Abstract
Seizures are frequent complications in brain tumor patients, yet the underlying neuronal mechanisms remain poorly defined. Here, we examined pathophysiological alterations in the peritumoral cortex of patients undergoing tumor resection. The synaptic activity, dendritic spine density, and gene expression of peritumoral pyramidal neurons differed significantly between patients with and without seizures. Using an inducible glioma rodent model, we characterized the progression of these alterations and their predictive value for seizure initiation. Computational simulations revealed that human cortical neurons are highly susceptible to synaptic and dendritic perturbations, which induce paroxysmal depolarizing shifts (PDS) in affected networks. Longitudinal analyses post-surgery showed that PDS were detectable prior to seizure onset in a subset of patients and reliably predicted post-resection seizure occurrence. These findings elucidate key neuronal substrates of tumor-associated seizures and suggest PDS as a potential biomarker for seizure risk, offering a foundation for targeted diagnostic and therapeutic strategies.
Similar content being viewed by others
Introduction
Patients with primary brain tumors have a high incidence of tumor-associated seizures (TAS) either as their presenting symptom or during disease progression1,2,3. Over half of the brain tumor patients experience TAS during their disease progression3. The occurrence of seizures considerably impacts disease burden and negatively affects the quality of life of brain tumor patients4. Treatment of TAS with antiepileptic drugs is often compromised by their side-effects and potential interactions with chemotherapeutics, which in turn can even further reduce seizure control5. Therefore, a better understanding of TAS pathogenesis is crucial for early diagnosis and for the development of better therapeutic approaches.
Even though the exact etiology of TAS remains elusive, recent studies have suggested that the peritumoral cortex (PTC), where tumor cells invade cortical tissue, is a key region for seizure development and progression6. The initiation zone for epileptic activity is usually located in the PTC, forming multiple seizure foci around the tumor7,8. Several cellular and circuit mechanisms in the PTC have been indicated to contribute to TAS. Glioma cells cause elevation of local glutamate concentration and alter the neuronal excitability via the non-synaptic xCT transporter system and/ or by impairing local glutamate reuptake9,10,11.
More recent evidence indicates that neuron-tumor crosstalk in the PTC is a crucial regulator of tumor progression and in turn tumor progression can alter neuronal functioning12,13,14,15. Neurons in the PTC can form excitatory electrochemical synapses with surrounding tumor cells, creating direct communication pathways within the peritumoral microenvironment, which aids in the progression and proliferation of tumor cells via direct and paracrine signaling pathways16,17,18,19,20. Furthermore, synaptogenic factors produced by specific astrocyte populations have also been linked to TAS21. Using an inducible glioma mouse model, a recent study illustrates that the progression of hyperexcitability in the PTC is accompanied by microglial inflammation during tumor growth22. In light of this emerging concept of tumor-neuron interaction in cancer neuroscience23, we postulate that tumor progression might alter the electrophysiological and structural properties of peritumoral neurons, and thereby directly drive seizure induction in PTC.
To date, the electrophysiological and structural characteristics of peritumoral neurons in patients with brain tumors remain insufficiently defined. Furthermore, the clinical significance of these alterations, particularly in relation to seizure initiation and suppression, requires further elucidation through detailed investigations of peritumoral cortical electrophysiology. Recent findings have demonstrated that tumor-induced neural remodeling activates the mTOR signaling pathway and disrupts the functional integrity of cortical pyramidal neurons in rodent models24. Notably, progressive functional impairments have been identified in layer 2/3 pyramidal neurons (L2/3 PCs) of the cerebral cortex using in vivo calcium imaging24. Given the established role of L2/3 PCs in the initiation and propagation of cortical seizures25,26, these alterations may contribute to the pathophysiology of TAS.
To directly test this hypothesis, we investigated the electrophysiological and morphological properties of human cortical neurons, with a specific focus on the L2/3 PCs, from a cohort of brain tumor patients. In addition, we followed up on the post-surgical seizure progression of these patients. Our results illustrate synaptic and dendritic alterations in the PTC neurons of TAS patients. To model the tumor progression and its impact on neuronal properties in the PTC prior to seizure onset, we examined the progressive neuronal alterations in an inducible, immunocompetent glioma mouse model27. Consistent with the aberrant neuronal properties in TAS patients, mice bearing cortical glioma also developed specific alterations in their intrinsic neuronal properties, which occurred during the early phase of tumor progression prior to seizure onset. Numeric simulation indicated that human cortical neurons are highly sensitive to enhanced dendritic inputs, which drive paroxysmal depolarizing shifts in the neurons of diseased circuits. By correlating the neuronal properties of individual patients with their long-term seizure prognosis, we indeed identified that the occurrence of paroxysmal depolarizing shifts precedes the onset of clinically overt seizures, which could be used as a biomarker for identification and intervention of post-surgical TAS in brain tumor patients.
Results
Altered intrinsic properties of L2/3 PCs in the peritumoral cortex
For this prospective study, we obtained cerebral cortical tissue from a cohort of brain tumor patients who underwent first time tumor resection from 2016 to 2020 (Supplementary Tables 1 and 2). We focused exclusively on tissue less than 1 cm away from solid tumor, i.e., the PTC, where tumor invades the surrounding cortex without clear signs of scarring or necrosis (Fig. 1a and Supplementary Fig. 1). Histological and molecular diagnosis for all cases was confirmed post-hoc by neuropathologists (Fig. 1b, c). Both high- and low-grade tumor patients were included in this study, and tissue samples were mostly collected from frontal and temporal cortical areas (Fig. 1c). Oncological and neurological symptoms of progression were prospectively followed up for a minimum of two years or until death. For our electrophysiological experiments, 37 patients were diagnosed with TAS prior to surgery (Supplementary Table 1). The non-TAS group consists of 19 patients who had no pre-surgical seizures and did not show any post-surgical seizure-related symptoms for last least 2 years or till death. Tissue was transferred to laboratory for ex-vivo electrophysiological examinations within 15 min after resection (see “Methods”).
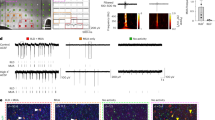
a Example MRI images aligned with intra-operative neuronavigation for tissues resected from two patients, locations of tissue removal indicated in green areas. b Hematoxylin-eosin (HE) staining of PTC from an anaplastic oligodendroglioma patient. Zone I indicates the peritumoral cortex with clear cortical layers. Zone II indicates the tumor area with giant nuclei (inset), mitoses, abnormal vasculature, and high cell density (right panel). Zone III with HE (bottom left) and Ki67 (bottom right) staining indicate the tumor invasion zone. c Scheme of experimental design. Peritumoral neurons in the PTC slices are recorded with whole-cell patch-clamp techniques. Right pie charts indicate distribution of tumor types within the cohort and anatomical sites of tissue resection. Gr WHO grade, Oligo oligodendroglioma, Astro astrocytoma, GBM glioblastoma. d Recorded cells are filled with biocytin and reconstructed using confocal imaging (staining used are streptavidin, magenta; SMI 32, yellow; GFAP, blue). Image shows L2/3 (left cells) and L5/6 pyramidal cells (right cells). Right panel: a higher magnification of the L2/3 pyramidal cell from dashed box. Inset: infrared differential interference contrast (IR-DIC) image showing the same cell and attached patch-clamp pipette. e Left: example voltage traces of a L2/3 pyramidal cells in response to incremental current injections. Right panels: distinct resting membrane potential (P = 0.0005) and input resistance (P = 0.0057) of L2/3 PCs from patients with TAS (red, n = 122 cells) and without TAS (grey, n = 55 cells). f Left: example voltage traces showing maximum firing frequency of TAS (red) and non-TAS PCs (grey). Right panel: quantifications of spike frequencies in response to depolarizing currents in TAS (red, n = 122 cells) and non-TAS groups (grey, n = 53 cells, linear regression with comparison of slope). Inset shows the maximum frequencies for both groups. Error bars show mean ± SEM. *P < 0.05, **P < 0.01, ***P < 0.001. ns not significant, PTC peritumoral cortex, AP action potential, PC pyramidal cell, TAS tumor-associated seizures. Statistics: e, two-sided Mann–Whitney test. f linear regression with comparison of slope. Source data are provided as a Source Data file.
We performed whole-cell intracellular recordings from the cortical neurons resected from peritumoral regions (Fig. 1c, d). All recorded cells were filled with biocytin for histological reconstruction to confirm their layers and cell types (Fig. 1d and Supplementary Fig. 1). In total, we recorded 177 L2/3 PCs. We examined the intrinsic neuronal properties of L2/3 PCs from brain tumor patients with and without TAS. L2/3 PCs from TAS patients had a more depolarized resting membrane potential, accompanied by a lower input resistance (Fig. 1e). We next examined their excitability in response to incremental current injection steps. Both the action potential threshold and the current intensity required to drive the first action potential (rheobase) were comparable between the TAS and non-TAS groups (Supplementary Fig. 2a–c). PCs from both groups had comparable current injection-action potential frequency relationships (Fig. 1f), as well as similar action potential properties (Supplementary Fig. 2d–i). Whereas the sag potential in response to hyperpolarizing current, an indication of hyperpolarization-activated inwardly rectifying current, was lower in the TAS patients (Supplementary Fig. 2j). Collectively, these results indicate specific TAS-associated alterations in the intrinsic neuronal properties of L2/3 PCs in brain tumor patients.
We next sought to examine whether the presence of tumor also affects other types of cortical neurons. Layer 5 pyramidal cells (L5 PCs) have distinct morphological and electrophysiological features from L2/3 PCs and could have their specific contributions to epileptogenesis28,29. We examined the intrinsic properties of L5 PCs (n = 26). In contrast to altered intrinsic properties in L2/3 PCs, we did not observe any significant difference in the membrane potential, input resistance, or action potential threshold of TAS L5 PCs (Supplementary Fig. 3a–c). In addition, action potential properties and firing frequency were comparable between these groups (Supplementary Fig. 3d–g), although the numbers of L5 PCs were relatively limited. Last, to clarify the contribution of cortical inhibitory interneurons to seizure progression30,31,32, we examined the intrinsic properties of 74 cortical interneurons. The resting membrane potential of interneurons was more depolarized in TAS patients, although not as pronounced as the L2/3 PCs. The other intrinsic properties were comparable between TAS and non-TAS groups (Supplementary Fig. 4a–f). Furthermore, we observed no difference in the intrinsic excitability of L2/3 interneurons (Supplementary Fig. 4g). Taken together, our data illustrates clear changes in intrinsic properties of peritumoral cortical neurons.
