Abstract
Eosinophils are innate immune cells with central roles in allergy, parasitic diseases and multiple inflammatory conditions. Moreover, their role in host-pathogen interactions has been well characterized. However, the role of eosinophils during fungal infection is poorly defined. In this study, we delineate the importance of eosinophils during C. albicans systemic infections. C. albicans is promptly phagocytosed by human eosinophils, but growing hyphae escape this mechanism by releasing the fungal toxin candidalysin, which causes eosinophil membrane damage and cell death. Concomitantly, eosinophil mediators, notably major basic protein 1 (MBP-1), released during cytolysis, inhibits C. albicans growth and viability. Moreover, systemic candidiasis in genetic (Δdbl/GATA) or anti–IL-5–mediated depletion of eosinophils results in increased fungal burden and decreased survival. We here identified CD48 as a major receptor of eosinophils and possibly of other immune cells involved in the recognition of C. albicans via agglutinin-like sequence 6 (Als6). CD48 is important for protection in a model of systemic candidiasis as shown in CD48−/− mice and it binds clinical isolates of C. albicans. In conclusion, we have defined a protective role for eosinophils in vitro and in mouse C. albicans infections through CD48/Als6 host-pathogen interaction axis.
Similar content being viewed by others
Introduction
Worldwide, invasive fungal diseases (IFD) affect increasing numbers of individuals, both immunocompromised (i.e. HIV) and immunocompetent patients, leading to a staggering estimate of 1.5 million deaths annually due to underdiagnosis and lack of effective treatments1. To foster research and intervention in this critical field, the World Health Organization (WHO) has established the first fungal pathogens priority list2. Candida albicans (C. albicans) and Candida auris (C. auris) together with Aspergillus fumigatus (A. fumigatus) and Cryptococcus neoformans constitute the “critical priority group”, while two other Candida species (C. parapsilosis and C. tropicalis) are among the seven members of the “high priority group”2. The commensal organism C. albicans, is a double-edged sword—contributing to both anti-fungal immunity3 and inflammatory responses. Additionally, its ability to switch between yeast and filamentous/ hyphae forms, which is linked to changes in virulence, highlights significant challenges in achieving accurate diagnosis and effective infection management.
In systemic candidiasis, innate immune cells such as neutrophils, macrophages, and natural killer (NK) cells are recruited to infected organs4 to clear the fungus5. C. albicans expresses mannoproteins that are typically recognized by innate immune cells via toll-like receptor 2 (TLR-2) and TLR-4. The dectin family receptors (dectin1/2/3) are also involved in the anti-fungal innate immunity by the recognition of β-glucans and α-mannans6. Moreover, C. albicans agglutinin-like sequence (Als) proteins were recently shown to bind the T cell immunoreceptor with Ig and ITIM domains (TIGIT) receptor and modulate the immune response of NK and T cells7.
Eosinophils are innate immune granulocytes, mainly known for their role in pathological conditions such as allergic diseases and parasitic infections8. Although eosinophil activation has been described mostly with allergenic fungi and therefore in the context of allergic diseases, a few studies indicate eosinophil function in fungal infections9. Recent data support a broader role for eosinophils in homeostasis10,11 and in the immune surveillance against bacterial, viral, and fungal infections12. Eosinophilia and candidiasis coexist in STAT3-loss of function hyper-IgE syndromes13, in pediatric IFD14, and in various other settings15,16,17. Moreover, a clinical study found a significant association between absolute eosinophil count and serum (1,3)-β-D-glucan in patients with invasive fungal infections, comprising 10% with invasive candidiasis and 50% of these with C. albicans strains18, suggesting a possible role for eosinophils in systemic candidiasis.
Eosinophils express several receptors that sense and bind pathogens, including TLR-2, TLR-419 and CD4820. Upon activation, eosinophils release granular lytic mediators including eosinophils peroxidase (EPX), eosinophil cationic protein (ECP), eosinophil-derived neurotoxin (EDN), and major basic protein 1 and 2 (MBP-1/2)21. Eosinophils also produce and release lipid membrane metabolites and an array of pre-formed and de novo synthesized pro-inflammatory cytokines and chemokines11. Eosinophils can also phagocytose several pathogens22 and form both nuclear and mitochondrial eosinophil extracellular DNA traps (EETs)23. Indeed, eosinophils were demonstrated more than 50 years ago to phagocytose C. albicans24, and more recently also to release EETs25 in response to this fungus.
In the present study, we show that recognition of C. albicans by human eosinophils leads to phagocytosis of the fungus. However, growing hyphae escape from the cells by secreting candidalysin, a recently described fungal toxin26 that induces damage to the eosinophil membrane, resulting in eosinophil cytolysis. Notably, the release of MBP-1 during this lytic process affects fungal growth and reduces fungal viability. In addition, we identified CD48 as a receptor, expressed by eosinophils and other leukocytes as well, that interacts with Als6 on C. albicans, leading to inhibition of fungal growth in vitro and in vivo. Together, our results delineate a possible role for eosinophils in invasive systemic candidiasis.
Results
Eosinophils affect C. albicans growth in a Candida-induced eosinophil death mechanism
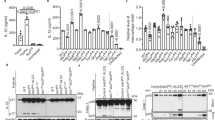
To determine the outcome of the interaction of eosinophils and C. albicans, human peripheral blood eosinophils were isolated and cultured with C. albicans in their yeast form at different multiplicities of infection (MOI) and time points. Eosinophil activation was assessed by measuring EPX release as a marker of degranulation. Significant levels of EPX were released after 3 h of incubation at a MOI of 1 (Fig. 1A), but not after 45 min (Fig. S1A). By observing trypan blue positive cells and measuring the release of lactate dehydrogenase (LDH) into the supernatants as a read-out for cell damage, we showed that C. albicans induced significant eosinophil death by 3 h at a MOI of 1 and after 20 h also at MOI of 0.1 (Fig. 1B, C). Nevertheless, eosinophil incubation with C. albicans significantly reduced the fungal viability by 60% as evaluated by colony-forming unit (CFU) count. Notably, this effect was observed after 20 h incubation (Fig. 1D) but not after 3 h (Fig. S1B). To assess whether eosinophil mediators released after interacting with C. albicans affect the viability of the fungus, supernatants of eosinophils incubated overnight alone or with C. albicans were added to fresh C. albicans cultures. A significant decrease in C. albicans CFU was observed only with the supernatants of eosinophils cultured with C. albicans, confirming that eosinophil released mediators affect C. albicans viability (Fig. 1E). Next, we assessed which eosinophil granule basic mediator was responsible for affecting C. albicans viability. We exposed C. albicans to several purified eosinophil granule proteins (EDN, MBP-1, EPX, ECP) at a concentration equivalent to the content found in 105 eosinophils21,27 (as used in the co-culture experiments described above) and found MBP-1 to be the most potent eosinophil basic protein responsible for the reduction of C. albicans viability, after 3 (Fig. S1C) and 20 h (Fig. 1F). Notably, C. albicans incubated with the four mediators combined reduced the CFU similarly to the eosinophils themselves (Fig. 1F).
