Abstract
Low-cost manganese-based oxides as promising cathodes for sodium-ion batteries still face significant challenges, including irreversible phase transition, air/water sensitivity, and low initial charge capacity. Herein, we precisely design a titanium-substituted Mn-based oxide cathode material with optimized local electronic structure distribution through a stepwise screening mechanism based on theoretical calculations, which enables suppression of irreversible phase transition and Jahn-Teller distortion by exerting spring effect and pinning effect. Notably, the optimized cathode fabricated using an aqueous binder exhibits stable electrochemical performance, retaining 96.16 % of its capacity after 500 cycles at 1 A g−1, along with reliable air/water stability under prolonged exposure, which is further confirmed by advanced characterization and theoretical calculations. In addition, Prussian blue analogs are proposed as a sodium supplement for full cell applications. The large-scale production and implementation of composite cathode materials have been successfully achieved and subsequently applied in practical pouch cells with non-presodiated hard carbon anodes. This work investigates an air/water-stable Mn-based layered oxide cathode for sodium-ion batteries, providing insights relevant to their future industrial development.
Similar content being viewed by others
Introduction
With increasing worldwide energy demand and growing concern about environmental preservation, research and development of sustainable energy storage technologies have become critically important1. In this context, sodium-ion batteries (SIBs) are a vital complement to lithium-ion batteries (LIBs) due to their abundant and easily accessible sodium resources, offering a viable pathway toward large-scale, cost-effective energy storage2,3. As the falling prices of lithium manganese oxide and lithium iron phosphate erode the cost advantage of nickel/cobalt-containing cathodes for SIBs, focusing on nickel/cobalt-free manganese (Mn)-based cathodes may be a promising route to cost-effective SIBs, owing to their high capacity, good conductivity, and environmental sustainability4,5. In general, layered oxides can be classified as O3-type and P2-type depending on the Na ions’ coordination environment and the stacking order of the oxygen layer. Among them, P2-type layered oxides with low sodium content face the challenges of irreversible phase transitions, poor air/water stability, and low initial charge capacity.
Firstly, during the sodium ion insertion and extraction, variations in stress in the internal lattice render Mn-based P2-type oxide cathodes susceptible to phase transitions such as P2-O2, P2-OP4, P2-Z, and P2-P2’ phase transitions5,6,7,8,9,10. The representative P2-type Na2/3MnO2 undergoes significant sodium ion deintercalation at a high state of charge (~4.1 V), causing transition metal layer slippage and inducing a P2-O2 phase transition with obvious volume changes, which adversely affects battery performance and cycle life11,12. Hence, researchers have attempted to use elements such as Li+, Mg2+, Cu2+, Zn2+, Al3+, Fe3+, and Sb5+ to suppress it13,14,15,16,17,18. Importantly, this phase transition typically evolves into a stacking fault OP4/Z phase rather than being completely suppressed. Qiao and Bruce et al. have shown that the OP4 phase consists of alternating P-O-P-O layers, while the Z phase represents an evolving intergrowth from P2 to O219,20. According to Yabuuchi et al. 21, the OP4 phase (space group: P6m2) possesses certain structural features of the P-type phase, including the mirror symmetry of the hexagonal crystal system. Compared to the O2 phases, this structural feature results in a smaller volume change during cycling and helps mitigate the degradation of electrochemical performance. Additionally, the P2-P2’ phase transition related to the Jahn-Teller distortion (J-T distortion) of Mn3+ often occurs at the end of the discharge process (~2.35 V), leading to severe capacity degradation22,23. Although researchers have proposed several strategies, including single-element doping, dual-element co-doping, multiple-ions synergistic effect, and surface chemical modification, to alleviate or suppress phase transitions and J-T distortion24,25, unfortunately, these strategies are not fully effective and are difficult to apply on a large scale due to the complexity of the procedure.
Secondly, in practical applications, the air stability of cathode materials affects their synthesis, electrode preparation, storage, transportation, battery performance, and commercialization potential26,27,28. When exposed to air, P2-type Mn-based oxide cathode materials undergo acid degradation mechanisms with water and carbon dioxide, leading to surface deterioration and bulk structural changes that adversely affect their electrochemical performance29. Besides, the particulate nature of P2-type Mn-based oxides makes it challenging to ensure uniform dispersion in the slurry. During slurry preparation with solvent-based polyvinylidene difluoride/N-methyl pyrrolidone (PVDF/NMP) binders, PVDF can be defluorinated with hydroxide ions (OH⁻) in highly alkaline environments, causing slurry gelation30,31. Meanwhile, the NMP solvent is water-sensitive and slightly toxic, posing environmental and health risks. Thus, designing Mn-based cathode materials with good air stability and compatibility with aqueous binders is essential32,33. Furthermore, the inherent defects of P2-type oxide cathode materials with low sodium content present a significant challenge for their application in full batteries. To solve these problems, researchers proposed several measures, including designing P2-type oxide cathodes with high sodium content, adopting the electrochemical precipitation method, and selecting self-sacrificial sodium supplements34,35,36. Although the electrochemical properties of the materials were improved, they still suffer from the complexity, high cost, and low utilization of the sodium compensation process. Consequently, developing sodium supplements that are operationally convenient, safe, low-cost, and highly utilized for sustainable P2 Mn-based oxide cathode SIBs remains a significant challenge.
In this work, we propose a stepwise screening mechanism to identify titanium (Ti) as the optimal element for modulating the electronic structure distribution of Mn-based oxide cathodes, employing Na0.6MnO2 as a representative model. Meanwhile, the Ti substitution content was precisely optimized through theoretical calculations combined with experimental measurements, and the designed Mn-based oxide cathode achieves a good balance between dynamic structure evolution, air stability, and full-cell applications37,38,39. In-situ X-ray diffraction (XRD) analysis and stress calculations reveal that the modulation of electronic structure distribution simplifies complex phase transitions and moderates the P2-OP4 transition, maintaining slight strain properties during cycling. Additionally, the specific electronic structure design changes the local electronic configuration around Mn due to the similar electronegativity and ionic radius, generating the spring effect to reduce unfavorable phase transition and constructing the pinning effect to suppress the J-T distortion of Mn3+, which is further confirmed by etched X-ray photoelectron spectroscopy (XPS), soft X-ray absorption spectroscopy (sXAS), and electron localization function (ELF) analyses. Significantly, the optimized Na0.6Mn0.9Ti0.1O2 (NMT-10) cathode exhibits enhanced electrochemical performance with aqueous-based binders compared to conventional solvent-based binders and maintains stable performance even after prolonged air exposure. First-principles calculations further confirm that surface-adsorbed and interlayer water molecules do not hinder Na⁺ migration in the Ti-substituted structure, consistent with the observed stability under ambient conditions. The optimized NMT-10 material with good air/water stability offers a promising cathode option for the practical application of SIBs. Moreover, ferromanganese-based Prussian blue analogs (PBAs), with inherent sodium-rich content, high capacity, and high operating voltage, are employed as a sodium source to construct PBAs/NMT-10 composite cathodes with unsodiated hard carbon anodes for full cells, and the feasibility of scaling this approach was demonstrated in pouch cells.
Results and discussion
Theoretical calculations and screening mechanisms
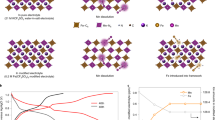
Manganese-based oxide cathode materials for sodium-ion batteries have been extensively studied due to their abundant resources, low cost, high theoretical capacity, and environmental friendliness. However, the practical application still faces challenges, including irreversible phase transitions, structural instability, and poor air stability, which necessitate modification strategies. Among these, element doping is widely considered a simple and effective method for modifying sodium transition metal oxide cathodes by tuning the band gap, adjusting layer spacing, enhancing structural stability, and tailoring ionic migration diffusion energy. However, screening suitable doping elements from all transition metals through traditional trial-and-error methods is time-consuming and labor-intensive, potentially requiring years to complete the necessary modifications. To address this problem, we propose a stepwise screening strategy to systematically investigate the effect of doping with 3 d and 4 d transition metal elements in the periodic table on structure stability (Fig. S1), considering factors such as cost, environmental friendliness, structural stress, ionic radius, and electronegativity (Fig. 1a and Table S1). Based on the density functional theory (DFT), a series of transition metals are doped in the crystal structure of Na0.6MnO2 with a supercell structure (4 × 4 × 1) and performed structural relaxation with a plane wave cutoff energy of 450 eV, the convergence criteria of system total energy as 10−6 eV and force on 0.02 eV Å−1. The corresponding optimized atomic structures are shown in Supplementary Data 1-17.
a Stepwise screening mechanisms. b Theoretical calculations of electron localization functions (ELF) for the pristine structure and doped structures with Ti, Cr, Fe, Cu, Zn. c Variation of TM-O bond lengths. d DFT calculations of sodium migration barriers for the Na0.6Mn1-xTixO2 (x = 0, 0.02, 0.05, 0.10, 0.15) cathodes. The solid lines represent the calculated energy profile along the full migration path, while data points indicate the corresponding peak values of the migration barriers for each composition. e–i Density of states (DOS) calculations of e NMO, f NMT-2, g NMT-5, h NMT-10, i NMT-15.
