Abstract
Radiotherapy (RT) is a clinical mainstay of cancer treatment that triggers tumor-specific immune responses. However, the effectiveness is usually hampered due to the hypoxic tumor microenvironment (TME) and the ambivalent impact of RT on the immune landscape of tumors. Herein, we develop an injectable hydrogel encapsulating interleukin-12 (IL-12)/anti-CTLA-4 (aCTLA-4) co-engineered red blood cells (RBC), which is in situ self-assembled within the TME to increase oxygen supply and instigate sequential aCTLA-4/IL-12 release, thus achieving Ba/O2 self-compensated radiosensitization and activating multistage immune responses. Once in the acidic TME, the in situ injected BaO2 undergoes hydrolysis to generate H2O2 and Ba2+, followed by the rapid reaction of Ba2+ with sodium alginate to afford a biocompatible hydrogel. Meanwhile, catalase presented on RBC converts H2O2 into O2, thereby alleviating hypoxia-induced radioresistance and inducing O2-mediated pore formation on RBC membrane for rapid release of aCTLA-4 to relieve tumor immunosuppression. Subsequently, IL-12 anchored on RBC is dilatorily released and interacts with T/NK cells within the TME to induce IFN-γ-dependent antitumor immunity. Taken together, the in situ self-assembled cell reservoir hydrogel offers a futuristic avenue to realize multistage radioimmunotherapy for effective tumor regression by programmable immunoregulation with significant clinical value.
Similar content being viewed by others
Introduction
Radiotherapy (RT) is a prominent clinical treatment modality for cancer by focusing a local beam of radiation at the tumor site1,2. Extensive investigations have recently revealed its dual natures in provoking DNA double-strand breaks (DSBs) in tumor cells as well as eliciting the release of damage-associated molecular patterns (DAMPs)3, thereby promoting the recruitment and maturation of dendritic cells (DCs) by instigating systemic antitumor immunity4. Of note, the effectiveness of RT is often restricted by tumor hypoxia5, which competitively reduces the DNA radicals (DNA·) to produce DNA-OO·6,7, thus resulting in less damage fixation and decreased DSBs. Moreover, due to the differential RT sensitivity among immune cells, RT may cause a direct decrease in the ratio of cytotoxic T cells to regulatory T cells (Tregs)8, accompanied by the upregulation of cytotoxic T-lymphocyte-associated protein 4 (CTLA-4)9. This inevitably restrains the DC migration and inhibits the transportation of tumor antigens to tumor-draining lymph nodes (TDLNs)10, thus greatly attenuating the therapeutic efficacy of RT. Consequently, it is a pressing priority to explore unparalleled combinational therapeutic approaches to increase intratumoral oxygen supply and alleviate tumor immunosuppression for ameliorating therapeutic outcomes of RT.
Cancer immunotherapy has been extensively investigated as a vital part of recent advent and revolutionized the profound outlook through immune checkpoint blockade (ICB) strategies11. Anti-CTLA-4 (aCTLA-4) serves as an immune modulator to inhibit CTLA-4 function12,13, thus dampening the activity of Tregs while simultaneously promoting DC maturation and even facilitating the migration of both cDC1 and cDC2 to TDLNs for CD8+ T and CD4+ T cells activation14,15. In recent years, significant research interests have been attracted to the concurrent administration of aCTLA-4 with RT to harness the potential synergism between the two treatment modalities16. Despite promising preclinical results and substantial mechanism evidence of the synergism, a majority of studies at the clinical level fail to verify the positive effect of the combined treatments17, possibly due to the simple immune checkpoint blockade insufficient to relieve cytotoxic T cell depletion and NK cell exhaustion within the TME18. Interleukin-12 (IL-12), a crucial immune cytokine, emerges as a pivotal immunostimulatory agent capable of stimulating both the innate and adaptive immune systems19. As one of the direct downstream molecules of IL-12, IFN-γ, secreted by natural killer (NK) and T cells20, promotes the recruitment of immune cells into tumors as well as instigates the DC cell-intrinsic IL-12/IFN-γ positive feedback loop21, thus bolstering antitumor responses via immune engine refueling. Hence, the integrated utilization of IL-12 and aCTLA-4 exhibits immense potential in augmenting antitumor efficacy by synergistic combination with RT22. Nevertheless, a major concern arises from the direct injection of the mixture of IL-12 and aCTLA-4 (IL/aC), which may result in uncontrolled activation of immune cells and an IL-12-induced cytokine storm23. Therefore, we postulate that orchestrating an in situ IL-12/aCTLA-4 multisite-fettering system could be a promising strategy to facilitate the application of dual immunostimulatory agents in combination with RT, thereby maximizing antitumor responses while minimizing adverse effects.
Hydrogels, defined as water-swollen and insoluble three-dimensional (3D) crosslinked networks similar to the tumor extracellular matrix24, can offer physical support for engineered cells and slow release of loaded cargos25, thus holding great potential in enabling long-term therapeutic effects and efficient tumor intervention. In comparison to synthetic hydrogels, natural hydrogels exhibit superior biodegradability26 and unexceptionable biocompatibility27, which are advantageous for applications requiring high biological compatibility and environmental sustainability. Sodium alginate (Alg), a natural polymer that remains stable within the biological environment, bears a strong resemblance to the extracellular matrix28, thus demonstrating low immunogenicity and excellent biocompatibility29. Furthermore, Alg can be degraded into nontoxic polysaccharides that do not participate in normal metabolic processes30,31. Due to the multiple merits of this natural polymer discussed above, Alg-based hydrogel is a potentially attractive alternative as a drug-encapsulating depot with negligible biosafety concerns32,33. However, traditional Alg-based hydrogels generally chelate with metal ions intrinsic in the body and lack precise control over the architecture formation34, which may result in drug leakage and potential toxicity. Hence, the development of a TME-responsive in situ-formed Alg-based hydrogel emerges as an urgent demand for spatiotemporally controlled drug release and unleashed antitumor efficacy.
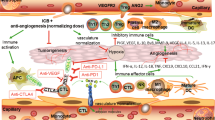
In this study, we devise an in situ self-assembled cell reservoir crosslinked network based on the introduction of barium dioxide (BaO2) into the synthesis of injectable hydrogels with locoregional delivery of IL-12/aCTLA-4 co-engineered red blood cells (IL-12/aCTLA-4@RBC-BaO2-Alg hydrogel, IL/aC@RBAH) to initiate multistage immune effects for amplified synergistic radioimmunotherapy (RIT) (Fig. 1). Upon intratumoral injection of the hydrogel precursors, BaO2 undergoes rapid hydrolysis within the acidic TME to release H2O2 and Ba2+, which quickly reacts with Alg to form hydrogels to entrap multiple cargos within the TME. Simultaneously, the generated H2O2 is decomposed under the catalysis of catalase (CAT) presented on RBC to produce oxygen for endogenous O2 furnishment. This multifaceted process not only alleviates tumor hypoxia-induced radioresistance, but also triggers pore formation on RBC membrane (RBCM) for rapid release of aCTLA-4. Subsequently, IL-12 anchored on RBC initiates activation of cytotoxic T/NK cells in the TME, with mitigated potential immunotoxicity by the immobilization on RBC surface and steady in situ retention of the hydrogel. The sequential aCTLA-4/IL-12-releasing hydrogel unleashes a therapeutic window in stepwise modulating immune networks, signified by significantly enhanced DC homing, boosted antitumor T cell infiltration, and NK cell recruitment for garnering an immunostimulatory TME. Notably, the synergy between Ba/O2 dual-compensated RT and sequential aCTLA-4/IL-12 release potentiates a benefit in both innate and adaptive immune activation to reshape the immune landscape of the TME and bolster the attack on both poorly immunogenic 4T1 triple-negative breast cancer (TNBC) and Panc02 pancreatic cancer. Collectively, the meticulous design of engineered RBC-encapsulating hydrogels offers a facile and feasible approach to synergize radiotherapy with immunotherapy to garner multistage radioimmunotherapy for reinvigorated antitumor immune responses and effective tumor regression.
a Upon intratumoral injection of the hydrogel precursors, BaO2 reacts with hydrogen ions within the TME to produce Ba2+ and H2O2 in situ. Subsequently, the generated H2O2 can be catalyzed by the CAT on RBC to supply oxygen for aerobic RT to augment sequential aCTLA-4/IL-12 release. b In the primary stage of RIT, aCTLA-4 is introduced to alleviate the tumor immunosuppression, thus primarily inducing DC homing and T cell activation. Afterward, IL-12 can initiate the immunoactivation in the relay stage to instigate the production of IFN-γ from T/NK cells for boosted antitumor immune responses.
Results
Design and characterization of IL/aC@RBC and BaO2
Numerous studies have demonstrated that native RBC (nRBC) harbors a range of active enzymes, notably CAT35,36, which catalyzes the production of oxygen from H2O2 for radiosensitization. Conventional methodologies for RBCM extraction led to substantial loss or inactivation of CAT37. In contrast, RBC processed through alternate hypotonic and hypertonic dialysis (designated as RBC) would preserve the structural integrity and enzyme activity of nRBC. Furthermore, in order to mask the immunotoxicity of IL-12 and facilitate sequential aCTLA-4/IL-12 release, RBC was engineered with IL-12 on the surface and aCTLA-4 within the cell for multisite drug encapsulation (Fig. 2a). Firstly, the sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) analysis confirmed that the composition of nRBC proteins was mostly reserved within IL/aC@RBC (Fig. 2b). In addition, to evaluate the influence of IL-12 feeding time points on the loading capacities of IL-12/aCTLA-4@RBC (IL/aC@RBC), IL-12 and aCTLA-4 were respectively labeled with AMCA and FITC to obtain IL-12-AMCA and aCTLA-4-FITC, as affirmed by the SDS-PAGE protein analysis and ultraviolet–visible (UV–Vis) absorption changes (Supplementary Fig. 1). Concurrently, sulfo-SMCC was connected with IL-12-AMCA to afford Mal-IL-12-AMCA, whose maleimide group was further clicked with DSPE-PEG2000-SH to generate DSPE-PEG2000-IL-12-AMCA for efficient IL-12 anchorage on aCTLA-4-encapsulated RBC (aC@RBC). Subsequently, IL/aC@RBC-1, IL/aC@RBC-2, and IL/aC@RBC-3 were prepared by varying the feeding time points of DSPE-PEG2000-IL-12-AMCA, which were respectively fed along with aCTLA-4-FITC, immediately subsequent to aCTLA-4-FITC, and followed by hypertonic sealing of aC@RBC. Compared with the fluorescence intensities of IL-12-AMCA and aCTLA-4-FITC in IL/aC@RBC-1 and IL/aC@RBC-2, that of IL-12-AMCA in IL/aC@RBC-3 was significantly elevated, concomitant with negligible discrepancy in the intensities of aCTLA-4-FITC (Supplementary Fig. 2). The result suggested superior IL-12/aCTLA-4 co-loading capabilities of IL/aC@RBC-3. Therefore, IL/aC@RBC-3 (denoted as IL/aC@RBC) was applied for subsequent experiments.
a Schematic showing the synthesis process of IL/aC@RBC. b SDS-PAGE protein analysis of nRBC, RBC, IL/aC@RBC, IL-12, and aCTLA-4. Marker indicates the molecular weight in kilodaltons (kDa). c Representative CLSM images of IL/aC@RBC. RBCM, IL-12, and aCTLA-4 were respectively labeled with DiI (red), AMCA (blue), and FITC (green). Scale bar, 2 μm. d Fluorescence colocalization spectra of RBCM-DiI, IL-12-AMCA, and aCTLA-4-FITC in IL/aC@RBC, as shown in (c). e SEM images of nRBC and IL/aC@RBC. Scale bar, 2 μm. f Relative enzyme activity analysis of CAT in nRBC, RBC, IL/aC@RBC and RBCM. g In vitro O2 production from nRBC and IL/aC@RBC. h IFN-γ concentrations secreted by murine splenocytes subjected to IL-12 and IL@RBC. i High-resolution TEM images of BaO2. Scale bars, 100 nm and 20 nm. j XPS spectrum of BaO2. k Release profiles of Ba2+ from BaO2 at different pH values. l H2O2 generation from BaO2 at different pH values. Experiments in (b, c, e, i) were independently repeated three times with comparable results. Data in (f, g, h, k, l) were presented as mean ± SD. n = 3 independent experiments. Statistical significance was determined using two-way ANOVA (k) and two-sided unpaired student’s t-test (l). Source data are provided as a Source Data file.
To visualize the loading sites of IL-12 and aCTLA-4, confocal laser scanning microscopy (CLSM) was used to analyze the colocalization of IL-12-AMCA and aCTLA-4-FITC with DiI-labeled RBCM (RBCM-DiI). The results revealed that IL-12 was effectively anchored on RBC surface, whereas aCTLA-4 was proficiently encapsulated into the cytosol of RBC (Fig. 2c, d). Moreover, the UV–Vis spectra presented the characteristic absorption peaks of both IL-12-AMCA and aCTLA-4-FITC in IL/aC@RBC, evidencing excellent dual-loading of IL-12 and aCTLA-4 in RBC, with corresponding encapsulation efficiency at 22.54% and 11.52%, respectively (Supplementary Fig. 3). Further investigations into the stability of IL/aC@RBC illustrated that IL-12-AMCA remained bound to RBC surface with aCTLA-4-FITC being retained inside IL/aC@RBC for an extended period of up to 7 days (Supplementary Fig. 4), confirming steady IL-12/aCTLA-4 co-loading capability of IL/aC@RBC. Next, to visualize the integrity of RBC in IL/aC@RBC, scanning electron microscopy (SEM) was employed to observe the morphology change. Although some IL/aC@RBC underwent a morphological transformation from the typical biconcave disc shape of nRBC to a spherical form with co-encapsulation of IL-12 and aCTLA-4, the images showed excellent integrity of RBC following the facile alternate hypotonic and hypertonic dialysis method (Fig. 2e). For further CAT enzyme activity evaluation, H2O2 catalytic activities were respectively monitored in nRBC, RBC, IL/aC@RBC and RBCM. The result indicated a negligible loss of CAT enzyme activity in RBC, yet a substantial decrease in RBCM compared to nRBC (Fig. 2f), indicating magnificent CAT preservation by using the alternate hypotonic and hypertonic dialysis method. Besides, a slight reduction in the enzyme activity value was observed in IL/aC@RBC, which was merely attributed to the elevation of total protein level with negligible loss in the active unit of CAT itself. Moreover, the catalytic O2 production from nRBC and IL/aC@RBC were quantitatively investigated to affirm CAT reservation on IL/aC@RBC. The results manifested that the O2 production from IL/aC@RBC and nRBC exhibited negligible difference, with the amount at 0.085 mg and 0.092 mg, respectively (Fig. 2g), demonstrating that the IL-12/aCTLA-4 encapsulation would not disrupt the CAT activity of RBC for O2 generation. Furthermore, the bioactivity of IL-12 in IL-12-anchored RBC (IL@RBC) was evaluated by the IFN-γ production during splenocyte activation. The half-maximal effective concentration (EC50) of IL@RBC was determined to be 2.302 pM, which was similar to that of free IL-12 at 1.698 pM (Fig. 2h), demonstrating effective preservation of IL-12 bioactivity in IL@RBC.
Given the lack of precise control over the drug payload and skeleton construction, most Alg-based hydrogels, chelating with metal ions intrinsic in the body38, show poor specificity between tumors and the surrounding tissues. Due to the high X-ray absorption coefficient of Ba element with a small K-edge value (37 keV) for nice X-ray attenuation character at low operating voltage39,40, BaO2 was chosen as a precursor to construct an acidity-responsive self-assembled hydrogel within the TME. During in situ injection, BaO2 was introduced for selective hydrolysis into Ba2+ and H2O2 in the acidic environment, subsequently facilitating the rapid in situ hydrogel formation through the cross-linking interaction between Ba2+ and Alg. Herein, a modified hydrolyzation-precipitation approach was employed to synthesize BaO2. Briefly, with the assistance of polyvinylpyrrolidone (PVP) as a colloidal stabilizer, BaCl2 was reacted with choline hydroxide to construct Ba(OH)2 as an intermediate product, which was further mixed with H2O2 in an alkaline methanol solution to afford BaO2 (Supplementary Fig. 5). Intriguingly, the morphology of the as-synthesized BaO2 was influenced by the feeding mass ratios of both PVP:H2O2 and H2O2:BaCl2. With the gradual decrease in the feeding mass ratio of PVP:H2O2 from 20:1 to 12:1, the morphology of the BaO2 products gradually changed from a smooth spherical shape to a rough bamboo-like shape with severe aggregation (Supplementary Fig. 6). While the ratio of PVP:H2O2 was further decreased to 2:1, bamboo-like BaO2 products with uniform dispersion were obtained. Owing to the low yield of spherical BaO2 nanoparticles formed at a high ratio of PVP:H2O2 (20:1), the feeding mass ratio of PVP:H2O2 (2:1) was ultimately chosen to synthesize smooth bamboo-like BaO2 for subsequent studies. Moreover, the influence of H2O2 on the formation of BaO2 was further investigated. At a feeding mass ratio of H2O2:BaCl2 = 1.2:1, the as-prepared BaO2 exhibited irregular morphologies with large aggregation. With an increased ratio of H2O2:BaCl2 to 1.8:1, the BaO2 products transitioned to a uniform, bamboo-like structure. Conversely, an elevation of the ratio of H2O2:BaCl2 to 3.6:1 resulted in the formation of agglomerated nanoparticles with a shortened bamboo-like structure. Consequently, a feeding mass ratio of H2O2:BaCl2 at 1.8:1 was the determined optimal choice for the synthesis of BaO2. Overall, BaO2 was meticulously synthesized at the feeding mass ratio of BaCl2:H2O2:PVP = 1:1.8:3.6 for further experiments with the morphology, dispersion, and yield into consideration.
