Abstract
The PD-L1 immune checkpoint ligates the PD-1 immune checkpoint, and antibodies against either are effective in treating selected human cancers. Although PD-L2 also ligates PD-1, αPD-L2 is little studied as cancer immunotherapy. We previously showed that αPD-L1 treated young B16-bearing mice but failed in aged. We show here that αPD-L2 fails in young B16-bearing mice but is effective in aged. αPD-L2 increases tumour-infiltrating interferon-γ+ immune cell prevalence and interferon-γ production in aged but not young melanoma-bearing hosts in an unexpectedly IL-17-dependent manner. We also show improved αPD-L2 efficacy with advancing age in another tumour type, and αPD-L2-responsive tumours in young hosts, but without IL-17 dependence. The immune B16 microenvironment in aged IL-17-deficient hosts resembles that of young hosts, and exogenous IL-17 elicits αPD-L2 melanoma efficacy in young hosts, demonstrating IL-17 influence on aged immune outcomes. Mechanistic insights into age-related αPD-L2 efficacy could improve immune checkpoint blockade efficacy including in younger patients, address immune checkpoint blockade-resistant tumours and improve understanding of age effects on anti-cancer immunity.
Similar content being viewed by others
Introduction
Immune checkpoint blockade (ICB) cancer immunotherapy has greatly improved cancer treatment, with FDA approval of numerous monoclonal antibodies blocking the PD-1, PD-L1, CTLA-4 or Lag3 immune checkpoints1, with others in trials. While approved ICB antibodies successfully treat diverse cancers, under 15% of all cancers respond, warranting testing additional ICB targets. Furthermore, although age is the biggest cancer risk factor2, pre-clinical studies testing ICB efficacy were initially and still predominantly done on young hosts. Age effects on ICB efficacy in melanoma or non-small cell lung cancer are contradictory, with some studies suggesting age worsens, whereas others show it improves ICB efficacy3,4. A meta-analysis found that aged (> 75 years) patients did not benefit from αPD-1/αPD-L1 ICB except those with melanoma5. αPD-1 (pembrolizumab) efficacy in patients with advanced melanomas improved as a function of age, also modeled in mice6, in contrast to age-related declines in efficacy of targeted small molecule melanoma therapies7.
Here, we show that αPD-L2 ICB treats distinct murine melanoma models in aged mice, but remarkably, not in young mice and treated a bladder cancer model more effectively in aged. αPD-L2 promoted IL-17-driven interferon-γ+ (IFNγ+) cell accumulation and IFNγ production in aged hosts that drove efficacy, and required both γδ T cells and conventional CD8+ T cells whereas γδ T cells were dispensable in young hosts.
Consistent with our prior report8, aged mice express higher tumour infiltrating immune cell PD-1, PD-L1, and PD-L2 and higher tumour PD-L1 and PD-L2 versus young. However, we find melanoma PD-L2 dispensable for αPD-L2 melanoma efficacy. αPD-L2 efficacy in other cancer models tested is not age- or melanoma-specific, and is independent of IL-17 and γδ T cells, distinct from anti-melanoma mechanisms. Thus, αPD-L2 exhibits mechanistic efficacy differences divergent from other ICB antibodies that are exploitable in distinct settings and suggest combinatorial efficacy with other ICB agents.
Results
PD-1/PD-L2 signals are immunopathologic in aged mice
We reported that αPD-1 significantly reduced subcutaneous B16F10 (“B16”) tumour growth in young and aged mice whereas αPD-L1 was efficacious only in young8,9. PD-1 ligates PD-L2 in addition to PD-L110,11, which could account for these dichotomous outcomes. We previously showed that tumour infiltrating CD45+ immune cell and B16 tumour PD-L1 expression are significantly higher in the aged B16 tumour microenvironment (TME) versus young8. To investigate the role of TME PD-L1 in αPD-1 treatment efficacy further, we eliminated all PD-L1 signals in vivo by challenging aged PD-L1KO mice with PD-L1KO B16 cells and treating with αPD-1. Despite total lack of PD-L1, αPD-1 effectively reduced tumour growth (Fig. 1a, Supplementary. Figure 1a).
Tumour growth curves of a PD-L1KO aged mice challenged subcutaneously with PD-L1KO B16 murine melanoma and treated with αPD-1 (200 µg/mouse) or isotype control (200 µg/mouse) intraperitoneally every 4 days starting on day 7 (isotype n = 3 mice, αPD-1 n = 3 mice), b wild type young (isotype n = 5 mice, αPD-L2 n = 5 mice), c wild type aged mice (isotype n = 6 mice; αPD-L2 n = 6 mice) challenged subcutaneously with B16 murine melanoma and treated with αPD-L2 (200 µg/mouse) or isotype control (200 µg/mouse) intraperitoneally every 4 days starting on day 3, d wild type young (isotype n = 8 mice, αPD-L2 n = 6 mice), e wild type aged mice (isotype n = 6 mice; αPD-L2 n = 7 mice) challenged subcutaneously with NCH1 murine melanoma and treated with αPD-L2 or isotype control as above, f wild type young (isotype n = 6 mice, αPD-L2 n = 5 mice) or g wild type aged mice (isotype n = 5 mice, αPD-L2 n = 5 mice) harboring subcutaneous MB49 murine bladder cancer tumours treated with αPD-L2 or isotype control as above. Flow cytometry data of intratumoural h prevalence of IFNγ+CD45+ immune cells, i mean fluorescence intensity (MFI) of IFNγ in CD45+ immune cells, j prevalence of IFNγ+CD4+ T cells (CD45+CD3+B220-), k MFI of IFNγ in CD4+ T cells, l prevalence of IFNγ+CD8+ T cells (CD45+CD3+B220-), m MFI of IFNγ in CD8+ T cells, n prevalence of IFNγ+γδ TCR+γδ T cells (CD45+CD3+B220-), and o MFI of IFNγ in γδ TCR+ γδ T cells (CD45+CD3+B220-) from wild type young or wild type aged mice as indicated harboring MB49 tumours as above. Tumour growth curves of p wild type young (isotype n = 4 mice, αPD-L2 n = 4 mice) or q wild type aged mice (isotype n = 3 mice, αPD-L2 n = 4 mice) harboring orthotopic ID8agg murine ovarian cancer tumours treated with αPD-L2 (200 µg/mouse) or isotype control (200 µg/mouse) intraperitoneally every 4 days starting on day 3. r Comparison of tumour growth curves of ID8agg tumours in wild type aged versus young mice (young mice n = 4 mice; aged mice n = 3 mice). Standard error of mean indicated; p-values, (a–g, p–r) two-way ANOVA, (h–o) unpaired Student’s t-test.
αPD-L2 is effective in aged, but not young mice bearing distinct melanoma types
To test PD-L2 signalling directly, we challenged young wild type (WT) mice with B16 subcutaneously and treated with αPD-L2, but tumour growth was unaffected (Fig. 1b, Supplementary Fig. 1b). Remarkably, yet consistent with Fig. 1a data, αPD-L2 significantly reduced B16 tumour growth in aged WT mice (Fig. 1c, Supplementary Fig. 1c). To exclude B16-specific effects, we tested a distinct NrasQ61R-mutant NCH1 melanoma line that we derived from a well-established autochthonus melanoma model12. αPD-L2 also significantly reduced its subcutaneous growth in aged but not young mice (Fig. 1d, e, Supplementary Fig. 1d,e), indicating a strong age effect in αPD-L2 efficacy in two independent melanoma models. To exclude tumour PDL1 as a mechanism for αPDL2 efficacy, we genetically deleted PD-L1 from NCH1 and B16, challenged young WT mice, and treated with αPD-L2, but it was ineffective (Supplementary Fig. 2a,b).
αPD-L2 anti-tumour efficacy is improved with age in distinct cancer types
To assess if αPD-L2 exerted age-related efficacy in other tumours, we used heterotopic subcutaneous MB49 bladder cancer challenged in young and aged wild type mice. Although αPD-L2 was effective in both young and aged, its efficacy was better in aged (Fig. 1f, g, Supplementary Fig. 1f,g) demonstrating that αPD-L2 efficacy is not melanoma or aged skin-specific, consistent with recent reports of αPD-L2 efficacy in distinct tumour models in young mice13,14. Aged and young WT mice bearing MB49 tumours had similar, but extremely low infiltration of IFNγ+ CD45+ immune cells (Fig. 1h). However, per cell production of IFNγ was significantly higher in total CD45+ immune cells in young WT (Fig. 1i). Among CD4+, CD8+ and γδ T cells, IFNγ+ prevalence was low and per-cell IFNγ was negligible in young and aged WT (Fig. 1j-o), suggesting that αPD-L2 efficacy relies little on IFNγ in subcutaneously challenged MB49 bladder cancer. By contrast, orthotopic ID8agg ovarian cancer did not respond to αPD-L2 in aged or young mice and the tumours grew significantly faster in aged (Fig. 1p–r, Supplementary Fig. 1h,i), which could reflect properties of ovarian cancer cells, the peritoneal environment or other factors.
Based on the essential role for IFNγ in ICB efficacy in younger hosts15,16,17,18,19, we tested mechanistic IFNγ contributions to αPD-L2 melanoma efficacy using aged IFNγKO mice. αPD-L2 did not control B16 tumour growth or improve mouse survival in aged IFNγKO mice (Fig. 2a,b), consistent with a major mechanistic role for IFNγ in age-related αPD-L2 efficacy against melanoma.
a Overall and individual tumour growth plots and b survival of aged IFNγKO mice bearing B16 melanomas treated with isotype (200 µg/mouse) or αPD-L2 (200 µg/mouse) (isotype n = 4 mice, αPD-L2 n = 5 mice). c Tumour growth curve of IL-17KO aged mice harboring B16 melanomas treated with αPD-L2 or isotype control as above (isotype n = 5 mice, αPD-L2 n = 4 mice). d Tumour growth curve of wild type aged mice harboring B16 melanomas treated with αPD-L2 or isotype control ± αIL-17 (100 µg/mouse) administered intraperitoneally starting on day 2, then twice weekly until day 19 (αPD-L2+isotype n = 5 mice, αPD-L2 + αIL-17 n = 4 mice). e Tumour growth curve of IL-17KO aged mice harboring NCH1 melanomas treated with αPD-L2 or isotype control as above (isotype n = 5 mice, αPD-L2 n = 5 mice). Standard error of mean indicated; p-values by (b) log-rank test for Kaplan-Meier survival curve, or (a, c–e) two-way ANOVA.