As an initial screening step, environmental impact assessment (Supplementary Table 2) was evaluated, and several candidate elements: V, Pd, Rh, Ru, Cd, and the radioactive Tc were excluded due to toxicity concerns. Elements such as Co, Ni, Mo, Nb, Y, Sc, and Ag were further eliminated owing to their high market costs. Consequently, Ti, Cr, Fe, Cu, Zn, and Zr were identified as potential dopants for further investigation. Second, theoretical calculations of stress variations (Fig. S2) indicate that Zr substitutions induce more significant stress changes among the investigated elements, whereas the incorporation of Ti, Cr, Fe, Cu, and Zn at low concentrations introduces minimal structural stress, suggesting minor disruption to the crystal lattice. Then, electron localization function (ELF) analysis (Figs. 1b, S3) demonstrated that Ti substitution causes significant electron localization around the dopant site, while Cr exhibits relatively weak electron localization, indicating that Ti forms stronger chemical bonds with neighboring oxygen atoms than Cr. In comparison, electrons surrounding Fe, Cu, and Zn are largely delocalized, implying metallic or ionic bonding characteristics. This contrast arises from the electronic configuration of Ti4+ (3d04s0), which provides empty orbitals for forming strong coordination covalent bonds with oxygen atoms, stabilizing the crystal structure40. It is worth noting that the close electronegativity and comparable ionic radii between Ti4+ (0.605 Å) and Mn3+ (0.645 Å) enable effective local adjustment of TM-O bond lengths. Figures 1c, S4 show that the Ti-O bond length is the shortest, corresponding to a large bond energy (~666.5 kJ/mol), which contributes to crystal structure stabilization. Therefore, after DFT calculation and stepwise screening element criteria, Ti is identified as the optimal dopant to regulate the local electronic structure of the Na0.6MnO2 cathode41,42,43,44.
To investigate the effects of varying Ti substitution contents (Na0.6TixMn1-xO2, x = 0.02, 0.05, 0.1, and 0.15, with corresponding DFT-optimized atomic structures provided in Supplementary Data 18-21 on the structure and properties of the materials, the climbing-image nudged elastic band (CI-NEB) method was employed to assess the Na+ migration energy barrier, as illustrated in Supplementary Videos 1-545,46,47. Compared to the pristine Na0.6MnO2 cathode with a high barrier of 0.666 eV, all the Ti substitution samples exhibited significantly reduced barriers, especially the NMT-10 sample, which was reduced to 0.227 eV (Figs. 1d, S5). The associated density of states (DOS) and projected density of states (PDOS) of Mn/Ti 3 d and O 2p were calculated to evaluate their contributions to the electronic structure and the effects on electrical conductivity and crystal stability, with schematic diagrams in Fig. 1e-i. Ti 3 d states were concentrated above the Fermi energy level (~2.0 eV), indicating that Ti remains electrochemically inactive in the cathodes, while the Mn 3 d states, predominantly located near the Fermi energy level, play a crucial role in charge compensation43. The broadened and flattened overlap between Mn 3 d and O 2p states near the Fermi level, particularly in the NMT-10 sample, indicates enhanced Mn-O hybridization and increased electronic delocalization, which contribute to stronger covalent bonding and improved electronic conductivity. These conclusions are primarily supported by experimental observations and further validated by advanced characterization and first-principles simulations.
Structural characterization and electrochemical performance of cathodes prepared with different binders
Morphology and structural characterization of the Na0.6Mn1-xTixO2 (x = 0, 0.02, 0.05, 0.10, 0.15, 0.20, 0.30, 0.45 denoted as NMO, NMT-2, NMT-5, NMT-10, NMT-15, NMT-20, NMT-30, NMT-45, respectively) cathode materials are displayed in Figs. S6-15 and Tables S3-9. The results show that the crystal structure can be transformed from layered to tunnel structure with increasing Ti content, which is demonstrated by structural characterization, refinement results, and thermodynamic theoretical calculations (Fig. S16). Tunnel-type oxides offer good structural stability but limited capacity, whereas layered structures provide high energy density but poor stability. The formation of a layered-tunnel composite enables the integration of cathode materials with enhanced capacity and improved structural integrity. In addition, the solvent-based PVDF/NMP binder prepared Na0.6Mn1-xTixO2 cathodes exhibited good electrochemical performance (Figs. S17-27 and Tables S10-13), and the in-situ XRD test of NMT-5 was performed to further comprehend the crystal structure evolution (Figs. S28-31). It undergoes the highly reversible P2/Tunnel-OP4/Tunnel-P2/Tunnel-P2 + P2’/Tunnel phase transition. Consequently, appropriate Ti substitution can mitigate the highly reversible P2-OP4 phase transition and stabilize the crystal structure of the cathode, thus exhibiting improved electrochemical performance. A comprehensive discussion and detailed analysis of all the above data are provided in the Supplementary Information.
However, in practical production applications, cathodes prepared with solvent-based PVDF/NMP binders are costly, toxic, and prone to structural degradation as well as slurry gelation in alkaline environments, making them inappropriate for large-scale industrialization. These limitations motivate the use of environmentally benign aqueous binder systems based on carboxymethyl cellulose (CMC) and deionized water. Here, the Na0.6Mn1-xTixO2 cathodes were fabricated using this aqueous binder (x = 0.02, 0.05, 0.10, 0.15 denoted as W-NMT2, W-NMT5, W-NMT10, W-NMT15, respectively); the related schematic is shown in Fig. S32. The electrochemical performance of the pristine NMO cathode is presented in Fig. S33 for comparison purposes. The first five cycles and different rate of galvanostatic charge/discharge (GCD) curves for all samples at different electrochemical windows are shown in Fig. 2a and Figs. S34-36. W-NMT10 shows enhanced reversibility, with capacities of 158.33 mAh g−1 (2.0-4.0 V) and 178.5 mAh g−1 (1.5-4.3 V) at 0.02 A g−1. The rate and cycle performance (Figures S37, 2b) suggest that the W-NMT2 and W-MNT10 electrodes exhibit capacity retention of 71.71 % (97.35 to 69.81 mAh g−1) and 96.72 % (94.61 to 91.51 mAh g−1) at 1 A g−1 after 600 cycles. Meanwhile, they maintain a favorable rate and cycle performance at 1.5-4.3 V, especially for W-NMT10 with a capacity retention of 69.08 % after 600 cycles at 0.4 A g−1 (Fig. S38). The GCD curves of the W-NMT10 cathode demonstrate a smooth profile, reflecting the suppression of multiple voltage plateaus, as indicated by the dQ/dV curves (Figs. S39-40). GITT at different voltage windows (Figs. S41a-d, S42a-d) reveals that the W-NMT10 cathode demonstrates lower polarization and enhanced kinetics during cycling. It is worth noting that cathodes fabricated with low-cost, environmentally friendly, and easily recyclable aqueous binder exhibit better electrochemical performance compared to their counterparts prepared with a conventional solvent-based binder, consistent with previous reports on Na0.44Mn1-xTixO2 cathodes48. The enhanced cycling stability can be attributed to the fact that the air/water stabilized tunnel structure produced by appropriate Ti substitution, as supported by structural refinement analyses and electrochemical testing before and after modification.
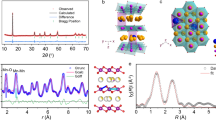
a The GCD curves of W-NMT2 and W-NMT10 measured at different voltage ranges. b Cycle performance at 1 A g−1 within 2.0-4.0 V. The capacity retention after 600 cycles was calculated with respect to the 46th cycle, following the rate capability test. c, d Evolution of lattice parameters a, c, volumes, and lattice strain calculation of W-NMT10 electrode at (c) 2.0-4.0 V and (d) 1.5-4.3 V. Comparison between FAULTS fitting and XRD experimental results of the W-NMT10 electrode at (e) 2.0-4.0 V and (f) 1.5-4.3 V.