Next, high-resolution transmission electron microscopy (TEM) was employed to determine the size of bamboo-like BaO2 (Fig. 2i), which revealed that the average length was approximately 140 nm (Supplementary Fig. 7). Besides, both the energy-dispersive X-ray spectroscopy (EDS) element mapping and spectrum presented the physical composition of BaO2, including O and Ba, each of which was uniformly distributed within the skeleton of BaO2 (Supplementary Fig. 8). Moreover, the X-ray diffraction (XRD) pattern revealed that the as-prepared BaO2 exhibited characteristic diffraction peaks aligned with those of tetragonal BaO2 crystal, confirming the similar crystallinity structure of as-synthesized BaO2 to the standard one (Supplementary Fig. 9). As expected, the valence states presented the coexistence of Ba2+ and O− within BaO2, as evidenced by the Ba 3 d peaks at 794.8/779.4 eV and O 1 s peak at 530.4 eV in the X-ray photoelectron spectroscopy (XPS) spectra (Fig. 2j and Supplementary Fig. 10). To further investigate the acidity responsiveness of BaO2, a comprehensive study was conducted on its degradation behavior under various acidic conditions. It is revealed that the Ba2+ release and H2O2 generation from BaO2 were notably slow in the neutral buffer (Fig. 2k, l). By contrast, a significant increase in the release rates was observed in the acidic buffer at pH 6.5 (Fig. 2k, l), probably because the acidic environment provided an abundance of protons to facilitate the rapid decomposition of BaO2. Furthermore, the IL/aC@RBC treated with BaO2 presented notable pores with distortions in appearance in a mildly acidic environment (pH 6.5) (Supplementary Fig. 11), which was attributed to CAT-mediated O2 production from BaO2-derived H2O2. Excitingly, the methyl thiazolyl tetrazolium (MTT) assay revealed negligible cytotoxicity of BaO2 to both 4T1 and Panc02 cells at a high concentration of even 150 μg/mL (Supplementary Fig. 12), suggesting relatively high biosafety of BaO2. Collectively, all these findings demonstrated that biocompatible BaO2 was successfully prepared and could respond to the acidic TME to generate sufficient Ba2+ and H2O2 for subsequent in situ hydrogel formation and local O2 supply.
Construction and characterization of biodegradable IL/aC@RBAH
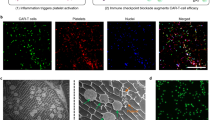
Previous studies have reported that Alg could chelate with metal ions to form crosslinked hydrogels for efficient encapsulation of biomacromolecules41,42 and cells43,44. Herein, to construct a self-assembled IL/aC@RBC reservoir hydrogel in response to the acidic TME, we introduced the scrupulously orchestrated BaO2 as the hydrogel precursor. Inspired by the above acidity-responsiveness of BaO2, we speculated that BaO2 could undergo hydrolysis in the acidic environment, resulting in the generation of Ba2+ and H2O2. Then, Ba2+ reacted rapidly with Alg to form a biocompatible 3D crosslinked network to in situ encapsulate IL/aC@RBC for constructing IL/aC@RBAH. Meanwhile, the generated H2O2 was catalyzed by primordial CAT on RBC to increase O2 supply, which also induced pore formation on RBCM for rapid aCTLA-4 release. Subsequently, with the slow degradation of the constructed Ba-Alg hydrogel, IL-12 anchored on RBC was steadily released through the enlarged crosslinked channels of the hydrogel (Fig. 3a). To testify the acidity-responsive self-assembly characteristics, the formation of IL/aC@RBAH was monitored at different pH values. As expected, IL/aC@RBAH was successfully gelated at pH 6.5, indicating that the generated Ba2+ from decomposed BaO2 could effectively chelate with Alg under acidic conditions (Fig. 3b). To gain further insight into the rheological and mechanical properties of IL/aC@RBAH, we conducted dynamic oscillation tests at first. The gelatin time of IL/aC@RBAH at pH 6.5 and 7.4 was measured using a rheometer. As shown in Fig. 3c, the hydrogel formed in merely 2.39 min at pH 6.5, while no gel formation was observed at pH 7.4, which demonstrated that the IL/aC@RBAH hydrogel could be rapidly formed in response to the acidic condition. Moreover, IL/aC@RBAH fed with various concentrations of BaO2 underwent strain amplitude sweeps and dynamic oscillatory frequency sweeps. It is noteworthy that a significant enhancement in the mechanical strength was observed when the feeding concentration of BaO2 increased from 2 to 6 mg/mL (Fig. 3d, e), which indicated increased toughness and mechanical strength of the Ba-Alg crosslinked network. With a further increase in the feeding concentrations of BaO2 from 6 to 8 mg/mL, the elevation of mechanical strength was less pronounced. Consequently, IL/aC@RBAH with feeding BaO2 at 6 mg/mL was selected for further experiments with both biosafety and rheological properties of Ba-Alg hydrogel into consideration. In order to observe the in situ encapsulation of multiple cargos within the Ba-Alg hydrogel, IL-12, aCTLA-4, and RBCM were labeled with AMCA, FITC, and DiI, respectively. Both the fluorescences of IL-12-AMCA (blue) and aCTLA-4-FITC (green) were successfully overlapped with RBCM-DiI (red), and all of them presented a uniform distribution within the hydrogel, as visualized via CLSM observation (Fig. 3f). The findings manifested successful IL-12/aCTLA-4 engineering of RBC, which was uniformly distributed in the Ba-Alg hydrogel to yield IL/aC@RBAH. Meanwhile, the cryo-scanning electron microscopy (cryo-SEM) image of IL/aC@RBAH showed that IL/aC@RBC with smoothly spherical morphology lay in the crosslinked networks structure of the hydrogel (Fig. 3g). All these results verified IL/aC@RBAH to be an excellent IL/aC@RBC reservoir hydrogel.
a Scheme for the in situ synthetic route and biodegradation performance of IL/aC@RBAH. b Photographs of IL/aC@RBAH following the pH adjustment. c Changes of G’ and G” of IL/aC@RBAH at pH 7.4 and pH 6.5. d Strain amplitude sweeps of IL/aC@RBAH with different feeding concentrations of BaO2 at a frequency of 1 Hz. e Dynamic oscillatory frequency sweeps of IL/aC@RBAH with different feeding concentrations of BaO2 at a constant strain of 2.5%. f Representative CLSM images of IL/aC@RBAH. RBCM, IL-12, and aCTLA-4 were respectively labeled with DiI (red), AMCA (blue), and FITC (green). Scale bar, 50 μm. g Cryo-SEM image presenting the microstructure of IL/aC@RBAH. Scale bar, 2.5 μm. h Degradation behavior of IL/aC@RBAH within 14 days. i Cumulative release profiles of aCTLA-4 and IL-12 from IL/aC@RBAH at pH 7.4 and pH 6.5 within 48 h. j Cumulative release profile of Ba2+ from IL/aC@RBAH within 48 h. k Representative CLSM images for live/dead staining of 4T1 cells subjected to IL/aC@RBAH for 24 h. Green, Calcein-AM, live cells. Red, PI, dead cells. Experiments in (b, f, g, k) were independently repeated three times with comparable results. Data in (h–j) were presented as mean ± SD. n = 3 independent experiments. Statistical significance was determined using two-way ANOVA (i). Source data are provided as a Source Data file.
Next, the degradation of IL/aC@RBAH was detected at pH 6.5, mimicking the mildly acidic TME. The result revealed a two-phase degradation pattern of IL/aC@RBAH, comprising an initial rapid phase and a subsequent slower rate, with the degradation process reaching equilibrium over time (Fig. 3h). Within the first day of immersion, the residual liquid and BaO2, which were originally presented on the hydrogel surface during preparation, exhibited rapid depletion, indicative of the swift degradation kinetics of the hydrogel. Subsequently, the degradation rate stabilized, with approximately 70% degradation observed after 14 days (Fig. 3h). The cumulative release behaviors of IL-12 and aCTLA-4 from IL/aC@RBAH were also investigated to prove the sequential release performance. It was obvious that aCTLA-4 was rapidly released (Fig. 3i), which demonstrated a primary liberation of aCTLA-4 through the O2-induced pores on RBCM. Afterward, IL-12 anchored on RBC was dilatorily released through the enlarged crosslinked channels of the degrading IL/aC@RBAH. Notably, the release rate of aCTLA-4 was significantly accelerated at pH 6.5 compared to that at pH 7.4, which was possibly attributed to the acidity-induced residual BaO2 degradation to trigger H2O2 production for continuous O2-mediated pore formation on RBC. Contrarily, the pores on RBC were no longer increased at pH 7.4, thus delaying the release kinetics of aCTLA-4 in the neutral environment. Moreover, the cumulative release of Ba2+ was further investigated. After 48 h of incubation at pH 6.5, approximately 50% of Ba2+ was liberated from the network structure of IL/aC@RBAH, slightly higher than that in the neutral environment (Fig. 3j), possibly due to the continuous degradation of superfluous BaO2 in the hydrogel in the mildly acidic condition. To further evaluate its biocompatibility, we conducted a calcein-AM/propidium iodide (PI) co-staining assay, showing negligible red fluorescence compared with green signals (Fig. 3k), which revealed little toxicity of IL/aC@RBAH, fitting well with the result of the MTT assay conducted on 4T1 and Panc02 cells (Supplementary Fig. 13). Overall, all the results above underscored that the acidity-responsive self-assembled IL/aC@RBAH hydrogel exhibited excellent biocompatibility and controlled sequential drug release.
In vitro Ba/O2 dual-compensated radiosensitization of IL/aC@RB
IL/aC@RBC was dexterously combined with BaO2 for IL/aC@RB to initiate O2 production for Ba/O2 self-compensated radiosensitization. Mechanically, BaO2 could respond to the acidic environment to provoke H2O2 production, which was then catalyzed by CAT inherent on RBC for O2 supply. Meanwhile, Ba element with a high X-ray absorption coefficient could ally with self-supplied O2, thus inducing Ba/O2 dual-compensated radiosensitization for augmented antitumor efficacy. To verify the underlying mechanism of dual-compensated radiosensitization, a colony formation assay was conducted to evaluate the sensitivity of 4T1 cells to RT with different formulations in vitro. The result showed that RT + IL/aC@RBC exhibited an imperceptible change in the cytotoxicity of 4T1 cells compared to the sole RT group (Supplementary Fig. 14), demonstrating that single IL/aC@RBC intervention scarcely promoted the efficacy of RT. Nevertheless, the treatment of RT + BaO2 considerably decreased the cloning numbers of 4T1 cells compared to the groups of RT and RT + IL/aC@RBC, suggesting the ameliorated RT by the existence of Ba element in BaO2. Interestingly, the cell viability was further constrained with the slightest clones in the RT + IL/aC@RB group, inferring excellent radiosensitization induced by the joint of Ba element and O2 from the alliance of RBC with BaO2. Furthermore, the intracellular ROS level was evaluated by employing 2’,7’-dichlorofluorescein diacetate (DCFH-DA) as a ROS probe45 and investigated via both CLSM and flow cytometry. As expected, RT + BaO2-treated cells presented moderately elevated ROS generation in contrast to the RT and RT + IL/aC@RBC groups, which indicated mild radiosensitization by Ba element from BaO2 (Supplementary Figs. 15 and 16). Remarkably, a significantly stronger fluorescence signal of ROS could be detected in the RT + IL/aC@RB group compared to that in the RT, RT + BaO2, and RT + IL/aC@RBC groups (Supplementary Figs. 15 and 16), attributing to effective Ba/O2-supplied radiosensitization through Ba element introduction and in situ O2 generation. Collectively, these findings indicated that IL/aC@RB could be deemed to be an efficient Ba/O2 depot for radiosensitization via the tactical incorporation of RBC with BaO2.
Immunogenic cell death (ICD) induced by RT causes the release of a serious of DAMPs, including calreticulin (CRT), high mobility group box 1 protein (HMGB1), and adenosine triphosphate (ATP), thus collectively activating the immunogenicity of tumor cells for promoted antigen presentation46. Therefore, the exposure of CRT, and the release of both HMGB1 and ATP after different treatments were analyzed to further substantiate amplified ICD effects of Ba/O2 dual-compensated radiosensitization. As expected, the immunofluorescence staining showed that RT + BaO2 could increase the CRT exposure on 4T1 cells, accompanied by HMGB1 release from cell nuclei in 4T1 cells in contrast to sole RT and RT + IL/aC@RBC (Supplementary Figs. 17 and 18). Noteworthily, the irradiated cells in the RT + IL/aC@RB group displayed the strongest CRT signals on the membrane surface and the lowest HMGB1 retention in the nucleus of 4T1 cells (Supplementary Figs. 17 and 18), affirming boosted in vitro ICD induction efficacy of Ba/O2-driven radiosensitization. In addition, concomitant with the decreased intracellular ATP content, the extracellular level of ATP in the RT + IL/aC@RB group was nearly 2.08-fold, 2.02-fold, and 1.24-fold of that in RT, RT + IL/aC@RBC, and RT + BaO2 groups, respectively (Supplementary Fig. 19). These results jointly clarified prominent ICD efficacy by RT + IL/aC@RB-mediated radiosensitization.
