Abstract
C-type natriuretic peptide (CNP) is released from endothelial cells and acts as an autocrine/paracrine mediator, regulating systemic blood pressure and vascular remodeling via guanylyl cyclase-B (GC-B) and natriuretic peptide receptor-C. We investigate the impact of vascular CNP/GC-B signaling on the development of pulmonary arterial hypertension (PAH). Mice developing pulmonary hypertension (PH) show reduced pulmonary NPPC and NPR2 expression than mice without PH. Similarly, endothelial cells (EC) from patients with idiopathic PAH exhibit lower NPPC and NPR2 expression than control EC. EC-specific CNP or GC-B conditional knockout (CNP ecKO or GC-B ecKO) mice, but not smooth muscle cell-specific GC-B conditional knockout (GC-B smcKO) mice, show more severe PH and greater expression of Edn1, Il6, Ccl2 and Tgfb1 mRNAs than their genetic controls in PAH models. CNP suppresses hypoxia-induced increases in expression of these mRNAs and restored SMAD2/3-SMAD1/5/9 balance in cultured human pulmonary arterial EC. Moreover, CNP administration prevents PH in genetic control and GC-B-smcKO mice but not in GC-B ecKO mice. CNP administration also has therapeutic effects against Sugen5416-hypoxia PAH models as well as additive benefits with established therapies. Endothelial CNP/GC-B signaling thus exerts pivotal preventative effects against development of PH, suggesting the therapeutic potential of CNP for PAH.
Similar content being viewed by others
Introduction
Pulmonary arterial hypertension (PAH) is a life-threatening disease that leads to progressive right heart failure and death. The pathogenesis of PAH is diverse and includes pathological conditions defined by constriction and remodeling of the small pulmonary arteries1. Although new drug development and advances in treatment strategies over the last few decades have steadily improved the overall prognosis of PAH patients, those severely affected by the disease continue to have a poor prognosis and may even require lung transplantation2,3. Currently, drugs acting on three different systems are in clinical use for the treatment of PAH. However, all these drugs largely act to lower pulmonary arterial pressure through vasodilation4. As the prognostic value of this combination of drugs remains unsatisfactory, the development of drugs based on novel therapeutic mechanisms is an urgent issue in the treatment of PAH5.
The natriuretic peptide system consists of three ligands, atrial natriuretic peptide, brain (or B-type) natriuretic peptide (BNP) and C-type natriuretic peptide (CNP), and three receptors, natriuretic peptide receptor-A (NPR-A, also known as guanylyl cyclase-A, GC-A), natriuretic peptide receptor-B (NPR-B, also known as natriuretic peptide receptor-2, NPR2 and guanylyl cyclase-B, GC-B) and natriuretic peptide receptor-C (NPR-C, also referred to as the clearance receptor)6. GC-A and GC-B are transmembrane receptors that bind their ligand and function as particulate guanylyl cyclases to enhance cyclic guanosine monophosphate (cGMP) generation6. CNP is widely expressed in multiple organs, including the central nervous system, osteochondral system and cardiovascular system6. CNP exerts its biological effects via two receptors, GC-B and NPR-C, though mice globally lacking NPR-C exhibit longer bones and mild hypotension, which is consistent with the idea that NPR-C functions primarily as a clearance receptor for natriuretic peptide degradation6,7.
Within the vascular system, GC-B and NPR-C are expressed in multiple cell types, including vascular endothelial and smooth muscle cells8. However, CNP is predominantly released from endothelial cells8. Available evidence indicates endothelium-derived CNP plays a significant role in regulating vascular tone and systemic blood pressure (BP), while also exerting protective effects against pathological vascular remodeling. We previously reported that endothelium-derived CNP contributes to the chronic regulation of vascular tone and systemic BP, in part by contributing to the maintenance of endothelial function9. Studies by other groups also showed that CNP-induced GC-B/cGMP signaling in microvascular smooth muscle cells and pericytes is essential for maintaining normal microvascular resistance and systemic BP and that endothelium-derived CNP preserves vascular homeostasis via a CNP/NPR-C pathway within the systemic circulation10,11.
Still, the roles of endothelial cell-derived CNP and its target cell types and receptor-mediated signaling pathways in maintaining pulmonary vascular homeostasis and the development of PAH remained uncertain. Earlier studies yielded conflicting results regarding the therapeutic effects of continuous CNP infusion in rats with pulmonary hypertension (PH)12,13. In the present study, we used vascular endothelial-specific CNP knockout (CNP ecKO) mice to investigate the role of the endothelium-derived CNP in the pathophysiology of PAH and used endothelial- or smooth muscle cell-specific GC-B knockout (GC-B ecKO or GC-B smcKO, respectively) mice to determine its target cells and dissect its receptor signaling in two mouse PAH models, monocrotaline pyrrole (MCTp)-induced14 and hypoxia (Hx)-induced15 PH and in human pulmonary endothelial cells (HPAECs). We also examined the ability of CNP administration to prevent the development of PH and determined its target cells in GC-B ecKO and GC-B smcKO mice in the two PAH models. In addition, we evaluated the therapeutic effect of CNP administration on SU5416 combined with hypoxia (SuHx) rat and mouse PAH models16,17. Our results demonstrate the pivotal protective role played by endothelial CNP/GC-B signaling against the progression of PH and suggest the therapeutic potential of CNP for the treatment of PAH.
Results
Endothelial cell-derived CNP prevents progression of PH in mouse models
We evaluated the effect of endogenous endothelial-derived CNP on the development of PAH. Because global deletion of CNP leads to early death due to osteochondral impairment18, we generated CNP endothelial conditional knockout (ecKO) mice by crossing Nppcflox/flox mice with transgenic mice expressing Cre recombinase under the control of the Tie2 promotor (Tie2-Cre mice). We then confirmed that Nppc mRNA levels in the lungs of CNP ecKO mice were reduced to less than 20% of that in the lungs of control Tie2-Cre mice, indicating CNP expression in the lungs occurs primarily in pulmonary endothelial cells (Supplementary Fig. 1a). CNP ecKO mice showed normal fertility and growth, though their systemic BP was significantly higher than in control mice, as we previously reported9 (Tie2-Cre mice vs. CNP ecKO; 111.24 vs. 116.24 mmHg, p = 0.0236, Supplementary Fig. 1b). There were no significant differences in echocardiographic parameters, including interventricular septal wall thickness (IVS), left ventricular end-diastolic diameter (LVDd) and left ventricular ejection fraction (LVEF), between CNP ecKO and control mice (Supplementary Fig. 1c–e).
We next produced MCTp-induced PH14 in CNP ecKO and control Tie2-Cre mice. Two weeks after the injection of MCTp into the tail vein, right ventricular systolic pressures (RVSP) measured using right heart catheterization and right ventricle weight/left ventricle plus interventricular septum weight [RV/(LV + IVS)] ratios were significantly higher in MCTp-treated than untreated Tie2-Cre mice (Fig. 1a, b). Levels of Nppc and Npr2 mRNA expression in the lungs of MCTp-treated mice were significantly reduced to about 65% and 85% of that in the untreated mice (Fig. 1c, d), which suggests the CNP-GC-B system was impaired in MCTp-induced PH. Although the RVSP and RV/(LV + IVS) ratios in untreated CNP ecKO mice were similar to those in untreated Tie2-Cre mice, MCTp-treated CNP ecKO mice showed significantly higher RVSP and RV/(LV + IVS) ratios than MCTp-treated Tie2-Cre mice (Fig. 1a, b). There were no significant differences in BP, IVS, LVDd or LVEF between MCTp-treated and untreated mice, irrespective of genotype (Supplementary Fig. 1b–e). Histological analysis showed that the pulmonary arterial medial wall thickness (percent medial wall thickness), which is an indicator of vascular remodeling, was also significantly higher in MCTp-treated CNP ecKO mice than in MCTp-treated Tie2-Cre mice (Fig. 1e, f).
RVSP (a) and RV/(LV + IVS) ratios (b) in CNP ecKO mice and Tie2-Cre controls following MCTp treatment (10-week-old; n = 5 per group). Whole-lung mRNA expression of Nppc (c) and Npr2 (d) in Tie2-Cre mice with or without MCTp treatment (10-week-old; n = 5 per group). Representative pulmonary artery sections stained with hematoxylin-eosin and Victoria blue (e; scale bar, 50 μm) and quantification of percent medial wall thickness (f) in MCTp-treated CNP ecKO and Tie2-Cre mice. RVSP (g) and RV/(LV + IVS) ratios (h) in CNP ecKO and Tie2-Cre mice exposed to Hx (12-week-old; n = 6 per group). Whole-lung mRNA expression of Nppc (i) and Npr2 (j) in Tie2-Cre mice under normoxia or Hx (12-week-old; n = 6 per group). Representative pulmonary artery sections stained with hematoxylin-eosin and Victoria blue (k; scale bar, 50 μm) and quantification of medial wall thickness (l) in normoxic and Hx mice (12-week-old; n = 5 for normoxia, n = 10 for Hx). Data are presented as the mean ± SEM. P values were calculated using one-way ANOVA with Tukey’s multiple comparisons test.
We also generated a Hx-induced mouse PH model. CNP ecKO and control Tie2-Cre mice were housed in a Hx chamber (10% O2) for 4 weeks19 and then evaluated. RVSP and RV/(LV + IVS) ratios were significantly higher in Hx-exposed than untreated Tie2-Cre mice (Fig. 1g, h). Levels of Nppc and Npr2 mRNA expression in the lungs of Hx-exposed Tie2-Cre mice were significantly reduced to about 62% and 85% of that in the untreated Tie2-Cre mice (Fig. 1i, j), suggesting the CNP-GC-B system was also impaired in Hx-induced PH. Notably, RVSP was significantly higher in Hx-exposed CNP ecKO mice than Hx-exposed Tie2-Cre mice (Fig. 1g). Moreover, RV/(LV + IVS) ratios and pulmonary arterial medial wall thickness were also significantly higher in Hx-exposed CNP ecKO mice than Hx-exposed Tie2-Cre mice (Fig. 1h, k, l). There were no significant differences in BP, IVS, LVDd or LVEF between untreated and Hx-exposed mice, irrespective of genotype (Supplementary Fig. 1f–i). These results demonstrate the impairment of the CNP-GC-B system in both the MCTp-induced PAH model and Hx-induced PH model and the protective role of endothelial cell-derived CNP against the progression of PH in those models.
