Abstract
Thermosensation is evolutionarily conserved for survival, yet the roles of glia in temperature coding and circuit dynamics remain poorly understood. Here, we identify C. elegans AMsh glia as dual-mode thermosensory hubs that autonomously detect temperature fluctuations by co-expressing the heat receptor GCY-28 (guanylate cyclase) and cold receptor GLR-3 (ionotropic glutamate receptor). Thermal changes induce spatiotemporal calcium dynamics in glia, driving GABA release to bidirectionally modulate neural circuits: enhancing AFD-mediated warmth detection through the excitatory receptor EXP-1 and suppressing ASH-dependent cold avoidance via the inhibitory receptor LGC-38. This GABAergic hub-and-spoke architecture regulates a broad range of thermal behaviors, including thermal avoidance, thermal resistance, and thermal preference. These findings establish glia as critical interpreters of environmental cues, highlighting their dual roles as sensors and modulators in sensory processing and providing a paradigm for understanding conserved glial mechanisms in neural circuit dynamics and behavioral plasticity.
Similar content being viewed by others
Introduction
The ability to interpret environmental thermal gradients, termed thermosensation, represents an evolutionarily conserved survival compass, enabling organisms to navigate thermal fluctuations through coordinated metabolic and behavioral recalibration1,2,3. Although molecular entities such as transient receptor potential (TRP) channels, guanylyl cyclases (GCs), and ionotropic glutamate receptors (e.g., GLR-3/GLUK2) have been identified as conserved thermosensory transducers1,2,3,4,5, the circuit-level mechanisms by which thermal signals are transduced into adaptive behavioral outputs remain incompletely characterized.
As a main component of the nervous system and brain, glial cells are increasingly recognized for their active involvement in neural computation, as well as in the modulation of synaptic plasticity, network dynamics, and sensory information processing6,7,8,9,10,11,12,13,14,15. However, the extent to which glia directly mediate sensory perception and how they orchestrate behavioral responses remain largely unknown.
The nematode Caenorhabditis elegans, adapted to habitats characterized by steep thermal gradients and variable temperatures, has evolved a highly sensitive thermosensory system capable of detecting minute temperature fluctuations ( < 0.01°C)16, thereby driving preferential orientation toward thermally optimal microenvironments while evading noxious extremes1,17,18. Central to this capability is a diverse set of thermosensory neurons that respond to distinct temperature ranges, spanning noxious cold, ambient living temperatures, and noxious heat. Activated by either warming or cooling stimuli, these neurons regulate temperature-dependent behaviors and physiological responses, such as cold/hot avoidance, temperature preference, and tolerance to extreme temperatures1,18,19.
The first identified thermosensory neuron in C. elegans is the AFD neuron, located in the head amphid. It perceives heat via guanylyl cyclases and stands as the most critical heat-sensing neuron, participating in behaviors including temperature memory, temperature preference, and heat tolerance20,21,22,23,24. The polymodal amphid sensory neuron ASH was shown to play a central role in cold avoidance responses25. Among other head amphid sensory neurons, AWC, ASI, and ASJ were identified relatively early as temperature-sensitive neurons. ADL and ASG respond to warming and contribute to cold tolerance, while ASER detects cold through the glutamate receptor GLR-31,19. In addition, other neurons such as PHC, PVD, FLP, and OLL are also involved in temperature sensation19,26.
Central to the amphid sensory architecture are AMsh glial cells, which form ensheathing processes around the dendrites of all amphid sensory neurons11,15,27. Interestingly, the expression of ver-1 in AMsh glia has been previously reported to be modulated by temperature, suggesting these glial cells are sensitive to temperature28,29. Despite their strategic localization at the interface of sensory transduction and their established association with thermal modulation30,31,32, how AMsh glia contribute to thermotransduction and the downstream circuit coding mechanism remains largely unexplored.
Here, we demonstrate that AMsh glia autonomously detect bidirectional thermal cues through co-expression of complementary thermoreceptors: GCY-28, a heat-activated guanylyl cyclase mediating warm stimulus detection, and GLR-3, a cold-sensitive ionotropic glutamate receptor transducing noxious cold signals. This cellular ensemble constitutes a GABAergic “hub-and-spoke” microcircuit, enabling differential modulation of downstream neural pathways: excitatory signaling via the EXP-1 GABA receptor potentiates AFD-mediated warm detection, whereas inhibitory signaling through the LGC-38 receptor attenuates ASH-dependent cold aversion. These findings establish a parsimonious mechanistic paradigm, wherein glial cells, in collaboration with neurons, jointly detect environmental thermal cues and serve as integrative hubs to synthesize sensory information, orchestrating adaptive behavioral outputs that optimize organismal fitness.
Results
AMsh glia are bona fide thermal receptor cells that respond to both warm and cold stimuli
Consistent with previous studies, we observed calcium responses in AFD neurons induced by warmth and in ASH neurons triggered by cold, using transgenically expressed GCaMP5 as the calcium indicator25,33(Fig. 1a,b). Interestingly, calcium increases were also detected in AMsh glia, the sheath glial cells surrounding the receptor endings of 12 pairs of amphid sensory neurons, including AFD and ASH27, in response to both warm and cold stimuli (Fig. 1a-d). This suggests that these glial cells may actively participate in thermosensation.
a, Time-lapse heatmap images show GCaMP5.0-based calcium dynamics in ASH neurons and AMsh glia upon cooling. The bath solution was perfused towards the worm, maintaining a temperature of 20 °C, and then cooled from 20 °C to 10 °C within 20 seconds. b, Time-lapse heatmap images show GCaMP5.0-based calcium dynamics in AFD neurons and AMsh glia upon heating. The bath solution was perfused towards the worm, maintaining a temperature of 20 °C, and then heated from 20 °C to 28 °C within 30 seconds. c, Cold-induced calcium responses in AMsh glia in mutants defective in neurotransmitter and neuropeptide release. Left, ΔF/F0 of GCaMP5.0, with solid lines representing the average fluorescence changes and shaded areas indicating standard error of the mean (SEM). Right, Maximum ΔF/F0 of GCaMP5.0, indicating peak calcium responses. Numbers on the columns indicate number of worms tested. d, Heat-induced calcium responses in AMsh glia in mutants defective in neurotransmitter and neuropeptide release. Left, ΔF/F0 of GCaMP5.0. Right, Maximum ΔF/F0 of GCaMP5.0. Numbers on the columns indicate number of worms tested. e-g, Cold and heat-induced calcium responses in isolated cultured AMsh glial cells. e, Time-lapse heatmap images show GCaMP5.0-based calcium dynamics. f, ΔF/F0 and Maximum ΔF/F0 of GCaMP5.0 upon cooling. g, ΔF/F0 and Maximum ΔF/F0 of GCaMP5.0 upon heating in wild type and pde-1,pde-2,pde-3,pde-5 quadruple mutants. Numbers on the columns indicate number of cells tested. Unpaired Mann-Whitney U test, two-tailed. All error bars represent SEM. Unpaired two-tailed t test except Fig.1g. **P < 0.05, and **P < 0.01. Source data are provided as a Source Data file.
We then asked whether temperature-induced calcium responses in AMsh glia are triggered by the activation of neighboring sensory neurons. AMsh glia retained both cold- and warm-induced calcium responses in unc-13 and unc-31 mutant animals, in which neurotransmitter and neuropeptide release are deficient (Fig. 1c,d)34,35,36. This finding suggests that the observed responses in AMsh glia are likely cell-autonomous. The thermosensory responses of AMsh glia were even more robust in unc-13 mutants than in wild type, accompanied by altered response kinetics. These observations indicate that AMsh glial activity is mildly suppressed by circuit inputs, which are likely mediated by surrounding neurons (Fig. 1c, d).
It has been reported that AMsh glia actively engulf fragments of AFD neurons and extracellular vesicles (EVs) from ciliated amphid neurons15,37,38. Thus, it is plausible that thermosensory neurons modulate the temperature response of AMsh glia via an UNC-13/UNC-31-independent pathway. To test this possibility, we transgenically expressed TRPV1 in ASH and AFD neurons, respectively. While capsaicin reliably activated ASH and AFD in these transgenic lines9,39, no corresponding calcium responses were detected in AMsh glia (Supplementary Fig. 1). These results reinforce the notion that AMsh glia may detect temperature in a cell-autonomous manner. In our previously reported work, we demonstrated that isolated single AMsh glial cells respond to odorant stimulation9. Building on this, we recorded calcium responses elicited by temperature stimuli in individual isolated AMsh glia cells. To rule out intercellular crosstalk, dissociated cells were seeded at a low plating density. Resemblance to those recorded in the intact organism, we observed in vitro cultured AMsh glial cells exhibited pronounced calcium elevations when subjected to either cold or heat stimuli (Fig. 1e–g).
