Abstract
Identifying biomarkers that precisely track the neurodegenerative component of Alzheimer’s disease (AD) is essential for effective clinical management. Here we show that cerebrospinal fluid (CSF) levels of the synaptic proteins NPTX1 and NPTXR are robust indicators of disease severity and future clinical progression. In two independent, multi-ethnic cohorts spanning the AD continuum (n = 635), lower CSF NPTX levels correlate strongly with cognitive impairment and cortical thinning in AD-vulnerable regions. Longitudinally, baseline NPTX levels predict accelerated brain atrophy and the clinical transition from mild cognitive impairment to dementia, frequently outperforming or complementing established markers such as pTau181 and neurofilament light chain. These findings establish NPTX1 and NPTXR as sensitive, stage-specific markers of synaptic integrity and neurodegeneration. By accurately forecasting disease progression, these biomarkers offer significant potential to enhance patient stratification and provide a crucial tool for monitoring the efficacy of disease-modifying therapies in clinical trials.
Similar content being viewed by others
Introduction
Alzheimer’s disease (AD) is a progressive neurodegenerative disorder pathologically defined by extracellular amyloid-β (Aβ) plaques and intracellular neurofibrillary tangles composed of hyperphosphorylated tau protein1. These pathologies initiate cascades involving synaptic dysfunction, neuronal death, and brain atrophy, culminating in progressive cognitive decline and dementia. The advent of validated fluid biomarkers such as CSF Aβ42/40 ratio2 and the CSF pTtau/Aβ42 ratio3, including FDA-approved versions on fully automated instruments, and more recently blood-based phosphorylated tau isoforms (e.g., pTau181, pTau217)4,5,6, has significantly advanced AD diagnostics by enabling in vivo detection of core pathology years before symptom onset. This progress underpins the AT(N) framework, which classifies individuals biologically based on Aβ pathology (A), tau pathology (T), and neurodegeneration or neuronal injury (N)7,8. Accessible fluid biomarkers—primarily in CSF but increasingly measurable in plasma (e.g., Aβ42/40 ratio, tau variants)—now reliably determine ‘A’ (amyloid) and ‘T’ (tau) status, enabling a biological definition of AD8.
However, robustly tracking the “N” component, particularly the synaptic damage closely linked to cognitive symptoms, remains challenging using fluid biomarkers. Established “N” markers such as structural MRI (atrophy) and FDG-PET (hypometabolism)7, offer valuable staging information but face limitations in accessibility, cost, and molecular specificity. While neurofilament light chain (NFL) is an accessible fluid marker of axonal injury, its utility for tracking AD-related neurodegeneration is hampered by substantial inter-individual variability9 and sensitivity to concomitant pathologies like cerebrovascular disease and major age-related changes10. Consequently, there is a critical need for accessible fluid biomarkers that sensitively reflect synaptic degeneration and its functional consequences—cognitive impairment—across the AD continuum. Validating such biomarkers across diverse populations is essential for advancing precision medicine, enabling accurate disease staging, identifying optimal therapeutic windows and improving prognostic accuracy.
Synapses, the fundamental units of neuronal communication, are essential for learning, memory formation, and cognitive function11. Synaptic loss is an early pathological hallmark of AD, correlating more strongly with cognitive decline severity than plaques or tangles alone12,13. Biomarkers reflecting synaptic integrity are therefore promising candidates for staging neurodegeneration and cognitive impairment. Neuronal pentraxins (NPTXs)—including neuronal pentraxin 1 (NPTX1), neuronal pentraxin 2 (NPTX2), and neuronal pentraxin receptor (NPTXR)—are secreted synaptic proteins crucial for synapse plasticity14, circuit maturation15, and the modulation of microglia-mediated synapse elimination16. While reduced CSF NPTX2 levels have been consistently reported in AD17,18,19,20 and other neurodegenerative disorders21,22, data on NPTX1 and NPTXR in AD remain sparse and inconsistent. Prior studies are limited, reporting decreases only late in the disease23,24, finding no change18, or lacking systematic evaluation across the biologically defined AD continuum (from preclinical to dementia stages) and longitudinal assessment of predictive capacity.
Therefore, we hypothesized that CSF NPTX1 and NPTXR levels serve as sensitive indicators of synaptic degeneration that track neurodegeneration severity and cognitive decline across the AD continuum and predict future clinical progression. To rigorously test this, we leveraged two independent, ethnogeographically distinct cohorts—the China Aging and Neurodegenerative Disorder Initiative (CANDI, n = 435) and the Norwegian Dementia Disease Initiation (DDI, n = 200)—encompassing the full AD spectrum (cognitively unimpaired [CU], mild cognitive impairment [MCI], AD dementia) and individuals with non-AD dementia (non-ADD). We specifically aimed to: (1) Characterize CSF NPTX1 and NPTXR level trajectories across cognitive impairment stages; (2) Determine their cross-sectional associations with cognitive performance and structural MRI measures, comparing them to established AD biomarkers (Aβ42/40, pTau181, tTau, NFL); and (3) Assess the prognostic value of baseline NPTX1 and NPTXR levels for longitudinal brain atrophy, cognitive decline and MCI-to-Dementia conversion. This study provides comprehensive, validated evidence positioning CSF NPTX1 and NPTXR as sensitive and specific fluid biomarkers for staging cognitive impairment in AD and predicting its clinical course.
Results
Participant demographics and baseline characteristics
The study included 635 participants from two independent cohorts: the CANDI (n = 435, mean age 62.1 ± 8.5 years; 59.5% female) and the DDI (n = 200, mean age 65.9 ± 8.5 years; 51.5% female).