GC-B signaling in endothelial cells, but not smooth muscle cells, prevents the progression of PH in mouse models
CNP exerts its effects primarily by binding to GC-B, which is expressed in several cell types, including vascular endothelial and smooth muscle cells. Indeed, global GC-B KO and CNP KO mice exhibit similar phenotypes that include, for example, severely impaired bone growth18,20. We then sought to clarify whether it was vascular endothelial cells or smooth muscle cells that mediate the protective effect of endothelial-derived CNP against the development of PH by selectively knocking out GC-B in vascular endothelial or smooth muscle cells.
We generated GC-B ecKO mice by crossing Npr2flox/flox mice with Tie2-Cre mice, and GC-B smooth muscle conditional KO (smcKO) mice by crossing Npr2flox/flox mice with transgenic mice expressing Cre recombinase under the control of the SM22α promotor (SM22α-Cre mice). Npr2 mRNA levels in both GC-B ecKO and GC-B smcKO mice were reduced to ~50% of those in their control mice (Tie2-Cre mice and Sm22α-Cre mice, respectively), indicating comparable expression of Npr2 mRNA in pulmonary endothelial and smooth muscle cells (Supplementary Figs. 2a and 3a). In addition, immunohistochemistry (IHC) confirmed the protein level expression of NPR2 in the pulmonary arteries. To validate the ability of the GC-B antibody to distinguish between GC-A and GC-B, given their sequence similarity (61.99% homology, as confirmed by the manufacturer), we performed siRNA-mediated knockdown of GC-B (siGCB) in HPAECs. RT-PCR confirmed a marked and selective reduction in Npr2 mRNA levels (Supplementary Fig. 4a, b). Moreover, Western blot analysis showed a corresponding decrease in GC-B protein levels in siGCB-treated cells. Importantly, the signal detected with an NPR-A antibody remained unchanged under these conditions, which is indicative of the minimal cross-reactivity and the specificity of the GC-B antibody (Supplementary Fig. 4c–e). IHC showed that within pulmonary arterioles of the size mainly affected by PH, NPR2 was expressed in both endothelial and smooth muscle cells in control mice (Supplementary Fig. 4f) but was barely detectable in endothelial cells from GC-B ecKO mice (Supplementary Fig. 4g) or in smooth muscle cells from GC-B smcKO mice (Supplementary Fig. 4h). We also confirmed that SM22α is similarly expressed in smooth muscle cells in the relevant sized vessels (Supplementary Fig. 4i). These cell type-specific KO mice showed normal fertility and growth (Supplementary Figs. 2b and 3b), and there were no significant differences in BP between GC-B ecKO and GC-B smcKO mice and their respective control mice (Supplementary Figs 2c, g and 3c, g respectively). Likewise, there were no significant differences in IVS, LVDd or LVEF between GC-B ecKO or GC-B smcKO mice and their controls (Supplementary Figs. 2d–f, h–j and 3d–f, h–j, respectively).
When we then generated MCTp-induced PH in GC-B ecKO and GC-B smcKO mice, MCTp-treated GC-B ecKO mice showed significantly higher RVSP than MCTp-treated Tie2-Cre mice (Fig. 2a). By contrast, no significant difference in RVSP was detected between MCTp-treated GC-B smcKO and MCTp-treated SM22α-Cre mice (Fig. 2b). Similarly, RV/(LV + IVS) ratios and percent medial wall thicknesses were significantly greater in MCTp-treated GC-B ecKO than MCTp-treated Tie2-Cre mice, whereas they were comparable between MCTp-treated GC-B smcKO and MCTp-treated SM22α-Cre mice (Fig. 2c–e, f–h, respectively). There were also no significant differences in BP, IVS, LVDd or LVEF between control mice (Tie2-Cre mice or SM22α-Cre mice) and GC-B ecKO or GC-B smcKO in the MCTp-induced PAH model (Supplementary Figs. 2d–g and 3d–f). In addition, cardiac output (CO) assessed based on echocardiography did not significantly differ among genotypes in the MCTp model under either non-PH or PH conditions (Supplementary Fig. 5a).
RVSP in GC-B ecKO versus Tie2-Cre mice (a; 10-week-old; n = 5 per group) and in GC-B smcKO versus SM22α-Cre mice (b; 10-week-old; n = 4 per group) following MCTp treatment. c–e GC-B ecKO versus Tie2-Cre mice following MCTp treatment. RV/(LV + IVS) ratios (c), representative pulmonary artery sections stained with hematoxylin–eosin and Victoria blue (d; scale bar, 50 μm), and quantification of medial wall thickness (e) (10-week-old; n = 5 for MCTp (–), n = 10 for MCTp (+)). f–h GC-B smcKO versus SM22α-Cre mice following MCTp treatment. RV/(LV + IVS) ratios (f), representative pulmonary artery sections stained with hematoxylin–eosin and Victoria blue (g; scale bar, 50 μm), and quantification of medial wall thickness (h) (10-week-old; n = 5 for MCTp (–), n = 10 for MCTp (+)). Data are presented as the mean ± SEM. P values were calculated using one-way ANOVA with Tukey’s multiple comparisons test.
Generation of Hx-induced PH in GC-B ecKO and GC-B smcKO mice yielded results similar to those in the MCTp model. RVSP and RV/(LV + IVS) ratios were significantly higher in Hx-exposed GC-B ecKO than Hx-exposed Tie2-Cre mice (Fig. 3a, b). In addition, pulmonary arterial medial wall thicknesses were significantly greater in Hx-exposed GC-B ecKO than Hx-exposed Tie2-Cre mice (Fig. 3c, d). On the other hand, RVSP, RV/(LV + IVS) ratios and pulmonary arterial medial wall thickness did not significantly differ between Hx-exposed GC-B smcKO mice and Hx-exposed SM22α-Cre mice (Fig. 3e–h). Similarly, CO assessed based on echocardiography remained unchanged across all genotypes under both non-PH and PH conditions (Supplementary Fig. 5b). Together, these findings indicate that the protective effects of endothelial-derived CNP against the progression of PH are predominantly mediated through endothelial GC-B in both the MCTp-induced PAH model and Hx-induced PH model.
a–d GC-B ecKO versus Tie2-Cre mice following Hx exposure. RVSP (a), RV/(LV + IVS) ratios (b), representative pulmonary artery sections stained with hematoxylin-eosin and Victoria blue (c; scale bar, 50 μm), and quantification of medial wall thickness (d) (12-week-old; n = 5 per group). e–h GC-B smcKO versus SM22α-Cre mice following Hx exposure. RVSP (e), RV/(LV + IVS) ratios (f), representative pulmonary artery sections stained with hematoxylin-eosin and Victoria blue (g; scale bar, 50 μm), and quantification of medial wall thickness (h) (12-week-old; n = 5 for normoxia, n = 10 for Hx). Data are presented as the mean ± SEM. P values were calculated using one-way ANOVA with Tukey’s multiple comparisons test.
As described above, the protective actions of the endothelial CNP/GC-B pathway against PH progression were investigated using male mice. Because human PAH shows sex-related differences with higher prevalence and survival rates in females21, we compared PH progression between male and female mice of each genotype. We found no significant difference in RVSP or RV/(LV + IVS) ratios between the sexes, even under PH conditions (Supplementary Fig. 6a–d).
Endothelial CNP/GC-B signaling represses expression of endothelin-1 and proinflammatory cytokines
When we evaluated whole lung expression of Nppc and Npr2 mRNA in control and MCTp-induced or Hx-induced PH model mice, we found that GC-B ecKO and GC-B smcKO mice showed no significant changes in Nppc mRNA levels as compared to their genetic control mice (Tie2-Cre or SM22α-Cre mice) (Fig. 4a, b). In both PH models, pulmonary Nppc mRNA expression in both Tie2-Cre and SM22α-Cre mice was significantly lower in mice with PH than in untreated mice without PH (Figs. 1c, i and 4a, b). Similarly, in both PH models, pulmonary Npr2 mRNA expression in both control Tie2-Cre and SM22α-Cre mice was significantly lower in mice with PH than in untreated mice without PH (Figs. 1d, j and 4c, d). This indicates a decrease in pulmonary vascular CNP/GC-B signaling in these two PH models. In both models, pulmonary Npr1 mRNA expression was significantly lower in mice with PH than in untreated mice with PH mice, irrespective of genotype, but there were no significant differences in Npr1 mRNA expression among Tie2-Cre, SM22α-Cre, CNP ecKO, GC-B ecKO and GC-B smcKO mice, with or without PH (Fig. 4e, f).
RT-qPCR analyses of the whole lung expression of Nppc (a, b), Npr2 (c, d), Npr1 (e, f), Edn1 (g, h), Il6 (i, j), Ccl2 (k, l), and Tgfb1 (m, n) in Tie2-Cre, SM22α-Cre, CNP ecKO, GC-B ecKO and GC-B smcKO mice in the MCTp-induced model (10-week-old, a, c, e, g, i, k, m) and Hx-induced model (12-week-old, b, d, f, h, j, l, n) compared with untreated controls (10-week-old of MCTp (–) and 12-week-old normoxia; n = 8 per group). GAPDH was used as the internal control. Data are presented as the mean ± SEM. P values were calculated using one-way ANOVA with Tukey’s multiple comparisons test.
We next examined the expression of endothelin-1 (ET-1), a key vasoconstrictive and mitogenic mediator implicated in the development of PAH22. At baseline, levels of endothelin 1 (Edn1) mRNA were significantly higher in CNP ecKO and GC-B ecKO mice, but not in GC-B smcKO mice, than in their respective genetic controls (Fig. 4g, h). In both the MCTp- and Hx-induced PH models, expression of Edn1 mRNA was greater in mice with PH than in untreated mice without PH, irrespective of genotype (Fig. 4g, h). Importantly, in both the MCTp- and Hx-induced PH models, the increased expression in Edn1 mRNA was more enhanced in CNP ecKO and GC-B ecKO mice with PH than in Tie2-Cre mice with PH (Fig. 4g, h). By contrast, the increase in Edn1 mRNA expression in MCTp- or Hx-treated GC-B smcKO mice did not significantly differ from the increase in MCTp- or Hx-treated SM22α-Cre mice (Fig. 4g, h). In addition, the whole lung mRNA expression of the pro-inflammatory cytokines of interleukin (IL) 6 (Il6), C-C motif chemokine ligand 2 (CCL2) (Ccl2), and transforming growth factor-β1 (TGFB1) (Tgfb1), which are known to facilitate the PAH progression, was significantly induced by MCTp or Hx treatment in each genotype (Fig. 4i–n). Moreover, the MCTp- and Hx-induced increases in pulmonary mRNA expression of these cytokines was greater in CNP ecKO and GC-B ecKO mice, but not GC-B smcKO mice, than in their genetic controls (Fig. 4i–n). These results suggest endothelial CNP/GC-B signaling prevents the progression of PH in mice, at least in part, by suppressing expression of Edn1 and several pro-inflammatory cytokines.
