Abstract
Existing evidence regarding the impact of vaccination on the natural history of high-risk human papillomavirus (HPV) infections remains limited, understanding such effects is essential for optimizing cervical cancer screening in post-vaccination era. Using 10-year follow-up data from a phase 3 randomized trial of the Escherichia coli-produced HPV-16/18 bivalent vaccine (NCT01735006) and its extension study (NCT05045755, NCT04969445), we compared the spectra and natural history (persistence, clearance, and progression) of high-risk HPV infections between vaccinated and unvaccinated females aged 18-45 years. Data was analyzed using the Cox regression and the competing risk model. Our findings indicate that vaccination reduces the burden of HPV-16/18-associated lesions (HR = 0.12, p = 0.0041) primarily by preventing incident infections (HR = 0.45, p < 0.0001) and modifying the natural history of breakthrough infections (enhancing clearance: 98.5% vs. 93.8%, p < 0.0001; and attenuating progression: 1.5% vs. 6.2%, p = 0.0420). Conversely, the elevated burden of HPV-52-associated lesions (HR = 3.06, p = 0.0303) observed in the vaccine group stems mainly from altered natural history (reduced clearance: 90.3% vs. 97.9%, p = 0.0144; and increased progression: 9.7% vs. 2.1%, p = 0.0421), rather than an increase in incidence (HR = 1.09, p = 0.2669). In this work, the observed shifts in HPV infection profiles and natural history between vaccinated and unvaccinated populations suggest that cervical cancer screening recommendations may warrant adjustment for vaccinated individuals.
Similar content being viewed by others
Introduction
Cervical cancer is the fourth most prevalent cancer among women globally1, the primary cause of which is human papillomavirus (HPV) infection. More than 80% of women acquire an HPV infection during their lifetime2,3. HPV vaccination is recognized as the most effective measure for preventing cervical cancer4. As of July 17, 2025, 149 countries have incorporated the HPV vaccine into national immunization programs5. Although a significant decline in the infection with vaccine-protected types and associated cervical lesions has been consistently demonstrated6,7,8, type replacement has been proposed to occur post-vaccination, whereby the marked reduction of vaccine-protected types creates an ecological niche that may permit expansion of non-vaccine-protected types. However, reported shifts in the prevalence of non-vaccine-protected types exhibit heterogeneity with no consensus: some studies indicate increases in HPV-39 and HPV-529, as well as HPV-56 and HPV-688, while others report minimal or no change10,11.
Screening is another crucial measure against cervical cancer12. The 2021 World Health Organization (WHO) guidelines recommend transitioning the primary screening method from traditional cytology or visual inspection to HPV DNA testing, given its superior sensitivity, higher negative predictive value, objectivity of molecular diagnostics and reduced reliance on cytology expertise13,14. Within this framework, the management of HPV-positive individuals primarily depends on the natural history of different HR-HPV types. Screening and triage recommendations across countries differ15,16,17. In the United States, Australian and China, detection of HPV-16/18 infection warrants immediate referral to colposcopy, whereas other HR-HPV types necessitate cytological or p16/Ki67 triage.
Almost all guidelines emphasize that vaccinated individuals should continue regular cervical cancer screening. As the vaccinated population continues to expand, developing evidence-based screening and triage recommendations tailored to vaccinated cohorts become increasingly important. However, existing studies focus on outcome events post-vaccination—shifts in each HR-HPV type infection incidence or related cervical lesions—while neglecting viral kinetics alterations, such as the vaccine’s impact on viral persistence, clearance, or progression. The latter is essential for designing proper screening strategies for vaccinated populations. If subsequent changes in cervical lesion burden result exclusively from shifts in infection incidence, then existing strategies for the general population may remain applicable for vaccinated population. However, if vaccination alters viral persistence or progression capabilities, adjusting the protocols for screening and abnormality management accordingly will be required.
In this work, we aim to unveil the impact of HPV-16/18 bivalent vaccine immunization on the HR-HPV infection spectra and natural history, providing novel insights for developing cervical cancer screening strategies in the post-vaccination era.
Results
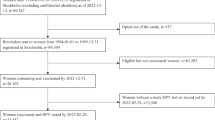
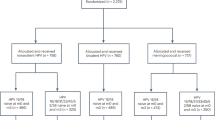
A total of 7372 participants were enrolled in the Phase 3 trial. Among them, 3691 received the HPV vaccine and 3681 received the control vaccine (Fig. 1). After the end of the Phase 3 trial, 1648 participants consented and were enrolled in the extension study, with 806 in the vaccine group and 842 in the control group (Supplementary Fig. 1). The baseline characteristics were similar between the vaccine and control groups in both the trial and extension study (Table 1). In the Phase 3 trial, the mean age (standard deviation, SD) was 30.0 (7.4) years in the vaccine group and 29.9 (7.3) years in the control group. The median follow-up time (interquartile range, IQR) was 5.6 (5.5, 5.7) years for women in both the vaccine and control groups. In the extension study, the mean age (SD) was 30.2 (7.4) years in the vaccine group and 30.1 (7.4) years in the control group. The median (IQR) follow-up time for women in both groups was 10.2 years (10.2, 10.3). In both studies, the baseline prevalence rates of HR-HPV types were similar between the vaccine and control groups, with HPV-52, −16, and −51 being the most common types.