Collectively, these results support that AMsh glia detect temperature in a cell-autonomous manner.
GCY-28 acts as a heat sensor in AMsh glia
We next investigated how temperature fluctuations activate AMsh glia. AFD neurons detect heat through membrane-bound guanylyl cyclases GCY-8, GCY-18, and GCY-23, which synthesize cGMP, activating cyclic nucleotide-gated (CNG) channels to mediate temperature-induced depolarization. Phosphodiesterases (PDEs) subsequently hydrolyze cGMP, lowering its concentration and thereby closing the CNG channels22,33. To investigate the role of cGMP in AMsh glia, we utilized two distinct AMsh glia-targeting promoters (vap-1 and R11D1.3)40,41 to transgenically express a blue light-inducible guanylyl cyclase (BlgC) alongside GCaMP5.042,43. Blue light induction of BlgC resulted in robust calcium increases in AMsh glia, suggesting that the increase of cGMP level can activate AMsh glia (Fig. 2a, b). Several amphid neurons respond to blue light through the light receptors LITE-1 and GUR-344,45. We found that blue light-induced calcium elevations in BlgC-expressing AMsh glia remained intact in lite-1;gur-3 double mutants (Fig. 2a, b). This indicates that blue light stimulation activates AMsh glia specifically via BlgC.
a, b Blue light induced calcium responses in AMsh glia expressing BLGC. Blue light was delivered in a series of 100 ms pulses with 100 ms intervals, while GCaMP5.0 was sampled at a frequency of 5 Hz. a, Time-lapse heatmap images. b Maximum ΔF/F0 of GCaMP5.0. c–e Heat-induced calcium responses in AMsh glia require CNG channel subunits. Time-lapse heatmap images c, ΔF/F0 of GCaMP5.0 d, and Maximum ΔF/F0 of GCaMP5.0 e. f–h, Heat-induced calcium responses in AMsh glia in wild type, gcy-8, gcy-18, gcy-23 triple mutants, gcy-28 mutants and gcy-28 mutants with AMsh glial restoration of GCY-28. Time-lapse heatmap images g, ΔF/F0 of GCaMP5.0 h, and Maximum ΔF/F0 of GCaMP5.0 i. i, j Ectopic expression of gcy-28 confers heat sensing to AWB neurons. ΔF/F0 of GCaMP5.0 (j), Maximum ΔF/F0 of GCaMP5.0 k. k GCY-28-transfected HEK293T cells exhibited markedly elevated intracellular cGMP levels under two distinct thermal challenges: a direct heat-up from 37 °C (the culture temperature of HEK293T cells) to 45 °C for 30 min, or a precooling step (from 37 °C to 20 °C for 2 h) followed by heating to 28 °C for 30 min. Numbers on the columns indicate number of trials with three wells of cells for each trial. Unpaired Mann-Whitney U test, two-tailed. l, m Expression of cng-1, cng-3, tax-4 and gcy-28 in AMsh glia revealed by 7×GFP knock-in and transcriptional reporter lines. GFP fluorescence were observed in AMsh glia in the [Pvap-1::spGFP1-10::sl2::CeBFP+cng-1::GFP11*7], [Pvap-1::spGFP1-10::CeBFP+cng-3::GFP11*7], and [Pvap-1::spGFP1-10::CeBFP +tax-4::GFP11*7] transgenic lines. GFP driven by the gcy-28 promoter colocalized with Pvap-1::mCherry. Large arrows indicate the somas, and small arrows denote the anterior processes of AMsh glia. Scale bars represent 20 µm.Numbers on the columns indicate number of animals tested. All error bars represent SEM. Unpaired two-tailed t test. ns, not significant. **P < 0.01, and ***P < 0.001. Source data are provided as a Source Data file.
In AFD neurons, CNG channel subunits TAX-2 and TAX-4 act downstream of cGMP22. Six CNG channel subunits are present in C. elegans: TAX-2, TAX-4, and CNG-1 through CNG-446,47. BlgC induced calcium increases in AMsh glia were slightly reduced in either tax-4 mutants or cng-1, cng-3 double mutants, and were completely abolished in cng-1, cng-3, tax-4 triple mutant background, but were rescued by restoring tax-4 expression in AMsh glia (Fig. 2a, b). Consistently, heat responses in AMsh glia were abolished in cng-1, cng-3, tax-4 triple mutant worms and rescued by AMsh glial expression of tax-4 (Fig. 2c-e). These results suggest that the CNG channels CNG-1, CNG-3, and TAX-4 act downstream of cGMP in AMsh glia. C. elegans genome encodes six PDE genes. Of these PDEs, PDE-4 and PDE-6 are cAMP-specific, while the other four PDEs can hydrolyze both cAMP and cGMP46. We found warm-induced calcium responses in AMsh glia remained intact in pde-4, pde-6 double mutants but were enhanced in pde-1, pde-2, pde-3, pde-5 quadruple mutants (Fig. 2c–e). Moreover, heat-induced calcium responses in isolated single AMsh glial cells were augmented in the pde quadruple mutants (Fig. 1e,g). These results are consistent with a role for cGMP in regulating AMsh glial thermosensory responses.
We then sought to identify the membrane GC proteins responsible for temperature sensing in AMsh glia. In gcy-8, gcy-18, gcy-23 triple mutants, the warm-induced calcium responses in AMsh glia were not reduced compared to wild-type worms (Fig. 2e–g). These findings imply that other guanylyl cyclases might be responsible for sensing heat in AMsh glia. Through single-cell RNA sequencing, the expression of several membrane GCs such as GCY-28, GCY-12, and GCY-18 has been implicated in AMsh glia48. We revealed that the absence of GCY-28 abolished warm responses in AMsh glia. Restoration of GCY-28 expression in AMsh glia fully rescued these responses, indicating that GCY-28 may function as a heat sensor in these glial cells (Fig. 2f–h).
To gain more evidence that GCY-28 is a heat sensor, we ectopically expressed GCY-28 in the chemosensory AWB neurons as a previous study described33. Intriguingly, warm induced a calcium decline in AWB neurons. Nevertheless, AWB neurons with ectopic GCY-28 expression exhibited warm-induced calcium increases (Fig. 2i).
We next performed heterologous expression of GCY-28 in HEK293T cells and quantified intracellular cGMP levels using a commercial ELISA kit. Compared to empty vector-transfected controls, GCY-28-expressing cells showed markedly elevated intracellular cGMP levels under two distinct thermal stimuli: either a direct heat challenge from 37 °C (the standard culture temperature for HEK293T cells) to 45 °C for 30 min, or a two-step protocol involving precooling from 37 °C to 20 °C for 2 h (mimicking the nematode’s culture temperature) followed by heating to 28 °C for 30 min (Fig. 2k).
Using 7×GFP knock-in and transcriptional reporter lines, we confirmed that cng-1, cng-3, tax-4, and gcy-28 are all endogenously expressed in AMsh glia (Fig. 2l, m). Taken together, these findings suggest that GCY-28 acts as a heat sensor in AMsh glia, mediating warm responses by increasing intracellular cGMP levels. The elevated cGMP then activates the CNG channels CNG-1, CNG-3, and TAX-4, leading to calcium influx and triggering downstream responses in AMsh glia.
GLR-3 functions as a cold sensor in AMsh glia
It has been implicated that GCY-20 and the TRPA-1 channel transduce cold signals in C. elegans42,49,50, but we observed no defects in the cold responses of AMsh glia in either gcy-20 or trpa-1 mutants. A previous study identified GLR-3 as a cold receptor within the ASER sensory neuron, playing a crucial role in normal cold avoidance behaviors51. Similarly, the mammalian homolog of GLR-3, GluK2, is essential for detecting noxious cold temperatures and facilitating cold nociception in mice4. Remarkably, the loss of GLR-3 significantly reduced cold-induced calcium responses in AMsh glia and this deficit was rescued by restoring glr-3 expression in AMsh glia (Fig. 3a, b, e). Supporting this, AMsh glia-specific knockdown of glr-3 also resulted in reduced cold-induced calcium responses in these glia (Fig. 3a, b, e).
a–e Cold-induced calcium responses in AMsh glia in various genetic background. Time-lapse heatmap images a, ΔF/F0 of GCaMP5.0 b–d, Maximum ΔF/F0 of GCaMP5.0 e. f, g Expression of glr-3 in AMsh glia revealed by 7×GFP knock-in f and transcriptional reporter g lines. Arrows indicate the somas. Scale bars represent 20 µm. Numbers on the columns indicate number of animals tested. All error bars represent SEM. Unpaired two-tailed t test. ns, not significant. *P < 0.05, **P < 0.01, and ***P < 0.001. Source data are provided as a Source Data file.