We then performed in vitro experiments to determine whether the observed CNP/GC-B signaling-dependent changes in mRNA expression occurred in vascular endothelial cells. We first examined expression of Nppc and Npr2 mRNA in Hx-exposed HPAECs. Expression levels of both Nppc and Npr2 mRNA were significantly decreased by Hx, which is consistent with the in vivo data showing that CNP/GC-B signaling is attenuated under the pathological conditions associated with PH (Figs. 4a–d and 5a, b). We also assessed expression of EDN1, IL6, CCL2 and TGFB1 mRNA in Hx-exposed HPAECs transfected with control scrambled small interfering RNA (siRNA) or GC-B siRNA (siGC-B). Consistent with the mouse models, levels of EDN1, IL6, CCL2 and TGFB1 mRNA expression were higher in Hx-exposed HPAECs than in untreated control cells following transfection of control siRNA (Fig. 5c–f). After GC-B knockdown using siGC-B (Fig. 5g), expression of EDN1 and IL6 mRNA was significantly higher in the knockdown HPAECs than in HPAECs transfected with control siRNA, even in untreated cells (Fig. 5c, d). Knocking down GC-B also led to increases in CCL2 and TGFB1 expression in these cells under untreated conditions, though the effect was not significant (Fig. 5e, f). The increases in EDN1, IL6, CCL2 and TGFB1 mRNA expression induced by Hx were greater in HPAECs transfected with siGC-B than in those transfected with control siRNA (Fig. 5c–f). Expression of NPRC mRNA was detectable in HPAECs, but siRNA-mediated knockdown of NPR-C did not significantly alter EDN1, IL6 or CCL2 expression under normoxic conditions (Supplementary Fig. 7a–d).
Quantitative real-time RT-PCR analyses of NPPC (a) and NPR2 (b) mRNA expression in HPAECs exposed to Hx. mRNA expression of EDN1 (c), IL6 (d), CCL2 (e), and TGFB1 (f) in HPAECs transfected with control siRNA or siGC-B under normoxic and Hx conditions, with or without CNP-53 treatment. Hx-induced upregulation of these genes was abolished by CNP-53 in control cells, but not in siGC-B-transfected cells. g Knockdown efficiency of GC-B shown as reduced NPR2 mRNA levels in siGC-B–transfected HPAECs compared to scramble controls. (n = 8 biological replicates, independent culture wells, per group). GAPDH was used as the internal control. Data are presented as the mean ± SEM. P values were calculated using one-way ANOVA with Tukey’s multiple comparisons test.
CNP-53 is an endogenous molecular form of CNP with longer lasting biological activity than CNP-22 in vivo because it is less sensitive to neutral endopeptidase (NEP)23,24,25. To compare CNP-22 and CNP-53 for their ability to elevate cGMP levels, mice were infused for 7 days with vehicle, CNP-22 or CNP-53 (0.2 mg/kg/day) using osmotic pumps. CNP-53 significantly increased urinary cGMP concentrations as compared to vehicle. The increase in urinary cGMP induced by CNP-22 was comparable to that induced by CNP-53 but did not significantly differ from that induced by vehicle (Supplementary Fig. 8). Addition of CNP-53 to the culture medium of HPAECs significantly suppressed the increases in EDN1, IL6, CCL2 and TGFB1 mRNA expression induced by Hx in HPAECs transfected with control siRNA (Fig. 5c–f). On the other hand, the suppressive effect of CNP-53 on the Hx-induced increases in EDN1, IL6, CCL2 and TGFB1 mRNA expression was nearly completely abolished in HPAECs transfected with siGC-B (Fig. 5c–f). Taken together, these findings indicate that an endothelial CNP/GC-B axis suppresses the increase in endothelial expression of EDN1, IL6, CCL2 and TGFB1 mRNA induced by pathological stimuli related to the progression of PAH.
CNP corrects pathological alterations in SMAD signaling pathways in endothelial cells
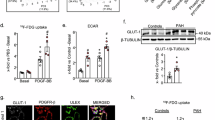
It was recently proposed that PAH is associated with an imbalance between the bone morphogenetic protein receptor type II (BMPRII)-SMAD1/5/9 pathway, which is an antiproliferative signaling pathway, and the activin receptor-SMAD2/3 pathway, which is a pro-proliferative signaling pathway, leading to pathological pulmonary arterial remodeling26,27. We therefore evaluated the effects of CNP/GC-B signaling on SMAD signaling pathways in Hx-exposed HPAECs. Whereas Hx-exposure significantly increased phosphorylated (phospho)-SMAD2/3 levels (normalized to total SMAD2/3), addition of CNP-53 significantly suppressed those increases in phosho-SMAD2/3 levels, and this suppressive effect of CNP-53 on the activation of SMAD2/3 was abolished by Rp-8-Br-cGMP, a cGMP-dependent protein kinase (PKG) inhibitor (Fig. 6a, b). By contrast, Hx significantly suppressed phospho-SMAD1/5/9 levels in HPAECs, but CNP-53 significantly reversed the Hx-induced suppression of phosho-SMAD1/5/9 levels (normalized to SMAD1), and these effects were abolished in the presence of PKG inhibitor (Fig. 6c, d). In addition, in HPAECs transfected with control siRNA, mRNA expression of inhibitor of differentiation 1 (ID1), whose expression is induced by activation of the SMAD1/5/9 pathway28, was significantly decreased by Hx, and CNP-53 significantly reversed the Hx-induced decrease in ID1 mRNA expression. Moreover, this effect of CNP on the Hx-induced decrease in expression of ID1 mRNA was completely abolished in HPAECs transfected with siGC-B (Fig. 6e). These changes of ID1 mRNA expression were consistent with the changes in SMAD1/5/9 phosphorylation. Expression of both BMPR2 mRNA and the BMPRII protein it encodes were reduced by Hx in HPAECs, and CNP-53 administration did not affect those Hx-induced decreases in these expression (Fig. 6f–h). In vivo, treatment with MCTp or Hx significantly decreased pulmonary Id1 mRNA expression in Tie2-Cre and SM22α-Cre mice, and those effects were greater in CNP ecKO and GC-B ecKO mice, but not in GC-B smcKO mice, than in Tie2-Cre or SM22α-Cre mice (Fig. 6i, j). On the other hand, the reductions in Bmpr2 mRNA expression seen in MCTp- and Hx-treated mice did not differ among the tested genotypes (Fig. 6k, l). These in vivo effects are consistent with those seen with Hx-exposed HPAECs.
Representative western blots of phosphorylated SMAD2/3 and total SMAD2/3 (a), and quantitative analysis of phosphorylated SMAD2/3 normalized to total SMAD2/3 (b) in HPAECs exposed to Hx with or without CNP or the PKGi (n = 4 biological replicates, independent culture wells, per group). Representative blots of phosphorylated SMAD1/5/9 and total SMAD1 (c) and quantitative analysis of phosphorylated SMAD1/5/9 normalized to total SMAD1 (d) under the same conditions (n = 4 biological replicates, independent culture wells, per group). e Effect of CNP and GC-B knockdown on Hx-induced expression of ID1 mRNA in HPAECs (n = 8 biological replicates, independent culture wells, per group). f Effect of CNP on Hx-induced expression of BMPR2 mRNA in HPAECs (n = 8 biological replicates, independent culture wells, per group). Representative blots of BMPRII and GAPDH (g) and quantitative analysis of BMPRII normalized to GAPDH (h) in HPAECs exposed to Hx with or without CNP (n = 3 per biological replicates, independent culture wells, group). Expression of Id1 (i, j) and Bmpr2 (k, l) mRNA in whole lungs from MCTp- or Hx-treated CNP ecKO, GC-B ecKO and GC-B smcKO mice and their controls (Tie2-Cre for CNP ecKO and GC-B ecKO; SM22α-Cre for GC-B smcKO) (n = 6 per group). m, Effect of CNP on Hx-induced expression of INHBA mRNA in HPAECs (n = 8 biological replicates, independent culture wells, per group). Expression of Inhba mRNA in whole lungs from MCTp- (n) and Hx-treated (o) CNP ecKO, GC-B ecKO, and GC-B smcKO mice and their respective controls (10-week-old MCTp (–)/(+) and 12-week-old normoxia/Hx; n = 6 per group). CNP and PKGi were added to culture medium at final concentrations of 1.0 μmol/L and 30 μg/mL, respectively. GAPDH was used as the internal control. Data are presented as the mean ± SEM. P values were calculated using one-way ANOVA with Tukey’s multiple comparisons test.
Moreover, mRNA expression of inhibin-β-a (INHBA) mRNA, which encodes the protein activin-A, a TGF-β superfamily cytokine that activates the SMAD2/3 pathway29, was significantly increased by Hx in HPAECs transfected with control siRNA (Fig. 6m). CNP-53 significantly suppressed the Hx-induced increase of INHBA expression in HPAECs transfected with control siRNA, but this suppressive effect was abolished in HPAECs transfected with siGC-B (Fig. 6m). Consistent with that finding, pulmonary expression of Inhba mRNA was significantly induced by MCTp or Hx treatment in genetic control mice (Fig. 6n, o). The increases in pulmonary Inhba mRNA expression induced by MCTp or Hx were greater in CNP ecKO and GC-B ecKO mice, but not in GC-B smcKO mice, than in the genetic control mice (Fig. 6n, o). These results suggest that preventing the pathological imbalance between the SMAD2/3 and SMAD1/5/9 signaling pathways and the pathological increase in INHBA gene expression may also underlie the molecular mechanisms by which endothelial CNP/GC-B/PKG signaling prevents the progression of PH.