A Eosinophil peroxidase (EPX) release in supernatants of eosinophils co-cultured with C. albicans (Ca) for 3 h at the indicated multiplicity of infection (MOI). EPX levels were assessed by a colorimetric assay and quantified using purified human EPX. Data is representative of 1 out of 4 experiments performed with eosinophils obtained from 4 independent donors. Each experiment was performed in technical triplicates. B, C Viable eosinophil numbers (B) and LDH levels (C) following incubation of eosinophils with C. albicans (MOI 0.1 and 1) for 0.5, 3, and 20 h. Viable cells were assessed by trypan blue exclusion test, and LDH was evaluated in supernatants by colorimetric assay; n = 3 biological replicates. D Colony forming unit (CFU) counts of C. albicans incubated with eosinophils (MOI 1) or in medium for 20 h; n = 3 biological replicates. Each experiment was performed in technical duplicates. E CFU counts of C. albicans incubated with supernatants of eosinophils following overnight incubation with medium alone (Eos sup) or with C. albicans (MOI 1; Eos+ Ca sup); n = 3 biological replicates. Each experiment was performed in technical triplicates. F CFU counts of C. albicans incubated with eosinophils (MOI 1) or with purified eosinophil granule proteins or with their combination for 20 h. Eosinophil granule proteins were added at a concentration corresponding to their content in 105 eosinophils; n = 3 biological replicates. Each experiment was performed in technical triplicates (G) Fluorescent microscopy image of eosinophils incubated with FITC-labeled C. albicans (MOI 1) for 30 min. Eosinophil nuclei were stained with DAPI. White arrows show engulfment of C. albicans by eosinophils (×400 magnification). H Scanning electron microscopy (SEM) image of eosinophils incubated with C. albicans (MOI 1) for 30 min. White arrows show the engulfment of C. albicans by eosinophils and red arrows show eosinophil extracellular traps (×10,000 magnification). I EPX release in supernatants of naive or cytochalasin D-pretreated (5 µg/ml) eosinophils incubated for 3 h with C. albicans (MOI 1); n = 3 biological replicates. J CFU counts of C. albicans incubated for 20 h with naive or cytochalasin D-pretreated (5 µg/ml) eosinophils (MOI 1); n = 3 biological replicates. Each experiment was performed in technical triplicates. K, L EPX (K) and LDH (L) release in supernatants of eosinophils co-cultured respectively for 3 and 20 h with WT C. albicans (WT), re-integrant strain (ece1Δ/Δ + ECE1), candidalysin mutant strains (ece1Δ/Δ, ece1Δ/Δ + ECE1Δ184–279; all at MOI 1) or with synthetic candidalysin at the indicated concentrations. Data is representative of 1 out of 3 experiments with eosinophils obtained from 3 independent donors. Each experiment was performed in technical triplicates. Data show the mean ± SD. Significance was analyzed by one-tail Anova (A, B, F, I–L) or two-tailed student t test (C–E) as described in “Materials and Method” section; ns not-significant, *P ≤ 0.05; **P ≤ 0.01; ***P ≤ 0.005; ****P ≤ 0.001.
To investigate the mechanism of eosinophil-induced C. albicans killing, host-fungal interaction was investigated by confocal fluorescence and scanning electron microscopy (SEM) analyses. Eosinophils phagocytosing C. albicans were observed 30 min after co-culture (Fig. 1G, H) together with EETs, that were visualized surrounding the yeast (Fig. 1H). Importantly, after 3 h of incubation (MOI 1), phagocytosed C. albicans was still capable of forming hyphae, thereby inducing damage to the eosinophils, resulting in cell death (Supplemental Movie 1). This correlated with the increase in LDH levels as described above (Fig. 1C). Therefore, we hypothesized that phagocytosis of C. albicans was the inducing mechanism of eosinophil mediator release as well as C. albicans death. To elucidate this, eosinophils were pre-treated with cytochalasin D, a well-characterized cytoskeleton inhibitor, and incubated with C. albicans. However, only a slight decrease in degranulation (Fig. 1I) and a minor increase in C. albicans viability (CFU; Fig. 1J) were detected in treated eosinophils, in comparison to untreated cells. Importantly, confocal microscopy showed that cytochalasin D treated eosinophils still interacted with C. albicans but did not phagocytose the fungus (Supplemental Movie 2).
Finally, we wondered if candidalysin, a fungal toxin secreted by C. albicans during hyphae formation, might be responsible for eosinophil activation and death. Therefore, we investigated the capacity of C. albicans mutants lacking candidalysin to induce eosinophil activation and death. Eosinophils were incubated with synthesized candidalysin, or mutant strains missing either the entire ECE1 gene (ece1Δ/Δ) or only the candidalysin-encoding sequence (ece1Δ/Δ + ECE1Δ184–279), with the control ECE1 re-integrant strain (ece1Δ/Δ + ECE1)26 or with the wild type (WT; SC3514) C. albicans strain. As shown by EPX levels (Fig. 1K), candidalysin triggered a dose-dependent degranulation of eosinophils. However, the candidalysin-deficient strains caused EPX released levels similar to those induced by C. albicans WT strain (Fig. 1K), suggesting that other mechanisms besides candidalysin may be involved in the degranulation of eosinophils. Moreover, eosinophils incubated with the wild type (WT) strain, or the different mutants showed a significant increase in LDH released after 20 h incubation. Interestingly only the highest candidalysin concentration (60 µM) induced a significant increase in LDH release (Fig. 1L).
Eosinophils decrease the fungal burden and improve survival in a murine model of systemic candidiasis
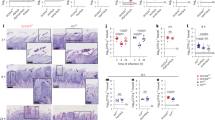
To investigate the role of eosinophils in systemic candidiasis in vivo, eosinophil deficient (ΔdblGATA) and WT mice were injected intravenously (i.v.) with 5 × 105 viable C. albicans yeast and euthanized 2 days post-infection (dpi). Fungal burdens were then assessed in infected organs (kidneys, liver, and spleen) by CFU (Fig. 2A). A significantly higher fungal load was found in the kidneys of the eosinophil deficient mice but not in the liver and the spleen (Fig. 2B). Kidneys removed from C. albicans infected mice were enlarged and appeared visually more infected in ΔdblGATA mice as compared with WT mice (Fig. S1D). Staining of the kidneys with hematoxylin and eosin (H&E) revealed few intact eosinophils (Fig. S1E) and immunofluorescence staining of eosinophils by anti-Siglec-F showed mostly debris of cells surrounding the hyphae (Fig. S1E). Furthermore, levels of IL-6, a general inflammation marker, and of IFN-γ, a type 1 cytokine effective for anti-fungal defense, were assessed 2 dpi. ΔdblGATA mice presented significantly elevated levels of IL-6 and decreased levels of IFN-γ in serum (Fig. 2C) and homogenized kidneys (Fig. 2D) in comparison to WT mice. In addition, ΔdblGATA mice showed a significantly decreased survival rate compared to WT mice (Fig. 2F). Finally, we found that incubation of human eosinophils with C. albicans for 3 h induces the secretion of IFN-γ (Fig. 2G).
A Diagram illustrating the systemic candidiasis model to assess the fungal burden (5 × 105 yeast/mouse). Created in BioRender. Zaffran, I. (2025) https://BioRender.com/9xkicks. B C. albicans CFU in the kidneys, liver, and spleen, of WT and Δdbl/GATA mice 2 days post-infection (dpi); n = 10 (WT) and 11 (Δdbl/GATA) mice/group for kidneys and liver samples and n = 9 for spleen samples, all collected from 3 independent experiments. C, D IL-6 (n = 7 for WT; n = 11 for Δdbl/GATA mice) and IFN-γ (n = 6) levels in serum (C) and IFN-γ levels in kidneys (D; n = 11 for WT; n = 14 for Δdbl/GATA mice) of C. albicans-infected WT and Δdbl/GATA mice. E Diagram illustrating the “survival” systemic candidiasis model (5 × 104 cells/mouse). Created in BioRender. Zaffran, I. (2025) https://BioRender.com/9xkicks. F Survival rate (percent survival) of WT (black curve) and Δdbl/GATA (red curve) mice; n = 12 mice/group in 2 independent experiments. G IFN-γ levels produced by human eosinophils culture in medium or with C. albican at 0.1 and 1 MOI for 3 h; n = 2 biological replicates. Data show the mean ± SEM (B–D) or mean ± SD (G). Significance was analyzed by two-tailed student t test (B–D and G), survival curve was analyzed by Log-rank (Mantel Cox) test; *P ≤ 0.05; **P ≤ 0.01.
CD48 binds C. albicans and regulates fungal growth
To investigate the mechanism involved in the interaction between human eosinophils and C. albicans, first we assessed the expression of TLR-2, TLR-4, and Dectin-1, which are involved in the innate immune response to C. albicans6. We also investigated CD48, an eosinophil surface protein that binds to bacterial surface molecules20. In line with previous observations, CD48 but not TLR-2, TLR-4, or Dectin-1 was found to be expressed on freshly isolated eosinophils19,28, (Fig. S2A). To evaluate whether C. albicans binds to CD48, we produced murine (mCD48-Fc) and human (hCD48-Fc) CD48 fusion proteins and used them to stain C. albicans yeast by flow cytometry (Fig. S2B). C. albicans effectively bound both human and mouse CD48 in comparison to the negative construct control (D1-Fc; Fig. 3A). Binding to hCD48-Fc and mCD48-Fc was further confirmed using plate-bound C. albicans (Fig. 3B) and visualized using immunofluorescence microscopy with both yeast (Fig. 3C) and hyphae (Fig. 3D).