CIN2 + , cervical intraepithelial neoplasia grade 2 or greater; HR-HPV, high-risk human papillomavirus; 6mPI, persistent infections (over 6 months); 12mPI, persistent infections (over 12 months). Participants who were negative for the corresponding HPV type at day 0, as well as those who attended at least one follow-up visit, were included in the HPV incident infection analysis set. Among them, participants who had not completed colposcopy referral were excluded from the cervical lesion analysis set. Participants who were negative for the corresponding HPV type at day 0, and attended two follow-up visits at intervals of at least 150 days (300 days), were included in the 6mPI (12mPI) analysis set.
The impact of HPV-16/18 vaccination on the incidences of HR-HPV infections and cervical lesions
The analysis sets are presented in Fig. 1. There were 7146, 7009, 6942, and 7143 participants included in the analysis sets of incident infections, persistent infections (over 6 months, 6mPIs), persistent infections (over 12 months, 12mPIs), and cervical lesions, respectively. Overall, the HPV-16/18 bivalent vaccine immunization has the most significant impact on the infections and cervical lesions associated with direct-protection types (HPV-16/18), cross-protection types (HPV-31/33/45), and one of the remaining high-risk types (HPV-52). Specifically, the incidences (per 1000 person-years [PYs]) of HPV-16/18 and HPV-31/33/45 incident infections were lower in the vaccine group compared to the control group, at 7.2 vs 16.0 (hazard ratio [HR] = 0.45, p < 0.0001, q = 0.0002) and 6.6 vs 11.0 (HR = 0.60, p < 0.0001, q = 0.0002; Table 2). The incidences (per 1000 PYs) of HPV-16/18 and HPV-31/33/45 6mPIs were 0.6 vs 4.9 (HR = 0.13, p < 0.0001, q = 0.0003) and 1.9 vs 3.4 (HR = 0.55, p = 0.0025, q = 0.0038; Table 3). 12mPIs endpoint also showed similar trends (Supplementary Table 1). The incidences (per 1000 PYs) of HPV-16/18 and HPV-31/33/45 associated cervical intraepithelial neoplasia grade 2 or greater (CIN2 + ) in the vaccine and control groups were 0.1 vs 0.8 (HR = 0.12, p = 0.0041, q = 0.0123) and 0.3 vs 0.4 (HR = 0.82, p = 0.7257, q = 0.7257; Table 4). In contrast, the incidence (per 1000 PYs) of HPV-52 incident infection was similar between the vaccine and control groups (18.0 vs 16.6, HR = 1.09, p = 0.2669, q = 0.3855; Table 2), while the incidences (per 1000 PYs) of 6mPIs (8.1 vs 5.7, HR = 1.43, p = 0.0031, q = 0.0134; Table 3) and CIN2+ (0.7 vs 0.2, HR = 3.06, p = 0.0303, q = 0.1515; Table 4) were higher in the vaccine group compared to the control group. In the sensitivity analysis excluding participants with multiple infections (Supplementary Table 2), nine HPV-52-associated CIN2+ cases were observed in the vaccine group and one in the control group, consistent with the main analysis (15 vs 5; Table 4).
In the vaccine group, HPV-52 related CIN2+ had the highest incidence, accounting for 38.5% of newly diagnosed HR-HPV related CIN2+ during the follow-up period, whereas in the control group, HPV-16 was the highest at 24.6%, with HPV-52 ranking sixth at 7.7%. (Supplementary Fig. 2). After excluding HPV-16/18 related cases, HPV-52 related CIN2+ accounted for 40.5% (ranking first) in the vaccine group and 10.4% (ranking fourth) in the control group, respectively (Supplementary Fig. 3). In addition, for overall HR-HPV, the incidences (per 1000 PYs) of incident infections, 6mPIs, and CIN2+ were 77.7 vs 88.3 (HR = 0.89, p = 0.0068, q = 0.0068), 26.7 vs 28.5 (HR = 0.94, p = 0.2781, q = 0.2781), and 1.8 vs 3.0 (HR = 0.62, p = 0.0396, q = 0.0594; Table 4), respectively.
