Abstract
Reducing the electrical energy consumption for formic acid electro-synthesis is indispensable for advancing its industrial implementation. In a conventional CO2 electrolyzer, most input electrical energy is consumed by the unprofitable anodic oxygen evolution reaction (OER) and ohmic drop. Electrolyzer engineering provides a promising platform to boost electrical energy utilization efficiency beyond catalyst optimization. Herein, we demonstrate a membrane-free CO2 electrolyzer design that pairs electrochemical CO2 reduction (CO2R) with an all-liquid-phase anodic reaction, enabling dual production of formate at both electrodes with significantly reduced cell voltage. The optimized design exhibits the lowest electrical energy consumption (< 310 kJ mol-1formate) at cell voltages below 2.7 V across a current density range of 0.05–0.4 A cm-2. This cell also maintains stable operation at 2.25 V for 313 h with a < 20 % increase in electrical energy consumption. Systematic techno-economic analysis (TEA) evaluates the economic viability of this design for formic acid electro-synthesis, revealing a potential roadmap towards low-cost formic acid production. This strategy provides guidelines for CO2R electrolyzer engineering toward energy-efficient, economically viable production of valuable chemicals.
Similar content being viewed by others
Introduction
Electrochemical CO2 reduction (CO2R) driven by renewable electricity is a promising route to substitute petroleum in the production of carbonaceous fuels and chemicals, enabling both electricity storage and a closed carbon loop1,2,3. Among the various CO2R products, the electroreduction of CO2 to C1 products requires the least charge passed, thus possessing the highest utilization of electrons (i.e., electricity) for carbon fixation4. Particularly, formic acid (HCOOH) has significant advantages of superior volumetric energy density and easy storage/transportation, making it a widely used industrial feedstock and energy carrier5,6,7. Nevertheless, the high energy demand for CO2-to-HCOOH conversion in current membrane-based flow cells limits the scalability of the technology. Techno-economic analysis (TEA) has highlighted that the electrolysis process consumes the majority of energy in HCOOH production4. Established approaches have primarily focused on catalyst optimization to enhance selectivity and activity, thereby reducing energy demand for the electrolysis process8,9,10,11. However, system design to improve energy and economic efficiency has not been fully exploited.
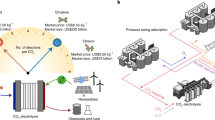
In the electrolysis process, the input electrical energy drives the cathodic and anodic half-reactions and is partly dissipated as thermal energy due to system resistance12,13. In a conventional membrane-based flow cell, CO2R is paired with the oxygen evolution reaction (OER), which has a high standard thermodynamic potential (1.23 V vs reversible hydrogen electrode, RHE) and yields an unprofitable product (O2) at the anode (Fig. 1a). Consequently, the electrical energy consumed in the OER process adds to operational costs without creating value. By contrast, replacing OER with an alternative organic oxidation reaction (OOR) presents an exciting opportunity to reduce electrical energy consumption and generate valuable products at the anode (Supplementary Fig. 1). Candidates for OOR include the oxidation of glycerol, glucose, alcohols, furfural, and other biomass polyols14. Among these, the glycerol oxidation reaction (GOR) is particularly attractive due to its lower standard thermodynamic potential (0.136 V vs RHE) and the abundantly available glycerol–a low-cost by-product of biodiesel and oleochemical industries. Similar to CO2R, the products of GOR are highly diverse, such as glyceric acid, glycolic acid, and formic acid15,16. Notably, the conversion of glycerol to formate (HCOO-, the predominant form of HCOOH in alkaline electrolyte) can achieve nearly 100 % selectivity with non-noble-metal catalysts17,18. Moreover, the complete conversion of 1 kg of glycerol (market price: US$ 0.2–0.4 kg–1) can yield 1.43 kg of formic acid (market price: US$ 0.6–0.9 kg–1), underscoring the economic viability of GOR. Consequently, pairing CO2R with GOR provides an efficient route to valorize both CO2 and glycerol, and represents a promising strategy for integrated electrobiorefinery systems19.
Energy and mass flows of (a) the co-electrolysis of CO2 and H2O in an anion exchange membrane (AEM)-based flow cell and b the co-electrolysis of CO2 and glycerol in a membrane-free flow cell. c Impacts of Q/NHCOOH value and cell voltage on the electrical energy consumption for producing per mole of HCOOH. The contour lines (solid lines) and circles represent equal electrical energy consumption and reported values in the literature, respectively, and the bubble size stands for the current density. The horizontal dashed lines represent the standard thermodynamic cell voltage, and the vertical dashed lines represent the minimum Q/NHCOOH value for the CO2 and H2O co-electrolysis (gray), the CO2 and methanol co-electrolysis (pink), as well as the CO2 and glycerol co-electrolysis (yellow), respectively. Source data are provided as a Source Data file.
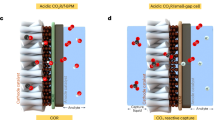
Ohmic losses from system resistance remain a major challenge in conventional membrane-based flow cells, with the electrolyte layer and ion exchange membrane being the primary contributors. Minimizing these resistances via thoughtful system design is essential for achieving energy-efficient CO2 electrolysis. A membrane-free CO2 electrolyzer featuring a thin electrolyte layer could potentially offer minimal system resistance, thereby reducing the associated electrical energy penalties. However, the conventional flow cell for the co-electrolysis of CO2 and H2O is unsuitable for the membrane-free design due to the crossover of electrolysis products that interfere with the counter electrodes (e.g., O2 from anode being reduced at cathode and CO2R products from cathode being oxidized at anode)20. Ager and colleagues designed a membrane-free co-laminar flow reactor incorporating a Y-split at the outlet to isolate the product before it diffuses across the reactor’s midline21. However, this strategy is highly dependent on maintaining a stable laminar condition, which is susceptible to disturbance. In contrast, a fundamentally different approach (i.e., utilization of paired electrolysis), in which both cathodic and anodic reactions produce valuable, mutually compatible products, might effectively mitigate anode-cathode interference and enable the operation of a membrane-free CO2 electrolyzer. As a representative example, the primary product of Ni-catalyzed GOR–HCOO-/HCOOH–is also a common CO2R product, which is a stable chemical with high resistance to further electrochemical reduction and oxidation in a glycerol-containing electrolyte (Fig. 1b)22. Moreover, all-liquid-phase GOR without involving gaseous species simplifies the electrolyzer structure and gas separation process at the anode20.
As a proof-of-concept, the CO2R and GOR are coupled to achieve dual HCOOH production in the proposed membrane-free flow cell. Energy analysis reveals that reducing cell voltage and charge passed for producing per mole of HCOOH would decrease the electrical energy consumption in the electrolysis process. Experimentally, the optimized membrane-free flow cell with an electrolyte layer thickness of 1.3 mm (NOM-CO2R/GOR1.3) achieves a low electrical energy consumption (< 310 kJ mol–1) across a current density range of 0.05–0.4 A cm–2, operating at cell voltages below 2.7 V with formate selectivity over 90%. This design achieves more than 56% reduction in electrical energy consumption compared to the conventional anion exchange membrane (AEM)-based flow cell (AEM-CO2R/OER) while demonstrating an overall energy conversion efficiency exceeding 45%. Moreover, the NOM-CO2R/GOR1.3 system demonstrates a stable operation over 310 h at 2.25 V. Techno-economic analysis (TEA) reveals that the NOM-CO2R/GOR1.3 system achieves a 29.1–48.3% reduction in HCOOH production cost compared to the AEM-CO2R/OER benchmark across major global economies, demonstrating its significant economic advantage. Furthermore, a detailed roadmap delineates the key factors and processes that need to be prioritized to enable cost-effective HCOOH electro-synthesis, highlighting a competitive production cost of US$ 0.379 kg–1HCOOH. Collectively, our work offers a promising strategy for enabling a membrane-free flow cell, delivers new insights into CO2R electrolyzer engineering, and paves the way toward energy-efficient, economically viable, and scalable electro-synthesis of HCOOH.