αPD-L2 efficacy against B16 melanoma in aged hosts is also IL-17-dependent
We20 and others21 have described age-related increased IL-17. As IL-17 is reportedly often detrimental to anti-tumour immunity22, including in B16 melanoma in young mice23,24, we considered that age-related increased IL-17 could impede αPD-L2 efficacy. We challenged aged IL-17KO mice with subcutaneous B16 and treated with αPD-L2, but unexpectedly found that absence of host IL-17 completely abrogated αPD-L2-mediated tumour growth control (Fig. 2c), suggesting an IL-17-dependent efficacy mechanism. Cytokine knock-out mice can develop compensatory mechanisms over time. We thus independently confirmed IL-17-driven αPD-L2 efficacy by challenging aged WT mice with B16 and treating with αPD-L2 plus IL-17 neutralizing antibody, which abrogated αPD-L2 efficacy in B16 challenge (Fig. 2d). To confirm that age-dependent, IL-17-dependent αPD-L2 efficacy was not peculiar to B16, we challenged aged IL-17KO mice with NCH1 to demonstrate that αPD-L2 efficacy was also entirely abrogated (Fig. 2e). Altogether, these data demonstrate an IL-17-dependent mechanism for αPD-L2 efficacy in distinct melanoma models in aged hosts.
Although B16 reportedly grows faster in young IFNγKO versus WT24, B16 tumour growth was similar in aged IFNγKO versus aged WT (Supplementary Fig. 2c). B16 tumour growth is reportedly reduced in young IL-17KO mice versus young WT24. Similarly, B16 tumour growth appeared slowed in our aged IL-17KO versus aged WT (e.g., compare Figs. 1c to 2c) suggesting that basal IL-17, but not IFNγ contributes to endogenous B16 immune control.
IFNγ is a predominant feature of the aged melanoma tumour microenvironment
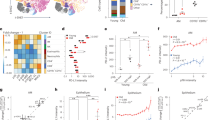
To understand IFNγ and IL-17 contributions to the tumour microenvironment (TME), we performed uniform manifold approximation and projection (UMAP) analysis of high dimensional flow data of B16 tumours and found that aged B16-bearing mice had a distinct immune TME versus young in isotype-treated controls as expected. Tumour-infiltrating immune cells clustered more prominently by age than by treatment (Fig. 3a), indicating strong age effects on the TME. UMAP analysis of B16 tumour-bearing mice demonstrated more total immune cell IFNγ expression and per-cell production in aged versus young (Fig. 3a), confirmed by parameter gating which also confirmed higher basal total CD8+ and CD4+ T cell IFNγ+ prevalence and per-cell expression in aged versus young (Fig. 3b–g).
a Flow cytometry UMAP color-continuous overlays of overall tumour-infiltrating CD45+ immune cell density, IFNγ+ CD45+ tumour-infiltrating immune cells, and IL-17+ CD45+ tumour-infiltrating immune cells in wild type aged (isotype n = 4 mice, αPD-L2 n = 4 mice), wild type young (isotype n = 5 mice, αPD-L2 n = 4 mice), and IL-17KO aged mice (isotype n = 5 mice, αPD-L2 n = 4 mice) challenged subcutaneously with B16 murine melanoma and treated with αPD-L2 (200 µg/mouse) or isotype control (200 µg/mouse) intraperitoneally every 4 days starting on day 3. b Prevalence of IFNγ+ CD45+, c IFNγ+ CD8+ and d IFNγ+ CD4+ tumour-infiltrating immune cells in isotype or αPD-L2 treated young or aged mice as indicated. e Mean fluorescence intensity (MFI) of IFNγ in CD45+, f CD8+ and g CD4+ tumour-infiltrating immune cells in isotype or αPD-L2 treated young or aged mice as indicated. h Prevalence of IL-17+ CD45+ tumour-infiltrating immune cells in isotype or αPD-L2 treated young or aged mice as indicated. i MFI of IL-17 in CD45+ tumour-infiltrating immune cells in isotype or αPD-L2 treated young or aged mice as indicated. Standard error of mean indicated; p-values by unpaired Student’s t-test.
UMAP analyses also defined increased total immune cell IL-17 in aged versus young B16-bearing, isotype-treated WT mice (Fig. 3a), confirmed with parameter gating (Fig. 3h,i), consistent with our prior report20. Aged TME immune cells in IL-17KO mice clustered similarly to young WT but not aged WT (Fig. 3a) suggesting that IL-17 drives the aged TME similar to reported IL-17-driven immune landscape shifts in aged skin21.
αPD-L2 promotes IL-17-dependent IFNγ+ tumour-infiltrating cell prevalence and production in aged B16 melanoma
Despite reports that IFNγ antagonizes T cell IL-17 production in young hosts25, aged IL-17KO mice surprisingly exhibited substantially lower TME immune cell IFNγ expression, similar to young WT and distinctly lower than aged WT by UMAP (Fig. 3a). To understand how IL-17 drives αPD-L2 efficacy, we investigated immune outcomes of αPD-L2 treatment in WT versus IL-17KO mice. TME IFNγ content was significantly higher in aged WT mice versus aged IL-17KO, similar to young WT (Fig. 3a), confirmed with parameter gating showing increased IFNγ+ total CD45+ immune cell, CD4+ T cell prevalence, and IFNγ+CD8+ T cell prevalence (Fig. 3b–d). Although αPD-L2 did not significantly enhance total IFNγ+ immune cell infiltration in aged WT mice, it specifically significantly enhanced IFNγ+CD8+ T cell prevalence while having no effect in aged IL-17KO or young WT, supporting that host IL-17 increases IFNγ+ cell content for αPD-L2 efficacy specifically in aged hosts in this model (Fig. 3b–d). Per-cell IFNγ production was significantly higher in total CD45+ immune cells in aged WT but not young WT treated with isotype, which depended on host IL-17 (Fig. 3e) and varied in specific cell subsets such as CD8+ and CD4+ T cells (Fig. 3f,g). Generally, αPD-L2 enhanced host IL-17-dependent per-cell IFNγ production in total CD45+ immune cells, CD8+, and CD4+ T cells in aged, but not young WT, but did not reach statistical significance (Fig. 3e–g).
To distinguish IFNγ production from IFNγ+ cell trafficking, we found that cells expressing IFNγ-associated CXCR3 were significantly reduced in aged IL-17KO versus WT, including CD8+CXCR3+IFNγ+ and CD4+CXCR3+IFNγ+ T cells, although αPD-L2 did not improve this effect (Fig. 4a,b). We used Ki67 expression to assess proliferating cells. Tumour infiltration of proliferating Ki67+CD8+CXCR3+IFNγ+ and Ki67+CD4+CXCR3+IFNγ+ T cells was comparable in young and aged WT and aged IL-17KO and unaffected by αPD-L2 in aged WT. Strikingly, αPD-L2 suppressed accumulation of proliferating Ki67+CD8+CXCR3+IFNγ+ T cells in aged IL-17KO and young WT mice with minimal effect on proliferating Ki67+CD4+CXCR3+IFNγ+ T cells in aged hosts (Fig. 4c, d). Prevalence of T cells expressing the CCR2 chemokine receptor associated with IL-17-driven chemotaxis26 was unaffected by IL-17KO (Fig. 4e, f). These data accord with IL-17-induced IFNγ that promotes conventional IFNγ+ T cell trafficking through CXCR3, but not that αPD-L2 improves this effect. As CCR2 is not specific for IL-17-mediated trafficking, IL-17 directly mediating immune cell trafficking through other factors is not excluded. These data together suggest that IFNγ+ T cell proliferation or trafficking are not major effector mechanisms for αPDL2-mediated efficacy in aged melanoma.
a–g Flow cytometry data for wild type aged (isotype n = 4 mice, αPD-L2 n = 4 mice), wild type young (isotype n = 5 mice, αPD-L2 n = 4 mice), and IL-17KO aged mice (isotype n = 5 mice, αPD-L2 n = 4 mice) challenged subcutaneously with B16 melanoma and treated with αPD-L2 (200 µg/mouse) or isotype control (200 µg/mouse) intraperitoneally every 4 days starting on day 3. a Prevalence of tumour-infiltrating CXCR3+IFNγ+CD8+ T cells (CD45+CD3+B220-), b CXCR3+IFNγ+CD4+ T cells (CD45+CD3+B220-), c Ki67+CXCR3+IFNγ+CD8+ T cells (CD45+CD3+B220-) d Ki67+CXCR3+IFNγ+CD4+ T cells (CD45+CD3+B220-) e CCR2+CD8+ T cells (CD45+CD3+B220-) f CCR2+CD4+ T cells (CD45+CD3+B220-). g SPICE-generated graphical pie chart output with overalying phenotype arcs for tumour-infiltrating CD8+ T cells. Boolean-gated prevalences within OMIQ were formatted within Pestle and exported into SPICE. Standard error of mean indicated; p-values by (a–f) unpaired Student’s t-test (g) SPICE permutation test.
αPD-L2 efficacy in aged melanoma includes promoting CD8+ T cell functions
CD8+ T cell polyfunctionality improves ICB outcomes27,28,29,30,31. We found that αPD-L2 significantly increased polyfunctional CD8+ T cell prevalence in aged WT B16-bearing mice but not aged IL-17KO or young WT mice (Fig. 4g). For functional confirmation of CD8+ T cell contributions, we depleted CD8+ T cells in aged B16-bearing mice to find that αPD-L2 efficacy was lost as expected (Supplementary Fig. 3a–c). Altogether, these data support a CD8+ T cell-mediated αPD-L2 efficacy mechanism largely through enhancing T cell anti-tumour effector polyfunctionality.
αPD-L2 efficacy in aged melanoma also depends on γδ T cells
γδ T cell prevalence was higher in isotype-treated aged WT versus young WT and aged IL-17KO mice, and αPD-L2 further increased total and IFNγ+ tumour-infiltrating γδ T cell prevalence in aged WT, but not in young WT or aged IL-17KO mice (Fig. 5a,b) or per-cell IFNγ (Fig. 5c), suggesting a potential role for γδ T cells in αPD-L2 efficacy. IFNγ+ CXCR3+ and proliferating Ki67+ γδ T cells were significantly enriched in aged WT versus aged IL-17KO and young WT mice but were not affected by αPD-L2 treatment (Fig. 5d,e) suggesting that γδ T cell contributions to αPD-L2 efficacy are not likely due to inducing CXCR3-mediated γδ T cell trafficking, proliferation or prevalence. Further, intratumoural γδ T cell polyfunctionality was not significantly improved by αPD-L2 in aged WT, aged IL-17KO or young WT mice (Supplementary Fig. 3d). While the role of IFNγ+CD8+ T cells in tumour immune surveillance and efficacy of ICB agents is well-established19, we32,33 and others34 have shown that γδ T cells are also important mediators of anti-tumour immunity, including through IFNγ. We tested a functional role for γδ T cells directly by challenging aged δTCRKO mice (lacking all γδ T cells but retaining conventional CD4+ and CD8+ T cells) with B16 and treated with αPD-L2, which abrogated treatment efficacy (Fig. 5f, Supplementary Fig. 3e). Prevalence of proliferating Ki67+CD8+ T cells was significantly higher in aged WT versus aged IL-17KO and young WT mice but αPD-L2 did not enhance this effect (Supplementary Fig. 3f) suggesting a role for IL-17 support of γδ T cell homeostasis, consistent with their role in αPD-L2 efficacy here. As both CD8+ and γδ T cells were essential for αPD-L2 efficacy in aged B16-bearing mice, we considered that one population could support function or content of the other. Although γδ T cell depletion abolished αPD-L2 efficacy, it did not affect tumour-infiltrating CD8+ T cell prevalence (Fig. 5f,g), but abrogated aged host αPD-L2-mediated IFNγ+CD8+ T cell infiltration (compare Figs. 5h to 3c). CD8+ T cell production of IFNγ, and TNFα was decreased by γδ T cell depletion but was unaffected by αPD-L2 treatment in aged WT or aged γδTCRKO mice (compare Figs. 5i to 3f and Supplementary Fig. 4a). Strikingly, CD8+ T cell production of granzyme B was significantly increased by αPD-L2 in aged WT but significantly decreased in aged γδTCRKO mice, suggesting a γδ T cell-specific role for enhancement of αPD-L2-driven CD8+ T cell-mediated anti-tumour immunity (Fig. 5j). These findings suggest γδ T cells help mediate αPD-L2 efficacy against subcutaneous B16 melanoma in the aged host by promoting CD8+ T cell effector functions but not accumulation.