Endothelial CNP-GC-B signaling affects PASMC proliferation and migration
Hypoxia-induced PASMC proliferation was suppressed in conditioned medium (CM) from control HPAECs, but this suppressive effect was lost when the CM was derived from GC-B-deficient HPAECs (Fig. 7a). By contrast, GC-B knockdown in PASMCs did not alter their proliferative response to control CM. The knockdown efficiency of siGC-B in HPAECs and PASMCs was confirmed by qRT-PCR (Supplementary Fig. 9a, b).
a PASMC proliferation assessed using CCK-8 assays under hypoxia. CM from GC-B-deficient HPAECs (EC+siGC-B) significantly increased PASMC proliferation compared with CM from control HPAECs (EC control). Proliferation was quantified in PASMCs (n = 12 biological replicates, independent culture wells, per group). b Schematic representation of the Boyden chamber co-culture assay. PASMCs (blue) were seeded into the upper insert, while HPAECs (red) were cultured in the lower chamber to evaluate endothelial-smooth muscle paracrine interactions. c PASMC migration measured by Boyden chamber assays under hypoxia. Migrated cells were quantified based on fluorescence intensity. Migration was significantly increased in response to CM from EC+siGC-B compared with control CM (n = 6 biological replicates, independent culture wells, per group). Representative Ki67 staining (d) and quantification (e) of PASMC proliferation in small pulmonary arteries from CNP ecKO mice in the MCTp-induced PAH model, treated with vehicle (Control), MCTp (PAH model) or MCTp + CNP (PAH model + CNP). Arrowheads indicate Ki67-positive nuclei (n = 10 per group). Representative TUNEL staining (f) and quantification (g) of PASMC apoptosis in the same groups. Arrowheads indicate TUNEL-positive nuclei (n = 10 per group). Data are presented as the mean ± SEM. P values were calculated using one-way ANOVA with Tukey’s multiple comparisons test.
Moreover, PASMC migration was significantly increased in response to CM from GC-B-deficient HPAECs as compared to CM from control HPAECs, whereas GC-B knockdown in PASMCs had no effect on migration (schematically illustrated in Fig. 7b and quantified in Fig. 7c). Consistent with these in vitro findings, pulmonary arteries from CNP ecKO mice in the MCTp-induced PAH model displayed a higher proportion of Ki67-positive nuclei and a lower proportion of TUNEL-positive nuclei than controls, and both of those effects were significantly ameliorated by CNP treatment (Fig. 7d–g).
NPPC and NPR2 expression is reduced in human pulmonary endothelial cells affected by PAH
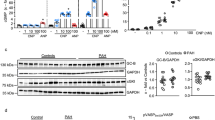
To further evaluate whether endothelial CNP-GC-B signaling is altered in human PAH, we analyzed scRNA-seq data from IPAH (n = 3) and control (n = 3) lung tissues (GSE16947130). UMAP visualization of endothelial cells revealed distinct subclusters (EC1-3) (Fig. 8a). FeaturePlots revealed sparse NPPC expression and broader NPR2 expression across the endothelial population (Fig. 8b, c). DotPlot analysis showed heterogeneous expression of NPPC and NPR2 across endothelial subclusters, suggesting potential functional specialization (Fig. 8d). And violin plots revealed that pulmonary endothelial cells affected by IPAH show significantly lower NPPC and NPR2 expression than control endothelial cells (Fig. 8e, f; NPPC: p = 7.9 × 10⁻⁴; NPR2: p = 0.043; Wilcoxon rank-sum test). These findings reveal diminished human pulmonary endothelial expression of CNP and GC-B that is consistent with the effects seen in mouse PH models and support the clinical relevance of endothelial CNP-GC-B signaling in human PAH.
a UMAP of endothelial cells (EC1-3) from control and IPAH lung samples. FeaturePlots of NPPC (b) and NPR2 (c) expression within endothelial cells. d DotPlot showing NPPC and NPR2 expression across endothelial subclusters. Violin plots comparing NPPC (e) and NPR2 (f) expression in endothelial cells between control and IPAH samples. Data are presented as the mean ± SEM. P values were calculated using the two-sided Wilcoxon rank-sum test.
CNP-53 administration suppresses the PH development via endothelial GC-B
To evaluate the therapeutic potential of CNP, we implanted osmotic mini-pumps that continuously administered CNP-53 (0.2 mg/kg/day) for 2 and 4 weeks to genetic control, GC-B smcKO and GC-B ecKO mice in the MCTp and Hx models, respectively. The dose of CNP-53 was selected based on the results of an earlier study in which CNP-53 promoted angiogenesis and vascular remodeling in vivo31. CNP-53 administration did not affect systemic BP in any genotype (Fig. 9a, b). In Tie2-Cre mice, whose phenotype is comparable to SM22α-Cre mice, CNP-53 significantly suppressed MCTp- or Hx-induced increases in RVSP and RV/(LV + IVS) ratios (Fig. 9c–f). This preventive effect of CNP-53 on the progression of PH was totally abolished in GC-B ecKO mice in both PH models (Fig. 9g–j). By contrast, in GC-B smcKO mice, as in Tie2-Cre mice, CNP-53 administration significantly suppressed MCTp- or Hx-induced increases in RVSP and RV/(LV + IVS) ratios (Fig. 9g–j). These results demonstrate that CNP-53 acts exclusively via endothelial GC-B to suppress PH development.
Effect of continuous subcutaneous infusion of CNP-53 (0.2 mg/kg/day) on systemic BP in Tie2-Cre control, GC-B ecKO and GC-B smcKO mice in the MCTp-induced model (a; 10-week-old) and Hx-induced model (b; 12-week-old) (n = 3 per group). Effects of CNP-53 infusion on RVSP (c, e) and RV/(LV + IVS) ratios (d, f) in Tie2-Cre mice under MCTp (c, d; 10-week-old) and Hx (e, f; 12-week-old) conditions (MCTp model: n = 5 for untreated [MCTp (–)/CNP (–)], n = 5 for MCTp (+)/CNP (–), n = 3 for MCTp (+)/CNP (+); Hx model: n = 5 for normoxia/CNP (–), n = 5 for Hx/CNP (–), n = 3 for Hx/CNP (+)). Effects of CNP-53 infusion on RVSP (g, i) and RV/(LV + IVS) ratios (h, j) in GC-B ecKO and GC-B smcKO mice under MCTp (g, h; 10-week-old) and Hx (i, j; 12-week-old) conditions (MCTp model: n = 5 for MCTp (–)/CNP (–), n = 5 for MCTp (+)/CNP (–), n = 3 for MCTp (+)/CNP (+); Hx model: n = 5 for normoxia/CNP (–), n = 5 for Hx/CNP (–), n = 3 for Hx/CNP (+)). Data are presented as the mean ± SEM. P values were calculated using one-way ANOVA with Tukey’s multiple comparisons test.
CNP-53 administration ameliorates PAH and exhibits additive effects with existing drugs in rodent SuHx PAH models
We also evaluated continuous CNP-53 administration using subcutaneous implanted osmotic minipumps in the SuHx rat model17, where severe PAH was induced by a single injection of the VEGF receptor inhibitor SU5416 followed by 3 weeks of hypoxia. After establishing PH in the SuHx-exposed rats, CNP-53 or vehicle administration via osmotic pumps (0.2 mg/kg/day) was initiated and continued for 3 weeks under normoxic conditions. CNP-53 significantly reduced RVSP and RV/(LV + IVS) ratios as compared to vehicle without affecting systemic BPs (Fig. 10a–c).
SuHx rat model. RVSP (a), RV/(LV + IVS) ratios (b) and BP (c) in SuHx rats treated with vehicle or CNP-53 (0.2 mg/kg/day) (n = 5 per group). d–g SuHx mouse model (wild-type). RVSP (d) and RV/(LV + IVS) ratios (e) in SuHx mice treated with vehicle, CNP-53 (0.2 mg/kg/day), macitentan (20 mg/kg/day) or the combination of macitentan and CNP-53. RVSP (f) and RV/(LV + IVS) ratios (g) in SuHx mice treated with vehicle, CNP-53, sotatercept (10 mg/kg twice weekly) or the combination of sotatercept and CNP-53 (controls and SuHx+vehicle groups, n = 5 each; macitentan group, n = 6; sotatercept group, n = 7). h–k SuHx mouse model (CNP ecKO). RVSP (h) and RV/(LV + IVS) ratios (i) in SuHx mice treated with vehicle, CNP-53 (0.2 mg/kg/day), macitentan (20 mg/kg/day) or the combination of macitentan and CNP-53. RVSP (j) and RV/(LV + IVS) ratios (k) in SuHx mice treated with vehicle, CNP-53, sotatercept (10 mg/kg twice weekly) or the combination of sotatercept and CNP-53 (controls and SuHx+vehicle groups, n = 5 each; macitentan group, n = 5; sotatercept group, n = 6). Data are presented as the mean ± SEM. P values were calculated using one-way ANOVA with Tukey’s multiple comparisons test.
We then used the SuHx mouse model of PAH16 to assess the therapeutic potential of CNP-53 administration during administration of drugs currently used for treatment of PAH. After the induction of PH with weekly SU5416 injections during 3 weeks of hypoxia, mice were returned to normoxia and randomized into treatment groups: vehicle, CNP-53 (0.2 mg/kg/day), macitentan (20 mg/kg/day)32, sotatercept (10 mg/kg, twice weekly)33, or their respective combinations with CNP-53. A non-SuHx control group was also included. Monotherapy with CNP-53, macitentan or sotatercept significantly reduced RVSP and RV/(LV + IVS) ratios as compared to SuHx treated with vehicle (Fig. 10d–k). These therapeutic effects were observed both in Tie2-Cre mice (Fig. 10d–g) and CNP ecKO mice (Fig. 10h–k), despite the greater baseline severity of PAH in CNP ecKO mice. Additive administration of CNP-53 with macitentan or sotatercept further improved RVSP as compared to monotherapy in both genotypes. Adding CNP-53 to macitentan or sotatercept also further reduced RV/(LV + IVS) ratios numerically as compared to monotherapy, but the difference did not reach statistical significance due to the already significant improvement with monotherapy in this model. Together, these findings demonstrate that exogenous CNP-53 administration has therapeutic efficacy in rodent models of severe PAH and shows additive benefit when combined with existing PAH therapies.