Results and discussion
Energy analysis of the electrolysis process
The factors determining the electrical energy consumption of the electrolysis process are explicitly analyzed. The electrical energy consumption for producing per mole of HCOOH is calculated using Eq. (1), which depends on Q/NHCOOH (i.e., charge passed for per mole of HCOOH production) and cell voltage. The contour map in Supplementary Fig. 2 communicates the impact of HCOOH yield and applied current on Q/NHCOOH. An increase in HCOOH yield at a given current density would lead to a reduced Q/NHCOOH value, which is independent of applied current. Under the assumption of 100 % Faradaic efficiency (FE) for HCOOH with no competing by-products, the Q/NHCOOH value in the CO2 and H2O co-electrolysis system maintains a constant minimum of 1.93 × 105 C mol–1. Substituting OER with OOR, such as GOR and methanol oxidation reaction (MOR) for HCOOH production, enables a higher HCOOH yield at equivalent current density. As a result, a substantial reduction in the minimum Q/NHCOOH value is observed, dropping to 1.29×105 C mol–1 and 1.1 × 105 C mol–1 when coupling CO2R with MOR and GOR, respectively.
The contour map in Fig. 1c illustrates the electrical energy consumption with variable Q/NHCOOH values and cell voltages. Reducing the Q/NHCOOH and cell voltage decrease the consumed electrical energy for HCOOH production. The dashed lines in Fig. 1c represent the standard thermodynamic cell voltage and the minimum Q/NHCOOH value of specific overall reactions. Pronounced declines are observed in both factors within the CO2 and organic co-electrolysis systems. The state-of-the-art systems for HCOOH production from CO2 and H2O demonstrate electrical energy consumption in the range of 500–1200 kJ mol–1, operating at cell voltages of 2–4 V while maintaining Q/NHCOOH values above 2 × 105 C mol–1 (solid circles in Fig. 1c)23,24,25,26,27,28,29,30,31,32,33,34,35,36. In contrast, HCOOH production from CO2 and organic demonstrates substantially reduced electrical energy consumption (300–500 kJ mol–1, hollow circles in Fig. 1c), which can be ascribed to the reduced Q/NHCOOH (below 2 × 105 C mol–1) and lower operating voltage (2–3 V)37,38,39,40. The energy analysis validates that designing a membrane-free flow cell for simultaneous HCOOH production with reduced ohmic drop offers an exciting pathway towards energetically and economically efficient HCOOH electro-synthesis.
Efficient CO2-to-formate and glycerol-to-formate conversion
The development of efficient electrocatalysts for simultaneous HCOOH production from CO2 and glycerol is a prerequisite for enabling membrane-free flow cells operation. Alkaline electrolytes are commonly employed to ensure high activity and selectivity of both CO2R and GOR, with formate emerging as the primary product. Metal-based catalysts (Bi-, In-, Sn-based) have been demonstrated as excellent candidates for CO2R, achieving nearly 100 % selectivity for HCOO-/HCOOH41. Among these, low-cost Bi-based electrocatalysts are of particular interest in view of the industrially relevant current density (>0.1 A cm–2)42. Therefore, commercial Bi2O3 particles are adopted as the CO2-to-formate catalyst. Supplementary Figs. 3–4 reveal that micron-sized Bi2O3 particles transform into Bi nanoparticles with a size of around 50 nm after the CO2R test. Linear sweep voltammetry (LSV) curves in Supplementary Fig. 5a show that commercial Bi2O3 delivers higher current densities under a CO2 atmosphere in an AEM-based flow cell, indicating good performance for CO2R. Figure 2a reveals that the FE of formate consistently exceeds 95% at a given current density, with minor H2 and CO by-products (<4 %). Due to the electromigration of formate anions, a small fraction of formate is detected in the anolyte (Supplementary Fig. 5b)43. We also evaluate the glycerol tolerance of CO2R using a mixture of 1 M KOH and 0.5 M glycerol as catholyte. The FEs of formate are comparable to those in pure 1 M KOH electrolyte, despite a marginal increase in working potential (Supplementary Figs. 5c, d). As a result, the presence of glycerol in the electrolyte has a negligible impact on the CO2R performance of commercial Bi2O3.
a, b Half-reaction performance evaluated using a membrane-based flow cell with a three-electrode system. a The FEs of cathodic products and corresponding working potentials (E-iR; 85%-iR compensated) for Bi2O3 in 1 M KOH. b The FEs of GOR products and corresponding E-iR for NiV/Ni foam in 1 M KOH with 0.5 M glycerol addition. c, d Overall reaction performance evaluated using various flow cells with a two-electrode system across a current density range of 0.05–0.4 A cm–2. c Formate selectivity of the CO2 and glycerol co-electrolysis systems. d Electrical energy consumption for producing per mole of formate. e The stability test of the NOM-CO2R/GOR1.3 system for 313 h at 0.1 A cm–2 in 1 M KOH with 0.5 M glycerol addition at a flow rate of 5 mL min−1, and the inset in e corresponds to the optical image of channels in the graphite flow field at the cathode. Values are means, and error bars represent the standard deviation of three independent experiments. Source data are provided as a Source Data file.
GOR is a representative anodic half-reaction to pair with CO2R in the membrane-free flow cell. NiV layered double hydroxide nanosheets with a Ni/V atomic ratio of 3.6:1 are grown on Ni foam (denoted as NiV/Ni foam) and used as the electrocatalyst for GOR (Supplementary Fig. 6)18. The LSV curves (Supplementary Fig. 7) show that the NiV/Ni foam electrode exhibits a much lower onset potential in glycerol-containing electrolyte compared to that in glycerol-free electrolyte, indicating the occurrence of GOR. The NiV/Ni foam electrode achieves over 95 % FE for formate with minor CO and lactate by-products ( < 2%) across a current density range of 0.05–0.4 A cm–2 (Fig. 2b and Supplementary Fig. 8). The working potentials of the NiV/Ni foam electrode in glycerol-containing electrolyte are significantly lower than those in glycerol-free electrolyte, implying that replacing OER with GOR reduces the anodic potentials (Supplementary Fig. 9a). Moreover, the introduction of CO2 gas behind the NiV/Ni foam electrode does not compromise the GOR performance, confirming the robust CO2 tolerance of GOR (Supplementary Fig. 9b). The mutual tolerance between CO2R (glycerol tolerance) and GOR (CO2 tolerance) ensures their efficient co-electrolysis in a reaction environment where CO2 and glycerol coexist.
Proof-of-concept of the membrane-free flow cell
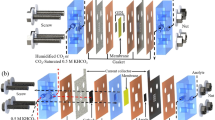
The successful preparation of commercial Bi2O3 and NiV/Ni foam electrodes with robust CO2R and GOR performance sets the stage for the efficient and simultaneous production of formate at both the cathode and anode in membrane-free flow cells (NOM-CO2R/GORx, where x represents electrolyte layer thickness) (Supplementary Fig. 10). For comparison, membrane-based flow cells with an electrolyte layer thickness of 9.5 mm, where CO2R pairs with either OER (AEM-CO2R/OER) or GOR (AEM-CO2R/GOR), are also implemented for formate electro-synthesis (Supplementary Fig. 11).
Experimentally, substituting OER with thermodynamically favorable GOR in the membrane-based flow cell reduces the cell voltage to some extent (Supplementary Fig. 12a). The membrane-free flow cell with an identical electrolyte layer thickness (NOM-CO2R/GOR9.5) exhibited even smaller cell voltage. Further decreasing the electrolyte layer thickness in membrane-free flow cells leads to a substantial decline in system resistance, resulting in a decreased ohmic drop (Supplementary Fig. 12b). As a result, the NOM-CO2R/GOR1.3 system shows the lowest cell voltage to achieve the same current density response, demonstrating a 19.7–51.6% reduction in cell voltage compared to the AEM-CO2R/OER system (Supplementary Fig. 13). Notably, the cell voltage remains below 2.7 V up to a current density of 0.4 A cm–2.
