Abstract
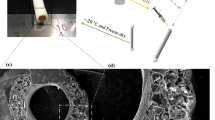
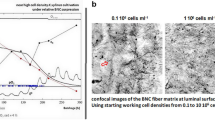
The increasing prevalence of cardiovascular diseases and haemodialysis access drive high clinical demand for vascular grafts. Bioinert synthetic and decellularized or cryopreserved artery grafts exhibit poor regeneration and associated complications. Here, we present an approach that combines in vivo bioengineering and decellularization to prepare polycaprolactone skeleton-reinforced biotubes (dPB), which demonstrate appropriate mechanical strength, anti-kinking properties, resistance to repeat puncture, and are compatible with long-term storage at 4 °C. The loose and porous microstructure and immunomodulatory bioactivity of dPB induce rapid recellularization and promote pro-regenerative M2 macrophage polarization, which together facilitate remodelling to native artery-like tissue. In porcine coronary artery bypass grafting, canine carotid artery replacements and arteriovenous grafting models, ovine-derived dPB show high patency without dilatation, calcification and intimal hyperplasia. In summary, dPB offer a readily available and pro-regenerative vascular graft with a broad application prospect for patients with unavailable autologous vessels.
Similar content being viewed by others
Data availability
The proteomics data generated in this study have been deposited in the ProteomeXchange database under accession code PXD051651. Raw data and processed data required to reproduce the analyses are available through this accession. Source data underlying the main and Supplementary Figs. are provided with this paper as a Source Data file. Source data are provided with this paper.
References
Niklason, L. E. & Lawson, J. H. Bioengineered human blood vessels. Science 370, eaaw8682 (2020).
Breuer, T., Jimenez, M., Humphrey, J. D., Shinoka, T. & Breuer, C. K. Tissue engineering of vascular grafts: a case report from bench to bedside and back. Arterioscler. Thromb. Vasc. Biol. 43, 399–409 (2023).
Gupta, P. & Mandal, B. B. Tissue-engineered vascular grafts: emerging trends and technologies. Adv. Funct. Mater. 31, 2100027 (2021).
Mehta, R. I., Mukherjee, A. K., Patterson, T. D. & Fishbein, M. C. Pathology of explanted polytetrafluoroethylene vascular grafts. Cardiovasc. Pathol. 20, 213–221 (2011).
Roina, Y., Auber, F., Hocquet, D. & Herlem, G. ePTFE-based biomedical devices: an overview of surgical efficiency. J. Biomed. Mater. Res. B Appl. Biomater. 110, 302–320 (2022).
Fang, S., Ellman, D. G. & Andersen, D. C. Tissue engineering of small-diameter vascular grafts and their in vivo evaluation in large animals and humans. Cells 10, 713 (2021).
Halbert, R. J., Nicholson, G., Nordyke, R. J., Pilgrim, A. & Niklason, L. Patency of ePTFE arteriovenous graft placements in hemodialysis patients: systematic literature review and meta-analysis. Kidney360 1, 1437–1446 (2020).
Bachleda, P. et al. Infectious complications of arteriovenous ePTFE grafts for hemodialysis. Biomed. Pap. Med. Fac. Univ. Palacky. Olomouc Czech Repub. 154, 13–19 (2010).
Zizhou, R., Wang, X. & Houshyar, S. Review of polymeric biomimetic small-diameter vascular grafts to tackle intimal hyperplasia. ACS Omega 7, 22125–22148 (2022).
Gaharwar, A. K., Singh, I. & Khademhosseini, A. Engineered biomaterials for in situ tissue regeneration. Nat. Rev. Mater. 5, 686–705 (2020).
Hussey, G. S., Dziki, J. L. & Badylak, S. F. Extracellular matrix-based materials for regenerative medicine. Nat. Rev. Mater. 3, 159–173 (2018).
Syedain, Z. H. et al. Pediatric tri-tube valved conduits made from fibroblast-produced extracellular matrix evaluated over 52 weeks in growing lambs. Sci. Transl. Med. 13, eabb7225 (2021).