In situ retention and O2 self-compensation of IL/aC@RBAH
The in vivo degradation of the IL/aC@RBAH was evaluated by monitoring its residues within the skin of mice over a period of 14 days. Consistent with the in vitro degradation behaviors (Fig. 3h), the hydrogel displayed a progressive deterioration with slight debris observed on the 14th day post-implantation (Fig. 4a). To further assess the local retention ability of IL/aC@RBAH, we introduced a near-infrared (NIR) fluorescent probe Cypate and intratumorally injected the mixture of Cypate-labeled IL-12 and aCTLA-4 (IL-Cp&aC), IL-Cp/aC@RBC, or IL-Cp/aC@RBAH into the tumors of orthotopic 4T1 tumor-bearing mice for real-time monitoring in situ retention via whole-animal in vivo fluorescence imaging system (IVIS). By comparison with the rapid fluorescence elimination of IL-Cp&aC within 1 day and IL-Cp/aC@RBC within 5 days, strong Cypate fluorescence of IL-Cp/aC@RBAH was still detected on day 14 postinjection (Fig. 4b, c), similar to the local retention behaviors of the formulations implanted under the skin (Supplementary Fig. 20). The difference claimed the prominent in situ retention capability of IL/aC@RBAH compared to free IL-12 and IL/aC@RBC, possibly due to the in situ IL-12/aCTLA-4 multisite-fettering system. Moreover, the IL-12 retention in the orthotopic 4T1 tumors was also monitored. Predictably, the IL-12 levels in the IL-aC (IL-12 prior to aCTLA-4) and aC-IL (IL-12 subsequent to aCTLA-4) groups both remarkably declined in one day (Supplementary Fig. 21), indicating rapid diffusion loss of free IL-12 from tumors. Compared to the time-dependent moderate reduction in the IL&aC@RBAH group (mixture of IL-12 and aCTLA-4 in RBC) due to the pore formation on RBC, the majority of IL-12 content still remained at the tumor site on 3 days post-injection in the IL/aC@RBAH group, possibly attributed to the excellent hydrogel stability and effective IL-12 anchorage on the RBC surface. Besides, the biodistribution of BaO2 in orthotopic 4T1 tumor-bearing mice was detected. Once IL/aC@RBAH was injected into the tumors, the level of Ba in the tumors was rapidly elevated at 0 h, followed by a moderate reduction at 24 h post-injection (Supplementary Fig. 22). During the subsequent 2-day monitoring period, the Ba content in the tumors maintained over 20% ID/g with a slight declining trend, manifesting the favorable in situ retention capability of IL/aC@RBAH with minimal leakage of Ba. Contrarily, the levels of Ba in the livers and kidneys were both imperceptible post the local administration during 72 h of monitoring, indicative of excellent in situ retention capacity of IL/aC@RBAH. To further visualize the RBC distribution in orthotopic 4T1 tumor-bearing mice post local injection, the RBC in IL/aC@RBAH was labeled with DiR (a cell membrane probe) to obtain DiR-RBC for real-time monitoring. During the 72 h of the tracking process, the total fluorescence of DiR-RBC at the tumor site remained persistently above 8 × 109 p/s with imperceptible changes (Supplementary Fig. 23). Moreover, the tumors and major organs, including heart, liver, spleen, lung, and kidney, were collected at 72 h for ex vivo imaging. As anticipated, in sharp contrast to the negligible fluorescence detected in the major organs, substantial DiR signals were tracked in the tumors. Collectively, all those results confirmed the brilliant in situ retention capability of IL/aC@RBAH with favorable stability.
a Photographs showing the in vivo degradation behavior of subcutaneous IL/aC@RBAH on different days. b, c Whole-animal in vivo fluorescence imaging (b) and corresponding fluorescence quantification (c) of orthotopic 4T1 tumor-bearing mice after intratumoral injection of IL-Cp&aC, IL-Cp/aC@RBC, and IL-Cp/aC@RBAH. d Concentrations of aCTLA-4 in serum of 4T1 tumor-bearing mice after intratumoral injection of saline, aCTLA-4, and IL/aC@RBAH. e Concentrations of IL-12 in serum of 4T1 tumor-bearing mice after intratumoral injection of saline, IL-12, and IL/aC@RBAH. f–i Detection of liver function indexes including ALT (f) and AST (g), and kidney function indexes, including BUN (h) and CREA (i) in 4T1 tumor-bearing mice after intratumoral injection of saline, IL&aC, RBAH, and IL/aC@RBAH. The normal range of liver function and kidney function indexes was marked by dash lines. j Representative PA images and corresponding quantification of sO2 levels in 4T1 tumors before and after injection of IL/aC@RBC and IL/aC@RB. k Representative PA images and corresponding quantification of sO2 levels in 4T1 tumors at different time points after injection of IL/aC@RB and IL/aC@RBAH. l Representative CLSM images of immunostaining (HIF-1α, CD31) in 4T1 tumors after different treatments. Green, HIF-1α; red, CD31; blue, DAPI. Scale bar, 100 μm. m Representative γ-H2Aχ fluorescence staining images of 4T1 tumors after different treatments. Green, γ-H2Aχ; blue, DAPI. Scale bar, 200 μm. Experiments in (a, j, k, l, m) were independently repeated three times with comparable results. Data in (c–k) were presented as mean ± SD. n = 3 mice per group. Statistical significance was determined using two-way ANOVA (c) and two-sided unpaired student’s t-test (d, e, g, j, k). Source data are provided as a Source Data file.
The excellent long-term retention performance of IL/aC@RBAH might favor to restrict the potential toxicity and enhance the biocompatibility of IL-12. In light of the prolonged in situ retention characteristic inherent to IL/aC@RBAH in vivo, the concentrations of aCTLA-4 and IL-12 in the serum were testified. The concentrations of these immunological agents exhibited a significant decline in comparison to those in the direct intratumoral administration aCTLA-4 of and IL-12 (Fig. 4d, e), signifying the rapid in situ information of IL/aC@RBAH to afford low leakage and in situ retention of both aCTLA-4 and IL-12. Considering the reported toxicity of free IL-12 to liver damage and kidney injury47, liver damage indexes, including alanine aminotransferase (ALT) and aspartate aminotransferase (AST), as well as kidney function indexes compromising blood urea nitrogen (BUN) and creatinine (CREA), were respectively measured. Compared to the liver and kidney indexes in the IL&aC group, in which the mixture of IL-12 and aCTLA-4 was injected intratumorally, those indexes in the RBC/BaO2/Alg hydrogel (RBAH) and IL/aC@RBAH groups were lower and maintained within the normal range (Fig. 4f–i), probably ascribed to the biosafety of RBAH, firm immobilization of IL-12 on RBCM, and steady retention of IL/aC@RBAH within the TME. Collectively, these findings provided compelling evidence supporting the favorable biosafety of IL/aC@RBAH.
The outcome of RT is severely hampered by the hypoxic TME48,49. It is anticipated that the designed IL/aC@RBAH possessed the ability of O2 self-production for tumor hypoxia amelioration. To unveil the underlying mechanism of O2 self-supply by IL/aC@RBAH, we assessed the vascular saturated oxygen (sO2) levels in tumors after local injection of IL/aC@RBC and IL/aC@RB. The result showed that IL/aC@RBC without BaO2 hardly afforded a statistically significant change in oxygen levels (Fig. 4j), possibly originating from restricted H2O2 in TME. Excitingly, with the incorporation of BaO2, IL/aC@RB could engender a notable elevation in the intratumoral sO2 levels (Fig. 4j), which affirmed that the integration of BaO2 into IL/aC@RB facilitated the generation of H2O2 and subsequent breakdown of H2O2 by CAT for O2 provision. Moreover, tumors injected with IL/aC@RB and IL/aC@RBAH were monitored over a 48-h period. The in situ O2 production from H2O2 by IL/aC@RB was slightly increased during the first 24 h (Fig. 4k). However, the sO2 content was decreased within 48 h, possibly owing to the rapid O2 generation and consumption. In contrast, the sO2 levels in the tumors subjected to IL/aC@RBAH showed an increase in sO2 levels within 48 h, which was attributed to the slow diffusion of H2O2 and the long-term retention of IL/aC@RBC during the hydrogel formulation. All the results above suggested that the IL/aC@RBAH could be an effective means to alleviate tumor hypoxia over an extended period. The expression of hypoxia-inducible factor-1α (HIF-1α) is recognized as an indicator of hypoxic conditions within biological systems50,51,52. In both the IL/aC@RB and IL/aC@RBAH groups, a discernible decrease in the HIF-1α expression was observed, and the tumors exposed to IL/aC@RBAH showed negligible signals of HIF-1α in comparison to those treated with BaO2 or IL/aC@RBC alone (Fig. 4l and Supplementary Fig. 24). The results collectively evidenced the efficacy of IL/aC@RBAH in persistently increasing O2 supply within the TME, and palliating intratumoral hypoxia dilemma. Moreover, the expression levels of γ-H2Aχ in tumor tissues were measured to examine the DNA damage induced by different treatments. Compared to the fluorescence intensity of γ-H2Aχ in the RT and RT + IL/aC@RA groups, that in the RT + IL/aC@BaO2/Alg (RT + IL/aC@BAH) group was significantly elevated (Fig. 4m and Supplementary Fig. 25), potentially attributed to augmented X-ray absorption of Ba element for radiosensitization. Notably, the green signal of γ-H2Aχ was the utmost in the RT + IL/aC@RBAH group, thus demonstrating excellent Ba/O2 dual-compensated radiosensitization for instigating DNA damage by the ingenious marriage of RBC and BaO2.
In vivo antitumor efficacy and multistage immune responses in 4T1 tumor-bearing mice
Motivated by the aforementioned findings, we proceeded to assess the antitumor efficacy of IL/aC@RBAH in vivo. Orthotopic 4T1 breast tumor-bearing mice were randomly allocated into six groups: saline (G1), RT (G2), RT + RBAH (G3), RT + aC@RBAH (G4), RT + IL@RBAH (G5), and RT + IL/aC@RBAH (G6). Prior to exposure to X-ray irradiation (8 Gy) in pre-designed groups, these mice were respectively intratumorally administered with saline, RBAH, aC@RBAH, IL@RBAH, and IL/aC@RBAH. The tumor volumes were recorded every other day and corresponding bioluminescence intensities were monitored every week. It was found that the tumors in the RT + RBAH group were moderately inhibited compared to those in the RT group (Fig. 5a–d), probably due to Ba/O2-supplied radiosensitization by RBAH. With aCTLA-4- or IL-12-engineered RBC encapsulation within RBAH in the RT + aC@RBAH or RT + IL@RBAH group, the mice showed longer survival with more notable tumor suppression in contrast to those in the RT + RBAH group (Fig. 5a–e), possibly attributed to the potential antitumor immune efficacy by the rapid release of aCTLA-4 or sustained liberation of IL-12 alone. As expected, the administration of RT + IL/aC@RBAH resulted in superior tumor regression and prolonged lifespan of at least 2 months in 60% of the mice compared with the RT + aC@RBAH and RT + IL@RBAH groups (Fig. 5a–e). Moreover, the necrosis and proliferation of tumor cells were respectively assessed by H&E staining and Ki67 immunofluorescence. Similarly, tumors derived from the RT + IL/aC@RBAH group manifested the most extensive necrosis and weakest green fluorescence of Ki67 (Fig. 5f–i and supplementary Fig. 26), indicative of the most pronounced therapeutic effect of RT + IL/aC@RBAH-mediated RIT for effective tumor regression. These findings verified the exceptional antitumor effect of RT + IL/aC@RBAH in the synergy of Ba/O2-supplied radiosensitization and aCTLA-4/IL-12-mediated multistage immunotherapy.
a, b Whole-animal in vivo bioluminescence imaging (a) and corresponding quantification (b) of orthotopic 4T1-luciferase (4T1-luc) tumor-bearing mice after different treatments. c, d Average tumor volume changes (c) and individual tumor growth kinetics (d) of 4T1-luc tumor-bearing mice after different treatments. CR complete tumor regression. e Survival curves of orthotopic 4T1-luc tumor-bearing mice after different treatments. f, g H&E staining of orthotopic 4T1 tumors after different treatments (f) and corresponding tumor necrosis analysis (g) (n = 3 independent experiments). Scale bar, 500 μm. h, i Ki67 immunofluorescence staining (green, Ki67; blue, DAPI) of orthotopic 4T1 tumors after different treatments (h) and corresponding quantification of tumor cell proliferation (i) (n = 3 independent experiments). Scale bar, 100 μm. j Representative flow cytometry plots and quantifications of intratumoral cytotoxic T lymphocytes (CD8+ in CD3+ cells) in orthotopic 4T1 tumors after different treatments. k–m Quantitative analyses of the intratumoral antigen-specific cytotoxic T lymphocytes (CD8+IFN-γ+ cells) (k), Th1 cells (CD4+IFN-γ+ cells) (l), and Tregs (CD4+CD25+ cells) (m) among CD3+ cells in orthotopic 4T1 tumors after different treatments. n–p Quantitative analyses of the ratios of CD8+ T/Treg (n) and Th1/Treg (o) and the populations of NK cells (CD3−CD49b+ in CD45+ cells) (p) in orthotopic 4T1 tumors after different treatments. q Quantification of IFN-γ+ NK cells in orthotopic 4T1 tumors after different treatments. r Quantification of cDC1 (CD103+ cells) and cDC2 (CD11b+ cells) among CD11c+MHC Ⅱ+ cells in TDLNs of orthotopic 4T1 tumor-bearing mice post different treatments. G1, Saline; G2, RT; G3, RT + RBAH; G4, RT + aC@RBAH; G5, RT + IL@RBAH; G6, RT + IL/aC@RBAH. Experiments in (f, h) were independently repeated three times with comparable results. Data in (b, c, g, i, j–r) were presented as mean ± SD. n = 5 mice per group were used in (b–e). n = 3 mice per group were used in (f–r). Statistical significance was determined using two-way ANOVA (b, c) and two-sided unpaired student’s t-test (g, i, j–r). Source data are provided as a Source Data file.
The biotoxicities of different treatments were further evaluated by monitoring the body weights of mice, which presented no discernible discrepancy among these groups (Supplementary Fig. 27). The typical hematoxylin and eosin (H&E) staining of major organs including heart, liver, spleen, lung and kidney from mice after different treatments were also observed, which showed excellent biosafety of all these treatments with negligible histomorphological changes (Supplementary Fig. 28). Besides, long-term toxicities were measured to affirm the cytokine storm mitigation of IL-12 by IL/aC@RBAH. Healthy BALB/c mice were randomly allocated into 4 groups and subcutaneously injected with saline, IL&aC (the mixture of IL-12 and aCTLA-4), RB (the mixture of RBC and BaO2), and IL/aC@RBAH once a week. During 2 months of continuous treatments, the mice treated with IL/aC@RBAH exhibited negligible body weight changes similar to those in the saline group (Supplementary Fig. 29a), indicating minimal systemic toxicity or metabolic perturbations induced by IL/aC@RBAH. Moreover, the blood and major organs were collected after 2 months of monitoring. The blood biochemical parameters of BUN (Supplementary Fig. 29b), CREA (Supplementary Fig. 29c), and ALT (Supplementary Fig. 29d) were maintained within the normal range in all the groups. Moreover, distinct from the abnormal AST level in the RB and IL&aC groups, that in the IL/aC@RBAH group was significantly lowered within safe bounds (Supplementary Fig. 29e). All the above results evidenced the normal hepatic and renal function in the IL/aC@RBAH-treated mice. Additionally, H&E analysis presented negligible histomorphological abnormalities of major organs in the IL/aC@RBAH group, in sharp contrast to moderate inflammation of the liver tissues in the IL&aC and RB groups (Supplementary Fig. 29f), probably triggered by the burst release of aCTLA-4/IL-12 and Ba2+. All the above studies confirmed the favorable long-term biosafety of IL/aC@RBAH with negligible risks of IL-12-related cytokine storm.
Besides, we conducted a comprehensive investigation into the impact of RT + IL/aC@RBAH on the in situ immunomodulatory TME. An established orthotopic 4T1 tumor model was employed to harvest tumors to scrutinize the immune cell composition within the TME. Remarkably, the treatment of RT + IL/aC@RBAH exhibited a notable elevation in the proportion of CD8+ T cells in company with the increased level of CD8+IFN-γ+ T cells within the tumors in comparison to the treatments of RT + RBAH, RT + aC@RBAH, and RT + IL@RBAH (Fig. 5j, k and Supplementary Fig. 30). The results illuminated exceptional RT-associated antitumor immune responses triggered by the synergistic therapeutic strategy of aCTLA-4 and IL-12. In addition, the proportion of Th1 cells (CD4+IFN-γ+ T cells) was increased, while that of Tregs was decreased in the RT + IL/aC@RBAH group (Fig. 5l, m and Supplementary Fig. 30), indicating effective comovements of aCTLA-4 and IL-12 by IL/aC@RBAH in combination with RT. Meanwhile, both the ratios of CD8+ T/Treg and Th1/Treg were significantly increased in the RT + IL/aC@RBAH group, which were approximately twofold higher than those in the RT + aC@RBAH and RT + IL@RBAH groups, and nearly threefold increase compared to those in the RT + RBAH group (Fig. 5n, o and Supplementary Fig. 30). Moreover, RT + IL/aC@RBAH was also verified to significantly enhance the proportion of NK cells and stimulate NK cells to produce IFN-γ (Fig. 5p, q and Supplementary Fig. 31). Furthermore, the cellular populations within TDLNs were investigated. Typically, RT + aC@RBAH triggered moderately upregulated infiltration of cDC1 and cDC2 in TDLNs, a better result distinct from the treatments of RT, RT + RBAH, and RT + IL@RBAH (Fig. 5r and Supplementary Fig. 32), demonstrating the enhanced DC homing efficacy of aCTLA-4. Notably, after exposure to RT + IL/aC@RBAH, both the levels of cDC1 and cDC2 in TDLNs were significantly elevated compared to those in the RT + IL@RBAH and RT + aC@RBAH groups. These findings collectively underscored the potent activation of antitumor immune responses and thus the robust therapeutic efficacy achieved by RT + IL/aC@RBAH-mediated RIT in 4T1 breast tumors.