One challenge in operating membrane-free flow cells is possible anode-cathode interference that compromises formate yield. Supplementary Fig. 14 displays that formate–with yields comparable to those of the AEM-CO2R/GOR system–is the dominant product in membrane-free flow cells, accompanied by minor by-products. The employment of a gas diffusion electrode (GDE) facilitates the extraction of gaseous by-products and unreacted CO2, preventing both product crossover and CO2 loss (Supplementary Table 5). Moreover, the NiV/Ni foam electrode exhibits a preference for glycerol oxidation over formate oxidation, ensuring formate stability within the potential window for GOR (Supplementary Fig. 15a). Notably, the formate yields increase inversely with cell voltage in the membrane-free flow cells operated at 0.4 A cm–2, reaching the highest value in the NOM-CO2R/GOR1.3 system at 2.66 V. Similarly, at low cell voltages, the increase in formate concentration in the membrane-free flow cell approaches the theoretical value despite pre-existing formate in the electrolyte. However, the formate over-oxidation inevitably occurs under higher cell voltages (Supplementary Fig. 15b). These results underscore the importance of operating a membrane-free CO2 electrolyzer at low cell voltages.
Owing to the simultaneous formate production at both the cathode and anode, the Q/Nformate values drop markedly in the CO2 and glycerol co-electrolysis systems (Supplementary Fig. 16). To quantify the efficiency of formate production in the CO2 and glycerol co-electrolysis system, we define formate selectivity as the ratio of formate yield to its theoretical yield (assuming 100% electron utilization efficiency for formate production). The membrane-free flow cells, except NOM-CO2R/GOR9.5, deliver higher selectivity of formate than the AEM-CO2R/GOR system (Fig. 2c). Notably, the formate selectivity remains consistently above 90 % across a current density range of 0.05–0.4 A cm–2 in the NOM-CO2R/GOR1.3 system, highlighting its electron utilization efficiency for formate production.
The reduction in cell voltage and the enhancement of formate yield resulting from the engineering of the cell system align with our assumptions. As expected, the AEM-CO2R/GOR system delivers a lower value of electrical energy consumption for producing per mole of formate at the same current densities in comparison to the AEM-CO2R/OER system (Fig. 2d). The membrane-free flow cells with decreased cell voltage further reduce electrical energy consumption. Among these systems, NOM-CO2R/GOR1.3 demonstrates the lowest electrical energy consumption ( < 310 kJ mol–1) across a current density range of 0.05–0.4 A cm–2, operating at cell voltages below 2.7 V while delivering the highest formate yield. Compared to the AEM-CO2R/OER system, the NOM-CO2R/GOR1.3 system achieves a 56.1–73% reduction in electrical energy consumption for producing per mole of formate within the applied current density range (Supplementary Fig. 17).
We also compare the stability of membrane-based and membrane-free flow cells for CO2 and glycerol co-electrolysis at 0.1 A cm–2. The NOM-CO2R/GOR1.3 system demonstrates notable voltage stability, maintaining 2.25 V over 310 h of continuous operation (Fig. 2e and Supplementary Fig. 18). The evolution of H2 FE during long-term stability tests also differs between the two configurations (Supplementary Fig. 19). In the AEM-CO2R/GOR system, H2 FE surges from 2.3 to 24.8% accompanied by a voltage fluctuation, whereas membrane-free flow cells exhibit a gradual rise in H2 FE after ~130 h of operation. This difference suggests distinct degradation mechanisms, as discussed shortly. As both cell voltage and Q/Nformate increase over prolonged operation, the electrical energy consumption increases in all three flow cells (Fig. 2e and Supplementary Fig. 18). Among them, the NOM-CO2R/GOR1.3 system exhibits the smallest electrical energy consumption increase rate (0.157 kJ mol–1 h–1), corresponding to only a 19.3% increase relative to the initial performance.
The post-stability Bi2O3 and NiV/Ni foam electrodes display analogous morphologies in all three flow cells, characterized by uniformly covered nanosheets on the NiV/Ni foam electrode surface and stable Bi2O3-derived nanoparticles (Supplementary Fig. 20). Notably, distinct Ni deposition (0.03 mg cm–2) occurred on the post-stability Bi2O3 electrode in the NOM-CO2R/GOR1.3 system, while no Ni is detected on the cathode in the AEM-CO2R/GOR system after stability test (Supplementary Table 12 and Supplementary Fig. 21). The dissolution-deposition of Ni from the NiV/Ni foam electrode onto the Bi2O3 electrode likely acts as an active catalyst for the hydrogen evolution reaction (HER), explaining the gradual increase of H2 FE in membrane-free flow cells. Thus, developing a corrosion-free Ni-free anode might eliminate such chemical crosstalk between the two electrodes in membrane-free systems. In the AEM-CO2R/GOR system, the GDE flooding (Supplementary Fig. 22) and carbonate precipitation (Supplementary Fig. 18) are likely the primary causes of the abrupt increase in H2 FE within 60 h. However, these issues are difficult to avoid in the existing system.
Universality and scalability demonstration
The membrane-free flow cells are versatile to support the coupling of CO2R with other organic oxidation reactions. For instance, a CuSn/Cu foam electrode for MOR works with a Bi2O3 electrode in a membrane-free flow cell (inset in Fig. 3a and Supplementary Fig. 23). Formate is the only detected MOR product, with an FE over 90 % across a current density range of 0.05–0.25 A cm–2 (Supplementary Fig. 24a). The NOM-CO2R/MOR1.3 system achieves nearly 100 % formate selectivity, verifying the successful co-electrolysis of the CO2 and methanol in the membrane-free flow cell (Supplementary Fig. 24b). In particular, the cell voltage of the NOM-CO2R/MOR1.3 system is lower than 2.4 V, with the electrical energy consumption below 320 kJ mol–1 at a given current density (Fig. 3a). In comparison, coupling CO2R with GOR is more appealing than MOR, as it enables lower electrical energy consumption due to the reduced Q/Nformate (Fig. 3b and Supplementary Fig. 25).
a Contour plot of the electrical energy consumption for producing per mole of formate in the NOM-CO2R/MOR1.3 system as a function of measured cell voltage and formate yield across a current density range of 0.05–0.25 A cm–2. The insets in a correspond to the SEM image of CuSn/Cu foam (up) and LSV curves of CuSn/Cu foam electrode evaluated in 1 M KOH with or without 1 M methanol addition (down, 85%-iR compensated). b Comparison of the electrical energy consumption between the NOM-CO2R/GOR1.3 and NOM-CO2R/MOR1.3 systems. c FEs of by-products and formate selectivity at a given current in the scaled-up electrolyzer. d pH distribution between the cathode (left) and anode (right) in membrane-free flow cells with reaction areas of 2 cm2, 25 cm2, and 100 cm2, respectively, operated at 0.2 A cm–2 with an electrolyte flow rate of 15 mL min–1. e FEs of H2, and f electrical energy consumption for producing per mole of formate at a given current in the scaled-up electrolyzer under different operation conditions. Values are means, and error bars represent the standard deviation of three independent experiments. Source data are provided as a Source Data file.
To evaluate the scalability of membrane-free flow cells for large-scale HCOOH electro-synthesis, a scaled-up electrolyzer with an expanded reaction area of 22.5 cm2 is assembled (Supplementary Fig. 26a). As shown in Fig. 3c, the electrolyzer maintains >80 % formate selectivity at currents below 6 A. When further increasing the current to 9 A, the elevated cell voltage and intensified HER together lead to a decline in formate selectivity (Supplementary Fig. 26b). Finite element simulations using COMSOL Multiphysics are performed to probe pH and glycerol concentration variations during scale-up (Fig. 3d and Supplementary Figs. 26c–e). As the reaction areas expand to 25 cm2 and 100 cm2, the pH near both cathode and anode surfaces progressively decrease along the electrolyte flows (Fig. 3d). In a scaled-up system with a length of 100 mm, the average pH at the outlet slightly decreases from 14 to 13.88 (Supplementary Fig. 26c). Experimentally, electrolyte collected from the 22.5 cm2 electrolyzer at 0.22 A cm-2 exhibits a 0.1 pH drop–larger than that in the 2 cm2 electrolyzer–in agreement with simulation results (Supplementary Fig. 26f). Scaling up extends the average electrolyte flow path, increasing glycerol consumption and gradually reducing its concentration at the outlet, yet such variations are not significant in medium-size electrolyzers (Supplementary Figs. 26d, e).