TAS-associated alterations in excitatory synaptic activity, dendritic spine density, and transcriptomic profiles
Based on the electrophysiological findings in L2/3 PCs, we examined the excitatory inputs from local networks to L2/3 PCs (Fig. 2a). L2/3 PCs from TAS patients had a significantly higher spontaneous excitatory postsynaptic current (sEPSC) frequency (Fig. 2b), whereas the sEPSC amplitude, rise, and decay times were comparable to the non-TAS group (Fig. 2b). When groups were separated based on the brain regions from which tissue was collected, the TAS group consistently showed higher excitatory postsynaptic current (EPSC) amplitudes across different cortical areas. However, these differences did not reach statistical significance, likely due to the small number of neurons analyzed within each regional subgroup (Supplementary Fig. 5). No difference was observed for the sEPSC properties in L5 PCs and L2/3 interneurons (Supplementary Fig. 6b–e). In our initial analysis, we included a small group of patients with metastatic tumor (N = 6 for Non-TAS; N = 4 for TAS). As glioma and metastatic tumor may have different effects on peritumoral neurons, we next focused our analysis on the cells from glioma patients. Consistently, the EPSC amplitudes were significantly higher in the glioma patient group with TAS (Supplementary Fig. 5a, b). Finally, we recorded inhibitory postsynaptic currents (IPSCs) in a subset of glioma patients to assess the effect of inhibitory inputs on peritumoral neurons. In these subgroups of recordings, we did not observe a significant change in IPSC frequency or amplitude (Supplementary Fig. 5c). Altogether, our data revealed specific alteration of excitatory inputs in the PTC, indicating their potential contribution to TAS.
a Example sEPSC traces of L2/3 PCs from patients without TAS (no seizures, grey) and patient with seizures (seizures, red). b Quantifications of the sEPSC frequency (P = 0.0011), amplitude (P = 0.6898), rise time (P = 0.1299), and decay time (P = 0.9164) of L2/3 PCs from patients without TAS (grey, n = 46 cells) and with TAS (red, n = 88 cells). c Left: Confocal image of a labelled L2/3 PC with various orders of branches indicated (1st order branch, 2nd order branch, apical dendrite, basal dendrite). Middle: high magnification example images of dendritic spines from the four dendritic zones indicated. Right panel: Quantifications of dendritic spine density for cells from non-TAS patients (grey, n = 21 cells) and TAS patients (red, n = 35 cells). TAS patients have higher spine densities overall with significant differences in the apical (AD) (P = 0.0349) and 2nd branch (2nd br) (P = 0.016) dendrites. d RNA sequencing results for excitatory neuronal markers and proliferation marker Ki67 in 6 non-TAS patients (patient 1–6, x-axis) and 6 TAS patients (patient 7–12), bar shows log scale of number of reads. e comparative transcriptomic analysis for all patients with top 10 upregulated and top 10 downregulated genes. Colors represent gene functions, legends are shown below figure, bar shows log scale of number of reads. TAS tumor-associated seizures, sEPSC spontaneous excitatory post synaptic current. Error bars show mean ± SEM. *P < 0.05, **P < 0.01, ***P < 0.001. Statistics: b, two-sided Mann–Whitney test. c two-sided unpaired t-tests per dendritic region. Source data are provided as a Source Data file.
The higher frequency of synaptic inputs to L2/3 PCs could be related to structural alterations in L2/3 PCs and therefore, enhance connectivity. We assessed the morphological properties of L2/3 PCs. The overall dendritic arborization showed no obvious differences between neurons from TAS and non-TAS patients (Supplementary Fig. 6a). However, quantification of the dendritic spine densities of different dendritic regions revealed that the dendritic spines densities were generally higher in PCs of TAS patients, especially in the apical dendrites and second order branches (Fig. 2c).
To investigate the possible molecular mechanisms underlying the electrophysiological and morphological alterations observed in patients with TAS, we conducted single-cell RNA sequencing (scRNAseq) of L2/3 PCs from patients with and without seizures. Individual neurons were harvested using patch-clamp electrodes, followed by Smart-seq2 library preparation and sequencing via the well-established patch-seq protocol33,34. In total, 12 samples, 6 from TAS patients and 6 from non-TAS patients (all diagnosed with high-grade glioma), were successfully sequenced. All sequenced samples exhibited high expression of excitatory neuronal markers and undetectable levels of the proliferation marker Ki67 (Fig. 2d), confirming that these were peritumoral L2/3 neurons rather than recently characterized spiking glioma cells35. Comparative transcriptomic analysis between the TAS and non-TAS groups revealed a distinct set of genes that were significantly upregulated or downregulated in the TAS group. Notably, many of these differentially expressed genes are implicated in the regulation of neuronal homeostasis, synaptic transmission, and synaptic plasticity (Fig. 2e and Supplementary Fig. 7). These findings suggest that specific transcriptional alterations in peritumoral neurons, consistent with other recent reports24,36, may contribute to seizure susceptibility and progression in TAS.
Computational simulations indicate that human L2/3 PCs are highly sensitive to synaptic alterations
We next used computational simulation of human L2/3 PCs to reveal the impacts of synapto-dendritic alterations on neuronal activities. We constructed a detailed compartmental model of a L2/3 PC that consists of a somatic compartment, 101 basal dendritic sections, 81 apical tree sections, and an axonal cable to replicate the electric and morphological characteristics of human L2/3 PCs (Fig. 3a, see “Methods”)37. To simulate the pathological alterations of dendritic spine densities, we varied the number of synapses that project to different dendritic subregions of this neuron (Fig. 3b). The maximum strength per synaptic input was set to 0.5 pA (see “Methods”), which matches the sEPSC amplitudes we recorded from human neurons. Increasing dendritic spine densities consistently enhanced the synaptic inputs in 4 distinct dendritic subregions. Increasing the dendritic spine density from 10 to 15, which matches our observation from TAS patients, doubled the spike outputs (Fig. 3c). Therefore, our results illustrate that increased dendritic spine densities in L2/3 PCs cause substantial increase in excitatory synaptic inputs, which together could lead to higher seizure susceptibility.
a Human L2/3 PC modeled with the whole-cell morphology and four dendritic regions of interest, 1st order branch (grey), 2nd order branch (green), apical dendrite (yellow), and basal dendrite (magenta). b Simulations of the somatic membrane potential traces and the corresponding EPSC traces when 11 (left) and 15 (right) synapses are distributed at four dendrites, respectively. c Input-output curves when 7 (red), 10 (blue), and 15 (cyan) synapses are distributed at the dendritic regions of interest, respectively. The x-axis represents stimulation frequency from 10 Hz to 100 Hz with step size of 10 Hz. The y-axis represents the somatic spiking response frequency (output rate in Hz). The curve is fitted by the Hill function. EPSC excitatory post synaptic current. Error bars show mean ± SEM (Each simulation described was run 10 times, duration of each simulation 1000 ms). Source data are provided as a Source Data file.
Tumor progression and seizure development in a clinically relevant glioma mouse model
Even though epilepsy is one of the most common initial symptoms associated with brain tumor diagnosis3, the key neuronal alterations occurring at the early phase of tumor progression are currently unclear. To uncover the temporal and spatial development of neuronal alterations during tumor progression, we examined the neuronal properties of PTC neurons in a clinically relevant mouse model of high-grade glioma. We made use of an inducible, highly reproduceable cortical glioma mouse model with intact immune response, a critical factor for both brain tumor biology and epileptogenesis38,39,40,41. We injected AAV1-GFAP-cre virus in the primary motor cortex of the mice carrying floxed Pten, floxed Ink4a/Arf, and lox-Stop-lox K-Rasv12 transgene (namely: Pten;Ink4a/Arf;K-Rasv12 mouse)27 (Fig. 4a). Cre-recombinase expression under a GFAP promoter, resulting in glial-specific deletion of Pten, Ink4a/Arf, and overexpression of K-Rasv12. All AAV1-GFAP-cre injected mice developed focal brain tumors in motor cortex 21 days post injection (DPI) and displayed histopathological hallmarks of high-grade glioma (WHO grade 3 and 4), including an invasive growth pattern and increased cell density with nuclear atypia such as giant cells and mitoses (Fig. 4b and Supplementary Fig. 8a). In contrast, none of the conditional mice injected with a control viral vector AAV1-CB7-GFP developed a detectable tumor (Supplementary Fig. 8c).
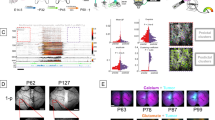
a Schematic illustration of experimental design for glioma induction and measurements in the glioma mouse model. Deletion of PTEN, Ink4a/Arf, and overexpression of K-RasV12 were induced by AAV-GFAP-cre injection in the primary motor cortex of triple conditional mouse. Measurements were carried out on 7, 14, and 21 days post injection (DPI). Brain outlines from Allen Mouse Brain Atlas (atlas.brain-map.org). b Representative hematoxylin-eosin (HE) staining and Ki67 immunolabelling of mouse brain with tumor-bearing cortex. Tumor resembles features of human high-grade glioma, including high cell density, mitoses, giant nuclei, and high density of Ki67 expression. White arrows indicate giant nuclei. c Confocal imaging series of tumor-bearing cortex on 7, 14, and 21 DPI (see “Methods”). Neurons indicated by NeuN (magenta), glial reaction indicated by GFAP (grey), and proliferating cells of growing tumor indicated by Ki67 (green). d Progression of tumor size on 7 DPI (green, N = 3 animals), 14 DPI (blue, N = 7 animals), and 21 DPI (magenta, N = 9 animals). (7 vs 14 DPI P = 0.7733; 14 vs 21 DPI P = 0.0067; 7 vs 21 DPI P = 0.0063) e Progressive changes of cell composition during tumor progression. Top row: Representative images from tumor-bearing cortical regions used for intensity analysis. Bottom row: fluorescent intensity analysis showing progressive reduction of neuron density (NeuN, magenta), expanding GFAP reactive rim (grey), and growing Ki67-positive tumor core (green) in time. Thin lines indicate individual animals (N = 2 animals per timepoint, see “Methods”), thick lines represent composite mean signal. The center of the tumor is indicated with dashed and the distance from tumor core is indicated on x-axis. f Confocal image of 21 DPI tumor-bearing cortex showing distinct borders of tumor core, infiltrated zone, and outer ridge. g Left: Example images of VGlut1/2 stainings (magenta), NeuN (cyan), Ki67 (green), and GFAP (blue) in peritumoral regions. Right: quantification of the densities of VGlut1/2 positive puncta per 25600 µm2 in the peritumoral regions. 7-DPI (N = 2, n = 9), 14-DPI (N = 2, n = 11), 21-DPI (N = 2, n = 10); peritumoral regions defined by contralateral (dark blue), outer ridge (grey), infiltrated zone (green), tumor core (light blue). DPI Days post injection. Error bars show mean ± SEM. *P < 0.05, **P < 0.01, ***P < 0.001. Statistics: d, g, one-way ANOVA with Sidak’s multiple comparisons test. Source data are provided as a Source Data file.
We sacrificed mice at 7-, 14-, and 21-DPI and examined the expression of neuronal cell marker NeuN, glial cell marker GFAP, and cell proliferation marker Ki67 (Fig. 4c and Supplementary Fig. 8b). Tumor volume as indicated by GFAP and Ki67 labelling, increased rapidly between 7- and 21-DPI (Fig. 4d). At 7-DPI, the NeuN+ population remained largely intact, with GFAP-positive cells developing around the Ki67+ region. From 14- to 21-DPI, NeuN+ cell intensity gradually decreased in the Ki67+ region, together with a strong increase in both GFAP and Ki67 intensity (Fig. 4e). Based on these histological hallmarks, we segmented the tumor-bearing cortex into 3 zones: the Ki67+ “tumor core”; the “infiltrated zone” with high GFAP intensity and occasional Ki67 infiltration; and the “outer ridge” with a dim GFAP reactive area and sparsely diffused tumor cells in the PTC (Fig. 4f). Notably, enhanced GFAP responses were also observed in the contralateral motor cortex (Supplementary Fig. 8d). Despite the gradual loss of neuronal cells in tumor core, no significant difference in the numbers of excitatory and inhibitory synapses during the course of tumor development were found (Fig. 4e and g). Thus, our mouse model captures the main pathological features of cortical high-grade glioma and reveals the specific spatiotemporal characteristics of peritumoral regions with an enhanced immune response in peritumoral and contralateral cortex. This well-timed tumor progression is therefore ideal for longitudinal examinations of neuronal properties within PTC.