Inspired by the above excellent antitumor immune responses within the TME triggered by RT with multiplex immunotherapy by aCTLA-4 and IL-12, we further exploited the underlying mechanism of IL/aC@RBAH-mediated RIT. Previous studies have reported that RT-induced ICD may promote antitumor immune responses by tumor antigen presentation. Nevertheless, owing to the concomitant upregulation of CTLA-4 and differential RT sensitivities among immune cells in the TME53,54, we speculated that the ratio of CD8+ T/Treg would be decreased in tumors due to the stronger radioresistance of Treg than CD8+ T cells (Fig. 6a). After exposure to RT, the proportion of CTLA-4 in CD3+ T cells was remarkably increased (Fig. 6b). As speculated, the ratio of CD8+ T/Treg within tumors was also significantly downregulated following single RT, accompanied by a discernible decrease in the overall number of CD8+ T cells and a slight decline in the quantity of Treg compared to those in the saline group (Fig. 6c, d). The increased CTLA-4 expression and decreased CD8+ T/Treg ratio would potentiate DC tolerance and prevent antigen presentation for constrained antitumor immune effects55. Due to the immunosuppressive TME in the initial phase after single RT, simple RIT is limited without blocking immune checkpoints first. Therefore, we introduced a stepwise immune network regulation strategy for sequentially promoting antigen presentation and activating immune cells to generate multistage immune effects. Initially, the rapid release of aCTLA-4 from IL/aC@RBAH might relieve RT-induced immunosuppressive TME and promote the migration of DCs to TDLNs to present tumor antigens for priming T cells. Subsequently, the dilatory release of IL-12 from IL/aC@RBAH would further promote the antitumor effector cell infiltration and elevate IFN-γ production to initiate fortified antitumor immunity.
a Schematic illustration showing immunosuppression within the TME after monotherapy of RT. b Representative flow cytometry plots and corresponding quantification of CTLA-4 expression in CD3+ T cells within tumors from orthotopic 4T1 tumor-bearing mice at 24 h post the treatments of saline and RT. c, d Representative flow cytometry plots (c) and quantifications of CD3+CD8+ T cells, CD4+CD25+ T cells, and the ratio of CD8+ T/Treg (d) in tumors from orthotopic 4T1 tumor-bearing mice at 24 h. e–g Quantification of CD8+ T cells (e), NK cells (f), and Tregs (g) in orthotopic 4T1 tumors after different treatments. h–j Quantification of IFN-γ production by CD8+ T cells (h), CD4+ T cells (i), and NK cells (j). k Quantification of mature DC (CD80+CD86+ in CD11c+MHC Ⅱ+ cells) in orthotopic 4T1 tumors after different treatments. l–n Quantitative analyses of cDC1 (l) and cDC2 (m) and activated CD8+ T lymphocytes (CD69+ in CD3+CD8+ cells) (n) in TDLNs of orthotopic 4T1 tumor-bearing mice post different treatments. o Schematic for the tumor rechallenge model establishment and RT + IL/aC@RBAH-mediated therapeutic procedure. p Average tumor growth curves of subcutaneous rechallenge mice receiving the RT + IL/aC@RBAH treatment. Healthy mice implanted with 4T1 tumor cells on day 26 were indicated as control. q Quantification of Tem cells (CD62L−CD44+) and ratios of Tcm/Tem cells gating on CD8+ T cells and CD4+ T cells in spleens of 4T1 tumor-bearing mice at the end of different treatments. G1, Saline; G2, RT; G3, RT + RBAH; G4, RT + aC@RBAH; G5, RT + IL@RBAH; G6, RT + IL/aC@RBAH. Experiments in (b, c) were independently repeated three times with comparable results. Data in (b, d–n, p, q) were presented as mean ± SD. n = 3 mice per group were used in (b, d–n, q). n = 5 mice per group were used in (p). Statistical significance was determined using two-sided unpaired student’s t-test (b, d–n, q) and two-way ANOVA (p). Source data are provided as a Source Data file.
To affirm the above speculated multistage immune mechanism of RT + IL/aC@RBAH, we comprehensively tested the kinetics of aCTLA-4 and IL-12 release, as well as the respective effects of their administration sequence on immune cell populations and function. IL-12 and aCTLA-4 were jointly entrapped in RBC to obtain IL&aC@RBAH as control. The release behaviors of IL-12 and aCTLA-4 from IL&aC@RBAH and IL/aC@RBAH in the mildly acidic environment (pH 6.5) were investigated. As expected, both IL-12 and aCTLA-4 in IL&aC@RBAH were released rapidly in a similar trend within 48 h (Supplementary Fig. 33), possibly due to the simple diffusion of the mixture of IL-12 and aCTLA-4 from the formed pores on RBC. Contrarily, IL/aC@RBAH presented a sequential release behavior with rapid release of approximately 40% aCTLA-4 from the oxygen-induced pores on the RBC membrane within 48 h and slow release of less than 20% IL-12 from the enlarged channels in the crosslinked network of the degrading hydrogel within 48 h. Collectively, all those results affirmed the sequential timing and kinetics of aCTLA-4 and IL-12 release from IL/aC@RBAH. To further unveil the impact of sequential aCTLA-4/IL-12 release on the multistage immune effects, we investigated the immune cell populations in orthotopic 4T1 tumor-bearing mice exposed to different treatments. Briefly, the orthotopic 4T1-tumor bearing mice were randomly divided into 5 groups and then respectively treated with saline, RT, RT + IL-aC (IL-12 prior to aCTLA-4), RT + IL&aC (the mixture of IL-12 and aCTLA-4), and RT + aC-IL (IL-12 subsequent to aCTLA-4). Compared to the slight upregulation of CD8+ T cells induced by RT-mediated ICD, the intratumoral level of those cells was moderately upregulated in the RT + IL-aC and RT + IL&aC groups (Fig. 6e), demonstrating the modest immune activation in the primary and relay stages. This limitation was likely attributed to the rapid diffusion and loss of IL-12 from tumors, which was insufficient to sustain downstream activation during the relay stage. Of note, the RT + aC-IL treatment elicited the utmost increase in CD8+ T cells, along with the lowest Treg ratio and the highest NK cell level among all the groups (Fig. 6f, g), indicating the strongest antitumor immune initiation by the sequential aCTLA-4/IL-12 supply. Moreover, the IFN-γ production in CD8+ T cells, CD4+ T cells, and NK cells in the RT + aC-IL group were all at the highest level among all the groups (Fig. 6h–j), further affirming the most potent immune responses through aCTLA-4-mediated primary immunosuppression alleviation and relay IL-12-induced immunoactivation. It is noteworthy that the RT + aC-IL treatment likewise elicited the uppermost level of mature DCs, which was almost 1.44- and 1.27-fold higher than that in the RT + IL-aC and RT + IL&aC groups (Fig. 6k). The result further elucidated the most powerful antigen-presenting initiation of the sequential aCTLA-4/IL-12 supply in the RT + aC-IL treatment for antitumor immunity through the DC cell-intrinsic IL-12/IFN-γ positive feedback loop. Given the intratumoral immune responses elicited by RT + aC-IL, the impacts of sequential cytokine supply on antigen presentation process were further investigated through the immune cell populations in the tumor-draining lymph nodes (TDLNs) of orthotopic 4T1-tumor bearing mice subjected to different treatments. The amounts of both cDC1 and cDC2 in TDLNs were maximally elevated in the RT + aC-IL group, quite different from those in the other groups (Fig. 6l, m). Meanwhile, both the levels of CD8+ T cell infiltration and activation were at the peak in the RT + aC-IL group (Fig. 6n and Supplementary Fig. 34), further demonstrating the supreme antitumor initiation by the sequential aCTLA-4/IL-12 supply. Overall, all the results firmly affirmed the virtues of systemic multistage immune responses induced by the sequential aCTLA-4/IL-12 release from RT + IL/aC@RBAH.
Building on the multistage immune response initiated by RT + IL/aC@RBAH, local injection of IL-12 and aCTLA-4 plus RT (RT + IL&aC) were further set as a control for comprehensive therapeutic efficacy evaluation. The orthotopic 4T1 tumor-bearing mice were randomly allocated into 6 groups and respectively treated with saline, RT, RT + RBAH, IL&aC, RT + IL&aC, and RT + IL/aC@RBAH. Compared to the slight tumor inhibition in the RT group, the tumor volumes in the RT + RBAH and IL&aC groups were moderately suppressed (Supplementary Fig. 35a–d), elucidating the inadequate antitumor efficacy of the sole Ba/O2− supplied radiosensitization by RBAH or the single antitumor immune responses induced by IL&aC. Comparatively, the RT + IL&aC treatment showed much more prominent tumor inhibition than the RT + RBAH and IL&aC groups, possibly ascribed to the combined therapeutic strategy of RT and immunotherapy. Notably, the mice in the RT + IL/aC@RBAH group presented the utmost tumor regression with a prolonged lifetime of at least 2 months in about 80% of the mice (Supplementary Fig. 35e), thus substantiating the supreme antitumor efficacy of RT + IL/aC@RBAH-induced multistage RIT. In addition, the immune cell populations in the RT + IL&aC and RT + IL/aC@RBAH groups were characterized to elucidate the immunological mechanisms underlying the superior antitumor efficacy of multistage RIT. In contrast to the modest increases in intratumoral CD8+ T cells and NK cells induced by RT + IL&aC, a marked accumulation of both effector populations was instigated by RT + IL/aC@RBAH (Supplementary Fig. 36a, b). Concordantly, Tregs were reduced to the lowest levels, approximately 0.46-fold relative to those in the RT + IL&aC group (Supplementary Fig. 36c), thereby sharpening the intratumoral effector-to-suppressor balance. These findings were aligned with hydrogel-enabled sequential release of aCTLA-4 followed by IL-12, producing early relief of checkpoint and Treg constraints and later reinforcement of immune cell-mediated antitumor activities. Furthermore, the migration of both cDC1 and cDC2 to TDLNs was highest in the RT + IL/aC@RBAH group, exceeding that in the RT + IL&aC group (Supplementary Fig. 36d, e), consistent with enhanced antigen transport and cross-priming, thus providing a mechanistic basis for the superior initiation of antitumor immunity conferred by the sequential release of aCTLA-4 and IL-12 from the hydrogel.
Tumor relapse is the major cause of cancer-related mortality56. Given 60% of tumor regression by RT + IL/aC@RBAH in the above in vivo antitumor results (Fig. 5a–e), we pondered whether the therapeutic strategy of RT + IL/aC@RBAH could prevent tumor relapse. Herein, orthotopic 4T1 tumor-bearing mice with complete tumor regression after receiving the RT + IL/aC@RBAH treatment were rechallenged with 4T1 tumor cells in the right mammary fat pad to simulate the relapse. Following the re-implantation, the tumor re-growth process was monitored until day 52 following primary tumor inoculation (Fig. 6o). Strikingly, RT + IL/aC@RBAH-treated mice exhibited superior tumor suppression with almost no tumor occurrence (Fig. 6p). However, discernible tumor growth in uncontrollable trends was monitored in the 4T1-implanted healthy mice in the control group. The result verified that the treatment of RT + IL/aC@RBAH instigated long-term immunity against tumor occurrence. Emerging studies have unveiled effector memory T (Tem) cells as the direct antigen-primed cells that migrated into peripheral tissues to initiate inflammatory responses or cytotoxic effects57. However, central memory T (Tcm) cells, produced by naive T cells after antigen activation, could not directly initiate cytotoxic antitumor effects until secondary stimulation triggered effective differentiation into Tem57. To further explore the mechanism of durable immune responses elicited by RT + IL/aC@RBAH, we distinguished the levels of memory T cells, comprising Tem and Tcm in CD4+ T cells and CD8+ T cells within spleens. Excitingly, the numbers of CD8+ Tem and CD4+ Tem cells were entirely increased by the treatment of RT + IL/aC@RBAH, along with the lower ratio of Tcm/Tem in both CD8+ T cells and CD4+ T cells compared to those in the control group (Fig. 6q and supplementary Fig. 37). Taken together, the RT + IL/aC@RBAH-mediated RIT strategy based on Ba/O2 self-compensated radiosensitization and sequential aCTLA-4/IL-12 release could effectively elicit the establishment of tumor-specific immune memory against tumor relapse. Although short-term antigenic or inflammatory exposure prompts immune cells to undergo memory differentiation, persistent stimulation drives them toward an exhausted phenotype. Immune cell exhaustion is characterized by diminished effector function and increased inhibitory receptor expression, which is associated with detrimental drug resistance58,59. Studies have shown that key cytokines driving immune cell exhaustion in vivo include IL-10, IFN-I, TGF-β, and IL-23, while IL-12 often acts to reverse or alleviate the exhaustion in the majority of settings60,61,62,63. Despite its generally restorative role in the immune recovery, sustained intratumoral IL-12 exposure at a high level from the hydrogel might paradoxically promote immune cell exhaustion, accompanied by heightened metabolic demand and increased susceptibility to activation-induced dysfunction. Nevertheless, the critical driving factor of exhaustion remains persistent antigen exposure64. By rapidly suppressing tumor growth and thereby markedly reducing the antigen exposure of intratumoral immune cells, RT + IL/aC@RBAH is expected to prevent immune cell exhaustion, thereby outweighing any potential IL-12-associated pro-exhaustion effect.
Immune checkpoint blockade (ICB) targeting PD-1/PD-L1, a pivotal approach to inhibiting immune evasion, has achieved durable clinical benefits and favorable antitumor efficacy when combined with RT65. Hence, the RT + IL/aC@RBAH treatment was compared with RT combined with conventional PD-1/PD-L1-targeting ICB therapy to evaluate its deeper antitumor potential. Distinct from the rapid tumor progression in the saline group, the tumor volumes in the RT + aPD-1 and RT + aPD-L1 groups were moderately suppressed (Supplementary Fig. 38a–c), indicating the deficient antitumor efficacy of RT combined with ICB therapy. Intriguingly, the RT + IL/aC@RBAH treatment showed much more prominent tumor inhibition than the RT + aPD-1 and RT + aPD-L1 groups, thus illustrating the superior antitumor efficacy of RT + IL/aC@RBAH-mediated multistage RIT over RT combined with ICB therapy. To further elucidate the underlying immune mechanisms, the immune effects of RT + IL/aC@RBAH were compared with those of RT combined with either aPD-1 or aPD-L1. Relative to the moderate changes in immune cell levels observed with RT + aPD-1 and RT + aPD-L1, RT + IL/aC@RBAH markedly increased CD8⁺ T cells and NK cells infiltration while reducing intratumoral Tregs, thereby enhancing the effector-to-suppressor balance in the TME (Supplementary Fig. 38d–f). Taken together, these results indicated that RT synergized with the sequential release of IL-12 and aCTLA-4 was superior over its combination with anti-PD-1/PD-L1 in priming and amplifying antitumor immunity and in reshaping the TME toward an effector-dominant state that favored immune-mediated antitumor efficacy.