The impact of HPV-16/18 vaccination on the natural history of HR-HPV infections
During the follow-up period, a total of 3695 HR-HPV incident infection events were observed in participants who were negative for the corresponding HPV type at day 0 (Fig. 1). Of these, 604 infections were identified in the participant’s last follow-up visit, and subsequent progression could not be observed. Ultimately, 3091 events were included in the analysis set of progression, with 1446 in the vaccine group and 1645 in the control group (Supplementary Table 3). Overall, the HPV-16/18 bivalent vaccine immunization has the most significant impact on the natural history of HPV-16/18/52, while no evident effect was observed on the cross-protection types (HPV-31/33/45) and other HR-HPV types. For HPV-16/18, HPV-31/33/45, and HPV-52 incident infections, the persistent proportions in the vaccine and control groups were 10.8% vs 40.0% (p < 0.0001, q = 0.0003), 36.4% vs 35.7% (p = 0.9109, q = 0.9109), and 53.5% vs 41.9% (p = 0.0052, q = 0.0225), respectively (Supplementary Table 3). The incidences of clearance (per 100 PYs) were 90.9 vs 61.5 (HR = 1.61, p < 0.0001, q = 0.0003), 76.9 vs 69.5 (HR = 1.17, p = 0.1956, q = 0.2209), and 45.0 vs 54.3 (HR = 0.83, p = 0.0488, q = 0.2115), respectively (Supplementary Table 4). The persistence durations and times to clearance were detailed in Supplementary Tables 5-6. Competing risk models showed that for the HPV-16/18 (Fig. 2 and Supplementary Figs. 5A-6A), the vaccine group had a lower cumulative risk of progression (1.5% vs 6.2%, Gray’s test p = 0.0420, q = 0.0803) and a higher cumulative clearance rate (98.5% vs 93.8%, Gray’s test p < 0.0001, q = 0.0003) compared to the control group. Conversely, for HPV-52 (Fig. 3), the cumulative risk of progression was significantly higher in the vaccine group compared to the control group (9.7% vs 2.1%, Gray’s test p = 0.0421, q = 0.2526), but the cumulative clearance rate was significantly lower (90.3% vs 97.9%, Gray’s test p = 0.0144, q = 0.0776). Comparable progression risks and clearance rates between vaccine and control groups were observed for the HPV-31/33/45 types (Fig. 4 and Supplementary Figs. 7A-9A) and other HR-HPV types (HPV-35, -39, -51, -56, -58, -59, and -68, Supplementary Figs. 10A-16A). The sensitivity analyses excluding participants with multiple HR-HPV infections (Supplementary Tables 7-10, Supplementary Fig. 4 and Supplementary Figs. 5B-16B) and applying a stricter definition of clearance (Supplementary Tables 11-12) yielded results consistent with the main analysis.
CIN2 + , cervical intraepithelial neoplasia grade 2 or greater; HR-HPV, high-risk human papillomavirus; FDR, false discovery rate. In competing risk analyses, the estimates for progression (to CIN2 + ), clearance, and persistence add up to 100% at each time point. A stratified Benjamini-Hochberg FDR procedure was used to adjust for multiple comparisons. All statistical tests were two-sided, with a significance level of α = 0.05.
CIN2 + , cervical intraepithelial neoplasia grade 2 or greater; HR-HPV, high-risk human papillomavirus; FDR false discovery rate. In competing risk analyses, the estimates for progression (to CIN2 + ), clearance, and persistence add up to 100% at each time point. A stratified Benjamini-Hochberg FDR procedure was used to adjust for multiple comparisons. All statistical tests were two-sided, with a significance level of α = 0.05.
CIN2 + , cervical intraepithelial neoplasia grade 2 or greater; HR-HPV, high-risk human papillomavirus; FDR false discovery rate. In competing risk analyses, the estimates for progression (to CIN2 + ), clearance, and persistence add up to 100% at each time point. A stratified Benjamini-Hochberg FDR procedure was used to adjust for multiple comparisons. All statistical tests were two-sided, with a significance level of α = 0.05.
Exploratory analyses were conducted to investigate the natural history of HPV-52 in participants with single infection and those with multiple infection of HPV-52. In the control group, HPV-52 multiple infection events demonstrated comparable persistence (36.7% [18/49] vs 36.1% [60/166], p = 0.9398; Supplementary Table 13) and clearance proportions (85.7% [42/49] vs 87.4% [145/166], p = 0.7651; Supplementary Table 13) to single infection events. Both persistence and clearance times showed non-significant differences between the HPV-52 multiple infection and single infection events (p > 0.05; Supplementary Table 13). Multiple infection with other HR-HPV types does not affect the persistence and clearance capacities of HPV-52 infection. Compared to the control group, HPV-52 single infection events in the vaccine group exhibited a significantly higher persistence proportion (52.4% [99/189] vs 36.1% [60/166], p = 0.0021; Supplementary Table 7), alongside a lower clearance proportion (78.3% [148/189] vs 87.4% [145/166], p = 0.0252; Supplementary Table 8). Persistence duration was significantly prolonged (p = 0.0004; Supplementary Table 9) while clearance time was delayed (p = 0.0428) in the vaccine group (Supplementary Table 10).
