sexual satisfaction, and stigma among people with HIV and key populations. Accepted for publication in Communications Medicine. https://doi.org/10.21203/rs.3.rs-4311908/v1 (2024).
Boender, T. S. et al. AIDS therapy evaluation in the Netherlands (ATHENA) national observational HIV cohort: cohort profile. BMJ Open 8, e022516 (2018).
ECDC HIV Platform tool [Software Application]. Version 3.0.3 stockholm: European centre for disease prevention and control https://www.ecdc.europa.eu/en/publications-data/hiv-platform-tool (2021).
Kayaert, L. et al. Sexually transmitted infections in the Netherlands 2022. Bilthoven: National Institute for Public Health and the Environment, RIVM https://www.rivm.nl/bibliotheek/rapporten/2023-0161.pdf (2023).
Rozhnova, G., van der Loeff, M. F. S., Heijne, J. C. M. & Kretzschmar, M. E. Impact of heterogeneity in sexual behavior on effectiveness in reducing HIV transmission with test-and-treat strategy. PLoS Comput. Biol. 12, e1005012 (2016).
De Bellis, A. et al. Model-based evaluation of the impact of a potential HIV cure on HIV transmission dynamics. hivcure_rev v1.0.0 - public release https://doi.org/10.5281/zenodo.15031608 (2025).
Acknowledgements
The authors gratefully acknowledge funding by the Aidsfonds Netherlands, grant number P-53902, awarded to G.R., and funding by Aidsfonds & NWO, the SPIRAL project KICH2.V4P.AF23.001, awarded to G.R. and M.N. The survey data collection was funded by the Aidsfonds Netherlands, grant number P-52901, awarded to G.R. We extend our gratitude to the survey participants and thank the staff of the Amsterdam Cohort Studies, the AGEhIV Cohort Study, and the infectious diseases outpatient clinic at the University Medical Center Utrecht for their assistance in participant recruitment. We extend our sincere gratitude to Thijs Albers, Bertus Tempert, and Renee Finkenflügel from the Dutch HIV Association for their valuable contributions. We also appreciate the valuable discussions with Franco Romero Gonzalez, Maartje Dijkstra, Maartje Basten, Laura de Groot, Sigrid Vervoort, Berend van Welzen, Udi Davidovich, Amy Matser, Myrthe Verburgh, Pythia Nieuwkerk, Godelieve de Bree, and Janneke Heijne.
Author information
Authors and Affiliations
Contributions
G.R. conceived and supervised the study. A.D.B. and G.R. developed the mathematical model. A.D.B., G.R., and G.G. developed the fitting procedure. K.A.G.J.R. and A.v.S. collected and provided data. K.A.G.J.R., P.R., M.F.S.v.d.L., A.D.B., M.N., and G.R. developed cure scenarios. M.S.W. performed a literature review. A.D.B. conducted analyses and prepared figures. A.D.B., M.S.W., and G.R. wrote the manuscript. A.D.B. and M.S.W. prepared the resubmission supported by G.R. and G.G. A.D.B., M.S.W., G.G., A.v.S., K.A.G.J.R., P.R., M.F.S.v.d.L., J.H.H.M.v.d.W., M.N., M.E.E.K., and G.R. contributed to the interpretation of the results, commented on the manuscript, and gave final approval for publication.
Corresponding authors
Ethics declarations
Competing interests
A.v.S. reports grants, paid to his institution, from the Dutch Ministry of Health, Welfare and Sport through the Centre for Infectious Disease Control of the National Institute for Public Health and the Environment, and from the European Centre for Disease Prevention and Control (ECDC). All other authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Daniel Reeves and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
De Bellis, A., Willemsen, M.S., Guzzetta, G. et al. Model-based evaluation of the impact of a potential HIV cure on HIV transmission dynamics. Nat Commun 16, 3527 (2025). https://doi.org/10.1038/s41467-025-58657-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-58657-x