In vivo antitumor mechanism of RT + IL/aC@RBAH in 4T1 tumor-bearing mice
To elucidate the underlying mechanism governing the modulation of antitumor immune responses by the treatment of RT + IL/aC@RBAH, we conducted a comprehensive analysis of the transcriptomic landscape post-treatment. Differential expression gene (DEGs) analysis, in conjunction with enrichment analysis, including Gene Ontology (GO) term and Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment, was employed to delineate the transcriptomic alterations within tumors from orthotopic 4T1 tumor-bearing mice following diverse therapeutic interventions. Compared with the control group, there were 1071 differentially expressed genes (DEGs) with 849 upregulated ones and 222 downregulated ones in the RT + IL/aC@RBAH group (Fig. 7a). In comparison to the components in the RT group, a discernable gene expression disparity was also signified in the tumors after exposure to RT + IL/aC@RBAH (Fig. 7b). Analysis of the Volcano plot revealed significant expression differences in 770 genes, comprising 557 upregulated genes and 213 downregulated genes after treatment with RT + IL/aC@RBAH. The group cluster heatmap also delineated that there were gene expression distinctions among the control, RT, and RT + IL/aC@RBAH groups (Fig. 7c). To further explore these transcriptional changes, the enrichment analysis was conducted to compare the control or RT group with the RT + IL/aC@RBAH group. The DEGs in the RT + IL/aC@RBAH group, as shown by the KEGG enrichment analysis, were enriched in antitumor immune response-associated pathways, including cytokine-cytokine receptor interaction, NOD-like receptor, TNF, MAPK, B cell receptor, NF-κB, natural killer cell-mediated cytotoxicity, and Toll-like receptor signaling pathway (Supplementary Fig. 39a). In particular, the KEGG pathway enrichment also revealed pronounced upregulation of immune response-related and inflammatory signaling pathways in the RT + IL/aC@RBAH group compared to the RT group, notably encompassing cytokine-cytokine receptor interaction, TNF, IL-17, NOD-like receptor, and chemokine signaling pathways (Fig. 7d, e). In addition, GO enrichment analysis manifested that the upregulated genes in the RT + IL/aC@RBAH group compared to control group were mainly involved in response to IFN-γ, B cell activation, lymphocyte-mediated immunity, T cell proliferation, cell activation involved in immune response, and lymphocyte proliferation (Supplementary Fig. 39b). Consistently, the pronounced enrichment of related genes in immune-related signaling pathways under the RT + IL/aC@RBCH treatment in the contrast to the RT group (Fig. 7f). Notably, the genes, such as Il21, Cxcl2, Il1a, Ccl3, Il1b, Il1f9, Il6, Tnf, and Il12a, were significantly upregulated in the RT + IL/aC@RBAH group, which were in accordance with the activation of leukocyte migration, leukocyte cell-cell adhesion, cell chemotaxis, cytokine-mediated signaling pathway, and T cell activation. Furthermore, in comparison with control group, the multistage RIT initiated by RT + IL/aC@RBAH could activate antitumor immune effect-related signaling pathways, such as C-type lectin receptor signaling pathway, natural killer cell-mediated cytotoxicity, TNF signaling pathway, and NOD-like receptor signaling pathway (Supplementary Fig. 40), as evidenced by the Gene Set Enrichment Analysis (GESA). Notably, as further presented by the GSEA, quite different from the RT group, RT + IL/aC@RBAH could collectively upregulate immune response-related pathways, including Toll-like receptor signaling pathway (Fig. 7g), NOD-like receptor signaling pathway (Fig. 7h), IL-17 signaling pathway (Fig. 7i), and chemokine signaling pathway (Fig. 7j). The upregulation of these immunes-related pathways delineated above was attributed to the Ba/O2-supplied radiosensitization and multistage immune activation by sequential aCTLA-4/IL-12 release. In summary, the presented transcriptomic profiles provided reliable evidence that the synergistic RIT strategy based on sequential aCTLA-4/IL-12 release and Ba/O2− aided radiosensitization facilitated the potentiation of multistage immune activation and thus induction of tumor eradication.
a Volcano plot of DEGs between the RT + IL/aC@RBAH and control groups. b Volcano plot of DEGs between the RT + IL/aC@RBAH and RT groups. c Heat map analysis of DEGs among the control, RT, and RT + IL/aC@RBAH groups. d KEGG enrichment bubble chart of DEGs in the RT and RT + IL/aC@RBAH groups. e KEGG enrichment radar map of DEGs within the associated pathways after treatment with RT + IL/aC@RBAH. f GO enrichment chord plot of DEGs in the RT and RT + IL/aC@RBAH groups. g–j GSEA analysis of representative immune signaling pathways about Toll-like receptor (g), NOD-like receptor (h), IL-17 (i), and Chemokine (j) after treatment with RT + IL/aC@RBAH. n = 3 mice per group. DEGs were analysed using DESeq2 (two-sided Wald test) (a, b). KEGG pathway enrichment (d, e) and GSEA (g–j) were performed based on DESeq2 results. No additional multiple-comparisons adjustment was applied.
In vivo antitumor efficacy and multistage immune responses in Panc02 tumor-bearing mice
To validate the broad applicability of the treatment of RT + IL/aC@RBAH, a highly aggressive subcutaneous Panc02 tumor model was established to assess the potential of RT + IL/aC@RBAH-mediated multistage RIT. When the tumor volume reached approximately 100 mm3, C57BL/6 mice bearing Panc02-luciferase (Panc02-luc) tumors were randomly divided into six groups and the treatments were commenced as delineated above. Different from the cases of saline, RT, and RT + RBAH, the treatments of RT + aC@RBAH and RT + IL@RBAH could respectively lead to considerably deferred tumor growth (Fig. 8a–d). Remarkably, RT + IL/aC@RBAH resulted in a superior therapeutic outcome with substantial tumor suppression compared to other treatments (Fig. 8a–d), possibly induced by locally multistage immune activation via sequential release of aCTLA-4 and IL-12. Owing to the excellent tumor inhibition efficacy, the treatment of RT + IL/aC@RBAH generally resulted in a discernibly prolonged lifespan of mice (Fig. 8e). Body weights of all the treated mice were recorded every 4 days and no obvious weight changes were observed during the surveillance period (Supplementary Fig. 41). Moreover, a histopathological examination of tumor specimens was conducted to determine the in vivo antitumor efficacy of RT + IL/aC@RBAH by H&E staining and terminal deoxynucleotidyl transferase-mediated dUTP nick-end labeling (TUNEL) immunofluorescence. Both of the staining results revealed predominant tumor tissue apoptosis and necrosis with the most extensive damage of tumor cells in the RT + IL/aC@RBAH group (Fig. 8f–i and supplementary Fig. 42). To reveal the underlying mechanism of the therapeutic efficacy, the immunophenotyping of tumors from mice subjected to different treatments was examined. As expected, after the synergistic RIT by RT + IL/aC@RBAH, the numbers of NK and CD8+ T cells were increased, accompanied by decreased Tregs compared with those in the other groups (Fig. 8j–l and supplementary Fig. 43). The results collectively demonstrated efficient tumor immunomodulation by Ba/O2-mediated radiosensitization and multistage immune responses. Moreover, compared to the other groups, the treatment of RT + IL/aC@RBAH significantly increased the IFN-γ levels in CD4+ T, CD8+ T, and NK cells (Fig. 8m–o and supplementary Fig. 43). Overall, all these findings comprehensively affirmed excellent antitumor immune responses and robust treatment efficacy by the synergistic therapeutic strategy of RT + IL/aC@RBAH-induced multistage RIT in pancreatic tumors.
a, b Whole-animal in vivo bioluminescence imaging (a) and quantification (b) of tumors in subcutaneous Panc02-luc tumor-bearing mice after different treatments. c, d Average tumor growth curves (c) and corresponding individual tumor growth kinetics (d) of Panc02-luc tumor-bearing mice after different treatments. e Survival curves of Panc02-luc tumor-bearing mice after various treatments. f Tumor histological images of H&E of Panc02 tumors after different treatments. Scale bar, 500 µm. g Necrosis analysis of tumor cells according to the H&E images in (f). h Tumor histological images of TUNEL staining of Panc02 tumors after different treatments. Red, TUNEL; Blue, DAPI. Scale bar, 50 µm. i Tumor apoptosis analysis corresponding to the TUNEL images in (h). j–l Quantification of CD8+ T cells (j), Tregs (k), and NK cells (l) in Panc02 tumors after different treatments. m–o Quantification of IFN-γ gating on CD45+CD4+ T cells (m), CD45+CD8+ T cells (n), and NK cells (o) in Panc02 tumors after different treatments. G1, Saline; G2, RT; G3, RT + RBAH; G4, RT + aC@RBAH; G5, RT + IL@RBAH; G6, RT + IL/aC@RBAH. Experiments in (f, h) were independently repeated three times with comparable results. Data in (b, c, g, i–o) were presented as mean ± SD. n = 5 mice per group were used in (b–e). n = 3 mice per group were used in (f–o). Statistical significance was determined using two-way ANOVA (b, c) and two-sided unpaired student’s t-test (g, i–o). Source data are provided as a Source Data file.
Furthermore, antitumor immune cell infiltration within tumors was also visualized by immunofluorescence. Evidently, the treatment of RT + aC@RBAH and RT + IL@RBAH could temperately relieve RT-induced tumor immunosuppression by increasing CD4+ T and CD8+ T cell infiltration (Fig. 9a–c). Intriguingly, both above antitumor T cells were uppermost elevated in the tumors subjected to the therapeutic modality of RT + IL/aC@RBAH (Fig. 9a–c), emphasizing amplified antitumor immunity through the remedy of Ba/O2-enhanced RT with sequential aCTLA-4/IL-12 release. Similarly, the impact of RT + IL/aC@RBAH on the immune cellular compositions of TDLNs and spleens in Panc02 tumor-bearing mice was decoded to determine whether systemic antitumor responses were induced. Resembled with the case of 4T1 breast tumor-bearing mice, cDC1 and cDC2 cells in TDLNs were respectively sorted to investigate the capacities of RT + IL/aC@RBAH to induce antigen presentation. As predicted, the RT + IL/aC@RBAH group exhibited a significant increase in the numbers of cDC1 and cDC2 cells in TDLNs compared with the other groups (Supplementary Figs. 44 and 45), demonstrating that systemic antitumor efficacy was remarkably enhanced by the tactics of aCTLA-4-alleviated immunosuppression and IL-12-induced immunoactivation. Moreover, similar to the results of the previous antitumor immunity in subcutaneous Panc02 tumors, the RT + IL/aC@RBAH treatment exhibited significantly elevated IFN-γ secretion by CD4+ T, CD8+ T, and NK cells in spleens (Fig. 9d–i and supplementary Fig. 46). The levels of IFN-γ secretion in CD8+ T cells were approximately 1.5-fold higher than those in the RT + IL@RBAH and RT + aC@RBAH groups, and increased even more than 2.6 times compared to the saline group (Fig. 9f, g and supplementary Fig. 46). All the findings collectively corroborated that RT + IL/aC@RBAH elicited remarkable systemic immune responses and robust antitumor efficacy for prompt regression of multiple tumors.
a Representative immunofluorescence images of CD8+ T cells and CD4+ T cells in tumors from Panc02 tumor-bearing mice after different treatments. Scale bar, 200 µm. b, c Quantifications of CD8+ T cells (b) and CD4+ T cells (c) in tumors from subcutaneous Panc02 tumor-bearing mice corresponding to immunofluorescence images in (a). d–i Representative flow cytometry plots and quantifications of IFN-γ+ cells gating on CD3+CD4+ T cells (d, e), CD3+CD8+ T cells (f, g), and NK cells (h, i) in spleens from Panc02 tumor-bearing mice after different treatments. G1, Saline; G2, RT; G3, RT + RBAH; G4, RT + aC@RBAH; G5, RT + IL@RBAH; G6, RT + IL/aC@RBAH. Experiments in (a, d, f, h) were independently repeated three times with comparable results. Data in (b, c, e, g, i) were presented as mean ± SD. n = 3 mice per group. Statistical significance was determined using two-sided unpaired student’s t-test (b, c, e, g, i). Source data are provided as a Source Data file.
Discussion
In conclusion, this study developed an in situ self-assembled cell reservoir hydrogel based on the ingenious introduction of BaO2 into the construction of a crosslinked network for encapsulation of IL-12/aCTLA-4 co-engineered RBC, thus instigating Ba/O2-mediated radiosensitization and initiating multistage immune effects for synergistic RIT. Once intratumoral injection, BaO2 was preliminarily hydrolyzed into H2O2 and Ba2+ in response to the acidic TME. Subsequently, Ba2+ rapidly chelated with Alg to in situ form the RBAH within tumors, thus simultaneously encapsulating IL/aC@RBC within the 3D crosslinked network for IL/aC@RBAH. During the self-assembly process of the IL/aC@RBAH, the generated H2O2 would be catalyzed by the intrinsic CAT on RBC to elicit O2 supply, which concurrently facilitated O2-sensitized RT and triggered hole formation on RBCM for rapid release of the entrapped aCTLA-4 through the swollen pores. The priorly liberated aCTLA-4 could specifically target the overexpressed CTLA-4 on T cells, thus alleviating the RT-driven suppression of the antigen presentation. Therefore, DC homing to the TDLNs was promoted by aCTLA-4 in the primary stage of RT for initially instigating an immunomodulation niche. Accompanied by the dilatory degradation of the hydrogel, IL-12 anchored on RBC was subsequently released to the TME to bolster the DC cell-intrinsic IL-12/IFN-γ positive feedback loop and induce antitumor effecter immune cell infiltration for augmented tumor regression. Overall, the sequential aCTLA-4/IL-12-releasing hydrogel points toward tremendous potential utility in cancer treatments by sensitizing RT and fortifying multistage antitumor immunity in a safe and effective manner.
Distinct from traditional RT and immunotherapy combinations, the therapeutic modality of RT + IL/aC@RBAH compensates for the shortcomings by minimizing off-target effects and reversing RT-induced periodical tumor immunosuppression, with the potential application in superficial tumors (e.g., skin cancer, oral cancer, etc.) and deeper tumors (e.g., hepatocellular carcinoma, gastric cancer, colon cancer, etc.) through the integration of precise localized delivery and advanced interventional techniques. Moreover, the elaborately designed self-assembled hydrogel is a facile platform with comprehensive economic, logistical, and clinical considerations. Economically, the engineered RBC exhibit distinct advantages, including wide availability, high prevalence, and low-cost chemical modification66. The chemically engineered RBC exhibit enhanced safety and controllability, with significantly lower production costs compared to genetically engineered cells. Notably, the quality and yield of RBC are both stable and reproducible at laboratory and bench scales. Additionally, the introduced barium peroxide (BaO2) is synthesized via a hydrolytic precipitation method, which also offers the advantages of low-cost and economical production67. The as-prepared BaO2 nanoparticles present a highly consistent bamboo-like morphology and excellent batch-to-batch uniformity during our study. Moreover, sodium alginate exhibits excellent potential as a biomaterial due to its high structural stability, mature production processes, and wide availability from brown algae. This low-cost material offers convenient transportability and superior economic cost. Moreover, the degraded nontoxic polysaccharides from Alg-based hydrogel do not participate in normal metabolic processes, which are conducive to the development requiring high biological compatibility and environmental sustainability. Therefore, alginate-based hydrogels, featuring low production costs and FDA-approved excellent biocompatibility, illuminate a promising feasibility for clinical translation68. In the future, the self-assembled hydrogel is promising to be a generic platform to offer physical support for diverse engineered cells with compartmentalized encapsulation of various cytokines for potentiating multistage antitumor immune responses in diverse types of cancers.
Despite the potent immunotherapeutic efficacy demonstrated by RT + IL/aC@RBAH in multiple preclinical tumor models, clinical translation remains challenging. Foremost among these considerations is the quality control during manufacturing. Although the preparation of engineered RBC is relatively complex, several companies have already advanced such products (e.g., RTX-224, RTX-240, etc.) into clinical development, suggesting a degree of feasibility for large-scale production of IL/aC@RBC69,70. Nevertheless, the additional encapsulation of immunomodulatory factors within RBC further increases manufacturing complexity and quality-control challenges for clinical application of IL/aC@RBC. Furthermore, beyond the concerns regarding the stability and batch-to-batch consistency of BaO2 nanoparticles during large-scale production, the formation of highly uniform cell-encapsulating hydrogels in patients requires in-depth exploration integrating chemical technologies with biosynthetic techniques. Apart from the quality control issues discussed above, the intratumoral injection route somewhat limits the application of IL/aC@RBAH in deep-seated or metastatic tumors. Future efforts should focus on developing an in vivo platform for in situ tumor-site assembly of intravenously injected nanogels into a bulk hydrogel or combining interventional therapeutic techniques with stereotactic radiotherapy to expand the therapeutic applicability of multistage RIT elicited by RT + IL/aC@RBAH.
Methods
Chemicals and reagents
Polyvinylpyrrolidone, choline hydroxide solution, glutathione, adenosine triphosphate, creatinine, 7-Amino-4-methyl-3-coumarinylacetic acid, fluorescein isothiocyanate, and sodium pyruvate were purchased from Shanghai Aladdin Biochemical Technology Co., Ltd. Sodium alginate and barium chloride were purchased from Shanghai Macklin Biochemical Technology Co., Ltd. 4-(N-Maleimidomethyl)cyclohexane-1-carboxylic acid 3-sulfo-N-hydroxysuccinimide ester sodium salt (sulfo-SMCC) was purchased from Sigma-Aldrich. DSPE-PEG2000-SH was purchased from Chongqing Yusi Pharmaceutical Technology Co., Ltd. Mouse IFN-γ (Interferon Gamma) ELISA kit (Cat# E-EL-M0048) was purchased from Elabscience. Catalase assay kit (Cat# S0051), calreticulin rabbit monoclonal antibody (Cat# AF1666, Clone N/A, 1:100), HMGB1 rabbit polyclonal antibody (Cat# AF0180, Clone N/A, 1:100), ATP assay kit (Cat# S0026), and Reactive oxygen species assay kit (Cat# S0033S) were purchased from Beyotime Biotechnology. Horseradish peroxidase (HRP) (Cat# P8020), BCA protein assay kit (Cat# PC0020), live/dead cell double stain kit (Cat #CA1630), and red blood cell lysis buffer (Cat #R1010) were purchased from Beijing Solarbio Science & Technology Co., Ltd. Recombinant Mouse IL-12 (Cat# CM39) was purchased from Novoprotein. InVivoMAb anti-mouse CTLA-4 (CD152) antibody (Cat# BE0131, Bio X Cell) was purchased from BioXcell. The aCTLA-4 ELISA detection kit (Cat# HS1392-Mu) was purchased from Shanghai Hengyuan Biotechnology Company. Purified Rat anti-CD16/CD32 Antibody (Cat# 553141, Clone 2.4G2, 1:1000), fixable viability stain 510 (Cat# 564406), and BV421 anti-CD8α Antibody (Cat# 563898, Clone 53-6.7, 1:100) were purchased from BD Bioscience. FITC anti-CD45 Antibody (Cat# 103108, Clone 30-F11, 1:200), APC-cy7 anti-CD4 Antibody (Cat# 100526, Clone RM4-5, 1:20), APC anti-CD11c Antibody (Cat# 117310, Clone N418, 1:80), PE-cy7 anti-CD25 Antibody (Cat# 102016, Clone PC61, 1:20) PE anti-IFN-γ Antibody (Cat# 505807, Clone XMG1.2, 1:80), and APC anti-CD49b Antibody (Cat# 108909, Clone DX5, 1:80) were purchased from BioLegend. PE-EF610 anti-CD44 Antibody (Cat# 61-0441-82, Clone IM7, 1:20), SB600 anti-CD62L Antibody (Cat# 63-0621-80, Clone MEL-14, 1:150), FITC anti-CD11b Antibody (Cat# 11-0112-81, Clone M1/70, 1:100), PE anti-MHC Ⅱ Antibody (Cat# 12-5321-82, Clone M5/114.15.2, 1:1000), and EF450 anti-CD103 Antibody (Cat# 48-1031-82, Clone 2E7, 1:20) were purchased from Ebioscience.