Shanberg, D. A., Stark, K., Kujath, S. & Hsiung, I. Evaluation of the Artegraft conduit for lower extremity revascularization: retrospective analysis of outcomes when used in the situation of no available autologous saphenous vein for conduit in lower extremity bypass procedures. J. Vasc. Surg. 76, e51 (2022).
Bibevski, S. et al. Performance of SynerGraft decellularized pulmonary allografts compared with standard cryopreserved allografts: results from multiinstitutional data. Ann. Thorac. Surg. 103, 869–874 (2017).
Heo, S. H. et al. Recent results of in situ abdominal aortic reconstruction with cryopreserved arterial allograft. Eur. J. Vasc. Endovasc. Surg. 53, 158–167 (2017).
Pashneh-Tala, S., MacNeil, S. & Claeyssens, F. The tissue-engineered vascular graft—past, present, and future. Tissue Eng. Part B Rev. 22, 68–100 (2016).
Behr, J.-M., Wong, Y. S. & Venkatraman, S. Small-diameter blood vessel substitutes: biomimetic approaches to improve patency. Biomimetics 9, 97 (2024).
Dahl, S. L. M. et al. Readily available tissue-engineered vascular grafts. Sci. Transl. Med. 3, 68ra69 (2011).
Syedain, Z. H. et al. A completely biological “off-the-shelf” arteriovenous graft that recellularizes in baboons. Sci. Transl. Med. 9, eaan4209 (2017).
Geelhoed, W. J., Moroni, L. & Rotmans, J. I. Utilizing the foreign body response to grow tissue engineered blood vessels in vivo. J. Cardiovasc. Transl. Res. 10, 167–179 (2017).
Lawson, J. H. et al. Bioengineered human acellular vessels for dialysis access in patients with end-stage renal disease: two phase 2 single-arm trials. Lancet 387, 2026–2034 (2016).
Kirkton, R. D. et al. Bioengineered human acellular vessels recellularize and evolve into living blood vessels after human implantation. Sci. Transl. Med. 11, eaau6934 (2019).
Nakayama, Y., Kaneko, Y., Takewa, Y. & Okumura, N. Mechanical properties of human autologous tubular connective tissues (human biotubes) obtained from patients undergoing peritoneal dialysis. J. Biomed. Mater. Res. B Appl. Biomater. 104, 1431–1437 (2016).
Jankovic, M. G. et al. Scaling up human mesenchymal stem cell manufacturing using bioreactors for clinical uses. Curr. Res. Transl. Med. 71, 103393 (2023).
Shuto, T. et al. Safety and efficacy of an iBTA-induced autologous Biotube vascular graft and its preparation device BTM1 in below-the-knee bypass surgery for chronic limb threatening ischemia: a protocol for an open-label, single-arm, multicenter clinical trial. PLoS One 20, e0335900 (2025).
Higashita, R. et al. Acute phase pilot evaluation of small diameter long iBTA induced vascular graft “Biotube” in a goat model. EJVES Vasc. Forum 54, 27–35 (2022).
Abdollahi, S., Boktor, J. & Hibino, N. Bioprinting of freestanding vascular grafts and the regulatory considerations for additively manufactured vascular prostheses. Transl. Res. 211, 123–138 (2019).
Devillard, C. D. & Marquette, C. A. Vascular tissue engineering: challenges and requirements for an ideal large scale blood vessel. Front. Bioeng. Biotechnol. 9, 721843 (2021).
Zhi, D. et al. Mechanically reinforced biotubes for arterial replacement and arteriovenous grafting inspired by architectural engineering. Sci. Adv. 8, eabl3888 (2022).
Konig, G. et al. Mechanical properties of completely autologous human tissue engineered blood vessels compared to human saphenous vein and mammary artery. Biomaterials 30, 1542–1550 (2009).
Obiweluozor, F. O. et al. Considerations in the development of small-diameter vascular graft as an alternative for bypass and reconstructive surgeries: a review. Cardiovasc. Eng. Technol. 11, 495–521 (2020).
Mugnai, D. et al. Experimental noninferiority trial of synthetic small-caliber biodegradable versus stable vascular grafts. J. Thorac. Cardiovasc. Surg. 146, 400–407 (2013).
Mayorca-Guiliani, A. E. et al. ISDoT: in situ decellularization of tissues for high-resolution imaging and proteomic analysis of native extracellular matrix. Nat. Med. 23, 890–898 (2017).