Spatiotemporal effects of tumor growth on peritumoral neuronal activity of glioma mice
Next, we performed electro-corticographic activity (ECoG) recordings from the mouse motor cortex to monitor neuronal activity during tumor progression (Fig. 5a, see “Methods”). All glioma-bearing mice show seizure-like activity starting around 14-DPI, such as high power in the theta and gamma frequency bands, spike wave discharges (SWD), and interictal spiking (Fig. 5b, c). Behaviorally, tumor-bearing mice showed abnormal movements such as jaw clapping and tail suspension, corresponding to subclinical focal seizure activity (Supplementary Movie 1). In mice where larger tumors were grown after 21-DPI, we observed generalized seizures often leading to the death of these mice (Supplementary Fig. 9a and Supplementary Movie 2).
a Schematic representation of tumor induction and ECoG recording protocol. b Representative ECoG traces of glioma-bearing mice showing interictal spikes (top) and spike wave discharges (SWD, bottom). c Top panel: power spectral analysis of SWD (red) and baseline (blue) epochs from a tumor-bearing mouse. Bottom panel: numbers of SWD per 20 min at three timepoints, 7 DPI (P = 0.0375), 14 DPI (P = 0.0363), 21 DPI (P = 0.2712) (control mice, grey, N = 4, glioma mice, red, N = 13). d Experimental design with coronal mouse brain section showing tumor induction and electrophysiological recordings on 14 (light blue) and 21 DPI (dark blue). Lower panel: a representative biocytin-filled L2/3 PC. e Comparisons of the intrinsic properties of the L2/3 PCs from control, 14-and 21-DPI mice. Progressive changes were found in membrane potential (control vs glioma 21-DPI P = 0.0026; 14-DPI vs 21-DPI P = 0.0236) (control, grey, n = 49; 14-DPI, light blue, n = 47; 21-DPI, dark blue, n = 70) and action potential threshold (control vs glioma 21-DPI P = 0.007) (control, n = 47; 14-DPI, n = 28; 21-DPI, n = 67), but not for input resistance (control, n = 48; 14-DPI, n = 29; 21-DPI, n = 70). f Slower spike decay time for 21 DPI group (control vs glioma 21-DPI P = 0.0409; 14-DPI vs 21-DPI P = 0.0038) (control, n = 42; 14-DPI, n = 28; 21-DPI, n = 64). g Current injection-frequency plot showing increased firing frequency slope (P < 0.0001) and maximum firing frequency in 14-DPI mice vs control (P = 0.0143) and 21-DPI (P = 0.0385), but not in control vs 21-DPI mice (P = 0.7719) (control, grey, n = 42; 14-DPI, light blue, n = 27; 21-DPI, dark blue, n = 66). h Zonal segmentation of tumor-bearing cortex and an example PC recorded from the outer ridge. i Unique spatial features of resting membrane potential alterations in different peritumoral zones: tumor core (magenta, n = 9), infiltrated zone (yellow, n = 27), outer ridge (green, n = 24) and control (grey, n = 49) (control vs infiltrated zone P = 0.0015; control vs core P = 0.0027; outer ridge vs core P = 0.0263) j Spiking frequency in response to depolarizing current steps showed different slope for core versus other groups (P = 0.0168) but no difference in maximum firing frequency (core, n = 9; infiltrated zone, n = 28; outer ridge, n = 22; control, n = 43). ECoG electrocorticogram, DPI days post injection, PC pyramidal cell, SWD spike wave discharge. Error bars show mean ± SEM. *P < 0.05, **P < 0.01, ***P < 0.001. Statistics: (c), multiple unpaired t-tests with Welch correction per time point, e and i Kruskal–Wallis with Dunn’s multiple comparisons test (f). Ordinary one-way ANOVA with Tukey’s multiple comparisons test for f decay time, g max firing frequency, and j max firing frequency. g and j simple linear regression for firing frequency slope comparisons. Source data are provided as a Source Data file. Brain outlines from Allen Mouse Brain Atlas (atlas.brain-map.org).
To evaluate the development of neuronal deficits during the initial phase of seizure development, we recorded from the L2/3 PCs in the PTC of 14- and 21-DPI mouse (Fig. 5d). The resting membrane potential showed a gradual shift during tumor progression (Fig. 5e) and was significantly more depolarized in 21-DPI tumor-bearing mice, which is consistent with our observations from the L2/3 PCs of TAS patients (Fig. 1e). We observed a similar progressive change for the action potential threshold (Fig. 5e). In addition, a longer action potential decay time was found in 21-DPI mice (Fig. 5f), consistent with a similar trend found in TAS patients (Supplementary Fig. 2h). Other action potential properties did not differ among groups (Fig. 5e and Fig. 9b–d). Interestingly, a transient elevation of intrinsic excitability was uncovered in the 14-DPI group, as evidenced by higher firing frequency slopes and higher maximum firing frequencies (Fig. 5g). The PCs in the contra-tumoral hemisphere had normal membrane potential (Supplementary Fig. 10), suggesting restricted local neuronal alterations in response to tumor progression. No difference in intrinsic properties, firing frequency, and action potential properties were observed in the ipsi- and contralateral motor cortex of the control group (Supplementary Table 3). These findings indicate that neurons in PTC display progressive alterations of their intrinsic properties in response to tumor progression, potentially leading to the development of hyperexcitable networks in the PTC and driving seizure onset.
Previous studies in patients showed that seizures could initiate from different peritumoral regions42 (Fig. 5h, see also Fig. 4f). We therefore compared the intrinsic properties of the L2/3 PCs located in the tumor core, the infiltrated zone and the outer ridge of the PTC (Fig. 5h). The distance from tumor core linearly correlated with the impairments of membrane potential, in that the PCs in tumor core had the most depolarized membrane potential (Fig. 5i). Notably, a similar trend was observed for the current injection–spike frequency relationship, with the strongest correlation in the neurons recorded closest to the tumor core (Fig. 5j). The PCs in the infiltrated zone had a more depolarized action potential threshold (Supplementary Fig. 9f). Therefore, PCs located at different peritumoral regions display distinct intrinsic properties, likely to reflect complex neuron-tumor interactions during tumor progression. Taken together, using a clinically relevant glioma mouse model, we identified specific spatiotemporal developments of neuronal phenotypes in the PTC during the pre-clinical phase of epileptogenesis.
TAS-associated synaptic and morphological alterations of L2/3 pyramidal cells in the glioma mouse model
We observed enhanced synaptic activity with concurrent morphological differences in the L2/3 PCs of TAS patients, compared with the non-TAS patients (Fig. 2). To investigate the progression of these changes over time in the mouse model, we recorded sEPSCs from the L2/3 PCs of control, 14- and 21-DPI mice (Fig. 6a). We observed a progressive increase in sEPSC frequency following tumor induction (Fig. 6b), whereas the sEPSC amplitudes, rise and decay times were unaltered (Supplementary Fig. 11). Increased sEPSC activity was also observed in the PCs of the hemisphere contralateral to tumor, but not in control mice (Supplementary Fig. 11b, c). Consistent with the patient data, progressively increased spine counts were observed in all 4 dendritic compartments (Fig. 6c). The spine densities of 1st branches and basal dendrites were significantly higher in the 21-DPI group (Supplementary Fig. 11d). The dendritic arborizations were unaltered in 14-DPI and 21-DPI mice, except for a tendency towards increased intersections in proximal areas. Together, these findings indicate a time-dependent progression of synaptic and morphological alterations of the L2/3 PCs during tumor progression and suggest that these morphological alterations lead to, rather than being the consequence of, seizures.
a Example sEPSC traces of L2/3 PCs from control, 14-DPI, and 21-DPI mice. b Quantifications of sEPSC frequencies for control (grey, n = 30 cells), 14-DPI (light blue, n = 22 cells), and 21-DPI (dark blue, n = 62), cells show progressive change in sEPSC frequency (control vs 21-DPI P = 0.0073). c Left panel: confocal image of reconstructed L2/3 PC. Red boxes around dendritic regions indicate the analyzed segments. Middle panel: example images of dendritic spines from the four indicated dendritic regions. Right panel: quantification of dendritic spines per dendritic branch in control animals (grey, n = 28 cells), 14-DPI (light blue, n = 10), and 21-DPI cells (dark blue, n = 30 cells), revealing progressive increase in spine densities during tumor growth (1st branch control vs 21-DPI P = 0.0043). d Schematic drawing of experimental design on coronal mouse brain section. Sulfasalazine (SAS) was added to the bath solution during recording of mouse L2/3 pyramidal cells (PCs). The membrane potential of glioma mice PCs treated with SAS was no longer significantly depolarized (control vs glioma 21-DPI P = 0.0005; control vs 21-DPI + SAS P = 0.1784) (Glioma 21 DPI, dark green, n = 70 cells, glioma + SAS, cyan, n = 11; control, dark grey, n = 49) e Excitability was measured by sEPSC frequency and was found reduced in the mice treated with sulfasalazine, no significant difference was found with control mice (control vs 21-DPI P = 0.0098; control vs 21-DPI + SAS P = 0.999) (Glioma 21 DPI, dark green, n = 62 cells, glioma + SAS, cyan, n = 10; control, dark grey, n = 30). sEPSC: spontaneous excitatory post-synaptic current, DPI days post-injection, SAS sulfasalazine. Error bars show mean ± SEM. *P < 0.05, **P < 0.01, ***P < 0.001. Statistics: b and e Kruskall–Wallis test with Dunn’s multiple comparisons test. c Kruskal–Wallis test with Dunn’s multiple comparisons test per dendritic region. d one-way ANOVA with Sidak’s multiple comparisons. Source data are provided as a Source Data file. Brain outlines from Allen Mouse Brain Atlas (atlas.brain-map.org).
Sulfasalazine regulates neuronal functioning in L2/3 PCs
We explored the possibility of treating the synaptic aberrations as a potential therapeutic target for TAS. The FDA-approved drug sulfasalazine has been shown to inhibit the non-synaptic xCT glutamate transporter system and thereby to attenuate late phase seizures both in vitro and in vivo9,20,25. We analyzed the effects of sulfasalazine on the intrinsic properties of L2/3 PCs in control and 21-DPI glioma mice. Bath application of sulfasalazine rapidly reversed depolarized membrane potential deficits in the 21-DPI group (Fig. 6d). Other properties, such as action potential threshold or firing frequency, were not changed. The EPSC frequency was greatly reduced and returned to a similar level as the control group, when comparing sulfasalazine-treated group to the 21 DPI glioma group (Fig. 6e). These data further confirm that the aberrant synaptic transmission in the PTC could be a potential therapeutical target for treating TAS patients.