GLR-3 and GluK2 act as cold receptors through a noncanonical metabotropic mechanism, rather than through their ion channel function4,51. Our previous study demonstrated that odorant-induced calcium responses in AMsh glia involve inositol 1,4,5-trisphosphate receptor (IP3R)-mediated calcium release from the endoplasmic reticulum (ER)9. We observed that cold-induced responses in AMsh glia were diminished in both itr-1 mutants and worms with AMsh glial itr-1 RNAi, indicating that IP3R-mediated ER calcium release is involved in cold sensing by AMsh glia (Fig. 3c, e). Notably, glr-3;itr-1 double mutants exhibited defects comparable to those of the respective single mutants (Fig. 3c, e). Additionally, cold-induced calcium responses in AMsh glia were unaltered both in worms expressing the tetanus toxin light chain (TeTx) in ASER neurons (to block their synaptic transmission)25,42 and in che-2 mutants, in which animals have extremely short cilia and abnormal posterior projections of amphid sensory neurons and exhibit defects in behaviors mediated by these neurons52(Fig. 3d, e). Collectively, these results indicate that cold stimulation activates GLR-3 in AMsh glia, a process that trigger the release of calcium ions from the ER or the influx of extracellular calcium ions.
To confirm the expression of GLR-3 in AMsh glia, we generated a 7×GFP knock-in line, which demonstrated GLR-3 expression in these cells (Fig. 3f). A transcriptional reporter line containing a 2.9 kb glr-3 promoter driving fluorescent protein expression further confirmed the presence of GLR-3 in AMsh glia (Fig. 3g).
AMsh glia modulate thermal avoidance and tolerance
The observation that AMsh glia are activated by temperature fluctuations prompted us to investigate whether these glial cells contribute to thermal behaviors. To avoid the developmental deficits of neurites may be caused by ablation of glial cells, we transgenically expressed the histamine-gated chloride channel HisCl1 from Drosophila, which has been shown to be an effective chemogenetic tool for the inducible silencing of C. elegans neurons and other cells in intact animals42,53. By applying histamine, we were able to temporally inhibit the activity of AMsh glia within minutes (Supplementary Fig. 2).
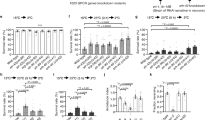
A well-known behavior of animals in response to temperature changes is the avoidance of cold and hot stimuli. To assess temperature avoidance, we performed cold- and hot-plate assays. In a 6 cm diameter NGM plate, the central 1.5 cm circular region was temperature-controlled to either 10 °C or 30 °C using a plate temperature regulator (Fig. 4a). We placed the worms at the center of this area and, after 10 min, counted the number of worms remaining within the central region of this assay plate. Next, we transgenically expressed HisCl1 with mCherry in AMsh glia and used histamine to inhibit the activity of these cells. While the application of histamine (10 mM) to the assay plates did not affect control worms expressing only mCherry in AMsh glia, inhibition of AMsh glia via histamine significantly reduced the number of worms remaining in the cold circle (10 °C) after 10 min (Fig. 4b,c). Conversely, histamine-mediated inhibition of AMsh glia increased the number of worms staying in the hot (30 °C) circle (Fig. 4d). We observed some degree of difference in thermal behaviors between control worms and HisCl1-expressing worms. This is not unexpected, as ectopic expression of an ion channel may induce basal activity changes. In particular, ectopic expression of HisCl1 has been implicated in a weak chloride ion leak effect42. Nevertheless, HisCl1 has been established as a relatively stable method for the real-time manipulation of neural activity25,53. Since histamine application had no effect on the locomotion of control worms (Fig. 4b–d), the altered thermal avoidance observed in histamine-treated AMsh glial HisCl1 worms indicate that AMsh glia function to suppress cold avoidance while enhancing hot avoidance.
a–d AMsh glia regulate thermal avoidance behavioral. Schematic of cold and hot plate assays a, Number of worms remaining within the central room temperature region b, the central cold region c, or the central hot region d. Numbers on the columns indicate the number of trials. Unpaired two-tailed t test. Ten well-fed day 1 adult hermaphrodite worms were used in each trial. e AMsh glia regulate cold tolerance. Numbers on the columns indicate the number of trials. Unpaired two-tailed t test. Thirty well-fed day 1 adult hermaphrodite worms were used in each trial. f AMsh glia regulate heat tolerance. Left, Inhibition of HisCl1-expressing AMsh glia via histamine. Right, gcy-28 mutants are defective in heat tolerance. Thirty well-fed day 1 adult hermaphrodite worms were used in each trial, with six trials performed for each condition (n = 6). Data were analyzed statistically using Two-way ANOVA. g Schematic of the linear thermotaxis (TTX) assay. h, i The distribution of 23 °C- cultured worms without (Left) and with (Right) HisCl1 expression h and TTX index i. Gradient: 0.5 °C/cm. Numbers on the columns indicate the number of trials (n = 18, 19, 20, 19). 250–500 well-fed day 1 adult hermaphrodite worms were used in each trial. Unpaired two-tailed t test. j, k The distribution of 23 °C- precultured worms without (Left) and with (Right) HisCl1 expression j and TTX index k. Gradient: 1.0 °C/cm. Numbers on the columns indicate the number of trials (n = 34, 30, 36, 43). 250–500 well-fed day 1 adult hermaphrodite worms were used in each trial. Unpaired two-tailed t test. l, m The distribution of 17 °C- precultured worms without (Left) and with (Right) HisCl1 expression l and TTX index m. Gradient: 1.0 °C/cm. Numbers on the columns indicate the number of trials (n = 19, 18, 29, 29). 250–500 well-fed day 1 adult hermaphrodite worms were used in each trial. Unpaired two-tailed t test. All error bars represent SEM. ns, not significant. *P < 0.05, **P < 0.01 and ***P < 0.001. Source data are provided as a Source Data file.
Tolerance to extreme cold and hot temperatures is essential for an animal’s survival. To investigate this, we placed the NGM plates in a 2 °C incubator and counted the number of worms alive after 72 h. We observed that while histamine (2 mM) application did not affect the mortality rate of wild-type worms at 2 °C, inhibition of HisCl1-expressing AMsh glia via histamine significantly reduced the mortality rate of the worms (Fig. 4e). We next placed NGM plates in a 37 °C incubator and monitored the survival ratio of the worms every two hours. Histamine treatment slightly increased the mortality rate in wild-type worms without HisCl1 expression under these conditions, but significantly increased the mortality rate in worms with HisCl1 expression (Fig. 4f). We then examined whether the absence of GCY-28, which we identified as a heat receptor in AMsh glia, affected hot tolerance. Remarkably, loss of gcy-28 significantly reduced the worms’ tolerance to heat, but this defect could be significantly rescued by restoring gcy-28 expression specifically in AMsh glia (Fig. 4f). Consistent with the role of GCY-28 in warm sensing of AMsh glia, no difference in cold tolerance was observed between WT and gcy-28 mutants (Supplementary Fig. 3a). It has been reported that sensory-deficient mutants often exhibit extended lifespan54. However, as demonstrated in both the present study and previous work, GCY-28 displays a broad expression pattern beyond AMsh glia (Fig. 2m)55. Given this wide expression profile, it is not surprising that gcy-28 mutants, essential for Amsh glial thermosensation, show reduced survival in extreme heat conditions.
Taken together, these results suggest that AMsh glia play a role in inhibiting cold tolerance while enhancing hot tolerance.