a–e Flow cytometry data for wild type aged (isotype n = 4 mice, αPD-L2 n = 4 mice), wild type young (isotype n = 5 mice, αPD-L2 n = 4 mice), and IL-17KO aged mice (isotype n = 5 mice, αPD-L2 n = 4 mice) challenged subcutaneously with B16 melanoma and treated with αPD-L2 (200 µg/mouse) or isotype control (200 µg/mouse) intraperitoneally every 4 days starting on day 3. f Tumour growth curve (isotype n = 8 mice, αPD-L2 n = 8 mice) and g–i flow cytometry data of tumour-infiltrating g CD8+ T cells (CD45+CD3+B220-) h IFNγ+CD8+ T cells and i mean fluorescence intensity (MFI) of IFNγ in CD8+ T cells from δTCRKO aged mice (isotype n = 4 mice; αPD-L2 n = 4 mice) as indicated harboring B16 melanomas treated with αPD-L2 or isotype control as above. j MFI of Granzyme B in tumour-infiltrating CD8+ T cells from wild type aged (isotype n = 4 mice, αPD-L2 n = 4 mice), wild type young (isotype n = 5 mice, αPD-L2 n = 4 mice), IL-17KO aged mice (isotype n = 5 mice, αPD-L2 n = 4 mice), and δTCRKO aged mice (isotype n = 4 mice, αPD-L2 n = 4 mice) as indicated harboring B16 melanomas treated with αPD-L2 or isotype control as above. k Tumour growth curve and l–n flow cytometry data of tumour-infiltrating l CD8+ T cells (CD45+CD3+B220-), m IFNγ+CD8+ T cells (CD45+CD3+B220-), and n MFI of IFNγ in CD8+ T cells (CD45+CD3+B220-) from wild type young mice harboring subcutaneous MB49 murine bladder cancer tumours treated with αPD-L2 or isotype control as above ± α-δTCR (500 μg/mouse loading dose followed by 200 μg/mouse for remaining doses) or respective isotype control administered every 3 days beginning on day 2 (α-δTCR n = 5 mice, α-δTCR + αPD-L2 n = 5 mice). Standard error of mean indicated; p-values by (a–e, g–j, l–n) unpaired Student’s t-test (f, k) two-way ANOVA.
To assess γδ T cell contributions in young mice, we did not use B16 as αPD-L2 was ineffective in them. We instead challenged young mice with subcutaneous MB49 and treated with α-δTCR to deplete γδ T cells ± αPD-L2, but αPD-L2 was still efficacious (Fig. 5k, Supplementary Fig. 4b) despite confirmed γδ T cell depletion (Supplementary Fig. 4c). In the setting of γδ T cell depletion, αPD-L2 was effective despite not significantly altering TME CD8+ T cell prevalence or per-cell IFNγ production (Fig. 5k–n). Altogether, these data support distinct αPD-L2 efficacy mechanisms in MB49 bladder cancer versus melanoma and further suggest that γδ T cell effects differ by age as expected and possibly by tumour.
Human γδ T cells experience immune changes with age
To understand age-related γδ T cell differences, we mined publicly available human scRNA-seq data (GSE120575, GSE72056, GSE115978) and stratified γδ T cells by age and by distinct Tγδ1 and Tγδ17 subsets producing primarily IFNγ or IL-17, respectively32 (Fig. 6a, b). Tγδ17 prevalence trended higher in aged versus young (Fig. 6c), but expression of IFNγ and IL-17 in Tγδ1 and Tγδ17 populations, respectively, did not differ notably based on host age (Fig. 6d). γδ T cell PD-L2 expression in Tγδ1 was significantly lower in aged and Tγδ17 PD-L2 expression trended to be lower in aged (Fig. 6e, f), supporting that γδ T cells might not be initial αPD-L2 targets or principal IL-17-producing cells for treatment efficacy. Antigen presenting pathways were increased in aged γδ T cells (Fig. 6g), which could be a mechanism by which they boost CD8+ T cell functions, as γδ T cells as antigen presenting cells has been reported35 including cross presenting tumour specific antigens36.
a–g Data from three pooled human melanoma scRNA-seq datasets (GSE120575 n = 32 human donors, GSE72056 n = 19 human donors, GSE115978 n = 16 human donors). a UMAP projection of γδ T cells stratified into Tγδ1 and Tγδ17 subsets using IFNG and IL17A gene expression, respectively. b Differential gene expression of markers validating the classification of γδ T cells, Tγδ1/Tγδ17 subsets and expression of PDCD1LG2 (PD-L2) in each subset. c Overall prevalence of γδ T cell subsets stratified by age group (young <67 years, old >67 years). d IL17A and IFNG gene expression in Tγδ1/Tγδ17 subsets stratified by patient age. e PDCD1LG2 gene expression in all γδ T cells and Tγδ1/Tγδ17 subsets stratified by patient age. f Statistical analysis of PDCD1LG2 gene expression in all γδ T cells and Tγδ1/Tγδ17 subsets with a median age cutoff of 67 years (young <67 years, old >67 years). g Gene Ontology (GO) analysis of pathways enriched in tumour-infiltrating γδ T cells old versus young patients. p-values by (f) unpaired Student’s t-test (g) GO analysis.
Host IL-17 and IFNγ regulate TME γδ T cell content and functions
Tumour-infiltrating total and CCR2+ γδ T cell prevalence was lower in aged IL-17KO whereas CXCR3+ γδ T cells were significantly increased versus aged WT, although αPD-L2 had no effect on these distribuitions (Supplementary Fig. 4d-f). Total γδ T cell prevalence was specifically enhanced in aged WT mice by αPD-L2 but not in any other group (Supplementary Fig. 4d). Tumour-infiltrating total, CXCR3+, and CCR2+ γδ T cell prevalence was lower in aged IFNγKO versus aged WT mice with αPD-L2 enhancing CXCR3+ and CCR2+ γδ T cell prevalence in aged IFNγKO mice (Supplementary Fig. 4d–f). Altogether, these data suggest that IFNγ and IL-17 regulate age-related TME γδ T cell functions that affect ICB efficacy but consequences for anti-cancer immune surveillance are less clear.
CD4+ T cell IL-17 elicits αPD-L2 efficacy in aged melanoma-bearing mice
As γδ T cells appeared unlikely to be mechanistic IL-17 sources, we assessed other IL-17 sources in the aged TME promoting αPD-L2 efficacy. Supernatants of cultured B16 and MB49 cells had negligible IL-17 (Supplementary Fig. 4g). As CD4+ T cells, not epithelial cells, produce significant IL-17, we assessed the EL4 CD4+ T cell lymphoma line to find strikingly high IL-17 production (Supplementary Fig. 4g). Nonetheless, it did not respond to αPD-L2 following subcutaneous challenge into young mice (Supplementary Fig. 4h). These data do not support differential tumour IL-17 production as a basis for αPD-L2 efficacy, although the possibility is not excluded. Further, recombinant IL-17, induced significant proliferation in B16 and NCH1 melanoma cells (Supplementary Fig. 4i), suggesting that direct effects of αPD-L2-elicited IL-17 on tumour cells do not account for anti-melanoma efficacy.
We examined IL-17 content in conventional and γδ T cells, natural killer cells, and myeloid cells in aged and young WT mice, including distinct subpopulations by flow cytometry. Although B cells and CD11b+ myeloid cells were the most prevalent cells in the aged B16 TME, along with many other IL-17-producing cells, αPDL2 did not clearly increase IL17 in any population (Supplementary Fig. 5a-h). We thus assessed CD4+ T cell IL-17 in αPD-L2 efficacy based on their known contributions to other IL-17-mediated processes26.
We transferred age-matched WT CD4+ T cells into aged IL-17KO hosts, challenged B16 subcutaneously the next day, and treated with αPD-L2. WT CD4+ T cell transfer, whose IL-17 production we validated by flow, elicited αPD-L2 efficacy. When the tumour curves had significantly separated on day 18 (p = 0.024), we administered αIL-17AR, which abrogated treatment efficacy (Fig. 7a, Supplementary Fig. 6a-b). Flow cytometry confirmed a significant increase in CD4+ T cell IL-17 following cell transfer in aged IL-17KO mice (Supplementary Fig. 6c,d). CD8+ T cell IFNγ trended to be increased without reaching statistical significance, possibly owing to IL-17 neutralization (Supplementary Fig. 6e,f).
a Overall tumour growth curve of B16 melanoma tumours from IL-17KO aged mice harboring subcutaneous B16 melanoma tumours given 1×106 naive wild type aged CD4+ T cells day -1 before tumour challenge treated with isotype (200 µg/mouse) or αPD-L2 (200 µg/mouse) intraperitoneally every 4 days starting on day 3 ± α-IL-17R (25 μg/mouse intraperitoneally on day 18 post-tumour challenge) (isotype n = 3 mice, αPD-L2 n = 4 mice). b Tumour growth curves of wild type young mice challenged subcutaneously with B16 melanoma and treated with αPD-L2 (200 µg/mouse), isotype control (200 µg/mouse) intraperitoneally every 4 days starting on day 3 ± recombinant IL-17A (rIL17 1 µg/mouse) daily from day -10 to day +11 pre- and post-tumour challenge. (isotype n = 4 mice, αPD-L2 n = 5 mice, rIL-17 n = 4 mice, rIL-17 + αPD-L2 n = 4 mice). Flow cytometry data of intratumoural prevalence of c CXCR3+CD8+ T cells (CD45+CD3+B220-), d CXCR3+CD4+ T cells (CD45+CD3+B220-), and e CXCR3+γδ TCR+ γδ T cells (CD45+CD3+B220-) from tumours in b. f NY-ESO-1 TCR expressing HLA-A2+ human T cells cultured with HLA-A2+ U251-nGFP-NY-ESO-1+ human glioblastoma cells treated with isotype control (50 μg/mL) or αPD-L2 (50 μg/mL) with GFP expression quantified (n = 3 human donors). g Tumour growth curve of wild type aged mice challenged subcutaneously with PD-L2KO B16 murine melanoma and treated with αPD-L2 or isotype control as above (isotype n = 7 mice; αPD-L2 n = 6 mice).Standard error of mean indicated. p-values (a, b, g) two-way ANOVA (c–f) unpaired Student’s t-test.