Optimizing operation conditions provides a straightforward strategy to mitigate electrolyte composition changes during scale-up and minimize their impact on performance. Simulations reveal a more stable electrolyte composition in a 100 cm2 electrolyzer operating at an electrolyte flow rate of 50 mL min–1 (Supplementary Figs. 27a, b). Experimentally, increasing electrolyte flow rate and initial KOH concentration suppresses competing hydrogen evolution by ensuring a rapid and sufficient feedstock supply (Fig. 3e). Nevertheless, the FE of H2 still exceeds 10% at currents above 6 A, primarily due to the limited CO2R activity of Bi2O3 (Supplementary Fig. 27c). As expected, higher flow rate reduces pH variation at the outlet, and formate selectivity remains >80 % across a current range of 0.5–8 A (Supplementary Figs. 27d, e). Together with reduced cell voltages (Supplementary Fig. 27f), the scaled-up system enables energy-efficient formate production of 0.216 mol h–1 with an electrical energy consumption below 400 kJ mol–1 at 8 A, remaining competitive with conventional CO2 electrolysis systems in both productivity and energy efficiency (Fig. 3f).
Performance comparison with state-of-the-art CO2-to-HCOOH conversion systems
Membrane-based electrolyzers, such as flow cells and membrane electrode assembly (MEA) electrolyzers, have been widely employed for HCOOH production from CO2. Of note, formate generated in alkaline or neutral electrolytes can be converted into the target product, HCOOH, through an acidification process, with the associated costs being considered in the TEA, as discussed shortly. Compared with the reported state-of-the-art CO2-to-HCOOH conversion systems (Fig. 4a, b, see Supplementary Table 13 for details), the NOM-CO2R/GOR1.3 system achieves the lowest electrical energy consumption and the highest electricity-to-HCOOH conversion efficiency, highlighting its advantage in electricity utilization. In detail, the solid-state electrolyte-MEA (SSE-MEA, hollow dots in Fig. 4a, b) electrolyzers are capable of producing high-concentration formic acid24,26,44, yet such MEA design typically results in high cell voltage (e.g., 2.5–4 V) for CO2 and H2O co-electrolysis due to the complex membrane structure. The high electrical energy consumption (exceeding 500 kJ mol–1) and low electricity-to-HCOOH conversion efficiency (below 40%) at a current density above 0.1 A cm–2 counteract the advantage of downstream separation. Conventional MEA devices for CO2 and H2O co-electrolysis (solid dots in Fig. 4a, b) also encounter the challenges of high electrical energy consumption and low conversion efficiency25,32,45.
a Cell voltage versus electrical energy consumption. Bubble size reflects applied current densities. b Amount of passed charge versus electricity-to-HCOOH conversion efficiency. Bubble size reflects the HCOOH yield. The reference number corresponds to that in Supplementary Table 13. Source data are provided as a Source Data file.
Replacing OER with hydrogen oxidation reaction (HOR) in the MEA system significantly reduces the cell voltage for CO2 electrolysis46,47. For instance, cation exchange membrane (CEM)-based MEA achieves a current density of 0.6 A cm–2 for CO2 and H2 co-electrolysis at 2.2 V with an electrical energy consumption below 450 kJ mol–1, demonstrating advantageous performance compared to conventional MEA systems. However, this system incurs additional costs for sustainable hydrogen supply and only produces low-value products. Despite the high mass-energy density of H2, the prevailing high market price of green hydrogen (US$ 3–6 kg–1) results in higher feedstock costs for oxidizing H2 over glycerol at the anode, when fixing an equivalent amount of CO2.
In contrast, the simultaneous production of HCOOH at both the cathode and anode offers an exciting opportunity to maximize the energy value of the anode reaction, exhibiting an increased HCOOH yield under the identical charge passed (hollow dots with lines in Fig. 4b)37,38,48,49. However, existing co-electrolysis systems utilizing CO2 and glycerol struggle to achieve a high current response at low cell voltage (Fig. 4a). In sharp contrast, the NOM-CO2R/GOR1,3 system operates within a narrow cell voltage window (1.98–2.66 V), enabling a high current response (0.05–0.4 A cm–2) and a high yield. The lowest electrical energy consumption for HCOOH electro-synthesis and the highest electricity-to-HCOOH conversion efficiency of this system enable significant potential for industrial deployment. Notably, the co-electrolysis of CO2 and organics or H2 also utilizes the chemical energy of anode feedstocks, a factor that should not be overlooked when evaluating the overall energy conversion efficiency. Taking the chemical energy input into account, the NOM-CO2R/GOR1.3 system achieves an overall energy conversion efficiency above 45% across a current density range of 0.05–0.4 A cm–2 (Supplementary Fig. 28).
Techno-economic analysis for formic acid electro-synthesis
The economic feasibility of the membrane-free flow cell for HCOOH electro-synthesis is evaluated through a techno-economic analysis (TEA) based on the performance at 0.2 A cm–2 (see Supplementary Note 1). As shown in Fig. 5a, our TEA first evaluated the impact of external market parameters and system technical parameters on HCOOH production cost. External market parameters–including feedstock costs, energy prices, and carbon credits–exhibit significant regional variations. When powered by renewable electricity in China–characterized by low electricity price and average carbon emission factor–the production costs of HCOOH are significantly reduced across all device configurations. In the case of identical production areas, cost variations are also observed due to differences in system technical parameters (e.g., cell voltage, HCOOH selectivity) induced by device configurations. Among these systems, the NOM-CO2R/GOR1.3 system exhibits the lowest HCOOH production cost, achieving a 29.1–48.3% reduction compared to the AEM-CO2R/OER benchmark across major global economies (e.g., US, EU, China).
a The HCOOH production cost in four various flow cells across major global economies (e.g., US, EU, China) based on the performance at a current density of 0.2 A cm-2 with a CO2 flow rate of 28.5 mL min–1. b Single-variable sensitivity analysis for the production cost of HCOOH in the NOM-CO2R/GOR1.3 system (the base HCOOH production cost is set at US$ 0.573 kg–1). c HCOOH production cost comparison between the NOM-CO2R/GOR1.3 system (varying CO2 SPCE) and the CEM-CO2R/OER system (based on the performance in the work of Yang et al.45). d Waterfall chart showcasing the cumulative impact of key factors and processes optimization on the HCOOH production cost. Source data are provided as a Source Data file.
A single-variable sensitivity analysis is conducted to investigate the main parameters that govern the economic efficiency of HCOOH electro-synthesis in the NOM-CO2R/GOR1.3 system. As shown in Fig. 5b, the HCOOH production cost is most sensitive to electricity price, with a 50% decrease in electricity price yielding a 16.9% cost reduction. The HCOOH volume concentration is another critical variable, exhibiting a distinctly nonlinear relationship with HCOOH production cost. Continuous improvements in HCOOH volume concentration yield diminishing returns in terms of cost reduction, and thus maintaining 5–10% v/v is sufficient to avoid distillation and crystallization cost escalation. The single-pass conversion efficiency (SPCE) of CO2, which determines CO2 recovery costs, is also worth optimizing. Increasing SPCE from 10 to 50% reduces production cost by US$ 0.053 kg–1HCOOH (Fig. 5c and Supplementary Fig. 29). However, alkaline CO2 electrolysis has an inherent upper limit in CO2 SPCE and requires an additional acidification process to convert formate into HCOOH. Acidic CO2 electrolysis offers a promising alternative to address these issues. Nevertheless, even the most advanced CEM-CO2R/OER system45 still suffers from high electrical energy consumption (i.e., high electricity cost), counteracting these advantages in reduced acidification and CO2 recovery cost. For the membrane-free flow cell, replicating the performance metrics–particularly cell voltage and selectivity of glycerol-to-formate conversion–achieved in the alkaline electrolyte under acidic electrolytes remains a formidable challenge. Moreover, variations in other parameters have limited impacts on production cost, with each improvement resulting in less than a 10% reduction.