Yang, Y., Ritchie, A. C. & Everitt, N. M. Using type III recombinant human collagen to construct a series of highly porous scaffolds for tissue regeneration. Colloids Surf. B Biointerfaces 208, 112139 (2021).
Izu, Y. & Birk, D. E. Collagen XII mediated cellular and extracellular mechanisms in development, regeneration, and disease. Front. Cell Dev. Biol. 11, 1129000 (2023).
Jayadev, R. & Sherwood, D. R. Basement membranes. Curr. Biol. 27, R207–R211 (2017).
Reggio, S. et al. Increased basement membrane components in adipose tissue during obesity: links with TGFβ and metabolic phenotypes. J. Clin. Endocrinol. Metab. 101, 2578–2587 (2016).
Sadtler, K. et al. The scaffold immune microenvironment: biomaterial-mediated immune polarization in traumatic and nontraumatic applications. Tissue Eng. Part A 23, 1044–1053 (2017).
Rodríguez-Morales, P. & Franklin, R. A. Macrophage phenotypes and functions: resolving inflammation and restoring homeostasis. Trends Immunol. 44, 986–998 (2023).
Zhang, F. & King, M. W. Immunomodulation strategies for the successful regeneration of a tissue-engineered vascular graft. Adv. Healthc. Mater. 11, 2200045 (2022).
Hibino, N. et al. A critical role for macrophages in neovessel formation and the development of stenosis in tissue-engineered vascular grafts. FASEB J. 25, 4253–4263 (2011).
Zilla, P., Bezuidenhout, D. & Human, P. Prosthetic vascular grafts: wrong models, wrong questions and no healing. Biomaterials 28, 5009–5027 (2007).
Hsia, K. et al. Decellularized human umbilical artery exhibits adequate endothelialization in xenogenic transplantation. Biotechnol. Bioprocess Eng. 28, 439–450 (2023).
Wajihah, S. A., Sankar, D. S. & Nagar, A. K. Influence of thrombosis, stenosis and catheter on rheological characteristics of blood: a systematic review. Arch. Appl. Mech. 93, 4279–4304 (2023).
Martin, K. E. & García, A. J. Macrophage phenotypes in tissue repair and the foreign body response: implications for biomaterial-based regenerative medicine strategies. Acta Biomater. 133, 4–16 (2021).
D’Hondt, S. et al. Type III collagen affects dermal and vascular collagen fibrillogenesis and tissue integrity in a mutant Col3a1 transgenic mouse model. Matrix Biol. 70, 72–83 (2018).
Xue, M. & Jackson, C. J. Extracellular matrix reorganization during wound healing and its impact on abnormal scarring. Adv. Wound Care 4, 119–136 (2015).
Nikolova, G., Strilic, B. & Lammert, E. The vascular niche and its basement membrane. Trends Cell Biol. 17, 19–25 (2007).
Yu, Y. et al. Sulfated polysaccharide directs therapeutic angiogenesis via endogenous VEGF secretion of macrophages. Sci. Adv. 7, eabd8217 (2021).
Fang, J. et al. Skeletal muscle stem cells confer maturing macrophages anti-inflammatory properties through insulin-like growth factor-2. Stem Cells Transl. Med 9, 773–785 (2020).
Li, Q. et al. Proteomic analysis of naturally-sourced biological scaffolds. Biomaterials 75, 37–46 (2016).
Roy-Chaudhury, P. et al. Venous neointimal hyperplasia in polytetrafluoroethylene dialysis grafts. Kidney Int 59, 2325–2334 (2001).
Takahashi, E. A., Kilari, S. & Misra, S. clinical therapies and technologies in dialysis vascular access. Kidney360 2, 1373–1379 (2021).
Matsuzaki, Y., John, K., Shoji, T. & Shinoka, T. The evolution of tissue engineered vascular graft technologies: from preclinical trials to advancing patient care. Appl. Sci. 9, 1274 (2019).
Sant, S., Iyer, D., Gaharwar, A. K., Patel, A. & Khademhosseini, A. Effect of biodegradation and de novo matrix synthesis on the mechanical properties of valvular interstitial cell-seeded polyglycerol sebacate–polycaprolactone scaffolds. Acta Biomater. 9, 5963–5973 (2013).
