Abstract
The durability and protective effect of naturally acquired antibodies against hepatitis E virus (HEV) reinfection and clinical progression remain unclear in humans. In a 103-month longitudinal analysis of 7032 adult placebo recipients (aged 16 to 65 years) from a phase 3 HEV vaccine trial in China, we demonstrated that baseline anti-HEV IgG seropositivity (n = 3194) conferred over 50% higher protection against reinfection compared with seronegative individuals (n = 3838), with this protective effect remaining consistent over 8.5 years. A non-linear dose-response relationship was observed, whereby baseline anti-HEV IgG concentrations ≥0.25 WHO units/mL were associated with at least a 50% reduction in infection risk, with higher baseline antibody levels correlated with a lower risk of infection. Natural immunity provided approximately 70% protection against clinically apparent hepatitis E in the cohort, with 10 symptomatic cases identified over a decade of active surveillance. Six were hospitalized, all of whom were baseline seronegative. These findings establish that natural HEV immunity provides durable, though incomplete, protection.
Similar content being viewed by others
Introduction
Hepatitis E virus (HEV) ranks among the leading causes of acute viral hepatitis worldwide1. An estimated 20 million HEV infections occur annually, resulting in approximately 3.3 million symptomatic cases, 70,000 deaths, and 3000 stillbirths2. HEV seroprevalence varies substantially, influenced by the sensitivity of the diagnostic assays used3. In low- and middle-income countries, seroprevalence ranges from 4% to 94%, in contrast to 1–52% in high-income countries1. In Asia, including India, Bangladesh, China, and Malaysia, as well as in most African countries, HEV antibody prevalence in natural population commonly exceeds 25%. In Europe, although seroprevalence is generally below 20%, recent increases have been reported in countries such as Germany, Greece, and France4. A meta-analysis estimates that about 939 million individuals—around one-eighth of the global population, have been infected with HEV, with 15–110 million experiencing recent or ongoing infections3.
Anti-HEV IgG antibodies can persist for many years post-infection5. In a 1978 outbreak in India, 47% (21 of 45) of affected individuals still had detectable anti-HEV IgG antibodies 14 years later6. A modeling study similarly predicted that approximately half of subclinically infected individuals would retain detectable antibodies after 14.5 years7. Although anti-HEV IgG antibodies induced by viral challenge confer protection against HEV reinfection in pigs8 and non-human primates9,10,11,12, the protective role of naturally acquired immunity in humans remains poorly defined. This knowledge gap largely reflects the lack of population-based studies with long-term serological follow-up and comprehensive hepatitis E case surveillance. Previous studies have suggested that individuals with naturally acquired anti-HEV antibodies or a history of hepatitis E may be prevented from severe disease during outbreaks, but this protection has not been quantitatively assessed, primarily due to constraints in research conditions and laboratory capacity13,14,15. Our earlier report demonstrated that anti-HEV IgG antibodies provide short-term (~12 months) protection against reinfection (RR = 0.25, 95% confidence interval [CI] 0.15–0.42); even marginal antibody levels (0.077–0.25 WU/mL) can significantly reduce the risk of subsequent HEV infection16. However, the duration of this protective effect remains uncertain.
Understanding the durability of naturally acquired HEV antibodies and their influence on population-level infection risk is essential for formulating effective hepatitis E prevention and control strategies, particularly in high-prevalence settings. Here, we present the findings from a 103-month longitudinal serological follow-up of placebo recipients enrolled in the phase 3 efficacy trial of the hepatitis E vaccine (Hecolin®, Xiamen Innovax Biotech, China)17,18,19. Our aim was to systematically investigate the protective effect of pre-existing anti-HEV antibodies across the hepatitis E disease spectrum (from subclinical infections to severe diseases) and to explore the duration of this protection.