AMsh glia modulate temperature preference
C. elegans exhibit thermotactic navigation (thermotaxis, TTX) by moving toward and maintaining their position near their cultivation temperature within thermal gradients, helping them stay within their adaptive thermal range while avoiding potentially harmful conditions. This behavior serves as a model for neural computation, integrating temperature sensing, adaptive memory, and locomotor control to reveal fundamental principles of how organisms respond to environmental temperature cues17,56. We then established a linear temperature gradient of 0.5 °C/cm and monitored the migration of worms. The worms were cultured at 23 °C and initially placed in the zero subzone (20 °C) of the gradient plate. After one hour, we quantified the distribution of worms across the gradient subzones (Fig. 4g). Consistent with previous studies, worms preferentially migrated toward higher temperatures than that in the zero subzone. When AMsh glial HisCl1-expressing worms were transferred to a histamine (2 mM)-containing temperature gradient plate (TTX plate), they migrated toward their cultivation temperature, indicating that inhibition of AMsh glia did not block temperature memory retrieval. However, AMsh glial HisCl1-expressing worms in the histamine-containing plate exhibited a higher TTX index compared to those in a histamine-free plate, indicating that thermal preference was modulated by AMsh glia (Fig. 4h, i).
The steepness of the temperature gradient plays a crucial role in worm thermotaxis. When the gradient reaches approximately 1.0 °C/cm or surpasses this threshold, worms cultured at low temperatures (15–17 °C) tend to accumulate near their cultivation temperature. In contrast, worms cultured at high temperatures (23–25 °C) exhibit an almost athermotactic phenotype, showing minimal or no preference for the thermal gradient. Some worms even become trapped in the cold region, displaying cryophilic behavior17,56,57. Consistently, we observed that worms cultured at 23 °C displayed cryophilic behavior in a 1.0 °C/cm gradient. However, fewer AMsh glial HisCl1-expressing worms were trapped in the cold region (12–14 °C) on histamine-containing TTX plates compared to those on histamine-free plates (Fig. 4j, k). Additionally, AMsh glial HisCl1-expressing worms cultured at 17 °C migrated toward their cultivation temperature in the histamine-containing TTX plates in a 1.0 °C/cm gradient, and again showed a higher TTX index compared to those in the histamine-free plates (Fig. 4l,m). As a control, we cultured gcy-8,gcy-18,gcy-23 triple mutants in either 17 °C or 23 °C, and examined their distribution in TTX plates in a 1.0 °C/cm or 0.5 °C/cm gradient. These triple mutants failed to migrate to their cultured temperature as previously reported (Supplementary Fig. 3b)58. These findings suggest that AMsh glia are involved in modulating thermotactic behavior, influencing the worm’s ability to track and respond to thermal gradients.
AMsh glia differentially regulate thermosensory neurons to modulate thermal responses
To investigate whether AMsh glia modulate thermal behaviors through interactions with thermosensory neurons, we focused on AFD and ASH neurons, which play distinct roles in temperature sensing. AFD neurons detect temperature changes within the nematode’s optimal living range, mediating thermotactic behavior and contributing to heat avoidance and thermal tolerance21,22, whereas ASH neurons primarily respond to cold stimuli and drive cold avoidance25. Using worms expressing HisCl1 specifically in AMsh glia, we found that histamine treatment significantly attenuated warm-induced calcium responses in AFD neurons (Fig. 5a–d). To further validate AMsh glia’s role in AFD activation, we subjected these worms to a cold stimulus (10 °C, 4 min), which activates AMsh glia in histamine-free but not histamine-containing solutions, followed by a heat stimulus. Histamine treatment strongly suppressed subsequent heat-induced AFD responses (Fig. 5e–g), confirming that AMsh glia enhance warm sensitivity in AFD neurons. Consistent with this, warm-induced AFD responses were significantly reduced in gcy-28 mutants, which can be rescued by restoring GCY-28 expression specifically in AMsh glia (Fig. 5h, i). Conversely, in worms expressing HisCl1 in AMsh glia, histamine treatment prolonged cold-induced calcium responses in ASH neurons compared to untreated controls (Fig. 5j–m). These findings demonstrate that AMsh glia bidirectionally regulate thermosensory neurons—potentiating warm responses in AFD while suppressing cold responses in ASH.
a-d Inhibition of HisCl1-expressing AMsh glia via histamine modulated warm-induced calcium responses in AFD neurons. ΔF/F0 of GCaMP5.0 in worms without a or with b, c HisCl1 expression. Maximum ΔF/F0 of GCaMP5.0 in AFD d. e–g Inhibition of HisCl1-expressing AMsh glia via histamine modulated warm-induced calcium responses in AFD neurons in cold- pretreated worms. The bath solution was perfused towards the worm, maintaining a temperature of 10 °C for four minutes, and then warmed from 10 °C to 28 °C within 30 s. d, e Worms without e or with f HisCl1 expression. Maximum ΔF/F0 of GCaMP5.0 in AFD g. h, i Heat-induced calcium responses in AFD neurons in gcy-28 mutants, and gcy-28 mutants with AMsh glial restoration of GCY-28. ΔF/F0 of GCaMP6s h. Maximum ΔF/F0 of GCaMP6s in AFD i. j–m Inhibition of HisCl1-expressing AMsh glia via histamine modulated cold-induced calcium responses in ASH neurons. ΔF/F0 of GCaMP5.0 in worms without j or with k, l HisCl1 expression. Maximum ΔF/F0 of GCaMP5.0 in ASH neurons m. Numbers on the columns indicate number of animals tested. All error bars represent SEM. Unpaired two-tailed t test. ns, not significant. *P < 0.05, **P < 0.01, and ***P < 0.001. Source data are provided as a Source Data file.
AMsh glia differentially regulate temperature-sensitive neurons through distinct GABAA receptor pathways
How does histamine-mediated inhibition of HisCl1-expressing AMsh glia differentially regulate the activity of AFD and ASH neurons? One possibility is that the opening of HisCl1 channels disrupts chloride homeostasis in the glial-neuronal extracellular space, a perturbation that has been shown to impact AFD neuron activity8,32,33. Another potential mechanism is that AMsh glia release transmitters to modulate the activity of these neurons. Previous studies have demonstrated that AMsh glia release GABA in response to nose touch or aversive odorant exposure, activating the inhibitory GABAA receptor LGC-38 in ASH neurons to promote olfactory and mechanical adaptation8,9,59. Extending these findings, we observed significantly impaired adaptation of cold-induced calcium responses in ASH neurons of both unc-25 mutants (GABA biosynthesis-deficient46 and lgc-38 mutants compared to wild-type animals (Fig. 6a, b). Importantly, these adaptation deficits were rescued by restoring unc-25 expression specifically in AMsh glia or by expressing lgc-38 in ASH neurons of the respective mutants. These results demonstrate that GABAergic signaling from AMsh glia is critical for proper cold adaptation in ASH neurons.
a, b Cold- induced calcium responses in ASH neurons in wild type, unc-25 mutants, lgc-38 mutants and rescue lines. ΔF/F0 changes a, Average ΔF/F0 between 120 s to 180 s after cooling b. c, d Heat-induced calcium responses in AFD neurons in wild type, wild type with GABA treatment, unc-13 mutants, unc-25 mutants, and rescue lines. ΔF/F0 of GCaMP5.0 c, Maximum ΔF/F0 of GCaMP5.0 in AFD d. e, f Heat-induced calcium responses in AFD neurons in wild type, exp-1 mutants and exp-1 mutants with AFD-specific rescue. ΔF/F0 of GCaMP5.0 e, Maximum ΔF/F0 of GCaMP5.0 in AFD f. g Expression of exp-1 in AFD neurons. Scale bars represent 20 µm. Numbers on the columns indicate number of animals tested. All error bars represent SEM. Unpaired two-tailed t test. ns, not significant. *P < 0.05, **P < 0.01, and ***P < 0.001. Source data are provided as a Source Data file.
We next investigated how AMsh glia enhance the activity of AFD neurons. Warm responses in AFD neurons were reduced in unc-13 mutants and rescued by restoration of unc-13 in AMsh glia, suggesting a role for neurotransmitter(s) (Fig. 6c, d). Further screening of neurotransmitter pathway mutants revealed that unc-25 mutants also exhibited significantly weaker warm responses in AFD neurons compared to wild-type animals. Importantly, rescuing unc-25 expression specifically in AMsh glia restored these responses (Fig. 6c, d), indicating that GABA release from AMsh glia is required for AFD neuron activation. Consistent with this, exogenous GABA application enhanced warm responses in AFD neurons (Fig. 6c, d). These findings suggest that heat stimulation triggers GABA release from AMsh glia, which in turn potentiates AFD neuron activity.