A Flow cytometry staining of C. albicans with hCD48-Fc (red open histogram), mCD48-Fc (blue open histogram) or the negative control D1-Fc (gray filled histogram). Histograms are representative of 3 independent experiments. B Binding of. C. albicans to human and mouse CD48 as evaluated by ELISA. Plate-bound C. albicans were incubated with hCD48-Fc, mCD48-Fc, or the negative controlfig D1-Fc all at 1 and 1.5 µg/ml followed by incubation with secondary antibody, n = 3 biological replicates. Each experiment was performed in technical duplicates. C, D Immunofluorescence microscopy images of FITC-stained C. albicans yeast (C) and hyphae (D) stained with hCD48-Fc (red) or the negative control D1-Fc. Fungi were stained with FITC (green) and nuclei with DAPI (blue). Shown images are representative of 3 independent experiments. E Flow cytometry staining of C. tropicalis, C. parapsilosis, and C. lusitaniae with hCD48-Fc (red open histograms) or D1-Fc (black open histograms). Histograms are representative of 3 independent experiments. F EPX release in supernatants of eosinophils co-cultured for 3 h with C. albicans (Ca; MOI 1) following their pre-incubation with hCD48-Fc (red column) or D1-Fc (dark gray column; 5 µg/ml). Data is representative of 1 out of 3 experiments with eosinophils obtained from 3 independent donors. G p-Lyn expression in eosinophils incubated with C. albicans (Ca, MOI 1, blue open histogram) or in medium for 15 min. Protein expression was evaluated by intracellular flow cytometry staining. Black open Histogram shows the staining with IgG1 isotype control. The histograms are representative of 1 out of 3 experiments. The right panel represents the relative fluorescence of the pooled experiments. n = 3. H CFU counts of C. albicans incubated with CD48–/– or WT BMEos for 20 h; n = 3 biological replicates. Each experiment was performed in technical triplicates. I C. albicans CFU in the kidneys and liver of CD48–/– and WT mice 2 dpi. Fungal load was assessed by CFU as described in Fig. 2A. n = 12 for WT; n = 10 for CD48–/– mice, collected in 2 independent experiments. J IL-6 (n = 9 for WT; n = 8 for CD48–/– mice) and IFN-γ levels (n = 14 for WT; n = 16 for CD48–/– mice) in serum of C. albicans-infected WT and CD48–/– mice. IFN-γ was performed in technical duplicates. K Survival rate (percent survival) of WT and CD48–/– mice in the “survival” systemic infection model as described in Fig. 2E. n = 6 mice/group in 2 independent experiments. Data show the mean ± SEM (I and J) or mean ± SD (B, F–H). Significance was analyzed by one-tail Anova (B and F) or two-tailed student t test (G–J), survival curve was analyzed by Log-rank (Mantel Cox) test; **P ≤ 0.01; ***P ≤ 0.005, ****P ≤ 0.001.
Interestingly, other Candida species including C. lusitaniae and C. tropicalis, but not C. parapsilosis, also bound to CD48 (Fig. 3E), although to a far lesser degree than C. albicans.
To determine whether binding of C. albicans to CD48 could activate eosinophils, the degranulation of eosinophils induced by C. albicans preincubated with hCD48-Fc was measured. Under these conditions decreased EPX levels were observed in comparison to those released by eosinophils incubated with C. albicans alone (Fig. 3F). Moreover, eosinophil incubation with C. albicans significantly increased the phosphorylation of Lyn at 15 min, a signal transduction protein linked to CD48 (Fig. 3G), and slightly increased CD48 expression on eosinophils at 120 min (Fig. S2C).
To confirm the role of CD48 in C. albicans-eosinophil interactions, bone marrow derived eosinophils (BMEos) obtained from WT and CD48–/– mice (Fig. S2D) were incubated with C. albicans (MOI 1) for 20 h and fungal viability was assessed. As with human eosinophils, WT BMEos co-cultured with C. albicans reduced significantly the fungal CFU, while no difference in CFU was found when the fungus was incubated with CD48–/– BMEos (Fig. 3H), thus confirming a role for CD48 in C. albicans interactions with eosinophils.
Next, using CD48–/– mice, we evaluated the involvement of CD48 in systemic candidiasis. CD48–/– mice displayed a significant increase in fungal burden in the kidneys and a higher mortality rate than WT mice (Fig. 3I, K). Moreover, IL-6 but not IFN-γ levels were found to be significantly increased in the serum of the infected mice (Fig. 3J).
Antibody-mediated depletion of eosinophils increases the fungal burden of WT but not of CD48–/– mice
To determine whether the CD48-dependent effect of fungal clearance is specific for eosinophils, we depleted eosinophils by injecting anti-IL-5 or rat IgG1 (as isotype control; Fig. S3A) in WT and CD48–/– mice and assessed if this resulted in significant exacerbation of the infection. Similarly to ΔdblGATA mice, eosinophil depletion in WT mice led to a significantly higher fungal load in the kidneys (Fig. 4A). However, unlike WT mice, in which eosinophil depletion led to significantly higher fungal load, eosinophil depletion in CD48–/– mice did not result in significant elevation in CFU in comparison to isotype control treated CD48–/– mice, suggesting that CD48 expression on eosinophils is critical for their antifungal function. Notably, the fungal load in eosinophil-depleted CD48⁻/⁻ mice was significantly higher than in eosinophil-depleted WT mice, indicating that CD48 also plays an important role in antifungal defense through other CD48-expressing immune cells. Finally, IFN-γ levels in serum and kidney homogenates decreased following eosinophil depletion, confirming the production of IFN-γ by eosinophils (Fig. 4B, C).
A C. albicans CFU in the kidneys and liver of WT and CD48–/– mice injected with isotype control (rIgG1; n = 8 for kidneys; n = 5 for liver) or with anti-IL-5 (α-IL-5; n = 11 for kidneys; n = 7 for liver), at 2 dpi. B IL-6 (n = 8) and IFN-γ levels (n = 10) in serum. C IFN-γ levels in kidneys of WT (n = 8 and 11) and CD48–/– (n = 7) mice depleted for eosinophils. Data show the mean ± SEM. Significance was analyzed by two-tailed student t test; *P ≤ 0.05, **P ≤ 0.01.
CD48 recognizes Als6 as a ligand on C. albicans
We next sought to identify the fungal ligand of CD48. S. cerevisiae α-agglutinin protein has sequence characteristics of immunoglobulin (Ig)-like protein and homology to human CD2 protein, a low-affinity ligand of CD4829. Using a computational approach, we aligned the agglutinin sequences of S. cerevisiae and C. albicans with CD2 and detected 44% similarity between C. albicans agglutinin and CD2. Therefore, we assessed whether any of Als proteins could be CD48 ligand. For this purpose, we generated BW-hCD48 as reporter cells. BW (BW5147.3 cell line) cells were transfected with the extracellular domain of CD48 conjugated to mouse CD3ζ. Consequently, binding of CD48 led to the release of mouse interleukin-2 (mIL-2) detectable by enzyme-linked immunosorbent assay (ELISA)30. C. albicans knock out strains for each of the ALS gene family (als1-9Δ/Δ) or WT (SC5314) strains were incubated with either BW-hCD48 or BW parental cells as a control (Fig. S4A) for 48 h, and mIL-2 levels in the supernatant were assessed as read outs of CD48 binding (Fig. 5A). As expected, BW parental cells did not release mIL-2 while BW-hCD48 cells incubated with WT C. albicans released mIL-2. Notably, incubation with als5Δ/Δ, als6Δ/Δ, and als7Δ/Δ strains showed significantly decreased levels of mIL-2, indicating that these Als proteins could also act as biologically relevant ligands for CD48 (Fig. 5B). To confirm this result, we assessed by ELISA the binding of hCD48-Fc to plate bound WT, als5Δ/Δ, als6Δ/Δ, and als7Δ/Δ C. albicans strains. As with BW system, WT but not als5Δ/Δ, als6Δ/Δ, and als7Δ/Δ was found to bind hCD48-Fc (Fig. 5C), confirming that Als5, Als6, and Als7 are ligands for CD48. Focusing on Als6 as the most compelling interactor, we confirmed the binding of CD48 to Als6 by a lack of staining of als6Δ/Δ with hCD48-Fc by flow cytometry (Fig. 5D).