Results
Analysis population
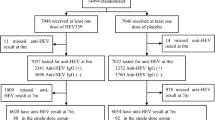
This study included 7032 placebo recipients from the serological follow-up cohort of the phase 3 Hecolin® trial20. According to the endpoints (HEV infection or hepatitis E disease) and the sampling intervals, we defined three analysis sets: the Disease Endpoint Analysis set (DE-A, n = 7032) for the symptomatic hepatitis E, the Overall Infection Analysis set (OI-A, n = 6743) for infections detected over 0–103 months, and the Precise Infection Analysis set (PI-A, n = 6369) for infections identified over 0–55 months using a more stringent definition (Fig. 1). In brief, participants in the DE-A with at least one valid serological follow-up after month 0 were included in the OI-A, and those in the OI-A with at least one pair of samples collected ≤13 months apart were further included in the PI-A. In each analysis set, participants were stratified into seropositive and seronegative groups based on their baseline anti-HEV antibody status. In the PI-A, demographic analysis revealed significant differences between groups in both sex distribution and age, with the seropositive group showing a higher male proportion (48.1% vs 33.7%, P < 0.0001) and an older mean age (46.7 years vs 43.9 years, P < 0.0001). Similar demographic patterns were observed in the OI-A and DE-A sets, with consistent and significant differences in sex ratios and age distributions between seropositive and seronegative subgroups (all P < 0.0001) (Table 1).
In the phase 3 clinical study of the hepatitis E vaccine Hecolin®, 7032 participants from the placebo arm of the immunogenicity subset (participants from Town QD and Town AF) who had valid baseline serostatus data were included in the analysis set for natural infection. Serum samples were collected at baseline (0 m) and scheduled blood collection time points including months 0, 7, 13, 19, 31, 43, 55, 67, 79, 91, and 103, and anti-HEV IgG antibodies were tested. In the PI-A set, the intervals between the paired samples include 6, 7, 12, and 13 months, with a median interval of 12 months. In the OI-A set, the intervals between the two consecutive samples range from 6 to 103 months, with a median interval of 24 months. The presence of HEV infection was determined by the dynamic changes in anti-HEV IgG antibodies across the series of serum samples. Hepatitis E cases were captured through an active hepatitis surveillance system established for the phase 3 clinical trial from 2007 to 2017.
Protection against HEV infection
The protective effect of pre-existing antibodies against HEV reinfection during 103 months is presented in Fig. 2 and Table S1. Among participants in the PI-A, 249 HEV infections were identified within the first 55 months, including 69 cases in the seropositive group (0.8 per 100 person-years) and 180 in the seronegative group (1.8 per 100 person-years). During this follow-up period, the crude protection of pre-existing antibodies against HEV infection was 55.8% (95%CI: 41.7, 66.5), and the adjusted estimate was 58.5% (44.8, 68.7). In the OI-A set, 381 HEV infections were identified during the 103-month period, with 113 cases in the seropositive group (0.7 per 100 person-years) and 268 in the seronegative group (1.5 per 100 person-years). The crude and adjusted protection estimates were 54.0% (42.7, 63.1) and 56.2% (45.1, 65.1), respectively. It should be noted that only one individual in the OI-A set was found to have two infections during the entire follow-up period; only the first infection was included in the analysis. The protective effects in the PI-A and OI-A groups across different follow-up periods indicated that protection remained stable over time, e.g., the adjusted protection in the OI-A set was 61.9% (37.8, 76.7) within 7 months and 57.6% (45.4, 67.0) within 55 months, which were comparable to that observed at 103 months (56.2%). We also conducted non-overlapping, time-stratified analyses that supported the stability of protection, although point estimates fluctuated and confidence intervals were wide due to the low incidence (Table S2).
The y-axis in the figure represents the protection rate over the entire follow-up period (from month 0 to the specified time point). The error bars in the figure represent the 95% confidence interval of the protection rate. a In the Precise infection analysis (PI-A) set. b In the Overall infection analysis (OI-A) set. A Poisson regression model was utilized to evaluate the relative risk (RR) of the incidence between the groups. The protection rate (%) was calculated as (1 − RR) ×100. The adjusted protection rate was calculated by including age, sex, and township as covariates in the model. Source data are provided as a Source Data file.
Additionally, the baseline characteristics comparisons between HEV infection cases and non-cases, as well as the protective effect across different sex and age groups, were analyzed. In the seropositive group, infected cases had a lower geometric mean concentration (GMC) of baseline antibody than non-cases (0.38 WU/mL vs. 0.57 WU/mL, P = 0.0022) (Table S3). Subgroup analyses indicated that younger participants appeared to benefit from higher protection. Notably, substantial protection was observed among women of childbearing age (16–45 years), with adjusted protection of 67.5% (39.3, 82.6) in the PI-A and 57.9% (33.5, 73.3) in the OI-A (Table S4).