In-situ XRD uncovers a reversible phase transition and tiny lattice strain for the NMT-10 cathode
In-situ XRD measurements were conducted to elucidate the crystal structure evolution of the NMT-10 electrodes prepared with an aqueous-based binder during the charge/discharge process. The contour maps are displayed in Fig. S43 (2.0-4.0 V) and Fig. S44 (1.5-4.3 V), the corresponding profiles and intensity contour plots are shown in Figs. S45-46, respectively. According to the first cycle of in-situ charge and discharge curves, the (002) and (004) peaks of the P2 phase gradually shift to a lower and then turn to higher angles during the Na+ extraction, the OP4 peak emerges around charging to 3.5 V when sodium ions are extracted to a critical concentration (~1/3 of the initial sodium content). Upon discharging, the (002)/(004) peaks return to their original positions. However, in the second cycle, the (002)/(004) peaks shift slightly, and the OP4 phase transition nearly disappears, attributed to the buffering effect of mechanically stabilized TiO6 octahedra, which will be discussed further. The overall P2-OP4 phase transition demonstrates high reversibility over the cycles, the phase transition at 2.0-4.0 V is more moderated than that in a wide range of 1.5-4.3 V. This is evidenced by the Rietveld refinement and strain calculations in Fig. 2c, d. During the second phase transition at 2.0-4.0 V, the cell parameters changed to 0.45 % for a/b, 0.14 % for c, and 0.39 % for cell volume, compared to 1.60 %, 0.33 %, and 0.96 %, respectively at 1.5-4.3 V. The microscopic stresses calculated based on the Gaussian function remain slightly changed, ranging from 0.011 to 0.015 at 2.0-4.0 V and 0.028 to 0.043 at 1.5-4.3 V. Furthermore, the variations in cell parameters are smaller during the second cycle compared to the first cycle, consistent with the moderate OP4 phase transition and slight strain process in the second cycle49.
To elucidate the origin of the moderated structural and stress variations, the structural evolution during O/P-type intercalation was further investigated by simulating diffraction patterns of different stacking configurations using the FAULTS program, a tool designed for structural refinement in the presence of extended defects50 (Fig. 2e-f and Supplementary Information Methods). By integrating the simulated patterns with experimental XRD data, the extent of sodium deintercalation during phase transitions was evaluated. A gradual shift of the (002) peak to higher angles reflects a transition from P2 to OP4 stacking. Meanwhile, quantitative analysis of stacking probabilities under high-voltage conditions reveals a transition trend from P-type to O-type stacking. The NMT-10 cathode maintains a dominant proportion of P-type stacking at various voltages, with 71 % at 2.0-4.0 V and 79 % at 1.5-4.3 V, corresponding to Na content variations of 0.39 → 0.25 → 0.52 and 0.32 → 0.19 → 0.34, respectively. Notably, no P2-P2’ phase change is observed across the whole electrochemical process under different voltage ranges. This further corroborates that a certain amount of Ti substitution effectively stabilizes the crystal structure and mitigates the P2-P2’ phase transition related to the J-T distortion of Mn3+ (t2g3eg1) at the end of discharge, which will be discussed in detail later.
Revealing the dynamical phase transition and its spring effect
To provide an intuitive and in-depth understanding of the more moderate phase transition in the second cycle compared to the first, in situ XRD patterns of NMT-5 (Figs. 3a-h) and NMT-10 (Figs. 3i-p) are projected in three-dimensional (3D), with the corresponding two-dimensional (2D) contour plots. At various voltage windows, the cathodes consistently exhibit a reversible P2-OP4 phase transition during charging, returning to the P2 phase upon discharge. In the second cycle, this transition becomes more moderate, corresponding with minor changes in cell parameters, volume, and stress, as previously discussed. The similarity in electronegativity and ionic radius of Ti4+ (0.605 Å) and Mn3+(0.645 Å) results in minimal impact on the crystal structure, and Ti enhances the distribution and transfer of the sodium ions, especially in the second cycle, allowing more sodium deintercalation while retaining some to support the structure. Moreover, the ionic radius of Ti4+ (0.605 Å), located between Mn3+ (0.645 Å) and Mn4+ (0.53 Å), enables the Ti4+O6 octahedra to stabilize the crystal structure and buffer the structural transition between Mn3+O6 and Mn4+O6, thus alleviating the lattice stresses and decreasing the degree of distortion to maintain the structural reversibility. Specifically, sodium ions are extracted from the layered structure during the charging process, accompanied by the oxidation of Mn3+ to Mn4+; the electrochemically inactive Ti acts as a stabilizer and buffer, significantly mitigating lattice stress variations and minimizing the slippage of the transition metal (TM) layer into the OP4 phase. In the subsequent discharge process, Mn4+ is reduced to Mn3+, and the Ti4+ substitution effectively suppresses J-T distortion, restoring the crystal to its original P2 phase. Overall, the TiO6 octahedra create a stable space for Na+ insertion/extraction, effectively buffering the distortion of the transition metal layer and minimizing overall deformation during cycling, which could be explained by the “spring effect” mechanism (Fig. 3q).
a–d NMT-5 within 2.0-4.0 V: (a, c) 3D and (b, d) 2D projections of (002) and (004) reflections. e–h NMT-5 within 1.5-4.3 V: (e, g) 3D and (f, h) 2D projections of (002) and (004) reflection. i-l NMT-10 within 2.0-4.0 V: (i, k) 3D and (j, l) 2D projections of (002) and (004) reflections. m–p NMT-10 within 1.5-4.3 V: (m, o) 3D and (n, p) 2D projections of (002) and (004) reflections. q Schematic of the spring effect mechanism to explain the moderation of the OP4 phase transition in the first and second cycles.
Insight into the suppression of J-T distortion
To further elucidate the suppression mechanism of the J-T distortion, we conducted a comprehensive analysis by investigating its underlying causes and combining advanced characterization techniques, including etched XPS and X-ray Absorption Near Edge Structure (XANES) during different charge/discharge states, and theoretical calculations. Firstly, as shown in Fig. 4a, Mn3+ exhibits an outermost electron orbital configuration of 3d4 (t2g3eg1), with an asymmetric distribution of electrons in orbitals of equal energy. Consequently, the spontaneous J-T distortion occurs to redistribute the electron cloud uniformly to minimize the system energy. When a single electron occupies the eg orbital, the system undergoes stretching or compression distortions, depending on the electron arrangement within the split energy level51,52. It is reported that J-T distortion in manganese-based oxide cathode materials typically exhibits a stretching distortion with an elongated axial Mn-O bond53,54,55. To mitigate this distortion, Ti substitution introduces a stronger Ti-O bond (~666.5 kJ/mol) compared to the Mn-O bond (~402 kJ/mol), effectively anchoring Ti within the TM layer and creating a local pinning effect (Fig. 4b), thus stabilizing the lattice and mitigating undesirable sliding56,57. Moreover, the similar ionic radius of Ti4+ and Mn3+ enables substitution with minimal lattice strain, effectively lowering the Mn3+ content and alleviating J-T distortion. Furthermore, the empty d-orbital of Ti4+ creates a highly stable electronic configuration with little deformation, while its substitution modifies the local electronic configuration and raises the average valence state of Mn ions, further suppressing the J-T distortion58,59. This is confirmed by the rightward shift in the Mn K-edge absorption edge for NMT-10, analyzed by the XANES in Fig. 4c. Meanwhile, XPS with different depths of etching was conducted (Figs. S47a-f, S48a-d), and Figs. 4d-e presents the XPS etching plots for Mn 2p and Ti 2p. The distribution of Mn3+ and Mn4+ determined by the peak splitting of Mn 2p1/2 and 2p3/2 (Fig. 4f) reveals a higher abundance of Mn4+ relative to Mn3+ at the surface and deeper bulk. Similarly, the corresponding contents of Ti 2p1/2 and 2p3/2 from splitting peaks (Fig. 4g) indicate a consistent and uniform distribution of Ti content throughout the material.
a, Schematic illustration of the J-T distortion of Mn3+. bSchematic diagram of Ti pinning effect in the crystal structure. c The Mn K-edge absorption edge for the NMT-10 electrode. d, e XPS depth profile spectrum of (d) Mn 2p and (e) Ti 2p. Sputtering was performed from 0 to 90 s with 10 s intervals, and the depth positions of each layer are indicated in the spectra. f Relative Mn3+/Mn4+ content obtained by Mn 2p peak splitting. g Relative Ti4+ content. h, i sXAS spectra of NMT-10 electrode at different voltage states: (h) Mn L-edge iPFY mode and (i) O K-edge PFY spectra mode. j-m Theoretical calculations of electron localization functions for (j) NMT-2, (k) NMT-5, (l) NMT-10, and (m) NMT-15 samples.