Discussion
This report shows that mRNA expression of CNP and GC-B is decreased in the lungs of two different mouse PAH models, Hx-exposed HPAECs and pulmonary endothelial cells affected by human IPAH, and that endothelial CNP/GC-B signaling exerts preventive effects against disease progression in several PH models. These results indicate that a decrease in endothelial CNP/GC-B signaling plays an important role in the progression of PAH. The preventive effects of CNP/GC-B signaling appear to be mediated by at least two mechanisms: (1) suppression of inflammatory cytokine and ET-1 expression and (2) PKG-mediated correction of an imbalance between SMAD2/3 and SMAD1/5/9 signaling in endothelial cells. We also found that CNP administration prevents the development of PH in mice in an exclusively endothelial GC-B-dependent manner and ameliorated PH in a Su/Hx rat PAH model, even after establishment of PH.
Nitric oxide (NO)/soluble guanylyl cyclase (sGC)/cyclic guanosine monophosphate (cGMP)-dependent signaling exerts important protective effects against the development of PAH. Indeed, phosphodiesterase 5 inhibitors and sGC activators are clinically used in the treatment of PAH patients. PKG1 in vascular smooth muscle cells is known to be a critical mediator of NO- and cGMP-induced vasodilation34. Indeed, deletion of Prkg1 gene, encoding PKG1, induces pulmonary hypertension along with increased vasoconstriction in mice35. Although PKG1 is also known to mediate CNP/GC-B signaling36, the roles played by endothelial CNP/GC-B/PKG1 signaling in the progression of PAH remained unclear. Our study demonstrates the role of endothelial CNP/GC-B/PKG1 signaling in the pathophysiology of PAH. This endothelial CNP/GC-B signaling provides an additional therapeutic axis alongside current therapies. This is consistent with the additive effects of CNP when combined with macitentan or sotatercept in this study.
The TGF-β family/SMAD pathway plays a critical role in the development of PAH26. During the development of PAH, the balance among the activities of the SMAD signaling pathways is altered downstream of activin and bone morphogenetic protein (BMP), two members of the TGF-β superfamily, with SMAD2/3 phosphorylation and activation being enhanced and SMAD1/5 phosphorylation and activation being suppressed37. An earlier study showed that NO and Rp-8-GMPS, a cGMP analog, inhibit TGF-β-dependent phosphorylation and nuclear translocation of SMAD2/3 via PKG1 in endothelial cells38. In addition, another group showed that BMP2-dependent phosphorylation of SMAD1/5/9 is suppressed by 8-pCPT-cGMP, a PKG inhibitor, in vascular smooth muscle cells39 and that BMP signaling via Smad1/5/9 and PKG1 is required to maintain the differentiated and low proliferative phenotype of pulmonary arterial smooth muscle cells (PASMCs)40. Most recently, it was reported that activation of cGMP/PKG signaling induced by natriuretic peptides suppresses phosphorylation of SMAD2/3 in cardiac fibroblasts41. However, the relationship between CNP and SMAD signaling pathways in endothelial cells remained undefined. In the present study, we demonstrated that CNP/GC-B/PKG signaling participates in maintaining the normal balance between SMAD2/3 and SMAD1/5/9 signaling in endothelial cells and that attenuation of endothelial CNP/GC-B signaling leads to an imbalance in SMAD signaling resulting in the progression of PAH. Notably, although expression of BMPR2 mRNA was significantly reduced in Hx-exposed HPAECs, CNP did not affect its expression levels. Similarly, pulmonary BMPR2 mRNA expression was significantly decreased in mice with PH, irrespective of the genotype, which suggests CNP/GC-B/PKG signaling enhances SMAD1/5/9 signaling and modulates the balance among SMAD signaling pathways downstream of the BMPRII receptor. This also suggests the therapeutic potential of CNP administration in the treatment of human PAH caused by heterozygous BMPR2 loss of function mutations.
Expression of Inhba gene, which encodes proteins involved in the production of the TGF-β superfamily member activin-A, is used as a proxy for assessing expression of activin-A27. Activin-A is a ligand of activin receptor types IIA and IIB (ACTRIIA and ACTRIIB) and activates the SMAD2/3 signaling pathway. Several recent studies reported that activin/ACTRII signaling is enhanced in human PAH patients and rodent PAH models, including MCT and SuHx-induced PH in rats and Hx-induced PH in mice29,42,43, and that plasma activin-A levels correlate with the disease severity and prognosis in patients with PAH43,44. This study clearly demonstrates that activation of endothelial CNP/GC-B signaling is sufficient and necessary to attenuate increases in pulmonary Inhba gene expression in response to pathological stimuli related to PAH. This effect may also be involved in endothelial CNP/GC-B signaling-induced down-regulation of SMAD2/3 activation.
Inflammation is now known to be an important factor contributing to the progression of PAH. Among the inflammatory factors involved, serum IL6 levels are elevated in PAH patients and correlate with their prognosis45. We found that endothelial CNP/GC-B signaling suppresses of pulmonary expression of IL6 mRNA induced by pathological stimuli related to PH both in vitro and in vivo. Previous reports demonstrated that in pulmonary smooth muscle cells from mice, BMPRII signaling suppresses Il6 gene expression in PH46. CNP/GC-B signaling may suppress Il6 gene expression by affecting BMPRII/SMAD1/5 signaling pathways. Earlier reports also revealed the inhibitory effect of CNP on expression of several inflammatory cytokines. For instance, protein expression CCL2 (also called monocyte chemoattractant protein-1, MCP-1) was significantly suppressed by CNP in lipopolysaccharide-exposed human glomerular mesangial cells47. CNP also inhibited secretion of CCL2 by cardiac fibroblasts48. We previously demonstrated that expression of Edn1 mRNA is significantly increased in PAECs isolated from CNP ecKO mice, and plasma ET-1 levels are significantly increased in CNP ecKO mice9. In addition, CNP suppresses angiotensin II- and arginine vasopressin-induced ET-1 production in cultured rat vascular smooth muscle cells49. ET-1 reportedly enhances expression of proinflammatory cytokines, including IL6, in addition to its potent vasoconstrictive action50. Those findings are consistent with our observations that CNP suppresses expression IL6, CCL2 and EDN1 mRNA in vivo and in vitro. Transcription of TGFB1 is reportedly regulated by AP1, SP1, NF-kB and STAT3, four transcriptional factors that can be activated by inflammatory cytokines. Expression of INHBA gene and Activin-A protein is also reportedly induced by inflammatory cytokines, including IL651,52,53. The inhibitory effect of CNP on expression of inflammatory cytokines may mediate CNP-induced suppression of TGFB1 and INHBA gene expression and, in turn, suppression of SMAD2/3 activity.
In addition to GC-B, CNP also binds to NPR-C, which is thought to primarily act as a clearance receptor involved in the degradation of CNP. However, several recent studies demonstrated that CNP exerts its effects by activating NPR-C in a cGMP-independent manner54,55,56,57. It was also reported that endothelial function is impaired and the antihypertensive effect of CNP is lost in NPR-C KO mice58. Previous studies have suggested that both GC-B–mediated and NPR-C–mediated actions of CNP may contribute to PH59, although the relative importance of each pathway appears to differ by disease context. In particular, recent reports have highlighted a potential role for NPR-C not only in experimental PAH models but also in PH secondary to left-heart disease60,61. In the present study, PH was exacerbated in CNP ecKO mice, as in GC-B ecKO mice, but not in GC-B smcKO mice. Moreover, the preventive effect of CNP against the progression of PH were nearly completely abolished in GC-B ecKO mice. We also confirmed that Hx-induced increases in EDN1, IL6, CCL2 and TGFB1 gene expression in HPAECs were significantly suppressed by CNP, and that effect was exclusively GC-B dependent. Indeed, knocking down NPR-C in HPAECs did not affect expression of EDN1, IL6 or CCl2 mRNA, whereas knocking down GC-B significantly increased expression of EDN1 and IL6 mRNA in those cells under normoxic conditions. The contribution of NPR-C to CNP-dependent signaling may differ among the various species used in experiments, depending upon the size and type of blood vessels/tissues studied and/or the different pathological conditions.
Notably, reducing endothelial CNP/GC-B expression alone was not sufficient to initiate PH. This suggests other pathological stimuli in addition to reduced endothelial CNP-GC-B signaling are required to initiate PH. It has also been demonstrated that CNP exerts anti-fibrotic effects in fibroblasts and anti-proliferative effects in lung pericytes through GC-B62,63. Although in the present study CNP administration prevented the development of PH in mice in an almost exclusively endothelial GC-B-dependent manner, there is a possibility that GC-B in pulmonary fibroblasts and/or pericytes activated during the development of PH may also play a role in pulmonary arterial remodeling. Further studies will be needed to address these issues. In summary, this study highlights the important role of endothelial CNP/GC-B signaling in the pathogenesis of PAH and suggests that modulation of this pathway may provide a potential therapeutic approach for PAH in humans.
Method
Animal experiments
The animal care and all animal experimental protocols were reviewed and approved by the Animal Research Committee at the Kyoto University Graduate School of Medicine (Approval number MedKyo22150) and conformed to the US National Institutes of Health Guide for the Care and Use of Laboratory Animals64. All efforts were made to minimize pain and distress. All mice used in this study were on a C57BL/6J background (Jackson Laboratory, Bar Harbor, ME, USA). Both male and female mice were examined in MCTp and Hx models, and no significant sex differences were observed in RVSP or RV/(LV + IVS) ratios. To maintain consistency with earlier work and minimize variability, only male mice were used in the main study.
Generation of CNP ecKO, GC-B ecKO, and GC-B smcKO mice
The Nppc-floxed and Npr2-floxed mouse lines used in this study were generated previously9. These lines were bred to homozygosity (Nppcflox/flox or Npr2flox/flox) in our facility. CNP ecKO mice were generated by crossing Nppcflox/flox mice with heterozygous Tie2-Cre transgenic (Tg) mice (Jackson Laboratory, Bar Harbor, ME, USA). GC-B ecKO mice were generated by crossing Npr2flox/flox mice with heterozygous Tie2-Cre Tg mice. GC-B smcKO mice were generated by crossing Npr2flox/flox mice with heterozygous SM22α-Cre knock-in mice also purchased from Jackson Laboratory. For all experiments, Tie2-Cre Tg or SM22α-Cre knock-in mice were used as controls for comparison with the corresponding conditional knockout mice. We confirmed that the phenotypes and quantitative RT-PCR results of floxed mice did not differ from those of Nppcflox/flox or Npr2flox/flox mice, supporting the validity of this control strategy.