The distribution of fold changes (increase or decrease) in anti-HEV IgG concentrations between consecutive samples, stratified by baseline serostatus, is shown in Fig. S1. Although a 4-fold rise in specific-antibody concentration is widely used to define viral infection, we conducted sensitivity analyses in the PI-A set using alternative thresholds to test the robustness of our findings. When infection was defined as a ≥3-fold increase or a ≥5-fold increase, the adjusted protection estimates remained consistent with those based on the 4-fold threshold over the study period, indicating that although the definition of HEV infection may influence estimated protection, the overall effect is modest (Fig. S2 and Table S5).
Furthermore, there was no difference in the GMCs of post-infection antibody between the seropositive and seronegative groups in both PI-A and OI-A sets, but the seronegative group exhibited a significantly higher fold increase in antibodies (PI-A: 446.6 vs. 34.6, P < 0.0001; OI-A: 330.0 vs. 28.5, P < 0.0001) (Fig. S3).
Protective antibody level
To evaluate the effect of antibody level on the protection against HEV infection, participants were stratified by anti-HEV IgG level at baseline. The HEV infection cumulative incidences of different baseline levels were shown in Fig. 3, with the seronegative group serving as the reference for calculating the relative risk (RR) (Table S6). In the PI-A set, we observed a dose–response relationship, where higher baseline antibody levels were associated with a lower risk of infection. Remarkably, no HEV infection was identified among individuals with baseline anti-HEV IgG ≥5.0 WU/mL. In the OI-A set, anti-HEV IgG antibody levels above 0.25 WU/mL were associated with a substantially reduced risk (0.33–0.39), though further increases in antibody concentrations did not yield additional reductions in infection risk. (Table S6, Fig. S4).
a In the Precise infection analysis (PI-A) set. b In the Overall infection analysis (OI-A) set. Participants were categorized into seven groups based on baseline anti-HEV IgG concentrations: not detectable (green line), [0.077, 0.25) (light blue line), [0.25, 0.5) (dark blue line), [0.5, 1.0) (light purple line), [1.0, 2.5) (dark purple line), [2.5, 5.0) (pink line), and ≥5.0 WU/mL (orange line). “Not detectable” indicates antibody concentrations below the cut-off of 0.077 WU/mL; for example, [0.077, 0.25) represents concentrations ≥0.077 but <0.25 WU/mL, [0.25, 0.5) represents ≥0.25 but <0.5 WU/mL, and so forth. Cumulative HEV infection incidence was shown for the PI-A set within 55 months and for the OI-A set within 103 months. P-value was calculated by the log-rank test for comparison across all groups. These figures of survival analysis (i.e., Kaplan–Meier) were generated using R software, version 4.5.1.
We further examined short-term protection by defining the “dynamic-baseline” as the antibody level measured in the first of any two consecutive samples. Participants were stratified by this dynamic baseline, and infection risk was assessed during the interval between the two consecutive samples. In the PI-A set, the median interval was 12 months, while the interval was broader in the OI-A set, with a median of 24 months. Across both sets, a consistent dose–response relationship was observed: higher antibody levels in the first sample predicted a lower risk of infection during the subsequent interval (Table S7, Fig. S4).
Protection against Hepatitis E
Over the 10-year observation period, ten confirmed cases of hepatitis E were identified(Table S8), including two cases in the seropositive group and eight in the seronegative group, which corresponded to an estimated natural immunity protection of 70.0% (−41.3, 93.6). After adjusting for sex, age, and township, the protection was 71.5% (−37.9, 94.1). Notably, none of the cases in the seropositive group required hospitalization, while six of the eight cases in the seronegative group did, resulting in an adjusted protection of 100.0% (39.0, 100.0) against hospitalized hepatitis E (Table 2).
The clinical characteristics of the 10 hepatitis E cases are summarized in Fig. S5. All cases had low anti-HEV IgG avidity (<40%). In the seronegative group, anti-HEV IgG levels significantly increased after disease onset, whereas in the seropositive group, IgG levels remained low (P = 0.0444); a similar trend was observed for IgM antibodies. Serum alanine aminotransferase (ALT) levels were significantly lower in the seropositive group compared to the seronegative group (P = 0.0444). Notably, six baseline seronegative hospitalized cases had ALT levels ≥15 times the upper limit of normal (ULN), whereas both seropositive cases had milder ALT elevations (2.5 and 3.1 ×ULN).