The CANDI cohort encompassed the full clinical spectrum and was stratified into six clinical-pathological groups based on clinical diagnosis and amyloid status (A + /A−), including non-AD dementia (Dementia A-).
The DDI validation cohort, which had a higher prevalence of APOE-ε4 carriers (57.0%, n = 114 vs. 41.4% in CANDI), comprised participants who were cognitively unimpaired (CU) or had mild cognitive impairment (MCI). It was stratified into four diagnostic groups: CU A- (n = 40), CU A+ (n = 43), MCI A- (n = 41), and MCI A+ (n = 76).
Comprehensive baseline characteristics and biomarker profiles for both cohorts are detailed in Table 1 and Supplementary Table 1.
CSF NPTXs levels across the AD continuum
In Aβ-negative (A−) individuals, NPTXR levels demonstrated a biphasic trajectory, showing significant elevation at the MCI stage (p = 0.038) relative to CU participants, followed by marked reduction in dementia-stage patients (p = 0.005) in the CANDI cohort (Fig. 1). NPTX1 levels mirrored this late-stage decline, being significantly lower in Dementia compared to both CU (p = 0.001) and MCI (p = 0.002) groups. Notably, while tTau and NFL exhibited progressive elevation with disease severity (Dementia vs CU: p = 0.033 and p = 0.034; vs MCI: p = 0.045 and p = 0.012), pTau181 levels remained stable across cognitive stages (Supplementary Fig. 1).
The box plots depict the median (horizontal bar), interquartile range (IQR, hinges), and the whiskers indicate the minimum and maximum values. CSF NPTXs levels were measured in the main (CANDI, n = 435) cohort (A) and validated in the replication cohort (DDI, n = 200) (B). P-values (two-sided) were assessed by a one-way analysis of covariance (ANCOVA) or t-Test, adjusting for age, sex, educational level, and apolipoprotein E (APOE)-ε4.
The Aβ-positive (A + ) population revealed distinct biomarker patterns. Both NPTXR and NPTX1 displayed monophasic decreases with cognitive decline in the CANDI cohort (Dementia vs CU: p = 0.007 and p = 0.002; vs MCI: p = 0.006 and p < 0.001). This progression was replicated in the independent DDI cohort (MCI vs CU: p = 0.024 and p = 0.027 for NPTXR and NPTX1, respectively) (Fig. 1). Concomitantly, pTau181 levels increased significantly with cognitive impairment (MCI vs CU: p = 0.008; Dementia vs CU: p = 0.002), while tTau elevations were specifically observed in dementia-stage participants (Dementia vs CU: p = 0.003). Notably, Aβ42/Aβ40 ratios and NFL levels remained stable throughout the A+ continuum (Supplementary Fig. 1).
Age-associated biomarker changes emerged specifically in A+ individuals: NPTX1 (β = 0.157, p = 0.035) and NFL (β = 0.105, p = 0.033) showed positive correlations with age, while pTau181 demonstrated an inverse relationship (β = −0.101, p = 0.044). In contrast, A- individuals only exhibited a modest positive association between pTau181 and age (β = 0.182, p = 0.020) (Supplementary Fig. 2). Notably, NPTX1 and NPTXR levels showed divergent correlations with core AD biomarkers: positive associations with pTau181 across both A- and A+ groups, but negative correlations with Aβ42/Aβ40 ratios exclusively in A+ individuals (Supplementary Fig. 3).
CSF NPTXs link to cortical thickness in AD regions
Whole-brain vertex-wise analysis revealed divergent spatial patterns of NPTX1 and NPTXR associations with cortical architecture (Fig. 2). In A+ individuals, NPTXR showed widespread positive correlations spanning frontal/temporal cortices and insula (FDR-corrected p < 0.05), contrasting sharply with the restricted insular positivity and cuneal negativity observed in A- counterparts. NPTX1 demonstrated selective preservation effects in the insula and dorsolateral prefrontal cortex (middle superior frontal gyrus). Notably, classical neurodegeneration markers showed inverse spatial patterning: tTau correlated with temporoparietal thinning in A- individuals, while serum NFL predicted insula and inferior temporal atrophy in A+ subjects (Supplementary Fig. 4). ROI-based quantification confirmed these differential associations across eight predefined regions (Supplementary Table 2).
Results of the surface vertex-wise analyses show a significant relationship between brain atrophy regions measured on MRI and CSF NPTXs after adjusting for age, sex, educational level and apolipoprotein E (APOE)-ε4. Color bar represents the r values. All results were adjusted for multiple comparisons using FDR (p < 0.05).
Total cortical thickness analysis uncovered biomarker-specific relationships across pathological strata (Supplementary Fig. 5). In A+ individuals, both NPTXR (β = 0.260, p < 0.001) and NPTX1 (β = 0.232, p = 0.004) demonstrated strong structure-preserving effects, outperforming classical AD biomarkers. A- individuals exhibited bimodal biomarker associations: protective effects of NPTXR and NPTX1 (NPTXR β = 0.144, p = 0.026; NPTX1 β = 0.179, p = 0.017) contrasted with neurodegenerative effects of tau proteins (pTau181 β = −0.283, p = 0.001; tTau β = −0.247, p = 0.008). Clinico-biological validation demonstrated consistent cognition-structure coupling (Supplementary Figs. 6–7). Higher expression levels of both NPTXR and NPTX1 presented positive correlations with enhanced cognitive performance across all subgroups within the CANDI and DDI cohorts, with the exception of NPTXR levels observed in the A- subgroup of the DDI cohort. And alterations in pTau181 (β = −0.197, p = 0.017), tTau (β = −0.330, p < 0.001), Aβ burden (β = 0.243, p = 0.003), and NFL (β = −0.370, p < 0.001) were correlated cognitive changes in the A- group, while NFL and tTau was negative correlated with cognitive function in A+ subgroup.