A Diagram illustrating the BW reporter assay employed. Binding of C. albicans to CD48 was evaluated by measuring levels of mIL-2 released in the supernatants of BW and BW-hCD48 cells. Created in BioRender. Zaffran, I. (2025) https://BioRender.com/9xkicks. B mIL-2 levels in supernatants of BW (white columns) and BW-hCD48 (dark columns) cells incubated with 2B4-Fc (positive control), WT (SC5314) or Als mutant C. albicans strains (als1-9Δ/Δ) for 48 h. n = 3 biological replicates. C Binding of hCD48-Fc or secondary antibody to plate bound WT, Als5, Als6, and Als7 mutant strains or to 2B4-Fc as positive control. Binding was assessed by ELISA at OD = 450 nm; n = 3 biological replicates. D Flow cytometry staining of WT and als6Δ/Δ C. albicans strains with hCD48-Fc (red open histogram) or the negative control D1-Fc (gray open histogram). Histograms are representative of 3 independent experiments. E CFU counts of als6Δ/Δ C. albicans strain incubated for 20 h with CD48–/– or WT BMEos (MOI 1); n = 3 biological replicates. Each experiment was performed in technical duplicates or triplicates. F CFU counts in the kidneys of CD48–/– (n = 8) and WT (n = 7) mice 2 dpi with WT or als6Δ/Δ C. albicans strains. Fungal load was assessed by CFU as described in Fig. 2A. Samples were collected in 2 independent experiments. G, H IL-6 (n = 6) levels in serum (G) and IFN-γ (n = 5) levels in kidneys (H) of WT and CD48–/– mice 2 dpi with WT or als6Δ/Δ C. albicans strains. I Binding of C. albicans WT (SC5314), als6Δ/Δ, or clinical isolates (n = 34) to CD48 as evaluated by released levels of mIL-2 in supernatants of BW-hCD48. Data are normalized and relative to those of WT strain. Significance was analyzed in comparison to BW-hCD48 cells incubated in medium alone; n = 1 in technical triplicates. Data show the mean ± SD and significance was analyzed by one-way Anova (E–H) or two-tailed student t test (C). For B and I one-way Anova with correction for multiple comparisons using Dunnett’s test was used. ns= non-significant, *P ≤ 0.05; **P ≤ 0.01; ***P ≤ 0.005, ****P ≤ 0.001.
We next sought to determine the functional relevance of CD48-Als6 binding in the antifungal activity of eosinophils. Therefore, C. albicans als6Δ/Δ was incubated with either CD48–/– or WT BMEos. In both cases, similar CFUs were observed (Fig. 5E), contrasting with our previous observation with the WT C. albicans strain, where WT but not CD48–/– BMEos showed a significant reduction of the fungi viability (Fig. 3H). Finally, to evaluate the relevance of these findings in vivo, we injected als6Δ/Δ C. albicans strain into WT and CD48–/– mice and evaluated the fungal load in the kidneys, the serum IL-6, and the IFN-γ levels in the kidneys 2 dpi. In contrast to data obtained with WT C. albicans strain where a significant increase in fungal load, serum IL-6 and IFNγ was found in CD48–/– mice, these parameters were all similar in WT and CD48–/– mice infected with als6Δ/Δ C. albicans (Fig. 5F, G, H).
Next, we sought to determine whether CD48 might play a general role in eosinophil-fungal interactions. We collected 34 clinical isolates of C. albicans from a cohort of patients with invasive candidiasis, and- using BW-hCD48 and BW cells we evaluated their CD48 binding capacity. We found that 55% of the clinical isolates bound significantly to BW-hCD48 cells as compared to BW-hCD48 cultured in medium alone (Fig. 5I), while incubation of BW parental cells with the same strains did not show any binding (Fig. S4B). To note, most clinical strains displayed similar or even higher binding levels than the WT strain, highlighting the potential clinical relevance of CD48.
Discussion
The role of eosinophils in allergic diseases, including those caused by fungal allergens, is well defined. However, eosinophil contribution in fungal infections, and particularly in candidiasis, remains unclear. A clinical study demonstrated that absolute eosinophil count is significantly associated with serum (1,3)-β-D-Glucan in patients with invasive fungal infections18, implying a possible role for eosinophils. Eosinophil depletion by biological drugs (mepolizumab, benralizumab) for treatment of severe eosinophilic asthma or other eosinophil-linked diseases (hypereosinophilic syndrome, eosinophilic esophagitis, etc.) is being increasingly employed. However, post market surveillance (pharmacovigilance) of these drugs highlights the increased risk of infections for patients31,32,33,34.
In the current study, we show a protective role of eosinophils against C. albicans in vitro and during systemic candidiasis in vivo. Using human peripheral blood eosinophils, we found that eosinophils were activated by C. albicans and inhibited fungal growth via a complex mechanism. C. albicans is phagocytosed by eosinophils, but the growing hyphae within the eosinophils and the subsequent secretion of candidalysin, allow the fungi to escape and to cause eosinophil degranulation, cytokine production (IFN-γ) and cytolysis. This dual killing mechanism has also been demonstrated in human macrophages after C. albicans phagocytosis35 and in eosinophils in response to A. alternata28. Importantly, another study also reported that stimulating human eosinophils with GM-CSF, IL-1, IL-3, and IL-5 augmented phagocytosis of C. albicans, but this paradoxically did not result in increased killing of the organism36 and in line with our findings confirms that C. albicans may escape from eosinophils phagocytosis.
The killing of C. albicans appears predominantly mediated by the release of the eosinophil basic granule proteins, mainly MBP-1. MBP-1 is a strongly basic protein (pI = 11.4)21 and is active against a wide range of microorganisms, including bacteria, helminths, and fungi37. The cationic property of MBP-1 affects the charge of surface membranes, resulting in their permeabilization and disruption, as shown with Staphylococcus. aureus (S. aureus) and E. coli38. In another study, it was reported that recombinant RNase3/ECP displays anti-C. albicans properties due to its basic characteristics39,40. Together, this suggests that strong positively charged compounds influence C. albicans growth/viability, even though C. albicans can tolerate a broad range of pH (from 2 to 10)41.
In our study eosinophil death appears to be mediated by hyphal growth and candidalysin secretion. Candidalysin directly permeabilizes host cell membranes, driving cell damage26, and is a key driver of inflammasome-independent cytolysis42. Candidalysin also induces immune responses in host cells including macrophages42, epithelial cells43, and mast cells44. In this study, we demonstrate that candidalysin induced eosinophil degranulation while its capacity to induce eosinophil death was limited only with the highest candidalysin concentration used (60 µM). Notably, eosinophils co-cultured with candidalysin-deficient strains displayed similar degranulation and killing as that induced by WT strain, suggesting that other mechanisms, for example those induced by growing hyphae, may contribute to eosinophil degranulation and cytolysis.
Moreover, SEM analysis after co-culture revealed EETs surrounding C. albicans. EETs formation in response to C. albicans, E-coli, S. aureus25, and A. fumigatus45 is a well-known phenomenon that occurs upon cytolytic eosinophil death46. Therefore, EET formation may be an additional contributor to anti-Candida activity and eosinophil death.
Using eosinophil deficient ΔdblGATA mice we demonstrate that eosinophils play an important anti-fungal role in a systemic candidiasis model. GATA-1 mutation causes not only eosinophil deficiency but also an impairment of hematopoietic stem and progenitor cells47, together with a concomitant reduction in IL-4 production, mast cell numbers, and IgE mediated-activation48. These features together with the absence of eosinophils might influence the outcome of systemic candidiasis in the ΔdblGATA mice. We therefore performed another model in which eosinophils were specifically depleted by injection of anti-IL-5 antibody. In both models, eosinophils deficient mice showed a higher fungal load in the kidneys together with increased IL-6 levels in serum, decreased IFN-γ in serum and homogenates kidneys and increased mortality. Importantly, the decreased IFN-γ levels found in both eosinophil deficient mice models, together with the release of this cytokine by human eosinophils in response to C. albicans in vitro, suggests that eosinophils may also affect the immune response to decrease the fungal burden. Notably, eosinophil staining by H&E and anti-Siglec-F in the infected kidneys revealed few intact eosinophils and mostly eosinophil debris as described by the Splendore–Hoeppli phenomenon, that is characterized by microorganisms (fungi, bacteria and parasites) surrounded by eosinophilic debris and material49,50. To note, this phenomenon was also described in microabscesses and granulomas throughout the body of patients with systemic candidiasis51. Moreover, the hyphal form was predominantly observed in infected kidneys, while the yeast form was predominantly observed in the liver and spleen42, consistent, with our in vitro data showing that eosinophil anti-fungal activity took place after hyphae formation.
In a lung infection model caused by intranasal inoculation of A. fumigatus, ΔdblGATA mice showed a higher fungal burden in the lungs52, similar to our data in the kidneys.