During the entire follow-up period, 147 women underwent surgical interventions: 69 (1.87%) vaccinated and 78 (2.12%) unvaccinated (Supplementary Fig. 17). During the 0-4 years, surgery was performed in 80 women (38 vaccinated [1.03%] and 42 unvaccinated [1.14%]). During the 5-11 years, 67 women received surgical treatment (31 vaccinated [0.84%] and 36 unvaccinated [0.98%]). Although the vaccine group showed slightly fewer surgical procedures than the control group, there was no statistical significance (all p > 0.05). Sensitivity analysis, limited to non-surgical and postoperatively censored participants, confirmed consistent patterns with the main population: HPV-52 infection events in the vaccine group exhibited significantly elevated persistence and reduced clearance capacities compared to that in the control group (Supplementary Table 14).
Discussion
To our knowledge, this report represents the first comprehensive evaluation of the differences in the HR-HPV infection spectra and natural history between HPV vaccinated and non-vaccinated populations. The findings indicate that immunization with the HPV-16/18 bivalent vaccine significantly altered the spectrum and natural history of some HR-HPV types. This is primarily manifested as a reduction in the incidence of HPV-16/18 infections, an increase in clearance capabilities, and a decrease in progression capabilities. In contrast, the incidence of HPV-52 infections remains unchanged, but clearance capabilities decrease, and progression capabilities increase.
Following nearly two decades of HPV vaccination implementation, birth cohorts of girl who were the primary target of HPV vaccination are gradually entering cervical cancer screening programs, but evidence-based screening guidelines for vaccinated population remain underdeveloped. As immunized females represent an increasing proportion of the screening-eligible population, adapting guidelines to this new paradigm is an urgent clinical necessity18. During the 10-year follow-up period, we observed that the incidence of HPV-52 related CIN2+ in the vaccine group was 3.06 times than that of the control group (p = 0.0303). Following vaccination, HPV-52 has emerged as the most frequently attributed type of high-grade cervical lesions, accounting for 38.5% of HR-HPV associated CIN2+ cases in the vaccine group. This marks a fivefold increase compared to the control group, where HPV-52 was responsible for only 7.7% of CIN2+ cases, making it the sixth most common type. This shift represents a complete reversal from the unvaccinated set, when HPV-16 was the most prevalent strain, comprising 24.6% of CIN2+ cases. Another study based on a Costa Rican cohort showed that, 7–11 years after receiving the bivalent HPV vaccine (Cervarix®), the incidence of CIN2 associated with non-vaccine-protected types (HPV-35/39/51/52/56/58/59) increased by 71%, and CIN3 increased by 135% compared to the control group19. This body of evidence is consistent with the possibility that the relative burden of lesions associated with non-vaccine HPV types may increase following widespread HPV vaccination.
The leading hypothesis concerning the spectrum variation of HR-HPV infection is the ecological niche hypothesis which cause type replacement as above mentioned20,21. However, the observations in our study do not support this hypothesis. Based on the data from this randomized controlled cohort, we observed that the incidences of HPV-16, -18, -31, -33, and -45 infections, which are directly or cross-protected by the vaccine, significantly declined in the vaccine group compared to the control group. However, no increase in the incidence of HPV-52 infections was observed in the vaccine group (HR = 1.09, p = 0.2669), which hints that the increase in the incidence of HPV-52 related CIN2+ (HR = 3.06, p = 0.0303) in the vaccine group is unlikely to be attributable to type replacement.
Additionally, the type interactions hypothesis posits that HPV vaccination significantly reduces vaccine-protected types infections in the vaccinated populations, the clearance and persistence capacities of non-vaccine-protected types with antagonistic or synergistic relationships to vaccine-protected types may undergo significant perturbation22. However, our data demonstrated no measurable effect of multiple infection with other HR-HPV types on HPV-52 viral kinetics. Similarly, the clinical unmasking hypothesis suggests that reductions in procedures among vaccinated women result in more at-risk women having intact cervical transformation zones, which leads to an increase of high-grade lesions caused by non-vaccine-protected HPV types that would have been removed by excisional or ablative treatment after the development of disease caused by vaccine-preventable type infections, or would be acquired after treatment but failed to develop because the tissue in which most high-grade lesions arise had been removed23. However, the results of sensitivity analysis in the non-surgical and postoperatively censored participants population were also consistent with the main analysis. Neither the clinical unmasking hypothesis nor type interactions hypothesis yielded conclusive supporting evidence in this study.
The differences in progression, clearance, and persistence observed for HPV-16/18 and HPV-52 incident infections in our study are intriguing and unexpected. Historically, neutralizing antibodies have been considered the principal correlate of protection for prophylactic HPV vaccines, preventing type-specific incident infections24. Although HPV VLP vaccines can also induce L1-specific cellular immune responses, there is little evidence, either from animal or clinical studies, that vaccination alters the course of existing infections or promotes their regression23. Hence, cell-mediated immunity is thought to play little direct role in protection after HPV vaccination25. In our study, vaccination was associated with lower progression and persistence for vaccine-covered types (HPV-16/18) but with higher persistence and progression for HPV-52, despite no detectable increase in HPV-52 incidence. Thus, classical antibody-mediated protection or simple type replacement alone does not satisfactorily explain these observations. The underlying biological mechanisms of these changes in natural history remain unclear and warrant further investigation. Potential explanations may involve indirect or non-canonical immune pathways, or interactions between host immunity and the mucosal microenvironment, but current data are insufficient to draw definitive conclusions.