Material characterizations
The morphologies of samples were observed by transmission electron microscopy (TEM, JEM-2100F, Japan) and scanning electron microscopy (SEM, JSM-7900F, JEOL, Japan). The element mapping (Ba, O) of BaO2 nanoparticles was characterized by energy-dispersive spectroscopy (EDS, FEI Talos F200X G2). X-ray diffraction (XRD) patterns of synthesized BaO2 nanoparticles and standard pattern of BaO2 were tested by X-ray diffractometer (X’ Pert Pro MPD, PANalytical B.V., Netherlands). The presence and valence state of the elements (Ba and O) in BaO2 nanoparticles were analyzed by X-ray photo spectrometer (XPS, EscaLab 250Xi, Thermo, USA).
Cells and animals
4T1 breast cancer cell line (purchased from the American Type Culture Collection, ATCC, Cat# CRL-2539) and Panc02 pancreatic ductal adenocarcinoma cell line (purchased from Procell, Cat# CL-0736) were respectively cultivated in RPMI 1640 medium (Gibico) supplemented with 10% FBS (Bioind) and DMEM (Gibco) supplemented with 10% FBS (Bioind) at 37 °C in a humidified atmosphere with 5% CO2.
Female BALB/c and male C57BL/6 mice were purchased from Yangzhou University (Yangzhou, China) and were 6–8 weeks old at the beginning of each experiment. All the mice were fed in the pathogen-free facility under a 12 h light/dark cycle at 20–26 °C and 40–70% humidity, with free access to food and water.
Ethics statement
All animal studies were performed under the guidelines for the care and use of laboratory animals in research approved by China Pharmaceutical University (SYXK(Su)2023-0018) and in accordance with all relevant ethical regulations. According to the institutional ethical regulations, subcutaneous tumors must not exceed a maximal volume of 1500 mm³. We confirm that no experimental animals exceeded this limit during the study.
Preparation of IL-12-AMCA-DSPE and IL-12-Cp-DSPE
To visualize the IL-12 anchorage on RBC in vitro and in situ retention of IL/aC@RBAH in vivo, IL-12 was respectively labeled with AMCA and Cypate. Briefly, AMCA (2.00 mg, 8.58 μmol) or Cypate (5.32 mg, 8.58 μmol) was dissolved in DMSO (100 μL). Then, NHS (1.19 mg, 10.30 μmol) and EDCI (1.97 mg, 10.30 μmol) were added and reacted at room temperature for 2 h to afford activated AMCA or Cypate. Subsequently, 5 mg of IL-12 was added into carbonate buffer solution at pH 9.2 (250 μL) and mixed with 25 μL of activated AMCA/Cypate. After stirring in dark overnight at 4 °C, the as-prepared IL-12-AMCA and IL-12-Cp were respectively ultrafiltered (3.5 kD molecular weight cut off (MWCO)), Millipore Sigma) to clear away the free AMCA/Cypate. Subsequently, IL-12-AMCA or IL-12-Cp was mixed with sulfo-SMCC (33 μg, 0.075 μmol) in PBS for 2 h, and further ultrafiltered (3.5 kD MWCO, Millipore Sigma) to afford Mal-IL-12-AMCA or Mal-IL-12-Cp.
5 mg of DSPE-PEG2000-SH was dissolved in DMSO (50 μL) and Mal-IL-12-AMCA or Mal-IL-12-Cp was dissolved in PBS (500 μL). 1.8 μL of DSPE-PEG2000-SH was subsequently added into Mal-IL-12-AMCA or Mal-IL-12-Cp solution and stirred at room temperature for 2 h. Finally, the resultant DSPE-PEG2000-IL-12-AMCA or DSPE-PEG2000-IL-12-Cp product was concentrated via ultrafiltration (3.5 kD MWCO, Millipore Sigma) and stored at 4 °C for use.
Preparation of aCTLA-4-FITC
100 μg of aCTLA-4 was dissolved in Na2CO3-NaHCO3 buffer at pH 9.0 (0.1 M, 100 μL). After 10 min of stirring at 4 °C, 1.03 μg of FITC was added and further stirred at 4 °C for 18 h. Finally, NH4Cl (5 M, 1 μL) was added to cease the reaction. Free FITC was removed by ultrafiltration (3.0 kD molecular weight cut off (MWCO), Millipore Sigma) with the endpoint as determined by the visual clarity of the filtrate.
Preparation of IL/aC@RBC
aC@RBC was prepared via an optimized “alternate hypotonic and hypertonic dialysis” approach as reported71. Briefly, the whole blood was collected from healthy BALB/c mice and mixed with PBS buffer with 5% EDTA. The obtained RBC was isolated by centrifugation (800 × g, 4 °C, 5 min) three times and resuspended in PBS buffer at 70% hematocrit. Then 500 μL of RBC suspension and 85 μL of FITC labeled aCTLA-4 (aCTLA-4-FITC, 5 mg/mL) were mixed and dialyzed (3.5 kD MWCO) against 40 mL of hypotonic buffer (deionized water containing 10 mM NaHCO3, 10 mM NaH2PO4, 20 mM glucose, 2 mM ATP, and 3 mM reduced glutathione (GSH)) at 4 °C for 90 min. Subsequently, the buffer was replaced with 45 mL of PBS buffer for 5 min of balance and then dialyzed with 50 mL of hypertonic buffer containing 10% PIGPA-NaCl buffer (deionized water containing 100 mM sodium pyruvate, 100 mM inosine, 100 mM glucose, 33 mM NaH2PO4, 12% (w/v) NaCl, and 2 mM ATP, pH 7.4) at 37 °C for 30 min. The resealed aC@RBC was collected by centrifugation (1000 × g, 4 °C, 10 min) and washed 3 times with PBS buffer to remove unentrapped aCTLA-4.
For efficient IL-12 anchorage and aCTLA-4 encapsulation in RBC for IL/aC@RBC, the loading capacities were investigated by varying the feeding time points of aCTLA-4.
IL/aC@RBC-1 was synthesized as follows: 500 μL of RBC suspension (4 × 109/mL), 85 μL of aCTLA-4-FITC (5 mg/mL), and 100 μL of DSPE-PEG2000-IL-12-AMCA (5 mg/mL) were mixed, and then dialyzed (3.5 kD MWCO) against 40 mL of hypotonic buffer. Subsequently, the buffer was replaced with 35 mL of PBS buffer for balance and then dialyzed with 50 mL of hypertonic buffer for further 30 min. The resealed IL/aC@RBC was collected by centrifugation (1000 × g, 4 °C, 10 min) and washed 3 times with PBS buffer to remove unentrapped aCTLA-4-FITC and DSPE-PEG2000-IL-12.
IL/aC@RBC-2 was synthesized as follows: 500 μL of RBC suspension (4 × 109/mL) was mixed with 85 μL of aCTLA-4-FITC (5 mg/mL) and then dialyzed (3.5 kD MWCO) against 40 mL of hypotonic buffer. Subsequently, the buffer was substituted with 35 mL of PBS buffer for balance. The resultant aC@RBC was added with 100 μL of DSPE-PEG2000-IL-12-AMCA (5 mg/mL) and dialyzed by 50 mL of hypertonic buffer. The resealed IL/aC@RBC was collected by centrifugation (1000 × g, 4 °C, 10 min) and washed 3 times with PBS buffer to remove unentrapped aCTLA-4-FITC and DSPE-PEG2000-IL-12.
IL/aC@RBC-3 was synthesized as follows: 500 μL of RBC suspension (4 × 109/mL) and 85 μL of aCTLA-4-FITC (5 mg/mL) were mixed and then dialyzed (3.5 kD MWCO) against 40 mL of hypotonic buffer. Subsequently, the buffer was substituted with 35 mL of PBS buffer for balance and then dialyzed by 40 mL of hypertonic buffer. The resealed aC@RBC was collected by centrifugation (1000 × g, 4 °C, 10 min) and washed 3 times with PBS buffer to remove unentrapped aCTLA-4-FITC. Then the resultant aC@RBC was added with 100 μL of IL-12-AMCA-DSPE (5 mg/mL) and incubated in PBS for IL-12 intercalation at 37 °C. The engineered IL/aC@RBC was collected by centrifugation (1000 × g, 4 °C, 10 min) and washed 3 times with PBS buffer to remove unentrapped DSPE-PEG2000-IL-12.
In vitro bioactivity assessment of Catalase (CAT) and IL-12
For CAT bioactivity evaluation, native RBC (nRBC) and RBC membrane (RBCM) were extracted as previous studies. Briefly, the whole blood was harvested from healthy BALB/c mice, collected in a tube, and then centrifuged (800 × g, 4 °C, 5 min) to discard plasma, leukocytes, and platelets in the upper layers. The precipitates were resuspended and washed three times by pre-cooled PBS buffer for nRBC. The resultant nRBC was diluted 40 times by pre-cooled lysate (0.01 M Tris-HCl, pH 8.0) containing 0.02 mM PMSF and stood at 4 °C for 2 h. Subsequently, RBCM was obtained by centrifugation (10,000 × g, 4 °C, 30 min), washed three times, and stored at −80 °C for further use. RBC and IL/aC@RBC were prepared via the optimized “alternate hypotonic and hypertonic dialysis” approach as introduced in “preparation of IL/aC@RBC”. Finally, the catalase bioactivities of nRBC, RBC, RBCM, and IL/aC@RBC were respectively assessed by Catalase Assay Kit (Beyotime, Shanghai, China), and normalized by the whole protein in the samples. In addition, the O2 generation from IL/aC@RBC and native RBC (nRBC) were quantitatively investigated to affirm CAT reservation on IL/aC@RBC. Briefly, 109 cells of IL/aC@RBC and nRBC were respectively dispersed in 5 mL of PBS buffer containing 500 μM H2O2. Then, the changes in O2 concentration were determined in 2 min by a dissolved oxygen analyzer.
For IL-12 bioactivity assessment, splenocytes were extracted for the evaluation of IFN-γ production stimulated by free IL-12 and IL@RBC. Briefly, spleens were collected from healthy BALB/c mice. The splenocytes were extracted and processed to remove RBC by lysis buffer (Beyotime, Shanghai, China). The obtained cells were seeded in a 96-well plate and subjected to free IL-12 or IL@RBC at pre-designated concentrations at 37 °C for 48 h. The culture supernatants were respectively collected for IFN-γ evaluation by ELISA (Mouse IFN-γ ELISA, Elabscience), and normalized by the whole protein in the samples.
Preparation of BaO2
Polyvinyl pyrrolidone (PVP, 0.35 × g, 6.03 μmol) was ultrasonically dissolved in 15 mL of methanol, followed by the addition of BaCl2 (0.1 × g, 0.48 mmol) and further stirring for complete dissolution. Then 20 μL of choline hydroxide (44% wt) was added to the solution. After 30 min of reaction, the mixture was dripped with 6 mL of 3% H2O2 at a constant rate of 0.05 mL/min and then stirred at room temperature for 1 h. The resultant BaO2 products were collected by centrifugation (10,000 × g, 4 °C, 15 min). After discarding the supernatant, the precipitates were resuspended by 15 mL of methanol and washed three times to afford white precipitates for further use.
Determination of Ba2+ and H2O2 generation from BaO2
In vitro release of Ba2+ from BaO2 was measured using the dialysis bag diffusion method. Typically, 1 mg of BaO2 nanoparticles in 1 mL of PBS (pH 7.4) were respectively dialyzed (1 kD MWCO) against 19 mL of PBS at pH 7.4 and pH 6.5. The dialysis was performed at 37 °C with a shaking speed of 100 rpm. At pre-designed time points of 0, 0.5, 1, 2, 4, 6, 8, 12, 24, 36, and 48 h, 1 mL of dialysate was collected and replaced with 1 mL of fresh medium. The obtained samples were diluted with 3% HNO3 and the Ba2+ concentration was quantified by Inductively Coupled Plasma-Mass Spectrometry (ICP-MS, iCAP RQplus, Thermo Scientific, USA).
To evaluate the acidity-induced generation of H2O2, 100 μL of BaO2 nanoparticles (0.1 mg/mL) was incubated with horseradish peroxidase (HRP) in 1.5 mL of PBS at pH 7.4 and pH 6.5, respectively. At pre-designed time points of 0, 4, 10, 20, and 30 min, 200 μL of tetramethylbenzidine (TMB) was added for 2–3 min of incubation. The absorption at 652 nm was measured by UV–vis spectrophotometer (3J1-0015, HITACHI, Japan) and H2O2 production was calculated according to the established standard curve of commercial H2O2 (0.05-1.0 mM).
Rheological test
Rheological characterizations of IL/aC@RBAH were observed using a rheometer (Discovery HR-2-TA, TA Instrument, American) with a parallel-plate geometry of 12 mm diameter and 0.6 mm gap at 25 °C. The changes in storage modulus and loss modulus of IL/aC@RBAH were monitored at a constant strain of 2.5% and a fixed frequency of 1 Hz for 20 min. Strain amplitude sweeps were conducted from (1% to 1000%) at a fixed frequency of 1 Hz. Dynamic oscillatory frequency sweeps were performed from 1 to 32 rad/s at a constant strain of 2.5% to estimate the elastic and viscous properties of IL/aC@RBAH.
In vitro degradation determination of IL/aC@RBAH hydrogel
The degradation profile of IL/aC@RBAH was investigated relying on the mass loss during the degradation process. Typically, BaO2 (0.1 mg), 3% Alg (100 μL), and IL/aC@RBC (2 × 108 cells in 100 μL) were mixed and incubated in PBS at pH 6.5 for IL/aC@RBAH formation. Afterward, the hydrogel was washed by deionized water three times and then dried under vacuum at −40 °C for 4–6 h. The initial mass of lyophilized IL/aC@RBAH was indicated as W0. Subsequently, the lyophilized IL/aC@RBAH was swollen in PBS at pH 6.5 and incubated at 37 °C. At designated time points on the 0.25, 0.5, 1.0, 1.5, 2, 4, 6, 8, 10, and 14 days, the hydrogel was collected, dried, and weighed (Wd). The degradation percentage (D%) of IL/aC@RBAH was calculated according to the formula as follows:
Release of IL-12, aCTLA-4 and Ba2+ from IL/aC@RBAH
IL-12 and aCTLA-4 in IL/aC@RBAH were respectively labeled with AMCA and FITC to obtain IL-12-AMCA and aCTLA-4-FITC. The release behaviors of IL-12 and aCTLA-4 were examined according to the previous report72. A 40 µm cell strainer containing IL/aC@RBAH was embedded in a six-well plate with 5 mL of PBS at pH 6.5 and pH 7.4. After 0, 1, 2, 4, 6, 8, 16, 24, and 48 h of incubation, the concentrations of IL-12-AMCA and aCTLA-4-FITC in PBS were respectively detected by a microplate reader (Molecular Devices, USA) at the absorption wavelength of 350 nm and 488 nm.
In vitro release of Ba2+ from IL/aC@RBAH was investigated via the dialysis method. Briefly, IL/aC@RBAH formed in PBS (pH 6.5) was washed with deionized water three times, and then 1 mL of the sample was dialyzed (3 kD MWCO) against 19 mL of PBS (pH 6.5) at 37 °C in a shaking culture incubator. After 0, 1, 2, 4, 6, 8, 16, 24, and 48 h of incubation, 1 mL of dialysate was taken and replenished with an equal volume of fresh buffer. The released Ba2+ amount was determined by ICP-MS (iCAP RQplus, Thermo Scientific, USA).