The decrease in eosinophil blood counts in WT mice infected with C. albicans and their absence in the kidney at 18 h post infection may indicate a role for these granulocytes in systemic candidiasis. Conversely, it is possible to hypothesize that the association of eosinophil plasma membrane markers of activation and the declining blood eosinophil counts are indicative of an eosinophil cytolytic process. A similar phenomenon was described in several infectious diseases, including bacterial infections53 and COVID-1954. Nevertheless, there is still a lack of evidence from human patients treated with biological drugs for depletion of eosinophils. It is possible to hypothesize that eosinophils and their effect in systemic candidiasis may be redundant in comparison to “professional” immune cells such as neutrophils, macrophages, or T cells. However, several case studies reveal that patients with candida infection and neutrophil impairment due to caspase recruitment domain-containing protein 9 (CARD9) deficiency showed an increased eosinophil count in blood and cerebrospinal fluid (CSF)55,56,57, suggesting a potential role for eosinophils.
Next, we report here on the role of CD48 in the interaction with C. albicans. CD48 is a glycosylphosphatidylinositol (GPI) anchored activatory receptor expressed by several myeloid and lymphoid cells and recognizes bacteria such as E. coli20 and Mycobacterium tuberculosis58. CD48 also plays a prominent role in the binding and activation of eosinophils by S. aureus20. Here, we demonstrated that CD48 binds C. albicans yeast and hyphae and that blocking the CD48-C. albicans axis, by incubation of C. albicans with hCD48-Fc, reduced eosinophil activation. Moreover, CD48–/– BMEos co-cultured with C. albicans could not induce reduction in C. albicans viability unlike WT BMEos.
CD48 plays a significant role in antifungal immunity in vivo, as evidenced by the increased kidney fungal burden, elevated serum IL-6 levels, and higher mortality observed in CD48–/– mice in models of systemic candidiasis. However, since CD48 is expressed on multiple leukocyte subsets, its antifungal function cannot be attributed solely to eosinophils. To explore this further, we depleted eosinophils in both CD48–/– and WT mice and compared their fungal burdens. Our results demonstrated that while CD48 is essential for eosinophil-mediated antifungal activity, its role extends beyond eosinophils. Overall, these findings identify CD48 as an antifungal receptor that contributes to host defense across various CD48-expressing immune cells.
The biological and clinical relevance of the CD48-C. albicans interaction was also assessed in this study. We report here two other Candida species including C. lusitaniae and C. tropicalis, but not C. parapsilosis to bind CD48. Furthermore 19 out of the 34 systemic C. albicans clinical isolates that were assessed significantly bound CD48.
C. albicans infections often exacerbate inflammatory diseases such as IBD59, asthma60 and COPD61. Zhang et al.62 suggested that the ability of certain fungi to exacerbate asthma symptoms is dependent on their immunomodulatory functions and independent of their ability to act as sensitizing allergens. Based on our results, we might assume that C. albicans affect the inflammatory milieu in the lungs and exacerbate disease via CD48, which we previously reported as being a pivotal eosinophil receptor in allergic eosinophilic airway inflammation63,64.
The Als family is cell surface glycoproteins required for adhesion of C. albicans to host cells, including endothelial, vaginal and buccal epithelial cells65. Here, we identified Als5, Als6, and Als7 as potential ligands of CD48. Focusing on Als6, we found that its interaction with CD48 was required to induce eosinophil mediated C. albicans killing in vitro and an anti-fungal activity in vivo. In contrast with the data observed with the WT C. albicans, CD48–/– mice injected with als6Δ/Δ mutant showed comparable kidney fungal burdens and IL-6 serum levels as WT mice. This indicates that Als6-CD48 interaction is crucial for a protective anti-fungal response.
Importantly, it was previously reported that C. albicans binds the TIGIT receptor via Als6, Als7 and Als9, inhibiting NK and T cell activity7. This shows that the Als proteins may function as adhesion molecules as well as immune modulators by binding activatory and inhibitory receptors.
In conclusion, we demonstrate that eosinophils, by a distinct mechanism, (Fig. 6) are important in host protection against C. albicans infection, and that the CD48-Als6 axis is a key driver of the anti-fungal immune cell response.
Eosinophils and Candida albicans interactions: from recognition to its death. Created in BioRender. Zaffran, I. (2025) https://BioRender.com/mlitkbu.
Methods
Fungal strains
The wild type C. albicans strain (SC5314) used in this study was obtained from ATCC. Candida parapsilosis, Candida tropicalis, and Candida lusitaniae were obtained from Prof. Daniel Kornitzer (Israel Institute of Technology, Haifa, Israel). Candidalysin-mutant strains: ECE1 deletion strain (ece1Δ/Δ), ECE1-complemented strain (ece1Δ/Δ + ECE1), and C. albicans strain lacking only the candidalysin-encoding region in Ece1 (ece1Δ/Δ + ECE1 Δ184–279) were produced as described in ref. 26. Als mutant strains were als1Δ/Δ1467, als2Δ/Δ2757, als3Δ/Δ1843, als4Δ/Δ2034, als5Δ/Δ2373, als6Δ/Δ1420, als7Δ/Δ1429 and als9Δ/Δ2028, which are all derivatives of SC53147. For all experiments, fungal cultures were inoculated overnight in Sabouraud dextrose (Sigma-Aldrich, Jerusalem, Israel) medium at 30 °C in aerobic conditions. The culture was then diluted 1:50 in a fresh secondary culture in Sabouraud dextrose medium for 2 h. Cells were then washed 3 times with ice cold PBS before their introduction into the experiment.
Systemic candidiasis mouse models
For the systemic candidiasis studies, 8-12-week-old males C57Bl/6 (Harlan, ENVIGO, UK), ΔdblGATA (backcrossed to the C57Bl/6 background for seven generations, a kind gift of Prof. Ariel Munitz, Department of Clinical Microbiology and Immunology, Faculty of Medicine, Tel Aviv University, Tel Aviv, Israel) and CD48–/– mice (a kind gift of Prof. Arlene H. Sharpe Department of Microbiology and Immunobiology, Evergrande Center for Immunologic Diseases, Harvard Medical School, Boston, MA, USA) were injected via the lateral tail vein with either 5 × 104 (for survival assay) or 5 × 105 (fungal burden assay) C. albicans yeast for the determination of fungal burden respectively. C. albicans (strain SC5314 or als6Δ/Δ) was grown at 30 °C in yeast Sabouraud dextrose broth as described above. On the day of the experiment the fungal cells were washed 3 times in ice-cold PBS and taken to the animal facility on ice.
For fungal burden experiments, mice were sacrificed with 48 h post-infection and relevant organs were harvested. Organs were homogenized, filtered through a 70 μm strainer, serially diluted in ice-cold PBS and plated on Sabouraud dextrose agar plates (Sigma-Aldrich). The plates were incubated for 48 h in a stationary 30 °C incubator and once colonies were visible, they were counted.
For survival experiments, mice were weighed, clinically evaluated daily and euthanized once their initial weight was reduced by 20%.
For the depletion of eosinophils, WT and CD48–/– mice were. injected i.p with 0.3 mg/100 µl of anti–IL-5 antibody in (TREK5; BioXCell) or with anti–horseradish peroxidase isotype control antibody (HRPN; BioXCell) at days −5, −3, and −1 before fungal infection.
All the animal experiments were performed according to Hebrew University ethical animal guidelines (MD-19-15990-5).
Human peripheral blood eosinophil purification
Human eosinophils were purified as previously described66 from the peripheral blood of mildly atopic volunteers (blood eosinophil levels 5–10%), asymptomatic and therefore not taking any drug. Written informed consent was obtained according to the guidelines of the Hadassah-Hebrew University Human Experimentation Helsinki Committee (0410-14-HMO). Briefly, venous blood (150 ml) was collected in heparinized syringes and left to sediment in 6% dextran (Sigma-Aldrich). Leukocytes were centrifuged on Ficoll-Hypaque (density, 1.077; 25 min, 700 × g, 22 °C; Amersham Bioscience, Amersham, Buckinghamshire, UK). Neutrophils and contaminating lymphocytes were tagged in the granulocyte-enriched pellet with micromagnetic beads bound to anti-CD16 and anti-CD3 (Miltenyi Biotec, North Rhine-Westphalia, Germany). Eosinophils were purified by passing the cell suspension through a magnetic column (LS-MACS). Eosinophils were evaluated by flow cytometry using APC-anti-human CCR3 (Abcam, Cambridge, UK) and PE anti-human Siglec-8 (BioLegend, CA, USA) and collected at a purity of >98% (Kimura staining), with viability of >98% (trypan blue staining). Then, eosinophils were re-suspended (1 × 106 cells/ml) in eosinophil medium consisting of RPMI-1640 supplemented with 10% heat-inactivated fetal calf serum (FCS; HyClones; Logan, Utah, USA), penicillin-streptomycin solution (100 u/ml; Biological Industries, Beit Haemek, Israel), and GM-CSF (5 ng/ml; Peprotech, Rocky Hill, NJ, USA).