Pre-epileptic hyperactivity in peritumoral neurons predicts occurrence of post-surgical seizures
The consistent neuronal alternations found in the glioma patient cohort (Figs. 1 and 2), the glioma mouse model (Fig. 5e–g), and the computational simulation (Fig. 3) led us to postulate that patients without clinically identifiable seizures prior to surgery might have already developed the epileptogenic networks in the PTC. Although tumor surgery often diminishes seizure activity43, patients with undetected and unresected epileptogenic cortex are likely to develop post-surgical seizure despite surgery. Indeed, within our patient cohort we identified a group of patients (N = 14) who were seizure free before and during surgery, but developed seizures starting from 1 to 15 months after surgery (Fig. 7a). As this delayed onset seizures (DOS) group initially did not display any features of clinically detectable epilepsy, they were not treated with anti-epileptic drugs prior to surgery. Intrinsic neuronal properties were measured from 9 DOS patients. Except for the moment of onset of the seizures, the DOS group was comparable to the TAS group in that it also concerned mainly high-grade gliomas, located at comparable brain regions and showing similar histopathological characteristics (Supplementary Table 4).
a Schematic overview of the seizure onset timing in non-TAS, delayed onset seizure, and TAS patient groups, in relation to the timing of tissue resection and electrophysiological recordings. b Example traces of sEPSCs in three patient groups. c sEPSCs frequency and amplitude of DOS group (light blue, n = 57) fall in between seizure (red, n = 88) and no seizure groups (grey, n = 46) (no seizures vs seizures P = 0.0045; control vs. DOS P = 0.46). d Example traces of paroxysmal depolarizing shifts (PDS) complexes from 3 DOS patients (patient 1–3). Note the long-lasting PDS clusters in patient 3. e Total numbers of PDS complexes recorded per 5 min in non-TAS (grey, n = 49 cells), DOS patients (blue, n = 25 cells), and TAS patients (red, n = 98 cells) (no seizures vs DOS P = 0.0031, no seizures vs seizures P = 0.0689). f Left panel: percentage of total neurons presenting PDS complexes per patient group. Yellow = PDS, white = no PDS, NS = no seizure (grey, 8/49), DOS = delayed onset seizures (blue, 12/25), and S = seizure patients (red, 36/98) (NS vs DOS P = 0.0170; NS vs S P = 0.0255). Right panel: percentage of patients per group with at least one neuron presenting PDS complexes (NS vs DOS P = 0.0029; DOS vs S P = 0.0476). g Cumulative proportion of the occurrence of post-surgical seizure in patients without pre-surgical seizures (DOS), as a function of with and without PDS complexes. Patients with PDS complexes in their neurons (N = 20 patients) develop seizures more often than the patients without PDS (N = 12 patients) (P = 0.0027, hazard ratio 5.05, Mantel–Haenszel test), yellow line = PDS, blue line = no PDS. TAS tumor-associated seizures, DOS delayed onset seizures, PDS paroxysmal depolarizing shift. Error bars show mean ± SEM. *P < 0.05, **P < 0.01, ***P < 0.001. Statistics: c, e, Kruskal–Wallis with Dunn’s multiple comparisons test, two-sided Fisher’s exact test with Holm–Šídák correction for multiple comparisons, g Log-rank (Mantel–Cox) with Mantel–Haenszel hazart ratio test. Source data are provided as a Source Data file.
L2/3 PCs from DOS patients had intrinsic property values in between those of the TAS and non-TAS groups. The resting membrane potential of DOS neurons was comparable with the non-TAS group (Supplementary Fig. 12a), whereas the action potential threshold, as well as the action potential decay slope and decay time, were significantly different from the non-TAS group (Supplementary Fig. 12b–e). On the other hand, the input resistance of DOS PCs was significantly larger than that of the TAS group, but not different from the non-TAS group (DOS vs. non-TAS p = 0.42, Supplementary Fig. 12b). The other intrinsic excitability parameters, including rheobase, firing frequency slopes, and maximal firing frequency, were comparable in all three groups (Supplementary Fig. 12b, c). The frequency and amplitude of sEPSCs of DOS PCs were both similar to those of the non-TAS neurons (Fig. 7b, c). Moreover, morphological parameters, including spine density and dendritic arborization of the PCs, were also comparable among DOS and non-TAS patients (Supplementary Fig. 12f, g). Taken together, these measurements revealed specific differences in neuronal properties among DOS, non-TAS, and TAS groups, with altered intrinsic properties in the DOS group likely to be an indication of a pre-clinical phase before seizure onset.
Identification of abnormal neuronal features specific for the early phase of seizure progression could be an important biomarker for diagnosis and treatment of TAS. Consistent with the prediction of our circuit model, we observed a high incidence of paroxysmal depolarizing shifts (PDS) in the L2/3 PCs of DOS patients. These complex discharges appeared as depolarizing shifts containing small excitatory peaks within larger depolarizing events, and as episodes of rapidly recurring large EPSCs without returning to normal baseline activity for several hundreds of milliseconds (Fig. 7d). Frequencies of PDS complexes varied greatly among neurons, on average 4.4 PDS per min, with a max of 19 per min in a patient with DOS, in contrast to <1 per min in TAS and non TAS patients (Fig. 7e). At the population level, the overall percentage of neurons displaying PDS activity was significantly higher in the DOS group, compared to both the non-TAS and TAS groups (48.0 vs. 16.3% and 37%; Fig. 7f). Interestingly, 100% of patients from the DOS group had at least one cell displaying PDS like activities, much more frequent than the incidences from the TAS and the non-TAS patients (62.2 and 36.8% respectively) (Fig. 7f). The frequency of PDS complexes was also uncorrelated with other intrinsic properties, such as membrane potential and firing frequency (Supplementary Fig. 12h, i).
The occurrence of PDS complexes appeared to be a strong indicator of post-surgical seizure onset. Patients who presented with frequent PDS complexes in their peritumoral neurons (N = 20 patients) turned out to develop post-operative seizures 5 times more likely, compared to the patients without PDS complexes (N = 12 patients, Fig. 7g). Therefore, peri-surgical electrophysiological and morphological analysis of peritumoral neurons could have implications for the decision-making for anti-epileptic treatment in brain tumor patients.
To mechanistically determine whether the PDS complex is related to enhanced synaptic connectivity and synaptic transmission (see Figs. 2 and 3), we constructed connected pairs of L2/3 PCs using computational simulation (Fig. 8). The neural activity of the pre-synaptic neuron was driven by 40 Hz Poisson distribution inputs to 20 synapses randomly distributed across dendrites (Fig. 8a–c). We varied the numbers and distribution of synaptic connections from the pre- to post-synaptic neurons. Large paroxysmal depolarizing shifts (PDS) were observed when we increased the numbers of synaptic connections to 12 (Fig. 8d, e). Our numeric simulations thus clearly indicate that the human L2/3 PCs are highly sensitive to synapto-dendritic alterations. Tumor-related increase in synaptic inputs is like to induce pronounced paroxysmal depolarizing shifts in peritumoral circuits.
a Current is injected in a simulated neuron, when the injected current is increased, the neuron displays depolarizing complexes. b A connected pair of L2/3 pyramidal cells. The pre-synaptic neuron was injected with current, number of synapses on the post-synaptic neuron was kept at 8. c Different dendritic distribution on the post-synaptic neuron leads to area-specific response. Increasing input current leads to depolarizing complexes in all dendritic areas; 1st branch (grey), second branch (green), apical dendrite (yellow), basal dendrite (magenta). d Connected L2/3 PCs where the pre-synaptic neuron is driven by 40 Hz Poisson distribution inputs to 20 synapses randomly distributed across dendrites. e The numbers and distribution of synaptic connections from the pre- to post-synaptic neurons were varied. When synaptic number increased, large paroxysmal depolarizing shifts (PDS) were observed. Each simulation described was run 10 times, duration of simulation was 1000 ms. Source data are provided as a Source Data file.
Discussion
TAS are a difficult to treat and detrimental complication of brain tumors with significant impact on the quality of life3,44. In this study, we examined the neuronal properties in the PTC prior to and during TAS development. Comparisons between TAS and non-TAS patients uncovered several intrinsic and synaptic properties of peritumoral neurons that correlate with the occurrence of TAS (Figs. 1–3). Leveraging a highly reproducible glioma mouse model, we characterized the unique spatiotemporal features of these alterations at the early phases of TAS onset (Figs. 4–6). Using computational simulations and longitudinal analyses during course of treatment, we show that pyramidal cells in PTC display PDS complexes. We demonstrate that PDSs were detectable prior to seizure onset in a subset of patients and reliably predicted post-resection seizure occurrence (Figs. 7 and 8). These findings form newly identified neuronal substrates of TAS and make PDS complexes a potential biomarker for seizure risk, offering a foundation for targeted diagnostic and therapeutic strategies.
Our results suggest that intrinsic and synaptic properties of peritumoral neurons are altered in patients with TAS (Fig. 1). The depolarization of the resting membrane potential in L2/3 PCs is consistent with findings from primary epilepsy research45,46,47. Additionally, the difference in sag current in TAS patients can also affect the intrinsic excitability and lead to seizure progression48,49. Alternatively, altered input resistance may reflect a circuit adaptation to a hyperexcitable microenvironment50. Indeed, lowered input resistance of hippocampal granule cells contributes to the attenuation of epileptogenesis in temporal lobe epilepsy51. These altered intrinsic and synaptic properties could be the results of either intracellular mechanisms, including changes in channel conductance31 and ion homeostasis30 or extracellular factors like enhanced synaptic inputs and extracellular glutamate concentration9,10. Given that the tissues resected from the PTC were perfused with ACSF during the recordings, which minimizes extracellular impact, our results are more likely to reveal the intracellular mechanisms that lead to changes in intrinsic properties of TAS neurons. Using single-cell RNAseq, we identified a large group of genes that are differentiated expressed between the TAS and non-TAS groups. Interestingly, many of these genes are closely related to the regulations of intrinsic and synaptic neuronal functions. Therefore, our molecular analysis also supports the possibility that intracellular mechanisms are the major determinant for seizure progression. Using single-cell RNA sequencing, we identified a large set of genes that are differentially expressed between the TAS and non-TAS groups. Notably, many of these genes are closely associated with the regulation of intrinsic neuronal properties and synaptic function. These molecular findings further support the hypothesis that intracellular mechanisms may play a central role in driving seizure progression.
Apart from altered intrinsic properties, L2/3 PCs of TAS neurons display increased EPSC frequency as well as an increased dendritic spine density (Fig. 2). We speculate that enhanced synaptic inputs and local circuit rearrangements also contribute to ictogenesis52. As specific mutation variants in glioblastoma increase the expression of synaptogenic factors that drive hyperexcitability of the network, the genetic composition of the tumor can be expected to contribute to this increase in synaptic inputs53. Disinhibition of the peritumoral cortical network before the onset of inter-ictal like discharges due to impaired interneuron functioning is probably another important factor in TAS pathophysiology30. Although we did not observe a change in IPSC frequency and amplitude, the limited sample size may have constrained the ability to detect subtle effects. Our analysis of L2/3 interneurons revealed a more depolarized resting membrane potential, which could promote hyperexcitability of interneurons and thereby facilitate inter-ictal like discharges in PCs. Whether and how alterations in inhibitory inputs contribute to the neurological changes in glioma patients require further investigation in the future.
Do these alterations in neuronal properties represent the underlying cause of seizures, or are they a consequence thereof? Longitudinal assessments in our glioma mouse model, coupled with findings from patients exhibiting delayed seizure onset, collectively suggest that distinct neurophysiological changes in the PTC precede the emergence of clinically manifest seizures. DOS neurons have action potential properties different from both TAS and non-TAS groups, indicating a distinct change in intrinsic neuronal function that could prime ictogenesis. This notion is supported by the gradual change in neuron density during tumor progression, with intact neuronal populations at 7 and 14 DPI and the observed transient elevation of intrinsic excitability in the 14-DPI, but not the 21-DPI, group of glioma mice. Consistently, neurons from DOS patients had a higher incidence of PDS complexes than both the TAS and non-TAS groups. The observed lower PDS complex occurrence in the TAS group is possibly the effect of long-term treatment of these patients with AEDs, whereas DOS patients received no AED treatment at time of recording. Studies of kindling-induced epilepsy have also confirmed the occurrence of PDS complexes as a cellular correlate for inter-ictal activity in developing epileptogenic networks54,55,56. As PDS complexes can arise from dendritic layers57 and induce long-term morphological and functional synaptic changes58 as well as activity-dependent gene expression59, it is likely that PDS complexes are an indication of and/or promote development of seizure-prone micro-circuits in the PTC.