Best, C. A. et al. Differential outcomes of venous and arterial tissue engineered vascular grafts highlight the importance of coupling long-term implantation studies with computational modeling. Acta Biomater. 94, 183–194 (2019).
de Valence, S. et al. Long term performance of polycaprolactone vascular grafts in a rat abdominal aorta replacement model. Biomaterials 33, 38–47 (2012).
Zhang, X. et al. Decellularized extracellular matrix scaffolds: recent trends and emerging strategies in tissue engineering. Bioact. Mater. 10, 15–31 (2022).
Fang, S. et al. Decellularized human umbilical artery: biocompatibility and in vivo functionality in sheep carotid bypass model. Mater. Sci. Eng. C. 112, 110955 (2020).
Bosque, B. A. et al. Ovine forestomach matrix in the surgical management of complex lower-extremity soft-tissue defects: a retrospective multicenter case series. J. Am. Podiatr. Med. Assoc. 113, 22–081 (2023).
Johnbosco, C. et al. Bioresponsive starPEG-heparin hydrogel coatings on vascular stents for enhanced hemocompatibility. Mater. Sci. Eng. C. 128, 112268 (2021).
Damiano, G. et al. Current strategies for tracheal replacement: a review. Life 11, 618 (2021).
Abou Chaar, M. K. et al. Complex esophageal reconstruction: challenges and techniques. Ann. Thorac. Surg. 120, 244–255 (2025).
Cao, Y. et al. Progress of 3D printing techniques for nasal cartilage regeneration. Aesthetic Plast. Surg. 46, 947–964 (2022).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (32222043 to K.W.; 82172106 to K.W.), the National Key R&D Program of China (2022YFA1105100 to K.W., X.Y. and Q.Z.; 2023YFC2412400 to M.Z.), the Natural Science Fund for Distinguished Young Scholars of Tianjin (24JCJQJC00180 to K.W.), the Shenzhen Science and Technology Program (KJZD20240903100504006 to K.W.), and the Tianjin Natural Science Foundation (23JCZDJC00980 to K.W.).
Author information
Authors and Affiliations
Contributions
Quhan Cheng (Q.C.), Dengke Zhi (D.Z.), Adam C. Midgley (A.C.M.), Meifeng Zhu (M.Z.), Kai Wang (K.W.) and Deling Kong (D.K.) conceived the study. K.W., Q.C., D.Z., Xiaorui Ping (X.P.), Baofa Sun (B.S.), Qining Fu (Q.F.), Xianhui Liang (X.L.), Qiuying Zhang (Q.Zhang) and Qiang Zhao (Q.Zhao) developed the methodology. Experiments and data acquisition were performed by Q.C., D.Z., Ye Wan (Y.W.), Yi Li (Y.L.), Ruixin Zhang (R.Z.), Yu Shi (Y.S.), Jiayao Zheng (J.Z.), Su Zhang (S.Z.), Boxin Liu (B.L.), Tingting Wei (T.Wei), Tengzhi Ma (T.M.), Muhammad Rafique (M.R.), Q.F., X.L., Beat H. Walpoth (B.H.W.), Pei Wang (P.W.), Yu Zhao (Y.Z.) and Ting Wang (T.Wang), Q.Zhang and Q.Zhao. Data visualization was carried out by X.P., B.S., A.C.M., D.Z. and Q.C. Funding was acquired by K.W., D.K., M.Z. and Xiaofeng Ye (X.Y.). Project administration was led by K.W., D.K., Q.C. and D.Z., and supervision was provided by K.W., D.K., M.Z., and X.Y. Q.C., D.Z. and A.C.M. wrote the original draft, and K.W., D.K., B.H.W., M.Z. and X.Y. reviewed and edited the manuscript. All authors discussed the results and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
D.K. and K.W. own stock or stock options in Leadbio Co. Ltd. D.Z. is an employee of Leadbio Co. Ltd. The other authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Cheng, Q., Zhi, D., Midgley, A.C. et al. Reinforced biotubes as readily available and regenerative vascular grafts. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70799-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70799-0