Experimental PH animal models
For the monocrotaline pyrrole (MCTp)-induced PAH model, 5-mg aliquots of MCTp (Lot J0917, I2818, SC-214861A; Santa Cruz Biotechnology, Dallas, TX, USA) were dissolved in N,N-dimethylformamide (DMF) and stored at –80 °C until use14,65. Eight-week-old mice were anesthetized with 2.5% isoflurane (Viatris Pharmaceuticals, Inc., Tokyo, Japan), and MCTp (5 mg/kg, diluted with saline) was injected into the tail vein. Control mice received an equivalent volume of saline-adjusted DMF. Age-matched littermates were randomly assigned to receive MCTp or DMF. Two weeks after the injections, we assessed body weights and BPs and performed RV catheter studies to measure right ventricular pressure. Subsequently, organ weights were measured and samples collected for mRNA extraction.
For the hypoxia (Hx)-induced PH model, 8-week-old mice were placed in a hypobaric chamber (Kyodo International, Kanagawa, Japan) and maintained under hypoxia (10% O₂ and 90% N₂) at normobaric pressure for 4 weeks15,19. Age-matched littermates were randomly assigned to Hx or normoxia (21% O₂; control). The mice were then assessed as described above for the MCTp-induced PAH model.
To generate the SU5416 combined with hypoxia (SuHx) rat model, we used 8-week-old male Sprague-Dawley rats (SLC, Shizuoka, Japan) weighing around 200 g. They were injected subcutaneously with SU5416 (20 mg/kg) which was followed by exposure to hypoxia (10% O2) for 3 weeks. They were then returned to normoxia (21% O₂) for an additional 3 weeks17, during which CNP-53 was administered. After these exposures, we assessed body weight and BP and performed RV catheter studies to measure right ventricular pressure. Subsequently, organ weights were measured and samples collected.
For the SuHx mouse model, 8-week-old male mice were injected subcutaneously with SU5416 (20 mg/kg) and exposed to hypoxia (10% O₂) for 3 weeks, followed by normoxia (21% O₂) for 3 weeks16. CNP-53, macitentan and sotatercept were administered in combination with existing PAH therapies during the normoxia period.
Non-invasive BP measurement
We measured BP and heart rates (HR) after the MCTp injection or hypoxia exposure. BP and HR were measured noninvasively in conscious mice using a tail-cuff (MK-2000ST, No. 100726C0311; Muromachi Kikai Co., Ltd., Japan).
Echocardiographic measurement
We performed echocardiography using a Fujifilm VisualSonics Vevo®2100 (FUJIFILM VisualSonics Inc. Canada) in mice under isoflurane anesthesia (1.5–2.0% in oxygen) after MCTp injection or hypoxia exposure. CO was measured in mice from the hypoxia and SuHx models using transthoracic echocardiography and calculated using the left ventricular outflow tract (LVOT) method66. Briefly, stroke volume (SV) was calculated as 0.785 × (LVOT diameter)² × LVOT velocity time integral (VTI), and CO was obtained as (SV × HR from LVOT)/1000.
Surgical preparation and right heart catheterization
Animals were anesthetized with isoflurane (Viatris Pharmaceuticals, Inc., Tokyo, Japan) after which the trachea was cannulated with a polyethylene tube, and the lungs were ventilated with a mouse ventilator. The inspired gas was indoor air, and the ventilator settings were adjusted to a tidal volume of 10 μL/g and a frequency of ~180/min. After skin incision, a diaphragm incision was made to open the thorax, and the right ribs were cut near the sternum to expose the heart. We measured RVSP with a Millar Mikro-Tip catheter (Millar Inc., Houston, Texas, USA) inserted through the heart’s apex. RV pressure tracings were obtained for more that 20 s. HR was derived from the peak RV systolic period and was usually 300–700 beats/min (bpm) under these conditions. When the HR fell below 300 bpm, the measurements were excluded. We acknowledge that open-chest catheterization may yield slightly lower RVSP values than closed-chest measurements67. Nevertheless, this approach allows detection of relative differences between experimental groups in rodent models of PH.
Morphometric analysis
After hemodynamic analysis, the mice were killed with an overdose of anesthesia, and the hearts and lungs were excised. After removing the atria, we separated the right ventricular wall from the left ventricle and septum and weighed it. We used Fulton’s index, which is the ratio of the weight of the right ventricle (RV) to that of the left ventricle (LV) plus the interventricular septum (IVS) [RV/(LV + IVS)], as an indicator of right heart hypertrophy. After weighing the excised lungs, the left lung was frozen in liquid nitrogen for subsequent mRNA analysis. The right lung was inflated with 10% formalin neutral buffer solution for morphometric analysis. After this procedure, samples were taken to the Center for Anatomical, Pathological and Forensic Medical Research, Graduated School of Medicine, Kyoto University, for histological processing. For pulmonary vascular morphometry, lung sections were cut from paraffin-embedded blocks and stained with hematoxylin-eosin plus Victoria blue. Images were then captured with a fluorescence microscope equipped with a camera (Zeiss; Carl Zeiss AG Corp). Morphometric studies were performed with lung sections from five or six mice randomly selected from each treatment group. Pulmonary remodeling was assessed based on the percent wall thickness of pulmonary arteries classified as small arteries and arterioles (<100 μm). Percent medial wall thickness was defined as the following formula: 100 × (external diameter – internal diameter)/external diameter determined with hematoxylin-eosin and Victoria blue-stained sections. We analyzed the medial wall thickness only to show a nearly circular profile in cross-section. We calculated the percent medial wall thickness in at least five small pulmonary arteries and arterioles from each mouse.
Blinding
All surgical procedures, histological analyses, and echocardiography were performed by individuals who were blinded to the genotype and treatment group through use of name and allocation concealment.
Immunohistochemistry (IHC)
Incubation and washing procedures were carried out at room temperature if they were not specified. After deparaffinization and antigen retrieval, endogenous peroxidase activity was blocked by 0.3% H2O2 in methyl alcohol for 30 min. The glass slides were washed in PBS (6 times, 5 each min) and mounted with 1% normal serum in PBS for 30 min. Sections were incubated overnight at 4 °C with primary antibodies against TAGLN/SM22α (Thermo Fisher, PA5-29767), NPR2 (Santa Cruz Biotechnology, SC-537749) or Ki67 (Abcam, ab15580), followed by biotinylated secondary antibodies and avidin–biotin–peroxidase complex (ABC-Elite, Vector Laboratories). Signals were visualized with DAB and counterstained with hematoxylin. Apoptotic cells were detected by TUNEL staining using a commercial apoptosis detection kit (MK500, Takara Bio Inc., Japan) according to the manufacturer’s instructions.
Reagents
Human CNP-53, a long-acting NPR-B activator that is refractory to NEP and has limited interaction with NPR-C, was purchased from Peptide Institute (No. 4241-s, Ibaraki, Osaka, Japan). For in vitro studies, CNP-53 was added to the culture medium to a final concentration of 1.0 μmol/L31. For in vivo studies, osmotic mini-pumps (Alzet model 2002, 2004, or 2ML4) were implanted intraperitoneally to deliver CNP-53 at 0.2 mg/kg/day31. In mice, CNP-53 was administered for 2 weeks in the MCTp model, 3 weeks in the SuHx models, and 4 weeks in the hypoxia model. In SuHx mouse and rat models, mini-pumps were implanted upon return to normoxia; in the MCTp and hypoxia models, pumps were implanted 24 h before disease induction.
Macitentan was purchased from Cayman Chemical (Item No. 23304, Ann Arbor, MI, USA) and administered by oral gavage at 20 mg/kg/day32. Sotatercept (mIgG2a) was purchased from MedChemExpress (Cat. # HY-P99590A, Monmouth Junction, NJ, USA) and administered at a dose of 10 mg/kg by subcutaneous injection twice weekly33. The PKG inhibitor Rp-8-Br-cGMP (Sigma-Aldrich, St. Louis, MO, USA) was added to the culture medium to a final concentration of 30 μg/mL68.
Urinary cGMP ELISA assay
To assess in vivo activation of GC-B signaling, urinary cyclic GMP (cGMP) levels were measured using a commercially available ELISA kit (Cyclic GMP ELISA Kit, Cayman Chemical, Ann Arbor, MI, USA, Cat# 581021) according to the manufacturer’s instructions. Absorbance was measured at 405 nm using a microplate reader (ARVO X3, PerkinElmer Inc., Massachusetts, USA), following the manufacturer’s instructions.
Human pulmonary arterial endothelial cell cultures
Human pulmonary arterial endothelial cells (HPAECs; CC-2530, Lonza, Walkersville, MD, USA; donor sex not specified) were maintained in EGM™-2 BulletKit™ medium (CC-3162, Lonza) according to the manufacturer’s instructions. Cells were plated at 5.0 × 10³ cells/cm² in 24-well plates and incubated for 24 h before treatment. For hypoxia experiments, cultures were exposed to 1% O₂ (balance N₂) with 5% CO₂ at 37 °C for 24 h, with or without CNP-53 (1.0 μmol/L) added at the onset of hypoxia. For GC-B knockdown, cells were transfected for 24 h with 100 nmol/L siRNAs targeting NPR2 (SASI_Hs01_00217023, Sigma-Aldrich) or NPR-C (s9690, 4390824, Thermo Fisher) or with non-targeting control siRNA (AM4635, Thermo Fisher) in Opti-MEM Medium (Opti-MEMTMⅠ Reduced Serum Medium, catalog No. 31985062, Gibco, United Kingdom), followed by the exposure to hypoxia for an additional 24 h. The cells were then harvested for RT-PCR.
Quantitative RT-PCR analysis
Total RNA was extracted from lungs, cultured HPAECs or human PASMCs using TRIzol reagent (Invitrogen) according to the manufacturer’s protocol. Quantitative reverse transcription PCR (qRT-PCR) was performed using a THUNDERBIRD Probe One-step qRT-PCR kit (TOYOBO Life Science) and a StepOnePlus™ Real-Time PCR System (Thermo Fisher Scientific, Massachusetts, USA). TaqMan Gene Expression Assays (Applied Biosystems) were used for target and reference genes, and relative mRNA levels were normalized to GAPDH. Primer sequences and probe IDs are listed in Supplementary Table 1.