Discussion
In this study, we provide long-term, cohort-based evidence characterizing the protective effect of naturally acquired anti-HEV immunity. Key findings show that natural infection confers sustained protection against HEV reinfection, with an estimated protection of over 50% in 103 months. Importantly, individuals with baseline anti-HEV IgG concentrations ≥0.25 WU/mL had markedly reduced infection risk (adjusted RR < 0.33). Against clinical symptomatic hepatitis E, protection reached ~70% over 10 years, with all six hospitalizations occurring exclusively among baseline seronegative individuals. Despite the limited number of symptomatic cases, these results indicate that prior natural infection not only reduces the likelihood of reinfection but may also attenuate disease severity. The findings enhance our understanding of naturally acquired immunity and provide a critical framework for interpreting HEV seroepidemiological data and guiding similar research in endemic regions.
A previous study suggested a correlation between anti-HEV IgG levels and protection in humans, but it included both naturally and vaccine-induced immunity, complicating interpretation due to differing immune response kinetics16. In contrast, our investigation in an entirely unvaccinated population establishes a dose–response relationship specific to naturally acquired antibodies, thereby addressing a key gap in HEV seroepidemiology.
Based on the findings of this study, the symptomatic proportion of HEV infections was estimated at 4% (10 confirmed cases among 249 infections in PI-A set) or 3% (10 among 381 infections in OI-A set), placing it at the lower bound of previously reported estimates (5–30%)21. Indeed, this proportion would be even lower if a less stringent threshold for antibody titer elevation were used to define infection. Notably, it is well established that HEV-1 is more likely to cause symptomatic disease than HEV-4, with most HEV-4 infections being asymptomatic. In our study area, HEV-4 was the predominant circulating genotype, accounting for more than 93% of genotyped cases over the 10-year period (all detected cases since 2012 were caused by HEV-4)18. As noted previously, our active hepatitis surveillance system, comprising 205 clinical sentinels, largely enhances case identification sensitivity17,18,19; however, we cannot exclude the possibility that some infections with mild symptoms did not lead to medical consultation and were thus missed by the system, potentially resulting in an underestimation of the true symptomatic infection rate.
The long-term protective effect of natural HEV antibodies parallels patterns observed for some other non-respiratory viral infections. Unlike antibody responses to respiratory pathogens—such as influenza, RSV, and SARS-CoV-2—which typically wane within weeks to months, systemic viral infections like measles and hepatitis A often elicit durable immunity that effectively prevents reinfection. For hepatitis A virus (HAV), lifelong protection is thought to arise from sustained IgG antibody levels in combination with robust memory B and T cell activity22,23. Although the exact antibody threshold required for protection against HAV infection remains undefined, even low or detectable concentrations of anti-HAV IgG are presumed to be protective24. And even when titers drop below the threshold sufficient to block breakthrough infection, anti-HAV antibodies may still reduce the severity of clinical disease23. In line with this, our study found that the presence of anti-HEV antibodies was associated with lower ALT levels among those infected. Over the 103-month follow-up period, the GMC of anti-HEV antibody among baseline seropositive individuals declined slowly, and more than 80% of unvaccinated, baseline seropositive participants remained anti-HEV IgG-positive at month 10318. These findings support the durability of the observed protection, which may be largely attributable to the persistence of circulating antibodies. Nevertheless, future studies are needed to elucidate the immunological mechanisms underlying long-term protection, including the potential roles of long-lived plasma cells, memory lymphocyte subsets, and antigen persistence.
Despite offering partial protection (over 50%), relying on naturally acquired antibodies to establish herd immunity is neither practical nor advisable. The findings of this study indicate that only half of individuals with a history of HEV infection may be protected from reinfection. Moreover, the true disease burden imposed by HEV is still not fully understood, as HEV remains an underestimated pathogen due to misdiagnosis or underdiagnosis. HEV infection in pregnant women can lead to serious consequences, including a high maternal fatality rate of up to 3.2–70%25, as well as adverse pregnancy outcomes such as intrauterine and stillbirth26. In recent years, rising HEV incidence in high-income countries and reports of chronic hepatitis E in immunocompromised individuals have challenged the notion of HEV as a self-limiting tropical illness4. Although this study observed similar levels of natural antibody-derived protection in women of childbearing age and the general population, it remains unknown whether such immunity can reduce the risk of reinfection or disease severity among pregnant women.