Our study is complementary to previous studies revealing that elevated extracellular glutamate secreted by tumor through the cystine–glutamate transporter (xCT) system promotes seizures, tumor proliferation, and excitotoxicity9,10. Peritumoral neurons from seizure-prone tissue, but not control tissue, are hyperexcitable in the presence of elevated glutamate levels10. This confirms a contribution of altered intrinsic neuronal properties to the peritumoral hyperexcitability in response to elevated glutamate concentration. We propose that enhanced extracellular glutamate and changes in intrinsic properties work in tandem during TAS progression. Glutamate receptor signaling mediates a plethora of plastic changes in the intrinsic and synaptic properties of neurons60. In turn, hyperactive neuronal circuits could directly drive tumor growth via synaptic and non-synaptic pathways16,17,20,61 and thereby form a vicious cycle to accelerate disease progression. Intriguingly, sulfasalazine appears to be effective in both reducing the xCT-dependent glutamate release9,17,22 and correcting the deficits in neuronal properties (Fig. 6). These findings are in line with other recent work demonstrating synaptogenic and neurotrophic cellular mechanisms that promote tumor-neuron interaction13. Transcriptome analysis of the PTC from TAS patients uncovered upregulation of genes involved in glutamatergic transmission and cell-to-cell signaling62. Suppressing neuronal activity and thereby neuron-tumor communication by the AMPA receptor antagonist Perampanel also suppresses tumor proliferation16,17,61. Simultaneously, the occurrence of PDS complexes potentially depends on AMPA receptor functioning, which indicates an intriguing option to treat patients in the pre-epileptic phase and assess the effects on both epileptogenesis and tumor progression. Although the clinical benefit is currently unclear9, therapeutics targeting the cellular mechanisms that affect both tumor progression and epileptogenesis could have the advantage of clinical outcome combined with lower adverse effects63.
The microenvironment in the PTC, in which neurons, glial cells, and tumor cells interact, could be another key factor for epileptogenesis. We identified specific spatial and temporal development of neuronal phenotypes in an immuno-competent glioma mouse model, which can be considered a model for the pre-clinical phase of tumor-associated epileptogenesis. Combining transgenic-viral injection strategies, highly reproducible tumor growth is restricted to specific cortical and subcortical regions in the immunocompetent hosts. We observed a progressive glial reaction around an invading tumor mass (Fig. 4), indicating a strong tumor-immune interaction64, as well as a dynamic change in the neuronal electrophysiological properties during disease progression41,65,66 (Fig. 5). Tumor-associated macrophages regulate the immune response in the tumor microenvironment and their activity is correlated with tumor malignancy and prognosis40,67. Inversely, inflammatory processes impact ictogenesis and glutamate management in cortex considerably68, which might also play a role in the observed effect of sulfasalazine on neuronal and cortical functioning. Therefore, our findings should be interpreted in the light of this strong effect of peritumoral microenvironment on neuronal functioning and excitability.
Recent studies have identified previously unrecognized alterations in the transcriptomic landscape of glioma cells, suggesting that these changes may contribute to hyperexcitability35,69,70. A subset of tumor cells displays distinct neuronal-like properties, characterized by the expression of canonical neuronal genes and excitability in response to current injection. These cells are typically interspersed with other tumor cells, particularly at the infiltrative margin36. Consistent with these findings, a recent single-nucleus RNAseq analysis of peritumoral cortical tissue supports the presence of complex neuronal features in glioma cells70. Collectively, these observations highlight a glioma-induced disruption of cell identities and suggest the emergence of aberrant neuronal-like states that may contribute to both tumor progression and neurological dysfunction. In the current study, we utilized single-cell RNAseq to investigate neuronal alterations associated with seizure development. The neurons captured in our dataset exhibited hallmark features of cortical neurons, including pyramidal morphology and canonical neuronal transcriptomic signatures, making it unlikely that they are neuronal-like glioma cells. This interpretation is further supported by the findings of Goldberg et al., demonstrating that glioma progression induces morphological changes and neuronal hyperexcitability24. Nevertheless, due to the limited yield of the patch-seq technique, we cannot entirely exclude the possibility that some neuronal-like glioma cells share features with peritumoral cortical neurons and may actively contribute to TAS progression. Future studies using high-throughput approaches will be essential to further establish causal relationships between specific cell types and TAS.
The clinical ramifications of TAS are multi-faceted. Seizure development in the post-operative phase is often associated with poor local tumor control and progression71,72. Early identification of subclinical and/or clinical epileptiform activity prior to overt seizure manifestation may offer a critical window for timely and potentially more aggressive therapeutic interventions. Effective seizure management is integral to improving patient quality of life and may influence overall survival rates. Studies have demonstrated that patients experiencing perioperative seizures often face longer hospital stays, higher readmission rates, and reduced survival, underscoring the importance of proactive seizure control in comprehensive cancer care73. Therefore, addressing the aberrant neuronal functions might just be as important as directly treating the tumor growth. Specific understanding of pathophysiologic mechanisms of TAS will also guide the optimization of treatment, selecting or developing more effective antiepileptic drugs for tumor patients. By addressing seizure control as an integral part of the treatment plan, clinicians can improve the patient’s overall condition, making them more resilient to undergo other therapies.
Moving forward, we advocate the application of perioperative electrophysiological recordings as a valuable preclinical and clinical tool for personalized diagnosis in brain tumor patients. The presence of PDS complexes in patients without preoperative seizures is highly predictive for their post-operative seizure development (Fig. 7), which could also be considered a therapeutic target for TAS prevention and these findings warrant need for multi-center validation. Notably, the emergence of PDS complexes and subsequent postoperative seizure development occurred independent of the extent of tumor resection. In combination with other biomarkers for TAS74,75,76, perioperative in-vivo and ex-vivo electrophysiological recordings provide a set of tools to identify specific clinical and subclinical alterations in individual brain tumor patients. Altogether, this work adds new insights into neuronal adaptation in seizure-prone PTC and provides strategies to detect and treat neuronal dysfunction in TAS patients. Within the rapidly evolving domain of cancer neuroscience, we anticipate that future investigations into the intricate dynamics of neuron–tumor interactions within the PTC will facilitate earlier detection of seizure onset, enable more precise diagnostic stratification, and inform the development of personalized therapeutic strategies for patients with brain tumors.
Methods
Patient cohort
All experimental procedures were performed under the approval of the Medical Ethical Committee of the Erasmus University Medical Center. Written informed consent was obtained prior to surgery in accordance with the Declaration of Helsinki. Surgical tissue samples were prospectively obtained from patients undergoing brain tumor surgery at the Department of Neurosurgery (Erasmus University Medical Centre Rotterdam, the Netherlands). The infiltrated peri-tumoral neocortical specimens used for this study were removed during a regular tumor resection (procedures involving tissue samples collection under protocol number MEC-2013-090). Never was resection of healthy cortex performed for research purposes. For this study, only patients who underwent first time surgery were included, patients with re-resections and patients treated previously with radiotherapy or chemotherapy were excluded. All specimens were taken out at the discretion of the neurosurgeons to ensure safe removal and intact cortex for electrophysiological experiments. Specimens were carefully taken out using scalpel cuts perpendicular to the pial surface to maintain cortical integrity across all layers with white matter present for orientation during processing. Cauterization was omitted prior to tissue resection to avoid excitotoxicity to cortical neurons.
All patients underwent preoperative MRI scans using a 3 Tesla (Discovery MR750, GE, Waukesha, WI, USA) whole-body scanner, equipped with a 32-channel head coil. For all patients, the locations of resected tissue were documented using the neuro-navigation images (BrainLab Origin software, Brainlab AG, Germany). On neuronavigation MRI, all specimens were taken <1 cm of the T1 Gadolinium enhancing tumor limits analyzed post-hoc with neuro-navigation images. Brain samples used for electrophysiology experiments were transported to laboratory within 10 min after resection. Remaining tissue was snap frozen in liquid nitrogen and stored as frozen sample at −80 °C or fixated with 4% paraformaldehyde for further histological analysis.
A unique number was given to each resected specimens and patients’ clinical information corresponding to this number was anonymously stored in a secured database within hospital. Patients (N = 70) were prospectively followed in our institution for the duration of the study. Patient demographic information was collected including age (range 25–86 years), sex (42 males [60%], 28 females), location and type of tumor, nature and duration of presenting symptoms, clinical and neurological symptoms, epilepsy status, medication, KPS and MMSE scores, pathology report and molecular analysis of tumor tissue and radiologic assessment of tumor specific traits. Post-surgical events were recorded, including (re-)occurrence of epilepsy, type and frequency of seizures, postsurgical adjuvant treatment, and survival. See Supplementary Table 2 for patient characteristics reported in accordance with the BRISQ requirements77. Sample sizes for experiments with human tissue are indicated in the figure legend of each experiment.
Tumor-associated seizures classification
The seizure status of each patient was determined by in-house neurophysiologists and neurosurgeons. Requirements for classification as “seizure free” included no documented seizure prior to surgery and no seizure at any time point during regular follow up after surgery for at least 2 years or till death. The seizure group consisted of patients with one or more seizures identified by either a neurologist or a neurosurgeon in the period before the surgery. Patients that had no seizure-like event prior to surgery but that developed seizures >4 weeks post-surgery were classified as the Delayed Onset Seizure (DOS) group. As post-surgical seizures could occur immediately after surgery, patients who presented with seizures immediately after surgery were excluded from this group. If seizures occurred within 3 weeks post-surgery, patients were only included in the DOS group if they displayed recurrent seizures. For all patients without seizures and those presenting with post-surgical seizures, detailed patient records and charts were reviewed by neuro-oncologists and neurologists to confirm the presence or absence of any symptoms related to epileptic seizures.
Surgical sample transport and processing
Resected neocortical specimens, typically cortical projecting samples, were immediately transferred to ice-cold oxygenated (95%O2/5%CO2) artificial cerebrospinal fluid (aCSF) containing (in mM): 124 NaCl, 24 NaHCO3, 22 D-glucose, 2.5 KCl, 1.25 NaH2PO4, 2 MgSO4, and 2 CaCl2. The time between surgical resection and tissue slicing was <10 min. Samples were trimmed to appropriate size before slicing. 350 μm slices were cut tangentially to the cortical surface on a vibratome (VT1200S, Leica) in oxygenated (95%O2/5% CO2) ice-cold N-methyl-d-glucamine (NMDG)-based slicing solution containing (in mM): 93 NMDG, 93 HCl, 25 D-glucose, 30 NaHCO3, 2.5 KCl, 0.5 CaCl2, 10 MgCl2, 20 HEPES, 1.25 NaH2PO4, 12 N-acetyl-cysteine, 2 thiourea, 5 Na-ascorbate, 3 Na-pyruvate (300 mOsm, pH 7.4). Slices were incubated in warm (34 °C) NDMG-based slicing solution for 5 min and subsequently transferred to a holding container in aCSF (34 °C) for 30 min. Slices were then allowed to recover at room temperature for at least 30 min before recordings and were used for recordings for up to 12 h.
Mouse model
Mice were housed and handled in compliance with institutional guidelines and Dutch legislation regarding animal experiments. All animal experiments were executed with approval of the animal ethics committee of the Erasmus MC. The animals used for ECoG and electrophysiology experiments were male and female mice between 9 and 14 weeks of age. For the generation of tumor-bearing animals, three conditional mice lines were intercrossed to generate the triple mutant mouse model carrying floxed Pten locus, floxed Ink4a/Arf locus, and lox-Stop-lox K-Rasv12 transgenes (Pten;Ink4a/Arf;K-Rasv12 mouse)31. Floxed Pten and floxed Ink4a/Arf were maintained as homozygous, whereas lox-Stop-lox K-Rasv12 was kept heterozygous. The generation and validation of this genetic model has been described previously27,78. Animals were housed individually in a temperature-controlled environment and were kept on a 12-h day/night cycle with food and water ad libitum.