To gain a comprehensive understanding of the inhibition mechanism of J-T distortion in NMT-10, soft X-ray absorption spectroscopy (sXAS) spectra were recorded to analyze the evolution of the electronic structures of Mn and O. The bulk-sensitive inverse partial fluorescence yield (iPFY, probing depth of 100-200 nm) modes of Mn L-edge sXAS spectra (Fig. 4h) were employed to determine bulk electronic structures at various charged/discharged states. The spectra reveal two distinct regions at 636-644 eV (L1) and 648-656 eV (L2), caused by the splitting of the 2p core-hole spin orbitals. The two spectral features (A1 and A2) in the L1 region correspond to the Mn t2g and eg subbands, respectively. Upon charging to 4.3 V, the intensity of A2 decreases, likely due to the reduction of valence electrons in the eg orbitals as the valence state of Mn increases, with a corresponding rise in the characteristic peaks belonging to Mn4+. During discharge to 1.5 V, the Mn3+ content is slightly restored, but Mn4+ remains dominant, confirming that Ti substitution effectively suppresses the J-T distortion caused by Mn3+. Besides, the O K-edge spectra in the partial fluorescence yield (PFY) and total electron yield (TEY) modes (Figs. 4i, S49) at various states show pre-edge features at 529.2 eV and 532.3 eV, assigned to the hybridization of the Mn 3 d t2g and eg orbitals with O 2p, respectively. The peak at 533.7 eV is attributed to carbonate species, while the peaks between 535 eV and 550 eV arise from the hybridization of the O 2p with the Mn 4sp orbitals60,61,62. When charged to 4.3 V, the peak around 529.2 eV shifted slightly to the right as Mn3+ ions oxidized to Mn4+, indicating enhanced hybridization of the orbitals between Mn 3 d t2g and O 2p. The decrease of the peak near 532.5 eV during oxidation corresponds to the loss of an eg orbital electron from Mn3+, which reappears at the end of the discharge to 1.5 V, aligning with the sXAS results in iPEY mode for Mn. Consequently, appropriate Ti substitution rivets in the TM layer can stabilize the crystal structure and regulate the symmetric distribution of Mn electron orbitals, suppressing the J-T distortion.
Moreover, DFT calculations were performed to investigate the impact of titanium substitution on the lattice distortion, bond length variation, and the electronic distribution states of manganese and its surrounding oxygen atoms63,64. Figure S50a-e depict the schematic crystal structures of various Ti-substituted contents, while Figs. 4j-m present the corresponding ELF to characterize the degree of electron localization or delocalization and the modulation of the Ti-O and Mn-O bonds65,66. The results indicate that increasing Ti substitution enhances the localization of electron cloud distributions between Ti and O, particularly in the NMT-10 sample, where stronger Ti-O bonds are formed compared to Mn-O bonds. Simultaneously, according to the Jahn-Teller distortion index (DI) formula proposed by Yu et al.67 (Supplementary Note 3), it is found that the distortion of MnO6 octahedra decreases significantly after Ti substitution, from the initial 0.069 to 0.006 (Fig. S51). Consequently, Ti substitution extends battery cycle life by enhancing Ti-O bond localization, modifying the coordination environment of Mn, alleviating the lattice distortion, and stabilizing the overall crystal structure68,69.
Exploration of air stability and its self-cleaning function
Air stability is crucial for maintaining battery performance, safety, longevity, and efficient production70,71. The air stability of the NMT-10 cathode material was evaluated after various air exposure periods (1, 3, 5, 7, 15, and 30 days). Figure 5a illustrates a schematic of structure changes on the material’s surface and within the crystal lattice after air exposure, with surface smoothing and increased interlayer spacing, likely resulting from a self-cleaning function and water molecule intercalation72. The SEM (Figs. 5b-e, S52) confirms the smooth surface after exposure, particularly after 15 and 30 days. High-resolution TEM images and the corresponding statistical analysis with error bars (Figs. 5f-j, S53) reveal a progressive increase in lattice spacing of the NMT-10 cathode material with prolonged air exposure, which corresponds to a gradual elongation along the crystallographic c-axis, resembling reported birnessite72,73. Crucially, selected area electron diffraction (SAED) patterns acquired along the [001] zone axis maintain consistent hexagonal symmetry (P63/mmc space group) and diffraction spot intensities throughout the exposure period, indicating that the atomic arrangement in this direction has been preserved to some extent after exposure to air, further revealing the reason why the electrochemical performance of the material has not decreased significantly upon air exposure. Meanwhile, the XRD patterns (Fig. 5k) display pronounced hydration peaks near 12.7 ° and 25.5°, with a shift of the (002) and (004) peaks to lower angles, suggesting intercalation of water molecules. XPS was employed to examine changes in the surface composition of pristine and exposed materials. The fitted C 1 s peaks showed Na2CO3 at 288.2 eV and NaHCO3 at 289.4 eV (Fig. 5l)74,75. Relative to the constant C-C content, the level of Na2CO3 and NaHCO3 decreased significantly after exposure to air. This observation was corroborated by the O 1 s characteristic peak (Fig. 5m), with a reduced area ratio of absorbed OH-/bulk and CO32-/bulk from 1.48 and 2.23 to 0.67 and 0.59, respectively, indicating a decrease in surface residual alkaline content. Such a decrease may be attributed to the decomposition of surface adsorbates by strong oxidizing hydroxyl radicals, indicating a self-cleaning function76,77. These findings are further validated by thermogravimetric analysis (Fig. S54) and pH measurements (Fig. S55) in the supplementary information.
a Schematic diagram of the changes in the surface and crystal structure of the material after exposure to air. SEM images exposure to air for (b) 1 day, (c) 7 days, (d) 15 days, (e) 30 days. Scale bar, 2 μm. HR-TEM, and SAED results of air exposure samples for (f) 1 day, (g) 7 days, (h) 15 days, (i) 30 days. Scale bar, 10 nm, 1/10 nm. j Statistical analysis based on TEM measurements after different exposure durations. Error bars represent the standard deviation (SD) from at least four measurements. k, XRD patterns: * indicates the hydrated phase in the sample. Pristine and exposed samples XPS of (l) C 1 s, (m) O 1 s. n Rate performance of exposed samples for different days at 0.02 A g−1 in 1.5-4.3 V. Schematic of sodium ion migration paths of water molecules on the surfaces of (o) NMO, (p) NMT-10, and water molecules at the interlayer of (q) NMO, (r) NMT-10. s The sodium ion migration energy barrier for water molecules at the surface and the interlayer.
The electrochemical performance of the NMT-10 cathode prepared with an aqueous-based CMC/deionized water binder after air exposure is listed with detailed parameters in Table S13. The GCD curves at 0.02 A g−1 show the material retains a high specific capacity of 181 mAh g−1 and exhibits stable rate and cycle performance, maintaining nearly 110 mAh g−1 at 1 A g−1 within the voltage range of 1.5-4.3 V (Figs. 5n, S56). After 500 cycles at 0.4 A g−1, the NMT-10 sample exposed to air for 30 days maintains a capacity retention of 74.24 % (Fig. S57), indicating improved air stability compared with some recently reported materials that suffered capacity fading upon air exposure78. This high capacity and sustained cycling stability is further supported by theoretical calculations (Figs. 5o-r), with the corresponding DFT-optimized atomic structures provided in Supplementary Data 22-25 and the Na+ migration processes illustrated in Supplementary Videos 6-9, which reveal that both surface-adsorbed and interlayer water molecules introduced during air exposure reduce Na+ migration energy barriers. Specifically, surface water decreases the diffusion barrier from 0.979 eV in NMO to 0.615 eV in NMT-10, while interlayer water reduces it from 0.295 eV to 0.284 eV (Fig. 5s). These results suggest that moderate water incorporation after Ti substitution can enhance structural stability and facilitate Na+ transport via structural and electronic modulation, aligning well with previous studies that highlight the beneficial role of lattice water in sodium-based cathode materials79,80,81. Moreover, the presence of interlayer water may serve as structural pillars that stabilize the TM-O framework and mitigate undesired lattice collapse during cycling, which has been reported in aqueous zinc batteries82. At the same time, this positive effect of water molecules sheds light on the underlying reason for the improved electrochemical performance of the cathode prepared by the aqueous binder. Electrochemical performance of NMT-10 cathodes prepared solvent-based PVDF/NMP binder after exposure to air is shown in Fig. S58, exhibiting reduced rate and cycling performance, especially within a wide voltage range of 1.5-4.3 V. This inferiority is primarily attributed to the chemical instability of the PVDF binder in alkaline environments, where defluorination by OH⁻ can lead to slurry gelation and degradation of the electrode structure after air exposure.