Currently, an available recombinant hepatitis E vaccine, Hecolin® (Xiamen Innovax Biotech, Xiamen, China), has demonstrated a favorable safety profile and sustained high efficacy for at least 10 years18,27. In 2022, to control a hepatitis E outbreak, the first hepatitis E reactive vaccination campaign was conducted in Bentiu internally displaced persons camp, South Sudan28. Real-world data from the campaign showed that the adjusted effectiveness of a two-dose regimen in the context of an outbreak was 89.4%29,30. To date, no evidence suggests that protection obtained from natural infection is superior to vaccination, nor is there evidence to support exempting previously infected individuals from vaccination. In contrast to the protection against hepatitis E disease conferred by pre-existing antibodies (71.5% [−37.9, 94.1]) in this study, the efficacy of a complete regimen of Hecolin® in the same region and period was 86.6% (73.0, 94.1) over 10 years18. In addition, modeling studies have shown that HEV vaccination elicits long-lasting, high-level anti-HEV IgG responses, whereas antibody levels acquired through natural infection decline progressively over time7. Furthermore, this study was conducted in a setting where hepatitis E occurred sporadically, and HEV genotype 4 was the predominant circulating strain18,31. While our data show a slow decline in antibody level over 8.5 years18, other studies in high-incidence settings have reported faster seroreversion (~15% annual risk)32, potentially due to differences in infecting genotype (HEV-1 vs. HEV-4), exposure intensity, or assay sensitivity. Further cohort studies across diverse settings are needed to clarify these discrepancies.
This study acknowledges several limitations. Firstly, blood collections after 43 m were conducted at an interval of 24 months, and the number of participants with valid results beyond 67 months was limited, potentially reducing the number of detected HEV subclinical infections. This limitation may restrict our ability to fully characterize the long-term protective effect against HEV infection. And the small number of symptomatic hepatitis E cases reduced the statistical power to robustly evaluate disease protection. Secondly, this study may be subject to bias due to unmeasured or unidentified confounding factors. For example, individuals with baseline anti-HEV IgG seropositivity might represent a higher risk of HEV exposure; antibody-negative individuals cannot be ruled out as having acquired non-antibody-mediated protection through subclinical HEV infections33. These factors could potentially lead to an underestimation of the protective effect of pre-existing naturally acquired immunity. Thirdly, it was not possible to determine the precise timing of natural infection in the seropositive group at baseline, precluding accurate assessment of the true durability of natural immunity.
In summary, this study demonstrates that anti-HEV antibodies acquired through natural infection offer meaningful protection against reinfection and severe symptomatic disease. However, the level of immunity remains insufficient to eliminate transmission or avert disease burden on a population level, underscoring the continued need for effective preventive strategies, including vaccination.
Methods
Study design and participants
In the phase 3 clinical trial of the hepatitis E vaccine started from 2007, 112,604 participants aged 16–65 from 11 townships in Dongtai county, Jiangsu Province, China, were randomly assigned to receive Hecolin® or placebo (hepatitis B vaccine) in a 1:1 ratio17,18,19. Among them, 14,094 participants from Qindong (QD) and Anfeng (AF) townships were enrolled for blood sample collection, which was used for immunogenicity analyses and related exploratory investigations. After excluding 14 placebo recipients lacking baseline anti- HEV testing (total placebo arm n = 7046), 7032 participants from the placebo arm with valid baseline serostatus data were considered a natural population cohort and included in the analysis for this study (Fig. 1). Serum samples were collected at baseline (0 m) and predetermined time points: 7, 13, 19, 31, 43, 55, 79 and 103 months for QD, and 7, 19, 31, 43, 67 and 91 months for AF. All the samples were tested for anti-HEV IgG antibody. Participants were categorized into the seropositive group and the seronegative group based on baseline antibody results. Baseline data on age, sex, and township were also collected for all participants.
The study was done in accordance with the Declaration of Helsinki. This report is an extension analysis of a 10-year follow-up study based on a large-scale phase 3 trial of Hecolin® vaccine (NCT01014845), approved by the ethics committee of the Jiangsu Provincial Center for Disease Control and Prevention, and Xiamen University, Xiamen, China. Written informed consent was obtained from each trial participant, and the protocol was provided previously18.
Definitions of HEV infection and hepatitis E
The presence of HEV infection was determined by the dynamic changes in anti-HEV IgG antibodies across the series of serum samples. A fourfold or greater increase in anti-HEV IgG antibody concentrations between the two consecutive samples was considered indicative of HEV infection.