In vitro biosafety evaluation
For the cytotoxicity analysis of BaO2, 4 × 103 4T1 cells and Panc02 cells were plated in each well of 96-well plates and incubated at 37 °C for 24 h. Different concentrations of BaO2 (0, 20, 40, 60, 80, 100, and 150 μg/mL) were added for 24 h of incubation. Afterward, MTT solution (0.5 mg/mL) was added and cocultured at 37 °C for 4 h. Then medium containing MTT was removed and replaced with 150 μL of DMSO in each well. After 30 min of constant temperature shaking at 37 °C, the optical density (OD) at 490 nm was detected. The experiment was repeated independently 5 times and the cell viability was calculated according to the formula: cell viability (%) = ODsample/ODcontrol × 100%.
For the biocompatibility evaluation of IL/aC@RBAH, hydrogel leaching solution was collected and diluted into different concentrations (0, 6.25%, 12.5%, 25%, 50%, and 100%). 4T1 cells and Panc02 cells were seeded in 96-well plates and incubated at 37 °C. After 24 h of culture, hydrogel leaching solutions at various concentrations were added and cocultured for 24 h. Subsequently, MTT solution (0.5 mg/mL) was added and incubated at 37 °C for 4 h. Then medium containing MTT was removed and replaced with 150 μL of DMSO in each well. After 30 min of constant temperature shaking at 37 °C, the OD at 490 nm was detected. The experiment was repeated independently 5 times for statistical analysis.
IL/aC@RBAH cytotoxicity was further investigated by live/dead cell staining assay. Briefly, sterile IL/aC@RBAH was placed in 48-well plates and 1 × 104 4T1 cells were seeded in the wells containing hydrogels, followed by 24 h of incubation at 37 °C. Subsequently, the cells were stained with calcein-AM (1 μg/mL) and PI (1 μg/mL) for confocal laser scanning microscopy (CLSM, Olympus Corporation, Japan) observation.
Colony formation assay
The cell viability exposed to different treatments was investigated via colony formation assay. Typically, one hundred cells were seeded in culture dishes (60 mm in diameter) and incubated at 37 °C for 24 h. Subsequently, cells were respectively subjected to saline, RT, RT + BaO2, RT + IL/aC@RBC, and RT + IL/aC@RB. The X-ray irradiation intensity was 1 Gy. Then cells were incubated for 14 days and replaced with fresh culture medium every 2–3 days. After colony formation in each dish, the cells were washed with PBS several times and fixed by 4% polyformaldehyde for 30 min at room temperature. Finally, colonies were dyed with 0.1% crystal violet for 30 min and washed by PBS for visual observation. The colonies were counted and calculated for survival fraction analysis.
Detection of intracellular ROS production
The ROS production inside cells was investigated by Reactive Oxygen Species Assay Kit (DCFH-DA, ROS fluorescence probe). 4T1 cells were cultured in confocal dishes and 12-well plates for 24 h at 37 °C and then added with DCFH-DA solution (10 μM). Subsequently, cells were treated with saline, RT, RT + BaO2, RT + IL/aC@RBC, and RT + IL/aC@RB, respectively cells and then incubated with DCFH-DA solution (10 μM) for 20 min at 37 °C. Afterward, the cells in 12-well plates were trypsinized and collected for flow cytometry. Meanwhile, the cells in confocal dishes were washed and stained with DAPI for observation by CLSM (Olympus Corporation, Japan).
In vivo degradation tests and in situ retention tests
In vivo degradation tests: The mixture of Ba2+, IL/aC@RBC, and Alg was injected into the right thorax of female BALB/c mice (6–8 weeks old, n = 3 mice per group). On days 7 and 14 after in situ administration, a small incision of about 1 cm was created on the injected site, and the residual hydrogel was photographed.
In situ retention tests: For real-time monitoring of in situ retention of IL/aC@RBAH in female BALB/c mice (6–8 weeks old, n = 3 mice per group), the IL-12 was pre-labeled by Cypate for IL-Cp&aC, IL-Cp/aC@RBC, and IL-Cp/aC@RBAH. At 1, 2, 3, 5, 7, 9, 11, and 14 days after subcutaneous injection and intratumoral injection, the fluorescence intensities were monitored by the IVIS Spectrum In Vivo Imaging System (IVIS Lumina XR, USA).
In vivo safety analysis
For cytokine analysis of serum, the orthotopic 4T1 tumor-bearing mice were intratumorally injected with saline, IL-12, aCTLA-4, and IL/aC@RBAH (n = 3 mice per group), respectively. After 4 h, the blood samples were collected and centrifuged (800 × g, 4 °C, 10 min) to remove cells. The concentrations of IL-12 and aCTLA-4 in the serum samples were determined by IL-12 and aCTLA-4 ELISA kit, and measured by microplate reader (Molecular Devices, USA).
For blood biochemical analysis, the orthotopic 4T1 tumor-bearing mice were intratumorally injected with saline, IL/aC, RBAH, and IL/aC@RBAH (n = 3 mice per group), respectively. After 3 days, the blood samples were acquired for quiescence at 4 °C overnight and further centrifugation (800 × g, 4 °C, 10 min) to remove cells. The serum samples were collected and sent to Servicebio Biotechnology Co., Ltd for analysis of liver function (Aspartate aminotransferase, ALT; Alanine aminotransferase, AST) and renal function (Creatinine, CREA; Blood urea nitrogen, BUN). The major organs (heart, liver, spleen, lung, and kidney) from the mice subjected to different treatments were collected for H&E staining analysis.
For long-term biosafety analysis, healthy BALB/c mice were randomly allocated into 4 groups (n = 3 mice per group) and subcutaneously injected with saline, IL&aC (the mixture of IL-12 and aCTLA-4), RB (the mixture of RBC and BaO2), and IL/aC@RBAH once a week. During 2 months of continuous treatments, the body weights of the mice were recorded every 7 days. After 2 months of monitoring, the blood and major organs were collected. Liver damage indexes involving alanine aminotransferase (ALT) and aspartate aminotransferase (AST), as well as kidney function indexes compromising blood urea nitrogen (BUN) and creatinine (CREA), were respectively detected. In addition, the major organs, including heart, liver, spleen, lung, and kidney, were collected for H&E analysis.
Evaluation of intratumoral oxygen contents
The female BALB/c mice (6–8 weeks old, n = 3 mice per group) were orthotopically inoculated with 4T1 cells (1 × 106 cells per mouse) to establish a 4T1 mouse breast tumor model. When the tumor volume reached approximately 100 mm3, the mice were subjected to treatments of IL/aC@RBC or IL/aC@RB. For quantification of intratumoral vascular oxygen saturation levels, the ratios of deoxygenated hemoglobin (λ = 750 nm) versus oxygenated hemoglobin (λ = 850 nm) were recorded, and the 2D photoacoustic (PA) images were monitored by PA imaging system (Vevo® LAZR, Fujifilm VisualSonics, Canada). In addition, for oxygen production sustainability evaluation, the orthotopic 4T1 tumor-bearing mice were respectively treated with IL/aC@RB and IL/aC@RBAH. At 0, 24, and 48 h post-treatment, the tumors were photographed by PA imaging system (Vevo® LAZR, Fujifilm VisualSonics, Canada) and the intratumoral vascular oxygen saturation levels were recorded.
For further evaluation of the intratumoral hypoxia relief, the orthotopic 4T1 tumor-bearing female BALB/c mice (6–8 weeks old, n = 3 mice per group) were respectively treated with saline, BaO2, IL/aC@RBC, IL/aC@RB, and IL/aC@RBAH. After 24 h, animals were sacrificed, and the tumors were collected. Subsequently, these tissues were successively frozen, sectioned, air-dried, fixed in acetone, and blocked. Immunofluorescence staining of HIF-1α and CD31 was conducted and then observed by CLSM (Olympus Corporation, Japan).
In vivo antitumor study on 4T1 tumor models and Panc02 tumor models
For orthotopic 4T1 tumor therapy, 1 × 106 4T1 expressing luciferase (4T1-luc) cells were injected into the left mammary fat pad of female BALB/c mice (6–8 weeks old, n = 5 mice per group). After the tumor volume reached 100 mm3, the mice were randomly divided into 6 groups (5 mice per group) and respectively treated with saline (G1), RT (G2), RT + RBAH (G3), RT + aC@RBAH (G4), RT + IL@RBAH (G5), and RT + IL/aC@RBAH (G6) (an equivalent aCTLA-4 amount of 500 μg/kg, IL-12 amount of 250 μg/kg, BaO2 amount of 150 μg/kg, and Alg amount of 3%). The X-ray irradiation treatment (8 Gy) in the pre-designed groups was imposed twice at the tumor sites after 0 and 2 days of in situ administration. On the 7, 14, and 21 days after the tumor inoculation, the tumor growth was monitored by the IVIS Spectrum In Vivo Imaging System (IVIS Lumina XR, USA). The tumor sizes and body weight were recorded every 2 days. The survival of mice was monitored over a 2-month period. The tumor volume (V) was calculated following the formula V = 0.5 × W2 × L, where L represents the long diameter of the tumor and W represents the width of the tumor. The tumors from the mice were collected for histological analysis, including H&E staining and Ki67 staining.
For evaluating the antitumor efficacy of RT + IL/aC@RBAH-induced multistage RIT, an orthotopic 4T1 tumor model was established. After the tumor volume reached 100 mm3, the female BALB/c mice were randomly divided into 6 groups (6–8 weeks old, n = 5 mice per group) and respectively treated with saline, RT, RT + RBAH, IL&aC, RT + IL&aC, and RT + IL/aC@RBAH (an equivalent aCTLA-4 amount of 500 μg/kg, IL-12 amount of 250 μg/kg, BaO2 amount of 150 μg/kg, and Alg amount of 3%). The X-ray irradiation treatment (8 Gy) in the pre-designed groups was imposed twice at the tumor sites after 0 and 2 days of in situ administration. On the 7, 14, and 21 days after the tumor inoculation, the tumor growth was monitored by the IVIS Spectrum In Vivo Imaging System. The tumor sizes and survival time were also recorded. The tumor volume (V) was calculated following the formula V = 0.5 × W2 × L.
For orthotopic 4T1 tumor relapse, 1 × 106 4T1 cells were injected into the left mammary fat pad of female BALB/c mice (6–8 weeks old, n = 5 mice per group). After 10 days, the mice were treated with RT + IL/aC@RBAH. Following 2 more days, the mice were re-treated with RT. On day 26, the mice presenting CR after RT + IL/aC@RBAH treatment were rechallenged with 1 × 106 4T1 cells into the right mammary fat pad. Meanwhile, healthy mice were orthotopically injected with 1 × 106 4T1 cells into the right flank of mice as the control. The tumor growth was tracked every 2 days and calculated following the formula V = 0.5 × W2 × L.
For comparing the antitumor efficacy between RT + aPD-1/aPD-L1 and RT + IL/aC@RBAH, an orthotopic 4T1 tumor model was established in female BALB/c mice. After the tumor volume reached 100 mm3, the mice were randomly divided into 4 groups (6–8 weeks old, n = 5 mice per group) and respectively treated with saline, RT + aPD-1 (10 mg/kg), RT + aPD-L1 (10 mg/kg), and RT + IL/aC@RBAH. The X-ray irradiation treatment (8 Gy) in the pre-designed groups was imposed twice at the tumor sites after 0 and 2 days of in situ administration. On the 7, 14, and 21 days after the tumor inoculation, the tumor growth was monitored by the IVIS Spectrum In Vivo Imaging System. The tumor growth was tracked every 2 days and calculated following the formula V = 0.5 × W2 × L.
For subcutaneous Panc02 tumor treatment, 1 × 106 Panc02 cells were injected into the right back of male C57BL/6 mice (6–8 weeks old, n = 5 mice per group). When subcutaneous tumor volume reached approximately 100 mm3, the mice were subjected to different treatments as designed in the orthotopic 4T1 tumor therapy. On days 7, 14, and 21 after the tumor inoculation, the tumor growth was monitored in real-time by the IVIS Spectrum In Vivo Imaging System (IVIS Lumina XR, USA). The tumor sizes and survival time were also tracked every 2 days. The tumors were taken for histological analysis, including H&E staining and TUNEL staining.
In vivo antitumor immune response analysis
For localized and systemic immune response analysis, orthotopic 4T1 tumor models were established in female BALB/c mice, and subcutaneous Panc02 tumor models were established in male C57BL/6 mice. When the tumors reached approximately 100 mm3, the mice were randomly divided into 6 groups (n = 3 mice per group) and treated with saline, RT, RT + RBAH, RT + aC@RBAH, RT + IL@RBAH, and RT + IL/aC@RBAH, respectively. Tumors and TDLNs were both collected 4 days after different treatments. The tissues were rinsed by PBS, minced and digested by Hank’s buffer containing DNase (0.1 mg/mL) and collagenase (1 mg/mL) for 45 min at 37 °C. Then the cells were filtered through 70 μm nylon cell strainers and centrifuged (500 × g, 4 °C, 5 min) to obtain single-cell suspensions. Afterward, 1 mL of erythrocyte lysate was added to each sample and lysed for 3 min at room temperature. Then, the cells were washed, blocked by anti-CD16/32 antibody for 5 min and stained with fixable viability stain 510 for 15 min at room temperature. Subsequently, fluorescein-labeled antibodies were added and incubated at 4 °C for 30 min, followed by washing with PBS. For the intracellular staining, the cell suspension was fixed and permeabilized by the fixation/permeabilization buffer (Thermo Fisher Scientific). Then the processed cells were stained with intracellular fluorescein-labeled antibodies and detected by flow cytometry.
For multistage immune effect analysis, the immune cell populations in orthotopic 4T1 tumor-bearing mice exposed to different treatments were investigated. Briefly, the orthotopic 4T1-tumor bearing female BALB/c mice were randomly divided into 5 groups (n = 3 mice per group) and then respectively treated with saline, RT, RT + IL-aC (IL-12 prior to aCTLA-4), RT + IL&aC (the mixture of IL-12 and aCTLA-4), and RT + aC-IL (IL-12 subsequent to aCTLA-4). Tumors and TDLNs were collected at 4 days after different treatments. The samples were processed as described above. Afterward, the processed cells were stained with intracellular fluorescein-labeled antibodies and detected by flow cytometry.
To further elucidate the antitumor efficacy between RT + IL&aC and RT + IL/aC@RBAH, the immune cell populations in orthotopic 4T1 tumor-bearing female BALB/c mice exposed to different treatments were investigated. Briefly, the orthotopic 4T1-tumor bearing mice were randomly divided into 3 groups (n = 3 mice per group) and then respectively treated with saline, RT + IL&aC, and RT + IL/aC@RBAH. The tumors were collected at 4 days after different treatments. The samples were processed as described above. Subsequently, the processed cells were stained with intracellular fluorescein-labeled antibodies and detected by flow cytometry.
For long-term immune memory evaluation, the orthotopic 4T1 re-challenge model was established as designed above. Briefly, 1 × 106 4T1 cells were injected into the left mammary fat pad of female BALB/c mice (n = 3 mice per group). After 10 days, the mice were treated with RT + IL/aC@RBAH. The X-ray irradiation treatment (8 Gy) was imposed twice at the tumor sites after 0 and 2 days of the treatment. On day 26, the mice presenting complete tumor regression after RT + IL/aC@RBAH treatment were rechallenged with 1 × 106 4T1 cells into the right mammary fat pad. Meanwhile, healthy mice were orthotopically injected with 1 × 106 4T1 cells into the right flank of mice as the control. On day 52, the spleens were collected and processed as above for analysis by flow cytometry.
To compare the antitumor efficacy between RT + aPD-1/aPD-L1 and RT + IL/aC@RBAH, the immune cell populations in orthotopic 4T1 tumor-bearing female BALB/c mice exposed to different treatments were investigated. Briefly, the orthotopic 4T1-tumor bearing mice were randomly divided into 4 groups (n = 3 mice per group) and then respectively treated with saline, RT + aPD-1 (10 mg/kg), RT + aPD-L1 (10 mg/kg), and RT + IL/aC@RBAH. The tumors and TDLNs were collected at 4 days after different treatments. The samples were processed as described above. Afterward, the processed cells were stained with intracellular fluorescein-labeled antibodies and detected by flow cytometry.