Subsequently, we used CSF NPTX1 and NPTXR stages to examine the distribution of cognitive function scores across the AD continuum (Fig. 3). We observed that the degree of cognitive impairment increased with lower CSF NPTX1 and NPTXR levels and individuals at the NPTX1high and NPTXRhigh stage had the best cognition and highest cortical thickness.
The box plots depict the median (horizontal bar), interquartile range (IQR, hinges), and the whiskers indicate the minimum and maximum values. n = 255 (NPTXR), n = 183 (NPTX1). P-values (two-sided) were assessed by a one-way analysis of covariance (ANCOVA), adjusting for age, sex, educational level, Aβ42/Aβ40, pTau181 and apolipoprotein E (APOE)-ε4.
CSF biomarkers shift with increasing cortical atrophy
We next utilized cross-sectional data incorporating varying cortical thickness to simulate biomarker alterations during brain atrophy. Within the entire cohort, we found that decreased levels of NPTX1 and NPTXR, alongside increased levels of pTau181, tTau, and Aβ burden (but not NFL), were significantly associated with the severity of cortical damage and atrophy (Fig. 4). Specifically, in A+ individuals, NPTXR and NPTX1 levels consistently decreased with brain atrophy, whereas pTau181 levels initially increased and then decreased. Aβ42/Aβ40, tTau, and NFL showed weak correlations with cortical thickness across the Alzheimer’s disease (AD) continuum. In the A- group, other biomarkers exhibited poorer performance than pTau181 and tTau in reflecting progressive neurodegeneration.
The graphs represent the z-score changes of each biomarker in the group. The resulting z-scores are shown as a function of mean thickness of cortex using a robust local weighted regression method. The vertical gray represent the 95% CI of the corresponding indicator values for CU patients. The horizontal dashed lines depict the threshold held at mean and 0.5 SD above/below the mean.
Baseline CSF NPTXs predict neurodegeneration and MCI conversion
In the CANDI cohort, higher baseline levels of NPTXR and NPTX1 were consistently associated with slower rates of cortical thinning and cognitive decline. Significant interactions between linear time and protein levels were observed for cortical thickness (NPTXR x Time: β = 0.291, p = 0.005; NPTX1 x Time: β = 0.269, p = 0.011), indicating a neuroprotective effect (Fig. 5A, Supplementary Fig. 8A). Similarly, trajectories of both MMSE and MoCA scores showed significantly attenuated decline in individuals with higher baseline protein levels (linear time × protein interactions: p < 0.05 for all models; Fig. 5B, Supplementary Fig. 8B). These protective associations remained significant after adjusting for APOE ε4 status, age, sex, and education, suggesting that synaptic integrity biomarkers may confer resilience against neurodegenerative processes. This pattern contrasted with the positive associations observed between pTau181/Aβ burden and progressive atrophy, where higher pathological markers were correlated with accelerated structural decline (Supplementary Fig. 9). However, these associations were absent in the DDI cohort (Supplementary Fig. 10), potentially attributable to the cohort’s baseline characteristics—comprising primarily CU and early MCI individuals with minimal neurodegeneration and limited cognitive/imaging progression observed during the follow-up period—rather than a contradiction of the biomarker’s potential.
Quadratic trajectories of cortical thickness (A) and cognitive performance (B) over time, demonstrating a significant interaction between the linear time term and baseline CSF levels of both NPTXR and NPTX1. The red lines represent individuals with high baseline protein levels ( + 1 SD), blue lines represent those with low baseline levels ( − 1 SD), and green lines represent the mean. Shaded areas indicate 95% confidence intervals. P-values (two-sided) of fixed effects were assessed by linear mixed-effects models with subject-specific random intercepts and random slopes for time, adjusting for age, sex, educational level, and apolipoprotein E (APOE)-ε4.
Additionally, we assessed how cognitive and imaging data changed over time based on the CSF NPTX1 and NPTXR stage classification (Supplementary Fig. 11). The rate of cognitive decline, measured with MMSE and MoCA, significantly increased at the NPTX1med and NPTXRmed stage compared to NPTX1high and NPTXRhigh stage and remained higher thereafter. On the other hand, the rate of cortical thinning started to significantly increase at the NPTXRmed stage until reaching a plateau.
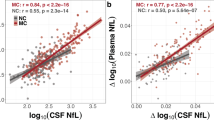
Finally, among 64 individuals diagnosed with MCI who were followed for an average of 2.5 ([SD],1.3) years, 42 remained stable (stable MCI, sMCI), while 22 progressed to dementia (progressive MCI, pMCI). Baseline CSF levels of NPTXR and NPTX1 were significantly higher in the sMCI group compared to the pMCI group (p = 0.002 and p = 0.003, respectively; Fig. 6A). Receiver operating characteristic (ROC) curve analysis demonstrated that baseline CSF levels of NPTXR and NPTX1 effectively discriminated between MCI progressors and non-progressors (AUC = 0.823 and AUC = 0.806, respectively). At the optimal cut-off points, both biomarkers achieved a sensitivity of 80%, with specificities of 83% (NPTXR) and 78% (NPTX1). The positive predictive value (PPV) was 75% for NPTXR and 71% for NPTX1, while the negative predictive value (NPV) was 86% for both biomarkers (Fig. 6B). These performances surpassed that of plasma pTau217 (AUC = 0.780, PPV = 65%, NPV = 89%; Supplementary Fig. 12).