To identify the GABA receptor mediating this effect, we screened GABA receptor mutants and found that exp-1 mutants exhibited reduced warm-induced responses in AFD neurons, and this defect was rescued by expressing exp-1 specifically in AFD neurons (Fig. 6e, f). EXP-1 is an excitatory cation channel previously implicated in enteric muscle contraction60,61. While EXP-1 is known to be expressed in intestinal muscles and some head neurons, our transcriptional reporter analysis confirmed its presence in AFD neurons (Fig. 6g). These findings demonstrate that AMsh glia differentially regulate temperature-sensitive neurons through distinct GABAA receptor pathways: EXP-1 mediates excitatory modulation of AFD neurons, while LGC-38 mediates inhibitory regulation of ASH neurons.
Discussion
Our study identifies AMsh glia as autonomous thermoreceptor cells capable of local sensory modulation. By co-opting molecular sensors traditionally attributed to neurons (GCY-28 for heat and GLR-3 for cold), these glial cells challenge the conventional paradigm of modality-specific sensory specialization. Through the co-expression of these receptors within a single cell, AMsh glia achieve dual-modality temperature detection, positioning them as input-layer modulators that balance opposing thermal cues via a GABAergic hub-and-spoke microcircuit (Supplementary Fig. 4). This architecture enables simultaneous modulation of heat-sensitive AFD and cold-sensitive ASH neurons—both resident in the sensory input layer—thereby regulating organismal thermal behaviors.
Previously recognized thermosensory systems universally segregate cold and heat detection into specialized neuronal subtypes (e.g., TRPV1⁺/TRPM8⁺ neurons in mammals; AFD/ASH neurons in C. elegans)62. In contrast, AMsh glia circumvent this organizational constraint by co-expressing GCY-28 and GLR-3, enabling concurrent heat and cold sensing through compartmentalized signaling cascades. This dual-receptor architecture suggests an evolved molecular strategy for integrating competing thermal signals at the input layer via a parsimonious coding mechanism. Such input-restricted integration likely enhances adaptive behavioral precision by facilitating real-time weighting of sensory inputs before signals reach higher-order processing centers. The discovery of glial-autonomous thermosensation raises intriguing questions about evolutionary conservation. Emerging evidence shows that mammalian glia and non- neuronal cells, like skin keratinocytes, express conserved thermoreceptors such as TRPV1, TRPV3, and TRPV4, suggesting their involvement in thermosensation63,64,65,66. However, currently, our understanding of the molecular mechanisms of thermal transduction within these cells, as well as their circuit- coding strategies and functional significance in thermal sensation, is still extremely limited. This implies that glia- like cells may have conserved roles in input- layer sensory detection and integration across different phyla.
Traditional hub-and-spoke models emphasize neuronal hubs integrating unimodal inputs67,68,69,70. Here, we describe a glia-centric input-layer circuit wherein AMsh glia act as local hubs, receiving raw thermal data and distributing processed signals via GABAergic transmission to adjacent sensory neurons (AFD and ASH). Among the twelve pairs of amphid sensory neurons ensheathed by AMsh glia, AWC, ASI, ASE, ADL, and ASG have also been reported to display thermosensitivity19. Our findings do not preclude the possibility that AMsh glia could modulate the activities of these neurons. Further investigations are essential to comprehensively map out how AMsh glia regulate thermosensory neural networks, which would offer profound insights into the underlying neural regulatory mechanisms.
The differential expression of GABA receptors—excitatory EXP-1 in AFD and inhibitory LGC-38 in ASH—establishes a push-pull regulatory mechanism, amplifying heat-seeking behaviors while dampening cold avoidance. This context-dependent GABAergic signaling exemplifies input-layer signal prioritization, whereby glia optimize sensory throughput according to environmental demands.
As the primary nociceptors in C. elegans, ASH neurons are tasked with detecting diverse noxious stimuli, including cold, harmful chemicals, and mechanical stress25,71,72. This study, in conjunction with prior work8,9,59, demonstrates that AMsh glia resolve sensory crosstalk via calcium-GABA gating, converting heterogeneous inputs into a unified GABAergic signal. By suppressing chronic nociceptive signaling, this mechanism may mitigate prolonged activation of energy-intensive interneuronal pathways, thereby promoting metabolic efficiency. Moreover, this unified GABAergic signal implements a sensory prioritization algorithm. For instance, under non-optimal temperatures, strong thermal cues dominate GABAergic output, attenuating ASH-mediated avoidance to reduce energy expenditure. Concurrently, AMsh glia sharpen AFD neurons—the principal thermosensory neurons that utilize GCY-family receptors (GCY-8/18/23) to detect thermal gradients and drive precise thermotaxis toward habituated temperatures—via excitatory EXP-1 receptors, enhancing their sensitivity to subthreshold thermal fluctuations. This glial facilitation could sharpen the signal-to-noise ratio at the input layer, enabling rapid reorientation toward preferred thermal microenvironments. Collectively, these findings illustrate how non-neuronal cells achieve polymodal sensory integration and input-layer homeostasis through spatially restricted, receptor-specific modulation.
Taken together, this work establishes AMsh glia as dual thermosensors and integrators, showcasing a paradigm where glial molecular machinery directly modulates thermal sensation, underscoring the critical role of glia in shaping adaptive neural computation.
Methods
C. elegans strains and media
All nematode strains were raised on nematode growth medium (NGM) plates seeded with E. coli OP50 at 20˚C following standard methods73. Day 1 (D1) adult hermaphrodites under well-fed conditions were utilized in all experiments. The Bristol N2 strain, obtained from the Caenorhabditis Genetics Center, was used as the wild-type. C. elegans strains used in this study are listed in the supplementary file.
Calcium Imaging
Worms were immobilized in a bath solution containing 145 mM NaCl, 2.5 mM KCl, 1 mM MgCl2, 5 mM CaCl2, 10 mM HEPES, and 20 mM glucose (pH 7.3, adjusted with NaOH). They were glued to a glass coverslip using medical-grade cyanoacrylate adhesive (Gluture Topical Tissue Adhesive, Abbott Laboratories). Intracellular calcium signals were monitored using the green fluorescent proteins GCaMP5.0. Fluorescent imaging was performed using an Olympus IX71 microscope fitted with a 20x water immersion objective lens and a Teledyne Photometrics Retiga E7 camera. Data were collected via Micro-Manager software. Fluorescence from GCaMP was excited with a ThorLabs blue LED lamp (emission wavelength, 460–480 nm) and collected at a sampling rate of either 1 Hz or 0.33 Hz using Micro-Manager software. For BLGC, blue light was delivered in a series of 100 ms pulses with 100 ms intervals, while GCaMP5.0 was sampled at a frequency of 5 Hz. Temperature control was achieved using the CL-100 Temperature Controller and the LCS-1 Liquid Cooling System (Warner Instruments).
Molecular biology
Promoters were amplified from N2 genomic DNA using PCR and subsequently recombined with specific donor vector fragments utilizing the In-Fusion PCR Cloning Kit (TaKaRa Inc.). The following promoter regions were analyzed to investigate expression patterns: for gcy-28, a 3.4 kb fragment located upstream of the start codon; for glr-3, a 2.9 kb fragment upstream of the start codon; for exp-1, a 4 kb fragment upstream of the start codon.
To achieve AMsh glia-specific knockdown, sense or antisense sequences targeting an exon-rich genomic region were expressed under the control of a 1245 bp promoter from the R11D1.3 gene upstream. The RNAi construct for itr-1 was generated using a 1134 bp PCR fragment spanning exons 3 to 6 (primers F: CTTGAGCACACTTGGGTTGG and R: GACATGTCCGGCTTGAGTTGTGG), while the construct for glr-3 used a 787 bp fragment spanning exons 2 to 3 (primers F: CGATGAGGTGAATCCCGTCTTGG and R: TACAGTACCCCACACATTGAC).
Cell-type-specific rescue or expression was performed by expressing genes under cell-specific promoters: vap-1p (2 kb) in AMsh glia, R11D1.3p (1.245 kb) in AMsh glia, sra-6p (3.8 kb) in ASH neurons, gcy-8p (0.7 kb) in AFD neurons, str-1p (3.7 kb) in AWB and gcy-5p (3.1 kb) in ASER neurons. The following coding sequences were cloned: gcy-28 isoform b (1914 bp cDNA), glr-3 (2511 bp cDNA), exp-1(1620 bp cDNA), unc-13 isoform e (5451 bp cDNA), unc-25 isoform a (1527 bp cDNA), and tax-4 (4016 bp genomic DNA).