Eosinophil viability and degranulation assay
For viability and degranulation assays, eosinophils (105 cells/well) were incubated at 37 °C, 5% CO2 for 1, 3, and 20 h in eosinophil medium with C. albicans at MOI 0.1, and 1 or with commercially synthesized candidalysin peptide toxin (SIIGIIMGILGNIPQVIQIIMSIVKAFKGNK; >98% pure; Peptide Protein Research Ltd, Hampshire, UK). Blocking of CD48 was assessed by pre-incubation of C. albicans with D1-Fc or hCD48-Fc (5 µg/ml) for 40 min on ice. Yeast was then washed with phosphate buffer saline (PBS) prior to the co-culture. After the indicated time points, viable cell count was determined by trypan blue exclusion test using a Neubauer chamber cell, and supernatants were collected to evaluate the levels of EPX and LDH. EPX levels were measured using a standardized colorimetric assay with purified human EPX (a kind gift of Prof. Gerald J. Gleich, University of Utah Health Sciences Center, Salt Lake City, UT, USA) and freshly prepared peroxidase substrate solution containing o-phenylenediamine dihydrochloride (OPD; Thermo-Fisher Scientific; Kyriat Shmona, Israel) as previously described67. LDH levels were determined using Lactate dehydrogenase assay kit (Abcam).
Generation of human and mouse CD48-Fc
Fusion proteins were generated as previously described50. In brief, the sequence encoding the extracellular part of human CD48 was amplified by PCR using the 5ʹ primer cccACCGGT GCCGCCACC (including AgeI restriction site) and the 3ʹ primer CATTCTTGGCCTGTTACTTACGGATCCccc (including BamHI restriction site). This PCR fragment was cloned into an expression vector containing a mutated Fc portion of human IgG1 (CSI-Ig Puro plasmid). The same protocol was performed for the generation of the mouse CD48-Fc (mCD48-Fc). All proteins were generated in 293 T cells and were purified on a protein G column (HiTrap TM protein G HP, GE Health Care, Chicago, IL, USA).
Generation of WT and CD48–/– BMEos
Bone marrow cells obtained from femurs of CD48–/– and WT mice were differentiated as previously described68. On days 10–12, eosinophil purity was examined by flow cytometry. Eosinophils were recognized by their expression of characteristic cell surface markers using PE-anti-mouse Siglec-F (BD Biosciences, NJ, USA) and APC-anti-mouse CCR3 (BioLegend). CD48 expression was also evaluated by flow cytometry using FITC-anti-mouse CD48 or hamster IgG isotype control (eBioscience, CA, USA).
Determination of fungal colony forming unit (CFU) in vitro
To evaluate C. albicans viability. human and murine eosinophils (1 × 105 cells/well) were co-cultured for 3 and 20 h with C. albicans at MOI 0.1 or 1. To assess the effect of eosinophil mediators, C. albicans (1 × 105 yeast/well) was incubated with purified eosinophil basic mediators, each at the concentration present in 1 × 105 eosinophils21,27. Cells were then collected and diluted in PBS by serial dilutions and plated onto Sabouraud agar plates, incubated at 30 °C for 48 h, and CFU numbers were counted.
Flow cytometry
For the staining of cell surface receptors, cells were harvested, centrifuged at 250 × g for mammalian cells and 3000 × g for fungal cells, washed twice in flow cytometry buffer (PBS, 0.1% bovine serum albumin (BSA)), and counted using a hemocytometer. Cells (1 × 105 cells/well) were plated into a 96 U-bottom plate and resuspended in blocking buffer (flow cytometry buffer supplemented with 5% goat serum, Biological Industries) for 15 min on ice. Human eosinophils were incubated with the following primary antibodies: anti-human CD48 (BioLegend), anti-human Dectin-1 (Abcam), anti-human TLR2 (BioLegend), anti-human TLR4 (BioLegend), anti-phospho-Lyn (Cell signaling, Danvers, MA USA) or the appropriate isotype control (mouse IgG1 or mouse IgG2a; all at 2.5 μg/ml) for 40 min on ice. Cells were then washed again and incubated with the secondary antibody Fluorescein (FITC)-goat anti-mouse IgG (1:200, Jackson ImmunoResearch, West Grove, PA, USA) for 30 min on ice in the dark. C. albicans staining was assessed using in-house murine and human CD48-Fc or D1-Fc (5 µg/ml), followed by FITC-goat anti-human IgG (1:2000 in FC buffer; Jackson ImmunoResearch). Afterward, the cells were washed twice, resuspended in 200 μl flow cytometry buffer, and analyzed. Samples were acquired by Cytoflex flow cytometer (Beckman Coulter, Brea, CA, USA) and analyzed using FlowJo (FlowJo X 10.0.7r2, Tree Star, Ashland, OR, USA) software.
Immunofluorescence microscopy
C. albicans yeast/hyphae were grown as described above and washed twice in ice cold PBS. Hyphal cells were grown overnight at 37 °C with RPMI-1640 supplemented with 10% FCS (Sigma-Aldrich) and 1% penicillin-streptomycin solution (100 U/ml; Biological Industries). Cells (2 × 105) were incubated in the presence of FITC (0.1 mg/ml, diluted in PBS) for 30 min, washed twice in ice-cold PBS (3000 G, 4 °C, 5 min) and blocked using PBS/5% BSA for 2 h at 4 °C. Next, the cells were washed again in PBS followed by incubation with hCD48-Fc or the negative control protein D1-Fc for 2 h at 4 °C. Unbound protein was then washed with ice cold PBS and incubated with the secondary antibody PE-anti-human IgG (1:500; Jackson ImmunoResearch) for 2 h at 4 °C and then washed with PBS. Finally, cells were mounted with DAPI mounting media and visualized using an Olympus Fluoview FV1000 confocal microscope.
Generation of BW-hCD48 cells
Reporter cells for hCD48 (BW-hCD48) were generated as previously described69. The inserts for the expression vectors encoding CD48 and zeta chain were generated by PCR using the following primers:
1-CD48 forward: cccAAGCTT GCCACC ATGTGCTCCAGAGGTTGGGA
2-CD48 reverse containing additional sequences complementary to zeta chain forward: TTCCATCTAGCAAGTAGCAGAG GGACCGGGCCAGGGTACAGGG
3-Zeta chain forward containing additional sequences complementary to CD48 reverse CCCTGTACCCTGGCCCGGTCC CTCTGCTACTTGCTAGATGGAA
4-Zeta chain reverse gggCTCGAG TTAGCGAGGGGCCAGGGTCTGC.
The full construct was then introduced into a plasmid with an antibiotic resistance (Puromycin) and inserted into BW5147.3 cell line (BW) using electroporation. Cells were cultured in RPMI-1640 supplemented with 10% FCS, penicillin-streptomycin solution (100 u/ml; Biological Industries) and gentamycin (5 µg/ml, G418; Sigma-Aldrich) for selection. Before each experiment, cells were tested by flow cytometry for CD48 expression as described above.
BW reporter assay
WT and C. albicans mutant strains: als1Δ/Δ1467, als2Δ/Δ 2757, als3Δ/Δ 1843, als4Δ/Δ 2034, als5Δ/Δ 2373, als6Δ/Δ 1420, als7Δ/Δ 1429, als9Δ/Δ 2028 and the C. albicans clinical strains were grown as described above. The fungal cells (5 × 104 yeast/well) were first incubated in a 96 flat-bottom plate for 2 h at 37 °C to adhere to the plate. Then, BW or BW-hCD48 cells were added (MOI 1) in a final volume of 200 μl RPMI-1640 growth media as described above and for an additional 48 h (37 °C; 5% CO2). The medium was supplemented with fluconazole (5 µg/ml) to prevent hyphae formation. After 48 h cells were centrifuged and supernatants were collected for measurement of released mIL-2 by ELISA.