A Comparison of baseline CSF NPTXs between MCI-MCI and MCI-dementia patients. The box plots depict the median (horizontal bar), interquartile range (IQR, hinges), and the whiskers indicate the minimum and maximum values. n = 51 (sMCI), n = 24 (pMCI). P-values (two-sided) were assessed by a one-way analysis of covariance (ANCOVA), adjusting for age, sex, educational level, and apolipoprotein E (APOE)-ε4. B Receiver operating characteristic curve analysis for differentiating individuals with progressing MCI from those with stable MCI using baseline CSF NPTXs. AUC: Area Under Curve, PPV: Positive Predictive Value, NPV: Negative Predictive Value.
Discussion
In the present study, we investigated CSF NPTX1 and NPTXR as biomarkers across the AD continuum, leveraging two independent ethnogeographically distinct cohorts: Chinese CANDI cohort and the Norwegian DDI cohort. Our primary finding, validated across both populations, is that lower CSF NPTX1 and NPTXR levels are robustly associated with greater disease severity, assessed by cognitive function and neuroimaging measures of atrophy, particularly within the AD continuum. Furthermore, longitudinal analyses in the CANDI cohort demonstrated that the associations of NPTX1 and NPTXR with longitudinal cognitive decline and neurodegeneration trajectories, and lower baseline NPTX1 and NPTXR levels predict accelerated subsequent brain atrophy, cognitive decline, and progression from MCI to dementia. These findings position CSF NPTX1 and NPTXR as valuable biomarkers reflecting the severity of neurodegeneration and synaptic dysfunction relevant to clinical outcomes in AD.
A key strength of our study is the replication of core cross-sectional findings in two diverse cohorts. The consistent observation that CSF NPTX1 and NPTXR levels progressively decrease with worsening cognitive status and correlate positively with cortical thickness in Aβ-positive individuals across both Eastern (CANDI) and Western (DDI) populations strongly supports their robustness as biomarkers.
The effect sizes for the associations between NPTX1 and NPTXR and cognition within the biologically defined AD (A + ) group were moderate (β = 0.333–0.460), reaching levels considered potentially clinically meaningful (β > 0.30)25. This consistency and magnitude underscore the potential utility of NPTX1 and NPTXR for specifically tracking AD-related neurodegeneration, complementing established AD core biomarkers. Our NPTX-based staging analysis further illustrated this, showing that even after accounting for Aβ and pTau pathology, lower NPTX levels corresponded to worse cognitive function and greater atrophy, suggesting they sensitively track neurodegenerative severity beyond core AD pathology.
Our observation of a nonlinear, accelerating trajectory of cortical thinning is consistent with seminal reports on the temporal dynamics of neurodegeneration in AD26,27. Furthermore, longitudinal data from the CANDI cohort provide compelling evidence for the prognostic value of CSF NPTX1 and NPTXR. Lower baseline levels not only predicted faster rates of cortical and hippocampal atrophy but also accelerated cognitive decline. Crucially, baseline NPTX1 and NPTXR levels effectively distinguished MCI participants who progressed to dementia (pMCI) from those who remained stable (sMCI) over ~2.5 years, achieving high sensitivity and specificity (AUCs ~0.80). This predictive capacity highlights their potential utility in identifying individuals at high risk for imminent clinical decline.
Comparing NPTX1 and NPTXR to established biomarkers revealed distinct patterns. Within the Aβ-positive AD continuum in our cohorts, CSF NPTX levels showed a clearer relationship with cognitive stage and atrophy severity than CSF Aβ42/40 ratio, pTau181, or serum NFL. While abnormal Aβ ratios signify underlying pathology and predict future risk28, they often plateau early and may not directly track concurrent cognitive status or atrophy severity29,30. Similarly, while CSF pTau isoforms are excellent markers of tau pathology, CSF pTau181 levels did not consistently track cognitive stages within the A+ group in our study, potentially reflecting complex dynamics not fully captured by this specific isoform alone31. Serum NFL, a marker of general axonal injury, also showed limited association with cognitive stage or longitudinal atrophy prediction within the A+ continuum in our cohorts, consistent with its known sensitivity to non-AD factors like cerebrovascular disease10 and potential stage-dependent behavior32,33. The distinct trajectory of NPTX1 and NPTXR—declining progressively with severity after Aβ pathology is established—suggests they specifically reflect downstream consequences of AD pathology on synaptic integrity and neurodegeneration, offering complementary information to AD core markers for staging disease progression.
Biologically, our findings align with the established role of synaptic loss as a core pathological feature closely linked to cognitive decline in AD12,13. The observed decrease in CSF NPTX1 and NPTXR likely reflects this underlying synaptic degeneration. The positive correlation between NPTX1 and NPTXR and pTau181 within the A+ group might seem counterintuitive but could reflect complex interactions where early pathological processes trigger compensatory responses or release mechanisms, before overwhelming synaptic loss leads to a net decrease in secreted synaptic proteins like NPTX1 and NPTXR34. The potential biphasic pattern of NPTXR observed in Aβ-negative individuals (initial increase in MCI, then decrease in dementia), also noted for other synaptic markers in some studies35, might suggest early, perhaps compensatory, synaptic changes preceding widespread degeneration, a phenomenon less apparent or masked by the dominant pathological drive in Aβ-positive AD.
This study has some limitations. First, while analyses of both CANDI and DDI cohorts confirmed associations between reduced baseline NPTX levels and neurodegeneration, the lack of significant longitudinal progression in the DDI cohort limited our ability to validate the predictive aspects of NPTX1 and NPTXR in that specific population, emphasizing the need for validation in cohorts with broader disease severity and longer follow-up. Second, although elevated levels of NFL in the CSF of AD patients are indicative of neuronal death and axonal degeneration36, our study measured only serum NFL and did not investigate CSF NFL levels or compare them with other CSF synaptic proteins. Third, the generally lower education levels (mean 7.7 years) in our CANDI cohort may affect cognitive test performance. However, we adjusted for education in all regression analyses, and the replication of core findings in the highly educated DDI cohort (mean 13.8 years) mitigates concerns about education confounding the primary NPTX associations. Lastly, larger sample sizes and extended follow-up durations are necessary to fully establish the clinical robustness and precise cut-offs for NPTX-based staging and prediction.