Exogenous IL-17 sensitizes young hosts with B16 to αPD-L2 immunotherapy
Exogenous IL-17 improves αPD-1 plus αCTLA-4 combination (but not individual agent) efficacy in Braf-mutated mouse melanoma models37. We found that exogenous IL-17 elicited αPD-L2 efficacy against B16 in young mice, although neither agent alone was efficacious (Fig. 7b, Supplementary Fig. 7a). Flow cytometry of tumour-infiltrating lymphocytes found that efficacy correlated with immune cell trafficking through CXCR3 specifically in CD4+ and γδ T cells but not CD8+ T cells (Fig. 7c–e). The Tbet transcription factor drives antigen-specific CD8+ T cell IFNγ production38. Tbet+CD8+ T cell prevalence significantly increased in mice given IL-17 plus αPD-L2 while prevalence of IFNγ+CD8+ T cells and their per-cell production of IFNγ remained unchanged (Supplementary Fig. 7b,c). Further, prevalence of IFNγ+CD45+ and IFNγ+CXCR3+CD45+ immune cells, CD4+ T cells, and γδ T cells and their per-cell IFNγ production was also unchanged by αPD-L2 irrespective of concomitant exogenous IL-17 (Supplementary Fig. 7d-f). Together, these findings suggest an IL-17-driven efficacy program in B16 melanomas in young WT mice that can elicit αPD-L2 efficacy, which could be mediated by IFNγ at some point during treatment, not detected at endpoint here by flow cytometry.
α-human PD-L2 elicits IL-17 and tumour cytotoxicity in vitro
In allogeneic mixed lymphocyte reactions of human dendritic cells and allogeneic peripheral blood mononuclear cells, αPD-L2 clone 24 F.10C12 elicited IL-17, whereas αPD-L2 clone MIH18 and αPD-1 (nivolumab) did not. Clone 24 F.10C12 also elicited simultaneous IFNγ, which was significantly greater than that elicited by αPD-1 (Supplementary Fig. 7g). No antibody elicited detectable IL-4, -10, -18, -21, -23 or -27.
Cytotoxicity of NY-ESO-1 TCR expressing HLA-A2+ human CD8+ T cells incubated with HLA-A2+ NY-ESO-1+ human tumour cells was significantly enhanced by αPD-L2 (Fig. 7f), demonstrating that αPD-L2 could mediate this function in vivo for efficacy, requiring further studies.
Host IL-17 drives immune cell PD-L2, PD-1 and RGMB but not PD-L1 expression in aged
IFNγ increases expression of immune and tumour cell PD-L1 associated with ICB efficacy and increases PD-L239,40 among other immune checkpoints, any of which could alter ICB efficacy. To address functional consequences of tumour PD-L2 expression, we genetically deleted PD-L2 from B16 melanoma (Supplementary Fig. 8a,b), challenged subcutaneously in aged mice and found that αPD-L2 remained effective (Fig. 7g, Supplementary Fig. 8c).
In studies of the B16 TME, young WT and aged IL-17KO mice had decreased per-cell PD-L2 expression in tumour-infiltrating total CD45+ immune cells, tumour, stromal, CD8+ T cells and B220+ cells (Supplementary Fig. 8d–h) versus aged WT, in line with our prior report8. αPD-L2 treatment significantly decreased per-cell PD-L2 expression in young WT and aged IL-17KO total CD45+ cells whereas specific subpopulations differed (Supplementary Fig. 8d-h). Prevalence of total PD-L2+CD45+ immune cells did not differ in aged WT mice versus young WT and aged IL-17KO mice and αPD-L2 significantly decreased total PD-L2+CD45+ immune cell prevalence only in aged IL-17KO and young WT mice while simultaneously increasing PD-L2+CD8+ T cell prevalence only in aged WT mice (Supplementary Fig. 8d–h).
PD-1 and RGMB are the two well-known PD-L2 receptors41,42. RGMB+ CD45+ immune cell and CD45- non-immune stromal cell prevalence was similar between aged WT, young WT, and aged IL-17KO mice while prevalence of RGMB+ tumour cells was significantly higher in young WT mice versus aged WT mice (Supplementary Fig. 9a–c). Per-cell RGMB expression trended higher in CD45+ and tumour cells in aged IL-17KO and young WT versus aged WT mice (Supplementary Fig. 9a–e). αPD-L2 treatment increased prevalence of RGMB+ tumour-infiltrating CD8+ T cells and B220+ cells and their per-cell RGMB in aged WT versus young WT and aged IL-17KO (Supplementary Fig. 9d,e). Isotype-treated aged WT mice exhibited per-cell PD-1 expression in tumour-infiltrating CD45+ cells similar to young WT and aged IL-17KO although the prevalence of PD-1+CD45+ cells was significantly higher in aged WT versus young WT as we reported8,20, and also versus aged IL-17KO (Supplementary Fig. 9f). αPD-L2 significantly increased PD-1+ CD8+ T cell prevalence and their per-cell PD-1 in aged WT mice in a host IL-17-dependent manner (Supplementary Fig. 9g).
PD-L2 protein expression increases in mouse and human cells with age
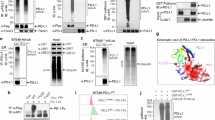
We mined a publicly available single cell RNA sequencing (scRNAseq) dataset examining host age effects on the immune B16 TME43 to determine whether specific immune populations differentially express PDCD1LG2 (PD-L2) in a host age-dependent manner. We projected the scRNAseq dataset onto a UMAP and clustered immune cells by expression of their characteristic gene signatures as described in the original dataset43 (Fig. 8a–c). Intratumoural NK cells, neutrophils, plasmacytoid dendritic cells, and conventional type I dendritic cells (cDC1) expressed higher PDCD1LG2 in aged versus young mice (Fig. 8c) suggesting a relationship between age and PD-L2 expression on specific murine immune cells.
a–c Data from mouse B16 scRNA-seq dataset accessed from the Broad Institute scRNA database repository SCP1261. a UMAP overlay displaying cell type annotation in murine B16 melanoma dataset. b Stratification of UMAP plot by host age. c PDCD1LG2 (PD-L2) gene expression stratified by host age and cell type. d–h Data from three pooled human melanoma scRNA-seq datasets (GSE120575 n = 32 human donors, GSE72056 n = 19 human donors, GSE115978 n = 16 human donors). d UMAP overlay displaying cell type annotation. e Subclustering of myeloid cell types including monocytes (Mono), conventional type 1 dendritic cells (cDC1), conventional type 2 dendritic cells (cDC2), plasmacytoid dendritic cells (pDC), migratory dendritic cells (migDCs), PLA2G7 high interstitial macrophages (PLA2G7.IMs), RNASE1 high interstitial macrophages (RNASE1.IMs), and cycling myeloid cells (Cycling). f Gene expression of markers defining clusters identified in e. g PDCD1LG2 gene expression stratified by cell types and host age (median host age 67 years). h Statistical analysis of PDCD1LG2 gene expression in myeloid subclusters identified in e stratified by host age > or <73. i Linear correlation plots of PD-L2+ immune cell subsets versus age in cancer patients versus healthy donors (tumour infiltrating [n = 10 human donors]; peripheral blood [healthy donor n = 3 human donors]; [cancer patient n = 10 human donors]). Standard error of mean indicated; p-values (i) linear regression (h) unpaired Student’s t-test (*, p < 0.05, ****, p < 0.0001).
To test human translational relevance of our findings, we examined PDCD1LG2 (PDL2) gene expression by age by pooling three human melanoma bulk RNAseq datasets (TCGA SKCM, GSE65185, GSE91061) to find that overall PDCD1LG2 expression did not significantly correlate with age, as it did in mice. To increase the resolution of our analysis, we further analyzed the three pooled human melanoma scRNAseq datasets used to generate Fig. 6 to identify immune cell-specific changes in PDCD1LG2 expression stratified by host age. We projected the datasets onto a UMAP and annotated individual cell subsets (Fig. 8d). To characterize myeloid subsets further, as we suspected dendritic cell contributions to host age-dependent αPD-L2 efficacy, we used our previously published custom myeloid marker panel44 to examine specific cell subset PDCD1LG2 expression stratified by host age (Fig. 8d–h). We found that myeloid subsets trended to exhibit higher PDCD1LG2 expression versus other immune subsets and identified PLA2G7.IMs, a subset of interstital tumour associated macrophages that are immunosuppresive in cancer45, and cDC1, as in mice (Fig. 8c), specifically expressed more PDCD1LG2 in aged versus young hosts (Fig. 8g,h). Intratumoural NK cell, and plasmacytoid dendritic cell PDCD1LG2 expression did not differ by age in human melanomas, highlighting species specificity of intratumoural PDCD1LG2 expression (Fig. 8i). We mined publicly available data from The Cancer Genome Atlas Pan-Cancer database and found that aged cancer patients, defined as >60 years old, expressed higher PDCD1LG2 (PD-L2), IL-17A (IL-17), and IFNG (IFNγ) than younger cancer patients ( < 60 years of age) (Supplementary Fig. 10a). To support our findings further, we studied peripheral blood mononuclear cells from normal subjects and cancer patients. Notably, PDCD1LG2 expression increased on lineage-DR+CD141+ cDC1, but not on cDC2 dendritic cells, plasmacytoid dendritic cells, other myeloid cells, T cells or B cells (Fig. 8i), suggesting a relationship between age and PD-L2 expression on specific human immune cells, including specifically cDC1, corroborating our scRNAseq analyses of human melanoma patients and aged mice. There was insufficient data available to make clear conclusions regarding age effects on PDL2 protein expression.
Discussion
ICB has significantly improved treatment efficacy in distinct tumour types, although most tumours fail to respond, prompting searches for additional targets. ICB antibodies blocking the PD-1 or PD-L1 immune checkpoints are FDA-approved and among the most successful cancer ICB agents. Strikingly, PD-L2, the other PD-1 ligand, has received little attention as cancer immunotherapy. Early reports that PD-L2 augmented T helper and cytotoxic functions46 suggested that its blockade might reduce anti-tumour immunity, supported by our earlier report that B7-DC+ (PD-L2+) B cells in aged mice promoted anti-tumour immunity47. However, reports emerged that tumour PD-L2 also negatively influenced anti-tumour immunity48,49, prompting additional studies. αPD-L2 did not control subcutaneous MC38 colorectal carcinoma challenge in young mice, but significantly enhanced αPD-L1 efficacy, correlated with PD-L2 expression on tumour-inflitrating macrophages50. Cytotoxic chemotherapy elicits senescent immune cell and tumour PD-L2 expression that improves αPD-L2 efficacy in combination with chemotherapy in young mice14. RGMB is the other PD-L2 receptor. αPD-L2 effectively treats cancers through PD-L2/RGMB interactions and microbiome effects13, although age effects were not studied.