Mouse surgery, viral vector injection, and ECoG recording
Mice were anesthetized using 2.5% isoflurane and oxygen and placed in a stereotactic frame (Harvard Apparatus, MA, USA). An incision was made, exposing the skull. For injection of the virus, a craniotomy was made above the primary motor cortex (1.0 mm anterior and 1.5 mm lateral to bregma) using a dental drill (Elcomed dental drill, W&H group, UK). Craniotomies of 0.3 mm in diameter were drilled 0.5 mm posterior and 0.5 mm lateral to bregma for the placement of two ECoG leads. A fourth craniotomy was drilled over the center of the cerebellar hemisphere for the placement of an ECoG reference electrode. 3 custom-made ECoG leads were fixated at these locations using dental composite Charisma (Kulzer, Mitsui chemicals, Japan). AAV viral vectors expressing cre-recombinase under the GFAP promoter (Addgene 105550-AAV5) was used for the induction of tumor growth. For control experiments an AAV1-CB7-GFP virus (Addgene 105542-AAV1) was used. The virus was loaded into borosilicate glass pipettes and virus injection was performed with the pipette mounted on a stereotactic manipulator. The pipette was moved into the cortex up until a depth of 1 mm from the cortical surface. 60–80 nL of virus was injected over a time period of 5–10 min and the pipette was kept in location for 10 min before retraction. The injection craniotomy was sealed with Charisma (Kulzer, Mitsui Chemicals, Japan). A head bar was mounted on top of the skull above lambda for head fixature during ECoG recordings.
Mice were left to recover for 3 days post-surgery and were habituated for at least 3 days on the ECoG recording setup prior to recording. Mice were recorded at day 7, 14, and 21 post-injection. All ECoG recordings were performed in head-restrained animals. Recordings were acquired using Intan RHD amplifier board and recorded using Intan software (Intan Technologies, California, USA). Video-recording was simultaneously made during ECoG recordings for a subset of mice. ECoG traces were analyzed using custom made scripts.
Mouse tissue preparation
Tumor-bearing mice were sacrificed at 14- or 21-days post-injection. Mice were sedated using isoflurane and decapitated. The brains were removed from the skull and transferred to ice-cold NMDG. A transverse cut was made at the level of lambda to create a flat surface for mounting and the brain was mounted onto the stage of the vibratome. 250 μm slices were cut tangentially to the cortical surface on a vibratome (VT1200S, Leica). Slicing was performed in ice-cold NMDG solution (see human tissue preparation section). Afterwards, slices were incubated in warm (34 °C) NDMG solution for 5 min and then transferred to warm aCSF for recovery for 30 min with subsequent recovery at room temperature for another 30 min.
Electrophysiological recordings of human and mouse tissue
Ex-vivo whole-cell patch-clamp recordings were made from the cortical slices of human and mice tissue specimens. Neurons were visualized by infrared differential interference contrast (IR-DIC) microscopy using a 40× water immersion objective (Fixed stage Axio Examiner, Carl Zeiss, Germany). Patching pipettes were made from borosilicate glass pipettes (World Precision Instruments, resistance range 3–6 mΩ), filled with intra-cellular solution containing (in mM): 140 mM K-gluconate, 5 mM Na2-phosphocreatine, 10 mM KCl, 0.1 mM BAPTA, 0.05 mM CaCl2, 4 mM MgATP, 0.4 mM Na3GTP, 10 mM HEPES, and 5 mg/mL biocytin (pH 7.2 with an osmolarity of 295 ± 5). For IPSC recordings, an internal solution (pH 7.3) containing: 150 mM CsCl, 15 mM CsOH, 1.5 mM MgCl2, 0.5 mM EGTA, 10 mM HEPES, 4 mM Na2ATP, and 0.4 mM Na3GTP were used. Recording was performed on slices submerged in aCSF at near-physiological temperatures (33–34 °C; perfusion rate of 10–14 mL/min) using HEKA EPC10 amplifiers and Patchmaster software (20 kHz sampling rates). Cells with series resistance \(\le\)20 mΩ were included. No correction was made for liquid junction potential. Data analysis was performed using custom-written code, Clampfit software (pClamp v11, Molecular Devices, CA, USA), and MiniAnalysis (Version 6.0.7, Synaptosoft Inc., GA, USA). Pipettes were kept in place for at least 30 min to ensure complete diffusion of biocytin throughout the dendrites and subsequently slowly retracted to reseal the neurons for immunohistological analysis.
Input resistance was determined from the peak current and steady state current in response to a negative voltage step of −10 mV. Passive and active membrane properties were determined using voltage responses to square-wave current pulses of 500 ms duration and 20 pA step size, ranging from −60 pA to +320 pA. The ratio of the maximum and steady-state voltage response after subtraction of resting membrane potential to a hyperpolarizing current injection of −60 pA was used to define the sag ratio. Single action potential (AP) properties were calculated from the first elicited action potential. AP threshold was defined as the point where the first derivative of the upward slope of the AP > 20 mV/ms. AP amplitude was defined as the difference between AP threshold and peak voltage. AP half-width was measured as the full width at 50% of the peak amplitude. AP rise and decay time was quantified as the duration from 10% to 90% and from 90% to 10% of the peak amplitude, respectively. The fast after-hyperpolarizing potential amplitude was defined as the difference between the AP threshold of the first AP and the maximum hyperpolarizing deflection directly following the AP down stroke.
Inclusion criteria of the specimens
As the use of human specimens naturally involves a certain degree of inter-patient variability, we implemented several strict criteria to ensure consistency across samples. Specimens were transferred directly from the operating room to the lab for electrophysiological experiments. All slices used in these experiments were first visually inspected under a microscope to confirm intact cortical layering; samples with disrupted cortical layers were excluded. Additionally, for each recorded neuron, we applied stringent criteria to assess cell membrane integrity and functionality before proceeding with recordings. In post-hoc analyses, intrinsic properties of all recorded neurons were evaluated, and those not meeting baseline criteria were excluded from further analysis. Neurons were also imaged using confocal microscopy to evaluate both cell and slice morphology for signs of tumor invasion. This included Ki67 staining and SMI32 staining to visualize neuronal layers.
Pharmacological application
Sulfasalazine (SAS; Sigma-Aldrich) was dissolved in 0.1 N NaOH solution to create a 10 mmol/L stock solution and was bath applied at 250 µmol/L. Slices treated with SAS were incubated for at least 30 min before whole-cell recordings started.
Patch-seq using Smart-seq2 protocol for human neurons
In a sub set of patient samples, we harvested pyramidal cells for modified patchSeq analysis34 using the protocol for Smart-seq233. For optimal RNA quality, we added 1 U/μl recombinant RNase inhibitor (Takara Cat.no. 2313 A) to the intracellular solution. From sliced cortical samples, cell contents were harvested and processed according to the methods as described34. Next, single-cell mRNA was converted to cDNA and sequencing libraries were created. DNA libraries for sequencing were prepared from cDNA libraries with the Illumina Nextera XT kit. The libraries were sequenced on an Illumina HiSeq2500 sequencer. Reads were generated of 50 base-pairs in length. Sequence adapters and poly-A were removed from the reads prior to alignment. The trimmed reads were aligned to the human GRCh38 reference genome using the HISAT2 aligner79. Gene quantification was performed using the htseq-count software. Statistic testing was performed using the DEseq280 with the ashr fold change shrinkage estimator81.
Immunohistochemistry analysis
For the reconstruction of human and mouse neurons after electrophysiology recordings, slices were fixated overnight in 4% paraformaldehyde (PFA). The following primary antibodies were used for immunohistochemical staining: anti-NeuN (Millipore/Mouse; 1:2000), anti-SMI-32 (Sternberger/Mouse; 1:5000), anti-GFAP (Abcam/Chicken; 1:2000), anti-Ki67 (Santa Cruz/Rabbit; 1:2000). Following primary antibody incubation, slices were rinsed with 0.1 M phosphate buffer and incubated for two hours at room temperature in secondary antibodies. Secondary antibodies used: Cy3 Streptavidin (Jackson, 1:400), anti-mouse Cy5 (Jackson/Donkey; 1:400), anti-rabbit Alexa Fluor 488 (Jackson/Donkey; 1:400), anti-chicken Alexa Fluor 405 (Jackson/Donkey; 1:200).
For tumor histology, 4 mm paraffin-embedded sections were made from human and mouse slices. For human tissue, resected samples were kept in 4% PFA overnight and subsequently embedded in paraffin. For mouse brain tissue, animals were trans-cardiac perfused with PBS and followed by 4% PFA. Hematoxylin and Eosin (HE) staining and immunohistochemical analysis of mouse and human tissues were performed and examined by pathologists according to the diagnostic standards of the Department of Pathology of the Erasmus Medical Centre.
Imaging and image processing
Confocal imaging was performed using a Zeiss LSM 700 microscope (Carl Zeiss, Germany) using ZEISS ZEN imaging software (Carl Zeiss, Germany). For illustration of different aspects in tumor tissue, single planes and maximum intensity projections were used. For representative animal histological images, example images were taken from individual animals. Stainings were repeated at least 2 times. For reconstruction of neurons for morphological analysis Z-stack images of complete neurons were taken and converted to maximum intensity projections (MIP). Imaging of light microscopy sections was performed on a NanoZoomer 2.0 HT digital slide scanner (Hamamatsu, Japan).
For the morphological analysis of neurons, images were prepared for analysis in FIJI ImageJ (Rasband, W.S., ImageJ, NIH). Dendritic arborization was analyzed using the ImageJ plugin for Sholl analysis, placing concentric rings starting from the cell soma. Dendritic spine analysis was performed using ImageJ82,83, spines were quantified across 20 µm sections for human and 15 µm sections for mouse neurons. Sholl analysis and dendritic spine density counts were analyzed by two independent experimenters. Experimenters were kept blind for experimental groups.
Quantification of the distribution of fluorescence intensity of the different cell populations was performed using FIJI ImageJ (Rasband, W.S., ImageJ, NIH). For each experimental group the mean pixel fluorescent intensity was calculated across the tumor hemisphere. A lateral-medial box, 2 mm in width and 0.5 mm in height, was manually placed over the tumor and peritumoral area covering the area of interest. Using GraphPad Prism (GraphPad Software, USA), a 2nd order smoothing was applied with 200 nearest neighbors prior to the normalization of each channel84. Experiments were replicated within animals and between animals. To estimate the excitatory synaptic densities, 40 µm sections from 7-DPI (N = 2), 14-DPI (N = 2), and 21-DPI (N = 2) mice were stained for synaptic marker VGlut1/2 and scanned with confocal imaging over a depth of 5 µm (12 sections at 0.35 µm) at 40× magnification. The ImageJ plugin Synapse Counter was used for quantification of synapses85. Illustration of brain sections were recreated by authors based on histological images from the Allen Brain Atlas (https://mouse.brain-map.org/static/atlas).
Computational model
We established a human L2/3 cortical neuron model with detailed neuronal morphology to explore how cortical neurons adapt to changes in synaptic distribution. The model represents multiple compartments for the membrane potential \(V\) using a set of nonlinear differential equations that account for ionic currents and leakage currents across the neuronal membrane. The dynamics of the membrane potential \({V}_{i}\) for the compartment \(i\) are given by:
where \({C}_{m,i}\) represents the membrane capacitance of compartment \(i\), \({I}_{{ion},i}\) denotes the ionic current in compartment \(i\), and \({I}_{L,i}\) corresponds to the leakage current in compartment \(i\). The term \({I}_{{syn},i}\) accounts for the synaptic current applied to compartment \(i\), while \({g}_{{ij}}\) represents the conductance between compartments \(i\) and \(j\). Finally, \({V}_{j}\) denotes the membrane potential of compartment \(j\). The human L2/3 neuron morphology was established from Gidon et al.37, consisting of a somatic compartment, 101 basal dendritic sections, 81 apical tree sections, and an axonal cable. For each section compartment lengths were at most \(30 \, \mu m\), Membrane capacitance and axial resistance (\({R}_{i}\)) were uniformly set to \(0.45\mu F{{cm}}^{2}\) and \(100\varOmega \times {cm}\), respectively, over the entire dendrite86. These values resulted in somatic input resistance (\({R}_{{in}}\)) and membrane time constant (\({\tau }_{m}\)) of 39 MΩ and 17 ms, respectively. Membrane resistance \({R}_{m}\) was \(37M\varOmega \times {{cm}}^{2}\) in all compartments.