Western blotting
Endothelial cells were lysed by sonication in lysis buffer (Cell Signaling Technology) and centrifuged to remove debris. Protein concentrations in the supernatant were measured, and samples were separated by 10% SDS-PAGE, transferred to PVDF membranes (Immobilon), and blocked with 5% BSA in TBS-T. The membranes were incubated first with primary antibodies overnight at 4 °C in Can Get Signal solution 1 (Nacalai Tesque) and then with appropriate secondary antibodies diluted 1:2000 in Can Get Signal solution 2 for 30 min at room temperature. The bands were detected using a Kaleidoscope ladder enhanced chemiluminescence kit (BIO-RAD, CA, USA). Band intensity was quantified using ImageJ. Detailed information on primary and secondary antibodies is provided in Supplementary Table 2.
To evaluate the role of endogenous PKG activity, HPAECs were treated with the PKG inhibitor Rp-8-Br-cGMP (30 μg/mL) alone or in combination with CNP-53 under hypoxic conditions.
Smooth muscle proliferation and cytotoxic stress assay
Control PASMCs were obtained from a healthy region of a lung resected from a non-PAH female patient and provided by the Okayama University Graduate School of Medicine. The patient underwent lung resection for localized lung cancer, and written informed consent for the use of resected tissue for research purposes was obtained prior to the procedure. All procedures were approved by the Ethics Committee of Okayama University Graduate School of Medicine, Dentistry, and Pharmaceutical Sciences (Approval number 1511-017) and the Kyoto University Medical Ethics Committee (Approval number R3091) and conformed to the Declaration of Helsinki.
To evaluate the effects of endothelial GC-B deficiency on smooth muscle cell responses, HPAECs were transfected with control or GC-B siRNA (100 nmol/L) and incubated under normoxic or hypoxic (1% O₂) conditions for 24 h. Culture supernatants were collected and transferred to PASMCs, which were seeded into 96-well plates and maintained in Dulbecco’s modified Eagle medium (DMEM; Thermo Fisher Scientific, Waltham, MA) supplemented with 10% fetal bovine serum (FBS; Sigma-Aldrich) and 1% penicillin-streptomycin (FUJIFILM Wako Pure Chemical Corporation, Osaka, Japan). Cell proliferation and cytotoxic stress were assessed using the Cell Counting Kit-8 (CCK-8; Dojindo Molecular Technologies Inc., Kumamoto, Japan) according to the manufacturer’s instructions. Absorbance was measured at 450 nm for CCK-8 using an ARVO X3 microplate reader (PerkinElmer Inc., Massachusetts, USA).
Boyden chamber migration assay
To assess the effect of endothelial GC-B signaling on smooth muscle cell migration, transwell migration assays were performed using a 24-well migration plate with a pore size of 8.0 µm (Cell Biolabs, Inc., San Diego, CA, USA), following the manufacturer’s instructions. HPAECs were transfected with control or GC-B siRNA (100 nmol/L) and cultured under hypoxic (1% O₂) conditions for 24 h. PASMCs were seeded into the upper chambers in serum-free medium, while HPAECs were cultured in the lower wells69. After incubation, non-migrated cells were removed, and migrated cells were lysed and quantified fluorometrically using a microplate reader.
Human scRNA-seq data analysis from public datasets
Publicly available single-cell RNA sequencing (scRNA-seq) data from human lung tissues (GSE16947130) were obtained from the Gene Expression Omnibus (GEO). We selected three IPAH samples (SC17, SC161, SC207) and three control samples (SC23F [SC155 and SC156 combined], SC45, SC59), all generated using the 10x Genomics platform. Data preprocessing and quality control were performed as described in the original publication. To identify vascular endothelial cells, we examined the expression of canonical marker genes (PECAM1, VWF, CDH5, CLDN5, EFNB2, NR2F2, PROX1, CA4)70. Endothelial clusters (EC1-3) were extracted and reanalyzed through UMAP embedding and reclustering. Expression levels of NPPC and NPR2 were compared between IPAH and control endothelial cells, and statistical differences were assessed using the Wilcoxon rank-sum test.
Statistical analysis
Because this was an exploratory study, and there had been no prior study that could be used as a reference, we calculated the sample size based on similar PH experiments reported in the literature before the start of this study. All data and bar graphs are presented as the mean ± standard error of the mean (SEM). Multiple comparisons were evaluated using repeated measures one-way ANOVA with Tukey’s multiple comparison test. Values of p < 0.05 were considered significant. Exact P values are reported whenever available, and values shown as “< 0.0001” reflect the lower reporting limit of the statistical software. All statistical analyses were performed using Prism9 (GraphPad Prism version9; GraphPad).
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The authors declare that all data supporting the findings of this study are available within the paper and its supplementary information files. All scRNA-seq datasets analyzed in this study are publicly available from the Gene Expression Omnibus under accession code GSE16947130. Source data are provided with this paper.
Code availability
No custom code was used. Analyses were conducted using standard R packages as detailed in the “Methods” section.
References
Humbert, M., Sitbon, O. & Simonneau, G. Treatment of pulmonary arterial hypertension. N. Engl. J. Med. 351, 1425–1436 (2004).
Pickles, H. & O’Grady, J. Side effects occurring during administration of epoprostenol (prostacyclin, PGI2), in man. Br. J. Clin. Pharmacol. 14, 177–185 (1982).
Humbert, M. et al. Survival in incident and prevalent cohorts of patients with pulmonary arterial hypertension. Eur. Respir. J. 36, 549–555 (2010).
Benza, R. L. et al. An evaluation of long-term survival from time of diagnosis in pulmonary arterial hypertension from the reveal registry. Chest 142, 448–456 (2012).
Chin, K. M. et al. Treatment algorithm for pulmonary arterial hypertension. Eur. Respir. J. 64, 2401325 (2024).
Nakagawa, Y., Nishikimi, T. & Kuwahara, K. Atrial and brain natriuretic peptides: hormones secreted from the heart. Peptides 111, 18–25 (2019).
Matsukawa, N. et al. The natriuretic peptide clearance receptor locally modulates the physiological effects of the natriuretic peptide system. Proc. Natl. Acad. Sci. 96, 7403–7408 (1999).
Vaandrager, A. B. et al. Differential role of cyclic GMP-dependent protein kinase II in ion transport in murine small intestine and colon. Gastroenterology 118, 108–114 (2000).
Nakao, K. et al. Endothelium-derived C-type natriuretic peptide contributes to blood pressure regulation by maintaining endothelial integrity. Hypertension 69, 286–296 (2017).
Špiranec, K. et al. Endothelial C-type natriuretic peptide acts on pericytes to regulate microcirculatory flow and blood pressure. Circulation 138, 494–508 (2018).
Moyes, A. J. et al. Endothelial C-type natriuretic peptide maintains vascular homeostasis. J. Clin. Investig. 124, 4039–4051 (2014).
Itoh, T. et al. C-type natriuretic peptide ameliorates monocrotaline-induced pulmonary hypertension in rats. Am. J. Respir. Crit. Care Med. 170, 1204–1211 (2004).
Casserly, B. et al. C-type natriuretic peptide does not attenuate the development of pulmonary hypertension caused by hypoxia and VEGF receptor blockade. Life Sci. 89, 460–466 (2011).
Hiraishi, K. et al. Substantial involvement of TRPM7 inhibition in the therapeutic effect of Ophiocordyceps sinensis on pulmonary hypertension. Transl. Res. 233, 127–143 (2021).
Rhodes, J. Comparative physiology of hypoxic pulmonary hypertension: historical clues from brisket disease. J. Appl. Physiol. 98, 1092–1100 (2005).
Vitali, S. H. et al. The Sugen 5416/hypoxia mouse model of pulmonary hypertension revisited: Long-term follow-up. Pulm. Circ. 4, 619–629 (2014).
Abe, K. et al. Formation of plexiform lesions in experimental severe pulmonary arterial hypertension. Circulation 121, 2747–2754 (2010).
Chusho, H. et al. Dwarfism and early death in mice lacking C-type natriuretic peptide. Proc Natl. Acad. Sci. USA 98, 4016–4021 (2001).
Hoshikawa, Y. et al. Hypoxia induces different genes in the lungs of rats compared with mice Hyp-oxia induces different genes in the lungs of rats compared with mice. Physiol. Genom. 12, 209–219 (2003).
Tamura, N. et al. Critical roles of the guanylyl cyclase B receptor in endochondral ossification and development of female reproductive organs. PNAS. 101, 17300–17305 (2004).
Alturaif, N., Attanasio, U. & Mercurio, V. Pulmonary arterial hypertension: sex-specific differences and outcomes. Ther. Adv. Respir. Dis. 19, 17534666251350493 (2025).
Giaid, A. del et al. Expression of endothelin-1 in the lungs of patients with pulmonary hypertension. N. Engl. J. Med. 328, 1732–1739 (1993).
Minamino, N., Kangawa, K. & Matsuo, H. N-terminally extended form of C-type natriuretic peftide (CNP-531 identified in porcine brain. Biochem. Biophys. Res. Commun. 170, 973–979 (1990).
Chen, Y. et al. C53: A novel particulate guanylyl cyclase B receptor activator that has sustained activity in vivo with anti-fibrotic actions in human cardiac and renal fibroblasts. J. Mol. Cell. Cardiol. 130, 140–150 (2019).
Wendt, D. J. et al. Neutral endopeptidase-resistant C-Type natriuretic peptide variant represents a new therapeutic approach for treatment of fibroblast growth factor receptor 3-related dwarfism. J. Pharmacol. Exp. Ther. 353, 132–149 (2015).
Humbert, M. et al. Sotatercept for the treatment of pulmonary arterial hypertension. N. Engl. J. Med. 384, 1204–1215 (2021).
Ryanto, G. R. T. et al. An endothelial activin A-bone morphogenetic protein receptor type 2 link is overdriven in pulmonary hypertension. Nat. Commun. 12, 1720 (2021).
Yang, J., Li, X. & Morrell, N. W. Id proteins in the vasculature: From molecular biology to cardiopulmonary medicine. Cardiovasc. Res. 104, 388–398 (2014).