Confocal microscopy
A 20 μL aliquot of 2–3% agarose solution was applied to a glass slide, which was then covered with another slide. After a few minutes, once the agarose had solidified, the slides were carefully separated to create an agar-coated surface. Next, 10 μL of M9 solution containing 200 mM sodium azide (NaN3) was added to the agar. An adult D1 worm was placed onto the agar surface, and a coverslip was gently placed on top. If the worm is damaged during this step, it should be discarded. The prepared slide was then placed under an inverted confocal laser microscope, and images were captured using FV10-ASW Viewer software (Olympus). For 7*GFP knock-in lines, a Leica STELLARIS high-resolution confocal microscope was employed to acquire images with improved signal-to-noise ratio.
Linear thermotaxis (TTX) assay
A linear thermal gradient was established on a rectangular aluminum platform (60 cm length × 2 cm thickness) using two plate thermal controllers set to distinct temperatures at either end. Ambient temperature and humidity were maintained at 20 °C and 25–40%, respectively. Temperature gradients of 0.5 °C/cm or 1.0 °C/cm were generated by adjusting the thermoregulator settings. A rectangular TTX plate (14 × 6 × 2 cm) containing 11 mL of TTX medium (2% agar, 0.3% NaCl, 25 mM potassium phosphate buffer, pH 6.0) was placed on the aluminum platform, with gaps sealed using conductive adhesive to ensure thermal contact. The temperature of the subzone 0 of the TTX plate was set to 20 °C, and the system was allowed to stabilize for 15 min prior to experiments (Fig. 4). A plastic cover was placed over the platform to minimize airflow interference and TTX evaporation.
Hermaphroditic worms were synchronized and cultured on histamine-free NGM plates seeded with E. coli OP50 at 17 °C or 23 °C until reaching day 1 adulthood. For TTX assays, worms were collected using ~5 mL of NG buffer (0.3% NaCl, 1 mM CaCl₂, 1 mM MgSO₄, 25 mM phosphate buffer, pH 6.0), pelleted in a conical tube, and washed twice with ~2 mL of NG buffer. All steps were performed in a 20 °C water bath with pre-equilibrated NG buffer. Approximately 250–500 worms were transferred to subzone #0 (20 °C) of the TTX plate. Excess buffer was carefully removed using tissue paper. After 1 h of incubation, worm distribution was quantified via two approaches: (1) calculating the percentage of worms in each subzone; (2) determining the thermotaxis (TTX) index, which is defined by the following equation:
where Si denotes the score of individual i, and N represents the total number of animals on the test plate.
We measured the actual temperature of each zone on individual assay plates prior to worm counting. Due to slight environmental fluctuations and plate-specific variations, the real temperature range of each plate deviated slightly from the theoretical gradient. Additionally, to more clearly and intuitively visualize the distribution range of worms, the temperature ranges showed in Fig. 4 were based on the actual worm distribution.
In histamine experiments, freshly made histamine stock was added to both the NG buffer and TTX medium to a final concentration of 2 mM. Previous studies have showed that 10 mM histamine induces paralysis within two minutes in worms with pan-neuronal expression of HisCl19,53. To minimize potential adverse effects of long-term histamine exposure, we employed 2 mM histamine in these specific assays.
Thermal avoidance assay
Cold- and hot-plate assays were performed to assess thermal avoidance. A 6 cm diameter NGM assay plate (0.2 cm thickness) was prepared with a 1.5 cm diameter circular central region temperature-controlled to either 10 °C (cold plate assay) or 30 °C (hot plate assay) using a plate thermal controller. The gap between central region of the assay plate and the plate of the thermal controller was sealed using conductive adhesive to ensure thermal contact. Room temperature and humidity were maintained at 20 °C and 25–40%, respectively. We calibrated the set surface temperature of the temperature-controlled plate to 10 °C, 20 °C, or 28 °C. However, during the actual avoidance behavior assay, the worms crawl on the surface of the NGM layer (where the surrounding air temperature is room temperature, 20 °C). Additionally, due to systematic factors, including the thermal conductive adhesive, the bottom of the plastic petri dish, and the NGM layer, that separate the worms from the temperature-controlled plate, the actual temperature perceived by the worms may slightly differ from the set temperature. For each experiment, 10 well-fed day 1 adult hermaphroditic worms were placed at the center of the temperature-controlled region. After 10 minutes, the number of worms remaining within the central zone was quantified. Histamine-containing assay plates were prepared by adding freshly made histamine stock to the sterilized, molten NGM medium when it had cooled to approximately 55 °C, achieving a final concentration of 10 mM.
Cold and heat tolerance assays
To evaluate cold tolerance, 30 well-fed day 1 adult hermaphroditic worms were transferred to an NGM plate seeded with E. coli OP50. Plates were incubated at 2 °C for 72 hours in a temperature-regulated chamber. Following cold exposure, plates were transferred to a 20 °C environment for a 1-hour recovery period to restore motility. Survival rates were quantified by counting motile worms responsive to gentle tactile stimulation. For heat tolerance assessment, 30 well-fed day 1 adult hermaphroditic worms were placed on an E. coli OP50-seeded NGM plate and exposed to 37 °C in a calibrated incubator. Survival was monitored at 2-hour intervals for up to 12 h. At each time point, live worms were identified by spontaneous movement or response to mechanical prodding, and survival ratios were calculated as the percentage of live worms relative to the initial population. Histamine-containing NGM plate were made by addition of freshly made histamine stock to the NGM plate to a final concentration of 2 mM.
HEK293T cell culture and transfection
HEK293T cells (ATCC CRL-3216) were maintained in Dulbecco’s Modified Eagle Medium (DMEM) supplemented with 10% fetal bovine serum (FBS) and 1% penicillin-streptomycin at 37 °C in a humidified 5% CO₂ atmosphere. Upon reaching approximately 80% confluence in 6-well plates, cells were transfected with either an empty vector or a GCY-28 expression plasmid using Lipofectamine 2000, following the manufacturer’s protocol.
Thermal stimulation and sample preparation
At 40 h post-transfection, cells were subjected to cGMP induction using one of two protocols. Protocol 1: Cells were switched to DMEM supplemented with 1 mM IBMX (3-isobutyl-1-methylxanthine, a phosphodiesterase inhibitor) and immediately incubated at either 37 °C (the standard cell culture temperature) or 45 °C for 30 min. Protocol 2: Cells were first pre-incubated at 20 °C for 2 h (mimicking the nematode’s living temperature) prior to medium replacement with IBMX-containing DMEM. Stimulation was then conducted at 30 °C for 30 min. Following stimulation, cells were immediately washed with PBS, scraped, and pelleted. To generate sufficient material for each biological replicate, cell pellets from three wells per condition were pooled and lysed in 300 μL of ice-cold lysis buffer (supplemented with protease and phosphatase inhibitors). Lysates were centrifuged at 12,000 × g for 15 min, and the clarified supernatants were collected for subsequent cGMP analysis.
cGMP quantification by ELISA
The concentration of cGMP in the cell lysates was determined using a commercial human cGMP ELISA kit (mlbio). Briefly, samples and standards were acetylated to enhance assay sensitivity, then incubated in antibody-coated wells. Following a washing step, a horseradish peroxidase (HRP)-conjugated tracer was added. Color development was initiated with tetramethylbenzidine (TMB) substrate and stopped with acid. The absorbance was measured at 450 nm using a multi-mode microplate reader (MD). A four-parameter logistic (4-PL) regression model was used to generate the standard curve for each assay according to the manufacturer’s instructions. All standard curves exhibited a coefficient of determination (R² ≥ 0.99), ensuring reliable quantification. Data are derived from at least three independent biological replicates.
Isolated AMsh glial cell culture
Isolation and culture of AMsh glia were performed based on a previously established primary C. elegans cell culture protocol9,74. Gravid worms were maintained on 8X peptone plates with E. coli OP50, and enriched eggs were collected for cell dissociation. Dissociated embryonic cells were plated at low density on peanut lectin-coated cover glasses. L-15 medium was changed daily to remove secretions. After 3 days in culture at 20 °C, isolated mature AMsh glia were recognized by co-expression of Pvap-1::mCherry and Pvap-1::GCaMP5.0.