ELISA assays
Cytokine levels in the serum or in homogenized kidneys of mice infected with C. albicans were determined 2 dpi by commercial kits for IFN-γ (Peprotech, Rehovot, Israel), IL-6 (ELISA MAX™ Deluxe Set Mouse IL-6, BioLegend) according to the supplier’s instructions.
mIL-2 levels in the supernatants from BW and BW-CD48 cells were determined by in-house ELISA. ELISA plates were initially coated with anti-mouse-IL-2 (0.1 μg/100 µl PBS; BioLegend) for 2 h, 37 °C. and blocked with PBS/1% BSA for an additional 2 h at room temperature. Supernatants were then added to the plates and incubated overnight at 4 °C. The plates were then washed 4 times with PBST (PBS/0.05% tween20) and a detection antibody biotinylated anti-mouse-IL-2 (0.1 μg/100 µl PBS; BioLegend) from a different clone than the coating antibody was added for 1 h at room temperature. Next the plates were washed 6 times with PBST, incubated for 30 min with Streptavidin-HRP (1:1000; Jackson ImmunoResearch) and again washed 6 times using PBST. Finally, the plates were developed using 3,3′,5,5′-tetramethylbenzidine (TMB) substrate (SouthernBiotech) and read at 650 nm using an ELISA reader (BIO-TEK, Winooski, VT, USA).
Quantification and statistical analysis
If not indicated otherwise, experiments were always performed in triplicates and repeated at least three times. Due to strong eosinophil donor variability, data from experiments with human eosinophils are shown as representative of three or more independent experiments performed with different donors. Results are expressed as mean ± SD or mean ± SEM and statistical tests were performed using Prism (GraphPad Prism 10.5.0.730) software. The unpaired two-tailed Student’s t test was used to compare differences between two groups. One-way ANOVA with Tukey’s test for multiple comparisons was used to evaluate experiments containing more than two groups. Survival curves were analyzed by Log-rank (Mantel Cox) test. P values less than 0.05 were considered significant for all analyses.
Ethics statement
Peripheral blood eosinophils were obtained according to the guidelines of the Hadassah-Hebrew University Human Experimentation Helsinki Committee (0410-14-HMO). All work involving human patients was approved by the Tel Aviv Sourasky Medical Center Institutional Ethics Committee (approval number 0729-16). The need for informed consent was waived given the retrospective observational nature of this study.
Mice experiments were performed according to the guidelines of the Hebrew University ethics committee for animal studies (MD-19-15990-5).
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
All the reagents used, and data supporting the findings of this study are available within the article and in the Supplementary Table 1, or available from the corresponding author upon request. Source data files are provided with this paper. Source data are provided with this paper.
References
Fisher, M. C. & Denning, D. W. The WHO fungal priority pathogens list as a game-changer. Nat. Rev. Microbiol. 21, 211–212 (2023).
WHO fungal priority pathogens list to guide research, development and public health action. https://www.who.int/publications/i/item/9789240060241.
Shao, T.-Y. et al. Commensal candida albicans positively calibrates systemic Th17 immunological responses. Cell Host Microbe 25, 404–417.e6 (2019).
Lionakis, M. S., Lim, J. K., Lee, C.-C. R. & Murphy, P. M. Organ-specific innate immune responses in a mouse model of invasive candidiasis. J. Innate Immun. 3, 180–199 (2011).
Netea, M. G., Joosten, L. A. B., van der Meer, J. W. M., Kullberg, B.-J. & van de Veerdonk, F. L. Immune defence against Candida fungal infections. Nat. Rev. Immunol. 15, 630–642 (2015).
Patin, E. C., Thompson, A. & Orr, S. J. Pattern recognition receptors in fungal immunity. Semin. Cell Dev. Biol. 89, 24–33 (2019).
Charpak-Amikam, Y. et al. Candida albicans evades NK cell elimination via binding of Agglutinin-Like Sequence proteins to the checkpoint receptor TIGIT. Nat. Commun. 13, 2463 (2022).
Klion, A. D., Ackerman, S. J. & Bochner, B. S. Contributions of Eosinophils to Human Health and Disease. Annu. Rev. Pathol. Mech. Dis. 15, 179–209 (2020).
Ghosh, S., Hoselton, S. A., Dorsam, G. P. & Schuh, J. M. Eosinophils in fungus-associated allergic pulmonary disease. Front. Pharmacol. 4, 8 (2013).
Weller, P. F. & Spencer, L. A. Functions of tissue-resident eosinophils. Nat. Rev. Immunol. 17, 746–760 (2017).
Wechsler, M. E. et al. Eosinophils in health and disease: A state-of-the-art review. Mayo Clin. Proc. 96, 2694–2707 (2021).
Gaur, P. et al. The regulatory role of eosinophils in viral, bacterial, and fungal infections. Clin. Exp. Immunol. (2022).
Farmand, S. et al. Eosinophilia and reduced STAT3 signaling affect neutrophil cell death in autosomal-dominant Hyper-IgE syndrome. Eur. J. Immunol. 48, 1975–1988 (2018).
Huang, J., Liu, C. & Zheng, X. Clinical features of invasive fungal disease in children with no underlying disease. Sci. Rep. 12, 208 (2022).
TSUNEMI, Y. et al. Secondary cutaneous candidiasis with eosinophilia. J. Dermatol. 37, 175–178 (2010).
Queiroz Filho, J. et al. Eosinofilia no sangue periférico de mulheres com candidiase vaginal recorrente. Rev. Bras. Ginecol. e Obs. 35, 453–457 (2013).
Jevtic, S. D. & Padmore, R. Eosinophils as a red herring for Candida. Hum. Pathol. Case Reports 20, 200367 (2020).
Zhu, P., Lv, J., Kong, Y., Xu, H. & Ming, L. Absolute eosinophil count was significantly associated with serum (1,3)-β-D-glucan in patients with invasive fungal infections. Clin. Lab. 63, 749–756 (2017).
Driss, V. et al. TLR2-dependent eosinophil interactions with mycobacteria: role of α-defensins. Blood 113, 3235–3244 (2009).
Minai-Fleminger, Y. et al. The CD48 receptor mediates Staphylococcus aureus human and murine eosinophil activation. Clin. Exp. Allergy 44, 1335–1346 (2014).
Acharya, K. R. & Ackerman, S. J. Eosinophil granule proteins: Form and function. J. Biol. Chem. 289, 17406–17415 (2014).
Shamri, R., Xenakis, J. J. & Spencer, L. A. Eosinophils in innate immunity: an evolving story. Cell Tissue Res. 343, 57–83 (2011).
Yousefi, S., Simon, D. & Simon, H.-U. Eosinophil extracellular DNA traps: Molecular mechanisms and potential roles in disease. Curr. Opin. Immunol. 24, 736–739 (2012).
Ishikawa, T., Condon Dalton, A. & Arbesman, C. E. Phagocytosis of Candida albicans by eosinophilic leukocytes. J. Allergy Clin. Immunol. 49, 311–315 (1972).
Ueki, S. et al. Eosinophil extracellular trap cell death–derived DNA traps: Their presence in secretions and functional attributes. J. Allergy Clin. Immunol. 137, 258–267 (2016).
Moyes, D. L. et al. Candidalysin is a fungal peptide toxin critical for mucosal infection. Nature 532, 64–68 (2016).
Venge P. Eosinophils in Human Disease. Eosinophils in Health and Disease 431–536 (2013).
Yoon, J., Ponikau, J. U., Lawrence, C. B. & Kita, H. Innate Antifungal Immunity of Human Eosinophils Mediated by a β 2 Integrin, CD11b. J. Immunol. 181, 2907–2915 (2008).
Grigorescu, A., Chen, M.-H., Zhao, H., Kahn, P. & Lipke, P. A CD2-Based Model of Yeast a-Agglutinin Elucidates Solution Properties and Binding Characteristics. IUBMB Life 50, 105–113 (2000).
Elias, S., Kahlon, S., Duev-Cohen, A. & Mandelboim, O. A BW Reporter System for Studying Receptor-Ligand Interactions. J. Vis. Exp. https://doi.org/10.3791/58685 (2019).
Khatri, S. et al. Assessment of the long-term safety of mepolizumab and durability of clinical response in patients with severe eosinophilic asthma. J. Allergy Clin. Immunol. 143, 1742–1751.e7 (2019).
Zou, F. et al. A real-world pharmacovigilance study of mepolizumab in the FDA adverse event reporting system (FAERS) database. Front. Pharmacol. 14, 1320458 (2023).
Giossi, R., Pani, A., Schroeder, J. & Scaglione, F. Exploring the risk of infection events in patients with asthma receiving anti-IL-5 monoclonal antibodies: A rapid systematic review and a meta-analysis. Heliyon 10, e23725 (2024).