Moreover, the findings also raise a concern: during the progression from initial HPV-52 infection (HR = 1.09) to 6mPIs (HR = 1.43), and then to high-grade precancerous lesions (HR = 3.06), the HRs between the two groups seem to show a trend of gradual increase. Ethical considerations, limited sample size, and the nature of cervical cancer progression prevent us from evaluating the differences in the incidence of HPV-52 related cervical cancer between vaccine and control groups. It remains unclear whether the HR of cervical cancer associated with HPV-52 will continue to increase as previously observed, which necessitates real-world studies.
In our study, a statistically significant reduction in the burden of CIN2+ associated with HPV-16/18 (HR = 0.12, p = 0.0041) and HR-HPV (HR = 0.62, p = 0.0396) following HPV vaccination was observed, which is highly consistent with the previous studies. The most compelling evidence regarding the benefits of HPV 16/18 vaccination programs comes from a meta-analysis which included 13 high-quality articles focused on CIN2+ and demonstrated a significant decline in CIN2+ incidence after 5–9 years of HPV vaccination26. Among screened females aged 15–19 years, the incidence decreased by 51%, while among women aged 20-24 years, it decreased by 31%. The aforementioned evidence highlights that, even with variations in the natural history of HPV-52 type, HPV 16/18 vaccination can still offer substantial benefits. Therefore, vaccines that cover HPV 16/18 types remain the best choice for low-resource areas. In the medium- to high-resource areas, women are encouraged to receive the second-generation (nonavalent) HPV vaccine to increase type coverage. Additionally, it is necessary to develop a third-generation HPV vaccine that provides comprehensive coverage against all high-risk HPV types.
HPV testing is increasingly recognized as the preferred approach to cervical cancer screening in many countries. HR-HPV types varied substantially in cumulative risk and annual rate of progression to precancer. HPV genotyping is an important part of accurate risk estimation and optimal clinical management. Hence, marking HPV-16/18 for immediate colposcopy is now the cornerstone of many national cervical cancer screening programs, including the United States, Australia, China, and so on. The other HR-HPV types were often triaged with cytological testing or p16/Ki67 dual stain16,17. However, the results of our study showed that there is a significant difference in the natural history (persistence, clearance, and progression) of HPV-16/18 and HPV-52 in vaccinated population compared with unvaccinated women. These findings suggest that current HPV DNA-based cervical screening algorithms, which were primarily developed for unvaccinated populations, may not be appropriate for vaccinated cohorts and could lead to substantial over-referral for colposcopy among individuals with HPV-16/18 infections. Revising screening recommendations will require integration of evidence from multiple studies and consensus among experts across disciplines. Therefore, we call for further research to elucidate the impact of HPV vaccination on the natural history of HR-HPV infections, and to accelerate the development of optimized cervical cancer screening guidelines in the post-vaccine era.
The strengths of this study include the randomized controlled design, consistent gynecological examination procedures, standardized HPV genotyping tests, a high follow-up rate, and regular, active follow-up over approximately 10 years. The randomized controlled design ensures well-balanced characteristics between the vaccine and control groups, minimizing the influence of sexual behavior and other confounding factors. This study had some limitations. First, this study was based on a phase 3 trial of an E. coli-produced HPV-16/18 bivalent vaccine among Chinese women aged 18–45 years. Therefore, the generalizability of our findings to adolescent vaccination schedules (e.g., 9–14 years) and to other vaccines (e.g., Cervarix® and Gardasil®) should be interpreted with caution. Further studies are warranted to confirm these findings in other populations and vaccine platforms. Second, causal attribution of CIN2+ lesions in the presence of HPV multiple infections remains inherently challenging in HPV research. Although we minimized potential misclassification by applying a predefined attribution rule based on the persistence of the same HPV type detected in preceding cytological samples, the possibility of misclassification cannot be completely excluded. However, a sensitivity analysis limited to participants without multiple HR-HPV infections produced results consistent with the main analysis. Third, although CIN2+ was recognized as a valid proxy for cervical cancer by regulatory agencies worldwide, CIN3+ was considered a better predictor of invasive cervical cancer. Due to ethical considerations, all high-grade cervical lesions (CIN2 + ) were treated promptly according to the clinical management after observation, this study did not accumulate sufficient cases of CIN3+ for analysis. Hence, we ultimately selected CIN2+ as one of the study endpoints, noting that the attribution to HPV 16/18 (44.1%) is lower than that for cervical cancer (69.1%)1. Consequently, the actual benefits of HPV-16/18 bivalent vaccination in this study may be underestimated. Fourth, after applying multiple-comparison correction, some associations (e.g., HPV-52-related CIN2 + ) lost statistical significance. Given that differences in persistent infection incidences and natural history characteristics of HPV-52 between the two groups remained significant, this likely reflects reduced power associated with limited event counts, rather than a true false-positive findings. Nevertheless, larger sample real-world studies are warranted to further validate this observation.