CRISPR/Cas9-engineered knock-in combined with issue-specific Split SfGFP
For tissue-specific tissue-specific Split SfGFP experiments, knock-in (KI) strains containing a tandem array of seven copies of the GFP11 β-strand (GFP11×7) inserted into the endogenous genomic loci of cng-1, cng-3, tax-4, or glr-3 were generated by the SunyBiotech company using CRISPR/Cas9 technology. We then independently microinjected these GFP11×7 knock-in strains with our constructed plasmid expressing the complementary split-GFP fragment (spGFP1-10) under the control of the vap-1 promoter. This approach enables specific labeling of the respective endogenous proteins in AMsh glia within their native cellular context9. Additionally, CeBFP driven by the vap-1 promoter was used as a control for labeling AMsh glia.
Statistical analysis
Calcium imaging data were acquired using Macro-Manager software and analyzed with ImageJ. Statistical analysis was performed in GraphPad Prism 8, and results are presented as mean ± standard error of the mean (S.E.M.). An unpaired two-tailed t-test was used to compare two groups of data. For non-normally distributed data within a group, the Wilcoxon test was applied. Two-way ANOVA was used for comparisons of heat resistance. A p-value of less than 0.05 was considered statistically significant, indicated by an asterisk (*), while p-values less than 0.01 and 0.001 were denoted by ** and ***, respectively. Sample sizes were chosen based on the reproducibility of the experiments and are consistent with standard practices in the field.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
Source data are provided with this paper. Any additional information required to reanalyse the data reported in this paper is available from the lead contact (kanglijun@zju.edu.cn) upon request. Source data are provided with this paper.
References
Xiao, R. & Xu, X. Z. S. Temperature sensation: from molecular thermosensors to neural circuits and coding principles. Annu Rev. Physiol. 83, 205–230 (2021).
Solinski, H. J. & Hoon, M. A. Cells and circuits for thermosensation in mammals. Neurosci. Lett. 690, 167–170 (2019).
Morrison, S. F. & Nakamura, K. Central mechanisms for thermoregulation. Annu Rev. Physiol. 81, 285–308 (2019).
Cai, W. et al. The kainate receptor GluK2 mediates cold sensing in mice. Nat Neurosci https://doi.org/10.1038/s41593-024-01585-8 (2024).
Chao, Y. C. et al. Receptor guanylyl cyclase-G is a novel thermosensory protein activated by cool temperatures. EMBO J. 34, 294–306 (2015).
Nagai, J. et al. Behaviorally consequential astrocytic regulation of neural circuits. Neuron 109, 576–596 (2021).
Kwak, H. et al. Astrocytes control sensory acuity via tonic inhibition in the thalamus. Neuron 108, 691–706 e610 (2020).
Fernandez-Abascal, J. et al. A glial ClC Cl(-) channel mediates nose touch responses in C. elegans. Neuron 110, 470–485 e477 (2022).
Duan, D. et al. Sensory glia detect repulsive odorants and drive olfactory adaptation. Neuron 108, 707–721 e708 (2020).
Abdo, H. et al. Specialized cutaneous Schwann cells initiate pain sensation. Sci. (NY) 365, 695–699 (2019).
Tang, Y., Illes, P. & Verkhratsky, A. Glial-neuronal sensory organs: evolutionary journey from Caenorhabditis elegans to mammals. Neurosci. Bull. 36, 561–564 (2020).
Won, W., Bhalla, M., Lee, J. H. & Lee, C. J. Astrocytes as key regulators of neural signaling in health and disease. Annu. Rev. Neurosci. https://doi.org/10.1146/annurev-neuro-112723-035356 (2025).
Cheng, H. et al. Phasic/tonic glial GABA differentially transduce for olfactory adaptation and neuronal aging. Neuron 112, 1473–1486 e1476 (2024).
Xin, Q. et al. Neuron-astrocyte coupling in lateral habenula mediates depressive-like behaviors. Cell 188, 3291–3309 e3224 (2025).
Singhvi, A., Shaham, S. & Rapti, G. Glia Development and Function in the Nematode Caenorhabditis elegans. Cold Spring Harbor perspectives in biology 16 https://doi.org/10.1101/cshperspect.a041346 (2024).
Ramot, D., MacInnis, B. L. & Goodman, M. B. Bidirectional temperature-sensing by a single thermosensory neuron in C. elegans. Nat. Neurosci. 11, 908–915 (2008).
Kimata, T., Sasakura, H., Ohnishi, N., Nishio, N. & Mori, I. Thermotaxis of C. elegans as a model for temperature perception, neural information processing and neural plasticity. Worm 1, 31–41 (2012).
Goodman, M. B. & Sengupta, P. How Caenorhabditis elegans senses mechanical stress, temperature, and other physical stimuli. Genetics 212, 25–51 (2019).
Glauser, D. A. Temperature sensing and context-dependent thermal behavior in nematodes. Curr. Opin. Neurobiol. 73, 102525 (2022).
Kobayashi, K. et al. Single-cell memory regulates a neural circuit for sensory behavior. Cell Rep. 14, 11–21 (2016).
Clark, D. A., Biron, D., Sengupta, P. & Samuel, A. D. The AFD sensory neurons encode multiple functions underlying thermotactic behavior in Caenorhabditis elegans. J. Neurosci. 26, 7444–7451 (2006).
Goodman, M. B. & Sengupta, P. The extraordinary AFD thermosensor of C. elegans. Pflug. Arch. 470, 839–849 (2018).
Mori, I. & Ohshima, Y. Neural regulation of thermotaxis in Caenorhabditis elegans. Nature 376, 344–348 (1995).
Takeishi, A. Environmental-temperature and internal-state dependent thermotaxis plasticity of nematodes. Curr. Opin. Neurobiol. 74, 102541 (2022).
Lin, C. et al. Molecular and circuit mechanisms underlying avoidance of rapid cooling stimuli in C. elegans. Nat. Commun. 15, 297 (2024).
Fan, Y. et al. Polymodal functionality of C. elegans OLL neurons in mechanosensation and thermosensation. Neurosci. Bull. 37, 611–622 (2021).
Singhvi, A. & Shaham, S. Glia-Neuron Interactions in Caenorhabditis elegans. Annu Rev. Neurosci. 42, 149–168 (2019).
Procko, C., Lu, Y. & Shaham, S. Sensory organ remodeling in Caenorhabditis elegans requires the zinc-finger protein ZTF-16. Genetics 190, 1405–1415 (2012).
Procko, C., Lu, Y. & Shaham, S. Glia delimit shape changes of sensory neuron receptive endings in C. elegans. Development 138, 1371–1381 (2011).
Ray, S., Gurung, P., Manning, R. S., Kravchuk, A. A. & Singhvi, A. Neuron cilia restrain glial KCC-3 to a microdomain to regulate multisensory processing. Cell Rep. 43, 113844 (2024).
Yoshida, A. et al. A glial K(+) /Cl(-) cotransporter modifies temperature-evoked dynamics in Caenorhabditis elegans sensory neurons. Genes Brain Behav. 15, 429–440 (2016).
Singhvi, A. et al. A Glial K/Cl Transporter Controls Neuronal Receptive Ending Shape by Chloride Inhibition of an rGC. Cell 165, 936–948 (2016).
Takeishi, A. et al. Receptor-type Guanylyl Cyclases Confer Thermosensory Responses in C. elegans. Neuron 90, 235–244 (2016).
Meunier, F. A. & Hu, Z. Functional Roles of UNC-13/Munc13 and UNC-18/Munc18 in Neurotransmission. Adv. Neurobiol. 33, 203–231 (2023).
Speese, S. et al. UNC-31 (CAPS) is required for dense-core vesicle but not synaptic vesicle exocytosis in Caenorhabditis elegans. J. Neurosci. 27, 6150–6162 (2007).
Richmond, J. E., Davis, W. S. & Jorgensen, E. M. UNC-13 is required for synaptic vesicle fusion in C. elegans. Nat. Neurosci. 2, 959–964 (1999).
Razzauti, A. & Laurent, P. Ectocytosis prevents accumulation of ciliary cargo in C. elegans sensory neurons. Elife 10 https://doi.org/10.7554/eLife.67670 (2021).
Raiders, S. et al. Glia actively sculpt sensory neurons by controlled phagocytosis to tune animal behavior. Elife 10 https://doi.org/10.7554/eLife.63532 (2021).
Tobin, D. M. et al. Combinatorial expression of TRPV channel proteins defines their sensory functions and subcellular localization in C. elegans neurons. Neuron 35, 307–318 (2002).
Wallace, S. W., Singhvi, A., Liang, Y., Lu, Y. & Shaham, S. PROS-1/Prospero Is a Major Regulator of the Glia-Specific Secretome Controlling Sensory-Neuron Shape and Function in C. elegans. Cell Rep. 15, 550–562 (2016).