Towards scale-up production of PBAs/NMT-10 cathode and sodium supplement strategy for full battery application
Given the low cost, environmental friendliness, and good air stability of the NMT-10 cathode, production has been scaled up to promote the industrialization of layered oxide cathodes for SIBs. Figures 6a and S59 depict the scale-up preparation setup and process flow from precursor synthesis to electrode fabrication, with procedures provided in Supplementary Videos 10-12. The precursor is calcined to yield cathode powders, which are ball-milled, made into a slurry, coated on Al foil, dried, and collected to obtain electrodes suitable for practical cell assembly. The XRD (Fig. S60a) and SEM (Fig. S60b) analysis of the large-scale samples (LS-NMT10) revealed a typical composite structure. The electrochemical performance of the LS-NMT10 (Fig. S61a-d) is consistent with that of the laboratory samples, with 66.7 % retention after 800 cycles at 0.2 A g−1.
a Large-scale preparation of the NMT-10 cathode material. (I-Ⅱ) The precursor is weighed, mixed, and calcined in a high-temperature furnace to yield cathode powders, which are subsequently ball-milled and dispersed with solvent, binder, and conductive additives to form a homogeneous slurry. (Ⅲ-Ⅳ) The slurry is uniformly coated onto aluminum foil, dried, and calendered to obtain electrodes with optimized thickness, density, and mechanical properties for practical cell assembly. Electrochemical properties of Na | |PBAs-NMT10 half cells. b GCD curves at 0.02 A g−1 in 1.5-4.3 V. c The detailed histograms of first charge specific capacity at different voltage ranges. d Rate performance of PBAs and NMT-10 with different composite ratios in 1.5-4.3 V. e Cycling performance of the Hard Carbon | |PBAs -NMT10 (1:2.52) coin-type full cell at 1.8-4.2 V.
Although the large-scale production of the cathode has been achieved, the inherent low initial charge capacity remains unresolved, limiting its application in full-cell systems. To address this, two sodium supplement strategies were developed for cathodes prepared with solvent and aqueous-based binders. Initially, sodium oxalate was employed as a sodium supplement for NMT-5 cathodes fabricated with solvent-based PVDF/NMP binders. However, this approach is not ideal for practical full-cell applications as the supplemental sodium on the cathode rarely returns after migrating to the anode, making the process irreversible. Detailed structural characterization and electrochemical performance are proven in Figs. S62-64.
Secondly, ferromanganese-based PBAs were utilized as a sodium supplement for NMT-10 electrodes prepared with aqueous-based binder, owing to their low cost, abundant sodium content, flexible three-dimensional framework, and high capacity83,84. The composite of PBAs and NMT-10 was initially carried out at a mole ratio of 1:3.75, with structural characterization and electrical performance detailed in Figs. S65-67. To achieve higher initial charge capacity, different mole ratios of PBAs to NMT-10 were experimented with: 1:3.75, 1:2.52, 1:2.03, and 1:1.88; the corresponding XRD and SEM are shown in Fig. S68a-f. The first charging specific capacity and their different composite ratios were tested (Figs. 6b, S69) with intuitive data in Fig. 6c, showing that increased PBAs content improved the initial charge specific capacity (from 58.12 to 115.71 mAh g−1 at 2.0-4.0 V and from 66.22 to 121.01 mAh g−1 at 1.5-4.3 V). A comparison of the electrochemical performances of different composite ratios is displayed in Fig. 6d. The composite cathode with a 1:2.52 mole ratio exhibits relatively better electrochemical performance, maintaining a discharge specific capacity of nearly 137.26 mAh g−1 in the first five cycles (Fig. S70a), with stable rates and retention capacity (Fig. S70b-c and Fig. S71a-c). Subsequently, a coin-type full cell was fabricated with PBAs-NMT10 (1:2.52) cathode and hard carbon anode. It demonstrates a stable rate and cycling performance with a retention rate of 89.22 % for 150 cycles at 60 mA g−1 (Figs. 6e and S72). The corresponding charge and discharge specific energies calculated from the cathode mass are shown in Figure S73. To advance composite cathode applications, a 2.25 A·h pouch-type sodium-ion battery was assembled, with an energy density of 86.29 Wh/kg (calculated based on the total pouch weight). The detailed parameters of the pouch cell are listed in Table S16, and the corresponding schematic diagram, photographs, and cross-sectional views are shown in Fig. S74a-c. Furthermore, the pouch cell demonstrated good reversibility in the first five GCD curves (Fig. S75). Consequently, the implementation of PBAs as a sodium supplement strategy effectively enhanced the initial charging specific capacity and improved full-cell electrochemical performance, suggesting potential benefits for the application of sodium-deficient transition metal oxide cathodes.
In summary, a screening mechanism based on economic and environmental considerations, stress analysis, and electron localization function calculations, Ti was identified as the promising dopant element to regulate the distribution of the local electronic structure to enhance the structural stability of Mn-based oxide cathodes for SIBs. The optimized Na0.6Mn0.9Ti0.1O2 (NMT-10) cathode demonstrates favorable electrochemical performance and air/water stability when fabricated using aqueous-based binders, retaining 74.24 % capacity after 500 cycles at 0.4 A g−1 even after 30 days of air exposure. In-situ XRD and stress analysis revealed that NMT-10 undergoes a highly reversible P2-OP4 phase transition with lower strain in the second cycling process, which is attributed to the “spring effect” of TiO6 octahedra that buffers lattice stresses. Meanwhile, strong Ti-O bonds enhance structure stability by modulating the local electronic configuration of Mn and increasing its average valence, effectively suppressing J-T distortion, as evidenced by the inhibited P2-P2’ phase transition. In particular, the sodium-rich, low-cost, and high-capacity ferromanganese-based PBAs are proposed as a promising candidate for sodium supplementation, effectively improving the initial charge capacity and confirming the feasibility of large-scale fabrication and practical full-cell application of PBAs/NMT-10 cathodes. This study provides insights into the possible application of sustainable manganese-based oxide cathodes for SIBs.
Methods
Preparation of pristine transition metal oxides
P2-Na0.6Mn1-xTixO2 was synthesized via a solid-state reaction. The Na2CO3 (99.99 %, Aladdin, 10% in excess), Mn2O3 (99.99 %, Aladdin), and TiO2 (99.00 %, Aladdin) were accurately weighed and combined in the mortar according to the stoichiometric ratios. The resulting mixture was ground for 45 min and pressed into pellets under a pressure of 16 MPa. The pellets were then calcined in air at 1000 °C for 15 h, cooled to room temperature, and stored in an argon-filled glovebox.
Synthesis of PBAs
Fe/Mn-based Prussian Blue Analogs (PBAs) were synthesized through a precipitation method, utilizing reagents sourced from Aladdin. This procedure dissolved 8 mmol of FeSO4·7H2O, 8 mmol of MnSO4·H2O, and 16 mmol of sodium citrate in 400 mL of deionized water to prepare Solution A. In a separate container, 8 mmol of Na4Fe (CN)6·10H2O was dissolved in 400 mL of deionized water to form Solution B. After stirring for 3 h, Solution A was gradually added to Solution B using a peristaltic pump, and the mixture was stirred for an additional 3 h before being aged for 24 hours. The resulting PBAs material was washed three times using deionized water and ethanol, followed by drying at 120 °C for 12 hours to obtain the final product.
PBAs sodium supplement strategy
The prepared Fe/Mn-based PBAs cathode and sintered NMT-10 cathode powder were mixed in a mortar in molar ratios of 1:3.75, 1:2.52, 1:2.03, and 1:1.88, respectively, and homogeneously milled for 10-30 min as a composite cathode material.
Air exposure experiments
The sintered NMT-10 cathode material was divided into six aliquots and placed in individual sample tubes. These samples were then exposed to air at a room temperature of 25-26 °C for varying durations of 1, 3, 5, 7, 15, and 30 days. After each designated exposure period, the samples were labeled and subjected to structure characterization and electrochemical performance testing to evaluate the influence of air exposure on the material’s properties.
Materials characterization
X-ray diffraction was supported by a PANalytical (Aeris) diffractometer equipped with a Cu Ka source (λ = 1.5418 Å) in the diffraction range of 10°-80° at a scanning rate of 2°/minute. Lattice parameters were obtained from the collected XRD data by FullProf. Scanning electron microscopy (SEM3100-CIQTEK) and transmission electron microscopy (JEOL JEM-2011 Limited Corporation, Japan) were used for morphological characterization, respectively. Deep etch XPS characterization was carried out on a Thermo Scientific Nexsa X-ray photoelectron spectrometer. In situ structural evolution throughout the electrochemical reaction was examined using XRD (Bruker D8 Advance) with Cu Kα radiation. The sXAS measurements were performed at beamline 02B02 of the Shanghai Synchrotron Radiation Facility. The XAS spectra in TFY and TEY modes were normalized to the incident beam intensity and subsequently to the pre- and post-edge absorption levels. The quantitative analysis of the Mn L-edge iPFY spectra was performed through a linear combination fitting technique, employing standard reference spectra for Mn2+, Mn3+, and Mn4+.