In this work, the observed shifts in HPV infection profiles and natural history between vaccinated and unvaccinated populations suggest that cervical cancer screening recommendations may warrant adjustment for vaccinated individuals. Importantly, despite the elevated progression risk of HPV-52, HPV bivalent vaccination still confers substantial net benefits. Further studies are needed to validate these findings in other age groups (e.g., 9–14 years) and with other vaccine platforms (e.g., Cervarix® and Gardasil®).
Methods
Study design and participants
The analysis was based on a multicenter, double-blinded, randomized, controlled phase 3 clinical trial of the E.coli-produced HPV-16/18 bivalent vaccine and its long-term extension study27,28. Briefly, 7372 eligible healthy women aged 18–45 years, who were non-pregnant, had no history of HPV vaccination or high-grade cervical lesions, and had 1–4 sexual partners, were enrolled from five sites in China. Eligible women were randomly allocated (1:1) to receive three doses of either the HPV vaccine or the control (Hepatitis E) vaccine. Following phase 3 trial completion, 1986 participants from two sites were invited to join the extension study. Participants who received at least one dose of the investigational vaccine in the phase 3 trial were eligible.
The phase 3 trial (NCT01735006) and extension study (NCT05045755, NCT04969445) were approved by independent ethics committees (Cancer Hospital Chinese Academy of Medical Sciences, Peking University People’s Hospital, Jiangsu Provincial Centre for Disease Control and Prevention, Guangxi Liuzhou Centre for Disease Control and Prevention, and Henan Cancer Hospital, all in China) and strictly adhered to Good Clinical Practice guidelines27,28. All participants provided written informed consent at the start of both studies.
Study procedures and laboratory testing
The same study procedures and laboratory testing for endpoint evaluation were used in both the phase 3 trial and extension study, ensuring consistent assessment throughout the 10-year follow-up. Gynecological examinations and cervical sample collection were regularly conducted during the phase 3 trial (at day 0 and months 7, 12, 18, 24, 30, 42, 54, and 66) and the extension study (at months 90 and 114) for ThinPrep Pap tests (Hologic, United States) and HPV DNA genotyping (Supplementary Fig. 1). The participant with abnormal cytological findings was referred for colposcopy, and then a biopsy was recommended if any suspicious lesions were observed.
Cytological diagnosis, histopathological diagnosis, and HPV DNA genotyping were performed at the Cancer Hospital of the Chinese Academy of Medical Sciences, Beijing, China. Cytology results were reported according to the Bethesda System. Histological diagnosis was performed on biopsy samples obtained from colposcopy or excision specimens following treatment. This diagnosis was conducted independently and blindly by three senior pathologists from an pathology panel. Samples were tested for HPV DNA and genotyped using the SPF10-PCR-DEIA/LiPA25 assay (Laboratory Biomedical Products, the Netherlands; Supplementary Method 1).
Endpoint definitions
HPV infections were categorized as incident infections, 6mPIs, and 12mPIs. Incident infection was defined as new HPV DNA detection during follow-up. 6/12mPI required at least 2 positive samples for the same HPV type spanning over 6/12 months (>150/300 days) with no intervening negative sample. Persistence time was defined as the interval between the first and last positive sample dates for the same HPV type. Clearance was defined as the transition from a positive to a negative result for the same HPV type in consecutive samples. The clearance time was defined as the interval between the initial positive and the first subsequent negative sample dates for the same HPV type. To assess the robustness of results, a stricter criterion requiring two consecutive negative results to confirm clearance was also applied in the sensitivity analysis.
CIN2+ encompassed CIN2, CIN3, adenocarcinoma in situ, or invasive cervical cancer. When a single HR-HPV type was detected in a lesion, that type was considered causative. When multiple HR-HPV types were present, causality was attributed to the type that persisted from the preceding HPV sample. In cases where multiple HR-HPV types were identified without preceding type, all detected types were considered potentially associated with the lesion.
Statistical analysis
Data from the phase 3 trial and the extension study were combined for analysis. No imputation was performed for missing data. Participants who were negative for the corresponding HPV type at day 0, as well as those who attended at least one follow-up visit, were included in the HPV incident infection analysis set. Among them, participants who had not completed colposcopy referral were excluded from the cervical lesion analysis set. Participants who were negative for the corresponding HPV type at day 0, and attended two follow-up visits at intervals of at least 150 days (300 days), were included in the 6mPI (12mPI) analysis set.