Ion channels are essential components of the neuronal membrane, playing a crucial role in generating and propagating electrical signals within neurons. The ionic current is described by:
where \({g}_{{ion}}\) represents the maximum ion conductance, \(m\) is the activation gating variable, and \(h\) is the inactivation gating variable. The exponents \(n\) and \(k\) denote the number of activation and inactivation gates, respectively. \(V\) is the membrane potential, and \({E}_{{ion}}\) is the ion reversal potential. The gating variables \(m\) and \(h\) follow standard first-order kinetics with voltage-dependent rate constants:
where \({\alpha }_{m,h}\) and \({\beta }_{m,h}\) are the rate constants governing ion channel dynamics. In addition to voltage-gated ion channels, the neuronal membrane also contains leakage ion channels, which allow passive ion flow and contribute to the resting membrane potential. The leakage current is given by:
where \({g}_{L}\) represents the leakage conductance, \(V\) is the membrane potential, and \({E}_{L}\) is the leakage reversal potential. The soma was modeled with sodium (\({I}_{{Na}}\)), potassium delayed rectifier (\({I}_{{Kdr}}\)) currents, with the corresponding maximal conductances, \({g}_{{Na}}=0.1S/{{cm}}^{2}\), \({g}_{K}=0.045S/{{cm}}^{2}\) and reversal potentials, \({E}_{K}=-80{mV},{E}_{{Na}}=90{mV}\). The rate functions for the sodium and potassium channels were taken from a previous study87. In addition, we also added L-type calcium with high-threshold of activation (\({I}_{{calh}}\))88 and slow sodium inactivation (\({I}_{{namr}}\))89 currents in all compartments (soma and dendrites), with the corresponding maximal conductances, \({g}_{{calh}}=0.0001S/{{cm}}^{2}\), \({g}_{{namr}}=0.001S/{{cm}}^{2}\) and reversal potentials, \({E}_{{calh}}=140{mV},{E}_{{namr}}= 50{mV}\). A hyperpolarization-activated cation current (\({I}_{h}\)) is added in all compartments (soma, dendrite) and reversal potentials \({E}_{h}=-25{mv}\), and implemented assuming a density gradient in the dendrite that reached its maximum \(500\mu m\) from the soma. We implemented a gradient of 1:7 based on previous physiological studies of \({I}_{h}\) in hippocampal CA1 pyramidal neurons90.
For synaptic input dynamics, two types of excitatory synapses, AMPA and NMDA, were incorporated into the neuronal model. The EPSC of each synapse is described by:
where \({I}_{{syn}}\) represents the receptor-mediated synaptic current, \({g}_{{syn}}\) is the synaptic conductance, \(V\) is the membrane potential, and \({E}_{{syn}}\) is the synaptic reversal potential. \({g}_{\max }\) denotes the maximum synaptic conductance, while \(w\) represents the synaptic weight. NMDA receptor-mediated currents exhibit voltage-dependent properties, as they require prior AMPA receptor activation to depolarize the membrane and remove the extracellular \({{Mg}}^{2+}\) blockade from NMDA-associated channels. These channels open only after the displacement of magnesium ions, allowing NMDA receptors to be activated in parallel. The scaling factor \(Y\) is set to 1 for AMPA receptors, whereas for NMDA receptors, it follows a nonlinear voltage-dependent function: \(Y=1/(1+\exp (-0.077*V)*0.28)\). The synaptic gating variable \(S(t)\) follows double-exponential kinetics and is defined as:
where \({\tau }_{{rise}}\) and \({\tau }_{{decay}}\) are the rise and decay time constants, respectively, and \({t}_{{spike}}\) represents the timing of the presynaptic spike. The rise time constant (\({\tau }_{{rise}}\)) was set to \(0.3{ms}\) for AMPA receptors and \(8{ms}\) for NMDA receptors, while the decay time constant (\({\tau }_{{decay}}\)) was set to \(1.8{{\rm{ms}}}\) for AMPA and \(35{{\rm{ms}}}\) for NMDA37, where the AMPA maximum conductance was set to \(7{nS}\) and NMDA maximum conductance is set to \(1.3{nS}\), resulting an amplitude of sEPSC around \(0.5{pA}\), matching experimental values of the recorded cells.
Statistical analysis and reproducibility
GraphPad Prism (version 10, GraphPad Software) was used for statistical analysis. To test for assumption of normality, Shapiro–Wilk’s W test was performed for each dataset. For normally distributed data, differences were analyzed by either unpaired Student’s t-test or one-way ANOVA, depending on the number of groups tested, with Sidak’s tests for multiple comparisons performed afterward. For non-parametric data, group differences were tested using the Mann–Whitney U test or Kruskal–Wallis test for more than two groups followed by Dunn’s post-hoc multiple comparisons tests. For repeated measurement data, significant differences between groups for slope and intercept of the best-fit line were determined using (non-)linear regression. Data are presented as mean ± SEM. p-values < 0.05 were considered significant.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
Source data are provided with this paper. The RNAseq dataset is available from the European Genome-Phenome Archive (EGA) under accession code EGAD50000001979. Access to data deposited to the EGA is controlled to comply with the General Data Protection Regulation and patient consent. To request access, a formal application must be submitted to the Data Access Committee (DAC) that is associated with the respective dataset(s) through the EGA. Further information can be found at https://ega-archive.org. The accession of files is limited to research purposes and regulated with a data use agreement. The timeframe for response to requests is as promptly as possible (expected within 30 working days), pending DAC approval. Source data are provided with this paper.
Code availability
The modelling and analysis codes are available online: https://github.com/ydtang/Seizure-modelhttps://github.com/kthvdk/GaoLabGlioma/branches.
References
Kerkhof, M. & Vecht, C. J. Seizure characteristics and prognostic factors of gliomas. Epilepsia 54, 12–17 (2013).
Gallagher, P., Leach, J. P. & Grant, R. Time to focus on brain tumor-related epilepsy trials. Neurooncol. Pr. 1, 123–133 (2014).
van Breemen, M. S., Wilms, E. B. & Vecht, C. J. Epilepsy in patients with brain tumours: epidemiology, mechanisms, and management. Lancet Neurol. 6, 421–430 (2007).
Taphoorn, M. J. Neurocognitive sequelae in the treatment of low-grade gliomas. Semin. Oncol. 30, 45–48 (2003).
van Breemen, M. S. et al. Efficacy of anti-epileptic drugs in patients with gliomas and seizures. J. Neurol. 256, 1519–1526 (2009).
Tantillo, E. et al. Molecular changes underlying decay of sensory responses and enhanced seizure propensity in peritumoral neurons. Neuro Oncol. 25, 1463–1473 (2023).
Senner, V. et al. A new neurophysiological/neuropathological ex vivo model localizes the origin of glioma-associated epileptogenesis in the invasion area. Acta Neuropathol. 107, 1–7 (2004).
Kohling, R., Senner, V., Paulus, W. & Speckmann, E. J. Epileptiform activity preferentially arises outside tumor invasion zone in glioma xenotransplants. Neurobiol. Dis. 22, 64–75 (2006).
Robert, S. M. et al. SLC7A11 expression is associated with seizures and predicts poor survival in patients with malignant glioma. Sci. Transl. Med. 7, 289ra286 (2015).
Buckingham, S. C. et al. Glutamate release by primary brain tumors induces epileptic activity. Nat. Med. 17, 1269–1274 (2011).
Ye, Z. C., Rothstein, J. D. & Sontheimer, H. Compromised glutamate transport in human glioma cells: reduction-mislocalization of sodium-dependent glutamate transporters and enhanced activity of cystine-glutamate exchange. J. Neurosci. 19, 10767–10777 (1999).
Monje, M. et al. Roadmap for the emerging field of cancer neuroscience. Cell 181, 219–222 (2020).
Krishna, S. et al. Glioblastoma remodelling of human neural circuits decreases survival. Nature 617, 599–607 (2023).
Numan, T. et al. Regional healthy brain activity, glioma occurrence and symptomatology. Brain 145, 3654–3665 (2022).
Hausmann, D. et al. Autonomous rhythmic activity in glioma networks drives brain tumour growth. Nature 613, 179–186 (2023).
Venkatesh, H. S. et al. Electrical and synaptic integration of glioma into neural circuits. Nature 573, 539–545 (2019).
Venkataramani, V. et al. Glutamatergic synaptic input to glioma cells drives brain tumour progression. Nature 573, 532–538 (2019).
Zeng, Q. et al. Synaptic proximity enables NMDAR signalling to promote brain metastasis. Nature 573, 526–531 (2019).
Venkatesh, H. S. et al. Neuronal activity promotes glioma growth through neuroligin-3 secretion. Cell 161, 803–816 (2015).
Venkatesh, H. S. et al. Targeting neuronal activity-regulated neuroligin-3 dependency in high-grade glioma. Nature 549, 533–537 (2017).
John Lin, C. C. et al. Identification of diverse astrocyte populations and their malignant analogs. Nat. Neurosci. 20, 396–405 (2017).
Hatcher, A. et al. Pathogenesis of peritumoral hyperexcitability in an immunocompetent CRISPR-based glioblastoma model. J. Clin. Invest. 130, 2286–2300 (2020).
Monje, M. & Winkler, F. Cancer research needs neuroscience and neuroscientists. Nat Neurosci. 28, 915–917 (2025).
Goldberg, A. R. et al. Glioma-induced alterations in excitatory neurons are reversed by mTOR inhibition. Neuron 113, 858–875.e810 (2025).
Wenzel, M., Hamm, J. P., Peterka, D. S. & Yuste, R. Reliable and elastic propagation of cortical seizures in vivo. Cell Rep. 19, 2681–2693 (2017).
Brodovskaya, A., Shiono, S., Sun, C., Perez-Reyes, E. & Kapur, J. Preferential superficial cortical layer activation during seizure propagation. Epilepsia 66, 929–941 (2025).
de Vries, N. A. et al. Rapid and robust transgenic high-grade glioma mouse models for therapy intervention studies. Clin. Cancer Res. 16, 3431–3441 (2010).
Polack, P. O. et al. Deep layer somatosensory cortical neurons initiate spike-and-wave discharges in a genetic model of absence seizures. J. Neurosci. 27, 6590–6599 (2007).
Jin, X., Prince, D. A. & Huguenard, J. R. Enhanced excitatory synaptic connectivity in layer v pyramidal neurons of chronically injured epileptogenic neocortex in rats. J. Neurosci. 26, 4891–4900 (2006).
Pallud, J. et al. Cortical GABAergic excitation contributes to epileptic activities around human glioma. Sci. Transl. Med. 6, 244ra289 (2014).
Campbell, S. L. et al. GABAergic disinhibition and impaired KCC2 cotransporter activity underlie tumor-associated epilepsy. Glia 63, 23–36 (2015).
Righes Marafiga, J., Vendramin Pasquetti, M. & Calcagnotto, M. E. GABAergic interneurons in epilepsy: more than a simple change in inhibition. Epilepsy Behav. 121, 106935 (2021).
Picelli, S. et al. Smart-seq2 for sensitive full-length transcriptome profiling in single cells. Nat. Methods 10, 1096–1098 (2013).
Cadwell, C. R. et al. Electrophysiological, transcriptomic and morphologic profiling of single neurons using Patch-seq. Nat. Biotechnol. 34, 199–203 (2016).