Electrochemical measurements
Throughout this manuscript, we follow battery conventions and use “anode” and “cathode”, which correspond to the “negative electrode” and “positive electrode”, respectively. The half-cell electrochemical performance of the materials was evaluated in coin cells (CR2032) with a sodium (Na) sheet (99.9 %, Sigma-Aldrich, thickness 0.5 mm, diameter 12 mm) as the counter electrode and porous glass fiber (Whatman GF/D, 260 μm thickness) as the separator. The Na sheet was cut in a glovebox under argon-filled atmosphere (O2 < 0.1 ppm, H2O < 0.1 ppm) before cell assembly. The electrolyte was a solution of 1 M NaClO4 (dissolved in 1:1 = ethyl carbonate (EC): diethyl carbonate (DEC) in 5 vol% solution of fluorinated ethyl carbonate (FEC) by volume) (Duoduo Chemical Technology Co., Ltd). The cathode film was fabricated with two different binders: the solvent-based binder film involved dispersing a slurry of active material (70 wt.%), Super P (20 wt.%), and polyvinylidene fluoride (PVDF, 10 wt.%) were thoroughly mixed in N-methyl pyrrolidone solvent (Aladdin, 99.9%), while the aqueous-based binder film involved dispersing a slurry of active material (70 wt.%), Super P (20 wt.%), and carboxymethyl cellulose (CMC, 10 wt.%) were thoroughly mixed in deionized water. All slurries were stirred for 30 min at 25 °C in the MSK-SFM-12M Micro-vibration ball mill (300 rpm, Shenzhen Kejing). Then, the obtained homogeneous slurry was applied using a scraper (gap 200-250 μm) for single-sided coating onto aluminum foil (15 μm, 99.6%, Hubei Fengsheng Electronic Technology Co., Ltd.) to prepare the electrode. The aluminum foil current collector was not pretreated before electrode preparation. After coating, the electrodes were dried at 80 °C in a vacuum oven for approximately 12 h, then cut into discs with a diameter of 10 mm (1.5-2 mg cm−2) using the MSK-T10 manual slicer (Shenzhen Kejing). All electrodes were further dried at 100 °C in a vacuum oven overnight before transferring to an argon-filled glovebox (O2 < 0.1 ppm, H2O < 0.1 ppm) for cell assembling.
In the coin full cell, the anode film consisted of 95 wt.% hard carbon and 5 wt.% sodium alginate (SA). The SA solution was prepared by dissolving 2 g of SA powder in ~98 g of deionized water to obtain a 2 wt.% solution. The slurry was cast onto Al foil (15 μm, 99.9 %) and dried under vacuum at 90 °C for 12 h, then cut into discs with a diameter of 11 mm (1.8-2.5 mg cm−2). The cathode was prepared as described above, and the N/P ratio (total reversible capacity of the anode divided by that of the cathode) was 1.05-1.2. A glass fiber separator was used, and 1 M NaClO4 in EC: DEC (1:1 v/v) with 2 % FEC was used as electrolyte (~100 μL per cell). All coin cells were assembled in an argon-filled glovebox (O2 < 0.1 ppm, H2O < 0.1 ppm). Galvanostatic charge/discharge measurements were carried out over a voltage window of 1.8-4.2 V at room temperature (25 ± 1 °C).
Electrochemical measurements were tested on a Neware battery test system (CT-4008Tn-5V10mA-164, Shenzhen, China) over the voltage range of 2.0-4.0 V and 1.5-4.3 V vs. Na+/Na (1 C = 200 mA g−1, 60 min). The specific capacity (mAh g−1) was calculated based on the measured discharge capacity of the full cell divided by the total mass of the active materials in the electrodes. For the galvanostatic intermittent titration technique (GITT) test, the cathodes were charged at 0.02 A g−1 for 30 min, followed by a 10 h open-circuit relaxation. Cyclic voltammetry (CV) measurement was performed on a CHI760D electrochemical workstation (Chenhua, China).
Pouch cell assembly details
To assemble the Hard Carbon‖PBAs-NMT10 (1:2.52) pouch cell, twelve high-loading positive electrodes (135 mm*77.5 mm, Al foil current collector) were paired with thirteen Hard Carbon negative electrodes (141 mm*83.5 mm, Al foil current collector) in a sandwich-type configuration. A polyolefin separator slightly larger than the electrode area (145 mm*88.5 mm) was employed to fully cover the electrodes. The N/P ratio was controlled at 1.085. Aluminum foil was used as the current collector for the cathode, and copper foil for the anode, with aluminum and nickel tabs serving as the current leads. Approximately 20 g of electrolyte was injected into the pouch cell, followed by a gas release step before final pouch sealing. During electrochemical cycling, an external pressure of 200 N was applied. No chemical presodiation of the anode was performed prior to cell assembly. The detailed pouch battery parameters are displayed in Table S16.
FAULTS simulations
FAULTS, a software tool developed by CIC Energigune (download link: https://cicenergigune.com/en/faults), which can be used to simulate planar intergrowth defects. This program enables refinement of X-ray diffraction (XRD) patterns for crystal systems containing coherent planar faults. Herein, FAULTS was employed to model O/P intergrowth structures with varying O/P ratios. The simulation process involved decomposing the lattice into two fundamental layers to extract the slip units corresponding to the pure P2 and OP4 phases. These units were then used to define appropriate slip vectors and stacking probabilities for constructing the intergrowth models. Finally, the corresponding spectral graphs were simulated by the FAULTS software.
DFT calculations
The theoretical calculations were performed by the projector augmented wave method using the CASTEP module through Materials Studio software with a cutoff energy of 450 eV and a k-point mesh of 4 × 4 × 1, ensuring convergence of total energy (<10−6 eV/atom) and forces (<0.02 eV/Å). The generalized gradient approximation (GGA) by Perdew-Burke-Ernzerhof (PBE) functional was selected for the exchange-correction functional, and the GGA + U approach was used to deal with the strongly correlated d electrons of transition metals85,86. Different Hubbard U parameters were applied to the TMs in the system based on references and the recommendation of the Materials Studio software87,88. For example, a Hubbard U correction was applied to Mn 3 d orbitals (Ueff = 4.0 eV) to account for strong electron correlation, as validated in prior studies on Mn-based oxides87. Ti 3 d orbitals were treated with standard PBE due to their empty nature (Ti4+), which minimizes self-interaction errors, and a high Ueff (6.4) was applied to Ni when doping in the supercell due to a longer Ni-O bond length89 (Ni has a valance electron configuration of 3d84s2, while Mn has a valance electron configuration of 3d64s2, showing a smaller ionic radius). The associated Hubbard U-value tables are shown in the Supplementary Table 15.
The supercells were constructed based on the experimental results. Generally, there are 21 Na sites, 32 Mn sites, and 64 O sites in each 4×4×1 supercell of Na0.6MnO2. For the transition metal screening in Na0.6Mn0.98TM0.02O2 (TM = Sc, Ti, V, Cr, Mn, Fe, Co, Ni, Cu, Zn, Y, Zr, Nb, Mo, Tc, Ru, Rh, Pd, Ag, and Cd), For the Ti substitution (Na0.6Mn1-xTixO2, x = 0, 0.02, 0.05, 0.10, and 0.15) in Na0.6MnO2, the number of Ti atoms in the 4×4×1 supercell was explicitly determined by the doping level x, simulating the experimental results. A 4 × 4 × 1 supercell of (001) plane was constructed with a thickness of vacuum of 15 Å. The study included comprehensive calculations of the total density of states (TDOS) and the projected density of states (PDOS) to analyze the effects of Ti doping on the band structure in detail. To accurately probe the electron localized/off-domain distribution, we performed electron localization function (ELF) calculations. Ion migration barriers were computed using the climbing-image nudged elastic band (CI-NEB) method, and all atoms were allowed to relax with the fixed lattice parameters during the process90,91. Additionally, the atomic positions, lattice parameters (a, b, c), and cell volume were fully relaxed. Constraints: Only symmetry (space group P63/mmc) was preserved to maintain the P2-type layered structure.
Data availability
The source data generated in this study are provided in the Source Data file. Source data are provided with this paper.
References
Wang, Q. et al. Fast-charge high-voltage layered cathodes for sodium-ion batteries. Nat. Sustain. 7, 338–347 (2024).
Wang, J. et al. Routes to high-performance layered oxide cathodes for sodium-ion batteries. Chem. Soc. Rev. 53, 4230–4301 (2024).
Wang, P. F. et al. Na+/vacancy disordering promises high-rate Na-ion batteries. Sci. Adv. 4, 6018 (2018).
Gao, A. et al. Topologically protected oxygen redox in a layered manganese oxide cathode for sustainable batteries. Nat. Sustain. 5, 214–224 (2021).
Han, M. H., Gonzalo, E., Singh, G. & Rojo, T. A comprehensive review of sodium layered oxides: powerful cathodes for Na-ion batteries. Energy Environ. Sci. 8, 81–102 (2015).
Fu, F. et al. Entropy and crystal-facet modulation of P2-type layered cathodes for long-lasting sodium-based batteries. Nat. Commun. 13, 2826 (2022).
Liu, Z. et al. Ultralow volume change of P2-type layered oxide cathode for Na-ion batteries with controlled phase transition by regulating distribution of Na+. Angew. Chem. Int. Ed. 60, 20960–20969 (2021).