The Cox regression model was used to estimate the HRs and the 95% confidence intervals (CIs) for the incidences of HR-HPV infection (incident infection, 6mPI and 12mPI), clearance and associated CIN2+ between the vaccine and control groups. For type-specific analyses, the standard Cox proportional hazards model was used. For the HPV-16/18 group (direct-protection types), HPV-31/33/45 group (cross-protection types), and HR-HPV group, the Wei, Lin, and Weissfeld (WLW) Cox model, was used to account for within-subject correlations by adjusting the variance using the ‘sandwich’ estimator29. Competing risk models were performed to estimate the competing cumulative risks of clearance, progression (to CIN2 + ), and persistence following HR-HPV infections. In competing risk analyses, progression (to CIN2 + ), clearance, and persistence of infections were considered as mutually exclusive endpoints, with their estimates adding up to 100% at each time point30. A stratified Benjamini-Hochberg false discovery rate (FDR) procedure was applied to control for multiple comparisons (Supplementary Method 2). Both original p values and FDR-adjusted q values were reported. Sensitivity analyses restricted to participants without multiple HR-HPV infections were performed to evaluate the robustness of the main findings. Additionally, exploratory analyses were conducted to examine whether the observed increase in HPV-52-related CIN2+ incidence could be interpreted within existing theoretical frameworks (ecological niche hypothesis, type interactions hypothesis, and clinical unmasking hypothesis)22,23,24.
The analyses were performed using SAS version 9.4 (SAS Institute Inc.). Statistical tests were two-sided, with a significance level (α) of 0.05.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The individual-level raw data in this study were collected as part of a clinical trial and are subject to confidentiality restrictions. The processed data are available under restricted access. Researchers may submit a detailed proposal for non-commercial purposes to the corresponding authors (wuting@xmu.edu.cn or zhangj@xmu.edu.cn) after publication. The corresponding authors will review the requests promptly and determine whether the data can be shared (within 2 months).
Code availability
All statistical analyses were performed using SAS software (version 9.4; SAS Institute, Cary, NC, USA). The SAS programs used for data processing and analysis are intrinsically linked to the individual-level raw data collected as part of the clinical trial and cannot be meaningfully interpreted or reused without access to these data. Therefore, the programs are available from the corresponding authors (wuting@xmu.edu.cn or zhangj@xmu.edu.cn) upon reasonable request, under the same access restrictions as the analyzed dataset.
References
ICO/IARC Information Centre on HPV and Cancer (HPV Information Centre). Human Papillomavirus and Related Diseases in the World. Available at https://hpvcentre.net/statistics/reports/XWX.pdf?t=1752544940758 (accessed on March 10, 2023).
Chesson, H. W. et al. The estimated lifetime probability of acquiring human papillomavirus in the United States. Sex Transm Dis 41, 660–664 (2014).
Woodman, C. B., Collins, S. I. & Young, L. S. The natural history of cervical HPV infection: unresolved issues. Nat Rev Cancer 7, 11–22 (2007).
World Health Organization. Human papillomavirus vaccines: WHO position paper, December 2022. Available at https://www.who.int/publications/i/item/who-wer9750-645-672 (accessed on December 16, 2022).
World Health Organization. HPV vaccine included in nationalimmunization programme. 2025. Available at https://app.powerbi.com/view?r=eyJrIjoiNDIxZTFkZGUtMDQ1Ny00MDZkLThiZDktYWFlYTdkOGU2NDcwIiwidCI6ImY2MTBjMGI3LWJkMjQtNGIzOS04MTBiLTNkYzI4MGFmYjU5MCIsImMiOjh9 (accessed on July 17, 2025).
Arbyn, M. et al. Prophylactic vaccination against human papillomaviruses to prevent cervical cancer and its precursors. Cochrane Database Syst Rev 5, Cd009069 (2018).
Markowitz, L. E. et al. Declines in HPV vaccine type prevalence in women screened for cervical cancer in the United States: Evidence of direct and herd effects of vaccination. Vaccine 37, 3918–3924 (2019).
Schlecht, N. F. et al. Incidence and Types of Human Papillomavirus Infections in Adolescent Girls and Young Women Immunized With the Human Papillomavirus Vaccine. JAMA Netw Open 4, e2121893 (2021).
Mesher, D. et al. Population-Level Effects of Human Papillomavirus Vaccination Programs on Infections with Nonvaccine Genotypes. Emerg Infect Dis 22, 1732–1740 (2016).
Kavanagh, K. et al. Introduction and sustained high coverage of the HPV bivalent vaccine leads to a reduction in prevalence of HPV 16/18 and closely related HPV types. Br J Cancer 110, 2804–2811 (2014).
Saccucci, M. et al. Non-Vaccine-Type Human Papillomavirus Prevalence After Vaccine Introduction: No Evidence for Type Replacement but Evidence for Cross-Protection. Sex Transm Dis 45, 260–265 (2018).
Bruni, L. et al. Cervical cancer screening programmes and age-specific coverage estimates for 202 countries and territories worldwide: a review and synthetic analysis. Lancet Glob Health 10, e1115–e1127 (2022).
World Health Organization. WHO guideline for screening and treatment of cervical pre-cancer lesions for cervical cancer prevention, second edition. Available at https://www.who.int/publications/i/item/9789240030824 (accessed on July 6, 2021).
Lindquist, S. et al. Comparative analysis of HPV testing versus cytology in Danish cervical cancer screening: Insights from a large-scale implementation study. Gynecol Oncol 191, 45–55 (2024).