Bacaj, T., Tevlin, M., Lu, Y. & Shaham, S. Glia are essential for sensory organ function in C. elegans. Sci. (N. Y., N. Y) 322, 744–747 (2008).
Li, R. et al. GCY-20 signaling controls suppression of Caenorhabditis elegans egg laying by moderate cold. Cell Rep. 43, 113708 (2024).
Ryu, M. H., Moskvin, O. V., Siltberg-Liberles, J. & Gomelsky, M. Natural and engineered photoactivated nucleotidyl cyclases for optogenetic applications. J. Biol. Chem. 285, 41501–41508 (2010).
Bhatla, N. & Horvitz, H. R. Light and hydrogen peroxide inhibit C. elegans Feeding through gustatory receptor orthologs and pharyngeal neurons. Neuron 85, 804–818 (2015).
Gong, J. et al. The C. elegans Taste Receptor Homolog LITE-1 Is a Photoreceptor. Cell 168, 325 (2017).
Hobert, O. The neuronal genome of Caenorhabditis elegans. WormBook, 1–106 (2013).
Wojtyniak, M., Brear, A. G., O’Halloran, D. M. & Sengupta, P. Cell- and subunit-specific mechanisms of CNG channel ciliary trafficking and localization in C. elegans. J. Cell Sci. 126, 4381–4395 (2013).
Taylor, S. R. et al. Molecular topography of an entire nervous system. Cell 184, 4329–4347 e4323 (2021).
Zhang, B. et al. Environmental Temperature Differentially Modulates C. elegans Longevity through a Thermosensitive TRP Channel. Cell Rep. 11, 1414–1424 (2015).
Chatzigeorgiou, M. et al. Specific roles for DEG/ENaC and TRP channels in touch and thermosensation in C. elegans nociceptors. Nat. Neurosci. 13, 861–868 (2010).
Gong, J. et al. A Cold-Sensing Receptor Encoded by a Glutamate Receptor Gene. Cell 178, 1375–1386 e1311 (2019).
Fujiwara, M., Ishihara, T. & Katsura, I. A novel WD40 protein, CHE-2, acts cell-autonomously in the formation of C. elegans sensory cilia. Development 126, 4839–4848 (1999).
Pokala, N., Liu, Q., Gordus, A. & Bargmann, C. I. Inducible and titratable silencing of Caenorhabditis elegans neurons in vivo with histamine-gated chloride channels. Proc. Natl. Acad. Sci. USA 111, 2770–2775 (2014).
Apfeld, J. & Kenyon, C. Regulation of lifespan by sensory perception in Caenorhabditis elegans. Nature 402, 804–809 (1999).
Tsunozaki, M., Chalasani, S. H. & Bargmann, C. I. A behavioral switch: cGMP and PKC signaling in olfactory neurons reverses odor preference in C. elegans. Neuron 59, 959–971 (2008).
Hedgecock, E. M. & Russell, R. L. Normal and mutant thermotaxis in the nematode Caenorhabditis elegans. Proc. Natl. Acad. Sci. USA 72, 4061–4065 (1975).
Jurado, P., Kodama, E., Tanizawa, Y. & Mori, I. Distinct thermal migration behaviors in response to different thermal gradients in Caenorhabditis elegans. Genes Brain Behav. 9, 120–127 (2010).
Inada, H. et al. Identification of guanylyl cyclases that function in thermosensory neurons of Caenorhabditis elegans. Genetics 172, 2239–2252 (2006).
Ding, G. et al. In vivo tactile stimulation-evoked responses in Caenorhabditis elegans amphid sheath glia. PLoS ONE 10, e0117114 (2015).
Beg, A. A. & Jorgensen, E. M. EXP-1 is an excitatory GABA-gated cation channel. Nat. Neurosci. 6, 1145–1152 (2003).
Lee, J., Song, H. O., Jee, C., Vanoaica, L. & Ahnn, J. Calcineurin regulates enteric muscle contraction through EXP-1, excitatory GABA-gated channel, in C. elegans. J. Mol. Biol. 352, 313–318 (2005).
Kashio, M. & Tominaga, M. TRP channels in thermosensation. Curr. Opin. Neurobiol. 75, 102591 (2022).
Yuan, J. et al. Controlled Activation of TRPV1 Channels on Microglia to Boost Their Autophagy for Clearance of Alpha-Synuclein and Enhance Therapy of Parkinson’s Disease. Adv. Mater. 34, e2108435 (2022).
Shibasaki, K., Ikenaka, K., Tamalu, F., Tominaga, M. & Ishizaki, Y. A novel subtype of astrocytes expressing TRPV4 (transient receptor potential vanilloid 4) regulates neuronal excitability via release of gliotransmitters. J. Biol. Chem. 289, 14470–14480 (2014).
Chung, M. K., Lee, H., Mizuno, A., Suzuki, M. & Caterina, M. J. TRPV3 and TRPV4 mediate warmth-evoked currents in primary mouse keratinocytes. J. Biol. Chem. 279, 21569–21575 (2004).
Sadler, K. E., Moehring, F. & Stucky, C. L. Keratinocytes contribute to normal cold and heat sensation. Elife 9 (2020).
Almoril-Porras, A. et al. Configuration of electrical synapses filters sensory information to drive behavioral choices. Cell 188, 89–103 e113 (2025).
Jang, H. et al. Dissection of neuronal gap junction circuits that regulate social behavior in Caenorhabditis elegans. Proc. Natl. Acad. Sci. USA 114, E1263–E1272 (2017).
Chatzigeorgiou, M. & Schafer, W. R. Lateral facilitation between primary mechanosensory neurons controls nose touch perception in C. elegans. Neuron 70, 299–309 (2011).
Vega-Zuniga, T. et al. A thalamic hub-and-spoke network enables visual perception during action by coordinating visuomotor dynamics. Nat. Neurosci. 28, 627–639 (2025).
Hilliard, M. A. et al. In vivo imaging of C. elegans ASH neurons: cellular response and adaptation to chemical repellents. EMBO J. 24, 63–72 (2005).
Kaplan, J. M. & Horvitz, H. R. A dual mechanosensory and chemosensory neuron in Caenorhabditis elegans. Proc. Natl. Acad. Sci. USA 90, 2227–2231 (1993).
Brenner, S. The genetics of Caenorhabditis elegans. Genetics 77, 71–94 (1974).
Christensen, M. et al. A primary culture system for functional analysis of C. elegans neurons and muscle cells. Neuron 33, 503–514 (2002).
Acknowledgements
This work was supported by grants from the National Foundation of Natural Science of China (32271017, 31771113 and 31471023 to L.K.; 32571180 to W.Z. and 82071851 to J.G.), the STI2030 Major Projects (2021ZD0203303 to L.K.), Zhejiang Provincial Natural Science Foundation (LZ22C090001 to L.K.; Y24C090003 to W.Z.), and the Non-profit Central Research Institute Fund of Chinese Academy of Medical Sciences (2023-PT310-01). The funding agencies were not involved in study design, data collection, analysis, publication decisions, or manuscript preparation. We thank Dr. Zhengxing Wu, Dr. Shangbang Gao, Dr. Cong Ma, Dr. Wei Zou, Dr. Lianfeng Wu, and Dr. Zhitao Hu for providing some of the strains and plasmids. We thank Sanhua Fang, Li Liu and Dan Yang from the Core Facilites, Zhejiang University School of Medicine for technical support, and thank the Caenorhabditis Genetic Center (CGC), which is supported by NIH Office of Research Infrastructure Programs (P40 OD010440), for providing strains.
Author information
Authors and Affiliations
Contributions
L.Z. and L.K. conceptually designed experiments; L.Z., R.L., M.Q., F.L., H.H., Y.L., Y.Z., Z.L., J.L., and W.Z. performed experiments; L.Z., R.L., J.G., and L.K. performed data analysis; L.Z., R.L., Y.Z., G.Z., H.M., and L.K. interpreted the data; L.K. supervised; L.Z., M.G., J.G., and L.K. prepared the manuscript with input from all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Laura Bianchi and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhu, L., Li, R., Qian, M. et al. A Glial Hub-and-Spoke Circuitry in C. elegans orchestrates bidirectional thermosensation. Nat Commun 17, 1899 (2026). https://doi.org/10.1038/s41467-026-68766-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-026-68766-w