Transcriptome analysis
An orthotopic 4T1 tumor model was established for transcriptome analysis. Briefly, 1 × 106 4T1 cells were injected into the left mammary fat pad of female BALB/c mice. After the tumor size reached 100 mm3, the orthotopic 4T1 tumor-bearing mice were randomly divided into 3 groups (n = 3 mice per group) and exposed to different treatments of saline, RT, and RT + IL/aC@RBAH, respectively. The tumor tissues were harvested on day 3 after treatments. Total RNA was extracted for transcriptome sequencing and analysis containing correlation analysis, differential analysis, GO analysis, KEGG analysis, and GSEA analysis on a Novogene platform (https://magic.novogene.com/).
Statistics and reproducibility
All numeric data are presented as mean ± SD deviation from at least three parallel experiments. No data were excluded from the analyses. The significance between two groups was determined by the two-sided unpaired students’ t-test. Two-way analysis of variance (ANOVA) with Turkey’s post hoc test was employed for multiple comparisons. Survival analysis was performed by log-rank (Mantel–Cox) test. All statistical analyses were performed using GraphPad Prism (v9.5.0). P values of less than 0.05 were considered significant. All flow cytometry data were analyzed on NovoExpress (v1.5.0, Agilent Technologies) and FlowJo software (v10, BD Biosciences). Living imaging software (v4.4, Perkin Elmer) was used to analyse bioluminescent and fluorescent images. Microscopic images were analyzed by OlympusMicro (FV30S-SW) and ImageJ (Version 1.52 k). Sex was not considered as a biological variable within each tumor model because 4T1 studies used only female mice and Panc02 studies used only male mice. Therefore, sex-disaggregated analysis within models is not applicable.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The experimental data generated in this study are available in the main article and supplementary information. The RNA-Seq data generated in this study have been deposited in the NCBI Gene Expression Omnibus database under the accession code PRJNA1283550. All other data supporting this work are available in the main article, supplementary information or source data file. Source data are provided with this paper.
References
Kong, X. et al. Revolutionizing the battle against locally advanced breast cancer: a comprehensive insight into neoadjuvant radiotherapy. Med. Res. Rev. 44, 606–631 (2024).
Vozenin, M. C., Bourhis, J. & Durante, M. Towards clinical translation of FLASH radiotherapy. Nat. Rev. Clin. Oncol. 19, 791–803 (2022).
Yi, G. et al. DNA polymerase θ-mediated repair of high LET radiation-induced complex DNA double-strand breaks. Nucleic Acids Res. 51, 2257–2269 (2023).
Herrera, F. G., Bourhis, J. & Coukos, G. Radiotherapy combination opportunities leveraging immunity for the next oncology practice. CA Cancer J. Clin. 67, 65–85 (2017).
Beckers, C., Pruschy, M. & Vetrugno, I. Tumor hypoxia and radiotherapy: a major driver of resistance even for novel radiotherapy modalities. Semin Cancer Biol. 98, 19–30 (2024).
Chen, Z., Han, F., Du, Y., Shi, H. & Zhou, W. Hypoxic microenvironment in cancer: molecular mechanisms and therapeutic interventions. Signal Transduct. Target Ther. 8, 70 (2023).
Liu, J. et al. Synergistic cerium oxide nanozymes: targeting DNA damage and alleviating tumor hypoxia for improved NSCLC radiotherapy efficiency. J. Nanobiotechnol. 22, 25 (2024).
Zhang, Y. et al. CD39 inhibition and VISTA blockade may overcome radiotherapy resistance by targeting exhausted CD8+ T cells and immunosuppressive myeloid cells. Cell Rep. Med. 4, 101151 (2023).
Persa, E., Balogh, A., Sáfrány, G. & Lumniczky, K. The effect of ionizing radiation on regulatory T cells in health and disease. Cancer Lett. 368, 252–261 (2015).
Esmaily, M. et al. Blockade of CTLA-4 increases anti-tumor response inducing potential of dendritic cell vaccine. J. Control Release 326, 63–74 (2020).
Ribas, A. & Wolchok, J. D. Cancer immunotherapy using checkpoint blockade. Science 359, 1350–1355 (2018).
Liu, Y. & Zheng, P. Preserving the CTLA-4 checkpoint for safer and more effective cancer immunotherapy. Trends Pharm. Sci. 41, 4–12 (2020).
Jenkins, K. A. et al. XTX101, a tumor-activated, Fc-enhanced anti-CTLA-4 monoclonal antibody, demonstrates tumor-growth inhibition and tumor-selective pharmacodynamics in mouse models of cancer. J. Immunother. Cancer 11, e007785 (2023).
van Pul, K. M. et al. Local delivery of low-dose anti-CTLA-4 to the melanoma lymphatic basin leads to systemic T(reg) reduction and effector T cell activation. Sci. Immunol. 7, eabn8097 (2022).
Davis-Marcisak, E. F. et al. Transfer learning between preclinical models and human tumors identifies a conserved NK cell activation signature in anti-CTLA-4 responsive tumors. Genome Med. 13, 129 (2021).
Formenti, S. C. et al. Radiotherapy induces responses of lung cancer to CTLA-4 blockade. Nat. Med. 24, 1845–1851 (2018).
Pointer, K. B., Pitroda, S. P. & Weichselbaum, R. R. Radiotherapy and immunotherapy: open questions and future strategies. Trends Cancer 8, 9–20 (2022).
Kalbasi, A. & Ribas, A. Tumour-intrinsic resistance to immune checkpoint blockade. Nat. Rev. Immunol. 20, 25–39 (2020).
Langrish, C. L. et al. IL-12 and IL-23: master regulators of innate and adaptive immunity. Immunol. Rev. 202, 96–105 (2004).
Nguyen, K. G. et al. Localized interleukin-12 for cancer immunotherapy. Front. Immunol. 11, 575597 (2020).
Ertel, W. et al. Inhibition of the defense system stimulating interleukin-12 interferon-gamma pathway during critical Illness. Blood 89, 1612–1620 (1997).
Azar, F. et al. TG6050, an oncolytic vaccinia virus encoding interleukin-12 and anti-CTLA-4 antibody, favors tumor regression via profound immune remodeling of the tumor microenvironment. J. Immunother. Cancer 12, e009302 (2024).
Eisenbeis, C. F. et al. Phase I study of the sequential combination of interleukin-12 and interferon alfa-2b in advanced cancer: evidence for modulation of interferon signaling pathways by interleukin-12. J. Clin. Oncol. 23, 8835–8844 (2005).
Raucci, M. G., D’Amora, U., Ronca, A. & Ambrosio, L. Injectable functional biomaterials for minimally invasive surgery. Adv. Health. Mater. 9, e2000349 (2020).
Hussey, G. S., Dziki, J. L. & Badylak, S. F. Extracellular matrix-based materials for regenerative medicine. Nat. Rev. Mater. 3, 159–173 (2018).
Shan, B. H. & Wu, F. G. Hydrogel-based growth factor delivery platforms: strategies and recent advances. Adv. Mater. 36, e2210707 (2024).
Zhang, D. et al. Architecturally designed sequential-release hydrogels. Biomaterials 303, 122388 (2023).
Xu, J. et al. A 3D bioprinted tumor model fabricated with gelatin/sodium alginate/decellularized extracellular matrix bioink. Int. J. Bioprint 9, 630 (2023).
Zhang, Y. et al. Highly conductive and tough polyacrylamide/sodium alginate hydrogel with uniformly distributed polypyrrole nanospheres for wearable strain sensors. Carbohydr. Polym. 315, 120953 (2023).
Karim, A. et al. Alginate-based nanocarriers for the delivery and controlled-release of bioactive compounds. Adv. Colloid Interface Sci. 307, 102744 (2022).
Sanchez-Ballester, N. M., Bataille, B. & Soulairol, I. Sodium alginate and alginic acid as pharmaceutical excipients for tablet formulation: structure-function relationship. Carbohydr. Polym. 270, 118399 (2021).
Ajayebi, F. S., Hassanzadeh Nemati, N., Hatamirad, A., Ghazli, M. & Attaran, N. Design and fabrication of alginate hydrogel nanohybrid as a promising cancer treatment. Iran. J. Basic Med. Sci. 27, 695–705 (2024).
Chen, P. et al. Photo-crosslinking modified sodium alginate hydrogel for targeting delivery potential by NO response. Int. J. Biol. Macromol. 253, 126454 (2023).
Wang, S. et al. Continuous and controllable electro-fabrication of antimicrobial copper-alginate dressing for infected wounds treatment. J. Mater. Sci. Mater. Med. 32, 143 (2021).
Melo, D. et al. Catalase, glutathione peroxidase, and peroxiredoxin 2 in erythrocyte cytosol and membrane in hereditary spherocytosis, sickle cell disease, and β-thalassemia. Antioxidants 13, 629 (2024).
Colvin, H. N. et al. An ex vivo model of oxidatively stressed red blood cells demonstrates a role for exogenous amino acids in enhancing red blood cell function and morphology. Blood 138, 922–922 (2021).
Hu, C. M. et al. Erythrocyte membrane-camouflaged polymeric nanoparticles as a biomimetic delivery platform. Proc. Natl. Acad. Sci. USA 108, 10980–10985 (2011).
Zhao, Z. et al. Capturing magnesium ions via microfluidic hydrogel microspheres for promoting cancellous bone regeneration. ACS Nano 15, 13041–13054 (2021).
Zhou, R. et al. Site occupancies, VUV-UV-vis photoluminescence, and X-ray radioluminescence of Eu(2 + )-doped RbBaPO(4). Inorg. Chem. 59, 17421–17429 (2020).
Wang, J. et al. BaHoF5 nanoprobes as high-performance contrast agents for multi-modal CT imaging of ischemic stroke. Biomaterials 71, 110–118 (2015).
Zhao, X. et al. Injectable antiswelling and high-strength bioactive hydrogels with a wet adhesion and rapid gelling process to promote sutureless wound closure and scar-free repair of infectious wounds. ACS Nano 17, 22015–22034 (2023).
Vázquez-González, M. & Willner, I. Stimuli-responsive biomolecule-based hydrogels and their applications. Angew. Chem. Int. Ed. Engl. 59, 15342–15377 (2020).
Behroozi Kohlan, T., Wen, Y., Mini, C. & Finne-Wistrand, A. Schiff base crosslinked hyaluronic acid hydrogels with tunable and cell instructive time-dependent mechanical properties. Carbohydr. Polym. 338, 122173 (2024).
Chen, T. et al. Rationally designed β-cyclodextrin-crosslinked polyacrylamide hydrogels for cell spheroid formation and 3D tumor model construction. Carbohydr. Polym. 339, 122253 (2024).
Sun, L. et al. Herceptin induces ferroptosis and mitochondrial dysfunction in H9c2 cells. Int. J. Mol. Med. 49, 17 (2022).
Chen, Q. et al. Nanoparticle-enhanced radiotherapy to trigger robust cancer immunotherapy. Adv. Mater. 31, e1802228 (2019).
Motzer, R. J. et al. Phase I trial of subcutaneous recombinant human interleukin-12 in patients with advanced renal cell carcinoma. Clin. Cancer Res. 4, 1183–1191 (1998).
Telarovic, I., Wenger, R. H. & Pruschy, M. Interfering with tumor hypoxia for radiotherapy optimization. J. Exp. Clin. Cancer Res. 40, 197 (2021).
Torrisi, F. et al. The role of hypoxia and SRC tyrosine kinase in glioblastoma invasiveness and radioresistance. Cancers 12, 2860 (2020).
Zhao, Y., Xing, C., Deng, Y., Ye, C. & Peng, H. HIF-1α signaling: essential roles in tumorigenesis and implications in targeted therapies. Genes Dis. 11, 234–251 (2024).
Zhang, H. et al. Maintaining hypoxia environment of subchondral bone alleviates osteoarthritis progression. Sci. Adv. 9, eabo7868 (2023).
Dai, W. et al. Hypoxia and the endometrium: an indispensable role for HIF-1α as therapeutic strategies. Redox Biol. 73, 103205 (2024).
Hou, Y. et al. Radiotherapy enhances metastasis through immune suppression by inducing PD-L1 and MDSC in distal sites. Clin. Cancer Res. 30, 1945–1958 (2024).
Wang, L. et al. Radiotherapy and immunology. J. Exp. Med. 221, e20232101 (2024).
Tekguc, M., Wing, J. B., Osaki, M. & Long, J. Sakaguchi S. Treg-expressed CTLA-4 depletes CD80/CD86 by trogocytosis, releasing free PD-L1 on antigen-presenting cells. Proc. Natl. Acad. Sci. USA 118, e2023739118 (2021).
Mahvi, D. A., Liu, R., Grinstaff, M. W., Colson, Y. L. & Raut, C. P. Local cancer recurrence: the realities, challenges, and opportunities for new therapies. CA Cancer J. Clin. 68, 488–505 (2018).
Liu, Q., Sun, Z. & Chen, L. Memory T cells: strategies for optimizing tumor immunotherapy. Protein Cell 11, 549–564 (2020).
McLane, L. M. et al. CD8 T cell exhaustion during chronic viral infection and cancer. Annu. Rev. Immunol. 37, 457–495 (2019).
Chow, A. et al. Clinical implications of T cell exhaustion for cancer immunotherapy. Nat. Rev. Clin. Oncol. 19, 775–790 (2022).
Baessler, A. et al. T Cell Exhaustion. Annu. Rev. Immunol. 42, 179–206 (2024).
Sumida, T. S. et al. Type I interferon transcriptional network regulates expression of coinhibitory receptors in human T cells. Nat. Immunol. 23, 632–642 (2022).
Wertheimer, T. et al. IL-23 stabilizes an effector Treg cell program in the tumor microenvironment. Nat. Immunol. 25, 512–524 (2024).
Kang, S. et al. Engineered IL-7 synergizes with IL-12 immunotherapy to prevent T cell exhaustion and promote memory without exacerbating toxicity. Sci. Adv. 9, eadh9879 (2023).
Zebley, C. C. et al. T cell dysfunction and therapeutic intervention in cancer. Nat. Immunol. 25, 1344–1354 (2024).
Zhang, Y. et al. Efficacy and safety of PD-1/PD-L1 checkpoint inhibitors versus anti-PD-1/PD-L1 combined with other therapies for tumors: a systematic review. Cancers 15, 682 (2023).
Nguyen, P. H. D. et al. Advances in drug delivery systems based on red blood cells and their membrane-derived nanoparticles. ACS Nano 17, 5187–5210 (2023).
Zhang, M. et al. Radiation-assisted metal ion interference tumor therapy by barium peroxide-based nanoparticles. Mater. Horiz. 6, 1034–1040 (2019).
Basu, T. et al. Cross-linker-free sodium alginate and gelatin hydrogels: a multiscale biomaterial design framework. J. Mater. Chem. B 10, 3614–3623 (2022).
McArdel, S. L. et al. Anti-tumor effects of RTX-240: an engineered red blood cell expressing 4-1BB ligand and interleukin-15. Cancer Immunol. Immunother. 70, 2701–2719 (2021).
Zhang, X. et al. Engineered red blood cells as an off-the-shelf allogeneic anti-tumor therapeutic. Nat. Commun. 12, 2637 (2021).
Robert, M. et al. Multiparametric characterization of red blood cell physiology after hypotonic dialysis based drug encapsulation process. Acta Pharm. Sin. B 12, 2089–2102 (2022).
Hu, Q. et al. Inhibition of post-surgery tumour recurrence via a hydrogel releasing CAR-T cells and anti-PDL1-conjugated platelets. Nat. Biomed. Eng. 5, 1038–1047 (2021).
Acknowledgements
This project is financially supported by the National Natural Science Foundation of China (82472157, 82302273, 92359304, 82330060, 82473791, 82150203, 32201127, 82270113, T2522033), the Natural Science Foundation of Jiangsu Province (BK20230839, BK20250102), the Specialized Research Funds from the State Key Laboratory of Natural Medicines, China Pharmaceutical University (SKLNMZZ2024JS36).
Author information
Authors and Affiliations
Contributions
Y.C. conceived and designed the concept of the experiments. Y.C. and Q.C. conducted the synthesis and characterization of materials. Y.C. and Y.M. conducted and performed in vitro and in vivo experiments. Y.C. and C.Z. analyzed data and verified the analytical methods. Y.C. and C.Z. provided important experimental insights and wrote the whole manuscript. J.X., K.L., Y.S., X.S., M.W., R.B., and T.D. aided in interpreting the results and worked on the manuscript. W.F., L.T., W.Y., and S.J. conceived the idea and supervised the entire project. All authors discussed, commented, and agreed on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Ke Cheng and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chen, Y., Chen, Q., Ma, Y. et al. In situ self-assembled cell reservoir hydrogel for maneuvering multistage radioimmunotherapy. Nat Commun 17, 1784 (2026). https://doi.org/10.1038/s41467-026-68490-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-026-68490-5