Jia, X. B. et al. Facilitating layered oxide cathodes based on orbital hybridization for sodium-ion batteries: marvelous air stability, controllable high voltage, and anion redox chemistry. Adv. Mater. 36, 2307938 (2023).
Li, C. et al. Hydrogels with amphiphilic chains and targeted adhesion for high-areal-capacity zinc batteries. Energy Stor. Mater. 60, 102858 (2023).
Ren, M. et al. Homeostatic solid solution in layered transition-metal oxide cathodes of sodium-ion batteries. J. Am. Chem. Soc. 145, 224–233 (2023).
Kubota, K., Asari, T. & Komaba, S. Impact of Ti and Zn dual-substitution in P2 type Na2/3Ni1/3Mn2/3O2 on Ni-Mn and Na-vacancy ordering and electrochemical properties. Adv. Mater. 35, 2300714 (2023).
Zhao, C. et al. Revealing high Na-Content P2-type layered oxides as advanced sodium-ion cathodes. J. Am. Chem. Soc. 142, 5742–5750 (2020).
Yang, L. et al. Lithium-doping stabilized high-performance P2-Na0.66Li0.18Fe0.12Mn0.7O2 cathode for sodium ion batteries. J. Am. Chem. Soc. 141, 6680–6689 (2019).
Wang, P. F. et al. Ti-substituted NaNi0.5Mn0.5-xTixO2 cathodes with reversible O3-P3 phase transition for high-performance sodium-ion batteries. Adv. Mater. 29, 1700210 (2017).
Wang, F. et al. Activating oxygen redox in layered NaxMnO2 to suppress intrinsic deficient behavior and enable phase-transition-free sodium ion cathode. Adv. Funct. Mater. 32, 2202665 (2022).
Cheng, Z. W. et al. Mitigating the large-volume phase transition of P2-type cathodes by synergetic effect of multiple ions for improved sodium-ion batteries. Adv. Energy Mater. 12, 2103461 (2022).
Liu, X. et al. Al and Fe-containing Mn-based layered cathode with controlled vacancies for high-rate sodium ion batteries. Nano Energy 76, 104997 (2020).
Wang, Q.-C. et al. Tuning sodium occupancy sites in P2-layered cathode material for enhancing electrochemical performance. Adv. Energy Mater. 11, 2003455 (2021).
Tang, Y. et al. Sustainable layered cathode with suppressed phase transition for long-life sodium-ion batteries. Nat. Sustain. 7, 348–359 (2024).
Somerville, J. W. et al. Nature of the “Z”-phase in layered Na-ion battery cathodes. Energy Environ. Sci. 12, 2223–2232 (2019).
Yabuuchi, N. et al. P2-type Nax[Fe1/2Mn1/2]O2 made from earth-abundant elements for rechargeable Na batteries. Nat. Mater. 11, 512–517 (2012).
Kumakura, S., Tahara, Y., Kubota, K., Chihara, K. & Komaba, S. Sodium and manganese stoichiometry of P2-Type Na2/3MnO2. Angew. Chem. Int. Ed. 55, 12760–12763 (2016).
Yang, T. et al. Stabilizing the deep sodiation process in layered sodium manganese cathodes by anchoring boron ions. Adv. Mater. 36, 2306533 (2023).
Kumakura, S., Tahara, Y., Sato, S., Kubota, K. & Komaba, S. P'2-Na2/3Mn0.9Me0.1O2 (Me = Mg, Ti, Co, Ni, Cu, and Zn): correlation between orthorhombic distortion and electrochemical property. Chem. Mater. 29, 8958–8962 (2017).
Zhang, G. H. et al. Suppressed P2-P2’ phase transition of Fe/Mn-based layered oxide cathode for high-performance sodium-ion batteries. Energy Stor. Mater. 51, 559–567 (2022).
Jia, S., Kumakura, S. & McCalla, E. Unravelling air/moisture stability of cathode materials in sodium ion batteries: characterization, rational design, and perspectives. Energy Environ. Sci. 17, 4343–4389 (2024).
Liu, Y. F. et al. Layered oxide cathodes for sodium-ion batteries: From air stability, interface chemistry to phase transition. InfoMat 5, e12422 (2023).
Wang, J. et al. Sustainable upcycling of spent LiCoO2 to an ultra-stable battery cathode at high voltage. Nat. Sustain. 6, 797–805 (2023).
Yang, Y. et al. Decoupling the air sensitivity of Na-layered oxides. Science 385, 744–752 (2024).
Seong, W. M., Kim, Y. & Manthiram, A. Impact of residual lithium on the adoption of high-nickel layered oxide cathodes for lithium-ion batteries. Chem. Mater. 32, 9479–9489 (2020).
Zhang, T. et al. Converting residual alkali into sodium compensation additive for high-energy Na-ion batteries. ACS Energy Lett. 8, 4753–4761 (2023).
Jian, Z.-C. et al. Solid-state synthesis of low-cost and high-energy-density sodium layered-tunnel oxide cathodes: dynamic structural evolution, Na+/vacancy disordering, and prominent moisture stability. Nano Energy 125, 109528 (2024).
Yuan, Y. et al. Understanding intercalation chemistry for sustainable aqueous zinc-manganese dioxide batteries. Nat. Sustain. 5, 890–898 (2022).
Jin, T. et al. Realizing complete solid-solution reaction in high sodium content P2-type cathode for high-performance sodium-ion batteries. Angew. Chem. Int. Ed. 59, 14511–14516 (2020).
Fang, K. et al. Injecting excess Na into a P2-type layered oxide cathode to achieve presodiation in a Na-ion full cell. Nano Lett. 23, 6681–6688 (2023).
Niu, Y.-B. et al. High-efficiency cathode sodium compensation for sodium-ion batteries. Adv. Mater. 32, 2001419 (2020).
Ding, Q. et al. W-doping induced efficient tunnel-to-layered structure transformation of Na0.44Mn1-xWxO2: phase evolution, sodium-storage properties, and moisture stability. Adv. Energy Mater. 13, 2203802 (2023).
Xiao, Y. et al. Deciphering an abnormal layered-tunnel heterostructure induced by chemical substitution for the sodium oxide cathode. Angew. Chem. Int. Ed. 59, 1491–1495 (2019).
Su, H. et al. Local spring effect in titanium-based layered oxides. Energy Environ. Sci. 13, 4371–4380 (2020).
Hu, H.-Y. et al. A universal strategy based on bridging microstructure engineering and local electronic structure manipulation for high-performance sodium layered oxide cathodes. ACS Nano 17, 15871–15882 (2023).
Tan, L. et al. Ti-substituted O3-type layered oxide cathode material with high-voltage stability for sodium-ion batteries. J. Colloid Interface Sci. 622, 1037–1044 (2022).
Goonetilleke, D. et al. Exploring the rate dependence of phase evolution in P2-type Na2/3Mn0.8Fe0.1Ti0.1O2. J. Mater. Chem. A 7, 12115–12125 (2019).
Linnell, S. F. et al. Enhanced oxygen redox reversibility and capacity retention of titanium-substituted Na4/7[ϒ1/7Ti1/7Mn5/7]O2 in sodium-ion batteries. J. Mater. Chem. A 10, 9941–9953 (2022).
Wang, Y., Xiao, R., Hu, Y.-S., Avdeev, M. & Chen, L. P2-Na0.6Cr0.6Ti0.4O2 cation-disordered electrode for high-rate symmetric rechargeable sodium-ion batteries. Nat. Commun. 6, 6954 (2015).
Bai, Q., Yang, L., Chen, H. & Mo, Y. Computational studies of electrode materials in sodium-ion batteries. Adv. Energy Mater. 8, 1702998 (2018).
Iddir, H. & Benedek, R. First-principles analysis of phase stability in layered-layered composite cathodes for lithium-ion batteries. Chem. Mater. 26, 2407–2413 (2014).
Meng, Y. S. & Arroyo-de Dompablo, M. E. First principles computational materials design for energy storage materials in lithium ion batteries. Energy Environ. Sci. 2, 589–609 (2009).
Liu, H. et al. Reviving sodium tunnel oxide cathodes based on structural modulation and sodium compensation strategy toward practical sodium-ion cylindrical battery. Adv. Mater. 36, 2407994 (2024).
Li, N. et al. Lowering sodium-storage lattice strains of layered oxide cathodes by pushing charge transfer on anions. Energy Environ. Mater. 7, e12671 (2023).
Casas-Cabanas, M., Reynaud, M., Rikarte, J., Horbach, P. & Rodríguez-Carvajal, J. FAULTS: a program for refinement of structures with extended defects. J. Appl. Crystallogr. 49, 2259–2269 (2016).
Zhao, X. et al. 3d-orbital regulation of transition metal intercalated vanadate as optimized cathodes for calcium-ion batteries. Adv. Funct. Mater. 34, 2309753 (2023).