In summary, this dual-cohort study provides robust, validated evidence that CSF NPTX1 and NPTXR levels track the severity of neurodegeneration and cognitive impairment across the AD continuum, particularly in Aβ-positive individuals. Lower baseline levels predict subsequent clinical and structural decline. Overall, these results strongly suggest that CSF NPTX1 and NPTXR are valuable biomarkers reflecting synaptic dysfunction and neuronal injury (‘N’) that offer complementary information to established AD core biomarkers. They hold promise for improving patient stratification, monitoring disease progression, and potentially assessing the efficacy of synaptically-targeted therapies in AD clinical trials.
Methods
Study participants and cohorts
China aging and neurodegenerative disorder initiative (CANDI)
This prospective cohort, established in 2018 at the First Affiliated Hospital of USTC37, contributed 435 participants ( > 50 years old), from part of the CANDI cohort. Inclusion required good general health, ability to undergo comprehensive assessments (clinical, neuroimaging, CSF/blood collection), and presence of a reliable informant (e.g., spouse or close family member) during clinical visits to provide collateral history. Key exclusion criteria encompassed individuals with: (1) any significant neurological disease other than dementia (e.g., Huntington’s disease, seizure disorder, brain tumor, multiple sclerosis); (2) any notable systemic illness that was not adequately controlled through a stable medication regimen (e.g., hypertension, hyperlipidemia, diabetes mellitus); or (3) a history of major depression, bipolar disorder, or schizophrenia that could potentially impact the reliability of cognitive assessments. Cognitively unimpaired (CU) individuals served as the control group. These participants did not meet diagnostic criteria for MCI or any form of dementia at baseline. Clinical diagnoses of MCI and AD were established following the NIA-AA criteria (2011)38 supported by Mini-Mental State Examination (MMSE), Clinical Dementia Rating (CDR) scale, and Montreal Cognitive Assessment (MoCA) assessments, incorporating informant input. MMSE scores ranged from 24–30 for CU and MCI participants and were ≤ 26 for dementia patients. CDR scores were 0 for CU participants, 0.5 for MCI participants, and 0.5 or 1 for dementia patients. Non-AD dementia (non-ADD) patients comprised Aβ-negative, cognitively impaired individuals diagnosed with conditions such as dementia with Lewy bodies, frontotemporal dementia (FTD), vascular dementia, or Parkinson’s disease dementia, based on clinical evaluation. This study was conducted under the framework of a Major International Joint Research Project from the NSFC. All laboratory analyses for the Chinese cohort—including CSF protein assays and APOE genotyping—were performed independently within China. Individual-level raw data from Chinese participants were not shared with international partner sites to ensure regulatory compliance. Ethics approval was obtained (First Affiliated Hospital of USTC: 2019KY-26, 2023KY-117), and written informed consent was secured from participants or their legally authorized representatives.
Dementia disease initiation (DDI)
This Norwegian multi-center cohort39 contributed 200 participants (40–80 years old), including CU and MCI individuals recruited from memory clinics and the community (2013-2022). All underwent standardized diagnostic assessments (medical history, neurological exams, neuropsychological testing, MRI, CSF/blood collection) with ~10-year follow-ups. Notably, MCI versus CU classification in DDI relied on an actuarial neuropsychological approach, independent of subjective cognitive complaints40. Ethics approval was granted by the Norwegian Regional Committees for Medical and Health Research Ethics, adhering with the Declaration of Helsinki and the Norwegian Health Research Act. Written informed consent was obtained from all participants.
Cognitive composite assessments
For the DDI cohort, we applied a recently published cognitive composite score comprising delayed verbal memory recall (Consortium to Establish a Registry for Alzheimer’s Disease (CERAD) word list test) and executive functions (Trail Making Test B (TMT-B)41. Briefly, the CERAD delayed verbal memory recall test was rescaled to a 0–1 range, where 0 represents the lowest possible performance and 1 the highest. For the TMT-B, where higher raw scores indicate worse performance, scores were inverted and similarly rescaled so that higher values reflect better performance. The standardized scores were then summed and averaged to create a composite score ranging from 0 to 1.
CSF biomarkers
CSF samples were collected via lumbar puncture and processed as outlined in a previous study37,42. Biomarker quantification was performed using platform-specific methodologies: For the CANDI cohort, CSF concentrations of Aβ40, Aβ42, total tau (tTau), and phosphorylated tau-181 (pTau181) were analyzed using Simoa kits on a Quanterix HD-X analyzer. In contrast, the DDI cohort measurements for Aβ42 and Aβ40 utilized the V-PLEX Aβ Peptide Panel 1 (6E10) kit (Meso Scale Discovery [MSD]) on a QuickPlex SQ 120 system in multiplex format.
Pathological classification criteria differed between cohorts: In the CANDI cohort, Aβ status was determined by the CSF Aβ42/Aβ40 ratio43, with threshold values set at 0.0642, as detailed in our earlier studies37. While for the DDI cohort, amyloid pathology (A+ threshold: CSF Aβ42/40 ≤ 0.077) was established through ROC curve analysis using [18 F]-flutemetamol PET visual reads as the reference standard42.