Yndestad, A. et al. Elevated levels of activin A in clinical and experimental pulmonary hypertension. J. Appl Physiol. 106, 1356–1364 (2009).
Saygin, D. et al. Transcriptional profiling of lung cell populations in idiopathic pulmonary arterial hypertension. Pulm. Circ. 10, 1–15 (2020).
Bubb, K. J. et al. Endothelial C-type natriuretic peptide is a critical regulator of angiogenesis and vascular remodeling. Circulation 139, 1612–1628 (2019).
Iglarz, M. et al. Pharmacology of macitentan, an orally active tissue-targeting dual endothelin receptor antagonist. J. Pharmacol. Exp. Ther. 327, 736–745 (2008).
Joshi, S. R. et al. Sotatercept analog suppresses inflammation to reverse experimental pulmonary arterial hypertension. Sci. Rep. 12, 7803 (2022).
Francis, S. H., Busch, J. L. & Corbin, J. D. cGMP-dependent protein kinases and cGMP phosphodiesterases in nitric oxide and cGMP action. Pharmacol. Rev. 62, 525–563 (2010).
Zhao, Y. D. et al. Protein kinase G-I deficiency induces pulmonary hypertension through Rho A/Rho kinase activation. Am. J. Pathol. 180, 2268–2275 (2012).
Nakagawa, Y. & Nishikimi, T. CNP, the third natriuretic peptide: its biology and significance to the cardiovascular system. Biology 11, 986 (2022).
Guignabert, C. & Humbert, M. Targeting transforming growth factor-β receptors in pulmonary hypertension. Eur. Respir. J. 57, 2002341 (2021).
Saura, M. et al. Nitric oxide regulates transforming growth factor-β signaling in endothelial cells. Circ. Res. 97, 1115–1123 (2005).
Schwappacher, R. et al. A molecular mechanism for therapeutic effects of cGMP-elevating agents in pulmonary arterial hypertension. J. Biol. Chem. 288, 16557–16566 (2013).
Schwappacher, R. et al. Novel crosstalk to BMP signalling: CGMP-dependent kinase I modulates BMP receptor and Smad activity. EMBO J. 28, 1537–1550 (2009).
Meng, L. et al. NPRC deletion attenuates cardiac fibrosis in diabetic mice by activating PKA/PKG and inhibiting TGF-β1/Smad pathways. Sci. Adv. 9, eadd4222 (2023).
Yung, L. M. et al. ACTRIIA-Fc rebalances activin/GDF versus BMP signaling in pulmonary hypertension. Sci. Transl. Med. 12, eaaz5660 (2020).
Guignabert, C. et al. Serum and pulmonary expression profiles of the activin signaling system in pulmonary arterial hypertension. Circulation 147, 1809–1822 (2023).
Hadwiono, M. R. et al. Increased serum activin A level in congenital heart disease-associated pulmonary artery hypertension: a comparative study from the COHARD-PH registry. Pulm. Circ. 13, e12280 (2023).
Humbert, M. et al. Increased interleukin-1 and interleukin-6 serum concentrations in severe primary pulmonary hypertension. Am. J. Respir. Crit. Care Med. 151, 1628–1631 (1995).
Hagen, M. et al. Interaction of interleukin-6 and the BMP pathway in pulmonary smooth muscle. Am. J. Physiol. Lung Cell Mol. Physiol. 292, 1473–1479 (2007).
Osawa, H. et al. C-type natriuretic peptide inhibits proliferation and monocyte chemoattractant protein-1 secretion in cultured human mesangial cells. Nephron. 86, 467–472 (2000).
Li, Z. Q. et al. Inhibitory effects of C.type natriuretic peptide on the differentiation of cardiac fibroblasts, and secretion of monocyte chemoattractant protein-1 and plasminogen activator inhibitor-1. Mol. Med. Rep. 11, 159–165 (2015).
Hanehira, T., Kohno, M. & Yoshikawa, J. Endothelin production in cultured vascular smooth muscle cells—Modulation by the atrial, brain, and C-type natriuretic peptide system. Metabolism 46, 487–493 (1997).
Kowalczyk, A., Kleniewska, P., Kolodziejczyk, M., Skibska, B. & Goraca, A. The role of endothelin-1 and endothelin receptor antagonists in inflammatory response and sepsis. Arch. Immunol. Ther. Exp. 63, 41–52 (2015).
Arai, K. Y. et al. IL-1beta stimulates activin betaA mRNA expression in human skin fibroblasts through the MAPK pathways, the nuclear factor-kappaB pathway, and prostaglandin E2. Endocrinology 152, 3779–3790 (2011).
Hübner, G. & Werner, S. Serum growth factors and proinflammatory cytokines are potent inducers of activin expression in cultured fibroblasts and keratinocytes. Exp. Cell Res. 228, 106–113 (1996).
Wu, B. et al. The TGF-β superfamily cytokine Activin-A is induced during autoimmune neuroinflammation and drives pathogenic Th17 cell differentiation. Immunity 54, 308–323.e6 (2021).
Murthy, K. S. et al. G(i-1)/G(i-2)-dependent signaling by single-transmembrane natriuretic peptide clearance receptor. Am. J. Physiol. Gastrointest. Liver Physiol. 278, G974–G980 (2000).
Moyes, A. J. & Hobbs, A. J. C-type natriuretic peptide: A multifaceted paracrine regulator in the heart and vasculature. Int. J. Mol. Sci. 20, 2281 (2019).
Ahluwalia, A. & Hobbs, A. J. Endothelium-derived C-type natriuretic peptide: more than just a hyperpolarizing factor. Trends Pharm. Sci. 26, 162–167 (2005).
Scotland, R. S. et al. C-type natriuretic peptide inhibits leukocyte recruitment and platelet-leukocyte interactions via suppression of P-selectin expression. Proc. Natl. Acad. Sci. 102, 14452–14457 (2005).
Li, N. et al. C-type natriuretic peptide (CNP) could improve sperm motility and reproductive function of asthenozoospermia. Int. J. Mol. Sci. 23, 10370 (2022).
Dignam, J. P. et al. Endothelium-derived C-type natriuretic peptide offsets the pathogenesis of pulmonary hypertension. Pharm. Res 219, 107870 (2025).
Egom, E. E., Maher, V. & El Hiani, Y. Evolving use of natriuretic peptide receptor type-C as part of strategies for the treatment of pulmonary hypertension due to left ventricle heart failure. Int. J. Cardiol. 281, 172–178 (2019).
Egom, E. E.-A. et al. A natriuretic peptides clearance receptor’s agonist reduces pulmonary artery pressures and enhances cardiac performance in preclinical models: New hope for patients with pulmonary hypertension due to left ventricular heart failure. Biomed. Pharmacother. 93, 1144–1150 (2017).
Werner, F. et al. Ablation of C-type natriuretic peptide/cGMP signaling in fibroblasts exacerbates adverse cardiac remodeling in mice. JCI Insight. 8, e160416 (2023).
Dabral, S. et al. C-type natriuretic peptide/cGMP/FoxO3 signaling attenuates hyperproliferation of pericytes from patients with pulmonary arterial hypertension. Commun. Biol. 7, 693 (2024).
National Research Council. The Guide for the Care and Use of Laboratory Animals. 8th Ed. (National Academies Press, Washington, DC, 2011).
Dumitrascu, R. et al. Characterization of a murine model of monocrotaline pyrrole-induced acute lung injury. BMC Pulm. Med. 8, 15 (2008).
Todd, E. A., Williams, M., Kamiar, A., Rasmussen, M. A. & Shehadeh, L. A. Echocardiography protocol: a tool for infrequently used parameters in mice. Front. Cardiovasc. Med. 9, 1038385 (2022).
Provencher, S. et al. Standards and methodological rigor in pulmonary arterial hypertension preclinical and translational research. Circ. Res. 122, 1021–1032 (2018).
Xu, F. et al. Icariside II, a PDE5 inhibitor, suppresses oxygen-glucose deprivation/reperfusion-induced primary hippocampal neuronal death through activating the PKG/CREB/BDNF/TrkB signaling pathway. Front. Pharmacol. 11, 523 (2020).
Ramadhiani, R. et al. Endothelial cell senescence exacerbates pulmonary hypertension by inducing juxtacrine Notch signaling in smooth muscle cells. iScience 26, 106662 (2023).
Vanlandewijck et al. A molecular atlas of cell types and zonation in the brain vasculature. Nature 554, 475–480 (2018).
Acknowledgements
We thank Ayako Ohta, Yukari Deguchi-Kubo, and Mebae Kobayashi for their excellent secretarial work, and Mizuho Takemura and Mari Sakamoto of Kyoto University for their excellent technical support. This research was supported by AMED under Grant Numbers JP21ek0109509h0001, JP22ek0109509h0001 and JP23ek0109509h0001_(K. Kuwahara, Y. Nakagawa, H. Kinoshita); Grants-in-Aid for Scientific Research from the Japan Society for the Promotion of Science to H. Inazumi (JP19K23963 and JP20K17077), K. Kuwahara (JP20H03673), Y. Nakagawa (JP22K08099), H. Kinoshita (JP20K08180) and T. Nishikimi (JP21K08126); and by grants from the Takeda Science Foundation and Kondou Kinen Medical Foundation (to Y. Nakagawa).
Author information
Authors and Affiliations
Contributions
K.K. and Y.N. conceived and designed the entire study. H.Y., H.K., H.I., T.K., T.N., M.O., K. Nakao (Kazuhiro), Y.U., A.Y., K. Nakao (Kazuwa), T.K., and K.O. was involved in experimental design. H.Y. performed experiments of generation and analysis of mice and rats PAH models in most of outlined studies. H.Y., T.K., H.K., H.I., K.M., D. Nakamura (Daisuke), K.S., K.Y., S.T., D. Nakajima (Daisuke), and I.S., assisted experiments of in vivo PAH models and in vitro studies. H.Y., K.K. and Y.N. prepared the manuscript with input from all authors. All authors reviewed and approved the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Sebastien Bonnet, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yanagisawa, H., Kuwahara, K., Nakagawa, Y. et al. Endothelial C-type natriuretic peptide/guanylyl cyclase-B signaling prevents pulmonary arterial hypertension. Nat Commun 17, 2331 (2026). https://doi.org/10.1038/s41467-026-70139-2
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-026-70139-2