Hepatitis E cases were captured through an active surveillance system established for the phase 3 clinical trial of Hecolin from 2007 to 201717. Briefly, this system involved 205 clinical sentinels covering 11 townships. Serum samples were collected from all individuals with hepatitis-like symptoms (fatigue or loss of appetite ≥3 days) for ALT concentration measurement. If ALT was ≥2.5 times the ULN, a second serum sample was collected 2–6 weeks later. If the anti-HEV IgM antibody was positive for at least one of the two serum samples, further regular follow-ups and blood sampling would be performed until ALT returned to normal or symptoms disappeared. All sequential serum samples were tested for HEV infection-related markers by the central laboratory, including HEV RNA, anti-HEV IgM antibody, and anti-HEV IgG antibody. If at least two of these three acute hepatitis E indicators (RNA positive, IgM positive, and IgG fourfold increase) were positive, a hepatitis E case was diagnosed. When a confirmed case required hospitalization, investigators would further collect relevant medical records.
Analysis set
This study focused on two endpoints—HEV infection and Hepatitis E (symptomatic disease)— each requiring separate analysis sets (Fig. 1).
Protection against HEV infection was evaluated among populations with baseline and follow-up serum samples. Because not all participants attended every scheduled blood collection visit, this resulted in variability in the number of valid serum samples and the time intervals between samples. These differences could influence the accuracy of infection ascertainment. To address this, we conducted parallel analyses using two infection analysis sets—the precise infection analysis (PI-A) set and the overall infection analysis (OI-A) set—defined as follows: (1) PI-A set: included participants with at least one pair of serum samples collected ≤13 months apart (within 0–55 months of follow-up). The infection was determined as ≥4-fold anti-HEV IgG increase within a ≤13 months interval. (2) OI-A set: included participants with at least one valid post-baseline anti-HEV IgG measurement (within 0–103 months follow-up). The infection was defined as a ≥4-fold increase in IgG levels between any two consecutive samples, regardless of the interval (range: 6–103 months).
Protection against symptomatic hepatitis E and related hospitalizations was evaluated in the disease endpoint analysis (DE-A) set, comprising all participants with valid baseline anti-HEV IgG data.
Laboratory measurements
The tests for anti-HEV IgG antibodies were done by commercial ELISA kits (Beijing Wantai Biological Pharmacy, Beijing, China), following the instructions provided by the manufacturer. Quantification of anti-HEV IgG was further performed using the WHO reference reagent (NIBSC code 95/584) and expressed in WHO units per mL (WU/mL). Based on laboratory methodological validation prior to the initiation of the large-scale trial, the assay cut-off recommended by the manufacturer was 0.077 WU/mL. The negative test results were arbitrarily set as 0.0385 WU/mL (half of the cut-off value) in the calculation of GMC.
Avidity of anti-HEV IgG antibody was tested on serum samples collected within 60 days of symptom onset from confirmed hepatitis E cases18. Avidity was determined by measuring the residual antibody level in the presence of 5 M urea, expressed as a percentage of the residual antibody level in the absence of urea.
Statistical analysis
A Poisson regression model was utilized to estimate the RRs and accompany a 95% confidence interval (CI) for the incidence of HEV infection and symptomatic hepatitis E. The protection rate (%) was calculated as (1 − RR) ×100, with the 95% CI transformed accordingly. The adjusted protection rate was calculated by including age, sex, and township as covariates in the Poisson regression model. Person-years at risk were calculated as the sum of follow-up time for each participant in the specific analysis set, with follow-up censored at the first occurrence of infection or disease. In the OI-A set, the follow-up ended at the date of the last available sample collection or the first detected infection; in the PI-A set, it ended at the date of collection of the second serum sample in the last pair or the first detected infection. In the DE-A set, the total person-years were roughly estimated as 120 months multiplied by the number of participants; for the calculation of the RR in the seropositive group where the event count is zero, the lower limit of the 95% CI and the corresponding one-sided P-value (α = 0.025) were computed using the Exact Poisson Regression model.
Two approaches were employed to investigate the association between antibody levels and protection against HEV infection. The first was the “fixed-baseline” method, which used antibody levels from the month 0 serum sample to stratify participants and evaluate the long-term protection conferred by naturally acquired antibodies over the entire follow-up period. The second was the “dynamic-baseline” method, which used the antibody level from the first of any two consecutive serum samples to stratify participants and assess short-term protection within the interval between the two samples.