Neuronal pentraxin measurements employed distinct analytical approaches: NPTX1 levels were quantified using a commercial MSD immunoassay (K151T8R-2) following manufacturer specifications, whereas NPTXR concentrations were determined via a custom-developed MSD-based assay (detailed methodology provided in Supplementary Methods and Supplementary Fig. 13). For A+ individuals, NPTX biomarker stages were stratified into tertile-based categories: low (NPTXR ≤ 1434.0 pg/mL, NPTX1 ≤ 6132.3 pg/mL), moderate (1434.0 pg/mL <NPTXR ≤ 2106.8 pg/mL, 6132.3 pg/mL <NPTX1 ≤ 8954.4 pg/mL), and high (NPTXR > 2106.8 pg/mL, NPTX1 > 8954.4 pg/mL).
Blood biomarkers
Blood samples were collected following an overnight fast, centrifuged, aliquoted, and stored at −80 °C. Serum NFL and plasma pTau217 were measured using the Simoa kits (Quanterix, 103520, 104570) on the HD-X platform, as indicated in a previous study44.
Neuroimaging
To address fundamental differences in ethnicity and study design between the cohorts, neuroimaging data were analyzed independently. Our primary strategy for assessing the generalizability of our findings across this population-level heterogeneity was a robust discovery (CANDI) and independent external validation (DDI) framework.
Aβ positron emission tomography (PET) imaging was performed using 18F-florbetapir (AV-45), and details of AV-45 PET imaging have been previously described37. Participants meeting AD criteria were classified as AV-45 PET positive (A + ), while others were designated as amyloid-negative. For subjects lacking PET data, amyloid status was determined through CSF Aβ42/40 ratio quantification using a validated threshold (A + : Aβ42/40 < 0.0642).
For the single-center CANDI cohort, a subset of 354 participants underwent T1-weighted structural magnetic resonance imaging (MRI). All scans were acquired on a single GE DISCOVER 750w 3.0 T MRI scanner (GE Healthcare, USA). T1-weighted images were processed using the automated recon-all pipeline in FreeSurfer version 7.2.0, from which cortical thickness values were extracted for 68 regions of interest (ROIs) based on the Desikan–Killiany atlas, along with subcortical volumes (e.g., hippocampus), and estimated total intracranial volume (eTIV). Images exhibiting poor quality were visually inspected and manually corrected if possible; otherwise, they were excluded. The associations between baseline biomarkers and brain measures were tested as follows: (a) Cortical Thickness: Multiple linear regression was performed for each ROI, with cortical thickness as the dependent variable, adjusting for age, sex, education level, eTIV, and APOE-ε4 carrier status. (2) Subcortical Volumes: For volumes of interest (e.g., hippocampus), the volume-to-eTIV ratio was used as the dependent variable in regression models, adjusting for age, sex, education level, and APOE-ε4 carrier status. Longitudinal analyses of change in brain measures followed the same covariate and ratio adjustment principles. The false discovery rate (FDR) method was applied to correct for multiple comparisons.
Within the multi-site DDI cohort, MRI data were acquired from six different sites (n = 200). To address this site-related technical heterogeneity prior to the validation analysis, we implemented the longitudinal ComBat harmonization method for cortical thickness measures, as described by Beer et al.45. The harmonization model included adjustment for age, sex, education, eTIV, and APOE-ε4 carrier status. The Destrieux atlas was used to parcellate the cortex into 148 ROIs. This cohort focused on the association between biomarkers and cortical measures. The association between baseline biomarkers and baseline cortical thickness was assessed using linear regression, with eTIV included as a covariate. The association with the rate of cortical atrophy (longitudinal change in thickness) was assessed using the harmonized longitudinal data, with the same set of covariates.
Statistical analysis
Statistical analyses were performed using SPSS (IBM, Chicago, USA) and R (R Foundation for Statistical Computing, USA). To justify our analytical approach, we formally assessed missing data patterns in the CANDI cohort (Supplementary Methods). This analysis revealed no significant associations (all p > 0.05) between biomarker missingness and any baseline clinical, demographic, or cognitive variables (Supplementary Table 3), providing empirical support for the use of complete-case and available-case analyses under a missing-completely-at-random (MCAR) assumption. For analyses involving individual fluid biomarkers, all available observations for each biomarker were included (available-case analysis). For analyses directly comparing multiple biomarkers or requiring a consistent sample across measures, only participants with complete data for all relevant biomarkers were included (complete-case analysis). Categorical variables were compared using Pearson’s chi-square tests. For continuous variables, group differences were assessed by one-way analysis of covariance (ANCOVA) and t-test, with adjustments for age, sex, education, and APOE ε4 status; analyses of cortical thickness were additionally adjusted for eTIV. Post-hoc comparisons were corrected using the Tukey–Kramer method. Linear regression models were employed to examine associations between fluid biomarkers and other variables, adjusting for age, sex, education, and APOE ε4 status. Cortical thickness analyses further included eTIV as a covariate. Biomarker values were standardized as z-scores and visualized along the brain atrophy continuum using a robust locally weighted regression (LOWESS) smoothing method. To evaluate the moderating effect of CSF levels of NPTX1 and NPTXR on longitudinal cortical thinning and cognitive decline, linear mixed-effects models were fitted. These models included orthogonal quadratic time terms, their interaction with baseline protein levels (scaled), and covariates (age, sex, education, APOE ε4 status). Subject-specific random intercepts and random slopes for time were incorporated to account for within-subject correlations. The significance of fixed effects was determined using Satterthwaite’s approximation. Finally, receiver operating characteristic (ROC) analysis was conducted to assess the ability of baseline CSF NPTX1 and NPTXR levels to predict progression from MCI to dementia during follow-up.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
Based on the EU General Data Protection Regulation (GDPR) and the DDI Ethics Review Board provisions, anonymized aggregated-level data from DDI cohort will be shared by request from a qualified academic investigator for the sole purpose of replicating procedures and results presented in the article. Data transfer will comply with applicable regulations and relevant ethical review decisions. Source data for the main figures are provided with this paper. However, in accordance with the Regulations on the Management of Human Genetic Resources of China, individual-level clinical and genetic data from the Chinese (CANDI) cohort cannot be deposited in public repositories. Summary-level data and the findings supporting the study’s conclusions are available from the corresponding author upon reasonable request and subject to regulatory approval. Source data are provided with this paper.