Despite numerous reports of immunotherapies that are effective in young but fail in aged3, we found that αPD-L2 controlled distinct, subcutaneous melanomas in aged but not young mice, the first example of single-agent cancer immunotherapy efficacy improved with advancing age to our knowledge. As age in the biggest overall cancer risk3, agents working preferentially in aged hosts could be highly useful. However, αPD-L2 failed to treat ID8agg ovarian cancer in peritoneum of aged mice, excluding age as the sole factor driving αPD-L2 efficacy. Further, αPD-L2 effectively controlled subcutaneous MB49 bladder cancer in young and aged WT hosts, although efficacy was superior in aged hosts, excluding aged skin effects on αPD-L2 efficacy and demonstrating that αPD-L2 efficacy is not melanoma-specific, further supported by recent reports13,14. Nonetheless, ICB efficacy in human cancers with αPD1 or αPDL1 does positively correlate with advancing age in some studies, and in mouse models, particularly in melanomas5,6, which could reflect a peculiarity of melanomas, or that they are most studied with ICB.
Although many cancers express PD-L2 that correlates with αPD-1 efficacy51, we showed that melanoma PD-L2 is dispensable for αPD-L2 efficacy. Immune cell PD-L2 thus appears to be a likely mechanistic candidate in αPD-L2 efficacy. Increased prevalence of PD-L2-expressing immune cells with age could help explain αPD-L2 efficacy in aged melanoma, but does not account for its efficacy in young MB49, where PD-L2 expression was low. RGMB expression in aged melanoma is also unlikely as the sole explanation owing to its higher expression in young, even though αPDL2 increased its expression in aged. The TY25 αPD-L2 treatment antibody clone that we used blocks both PD1 and RGMB interactions. Assessing PD1 versus RGMB contributions to efficacy in distinct contexts would be useful to understand these effects better. As RGMB blockade overcame host microbiome effects, which are known to affect ICB efficacy52 and change with age20, this avenue merits much additional investihgation.
IFNγ is critical to anti-tumour defenses53 and also specifically promotes efficacy of non-PD-L2-based ICB cancer immunotherapies15,16,17,18,19. It was thus unsurprising that αPD-L2 efficacy was IFNγ-dependent. Unexpectedly, though, αPD-L2 efficacy was also IL-17-dependent, which is unusual given the reportedly generally detrimental effects of IL-17 in cancers and specifically in B16 melanoma in young mice24. Mechanistically, IL-17 elicited IFNγ from CD8+ and γδ T cells, both of which contributed to anti-tumour immunity. As we showed that IL-17 promotes proliferation of both melanoma lines used in vitro, it is unlikely that IL-17 directly acts on tumour cells for tumour control.
Young IL-17-deficient versus WT mice reportedly controlled subcutaneous MC38 tumour growth poorly, with decreased IFNγ+CD8+ and CD4+ tumour-specific T cells in tumours and tumour-draining lymph nodes54, but a mechanistic link to tumour control or relationship between IL-17 and IFNγ was not reported. Adoptively transferred Th17-polarized CD4+ T cells protect from subcutaneous B16 melanoma in young mice in vivo, but effects were IFNγ- independent55 and thus distinct from endogenous IL-17 effects we now report. Mice challenged with IL-17-transduced hematopoietic tumour lines experienced better tumour control that was T cell-dependent and genetrated cytotoxic lymphocytes better than mice challenged with control tumours. In this study, no relationship to IFNγ was shown, and exogenous IL-17 in vitro did not increase lymphocyte expression of Perforin or Fas ligand, and IFNγ effects were not reported56. Thus mechanistic details are obscure and appear distinct from our study. We found that EL4, which spontaneously produced IL-17 in the same range as these reported transduced cells, was refractory to αPD-L2, and thus IL17 is not the sole determining factor to elicit αPDL2 efficacy in distinct settings.
Remarkably, we found that IL-17 drove both immune cell IFNγ production and accumulation of IFNγ-expressing, tumour-infiltrating cells. αPDL2-mediated increased intratumoural IFNγ+ cell accumulation appeared not to be related to cell trafficking or local proliferation. Nonetheless, IL-17 increased TME CXCR3+IFNγ+ immune cells, including CD8+ T cells known to mediate anti-tumour immunity in IL-17-replete mice, consistent with a recent report37, but αPD-L2 did not increase their prevalence. Tumour-infiltrating immune cell expression of CCR2, an IL-17-associated chemokine, was unaffected by IL-17KO. Thus, IL-17-driven IFNγ is a likely mechanism for accumulation of TME IFNγ+ anti-tumour effector cells, but αPD-L2 efficacy in aged subcutaneous melanoma challenge does not appear to depend on increasing their numbers. Efficacy mechanisms could include a cell co-localization effect or altered IL-17 sensitivity.
Increased IL-17 in aged hosts is reported20,21, but IL-17 contributions to ICB efficacy in the aged or young are little reported. In subcutaneous colon cancer challenges in young mice, IL-17 neutralization improved αPD-1 efficacy, attributed to reduced tumour PD-L1 and myeloid derived suppressor cells, and increased intratumoural CD3+ T cells (without report of IFNγ effects)57, in sharp contrast to IL-17-mediated improvement in αPD-L2 efficacy in aged melanoma we report. A recent report showed that a Th17 gene signature predicted efficacy of dual PD-1/CTLA-4 blockade (but not individual ICB) in patients with BRAFV600E-mutated, but not BRAF WT, melanomas37. In this study, recombinant IL-17 administration to mice improved tumour control from increased intratumoural IFNγ+ T cell trafficking through CXCR3, but effects on per-cell IFNγ production were not described. These data support our findings of IL-17-induced IFNγ and T cell trafficking, but differ in age effects, ICB agents studied, and that our tumours were all BRAF WT. Further, we noted a global IL-17, not just Th17 effect, as the age-related increase in IL-17 we observed was in many immune cell populations, including CD8+ T cells, B, natural killer, and γδ T cells, whereas Th17 relates specifically to CD4+ T cells26. Nonetheless, the preponderance of data point to a tendency for melanomas or selected other cutaneous tumours to experience beneficial IL-17 effects from ICB that are exploitable and bear further investigation.
Further, exogenous IL-17 administration to young mice rendered their B16 melanomas αPDL2-responsive. Thus, lack of αPDL2 response in young mice appears to relate to their defect in eliciting αPDL2-driven IL-17, rather than some defect in IL-17 effects on their immune cells. This outcome also differs from the report of exogenous IL-17 not improving single agent αPD1 or αCTLA4 in Braf-mutated melanomas, but only improving their combination37, supporting a distinct IL-17-dependent or -driven mechanism for distinct ICB agents which could also include age effects.
αPD-L2 efficacy against heterotopic MB49 bladder cancer in young hosts was not IL-17-dependent, and IL-17 depletion significantly slowed tumour growth, consistent with reports of orthotopic MB49 challenge in young hosts24. αPD-L2 did not elicit significant IL-17 or IFNγ in MB49 tumours suggesting tumour-specific effects requiring additional investigation. In aged melanoma we identified CD4+ T cells as providing mechanistic IL-17 for αPDL2 efficacy, but as many cells produced IL-17, other cells could also contribute. We previously reported a PD-L2+B220+ age-related B cell that elicited CD8+ T cell IL-17 and IFNγ for anti-tumour efficacy47 that merits investigation in this regard. Notably, αPD-L2 increased tumour-infiltrating γδ T cell anti-tumour cytokines and these γδ T cells supported tumour-infiltrating CD8+ T cell effector functions, as also seen under other conditions in young hosts58. Further, some γδ T cell subsets can increase with age in humans59. Our data support that γδ T cells contribute to αPD-L2 efficacy in these aged hosts. Γδ T cell participation in approved ICB efficacy is reported, notably in MHC class I low tumours60. By contrast, αPD-L2 effectively controlled subcutaneous MB49 in young mice independent of IL-17 and γδ T cells. We mined single cell RNA-seq data sets to establish distinctions in young versus aged γδ T cells specifically in the melanoma TME, but most outcomes did not clearly support a mechanistic contribution to αPD-L2 efficacy. The interesting finding that aged γδ T cells exhibited better antigen-presenting properties versus young γδ T cells bears additional study. Understanding these tumour- and organ-dependent αPD-L2 and IL-17 efficacy mechanisms should shed light on factors contributing to ICB outcomes, including in young hosts. Other potential contributions to tumour-related efficacy include immune checkpoint expression, production or induction of immunosuppressive factors, differential responses to cytokines, and tumour mutational burden.
Factors contributing to the aged tumour immune environment remain poorly understood. Our data identify IL-17 contributions to many age-related immune changes, consistent with a recent report in skin21, some of which are surprisingly beneficial, including inducing IFNγ and beneficial IL-17, and reducing Treg numbers or potential functions, e.g., through fragility. Contributions of IL-17 and IFNγ to age-related immune checkpoint expression and relationships to ICB efficacy warrant additional studies.
Our study has limitations. Although we showed that IL-17 induces IFNγ production, the mechanism remains undefined. We showed that tumour PD-L2 can be dispensable for αPD-L2 efficacy and implicated PD-L2+ cDC1 as a candidate immune cell αPD-L2 target, but the mechanistic target of αPD-L2 was undefined. Other important questions requiring further study include identifying the drivers of age-related IL-17 and IFNγ, and effects of Treg fragility.
Whether increasing age degrades ICB efficacy is controversial, with evidence from human studies that it can be beneficial in selected cases (recently reviewed3) which also depends on the cancer studied5,6. Nonetheless, studies of age effects on tumour immune surveillance are important as they help understand how anti-tumour immune responses change with age to improve efficacy of agents in elderly populations, help comprehend treatment resistance mechanisms. Further, if we can understand how age improves ICB efficacy, insights could be used to improve outcomes in younger patients.
Methods
All studies performed comply with IRB and IACUC regulations at UT Health San Antonio and Dartmouth. Human whole blood was obtained from healthy, consenting, and anonymized adult volunteers through Research Blood Components, LLC (Boston, MA) and peripheral blood mononuclear cells (PBMC) isolated by density centrifugation using Ficoll-Paque Plus (Fisher-Scientific). All blood collection followed American Association of Blood Banks guidelines and was approved by the New England Independent Review Board (NEIRB, 120160613).
Mice
Male and female wild type (WT), IFNγKO and δTCRKO C57BL/6 J (BL6) mice were purchased from Jackson Laboratory. PD-L1KO mice were a gift of Leiping Chen when at Mayo Clinic. IL-17KO mice were a gift from Seema Ahuja, UT Health San Antonio. The Tyrosinase:CreER inducible TN61R mouse melanoma model12,61 was a gift of Christin Burd, Ohio State. Other mice were purchased at ages 6-8 weeks or bred in-house, maintained under specific pathogen-free conditions and given food and water ad libitum, and aged at the South Texas Research Facility (San Antonio, TX) or at Dartmouth Cancer Center. Age- and sex-matched young (2.5-6 months) or aged (19-33 months) mice were used for all experiments.