Kyrgiou, M. et al. Cervical screening: ESGO-EFC position paper of the European Society of Gynaecologic Oncology (ESGO) and the European Federation of Colposcopy (EFC). Br J Cancer 123, 510–517 (2020).
Clarke, M. A. et al. Recommendations for Use of p16/Ki67 Dual Stain for Management of Individuals Testing Positive for Human Papillomavirus. J Low Genit Tract Dis 28, 124–130 (2024).
Mingzhu, L. et al. Guidelines for cervical cancer screening in China II. Gynecol Obstet Clin Med 5, e000205 (2025).
Bruni, L. et al. HPV vaccination introduction worldwide and WHO and UNICEF estimates of national HPV immunization coverage 2010-2019. Prev Med 144, 106399 (2021).
Shing, J. Z. et al. Precancerous cervical lesions caused by non-vaccine-preventable HPV types after vaccination with the bivalent AS04-adjuvanted HPV vaccine: an analysis of the long-term follow-up study from the randomised Costa Rica HPV Vaccine Trial. Lancet Oncol 23, 940–949 (2022).
Hampson, I. N. & Oliver, A. W. Update on Effects of the Prophylactic HPV Vaccines on HPV Type Prevalence and Cervical Pathology. Viruses 16, 1245 (2024).
Gray, P. et al. Long-term follow-up of human papillomavirus type replacement among young pregnant Finnish females before and after a community-randomised HPV vaccination trial with moderate coverage. Int J Cancer 147, 3511–3522 (2020).
Hampson, I. N., Oliver, A. W. & Hampson, L. Potential Effects of Human Papillomavirus Type Substitution, Superinfection Exclusion and Latency on the Efficacy of the Current L1 Prophylactic Vaccines. Viruses 13, 22 (2020).
Roden, R. B. S. & Stern, P. L. Opportunities and challenges for human papillomavirus vaccination in cancer. Nat Rev Cancer. 18, 240–254 (2018).
Schiller, J. T. & Lowy, D. R. Understanding and learning from the success of prophylactic human papillomavirus vaccines. Nat Rev Microbiol. 10, 681–692 (2012).
Schiller, J. T. & Müller, M. Next generation prophylactic human papillomavirus vaccines. Lancet Oncol. 16, e217–e225 (2015).
Drolet, M. et al. Population-level impact and herd effects following the introduction of human papillomavirus vaccination programmes: updated systematic review and meta-analysis. Lancet 394, 497–509 (2019).
Zhao, F. H. et al. Efficacy, safety, and immunogenicity of an Escherichia coli-produced Human Papillomavirus (16 and 18) L1 virus-like-particle vaccine: end-of-study analysis of a phase 3, double-blind, randomised, controlled trial. Lancet Infect Dis 22, 1756–1768 (2022).
Zhao, F. et al. Long-term efficacy and immunopersistence of an Escherichia coli-produced HPV-16/18 bivalent vaccine: an observational extension study following a randomised, double-blind Phase III clinical trial cohort. Lancet Reg Health West Pac. 61, 101668 (2025). Aug 21.
Lin, D. Y. & Wei, L. J. The Robust Inference for the Cox Proportional Hazards Model. J Am Stat Assoc 84, 1074–1078 (1989).
Demarco, M. et al. A study of type-specific HPV natural history and implications for contemporary cervical cancer screening programs. EClinicalMedicine 22, 100293 (2020).
Acknowledgements
We are grateful for the support of the National Key Research and Development Program of China (2023YFC2307602, J.Z.), National Natural Science Foundation of China (823B2086, Q.C.; 82273640, T.W.), Beijing Natural Science Foundation (L244091, T.W.) and Fundamental Research Funds for the Central Universities (20720250004, N-S.X.). The funders had no role in considering the study design or in the collection, analysis, interpretation of data, writing of the report, or decision to submit the article for publication. We thank all participants and investigators who participated in this study. AI-assisted technology (ChatGPT-4, OpenAI) was used to improve the readability and language of the report.
Author information
Authors and Affiliations
Contributions
J.Z., T.W., N-S.X., Y-Y.S. and Q.C. contributed to the study design; S-J.H., Q-Y.L., L-H.W., Q.C., J-L.Q., K-X.Z., L-C.L., J-X.L., L-F.Z., B.Z., G-H.Z., Z-F.B., and T.W. contributed to sample collection and experiment; Q.C., Y-Y.S., T.W., and J.Z. were the core team for data analysis and manuscript preparation. All the authors had access to a summary of all data. Y-Y.S. and Q.C. reviewed and verified the data in the study. All authors were responsible for the final decision to submit the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Iwao Kukimoto and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chen, Q., Quan, J., Zhu, K. et al. Variations in the Natural History of High-Risk HPV Types Following HPV-16/18 Bivalent Vaccination in Females Aged 18-45 Years. Nat Commun 17, 1677 (2026). https://doi.org/10.1038/s41467-026-68379-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-026-68379-3