Comparisons of clinical characteristics between groups of participants with hepatitis E were performed using the Mann–Whitney U test with exact P-value calculation. All statistical analyses were conducted using SAS software, version 9.4 (SAS Institute, Cary, North Carolina, U.S.). All reported P-values were two-sided with α of 0.05, except for the exact Poisson regression analyses described above, which employed a one-sided P-value (α = 0.025) when zero cases occurred in the seropositive group. The missing data were not imputed. The figures of survival analysis (i.e., Kaplan–Meier) were generated using R software, version 4.5.1.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
Source data are provided with this paper. The individual-level raw data in this study were collected as part of a clinical trial and subject to confidentiality restrictions. The processed data are available under restricted access. Researchers may submit a study proposal or a framework with a clear analytical purpose (non-commercial) to access processed data after publication, directed to zhangj@xmu.edu.cn. The corresponding authors will promptly review the request and determine whether the requested data can be shared, and will respond within 4 weeks of receiving the request. Any data that can be shared will be released via a material transfer agreement. The data will be available for one year once access has been granted. Source data are provided with this paper.
Code availability
The custom SAS analysis code supporting the findings of this study is publicly available in the Zenodo repository under the MIT License: https://doi.org/10.5281/zenodo.17346830. The repository includes annotated SAS programs (.sas files). Users may use, modify, and share the code for non-commercial purposes, provided appropriate credit is given.
References
Raji, Y. E., Toung, O. P., Taib, N. M. & Sekawi, Z. B. Hepatitis E virus: an emerging enigmatic and underestimated pathogen. Saudi J. Biol. Sci. 29, 499–512 (2022).
Rein, D. B., Stevens, G. A., Theaker, J., Wittenborn, J. S. & Wiersma, S. T. The global burden of hepatitis E virus genotypes 1 and 2 in 2005. Hepatology 55, 988–997 (2012).
Li, P. et al. The global epidemiology of hepatitis E virus infection: a systematic review and meta-analysis. Liver Int. 40, 1516–1528 (2020).
Geng, Y., Shi, T. & Wang, Y. Epidemiology of hepatitis E. Adv. Exp. Med. Biol. 1417, 33–48 (2023).
Krain, L. J., Nelson, K. E. & Labrique, A. B. Host immune status and response to hepatitis E virus infection. Clin. Microbiol. Rev. 27, 139–165 (2014).
Khuroo, M. S. & Khuroo, M. S. Seroepidemiology of a second epidemic of hepatitis E in a population that had recorded first epidemic 30 years before and has been under surveillance since then. Hepatol. Int. 4, 494–499 (2010).
Su, Y. Y. et al. Persistence of antibodies acquired by natural hepatitis E virus infection and effects of vaccination. Clin. Microbiol. Infect. 23, 336.e331–336.e334 (2017).
Sanford, B. J. et al. Prior infection of pigs with a genotype 3 swine hepatitis E virus (HEV) protects against subsequent challenges with homologous and heterologous genotypes 3 and 4 human HEV. Virus Res. 159, 17–22 (2011).
Huang, W. et al. Cross-protection of hepatitis E virus genotypes 1 and 4 in rhesus macaques. J. Med. Virol. 80, 824–832 (2008).
Arankalle, V. A., Chadha, M. S. & Chobe, L. P. Long-term serological follow up and cross-challenge studies in rhesus monkeys experimentally infected with hepatitis E virus. J. Hepatol. 30, 199–204 (1999).
Arankalle, V. A., Chadha, M. S., Chobe, L. P., Nair, R. & Banerjee, K. Cross-challenge studies in rhesus monkeys employing different Indian isolates of hepatitis E virus. J. Med. Virol. 46, 358–363 (1995).
Uchida, T. et al. An epidemic outbreak of hepatitis E in Yangon of Myanmar: antibody assay and animal transmission of the virus. Acta Pathol. Jpn. 43, 94–98 (1993).
Bryan, J. P. et al. Epidemic hepatitis E in Pakistan: patterns of serologic response and evidence that antibody to hepatitis E virus protects against disease. J. Infect. Dis. 170, 517–521 (1994).
Chadha, M. S., Walimbe, A. M. & Arankalle, V. A. Retrospective serological analysis of hepatitis E patients: a long-term follow-up study. J. Viral Hepat. 6, 457–461 (1999).
Shata, M. T. et al. Protective role of humoral immune responses during an outbreak of hepatitis E in Egypt. Trans. R. Soc. Trop. Med. Hyg. 106, 613–618 (2012).
Zhang, J. et al. Protection against hepatitis E virus infection by naturally acquired and vaccine-induced immunity. Clin. Microbiol. Infect. 20, O397–O405 (2014).
Zhu, F. C. et al. Efficacy and safety of a recombinant hepatitis E vaccine in healthy adults: a large-scale, randomised, double-blind placebo-controlled, phase 3 trial. Lancet 376, 895–902 (2010).