References
Frisoni, G. B. et al. The probabilistic model of Alzheimer disease: the amyloid hypothesis revised. Nat. Rev. Neurosci. 23, 53–66 (2022).
Gobom, J. et al. Validation of the LUMIPULSE automated immunoassay for the measurement of core AD biomarkers in cerebrospinal fluid. Clin. Chem. Lab Med 60, 207–219 (2022).
Palmqvist, S. et al. Performance of fully automated plasma assays as screening tests for Alzheimer disease-related beta-amyloid status. JAMA Neurol. 76, 1060–1069 (2019).
Mila-Aloma, M. et al. Plasma p-tau231 and p-tau217 as state markers of amyloid-beta pathology in preclinical Alzheimer’s disease. Nat. Med. 28, 1797–1801 (2022).
Karikari, T. K. et al. Blood phosphorylated tau 181 as a biomarker for Alzheimer’s disease: a diagnostic performance and prediction modelling study using data from four prospective cohorts. Lancet Neurol. 19, 422–433 (2020).
Ashton, N. J. et al. Diagnostic accuracy of a plasma phosphorylated Tau 217 immunoassay for Alzheimer disease pathology. JAMA Neurol. 81, 255–263 (2024).
Jack, C. R. Jr. et al. NIA-AA research framework: toward a biological definition of Alzheimer’s disease. Alzheimer’s. Dement J. Alzheimer’s. Assoc. 14, 535–562 (2018).
Jack, C. R. Jr. et al. Revised criteria for the diagnosis and staging of Alzheimer’s disease. Nat. Med. 30, 2121–2124 (2024).
Bavato, F. et al. Introducing neurofilament light chain measure in psychiatry: current evidence, opportunities, and pitfalls. Mol. Psychiatry 29, 2543–2559 (2024).
Korley, F. K. et al. Serum NfL (Neurofilament Light Chain) levels and incident stroke in adults with diabetes mellitus. Stroke 50, 1669–1675 (2019).
Sudhof, T. C. Towards an understanding of synapse formation. Neuron 100, 276–293 (2018).
Tzioras, M., McGeachan, R. I., Durrant, C. S. & Spires-Jones, T. L. Synaptic degeneration in Alzheimer disease. Nat. Rev. Neurol. 19, 19–38 (2023).
Colom-Cadena, M. et al. The clinical promise of biomarkers of synapse damage or loss in Alzheimer’s disease. Alzheimer’s. Res. Ther. 12, 21 (2020).
Sia, G. M. et al. Interaction of the N-terminal domain of the AMPA receptor GluR4 subunit with the neuronal pentraxin NP1 mediates GluR4 synaptic recruitment. Neuron 55, 87–102 (2007).
Pelkey, K. A. et al. Pentraxins coordinate excitatory synapse maturation and circuit integration of parvalbumin interneurons. Neuron 90, 661 (2016).
Zhou, J. et al. The neuronal pentraxin Nptx2 regulates complement activity and restrains microglia-mediated synapse loss in neurodegeneration. Sci. Transl. Med. 15, eadf0141 (2023).
Libiger, O. et al. Longitudinal CSF proteomics identifies NPTX2 as a prognostic biomarker of Alzheimer’s disease. Alzheimer Dement 17, 1976–1987 (2021).
Xiao, M. F. et al. NPTX2 and cognitive dysfunction in Alzheimer’s Disease. Elife 6 e23798 (2017).
Nilsson, J. et al. Cerebrospinal fluid biomarker panel for synaptic dysfunction in Alzheimer’s disease. Alzheimers Dement (Amst.) 13, e12179 (2021).
Oh, H. S. et al. A cerebrospinal fluid synaptic protein biomarker for prediction of cognitive resilience versus decline in Alzheimer’s disease. Nat. Med. 31, 1592–1603 (2025).
van der Ende, E. L. et al. Neuronal pentraxin 2: a synapse-derived CSF biomarker in genetic frontotemporal dementia. J. Neurol. Neurosurg. Psychiatry 91, 612–621 (2020).
Nilsson, J. et al. Cerebrospinal fluid biomarkers of synaptic dysfunction are altered in parkinson’s disease and related disorders. Mov. Disord. 38, 267–277 (2023).
Lim, B. et al. Liquid biopsy of cerebrospinal fluid identifies neuronal pentraxin receptor (NPTXR) as a biomarker of progression of Alzheimer’s disease. Clin. Chem. Lab Med 57, 1875–1881 (2019).
Begcevic, I. et al. Neuronal pentraxin receptor-1 is a new cerebrospinal fluid biomarker of Alzheimer’s disease progression. F1000Research 7, 1012 (2018).
Nieminen, P. Application of standardized regression coefficient in meta-analysis. BioMedInformatics 2, 434–458 (2022).
Jack, C. R. Jr. et al. Hypothetical model of dynamic biomarkers of the Alzheimer’s pathological cascade. Lancet Neurol. 9, 119–128 (2010).
Jack, C. R. Jr. et al. Tracking pathophysiological processes in Alzheimer’s disease: an updated hypothetical model of dynamic biomarkers. Lancet Neurol. 12, 207–216 (2013).
Aschenbrenner, A. J. et al. Comparison of plasma and CSF biomarkers in predicting cognitive decline. Ann. Clin. Transl. Neurol. 9, 1739–1751 (2022).