Additionally, we present the cumulative impact of key factors and process optimization on the HCOOH production cost in the form of a waterfall analysis (Fig. 5d). Operating expenditures (OPEX) account for more than 90% of the total HCOOH production cost, encompassing electrolyzer electricity cost, feedstock cost, downstream separation cost, etc. Among these, the electrolyzer electricity cost contributes 22.8% of the total cost, and its reduction depends on the availability of cheap electricity. If the electricity price drops from US$ 0.071 kWh–1 (baseline) to US$ 0.03 kWh–1 (representative of potential photovoltaic electricity), the corresponding production cost would be reduced by US$ 0.091 kg–1HCOOH. However, whether the annual average electricity price can remain below US$ 0.03 kWh–1 is still a matter for consideration in the near future, due to the intermittency of renewable energy. Moreover, the cell voltage, together with the electricity price, determines the electricity cost in the electrolysis process. Optimizing this technical parameter yields a 9.4% reduction in HCOOH production cost (US$ 0.048 kg–1HCOOH). In the present NOM-CO2R/GOR1.3 system, CO2R and GOR with large overpotential account for over 88% of the total cell voltage, thus dominating electrical energy consumption (Supplementary Fig. 30). This result highlights that developing high-performance catalysts, particularly for GOR, is crucial to further reducing electricity costs. Other technical parameters, including CO2 SPCE and HCOOH volume concentration, exhibit a direct correlation with downstream separation cost. For instance, improving the CO2 SPCE to 66.7 % can reduce CO2 recovery costs by US$ 0.024 kg–1HCOOH.
Notably, pairing CO2R with GOR introduces an additional feedstock cost of US$ 0.103 kg–1HCOOH (glycerol market price: US$ 0.36 kg–1), accounting for 20.2% of the total cost in the NOM-CO2R/GOR1.3 system. This burden could be alleviated by utilizing crude glycerol. However, another potential limitation is the considerable scale disparity between global glycerol production and excess CO2 emissions, as discussed by Verma et al.50. Specifically, even the complete utilization of global glycerol production (4.5–5 million metric tons per year, Mt yr–1) through co-electrolysis would fix only 8.6–9.6 Mt of CO2 annually (global CO2 emission: 14700 Mt yr–1). Looking ahead, the production of glycerol can scale with market expansion, and prices are likely to decline. Regardless of scale limitations, CO2 and glycerol co-electrolysis remains intrinsically valuable for upgrading two low-value/waste streams into value-added chemicals and advancing sustainable resource utilization. When combined with other non-equipment costs (assumed at US$ 0.1 kg–1HCOOH), including labor, insurance, packaging, transportation, storage, land, and taxation, the HCOOH production cost in a membrane-free flow cell would approach US$ 0.379 kg–1HCOOH, competing favorably with the traditional production method.
In summary, pairing CO2R with all-liquid-phase OORs, such as GOR and MOR, enables the efficient operation of the proposed membrane-free flow cell for HCOOH electro-synthesis. Energy analysis indicates that reducing cell voltage while increasing yield favors energy-efficient HCOOH production. Experimentally, the NOM-CO2R/GOR1.3 system achieves co-production of formate at both the cathode and anode, combined with minimized ohmic drop, resulting in the lowest electrical energy consumption (<310 kJ mol–1) at cell voltages below 2.7 V under industrially relevant current densities (0.05–0.4 A cm–2). This design saves up to 60.9% of electrical energy consumption compared to the AEM-CO2R/OER system, delivering an overall energy conversion efficiency of 53.7% and stable operation of 313 h at 0.1 A cm–2. Compared with the state-of-the-art systems, the NOM-CO2R/GOR1.3 system demonstrates competitive performance in terms of electrical energy consumption and electricity-to-HCOOH conversion efficiency. TEA highlights the economic viability of the membrane-free flow cell for HCOOH electro-synthesis across major global economies. A waterfall analysis outlines a viable roadmap for achieving low-cost formic acid production through further improving cell performance and taking advantage of cheap, renewable electricity and feedstocks. Overall, enabling low-voltage co-electrolysis of CO2 and glycerol in membrane-free flow cells will provide a promising and eco-friendly pathway for the profitable HCOOH electro-synthesis in the future.
Methods
Energy analysis
Electrical energy consumption \(\left({{\rm{kJ}}}{{{\rm{mol}}}}^{-1}\right)\) was calculated according to Eq. (1).
where Ecell is the cell voltage (in V), and Q (in C) is the amount of charge required to produce N (in mmol) of HCOOH. Experimentally, formate is the primary product in alkaline electrolytes; therefore, Q/NHCOOH is represented as Q/Nformate.
Catalyst synthesis and electrode preparation
Synthesis of NiV/Ni foam
The NiV/Ni foam electrode was synthesized by a hydrothermal method as reported18. At first, Ni foam (1 mm thickness, Kunshan GuangJiaYuan) was cut into 3 cm × 3 cm (or 5 cm × 5 cm for a scaled-up electrolyzer) in size and washed with HCl solution (2 M, Sinopharm Chemical Reagent, 36–38%) and deionized (DI) water (18.2 MΩ cm). Subsequently, 0.570 g of NiCl2·6H2O (Aladdin, 99%), 0.126 g of VCl3 (Energy Chemical, 99 %), 0.160 g of NH4Cl (Sinopharm Chemical Reagent, 99.5%), and 0.300 g of CH4N2O (Sinopharm Chemical Reagent, 99 %) were dissolved in 80 mL of DI water. The obtained solution was poured into a Teflon liner (100 mL), and a piece of Ni foam was put into the liner. Then, the liner was sealed in an autoclave and placed in an oven at 120 °C for 12 h. After cooling to 30 °C, the Ni foam was taken out, washed with DI water, and dried with N2 blowing. Finally, an electrochemical oxidation method was employed to activate the NiV/Ni foam electrode at 1.6 V vs RHE for 6000 s in 1 M KOH (Macklin, 95 %). To prepare the NiV/Ni foam electrode for the scaled-up electrolyzer, 160 mL of precursor solution with doubled concentration was poured into a Teflon liner (200 mL).
Synthesis of CuSn/Cu foam
The CuSn/Cu foam electrode was synthesized according to the method described in the literature37. At first, Cu foam (1 mm thickness, Kunshan GuangJiaYuan) was cut into 3 cm × 3 cm in size and washed with acetone (Sinopharm Chemical Reagent, 99.5%), HCl solution (2 M), and DI water. Subsequently, Cu foam was put into a solution containing 0.2 M (NH4)2S2O8 (Sinopharm Chemical Reagent, 98%) and 6 M NaOH (Sinopharm Chemical Reagent, 96%) for 20 min at 25 °C. The obtained Cu(OH)2 nanowires (NWs)/Cu foam was washed with DI water and dried with N2 blowing. Then, Cu(OH)2 NWs/Cu foam was annealed in a tube furnace at 200 °C in the air for 2 h with a heating rate of 5 °C min−1 to obtain CuO NWs/Cu foam. The Cu NWs/Cu foam was synthesized through electrochemical reduction of CuO NWs/Cu foam at –5 mA cm–2 for 5000 s. Then, Sn was electrodeposited onto the Cu NWs/Cu foam surface at –5 mA cm–2 for 2000 s in a solution containing 2 M KOH and 50 mM SnCl2 (Aladdin, 99%). Finally, the annealing process was performed in a tube furnace at 300 °C in a 5 % H2/Ar atmosphere with a flow rate of 30 mL min–1 for 3 h to obtain CuSn/Cu foam. The heating rate is 5 °C min–1 and the cooling rate is 1 °C min–1.