Huang, S. et al. Long-term efficacy of a recombinant hepatitis E vaccine in adults: 10-year results from a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet 403, 813–823 (2024).
Zhang, J. et al. Long-term efficacy of a hepatitis E vaccine. N. Engl. J. Med. 372, 914–922 (2015).
Zhu, K. et al. Persistence of hepatitis E vaccine-induced antibody response across different dosage schedules and baseline serostatus. NPJ Vaccines 9, 245 (2024).
Lhomme, S., Marion, O., Abravanel, F., Izopet, J. & Kamar, N. Clinical manifestations, pathogenesis and treatment of hepatitis E virus infections. J. Clin. Med. https://doi.org/10.3390/jcm9020331 (2020)
Van Damme, P. et al. Hepatitis A virus infection. Nat. Rev. Dis. Prim. 9, 51 (2023).
Walker, C. M. Adaptive immune responses in hepatitis A virus and hepatitis E virus infections. Cold Spring Harb. Perspect. Med. https://doi.org/10.1101/cshperspect.a033472 (2019).
Stapleton, J. T. Host immune-response to hepatitis-A virus. J. Infect. Dis. 171, S9–S14 (1995).
Berglov, A., Hallager, S. & Weis, N. Hepatitis E during pregnancy: Maternal and foetal case-fatality rates and adverse outcomes-a systematic review. J. Viral Hepat. 26, 1240–1248 (2019).
Bigna, J. J. et al. Burden of hepatitis E virus infection in pregnancy and maternofoetal outcomes: a systematic review and meta-analysis. BMC Pregnancy Childbirth 20, 426 (2020).
Zhuang, C. et al. Effectiveness of a hepatitis E vaccine against medically-attended symptomatic infection in HBsAg-positive adults from a test-negative design study. Nat. Commun. 16, 1699 (2025).
Ciglenecki, I. et al. The first reactive vaccination campaign against hepatitis E. Lancet Infect. Dis. 22, 1110–1111 (2022).
Nesbitt, R. C. et al. The effectiveness of two doses of recombinant hepatitis E vaccine in response to an outbreak in Bentiu, South Sudan: a case-control and bias indicator study. Lancet Infect. Dis. https://doi.org/10.1016/S1473-3099(24)00657-1 (2025).
Zhuang, C., Wu, T., Zhang, J. & Xia, N. Hecolin vaccination strategies for hepatitis E outbreak control in resource-constrained settings. Lancet Infect Dis. https://doi.org/10.1016/S1473-3099(24)00686-8 (2025).
Huang, X. et al. Active surveillance of hepatitis E: a 10-year epidemiological analysis in a city in eastern China. Emerg. Microbes Infect. 13, 2373315 (2024).
Dighe, A. et al. Annual risk of hepatitis E virus infection and seroreversion: Insights from a serological cohort in Sitakunda, Bangladesh. Epidemiol. Infect. 152, e52 (2024).
Amanna, I. J. & Slifka, M. K. Mechanisms that determine plasma cell lifespan and the duration of humoral immunity. Immunol. Rev. 236, 125–138 (2010).
Acknowledgements
This study was supported by the National Key Research and Development Plan (2023YFC2307602; 2024YFC2310602) (to J.Z.; to Z.Z.), National Natural Science Foundation of China (32370160; 823B2086) (to Z.Z.; to Q.C.), Fujian Provincial Natural Science Foundation (2022J02005) (to Z.Z.), the Fundamental Research Funds for the Central Universities (20720220006) (to N.X.), and the Postdoctoral Fellowship Program and China Postdoctoral Science Foundation (GZB20250195) (to C.Z.). The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
X.L. and X.Z. were co-first authors of this manuscript. T.W., S.H., C.Z., J.Z., and N.X. conceived of the study. X.Z., H.J., Y.W., C.Y., D.L., and W.Z. were investigators. K.Z., X.H., X-W.H., Z.B., Q.C., and Z.Z. were responsible for sample testing or management. Y.S. and S.H. managed the data. X.L., Y.S., and C.Z. performed the statistical analysis. X.L. drafted the manuscript and S.H., C.Z,. and J.Z. critically revised it.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Liu, X., Zang, X., Zhu, K. et al. Long-term protection from naturally acquired immunity against hepatitis E virus reinfection. Nat Commun 16, 11187 (2025). https://doi.org/10.1038/s41467-025-66188-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-66188-8