Curry, R. N. et al. Integrated electrophysiological and genomic profiles of single cells reveal spiking tumor cells in human glioma. Cancer Cell 42, 1713–1728.e6 (2024).
Curry, R. N. et al. Glioma epileptiform activity and progression are driven by IGSF3-mediated potassium dysregulation. Neuron 111, 682–695.e689 (2023).
Gidon, A. et al. Dendritic action potentials and computation in human layer 2/3 cortical neurons. Science 367, 83–87 (2020).
Kaneda, M. M. et al. PI3Kgamma is a molecular switch that controls immune suppression. Nature 539, 437–442 (2016).
Quail, D. F. et al. The tumor microenvironment underlies acquired resistance to CSF-1R inhibition in gliomas. Science 352, aad3018 (2016).
Takenaka, M. C. et al. Control of tumor-associated macrophages and T cells in glioblastoma via AHR and CD39. Nat. Neurosci. 22, 729–740 (2019).
Patel, D. C., Tewari, B. P., Chaunsali, L. & Sontheimer, H. Neuron-glia interactions in the pathophysiology of epilepsy. Nat. Rev. Neurosci. 20, 282–297 (2019).
Feyissa, A. M. et al. High-frequency oscillations in awake patients undergoing brain tumor-related epilepsy surgery. Neurology 90, e1119–e1125 (2018).
Yao, P. S., Zheng, S. F., Wang, F., Kang, D. Z. & Lin, Y. X. Surgery guided with intraoperative electrocorticography in patients with low-grade glioma and refractory seizures. J. Neurosurg. 128, 840–845 (2018).
Dinapoli, L. et al. Quality of life and seizure control in patients with brain tumor-related epilepsy treated with levetiracetam monotherapy: preliminary data of an open-label study. Neurol. Sci. 30, 353–359 (2009).
Williams, M. S. et al. Integrative properties and transfer function of cortical neurons initiating absence seizures in a rat genetic model. J. Physiol. 594, 6733–6751 (2016).
Chipaux, M., Vercueil, L., Kaminska, A., Mahon, S. & Charpier, S. Persistence of cortical sensory processing during absence seizures in human and an animal model: evidence from EEG and intracellular recordings. PLoS ONE 8, e58180 (2013).
Polack, P. O. & Charpier, S. Ethosuximide converts ictogenic neurons initiating absence seizures into normal neurons in a genetic model. Epilepsia 50, 1816–1820 (2009).
Jung, S. et al. Progressive dendritic HCN channelopathy during epileptogenesis in the rat pilocarpine model of epilepsy. J. Neurosci. 27, 13012–13021 (2007).
van Welie, I., van Hooft, J. A. & Wadman, W. J. Homeostatic scaling of neuronal excitability by synaptic modulation of somatic hyperpolarization-activated Ih channels. Proc. Natl. Acad. Sci. USA 101, 5123–5128 (2004).
Volman, V., Perc, M. & Bazhenov, M. Gap junctions and epileptic seizures-two sides of the same coin? PLoS ONE 6, e20572 (2011).
Stegen, M. et al. Adaptive intrinsic plasticity in human dentate gyrus granule cells during temporal lobe epilepsy. Cereb. Cortex 22, 2087–2101 (2012).
Paz, J. T. & Huguenard, J. R. Microcircuits and their interactions in epilepsy: is the focus out of focus? Nat. Neurosci. 18, 351–359 (2015).
Yu, K. et al. PIK3CA variants selectively initiate brain hyperactivity during gliomagenesis. Nature 578, 166–171 (2020).
Hellier, J. L. et al. Assessment of inhibition and epileptiform activity in the septal dentate gyrus of freely behaving rats during the first week after kainate treatment. J. Neurosci. 19, 10053–10064 (1999).
Chauviere, L. et al. Changes in interictal spike features precede the onset of temporal lobe epilepsy. Ann. Neurol. 71, 805–814 (2012).
White, A. et al. EEG spike activity precedes epilepsy after kainate-induced status epilepticus. Epilepsia 51, 371–383 (2010).
Schiller, Y. Inter-ictal- and ictal-like epileptic discharges in the dendritic tree of neocortical pyramidal neurons. J. Neurophysiol. 88, 2954–2962 (2002).
Staley, K. J. & Dudek, F. E. Interictal spikes and epileptogenesis. Epilepsy Curr. 6, 199–202 (2006).
Rakhade, S. N. et al. Activity-dependent gene expression correlates with interictal spiking in human neocortical epilepsy. Epilepsia 48, 86–95 (2007).
Reiner, A. & Levitz, J. Glutamatergic signaling in the central nervous system: ionotropic and metabotropic receptors in concert. Neuron 98, 1080–1098 (2018).
Izumoto, S. et al. Seizures and tumor progression in glioma patients with uncontrollable epilepsy treated with perampanel. Anticancer Res. 38, 4361–4366 (2018).
Feyissa, A. M. et al. Analysis of intraoperative human brain tissue transcriptome reveals putative risk genes and altered molecular pathways in glioma-related seizures. Epilepsy Res. 173, 106618 (2021).
Huberfeld, G. & Vecht, C. J. Seizures and gliomas-towards a single therapeutic approach. Nat. Rev. Neurol. 12, 204–216 (2016).
Henrik Heiland, D. et al. Tumor-associated reactive astrocytes aid the evolution of immunosuppressive environment in glioblastoma. Nat. Commun. 10, 2541 (2019).
Robel, S. et al. Reactive astrogliosis causes the development of spontaneous seizures. J. Neurosci. 35, 3330–3345 (2015).
Pombo Antunes, A. R. et al. Single-cell profiling of myeloid cells in glioblastoma across species and disease stage reveals macrophage competition and specialization. Nat. Neurosci. 24, 595–610 (2021).
Poon, C. C., Sarkar, S., Yong, V. W. & Kelly, J. J. P. Glioblastoma-associated microglia and macrophages: targets for therapies to improve prognosis. Brain 140, 1548–1560 (2017).
Barker-Haliski, M. L., Loscher, W., White, H. S. & Galanopoulou, A. S. Neuroinflammation in epileptogenesis: insights and translational perspectives from new models of epilepsy. Epilepsia 58, 39–47 (2017).
Varn, F. S. et al. Glioma progression is shaped by genetic evolution and microenvironment interactions. Cell 185, 2184–2199.e2116 (2022).
Hoogstrate, Y. et al. Transcriptome analysis reveals tumor microenvironment changes in glioblastoma. Cancer Cell 41, 678–692.e677 (2023).
Santos-Pinheiro, F. et al. Seizure burden pre- and postresection of low-grade gliomas as a predictor of tumor progression in low-grade gliomas. Neurooncol. Pr. 6, 209–217 (2019).
Ahmadipour, Y. et al. Preoperative and early postoperative seizures in patients with glioblastoma-two sides of the same coin? Neurooncol. Adv. 3, vdaa158 (2021).
Dewan, M. C., White-Dzuro, G. A., Brinson, P. R., Thompson, R. C. & Chambless, L. B. Perioperative seizure in patients with glioma is associated with longer hospitalization, higher readmission, and decreased overall survival. J. Neurosurg. 125, 1033–1041 (2016).
Sørensen, M. F. et al. High expression of cystine–glutamate antiporter xCT (SLC7A11) is an independent biomarker for epileptic seizures at diagnosis in glioma. J. Neuro-Oncol. 138, 49–53 (2018).
Kerkhof, M., Benit, C., Duran-Pena, A. & Vecht, C. J. Seizures in oligodendroglial tumors. CNS Oncol. 4, 347–356 (2015).
Zhong, Z., Wang, Z., Wang, Y., You, G. & Jiang, T. IDH1/2 mutation is associated with seizure as an initial symptom in low-grade glioma: a report of 311 Chinese adult glioma patients. Epilepsy Res 109, 100–105 (2015).
Moore, H. M. et al. Biospecimen reporting for improved study quality (BRISQ). Cancer Cytopathol. 119, 92–101 (2011).
Lin, F. et al. PI3K-mTOR pathway inhibition exhibits efficacy against high-grade glioma in clinically relevant mouse models. Clin. Cancer Res. 23, 1286–1298 (2017).
Kim, D., Langmead, B. & Salzberg, S. L. HISAT: a fast spliced aligner with low memory requirements. Nat. Methods 12, 357–360 (2015).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014).
Stephens, M. False discovery rates: a new deal. Biostatistics 18, 275–294 (2017).
Li, P. et al. Loss of CLOCK results in dysfunction of brain circuits underlying focal epilepsy. Neuron 96, 387–401.e386 (2017).
Sholl, D. A. Dendritic organization in the neurons of the visual and motor cortices of the cat. J. Anat. 87, 387–406 (1953).
Mandelbaum, G. et al. Distinct cortical-thalamic-striatal circuits through the parafascicular nucleus. Neuron 102, 636–652.e637 (2019).
Dzyubenko, E., Rozenberg, A., Hermann, D. M. & Faissner, A. Colocalization of synapse marker proteins evaluated by STED-microscopy reveals patterns of neuronal synapse distribution in vitro. J. Neurosci. Methods 273, 149–159 (2016).
Eyal, G. et al. Unique membrane properties and enhanced signal processing in human neocortical neurons. Elife 5, e16553 (2016).
Traub, R. D., Wong, R. K., Miles, R. & Michelson, H. A model of a CA3 hippocampal pyramidal neuron incorporating voltage-clamp data on intrinsic conductances. J. Neurophysiol. 66, 635–650 (1991).
Poirazi, P., Brannon, T. & Mel, B. W. Arithmetic of subthreshold synaptic summation in a model CA1 pyramidal cell. Neuron 37, 977–987 (2003).
Migliore, M. Modeling the attenuation and failure of action potentials in the dendrites of hippocampal neurons. Biophys. J. 71, 2394–2403 (1996).
Magee, J. C. Dendritic hyperpolarization-activated currents modify the integrative properties of hippocampal CA1 pyramidal neurons. J. Neurosci. 18, 7613–7624 (1998).
Acknowledgements
We thank Erika Sabel-Goedknegt for her contribution to all immunohistochemical analyses; Xiaolu Wang for his contribution to the analysis of scRNAseq data; Mandy Rutteman for the management of mouse models. We thank Dr. Marjolein Geurts for her contribution to patient data assessment, Dr. Maarten Titulaer, Dr. Geeske van Woerden and for comments on the manuscript. We thank the neurosurgeons of the Department of Neurosurgery for their cooperation with this study. Support was provided by Erasmus MC Fellowship, NWO VIDI grant (VI.Vidi.192.008), NWO-Klein grant (OCENW.KLEIN.007), ERC-Stg grant (852869), ERC-Cog grant (101170567), and the Convergence Flagship program to Z.G.
Author information
Authors and Affiliations
Contributions
Z.G., B.B., A.J.P.E.V., C.M.F.D. and C.I.D.Z. conceived this study and contributed to study design. O.v.T. contributed the mouse model used in this study. V.D. contributed to pathological assessment of tissues. B.B., A.B., A.v.d.S. and J.A.v.R. conducted experiments. B.B., A.B. and A.v.d.S. analyzed the data. Y.T., Z.Y. and J.K.L. performed the computational simulation. B.B. and Z.G. led the writing of the manuscript. A.J.P.E.V. and Z.G. jointly supervised the project. All authors contributed to the drafting of manuscript with important intellectual content.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Yuguo Yu and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Bouwen, B.L.J., Bolleboom, A., Tang, Y. et al. Aberrant neural activity in the peritumoral cortex underlies the progression of tumor-associated seizures. Nat Commun 16, 10846 (2025). https://doi.org/10.1038/s41467-025-66226-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-66226-5