Electrode preparation
The cathode electrode was prepared by a drop-coating method. A catalyst ink containing 10 mg of commercial Bi2O3 (Aladdin, 99.8%), 3 mL of ethanol, and 22.2 mg of Nafion solution (Dupont, 5% in a mixture of lower aliphatic alcohol and water) was first prepared and sonicated for 20 min. Subsequently, the catalyst ink was dropped onto a gas diffusion electrode (GDE, SGL 29BC, SGL CARBON) with an area of 2.2 cm × 2.2 cm (or 5 cm × 5 cm for a scaled-up electrolyzer) and dried using an infrared lamp. The loading of Bi2O3 on the GDE was maintained at 2 mg cm–2 by weighing the electrode. The NiV/Ni foam and CuSn/Cu foam with an area of 2.2 cm × 2.2 cm were used as anode electrodes directly.
Material characterization
Morphologies of catalysts were observed by scanning electron microscopy (SEM, Phenom Scientific) and transmission electron microscopy (TEM, FEI Tecnai G2 F20). The crystal structures of catalysts were confirmed by an X-ray diffractometer (XRD) equipped with an X’Pert PRO, PANalytical diffractometer with a Cu Kα radiation source at a scan speed of 2° min–1. X-ray photoelectron spectroscopy (XPS) was performed to analyze the Ni species on the post-stability Bi2O3 electrode surface. Inductively coupled plasma optical emission spectroscopy (ICP-OES) was employed to quantify the deposition mass of Ni on the post-stability Bi2O3 electrode.
Electrolyzer assembly and performance evaluation
Electrolyzer assembly
The electrochemical experiments were conducted in a homemade flow cell. Cathode and anode electrodes with a reaction area of 2 cm2 were placed between the gas compartment and the electrolyte compartment, and the catalyst layer faced the electrolyte compartment. A fresh GDE was placed on the back of the anode electrode, which is not in the scaled-up electrolyzer. For the membrane-based flow cell (i.e., AEM-CO2R/OER, AEM-CO2R/GOR), the cathode and anode electrodes were separated by an untreated anion exchange membrane (AEM, FAA-3-PK-130, FuMA-Tech) with a distance of 9.5 mm. The distance between the cathode and anode in membrane-free flow cells varied from 9.5 to 1.3 mm.
Performance evaluation
The overall reaction performances in membrane-based and membrane-free flow cells were evaluated by using a two-electrode system on an Autolab workstation (PGSTAT302N) equipped with a 10 A current booster at ambient temperature and pressure. The cell voltage was recorded by the workstation. CO2 and Ar were continuously passed through the back of cathode and anode electrodes with a flow rate of 28.5 mL min–1, with the flow precisely regulated by a mass flow controller (MFC), respectively, except where noted. The continuous flow of Ar through the back of the anode was to prevent the localized accumulation of GOR products within the Ni foam channels. Freshly prepared electrolyte was continuously supplied to the electrolyzer at 15 mL min–1 using a peristaltic pump in a non-recirculating single-pass mode, except where noted. The catholyte for AEM-CO2R/OER and AEM-CO2R/GOR was 1 M KOH. The anolyte for AEM-CO2R/OER and AEM-CO2R/GOR was 1 M KOH without and with 0.5 M glycerol addition, respectively. The electrolyte for NOM-CO2R/GORx was 1 M KOH with 0.5 M glycerol addition. The electrolyte for NOM-CO2R/MOR was 1 M KOH with 1 M methanol addition. Bi2O3 deposited on GDE was used as the cathode. NiV/Ni foam and CuSn/Cu foam electrodes were used as the anodes, respectively. The scaled-up membrane-free flow cell features a reaction area of 22.5 cm2 and an electrolyte chamber thickness of 4.5 mm. Under amper-level current, the CO2 flow rate and electrolyte flow rate are maintained at 100 mL min–1 and 15 mL min–1, respectively, except where noted.
The half-reaction performances, encompassing CO2R, OER, GOR, and MOR, were evaluated in the membrane-based flow cell by employing a typical three-electrode system. The Ag/AgCl electrode was used as the reference electrode. When evaluating the anodic half-reaction performances of NiV/Ni foam and CuSn/Cu foam, the Bi2O3-catalyzed HER half-reaction was paired with GOR and MOR by supplying Ar with a flow rate of 28.5 mL min–1 into the gas compartment at the cathode. All other operation conditions were maintained consistent with the overall reaction test. Finally, the working potential was 85%-iR compensated and converted to the RHE scale according to Eq. (2). The pHs of electrolytes in different systems were tabulated in Supplementary Tables 14, 15.
where i is the applied current (in A), Rs is the solution resistance (in Ω), \({E}^{\theta }({{\rm{Ag}}}/{{\rm{AgCl}}})\) is 0.198 V, and \({E}^{\theta }({{\rm{Hg}}}/{{\rm{HgO}}})\) is 0.098 V.
The LSV tests for the commercial Bi2O3 electrode were carried out in a membrane-based flow cell by employing a three-electrode system on a CHI 760E workstation. The Hg/HgO electrode and NiV/Ni foam electrode were used as the reference electrode and counter electrode. The electrolyte is 1 M KOH, and the gas flow rate is 28.5 mL min–1. The LSV tests for NiV/Ni foam and CuSn/Cu foam electrodes were carried out in a typical single-compartment electrolytic cell by employing a three-electrode system. The Hg/HgO electrode and Pt wire electrode were used as the reference electrode and counter electrode. The working potential was 85%-iR compensated and converted to the RHE scale according to Eq. (2)
For electrochemical impedance spectroscopy (EIS), all measurements were performed at open circuit potential (OCP) with a frequency range between 1 × 106 Hz and 0.1 Hz, with an AC amplitude of 10 mV. The system resistances were directly extracted from the EIS spectra without any fitting procedure.
Product detection and analysis
Detection
The gas products and liquid products were quantified by online gas chromatography and 1H nuclear magnetic resonance spectroscopy (1H-NMR, Bruker 400 M), respectively. The 0.2 mM Dimethyl sulfoxide (DMSO, Aladdin, 99.8 %) diluted in D2O (Energy Chemical, 99.9%) was used as an internal standard for 1H-NMR, and a water suppression mode was also adopted.
FEs of CO2R, GOR, and MOR products were calculated according to Eq. (3).
where Z is the number of electrons required to form 1 mol of product, F is the Faraday constant, N is the amount of product (in mol), \(i\) is the current (A), and t is the running time (in s). The Z is 2 for CO2-to-formate conversion, 8/3 for glycerol-to-formate conversion, and 4 for methanol-to-formate conversion.
Selectivity of formate for the co-electrolysis of CO2 and organic system was defined as the ratio of experimental formate yield to theoretical formate yield.
The experimental formate yield \(\left({{\rm{mmol}}}\,{{{\rm{cm}}}}^{-1}{{{\rm{h}}}}^{-1}\right)\) was calculated according to Eq. (4).
where Nformate is the total amount of formate in the catholyte and anolyte detected by 1H NMR (in mmol), s is the working area (in cm2), and T is the running time (in h).
The theoretical formate yield \(({{\rm{mmol}}}\,{{{\rm{cm}}}}^{-1}{{{\rm{h}}}}^{-1})\) was calculated according to the deformable Eq. (5).
where i is the current density (in A·cm-2). The theoretical formate yield of the CO2 and organic compound co-electrolysis system is the formate yield sum of CO2R and anode half-reaction.
Thus, the selectivity of formate for the co-electrolysis of CO2 and organic system was calculated according to Eq. (6).
Cell voltage reduction of the CO2R/GOR system was calculated according to Eq. (7).
All Ecell values incorporated in this equation are the means of three independent measurements.
Electrical energy consumption saving of the CO2R/GOR system was calculated according to Eq. (8).
All electrical energy consumption values incorporated in this equation are the means of three independent measurements.