In vivo tumour challenges, treatments and assessments
The NCH1 melanoma line was developed from a tumour developing in a TNQ61R, UVB-irradiated mouse derived from a well-characterized inducible melanoma model62. PD-L2KO B16 cells were generated by infection of parental B16 with a lentivirus containing the Cas9 nuclease and CRISPR gRNA targeting the PDCD1LG2 gene with the following gRNA sequence: 5’-CACACTGCTGCCGACGTCTA-3’ (GenScript). PD-L2 knockout was validated by Western blot and flow cytometry (Supplementary Fig. 8a,b). Tumour IL-17 secretion was assessed by ELISA per the manufacturer’s instructions (Invitrogen). 0.5 × 106 B16, PD-L2KO B16 or control PD-L2KO, 0.3 × 106 NCH1 or 0.5 × 106 PD-L1KO B16 cells in 150 μL sterile PBS were injected into flanks subcutaneously with a 27-gauge needle. Orthotopic ID8agg challenge was 4 × 106 cells intraperitoneally into females63. Heterotopic MB49 bladder cancer cell challenge was 0.5 × 106cells subcutaneously. Heterotopic EL4 murine lymphoma challenge was 3.5 × 105 cells subcutaneously. Cell lines were routinely tested monthly for mycoplasma using the Lonza MycoAlert Plus kit and confirmed mycoplasma-free but were not re-authenticated for this study. Treatments were αPD-1 (BioXCell, RMP1-14) 200 μg/mouse on days 7, 11, 15 or αPD-L2 (BioXCell, TY25) 200 μg/mouse on days 3, 7 and 11, or respective isotype controls. αIL-17 (BioXCell, 17F3) 100 μg/mouse was administered on day 2, and twice weekly until day 1964,65. αCD8 (BioXCell, 2.43) 100 μg/mouse or respective isotype control was administered every 3 days beginning on day 2. α-δTCR (Biolegend, UC7-13D5) 500 μg/mouse first dose followed by 200 μg/mouse for remaining doses or respective isotype control administered every 3 days beginning on day 2. Mice were sacrificed when tumours reached a maximum volume of 1500 mm3, which was not exceeded, or distress for survival studies or 500–1000 mm3 for immune studies as approved by UT Health San Antonio and Dartmouth College IACUCs9,66.
Recombinant IL-17A (1 μg/mouse; R&D Systems catalog # 421-ML-100/CF) was given to young WT C57/BL6 mice day -10 through day +11 with B16 challenge at day 0 (500,000 cells per flank subcutaneously). αPD-L2 was administered as above and tumours were monitored and harvested for flow cytometry as above.
Aged IL-17KO mice received 1 × 106 age-matched WT CD4+ T cells for adoptive transfer which were administered retro-orbitally on day -1 prior to B16 tumour challenge in a volume of 100 μL PBS. αPD-L2 was administered as above and tumours were monitored and harvested for flow cytometry as above. αIL-17R (R&D Systems catalog # MAB4481) was administered on day 18 (25 μg/mouse i.p.).
In vivo tumour growth monitoring
Subcutaneous tumour growth was measured using Vernier calipers and volume was calculated as (length x width2)/2. Bioluminescent imaging (IVIS Lumina Imaging System, Perkin Elmer) was used for in vivo tumour monitoring of ID8agg intraperitoneally challenges63. Briefly, mice were imaged 15 minutes after intraperitoneal injection of 3 mg PBS-dissolved d-luciferin K+ (Gold Biotechnology) with one-minute exposure, small binning, and F/stop = 1. Identical regions of interest were drawn over each mouse and average radiance (photons/second/cm2/region) was quantified with Living Image software version 4.2.
Western blot
CTRL and PDL2KO B16 cells were grown in vitro and lysed using TN1 lysis buffer containing 1x HALT protease and phosphatase inhibitor. Protein in each sample was quantified by BCA assay. 40 μg of protein was loaded for each sample on a 10 well TGX pre-cast polyacrylamide gel (Bio-Rad) and run at 90 V for approximately 90 min. Protein was transferred to nitrocellulose membranes using a Bio-Rad TransBlot Turbo set at 1-amp constant current for 30 min. Membranes were blocked for at least 1 h in 5% milk dissolved in 1x TBST. Membranes were incubated with primary antibody as indicated in 5 % milk/TBST rocking overnight at 4 °C. Membranes were washed 3x with 1x TBST and then incubated with appropriate HRP-conjugated secondary antibody for 1 h. Membranes were washed 3x with 1x TBST and then imaged using a ChemiDoc MP imager (Bio-Rad) and chemiluminescent ECL (Bio-Rad).
Flow cytometry
Mice were sacrificed by cervical dislocation after deep isoflurane anesthesia. Dissected tumours were placed in 70 μm strainers and filtered into single cell suspensions in serum-free RPMI-160 medium. Cells were counted with a Vi-cell XR (Beckman Coulter) and 5 × 106 cells/sample were transferred to 96-well plates. Dead cells were excluded by LIVE/DEAD Stain Kit (Thermo Fisher Scientific) or Ghost Dye Violet 510 (Tonbo Biosciences). Non-specific labeling was pre-blocked with anti-CD16/32 at 1:100 dilution (clone 93, Biolegend). Single cell suspensions in 1x PBS supplemented with 2% FBS were stained with monoclonal antibodies: αCD8 (BUV395, 53-6.7), αCD4 (BUV496, GK1.5), αB220 (BUV661, RA3-6B2), αCD11c (BUV805, HL3), αPD-L1 (BV421, MIH5), αNK1.1 (BV570, PK136), αTIM-3 (SB436, 8B.2C12), αPD-L2 (BV605, TY25), αRORγT (BV650, Q31-378), αCCR2 (BV711, 475301), αCD25 (BB515, PC61), αCD45 (AF532, 30-F11), αCD11b (PERCPCY5.5, MI/70), αPD-1 (PE-DAZZLE, RMP1-30), αTCRγ (PECY5, GL3), αFOXP3 (PECY7, FJK-16s), αGP100 (AF647), αCD3 (AF700, 17A2), αSA (APC-CY7), αRGMB (BIOTIN), αIFNγ (BUV737, XMG1.2), αTIM-3 (BV421, B8.2C12), αGZMB (Pacific Blue, QA16A02), αKi67 (BV510, B56), αIL-2 (BV605, JES6-5H4), αCXCR3 (BV650, CXCR3-173), αTNFα (BV750, MP6-XT22), αRORγT (APC, AFKJS-9), αIL-17 (AF700, TC11-18H10.1), αCD3 (APC/FIRE810, 17A2). When therapeutic TY25 clone αPD-L2 was used, PD-L2 was detected by cytometry using αPD-L2 (BV480, MIH37).
For intracellular staining, cells were fixed and permeabilized with FoxP3/transcription factor buffer (Tonbo) per manufacturer instructions and incubated at 4 °C for 45 min. For cytokine detection, cells were stimulated with Cell Activation Cocktail (Biolegend) containing phorbol 12-myristate 13-acetate, ionomycin, and brefeldin at 2 µL cocktail/mL CR10 medium (RPMI-1640 with 10% FBS, L-glutamine, sodium pyruvate, non-essential amino acids, penicillin/streptomycin, and HEPES buffer) for 6 h at 37 °C. After stimulation, intracellular staining was performed by incubating cells at 4 °C for 30-45 min with antibodies. Events were acquired using BD ARIA hardware or Cytek Aurora and analyzed by FlowJo (FlowJo LLC) or OMIQ software. For presentation of polyfunctionality, Boolean gates were used to determine the frequency of CD8+ T cells, NK cells or γδ T cells producing either 1, 2, 3, or 4 cytokines and data were analyzed using Pestle and SPICE67.
In vitro IL-17 proliferation assay
B16 or NCH1 melanoma cells were seeded at 3000 cells per well in 96-well flat bottom culture plates in 200 µl of culture medium (B16: 5% fetal bovine serum [FBS]-containing RPMI-1640 medium supplemented with 1% penicillin/streptomycin, L-glutamine, and 10 mM HEPES; NCH1: 10% FBS-containing DMEM medium supplemented with 1% penicillin/streptomycin, 4.5 g/L glucose, L-glutamine, and 10 mM HEPES) and cultured overnight. Recombinant IL-17a was purchased from R&D systems (421-ML) and used at the indicated concentrations. After the indicated incubation period, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium Bromide (MTT) reagent was added to each well to achieve a final concentration of 0.5 mg/mL and allowed to incubate for 3 h at 37 °C, 5% CO2 to allow the conversion of MTT to formazan crystals by viable cells. Formazan was solubilized by aspirating culture media and adding 100 µl of dimethyl sulfoxide (DMSO) to each well. Viability was assessed using a Biotek Synergy HT plate reader measuring absorbance at 540 nm.
In vitro human cell cultures
We generated monocyte derived dendritic cells from normal volunteer leukopaks as described68 and cultured them with allogeneic peripheral blood mononuclear cells (PBMC) from healthy donors ± αPD-1 (nivolumab) or each of two αPD-L2 clones (24 F.10C12 and MIH18, both from BioLegend) that bound distinct PD-L2 epitopes. Briefly, monocytes were enriched from PBMCs of healthy donors using the Pan Monocyte Isolation Kit (Miltenyi; Cat #130-096-537) according to manufacturer’s recommendations. The cells were then differentiated for 3 days at 37 oC, 5% CO2 with 0.2 μg/mL GM-CSF (PeproTech; Cat #300-03-250ug) and 0.2 μg/mL IL-4 (PeproTech; Cat #200-04-250ug), following by overnight maturation using a cytokine cocktail consisting of 10 ng/mL TNF-α (PeproTech; Cat #300-01a), 15 ng/mL IL-1β (PeproTech; Cat #200-01b), 150 ng/mL IL-6 (PeproTech; Cat #200-06-5ug) and 1 ug/mL PGE-2 (Sigma; Cat #45-P0409-5MG). Mature DCs were co-cultured with allogeneic PBMCs pre-labeled with 2 μM CellTraceTM Far Red (Invitrogen; Cat #C34564) at a DC:PBMC ratio of 1:10, in the presence of αPD-L2 antibody clones 24 F.10C12 and MIH18, αPD-1, or isotype control. Supernatant was collected at 72 hours, and an 18-plex Th1/Th2/Th9/Th17 Cytokine 18-Plex Human ProcartaPlex™ Panel (Thermo Fisher; Cat #EPX180-12165-901) was performed according to manufacturer’s recommendation. Data was acquired using Luminex® FLEXMAP 3D (Bio-Techne). We transduced human glioblastoma U231 cells with NY-ESO-1, nuclear GFP and HLA-A2. We induced NY-ESO-1 T cell receptor expression in HLA-A2+ human CD8+ T cells ( ~ 65% transduction efficiency) from PBMC and incubated with tumour cells at indicated ratios for 48 h. Cytotoxicity was estimated by reduction of GFP expression by digital imaging. Antibodies were used at concentrations shown.
TCGA data mining
Xena Functional Genomics Explorer69 was used to mine the TCGA Melanoma and Pan-Cancer datasets using TCGA datasets as parameter A, age at initial diagnosis as parameter B, and gene expression as parameter C in all inquiries. Samples with nulls were removed prior to analysis. Violin plots and statistics were generated within Xena.