Hou, Z. et al. Eliminating the Mn 3d orbital degeneracy to suppress the Jahn-Teller distortion for stable MnO2 cathode. Adv. Energy Mater. 14, 2302477 (2023).
Liu, Y. et al. Mitigation of Jahn-Teller distortion and Na+/vacancy ordering in a distorted manganese oxide cathode material by Li substitution. Chem. Sci. 12, 1062–1067 (2021).
Zhang, K. et al. Manganese based layered oxides with modulated electronic and thermodynamic properties for sodium ion batteries. Nat. Commun. 10, 5203 (2019).
Jin, J. et al. Annealing in argon universally upgrades the Na-storage performance of Mn-based layered oxide cathodes by creating bulk oxygen vacancies. Angew. Chem. Int. Ed. 62, e202219230 (2023).
Qing, R. P. et al. Enhancing the kinetics of Li-rich cathode materials through the pinning effects of gradient surface Na+ Doping. Adv. Energy Mater. 6, 1501914 (2015).
Chu, S. et al. Pinning effect enhanced structural stability toward a zero-strain layered cathode for sodium-ion batteries. Angew. Chem. Int. Ed. 60, 13366–13371 (2021).
Leube, B. T. et al. Activation of anionic redox in d0 transition metal chalcogenides by anion doping. Nat. Commun. 12, 5485 (2021).
Wang, Z. et al. Localized excitation of Ti3+ ions in the photoabsorption and photocatalytic activity of reduced rutile TiO2. J. Am. Chem. Soc. 137, 9146–9152 (2015).
Zhong, X. et al. Boosting oxygen reduction activity and enhancing stability through structural transformation of layered lithium manganese oxide. Nat. Commun. 12, 3136 (2021).
Xia, X. et al. Suppressing the dynamic oxygen evolution of sodium layered cathodes through synergistic surface dielectric polarization and bulk site-selective Co-doping. Adv. Mater. 35, 2209556 (2023).
Hu, N. et al. Unraveling the spatial asynchronous activation mechanism of oxygen redox-involved cathode for high-voltage solid-state batteries. Adv. Energy Mater. 14, 2303797 (2024).
Gan, L. et al. Highly symmetrical six-transition metal ring units promising high air-stability of layered oxide cathodes for sodium-ion batteries. Adv. Funct. Mater. 33, 2209026 (2023).
He, Q., Yu, B., Li, Z. & Zhao, Y. Density functional theory for battery materials. Energy Environ. Mater. 2, 264–279 (2019).
Shi, Q. et al. Niobium-doped layered cathode material for high-power and low-temperature sodium-ion batteries. Nat. Commun. 13, 3205 (2022).
Wang, C. et al. Tuning local chemistry of P2 layered-oxide cathode for high energy and long cycles of sodium-ion battery. Nat. Commun. 12, 2256 (2021).
Liu, S. et al. Reviving the lithium-manganese-based layered oxide cathodes for lithium-ion batteries. Matter 4, 1511–1527 (2021).
Shi, Y. et al. Slight compositional variation-induced structural disorder-to-order transition enables fast Na+ storage in layered transition metal oxides. Nat. Commun. 13, 7888 (2022).
Zhu, H. et al. Unblocking oxygen charge compensation for stabilized high-voltage structure in P2-type sodium-ion cathode. Adv. Sci. 9, 2200498 (2022).
Wang, S. et al. Air sensitivity and degradation evolution of halide solid state electrolytes upon exposure. Adv. Funct. Mater. 32, 2108805 (2021).
Yao, H.-R. et al. Designing air-stable O3-type cathode materials by combined structure modulation for Na-ion batteries. J. Am. Chem. Soc. 139, 8440–8443 (2017).
Zuo, W. et al. Engineering Na+-layer spacings to stabilize Mn-based layered cathodes for sodium-ion batteries. Nat. Commun. 12, 4903 (2021).
Zuo, W. et al. The stability of P2-layered sodium transition metal oxides in ambient atmospheres. Nat. Commun. 11, 3544 (2020).
Ji, P. et al. Unveiling the structural and chemical evolution of layered oxide cathode for Na-ion batteries induced by water vapor. Adv. Funct. Mater. 34, 2410485 (2024).
Sheng, H. et al. An air-stable high-nickel cathode with reinforced electrochemical performance enabled by convertible amorphous Li2CO3 modification. Adv. Mater. 34, 2108947 (2022).
Park, J. Y. How titanium dioxide cleans itself. Science 361, 753–753 (2018).
Hussain, H. et al. Structure of a model TiO2 photocatalytic interface. Nat. Mater. 16, 461–466 (2016).
Zuo, W. et al. Guidelines for air-stable lithium/sodium layered oxide cathodes. ACS Mater. Lett. 4, 1074–1086 (2022).
Nam, K. W. et al. Critical role of crystal water for a layered cathode material in sodium ion batteries. Chem. Mater. 27, 3721–3725 (2015).
Lim, S. Y. et al. Lattice water for the enhanced performance of amorphous iron phosphate in sodium-ion batteries. ACS Energy Lett. 2, 998–1004 (2017).
Zhu, K. et al. Tunable electrochemistry via controlling lattice water in layered oxides of sodium-ion batteries. ACS Appl. Mater. Interfaces 9, 34909–34914 (2017).
He, T. et al. Cation-deficient Zn0.3(NH4)0.3V4O10•0.91H2O for rechargeable aqueous zinc battery with superior low-temperature performance. Energy Stor. Mater. 38, 389–396 (2021).
Zhang, H. et al. Prussian blue analogues with optimized crystal plane orientation and low crystal defects toward 450 Wh kg-1 alkali-ion batteries. Angew. Chem. Int. Ed. 62, 202303953 (2023).
Zhang, H. et al. Long-cycle-life cathode materials for sodium-ion batteries toward large-scale energy storage systems. Adv. Energy Mater. 13, 2300149 (2023).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Paier, J., Hirschl, R., Marsman, M. & Kresse, G. The Perdew-Burke-Ernzerhof exchange-correlation functional applied to the G2-1 test set using a plane-wave basis set. J. Chem. Phys. 122, 234102 (2005).
Jain, A. et al. A high-throughput infrastructure for density functional theory calculations. Comput. Mater. Sci. 50, 2295–2310 (2011).
Daubner, S. et al. Combined study of phase transitions in the P2-type NaXNi1/3Mn2/3O2 cathode material: experimental, ab-initio and multiphase-field results. npj Comput. Mater. 10, 75 (2024).
Jia, H. et al. Persona of transition metal ions in solids: a statistical learning on local structures of transition metal oxides. Adv. Sci. 9, 2202756 (2022).
Chakraborty, A., Dixit, M., Aurbach, D. & Major, D. T. Predicting accurate cathode properties of layered oxide materials using the SCAN meta-GGA density functional. npj Comput. Mater. 4, 60 (2018).
Yin, W. et al. P2-type layered oxide cathode with honeycomb-ordered superstructure for sodium-ion batteries. Energy Stor. Mater. 69, 103424 (2024).
Acknowledgements
This work was supported by the National Key R&D Program of China (2024YFA1211900, Y.X.), National Natural Science Foundation of China (22579131, 52402301, Y.X.; 52472240, 52202284, Y.-F.Z.), Natural Science Foundation of Zhejiang Province (LQ23E020002, Y.X.), Wenzhou Key Scientific and Technological Innovation Research Project (ZG2023053, Y.X.), and China Scholarship Council (202106370062, J.-Y.L.). The authors thank BL02B02 of the Shanghai Synchrotron Radiation Facility (SSRF) for the beamtime and the University of Wollongong Electron Microscopy Center for the JEOL JEM-2011 electron microscope.
Author information
Authors and Affiliations
Contributions
X.-B.J., Y.-F.Z., X.-D.G., and Y.X. conceived and designed the experiments. X.-B.J., Z.-C.J., and N.-H.X. synthesized the materials and carried out the electrochemical analyses. H.-X.L., G.-Y.Z. performed full cell assembly and testing. Y.-F.L., J.-Q.W., and S.-X.D. provided manuscript revisions and suggestions. Q.-Q.P., J.-Y.L., and L.-Y.K. performed the in-situ synchrotron radiation XRD and deep-etch XPS analyses. C.C. and L.Z. collected and interpreted the XAS data. D.-C.C., H.-H.D., Y.S., and S.-Q.C. performed computational analyses. Y.X. supervised the work and contributed to the revision and finalization of the manuscript. All the authors participated in the discussion of the research.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Tanmoy K. Paul and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Jia, XB., Peng, QQ., Liu, YF. et al. Design principles of practical industrial-scale layered oxide cathodes with air/water stability for sustainable sodium-ion batteries. Nat Commun 16, 10477 (2025). https://doi.org/10.1038/s41467-025-65480-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-65480-x