Electricity-to-HCOOH conversion efficiency was calculated according to Eq. (9).
where \({\Delta H}_{{{\rm{HCOOH}}}}^{0}\) is the heating value of HCOOH ( = 254 kJ mol–1). Ntotal, Qtotal, and Ecell incorporated in this equation are the means of three independent measurements in the NOM-CO2R/GOR1.3 system.
Overall energy conversion efficiency was calculated according to Eq. (10).
where \({\Delta H}_{{{\rm{carrier}}}}^{0}\) is the heating value of energy carriers, such as glycerol ( = 1654 kJ mol–1), methanol ( = 638 kJ mol–1), and H2 ( = 242 kJ mol–1). \({N}_{{{\rm{carrier}}}}\) is the total amount of consumed energy carrier (in mol), which is calculated according to the formate yield. As the literature does not explicitly report the HCOOH yield at the anode, we estimated the contribution of the anodic reaction by proportionally distributing the total formate yield (listed in Supplementary Table 13) based on the ratio of formate production from the anodic and cathodic reactions. Ntotal, Qtotal, Ncarrier, and Ecell incorporated in this equation are the means of three independent measurements in the NOM-CO2R/GOR1.3 and NOM-CO2R/MOR1.3 systems.
Single-pass conversion efficiency (SPCE) of CO2R was calculated according to Eq. (11).
Where iproducts is the partial current of CO2R products (in A), t is the running time (in min), Vm is the molar volume ( = 24500 mL mol–1), and \(\upsilon\) is the flow rate of CO2 (in mL min-1).
COMSOL multiphysics simulation
To investigate the pH and glycerol concentration variations with increasing electrolyzer scale, two-dimensional multiphysics models were developed using COMSOL Multiphysics, integrating Laminar Flow, Diluted Species Transport, Chemical Reaction, and Secondary Current Distribution modules. The spatiotemporal distributions of OH⁻ and C₃H₈O₃ were evaluated in membrane-free flow cells with three reaction areas (2, 25, and 100 cm²) and two electrolyte flow rates (15 and 50 mL min–1). The inter-electrode gap was fixed at 1.3 mm, and the initial electrolyte consisted of 1 M KOH with 0.5 M glycerol.
A porous diffusion electrode layer was explicitly defined. CO₂ was introduced at the electrode boundary as a saturated aqueous solution, with its equilibrium concentration governed by Henry’s law:
where p is the CO2 partial pressure (atm); k is Henry’s constant (atm mol-1), and c is the dissolved CO₂ concentration.
Upon permeating the cathode diffusion layer, CO₂ underwent electrochemical reduction via a proton-coupled electron transfer process to produce HCOO⁻:
Meanwhile, glycerol oxidation occurred at the anode:
The local current density \(({i}_{{{\rm{loc}}}})\) distribution was resolved through the Butler-Volmer Eq. (15) coupled with the Nernst-Planck Eq. (16) for species flux.
where \({i}_{0}\) is the exchange current density (A m–²), \({C}_{{{\rm{R}}}}\) and \({C}_{{{\rm{O}}}}\) are the reduced and oxidized species concentrations, n is the number of electrons transferred, \({\alpha }_{{{\rm{a}}}}\) and \({\alpha }_{{{\rm{c}}}}\) are charge transfer coefficients, R is the universal gas constant (8.314 J mol–1·K–1), F is the Faraday constant (96485 C mol–1), and \(\eta\) is local overpotential (V).
The simulations were performed at an applied cell voltage of 2.31 V, sustaining a nominal current density of 0.2 A cm–². Unreacted CO₂ participated in homogeneous bulk electrolyte reactions with OH⁻, described by:
The forward rate constant \({k}_{{{\rm{f}}}}\) described by the Arrhenius equation51.
where A is the pre-exponential factor (m6 s–1·mol–2) and \({E}_{{{\rm{a}}}}\) is activation energy (J mol–1).
Species transport within the electrolyzer was dominated by convective-diffusive mechanisms, and the flow regime remained strictly laminar due to the low local Reynolds numbers.
The simulated pH profiles were determined from the steady-state OH- concentration via:
Techno-economic analysis
The techno-economic analysis proceeded in the following steps: (1) Determine the target product; (2) Determine basic plant parameters and market parameters; (3) Determine the reaction system (CO2R/OER or CO2R/GOR); (4) Determine intermediate parameters; (4) The capital expenditures (CAPEX) and the operating expenditures (OPEX) calculation; (5) Economic analysis. The TEA model and methodology were detailed in Supplementary Note 1.
Breakdown of the cell voltage
The cell voltage was divided into the standard thermodynamic cell voltage, the overpotentials of cathodic and anodic half-reactions, and the ohmic drop between the cathode and the anode. The calculation method for the breakdown of the cell voltage was described in Supplementary Note 2.
Data availability
The data supporting the findings of this study are available within the paper and Supplementary Information file, and are also provided in the Source Data file. Other relevant data are available from the corresponding authors upon request. Source data are provided with this paper.
References
Li, X., Wu, X., Lv, X., Wang, J. & Wu, H. B. Recent advances in metal-based electrocatalysts with hetero-interfaces for CO2 reduction reaction. Chem. Catal. 2, 262–291 (2022).
Whipple, D. T. & Kenis, P. J. A. Prospects of CO2 utilization via direct heterogeneous electrochemical reduction. J. Phys. Chem. Lett. 1, 3451–3458 (2010).
De Luna, P. et al. What would it take for renewably powered electrosynthesis to displace petrochemical processes?. Science 364, 350 (2019).
Shin, H., Hansen, K. U. & Jiao, F. Techno-economic assessment of low-temperature carbon dioxide electrolysis. Nat. Sustain. 4, 911–919 (2021).
Onishi, N. et al. Development of effective catalysts for hydrogen storage technology using formic acid. Adv. Energy Mater. 9, 1801275 (2018).
Agarwal, A. S., Zhai, Y., Hill, D. & Sridhar, N. The electrochemical reduction of carbon dioxide to formate/formic acid: engineering and economic feasibility. ChemSusChem 4, 1301–1310 (2011).
Aresta, M., Dibenedetto, A. & Angelini, A. Catalysis for the valorization of exhaust carbon: from CO2 to chemicals, materials, and fuels. Technological use of CO2. Chem. Rev. 114, 1709–1742 (2014).
Li, N. et al. In-situ formation of ligand-stabilized bismuth nanosheets for efficient CO2 conversion. Appl. Catal. B: Environ. 297, 120481 (2021).
Nankya, R. et al. Cobalt-doped bismuth nanosheet catalyst for enhanced electrochemical CO2 reduction to electrolyte-free formic acid. Angew. Chem. Int. Ed. 63, e202403671 (2024).
Dong, J. et al. Continuous electroproduction of formate via CO2 reduction on local symmetry-broken single-atom catalysts. Nat. Commun. 14, 6849 (2023).
Liu, X. et al. Mastering the lattice strain in bismuth-based electrocatalysts for efficient CO2-to-formate conversion. Adv. Funct. Mater. 34, 2400928 (2024).
Lai, W., Qiao, Y., Zhang, J., Lin, Z. & Huang, H. Design strategies for markedly enhancing energy efficiency in the electrocatalytic CO2 reduction reaction. Energy Environ. Sci. 15, 3603–3629 (2022).
Na, J. et al. General technoeconomic analysis for electrochemical coproduction coupling carbon dioxide reduction with organic oxidation. Nat. Commun. 10, 5193 (2019).
Vass, Á, Kormányos, A., Kószó, Z., Endrődi, B. & Janáky, C. Anode catalysts in CO2 electrolysis: challenges and untapped opportunities. ACS Catal. 12, 1037–1051 (2022).
Dodekatos, G., Schünemann, S. & Tüysüz, H. Recent advances in thermo-, photo-, and electrocatalytic glycerol oxidation. ACS Catal. 8, 6301–6333 (2018).
Li, Y., Wei, X., Chen, L., Shi, J. & He, M. Nickel-molybdenum nitride nanoplate electrocatalysts for concurrent electrolytic hydrogen and formate productions. Nat. Commun. 10, 5335 (2019).