Mouse scRNA-seq analysis
Data acquisition and preprocessing
The mouse scRNA-seq data used in this study were downloaded from the SCP1261 dataset (https://jitc.bmj.com/content/9/10/e002809), which includes samples from young and old mice with B16 melanoma tumour infiltrates. The data were preprocessed and normalized using the Seurat package in R v.4.2. Quality control measures included filtering cells with fewer than 200 genes or more than 10% mitochondrial gene content.
Differential expression analysis
Differentially expressed genes (DEGs) were identified using the FindAllMarkers function in Seurat. The data matrices from the SCT assay were used, with a minimal log fold change set to 0.25. Only genes detected in more than 25% of cells in either population were considered, and DEGs were computed using the Wilcoxon rank-sum test. Genes with a Bonferroni-adjusted P value < 0.05 were considered significant. DEGs were ranked by adjusted P value for downstream analysis.
Cell type identification
Cell clusters were identified using the FindClusters function with the Leiden algorithm at resolutions ranging from 0.5 to 2.0. The top DEGs of individual clusters were examined against known marker genes to annotate cell identities. Adjustments were made until each cluster displayed a unique gene expression pattern.
GO analysis
Gene Ontology (GO) analysis was performed using the TopGO package with Fisher’s exact test to identify enriched biological processes among DEGs. Background genes for enrichment assays included all detected genes in the datasets. The top five GO terms ranked by P value for each subset were selected, and overlapping terms were removed for visualization.
Data visualization
Data visualization was conducted using R and the ggplot2 package for bubble plots and bar graphs. Seurat was used to generate FeaturePlot, DimPlot, and VlnPlot visualizations.
Human bulk RNA-seq analysis
Data acquisition and preprocessing
Human bulk RNA-seq data were aggregated from three datasets (tcga_SKCM, GSE65185, and GSE91061) to enhance correlation and significance analysis, yielding a p-value of 0.055. Data preprocessing included normalization and batch effect correction to ensure comparability across datasets.
Differential expression analysis
DEGs were calculated using a standard linear model approach, adjusting for potential confounders such as age and tissue type. Genes with an adjusted P value < 0.05 were considered significant.
Pathway analysis
Pathway analysis was conducted using the Gene Set Enrichment Analysis (GSEA) tool to identify pathways significantly associated with age and PD-L2 expression. Enrichment scores and p-values were computed to rank pathways.
Data visualization
Results were visualized using ggplot2 for scatter plots and bar graphs, with significant pathways highlighted.
Human scRNA-seq analysis
Data acquisition and preprocessing
Human scRNA-seq data were combined from multiple datasets (GSE120575, GSE72056, and GSE115978) to ensure sufficient representation of dendritic cells (DCs) and interstitial macrophages (IMs). Quality control included filtering out cells with fewer than 200 genes or more than 10% mitochondrial gene content.
Cell type identification
Cell types were identified using the FindClusters function in Seurat with the Leiden algorithm at varying resolutions. Detailed subclustering of myeloid cell types was performed, including macrophages, monocytes, and DCs. Custom markers were used to define subtypes.
Differential expression analysis
DEGs were identified using the FindAllMarkers function in Seurat, with the same criteria as the mouse scRNA-seq analysis. Significant DEGs were determined based on a Bonferroni-adjusted P value < 0.05.
Pathway analysis
Pathway analysis was conducted using TopGO with Fisher’s exact test to identify enriched biological processes among DEGs. The top GO terms were selected and non-overlapping terms were visualized.
Data visualization
Visualization tools included ggplot2 for various plot types and Seurat for UMAP and violin plots. Expression levels of PDCD1LG2 across cell types were visualized with color representing expression level and size indicating the percentage of cells expressing PDCD1LG2.
Statistics and data analysis
An effect size of 0.85 to determine that the two-sample t-test achieves 80% power with 11 mice per B16 group to detect a survival difference between treatment and control with αPD-1 at 14 days with a 2-sided α = 0.05. Flow cytometry analysis including UMAP plots used OMIQ or FlowJo software. Statistical analyses were conducted with Prism software (GraphPad). Data in immune analyses are means ± SEM. For tumour growth, we used two-way ANOVA plus Bonferroni post-tests to compare replicate means. For all other analyses, one-way ANOVA or unpaired t-test was used as indicated. Statistically significant outliers were removed using the ROUT test for multiple outliers. P < 0.05 was considered significant.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The sequencing raw data and processed data used in this article are available in the Gene Expression Omnibus under accession codes: • Mouse scRNA-seq: Data were accessed from PMID: 34642245 according to instructions provided in the publication. Briefly, data were accessed from the Broad Institute scRNA database repository SCP1261 at the following persistent URL: https://singlecell.broadinstitute.org/single_cell/study/SCP1261. • Human bulk RNA-seq were aggregated from the following datasets: ○ TCGA_SKCM (https://portal.gdc.cancer.gov/projects/TCGA-SKCM) ○ GSE65185 ○ GSE91061 The combined human scRNA-seq datasets are: • GSE120575 • GSE72056 • GSE115978 TCGA human datasets analyzed using the Xena browser are: • TCGA Melanoma (SKCM) (https://xenabrowser.net/datapages/?dataset=TCGA.SKCM.sampleMap%2FHiSeqV2&host=https%3A%2F%2Ftcga.xenahubs.net&removeHub=https%3A%2F%2Fxena.treehouse.gi.ucsc.edu%3A443) • TCGA Pan-Cancer (PANCAN) (https://xenabrowser.net/datapages/?dataset=EB%2B%2BAdjustPANCAN_IlluminaHiSeq_RNASeqV2.geneExp.xena&host=https%3A%2F%2Fpancanatlas.xenahubs.net&removeHub=https%3A%2F%2Fxena.treehouse.gi.ucsc.edu%3A443) No new sequencing data were generated for this study. Source data are provided with this paper. Due to an existing Materials Transfer Agreement, we are unable to deposit the NCH1 cell line in a public repository. We will gladly provide the cell line to investigators upon reasonable request. Source data are provided with this paper.
References
Yang, Y. Cancer immunotherapy: harnessing the immune system to battle cancer. J. Clin. Investig. 125, 3335–3337 (2015).
Siegel, R. L., Miller, K. D. & Jemal, A. Cancer statistics, 2018. CA Cancer J. Clin. 68, 7–30 (2018).
Ontiveros, C. O., Murray, C. E., Crossland, G. & Curiel, T. J. Considerations and Approaches for Cancer Immunotherapy in the Aging Host. Cancer Immunol. Res. OF1-OF13 (2023). https://doi.org/10.1158/2326-6066.CIR-23-0121
Elias, R. et al. Efficacy of PD-1 & PD-L1 inhibitors in older adults: a meta-analysis. J. Immunother. cancer 6, 26 (2018).
Nie, R. C. et al. Efficacy of anti-PD-1/PD-L1 monotherapy or combinational therapy in patients aged 75 years or older: a study-level meta-analysis. Front. Oncol. 11, 538174 (2021).
Kugel, C. H. et al. Age correlates with response to anti-PD1, reflecting age-related differences in intratumoral effector and regulatory T-cell populations. Clin. Cancer Res. 24, 5347–5356 (2018).
Kaur, A. et al. sFRP2 in the aged microenvironment drives melanoma metastasis and therapy resistance. Nature 532, 250–254 (2016).
Garcia, M. G. et al. Immune checkpoint expression and relationships to anti-PD-L1 immune checkpoint blockade cancer immunotherapy efficacy in aged versus young mice. Aging Cancer 3, 68–83 (2022).
Padron, A. et al. Age effects of distinct immune checkpoint blockade treatments in a mouse melanoma model. Exp. Gerontol. 105, 146–154 (2018).
Latchman, Y. et al. PD-L2 is a second ligand for PD-1 and inhibits T cell activation. Nat. Immunol. 2, 261–268 (2001).
Schildberg, F. A., Klein, S. R., Freeman, G. J. & Sharpe, A. H. Coinhibitory pathways in the B7-CD28 ligand-receptor family. Immunity 44, 955–972 (2016).
Burd, C. E. et al. Mutation-specific RAS oncogenicity explains NRAS codon 61 selection in melanoma. Cancer Discov. 4, 1418–1429 (2014).
Park, J. S. et al. Targeting PD-L2-RGMb overcomes microbiome-related immunotherapy resistance. Nature 617, 377–385 (2023).
Chaib, S. et al. The efficacy of chemotherapy is limited by intratumoral senescent cells expressing PD-L2. Nat. Cancer https://doi.org/10.1038/s43018-023-00712-x (2024).
Higgs, B. W. et al. Interferon gamma messenger RNA signature in tumor biopsies predicts outcomes in patients with non-small cell lung carcinoma or urothelial cancer treated with durvalumab. Clin. Cancer Res. 24, 3857–3866 (2018).
Karachaliou, N. et al. Interferon gamma, an important marker of response to immune checkpoint blockade in non-small cell lung cancer and melanoma patients. Ther. Adv. Med Oncol. 10, 1758834017749748 (2018).
Garris, C. S. et al. Successful Anti-PD-1 Cancer Immunotherapy Requires T Cell-Dendritic Cell Crosstalk Involving the Cytokines IFN-gamma and IL-12. Immunity 49, 1148–1161.e1147 (2018).
Wang, W. et al. CD8(+) T cells regulate tumour ferroptosis during cancer immunotherapy. Nature 569, 270–274 (2019).
Topalian, S. L., Taube, J. M., Anders, R. A. & Pardoll, D. M. Mechanism-driven biomarkers to guide immune checkpoint blockade in cancer therapy. Nat. Rev. Cancer 16, 275–287 (2016).
Hurez, V. et al. Chronic mTOR inhibition in mice with rapamycin alters T, B, myeloid, and innate lymphoid cells and gut flora and prolongs life of immune-deficient mice. Aging Cell 14, 945–956 (2015).
Sola, P. et al. Targeting lymphoid-derived IL-17 signaling to delay skin aging. Nat. Aging 3, 688–704 (2023).
Qian, X. et al. Interleukin-17 acts as double-edged sword in anti-tumor immunity and tumorigenesis. Cytokine 89, 34–44 (2017).
Hayata, K. et al. Inhibition of IL-17A in tumor microenvironment augments cytotoxicity of tumor-infiltrating lymphocytes in tumor-bearing mice. PLoS One 8, e53131 (2013).
Wang, L. et al. IL-17 can promote tumor growth through an IL-6-Stat3 signaling pathway. J. Exp. Med. 206, 1457–1464 (2009).
Park, H. et al. A distinct lineage of CD4 T cells regulates tissue inflammation by producing interleukin 17. Nat. Immunol. 6, 1133–1141 (2005).
Kara, E. E. et al. CCR2 defines in vivo development and homing of IL-23-driven GM-CSF-producing Th17 cells. Nat. Commun. 6, 8644 (2015).