Lin, S., Luo, D., Gong, Z. & Zhan, Q. Updated insights into adverse events associated with mepolizumab: a disproportionality analysis from the FDA adverse event reporting system database. Front. Med. 11, 1449194 (2024).
Krysan, D. J., Sutterwala, F. S. & Wellington, M. Catching fire: Candida albicans, macrophages, and pyroptosis. PLoS Pathog. 10, e1004139 (2014).
Fabian, I. et al. Activation of human eosinophil and neutrophil functions by haematopoietic growth factors: comparisons of IL-1, IL-3, IL-5 and GM-CSF. Br. J. Haematol. 80, 137–143 (1992).
Soragni, A. et al. Toxicity of Eosinophil MBP Is Repressed by Intracellular Crystallization and Promoted by Extracellular Aggregation. Mol. Cell 57, 1011–1021 (2015).
Lehrer, R. I. et al. Antibacterial properties of eosinophil major basic protein and eosinophil cationic protein. J. Immunol. 142, 4428–4434 (1989).
Singh, A. & Batra, J. K. Role of unique basic residues in cytotoxic, antibacterial and antiparasitic activities of human eosinophil cationic protein. Biol. Chem. 392, 337–346 (2011).
Salazar, V. A. et al. Insight into the Antifungal Mechanism of Action of Human RNase N-terminus Derived Peptides. Int. J. Mol. Sci. 20, 4558 (2019).
Sherrington, S. L. et al. Adaptation of Candida albicans to environmental pH induces cell wall remodelling and enhances innate immune recognition. PLoS Pathog. 13, e1006403 (2017).
Kasper, L. et al. The fungal peptide toxin Candidalysin activates the NLRP3 inflammasome and causes cytolysis in mononuclear phagocytes. Nat. Commun. 9, 4260 (2018).
Richardson, J. P. et al. Candidalysin Drives Epithelial Signaling, Neutrophil Recruitment, and Immunopathology at the Vaginal Mucosa. Infect. Immun. 86, e00645-17 (2018).
Song, P. et al. Candidalysin, a virulence factor of candida albicans, stimulates mast cells by mediating cross-talk between signaling pathways activated by the dectin-1 receptor and MAPKs. J. Clin. Immunol. 42, 1009–1025 (2022).
Muniz, V. S. et al. Eosinophils release extracellular DNA traps in response to Aspergillus fumigatus. J. Allergy Clin. Immunol. 141, 571–585.e7 (2018).
Thompson-Souza, G. A., Vasconcelos, C. R. I. & Neves, J. S. Eosinophils: Focus on DNA extracellular traps. Life Sci. 311, 121191 (2022).
Hwang, D. et al. GATA1 controls numbers of hematopoietic progenitors and their response to autoimmune neuroinflammation. Blood Adv. 6, 5980–5994 (2022).
Jacobsen, E. A. et al. Eosinophil knockout humans: Uncovering the role of eosinophils through eosinophil-directed biological therapies. Annu. Rev. Immunol. 39, 719–757 (2021).
Gopinath, D. Splendore–Hoeppli phenomenon. J. Oral. Maxillofac. Pathol. 22, 161 (2018).
Leiferman, K. M. & Gleich, G. J. The true extent of eosinophil involvement in disease is unrecognized: the secret life of dead eosinophils. J. Leukoc. Biol. 116, 271–287 (2024).
Hussein, M. R. Mucocutaneous Splendore-Hoeppli phenomenon. J. Cutan. Pathol. 35, 979–988 (2008).
Lilly, L. M. et al. Eosinophil deficiency compromises lung defense against Aspergillus fumigatus. Infect. Immun. 82, 1315–1325 (2014).
Hogan, S. P., Waddell, A. & Fulkerson, P. C. Eosinophils in infection and intestinal immunity. Curr. Opin. Gastroenterol. 29, 7–14 (2013).
Xie, G. et al. The role of peripheral blood eosinophil counts in COVID-19 patients. Allergy 76, 471–482 (2021).
Lanternier, F. et al. Inherited CARD9 deficiency in otherwise healthy children and adults with Candida species–induced meningoencephalitis, colitis, or both. J. Allergy Clin. Immunol. 135, 1558–1568.e2 (2015).
Du, B. et al. Complete clinical remission of invasive Candida infection with CARD9 deficiency after G-CSF treatment. Comp. Immunol. Microbiol. Infect. Dis. 70, 101417 (2020).
Drummond, R. A. et al. CARD9-dependent neutrophil recruitment protects against fungal invasion of the central nervous system. PLOS Pathog. 11, e1005293 (2015).
Muñoz, S., Hernández-Pando, R., Abraham, S. N. & Enciso, J. A. Mast cell activation by mycobacterium tuberculosis: Mediator release and role of CD48. J. Immunol. 170, 5590–5596 (2003).
Li, X. V. et al. Immune regulation by fungal strain diversity in inflammatory bowel disease. Nature 603, 672–678 (2022).
Wu, Y. et al. Candida albicans elicits protective allergic responses via platelet mediated T helper 2 and T helper 17 cell polarization. Immunity 54, 2595–2610.e7 (2021).
Zuo, Y. et al. Candida in lower respiratory tract increases the frequency of acute exacerbation of chronic obstructive pulmonary disease: A retrospective case-control study. Front. Cell. Infect. Microbiol. 10, 538005 (2020).
Zhang, Z. et al. β-Glucan exacerbates allergic asthma independent of fungal sensitization and promotes steroid-resistant T H 2/T H 17 responses. J. Allergy Clin. Immunol. 139, 54–65.e8 (2017).
Munitz, A., Bachelet, I., Finkelman, F. D., Rothenberg, M. E. & Levi-Schaffer, F. CD48 is critically involved in allergic eosinophilic airway inflammation. Am. J. Respir. Crit. Care Med. 175, 911–918 (2007).
Gangwar, R. S. et al. CD48 on blood leukocytes and in serum of asthma patients varies with severity. Allergy 72, 888–895 (2017).
Zhao, X., Oh, S.-H. & Hoyer, L. L. Deletion of ALS5, ALS6 or ALS7 increases adhesion of Candida albicans to human vascular endothelial and buccal epithelial cells. Med. Mycol. 45, 429–434 (2007).
Munitz, A. et al. 2B4 (CD244) is expressed and functional on human eosinophils. J. Immunol. 174, 110–118 (2005).
Adamko, D. J. et al. The effect of cationic charge on release of eosinophil mediators. J. Allergy Clin. Immunol. 122, 383–390.e4 (2008).
Dyer, K. D. et al. Functionally competent eosinophils differentiated ex vivo in high purity from normal mouse bone marrow. J. Immunol. 181, 4004–4009 (2008).
Lankry, D., Gazit, R. & Mandelboim, O. Methods to identify and characterize different NK cell receptors and their ligands. Methods Mol. Biol. 612, 249–273 (2010).
Acknowledgements
This study was supported by the Israel Science Foundation (442/18), Aimwell Charitable Trust (UK), and the Emalie Gutterman Memorial Endowment Fund to F.L.S.; and by the Israeli Scholarship Education Foundation (ISEF) to I.Z. J.R.N. was supported by the Wellcome Trust (214229_Z_18_Z). J.V. was a Jacob and Lena Joels Memorial Foundation Visiting Professor at the HUJI. We thank Prof Ariel Munitz (Tel Aviv University) for his valuable comments on mice experiments and Dr. Eduard Berenshtein from the electron microscopy core facility of the Hebrew University for his assistance with scanning electron microscopy.
Author information
Authors and Affiliations
Contributions
I.Z., P.G., and P.O. performed the experiments, analyzed data, and wrote the manuscript; Y.C.A., B.H., J.R.N., R.B.A., N.O., and O.M. assisted with methodology; H.P., D.S., B.H., J.R.N., and R.B.A. provided reagents and fungal strains; J.V. edited the manuscript; Y.C.A., B.H., J.R.N., and O.M. reviewed the manuscript; F.L.S. designed the research, provided overall supervision, and edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
J.V. reports speaker and consultancy fees and travel support in the past 5 years from Astra Zeneca, HpVac, Novartis, L’Oréal, Sanofi, Stallergènes-Greer, and Thermo Fisher Scientific. The other authors have no financial competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zaffran, I., Gaur, P., Ofori, P. et al. Eosinophil CD48 interactions with Candida albicans Als6 is protective in vitro and in mouse systemic candidiasis. Nat Commun 16, 9291 (2025). https://doi.org/10.1038/s41467-025-64276-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-64276-3