Xia, Z. et al. Vacancy optimized coordination on nickel oxide for selective electrocatalytic oxidation of glycerol. ACS Catal. 14, 1930–1938 (2024).
Dong, L., Chang, G.-R., Feng, Y., Yao, X.-Z. & Yu, X.-Y. Regulating Ni site in NiV LDH for efficient electrocatalytic production of formate and hydrogen by glycerol electrolysis. Rare Met. 41, 1583–1594 (2022).
Bardow, A. & Wessling, M. Converting two wastes to value. Nat. Energy 4, 440–441 (2019).
Xie, K. et al. Eliminating the need for anodic gas separation in CO2 electroreduction systems via liquid-to-liquid anodic upgrading. Nat. Commun. 13, 3070 (2022).
Monroe, M. M., Lobaccaro, P., Lum, Y. & Ager, J. W. Membraneless laminar flow cell for electrocatalytic CO2 reduction with liquid product separation. J. Phys. D: Appl. Phys. 50, 154006 (2017).
van den Bosch, B. et al. Formate over-oxidation limits industrialization of glycerol oxidation paired with carbon dioxide reduction to formate. Chempluschem 88, e202300112 (2023).
Hou, P., Wang, X. & Kang, P. Membrane-electrode assembly electrolysis of CO2 to formate using indium nitride nanomaterials. J. CO2 Util. 45, 101449 (2021).
Xia, C. et al. Continuous production of pure liquid fuel solutions via electrocatalytic CO2 reduction using solid-electrolyte devices. Nat. Energy 4, 776–785 (2019).
Chen, Y. et al. A robust, scalable platform for the electrochemical conversion of CO2 to formate: identifying pathways to higher energy efficiencies. ACS Energy Lett. 5, 1825–1833 (2020).
Lin, L. et al. A nanocomposite of Bi clusters and Bi2O2CO3 sheets for highly efficient electrocatalytic reduction of CO2 to formate. Angew. Chem. Int. Ed. 62, 202214959 (2022).
Del Castillo, A. et al. Sn nanoparticles on gas diffusion electrodes: synthesis, characterization and use for continuous CO2 electroreduction to formate. J. CO2 Util. 18, 222–228 (2017).
Lee, W., Kim, Y. E., Youn, M. H., Jeong, S. K. & Park, K. T. Catholyte-free electrocatalytic CO2 reduction to formate. Angew. Chem. Int. Ed. 57, 6883–6887 (2018).
Li, M. et al. Local steric hindrance for CO2 electroreduction at a thermodynamic potential and wide working window. Adv. Energy Mater. 14, 2303073 (2024).
Yuan, C. Y. et al. Constructing metal(II)-sulfate site catalysts toward low overpotential carbon dioxide electroreduction to fuel chemicals. Angew. Chem. Int. Ed. 63, e202405255 (2024).
Li, W. et al. Beyond leverage in activity and stability toward CO2 electroreduction to formate over a bismuth catalyst. ACS Catal. 14, 8050–8061 (2024).
Huang, H. et al. CuO (111) microcrystalline evoked indium-organic framework for efficient electroreduction of CO2 to formate. Small, 20, 2400441 (2024).
Yu, X. et al. Coverage enhancement accelerates acidic CO2 electrolysis at ampere-level current with high energy and carbon efficiencies. Nat. Commun. 15, 1711 (2024).
Li, L. et al. Stable, active CO2 reduction to formate via redox-modulated stabilization of active sites. Nat. Commun. 12, 5223 (2021).
Li, L., Liu, Z., Yu, X. & Zhong, M. Achieving high single-pass carbon conversion efficiencies in durable CO2 electroreduction in strong acids via electrode structure engineering. Angew. Chem. Int. Ed. 62, e202300226 (2023).
Li, T., Lees, E. W., Zhang, Z. & Berlinguette, C. P. Conversion of bicarbonate to formate in an electrochemical flow reactor. ACS Energy Lett. 5, 2624–2630 (2020).
Li, Y. et al. Coupling CO2 reduction with CH3OH oxidation for efficient electrosynthesis of formate on hierarchical bifunctional CuSn alloy. Nano Energy 98, 107277 (2022).
Pei, Y., Pi, Z., Zhong, H., Cheng, J. & Jin, F. Glycerol oxidation-assisted electrochemical CO2 reduction for the dual production of formate. J. Mater. Chem. A 10, 1309–1319 (2021).
Guo, X. et al. Engineering hydrogen generation sites to promote electrocatalytic CO2 reduction to formate. ACS Catal. 12, 10551–10559 (2022).
Tan, D., Wulan, B., Ma, J., Cao, X. & Zhang, J. Interface molecular functionalization of Cu2O for synchronous electrocatalytic generation of formate. Nano Lett. 22, 6298–6305 (2022).
Han, N., Ding, P., He, L., Li, Y. & Li, Y. Promises of main group metal-based nanostructured materials for electrochemical CO2 reduction to formate. Adv. Energy Mater. 10, 1902338 (2019).
Guan, Y., Liu, M., Rao, X., Liu, Y. & Zhang, J. Electrochemical reduction of carbon dioxide (CO2): bismuth-based electrocatalysts. J. Mater. Chem. A 9, 13770–13803 (2021).
Wang, N. et al. Suppressing the liquid product crossover in electrochemical CO2 reduction. SmartMat 2, 12–16 (2021).
Elgazzar, A. et al. Electrochemical CO2 reduction to formic acid with high carbon efficiency. ACS Energy Lett. 10, 450–458 (2025).
Yang, K. et al. An acid-tolerant metal-organic framework for industrial CO2 electrolysis using a proton exchange membrane. Nat. Commun. 15, 7060 (2024).
Fan, L., Xia, C., Zhu, P., Lu, Y. & Wang, H. Electrochemical CO2 reduction to high-concentration pure formic acid solutions in an all-solid-state reactor. Nat. Commun. 11, 3633 (2020).
Fang, W. et al. Durable CO2 conversion in the proton-exchange membrane system. Nature 626, 86–91 (2024).
Zhu, Z. et al. Tailoring the electrolyte microenvironment of indium catalysts for enhanced formic acid electrosynthesis. J. Energy Chem. 105, 54–64 (2025).
Li, X., Chen, Q., Sun, W., He, C. & Wen, Z. Electron-efficient co-electrosynthesis of formates from CO2 and methanol feedstocks. Angew. Chem. Int. Ed. 63, e202412410 (2024).
Verma, S., Lu, S. & Kenis, P. J. A. Co-electrolysis of CO2 and glycerol as a pathway to carbon chemicals with improved technoeconomics due to low electricity consumption. Nat. Energy 4, 466–474 (2019).
Thee, H. et al. A kinetic study of CO2 capture with potassium carbonate solutions promoted with various amino acids: Glycine, sarcosine and proline. Int. J. Greenh. Gas. Con. 20, 212–222 (2014).
Acknowledgements
This work was supported by the National Key R&D Program of China (2023YFB4203900, H.B.W. and Q.L.), the Fundamental Research Funds for the Central Universities (226-2024-00075, H.B.W.), the National Natural Science Foundation of China Youth Fund (22402203, X.Li), and the China Postdoctoral Science Foundation (2024M763196, X.Li; GZC20232592, X.Li).
Author information
Authors and Affiliations
Contributions
X.Li, Q.L. and H.B.W. conceived the idea. X.Li performed the experiments and analyzed the results. K.G. performed the techno-economic analysis. M.Q. performed the finite element simulation. N.L. supplied catalysts. X.Lv and X.W. assisted in analyzing the results. X.Li, Q.L., and H.B.W. co-wrote the manuscript. Q.L. and H.B.W. supervised the project.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Bart van den Bosch and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Li, X., Gao, K., Qu, M. et al. Membrane-free CO2 electrolyzer design for economically efficient formic acid electro-synthesis. Nat Commun 16, 9237 (2025). https://doi.org/10.1038/s41467-025-64306-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-64306-0