Donia, M. et al. PD-1(+) polyfunctional T cells dominate the periphery after tumor-infiltrating lymphocyte therapy for cancer. Clin. Cancer Res. 23, 5779–5788 (2017).
Donia, M. et al. Aberrant expression of MHC class II in melanoma attracts inflammatory tumor-specific CD4+ T- cells, which dampen CD8+ T-cell antitumor reactivity. Cancer Res. 75, 3747–3759 (2015).
Berinstein, N. L. et al. Survivin-targeted immunotherapy drives robust polyfunctional T cell generation and differentiation in advanced ovarian cancer patients. Oncoimmunology 4, e1026529 (2015).
Tan, M. P. et al. Human leucocyte antigen class I-redirected anti-tumour CD4(+) T cells require a higher T cell receptor binding affinity for optimal activity than CD8(+) T cells. Clin. Exp. Immunol. 187, 124–137 (2017).
Tan, M. P. et al. T cell receptor binding affinity governs the functional profile of cancer-specific CD8+ T cells. Clin. Exp. Immunol. 180, 255–270 (2015).
Reyes, R. M. et al. CD122-directed interleukin-2 treatment mechanisms in bladder cancer differ from alphaPD-L1 and include tissue-selective gammadelta T cell activation. Jo. Immunother. Cancer 9, https://doi.org/10.1136/jitc-2020-002051 (2021).
Payne, K. K. et al. BTN3A1 governs antitumor responses by coordinating alphabeta and gammadelta T cells. Science 369, 942–949 (2020).
Yazdanifar, M., Barbarito, G., Bertaina, A. & Airoldi, I. Gammadelta T cells: the ideal tool for cancer immunotherapy. Cells 9, https://doi.org/10.3390/cells9051305 (2020).
Brandes, M., Willimann, K. & Moser, B. Professional antigen-presentation function by human gammadelta T Cells. Science 309, 264–268 (2005).
Muto, M., Baghdadi, M., Maekawa, R., Wada, H. & Seino, K. Myeloid molecular characteristics of human gammadelta T cells support their acquisition of tumor antigen-presenting capacity. Cancer Immunol. Immunother. 64, 941–949 (2015).
Varaljai, R. et al. Interleukin 17 signaling supports clinical benefit of dual CTLA-4 and PD-1 checkpoint inhibition in melanoma. Nat. Cancer 4, 1292–1308 (2023).
Sullivan, B. M., Juedes, A., Szabo, S. J., von Herrath, M. & Glimcher, L. H. Antigen-driven effector CD8 T cell function regulated by T-bet. Proc. Natl. Acad. Sci. USA 100, 15818–15823 (2003).
Keir, M. E., Butte, M. J., Freeman, G. J. & Sharpe, A. H. PD-1 and its ligands in tolerance and immunity. Annu. Rev. Immunol. 26, 677–704 (2008).
Garcia-Diaz, A. et al. Interferon Receptor Signaling Pathways Regulating PD-L1 and PD-L2 Expression. Cell Rep. 29, 3766 (2019).
Xiao, Y. et al. RGMb is a novel binding partner for PD-L2 and its engagement with PD-L2 promotes respiratory tolerance. J. Exp. Med. 211, 943–959 (2014).
Wojtowicz, W. M. et al. A Human IgSF Cell-Surface Interactome Reveals a Complex Network of Protein-Protein Interactions. Cell 182, 1027–1043 e1017 (2020).
Zhang, C. et al. Single-cell sequencing reveals antitumor characteristics of intratumoral immune cells in old mice. J. Immunother. Cancer 9, https://doi.org/10.1136/jitc-2021-002809 (2021).
Li, X. et al. Coordinated chemokine expression defines macrophage subsets across tissues. Nat. Immunol. 25, 1110–1122 (2024).
Zhang, F. et al. Inhibiting PLA2G7 reverses the immunosuppressive function of intratumoral macrophages and augments immunotherapy response in hepatocellular carcinoma. J. Immunother. Cancer 12, https://doi.org/10.1136/jitc-2023-008094 (2024).
Shin, T. et al. In vivo costimulatory role of B7-DC in tuning T helper cell 1 and cytotoxic T lymphocyte responses. J. Exp. Med. 201, 1531–1541 (2005).
Tomihara, K. et al. Aging-associated B7-DC(+) B cells enhance anti-tumor immunity via Th1 and Th17 induction. Aging Cell 11, 128–138 (2012).
Nakayama, Y. et al. Immune suppression caused by PD-L2 expression on tumor cells in gastric cancer. Gastric Cancer 23, 961–973 (2020).
Tanegashima, T. et al. Immune Suppression by PD-L2 against Spontaneous and Treatment-Related Antitumor Immunity. Clin. Cancer Res. 25, 4808–4819 (2019).
Umezu, D. et al. Inhibitory functions of PD-L1 and PD-L2 in the regulation of anti-tumor immunity in murine tumor microenvironment. Cancer Immunol. Immunother. 68, 201–211 (2019).
Yearley, J. H. et al. PD-L2 expression in human tumors: relevance to anti-PD-1 therapy in cancer. Clin. Cancer Res. 23, 3158–3167 (2017).
Fessler, J., Matson, V. & Gajewski, T. F. Exploring the emerging role of the microbiome in cancer immunotherapy. J. Immunother. cancer 7, 108 (2019).
Shankaran, V. et al. IFNgamma and lymphocytes prevent primary tumour development and shape tumour immunogenicity. Nature 410, 1107–1111 (2001).
Kryczek, I., Wei, S., Szeliga, W., Vatan, L. & Zou, W. Endogenous IL-17 contributes to reduced tumor growth and metastasis. Blood 114, 357–359 (2009).
Martin-Orozco, N. et al. T helper 17 cells promote cytotoxic T cell activation in tumor immunity. Immunity 31, 787–798 (2009).
Benchetrit, F. et al. Interleukin-17 inhibits tumor cell growth by means of a T-cell-dependent mechanism. Blood 99, 2114–2121 (2002).
Liu, C. et al. Blocking IL-17A enhances tumor response to anti-PD-1 immunotherapy in microsatellite stable colorectal cancer. J. Immunother. Cancer 9, https://doi.org/10.1136/jitc-2020-001895 (2021).
Ji, N. et al. gammadelta T Cells Support Antigen-Specific alphabeta T cell-Mediated Antitumor Responses during BCG Treatment for Bladder Cancer. Cancer Immunol. Res. 9, 1491–1503 (2021).
Clark, B. L. & Thomas, P. G. A cell for the ages: human gammadelta T cells across the lifespan. Int. J. Mol. Sci. 21, https://doi.org/10.3390/ijms21238903 (2020).
de Vries, N. L. et al. gammadelta T cells are effectors of immunotherapy in cancers with HLA class I defects. Nature 613, 743–750 (2023).
Hennessey, R. C. et al. Ultraviolet radiation accelerates NRas-mutant melanomagenesis: a cooperative effect blocked by sunscreen. Pigment Cell Melanoma Res. 30, 477–487 (2017).
Burd, C. E. et al. Monitoring tumorigenesis and senescence in vivo with a p16(INK4a)-luciferase model. Cell 152, 340–351 (2013).
Drerup, J. M. et al. CD122-selective IL-2 complexes reduce immunosuppression, promote Treg fragility, and sensitize tumor response to PD-L1 blockade. Cancer Res. (2020).
Xiong, H. et al. Innate lymphocyte/Ly6C(hi) monocyte crosstalk promotes Klebsiella pneumoniae clearance. Cell 165, 679–689 (2016).
Coffelt, S. B. et al. IL-17-producing γδ T cells and neutrophils conspire to promote breast cancer metastasis. Nature 522, 345–348 (2015).
Hurez, V. et al. Mitigating age-related immune dysfunction heightens the efficacy of tumor immunotherapy in aged mice. Cancer Res. 72, 2089–2099 (2012).
Roederer, M., Nozzi, J. L. & Nason, M. C. SPICE: exploration and analysis of post-cytometric complex multivariate datasets. Cytom. A 79, 167–174 (2011).
Zou, W. et al. Reciprocal regulation of plasmacytoid dendritic cells and monocytes during viral infection. Eur. J. Immunol. 31, 3833–3839 (2001).
Goldman, M. J. et al. Visualizing and interpreting cancer genomics data via the Xena platform. Nat. Biotechnol. 38, 675–678 (2020).
Acknowledgements
Flow cytometry experiments were carried out using equipment in DartLab, the Immune Monitoring and Flow Cytometry Shared Resource at the Norris Cotton Cancer Center at Dartmouth, with NCI Cancer Center Support Grant 5P30 CA023108-41. The Flow Cytometry Shared Resource at UT Health San Antonio is supported by the NCI (CA054174), the Cancer Prevention and Research Institute of Texas (RP210126), NIH (S10OD030432), and the Office of the Vice President for Research at UT Health San Antonio. We thank the UT Health San Antonio Department of Laboratory Animal Resources and the Dartmouth Center for Comparative Medicine and Research for caring for animals used in this study. C.O.O. was supported by NCI F31 CA281345. C.E.M. was supported by the Graduate Research in Immunology Program training grant NIH T32 AI138944 and the National Center for Advancing Translational Sciences, National Institutes of Health, through Grant TL1 TR002647. C.O.O, M.G.G, C.E.M, and R.M.R were supported by the South Texas Medical Scientist Training Program at UT Health San Antonio (T32GM113896, T32GM145432). T.J.C. received funding from NCI (CA054174, CA268641, CA023108, CA292467), The Clayton Foundation, The Skinner Endowment, the Owens Foundation, Guyre funds, Lawton Family Funds, and the Gmelich Chair. C.V.J. received funding from NIH (HL115334, HL135001, HL155458). M.J.T. received funding from NCI (CA254042, CA22502), the Knights of the York Cross of Honour, USA, and the O. Ross McIntyre, M.D. Endowment. M.S. received startup funding from the Dartmouth Cancer Center. We thank Agenus (K.T.S, S.K, and D.C) for assistance with performing some experiments used in this study.
Author information
Authors and Affiliations
Contributions
C.O.O., M.G.G., C.E.M., and T.J.C. conceived of the ideas, designed experiments, interpreted results and wrote the manuscript. C.O.O. and M.G.G. performed the experiments with assistance from C.E.M., Y.D., H.B., C.T., B.L., A.P., R.M.R., A.K., A.B., C.H., and H.B.G. X.L. performed bioinformatic analysis. K.T.S. and S.K. performed some experiments designed by T.J.C., D.C., C.O.O., and M.G.G. C.V.J., M.J.T., M.S., and J.C.G. provided scientific input and helped revise the manuscript. T.J.C. secured funding sources, provided supervision, and performed overall project management and coordination.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks anonymous reviewer(s) for their contribution to the peer review of this work. [A peer review file is available].
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Ontiveros, C.O., Garcia, M.G., Murray, C.E. et al. Anti-PD-L2 immunotherapy is efficacious against melanoma in aged hosts through IL-17 and IFNγ signalling. Nat Commun 16, 10176 (2025). https://doi.org/10.1038/s41467-025-65025-2
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-65025-2