Dansson, H. V. et al. Predicting progression and cognitive decline in amyloid-positive patients with Alzheimer’s disease. Alzheimer’s. Res. Ther. 13, 151 (2021).
Seidu, N. M. et al. Association of CSF biomarkers with MRI brain changes in Alzheimer’s disease. Alzheimers Dement (Amst.) 16, e12556 (2024).
Salvado, G. et al. Novel CSF tau biomarkers can be used for disease staging of sporadic Alzheimer’s disease. medRxiv (2023).
Lee, E. H. et al. Serum neurofilament light chain level as a predictor of cognitive stage transition. Alzheimer’s. Res. Ther. 14, 6 (2022).
Preische, O. et al. Serum neurofilament dynamics predicts neurodegeneration and clinical progression in presymptomatic Alzheimer’s disease. Nat. Med. 25, 277–283 (2019).
Wu, M. et al. The role of pathological tau in synaptic dysfunction in Alzheimer’s diseases. Transl. Neurodegener. 10, 45 (2021).
Duits, F. H. et al. Synaptic proteins in CSF as potential novel biomarkers for prognosis in prodromal Alzheimer’s disease. Alzheimer’s. Res. Ther. 10, 5 (2018).
Skillback, T., Zetterberg, H., Blennow, K. & Mattsson, N. Cerebrospinal fluid biomarkers for Alzheimer disease and subcortical axonal damage in 5,542 clinical samples. Alzheimer’s. Res. Ther. 5, 47 (2013).
Gao, F. et al. A combination model of AD biomarkers revealed by machine learning precisely predicts Alzheimer’s dementia: China Aging and Neurodegenerative Initiative (CANDI) study. Alzheimer’s. Dement. 0, 00–00 (2022).
McKhann, G. M. et al. The diagnosis of dementia due to Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimer’s. Dement. 7, 263–269 (2011).
Gonzalez-Ortiz, F. et al. Plasma brain-derived tau is an amyloid-associated neurodegeneration biomarker in Alzheimer’s disease. Nat. Commun. 15, 2908 (2024).
Nordengen, K. et al. Longitudinal cerebrospinal fluid measurements show glial hypo- and hyperactivation in predementia Alzheimer’s disease. J. Neuroinflamm. 20, 298 (2023).
Kirsebom, B.-E. et al. Repeated plasma p-tau217 measurements to monitor clinical progression heterogeneity. Alzheimer Dement. 21, e70319 (2025).
Fladby, T. et al. Detecting at-risk alzheimer’s disease cases. J. Alzheimer. Dis. 60, 97–105 (2017).
Jack, C. R. Jr. et al. A/T/N: an unbiased descriptive classification scheme for Alzheimer disease biomarkers. Neurology 87, 539–547 (2016).
Gao, F. et al. Blood-based biomarkers for Alzheimer’s disease: a multicenter-based cross-sectional and longitudinal study in China. Sci. Bull. (Beijing) 68, 1800–1808 (2023).
Beer, J. C. et al. Longitudinal ComBat: a method for harmonizing longitudinal multi-scanner imaging data. Neuroimage 220, 117129 (2020).
Acknowledgements
This work was supported by the National Natural Sciences Foundation of China (W2411069 and 82030034 to Y.S.; U23A20422 and 82501685 to L.D.; 82371418 and 82571610 to F.G.), the Chinese Academy of Sciences (XDB39000000 to Y.S.), the Fundamental Research Funds for the Central Universities (YD9110002027 and WK9100000057 to L.D.), Postdoctoral Science Foundation of Anhui Province (2023B723 to L.D.), Hefei Comprehensive National Science Center Hefei Brain Project (to Y.S.), the Major Frontier Research Project of the University of Science and Technology of China (LS9100000002 to Y.S.), USTC Research Funds of the Double First-Class Initiative (YD910002087 to F.G.), and the Norwegian Research Council (JPND/PMI-AD, NRC 311993 to T.F.).
Author information
Authors and Affiliations
Contributions
Y.S., K.B., F.G. and L.D. contributed to the conception and design of the study. L.D., C.W., M.Z., Q.W., M.N., B.E.K., F.G.-O., T.F. and J.S. contributed to the acquisition and analysis of data. L.D., F.G., B.E.K., F.G.-O., K.B. and Y.S. contributed to drafting the text or preparing the figures.
Corresponding authors
Ethics declarations
Competing interests
B.E.K. has served as a consultant for Biogen and medical advisory boards for Biogen and Eli Lilly. T.F. has served as a consultant and at the advisory boards for Biogen, Eisai, Novo Nordisk, Eli Lilly and Roche. K.B. has served as a consultant and at advisory boards for AbbVie, AC Immune, ALZPath, AriBio, Beckman-Coulter, BioArctic, Biogen, Eisai, Lilly, Moleac Pte. Ltd, Neurimmune, Novartis, Ono Pharma, Prothena, Quanterix, Roche Diagnostics, Sunbird Bio, Sanofi and Siemens Healthineers; has served at data monitoring committees for Julius Clinical and Novartis; has given lectures, produced educational materials and participated in educational programs for AC Immune, Biogen, Celdara Medical, Eisai and Roche Diagnostics; and is a co-founder of Brain Biomarker Solutions in Gothenburg AB (BBS), which is a part of the GU Ventures Incubator Program, outside the work presented in this paper. The remaining authors declare no competing interests
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Dai, L., Kirsebom, BE., Wang, C. et al. Cerebrospinal fluid NPTX1 and NPTXR predict neurodegeneration and clinical progression in Alzheimer’s disease. Nat Commun 17, 3674 (2026). https://doi.org/10.1038/s41467-026-70472-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-026-70472-6